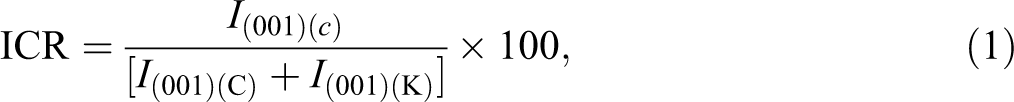Abstract
The intercalation complex marked as KAA was a modified kaolinite (KA) with potassium acetate as an intercalating agent, which was used as a reinforcement to prepare polyamide 1010 (PA1010) matrix nanocomposites (PKAA) by melt compounding. X-Ray diffraction results indicated that the interlayer basal spacing increased from 0.720 nm (KA) to 1.411 nm (KAA), after an intercalation process with an intercalation ratio of 99.7%. The nanocomposite with 2 wt% KAA exhibited the best comprehensive mechanical properties, including tensile strength, elongation at break, and notched impact strength. Furthermore, the thermal performance of these nanocomposites could be effectively improved, which manifested as the elevated glass transition temperature and thermal decomposition temperature in the test results of the dynamic mechanical thermal analysis and thermogravimetric analysis (TGA). The melting point and crystallization behavior of PKAA were also increased due to results from the differential scanning calorimetry. Besides, the bilayer inserting model was simulated by Materials Studios software to further understand the structure-function relationship of PKAA.
Introduction
Polymer nanocomposites are becoming research highlight because of their unique properties and wide potential applications, mainly including three types: nanoscaled particulates/platelets/fibrous fillers-reinforced polymer composites, polymer layered silicate (PLS) nanocomposites, and organic–inorganic nanohybrids. 1,2 Especially polymer nanocomposites based on layered silicates (clay minerals) relatively represent a large group of materials for market-oriented industrial and functional applications. 3 –5 These clay minerals are increasingly used as polymer nanofillers due to their cost advantage and processing/mechanical properties of final composites. 6,7 The study on polymer/clay nanocomposites can be traced back to the research of Toyota scientists in 1993, who prepared polyamide 6 (PA6)/montomorillonite (MMT) layered nanocomposites with improved mechanical and thermal properties. 8,9 The preparation procedure of these nanomaterials generally comprises two steps: (i) preparation of filler by intercalation of organic molecules into clay interlayer and (ii) compounding of fillers with polymer. 10 Besides enhancement of mechanical properties, it’s also worth noting that the gas barrier properties can be significantly improved for the PLS nanocomposites. 11,12
Kaolinite (KA) is an abundant natural clay mineral belonging to 1:1 group of phyllosilicates with the chemical composition Al2Si2O5(OH)4. KA is a good candidate as a filler for polymer nanocomposites because of its high crystallinity and unique structure when one side of KA interlayer is covered with hydroxyl groups of the Al2(OH)4 octahedral sheets, and the other side is covered by oxygen of the SiO4 tetrahedron. 13 The basal interlayer space of KA is relatively tight, characterized by interlayer distance 0.720 nm, and therefore, an important part of research is focused on the preparation of intercalates of KA with organic molecules (guest molecules). There are number of studies regarding the intercalation of KA using formamide (HCONH2), hydrazine (NH2NH2), urea (NH2CONH2), potassium acetate (CH3COOK), dimethyl sulfoxide (DMSO, [CH3]2SO), N-methylforamide (NMF), and ionic liquids based on organic cations such as imidazolium or pyridinium associated with a halide anion to extend the basal spacing of KA up to 1.420 nm. 14,15 The guest molecules acted as intercalating agents can be classified as three catalogs with different mechanisms. 16 Group I contains molecules including formamide, hydrazine, and urea that can form strong hydrogen bonds with the silicon tetrahedral sheets. Group II consists of dipole molecules including DMSO that have strong dipole interactions with the silicon sheets. Group III contains compounds including potassium acetate that surface hydroxyl groups on KA can form hydrogen bonds with short chain fatty acid salt.
Polyamide 1010 (PA1010) or nylon 1010 is an engineering thermoplastic with outstanding toughness, cold endurance, and lower water absorption compared with other types of polyamides like PA6 or nylon 6 and PA66 or nylon 66, but its mechanical strength, thermal stability, and dimensional stability are poorer than that of PA 6 and PA66. 17 Some research studies on KA-based nanocomposites with PA6 and PA66 have been already published, 18,19 but to the best of our knowledge, only PA1010/KA 17 and PA1010/MMT 20,21 nanocomposites were mentioned and they try to intercalate PA1010 chain into KA or MMT, however, it’s difficult. The PA1010/organic molecule intercalated KA nanocomposites have not been investigated.
In this study, potassium acetate has been selected as an intercalation agent to modify KA interlayer. A series of PA1010/KA nanocomoposites have been prepared by melt compounding. The blending components and conditions were explored to achieve optimal intercalating efficiency for the intercalating agent. After intercalation, the effects of the amount of intercalated KA on mechanical properties, crystalline behavior, and morphology of the PA1010/potassium acetate-intercalated KA nanocomposites were systematically studied. The results showed that the values of tensile strength of the PA1010/potassium acetate-intercalated KA nanocomposites are significantly increased in comparison with pure PA1010. KA served as a nucleating agent of PA1010 as evidenced by the drop of crystalline temperature using differential scanning calorimetry (DSC) measurements. The process of KA intercalation with potassium acetate was monitored by X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), and scanning electron microscopy (SEM) and also simulated by molecular modeling.
