Abstract
The present research investigates the morphology and rheological behavior of poly(butylene terephthalate)/polypropylene (PBT/PP) blends containing hydrophilic and hydrophobic organoclays. The distribution of nanoclays and morphology of nanocomposites were analyzed using X-ray diffraction (XRD) and transmission and scanning electron microscopies. The XRD patterns show that the level of intercalation structure in nanocomposites reinforced by hydrophilic nanoclay is significantly higher than nanocomposites filled by hydrophobic one. According to morphological analysis, both types of nanocomposites indicate the reduction of droplet size, whereas hydrophilic nanoparticles illustrate more compatibilization efficiency than hydrophobic. According to transmission electron microscopy, hydrophilic nanoclays are mainly localized in the PBT matrix and at the interface, whereas hydrophobic nanoparticles are confined in the PP-dispersed phase and at the interface. From the rheological point of view, the results show that nanocomposites with hydrophilic nanoclay show stronger pseudoplasticity, higher viscosity, and more elasticity than nanocomposites with the hydrophobic one. The localization of hydrophilic organoclay in the PBT matrix aids to finer morphology of the PBT/PP blend, whereas hydrophobic one resists the breakup of droplets by confinement in dispersed phase.
Introduction
Poly(butylene terephthalate) (PBT) is a semicrystalline engineering plastic with desirable mechanical features. Low shrinkage and electrical and thermal resistance of this substance are main reasons of its wide usage and applications (e.g. electrics and electronics as insulator). 1 –5 Polypropylene (PP) is one of the most important and widely used polymers that facilitates the crystallization of PBT by heterogeneous nucleation and improves the stiffness of polyesters. 6,7 Blending of polyolefins and poly(alkyl terephthalate) offers some advantages over its pure polymers in both cost and performance for industrial applications such as packaging, automotive, textiles, and so on, as well as for recycling technology. 8 PP is neither miscible nor compatible with PBT due to their considerable difference in polarity and chemical structure. Moreover, PP does not have necessary functional group to react with PBT. 9,10 As a result, the compatibilization of PBT and PP is an important point to achieve the appropriate properties.
Nanocomposites based on organically modified nanoclays (organoclays) are pretty appealing in both academic and industries. 11 –14 The extraordinary properties of nanocomposites greatly rely on the level of interactions between nanoclays and polymer chains. The intercalation and/or exfoliation structure of organoclays in the polymer matrix is responsible for satisfactory properties of polymer-layered silicate nanocomposites. 14 The localization of nanoclays in polymer blends is one of the main factors that can greatly affect the structure and ultimate properties of nanocomposites. 15 –18 Due to different affinity of nanoclays to various polymers, they can locate in one phase, two phases, or at the interface. 14 As shown by Hong and coworkers, 19 the most part of nanoclays locates in the PBT phase and some of the organoclay tactoids place at the interface of the PBT/polyethylene blend. Khonakdar et al. 20,21 showed that nanoclays (type of Cloisite 30B) confine in PET and at the interface of PET/PP. According to rheological investigation of Nazockdast et al., 22 there are good interactions between organoclays (Type of Cloisite 15A) and PP. Heydar et al. 12 studied the effect of nanoclay (Type of Cloisite 15A) on morphology, mechanical properties, and crystallization behavior of PP/nanoclay nanocomposites. According to their research, using the 5 wt% of Cloisite 15A indicates the maximum value of tensile strength and tensile modulus in PP. Additionally, the compatibilizers aided to nanoclay dispersion, which results in best performance in mechanical properties and crystallization behavior. Lipatov et al. 23,24 and Si et al. 25 recognized that in organic/inorganic systems, if one of the component is favorable, the inorganic particles tend to be localized at the interface and play the role of compatibilization.
According to literature review, organoclays with different polarities affect the morphology and rheological behavior of immiscible polymer blends, variously. Hence, the objective point of this research is built on this fact. It relies on the role of different types of organoclays, with different polarities (Cloisite 30B and Cloisite 15A), in morphology and viscoelastic behavior of the immiscible PBT/PP polymer blend.
Experimental
Materials
The polymeric constituents of samples used in this research are PBT (grade PB-NL70, density: 1.33 g cm−3, melting point: 225°C, product of Eurotec®, Turkey) and PP (homopolymer grade V30S, density: 0.9 g cm−3, melt flow index: 16 g/10 min, heat distortion temperature: 95°C, product of Marun Petrochemical Co. (Iran)). Organoclays (Cloisite 30B and Cloisite 15A) used in this study were supplied by Southern Clay Company (Gonzales, TX, USA). Cloisite 30B as hydrophilic nanoclay was modified with methyl tallow bis-2-hydroxyethyl quaternary ammonium with an amount of 90 mequiv./100 g. Cloisite15A as hydrophobic nanoclay was modified with dimethyl dihydrogenated tallow ammonium with modifier concentration of 125 mequiv./100 g. Irganox 1010, used as antioxidant, was obtained from Ciba (Singapore).
Sample preparation
The specimens were prepared by melt blending in a Brabender internal mixer (Germany) at 245°C and rotor speed of 60 r min−1. The formulation and code of samples are shown in Table 1. Firstly, PBT and PP were dry mixed and charged to the chamber. Melt blending lasted around 6 min. After the blending of PBT and PP, nanoclays varying in concentration (1, 3, and 5 phr) were introduced to the chamber and mixing took around 9 min. Moreover, 0.2 phr of Irganox 1010, as antioxidant, was used to prevent of degradation. For PBT/Cloisite 30B and PP/Cloisite 15A nanocomposites, polymeric component and nanoparticles were charged to the mixer, simultaneously, and melt blended for 10 min at 245°C and rotor speed of 60 r min−1. At last, the blends were hot pressed at 245°C for 10 min into a mold with the thickness of 1 mm and then annealed to room temperature.
Formulation and code of samples.
