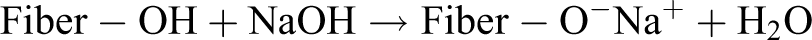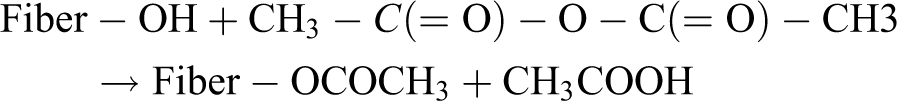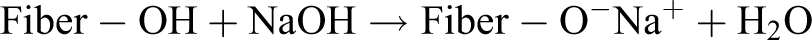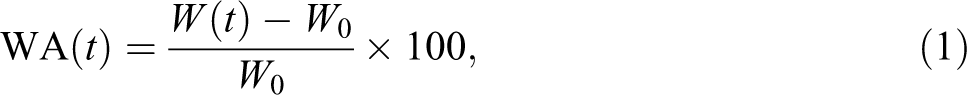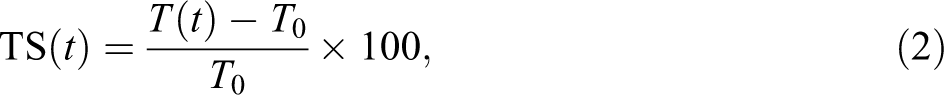Abstract
In this article, the different chemical modifications of natural fiber on the physical and mechanical properties of polypropylene (PP)/wood flour (WF)/nanoclay hybrid composites were studied. To meet this objective, the chemical treatments on WF, namely alkali, acetylation, and benzoylation, were carried out. Then, the composites were prepared through the melt mixing of WF and PP at 50% weight ratios, with various amounts of nanoclay (0, 3, and 6 per hundred compounds (phc)) in Haake internal mixer and the test specimens were prepared by injection molding. The amount of coupling agent was fixed at 2 phc for all formulations. The tensile strength, impact strength, and water uptake of these composites were analyzed and the reinforcing properties of the chemically treated composites were compared with those of untreated composites. The chemical modification efficiency was verified by Fourier transform infrared (FTIR) analysis, and the dispersion state of nanoclay in the composites was examined using X-ray diffractometer and transmission electron microscopy. The results showed that the tensile modulus, tensile strength, and impact strength of the composites increased by applying chemical treatments. Modified composites had lower water absorption (WA) and thickness swelling than unmodified ones. Furthermore, the highest mechanical properties and the lowest water uptake were observed in the composite modified with acetic anhydride. FTIR spectra show that the intensity of O–H bond at 3444.2 cm–1 and formation of ester bond at 1741.4 cm–1 were indications of changes in the chemical structure of the fibers. The tensile modulus and strength increased with increase of nanoclay up to 3 phc and then decreased. However, the impact strength and WA decreased with increase of nanoclay loading. The morphological findings showed that the samples containing 3 phc of nanoclay had higher order of intercalation and better dispersion.
