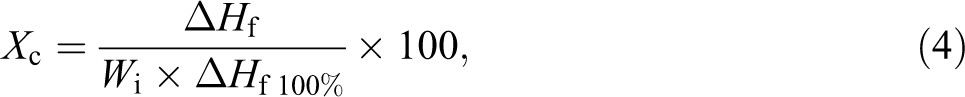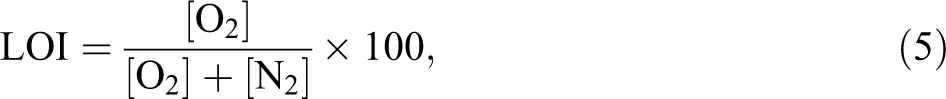Abstract
Poly(ethylene-co-vinyl acetate-co-carbon monoxide) (EVACO)/halloysite nanotube (HNT) nanocomposite films were solution cast. Dispersion of HNTs in the matrix was analyzed by elemental mapping and the role of HNTs on crystallizability, flammability and thermal, mechanical, and electrical properties of the polymer was evaluated. The nature of interaction between the EVACO matrix and HNTs was studied using Fourier transform infrared spectroscopy. The highest tensile strength was observed for the composite with 1% filler loading, whereas the highest crystallinity was observed for that with 3% filler loading. The decay in the tensile properties at higher filler loading was due to agglomeration of HNTs and debonding of polymer–filler interface. The electrical volume resistivity of the composites decreased with HNT loading because of the ionic charge transfer. The direct current electrical resistivity study of the composites proves that the addition of HNT can improve the antistatic properties of the polymer.
Introduction
Embedding rigid nanomaterials in polymer matrices is a widely accepted technique for improving several properties of polymer matrices, such as tensile strength, thermal stability, flame retardance, crystallinity, and so on. There is always a special interest on nanofillers, which are nontoxic, abundant, and inexpensive for use in polymer matrix composites. Nature is an abundant source of nanomaterials and naturally existing nanofillers satisfy all the aforesaid criteria and are important when large-scale production is considered. The never-ending search for nanofillers for polymer matrices resulted in the production of a large number of polymer nanocomposites (PNCs) filled with a huge variety of natural and synthetic nanofillers. Clay minerals and their derivatives are the most widely used nanofillers in polymer matrices, since clay is naturally and easily available nanomaterial that can be exfoliated to two-dimensional nanostructures.
The large surface area of nanomaterials plays a vital role in improving the interaction between the matrix and filler, thereby improving the properties of the matrices. Unlike macro- or micro fillers in polymer matrices, nanofillers act as multifunctional filler materials in many polymer composites. For instance, carbon nanotubes can simultaneously improve crystallization, 1 mechanical properties, 2 electrical conductivity, 3,4 and ultraviolet stability 5 of polymer matrices. Two-dimensional nanostructures, such as clays and layered hydroxides of which the majority is naturally occurring, are also capable of improving a number of properties of polymers except electrical conductivity. But, these can improve flame retardance 6 and barrier properties 7,8 of polymers. Every nanofiller has a unique functionality in a certain polymer matrix and it is incomparable with the same polymer matrix loaded with a different nanofiller. PNCs have widespread advanced applications since the scope of applications of virgin polymers is broadened by the addition of nanofillers with different functionalities.
Poly(ethylene-co-vinyl acetate-co-carbon monoxide) (EVACO) is a semicrystalline polymer with good adhesion to polar surfaces due to the carbonyl and acetate groups present in the backbone of the polymer chains. 9 The high polarity and the ability to form thin layers made EVACO as the ideal material for adhesion promoters in paints and coatings. Ethylene vinyl acetate (EVA) has an immediate analogy to EVACO; unlike EVA, the carbonyl groups in EVACO impart more polarity that may not be achievable by increasing vinyl acetate content in EVA. 10
The hazardousness of halogen and phosphorous compounds during their combustion resulted in the introduction of environmentally benign halogen-free nanofillers. Alumina trihydrate, montmorillonite, kaolinite, layered hydroxides, and halloysites are the best among the flame-retardant nanofillers. Halloysite nanotubes (HNTs) are formed by the weathering of aluminosilicate clay; its crystal structure has tetrahedral silicon tetroxide (SiO4) at the corners and octahedral aluminate (AlO6) at the edges in a stoichiometric ratio of 1:1. 11 In halloysite, the sheets of SiO4 and AlO6 are arranged in sequence and water molecules are present in between these layers. The unique performance of such nanofillers as flame retardants is due to the water molecules present in them, which will either dilute the free radicals at the flame front or act as barrier to the reactive species trying to reach the flame, thus extinguishing the fire. HNTs have found various applications, such as controlled release of drugs 12 and protective agents, 13 fillers in composites, 14 –17 emulsifiers, 18 organic pollutant sorbents, 11 and so on.
HNTs are used as nanofillers in a large number of polymer matrices to improve the mechanical, thermal, and crystalline properties of the polymers. In poly(lactic acid) (PLA)/HNT nanocomposite, the adhesion between the matrix and the HNTs was improved by the addition of maleic anhydride-grafted PLA and the resulting composites possessed improved mechanical properties as compared with pristine PLA. 19 The presence of HNTs improved not only the thermal and flame retardance properties of isotactic polypropylene 20 but also its crystallinity. 15 Chitosan/HNT nanocomposites are potential materials for tissue engineering scaffolds with good compressive strength and reduced water intake. 21 Addition of HNTs to poly(vinyl chloride) resulted in toughening and stiffening of the polymer. 22
Besides filler dispersion, filler–matrix interactions are also important for the performance of a PNC. The polymer chain dynamics are often affected by nanofillers, which in turn greatly influence the macroscopic characteristics of the polymer. 23 For instance, the majority of the properties of semicrystalline polymers are dependent on the amount of crystallinity in the matrix. Nanofillers, at low filler loading, are proven nucleating agents to improve crystallinity of polymer matrices. 15,24 The weak hydrogen bonding between filler and matrix can hinder the random movements of the polymeric chains, but it will help in arranging the chains in an organized fashion.
In this work, EVACO/HNT composites were fabricated through solution processing, since the industrial processing of EVACO is in the form of solution for the applications such as paints, adhesives, inks, and so on. The effect of HNT loading on the crystallinity and mechanical properties of the composites was analyzed. The ability of HNTs as flame-retardant filler in the EVACO matrix was also studied using UL-94 vertical burning test and limiting oxygen index (LOI) analysis.
Experimental details
Materials
EVACO (Elvaloy® 4924), (DuPont, Wilmington, DE, USA) with melt flow index = 35 g 10 min−1 and

TEM micrograph of HNTs. Inset shows the high-resolution image of the selected area. TEM: transmission electron microscopy; HNT: halloysite nanotube.
Preparation of EVACO/HNT nanocomposites
Four grams of EVACO was dissolved in 75 mL of DCM by continuous stirring at ambient temperature in a closed beaker. A good dispersion of predetermined quantity of HNTs in 25 mL of DCM was prepared by stirring and subsequent ultrasonication for 1 h. The dispersion of HNTs in DCM was slowly added to EVACO solution under stirring followed by ultrasonication for 1 h. 4 w/v% of polymer to solvent proportion was maintained throughout this study. The mixture was poured into glass Petri dishes to cast the sample films, the cast films were dried in a vacuum oven at 50°C for 6h, before further studies. EVACO/HNT composite films loaded with 1, 3, 5, 7, and 10 wt% of HNTs were prepared.