Experimental
Materials used
KA (SAK47) was supplied by LB Minerals, Czech Republic. Potassium acetate was purchased from Beijing Chemical Factory (China). PA1010 was purchased from Jiangsu Jianghu Xinglong Nylon Co. Ltd (China). 1,3,5-Tris(3,3,3-trifluoropropyl)methylcyclotrisiloxane (coupling agent) was purchased from Weihai Xinyuan Chemical Co. Ltd (China). Liquid paraffin was provided by Tianjin Fuchen Chemical Reagent Factory (China).
Preparation of KA intercalation complexes
The moisture-absorbing route was utilized to prepare the intercalated KA with potassium acetate KA and the procedure is as follows. KA and potassium acetate were mixed by 1:1 (wt/wt) under stirring. The mixture was kept at 25°C and 70% humidity for 24 h and then flushed residual potassium acetate with anhydrous ethanol. The mixture was dried at 60°C. The compounds were marked as KAA.
Preparation of PA1010/KA nanocomposites
Dried PA1010 at 90°C for 24 h in vacuum was mixed with 2–8 wt% KA intercalation complex (KAA), added 1.5 wt% of 1,3,5-tris(3,3,3-trifluoropropyl)methylcyclotrisiloxane (coupling agent) and 2.5 wt% lubricant liquid paraffin, then extruded them into pellets by twin-screw extrusion machine (TE-34 type, Kunshan Kexin Plast-Rubb Mechanery Co. Ltd, China) at the temperature zones among 230–250°C. Then, the pellets were injected into the composite samples by injection molding machine (HTF120X2 type, Ninghai Haitian Group Co. Ltd, China) at the temperature zones among 220–245°C. The molded nanocomposites were marked as PKAA-i, i = 1 is 2 wt%, i = 2 is 4 wt%, i = 3 is 6 wt%, and i = 4 is 8 wt% of KAA in PA1010.
Characterization, simulation, and mechanical tests
The structure of KAA was analyzed by (XRD) (Bruker AXS Company D8 Advance, Germany) with cobalt (Co) K α radiation source. XRD patterns were recorded using a generator voltage of 35 kV and a generator current of 30 mA. The scanning speed is 8° min−1 from 3° to 45°.
The morphology of KAA was observed by scanning electron microscopy (SEM, Philips XL30 type, Japan), and the SEM images were obtained using backscattered electrons at an operation voltage of 25 kV.
FTIR spectroscopy (Thermo Scientific Nicolet, USA) was utilized to characterize the chemical structure of intercalated kaolinite samples. The spectra were collected from 4000 to 400 cm−1 with potassium bromide pellets. The scan time is 32 with resolution of 8 cm−1.
Dynamic mechanical properties of nanocomposites were measured by a dynamic mechanical thermal analysis (DMTA, V type, Rheometric Scientific, Piscataway, New Jersey, USA) using single cantilever bending mode, fixed frequency of 1 Hz and heating rate of 3°C min−1 from −140°C to150°C. Both dissipation factor (tan θ) and storage modulus (E′) of the nanocomposites as a function of temperature can be received.
Thermagravimetric analysis (TGA) was used to evaluate the thermal stability of the nanocomposites by a synchronous thermal analyzer (Waters Sci-Tech Shanghai Co. Ltd, China) at a heating speed of 10°C min−1 from room temperature to 700°C under nitrogen atmosphere.
The melting and crystallization behavior of the nanocomposites were also determined by synchronous thermal analyzer but in DSC module (Waters SciTech Shanghai Co. Ltd, China) with heating and cooling rates of 10°C min−1 from room temperature to 250°C, kept at 250°C for 5 min, and then drop to room temperature.
Tensile properties of the nanocomposites were tested using an electronic materials testing machine (CMT4202 type, MTS System China Co. Ltd) according to China Standard GB 1040-2-2006. The tests were performed at the tensile speed of 50 mm min−1 and at room temperature. Notched impact strength of the nanocomposites was performed by Charpy impact machine (HTI-249 type, Chengde Jinjian Test Equipments Co. Ltd, China) according to China standard GB1043-2008.
Simulation of intercalation of KA with potassium acetate and urea was done by Materials Studio modeling environment (Forcite and Reflex modules) in this study. The structure model of KA unit cell with crystallochemical formula Al4Si4O10(OH)8 has been built under the periodic boundary conditions using the data by Neder et al. 22 with cell parameters: a = 5.14899 Å, b = 8.934 Å, c = 7.384 Å, α = 91.93°, β = 105.042°, and γ = 89.791°. Subsequently, the supercell structure with crystallochemical formula Al32Si32O80(OH)64 was built with the following parameters: a = 20.596 Å, b = 17.868 Å, c = 14.768 Å (i.e., doubled from the original value), α = 91.93°, β = 105.042°, and γ = 89.791°. Lattice parameters a, b, and γ were fixed during the geometry optimization procedure. Two sets of initial models were prepared: (i) models of KA containing only urea molecules (1.43–23.38 wt%) in the interlayer space; and (ii) models of KA containing both urea (2.25–16.43 wt%) and water (2.82–20.26 wt%) molecules in the interlayer space. In fact, more than 150 initial models with various interlayer contents were prepared, optimized, and examined. Charges of KA atoms were assigned using QEq method, 23 while charges of atoms in water and urea molecules were assigned using Gasteiger method. 24 All models have been optimized using Universal force field 25 in the Forcite module. A smart algorithm (i.e. the cascade of steepest descent, Conjugate gradient, and Quasi-Newton optimization algorithms) was used for the geometry optimization with 100,000 iteration steps. In order to monitor the changes of KA d 001 value in dependence on the interlayer content, simulated diffraction patterns have been calculated for each optimized model in Reflex module under the same conditions as in the experiment, like 2θ range of 5°–50°, Co source (λ = 1.78897 Å) and Bragg–Brentano geometry.
