Abstract
This article investigates three-layer co-extruded blown film comprised of low-density polyethylene (LDPE)/ethylene–vinyl alcohol (EVOH)/LDPE without adhesion layers. Various thicknesses of pure EVOH were sandwiched by outer LDPE layers blended with linear low-density polyethylene-grafted-maleic anhydride (LLDPE-
Introduction
Low-density polyethylene (LDPE) is a widely used packaging film in flexible packaging. In the case of blown film, it can be used as the sealing layer of a laminate due to its good moisture barrier, processability, moderate tensile strength, and of course, its heat sealing properties. 1 Unfortunately, LDPE has high permeability rates for oxygen and aromatics. However, with copolymerization of the vinyl alcohol functional group to an ethylene monomer, a converse effect is demonstrated indicating excellent barrier properties to oxygen permeability. With the recent push towards sustainability, the use of ethylene vinyl alcohol (EVOH) over aluminized polyester films has been growing in popularity due to its recyclability and transparent nature. However, EVOH tends to absorb moisture because of the presence of hydrophilic hydroxyl groups in its polymer structure, thus limiting its application primarily to the center of the laminates. 2 Therefore, combining LDPE and EVOH into one structure provides an ideal film structure for food packaging. The LDPE offers sealing and printing capabilities while acting as an outer layer to inhibit moisture diffusion into EVOH layer at the center. Improving what the LDPE lacks, the EVOH layer blocks the diffusion of oxygen molecules from one layer to the other.
EVOH resins have very poor adhesion to most polymers except nylons. A tie layer is usually needed to ensure LDPE and EVOH adhesion together in a lamination or extrusion process. A typical commercial LDPE/EVOH structure is a five-layered structure, namely LDPE/tie/EVOH/tie/LDPE. The tie resins used for EVOH and LDPE adhesion are based on graft polymers such as Plexar™ (Equistar Plastics), Binel™ (E. I. du Pont de Nemours Co.), and Modic™ (Mitsui Petrochemical Industries, Ltd). Applying a tie layer into a five-layer co-extrusion process is a complex and expensive process. Effort in research has been made to remove the adhesion layers from the co-extrusion process.
A number of investigations utilizing single-screw extruders were conducted in the late 90s to incorporate the grafted maleic anhydride (g-MAH) as a compatiblizer into LDPE/EVOH composite film. Kim and Chun
3
blended LDPE and EVOH by two different methods in an extrusion process: one using a two-stage technique that blends the EVOH with g-MAH first in a twin-screw extruder, which then feeds another extruder running LDPE. The second was to blend LDPE, EVOH, and g-MAH simultaneously using a single extruder, and they found that the latter method of blending all three together developed acceptable laminar structure with good barrier performance. Lee and Kim
4,5
developed LDPE/EVOH/LLDPE-
Furthermore, researchers added g-MAH into the co-extruded LDPE/EVOH/LDPE three-layer film to ensure proper bonding strength between LDPE layers and EVOH layer and at the same time retaining the barrier properties. Olmos et al.
7
blended the LLDPE-
This study developed the two combinations of LDPE/EVOH/LDPE without adhesion layers using a three-layer co-extrusion blown film line. The first group is a film with pure EVOH at the center layer and LDPE as outer layers without a tie layer. The purpose of this group is to study an effective EVOH gauge to block the oxygen barrier in a three-layer co-extruded blown film. The second group of the film is the modified structure from the 1st group where the outer layers of LDPE are blended with LLDPE-
Experimental
Materials
The Petrothene™ NA960 LDPE was acquired from LyondellBasell (Houston, Texas, USA) and is a blown film grade (
Each film layer contains 0–15 wt% of EVOH as center layer relative to the total mass of the film to achieve various thicknesses of pure EVOH. LLDPE-
Due to the difference of the densities between the LDPE and EVOH, the actual EVOH layer thickness cannot be derived by multiplying the weight percentage of the EVOH to the total film thickness. In order to determine the thickness of each layer of the multilayer film, the co-extruded film is sliced off cross sections in 90° angle with a special cutter and use of a microscope from AMSCOPE (Irvine, California, USA) to measure the resulting thickness. The targeted film thickness of all the three-layered films produced is 33 μ.
Processing
Table 1 summarizes all samples used in the study. The desired pure layer EVOH content as a percentage of final film thickness was adjusted by setting the extruders to the appropriate rotational speed ratios assuming that the screws had the same volumetric flow rate. The compatibilizer content LLDPE-
The processing table.