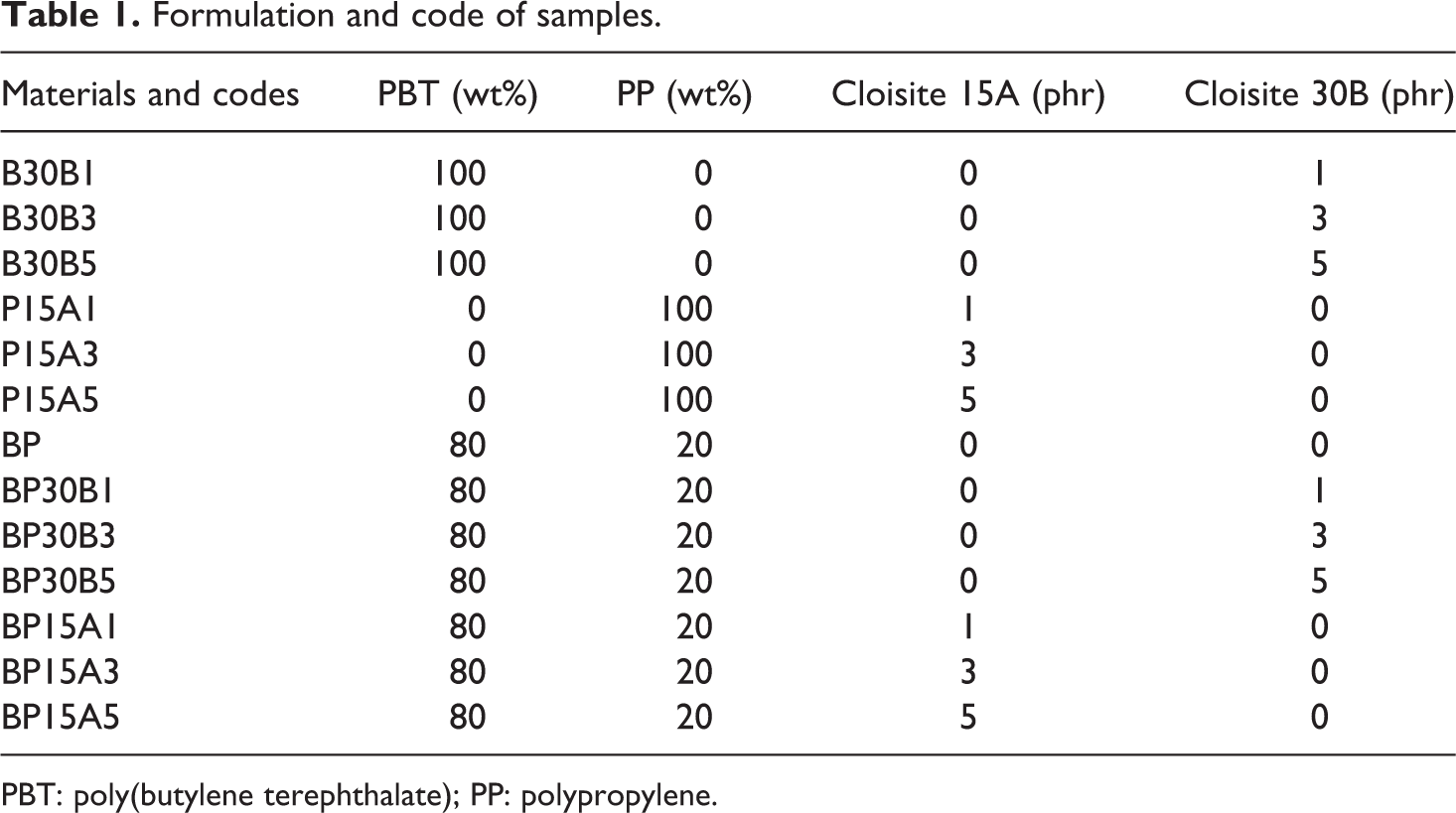
PBT: poly(butylene terephthalate); PP: polypropylene.
Characterization
X-ray measurements
The interlayer distance of Cloisite 30B and Cloisite 15A platelets in nanocomposites was determined by X-ray diffraction (XRD). The analyses were done by X′Pert MPD (Phillips, Netherlands). The diffractor was equipped with cobalt tube with the wavelength of 1.78 Å, voltage of 40 kV, and current of 40 mA. The diffractometer was scanned in the 2θ range from 1.5° to 10°, at the rate of 1 min−1, at ambient temperature, and measurements were recorded every 0.02°. The interlayer spacing of nanoclay layers can be specified by the following Bragg’s equation (equation (1)):

where d is the distance between atomic layers in crystals and λ is the wavelength of X-ray beam.
Morphology analysis
The morphology of the nanocomposites was observed using Vega/Tescan (Warrendale, Pennsylvania, USA) scanning electron microscope. The scanning electron microscopy (SEM) specimens were prepared by cryo fracturing of frozen nanocomposites in liquid nitrogen. The fractured surfaces were sputter coated with gold–palladium alloy for enhanced conductivity. To further insight into the role of organoclays on morphology of PBT/PP polymer blend, the droplet size was determined by using image analysis. Typically, 150 droplets were analyzed for each specimen. The number-average diameter (D n ), volume-average diameter (D v ), and polydispersity (PDI) were calculated as below:



In the above equations, N i is the number of droplets with the diameter of D i .
Transmission electron microscopy (TEM), Zeiss transmission electron microscope (Germany) using an accelerating voltage of 80 kV, was used to study the localization and dispersion state of organoclays in PBT/PP polymer blends. The thinnest part of nanocomposites, with thickness of 50–100 nm, was cryogenically microtomed with a diamond knife at temperature of about −70°C.
Rheological measurements
The rheological behavior of nanocomposites was investigated using a rheometric mechanical spectrometer (Paar Physica US200) equipped with a parallel plate geometry using 25 mm diameter plates on specimens with the thickness of 1 mm. The linear viscoelastic behavior of specimens was carried out using frequency sweep from 0.01 to 100 (rad s−1) in a small strain deformation at a temperature of 245°C.
