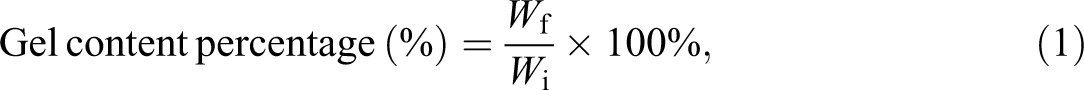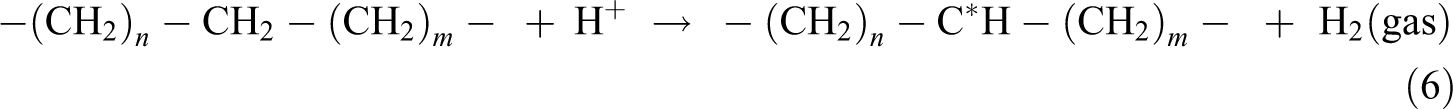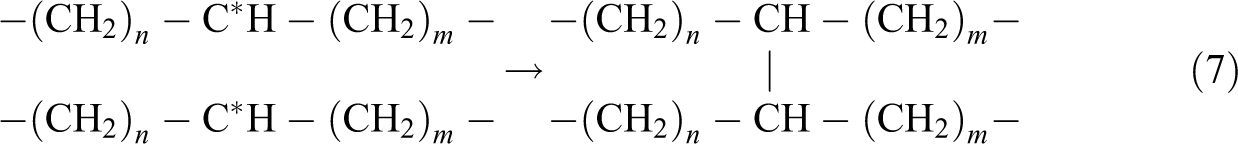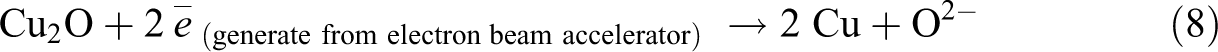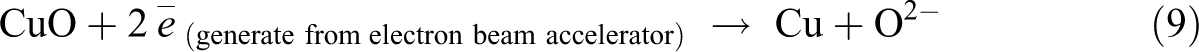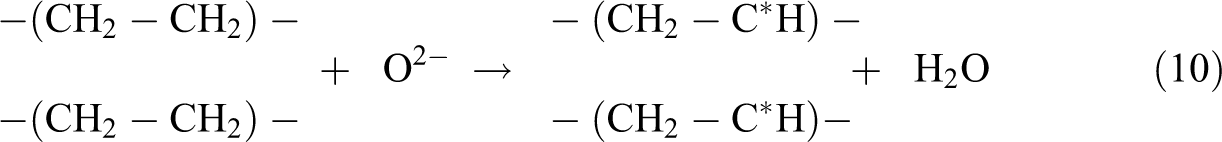Abstract
The aim of this research was to investigate the interaction of electron beam irradiation on the different valence of copper (I) and copper (II) oxides (Cu2O and CuO) added low-density polyethylene (LDPE) composites. The results showed the increasing of Cu2O loading level in replacing the CuO has significantly reduced the gel content (or degree of cross-linking networks) in LDPE matrix. This is due to the poorer effect of Cu2O in inducing the polymeric free radicals. Meanwhile, the application of low irradiation dosage (≤100 kGy) has significantly increased the crystallite size for crystallite peak (110) of all LDPE composites. However, further increment in irradiation dosages from 100 to 300 kGy has gradually reduced the crystallite size of deflection peak (110). The tensile strength of all LDPE composites was gradually decreased with increasing of Cu2O loading level due to agglomeration of Cu2O and CuO particles in LDPE matrix. In addition, the increasing of irradiation dosages on all Cu2O /CuO added LDPE composites has gradually increased the tensile strength by inducing the formation of the cross-linking networks in LDPE matrix. Nevertheless, the increasing of irradiation dosage has gradually decreased the elongation at break of all Cu2O /CuO added LDPE composites. This is due to the higher degree of cross-linking networks in LDPE matrix could restrict the mobility of LDPE macromolecular chains when subjected to straining stress.
Introduction
Over the years, the usage of metallic derivatives in polymer compound for application involving sliding motion has attracted the attention of researchers. 1 It was found that the introduction of additives especially the rigid fillers and reinforcements can affect the wear behavior of polymer compound. According to Bahadur and Schwartz, 2 wide range of inorganic compounds such as molybdenum disulfide, graphite, copper (II) oxide (CuO), copper (II) sulfide (CuS), lead oxide, ferric oxide, and metallic powders has been used as fillers in polymer to reduce wear corresponding to the reducing of coefficient of friction. Bahadur and Tabor 3,4 reported that the addition of CuO, CuS, lead(II, IV) oxide, and graphite can reduce the wear of high-density polyethylene for about 20%. For polytetrafluoroethylene, the rate of wear has significantly reduced with the incorporation of zinc, nickel, selenium, yttrium oxide, and silicon dioxide. 5 Generally, the reduction of wear by the addition of rigid metal filler is due to the modification of counterface surface and also because the fiber supported the applied load. 6 Report explained that the metal filler typically CuO can form microfragments to show roll-like structure than reduce the wear instantaneously. 7 Nevertheless, the addition of incompatible metal filler may lead to deterioration of the polymer composite. 8 This is especially important in the wear-engineering application that alternatives need to be taken such as through implementation of cross-linking structure in order to further enhance its ability to withstand harsh applications.
In addition to incorporation of additives to improve polymer properties, radiation cross-linking is also one of the methods used to enhance the properties of polymer by introducing the formation of cross-linking networks in polymer matrix. 9 –14 Generally, there are various forms of high-energy irradiations such as electron beam irradiation, gamma rays irradiation, and X-ray irradiation which are currently used to modify the properties of polymer materials. 15 The application of irradiation technique on polymers can induce the formation of cross-linking by generating polymeric free radicals in polymer matrix. These free radicals could cross-link two long macromolecule chains by forming C–C intermolecular bonds to modify the properties of polymers. In general, two important mechanisms, such as cross-linking reaction and chain scissioning reaction, would be occurred during the irradiation process of polymers. The dominant mechanism could affect the changes in matrix structure and also in mechanical properties of polymers. 16,17 The mechanical–structural properties of polymer matrix could be significantly enhanced when the cross-linking reaction is more dominance than chain scissioning reaction during the irradiation process. This is due to the formation of cross-linked networks in polymer matrix which could significantly increase the molecular weights of polymers. 18 On the other hand, the chain scissioning reaction is predominated during the irradiation process. The polymer matrix could be severely degraded by breaking down the macromolecules in polymer into smaller sizes. Thus, this could further reduce the molecular weight of polymer and weaken the mechanical–structural properties of polymer. 18 In order to achieve the predomination of cross-linking reaction compared to chain scissioning reaction during irradiation process, the application of suitable irradiation dosage is important for optimizing the release of high-energy free radicals to achieve the effective cross-linking.
According to previous studies, 11,19 low-density polyethylene (LDPE) tends to cross-link and form a three-dimensional polymer network structure when exposed to high-energy electron beam irradiation. The application of electron beam irradiation dosage up to 300 kGy on LDPE could induce the formation of cross-linking networks in LDPE matrix without undergoing degradation. LDPE polymer also can be easily cross-linked without the addition of any cross-linking agents due to its highly branched structure. 20 The high radiation resistance of LDPE enables the electron beam irradiation technique to be used in accomplishing the improvement of LDPE properties. Generally, the formation of cross-linking networks in LDPE matrix by high-energy irradiation involves two main mechanism stages. 21 The initial stage of cross-linking mechanism takes place by breaking down of C–H bond on LDPE chains to form free radicals by releasing hydrogen atom. This is followed by releasing the hydrogen atom that would combine with other hydrogen atom to form hydrogen gas. The generated free radicals from initial stage would react and combine together to form cross-linking network during the second stage of cross-linking mechanism. 21 In short, the electron beam irradiation of LDPE could easily generate free radicals and the generated free radicals could form three-dimensional networks in LDPE matrix by releasing hydrogen gas. The increasing of irradiation dosage could rapidly induce the degree of cross-linking in LDPE matrix. Such cross-linked LDPE exhibits outstanding mechanical properties, such as tensile strength, elongation, and so on, and electrical properties. 19,22,23 In this study, polymer matrix composite was prepared from blending of different valence of Cu2O and CuO with LDPE to investigate its reactivity when undergone electron beam irradiation. The outcomes of this work are important to provide an understanding on the reactivity of transition metal oxide in engineering material typically involving moving parts applications.
Experimental
Materials
LDPE (with commercial grade of NM 1209 P3Y) was manufactured by Petlin Sdn. Bhd (Malaysia) and used as polymer base in this study. Copper (I) oxide (Cu2O) and CuO with analytical reagent grade were supplied by ChemSolutions Sdn. Bhd (Malaysia). Cu2O and CuO were used as hybrid degradation promoter fillers in this study. All materials were used without further purification.
Preparation of samples
LDPE was compounded with Cu2O and CuO using Brabender mixer (Germany) at the heating temperature and rotor speed of 135°C and 50 r min−1, respectively, for a mixing time of 8 min. The loading level ratios of Cu2O:CuO used during the compounding process were 1 phr:4 phr, 2 phr:3 phr, 3 phr:2 phr, and 4 phr:1 phr, respectively. The compounded LDPE samples were further compression molded into 1-mm thickness sheet using compression molding machine at heating temperature of 185°C for 10 min. The compression molding machine was operated under compression pressure of 10 MPa. After that, the compression molded samples were further cooled down to room temperature at the pressure of 10 MPa for 2 min using cold press with cooling rate of 15°C min−1. The 1-mm thickness sheets were then cut into required shapes using suitable sample cutter. The cut samples were electron beam irradiated at the room temperature under accelerating voltage of 175 kV to 50 kGy, 100 kGy, 150 kGy, 200 kGy, and 250 kGy with the electron beam irradiation rate of 50 kGy per pass.
