Abstract
Kaolinite has often been intercalated before being introduced into polymers to improve its dispersibility; however, the conventional intercalation usually reduces the flame retardancy of the composite. This work reports our recent efforts on improving both the flame retardant efficiency and dispersibility of kaolinite in polypropylene (PP) by intercalating with ammonium sulfamate (AS). The intercalation had been performed through three steps: dimethyl sulfoxide was firstly introduced into kaolinite layers under supersonic wave, then it was replaced by potassium acetate-aqueous (KAc), and finally the intercalated KAc was replaced by AS to obtain AS-intercalated kaolinite. The structure of intercalated kaolinite was characterized by X-ray diffraction (XRD), Fourier-transform infrared spectroscopy, and thermogravimetric analysis (TGA). The flammability evaluation by limit oxygen index, vertical burning test (UL-94), cone calorimeters test (CONE), and TGA indicated that the fire resistance, thermal stability, and physical properties of PP can be effectively enhanced by the introduction of AS-intercalated kaolinite. The peak heat release rate (pHRR) value of PP composite containing only 1.5 wt% intercalated kaolinite (1169 kW m−2) had been reduced 13.2% compared with that of the sample containing 1.5 wt% raw kaolinite (1346 kW m−2). The morphology analysis from scanning electron microscope images and XRD patterns demonstrated that the compatibility and dispersibility of kaolinite in PP had been significantly improved by intercalation. The flame retardant mechanism of AS-intercalated kaolinite in PP was proposed.
Introduction
Nanocomposites have attracted much attention 1 –6 in the last two decades because of the significant improvement in polymer properties such as mechanical, barrier, and optical properties along with their fire performance compared with the corresponding neat polymer. 7 –10 A number of literatures focusing on preparation, characterization, and properties of nanocomposites articles have been published. 7,9 However, the natural layered clay mineral is not sufficient to generate well-intercalated or exfoliated morphologies, which cannot meet the requirement of good flame retardant and mechanical properties.
Kaolinite is an abundant mineral across the world, and it is widely applied in industrial fields such as paper fillers, ceramics, polymer additives, and so on. Kaolinite is a 1:1 non-expensive layered silicates with a chemical formula of Al2Si2O5(OH)4. 11,12 The stacked layers are linked by strong hydrogen bonds. The structural asymmetry of kaolinite, which is due to the superposition of the tetrahedral and octahedral sheets in the 1:1 layers, creates large superposed dipoles. Consequently, this asymmetry makes intercalation of guest organic molecules more challenging than that of other layered compounds such as layered double hydroxides and montmorillonite. 13,14 At the same time, the procedure of intercalation of kaolinite has received little attention and should be more developed. The preparation of the target intercalation kaolinite usually need three steps: Firstly, small or dipolar molecules (e.g. dimethyl sulfoxide (DMSO)) can be intercalated into the interlayers of kaolinite directly to break the strong hydrogen bonds of interlayers of kaolinite; secondly, the above intercalated kaolinite compounds are used as precursors for further intercalation by displacing previously intercalated precursor species 15 with potassium acetate-aqueous (KAc) 15,16 ; finally, target substance is intercalated into the layered space of kaolinite by replacing KAc. The conventional intercalation has mainly focused on improving the dispersion of kaolinite, and the intercalation surfactants usually contain aliphatic chains which might decrease the fire resistance of the polymer. Kaolinite has attracted more and more attention in fire science 17,18 in the last several years because of its environmental friendliness, low cost, and effective flame retardancy. Meanwhile, raw kaolinite or modified kaolinite and intumescent flame retardant was used to improve the flame retardancy of polymers in our recent research, 19 –23 surprisingly, the literature about introducing intercalated kaolinite to improve the fire performance of PP has not been found so far.
Polypropylene (PP) has been widely used in many areas due to its low cost, lightweight, chemical corrosion resistance, recycle ability, and so on. However, its application has greatly been restricted by its inherent flammability. 24,25 It is reported that nature clay could be an ideal alternative to the often used halogen-contained flame retardant formulations to enhance flame retardancy of polymer composites. 10 However, there is very strong commercial need to develop PP composite with both good flame retardancy and excellent mechanical properties by introducing low content (≤3 wt%) of flame retardant additives.
This work reported our recent efforts on improving both the flame retardant efficiency and dispersibility of kaolinite in PP by intercalating with ammonium sulfamate (AS). The flammability of PP containing low amount of AS-intercalated kaolinite was characterized and evaluated. Meanwhile, the morphology, thermal stability, and mechanical property of PP/kaolinite composite were analyzed.
Experimental
Materials
The commercial PP, with a melt flow index of 3 g/10 min, was kindly provided by Sinopec Maoming Company (Maoming, China). DMSO (the purity level >99.5%), KAc with a purity of 92%, and AS with a purity of 99.5% were purchased from Beijing Chemical Factory (Beijing, China). The raw kaolinite with a size range of 1–46 µm, a mean size of 12 µm, and a specific surface area of 17 m2 g−1 (the purity >95%) was kindly supplied by Xing Yi Mineral Processing Plant (Shijiazhuang, China).
Measurements
The X-ray diffraction (XRD) was recorded with a D/max-2500 diffraction (Rigaku Corporation). The copper Kα radiation source was taken using a generator voltage of 40 kV and a current of 20 mA (λ = 0.154 nm). Samples were scanned from 3° to 30° at a scan rate of 3° min−1.
Fourier-transform infrared (FTIR) spectroscopy was performed with a Nicolet IS5 Spectrophotometer (Thermo Fisher Scientific).
Thermogravimetric analysis (TGA) was carried out using a synchronous thermal analyzer (STA449C, Netzsch [Deutschland, Germany]) and heated from 30°C to 700°C at a heating rate of 10°C min−1 under nitrogen atmosphere.
Evaluation of flammability of the composites was tested using cone colorimeter (FTT Co., Ltd [Vereinigtes Königreich, UK]) according to the ISO 5660 standard with heat radiant flux density of 50 kW m−2. All samples with the dimensions of 100 × 100 × 3 mm3 were tested at horizontal position. Specimens were wrapped in aluminum foil, leaving the upper surface exposed to the radiator. The experiments were an average of three replicated measurements.
The limit oxygen index (LOI) value was obtained according to the standard oxygen index test method of ISO 4589-2. The dimension of each sample is 100 × 6.5 × 3 mm3. The vertical test was according to the UL-94 test standard. The specimens used were of dimensions 130 × 13 × 3 mm3. All samples were repeated three times.
The distributions of kaolinite in composites were observed by a Hitachi S-4700 (Hitachi Limited, Micromeritics Instrument Corporation) scanning electron microscope (SEM) under a voltage of 20 kV.
The tensile testing was carried out using a tensile tester (Zwick/Roell, Germany) at ambient temperature with a stretching rate of 50 mm min−1 according to the ISO 527-2 standard.
Specific surface area was determined by N2 adsorption in a Micromeritics Gemini 2.0 instrument, using the Brunauer–Emmett–Teller (BET) method. Particle size analysis was carried out by sedimentation and absorption of X-rays in a Micromeritics Sedigraph 5100 (LabX) apparatus.
Preparation of AS-intercalated kaolinite
Figure 1 shows the procedure of preparing AS-intercalated kaolinite, which includes three steps:
Step I (DMSO intercalation): Four grams of kaolinite (K0) was introduced to a three-neck flask containing a mixture of 40 mL of DMSO and 4.5 mL of deionized water. The suspension was reacted under ultrasonic with a power of 200 W for 4 h. The reacted mixture was washed by ethanol for three times and then filtrated before being dried at 60°C for 12 h. The dried product was DMSO-intercalated kaolinite (marked as K1).
Step II (KAc intercalation): Two grams of K1 was added to a three-neck flask containing 40 mL of saturated KAc water solution under stirring at 50°C for 24 h. The reacted mixture was washed by ethanol for three times and then filtrated before being dried at 60°C for 12 h. The product was KAc-intercalated kaolinite (marked as K2).