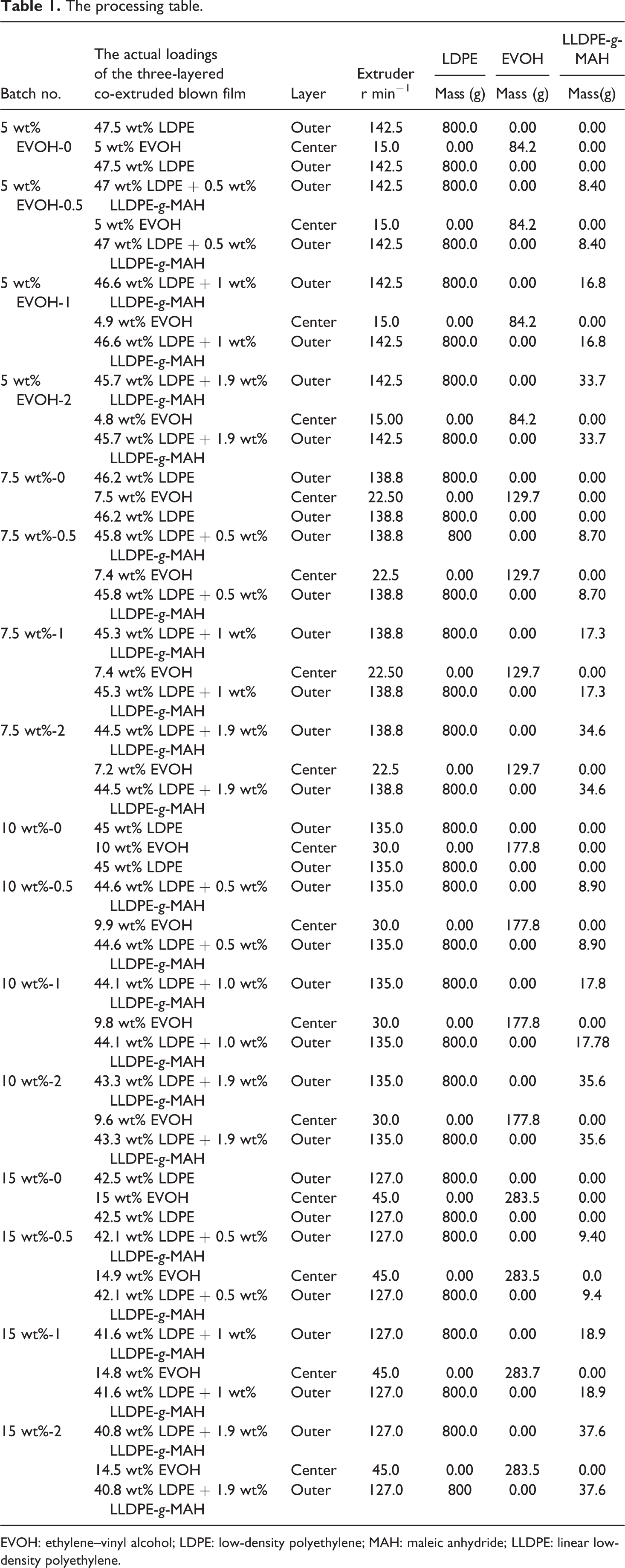
EVOH: ethylene–vinyl alcohol; LDPE: low-density polyethylene; MAH: maleic anhydride; LLDPE: linear low-density polyethylene.
Barrier and mechanical properties
Table 2 lists the typical packaging performance evaluation and the respective ASTM standards used. The sample size for tensile and peel strength were set at 10 for each analytical test. The barrier properties, water vapor transmission rate (WVTR), and oxygen transmission rate (OTR) were limited to two samples due to normal industry sampling practices, and as well as low standard deviation of results. The testing conditions for WVTR were 100°F (38°C) and 90% relative humidity (RH) and the testing conditions for OTR were 73°F (23°C) and 0% RH. The other testing including tensile and dart impact testing were conducted under the ambient environment at 21.4°C and RH 53.0%.
Testing methods of barrier and mechanical testing.
