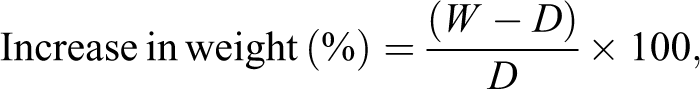Abstract
In this attempt, we have designed novel poly(ester–urethane) (PEU) using poly(di(ethylene glycol)/trimethylolpropane-alt-adipic acid), tolylene-2,4-diisocyanate, and novel diol. Later, PEU has been employed for the fabrication of nanocomposites and foams with hydroxyl-modified montmorillonite (MMT-OH) via in situ polymerization. Afterward, structure and physical properties of PEU/MMT-OH nanocomposites and foams have been explored using various techniques. Physical characteristics of nanocomposites and foams seemed to be dependent upon polyurethane structure, modification of layered silicate and physical interaction between matrix and organoclay platelets. Field emission scanning electron microscopy revealed distorted honeycomb morphology of PEU/MMT-OH 1–5 foams, while PEU/MMT-OH 1–5 nanocomposites depicted dispersed MMT-OH in the matrix. Increased cell density in nanocomposite foams was also observed relative to pure PEU foam. PEU/MMT-OH 5 (5 wt% MMT-OH) foam showed improved tensile strength of 58.1 MPa relative to PEU/MMT-OH 1 foam (56.8 MPa). The density of the foams was also increased (0.7–1.7 g cm−1) with clay loading. The 10% thermal decomposition temperature of PEU/MMT-OH 1–5 foams measured by thermogravimetric analysis was in the range of 431–465°C. Percentage of water absorption was also measured for the foam materials. Dynamic mechanical thermal analysis of PEU/MMT-OH 5 foam with 5 wt% nanofiller showed higher glass transition temperature (Tg) of 129°C relative to PEU/MMT-OH 1 (Tg 116°C). UL 94 and limiting oxygen index results showed that PEU/MMT-OH 1–5 foam had increased nonflammability (V-0 rating) with the clay loading. Silicate layers of MMT-OH were well exfoliated in PEU matrix due to chemical reaction between the hydroxyl of MMT-OH and functional groups of PEU. The morphology, mechanical, thermal, and flame retardant properties of PEU/MMT-OH 1–5 foams were found to be superior to those of PEU/MMT-OH 1–5 nanocomposites.
Introduction
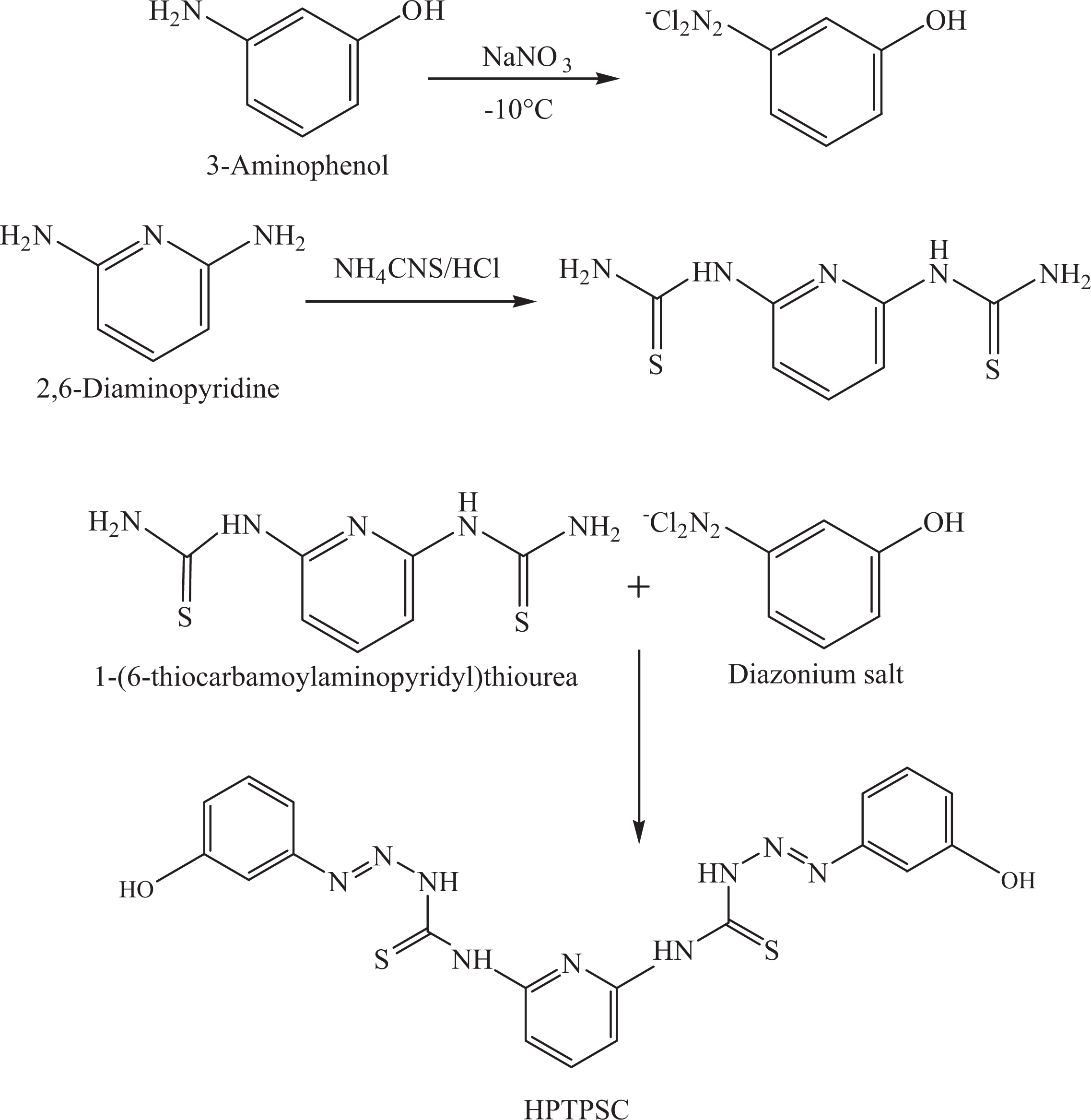
Fire retardants are frequently integrated into the polymeric materials to perimeter their flammability and to augment their frequent everyday use. 1 Success of the approach has been observed with low flammability polymeric materials rendering fine conciliation between price and properties. Scrupulous interest has been built-up regarding the development of nanocomposite technology consisting of polymer and organically modified clays since they often exhibit remarkably improved properties relative to virgin polymer. 2 In this regard, polyurethane foams have remarkably broad range of applications including thermal insulation, cushioning, buoyancy, energy absorption, and so on. Significant aerospace relevance of novel low-density foams embraces the design of light/stiff aircraft-interior panels, structural shapes such as bulkhead core, transom cores, stringers, motor mounts, and so on. Mechanical properties are also important considerations in fiber-reinforced plastic-based crash pads, boat building, composite foam cores, mold patterns and plugs, sports equipment core material, composite tooling, and so on. Polyurethane foams have been frequently obtained through reactive processing, in which polymerization and foam blowing occur concurrently. 3 Recent research has revealed polyurethane nanocomposites as capable materials in the area of flame retardancy, offering significant recompenses over traditional designs. Thermoplastic nanocomposite foam filled with clay has been found to decrease the cell size and boost the cell density in morphological patterns. Correspondingly, polyurethanes in conjunction with conventional flame retardants consequence optimistic effects on flame retardancy of clays. 4,5 Nevertheless, the total loading usually lies between 1 wt% and 15 wt% to achieve acceptable performance. Modification of other physical properties of polymers like mechanical and thermal profile has also been observed. The main rationale is the tremendously elevated aspect ratio and the equivalent length scale rendering clay clusters tranquil reinforcing agents. 6,7 Additionally, facile inclusion of polymer chains into interlayer spacing of the layered clay structure has been observed due to fine cation exchange capability. Consequently, splitting of clay platelets may initiate better interactions between clay and polymer matrix. Besides, vitals to reinforcement have been found to be strappingly dependent on the extent of clay exfoliation and dispersion within the polymer matrix. 8,9 Clay type, clay content, and polymer molecular structure have been investigated as significant characteristic implementation of the clay dispersion in these nanocomposites. 10 Fundamentally, montmorillonite (MMT) exchanged with long-chain molecules have good compatibility with polyol. The extent of gallery expansion of modified MMT is mainly determined by the chain length. 11 Thermal stability, mechanical properties, and gas permeability of polyurethane nanocomposites have been found to increase via fine dispersion of layered silicate structure. 12,13 Modification of MMT with hydroxyl active surfactants has also been adopted to attain fine dispersion of clays in polyurethane via in situ reaction. 