Step III (AS intercalation): Two grams of K2 was dispersed in a three-neck flask containing 40 mL of AS/H2O (8 mol/L) solution and stirred at 50°C for 24 h. The reacted mixture was washed by ethanol for three times and then was filtrated before being dried at 60°C for 12 h. The recovered solid sample was AS-intercalated kaolinite (
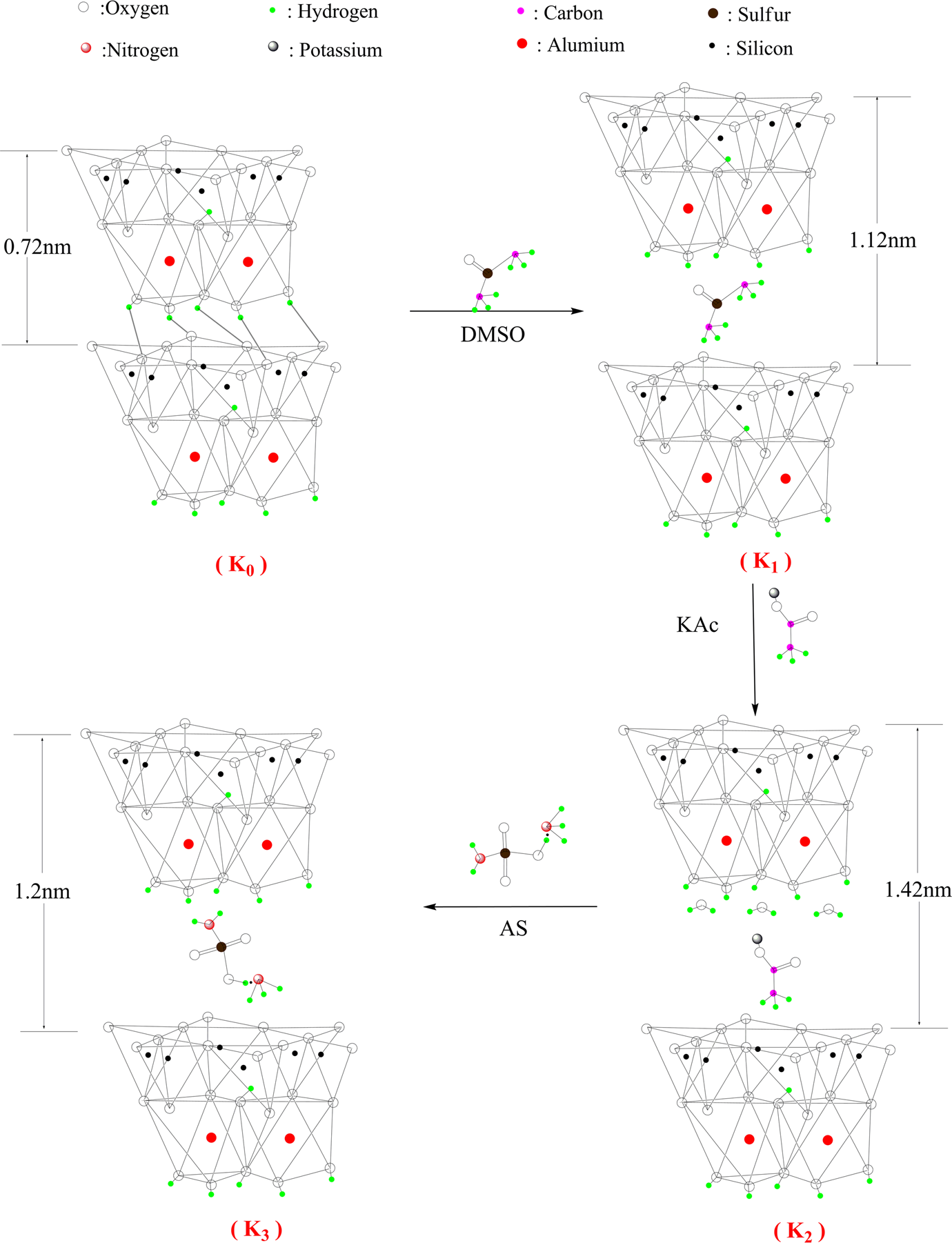
Preparation process for AS intercalated kaolinite. AS: ammonium sulfamate.
Preparation of composites
PP and PP composites containing K0 or K3 were prepared by melt blending with a micro twin-screw conical extruder (Wuhan Rui Ming Plastics Machinery Co. Ltd, (Wuhan City, China) the length, minimum diameter, and maximum diameter of screws of extruders is 190, 25, and 10 mm, respectively) at a screw speed of 40 r min−1. The processing temperature from hopper to die was set as 170, 180, and 190°C, respectively.
Table 1 presents the formulations of PP composite.
Formulation of PP composites (in mass percentage).
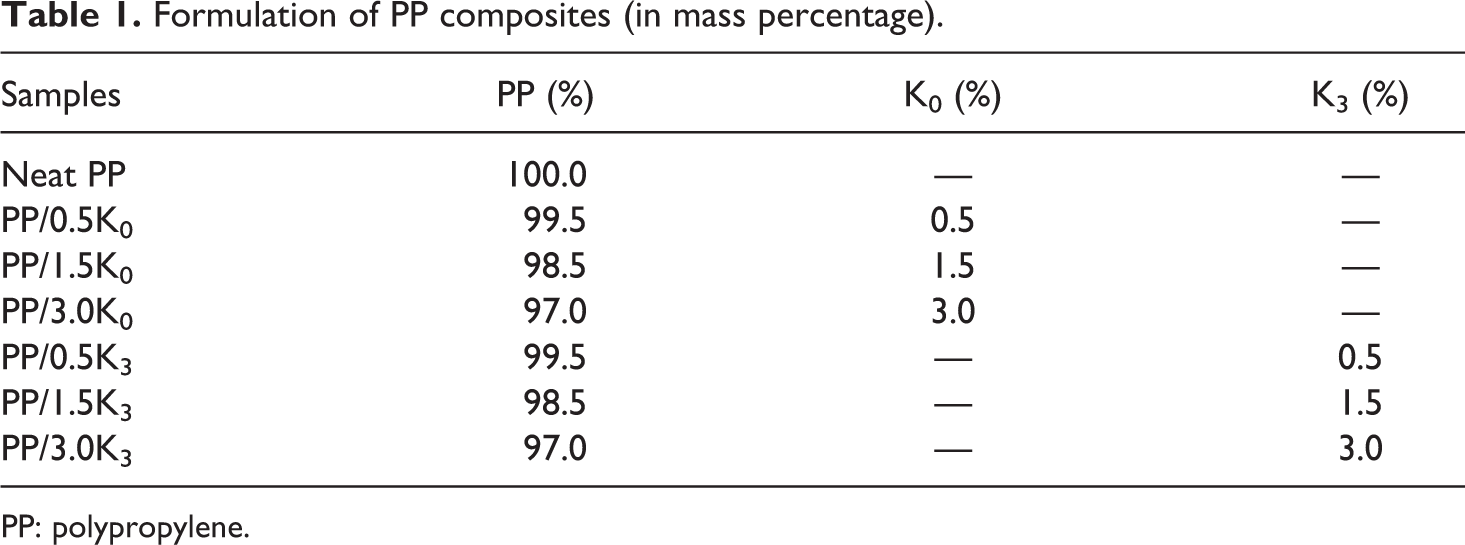
PP: polypropylene.
