Abstract
The lightweight economical bipolar plates for proton exchange membrane fuel cells were fabricated by using vinyl ester resin as matrix material, graphite powder as reinforcement and conducting polymer as additive. Effects of various contents of graphite and its particle size on electrical conductivity, physical and mechanical properties of the composite were investigated. The optimum composition of composite plates was determined at 55% graphite, 40% resin and 5% poly(1,4-phenylene sulphide) based on measurements of physical, electrical and mechanical properties with various graphite contents. The plate molded with 2.93 mm thickness gives density of 1.81 g cm−3 and compressive strength of 73 MPa, which is in consonance with reported standard. Thermogravimetric analysis–differential thermal analysis–derivative thermogravimetry of the composite was carried out in air atmosphere to find out mode of degradation and basis of its stability. The study reveals that composite is less stable than resin and graphite, but much more stable than operational fuel cell which works at 80–100°C.
Keywords
Introduction
In the search for environmentally clean alternative energy resources to substitute fossil fuels, the application of hydrogen fuel cells as an energy carrier is seen as a prospective issue. 1 –3 Proton exchange membrane fuel cells (PEMFCs) offer exceptional potential for a clean, efficient and reliable power source due to the increasing awareness of environmental factors and limited energy resources. 4 –6 Bipolar plate by weight, volume and cost is one of the key multifunctional components in PEMFCs and constitutes over 80% of the weight, 30% of the total cost and almost all of the volume in a typical fuel cell stack (Figure 1). 7 –9 Hence, bipolar plates must exhibit high electrical conductivity, high gas impermeability, good mechanical performance, good corrosion resistance and low cost in order to meet the optimum balance of properties established by the US Department of Energy (DOE). 9 Bipolar plates have been traditionally fabricated from high-density graphite on account of its superior corrosion resistance, high electrical conductivity, chemical stability and availability. The main problems with graphite plates are their brittle and porous structure, and the cost associated with machining channels into the plates. The thickness of the graphite plates cannot be reduced resulting in bulkiness and heaviness. 10 As a result, recent studies have moved away from graphite in the direction of developing and optimizing more cost-effective and feasible alternative materials such as metals and composites. The metal-based bipolar plates typically suffer from corrosion in the fuel cell environment, which results in the poisoning of the electrode catalysts in PEMFC. 7 –10

Schematic diagram of fuel cell assembled by using locally manufactured bipolar plates.
An alternative to graphite is the use of polymer composite bipolar plates, which is promising, and has the advantage of low cost, ease in machining and good corrosion resistance. Polymer composite bipolar plates are generally comprised of a polymer resin and conductive fillers. The main conductive filler is graphite powder due to its high conductivity. The two different types of polymer resins that have been used to fabricate composite plates are thermoplastic and thermosetting. 11 –38 Thermoplastic resins are used less in bipolar plate fabrication than thermosetting resins for various reasons. Thermosets in comparison to thermoplastics usually have higher strength, creep resistance and lower toughness. 8,9 In high-temperature fuel cells, thermosets can maintain their dimensional and thermal stabilities better than thermoplastics. Moreover, at higher temperatures, the viscosity of thermosets is lower than that of thermoplastics and thus those can be loaded with higher level of conductive fillers. This fact aids in enhancing electrical conductivity, mechanical strength and density of thermoset based composites. The composites of epoxy resin with various forms of carbon, prepared by various methods, have exhibited high in-plane conductivity. 14,21,22 Nevertheless, their through-plane conductivity and mechanical strengths are both well below the DOE targets. Recent literature has indicated that phenolic resin reinforced with two or three conductive fillers demonstrates higher electrical and mechanical properties compared to the composites containing single filler. 11 –13,15,20,23,24,28 –30,34 Therefore, the synergistic effect, which results from combining various types of conductive fillers, on electrical conductivity and mechanical properties is a topic of growing interest to researchers. Bipolar plates fabricated from vinyl ester resin have exhibited better properties than the epoxy plates. In addition to being well known for excellent resistance to corrosion, vinyl esters are lightweight, strong, tough and commercially available. Nevertheless, vinyl ester plates are also reported to have bulk conductivity and tensile and flexural strengths well below the DOE targets. 31 –33,35 –37 Moreover, the curing of epoxies has typically exhibited a time frame of at least 8 h, although vinyl ester matrices have shown to cure in only 10 min. 35
The characteristics of composite plates are governed by the type and relative amounts of conductive filler. Graphite creates a conductive network albeit it contributes to brittleness of the plate. 10 Electrical conductivity is dependent on graphite size, distribution and the ability to form a homogeneous mixture with the polymer resin. Electrical properties normally improve with increasing amounts of graphite or carbon black, while mechanical properties decrease, requiring a compromise in composition and a trade-off between conductivity and mechanical strength. 8 –10 During the recent years this trade-off is obtained by previously mentioned synergistic effect. Various studies have demonstrated that thermosetting and thermoplastic resin plates consisting of more than one additive result in enhanced electrical and mechanical properties. 11,12,14,20,23 –25,28 –30,38
Bipolar plates generally require a fairly large proportion of graphite particles. Such high concentration of graphite particles will eventually cause wetting problem. 8 –10 Dhakate et al. 27 also proposed that polymers containing polar groups favour the conductive paths, thereby enhancing the electrical conductivity of the composite. Poly phenylene sulphide has excellent chemical resistance, flame resistance, heat deflection temperature and mechanical strength; it is the representative engineering plastic that is commonly used in automobile parts, electric/electronic parts, electric heating parts, chemical devices, etc. 39 The objective of the present work is two-fold. First is to investigate the synergistic effects of poly(1,4-phenylene sulphide) as an additive on the physical, mechanical, electrical and thermal properties of vinyl ester resin/graphite composites. Secondly, electrical conductivity, physical and mechanical properties of poly(1,4-phenylene sulphide) added vinyl ester resin/graphite composite plates are evaluated to optimize the graphite and resin contents and graphite particle size in order to achieve a low-cost bipolar plate with high electrical conductivity with adequate physical and mechanical strength. Thermal stability of composite has been ascertained by thermoanalytical technique. Optical and scanning electron microscopy (SEM) is utilized to examine the morphology and porosity configuration in the fabricated composites.
