Abstract
This article deals with the investigation of electrical and mechanical properties of styrene–butadiene–styrene tri-block copolymer (SBS) nanocomposites containing SBS-grafted graphene oxide (SBS-g-GO) nanofillers dispersed in the SBS matrix through a solution processing method. In order to improve the compatibility of graphene oxide in SBS, graphene oxide was modified by maleic anhydride-grafted SBS to SBS-g-GO. The SBS-g-GO were characterized by X-ray diffraction, Raman spectroscopy, X-ray photoelectron spectroscopy, Fourier transform infrared spectroscopy, transmission electron microscopy, scanning electron microscopy, and thermogravimetric analysis. The results showed that the SBS molecules were homogeneously bonded onto the surface of the GO, leading to an improvement of the mechanical and electrical properties of SBS/SBS-g-GO composites due to the excellent interfacial adhesion and dispersion of SBS-g-GO in SBS.
Keywords
Introduction
To expand the applications of polymeric materials, the improvements in processability, durability, and performance are required. 1 –3 Polymer composites with carbon nanotubes and carbon nanofibers have attracted wide scientific and industrial interests due to the improved properties, such as electrical conductivity, thermal conductivity, and permeability resistance, provided by nanofiller materials. 4 –7 Styrene–butadiene–styrene triblock copolymer (SBS) possesses advantages of both conventional rubbers and plastic polymers, originated from its unique microphase separation, and has attracted much attention in recent years. 8 –10 However, with the rapid development in the innovative and advanced technology, there are needs of superior enough properties of SBS films to expand the application fields of SBS.
Graphene oxide (GO), a precursor of graphene, has attracted great attention due to its unique structure and outstanding physical and chemical properties. 11 –14 This material consists of graphene-derived sheets, heavily oxygenated, bearing hydroxyl, epoxide, carbonyl, and carboxylic functional groups. 15 The high density of oxygen functionalities at the GO sheet surface makes them highly hydrophilic, and very stable aqueous dispersions can be obtained. 16,17 However, aqueous dispersions are of limited interest in many applications. This is especially true regarding the preparation of polymer-based nanocomposites since most polymers are only soluble in organic solvents. To overcome the disadvantages, an immense effort has been done such as in situ polymerization and chemical functionalization. 18 –26 For instance, Aidan et al. reported an improvement in Young’s modulus at low GO content in composites. 27 Hui et al. achieved a relative low percolation threshold with well-dispersed graphene in SBS/graphene nanocomposites. 28 Borja et al. reported the influence from the sizes of nanofillers such as carbon nanotubes, GO, and graphene and concluded that GO is superior to graphene in increasing tensile strength of polyurethane. 29 Moreover, a method for growing polymers directly from the surface of GO was demonstrated by Ramanathan et al. 18 The technique involved the covalent attachment of an initiator followed by the polymerization of styrene, methyl methacrylate, or butyl acrylate using atom transfer radical polymerization (ATRP). Marques et al. also reported that poly (methyl methacrylate) chains could be grafted from the GO surface via ATRP, yielding a nanocomposite which was soluble in chloroform. 30 The ATRP method offers the possibility of preparing materials with good control over the end group functionality, and it provides a potential way to homogeneously disperse modified GO in polymeric dense matrices as well as to promote good interfacial adhesion. But for polymer/GO composites, it would introduce some functional groups that consist of elements like S and Br, which may not be beneficial to improve the properties of the composites. Therefore, more work about the functionalization of GO for the preparation of advanced polymer/GO composites should be conducted.
In this study, SBS/SBS-grafted GO (SBS-g-GO) composites with SBS-containing maleic anhydride (MAH; MAH-grafted SBS (MAH-g-SBS)) and GO were prepared. This covalent method could improve the dispersion of GO in SBS matrix as well as increase their interfacial adhesion. This study is devoted to prepare the nanocomposites from SBS and GO by a solution processing method and to explore the mechanical and electrical properties. The polymer-grafted GO filled with SBS showed improved mechanical performance compared with the pure SBS. We believe that the improvement was due to the covalent bonding of polymer chains to GO, where strong chemical bonds and a conjugated network between the GO layers and polymers were established. Moreover, the SBS-g-GO had obvious effects on the SBS elevated electrical conductivity, which made it a promising material in the field of electronic devices.
Experimental
Materials
The natural graphite powder (particle sizes ≤300 mesh; purity ≥98.0%) was purchased from Sinopharm Chemical Reagent Co., Ltd (China). The SBS (weight-average molecular weight = 70,000, 40% polystyrene and 60% polybutadiene) was produced by China Petroleum and Chemical Co. (Yanshan branch, China). MAH-g-SBS (2 wt% MAH) was prepared in this group through a common method. 31,32 All the other chemical reagents employed in the experiments were purchased from Beijing Chemical Works (China) and used as received.
Synthesis of the SBS-g-GO
Figure 1 shows the scheme for preparing SBS-g-GO. GO was prepared by an improved method. 33 A mixture of concentrated sulfuric acid/phosphoric acid (360:40 mL) was added to a mixture of graphite flakes (3.0 g) and potassium permanganate (18.0 g). The reaction was then heated to 50°C and stirred for 12 h. The reaction was cooled to room temperature and poured onto ice with 30% hydrogen peroxide (3 mL). For workup, the mixture was filtered through polyester fiber, then the filtrate was centrifuged (4000 r/min for 4 h), and the supernatant was decanted away. The remaining solid material was then washed in succession with 200 mL of water, 200 mL of 30% hydrochloric acid, and 200 mL of ethanol; for each wash, the mixture was filtered through polyester fiber with the filtrate being centrifuged (4000 r/min for 4 h) and the supernatant decanted away. The material remaining after this extended, multiple wash process was coagulated with 200 mL of ether, and the resulting suspension was filtered over a polytetrafluoroethylene membrane with a 0.45 μm pore size. The solid obtained on the filter was vacuum dried overnight at room temperature, obtaining 5.1 g of product.