Gel fraction
The gel content test was carried out by immersing the samples gravimetrically in hot xylene solvent in accordance to ASTM D2765 standard to determine the degree of cross-linking networks in polymer matrix induced by electron beam irradiation. Initially, the initial weight (W i) of samples were accurately weighed and measured by using analytical balance. The weighted samples were then immersed and heated in hot xylene solvent at the temperature of 120°C for 24 h extraction. The remaining samples after the extraction process were washed and rinsed with clean xylene for several times to remove the stain of soluble materials on the extracted samples. After that, these samples were rinsed again with methanol at least twice and soaked in methanol for 20 min. The washed and cleaned samples were dried in a vacuum oven to constant weight at temperature of 60°C for 6 h. The dried samples or insoluble materials were weighed using analytical balance to obtain the weight of insoluble materials (W f). The gel content percentage of the sample was calculated as shown in equation (1). The final value of gel content percentage for each sample was taken from the average of three specimens.
where W i is the initial weight of the samples before the extraction process (g) and W f is the final weight of the dried remaining samples after the extraction process (g).
Hot set test
The hot set test was conducted according to standard of DIN 57472 Part 602 VDE 0472 by using a hot set oven from Heraeus (UT 6050 HS, Germany). The hot set test is carried out to investigate the degree of cross-linking in polymer matrix of samples by determining the creep elongation of samples under a static load of 10 N mm−2 and heating at a temperature of 200°C. The 1-mm thickness sheet was cut into dumbbell shape specimens using dumbbell cutter. The initial gauge length was marked on the dumbbell specimens. The creep elongation between the two marks on the dumbbell specimens was measured after 15 min of heating process in hot set oven. The final creep elongation value of samples was taken and recorded as an average of three specimens.
XRD test
The crystallinity and crystalline structures of all Cu2O/CuO added LDPE composites were evaluated using X-ray diffraction (XRD). The XRD spectra of all LDPE samples were scanned using a Shimadzu XRD 6000 diffractometer (Japan) with copper K α (Cu-K α) radiation (λ = 0.1542 nm or 1.542 Å) for a 2θ range of 0° to 40°. All the LDPE samples were scanned at a scanning rate of 1.2° min−1. The Cu-Kα radiation generator was operated at accelerating voltage and current of 30 kV and 30 mA, respectively. The degree of crystallinity was calculated as the percentage of the scattered intensity of the crystalline phase over the scattered intensity of the crystalline and amorphous phase. The crystalline size, L was derived from the peak angle (2θ) on the XRD diffractogram in accordance to the Scherrer equation as shown in the following equation:
where k is Scherrer constant, β is the full width at half maxima of the diffraction peak, λ is wavelength of Cu irradiation (λ = 0.1542 nm or 1.542 Å), and 2θ is the angle of diffraction peak in radians. On the other hand, the d-spacing of crystallite structure was calculated using the Bragg’s equation as shown in equation (3), while the interchain separation, R of crystalline structure was calculated using Klug and Alexander equation as shown in the following equation:
where 2θ is the angle of diffraction peak in radians and λ is 0.1542 nm or 1.542 Å.
Tensile test
The tensile test was conducted to investigate the tensile properties of LDPE samples under straining stress. The 1-mm thickness sheets of LDPE samples were cut into dumbbell shape using dumbbell cutter in accordance to ASTM D1822 standard. The dumbbell specimens were tested using Instron Universal Testing Machine (model of 4302 Series IX) under straining load of 2 kN with crosshead speed of 50 mm min−1. The gauge length of all the dumbbell specimens was fixed at 10 mm. The gauge thickness and width of dumbbell specimens were measured prior the tensile testing. The final values of tensile strength and elongation at break were taken as an average of six specimens.
SEM analysis
Scanning electron microscopy (SEM) test was conducted using Hitachi S-3400 N Scanning Electronic Microscope (Japan). The fractured surfaces of the tensile tested samples were cut into small portions before performing the scanning. The cut samples were then placed and mounted onto the copper stub with the fractured surface facing up. The mounted samples were coated with a thin layer of palladium and gold using EMITECH SC7620 sputter coater. The scanning of the samples was performed at an electron beam voltage of 15 kV.
Results and discussion
Gel fraction
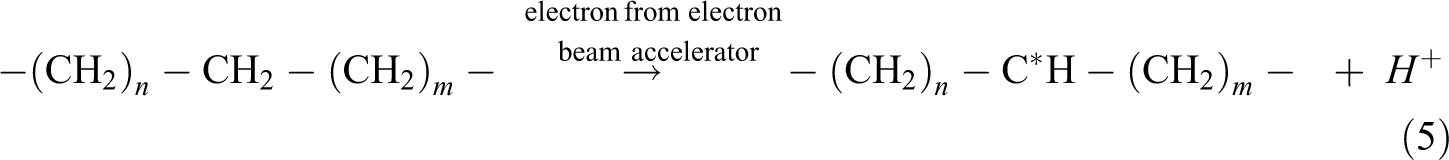
The gel content test was conducted to evaluate the degree of cross-linking formed by electron beam irradiation in polymer matrix. By referring to Figure 1, all the unirradiated Cu2O/CuO added LDPE composites were fully dissolved in hot xylene solution after 24 h of extraction process. This is due to the absence of cross-linking networks in all unirradiated Cu2O/CuO added LDPE matrix caused the LDPE matrix unable to withstand the solvent attack of hot xylene. 24,25 On the other hand, the gel content of all the Cu2O/CuO added LDPE composites was rapidly increased when electron beam irradiated up to 50 kGy as shown in Figure 1. The electron beam irradiation process could induce the formation of three-dimensional networks in LDPE matrix by generating polymeric free radicals in LDPE matrix. 26 The electron generated by electron beam accelerator could attack the C–H bonding on LDPE macromolecular chains in amorphous regions of LDPE matrix by generating polymeric free radicals and hydrogen ion as shown in equation (5). 24,27 The generated hydrogen ions could further extract the hydrogen atoms from the other LDPE macromolecular chains to form gas hydrogen by generating new free radicals as shown in equation (6). 24 –27 The generated polymeric free radicals would react together to form a three-dimensional network (also known as cross-linking networks) in LDPE matrix as shown in equation (7). 27 The formation of three-dimensional networks in LDPE matrix could highly reduce the solubility of LDPE matrix into hot xylene solvent. 26
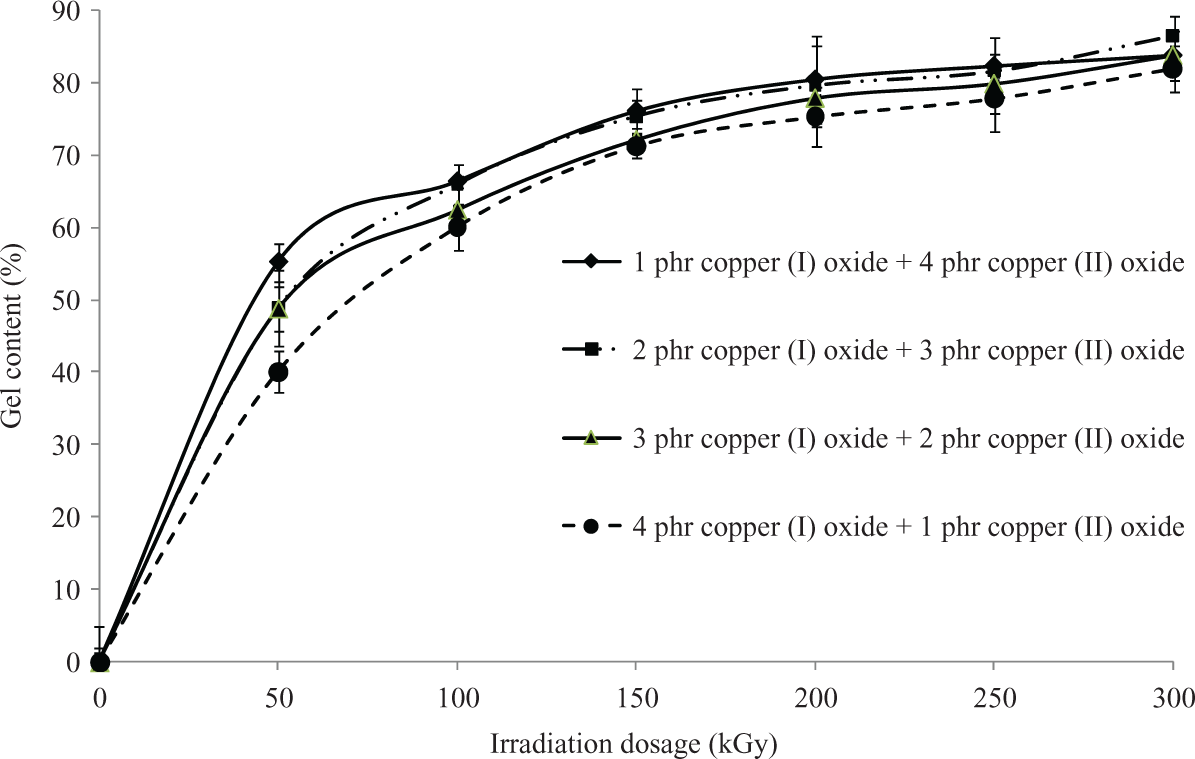
Gel content of LDPE composites added with various loading level ratio of Cu2O/CuO when subjected to increasing irradiation dosages. LDPE: low-density polyethylene; Cu2O: copper (I) oxide; CuO: copper (II) oxide.