Experimental
Materials
The chemicals, vinyl ester resin (methacrylated epoxy difunctional), cobalt naphthalate (Co-Nap) and methyl ethyl ketone peroxide (MEKP) procured from standard source suppliers were of commercial grade and used without further purification. Graphite powder (density 1.88) used in this process was of reactor grade, having the particle size ranging from 38 µm to 106 µm, on being graded these by screen mesh method. Conducting polymer, poly(1,4-phenylene sulphide) was of analytical grade.
The fabrication of carbon–polymer composite bipolar plate
First vinyl ester resin and Co-Nap were mixed thoroughly. Then MEKP was poured into the resin with intimate blending for catalysis. 5% conducting polymer, poly(1,4-phenylene sulphide), was then added and mixed thoroughly and finally graphite powder was gradually added to the formulated resin and the mixture was shaken to form a stiff paste of friable consistency. The paste was introduced into the mold and pressed in a preheated (80°C) hot press by applying a force of 50 kN. On raising the temperature of mold to 100°C, the conditions were maintained for 15 min. The hardened plate was then removed from the mold while hot and air-cooled for half an hour. Following compositions were used by incorporating the graphite of various particle sizes. 60% graphite, 40% resin 55% graphite, 45% resin 65% graphite, 30% resin and 5% poly(1,4-phenylene sulphide) 60% graphite, 35% resin and 5% poly(1,4-phenylene sulphide) 55% graphite, 40% resin and 5% poly(1,4-phenylene sulphide) 50% graphite, 45% resin and 5% poly(1,4-phenylene sulphide) 45% graphite, 50% resin and 5% poly(1,4-phenylene sulphide) 40% graphite, 55% resin and 5% poly(1,4-phenylene sulphide)
Physico-chemical methods
Density measurement
The density of material was measured by water immersion technique (Archimede’s principle). The sample was weighed in air and value was named as
Mechanical properties
A universal testing machine was used to investigate the flexural property of the composite bipolar plate. The flexural strength and flexural modulus test was performed according to the procedure by American Society for Testing and Materials E290-1997a with a specimen bar 55 mm in length, 10 mm in width and 3 mm in thickness. The supporting span is 55 mm and the rate of the cross head was 0.5 mm min−1. The compression testing of the composite bipolar plate at room temperature was carried out by reported standard method of American National Standard, American National Standards Institute (ANSI)/ASTM E. 40
Porosity determination
The ASTM C20 test procedure was used to measure the porosity of composite plate. The specimen was weighed in air and the value was recorded as 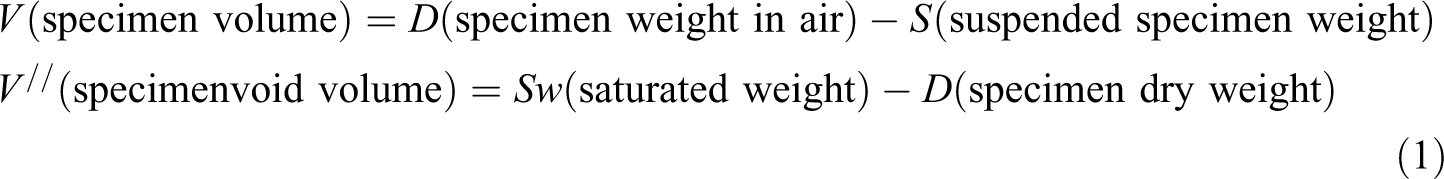
Structural characterization
The morphology of composites was examined using a SEM (LEO 440, M/s LEO Electron Microscopy, UK). The morphology of the composite bipolar plate was also examined using an optical microscope, LEICA MPS 30 (Germany). The specimen was shaped into required piece and after grinding and polishing, directly examined using the microscope to observe the surface morphology. Fourier transforms infrared (FTIR) spectra of starting materials and condensed products were recorded on a Shimadzu Prestige 21 spectrometer (Japan).
Conductivity measurement using four-probe method
Conductivity is directly calculated using four-probe method in which contacts were placed at four points on a specimen, that is, current and potential probes were mounted on a special holder. The constant current was allowed to pass through the sample using digital multi-meter Kyoritsu Model 1009 (Japan) The potential drop was measured across the probes using a 195A digital multimeter under ambient conditions where probe distance signifies the sample length. The potential drop observed was the average value obtained for both directions of current flow. Conductivity was calculated by the application 41 of following equation:
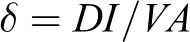
where
Thermal analysis
Thermoanalytical (thermogravimetric analysis–differential thermal analysis–derivative thermogravimetry (TGA-DTA-DTG)) investigation of solid samples was carried out under atmospheric air using NETZSCH Simultaneous Thermal Analyzer STA 409, Germany with a temperature-programmed furnace. Samples were contained in an aluminum crucible Al 203 (8 mm diameter × 10 mm depth) with central base recess. The crucible was then adjusted on palladium–ruthenium crucible support platform, which gave a proportional signal to the recorder and computer interface to plot the weight loss of sample against the temperature. The heating rate employed was 10°C min−1. All the experiments were performed in the temperature range of ambient to 1100°C. The evolved products were identified on weight loss curve coupled with the DTA result basis.