14 –16 The presence of hydroxyl groups has been known to improve the intragallery polymerization, which in sequence initiate improved clay dispersion. 17,18 The melt blending of organically modified MMT (OMMT) with ethylene vinyl acetate copolymer has been adopted to form nanocomposite foams. 19 The effect of OMMT content on the cellular structure and mechanical properties of foams has been investigated. In another attempt, the effect closite20A nanoclay content on cell microstructure, mechanical properties, thermal conductivity, and flame retardancy of nanocomposite foam (prepared by extrusion method) samples was studied. 20 The foam matrix used was low-density polyethylene with polyethylene graft maleic anhydride as compatibilizer. The average cell size was found to reduce, however, the cell density and microstructure uniformity was increased owing to the addition of compatibilizer in the nanocomposite foams. Presently, we have modified the physical properties of poly(ester–urethane) (PEU)/modified MMT (MMT-OH) nanocomposites and foams have been investigated. A novel dihydroxide monomer, (E)-1-(3-hydroxyphenyl)-4-(6-(3-(3-hydroxyphenyl)thioureido)-pyridin-2-yl)thiosemicarbazide (HPTPSC), was prepared in this regard. HPTPSC is a dihydroxy compound, (Figure 1), which acted as a chain extender during the synthesis of novel PEU. HPTPSC reacted with tolylene-2,4-diisocyanate and polyol through simple condensation to form PEU, as shown in Figure 2. In polyurethane, HPTPSC formed the hard segments to increase the structural rigidity and other physical properties. This short chain compound formed the hard segment in the PEU. Accordingly, PEU/MMT-OH nanocomposites in addition to their foams were prepared via in situ polymerization and foaming with varying modified clay contents. PEU was prepared using poly[di(ethylene glycol)/trimethylolpropane-alt-adipic acid], tolylene-2,4-diisocyanate (95%), and novel diol. Mechanical, morphological, thermal, and flame retardant properties of nanocomposites and foams were determined via Fourier transform infrared (FTIR) spectroscopy, field emission scanning electron microscopy, transmission electron microscopy (TEM), thermogravimetric analysis (TGA), dynamic mechanical thermal analysis (DMTA), tensile, limiting oxygen index (LOI), and UL 94 tests. Exploration of the influence of modified nanoclay content on morphology, flame retardancy, and other physical properties of nanocomposite foams exposed significant fallouts.
Synthesis of HPTPSC. HPTPSC: (E)-1-(3-hydroxyphenyl)-4-(6-(3-(3-hydroxyphenyl)thioureido)-pyridin-2-yl)thiosemicarbazide.
Synthesis of PEU. PEU: poly(ester–urethane).
Experimental
Instruments
FTIR spectra of thin films of block copolymer and nanocomposites were recorded using Excalibur Series FTIR spectrometer, Model No. FTSW 300 MX manufactured by Bio-Rad (Hercules, California, USA). Nuclear magnetic resonance (NMR) spectra were scanned at room temperature using Bruker spectrometer (300.13 MHz for proton nuclear magnetic resonance (1 H NMR); Billerica, Massachusetts, USA) in deuterated dimethyl sulfoxide (DMSO-d6). Stress–strain response of the hybrid samples (strips) with approximately 14 × 5.4–8.0 × 0.25–0.62 mm3 dimensions was monitored according to Deutsches Institut Rir Normung procedure 53455 at a crosshead speed of 5 mm min− 1 using Testometric Universal Testing Machine M350/550 (Lancashire, UK). To determine various mechanical properties, standard procedures and formulae were used. For phase morphological studies, composite films were cryogenically fractured in liquid nitrogen and the morphology was investigated by FEI Nova 230 field emission scanning electron microscope (Hillsboro, Oregon, USA). The average cell radius in nanometers was determined from the data of SEM observation. For TEM measurement, samples were prepared at −60°C with an Ultracut E ultramicrotome by Reichert and Jung (Vienna, Austria) using a diamond knife. Measurements were carried out on a LEO 912 Omega (120 kV).
The cell density was calculated using equation (1):
where Nc is the cell density (cells × cm−3), n is the number of cells observed in a scanning electron micrograph, A is the area of the micrograph (cm2), and M is the magnification factor. Thermal stability of the nanocomposites was determined by NETZSCH thermo gravimetric analyzer, model no. TG 209 F3 (Germany), using 1–5 mg of the sample in aluminum oxide crucible from 25°C to 800°C at a heating rate of 10°C min− 1 under nitrogen atmosphere with a gas flow rate of 30 mL min− 1. The DMTA was performed on nanocomposites in the temperature range of 0–300°C with DMTA Q800 (frequency of 5 Hz, heated at 10°C min− 1). The LOI value was measured on sheets (100 × 5.5 ×3 mm3) according to the standard oxygen index test ASTM D2863-77 via Flame Temperature Analyzer (FTA), IL, USA. UL-94 test was measured on sheets (110 × 11.1 × 3 mm3) according to ASTM D635-77 for UL-94 test. Moisture absorption test was performed according to ASTM C272 standards. Test specimens were prepared 3 ×3 × 0.5 in. 3 thickness. The samples were first dried and weighed on an analytical balance. After that the test samples were placed into deionized water for a given period of time (24, 48, 72, and 96 h). The specimens were removed from water and weighed for the assessment of water absorption percentage. The increase in mass was calculated as:
where W is the wet weight and D is the dry weight.