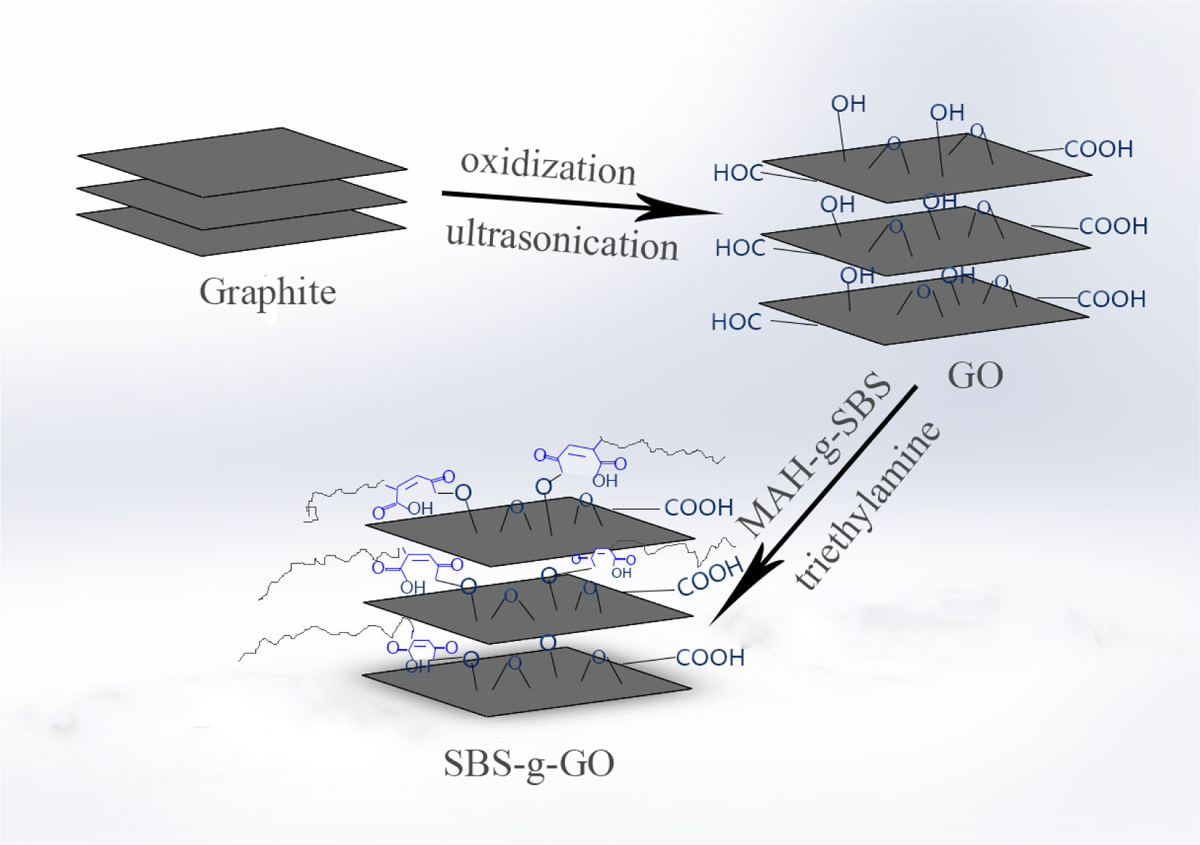
Schematic synthesis route for SBS-g-GO. SBS-g-GO: styrene–butadiene–styrene-grafted graphene oxide.
To synthesize SBS-g-GO, 1.5 g of GO would be dispersed in 150 mL of dimethylformamide under ultrasonication to form a uniform GO solution, followed by adding 15 g MAH-g-SBS which have completely dissolved in 300 mL of tetrahydrofuran in advance. Then the mixture was stirred in a three-necked flask at a speed of 80 r/min at 110°C for 1 h. After that, the temperature was turned down to 80°C, and 30 mL of triethylamine was added as catalyst for another 36 h of reaction, followed by hot-vacuum filtering with a 0.22 μm membrane and washing with tetrahydrofuran for removing unreacted MAH-g-SBS. Then the product was dried at 80°C in vacuo for 12 h. At last, solid powder of SBS-g-GO was obtained.
Preparation of SBS/SBS-g-GO composites
Raw SBS was dried at 80°C for 24 h. A mixture of tetrahydrofuran with SBS/SBS-g-GO nanocomposites was prepared, followed by mixing dissolved SBS and dispersed SBS-g-GO under ultrasonication. The dispersed solution was further kept under continuous stirring at room temperature for 4 h. Subsequently, the resulting solution was poured into a petri dish, and the resulting SBS/SBS-g-GO nanocomposites were obtained after all the tetrahydrofuran are evaporated. The composites were composed of an SBS matrix with different weight ratios of GO fillers that were respectively designed to be 0.5, 1, 3, 5, 7.5, and 10 wt%. The composites were finally compression molded at 15 MPa and about 180°C for 10 min to form disks of 10 mm in diameter and 4.2 mm in thickness for the test of mechanical properties.