Results and discussion
A composite bipolar plate, composed of vinyl ester resin as a matrix material and graphite as reinforcement, has been successfully prepared by compression molding technique. Curing of vinyl ester resin has been done by free radical polymerization, while organic catalyst MEKP is used as a curing agent. To further speed up the curing, cobalt naphtenate is added in addition to the catalyst. Five percent of poly(1,4-phenylene sulphide) is introduced as additive to increase the conductivity. Then compression molding of slurry produced is done to form a composite sheet.
Structural and physical properties
FTIR spectroscopy is useful to study the surface functional groups of organic materials. Figure 2 shows the FTIR spectra of fabricated carbon–polymer composite. Figure 2 indicates that it is somewhat difficult to identify a particular material or group in the final composite as the spectral bands from different components overlap. The peaks at 700, 830 and 891 cm−1 represent C–H out of plane bending vibrations. The peaks, positioned at 1250 and 1300 cm−1, are due to C–O stretching while peaks at 1400 and 1456 cm−1 are due to symmetric and asymmetric bending vibrations of the methyl group. Moreover, the peaks at 1510, 1550 and 1640 cm−1 are due to ring stretching vibrations of aromatic nuclei and the band appearing at 1720 cm−1 is due to ester carbonyl stretching. The bands at 910 and 945 cm−1 may be ascribed to vinyl ester double bonds. The band around 2400 cm−1 can be attributed to the O–C–O antisymmetrical stretching vibration of carbon dioxide (CO2) in air. The inset shows a large range between 3000 and 4000 cm−1 which is derived from O–H stretching vibration of carboxyl and hydroxyl groups. 37,42
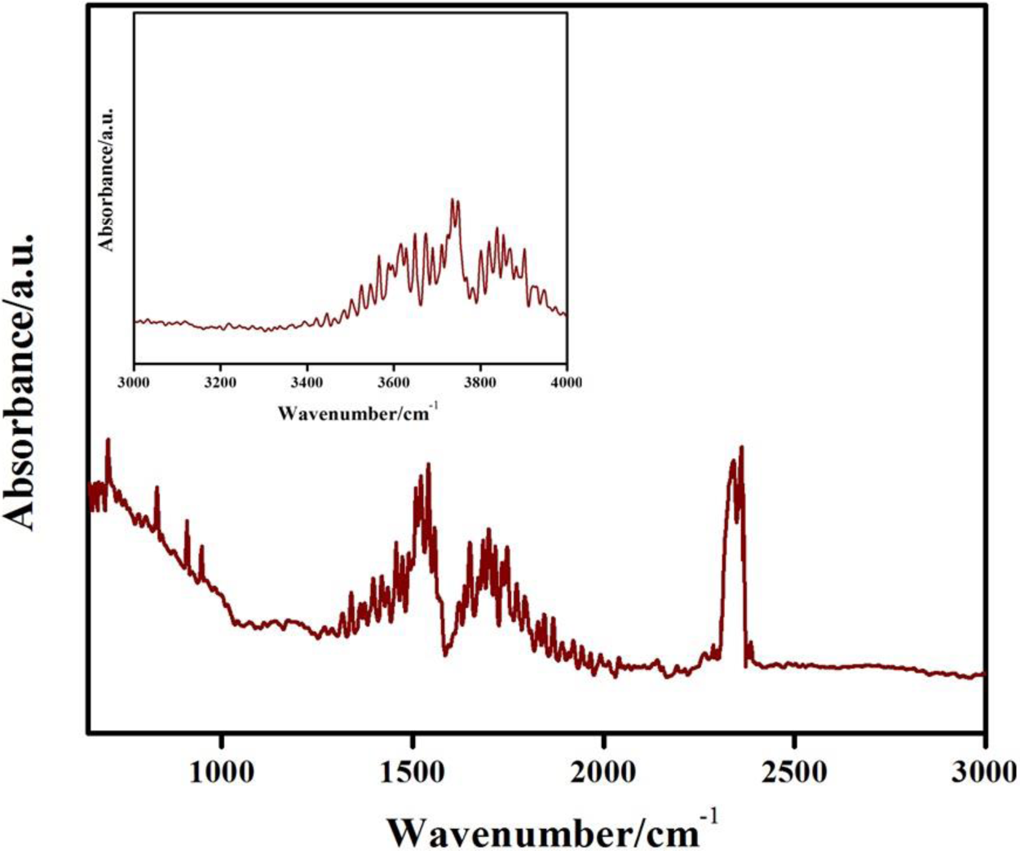
FTIR analysis of fabricated carbon-polymer composite. The inset shows the corresponding –OH stretching region.
The density of the composite bipolar plate increases with an increase in graphite contents. The density of the composite increases from 1.65 to 2.28 as the graphite contents are increased from 45% to 65% as shown in Figure 3. Since the density of graphite (1.88 g cm−3) is much greater than that of vinyl ester resin (1.03 g cm−3), therefore, when the graphite concentration is increased, the overall density of composite is increased significantly. It implies that the desired density of the present plate for a composite is found to be 1.81 g cm−3 with graphite percentage of 55% and particle size of 53 µm, which is lower than that of a pure-graphite bipolar plate viz. 1.88 g cm−3. Figure 4 depicts that the density of composite bipolar plate decreases with decrease in graphite particle size. It increases from 1.45 g cm−3 to 1.85 g cm−3 as the particle size is increased from 45 µm to 90 µm. Figure 4 also shows that the porosity of the composite plate increases with decrease in the graphite particle size. It increases from 3.9% to 22.42% as the particle size is decreased from 106 µm to 45 µm. As small-sized graphite particles contain more surface area than that of larger one, therefore, the graphite powder with a smaller size will have a stronger wetting ability than that of the larger graphite powder particles. Hence, the higher concentration of graphite with a smaller particle size can lead to insufficient wetting of graphite powder by vinyl ester resin which may result in an increased porosity. Moreover, insufficient resin may induce holes in the composite bipolar plate.