Characterization of nanocomposites
A scanning electron microscope (SEM; JSM-6380LA, JEOL, Japan) with energy dispersive X-ray spectroscopic (EDS; Link ISIS-300 Microanalytical System, Oxford instruments, UK) facility was used to quantify the degree of dispersion of HNTs in the EVACO matrix; elemental mapping of aluminum in the composite was carried out using EDS. The samples were sputtered with gold (JEOL JFC 1600), in an auto fine coater, prior to imaging. A transmission electron microscope (TEM, CM12 PHILIPS, Netherlands) was also used to image the finer morphological features of the HNTs and the cryomicrotomed sections of the nanocomposites.
X-ray diffraction (XRD) was used to determine the crystallinity of these nanocomposites. XRD patterns were recorded by JEOL XRD (DX-GE-2P) using copper K α (Cu K α) radiation. The samples were analyzed as sheets, at a scanning rate of 1°min−1. The degree of crystallinity (X c) of the virgin polymer as well as the composites was calculated by the XRD deconvolution method, where the amorphous and crystalline contributions to the diffraction spectrum were separated and the X c was calculated from the ratio of the integrated area of all crystalline peaks to the total integrated area under the XRD peaks. 25
The X c was measured using the following equation:
where I a and I c are the integrated intensities corresponding to the amorphous and crystalline phases, respectively.
Interplanar distance (d) and the approximate size of the crystallites (D) in the composites are obtained by the following equations:
where λ is wavelength of the X-rays (Cu K α = 1.5418 Å), β denotes the full width at half maximum (FWHM), θ is the Bragg angle, and approximately k = 0.9. The crystallite size was estimated using the reflections of the (110) plane.
Fourier transform infrared (FTIR) spectra (Jasco FTIR 4200, Japan) in attenuated total reflection mode were recorded in a wave number range of 650–4000 cm−1 for the representative samples at an average of 32 scans with a resolution of 0.5 cm−1.
Thermogravimetric analysis (TGA) measurements were performed under nitrogen flow of 25 mL min−1 using a TGA unit (TGA Q5000 V3.10 Build 258; TA instruments, New Castle, Delaware, USA) with near 6 mg samples. A constant heating rate of 10°C min−1 was maintained. TGA data collection for all the nanocomposites was done under identical conditions.
Differential scanning calorimetry (DSC) measurements were carried out for about 5 mg of the samples in sealed aluminum pans in a DSC analyzer (Mettler Toledo, Columbus, Ohio, USA), in nitrogen atmosphere from 0°C to 150°C at a heating rate of 10°C min−1.
The % crystallinity of EVACO was determined from the area under the endothermic peak using the following equation 26 :
where W i is the weight fraction of the polymer in the composite, X c is crystallinity (%), ▵H f is apparent melting enthalpy of crystallinity of EVACO (J g−1), and ▵H f100% is the extrapolated value of the enthalpy of crystallization of a 100% crystalline sample of EVA having a value of 68 J g−1. 27
The mechanical properties of the nanocomposites were tested by tensile measurements at ambient conditions in a Hounsfield Universal Testing Machine (H25KS; Hounsfield, UK). The testing was performed as per ASTM D638 standard for three dumbbell samples cut from the same composite sheets and their average is taken, at a crosshead speed of 50 mm min−1. Dumbbell specimens were prepared according to ASTM D412-B standard and an average of three tests was taken.
The relative flammability of the composite with respect to the control was determined by LOI and vertical burning test (UL-94). LOI corresponds to the minimum percentage of oxygen needed for the combustion of specimens. The LOI (equation 4) values were determined using a Dynisco LOI analyzer (Franklin, MA, USA) according to ASTM D2863 standard, and the vertical burning tests were performed according to the ASTM D3801 standard. LOI is defined as follows:
where [O2] is the volumetric flow rate of oxygen (cm3 s−1) and [N2] is that of nitrogen.
The direct current (DC) electrical resistivity measurements were made on thin films of 2 × 2 cm2 samples using two probes (MECO 81K; Mahape, Navi Mumbai, India) multimeter at ambient conditions following ASTM D257 standard.
Results and discussion
Morphology of the nanocomposites
SEM-EDS elemental maps
Figure 2 shows the aluminum elemental mapped images of the EVACO/HNT composites. The aluminum elemental maps help in estimating the dispersion of the HNTs in the EVACO matrix qualitatively. As the filler loading is increased, the number of HNTs is more and so is the number of agglomerations.

Aluminum elemental mapped SEM images at different levels of HNT loading: (a) 1%, (b) 3%, (c) 5%, (d) 7%, and (e) 10%. SEM: scanning electron microscopy; HNT: halloysite nanotube.
TEM analysis
Figure 3 shows the transmission electron microscopy (TEM) images of the EVACO/halloysite composite with 1% filler loading. The dense regions around the HNTs (Figure 3(a) and (b)) represent the crystallized polymer; it reveals the ability of nanotubes as nucleating agents. In the high-magnification image of the single HNT in EVACO matrix (Figure 3(c)), the boundaries of the tube are indistinguishable from that of the matrix, which may be due to good interaction of the matrix with the nanotube. Unlike the continuous diffraction rings in selected area diffraction pattern (SAED) for amorphous materials, the SAED pattern here is similar to that of a polycrystalline material (Figure 3(d)). The discontinuous diffraction rings are similar to that of polymer crystals with sheaf-like structure in which lamellae are oriented in parallel. 28

TEM images of EVACO/halloysite composite with 1% filler loading. TEM: transmission electron microscopy; EVACO: poly(ethylene-co-vinyl acetate-co-carbon monoxide).