Materials
Ammonium thiocyanate (98%) and dimethylformamide (99%) were supplied by Fluka (St Louis, Missouri, USA). Poly(di(ethylene glycol)/trimethylolpropane-alt-adipic acid), polyol (average Mn approximately 2300), tolylene-2,4-diisocyanate (TDI, 95%), 2,6-diaminopyridine (97%), and 3-aminophenol (98%) were provided by Sigma-Aldrich (St Louis, Missouri, USA). A mixture of cyclopentane and isopentane at 70:30 by weight was used as the blowing agent. 1,3,5-Triacryloylhexahydro-1,3,5-triazine 98% was used as the surfactant to prepare foams. The commercial nanoclay used in this study was provided by Southern Clay Products Inc, Cloisite 30B (MMT-OH) (Gonzales, Texas, USA), a MMT modified by bis-2-hydroxyethyl ammonium with a concentration of 90 mequiv./100 g clay.
Synthesis of HPTPSC
Firstly, 3-aminophenol (0.04 mol) was added to concentrated hydrochloric acid (HCL; 10 mL) and then dissolved in distilled water (50 mL). The mixture was placed in a freezing-bath to cool to 10°C to form precipitate. To the above suspension, 0.04 mol sodium nitrite solution (20 mL) was added with constant stirring for 1 h at 10°C to yield diazonium salt. 1-(6-Thiocarbamoylaminopyridyl)thiourea was prepared on a steam bath. The 2,6-diaminopyridine (0.2 mol), concentrated HCl (16 mL), ammonium thiocyanate (0.2 mol), and deaerated water (120 mL) were mixed and heated in a porcelain dish (2 h). The mixture was allowed to cool down to room temperature. The mixture was then evaporated to dryness to get 1-(6-thiocarbamoyl-aminopyridyl)thiourea (6–7 h). 21 The diazonium salt solution was added drop wise to the solution of 1-(6-thiocarbamoylaminopyridyl)thiourea (0.02 mol) in sodium hydroxide (NaOH; 10%) maintaining the temperature below −10°C (Figure 1). After stirring for 3 h, the mixture was acidified with dilute HCl (10%). The precipitate was filtered, washed with hot water, and then with HCl (3%). The precipitate was redissolved in NaOH (3%) followed by precipitation (HCl). The product was dried under vacuum at 80°C. Elemental analysis calculated: %C 48.81, %H 3.67, %N 26.96. Found = %C 43.44, %H 2.99, %N 22.68. FTIR (potassium bromide): 3422 cm−1 (–OH), 3365 and 1596 cm−1 (secondary (sec.) amine N–H stretch and bend), 3015 cm−1 (aromatic C–H stretch), 1419 cm−1 (–N=N– stretch), 1155 cm−1 (C=S stretch). 1 H NMR (300.13 MHz, DMSO-d6, δ (ppm): 5.11 (s, 2H, hydroxyl), 9.22 (s, 2H sec. amine), 8.13 (s, 2H sec. amine), 6.56 (d, 2H Ar), 6.74 (d, 2H Ar), 6.91 (s, 2H Ar), 7.23 (t, 2H Ar), 5.89 (d, 2H pyridine), 7.51 (t, 1H pyridine).
Synthesis of PEU/MMT-OH nanocomposites and foams
PEU was prepared employing TDI, diol and poly[di(ethylene glycol)/trimethylolpropane-alt-adipic acid] at the molar ratio of isocyanate/hydroxyl group ratio (NCO/OH) = 1 (Figure 2). The clay content was opted between 1% and 5% based on the overall weight of PEU. Firstly, poly(di(ethylene glycol)/trimethylolpropane-alt-adipic acid), TDI, and diol monomer were dehydrated under vacuum overnight at 40–50°C. MMT-OH was also dehydrated at 100°C for 24 h prior to use. The clay was first mixed with diol monomer for 2 h and then mixed with TDI for 0.5 h. Polymerization was carried out at ambient condition. The prepared hybrid was then post-cured in an oven at 100°C for 4 h. For reactive foaming, 1,3,5-triacryloylhexahydro-1,3,5-triazine was used as surfactant at 0.66 wt% and pentanes as the blowing agent at 5.5 wt%. Cyclopentane and isopentane (70:30) along with the above mixture were mixed by an impeller at 2000 r min−1 and foaming occurred in a closed plastic container with fixed volume at ambient temperature. The foams were then kept at 100°C for 4 h to ensure completion of the reaction.
Results and discussion
FTIR analysis of PEU/MMT-OH nanocomposite foams
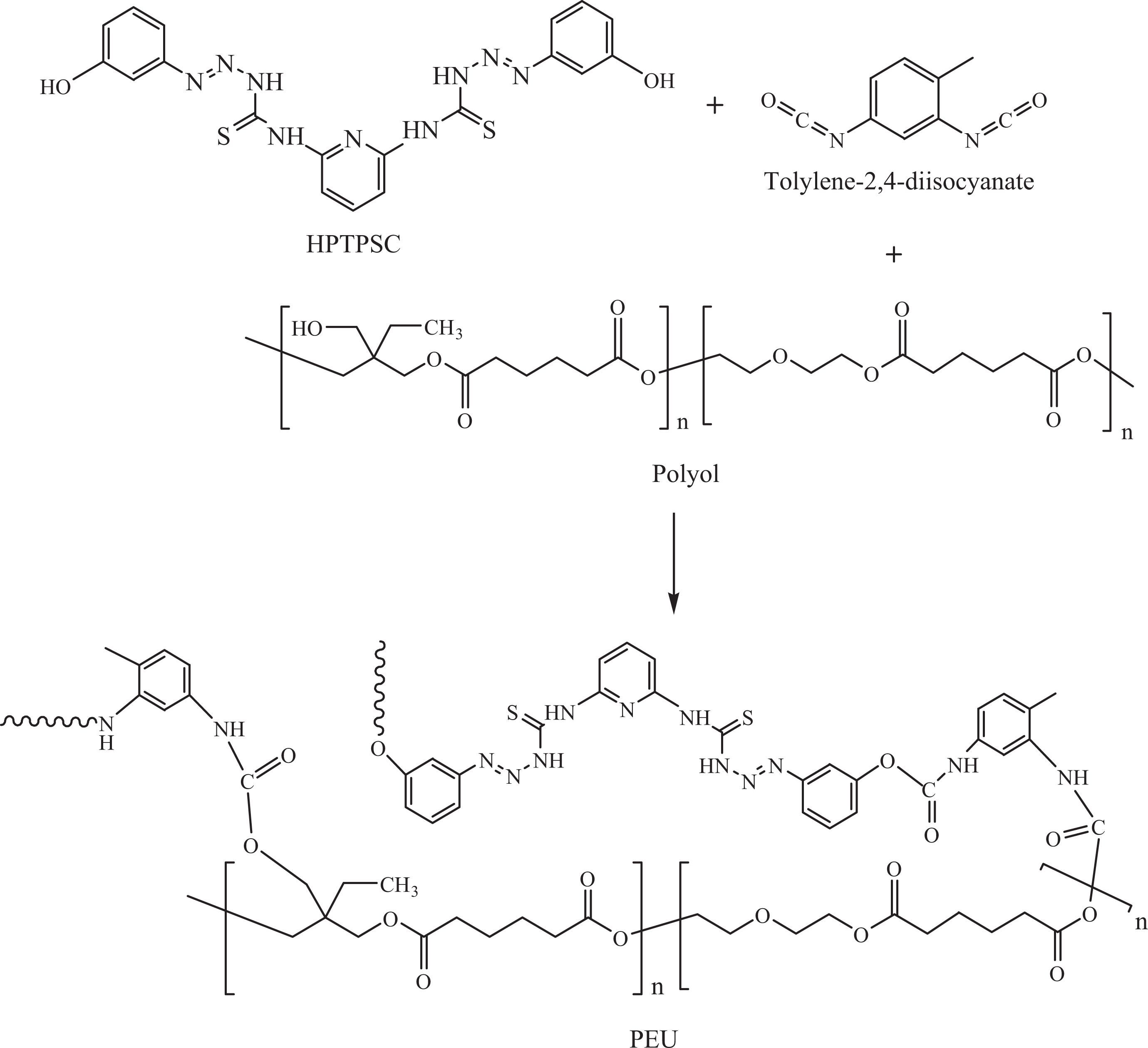
FTIR spectrum of PEU/MMT-OH 1 foam is given in Figure 3. All characteristic bands of polyurethane foam appeared in the corresponding spectrum. Accordingly, hydroxyl (O–H) stretching vibration appeared at 3435 cm−1. This band was related to free O–H groups of nonbonded polyol or O–H groups within the foams’ structure. NHCOO stretching and bending vibrations appeared at 3217 and 1594 cm−1, respectively. Additionally, aromatic protons were found at 3045 cm−1. The peaks for –CH2 were observed at 2961 and 2917 cm−1. Urethane carbonyl (C=O) in NHCOO was found at 1718 cm−1, while ester C=O appeared at 1745 cm−1. PUE/MMT-OH 1 also displayed peaks related to C–N and C–O at 1277 and 1262 cm−1 , respectively. Stretching vibrations of –N=N– and C=S were experiential at 1405 and 1151 cm−1, respectively.