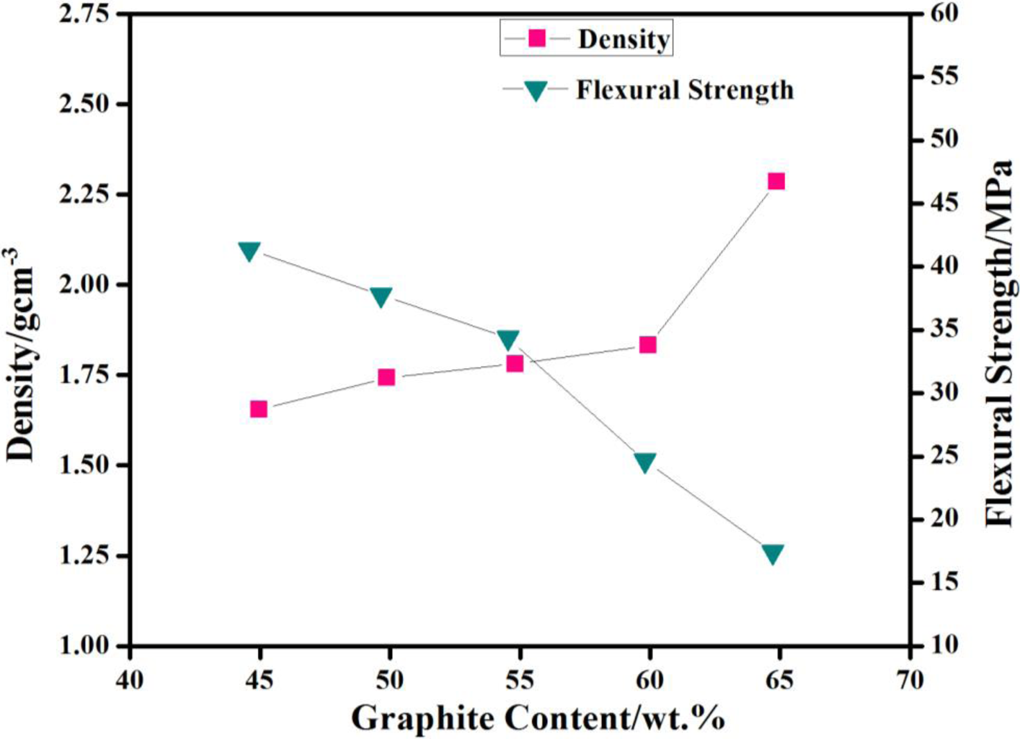
Density and flexural strength of composite bipolar plate with different graphite contents.
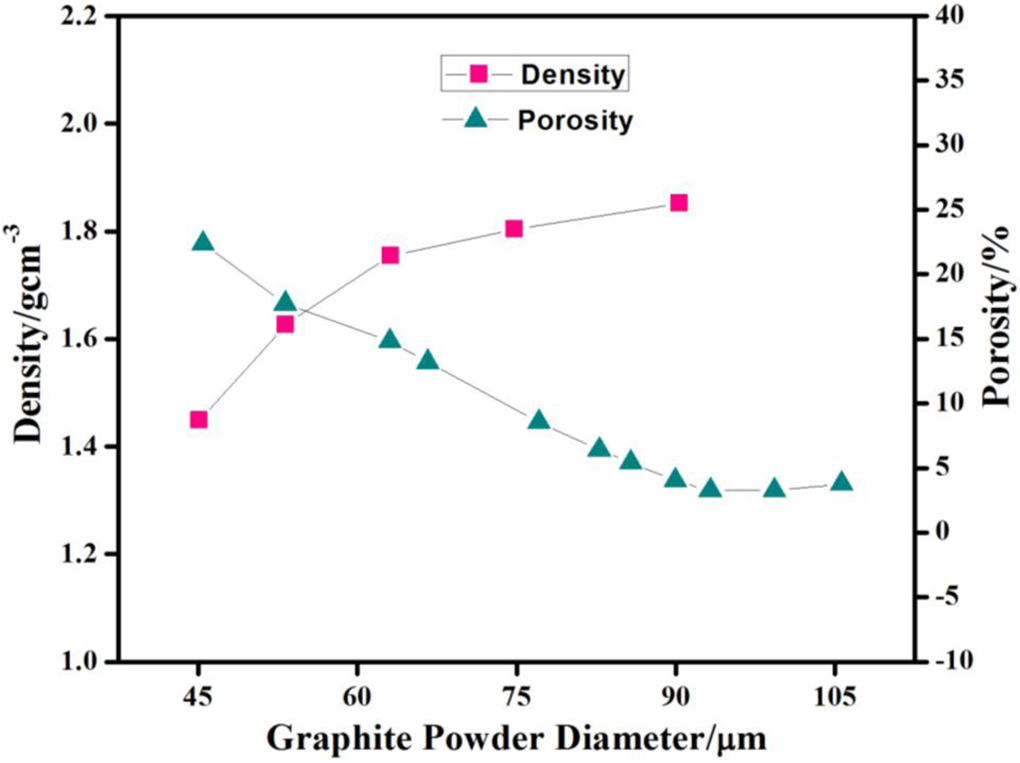
Density and porosity of composite bipolar plate with different graphite powder sizes.