Result and discussion
X-ray diffraction
The microstructure of PBT/PP/organoclay nanocomposites is typically characterized by XRD. The XRD patterns of neat nanoclay powders and nanocomposites containing various concentrations of organoclays are shown in Figure 1(a) and (b). As shown in Figure 1(a), the characteristic peak of Cloisite 30B (hydrophilic nanoclay) is observed at 4.85°, attributed to interlayer spacing of 21.19 Å. In nanocomposites reinforced with Cloisite 30B, the d001 peak location of Cloisite 30B is shifted to 2.88°, corresponding to interlayer spacing of 35.56 Å. The mentioned reduction of peak angle indicates the increment of Cloisite 30B gallery by which polymer chains can crawl into the interlayer spacing, more easily. It appears that the good affinity between hydroxyl and carboxyl end groups of PBT and hydroxyl groups of Cloisite 30B is responsible for the intercalation of PBT chains into the gallery of Cloisite 30B layers. The second peak observed at 5.5° < 2θ < 7° may be attributed to the thermal degradation of Cloisite 30B modifying alkyl ammonium groups 26 at 245°C (processing temperature). The XRD curves of neat Cloisite 15A (hydrophobic nanoclay) powder and nanocomposites based on Cloisite 15A are illustrated in Figure 1(b). Obviously, the characteristic peak of Cloisite 15A in nanocomposites is dropped to lower angles translated to increasing the d-spacing of Cloisite 15A layers from 38.86 Å to 54.60 Å. The similar hydrophobic nature of Cloisite 15A and PP could be responsible for the intercalation structure of PBT/PP/Cloisite 15A. The second peak in the XRD curve of Cloisite 15A organoclay at 6° < 2θ < 8° can be due to the presence of some non-intercalated platelets (non-modified). 27 Comparing Figure 1(a) and (b), the intercalated structure is more dominant in PBT/PP/Cloisite 30B nanocomposites than PBT/PP/Cloisite 15A. The higher level of intercalation structure in PBT/PP/Cloisite 30B nanocomposites could be relied on the strong polar interactions between hydroxyl groups of Cloisite 30B and hydroxyl and carboxyl groups of PBT (matrix phase).
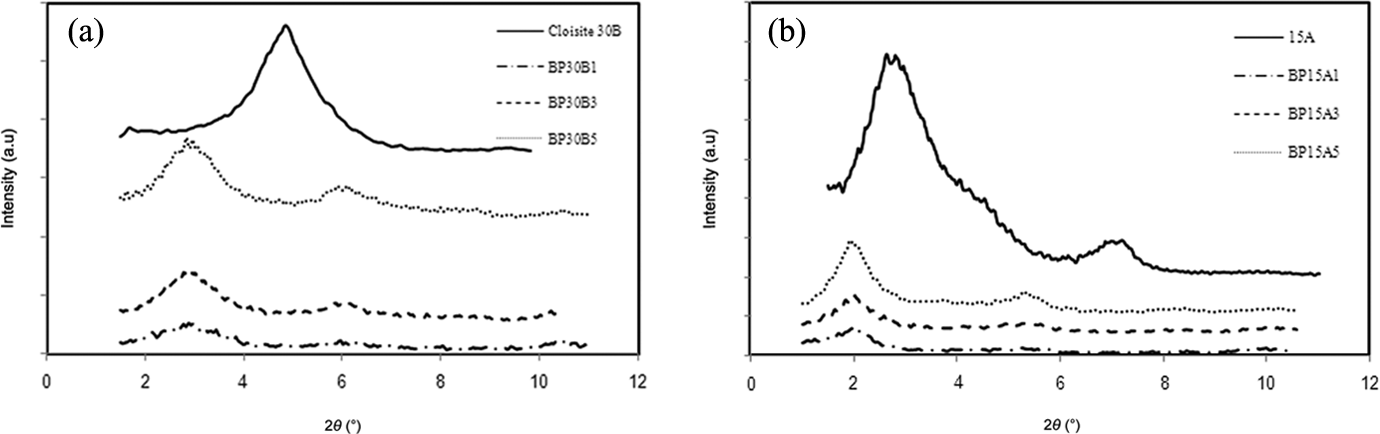
XRD patterns of (a) neat Cloisite 30B powder and nanocomposites based on Cloisite 30B and (b) neat Cloisite 15A powder and nanocomposites based on Cloisite 15A. XRD: X-ray diffraction.
Transmission electron microscopy
TEM micrographs of nanocomposites containing 5 phr of Cloisite 30B and Cloisite 15A are illustrated in Figure 2(a) and (b), respectively. Clearly, the hydrophilic nanoclays are distributed in the PBT matrix and at the interface. Besides, hydrophobic organoclays are located in PP-dispersed phase and at the interface. The heterogeneous distribution of both types of organoclays at the interface can result in reduction of interfacial tension and coalescence suppression of droplets. The present behavior is totally similar to trend of compatibilizers in immiscible polymer blends.
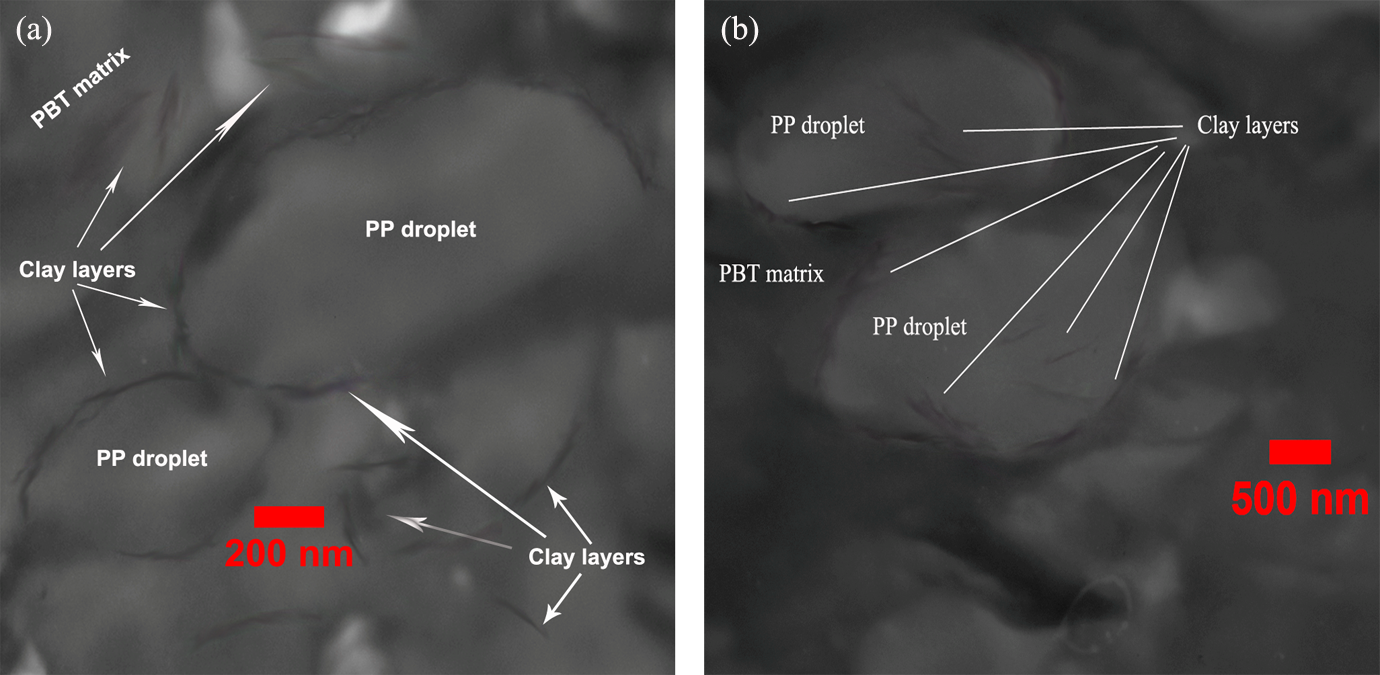
TEM images of (a) nanocomposite containing 5 phr of Cloisite 30B and (b) nanocomposite containing 5 phr of Cloisite 15A. TEM: transmission electron microscopy.
