Abstract
A macroinitiator was prepared by copolymerization of styrene (St) with 7-methacryloyloxy-4-chloromethylcoumarone (MAOCMC). Grafting studies of coumarone with methyl methylacrylate were carried out in the presence of the macroinitiator poly(7-methacryloyloxy-4-chloromethylcoumarone-co-styrene) and with the catalyst of copper(I) bromide/2,2′-bipyridyne at 110°C. The activation energy valuations of graft copolymers acquired by Coats–Redfern, Tang and Flynn–Wall–Ozawa methods were designated to be 212.69, 214.44 and 223.57 kJ mol−1, respectively. For the outcomes were compared with these valuation differential methods and discrepant integral were used. In terms of experiential outcomes, the reaction mechanism was a dimensional diffusion (Dn) deceleration type in the transformation range worked.
Keywords
Introduction
(Meth)Acrylic copolymers have reached foremost significance in a variety of ways in industrial practices. 1 – 3 For instance, copolymers comprising altered coumarone are used in the manufacture of superb fluorescent properties with high photoluminescence (PL) quantum efficiency, superb photostableness and ample spectrum range. 4,5
They are widely used in the areas of polymer science, medicine, laser dyes and biology.
6
Furthermore, copolymerization is beneficial and a significant way to advance novice materials. Copolymerization modifies both intermolecular and the intramolecular powers executed among polymer fragments. Hence, certain properties, such as, the glass transition temperature and the procedural decomposition temperatures with respect to thermal degradation may diversify within ample limits.
7
Because of the compatibilization ingredients in polymer blends, thermoplastic elastomers, surfactants, adhesives and adjuvants in high-impact materials, graft copolymers have been used in industrial practices and widely worked.
8
Grafting reactions have been used along with atom transfer radical polymerization to generate diversified graft copolymers in which the polymer involves a pendent group with atom transfer radical polymerization (ATRP) initiator functionality, like benzylic halides, α-halocarbonyls and aromatic sulphonyl chlorides.
9
Activated carbon–halogen (C-X) bond with many of the organic compounds can be used qua ATRP initiators. Allyl,
10
adenosine and uridine,
11
thiol,
12
acid
13
and hydroxyl
14
groups have been accomplishedly introduced into polymer ends by cautiously selecting the matching initiators. Some novice functional initiators were also offered by discrepant research groups. Bipyrridine,