Results and discussion
XRD analysis
Figure 2 shows the XRD patterns of K0 and its intercalated compounds of K1, K2, and K3. The intercalation rates (InR) are calculated according to equation (1), 16 where Ik (001) is the peak intensity of raw kaolinite, and Ii (001) is the peak intensity of intercalated products. Based on equation (1), InR value for K1, K2, and K3 was obtained as 92.6%, 95.2%, and 90.1%, respectively
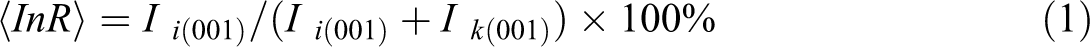
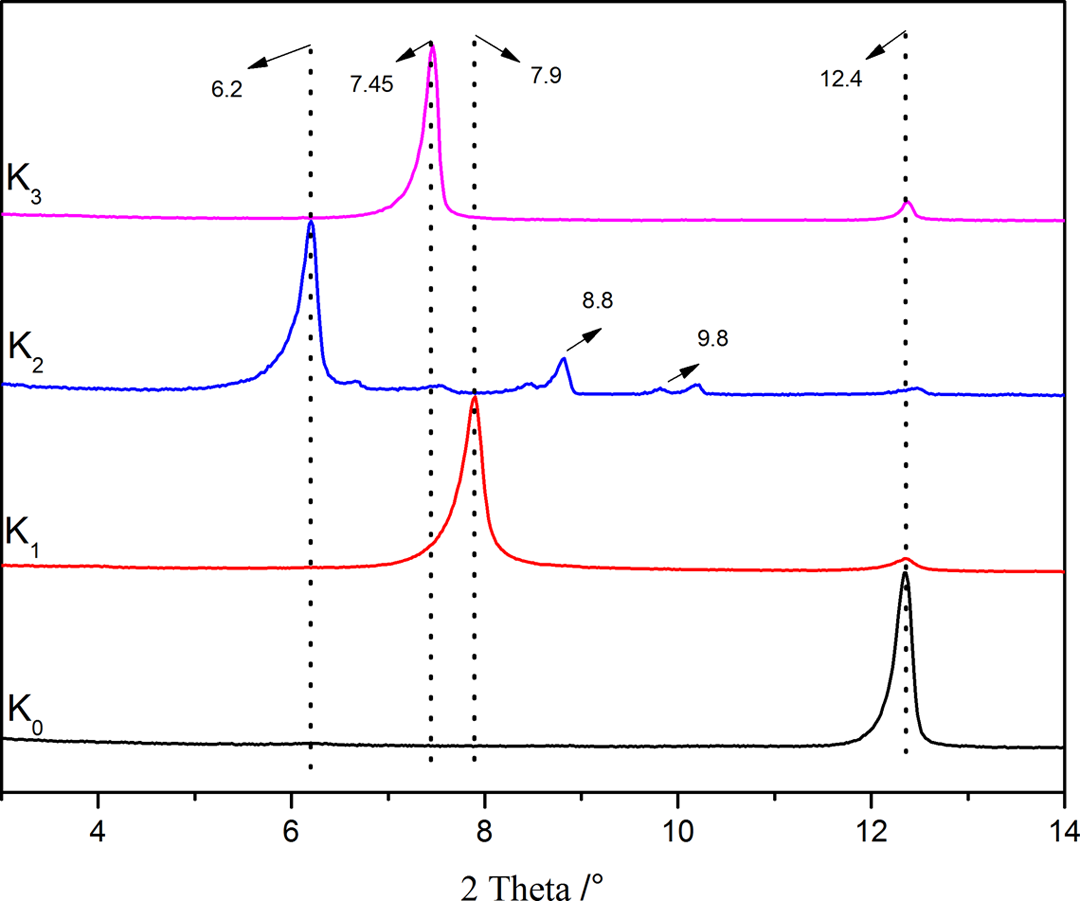
XRD patterns of K0 and different intercalated kaolinite.
The d 001 spacing was expanded from 0.72 nm (K0, 2θ = 12.4°) to 1.12 nm (K1, 2θ = 7.9°), which was in accordance with published research results. 15,16,26,27 After being treated with KAc in water, the basal spacing was expanded to 1.42 nm (K2, 2θ = 6.2°). The expansion space of 0.7 nm in K2 was in favor of further intercalation with AS. It should be noticed that there were two slight apices at 2θ = 8.8° and 9.8° in the XRD-trace of K2, indicating the release of KAc when it was dried under vacuum. 15 It was demonstrated that water and KAc existed in the monomolecular layers of kaolinite. 12 After being treated with AS in water, the d 001 space of K3 (1.2 nm, 2θ = 7.45°) showed a slight decrease compared with that of K2 (1.42 nm, 2θ = 6.2°). The decrease in d 001 space may be attributed to the orientation and configuration of AS in interlamellars.
FTIR analysis
Figure 3 shows the FTIR spectra of K0 and its intercalated compounds of K1, K2, and K3. The frequency and assignments of the main vibrations are summarized in Table 2. 26 –31 The four IR bands at 3694, 3670, 3654, and 3621 cm−1 were the characteristic bands of hydroxyl stretching vibration in K0. The bands at 3694, 3670, and 3654 cm−1 corresponded to interlayer hydroxyl stretching, while the band at 3621 cm−1 was attributed to the stretching vibrations of the internal hydroxyl groups. 28,29 The bands at 1115 (apical Si–O), 1033, and 1010 (Si–O–Si in-plane vibrations) were also typical bands for K0. 30 After being treated with DMSO (K1), new bands at 3663, 3022, 2937, 1319, and 905 cm−1 were observed, while the bands at 3670 and 3654 cm−1 were disappeared. It was noted that the guest molecules decreased the peak intensity at 3694 cm−1 but did not affect the peak intensity at 3621 cm−1. The appearance of new band at 3663 cm−1 and disappearance of bands at 3670 and 3654 cm−1 were attributed to the inner-surface hydroxyl groups which were hydrogen bonded to the DMSO. New band at 905 cm−1 was attributed to the hydroxyl deformation of the inner-surface hydroxyl groups that were hydrogen bonded to the S=O group in DMSO. Meanwhile, the bands appear at 3022, 2937, and 1319 cm−1 were assigned to CH3 stretching vibration. 31 The Si–O and Si–O–Si plane vibration bands of K1 were shifted to higher wavelengths (1018, 1040 and 1123 cm−1, respectively) compared with that of K0. After being treated with KAc (K2), the characteristic band at 3663 cm−1 vanished, and bands at 3694, 3607, 1602, and 1420 cm−1 were emerged. The reemerging of outer-surface hydroxyl band at 3694 cm−1 demonstrated that the DMSO molecules on outer surface were replaced by KAc, and the released hydroxyl groups on outer surface of K2 were chemically linked with KAc. The appearance of band at 3607 cm−1 indicated that KAc molecular was intercalated into the layers successfully. 16 At the same time, the appearance of bands at 1420 and 1602 cm−1 were the symmetric and asymmetric stretching of COO−. The vibration bands of the Si–O and Si–O–Si in K2 were slightly right shifted (1007, 1032, and 1114 cm−1, respectively) compared with that in K1.
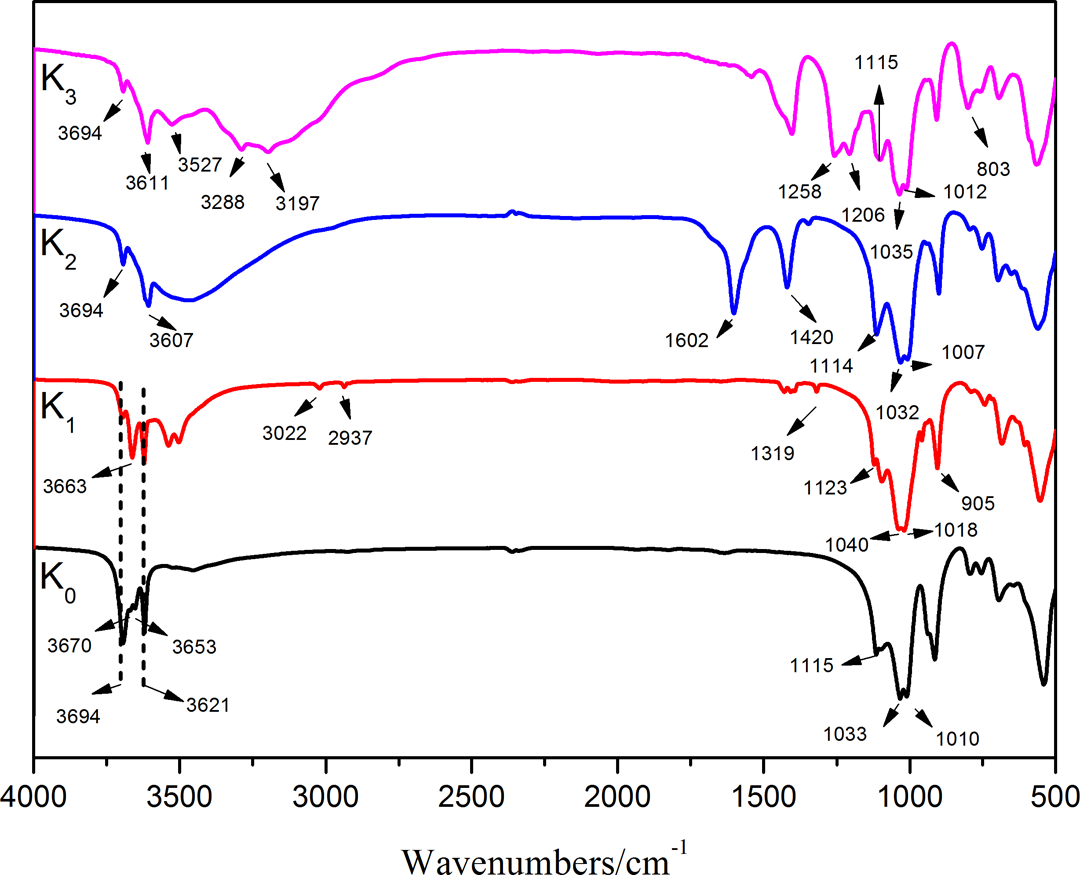
FTIR spectra of K0 and different intercalated kaolinite.