Results and discussion
Characterization of KAA
XRD patterns of KA and KA intercalation compound (KAA) are shown in Figure 1. Compared with KA, for which the lamellar spacing (d 001) is 0.720 nm, the diffraction peak (d 001) of KAA almost disappeared, instead of a strong new diffraction peak at 1.411 nm. It indicates that potassium acetate molecule has been successfully inserted into the interlamellar space of KA and the intercalation is completed. Intercalated ratio (ICR) values can be used for quantitative characterization of KA’s intercalation efficiency, as follows 26 :
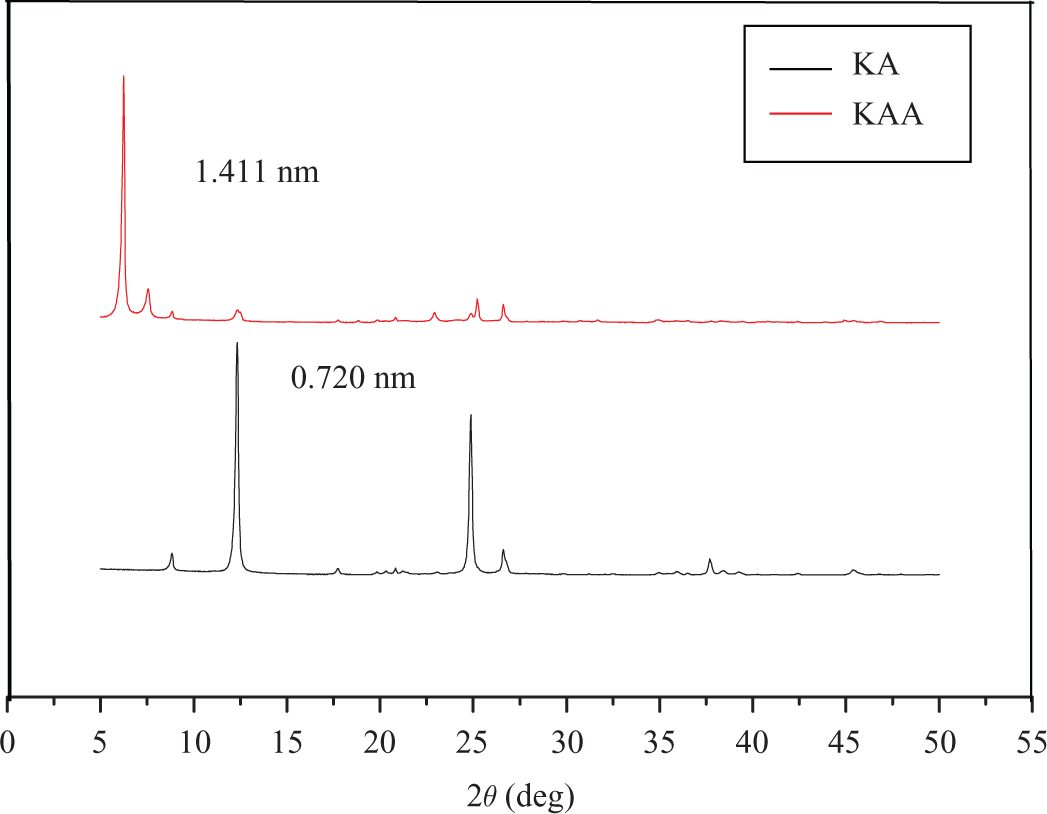
XRD patterns of KA and KAA. XRD: X-ray diffraction, KA: kaolinite; KAA: modified kaolinite.
where I(001)(C) is the new diffraction peak intensity from XRD pattern, I (001)(K)is the weakened diffraction peak intensity of original KA (d 001). According to the calculation, ICR value of KAA is 99.7%. Therefore, potassium acetate is one of the best organic guest molecules intercalating to host KA.
SEM images show that KA is a kind of relatively completed needle lamellar structure, as shown in Figure 2(a). Lattice edges and corners are obvious, but have an agglomeration phenomenon. The structure of KAA becomes lamellar structure (Figure 2(b)), proving that potassium acetate could enlarge the KA lamellae effectively, and the intercalation is more complete and make the KA lamellae exfoliate partly.

SEM images of (a) KA and (b) KAA. SEM: scanning electron microscopic; KA: kaolinite; KAA: modified kaolinite.
FTIR spectra of KA and KAA are shown in Figure 3. Typically, two major bands, 3697 and 3620 cm−1 in hydroxyl stretching vibration range related to KA can be observed. 3697 cm−1 is –OH stretching band corresponding to inner surface, while 3620 cm−1 is assigned to inner –OH stretching band. 27 There are three inner surface hydroxyl groups and one inner hydroxyl group in KA 28 where 1031 cm−1 is Si–O stretching band and 912 cm−1 is Al–O stretching band of KA. Both Si–O and Al–OH groups supply the sites for hydrogen bonding, therefore, after intercalation, the 3474 cm−1 band is intercalated water molecule’s absorption in KA. 29 Both 1604 and 1419 cm−1 are COO− antisymmetric and symmetric stretching bands, respectively. 30 FTIR spectra prove the intercalation of potassium acetate assisted by water molecules into KA.