FTIR spectrum of PEU/MMT-OH 1. FTIR: Fourier transform infrared; PEU: poly(ester–urethane); MMT-OH: hydroxyl-modified montmorillonite.
Mechanical property study
Table 1 shows the tensile test results of neat PEU, PEU/MMT-OH nanocomposites, and PEU/MMT-OH foams. The resultant stress–strain curves obtained are given in Figure 4. Tensile properties studied are tensile stress, tensile modulus, elongation at break, and toughness. In general, mechanical properties are enhanced with nanoclay addition, whereas tensile strain may decline in the nanocomposites. When studying PEU, tensile stress, elongation at break, tensile modulus, and toughness were found as 43.6 MPa, 6.61, 7 GPa, and 198 J m−3, respectively. In the case of PEU/MMT-OH 1-5 nanocomposites, tensile stress increased from 44.2 MPa to 46.7 MPa with the addition of clay content (Figure 4(a)). Tensile modulus of PEU/MMT-OH 1-5 nanocomposites was also found to increase from 14 GPa to 19 GPa. Correspondingly, the toughness was increased from 244 J m−3 to 288 J m−3 in PEU/MMT-OH nanocomposites. As expected, the elongation at break decreased from 6.98% to 6.77%. In the nanocomposites, MMT-OH was capable of developing interfacial interaction with the polyurethane chains. There existed hydrogen bonding interaction between the ester and urethane carbonyl (C=O) and thiocarbonyl (C=S) of PEU and O−H group of the modified MMT. These interactions were in turn responsible for the improved mechanical properties of the nanocomposite with modified MMT loading. The addition of filler further increased the mechanical strength of the foams due to the formation of rigid cellular structure. The cellular structure incorporating the nanoclay not only increased the mass/volume of the PEU but also the stiffness of the foam materials. In other words, the nanocellular structure became stiff with the MMT-OH addition increasing the overall strength of the foams. Tensile stress of PEU/MMT-OH 5 foam was detected as 58.1 MPa, which was higher relative to PEU/MMT-OH 1 foam (56.8 MPa) and PEU/MMT-OH 3 foam (57.7 MPa). Tensile modulus for PEU/MMT-OH 1–5 foam also demonstrated rising trend with the clay loading as 21–25 GPa. Similar to the nanocomposites, PEU/MMT-OH 1–5 foam exhibited decrease in elongation at break from 7.25% to 6.88% (Figure 4(b)). Toughness of the foams displayed increasing trend akin to tensile stress and tensile modulus. PEU/MMT-OH 1 foam had toughness of 308 J m−3, PEU/MMT-OH 3 foam had 317 J m−3 and PEU/MMT-OH 5 foam showed 325 J m−3. Increase in tensile strength with the nanoclay addition was due to structural homogeneity of the foams caused by chemical linkage amid matrix and clay and also due to high aspect ratio of the layered silicate filler. Moreover, increase in mechanical properties of the foams was due to the filling up of unreinforced regions between PEU foam with clay particles resulting in efficient stress transmission. 22 Density of PEU/MMT-OH foams was an important feature to examine (Table 2). The density of neat PEU foam was found to be 0.7 g cm−3. Analogous to the trend found for the mechanical properties, the density was also increased with clay loading from 1.1 g cm−3 (PEU/MMT-OH 1 foam) to 1.6 g cm−3 (PEU/MMT-OH 5 foam).
Mechanical properties of PEU and PEU/MMT-OH nanocomposite and foams.
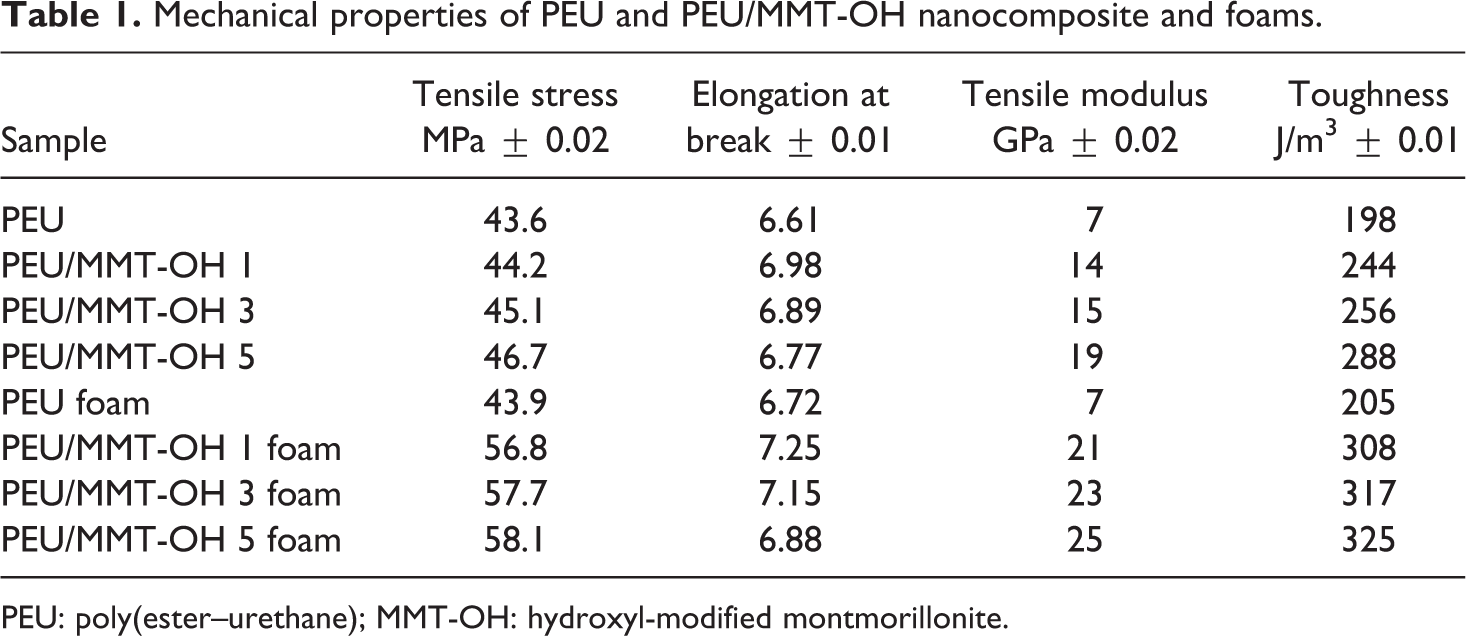
PEU: poly(ester–urethane); MMT-OH: hydroxyl-modified montmorillonite.