IR: infrared.
After being treated with AS (K3), additional bands at 3527, 3288, 3196, 1258, 1206, and 803 cm−1 were observed, while the bands at 1602 and 1420 cm−1 were disappeared. O–H stretching at 3527 cm−1, N–H stretching at 3288 and 3198 cm−1, S=O stretching at 1258 and 1206 cm−1, and S–O stretching at 803 cm−1 were characteristic absorbance bands of AS. In addition, the disappearance of characteristic bands of KAc at 1602 and 1420 cm−1 demonstrated that AS molecular had replaced KAc and intercalated into the clay layer successfully. The vibration bands of the Si–O and Si–O–Si in K3 were slightly left shifted (1012, 1035, and 1115 cm−1, respectively) compared with that in K2.
Thermal properties
Figure 4 presents TGA/differential thermal gravity (DTG) curves of K0, K1, K2, and K3. The major mass loss of K0 during 400–650°C in TGA curve was corresponding to the peak at 543°C in DTG curve, which was attributed to the dehydroxylation process of K0. 12 The minor mass loss during 220–280°C was attributed to the elimination of crystal water in the interlayer of K0. The residue of K0 at 700°C was 86.4%. For K1, the TGA curve had two major mass losses, centering at 181 and 528°C. 12 The former one during 130–200°C was due to the elimination of DMSO from K1 and the latter one during 450–650°C was due to the dehydroxylation of K1. The residue at 700°C was 71.8%. The TGA curve of K2 showed two major mass losses. The former one from 30°C to 120°C with a DTG peak of 76°C was attributed to the removal of absorbed water on the surface of K2. The latter step within 310–560°C with a DTG peak of 381°C was assigned to the removal of KAc 12 and the dehydroxylation of kaolinite from K2. The residue at 700°C was 72.5%.
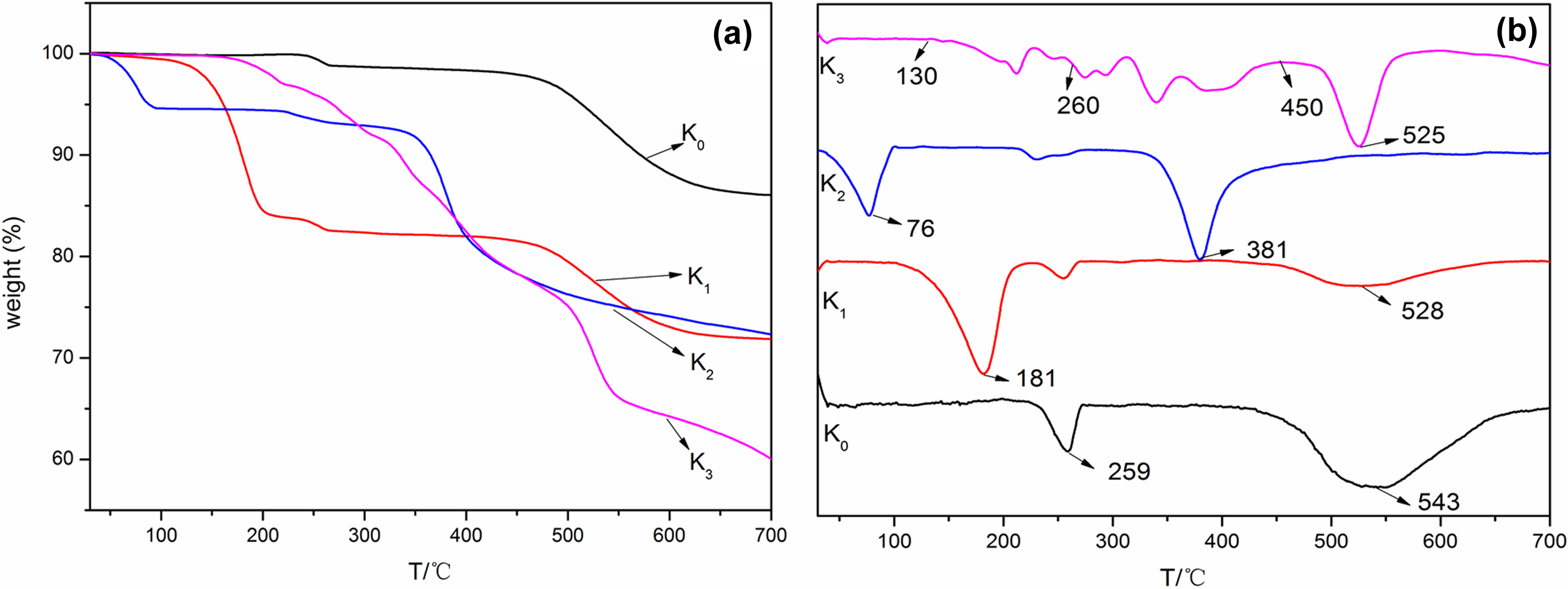
TGA (a) and DTG (b) curves of K0 and different intercalated kaolinite.
K3 undergoes seven stages during decomposition process. The first and second mass losses (between 130°C and 260°C) were assigned to the dimerization of AS associate to the release of ammonia 32 and removal of crystal water of K3 during heating. The next four steps occurred in the range of 270 and 600°C. The maximal degradation rate was reached 360°C, which was ascribed to the removal of sulfamic acid from the K3 interlamellar space. The last one in the range of 460–600°C with the DTG peak of 525°C was attributed to the dehydroxylation of clay. The residue at 700°C was 59.9%. It can be concluded that AS was successfully intercalated into kaolinite (K3) by the three-step reactions.