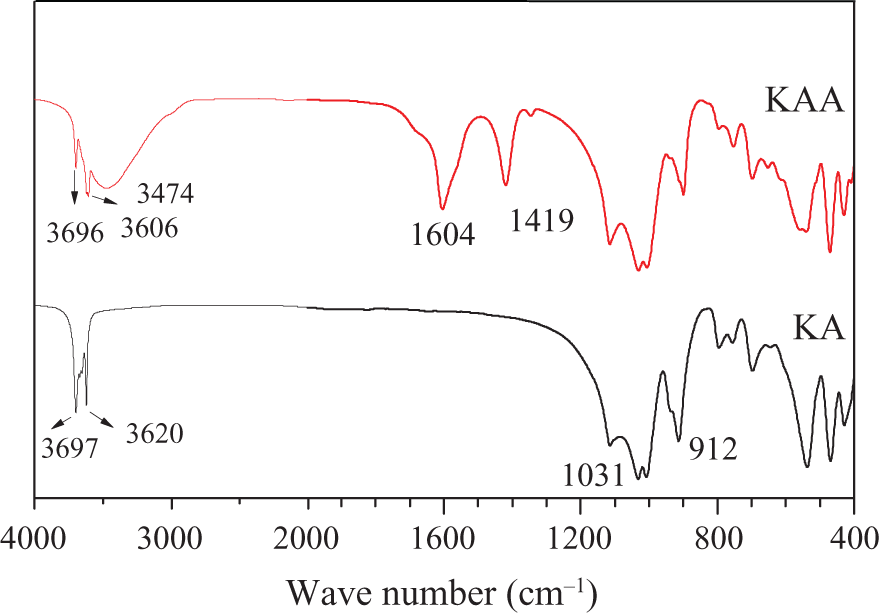
FTIR spectra of KA and KAA. FTIR: Fourier transform infrared; KA: kaolinite; KAA: modified kaolinite.
The mechanisms of the intercalation of KA have been discussed. Mostly, monolayer model and intercalation mechanisms were suggested by Wada, 31 White et al., 32 and Frost et al. 33 Water plays a critical role in Wada’s model. Three configurations of potassium acetate with or without water present within the KA were proposed by White’s and Frost’s monolayer models. However, our Materials Studio modeling results shown in Figure 4 revealed that lateral bilayers of potassium acetate inserted into KA is possible and both monolayer and bilayer structures can be coexisted. Other experimental observation and molecular simulation of bilayers of potassium acetate in KA were reported. 34,35
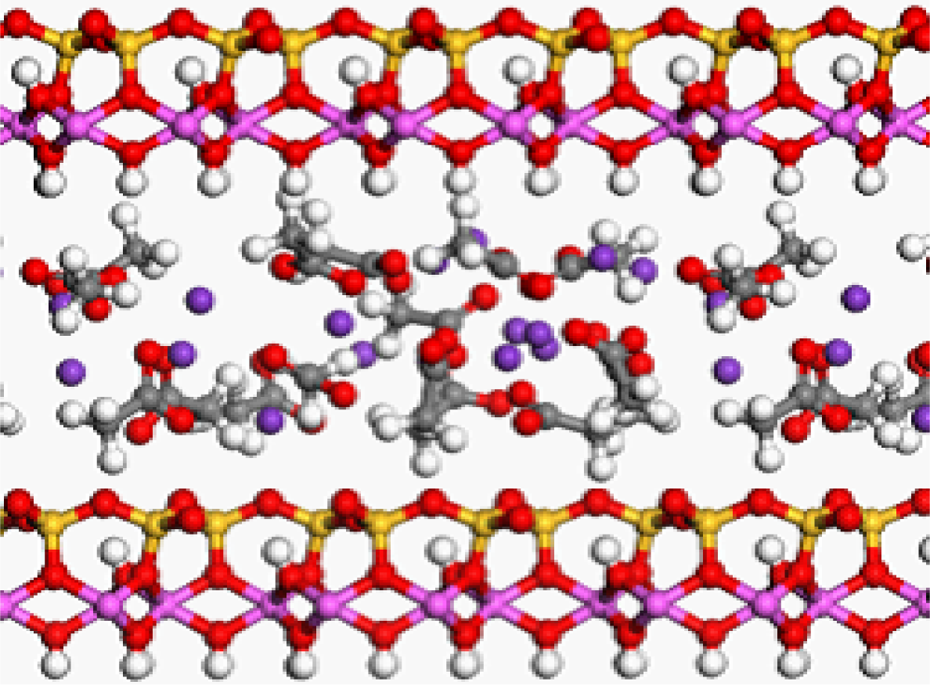
Simulation of potassium acetate intercalated to KA. KA: kaolinite.
Mechanical properties of PKAA
The tensile strength, elongation at break, and impact strength of PKAA are shown in Figure 5. To compare with PA1010, the tensile strength of the nanocomposites is all increased, and the maximum value can be increased to about 20% when the content of KAA is 8 wt%. KAA plays an important role in enhancing the tensile strength of PA1010. The elongation at break of the nanocomposites shows the best value when the content of KAA is 2 wt%. Elongation at break of PKAA-1 is increased by 66.3% than that of PA1010. Subsequently, when KAA content is greater than 4 wt%, the elongation at break of composites is decreased sharply with the increase of KAA content, until sacrificing nearly 75% of the toughness of nanocomposites. In addition, due to notched impact strength of PA1010 is sensitive, the notched impact strength of nanocomposites is lower than that of PA1010 and decreased with KAA content increasing.