Based on thermodynamic standpoint, the localization of nanoclays can be predicted by wetting parameter (W12) expressed in equation (5) 28 :

where, τ n-PBT is the interfacial tension between nanoclays and PBT, τ n-PP is the interfacial tension between nanoclays and PP, and τ12 is the interfacial tension between two polymers. When W12 > 1, nanoclays can be presented in PP. If W12 < −1, nanoclays tend to be located in PBT and if −1 < W12 < 1, nanoclays will be accumulated at the interface. 21 Additionally, Wu’s equation is usually used to estimate the interfacial tension between two polymeric components (equation (6)) 29 :
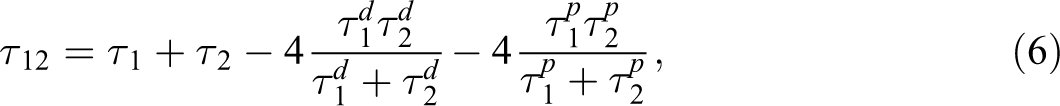
where, τ1 and τ2 are surface tension of polymer 1 and 2. The exponents of d and p are the dispersive and polar contribution of surface tension, respectively. The values of surface tension, dispersive, and polar contribution of surface tension for neat materials and nanoclays are illustrated in Table 2. 30 The values of surface tension for neat constituents and organoclays at 245°C are also calculated by temperature gradient of surface energy. Based on equation (5), the W12 value is −1.07 and 0.56 for nanocomposites filled by Cloisite 30B and Cloisite 15A, respectively. The present result indicates that hydrophilic organoclays can be located in PBT phase and hydrophobic nanoparticles accumulate at the interface. There is some inconsistency between TEM images and thermodynamic approximation. Elias et al.’s 31 discussion regarding the migration of nanoclays from one phase to other phase or to interface is listed below:
Surface tension, dispersive, and polar contribution of surface tension for different materials at 20°C. 30
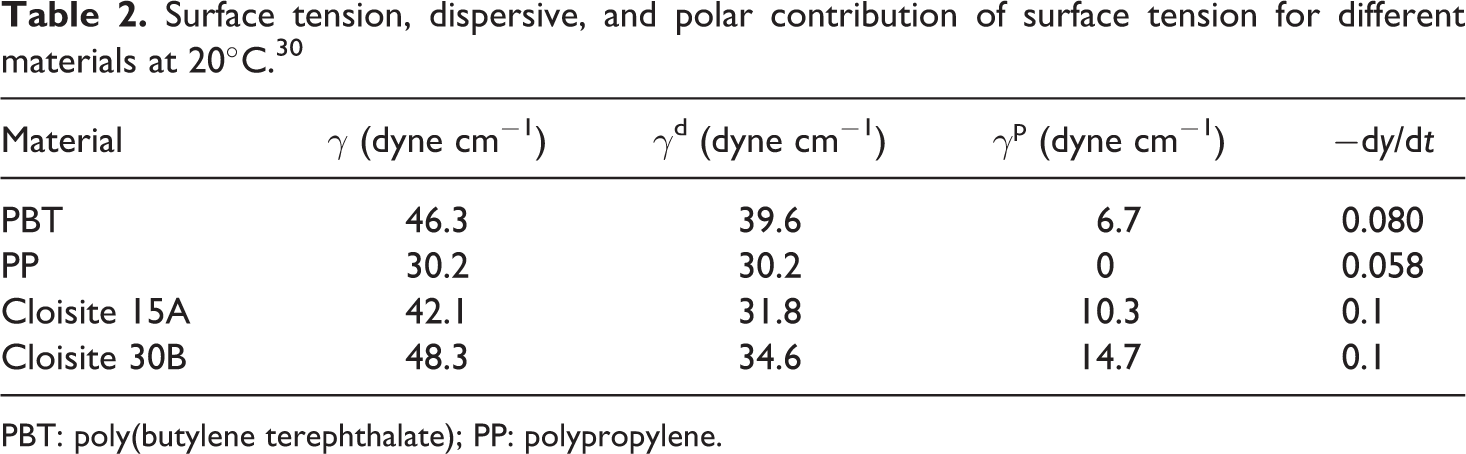
PBT: poly(butylene terephthalate); PP: polypropylene.
The first mechanism is self-diffusion of nanoparticles as a result of Brownian motion. The diffusion coefficient (D0) of a particle with the radius of a at the temperature of T in a fluid with the viscosity of η can be expressed by:

where KB is Boltzmann constant. In addition, the time required (t s ) for a particle to diffuse a distance equal to its radius is presented by:

Assuming that organoclays are circular plate-like with the radius of 250 nm, it can be estimated around 8.3 × 103 s (equal to 138 min) in PBT matrix with the viscosity of about 200 Pa s and the temperature of 518 K. As obvious as it is, this time is significantly higher than melt blending process time. So it can be concluded that Brownian motion is not the major mechanism of organoclay migration.