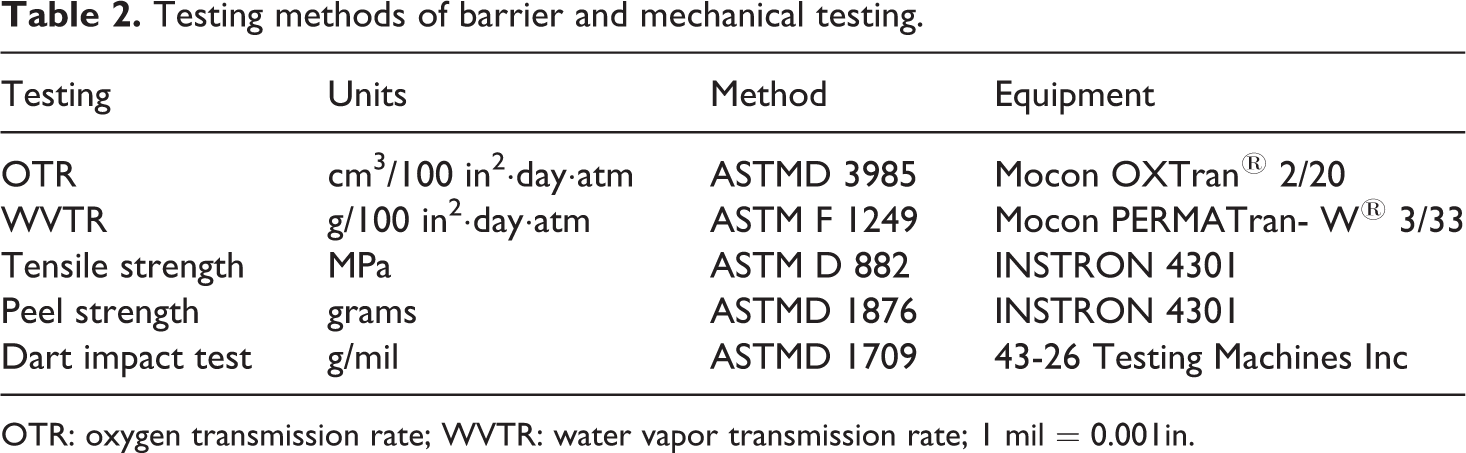
OTR: oxygen transmission rate; WVTR: water vapor transmission rate; 1 mil = 0.001in.
Thermal properties
The thermal properties of the three-layered co-extruded film were obtained through differential scanning calorimetry (DSC) model Q500 manufactured by TA Instruments (New Castle, Delaware, USA). The sample was equilibrated at 20°C, heated at a rate of 10°C min−1 from 20°C to 230°C. A melting endotherm, temperature, and melting enthalpy were obtained. The sample was cooled at a rate of 10°C min−1 from 230°C to 0°C, which results in crystallization with the crystallization temperature and crystallization enthalpy. Subsequently, the same heating and cooling process was repeated again for the sample. In addition to the temperature and enthalpy, the degree of crystallinity of the co-extruded film was also determined through the calculation of the enthalpy of fusion (Δ