The optical micrograph of composite bipolar plate is shown in Figure 5(a). The presence of voids is obvious in the composite, which is probably due to the evaporation of ingredients during the partial degradation of polymer and unwetted graphite powder. In order to provide further understanding, the samples with various compositions are studied under a SEM so as to observe their morphology. Figure 5(b) indicates that graphite powder is poorly dispersed in vinyl ester resin and graphite particles remain in the form of aggregates. There are many conducting graphite channels and networks in the composite. This phenomenon is attributed to graphite’s incompatibility with vinyl ester. It is noticed that a higher concentration of graphite is needed to create a conductive path throughout the entire composite. 43 With the increase of resin contents, the graphite particles do not make contact with the nearest neighbours due to partial wetting by the resin. Dawson et al. 44 have proposed that the conductivity in graphite-filled polymers is regulated by inter-particle electron transfer at low filler loadings and by intra-particle electron transfer at high filler loadings. From Figure 5(c) and (d), it is visible that the graphite particles are aligned parallel to the plane of the surface which is conducive in achieving high in-plane electrical conductivity. 24 It is also evident in Figure 5(d) that the resin distributes more uniformly and coats the graphite particles effectively when the resin content is higher. Moreover, the porosity may arise from the evaporation of polymer during partial degradation and cross-linking of polymer during the hot pressing process. It has already been stated that insufficient wetting of graphite powder by vinyl ester resin may result in an increased porosity. Therefore, the number and size of pores decrease with an increase in resin contents (Figure 5(c) and (d)).

(a) Optical micrograph of 55% graphite, 40% resin and 5% poly(1,4-phenylene sulphide) composite, SEM images of (b) 65% graphite, 30% resin and 5% poly(1,4-phenylene sulphide) composite, (c) 55% graphite, 40% resin and 5% poly(1,4-phenylene sulphide) composite and (d) 45% graphite, 50% resin and 5% poly(1,4-phenylene sulphide) composite.
Electrical properties
The electrical conductivity of composite bipolar plate increases with increasing the graphite contents (not shown here). Electrical conductivity increases from 40 S cm−1 to 55 S cm−1 as the graphite contents are increased from 50 wt% to 55 wt%. It has also been observed that there is a considerable increase in electrical conductivity from 55 S cm−1 to 90 S cm−1 as the graphite contents are increased from 55 wt% to 60 wt%. This might be due to the fact that increase in the graphite concentration may lead to a strong possibility of formation of strong electrical conductive matrix. This strong electrical conductive matrix results an increase in the electrical conductivity of composite bipolar plate. Figure 6 demonstrates that the electrical conductivity of the composite bipolar plate increases with increase in graphite size. There is a considerable increase in the electrical conductivity of plate as the graphite particle size is increased from 45 µm to 90 µm. Actually, compaction of powder enhances the conduction of electrons through graphite powder. Aggregation of the powder decreases as the particle size of the powder is increased. Consequently, the electrical conductivity of the composite bipolar plate increases as the particle size of the graphite powder increases. Furthermore, it is well understood that the polymer matrix is not just the network supporting the filler graphite rather it plays an essential role in many other aspects like providing the correct conditions for optimum performance of the end product. It favours particle interconnectivity (percolation). It also provides electrical conductivity through a tunnel type mechanism in the event of deficient percolation. Moreover, it tolerates the incorporation of large amount of additives facilitating both the conducting mechanisms and dimensional stability of the resulting composite. 10
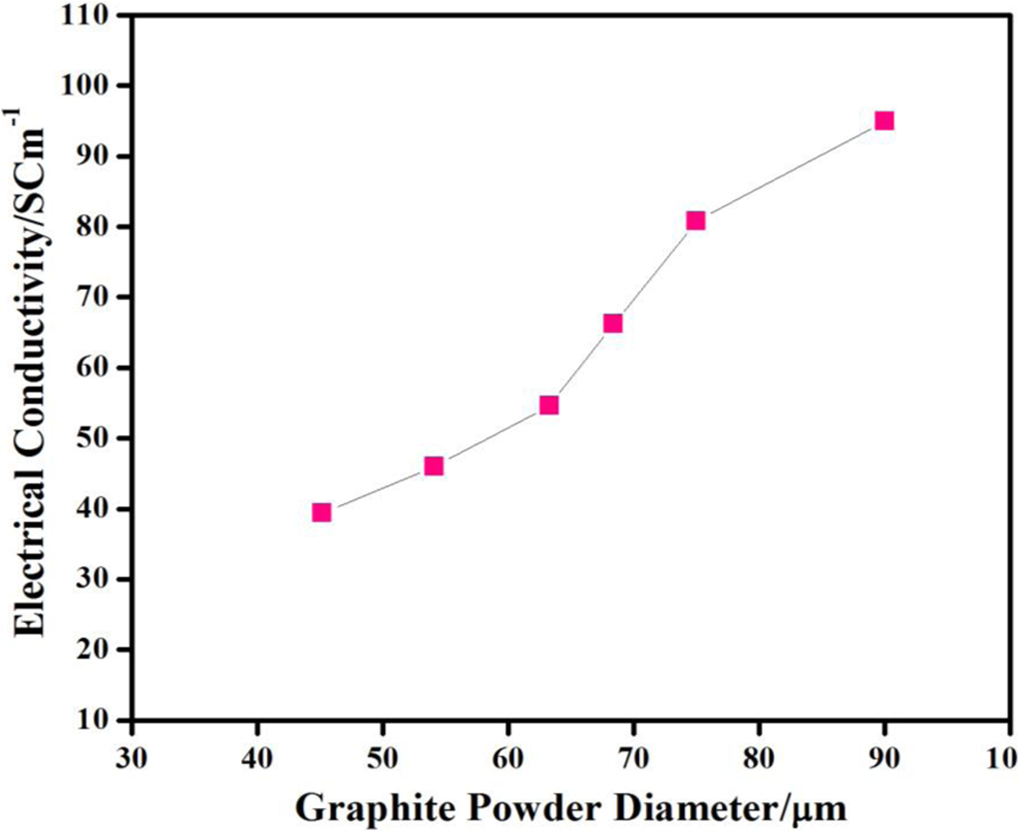
Electrical conductivity of composite bipolar plate with different graphite powder sizes.