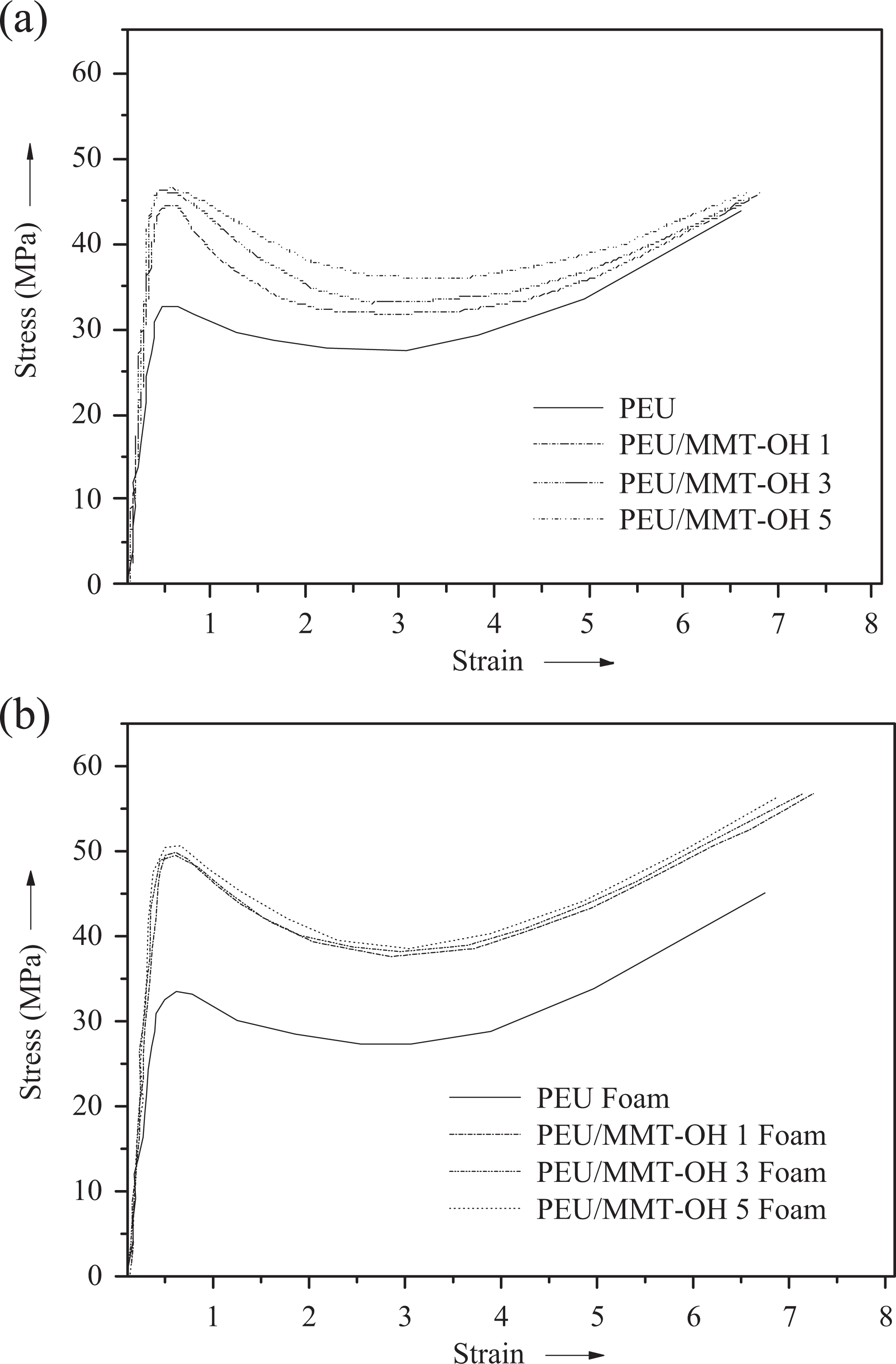
(a) Stress–strain curves of PEU and PEU/MMT-OH nanocomposite. (b) Stress–strain curves of PEU foam and PEU/MMT-OH foam. PEU: poly(ester–urethane); MMT-OH: hydroxyl-modified montmorillonite.
Cell density of PEU foam and nanocomposite foams.

PEU: poly(ester–urethane); MMT-OH: hydroxyl-modified montmorillonite.
Morphology investigation
Figure 5(a) and (b) shows the cryo-fracture surface images of PEU/MMT-OH nanocomposites. The micrographs showed somewhat layered structure of the nanocomposites with uniformly dispersed clay particles in PEU matrix. Micrographs of the foams are given in Figure 5(c) to (f). Neat PEU foam and PEU/MMT-OH 1-5 foams showed the expected cellular structure of foams. SEM image for neat polyurethane foam was observed as cellular structure having exceptionally large pentagonal cell constitution (Figure 5(c)). Nevertheless, PEU/MMT-OH 1–5 foams exhibited somewhat distorted cellular structures with adequately smaller cell size relative to neat PEU foam. Improved cell density and reduced cell size of nanocomposite foams were due to the chemical interaction between hydroxyl-modified clay and polyurethane during foaming process. Besides, some granular particles were visible inside the cells of nanocomposite foams (Figure 5(d) to (f)). The cellular structure with reduced cell size and improved density was accountable for the improved mechanical performance of new PEU/MMT-OH 1–5 foams. TEM images of PEU/MMT-OH 1 and PEU/MMT-OH 3 nanocomposites are displayed in Figure 6(a) and (b). Some of the clay layers were seemed to be dispersed in layers (Figure 6(a)). Relatively thicker clay fillers were found inside PEU/MMT-OH 3 containing more silicate layers. However, the dispersion pattern was approximately analogous having dark-layered structures representing clay platelets. The grown cells having diameter of 100 nm were localized along the dispersed nanoclay particles in the cell wall. The cell density of PEU/MMT-OH 1–5 was studied as an important factor. Cell densities were calculated to analyze the effect of clay loading on the mechanical properties and foaming behavior (Table 2). Cell densities of PEU/MMT-OH foam, PEU/MMT-OH 1 foam, PEU/MMT-OH 3 foam, and PEU/MMT-OH 5 foam were found to be 1.1 × 1010, 1.6 × 1010, 1.9 × 1010, and 2.1 × 1010 cells × cm−3, respectively. The cell density was increased with the clay loading while the cells’ size was somewhat decreased. The improved nanocellular density can also be related to the increased mechanical properties of the system.