15
oxazoline,
16
pyrrole,
17
norbornenyl,
18
aldehyde,
19
sole-walled carbon nanotubes,
20
pyrene
21
and
In this article, a photosensitive ATRP initiator and graft copolymers were prepared using ATRP method. The evident activation energy (
Experiential section
Materials
Before use, styrene (St) and methyl methacrylate (MMA) were distilled under vacuum. 2,2′-bipyridyne (bpy), copper(I) bromide (CuBr), 2,2′-azobisisobutyronitrile, tetrahydrofuran (THF), dichloromethane and ethanol were used without further purification.
Instrumentation
Infrared spectra were registered on a Perkin–Elmer Spectrum One Fourier transform infrared spectroscopy (FTIR; Waltham, Massachusetts, USA) spectrometer. Proton nuclear magnetic resonance ( 1 H NMR) spectra was acquired on a 400 MHz Bruker AVIII 400 machines (Billerica, Massachusetts, USA), using tetramethylsilane as an interior standard and deuterated chloroform as the solvent. Ultraviolet–visible (UV–vis) spectra were registered using a Shimadzu spectrophotometer (Japan). Thermal stability studies were carried out on a Shimadzu thermal gravimetric analyzer (TGA)-50 thermobalance under nitrogen (N2) flow with a heating rate of 10°C min−1.
Synthesis of macroinitiator poly(7-methacryloyloxy-4-chloromethylcoumarone-co-styrene) (P(MAOCMC-co-St))
Synthesis of poly(7-methacryloyloxy-4-chloromethylcoumarone-co-styrene) (P(MAOCMC-
Synthesis of graft copolymers by ATRP
Atom transfer radical graft copolymerizations were carried out in a 25-mL flask equipped with a condenser. The necessary amount of the macroinitiator P(MAOCMC-co-St) was dissolved in dichloromethane, and the following were then added to the solution: the suitable monomer (methyl methacrylate), macroinitiator, the catalyst 2,2′-bipyridyl and CuBr at a mole ratio of 100:1:2:1, respectively. The solution contained similar molar amounts of group MAOCMC and CuBr. Argon gas was moved through the solution for 15 min. The polymerization flask was permitted to react at 110°C for the required time, after which, the graft copolymer was precipitated into slender acidic ethanol. The graft copolymers, poly((7-methacryloyloxy-4-chloromethylcoumarone-co-styrene)-g-(methylmethacrylate)) P[(MAOCMC-co-St)-
Thermal decomposition kinetics
The practice of dynamic TGA methods keeps huge procurement as a tool for untying the mechanisms of chemical and physical process that happen throughout polymer degradation. Isothermal decomposition reactions of solid state can be denoted as follows:

where 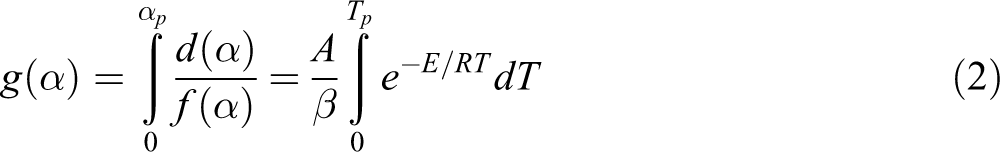
where
Algebraic expressions for

Flynn–Wall–Ozawa method
This method which can designate the 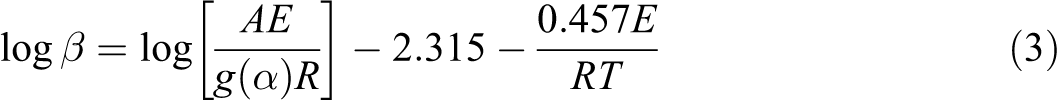
The
Tang method
With an approximation formula used and with the logarithms of the side taken for the solution of equation (2), the following equation can be acquired:
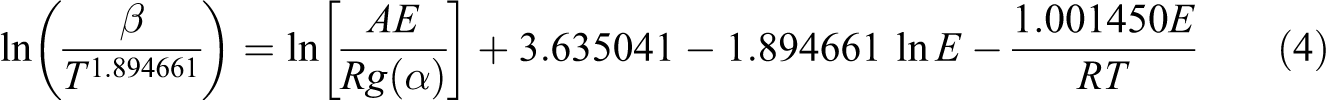
Plots of 1/
Coats–Redfern method
An asymptotic approach for the resolution of equation (2) uses the Coats–Redfern method:
32

The
Results and discussion
Synthesis and characterization of the macroinitiator
In this study, in order to carry out one-armed grafting on P(MAOCMC-co-St), resulting in P[(MAOCMC-co-St)-g-MMA] (Figure 1). Consequently, it was found that the chloro group in the macroinitiator was to be in ratio of 5% (by mole). The macroinitiator for ATRP, P(MAOCMC-co-St), was acquired by copolimerization. Characteristic bands of the phenyl in styrene units at 3050 and 3075 cm−1 (=C–H stretching vibration), at 1602 cm−1 (C=C stretching vibration) and 756 and 700 cm−1 (=C–H out-of-plane bending of phenyl ring) were observed. Figure 2 indicates that the FTIR spectra of P(MAOCMC-co-St) has a characteristic absorbance at 1737 cm−1 (C=O stretching vibration), 3073 (–CH stretching of the aromatic ring), 2986 (–CH3), 1737 (broad, C=O in ester), 1614 (C=C stretching of the aromatic ring), 1230 (asymmetric C–O–C) and 1142 (symmetric C–O–C). 1454 cm−1 and 1496 cm−1 were assigned to typical C–C vibrations of the polystyrene chain. 33
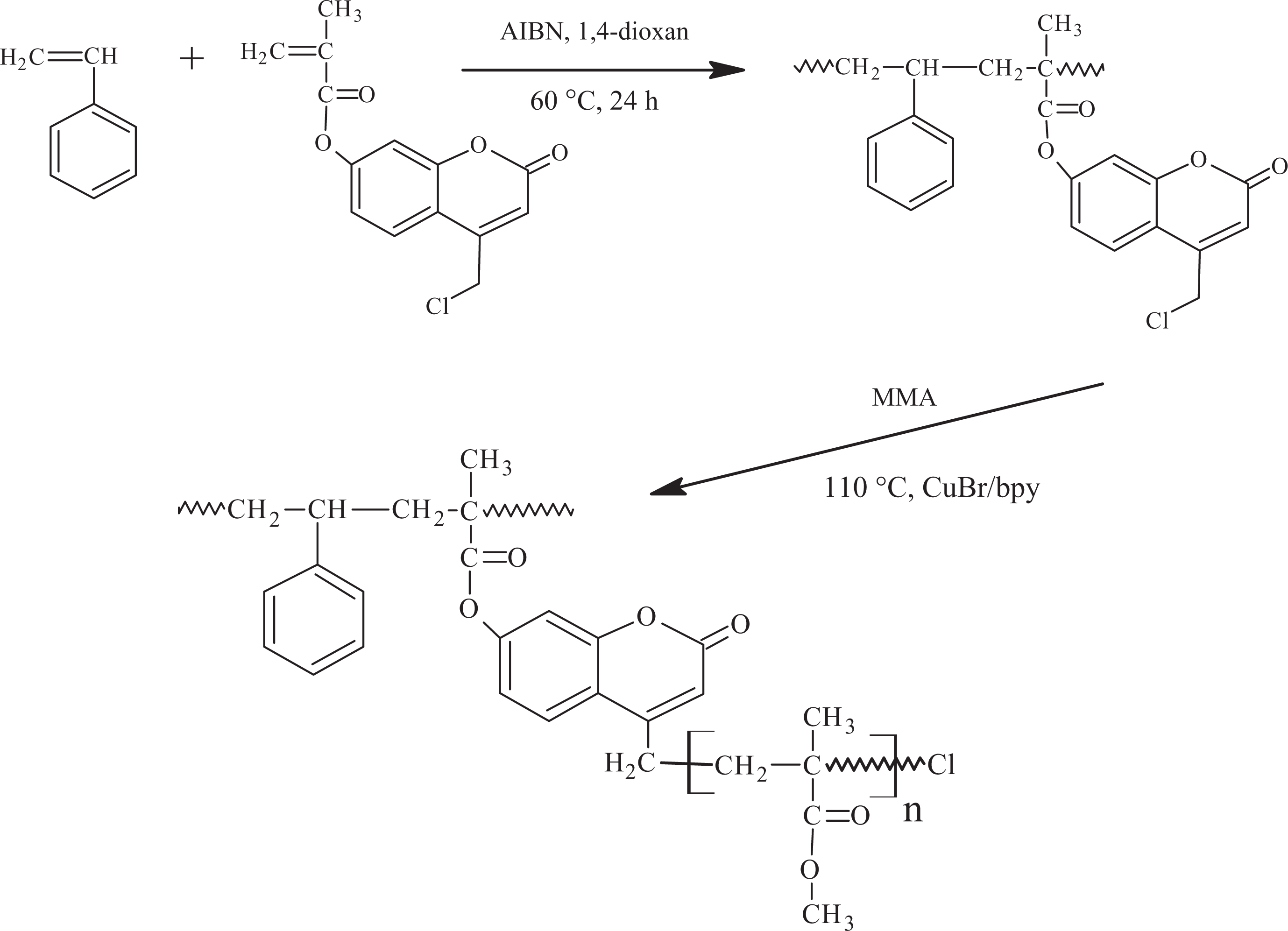
Synthesis of macroinitiator and graft copolymers.
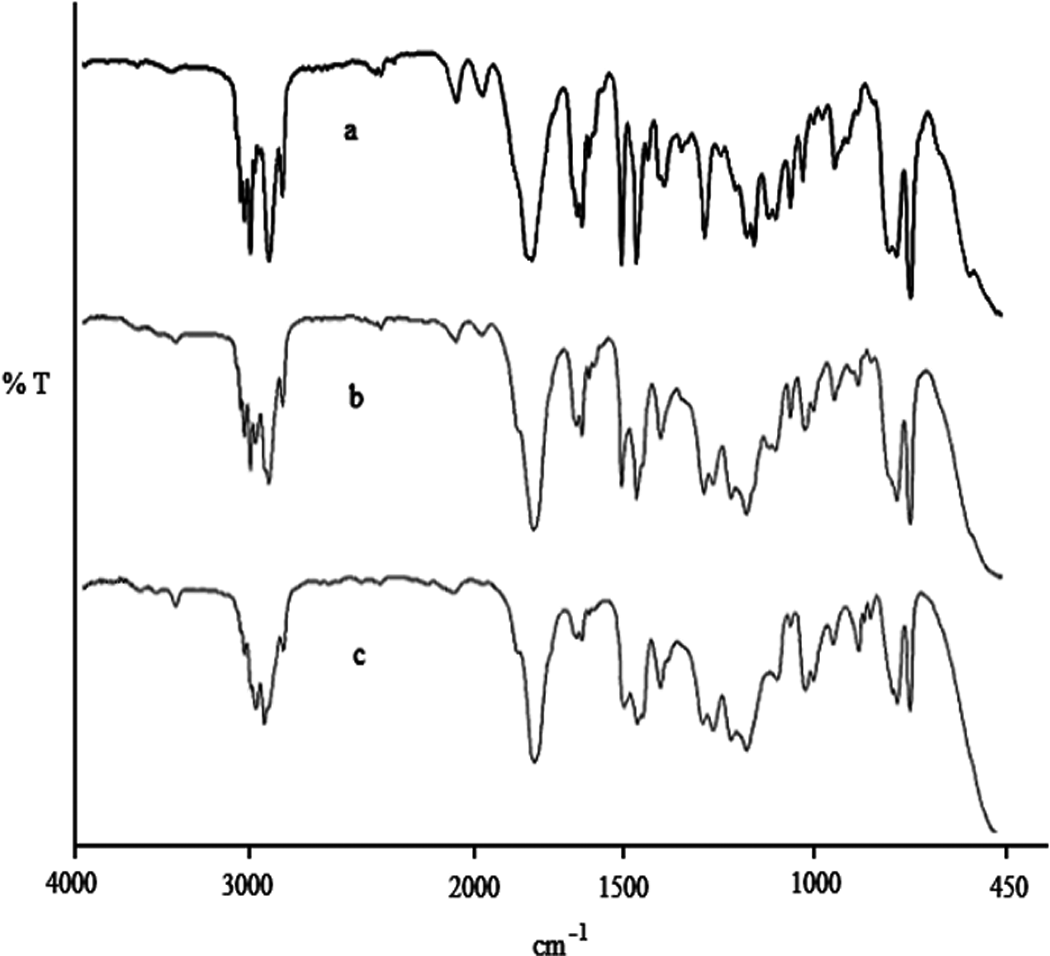
The FTIR spectra of (a) P(MAOCMC-co-St), (b) P[(MAOCMC-
Synthesis and characterization of the graft copolymers
The macroinitiator, P(MAOCMC-
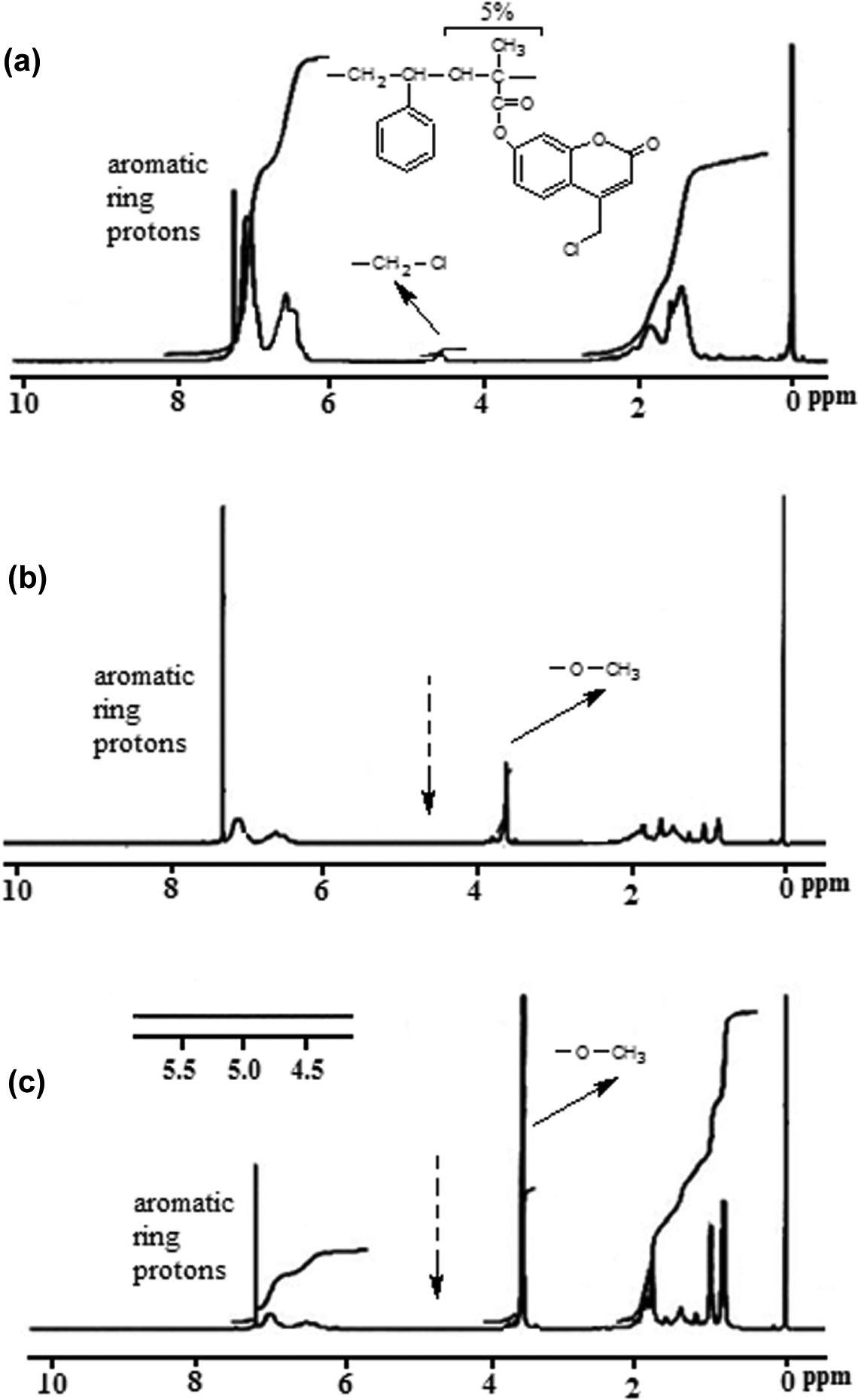
The
1
H NMR spectra of (a) P(MAOCMC-
UV–vis spectra of the P(MAOCMC-
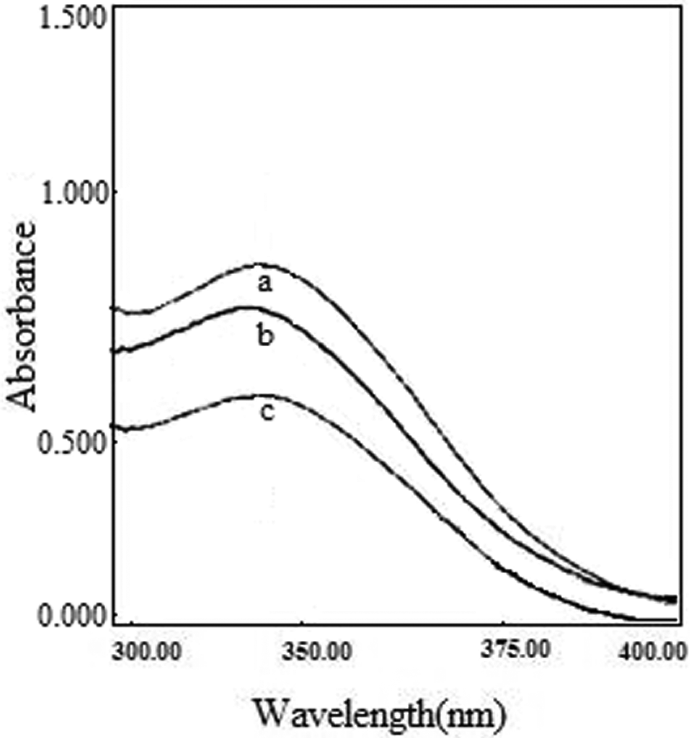
The UV–vis spectra of (a) P(MAOCMC-
Thermal decomposition kinetics
Thermal decomposition curves of the graft copolymers and macroinitiator were carried out at discrepant heating rates of 5, 15, 25, and 35°C min−1. After complete degradation, the initial decomposition temperature, decomposition temperature at 50% weight loss, weight loss (%) at 300°C and 350°C and residual mass at 500°C were designated from these curves and are presented in Tables 2 to 4.
TGA data of macroinitiator at different heating rates.