Flexural and compressive properties
Figure 3 indicates that the flexural strength of the composite bipolar plate decreases from 41.2 MPa to 17.35 MPa, as the graphite content is increased from 45 wt% to 65 wt%. It has also been observed that flexural modulus decreases with increase in graphite contents. Figure 7 shows that flexural strength and flexural modulus of composite bipolar plate decreases with decrease in graphite particle size. Flexural strength decreases from 30.85 MPa to 17.35 MPa, as the particle size is decreased from 125 µm to 45 µm. Flexural modulus decreases from 6.22 to 4.35 as the particle size decreases from 125 µm to 45 µm. When the graphite contents are increased, the concentration of the graphite becomes much greater than that of resin. That results in a weak adhesion between graphite and resin. Consequently, flexural strength and compressive strength of composite bipolar plate decrease as the graphite concentration is increased. Ideally the composite plate should have an adequate mechanical strength to be used in fuel cell stacks where these will have to withstand constant compressive load. The composite should have the compressive strength greater than 50 MPa (7250 psi) with test standard ASTM D695, which shows good results during stacking. 8 –10 The plate molded in the present work with 2.93 mm thickness, having composition of 55% graphite, 40% polymer and 5% conducting polymer, exhibits a compressive strength of 73 MPa (not shown here), which is in consonance with the reported standard. Furthermore, it has already been shown that the decrease in graphite size results in the density of composite bipolar plate. It implies that the decrease in graphite size results an increase in the porosity in the composite bipolar plate that ultimately leads to decrease in the flexural property and compressive strength of the bipolar plate.
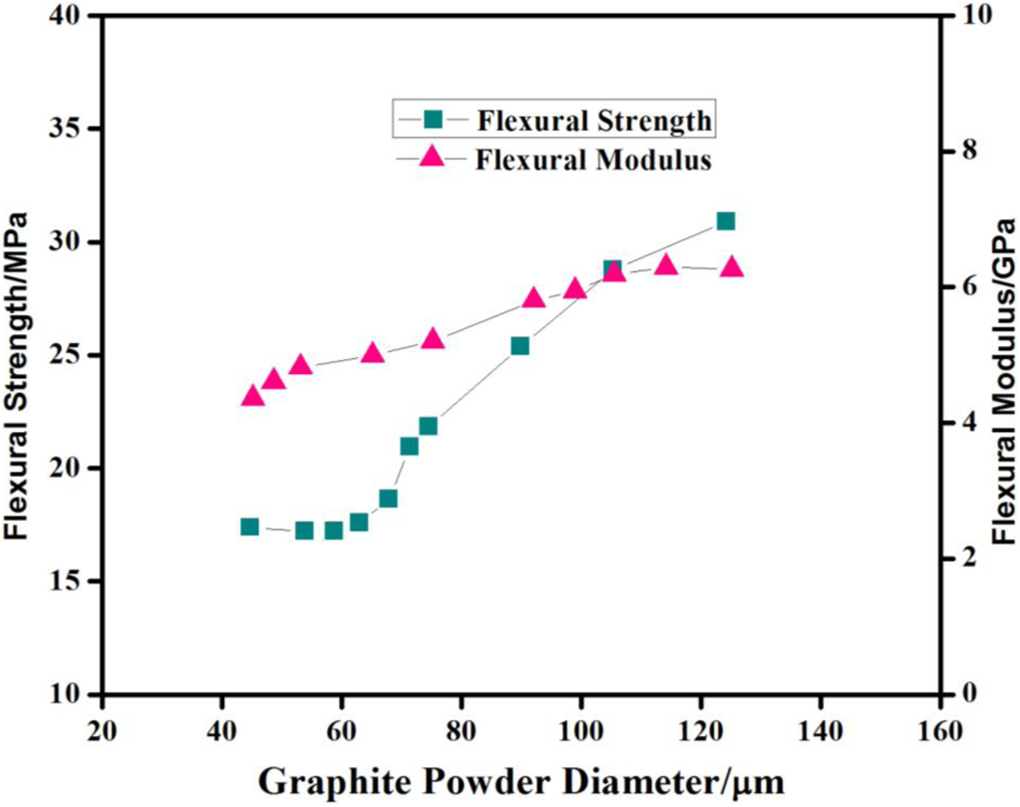
Flexural strength and modulus of a composite bipolar plate with different graphite powder sizes.