FE-SEM images of (a) PEU/MMT-OH 1, (b) PEU/MMT-OH 3, (c) neat PEU foam, (d) PEU/MMT-OH 1 foam, (e) PEU/MMT-OH 3 foam, and (f) PEU/MMT-OH 5 foam. FE-SEM: field emission scanning electron microscopy; PEU: poly(ester–urethane); MMT-OH: hydroxyl-modified montmorillonite.

TEM images of (a) PEU/MMT-OH 1 and (b) PEU/MMT-OH 3. TEM: transmission electron microscopy; PEU: poly(ester–urethane); MMT-OH: hydroxyl-modified montmorillonite.
Thermogravimetric analysis
Table 3 shows the thermal stability of PEU/MMT-OH 1–5 nanocomposites and PEU/MMT-OH 1–5 foams estimated via TGA and DMTA. TGA plots of PEU/MMT-OH 1–5 nanocomposites are shown in Figure 7. Thermograms have shown that all the materials prepared demonstrated major weight loss via wide single step. Pure polymer showed preliminary weight loss at initial decomposition temperature (T0) of 401°C, 10% decomposition temperature (T10) at 422°C and maximum decomposition temperature (Tmax) at 465°C. As expected, the thermal stability of PEU/MMT-OH 1–5 nanocomposites and PEU/MMT-OH 1–5 foams was enhanced with the increase in the clay content. Accordingly, PEU/MMT-OH 1 displayed T0 of 403°C, T10 of 424°C, and Tmax of 478°C. The values were increased in PEU/MMT-OH 3 as T0 = 405°C; T10 = 425°C, and Tmax = 489°C and to T0 = 407°C; T10 = 429°C, and Tmax = 499°C in PEU/MMT-OH 5. Compared with the thermal properties of PEU nanocomposites, the T0 (404°C), T10 (423°C), and Tmax (466°C) of the PEU foam were slightly increased. T0, T10, and Tmax of nanocomposite foams also increased with increasing clay dosage. The char yield of PEU/MMT-OH 1–5 nanocomposites was found to enhance with the silicate loading from 19% to 22%. PEU/MMT-OH 1 foam with 1 wt% nanoclay showed T0 of 410°C, which was increased to 416°C in PEU/MMT-OH 3 foam with 3 wt% nanoclay and to 437°C in PEU/MMT-OH 5 foam with 5 wt% nanoclay. Correspondingly, in PEU/MMT-OH 1–5 foam T10 and Tmax were increased to 431–465°C and 505–523°C, respectively. Char yield of the foam was increased from 27% to 35%. Interaction between the nanofiller and matrix in fact enhanced the dispersion of filler in PEU and so enhanced the thermal stability of the nanocomposites and foams. Glass transition of the nanocomposites and foams was investigated using DMTA (loss tangent vs temperature) as illustrated in Figure 8. Glass transition temperature (Tg) of pure PEU was found to be 104°C, however with foaming, the Tg was found to increase to 105°C. In PEU/MMT-OH 1–5 nanocomposites, Tg was enhanced from 107°C to 112°C and in PEU/MMT-OH 1–5 foam it was increased to 116–129°C. Improved structural rigidity of foams with nanoclay addition was responsible for increase in Tg. When compared with the literature foams, thermal properties of PEU/MMT-OH 1–5 foams and nanocomposites were found to be superior. 23,24
Thermal analyses data of PEU and PEU/MMT-OH nanocomposites and foams.
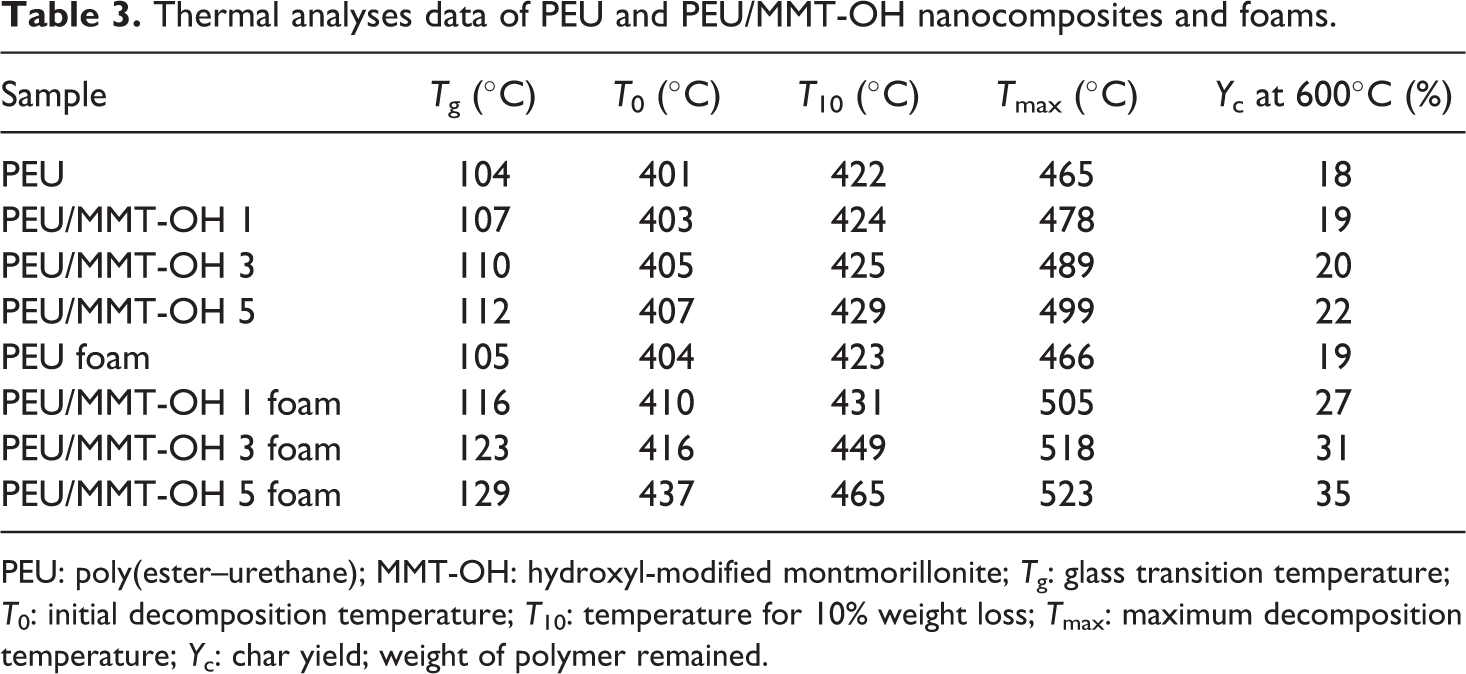
PEU: poly(ester–urethane); MMT-OH: hydroxyl-modified montmorillonite; Tg: glass transition temperature; T0: initial decomposition temperature; T10: temperature for 10% weight loss; Tmax: maximum decomposition temperature; Yc: char yield; weight of polymer remained.