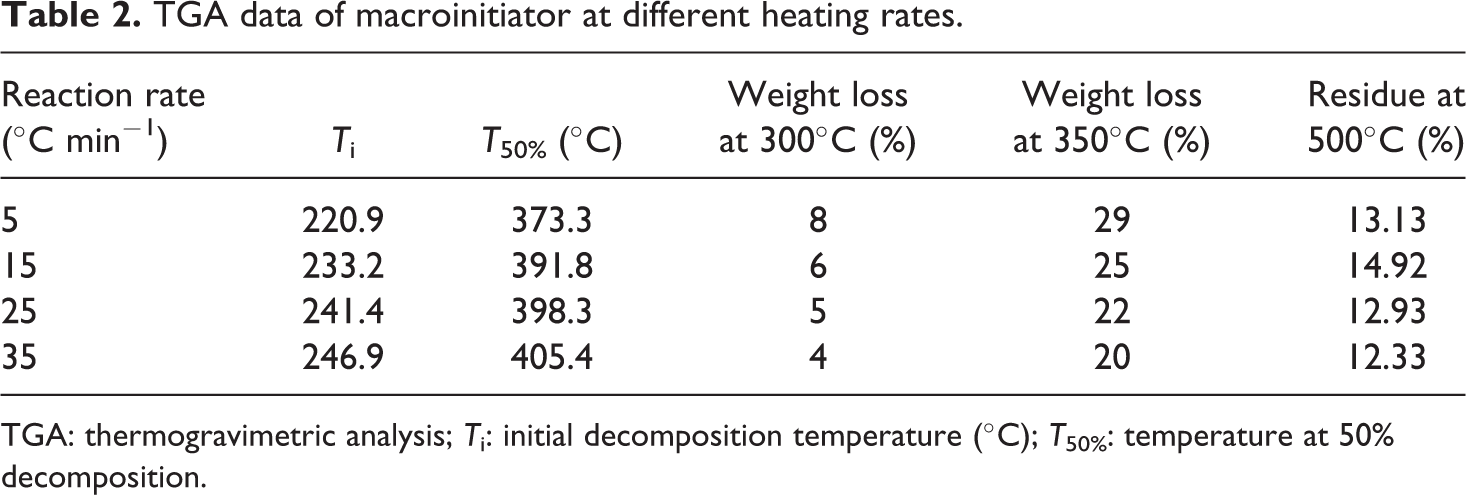
TGA: thermogravimetric analysis;
TGA data of graft copolymer (30 h) at different heating rates.

TGA: thermogravimetric analysis.
TGA data of graft copolymer (60 h) at different heating rates.

TGA: thermogravimetric analysis;
These curves indicate that, for graft copolymer (60 h) at 500°C, the residue decreased 3.5% with a 5°C min−1 heating rate. The 10°C min−1 intervals between measurements were chosen to avoid the imbrication of inflection point temperatures.
27
The
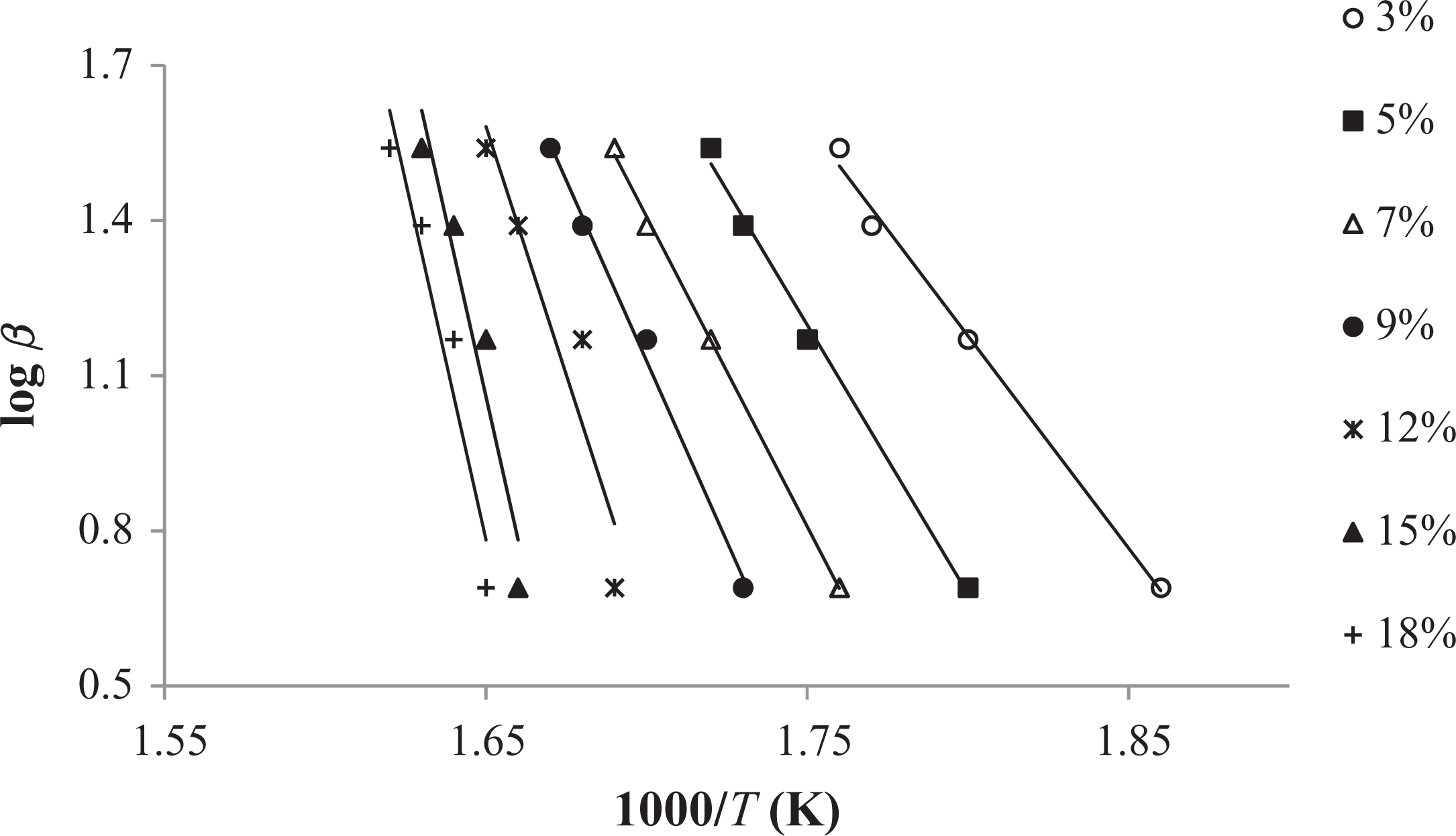
FWO method applied to the experimental data (3–18%) of macroinitiator. FWO: Flynn–Wall–Ozawa.