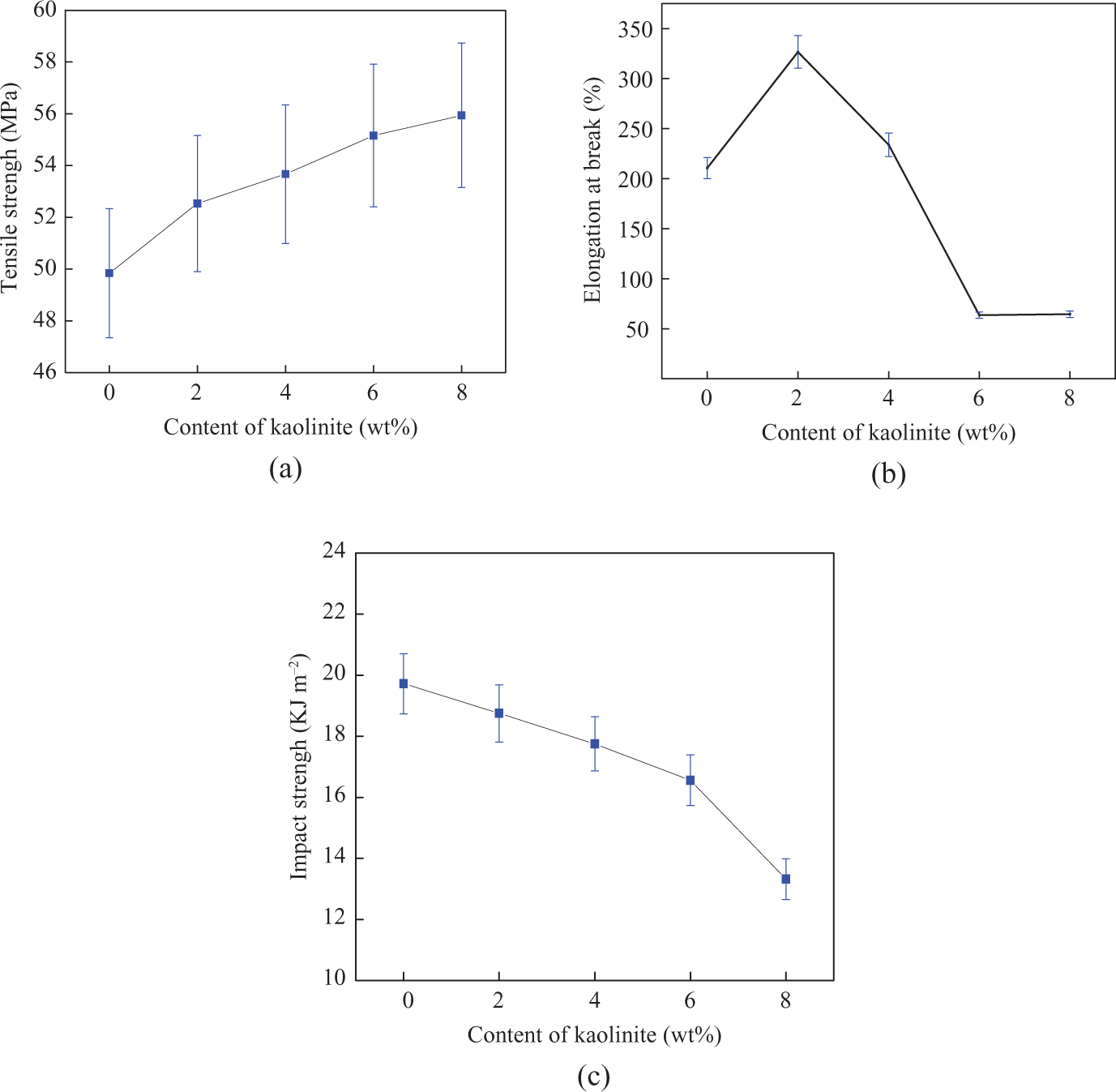
The tensile strength (a), elongation at break (b), and notched impact strength (c) of PA1010 and PKAA-i. PA1010: polyamide 1010; PKAA: polyamide 1010 matrix nanocomposite.
Dynamic mechanical properties of PKAA
The DMTA of PA1010 and PKAA nanocomposites is shown in Figure 6 and Table 1. Two transitions within the temperature range measured were observed from the curves of tan δ as a function of temperature. The α peak (glass transition) representing the segments movement in amorphous zone of PA 1010 appears at about 57.4°C and the β peak related to the –CONH– movement in amorphous zone of PA 1010 locates at about −67.2°C. The α transition is shifted to a higher temperature after the addition of PAA to PA1010, that is, the glass transition temperature (T g) of PKAA-i is increased when KAA content is increased due to the chains of PA1010 inserted into the layers of KA, and therefore, the segment movement of PA1010 was constrained. The β peaks were located almost at the same temperature. Also, the curves of E′ as a function of temperature indicates that the stiffness of the PKAA is higher than PA1010 when the temperature is larger than 57.4°C.