However, the extent of increment of gel content for all Cu2O/CuO added LDPE composites was observed to be significantly reduced with further increasing the irradiation dosage from 50 kGy to 300 kGy. The already cross-linked networks in LDPE matrix has entrapped the polymeric free radicals generated by electron beam irradiation by restricting the migration of generated polymer-free radicals to the amorphous regions of LDPE matrix. 28,29 This further caused the entrapped polymeric free radicals tended to form chemical bonds inside the already cross-linked networks. 24,29 Subsequently, the formation of cross-linking bonds inside the cross-linked networks did not provide a significant increment in the degree of cross-linking or gel content of all Cu2O/CuO added LDPE composites. Thus, the increment rate of gel content for all LDPE composites was significantly lower with further increasing of irradiation dosages from 50 kGy to 300 kGy.
By referring to Figure 1, the increasing of Cu2O loading level (from 1 phr to 4 phr) in replacing the loading level of CuO was observed to significantly reduce the gel content of LDPE composites at all irradiation dosages. The higher molecular weight of Cu2O in comparing to CuO is the main factor in causing the reduction of gel content of LDPE composites when added with increasing of Cu2O loading level. Under electron beam irradiation process, the Cu2O and CuO in LDPE matrix are tended to experience a direct decomposition process by an electron beam energy source. 30 The direct decomposition of Cu2O and CuO in LDPE matrix could produce an active radicals of O2− as illustrated in equations (8) and (9). 30 Subsequently, the production of these O2− radicals could attack the LDPE macromolecular chains in LDPE matrix to form polymeric free radicals as indicated in equation (10). These active O2− radicals could extract hydrogen atom from two LDPE macromolecular chains to form water vapor by generating two polymeric free radicals. 30 The addition of Cu2O and CuO into LDPE matrix has significantly induced the release of polymeric free radicals when subjected to electron beam irradiation. Thus, the degree of cross-linking networks formed in LDPE matrix (or gel content) also can be slightly increased when added with Cu2O and CuO as fillers.
By comparing the direct decomposition reactions of Cu2O and CuO (as shown in equations (8) and (9)), the Cu2O with higher molecular weight might need in higher concentration than CuO in order to produce the equivalent effective O2− radicals. The increasing of Cu2O loading level in replacing the CuO loading level in LDPE matrix has significantly reduced the amounts of effective O2− radicals produced in LDPE matrix. Subsequently, the polymeric free radicals produced by the attack of effective O2− radicals also have been reduced in amounts and this further decreased the degree of cross-linking networks formed in LDPE matrix. Thus, the gel content of LDPE composites was significantly reduced when the CuO loading level added into LDPE matrix was gradually replaced by Cu2O.
Hot set test
The effects of electron beam irradiation on the hot set results for all unirradiated and irradiated Cu2O/CuO added LDPE composites when subjected to static load of 10 N cm−2 and heating temperature of 200°C are illustrated in Table 1. All the unirradiated copper (1) oxide/CuO added LDPE composites were observed to fail hot set test immediately when subjected to static load of 10 N cm−2 under high heating temperature. This is due to the absence of cross-linking networks in the unirradiated LDPE matrix that has caused the LDPE macromolecular chains in polymer matrix to slide freely on each other without resistance when subjected to the static load under high heating temperature. 26,28 This finding also found to be in agreement with the gel content results, where no gel content (degree of cross-linking) was detected in the LDPE matrix of all unirradiated LDPE composites.
Hot set results of all different loading level ratios of Cu2O/CuO added LDPE composites when subjected to various electron beam irradiation dosages.
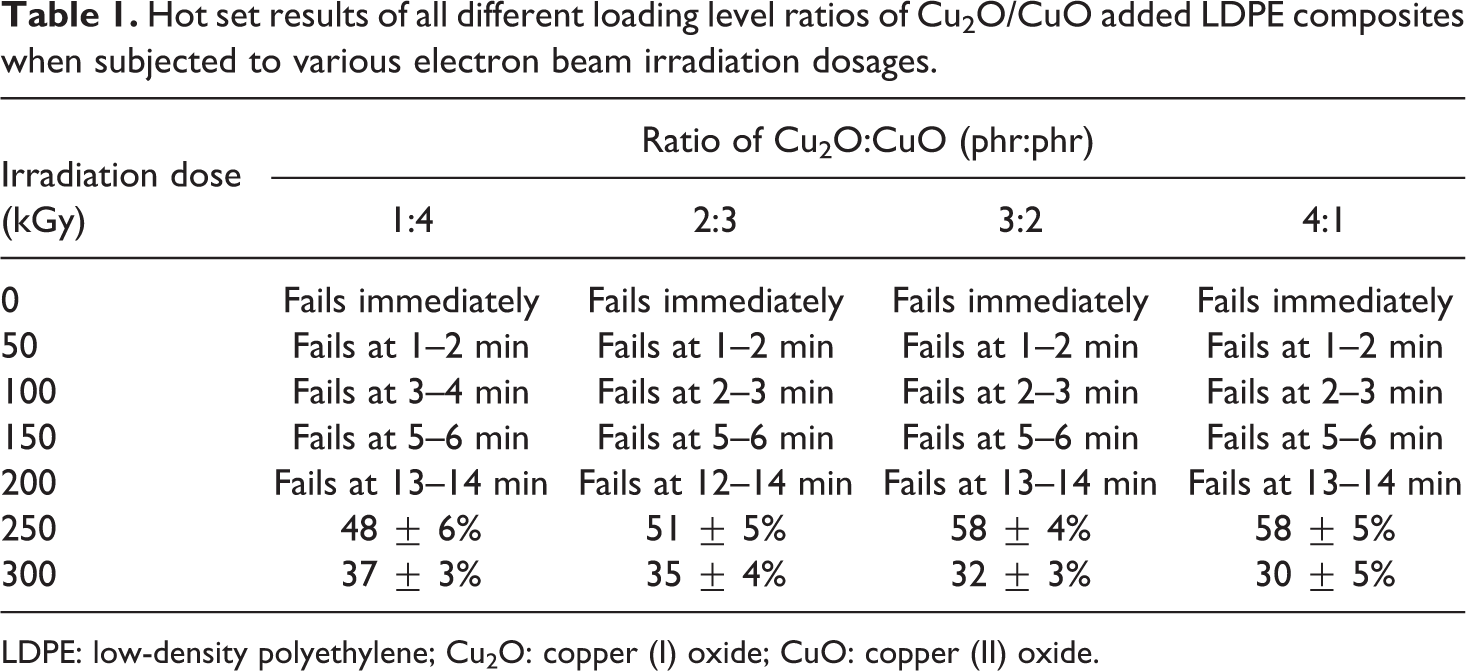
LDPE: low-density polyethylene; Cu2O: copper (I) oxide; CuO: copper (II) oxide.
By referring to Table 1, the failing time of all Cu2O/CuO added LDPE composites was observed to slightly delay by 1–2 min when electron beam irradiated up to 50 kGy. When the electron beam irradiation dosages increased from 50 kGy to 200 kGy, all the Cu2O/CuO added LDPE composites still remained falling the hot set test within 15 min when subjected to static load under high temperature. The failing time of all Cu2O/CuO added LDPE composites was gradually delayed up to 5–6 min when further irradiated from 50 kGy to 150 kGy as shown in Table 1. This also indicates that the increment in electron beam irradiation dosages from 50 kGy to 150 kGy could increase the resistance ability of LDPE matrix by increasing the degree of cross-linking formed in LDPE matrix. According to Table 1, further increment of electron beam irradiation dosage up to 200 kGy has significantly delayed the failure time of all Cu2O/CuO added LDPE composites up to 13–14 min. The increasing of electron beam irradiation dosages applied on LDPE matrix has effectively induced the formation of cross-linking networks in LDPE matrix by generating more polymeric free radicals inside LDPE matrix in accordance to reactions shown by equations (5) to (7). 24,31 This is due to the occurrence of cross-linking networks in LDPE matrix could highly restrict the mobility of LDPE macromolecular chains in LDPE matrix when subjected to static load and high temperature. Subsequently, the higher degree of cross-linking networks formed in LDPE matrix by electron beam irradiation has effectively prolonged the resistance time of LDPE matrix against the applied static load under high temperature.
By referring to Table 1, the creep elongation of all Cu2O/CuO added LDPE composites was observed to gradually decrease when all the Cu2O/CuO added LDPE composites were further electron beam irradiated from 200 kGy to 300 kGy. As discussed in earlier, the increasing of electron beam irradiation dosages could significantly induce the formation of polymeric free radicals inside LDPE matrix. The polymeric free radicals in LDPE matrix would further react to each other by forming the cross-linked networks in LDPE matrix. 24 The formation of cross-linked networks in LDPE matrix could further restrict the slippage effect of LDPE macromolecule chains in LDPE matrix when subjected to static load. 31 Thus, the elongation ability of all Cu2O/CuO added LDPE composites has been significantly reduced with increasing of electron beam irradiation dosages from 250 kGy to 300 Gy.