Flammability and thermal stability
LOI and UL-94 tests
LOI and UL-94 test results of PP composites containing K0 and K3 are listed in Table 3. The LOI value for neat PP was only 18.0 and it was lightly increased for PP containing K0 or K3. The LOI value reached 18.4 and 18.7 for PP samples containing 3 wt% K0 or K3, respectively. However, all samples cannot pass UL-94 tests, indicating the flame retardant cannot be effectively improved by the presence of clay only. It is suggested that the higher ignition propensity, due to the silicates could catalyze the degradation of the matrix and/or promote the dehydroxylation of the aluminosilicate lattice, 33,34 resulting a negative effect on their performance in flammability tests, such as the LOI and UL-94. 3,35
Flammability test results for neat PP and its composites.a
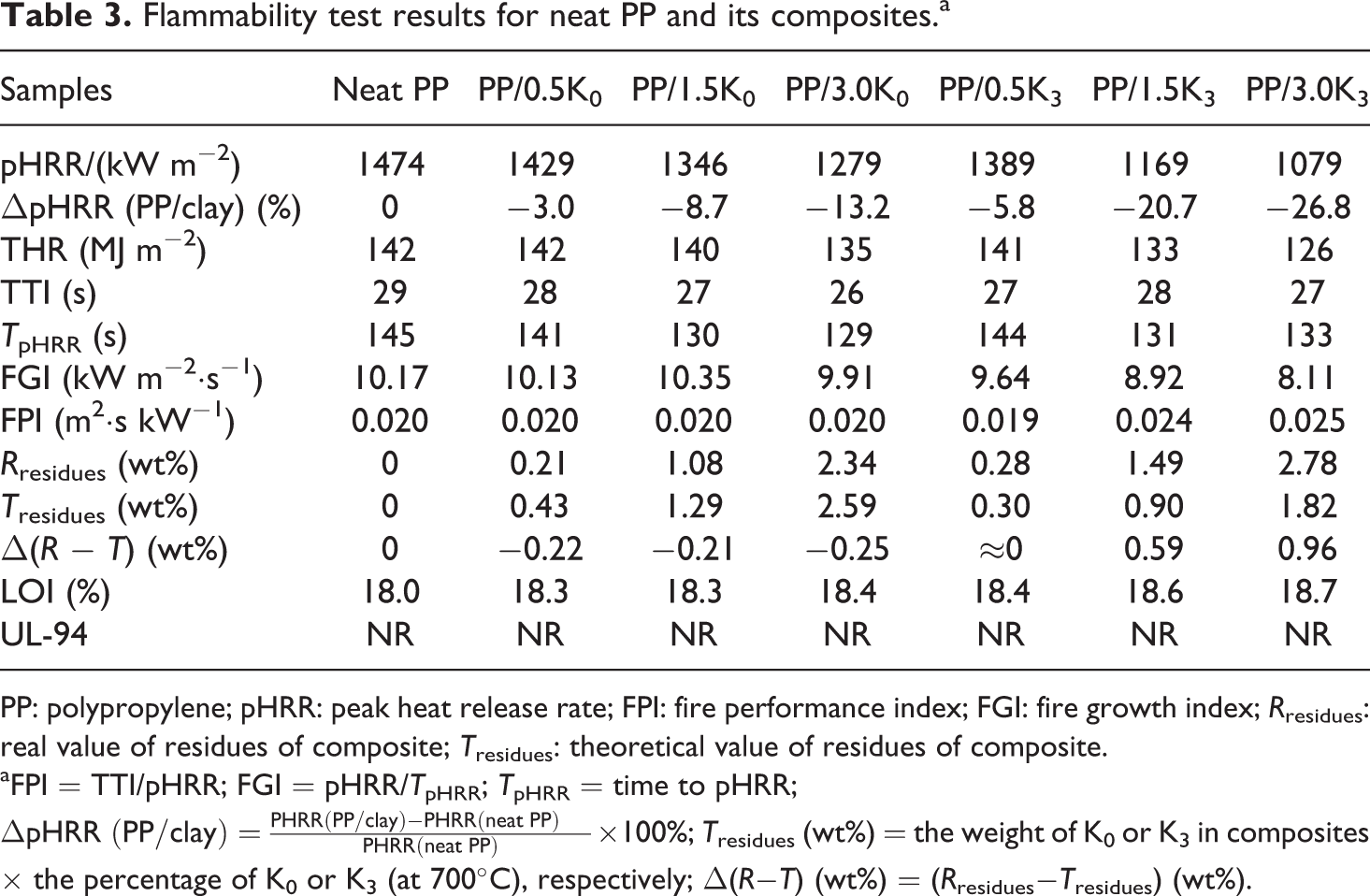
PP: polypropylene; pHRR: peak heat release rate; FPI: fire performance index; FGI: fire growth index; R residues: real value of residues of composite; T residues: theoretical value of residues of composite.
aFPI = TTI/pHRR; FGI = pHRR/T
pHRR; T
pHRR = time to pHRR;
Cone calorimetry
The values of the peak heat release rate (pHRR) and some key data from cone calorimeter are listed in Table 3. It was demonstrated that low amount of clay (0.5 wt% K0 or K3) could not effectively improve the flame retardancy of PP composite (the ΔpHRR (PP/0.5K0) and ΔpHRR (PP/0.5K3) were −3.0% and −5.8%, respectively), and the declining ratio of the ΔpHRR (PP/0.5K3)/ΔpHRR (PP/0.5K0) was only 1.93. The reduction in pHRR value for PP/3 wt% K3 (ΔpHRR (PP/3.0K3) = −26.8%) was significantly higher compared with that for PP/3K0 (ΔpHRR (PP/3.0K0) was −13.2%), and the declining ratio of the ΔpHRR (PP/3.0K3)/ΔpHRR (PP/3.0K0) was only 2.03. It was interesting to note that the declining ratio of the ΔpHRR (PP/1.5K3)/ΔpHRR (PP/1.5K0) was 2.38 after the addition of 1.5 wt% K3 or K0 (the ΔpHRR (PP/1.5K3) and ΔpHRR (PP/1.5K0) were −20.7% and −8.7%, respectively), which was greater than 2.03. Therefore, the presence of 1.5 wt% K3 was regarded as the efficient amount in this work in improving the fire resistance of PP, which was determined by the declining ratio of the ΔpHRR (PP/K3)/ΔpHRR (PP/K0), and the following discussion will focus on samples containing 1.5 wt% K0 or K3. It was suggested that filler could easily achieve homogenous exfoliation and random dispersion at a nanometer level at low contents, whereas the higher content filler loading could aggregate silicate layers. At the same time, nature clay could catalyze the degradation of polymer at high concentration, 1 which may result in the decrease in efficiency of the filler.
The HRR curves for samples containing 1.5 wt% K0 and K3, respectively, are also shown in Figure 5(a). The HRR value of PP/K0 composite was decreased to 1346 kW m− 2 which was only reduced 8.7% compared with that of neat PP (1474 kW m−2). However, the addition of 1.5 wt% K3 can significantly reduce the pHRR to 1169 kW m−2 which was 20.7% less than that of the neat PP. These results demonstrated that K3 was more effective on improving the flame retardancy of PP. The reason could be attributed to two aspects, one was the compatibility between kaolinite and PP was improved by AS intercalation. Good dispersion and good compatibility between modified kaolinite and PP composites could prevent the movement of PP molecular chain during burning; another was the modified kaolinite could not only release the inert gas that reduced the concentration of combustible gas but also provide better barrier effect than raw kaolinite during combustion.
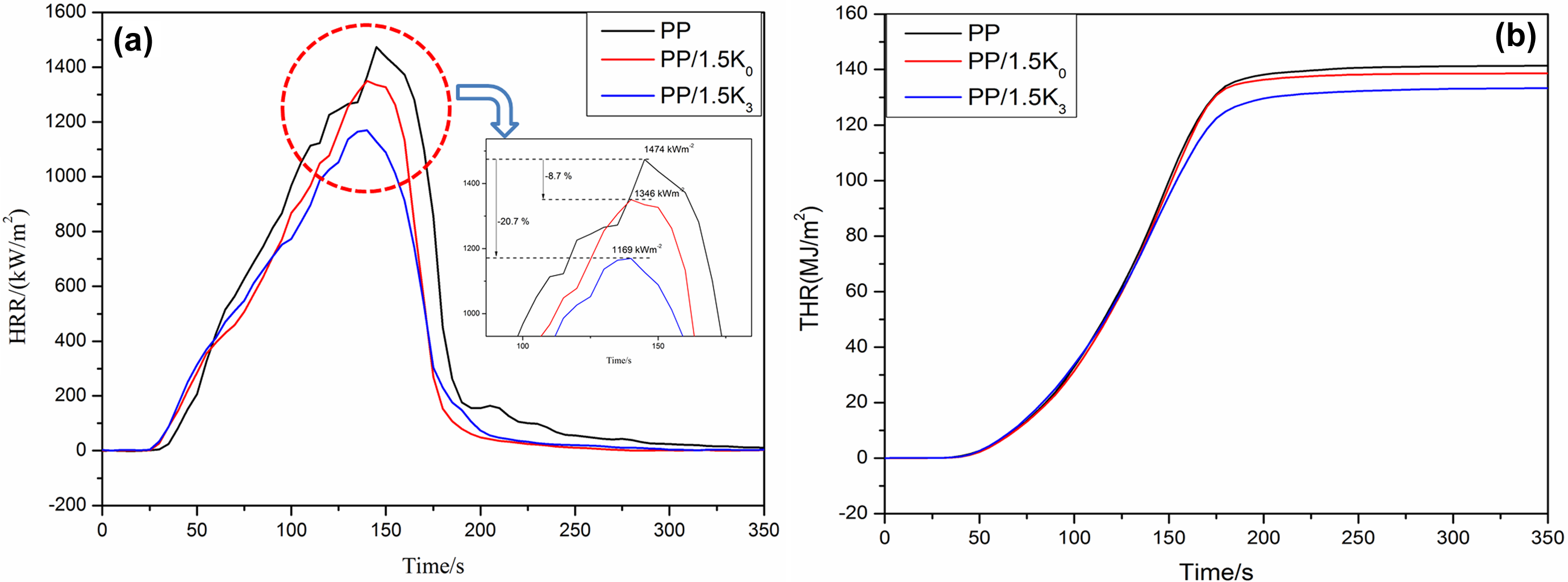
HRR and THR curves of neat PP and its composites.