Measurements
X-Ray diffraction (XRD) was performed in 2θ = 5°–60° at room temperature on a Bruker D8 X-ray diffractometer (Billerica, Massachusetts, USA) using copper K α radiation (λ = 0.15405 nm, 40 kV, 100 mA). Raman spectra were recorded from 800 cm−1 to 2500 cm−1 on a LABRAMHR800 confocal micro-Raman spectrometer using a 532 nm neodymium-doped yttrium aluminum (Al) garnet laser. X-Ray photoelectron spectroscopy (XPS) was recorded on a Kratos-AXIS ULTRA DLD with an Al K α X-ray source. Fourier transform infrared (FTIR) analysis was performed on a PerkinElmer spectrum (model 100, Waltham, Massachusetts, USA) FTIR spectrometer. Transmission electron microscopic (TEM) images of the nanocomposites were obtained with a JEM-2100 (JEOL, Japan; 200 kV). Scanning electron microscopic (SEM) images of the nanocomposites were obtained with a Hitachi S-4800 (Japan). Thermogravimetric analyses (TGAs) were conducted at 10°C min−1 from room temperature to 700°C under nitrogen flow using a TGA 209 F1 analyzer (Netzsch, Germany). The tensile strength and Young’s modulus of the composites were measured at room temperature with an R-9100 mechanical tester from Shen Zhen Rerer Instrument Co. Ltd (China). The strain rate was 5 mm min−1 with a load of 1 kN. The electrical resistivity was measured by an ZC36 megger from Shanghai No. 6 Electricity Meter Co. Ltd (China).
Results and discussion
XRD patterns of GO, MAH-g-SBS, and SBS-g-GO are shown in Figure 2. MAH-g-SBS is a kind of compatibilizer, which is synthesized by MAH and SBS. 31,32 The latter is an amorphous thermoplastic elastomer. Broad peaks can be observed in the XRD patterns corresponding to polybutadiene and polystyrene. The intense peak at 2θ = 19.5° corresponds to amorphous polybutadiene, and the weak peak at 2θ = 7.5° corresponds to polystyrene. 34 For GO, a strong peak at 2θ = 9.7° indicates the presence of residual stacked layers of graphene oxide with functional groups that contain oxygen and formed during oxidation. 35 However, this peak decreased and broadened significantly in SBS-g-GO, which indicates that interplanar spacing between GO and MAH-g-SBS broadened owing to the grafting by MAH-g-SBS and exfoliation of GO by treatment with MAH-g-SBS. Moreover, the formation of a conjugated network between the GO layers and the polymer could be proved by the diffraction peak at 2θ = 17.6°, which indicates the inducing behavior to MAH-g-SBS by the rich functional groups on the GO layers.
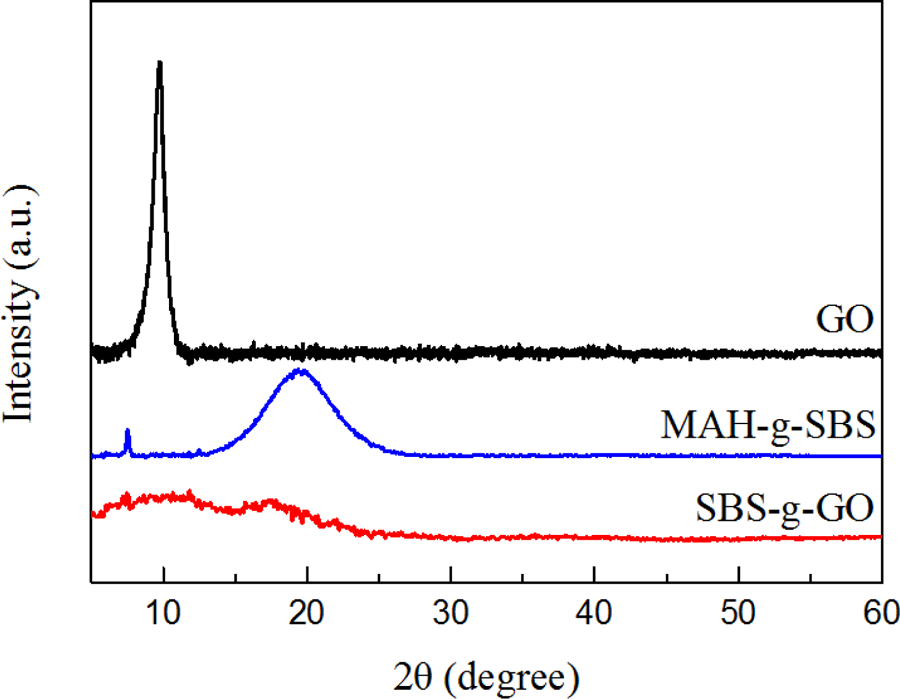
XRD patterns of GO, MAH-g-SBS, and SBS-g-GO. XRD: X-ray diffraction; GO: graphene oxide; MAH-g-SBS: maleic anhydride-grafted styrene–butadiene–styrene; SBS-g-GO: styrene–butadiene–styrene-grafted graphene oxide.