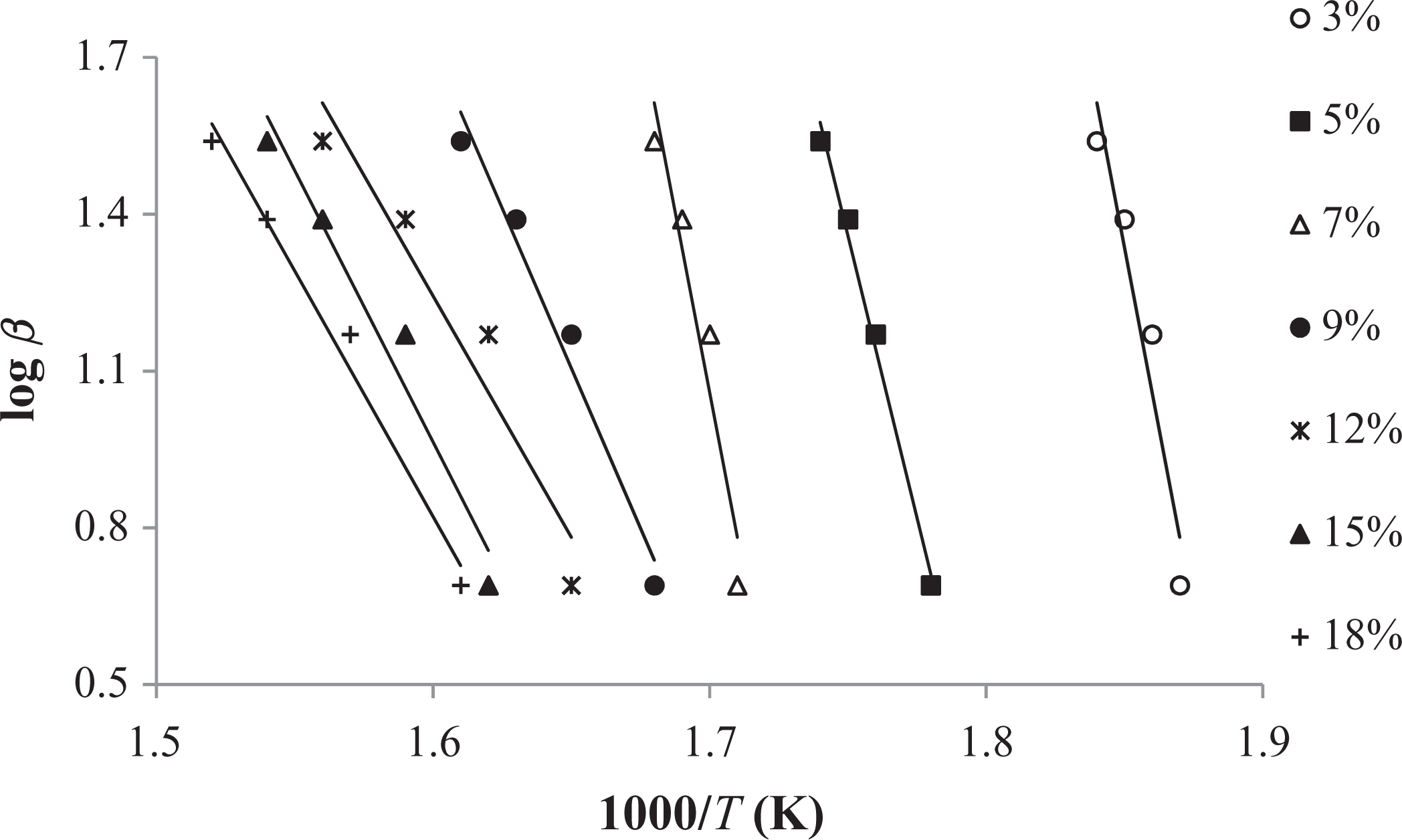
FWO method applied to the experimental data (3–18%) of graft copolymer (30 h). FWO: Flynn–Wall–Ozawa.
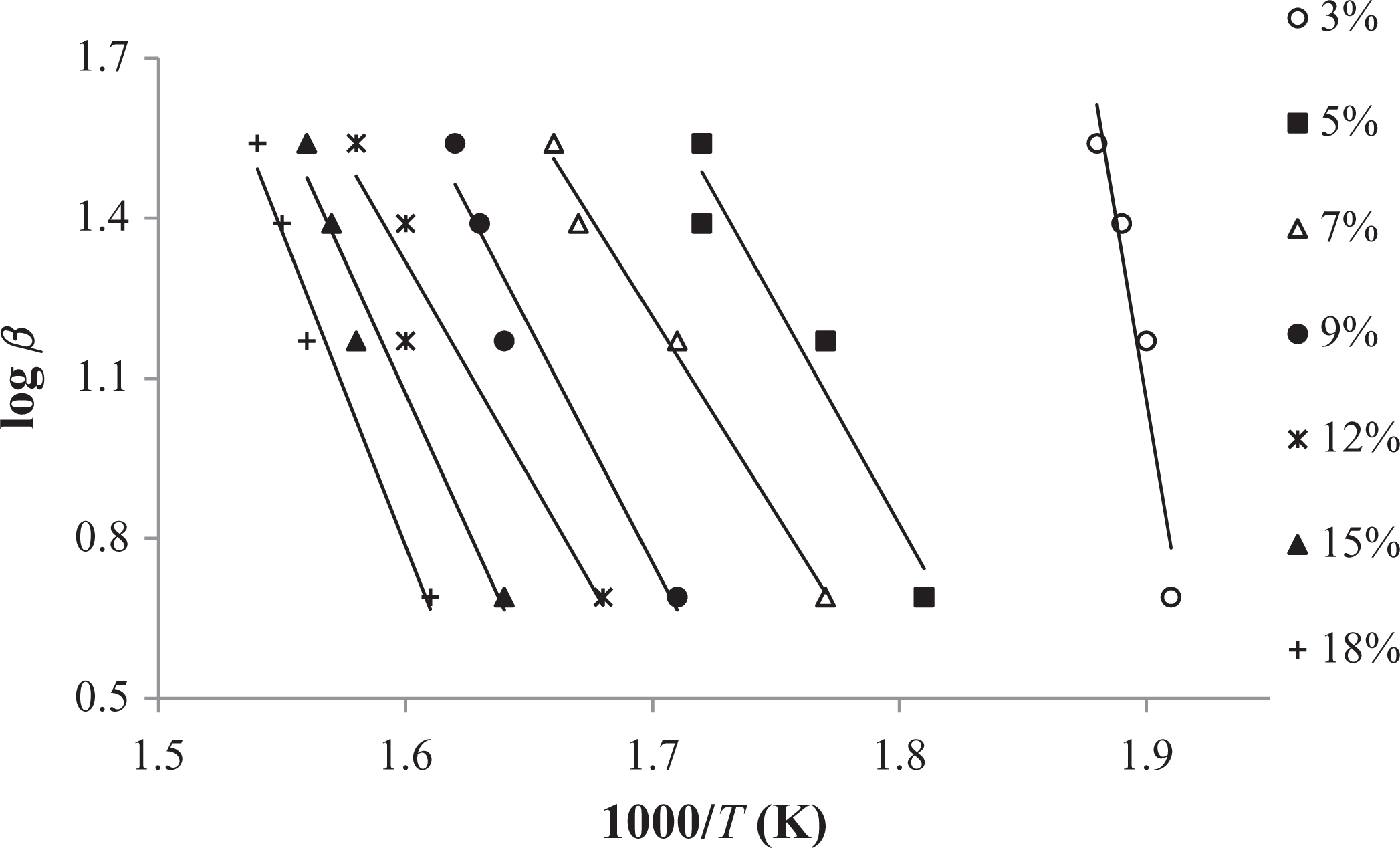
FWO method applied to the experimental data (3–18%) of graft copolymer (60 h). FWO: Flynn–Wall–Ozawa.
For the detection of
Tang method applied to the experimental data (3–18%) of macroinitiator.