FTIR spectral analysis
The FTIR spectra of two representative composite samples along with that of pure EVACO and HNTs are shown in Figure 4. In the spectrum of EVACO, the peak at 3418 cm−1 corresponds to the vibrations of adsorbed water molecules, as EVACO is hydrophilic due to the polar groups in it. The doublet at 2919 and 2850 cm−1 is attributed to the symmetric and asymmetric CH2 stretching vibrations, respectively. The peaks at 1736 and 1713 cm−1 are assigned to C=O stretching of vinyl acetate and carbon monoxide in the polymer backbone, respectively. The peaks at 1467 and 1375 cm−1 correspond to CH scissoring in –CH2– and symmetric deformation of CH in –CH3, respectively. The peak at 1231 cm−1 is due to the twisting and wagging of CH in –CH2–, the peak at 1019 cm−1 is due to C−O−C stretching, and the peak at 721 cm−1 can be attributed to the rocking vibration of CH. In the spectrum of halloysite, the peaks at 3697 and 3625 cm−1 are due to the aluminum hydroxide (Al2OH) stretching bands, each hydroxyl (OH) group is linked to two aluminum (Al) atoms, the peak at 1036 cm−1 is due to Si–O stretching, and the peak at 910 cm−1 corresponds to Al2OH bending. 29,30
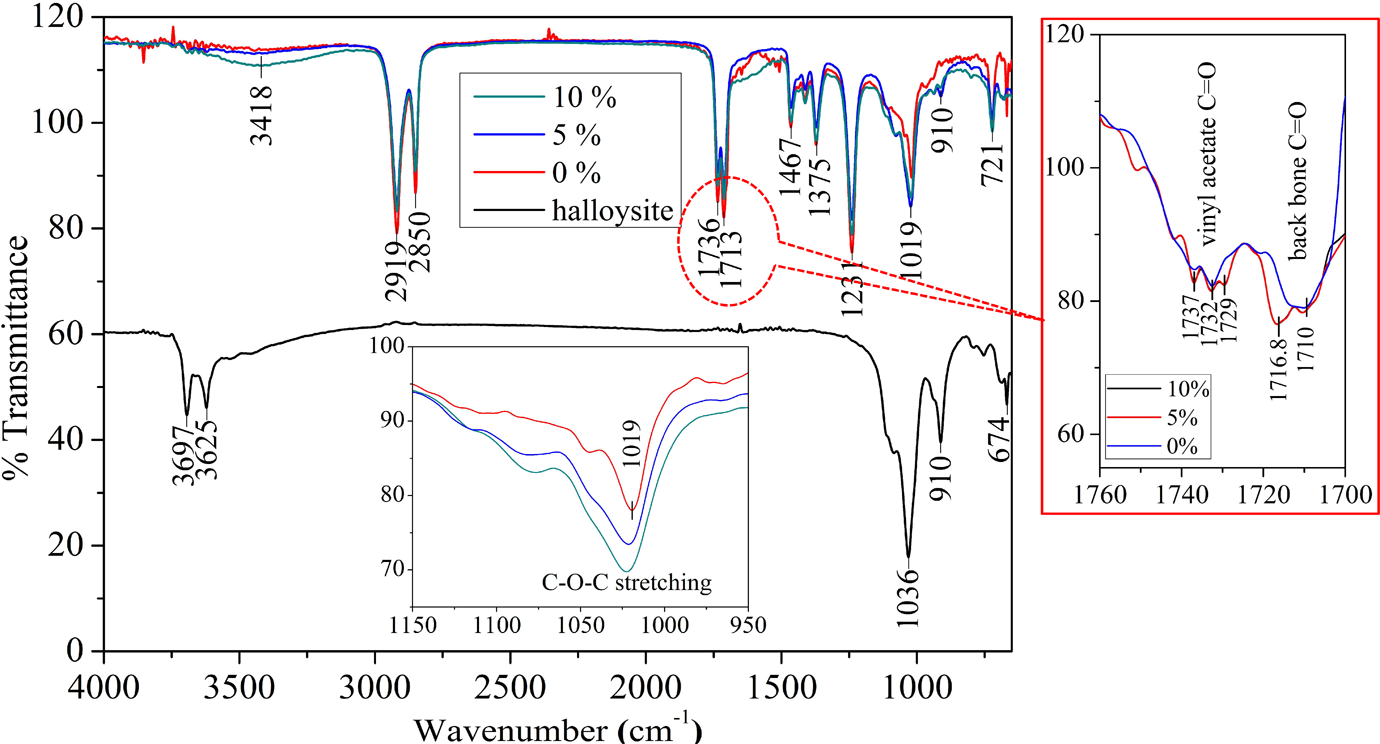
FTIR spectra of EVACO, HNT, and representative EVACO/HNT nanocomposites. FTIR: Fourier transform infrared; EVACO: poly(ethylene-co-vinyl acetate-co-carbon monoxide); HNT: halloysite nanotube.
The addition of halloysite to EVACO changes the intensity of OH stretching peaks in the spectra of the composite. The intermolecular hydrogen bonding between the polymer and the filler lengthens and weakens the O–H bond. 31 The peaks at 1732 and 1710 cm−1 correspond to C=O stretching of vinyl acetate and carbon monoxide repeating units in the polymer backbone (the enlarged region in Figure 4). In the spectra of the nanocomposites, the abovementioned peaks are split into multiplets, and some new peaks appear at slightly different wave numbers. This happens due to the intermolecular hydrogen bonding between the C=O groups of EVACO and the –OH groups of HNT. Also, broadening and shifting of the peak at 1019 cm−1 (inset of Figure 4), which corresponds to the C−O−C stretching of vinyl acetate, is an indication of the interaction of the Al–OH bonds of halloysite with EVACO. Also, the appearance of peak at 910 cm−1 represents the –OH groups in halloysite in the spectrum of the composite, which is not evident in the spectrum of pure EVACO. The types of interaction between the matrix and HNTs are represented schematically in Figure 5, and the interactions are highlighted by dotted circles.
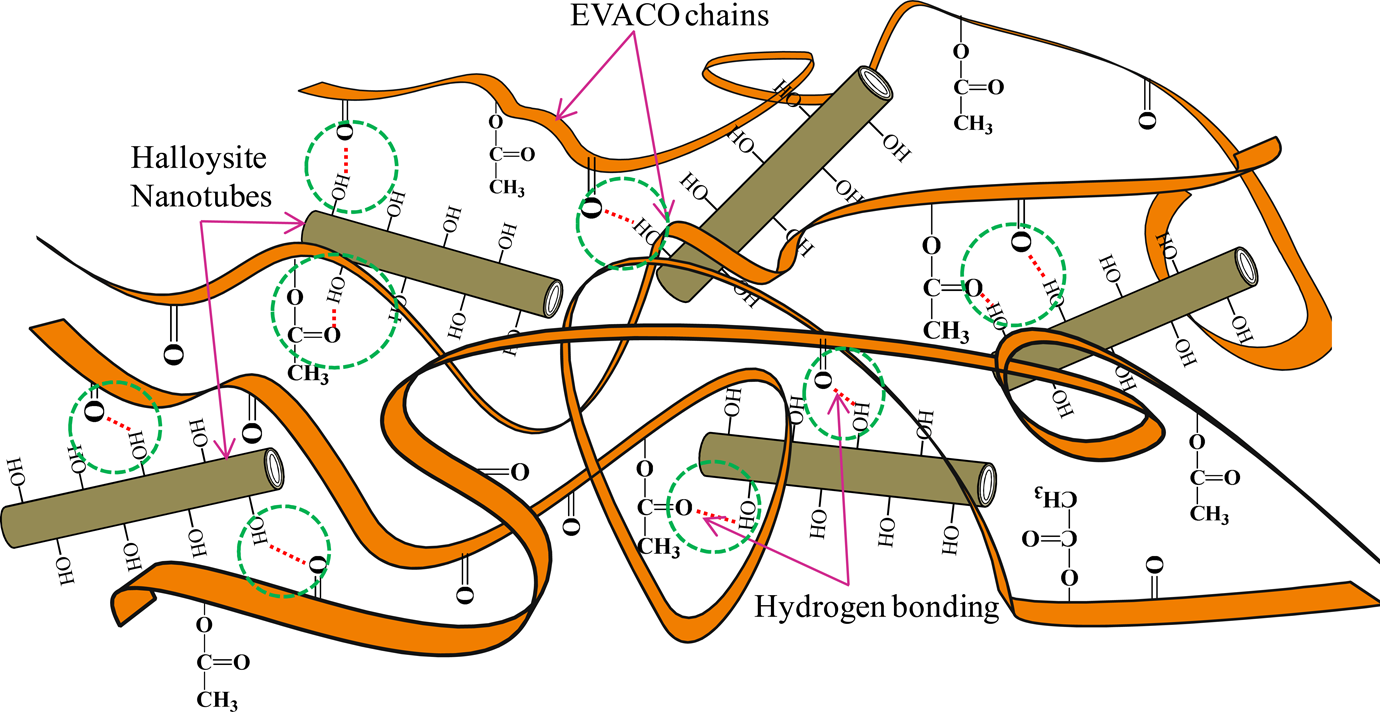
Schematic representation of types of interaction between HNTs and EVACO and the interactions are highlighted in dotted circles. HNT: halloysite nanotube; EVACO: poly(ethylene-co-vinyl acetate-co-carbon monoxide).