where, Δ
Results and discussion
Barrier and peel strength
In order to investigate the correlation between the permeation and bonding of the film after adding the LLDPE-
Barrier and peel strength of the LDPE/EVOH/LDPE without LLDPE-
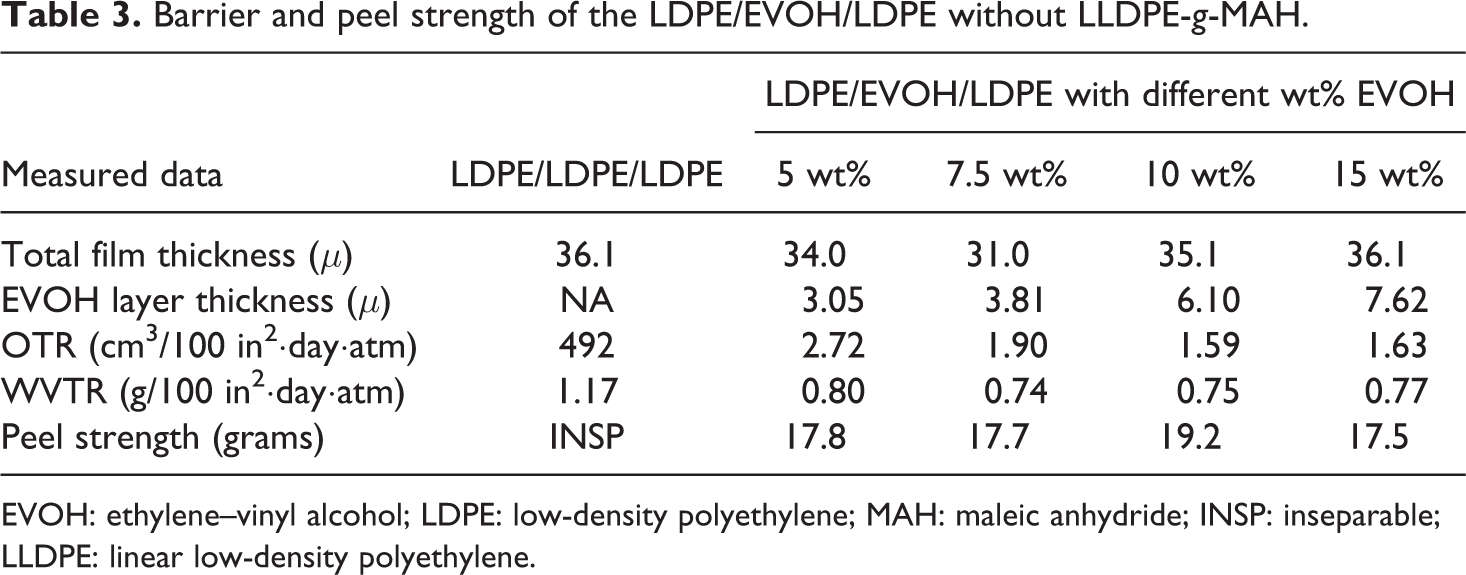
EVOH: ethylene–vinyl alcohol; LDPE: low-density polyethylene; MAH: maleic anhydride; INSP: inseparable; LLDPE: linear low-density polyethylene.
Barrier and peel strength of the LDPE/EVOH/LDPE with LLDPE-
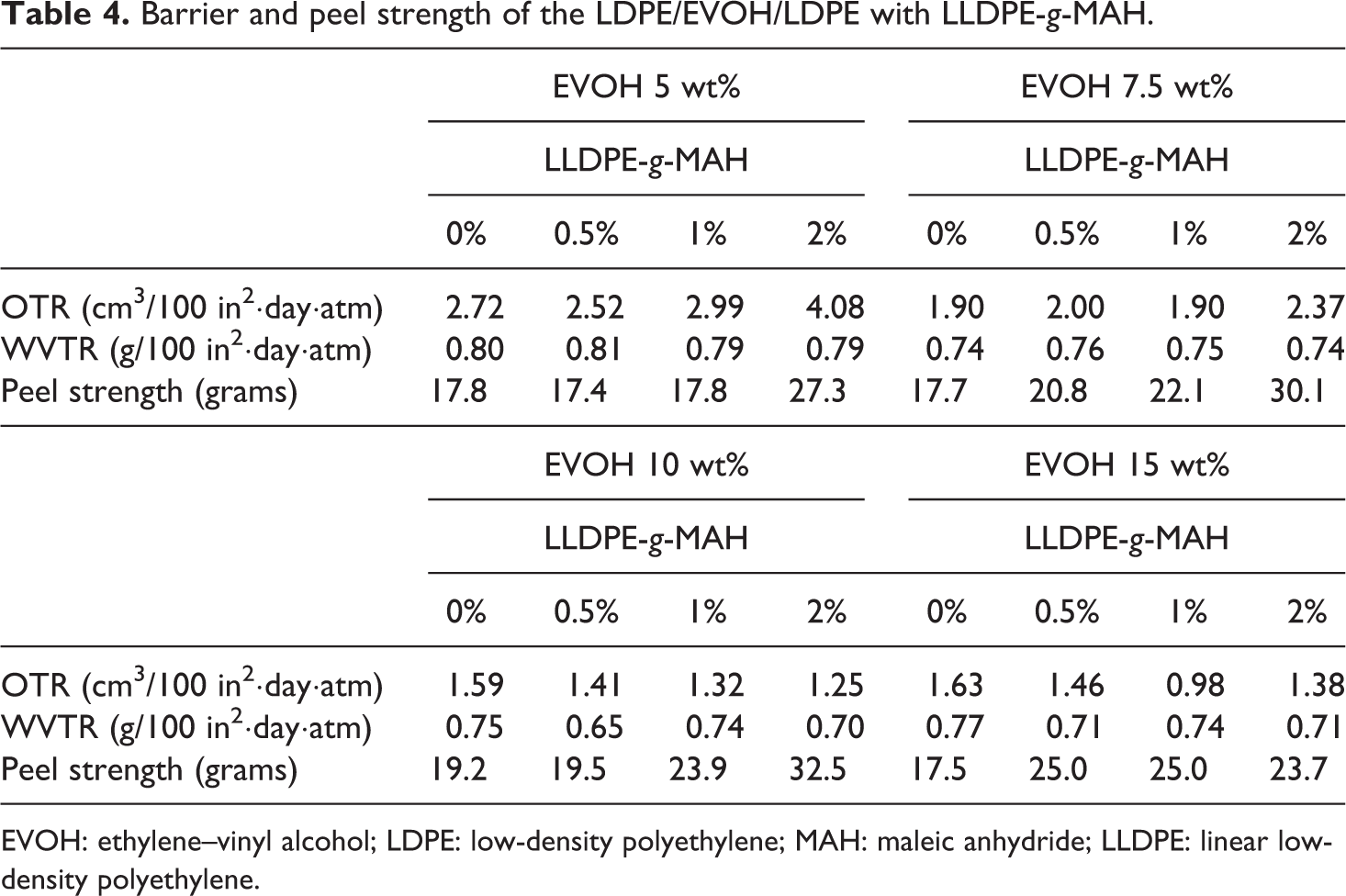
EVOH: ethylene–vinyl alcohol; LDPE: low-density polyethylene; MAH: maleic anhydride; LLDPE: linear low-density polyethylene.
Compared to the three-layered co-extruded blown LDPE film in Table 3, the OTR in the EVOH-centered film was reduced from control sample’s 491.82 cm3/100 in2·day·atm to 4.66 cm3/100 in2·day·atm with 5 