Raman spectroscopy was one of the most important methods in the structural characterization of graphitic materials, as it can provide useful information on the structure defects (D band) and in-plane vibration of sp2 carbon atoms (G band). 36,37 The Raman spectra of GO and SBS-g-GO are plotted in Figure 3. GO exhibits a G band and a D band at 1583 and 1355 cm−1, respectively. In case of SBS-g-GO, a shift of the G band to a high frequency was observed, suggesting that the number of stacked GO layers was decreased in the presence of MAH-g-SBS compared with that of GO. 38 The intensity ratio of D band to G band (ID/IG ) is further employed to evaluate the grafting of MAH-g-SBS onto GO. The value of I D /I G was increased from 0.90 for GO to 1.17 for SBS-g-GO, which was attributed to the formation new sp2 domains. 39
XPS spectra and C1s spectra for GO and SBS-g-GO are shown in Figure 4. The chemical compositions are listed in Table 1. For the GO, aside from the main C−C peak, presence of a peak at 284.6 eV (O 1s, atm%: 34.2) was attributed to surface oxygenic functional groups. 40 For SBS-g-GO sample, the C/O atomic ratio increases from 1.9 for GO to 3.2 for G-a-SBS, implying the MAH-g-SBS was successfully grafted onto the surface of GO. The C1s signal of pristine GO, as seen in Figure 4(b), consisted of six different chemically shifted components, which could be deconvoluted into C=C (284.6 eV) and C–C (285.1 eV) in aromatic rings and C atoms bonded to hydroxyl (C−OH, 286.5 eV), epoxide (C−O−C, 287.0 eV), carbonyl (> C = O, 287.9 eV), and carboxyl groups (COOH, 289.2 eV). 40 Although the C1s XPS spectra of SBS-g-GO (Figure 4(c)) also exhibit some different functionalities that have been assigned for GO, the peaks of hydroxyl and carbonyl groups vanished and a new peak at 286.0 eV appeared as attributed to the presence of ester groups. Carbonyl groups were decomposed at the temperatures of 100°C during grafting of SBS onto GO. 41 The diappearance of hydroxyl groups and the emergence of ester groups were both due to the reaction between hydroxyl groups on the surface of GO and MAH in the structure of MAH-g-SBS. The obvious increased intensity of C=C bonds was caused by the attached SBS chains on GO. These results, combined with Raman measurement, indicate the successful grafting of MAH-g-SBS onto GO.
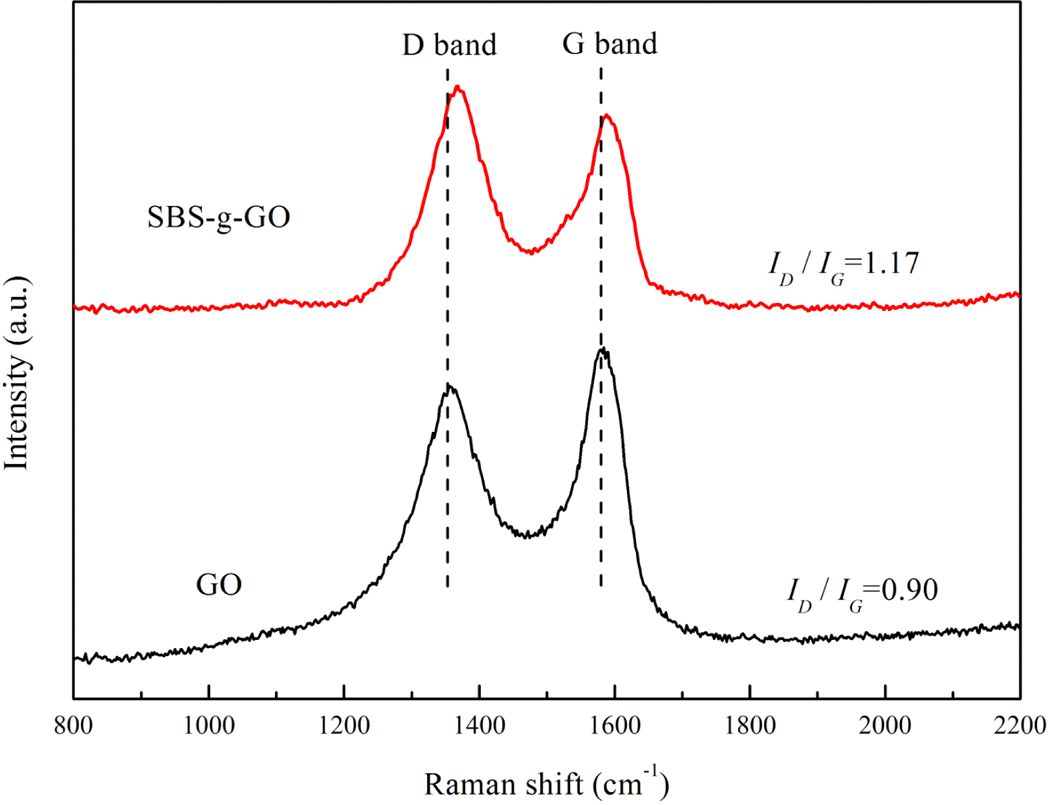
Raman spectra of GO and SBS-g-GO. GO: graphene oxide; SBS-g-GO: styrene–butadiene–styrene-grafted graphene oxide.