By referring to Table 1, the increasing of Cu2O loading level from 1 phr to 4 phr (to replace the CuO loading levels) in Cu2O/CuO added LDPE composites has gradually increased the creep elongation of LDPE composites from 48% to 58%. This could be due to higher concentration of Cu2O that is needed in order to produce the equivalent effective amounts of reactive O2− radicals as generated by CuO due to the higher molecular weight of Cu2O. The replacement of CuO with Cu2O could further reduce the polymeric free radicals generated in LDPE matrix when subjected to electron beam irradiation in accordance to reaction mechanism as shown in equations (8), (9), and (10). 30 The lower polymeric free radicals amounts generated in LDPE matrix has slightly decreased the degree of cross-linking networks in Cu2O/CuO added LDPE composites. Subsequently, the creep elongation also has been gradually reduced with increasing of Cu2O loading level. However, the creep elongation of all Cu2O/CuO added LDPE composite was observed to significantly reduce when further electron beam irradiated from 250 kGy to 300 kGy. This is due to the increasing of electron beam irradiation dosage up to 300 kGy could further inducing the release of polymeric free radicals inside the already cross-linked chains. The mobility of these polymeric free radicals has been highly restricted by the already cross-linked chains and this further caused these polymeric free radicals tended to form the cross-linking bonding inside the already cross-linked chains. Thus, the formation of more cross-linking bonds in already cross-linked has further restricted the mobility of LDPE macromolecular chains when subjected to static load at high temperature. However, the creep elongation of 300 kGy irradiated LDPE composites gradually increased when the loading level of Cu2O further increased from 1 phr to 4 phr. At higher irradiation dosages (≥300 kGy), the higher CuO loading level in LDPE matrix would generate more polymeric free radicals in compared to Cu2O. The generated polymeric free radicals tended to attack the main backbone of LDPE macromolecular chains where this process promotes the occurrence of chain scissioning process instead of cross-linking. 30,32 The restriction of LDPE chains mobility by cross-linking networks has significantly reduced due to the occurrence of chains scissioning process. Thus, the creep elongation of 300 kGy irradiated (higher irradiation dosages) LDPE composites was gradually increased when the Cu2O loading level increased from 1 phr to 4 phr.
XRD analysis
The XRD patterns of LDPE composites added with various loading levels ratios of Cu2O/CuO when irradiated at various irradiation dosages are shown in Figures 2 and 3. By referring to Figures 2 and 3, three significant diffraction peaks of (110), (200), and (020) can be observed on the XRD curves of Cu2O and CuO added LDPE composites at 2θ ≈ 21.5°, 23.7°, and 36.4°, respectively. 33 The increasing of Cu2O loading level from 1 phr to 3 phr (in replacing CuO loading level) has significantly improved the peak intensity of deflection peak (020) on XRD curves of LDPE composites as shown in Figure 2(a). The increasing in peak intensity of peak (020) represents the convergence of random polymer chains arrangement structures in LDPE matrix towards highly ordered arrangement. 34 Besides, the peak width of deflection peak (020) on XRD curves also observed to be narrower when the loading level of Cu2O added in LDPE matrix was gradually increased from 1 phr to 3 phr. The narrowing effect of deflection peak (020) is associated with the increment in the average crystallite size. 34 However, further increment of Cu2O loading level from 3 phr to 4 phr has significantly reduced the peak intensity of deflection peaks (020) as shown in Figure 2(a). This also indicates that the irregularity arrangement of Cu2O and CuO particles in LDPE matrix could slightly disturb the highly ordered chains structure arrangement in LDPE matrix. Besides, the intensity of deflection peak (020) for all 100 kGy and 300 kGy irradiated Cu2O and CuO added LDPE composites has significantly reduced with higher Cu2O loading level. This is because the replacement of CuO with Cu2O into LDPE matrix could help in converging the random chains arrangement into highly ordered arrangement inside polymer matrix. The increasing of Cu2O loading level also observed to broaden the deflection peaks (110) and (200) of unirradiated LDPE composites as shown in Figure 2(a). This also indicates that the increasing of Cu2O loading level could lead the reduction in average crystallite size. By referring to Figure 3, the peak intensity of deflection peak (110) of all Cu2O and CuO added LDPE composites has significantly reduced when subjected to increasing of irradiation dosages. Furthermore, the deflection peak of all Cu2O and CuO added LDPE composites also found to be broadened with increasing of irradiation dosages. This indicates that the application of electron beam irradiation on all Cu2O and CuO added LDPE composites could disturb and rupture the highly ordered chains in the crystallite of peak (110). This could further reduce the interaction effect between the existing crystallite in LDPE matrix and the newly formed crystallites and thus broaden the deflection peak of (110).
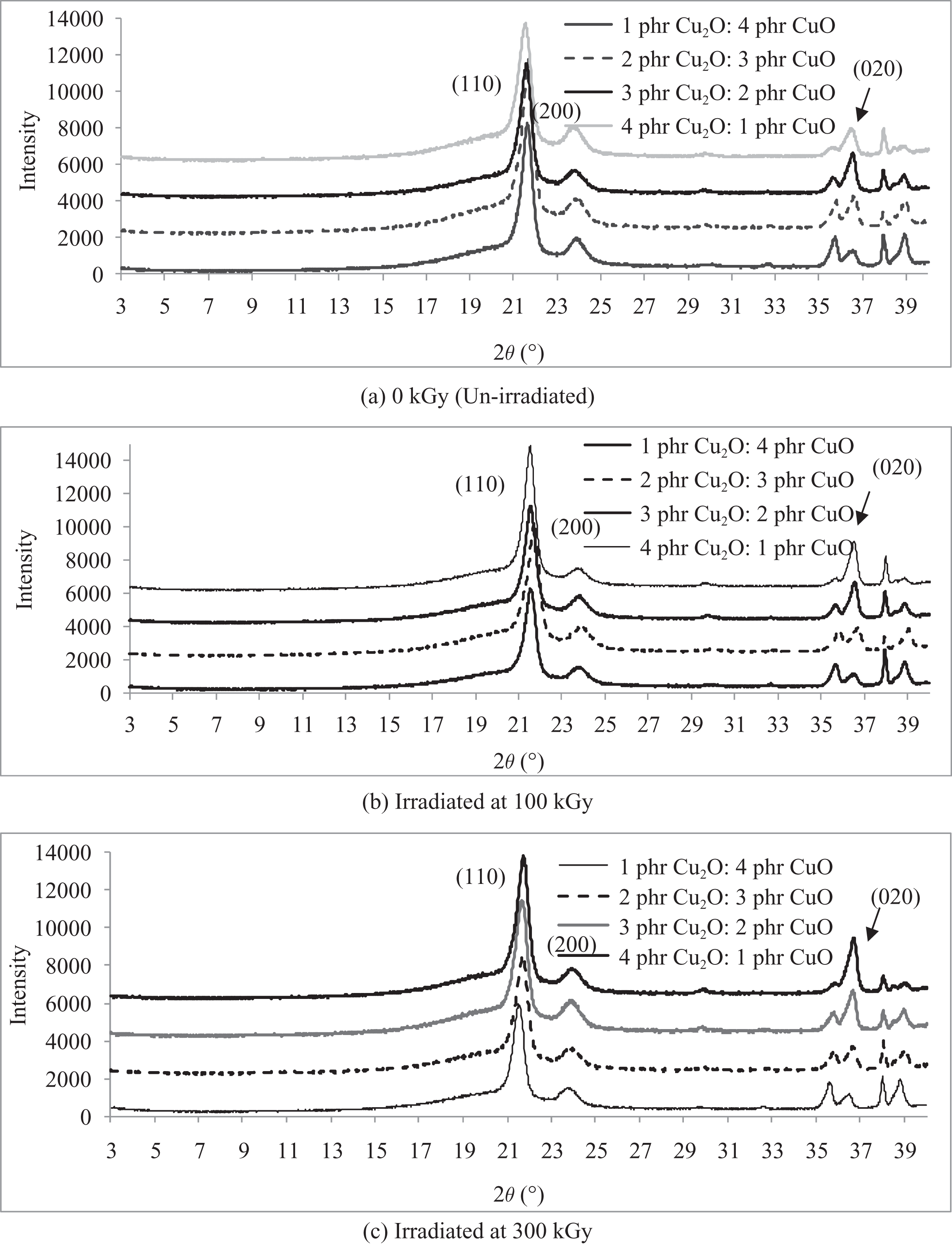
XRD pattern curves of LDPE composites added with various loading level ratio of Cu2O /CuO when subjected to (a) 0 kGy (unirradiated), (b) 100 kGy, and (c) 300 kGy. LDPE: low-density polyethylene; XRD: X-ray diffraction; Cu2O: copper (I) oxide; CuO: copper (II) oxide.