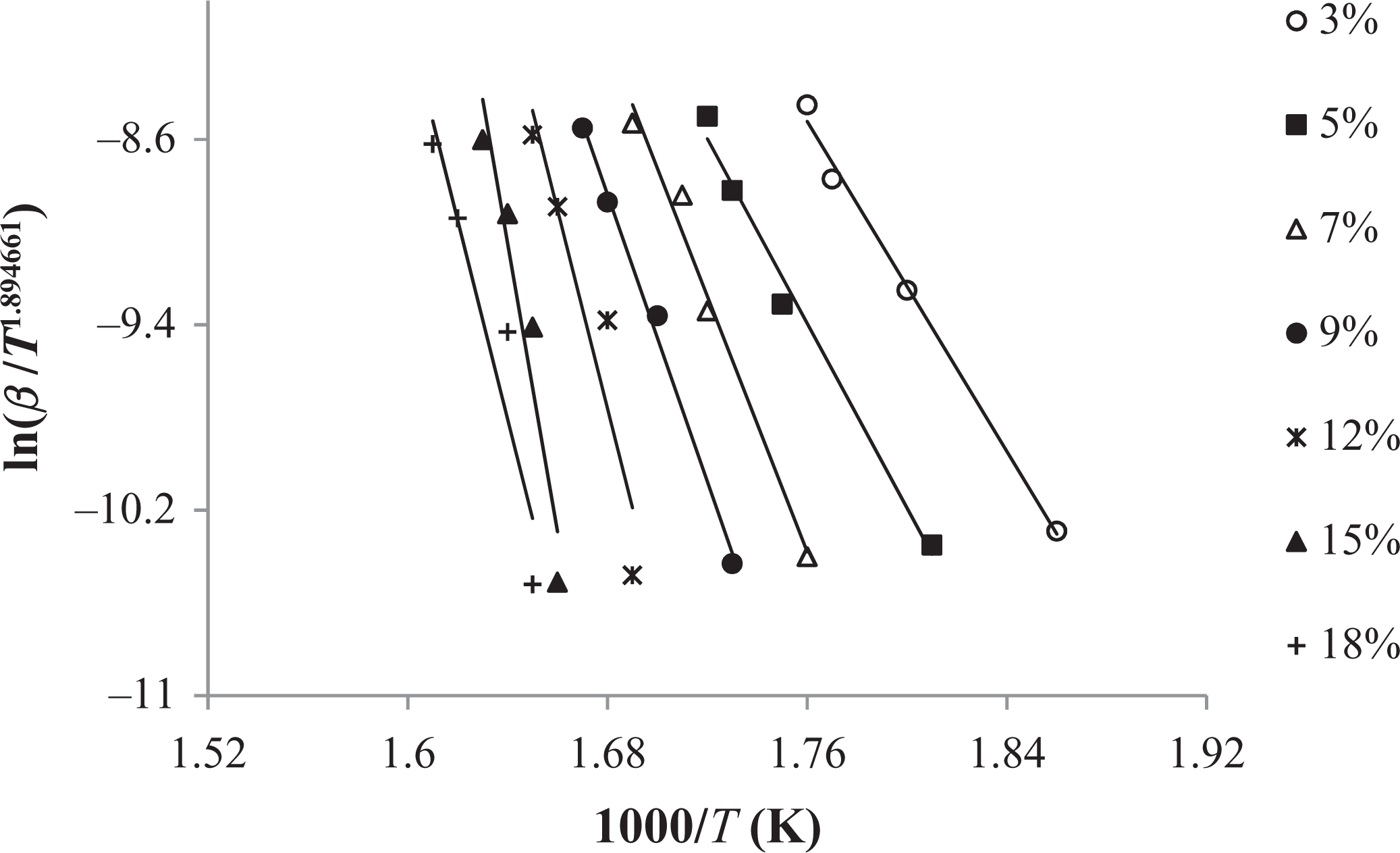
Tang method applied to the experimental data (3–18%) of graft copolymer (30 h).
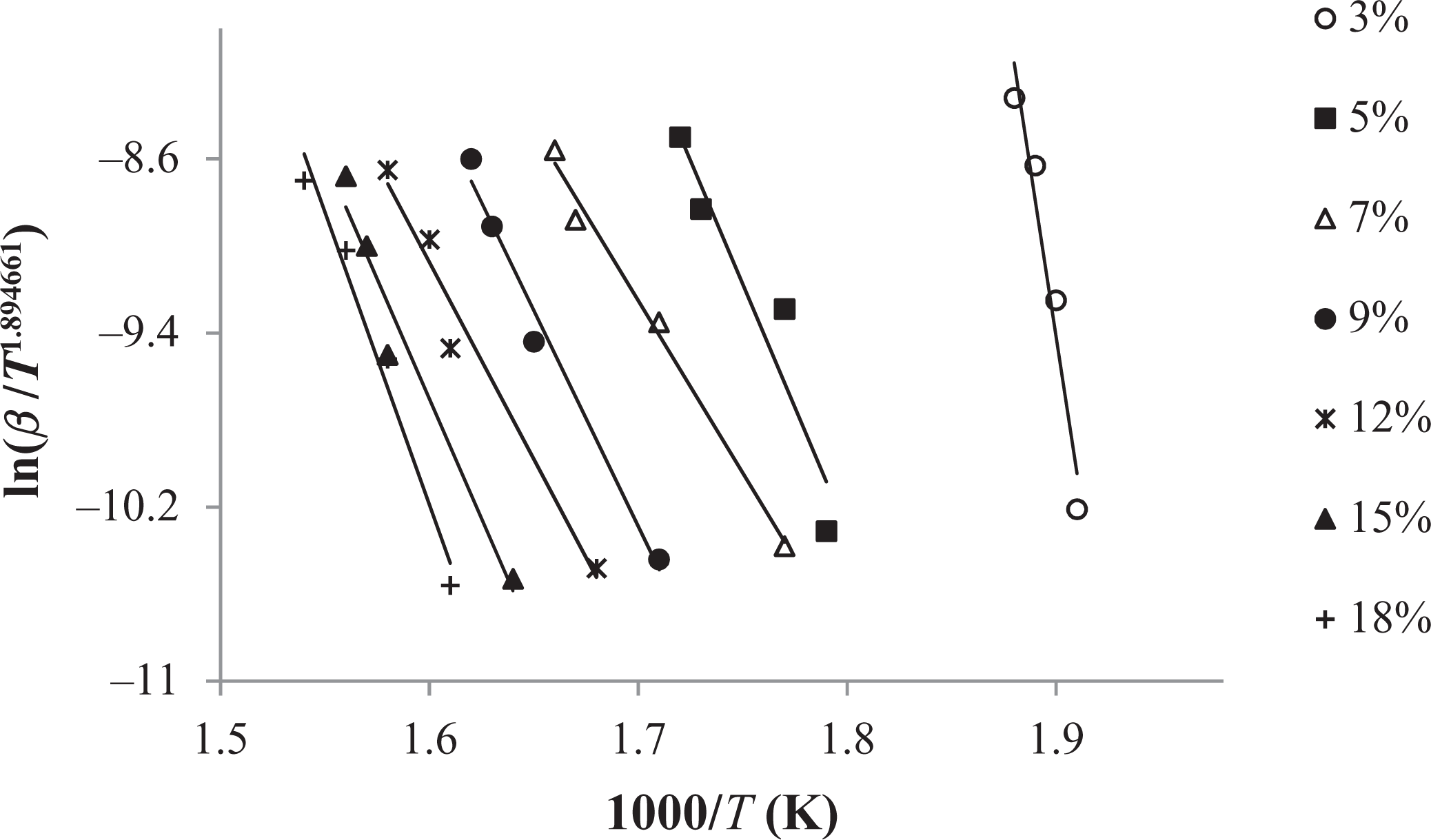
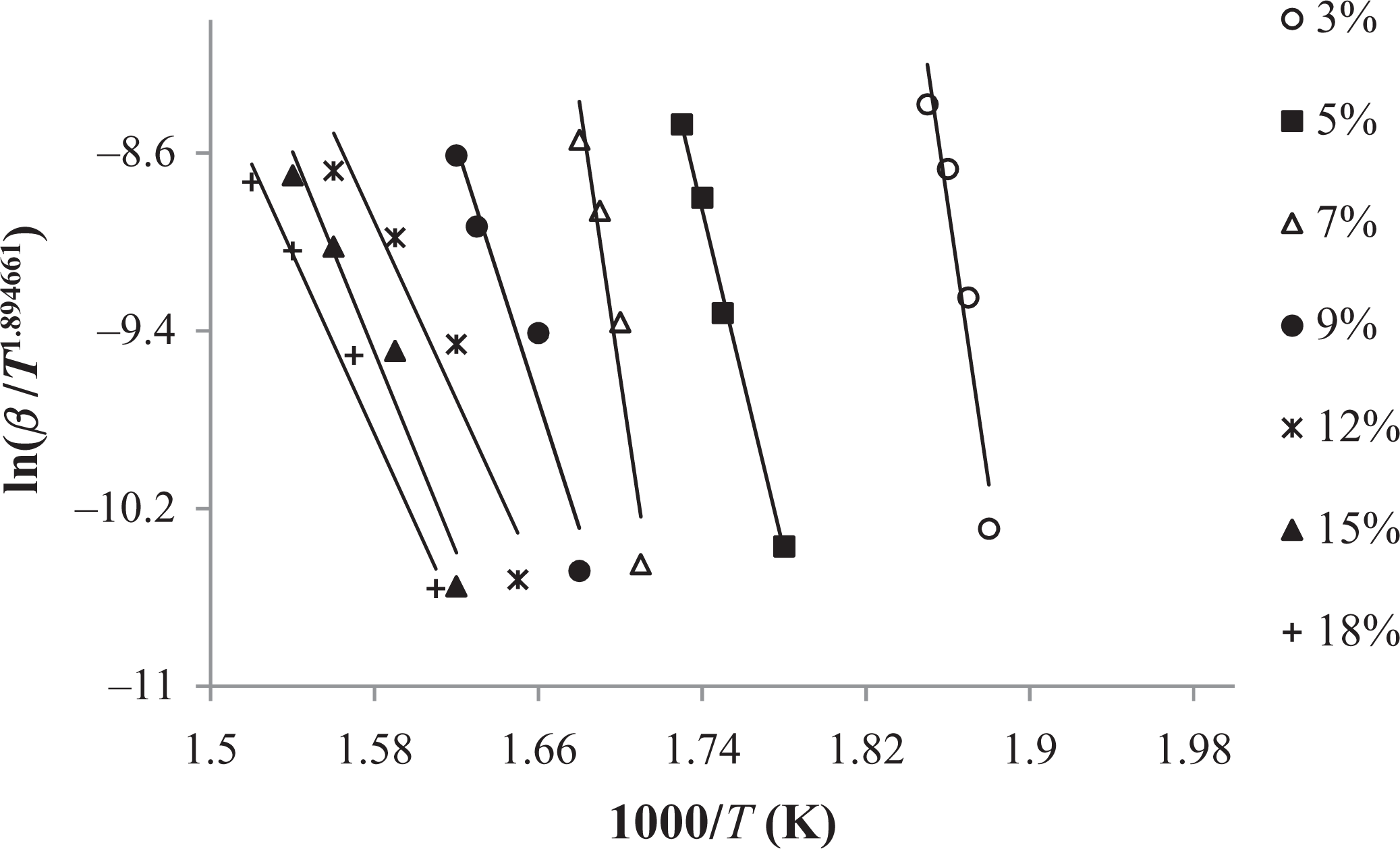
Tang method applied to the experimental data (3–18%) of graft copolymer (60 h).
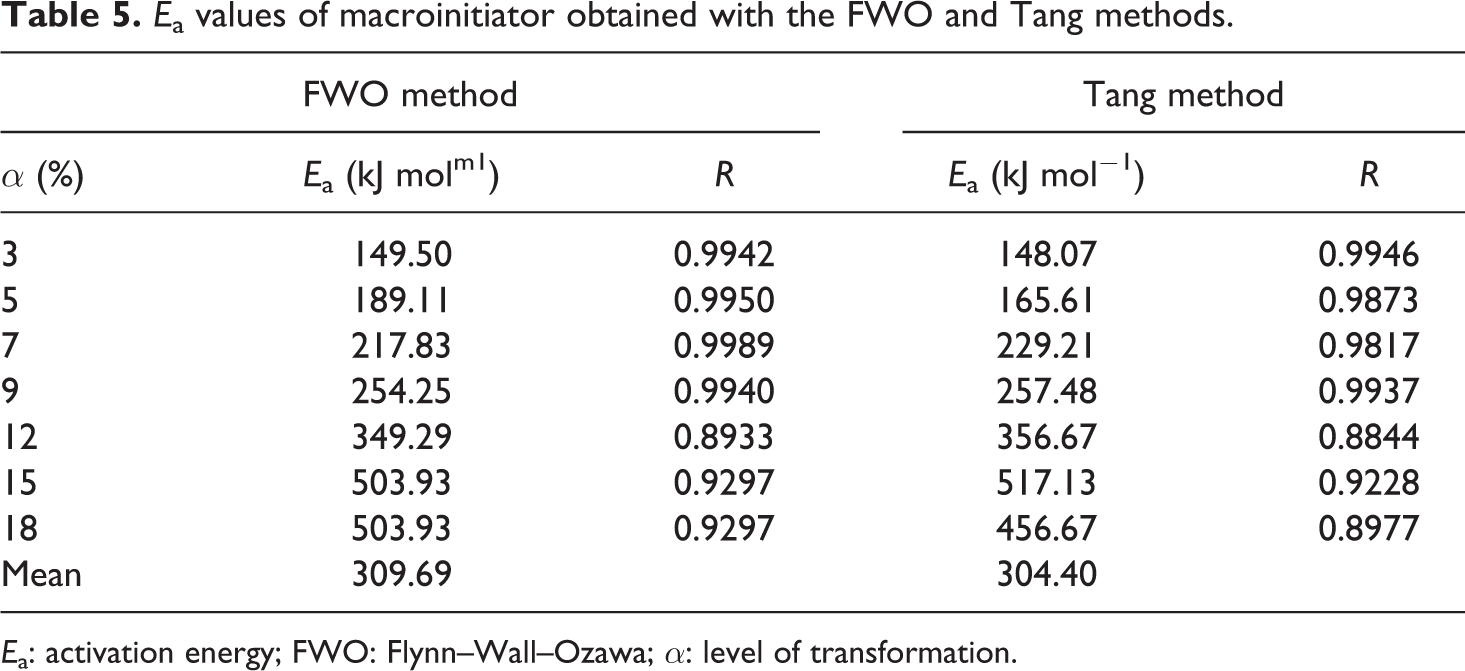
The conjectured outcomes are summarized in Tables 5
to 7. The mean value of the