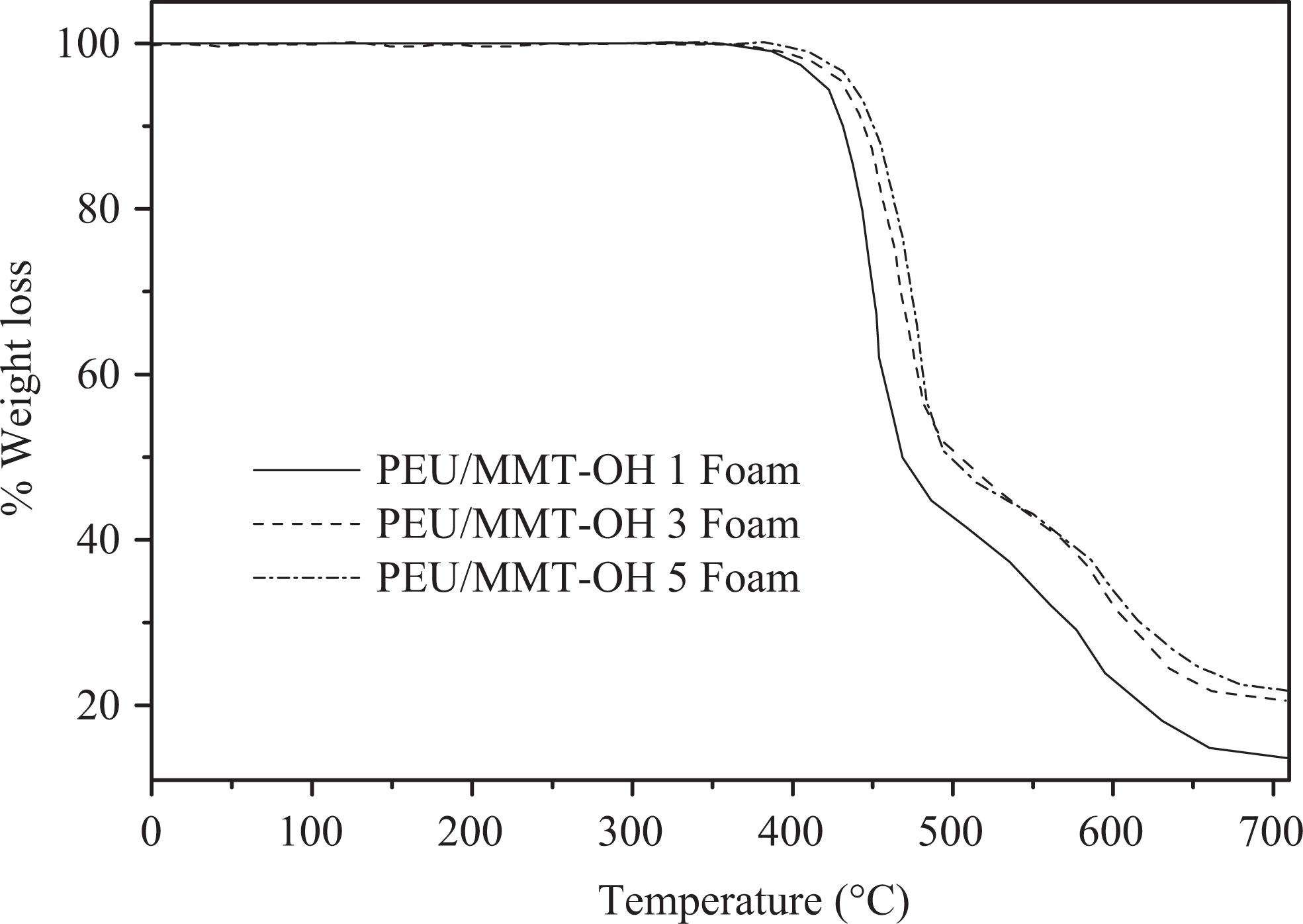
TGA curves of PEU/MMT-OH foams at 10°C min−1 (N2). TGA: thermogravimetric analysis; PEU: poly(ester–urethane); MMT-OH: hydroxyl-modified montmorillonite; N2: nitrogen.
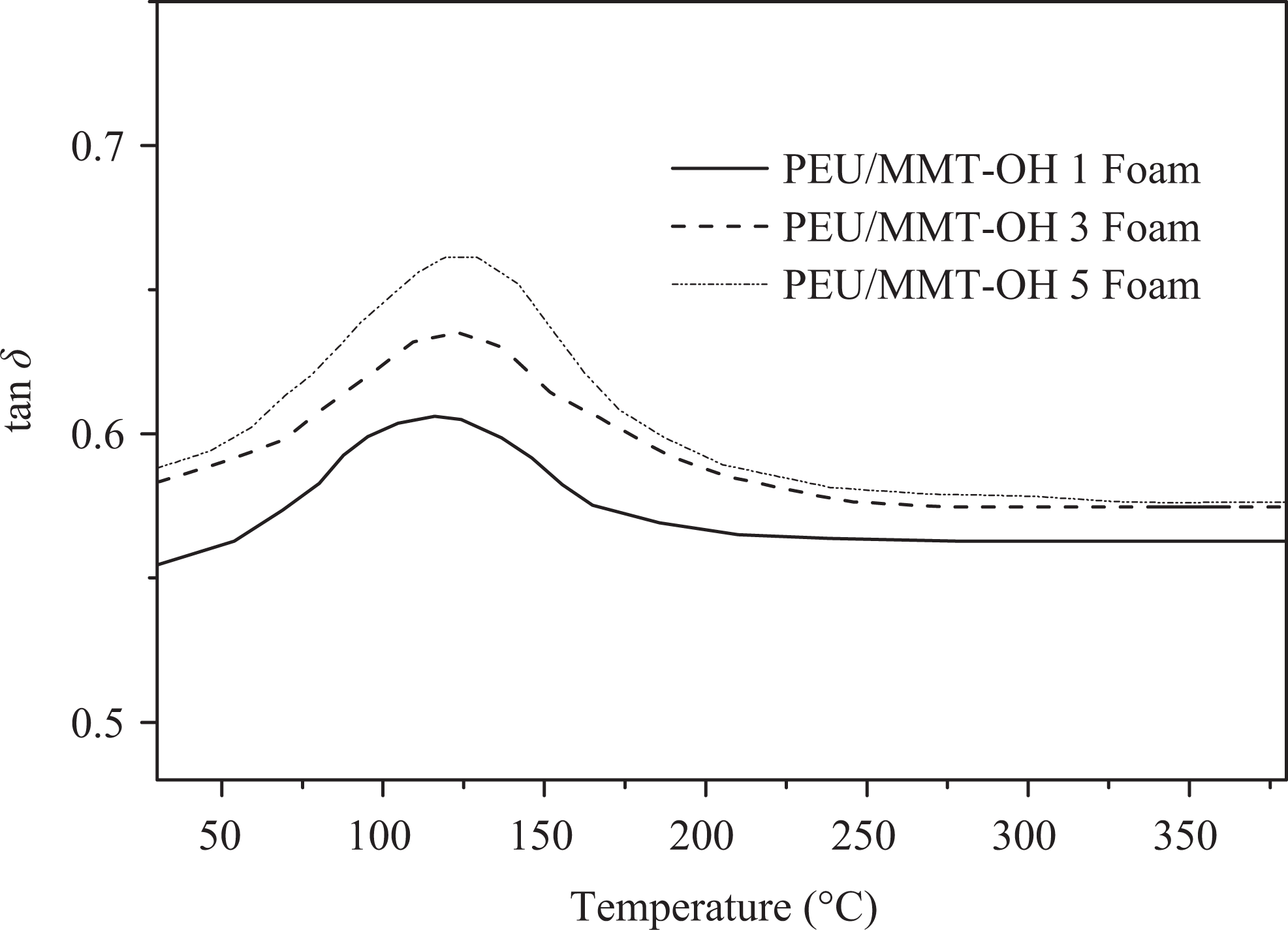
Variation of loss tangent with temperature for PEU/MMT-OH foams. PEU: poly(ester–urethane); MMT-OH: hydroxyl-modified montmorillonite.