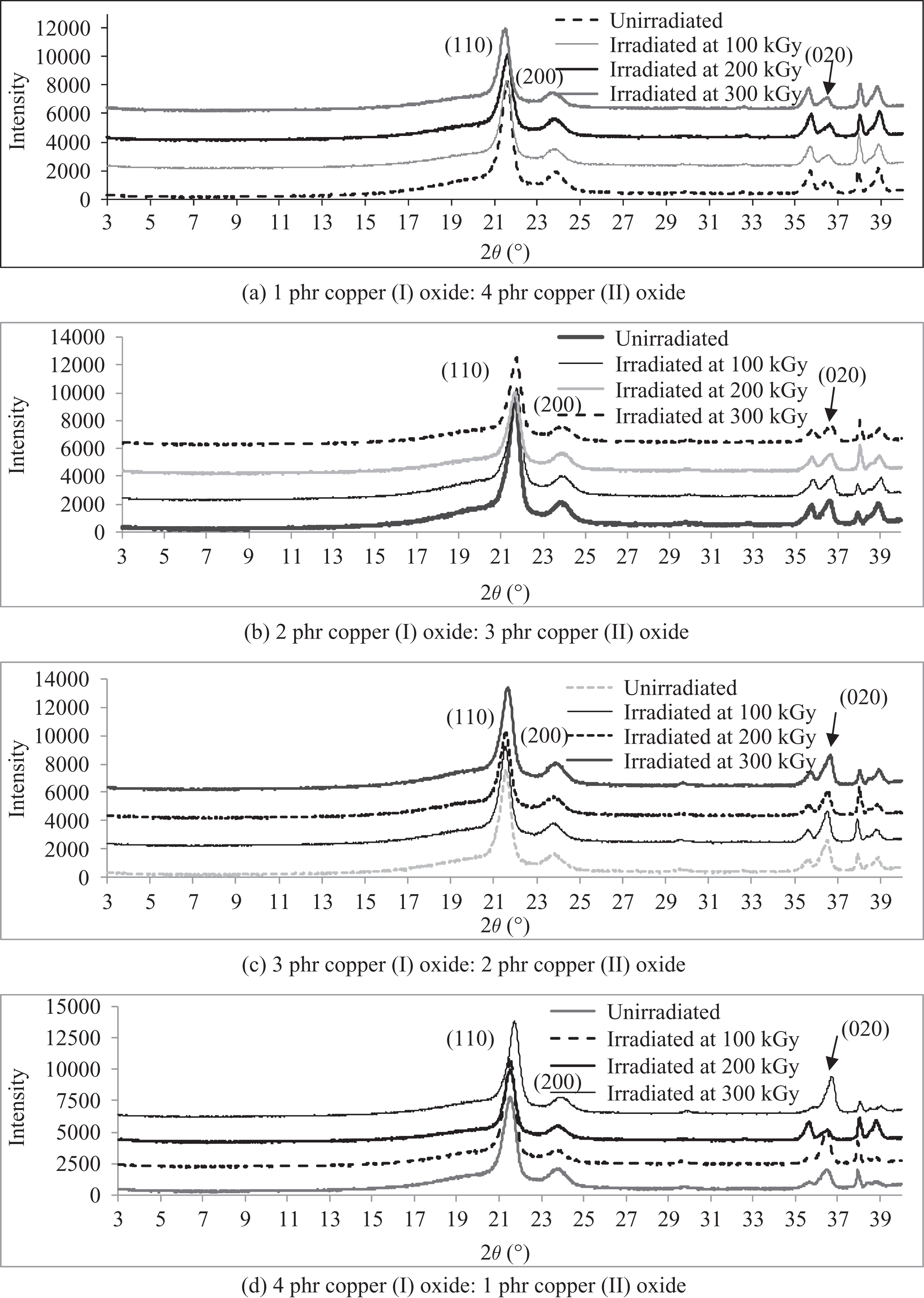
XRD pattern curves of LDPE composites added with (a) 1 phr Cu2O: 4 phr copper (II) oxide; (b) 2 phr Cu2O:3 phr CuO; (c) 3 phr Cu2O:2 phr CuO; and (d) 4 phr Cu2O:1 phr CuO when subjected to various irradiation dosages. LDPE: low-density polyethylene; XRD: X-ray diffraction; Cu2O: copper (I) oxide; CuO: copper (II) oxide.
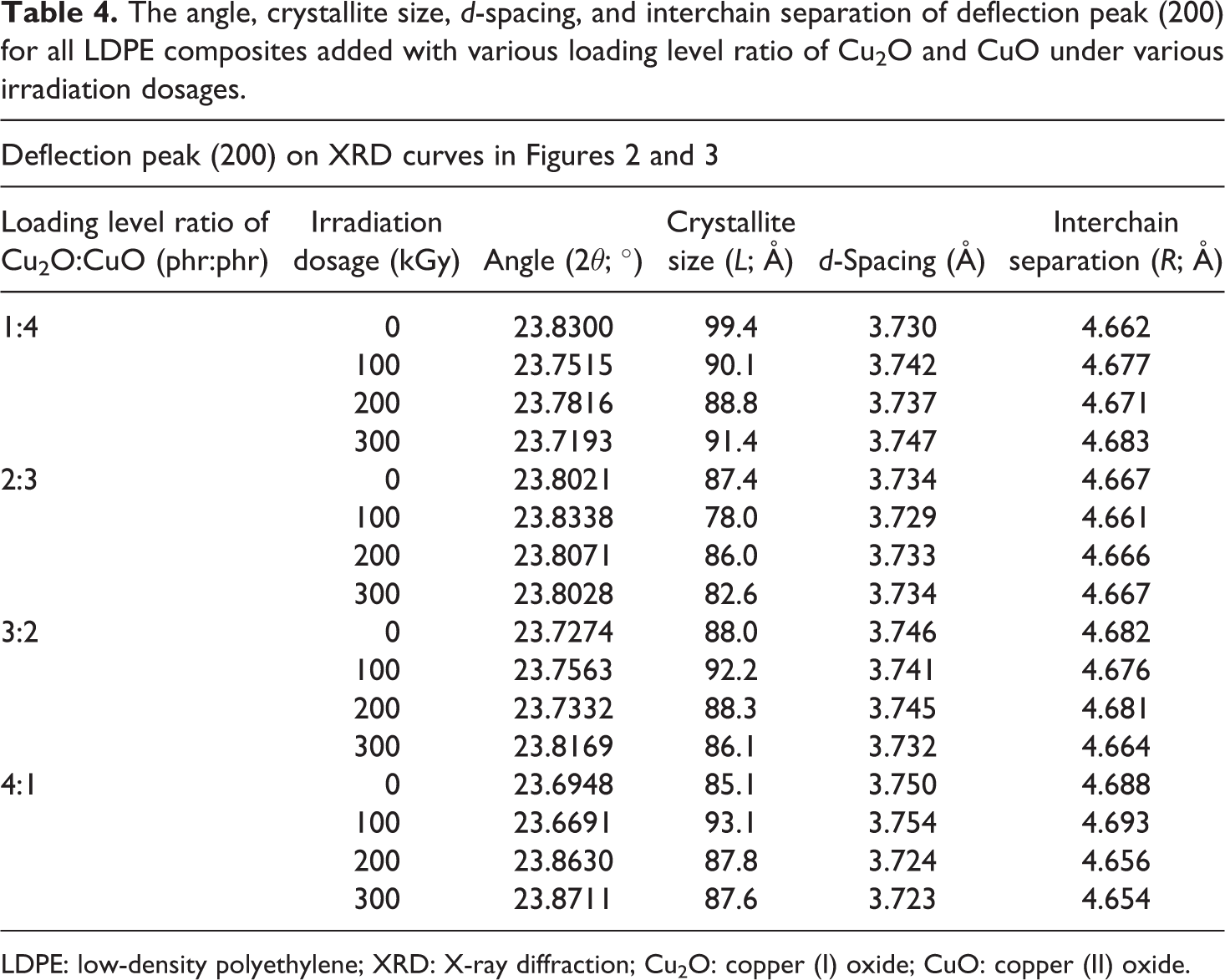
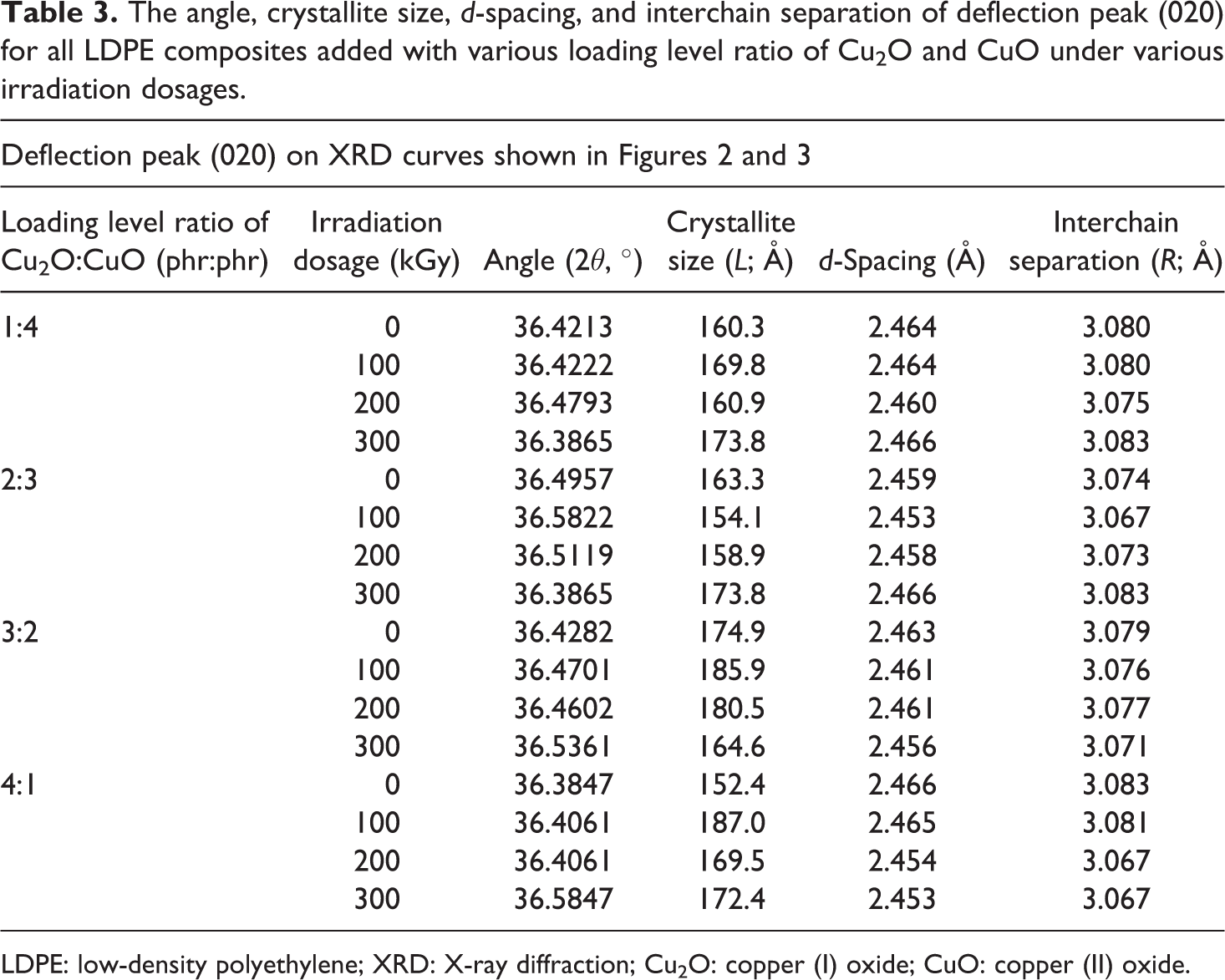
The crystallite size, d-spacing, and interchain separation for all Cu2O/CuO added LDPE composites under various irradiation dosages were calculated as corresponding to the deflection peaks of (110), (200), and (020) as shown in Figures 2 and 3. The increasing of Cu2O loading level from 1 phr to 2 phr (in replacing CuO loading level) into LDPE matrix decreased the crystallite size of deflection peaks (110) and (200). The increasing of Cu2O loading level from 1 phr to 2 phr (in replacing CuO loading level) into LDPE matrix could significantly reduce the size of the existing crystallite in amorphous regions of LDPE matrix by disturbing and rupturing the highly ordered chains arrangement in crystallite into more random arrangement. 34 Particularly, the rupture of the existing crystallite structures could significantly reduce the crystallite size of deflection peaks (110) and (200). However, the crystallite size of deflection peak (110) and (200) of un-rradiated LDPE composites increased from 117.7 Å to 133.2 Å as the loading level of Cu2O increased from 1 phr to 4 phr as depicted in Table 2. This is attributed to the increasing of Cu2O amounts in replacing the CuO in LDPE matrix could promote the formation of highly ordered arrangement structures by forming new crystallite structures. 34,35 The interaction effect between the newly formed crystallite structures and the existing crystallites in LDPE matrix could increase the crystallite size of deflection peaks of (110) and (200). By referring to Table 3, the crystallite size of the deflection peak of (020) for LDPE composites was found to reduce with the increasing of Cu2O loading level from 1 phr to 4 phr. The showed that the increasing of Cu2O loading level in replacing the loading level of CuO in LDPE matrix could induce the formation of new crystallites in amorphous regions of LDPE matrix. 15 These newly formed crystallites would interact with the existing crystallite and lead to increase the crystallite size of crystallite of peak (020).