The second mechanism is based on the migration of nanoparticle by the shear (convective diffusion). In this mechanism, the frequency of collisions between particles or droplets (C) in simple shear flow can be approximated by 32 :

where Ø is volume fraction of particles or dispersed droplets and
Scanning electron microscopy
The aim of this section is to study the role of nature and concentration of organoclays on the morphology of PBT/PP blend. Figure 3(a) to (d) presents the SEM micrographs of neat PBT/PP blend and nanocomposites filled by different concentrations of hydrophilic nanoclay. As it is clear, with the increment of Cloisite 30B loading, the size of PP droplets decreases and more homogeneously disperse in the PBT/PP blend. Due to high affinity of Cloisite 30B to PBT, the viscosity of PBT as matrix can be increased with the increment of Cloisite 30B concentration. As we know, an increase in matrix viscosity and elasticity results in the enhancement of capillary number (equation (10)). The droplets of dispersed phase can breakup more easily at higher capillary numbers. In equation (10), ηm is matrix viscosity,

SEM micrographs of (a) neat PBT/PP blend, (b) PBT/PP filled by 1 phr of Cloisite 30B, (c) PBT/PP filled by 3 phr of Cloisite 30B, and (d) PBT/PP filled by 5 phr of Cloisite 30B. SEM: scanning electron microscopy; PBT: poly(butylene terephthalate); PP: polypropylene.

Additionally, with the increment of matrix viscosity as a result of Cloisite 30B loading, the viscosity ratio of PBT/PP polymer blend will be changed and may go to unity. Based on Wu’s theory, 34 the droplets of dispersed phase can breakup more easily when the viscosity ratio of a blend is around unity. In addition to rheological parameter, the nonhomogeneous distribution of Cloisite 30B layers at the interface can result in the reduction of interfacial tension and aid to coalescence hindrance of droplets. Prevention of the coalescence of dispersed droplets can be very effective in stabilizing droplet size.
The SEM micrographs of the PBT/PP blend filled by different concentrations of Cloisite 15A (hydrophobic nanoclay) are shown in Figure 4(a) to (c). The droplet-matrix morphology is observed for these nanocomposites, as well. The size of droplets decreases with the increment of Cloisite 15A, whereas the nanocomposites based on Cloisite 30B show more homogeneous morphology with lower droplet size than nanocomposites filled by Cloisite 15A. Decreasing the size of droplets in the presence of Cloisite 15A can be relied on the localization of Cloisite 15A at the interface (as shown in TEM images). There are two opposite factors that affect the morphology of PBT/PP blends filled by hydrophobic organoclays. Localization of some parts of Cloisite 15A in the PP phase causes enhancement of PP viscosity and elasticity as a dispersed phase. The breakup of droplets becomes harder when the elasticity of dispersed phase increases. On the other hand, the localization of Cloisite 15A at the interface can lower the interfacial tension and suppress the coalescence of dispersed droplets. As Figure 4(a) to (c) illustrates, the competition between these two opposite factors results in lowering of droplet diameter. Then, it can be concluded that the contribution of interfacial features is greater than rheological parameters in morphology development of PBT/PP polymer blend. With the aid of image analysis, the values of Dn, Dv, and PDI are calculated for different samples. Table 3 indicates the particle size characterization of PP droplets in PBT/PP/organoclay nanocomposites. As obvious as it is, the droplet diameter and particle size distribution of nanocomposites filled by Cloisite 30B are lower than nanocomposites based on Cloisite 15A. The present result suggests that the efficiency of Cloisite 30B in dispersion and distribution of PP droplets is higher than Cloisite 15A. In PBT/PP/Cloisite 30B nanocomposites, the rheological parameters help to breakup the droplets, due to localization the some parts of nanoparticles in the matrix. However, the rheological parameters hinder the breakup of droplets in nanocomposites filled by Cloisite 15A, due to confinement of some parts of hydrophobic nanoclays in dispersed phase. As presented in Table 3, the PDI of BP15A1 sample is higher than other samples. As it was mentioned, the localization of Cloisite 15A organoclays in dispersed droplets and at the interface has vice versa effect on morphology of PBT/PP blend. Some parts of organoclays help to morphology development by localization at the interface and the other parts suppress the breakup of droplets. Hence, the dispersion homogeneity of droplets in PBT/PP/Cloisite 15A nanocomposites is lower than nanocomposites filled by Cloisite 30B. It seems that using 1 phr of Cloisite 15A is insufficient to cover the interface domains. As a result, BP15A1 nanocomposite shows larger droplet diameter and higher PDI value compared to other samples.
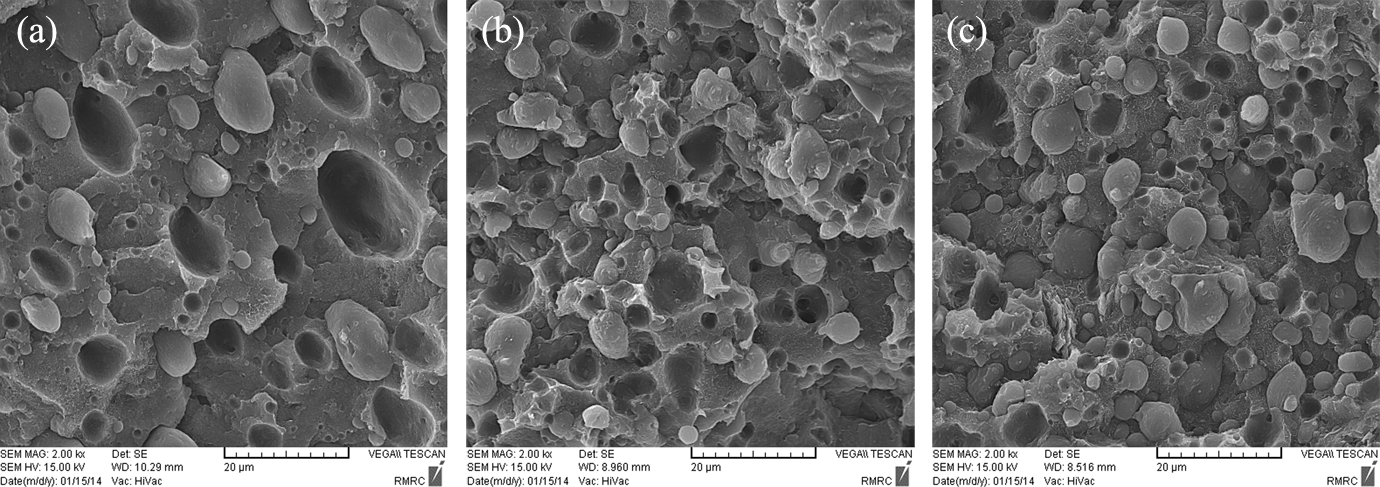
SEM micrographs of (a) PBT/PP filled by 1 phr of Cloisite 15A, (b) PBT/PP filled by 3 phr of Cloisite 15A, and (c) PBT/PP filled by 5 phr of Cloisite 15A. SEM: scanning electron microscopy; PBT: poly(butylene terephthalate); PP: polypropylene.