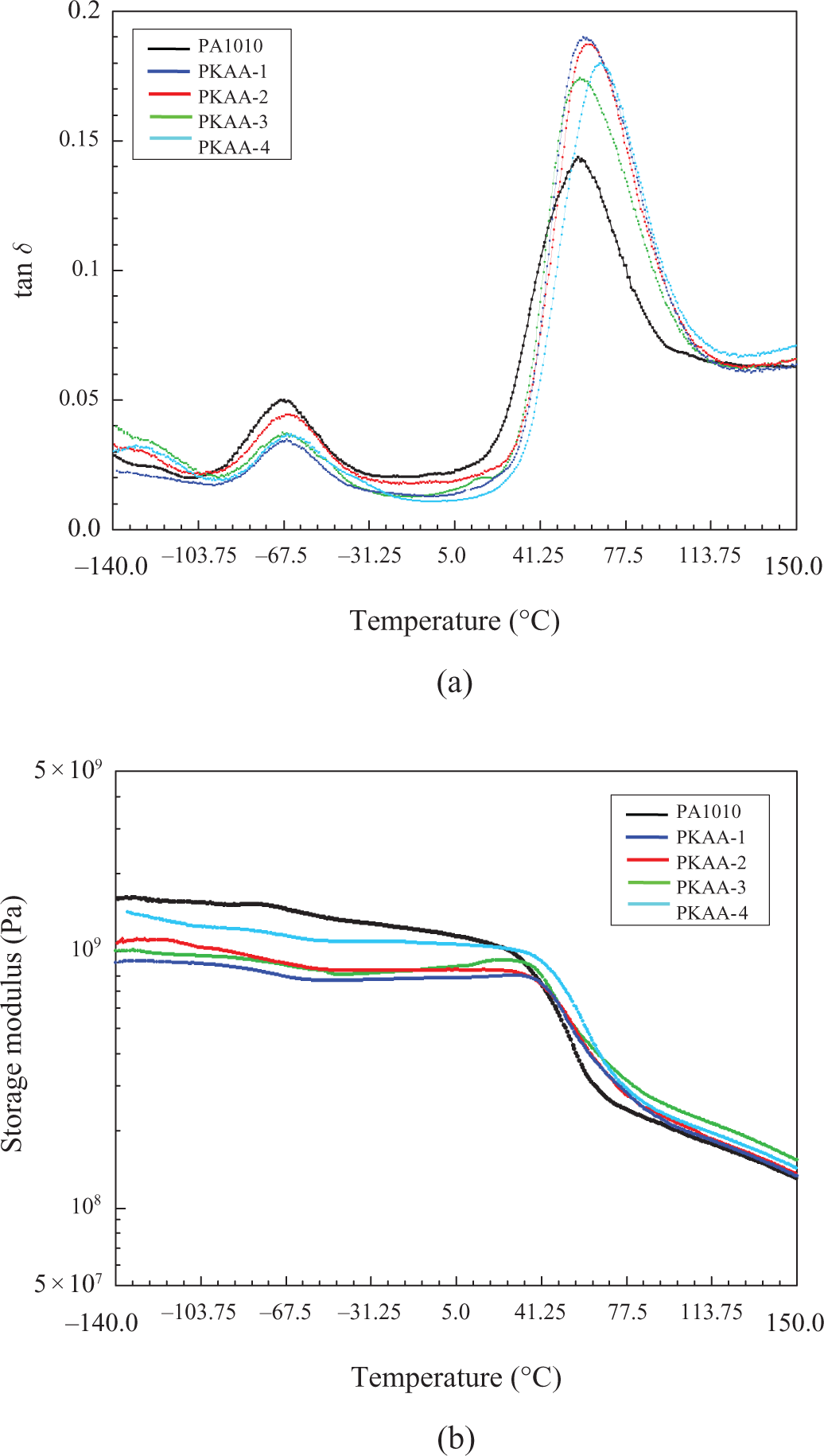
DMTA curves of PA1010 and PKAA-i (a) tan δ and (b) E′ as a function of temperature. DMTA: dynamic mechanical thermal analysis; PA1010: polyamide 1010; PKAA: polyamide 1010 matrix nanocomposite.
T g and T β of PA1010 and PKAA-i.
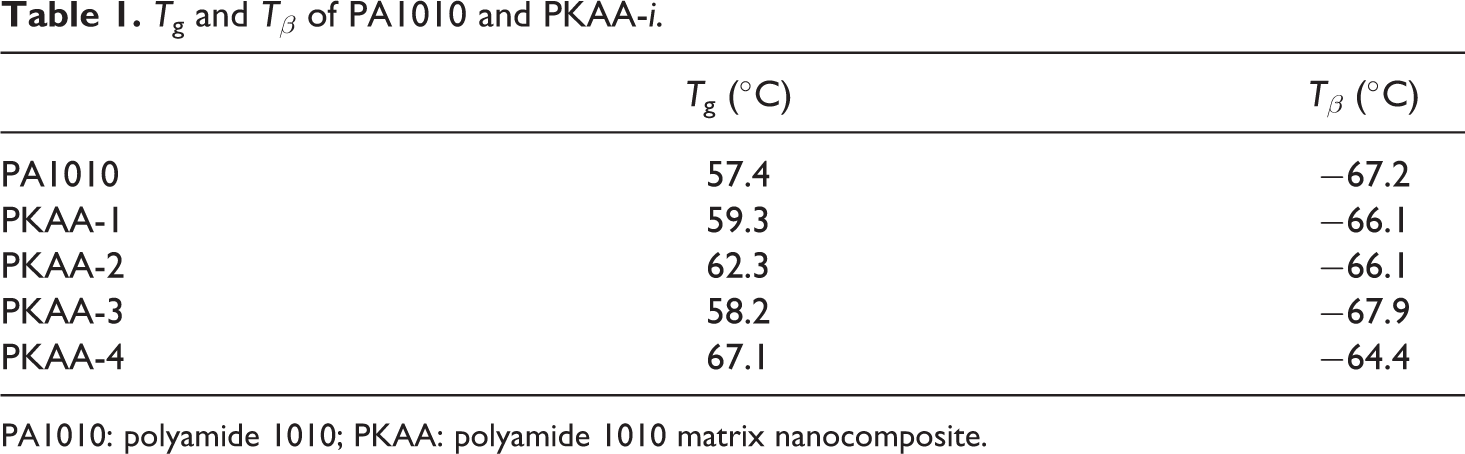
PA1010: polyamide 1010; PKAA: polyamide 1010 matrix nanocomposite.
Thermal stability of PKAA
TGA results (Figure 7 and Table 2) shows that thermal decomposition temperature (T d) of nanocomposites increased in contrast to neat PA1010 when the KAA content was ≤ 4 wt%, but Td of nanocomposites is decreased slightly with the increasing of KAA content. It indicates that a small amount of KAA can improve the thermal stability of the matrix better. Due to the movement of the polymer molecular chain was restricted in KA lamellae, 36 the thermal stability of PA1010/KA nanocomposites can be improved.