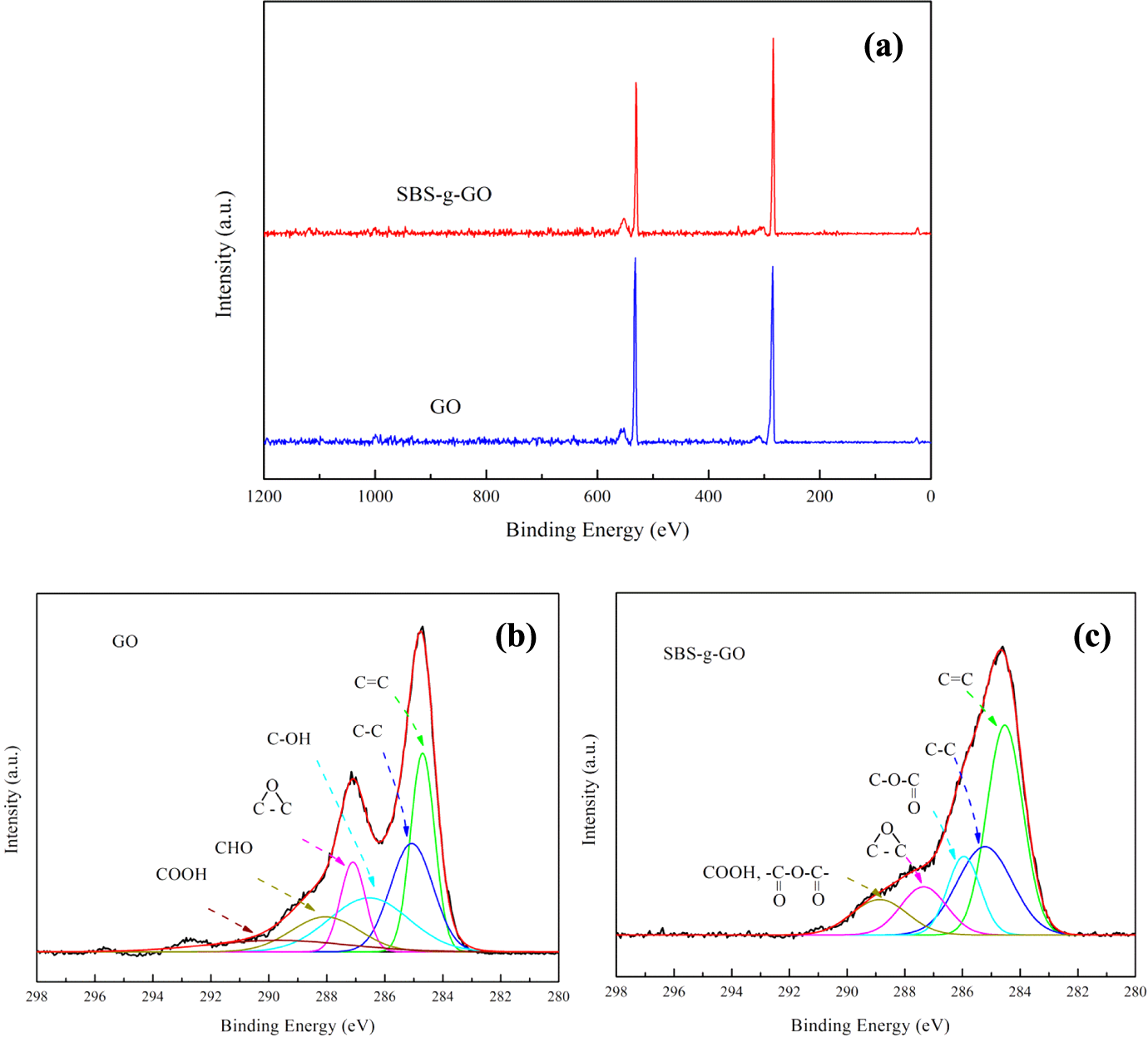
(a) XPS wide spectra of GO and SBS-g-GO samples and C1s XPS spectra of (b) GO and (c) SBS-g-GO samples. XPS: X-ray photoelectron spectroscopy; GO: graphene oxide; SBS-g-GO: styrene–butadiene–styrene-grafted graphene oxide.
Atomic percentages by XPS.
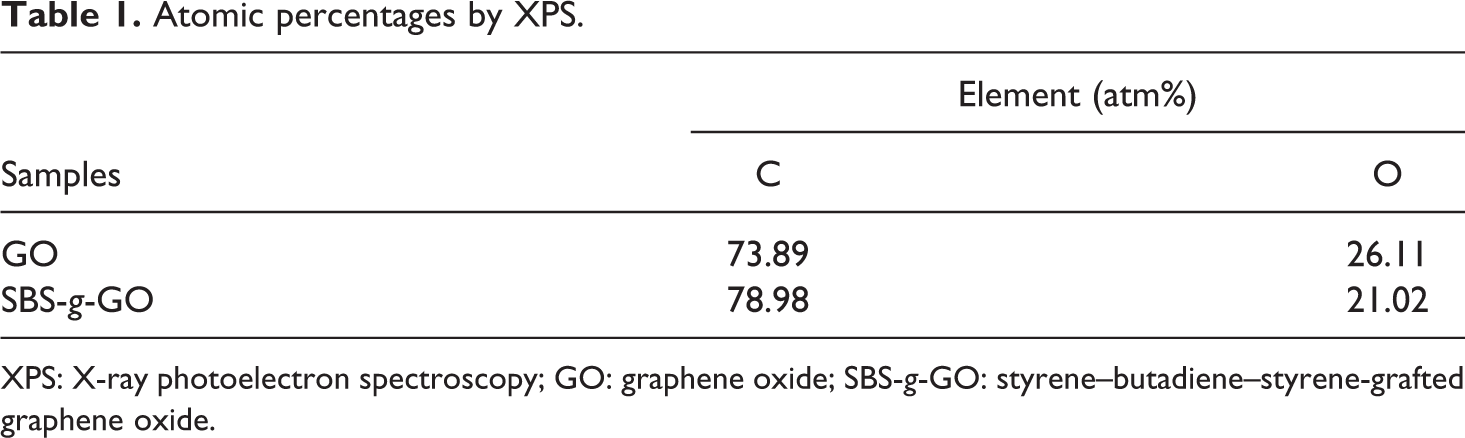
XPS: X-ray photoelectron spectroscopy; GO: graphene oxide; SBS-g-GO: styrene–butadiene–styrene-grafted graphene oxide.