LOI and UL-94 test of foam materials
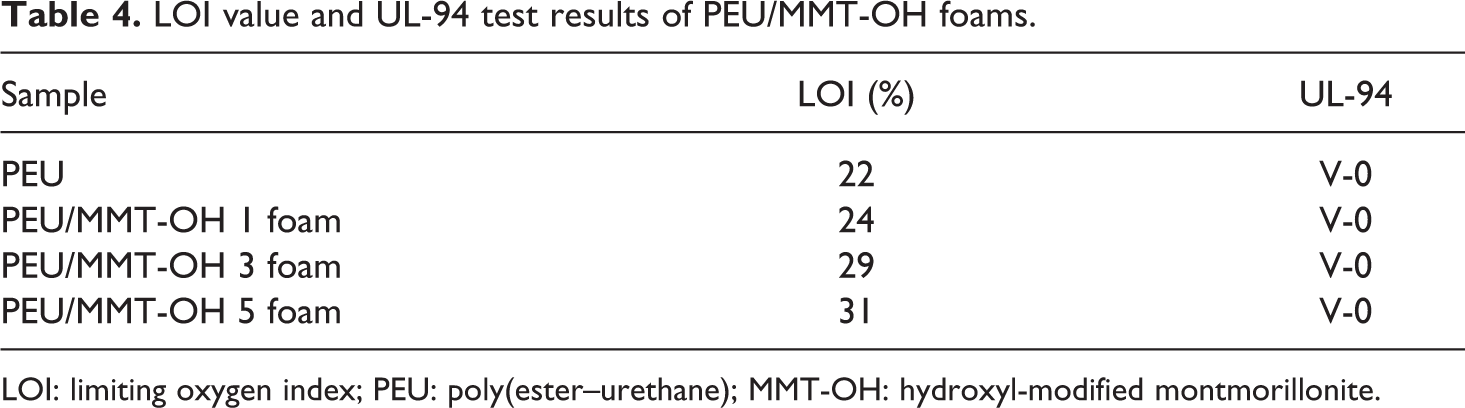
Flammability tests include LOI and UL-94 rating of PEU/MMT-OH foams as given in Table 4. In case of LOI measurement, adding 1 wt% clay in PEU/MMT-OH 1 foam resulted in increase in LOI value to 24% relative to pure PEU (22%). As predictable, further inclusion of nano-modified clay to PEU/MMT-OH 3 foam (3 wt %) improved the LOI value (29%). Similarly, inclusion of 5 wt% nano-modified clay in PEU/MMT-OH 5 increased LOI to 31%. Improvement in LOI value of the foams from 24% to 31% with 1–5 wt% nanoclay depicted that the material was adequately flame retardant. UL-94 rating of PEU/MMT-OH foams also revealed important results. V-0 rating in UL-94 test of PEU/MMT-OH 1–5 foam was indicative of the synergistic effect of the layered silicate and PEU structure in foams. In this regard, addition of clay might have contributed to the heat-shielding effect in rigid foams. 25 During vertical UL-94 test, a very rigid char layer was formed over the surface of PEU/MMT-OH 1–5 foam during the burning of material. The outer layer was sufficiently hard to block the release of combustible gases and protect the foam underneath. Increasing the nanoclay loading from 1 wt% to 5 wt% also exhibited V-0 rating in UL-94 test, thus, superb flame retardancy.
LOI value and UL-94 test results of PEU/MMT-OH foams.
LOI: limiting oxygen index; PEU: poly(ester–urethane); MMT-OH: hydroxyl-modified montmorillonite.
Moisture absorption test for PEU/MMT-OH foams
After weighing, the specimens were immersed completely in deionized water at specified time and temperature. After immersion period, the specimens were removed, dried, and weighed. Water absorption was reported as percentage of weight gain. The variation of percentage of moisture absorption versus time for rigid PEU/MMT-OH foams is shown in Table 5. It was observed that PEU/MMT-OH 5 foam showed maximum percentage of moisture absorption over the period of time. Water absorption percentage for PEU/MMT-OH 5 foam was found to be 8.11 after 96 h, which was higher among all the foam sample studied. On the other hand, for PEU/MMT-OH 1 foam the percentage of moisture absorption was found to be least, that is, 7.22 for 96 h. Furthermore, the percentage of moisture absorption for PEU/MMT-OH 1 foam was 11 times lower than that of PEU/MMT-OH 5 foam (over the period of 96 h). However, the water absorption capacity gradually increased over the period of time for all PEU/MMT-OH 1–5 foams.
Water absorption of PEU/MMT-OH 1-5 foams.

PEU: poly(ester–urethane); MMT-OH: hydroxyl-modified montmorillonite.
Conclusions
The aim of this work was to prepare some novel polyurethane/clay materials and to study the influence of clay modification and content on thermal decomposition trail, flammability, and tensile properties. For this purpose, PEU and hydroxyl-functionalized organoclay-based nanocomposites and foams have been fabricated. Accordingly, PEU foams were reinforced with varying MMT-OH content during processing to initiate in situ reaction. Modified MMT clay containing functional groups on platelets interacted chemically with polyurethane to generate an exfoliated structure. Influence of clay dispersion and content on the foam structure and properties revealed important results. Unlike the thermoplastic nanocomposite in which clay dispersion was observed, foams provided smaller cell size, higher cell density, and better nanoclay dispersion. The addition of nanoclay in the composites improved the thermal stability and increased the LOI values from 24% to 31%. Consequently, with the inclusion of 5 wt% functional organoclays, nanocomposite foams showed higher tensile, thermal, and nonflammable properties relative to other foams prepared and nanocomposites. To summarize, it can be concluded that the organo-modified clay loading have optimistic effects on the thermal and flammability properties of foams. New foams with improved heat-insulating ability may be employed for thermal insulation in ultimate energy savers.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.