Figure 5(b) presents the total heat release (THR) curves for neat PP, PP/1.5K0, and PP/1.5K3 samples. It can be observed that after 200 s, the THR value for neat PP was 142 MJ m−2 and it slightly decreased to 140 MJ m−2 for PP/1.5 K0, while it was further decreased to 133 MJ m−2 for PP/1.5K3.
Fire performance index (FPI); TTI/pHRR) and fire growth index (FGI; pHRR/ TPHRR) can also be used to evaluate the fire performance of polymer. 36 FPI value (Table 3) of neat PP, PP/1.5K0, and PP/1.5 K3 was 0.020, 0.020, and 0.024 m2·s kW−1, respectively. The FGI value was slightly increased from 10.17 kW (m·s)−1 for neat PP to 10.35 kW (m·s)−1 for the sample containing 1.5 wt% K0, and the induction value of FGI was minimal. However, the FGI value was reduced to 8.92 kW (m·s)−1 for the sample containing 1.5 wt% K3. It was concluded that K3 was more effective than K0 on improving the flame retardancy of PP than K.
Thermal stability
The TGA curves for the neat PP and PP/kaolinite composites in nitrogen are shown in Figure 6, and characteristic TGA data are collected in Table 4. One can see neat PP started to decompose (5% weight loss = T 0.05) at 420°C. The sample containing 1.5 wt% K0 started to decompose at 421.6°C, and the sample containing 1.5 wt% K3 started to decompose at 432.7°C. The temperature for 50% weight loss (T 0.5) of PP/1.5 K0 (458.9°C) showed a slight increase compared with that of neat PP (455.6°C); however, it showed the same value for the sample containing 1.5 wt% K3 (458.9°C), compared to PP/1.5 K0 composite.
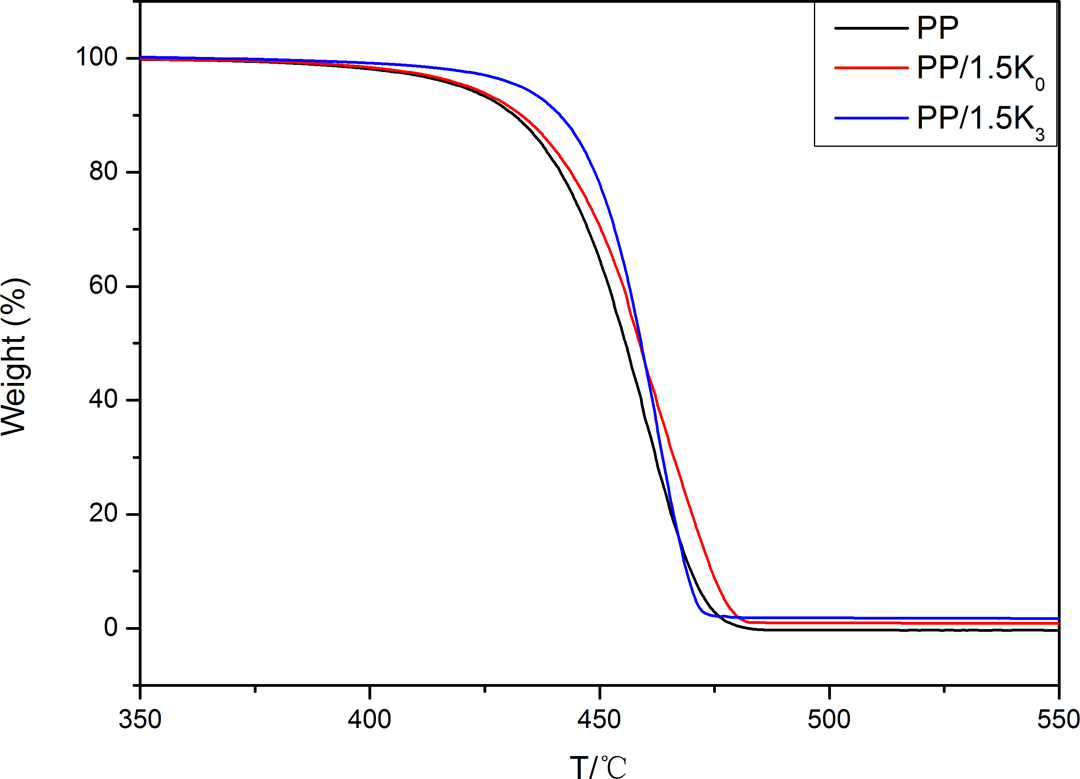
TGA curves of neat PP and its composites.
TGA data of PP/kaolinite composites in nitrogen.a
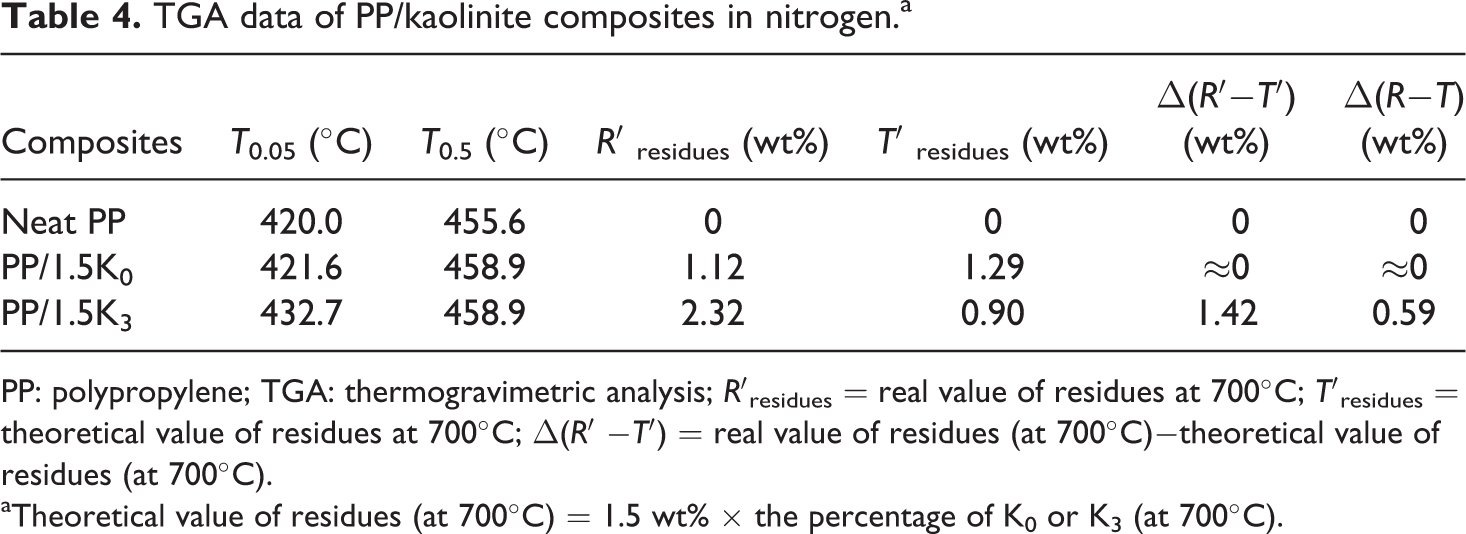
PP: polypropylene; TGA: thermogravimetric analysis; R′residues = real value of residues at 700°C; T′residues = theoretical value of residues at 700°C; Δ(R′ −T′) = real value of residues (at 700°C)−theoretical value of residues (at 700°C).
aTheoretical value of residues (at 700°C) = 1.5 wt% × the percentage of K0 or K3 (at 700°C).