To further gain insights into chemical structure of GO and SBS-g-GO, FTIR (Figure 5) was employed to analyze the existence of functional groups and the changes after the grafting of MAH-g-SBS. The improved method to prepare GO introduces large quantities of oxygen-containing groups. The peaks at approximately3420, 1730, 1280, and 1070 cm−1 signify the presence of –OH, C=O (carboxylic and ketone), C–O (carboxylic), and C–O (epoxy) functional groups, respectively. For SBS-g-GO, the new peaks at 620 cm−1, regarded as the special absorption peaks of SBS, were attributed to the bending vibration of C–H in benzene. The absorption at approximately1730 cm−1 greatly decreases in intensity as a result of decomposition of the carbonyl group and the decreases in intensity of carboxylic functional groups was due to the reaction between hydroxyl groups and MAH during the synthesis of the SBS-g-GO, which have also been proved by the analyze of C1s spectra above.
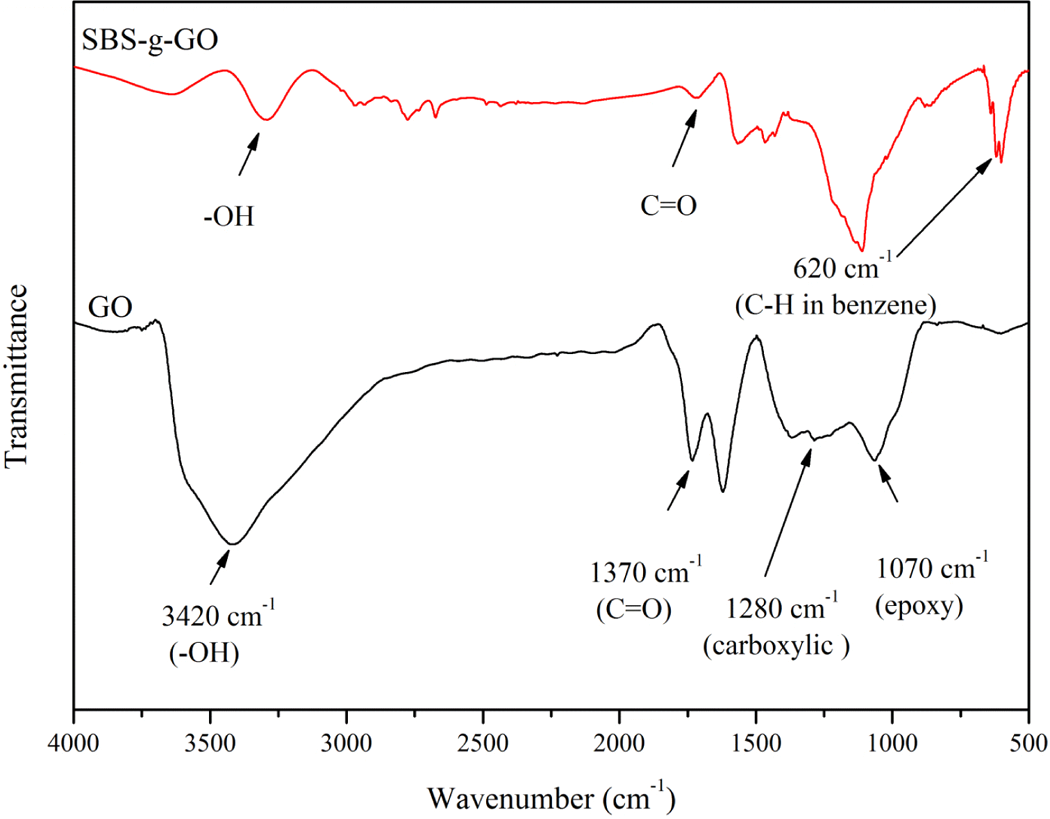
FTIR spectra of GO and SBS-g-GO. FTIR: Fourier transform infrared; GO: graphene oxide; SBS-g-GO: styrene–butadiene–styrene-grafted graphene oxide.
The structure and morphology of the GO and SBS-g-GO were characterized using TEM and SEM. The results are shown in Figures 6 and 7, respectively. The GO prepared here had a typically curved, layer-like structure with the size of tens of micrometers. For composites SBS-g-GO, SEM and TEM images showed that GO sheets were covered by MAH-g-SBS and formed a coated structure so that GO layer cannot be observed clearly. Moreover, MAH-g-SBS distributed both on the surface and between the GO sheets. What could also be observed was that the grafting of MAH-g-SBS process didn’t have a significant effect on the morphology of GO.

TEM micrographs of GO and SBS-g-GO. TEM: transmission electron microscopic; GO: graphene oxide; SBS-g-GO: styrene–butadiene–styrene-grafted graphene oxide.

SEM micrographs of GO and SBS-g-GO. SEM: scanning electron microscopic; GO: graphene oxide; SBS-g-GO: styrene–butadiene–styrene-grafted graphene oxide.
The amount of the MAH-g-SBS grafted onto the GO was determined using TGA under nitrogen atmosphere. As shown in Figure 8, GO and SBS-g-GO show a little mass loss around 100°C due to the deintercalation of solvent. GO showed a dramatic mass loss from around 200°C to 300°C due to the decomposition of oxygen-containing groups, and it showed little mass loss at 330–580°C. 35 When the temperature raised to 351°C, the weight of MAH-g-SBS started to decrease and the weight percentage of MAH-g-SBS dropped to as low as about 0 at the temperature of 550°C. Hence, weight loss of about 43 wt% occurring in the temperature range of 351–550° oC is considered as the degradation of the grafted MAH-g-SBS onto GO.