Crystallizability of EVACO/HNT nanocomposites
DSC analysis
The first heating and cooling DSC curves of EVACO and EVACO/halloysite nanocomposites at different filler ratios are shown in Figure 6. The segmental mobility of the ethylene chain sequences in EVACO determines the melting/softening temperature of EVACO. The maximum absorption of heat took place within the range of 20–100°C in all the composites. EVACO has a broad melting peak, which corresponds to the wide size distribution of the uneven and random crystallites in the virgin polymer, 32 due to the randomly placed crystalline polyethylene phases. An increase in the crystallinity of the composite over the pristine EVACO is observed at low filler loadings, 1 and 3%, which is due to the ability of HNTs to act as a nucleating agent as reported earlier. 15 However, at higher filler loading, the % crystallinity is reduced remarkably, which is due to the dilution of the spherulite growth front by the high-aspect ratio nanotubes. Also, these nanotubes are capable of arresting the movement of crystallizable polymer chains near the crystal growth front, which undergo crystallization if the nanotubes are absent.
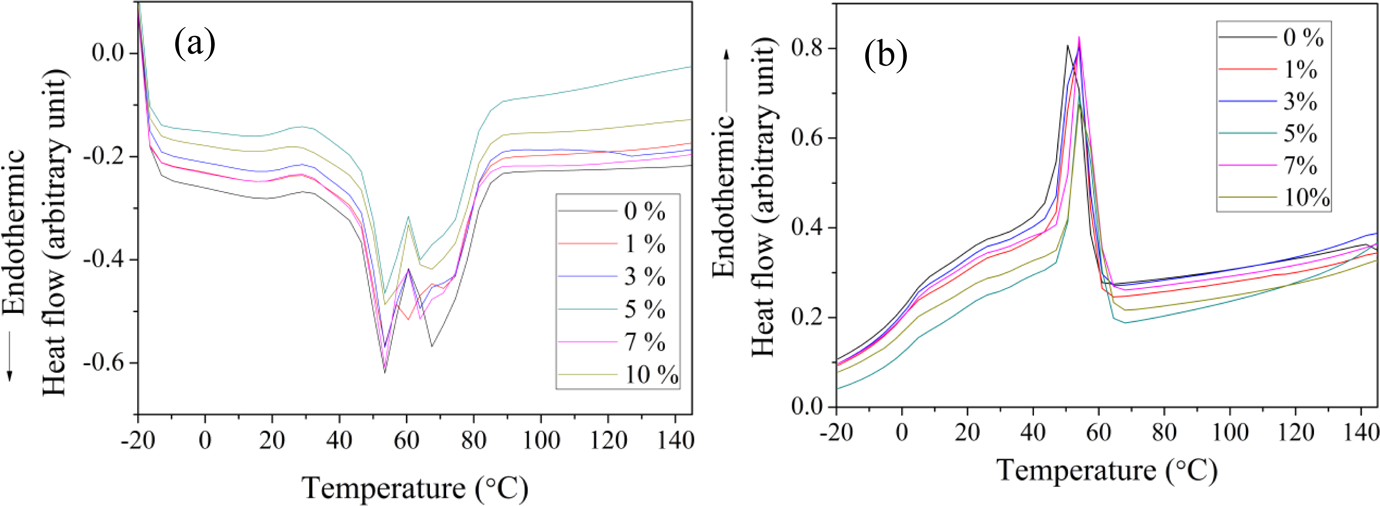
DSC curves of EVACO and EVACO/HNT nanocomposites: (a) heating and (b) cooling. DSC: differential scanning calorimetry; EVACO: poly(ethylene-co-vinyl acetate-co-carbon monoxide); HNT: halloysite nanotube.
During the first heating, the pristine EVACO has two major melting peaks. Unlike the pristine EVACO, the composites exhibit three melting peaks that correspond to α, β, and γ crystallites. The intensity of the second melting peak at high temperature is reduced in the presence of halloysite, which in turn is split into two peaks of lower intensities at low filler loading. Meanwhile, at high filler loadings (5, 7, and 10%), the γ peaks are absent and the intensity of β peaks has decreased in comparison with the pristine EVACO.
During the cooling process, only one melting peak was observed. During the solution casting process, the polymer chains are free to move and therefore thermodynamically favorable orientation of chains takes place before the complete evaporation of the solvent. This will ultimately result in the formation of different types of crystallites. But during melting, the chain movements are restricted and allow the formation of only one type of crystallite. Also, it is important to note that the addition of HNTs shifts the melting temperature of the composites to a slightly higher value. The % crystallinity and melting peak positions of the composites are presented in Table 1.
DSC results.
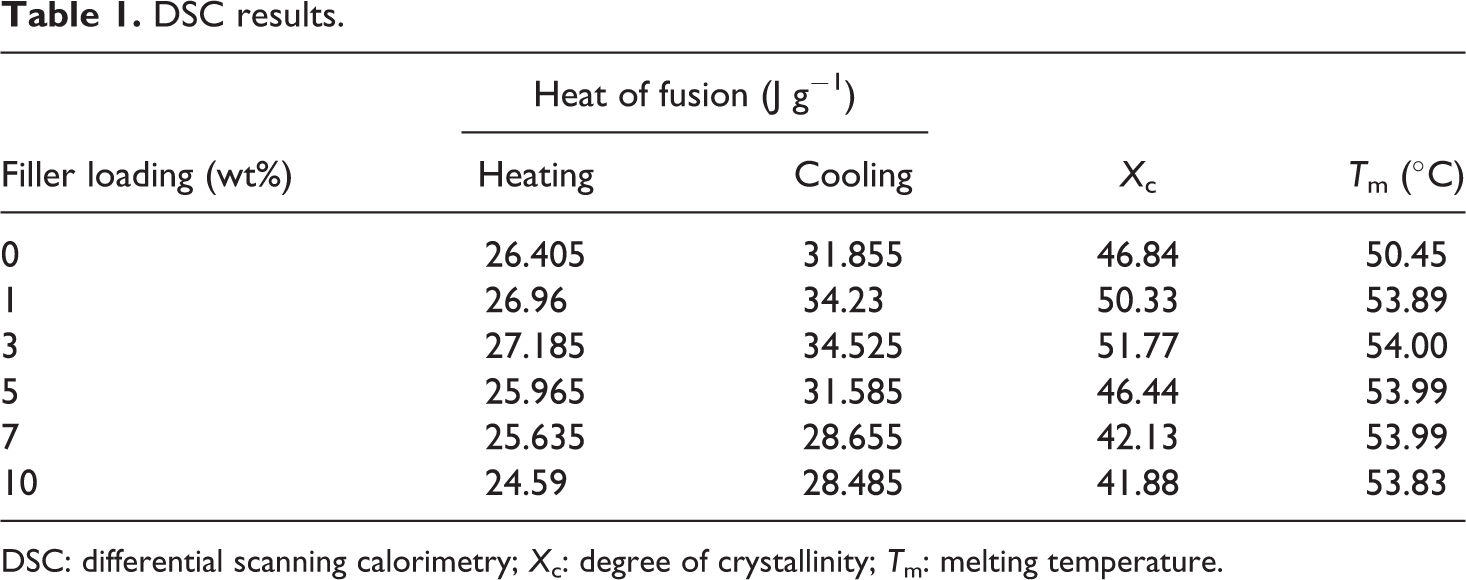
DSC: differential scanning calorimetry; X c: degree of crystallinity; T m: melting temperature.