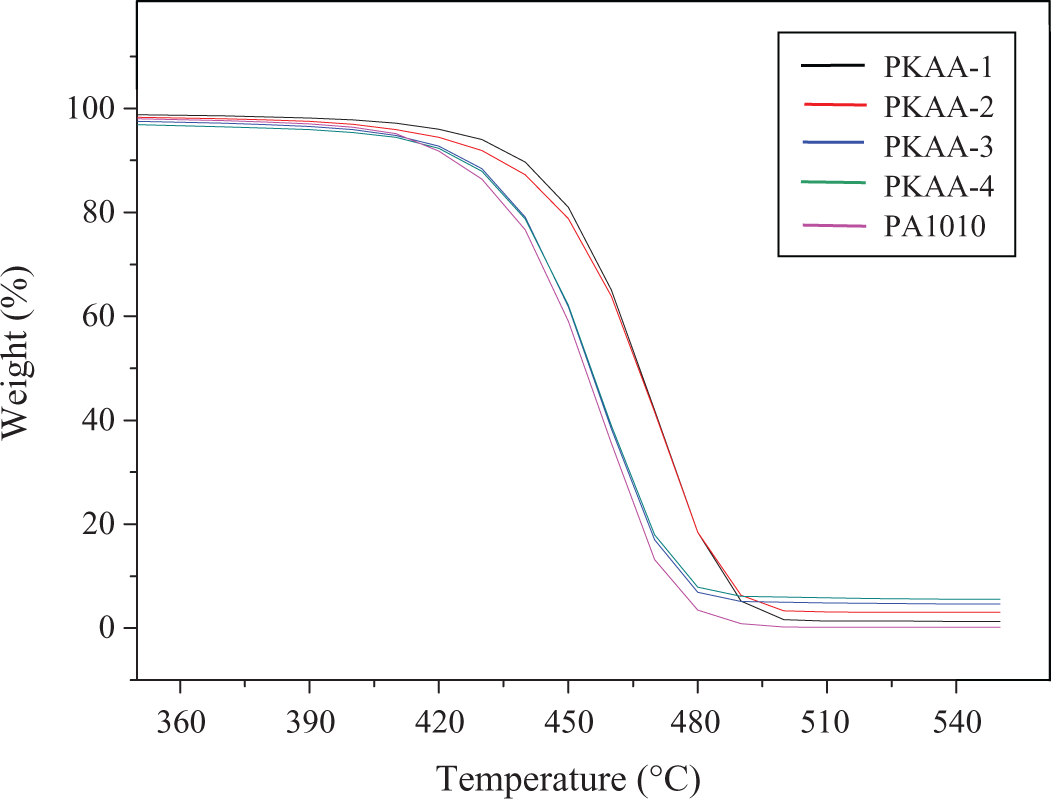
TGA curves of PA1010 and PKAA-i. TGA: thermogravimetric analysis; PA1010: polyamide 1010; PKAA: polyamide 1010 matrix nanocomposite.
Initial and thermal decomposition temperature of PA1010 and PKAA-i.

PA1010: polyamide 1010; PKAA: polyamide 1010 matrix nanocomposite; T i: initial decomposition temperature; T d: thermal decomposition temperature.
Melting and crystallization behavior of PKAA
The crystallization and melting curves or data of PA1010 and nanocomposites are shown in Figure 8 and Table 3. Single melting peak was appeared in PA1010, but double melting peaks of PKAA were observed. The melting points of PKAA-i are increased from 209.4 (PA1010) to about 225.1°C (PKAA-1). The lower melting peaks of PKAA-i may be caused by partly PA1010 chains inserted into kaolinite and some unperfected crystalline was formed within the layers of KA.
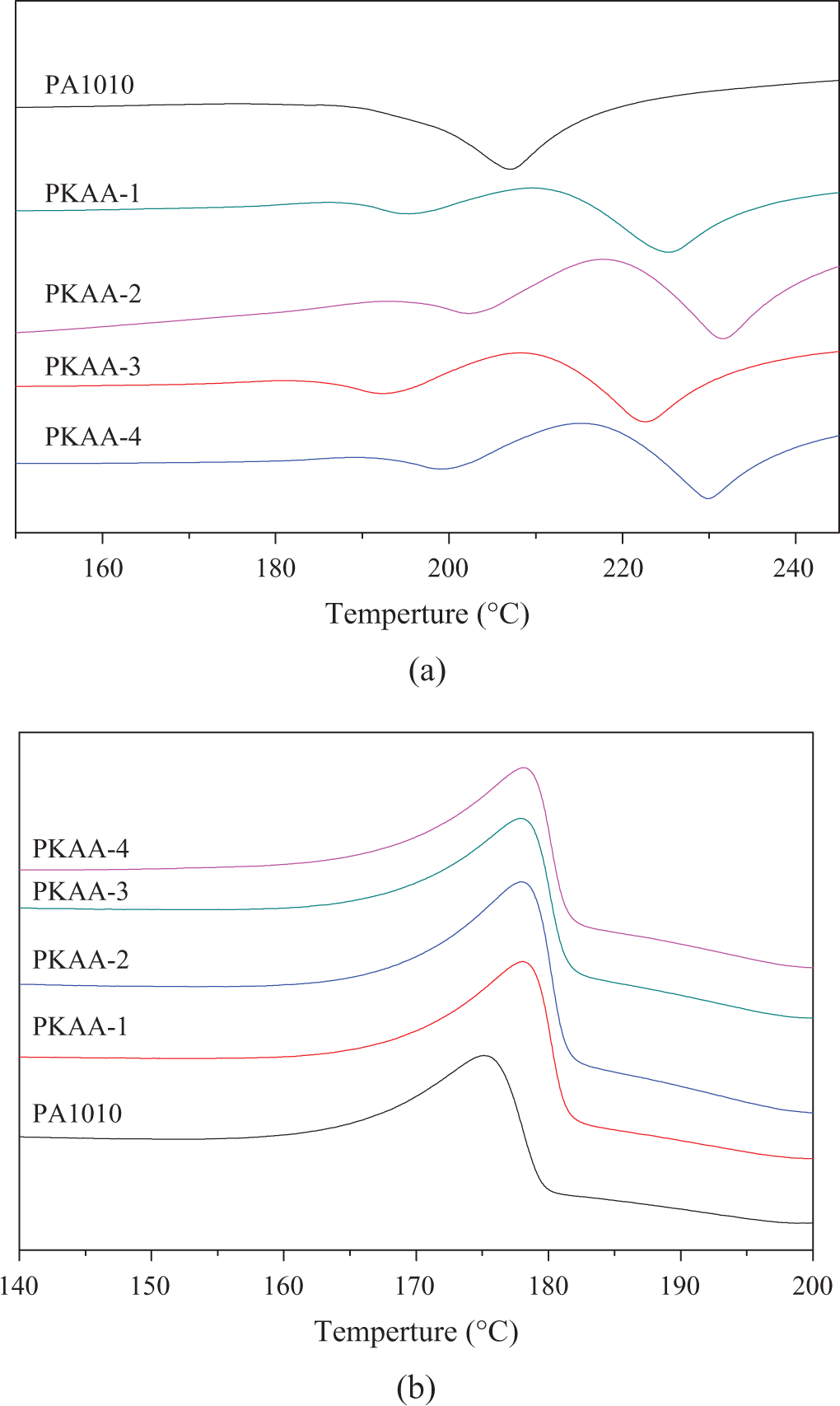
Melting (a) and crystallization (b) behavior of PKAA-i. PKAA: polyamide 1010 matrix nanocomposite.
Melting and crystallization data of PA1010 and PKAA-i.
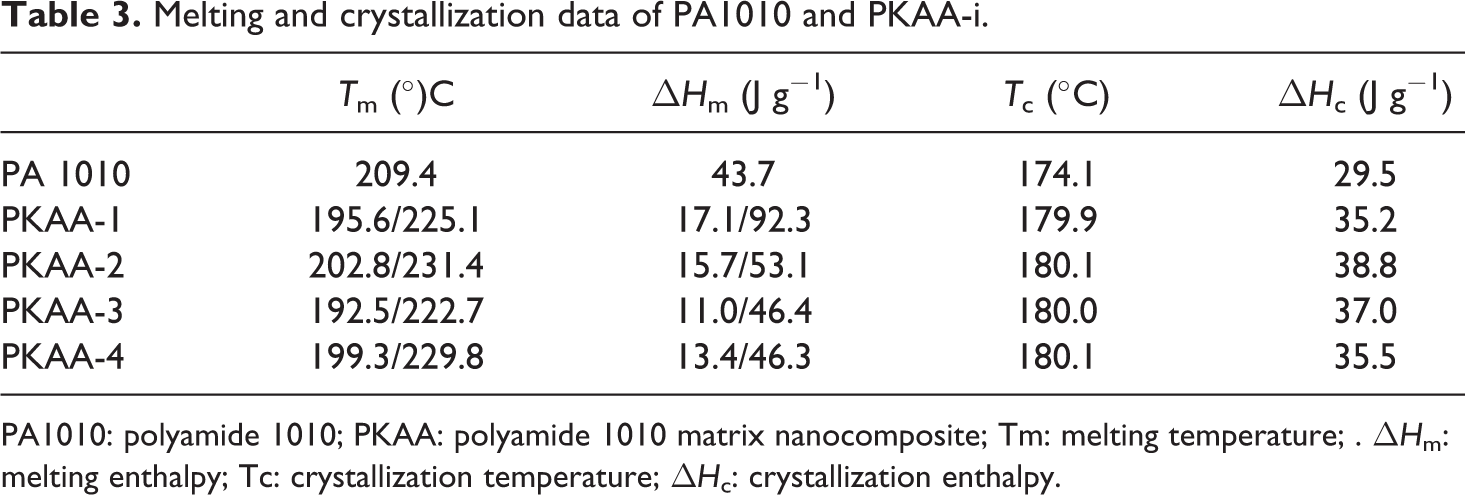
PA1010: polyamide 1010; PKAA: polyamide 1010 matrix nanocomposite; Tm: melting temperature; . ΔH m: melting enthalpy; Tc: crystallization temperature; ΔH c: crystallization enthalpy.
Conclusions
Potassium acetate can effectively expand the interlamellar spacing of KA from 0.720 nm to 1.411 nm, with an intercalation efficiency of 99.7%. Intercalated KA and PA1010 are feasible for preparing nanocomposites (PKAA) by a melt compounding method, and these nanocomposites generally show a good overall performance. Mechanical properties of the PKAA were improved by comparing with original PA1010. The best mechanical properties including tensile strength, elongation at break, and notched impact strength were found in PKAA-1 nanocomposite with 2 wt% KAA. Furthermore, the thermal performance of these nanocomposites could be effectively improved by the addition of KAA according to the DMTA, TGA, and DSC results, which manifested as the elevated T g, T d and melting point.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by China-Czech cooperation project (40-22) and project LH 12184 (Ministry of Education Youth and Sports of Czech Republic).