The angle, crystallite size, d-spacing, and interchain separation of deflection peak (110) for all Cu2O added LDPE composites under various irradiation dosages.
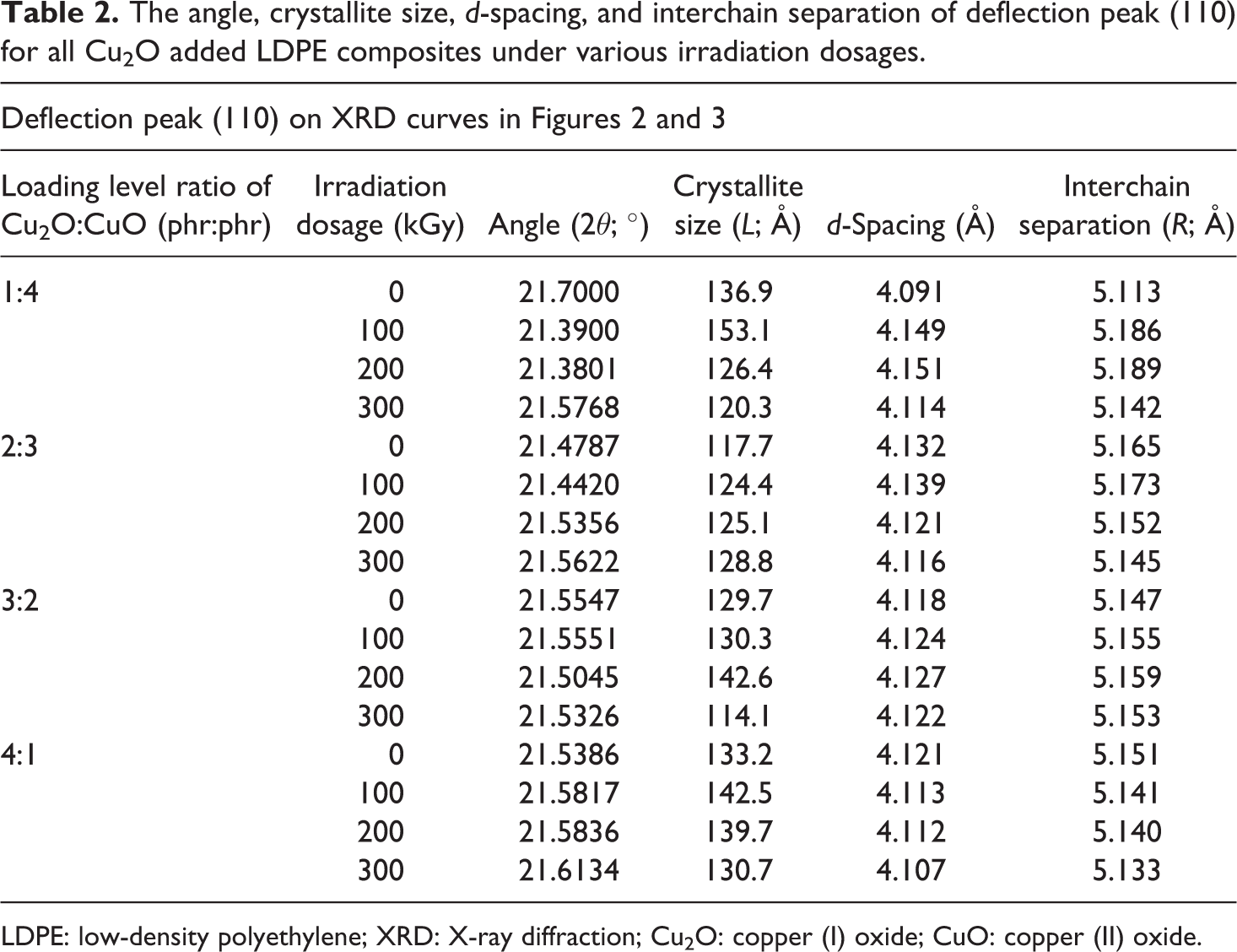
LDPE: low-density polyethylene; XRD: X-ray diffraction; Cu2O: copper (I) oxide; CuO: copper (II) oxide.
The angle, crystallite size, d-spacing, and interchain separation of deflection peak (020) for all LDPE composites added with various loading level ratio of Cu2O and CuO under various irradiation dosages.
LDPE: low-density polyethylene; XRD: X-ray diffraction; Cu2O: copper (I) oxide; CuO: copper (II) oxide.
By referring to Table 2, the increasing of electron beam irradiation dosages up to 100 kGy has significantly increased the crystallite size of peak (110) of all Cu2O/CuO added LDPE composites. However, the crystallite size for crystallite of peak (110) of all Cu2O and CuO added LDPE composites (except 2 phr Cu2O + CuO added LDPE composite) was smaller with further increasing the irradiation dosage from 100 kGy to 300 kGy as shown in Table 2. This also indicates that the application of higher irradiation dosages (≥200 kGy) could rupture the crystallites structures by disturbing the highly ordered chains arrangement into more random chains arrangement. Besides, the reduction in crystallite size also might be attributed to the occurrence of chain scissioning process at higher irradiation dosages which breakdown the large LDPE macromolecules chains into smaller chains. 35 The reduction of LDPE macromolecular chains size in LDPE matrix could disturb the highly ordered chains arrangement by converging to random chains arrangement. On the other hand, the increasing of electron beam irradiation dosage up to 300 kGy has gradually decreased the crystallite size of crystallite peak (200) of low Cu2O loading level added LDPE composites (1 phr Cu2O + 4 phr CuO and 2 phr Cu2O + 3 phr CuO added LDPE composites) as shown in Table 4. This might be attributed to the formation of cross-linking networks induced by electron beam irradiation did not improve the distribution and interaction of Cu2O (≤2 phr) and CuO (≥3 phr) particles in LDPE matrix. The poor interaction effect between the Cu2O and CuO particles with the cross-linking networks formed in LDPE matrix could cause an inferior effect to the highly ordered chains arrangement of crystallites in the amorphous regions of LDPE matrix. 34 The poor interaction between the existing crystallite and newly formed crystallite caused the crystallite size of crystallite for peak (200) to significantly decrease. Besides, the reduction of crystallite size also can be due to the occurrence of chains scissioning process during irradiation process. Thus, the chain scissioning process could disturb the ordered chains arrangement in LDPE matrix and reduce the crystallite size of these crystallites. 34 However, the crystallite size of peak (200) for higher Cu2O added LDPE composites highly increased when the electron beam irradiation increased up to 100 kGy as depicted in Table 4. This might be due to the formation of cross-linking networks by lower irradiation dosage could improve the distribution of Cu2O and CuO in LDPE matrix. This induces the interaction effect between the existing crystallite of peak (200) and the new crystallites formed by electron beam irradiation and thus increased the crystallite size of crystallite for peak (200). However, the crystallite size of crystallite peak (200) was gradually decreased with further increment in irradiation dosages up to 300 kGy as shown in Table 4. This can be due to the higher degree of cross-linking networks formed in LDPE matrix did not provide enhancement effect to the distribution of Cu2O and CuO particles in LDPE matrix. Thus, this could further reduce the interaction effect between the existing crystallites and newly formed crystallites and cause the crystallite size of peak (200) to gradually decrease. By referring to Table 3, the increasing of electron beam irradiation up to 300 kGy has marginally increased the crystallite size of peak (020) for lower Cu2O added LDPE composites. This indicates that the application of electron beam irradiation could increase the crystallite size of peak (020) by providing better interaction effect between the existing crystallites in LDPE matrix and the newly formed crystallites.