In comparison to other methods, Tang and FWO methods are advantageous, because they don’t require previous knowledge of the reaction mechanism for designating the
The
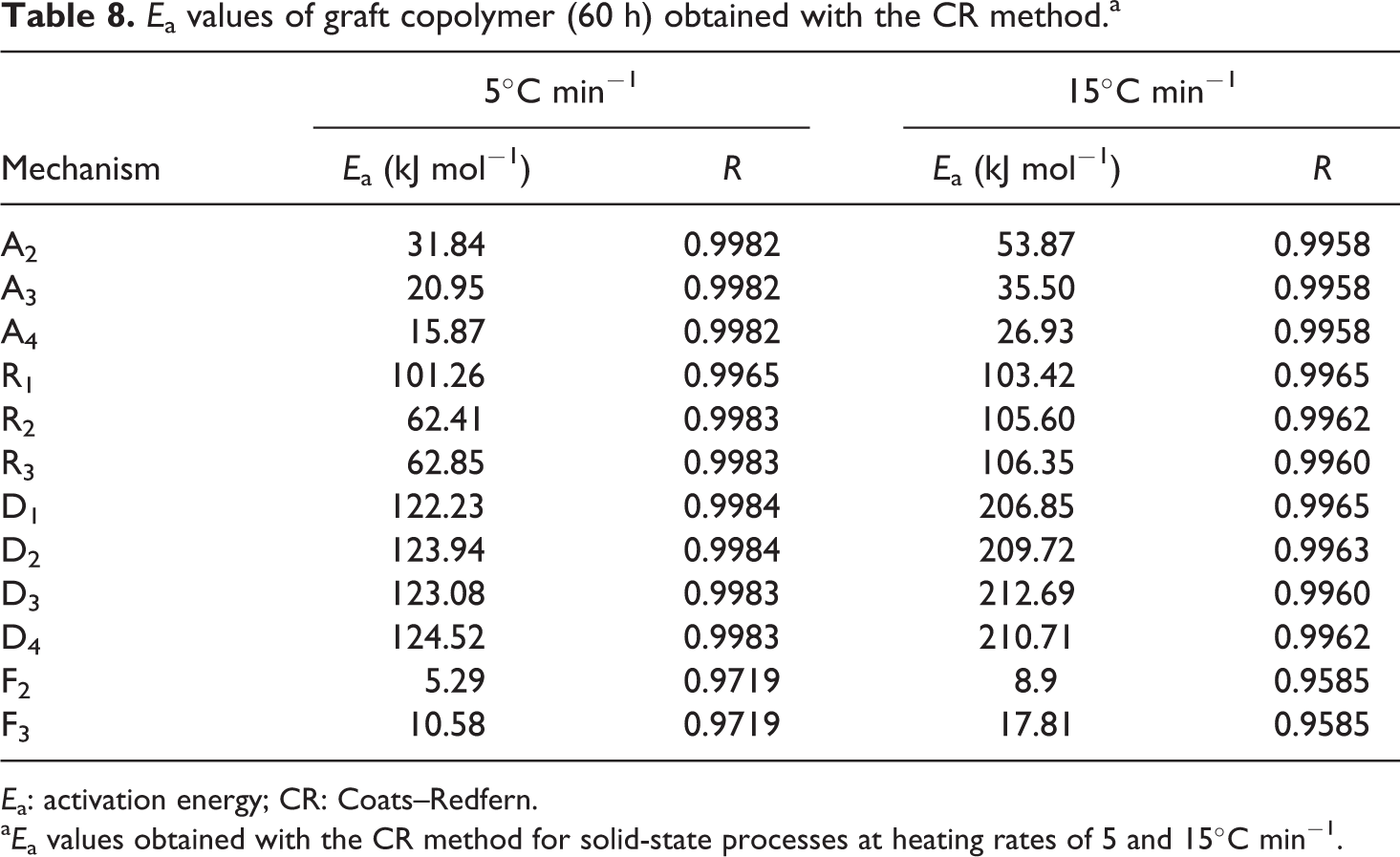
a
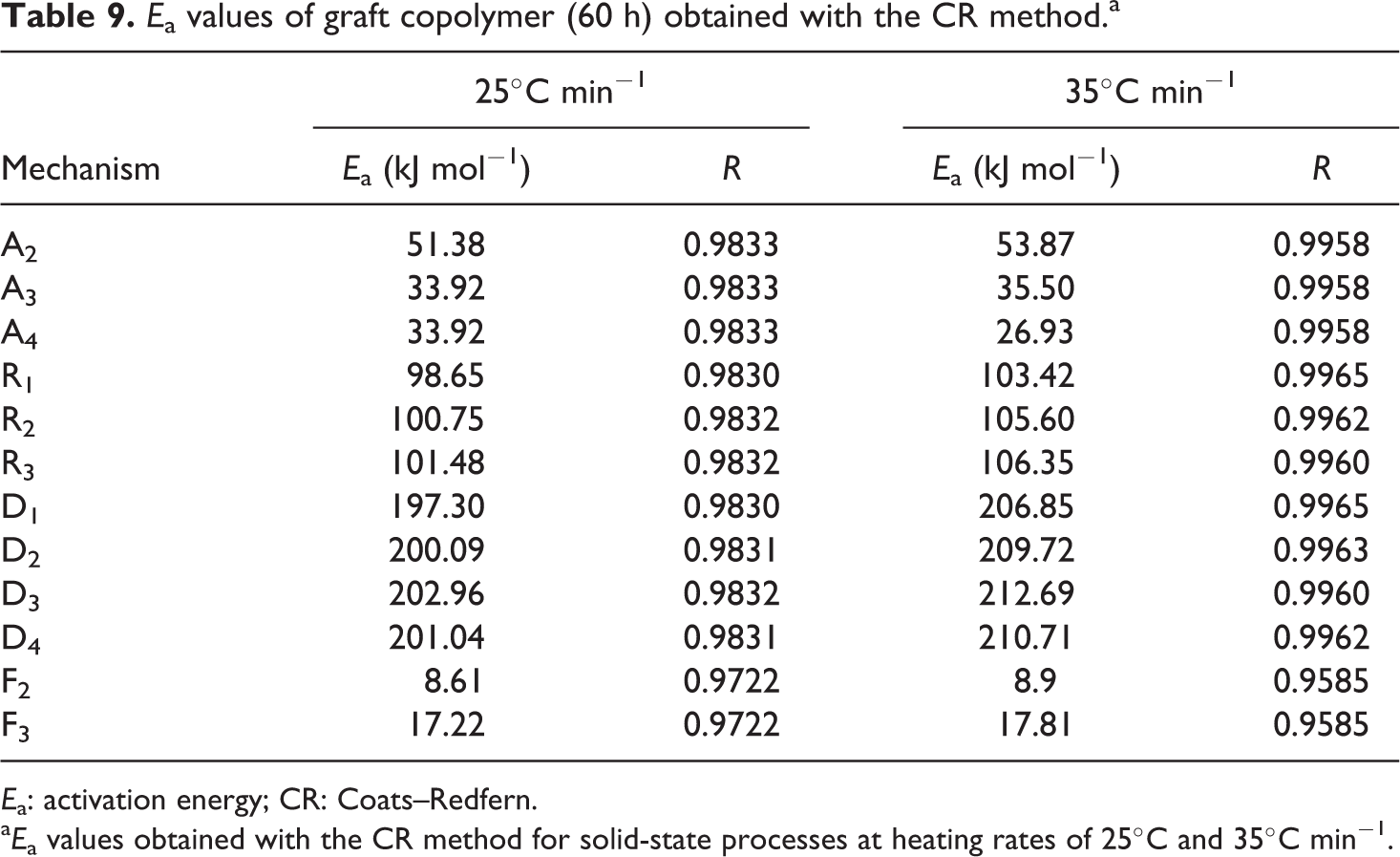
a
Conclusions
The ATRP macroinitiator was prepared by copolymerization of St with MAOCMC. The grafting of the macroinitiator with MMA was carried out at 110°C. The FTIR and
1
H NMR data confirmed the structures of all the graft copolymers and the starting materials. The thermal stability of the graft copolymers (30 h and 60 h) indicated higher thermal stability. The thermal degradation kinetics of the polymers was also investigated with diversified methods of TGA. The
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to thank FUBAP-1792 for financial support of this project.