Thermal properties
Table 1 presents the observations made from TG, DTG and DTA curves in Figure 8. One-step degradation for pure graphite in air takes place at 700–1100µC. At around 700°C, graphite exhibits weight loss due to oxidation. Up to 1100°C, 42% weight loss is observed. The corresponding endothermic peak in the DTA curve is at 829°C. The vinyl ester blend exhibits two-step degradation. However, rapid decomposition is observed in the temperature range of 185–440°C and approximately 86% of the sample is lost during this major decomposition step. This step is represented by an endothermic and an exothermic DTA peak at 332°C and 360°C, respectively. The probable composition of expelled moiety in this step may be hydrogen (H2), carbon monooxide, methane (CH4), monomer and ethane. Regardless of the polymer composition it is reported that at temperatures of around 300°C cured vinyl ester resins undergo spontaneous decomposition. This is a characteristic of vinyl polymers which are caused by their depolymerization to form monomeric species. 45 Following the completion of first stage around 440°C, the intermediate compound of cobalt exhibits some stability. 46 The second stage starts at 490°C and terminates at 600°C showing slow rate of disintegration and probably losing small molecules like Co, CO2, H2, CH4, O, –CH2, etc. This is ascribed to the advanced fragmentation of macromolecules formed in the first stage, secondary reactions of dehydrogenation, thermal cracking and disproponation and gasification process. 47,48 This decomposition is accompanied by DTA peak at 551°C and DTG minimum at 590°C. In the third stage, the residue may be cobalt oxide which has high thermal stability. 49 The ceramic materials have a significant effect on the physical and thermal properties of the fabricated polymer composites. Figure 8 shows that the graphite particle loading reduces the thermal stability of vinyl ester resin at lower temperatures except much less weight reduction arising from the less mobility of monomers. It is interesting to note that the thermal behaviour of composite indicates the physical combination of the components behaving almost independently of each other. This composite is thermally less stable than its components if the temperature of first weight loss is taken into account. Nevertheless, higher stability has been observed at higher temperatures in the high-temperature decomposition range. This phenomenon is similar to that observed in the vinyl ester resin composites reinforced with alumina, copper oxide and zinc oxide nanoparticles. 45,50,51 The thermal analysis of carbon–polymer composite shows two-step pyrolysis (Figure 8). The composite is thermally stable up to 160°C. During the first stage (160–382°C), the degradation of vinyl ester resin occurs and monomers are evolved accompanying the elimination of small molecules like H2, CO, water, CH3, C2H6 and ethylene. The conducting polymer ((poly(1,4-phenylene sulfide)) may also degrade towards the completion of first stage emitting benzene and hydrogen sulphide with the weight loss of 20%. 51 The intermediate, formed at the termination of the first stage, manifests little stability and starts decomposing around 410°C. It is clearly Co compound whose splitting gives off small molecules such as CO, CO2, oxygen, CH4, ethylene and H2. 46 This is accompanied by an endothermic DTA peak at 379°C and a DTG minimum at 365°C. Around 700°C, the disintegration pattern is that of graphite. The weight loss (38%) is very sharp till 1100°C due to the production of CO2 showing corresponding endothermic DTA peaks at 542°C and 700°C. The process goes on even at the completion of present run.
Thermoanalytical results of pure graphite, VER blend and composite material.
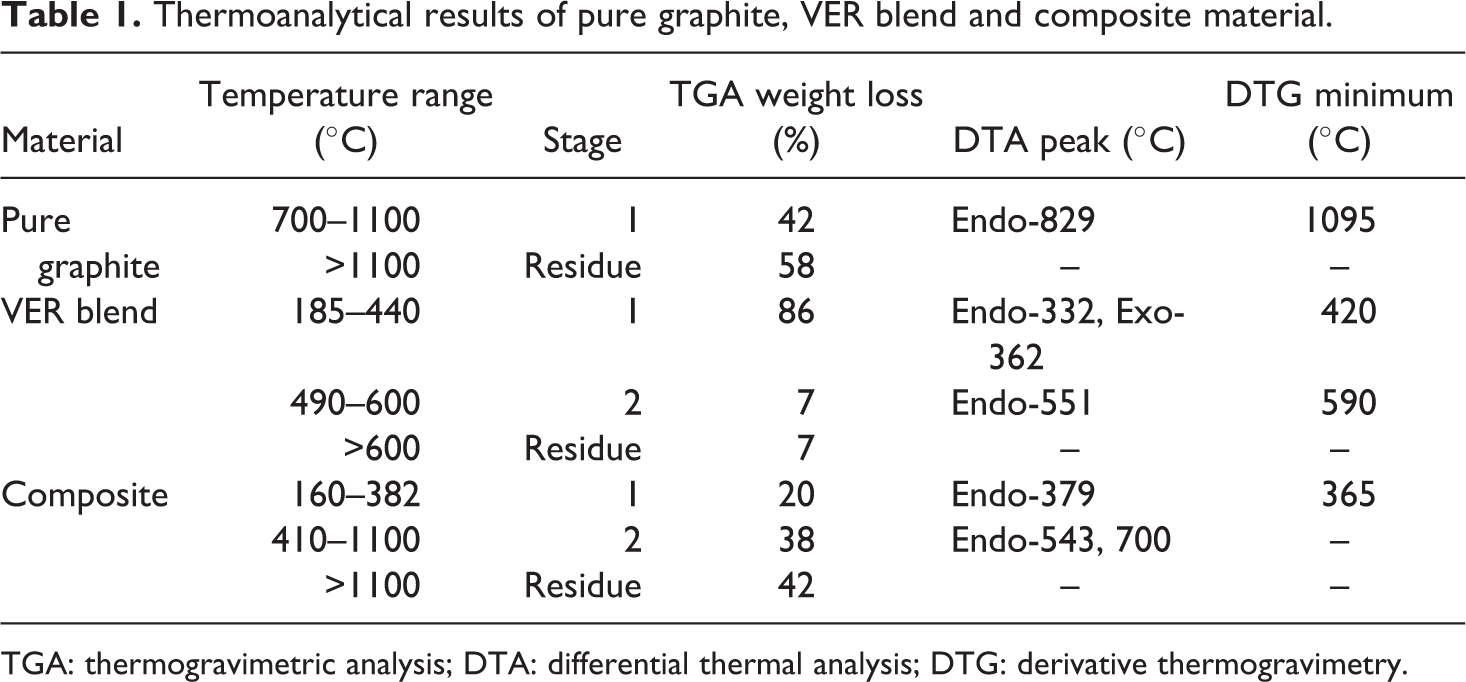
TGA: thermogravimetric analysis; DTA: differential thermal analysis; DTG: derivative thermogravimetry.