XRD analysis
XRD analysis also reveals a similar trend, as observed in the DSC results. All the XRD patterns exhibit two distinct crystalline peaks revealing the semicrystalline nature of the polymer (Figure 7). The peak position, d spacing, FWHM, crystallite size, and % crystallinity of the pristine EVACO and its composites are shown in Table 2. The filler addition affects the semicrystalline nature of EVACO. The intense diffraction peak at an angle of 20°−22° corresponds to the reflection from the (110) plane and the broadness is due to the presence of amorphous region. The XRD patterns of the EVACO/HNT nanocomposites at different filler loadings are also shown (Figure 7). In general, the polyethylene phase in ethylene-based copolymers exhibits semicrystalline nature, since it can undergo crystallization. The crystallinity of the EVACO/HNT composites is a function of their filler content and the calculated values of X c are presented in Table 2. At certain filler loadings, that is, at 1 and 3%, the X c of EVACO resin is increased, which then decreases as the filler loading is increased further. It is expected that at 3% filler loading the number of nucleation sites are critical and above that the excess HNTs added to the matrix destroy the spherulite front surrounding the nucleation sites.
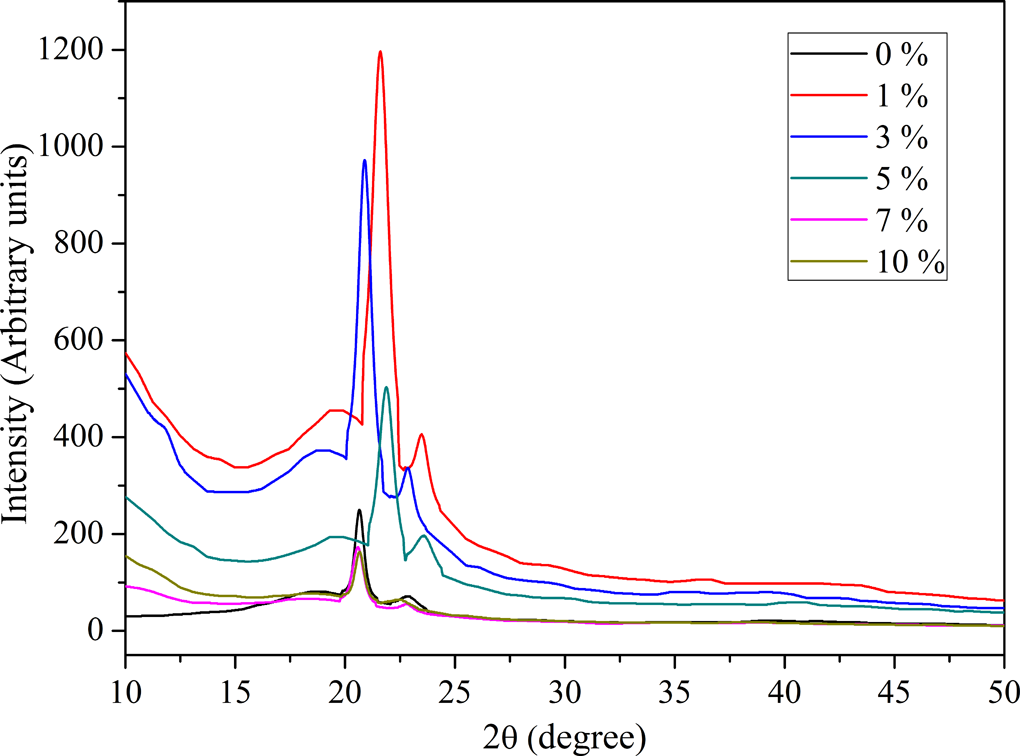
Crystalline peaks of the EVACO and its nanocomposites from XRD analysis. EVACO: poly(ethylene-co-vinyl acetate-co-carbon monoxide); XRD: X-ray diffraction.
Percentage crystallinity and d-spacing of each sample from XRD analysis.
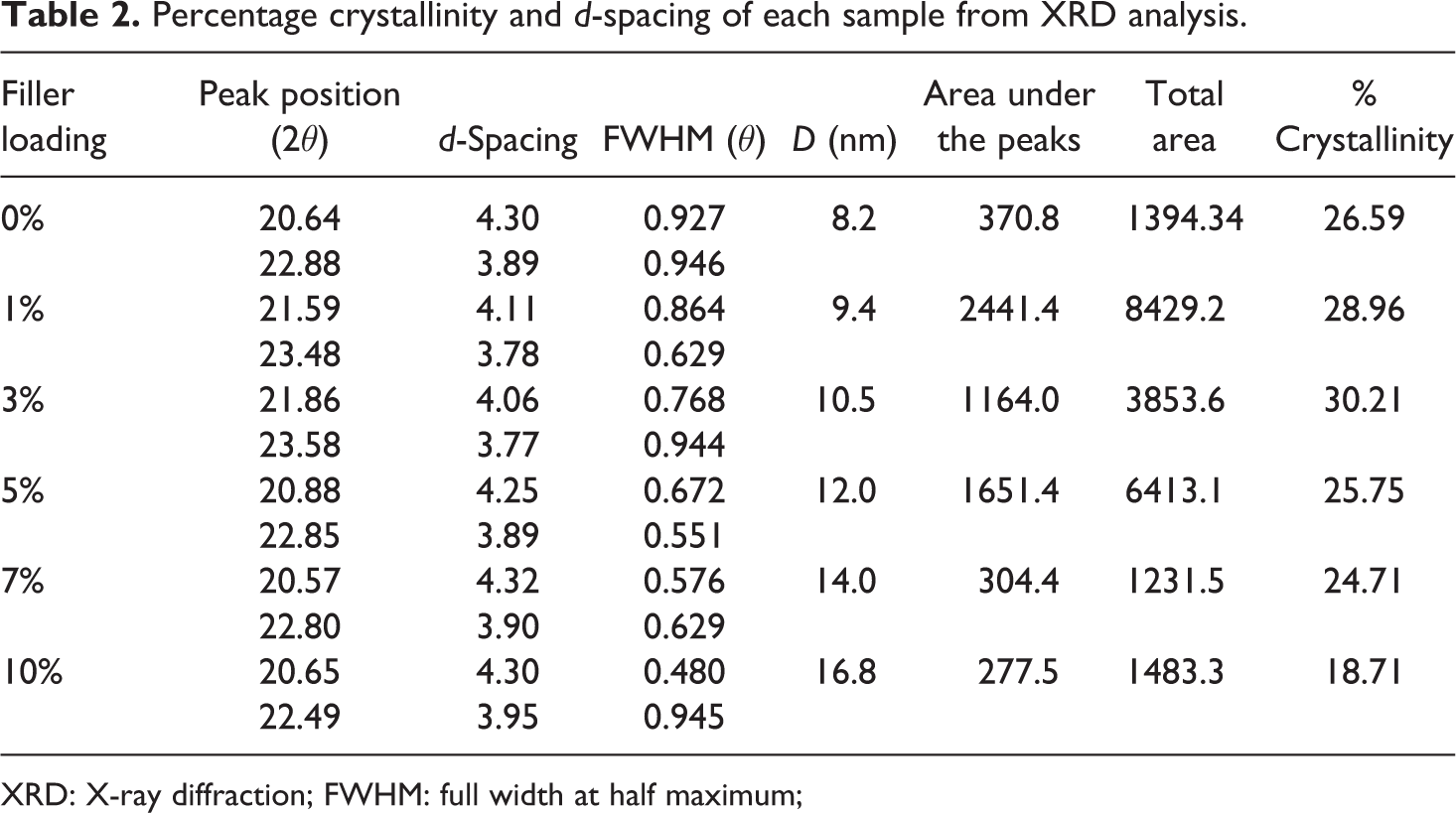
XRD: X-ray diffraction; FWHM: full width at half maximum;
Adding HNTs at a lower ratio brings the polymer chains close together by the weak pull exerted by the HNTs through hydrogen bonding, but addition of filler above a certain wt% reduces the crystallinity because the filler particles migrate to the crystalline phase of the polymer or impede the movement of crystallizable polymer chains near the nucleation sites. Also, the agglomerated nanoparticles at these filler loadings have a lower nucleating ability than the one with good dispersion. 15 The change in % crystallinity with filler ratio was also observed in DSC measurements. Therefore, the segmental motions of polymer chains are controlled by HNTs. The shifts in the crystalline peaks of the samples are due to the different orientation of the crystallites in the matrix. 33
The difference in % crystallinity in XRD and DSC analyses is due to the inevitable errors, which can happen during the calculation process. In XRD analysis, the error may be linked with the separation of crystalline and amorphous regions from the diffractogram; in DSC, it may be during the baseline correction, which is necessary for the evaluation of heat of fusion. 34
Thermal and flammability characteristics
TGA results