The angle, crystallite size, d-spacing, and interchain separation of deflection peak (200) for all LDPE composites added with various loading level ratio of Cu2O and CuO under various irradiation dosages.
LDPE: low-density polyethylene; XRD: X-ray diffraction; Cu2O: copper (I) oxide; CuO: copper (II) oxide.
By referring to Tables 2 to 4, the increasing of Cu2O loading level (in replacing the CuO) in LDPE matrix was observed to provide insignificant effect to the d-spacing and interchain separation of crystallites (110), (200), and (020). This indicates that the increasing of Cu2O loading level in LDPE matrix did not provide significant effect to the compactness of these crystallites. 36 On the other hand, the application of electron beam irradiation up to 300 kGy also found to exhibit insignificant effect on the d-spacing and interchain separation of crystallite (110), (200), and (020). This also indicates that the formation of cross-linking networks induced by electron beam irradiation did not provide significant effect to the compactness of polymer chains arrangement inside the crystallites in LDPE matrix. Thus, the d-spacing and interchain separation of the crystal planes for crystallite (020) did not show any significant change.
Mechanical properties analysis
By referring to Figure 4(a), the increasing of Cu2O loading level from 1 phr to 4 phr (in replacing CuO particles) into LDPE matrix has gradually decreased the tensile strength of unirradiated and irradiated LDPE composites. This indicates that the higher loading level of Cu2O particles in LDPE matrix has provided a poorer interaction effect with LDPE matrix than CuO particles by disturbing the LDPE chains structure arrangement in LDPE matrix as shown in Figure 5(a) to (d). This is because the increasing of Cu2O loading levels in LDPE matrix has significantly caused the agglomeration of Cu2O and CuO particles in LDPE matrix. According to SEM images shown in Figure 5(a) to (d), the agglomerated particles of Cu2O and CuO and the occurrence of cavities can be observed on the LDPE matrix. The agglomeration of Cu2O and CuO particles in LDPE matrix could also reduce the interfacial bonding between the LDPE macromolecules chains and the agglomerated particles of Cu2O and CuO. 37 The poor interfacial adhesion effect between the agglomerated of Cu2O and CuO particles and LDPE matrix also caused the occurrence of cavities in among the interfaces of the agglomerated Cu2O and CuO particles as well as LDPE matrix. This has significantly reduced the effectiveness in transferring the applied straining stress evenly from the polymer matrix to agglomerated particles of Cu2O and CuO where these agglomerated particles would act as stress concentration points when subjected to straining.
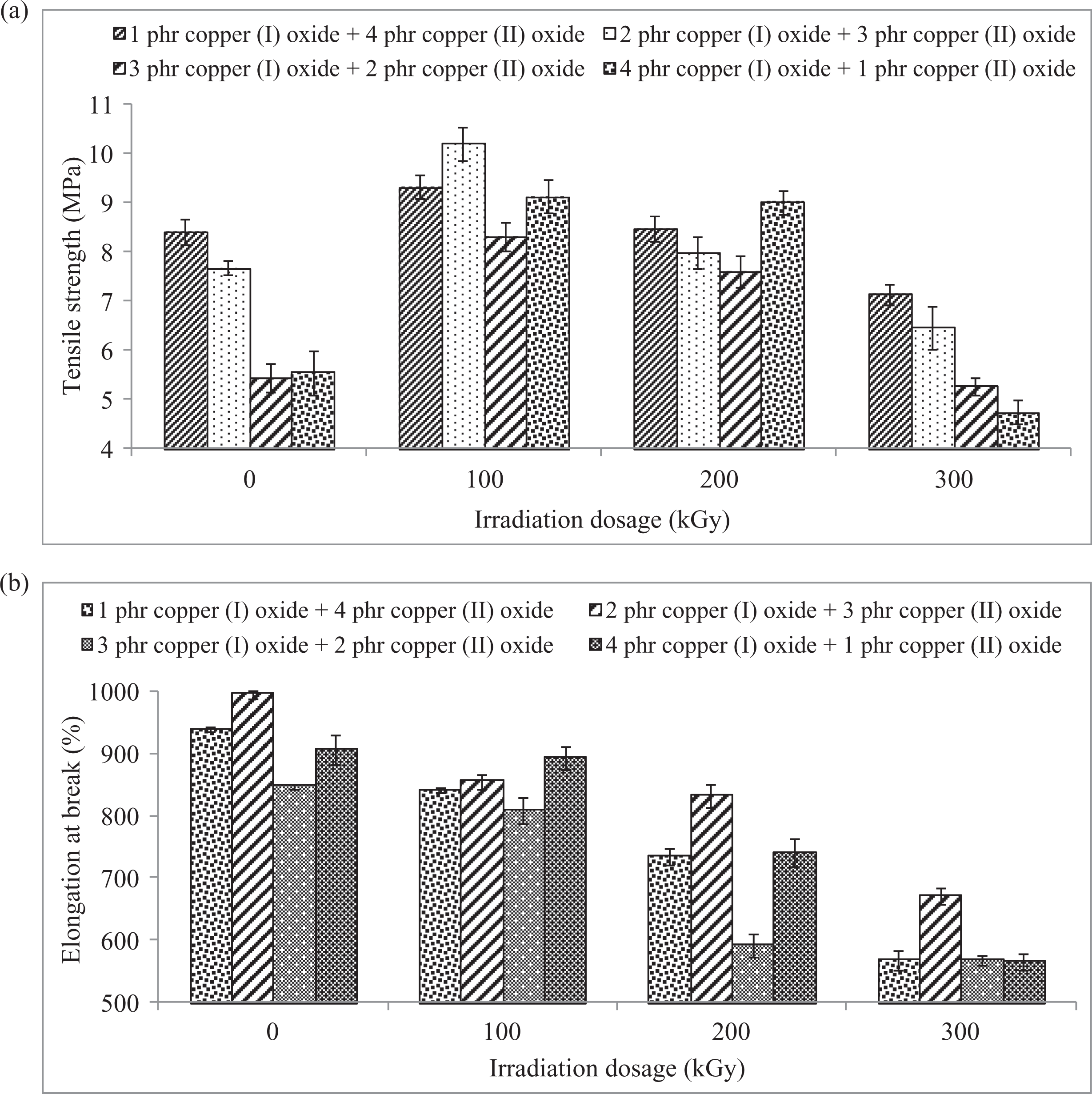
The effects of increasing electron beam irradiation dosages on the (a) tensile strength and (b) elongation at break of all different loading level ratios of Cu2O/CuO added LDPE composites. LDPE: low-density polyethylene; Cu2O: copper (I) oxide; CuO: copper (II) oxide.
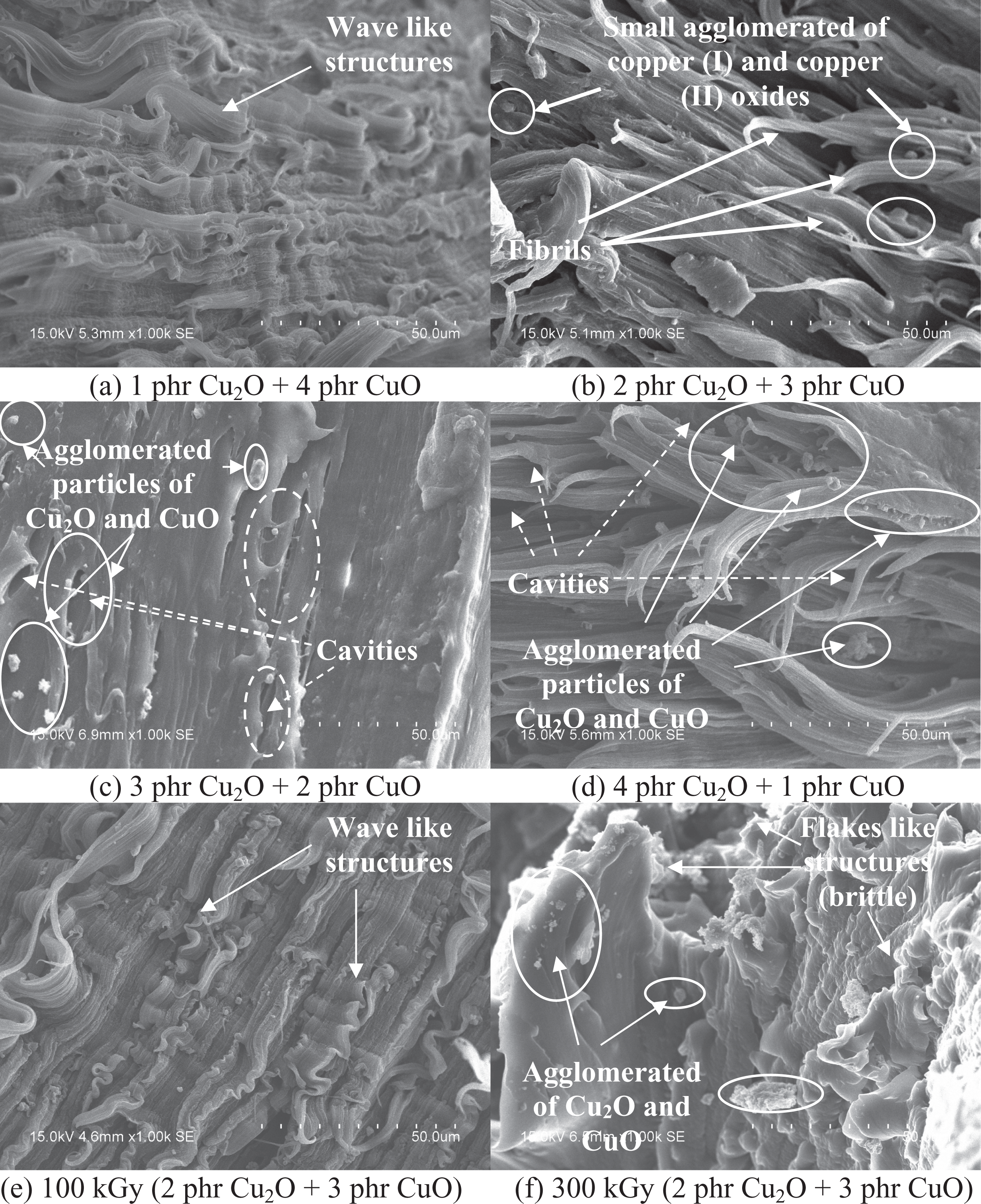
SEM images of (a–d) unirradiated LDPE composites added with different loading level ratios of Cu2O/CuO, and (e–f) 2 phr Cu2O + 3 phr CuO added LDPE composites when subjected to electron beam irradiation dosages of 100 kGy and 300 kGy, respectively. LDPE: low-density polyethylene; SEM: scanning electron microscopy; Cu2O: copper (I) oxide; CuO: copper (II) oxide.