Particle size characterization of PP in PBT/PP/organoclay nanocomposites.
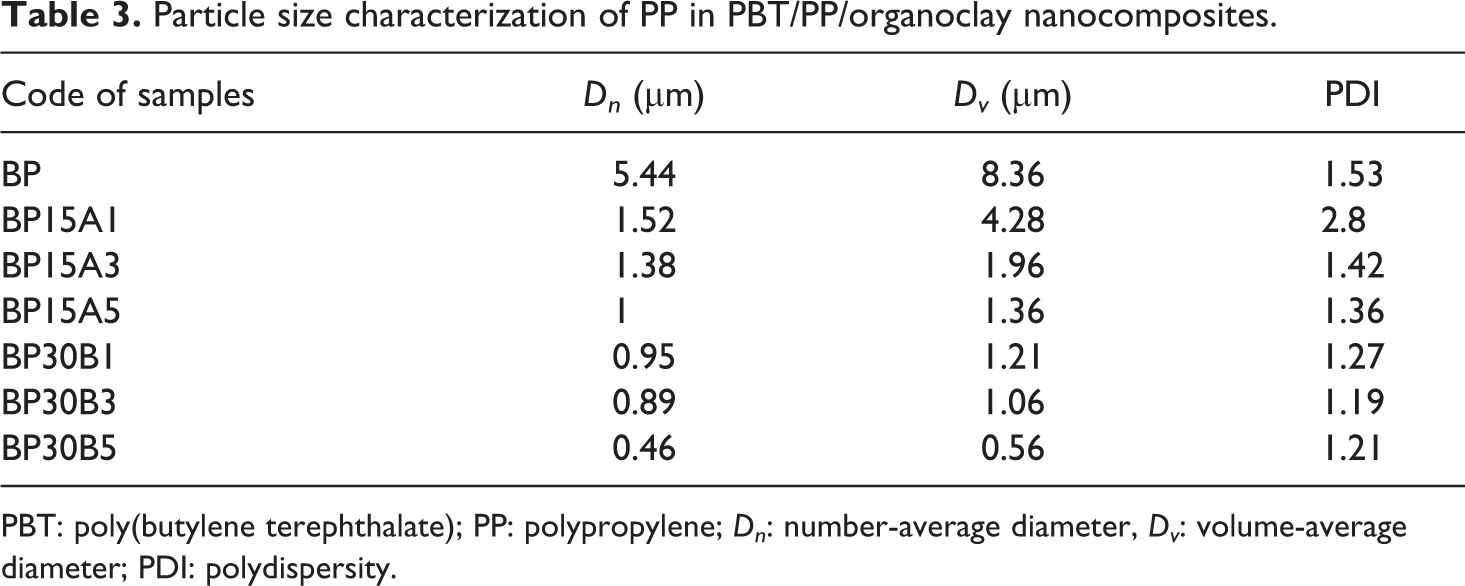
PBT: poly(butylene terephthalate); PP: polypropylene; D n : number-average diameter, D v : volume-average diameter; PDI: polydispersity.
Rheological behavior
Figure 5(a) and (b) illustrates the complex viscosity of neat PBT/PP blend and different nanocomposites versus frequency. Figure 5(a) shows that the complex viscosity of nanocomposites reinforced with Cloisite 30B is increased with the content of organoclays, dramatically. The complex viscosity of nanocomposite containing 5 phr of Cloisite 30B (BP30B5) grows about two orders of magnitude compared to neat PBT/PP polymer blend. Hydrogen bonding between hydroxyl groups of Cloisite 30B and hydroxyl and carboxyl end groups of PBT plays an important role in enhancement of complex viscosity, at low frequencies. The same results are observed for nanocomposites reinforced with various concentrations of Cloisite 15A. The tendency of Cloisite 15A to PP, due to the similar polarity, can be responsible for increasing the complex viscosity of blend. Comparison of Figure 5(a) and (b) indicates that Cloisite 30B is more effective than Cloisite 15A in increment of complex viscosity. In addition, the nanocomposites based on hydrophilic nanoclay show stronger pseudoplastic behavior than nanocomposites based on hydrophobic one. The present results are in good agreement with the outcomes of SEM and XRD analyses.