The characteristic weight loss of EVACO happens in two steps; the first step is due to the elimination of acetic acid through free radical and ionic β-elimination mechanisms 35 . During this process, C–O bond in the vinyl acetate is broken, which forms a free acetate radical, and the β-hydrogen atom from the ester is shifted to the acetate radical (ester pyrolysis) to form acetic acid and a complex polyene structure at approximately 350°C. The second step corresponds to the main chain degradation, which occurs at approximately 450°C. 9 Figure 8 shows the TGA and derivative thermogravimetric plots of pristine EVACO and its nanocomposites and two weight loss events are seen in all the curves. The char formation from EVACO is dependent on the polyene structure, 36 since the mechanism for the degradation behavior of EVA copolymer may also be extended to EVACO.
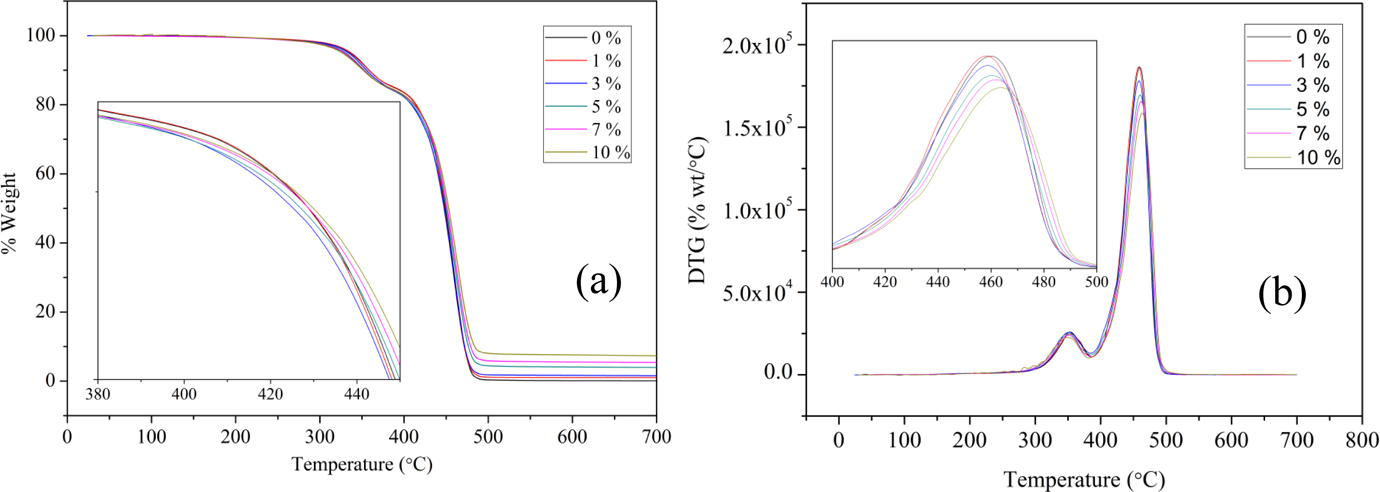
(a) TGA and (b) DTG curves of EVACO and its nanocomposites. TGA: thermogravimetric analysis; DTG: derivative thermogravimetric; EVACO: poly(ethylene-co-vinyl acetate-co-carbon monoxide).
In EVACO/HNT composites, the addition of HNTs in small quantities does not alter the degradation behavior of EVACO significantly; the onset of the second degradation step is slightly shifted toward higher temperature as the filler loading is increased (Figure 8(b)). An improvement in the thermal stability is observed for the composites with higher filler loadings (i.e. 5, 7, and 10%). The improvement in the thermal stability in the case of PNCs containing nanolayers such as clay can be attributed to the influence of the fillers on both the thermal and mass transport. 37 The thermal barrier will protect the polymer from the surrounding temperature and the mass barrier slows down the escape of volatile matter produced during the degradation. Since the aspect ratio of the HNTs is lower than that of layered fillers, their barrier properties also exhibit a similar trend. The thermal stability of the EVACO/HNT composites with higher filler loading, therefore, not only depends on the barrier properties but also on the entrapment of the free radicals produced during the degradation in the lumen of the HNTs that is rich in OH groups; this entrapment will dilute the degradation products. 20 The increasing amount of residue with filler loading after the final degradation step might improve the flammability properties of these nanocomposites.
Flammability properties
The vertical burning test and LOI analyses were performed for studying the flammability of the EVACO/HNT nanocomposites. During the vertical burning test, pure EVACO burnt easily and rapidly in air and the composites melted and produced flammable drops during their combustion, which ignited the cotton placed below the specimen. The addition of HNTs improves the flammability properties of these nanocomposites (Table 3). As the filler loading increases the flame resistance is enhanced, because of the evolution of water molecules present in the lumen of HNTs. These water molecules dilute the reactive species produced when the combustion progresses, which suppress the easy spreading of the flame in the composites.