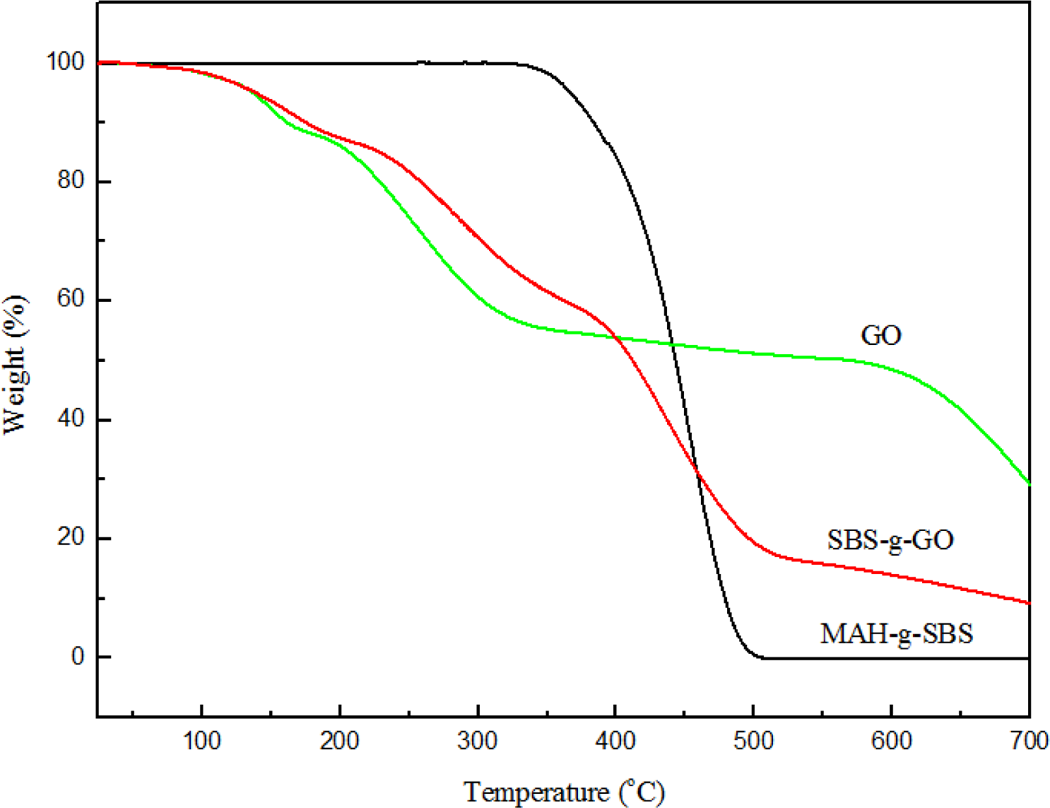
TGA traces of GO, SBS-g-GO and MAH-g-SBS. TGA: thermogravimetric analysis; GO: graphene oxide; MAH-g-SBS: maleic anhydride-grafted styrene–butadiene–styrene; SBS-g-GO: styrene–butadiene–styrene-grafted graphene oxide.
Mechanical and electrical properties of SBS/SBS-g-GO composites
Figure 9 illustrates the tensile strength and Young’s modulus of SBS/SBS-g-GO composites with various loadings of GO. The tensile strength with 3 wt% loadings of GO was 8.96 MPa increased by maxima of 43% compared to pure SBS (6.25 MPa). But the tensile strength obviously decreased with further increasing of GO, which was due to the aggregation of GO. The Young’s modulus of the composites increased with increasing GO content. The improved mechanical properties of the SBS/SBS-g-GO composite were due to the covalent chemical bonding of the SBS chains to the GO, where much stronger chemical bonds and conjugated network between the GO and SBS were established. As a result, the stronger interfacial adhesion developed a better dispersion system between the GO and SBS and the mechanical properties of the SBS/SBS-g-GO were improved in return. 42
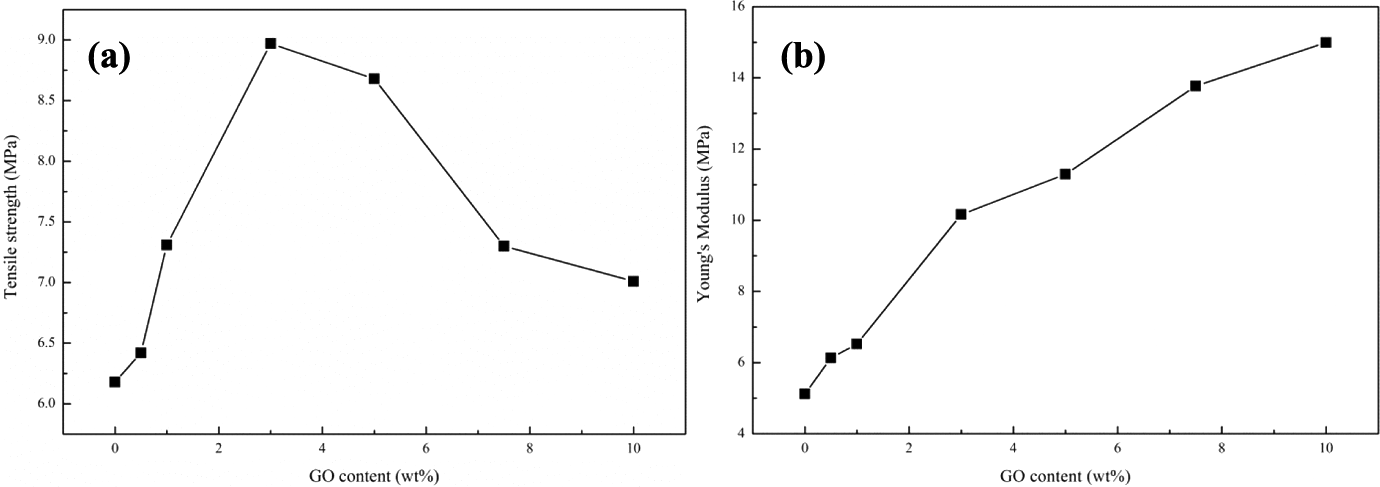
(a) Tensile strength and (b) Young’s modulus curves of SBS/SBS-g-GO composites. SBS: styrene–butadiene–styrene; SBS-g-GO: styrene–butadiene–styrene-grafted graphene oxide.