wt% EVOH, 2.56 cm3/100 in2·day·atm for 10 wt% EVOH, and 2.09 cm3/100 in2·day·atm for 15 wt% EVOH, respectively. Noticeably, the initial 5 wt% EVOH brought down the permeation significantly where diminishing improvements for EVOH concentrations above 10 wt% and 15 wt%. The WVTR slightly improved from the original 1.17 g/100 in2·day·atm to 0.80 g/100 in2·day·atm for 5% EVOH, 0.75 g/100 in2·day·atm for 10 wt% EVOH and 0.77 g/100 in2·day·atm for 15 wt% EVOH. The barrier effect of the EVOH to moisture is diminished in the LDPE/EVOH/LDPE film due to the excellent moisture barrier of the LDPE film. As expected, the LDPE/EVOH co-extrusion film without tie layer showed weak bonding strength between EVOH and LDPE layers. The film thicknesses in Table 3 were averaged by ten measuring points for each of the variable.
Table 4 demonstrated that the peel strength of the LDPE/EVOH/LDPE films increased moderately when the LLDPE-
The cross machine direction (CD) profile of the film was observed by the use of scanning electron microscopy (SEM). Results in Figure 1 indicated that both 5 wt% EVOH-0% LLDPE-
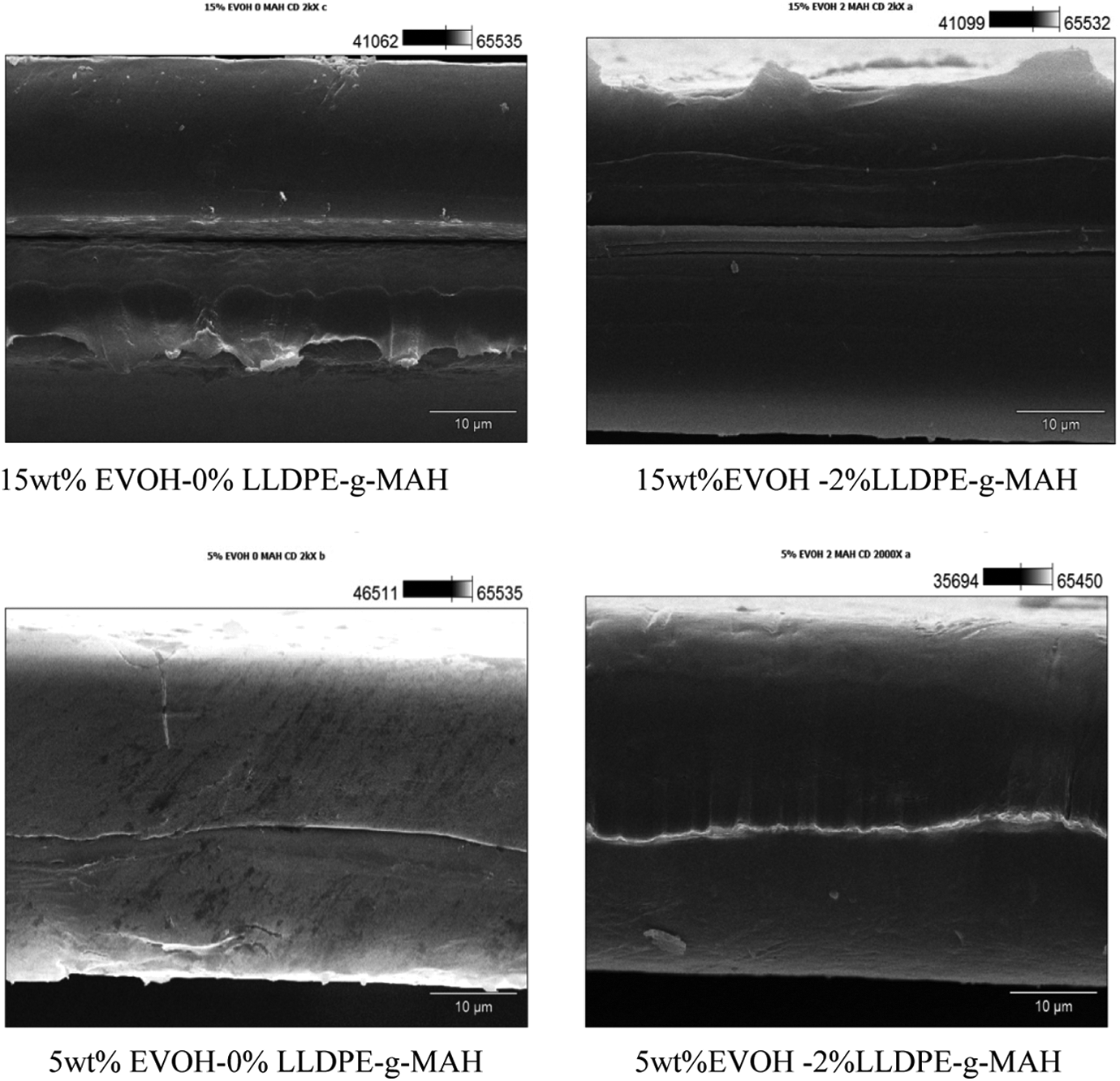
Scanning electron microscopy micrographs (×2000) for film’s cross section.
Compared to the samples without LLDPE-
Tensile and dart impact strength
Overall, the tensile strength of the film in both machine direction and CD increased when the EVOH was acted as the center layer. LLDPE-
Tensile and dart impact strength.
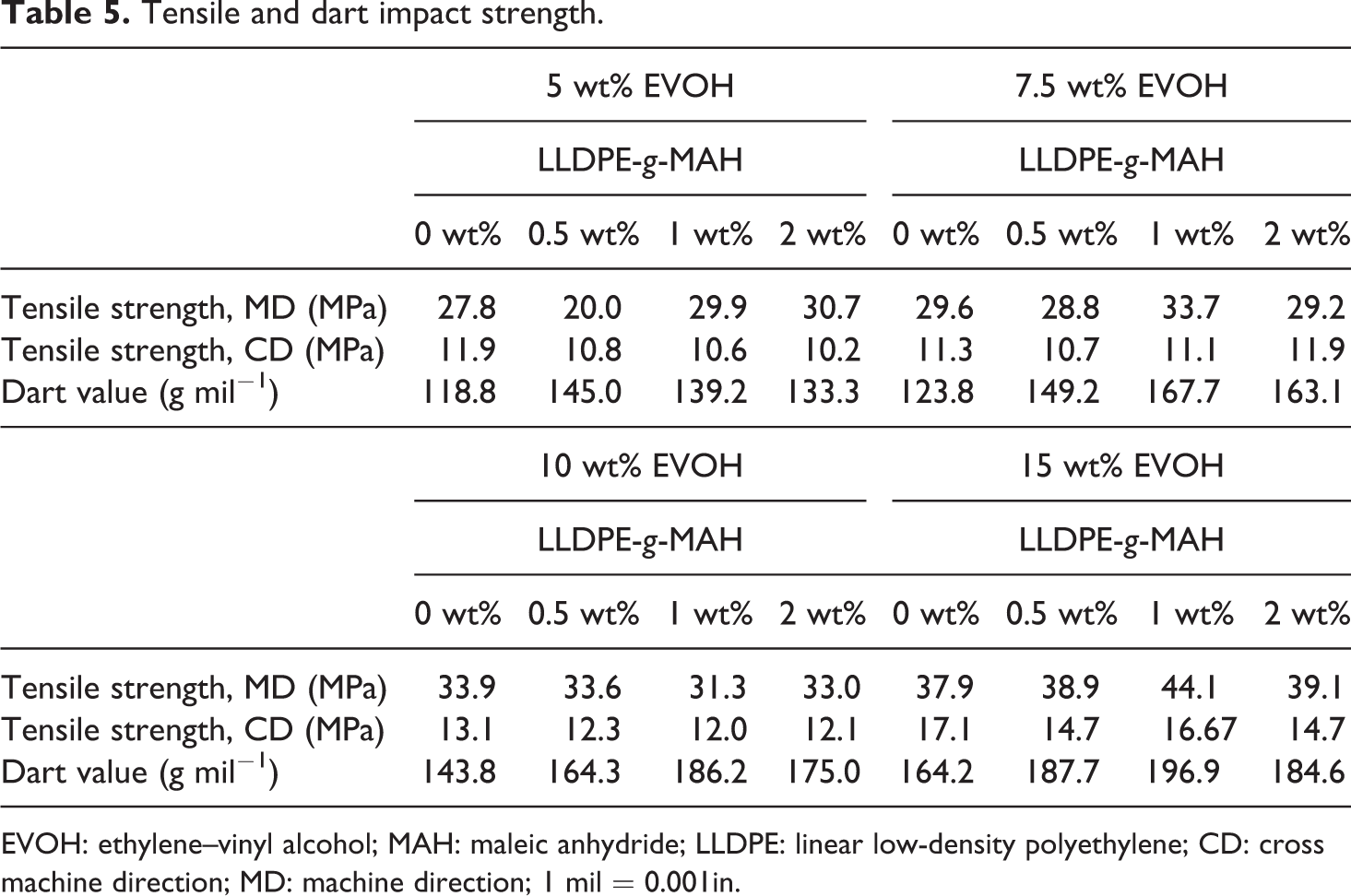
EVOH: ethylene–vinyl alcohol; MAH: maleic anhydride; LLDPE: linear low-density polyethylene; CD: cross machine direction; MD: machine direction; 1 mil = 0.001in.
For the dart drop test, adding a pure EVOH center layer in the middle did increase the impact resistance of the co-extruded film. Similar to the tensile strength, LLDPE-
Thermal properties
Table 6 summarizes the thermal properties of the film derived from the two-cycle heating and cooling scan based on DSC. The crystallinity calculated by the 1st heating scan curve reflected crystallinity as a result of the cooling rate during the blown film process. The crystallinity presented in 2nd cycle mirrored the crystallization process during the 1st cycle in DSC. The LDPE crystallinity of the 2nd cycle showed little difference from the 1st cycle, ranging from 0.32 to 0.37. In contrast, the EVOH crystallinity of the 2nd DSC cycle showed much less crystallinity due to the difference in quench rates. The relatively rapid cooling speed of the blown film reflected in the 1st cycle of DSC versus the slow cooling rate in the second DSC cycle.
Thermal properties of the LDPE/EVOH/LDPE.