The effect of electron beam irradiation on tensile strength of Cu2O/CuO added LDPE composites is illustrated in Figure 4(a). The increasing of electron beam irradiation dosages from 0 kGy to 100 kGy has gradually increased the tensile strength of all Cu2O and CuO added LDPE composites. The application of electron beam irradiation on all LDPE composites could induce the formation of cross-linking networks in polymer matrix by releasing the polymeric free radicals in LDPE matrix as discussed earlier in gel content. 31 The polymeric free radicals were generated in LDPE matrix by attacking the C–H bonding of LDPE macromolecules chains to release gas hydrogen in accordance to the mechanism as shown in equations (5) and (6). 27,38 The generated polymeric free radicals in LDPE matrix could react together to form cross-linking networks in LDPE matrix. The cross-linking networks formed in LDPE matrix could further improve the tensile strength by enhancing the reinforcement effect of LDPE matrix in resisting the straining stress. The formation of cross-linking networks could significantly increase the molecular weight of LDPE chains and this could further lead to stronger intermolecular bonding of the LDPE composites. Subsequently, the strong intermolecular bonding in LDPE matrix could provide outstanding strength to the LDPE matrix when subjected to straining stress. 39 The formation of cross-linking networks in LDPE matrix could also restrict the slippage effect of the LDPE macromolecular chains in LDPE matrix and thus the ability of LDPE matrix to resist the straining effect also significantly improved. 27,39 Furthermore, the formation of cross-linking networks in LDPE matrix could further improving the structural rearrangement of Cu2O and CuO particles in LDPE matrix due to the irregularity shapes of Cu2O and CuO particles. The formation of cross-linking networks in LDPE matrix could prevent and resist the structural reorganization of polymer matrix when subjected to straining stress. 26,40
However, subsequently increment in electron beam irradiation dosages from 100 kGy to 300 kGy have gradually decreased the tensile strength of all Cu2O and CuO added LDPE composites as illustrated in Figure 4(a). Generally, the cross-linking and chain scissioning processes would simultaneously take place when subjected to irradiation dosages. However, the available C–H bonding in LDPE macromolecule chains at higher irradiation dosages (>100 kGy) to generate polymeric free radicals in LDPE matrix has significantly reduced. This could significantly reduce the domination of cross-linking networks in LDPE matrix under irradiation and promote the occurrence of chain scissionning process. 41 The accelerated electron released by electron beam accelerator would attack and breakdown the C–C bonding of the backbone chains of LDPE macromolecules to generate polymeric free radicals in LDPE matrix. 41 This could further reduce the LDPE macromolecular chains in LDPE matrix and lowering the molecular weight of LDPE chains. Thus, the tensile strength has significantly reduced.
Elongation at break
Figure 4(b) illustrates the effect of electron beam irradiation dosages on the elongation at break of different loading level ratios of Cu2O/CuO added LDPE composites. By referring to Figure 4(b), the increasing of Cu2O loading level in LDPE matrix from 1 phr to 2 phr (in replacing the CuO loading level) was observed to significantly increase the elongation at break of all unirradiated and irradiated LDPE composites. This is due to the irregular shapes of Cu2O and CuO particles in this loading level ratio could disrupt the structural arrangement of polymer chains in LDPE matrix which reduce the interfacial bonding between the Cu2O and CuO particles with LDPE matrix. The reduction in interfacial bonding between the Cu2O and CuO particles and LDPE matrix could slightly decrease the restriction on the slippage effect of LDPE macromolecules chains in LDPE matrix when under straining stress. 26 On the other hand, the further increment of Cu2O particles from 2 phr to 3 phr (or decreasing CuO particles) in LDPE matrix also could significantly reduce the continuities of LDPE matrix by promoting the agglomeration of Cu2O and CuO particles in LDPE matrix as shown in Figure 5. By referring to Figure 5(c), the occurrence of fibrils were found to be disappeared from fractured surface of 3 phr Cu2O + 2 phr CuO added LDPE composites which indicates the loss of plastic deformation ability of polymer matrix. The occurrence of agglomerated Cu2O and CuO particles in LDPE matrix could reduce the plastic deformation ability of polymer matrix. 26,42 The poor compatibility between the agglomerated Cu2O CuO particles and LDPE matrix could cause these agglomerated particles to act as stress concentration points in LDPE matrix which caused LDPE matrix unable to elongate easily before the breakdown of polymer matrix. Thus, the elongation at break was significantly reduced.
By referring to Figure 4(b), the increasing of electron beam irradiation dosages up to 300 kGy has decreased the elongation at break of all Cu2O/CuO added LDPE composites. This is attributed to the electron beam irradiation process could induce the formation of cross-linking networks in LDPE matrix by generating the polymeric free radicals. The polymeric free radicals generated in LDPE matrix would react together to form cross-linking networks in LDPE matrix. 40 The formation of cross-linking networks in LDPE matrix could significantly extend the macromolecules size of LDPE chains by increasing the molecular weight of LDPE macromolecular chains. The increasing of electron beam irradiation dosages could highly induce the generation of polymeric free radicals in LDPE matrix. The higher polymeric free radicals generated in LDPE matrix could promote formation of higher degree of cross-linking networks in LDPE matrix. 39,43 The higher degree of cross-linking networks formed in LDPE matrix could further restrict the mobility or slippage effect of LDPE macromolecular chains when subjected to straining stress. 39 Thus, the increasing of electron beam irradiation on LDPE matrix could decrease the extendibility of the LDPE matrix. In addition, the increasing of macromolecules size for LDPE macromolecular chains in LDPE matrix could lead to stronger intermolecular bonding in Cu2O/CuO added LDPE composites. The stronger intermolecular bonding in LDPE matrix could further increase the restriction effect on the slippage effect or mobility of LDPE chains in LDPE matrix under straining. 40
Conclusions
This study has investigated the effect of electron beam irradiation dosages on physicomechanical characteristics of different loading level ratios of Cu2O/CuO added LDPE composites. The application of electron beam irradiation has gradually increased the gel content (or cross-linking networks) of all LDPE composites by generating more polymeric free radicals in LDPE matrix. Besides, the increasing of Cu2O loading level (in replacing CuO) has significantly reduced the gel content of all LDPE composites at all irradiation dosages. It was found that Cu2O required higher concentration level to promote the formation of cross-linking networks in LDPE matrix. On the other hand, the formation of cross-linking networks in LDPE matrix also has significantly improved the durability of LDPE matrix when subjected to static load at high temperature. In addition, the application of irradiation dosage up to 100 kGy on all Cu2O/CuO added LDPE composites has increased the crystallite size of deflection peak (110). This might be due to the formation of lower degree of cross-linking networks could converge the random chains arrangement into more ordered chains arrangement in LDPE matrix. However, further increment in irradiation dosages (>100 kGy) has significantly reduced the crystallite for deflection peak (110) by rupturing the existing crystallite structure. Whereas the increasing of Cu2O loading level has gradually decreased the tensile strength of all LDPE composites due to the irregularity shapes of Cu2O and CuO particles in LDPE matrix. The irregularity shapes of Cu2O and CuO particles tended to promote the agglomeration of Cu2O and CuO particles which reduce the interfacial bonding between LDPE matrix and copper oxides particles. The applied straining stress on LDPE matrix was unable to be transferred effectively from LDPE matrix to the agglomerated particles and thus decreasing the tensile strength. The increasing of irradiation dosage has effectively increased the tensile strength of all LDPE composites by introducing the formation of cross-linking networks. However, the formation of cross-linking networks induced by electron beam irradiation has gradually decreased the elongation at break of all LDPE composites by restricting the mobility of LDPE macromolecular chains.
Footnotes
Acknowledgments
The authors are very appreciating with the kindliness of Malaysian Nuclear Agency, Bangi, Selangor for allowing usage their equipments on performing this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.