The char residue for neat PP could not be detected at 500°C, whereas the remained char for the sample containing 1.5 wt% K0 or K3 was about 1.1 and 2.3 wt%, respectively, at the same temperature. This showed that K3 could decrease the weight loss and increase the char residue of the PP composite, indicating that K3 could form a better barrier on the matrix than K0 when heated.
Even if AS could be removed from the interlayers of K3 before 400°C (Figure 4(a)), the addition of K3 was so low (only 1.5 wt%) that it was difficult to observe the degradation of K3 in the composite. Therefore, it was suggested that the presence of kaolinite could enhance the initial thermal stability and promote the char formation of PP composites. It was interesting to calculate the mass difference between real value and theoretical value (defined as Δ(R′−T′)) of residues of PP composites. It was found that the Δ(R′−T′) value of PP/K0 composite was near to zero. However, the Δ(R′−T′) value of PP/K3 composite was 1.42 wt%, which was more than the residue amount measured in cone (Δ(R−T) = 0.59 wt%). It was also demonstrated that K3 was more effective than K0 in improving the fire performance of PP.
Digital photos of residues after CONE test
The digital photos of the residues for neat PP, PP/1.5K0, and PP/1.5K3 samples after CONE test are shown in Figure 7. There was no residue left for neat PP after combustion; while some residue was left for PP/1.5K0 and PP/1.5K3. This demonstrated that kaolinite improved the fire performance of PP through barrier effect.

Digital photographs of residues after CONE test.
Distribution of kaolinite
XRD analysis of PP composites
XRD is used to evaluate the effect of K0 and K3 on the crystalline structure of PP matrix and dispersion of K0 and K3 in PP. The XRD patterns of neat PP and PP composites with 1.5 wt% K0 or K3 are shown in Figure 8. The peaks of neat PP at approximately 14.0°, 16.8°, and 18.5° corresponded to the (110), (040), and (130) planes of α crystal of PP, respectively. 37 However, the XRD pattern of PP/1.5K0 was similar to that of PP; the appearance of a new peak at 16.0° in PP/1.5K3 demonstrated the formation of β phase. 21,38,39 It was suggested that modified kaolinite could induce the formation of β phase.
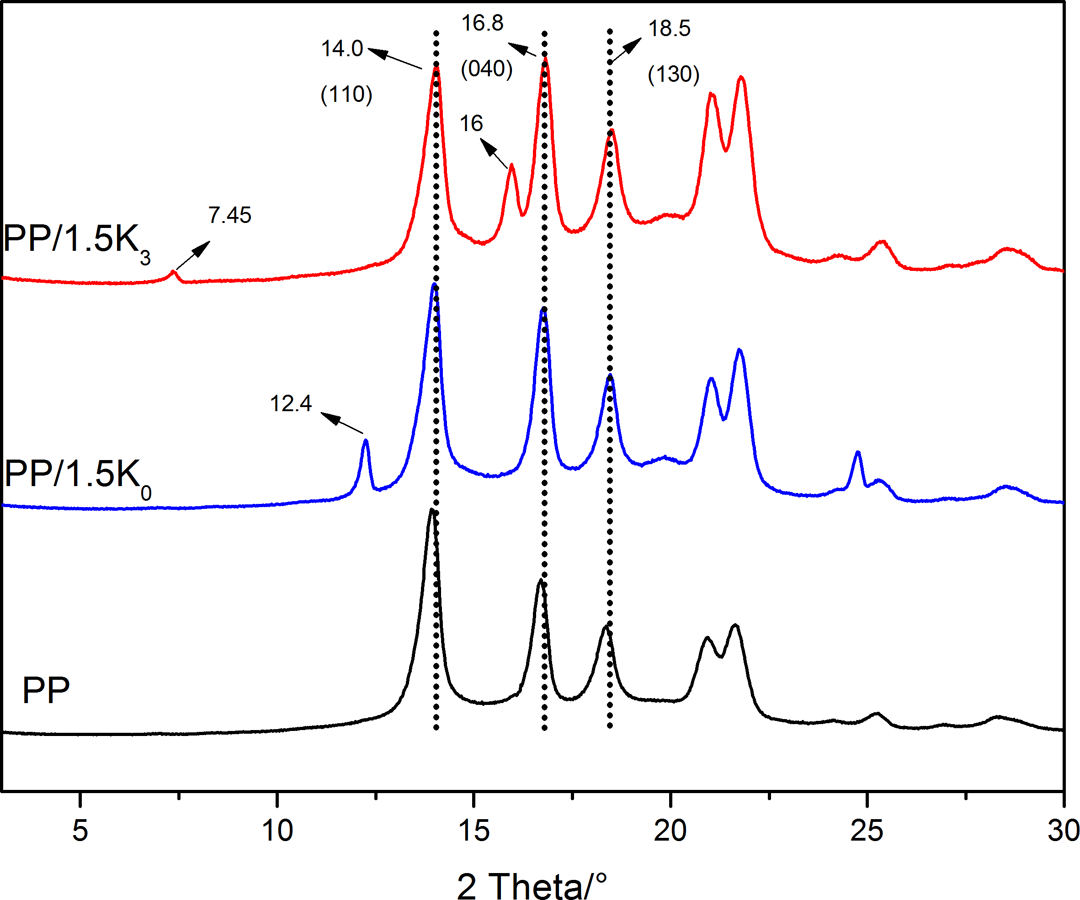
XRD patterns of neat PP and its composites.
It was easily found that the (001) plane of K0 (2θ = 12.4°) still exists in the composite containing K0, which indicated that it was difficult for PP molecular chains to intercalate into the layer of raw kaolinite. However, the 001 plane of K3 (2θ = 7.45°) almost disappeared in the composite containing K3, which indicated the formation of exfoliation structure of K3 in PP during the process of extrusion.
SEM of PP composites
The microstructure of neat PP and PP composites is investigated by SEM. Figure 9 shows the SEM images of PP/1.5K0 and PP /1.5K3. K0 particles with a size range of around 10–20 µm were observed in Figure 9(a), whereas K3 particles could uniformly distribute in the matrix with an average size of less than 5 µm (Figure 9(b)). The images suggested that modified kaolinite had better dispersion in PP matrix than K0, which was consistent with the analysis above.

SEM images of PP composites: (a) PP/1.5K0 and (b) PP/1.5K3.
The SEM and XRD results showed that despite the size of K3 particles dispersed in PP matrix was very small, not all the K3 was exfoliated in PP matrix after the process of extrusion. There still existed aggregate of K3 in the composites.
Mechanical properties of PP composites
The tensile strength of neat PP and PP composites is summarized in Table 5. The tensile strength of neat PP was 35.3 MPa and it was improved by the introduction of kaolinite. K3 was more effective than K0 (40.3 MPa for PP/1.5K3 and 37.1 MPa for PP/1.5 K0) in improving the tensile strength of PP. Improvement of the tensile strength of PP composite was closely related to the small disperser particles, the larger the surface area is, the more the interactions between filler and polymer is. A better dispersion can increase the entanglement that acts as stress arrestor. 37 At the same time, good compatibility between matrix and filler could effectively prevent the movement of molecular chain during the stretching process. The elongation at break of PP composites was reduced from 18.9% of neat PP to 17.4% and 16.5% of PP containing 1.5 wt% K0 and K3, respectively. It was suggested that the rearrangement and orientation of polymer molecules during deformation was inhibited at the presence of K0 or K3, which could cause the reduction in elongation at break.