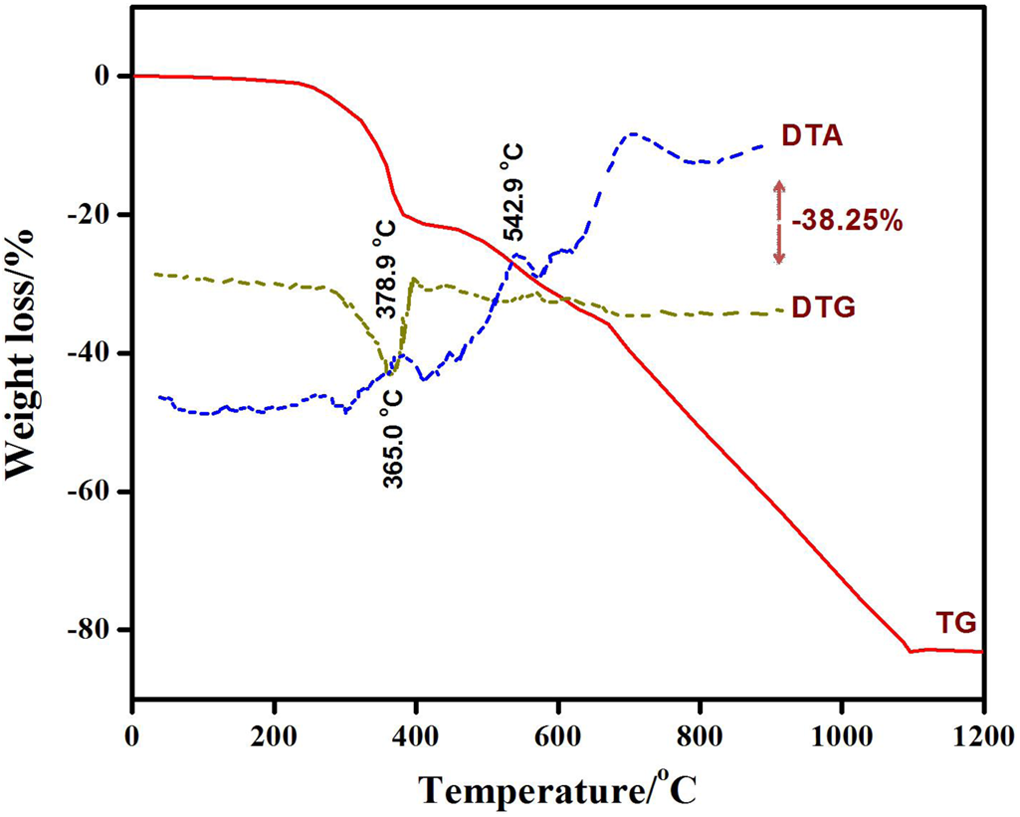
Thermoanalytical curves of fabricated carbon–polymer composites.
Conclusions
A carbon–polymer composite sheet consisting of commercially available graphite, vinyl ester resin (VER), MEKP, Co-Nap and poly(1,4-phenylene sulphide), has been successfully prepared by the compression molding technique. The effects of graphite contents, graphite particle size and resin contents on the electrical, physical and mechanical properties of poly(1,4-phenylene sulphide) added vinyl ester/graphite composite are investigated. The results indicate that poly(1,4-phenylene sulphide) is effective as the secondary additive in order to enhance the electrical conductivity and flexural strength of the composite plates. It is deduced that the graphite particle size has a significant influence on the physical, electrical and mechanical performances of composite plates. A larger graphite particle size is advantageous for increasing the density, electrical conductivity and flexural strength. The conflict between the electrical property and the mechanical property is eliminated by optimizing the graphite and resin contents. The density of composite increases from 1.65 g cm−3 to 2.28 g cm−3 as the graphite contents are increased from 45% to 90%. It is also observed that there is a considerable increase in electrical conductivity from 55 S cm−1 to 90 S cm−1 as the graphite contents are increased from 55 wt% to 60 wt%. The flexural strength of the composite bipolar plate decreases from 41.2 MPa to 17.35 MPa, as the graphite contents are increased from 45 wt% to 65 wt%. The morphological analysis of the composite bipolar plate has showed good correlation with the effects of graphite and resin contents on electrical conductivity and porosity of the bipolar plate. The first thermal study of such composites reveal that composite is less stable than VER and pure graphite, but much more stable than the operational fuel cell which works at 80–100°C. The detailed comparison of the developed vinyl ester resin/graphite composite bipolar plate, with those reported by various researchers, shows that the composite is a good candidate for bipolar plates in PEMFC meeting the electrical and mechanical targets of DOE simultaneously.
Footnotes
Acknowledgements
The authors would like to express their gratitude to Chemistry Division, Directorate of Science, Pakistan Institute of Nuclear Science and Technology (PINSTECH), Islamabad, Pakistan for providing the opportunity to undertake this research work. Thanks are also due to Mr Fahad Ali, SE, Material Division, Pakistan Institute of Engineering and Applied Sciences (PIEAS), Islamabad, for his assistance in evaluating various physical and mechanical parameters.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