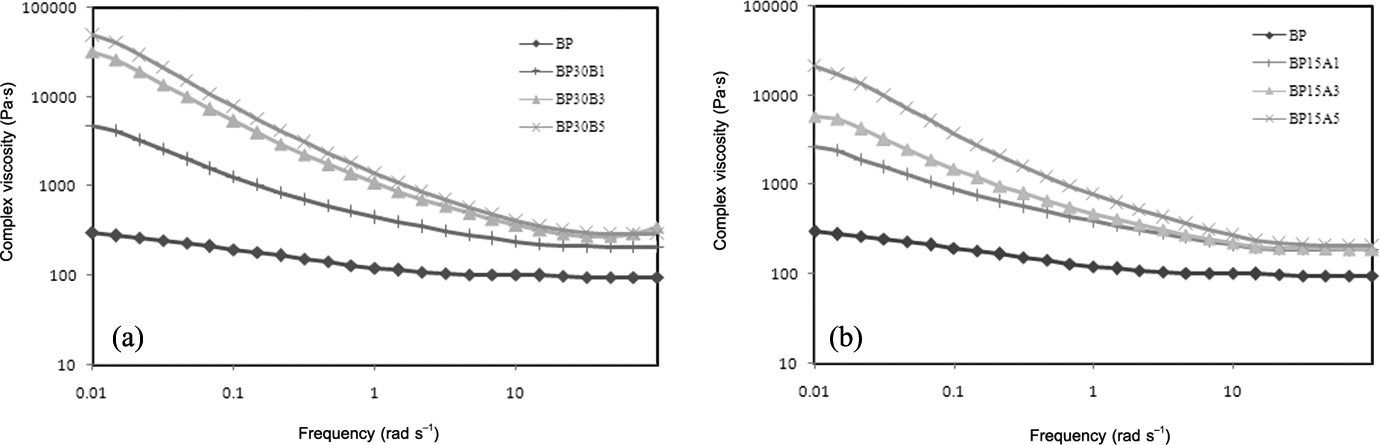
Complex viscosity versus frequency for (a) nanocomposites filled by Cloisite 30B and (b) nanocomposites filled by Cloisite 15A.
The storage and loss moduli of various nanocomposites are illustrated in Figure 6(a) to (d). By incrementing the concentration of Cloisite 30B, the values of storage and loss moduli increase greatly, at low frequencies. Moreover, by increasing the extent of Cloisite 30B to 5 phr, the slope of storage and loss modulus curves decreases, at low frequencies. The nonterminal behavior of nanocomposite containing 5 phr of Cloisite 30B can be relied on the transition of rheological behavior from liquid-like to solid-like. Transition of the rheological behavior from liquid-like to solid-like is the indication of three-dimensional physical networks formed in this nanocomposite. As it is obvious, the viscoelastic behavior of sample containing 1 phr of Cloisite 15A remains very similar to neat PBT/PP blend. The viscoelastic behavior of sample reinforced with 5 phr of Cloisite 15A shows adequate enhancement in storage and loss moduli compared to neat polymer blend, at low frequency region. Moreover, the primary slope of storage and loss modulus curves for nanocomposites based on Cloisite 30B is lower than specimens filled by Cloisite 15A. Also, the nonterminal behavior observed for nanocomposite based on 5 phr of Cloisite 30B is not recognized explicitly in nanocomposites based on Cloisite 15A. Based on linear viscoelastic investigation, the sample containing 5 phr of Cloisite 30B shows more solid-like behavior at low-frequency regions. Then, the degree of elastic interactions formed between PBT chains and Cloisite 30B layers in BP30B5 is higher than other samples. The outcomes of rheological properties together with morphological features prove that both types of organoclays represent compatibilization ability in PBT/PP blend, whereas the efficiency of hydrophilic nanoclay is higher than hydrophobic one. In addition, hydrophilic organoclays indicate higher strengthened effect than hydrophobic nanoparticles. In agreement with the research studies of Lipatov et al. 23,24 and Si et al., 25 if there is good affinity between nanoparticles and one of the components in immiscible polymer blends, these particles tend to be distributed around the interface and play the role of compatibilization. Besides, the exceed parts of nanoparticles usually take place in component with higher affinity to it. Based on this research, if the nanoparticles have higher affinity to matrix phase, the rheological parameter will aid to morphology development of immiscible polymer blends besides the interfacial features.
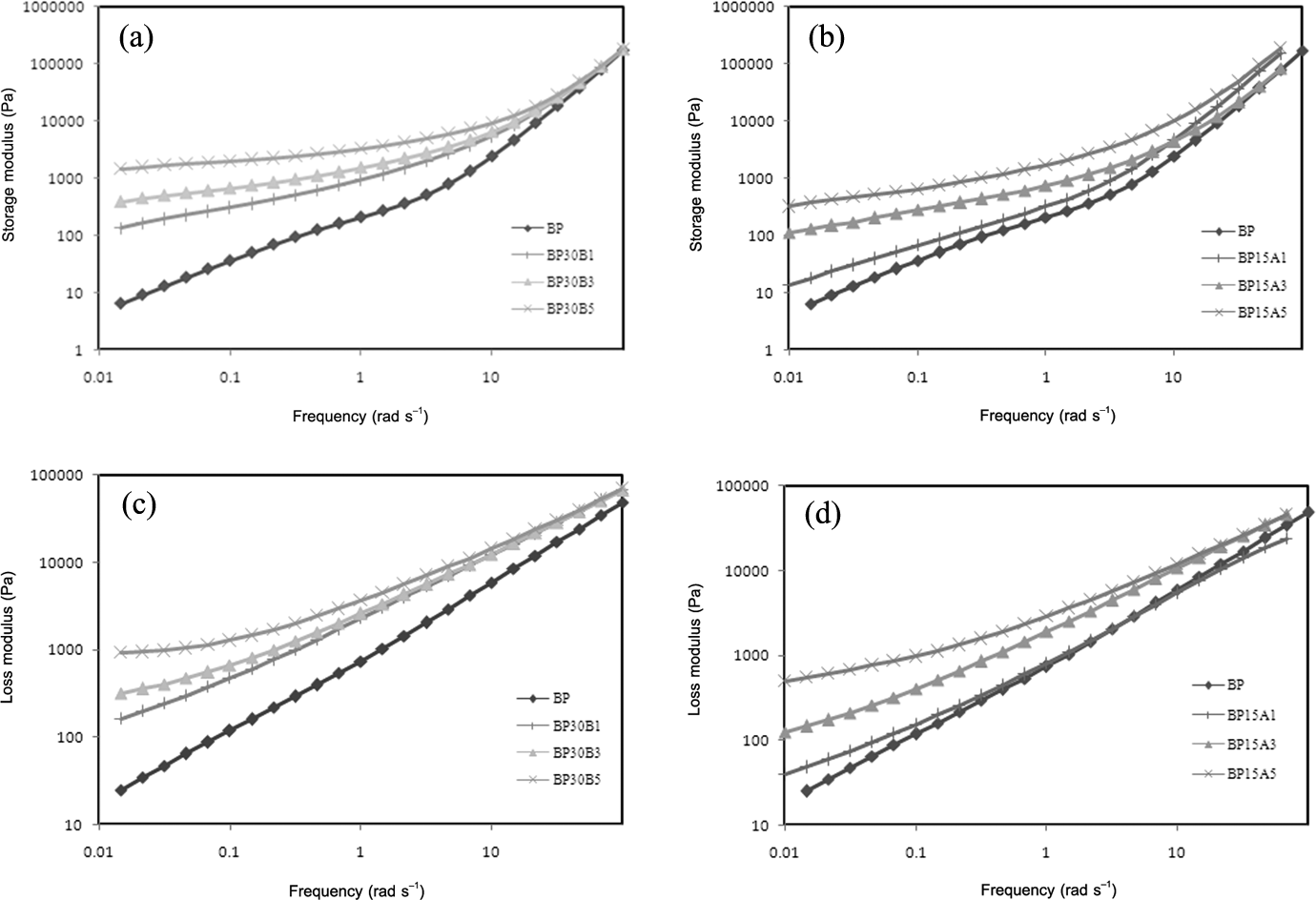
(a) Storage modulus of nanocomposites based on Cloisite 30B, (b) storage modulus of nanocomposites based on Cloisite 15A, (c) loss modulus of nanocomposites based on Cloisite 30B, and (d) loss modulus of nanocomposites based on Cloisite 15A, as a function of frequency.