Tensile strength and the elongation at break of PP and its composites.
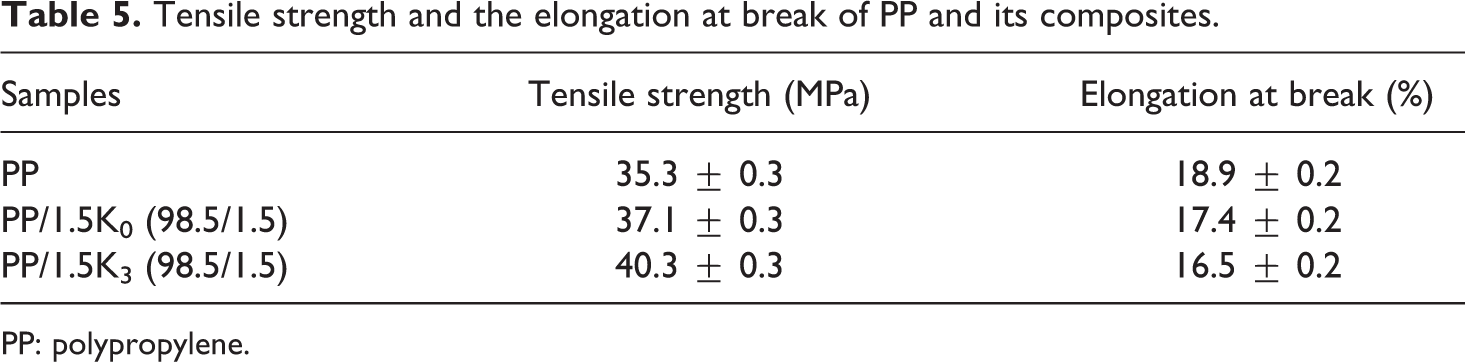
PP: polypropylene.
Flame retardant mechanism
Based on the above discussion, a flame retardant mechanism of PP containing AS-intercalated kaolinite is proposed and illustrated in Figure 10.
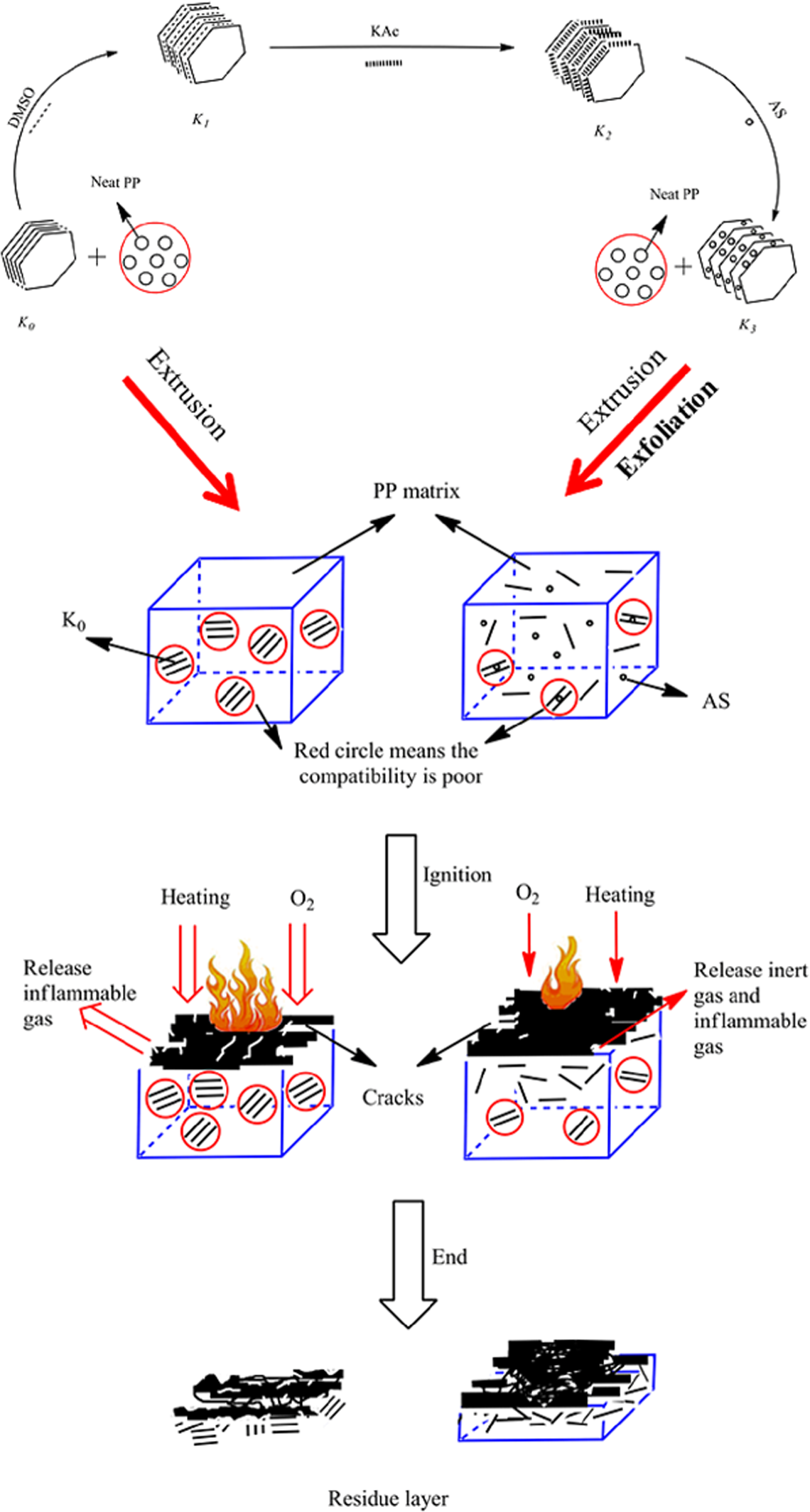
Possible flame retardant mechanism of kaolinite in PP.
The strong shear stress during extrusion was high enough to transform K3 from platy to exfoliation flakes due to the AS intercalation. Moreover, K3 achieved even distribution in PP by the aid of nano-scattered kaolinite lamellas. This variation could effectively prevent the polymer from further degradation mainly through physical barrier 18 during burning.
The results showed that K3 mainly took effect through condensed phase. It was proposed that the modified kaolinite was mainly transformed to exfoliation flakes during extrusion, which was more inclined to emigrate to the surface. 40,41 The clay was emigrated to the surface to form barrier layer during combustion. Similar to the flame retardant mechanism of other nano-lamellas, the emigration was helpful to form thicker and denser char layer, protecting PP from further degradation, 42 and restraining release of toxic gas such as carbon monoxide. Meanwhile, AS could also release ammonia and other inert gas during combustion 43 ; however, the amount of K3 was too low to make significant effect on reducing the flame spreading.
Conclusions
AS-intercalated kaolinite was successfully prepared by a three-step ion exchange reaction. The basal spacing of kaolinite in the XRD curve was increased from 0.72 nm (K0) to 1.2 nm (K3).
The introduction of AS-intercalated kaolinite into PP could significantly improve the flame retardancy and tensile strength of the composite. AS-intercalated kaolinite was more effective than raw kaolinite on reducing the pHRR value of PP composite. The tensile strength of PP composite containing 1.5% K3 (40.3 MPa) was superior to that of the sample containing the same load of K0 (37.1 MPa). It was proposed that AS-intercalated kaolinite could take effect in both solid phases by enhancing the char formation and gas phase through releasing inert gases during burning process.
Footnotes
Authors’ note
HL and WT are joint first authors.
Acknowledgement
The authors would like to thank the National Natural Science Foundation of China and Fundamental Research Funds for the Central Universities for their financial support of this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received their financial support for this research from the National Natural Science Foundation of China (Nos. 21374004 and51373018) and Fundamental Research Funds for the Central Universities (YS201402).