The electrical resistivity of SBS/SBS-g-GO composites are shown in Figure 10. The electrical resistivity decreases with increasing GO loading. A sudden decrease in the resistivity, which is considered the percolation phenomenon, was observed as the mass fraction of GO varies from 3 wt% to 5 wt%, indicating the formation of a preliminary conductive network which, however, was higher than the previous work achieved by loading graphene in composites. 28 This may be due to the difference in the formation of graphene and GO nanomesh. 43 When the content of GO reaches 10 wt%, the electrical resistivity of the composite attains a value of 3.47 × 105 Ω m, which means the polymer composites experienced a transition from electrical insulator to semiconductor. In particular, the functionalization of GO with SBS moieties is expected to improve the chemical affinity, the dispersion and their interactions with SBS, modifying therefore in a strong way the electrical conductivity.
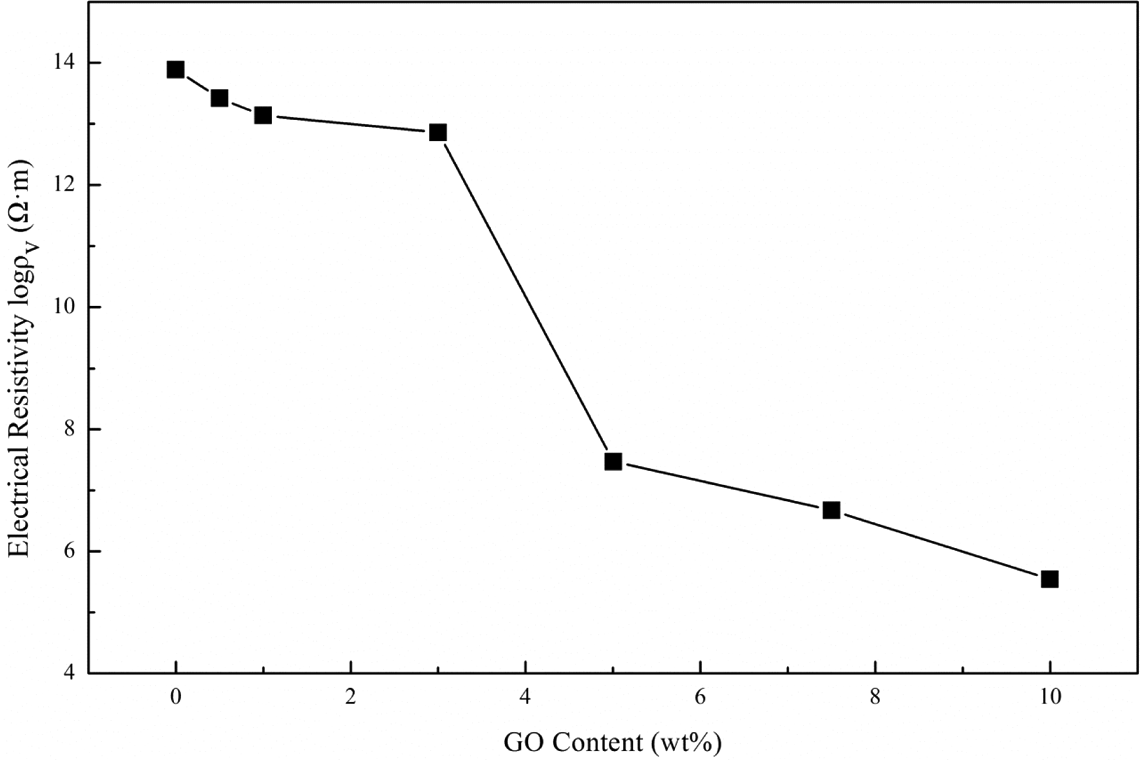
Electrical resistivity of SBS/SBS-g-GO composites with 0.5 wt%, 1 wt%, 3 wt%, 5 wt%, 7.5 wt%, and 10 wt% loadings of GO. SBS: styrene–butadiene–styrene; GO: graphene oxide; MAH-g-SBS: maleic anhydride-grafted styrene–butadiene–styrene; SBS-g-GO: styrene–butadiene–styrene-grafted graphene oxide.
Conclusions
In summary, the homogeneous composites of chemically modified GO and SBS is prepared by solution processing method. Meanwhile, the morphology and structure of GO and SBS-g-GO are fully characterized by different techniques such as XRD, Raman spectra, FTIR, SEM, TEM, TGA, and XPS. It is clear that the chemically modified GO form a homogeneous composite with the MAH-g-SBS distributed on the surface or between the GO sheets. The amount of the MAH-g-SBS grafted to GO reached as high as 43 wt%. For SBS/SBS-g-GO composite, the tensile strength, Young’s modulus, and electrical conductivity were all improved upon the addition of the SBS-g-GO.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the National Natural Science Foundation of China (contract grant number 31270608) and the Heilongjiang Educational Committee (contract grant number 1511385).