Figure 7(a) and (b) represents the effect of Cloisite 30B and Cloisite 15A on storage modulus of PBT and PP, respectively. As clear as it is, Cloisite 30B has dramatic effect on enhancement of the elasticity of PBT phase. The plateau region of storage modulus in PBT containing 5 phr of Cloisite 30B, at low frequencies, is the indication of physical networks formed in this phase. Based on Figure 7(b), Cloisite 15A nanoparticles increase the elasticity of PP phase but the behavior of storage modulus is completely similar to PP component. Then, it can be said that hydrophobic organoclays don’t alter the viscoelastic behavior of PP so much and only play the role of thickening agent. Comparing Figures 6(b) and 7(b), it can be observed that the effect of hydrophobic nanoclay on enhancement of the elasticity of PBT/PP blend is higher than sole PP. Due to localization of Cloisite 15A nanoclays in PP phase and at the interface, the exceed values of storage modulus (elasticity) in PBT/PP/Cloisite 15A nanocomposites origin from the distribution of these nano layers at the interface. As Sundararaj and Macosko 35 declared, increment of the elasticity of matrix phase aids to easier breakup of droplets while the enhancement of dispersed-phase elasticity prevents the breakup of droplets. As a result, hydrophilic nanoclays help to morphology development of PBT/PP blend with increasing of matrix elasticity. On the other hand, hydrophobic organoclays hinder the breakup of droplets by the increment of dispersed-phase elasticity.
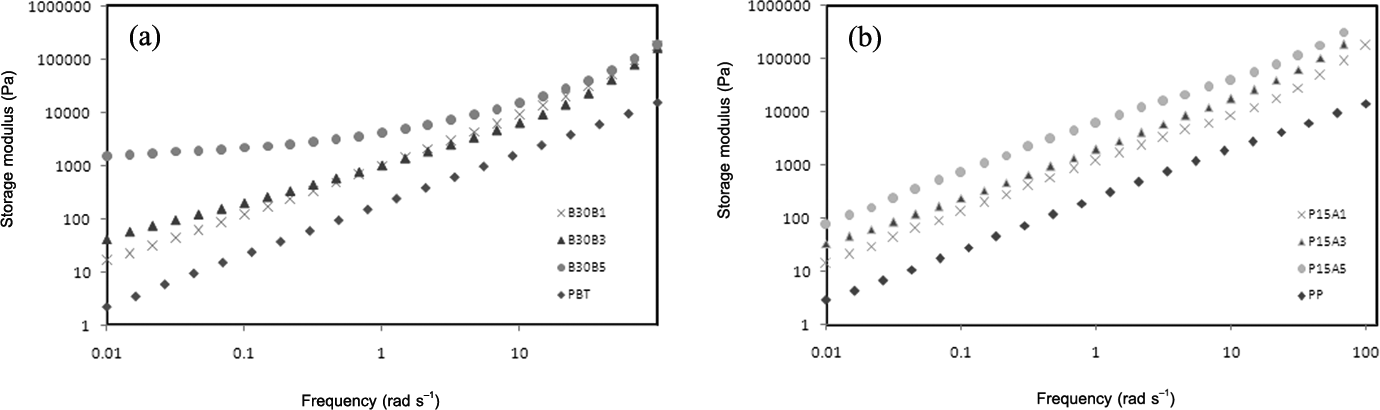
Storage modulus against frequency for (a) PBT and PBT/Cloisite 30B nanocomposites and (b) PP and PP/Cloisite 15A nanocomposites. PBT: poly(butylene terephthalate); PP: polypropylene.
Conclusion
The morphology and rheological behavior of hydrophilic and hydrophobic organoclay-containing PBT/PP polymer blend were investigated in this research. It is found that the intercalation structure in nanocomposites reinforced by hydrophilic nanoclay is higher than samples filled by hydrophobic one. Based on TEM, hydrophilic nanolayers are confined in the PBT matrix and at the interface, whereas the hydrophobic organoclays are located in the PP dispersed phase and at the interface. The nonhomogeneous distribution of both types of organoclays at the interface results in coalescence suppression of dispersed droplets and lowers the interfacial tension. On the other hand, the rheological parameters (viscosity and elasticity) aid morphology development of nanocomposites reinforced with hydrophilic organoclay, whereas the rheological parameters hinder the breakup of droplets in nanocomposites filled by hydrophobic one. As shown by SEM micrographs, hydrophilic nanoparticles indicate greater compatibilization efficiency than hydrophobic in PBT/PP blends. From the rheological point of view, hydrophilic organoclays affect more pronounced than hydrophobic, on complex viscosity and elasticity of PBT/PP blend. Solid-body viscoelastic behavior is the indication of three-dimensional physical networks formed in nanocomposite containing 5 phr of hydrophilic nanoclay showed stronger pseudoplasticity than other samples.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