Vertical UL-94 flammability properties and LOI of the samples.

LOI: limiting oxygen index; HNT: halloysite nanotube; NA: not applicable.
EVACO itself is self-extinguishing in nature. 38 As the HNT content is increased the amount of oxygen required for the combustion also increases. During LOI analysis, the samples are burnt from the top and the flammable polymer melts running toward the bottom and spread the flame to the lower parts of the samples easily. The residual char formation is increased with increasing filler loading, which may ultimately act as a thermal barrier when these composites are used for coatings.
Electrical properties
The effect of HNT nanotubes on the electrical resistivity of EVACO was studied by DC volume resistivity measurement. The resistivity of the composite decreases with the filler content exponentially, as shown in Figure 9, and the relationship between the filler content and conductivity is found to be:
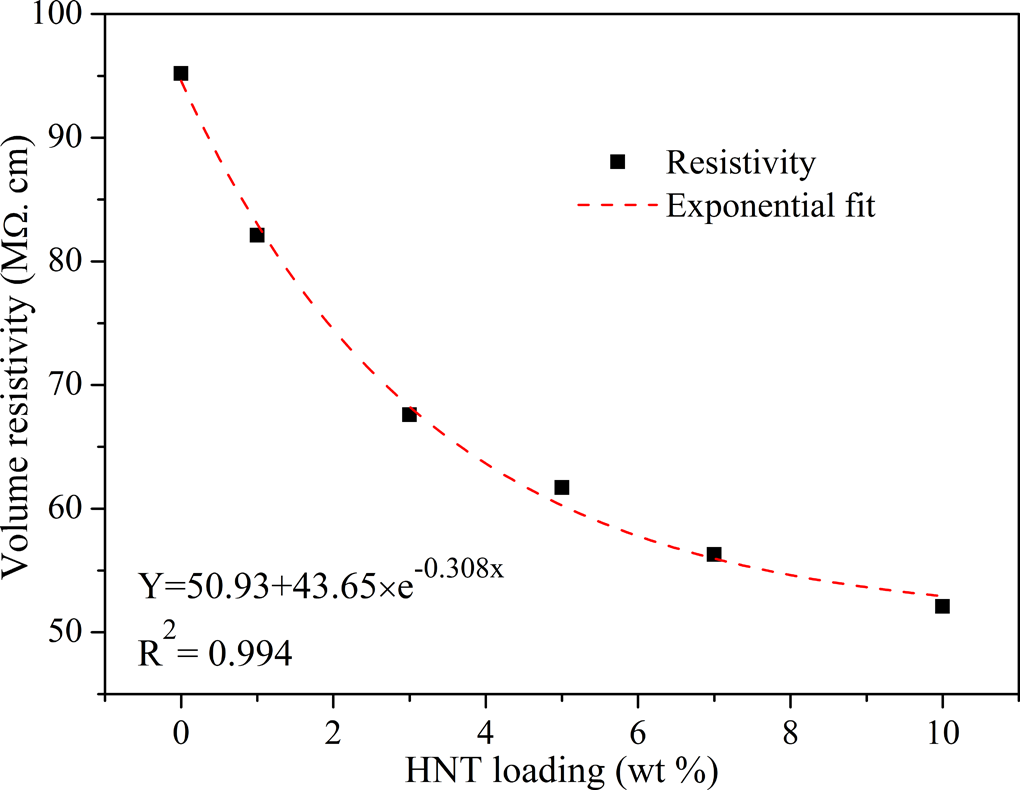
Change in DC electrical resistivity of the nanocomposites with HNT loading. DC: direct current; HNT: halloysite nanotube.

where y is the resistivity (MΩ cm) and x is the filler loading in percentage.
The decrease in resistivity of the EVACO/HNT composites with increasing filler content is due to the presence of cations and anions in HNTs. The movement of these cations and anions present in HNTs favors the electron transfer, which ultimately results in the reduction of resistivity of these composites. The vinyl acetate content in EVA helps in ionic movements, and it has been used as gel polymer electrolytes. 39 The presence of vinyl acetate in EVACO and its immediate analogy to EVA confirms that EVACO can also act as a medium for ionic movements. It is factual that as the HNT content is increased the resistivity of the composite decreases because of the availability of more ions for the charge transfer.
Mechanical properties
The tensile stress–strain plots of EVACO and its nanocomposites are shown in Figure 10 and the values of ultimate strength, elongation at failure, stress at 100% strain, and toughness (area under the stress–strain curve) are given in Table 4. The nanocomposite with 1% filler loading has the highest tensile strength because of the uniform dispersion of the HNTs in it. At 3% loading, the maximum crystallinity is observed, but the dispersion and distribution of the HNTs are poorer than that of the composite with 1% HNT loading. Therefore, the stress distribution also will not be uniform, ultimately leading to a slight reduction in the ultimate tensile strength. This difference is due to the presence of agglomerations and it is increasing with HNT loading. Agglomerations can act as points for stress concentration, from where the crack will be initiated. 40 A similar trend is observed for elongation at break and toughness values, which can be accounted by the same reasoning. A maximum elongation at break is observed for the composite loaded with 1% HNT, which is elucidated as the synergic effect of good filler dispersion and crystallinity at this composition.
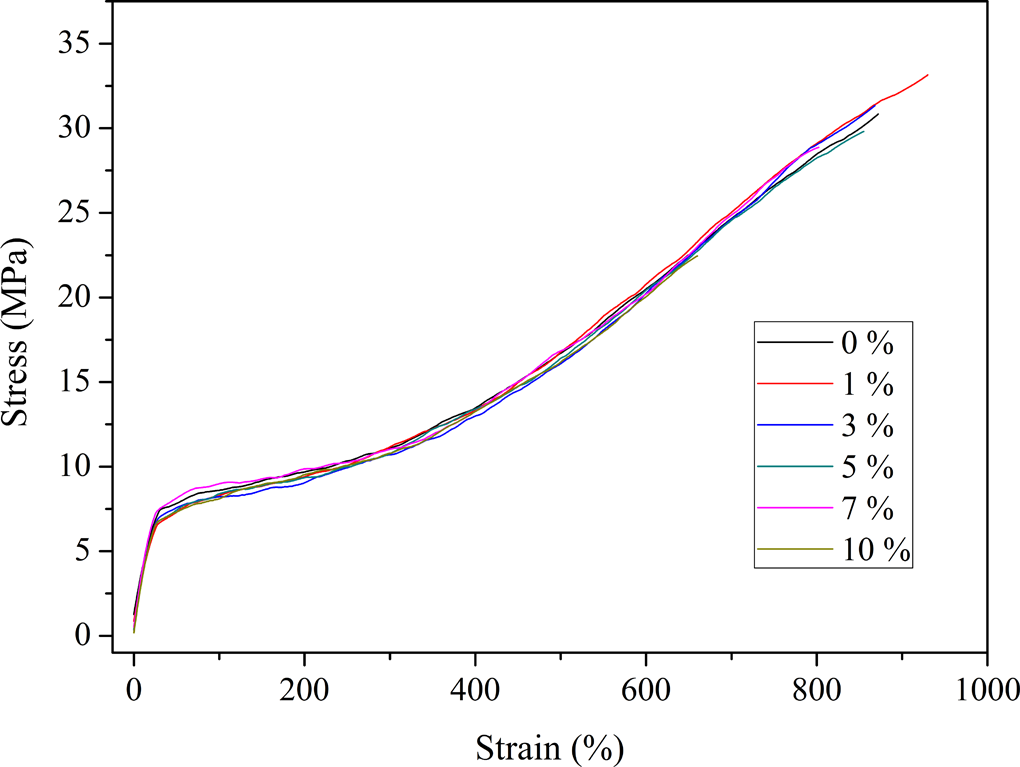
Stress versus strain curves of EVACO and EVACO/HNT nanocomposites. EVACO: poly(ethylene-co-vinyl acetate-co-carbon monoxide); HNT: halloysite nanotube.