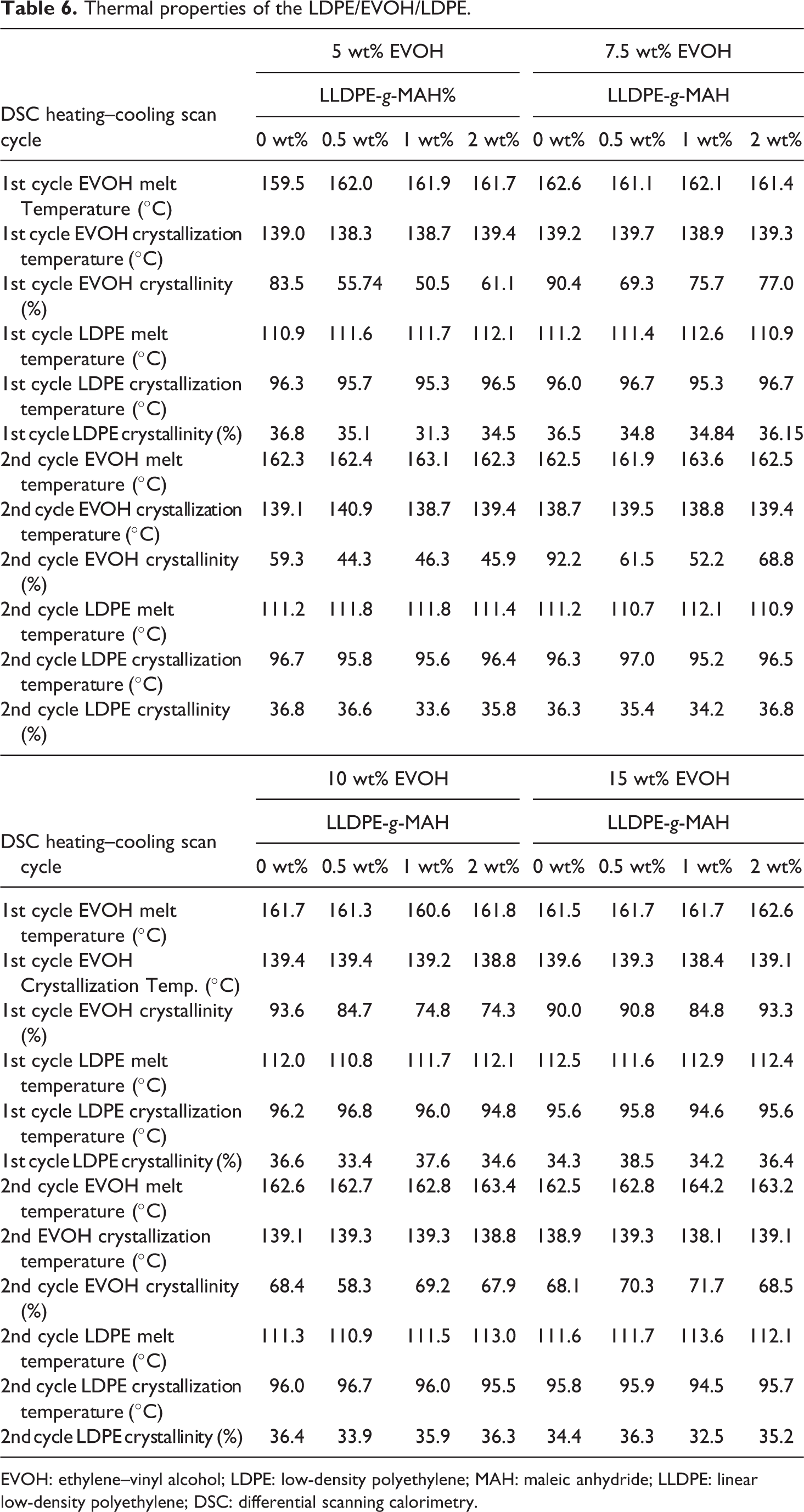
EVOH: ethylene–vinyl alcohol; LDPE: low-density polyethylene; MAH: maleic anhydride; LLDPE: linear low-density polyethylene; DSC: differential scanning calorimetry.
The g-MAH showed no effect to the LDPE crystallinity. Compared to LDPE, the EVOH crystallinity is significantly higher, ranging from 0.84 to 0.90 without LLDPE-
Conclusion
A 3-μ thin EVOH-centered film resulted in a dramatic increase of the oxygen barrier, from the original OTR 491.83 cm3/100 in2·day·atm of the LDPE/LDPE/LDPE sample down to 2.72 cm3/100 in2·day·atm for the LDPE/EVOH/LDPE film. Although subsequent increase of the EVOH amount continued the enhancement on the barrier, the magnitude of the increasing is not as dramatically as the initial adding of the EVOH.
The ratio of the EVOH/LLDPE-
The introduction of the LLDPE-
The bonding between LDPE and EVOH layer in the presence of LLDPE-
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