Mechanical properties of virgin EVACO and the nanocomposites.
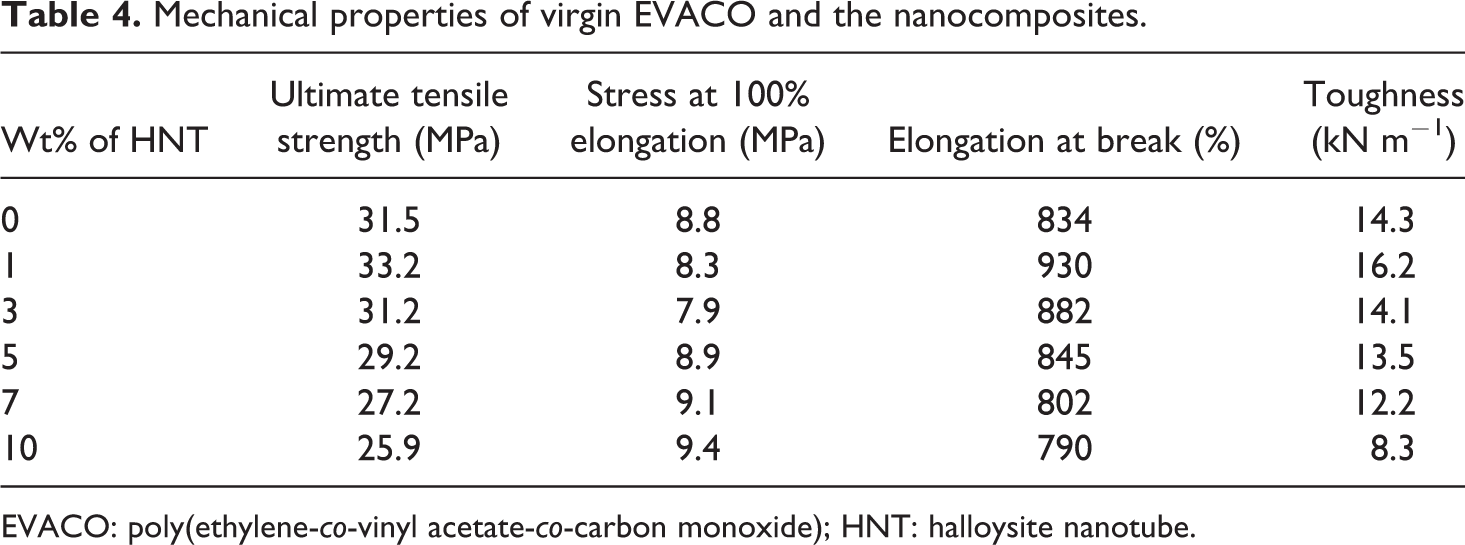
EVACO: poly(ethylene-co-vinyl acetate-co-carbon monoxide); HNT: halloysite nanotube.
Fractography
The fracture surfaces of the nanocomposites after tensile test were imaged using SEM and the micrographs are shown in Figure 11. As the filler loading is increased the smoothness of the fracture surfaces is lost. The fracture surface of pristine EVACO has a smooth surface and the continuous crack propagation trajectories confirm its ductile fracture. A similar trend is observed in the case of nanocomposites with filler loadings of 1 and 3%. But, as the filler loading is increased, the adhesion between the matrix and filler becomes poorer and stress concentrates at these regions in the matrix, where nanoparticles are agglomerated. Due to stress concentration, the amount of the stress-whitened regions increases as the filler loading is increased. The weak adhesion between the HNTs and the matrix could be due to the difference in polarities of the matrix and the filler. If the difference in polarity is high, the strength of the hydrogen bonding will also be less. Also, the chances of hydrogen bond formation are higher between the filler particles rather than the filler particles and the polymer chains.

SEM micrographs of tensile fracture surfaces of (a) neat EVACO and EVACO/HNT nanocomposites with (b) 1%, (c) 3%, (d) 5% (e) 7%, and (f) 10% of HNT. SEM: scanning electron microscopy; EVACO: poly(ethylene-co-vinyl acetate-co-carbon monoxide); HNT: halloysite nanotube.
At low filler loading, the polar groups in the polymer chains and the fillers particles are closely matching, and as the filler loading is increased, the mismatch between the number of surface polar groups on the filler particles and the polymer chains is increased tremendously, which has ultimately resulted in the agglomeration of particles at high filler loading. The polarity, in turn, reflects the surface energy or surface tension of the materials. In the case of polymers, the surface energy is in the order of tens whereas for fillers it is in hundreds. 41 Due to this mismatch in the polarity of matrix and fillers, the agglomerated nanoparticles can easily be dislodged from the polymer matrix. 38 Micropores are observed in the fracture surfaces of composites with higher filler loading, which were previously occupied by the agglomerated HNTs.
Ductile fracture occurs in all the nanocomposites as the crack propagation leaves a continuous track on the fracture surfaces. As filler loading is increased above 3%, the continuity of the crack propagation trajectories is lost; they become short or intermittent. Therefore, it can be said that as the filler loading is increased the failure of the samples tends to be brittle in nature, as shown in Figure 11. The fracture surface roughness of the samples increases as the filler loading is increased. In the case of pure EVACO, yielded fibrils are absent, but crazing is present in the contours of the crack propagation trajectories.
Conclusions
In summary, the crystallizability and mechanical properties of the EVACO/HNT nanocomposites are dependent on the dispersion and distribution of HNTs in the EVACO matrix. The reductions in mechanical and crystalline properties of the composites with the HNT loading are due to the presence of agglomerates and poor dispersion of HNTs in the matrix. The best mechanical properties and crystallinity are observed for the composite with a homogenous dispersion of HNTs, which are achieved at 1% HNT loading, as in the elemental maps. The crystallinity of EVACO is influenced by the presence of HNTs at low weight fraction, which reveals the ability of HNTs as a nucleating agent. The flammability of the composites is improved with an increase in HNT loading. The DC resistivity studies show that the electrical resistivity of the composites is decreasing exponentially with HNT loading. The expected reason behind the reduction in resistivity is the cations and anions present in the HNTs and the ability of the polymer to support ionic transport.
Footnotes
Acknowledgments
GG would like to thank the Department of Metallurgical and Materials Engineering, National Institute of Technology of Karnataka (NITK), Karnataka, India, for a research fellowship. The authors thank Ms U Rashmi and Ms Akshata G Patil for their kind assistance in scanning electron microscopy and X-ray diffraction, respectively. The authors are very grateful to Mr Hariharan, DuPont Dow elastomers, India, for the free supply of EVACO.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.