Introduction
In recent years, wood-derived fillers have made significant contributions to the thermoplastic industry, which has led to the emergence of wood–plastic composites (WPCs) in the construction industry. Products such as decking, fencing, siding, window framing, and roof tiles are being introduced into the market. The uses of WPCs are also increasing in construction, transportation, industrial, and consumer industries. Growing interest of renewable resources-based products is due to social and environmental concerns. Natural fibers have several advantages, such as being inexpensive, being renewable, being lower density, undergoing little damage during processing, and their disposal causing minor ecological impact. Commodity thermoplastics such as polyethylene, polypropylene (PP), polyvinyl chloride, and polystyrene are more commonly used in the manufacture of plastic/wood fiber composites. 1 –4
Natural fibers have some disadvantages, such as lower processing temperature and incompatibility between the hydrophilic fibers and the hydrophobic polymers. The incompatibility results in a poor interfacial adhesion between hydrophilic fibers and the hydrophobic plastic matrix, which results in poor adhesion and therefore poor ability to transfer stress from the matrix to the fiber, reducing mechanical strengths and ductility. 3,4 Therefore, it is imperative that natural fibers be subjected to chemical modification to increase the compatibility and adhesion between fibers and the matrix. Chemical modifications may activate these groups or can introduce new moieties that can effectively interlock with the matrix. Some important fiber chemical modifications include mercerization, isocyanate treatment, acrylation, permanganate treatment, acetylation, silane treatment, and peroxide treatment with various coupling agents, and other pretreatments of natural fibers have achieved various levels of success in improving fiber strength, fiber fitness, and fiber–matrix adhesion in natural fiber-reinforced composites. Simple chemical treatments can be applied to the fibers to change surface tension and polarity through modification of fiber surface. 5,6
Jacob and Anandjiwala 7 studied the chemical modification of flax-reinforced PP composites. They found that composites containing chemically modified flax fibers were found to possess improved mechanical properties. Also chemical modification was found to increase the storage modulus due to enhanced interfacial adhesion. 7 Dányádi et al. 8 investigated the effect of various surface modifications of wood flour (WF) on the properties of PP/wood composites. They found that surfactants improve homogeneity and processability and clearly prove that the proper selection of the approach and level of surface modification may lead to considerable improvement in targeted properties. 8 Ghasemi and Farsi 9 reported that the chemical treatment of lignocellulosic fillers results in better dispersion of the filler into the polymer matrix and leads to higher impact resistance in comparison with samples containing filler with no chemical treatment. In addition, the scanning electron micrographs showed that the best encapsulation of WF by polymer matrix is observed in samples of chemically treated fibers. 9
Recently, nanoscience and nanotechnology have opened up a completely new way to develop polymer composites. 10 Nanotechnology is a very promising field for improving the properties of polymer composites using nanosized fillers. Nanocomposite technology with layered silicate clays as in situ reinforcement has been intensively investigated. Essential improvements of physical and mechanical properties, thermal stability, flame resistance, and barrier resistance have been observed for various thermoplastic and thermoset nanocomposites at low silicate content. 10 –13 Nanoclays are the most commonly used layered silicate because of their natural occurrences and beneficial properties (high cationic exchange capacity, high surface area, and large aspect ratio). Using nanoclay filler in WPC composite has been reported in the literature. 14 –20 Many efforts have been made in the formation of wood polymer nanocomposite to improve such properties so as to meet specific end-use requirements.
The objective of this work was to investigate the effect of chemical modification of WF on the physical and mechanical properties of PP/WF/nanoclay hybrid composites.
Experimental
Materials
The polymer matrix used in this study was PP with a melt flow index of 18 g/10 min, and a density of 0.2 g cm−3 (supplied by Arak Petrochemical Co., Iran). WF that was used as the reinforcing material was supplied by Cellulose Aria Co. (Iran); the average size of WF particles was about 425 μm. Maleic anhydride-grafted polypropylene (PP-g-MA) provided by Solvay with trade name of Priex 20070 (MFI = 64 g/10 min, grafted maleic anhydride 1 wt%) was used as a coupling agent. Montmorillonite (MMT) modified with a quaternary ammonium salt (methyl ammonium chloride) of bis-2-hydroxyethyl tallow as an organic modifier, having a cation-exchange capacity of 90 meq/100 g clay, a density of 1.98 g cc−1, and a d-spacing of d001 = 18.5 Å, was obtained from Southern Clay Products Co. (Gonzalez, Texas, USA), with the trade name Cloisite 30B.
Method
Chemical modification
Mercerization
The wood fibers were soaked in 5 wt% sodium hydroxide (NaOH) solution at ambient temperature, maintaining a solution:fiber ratio of 20:1 (by weight). The fibers were immersed in the solution for 30 min. After treatment, fibers were copiously washed with distilled water to remove any traces of alkali on the fiber surface and subsequently neutralized with 1 wt% acetic acid solution. The treated fibers were then dried in an oven at 80°C for 24 h. The proposed reaction for this treatment is given in the following equation:
5–6
Acetylation
The wood fibers pretreated with NaOH solution were soaked in 55% solution of glacial acetic acid for 1 h at 30°C; it was decanted and soaked in acetic anhydride containing one drop of concentrated sulfuric acid for 5 min. Then, the fibers were placed in a preheated oven at 120°C for 5 h with the aim of encouraging esterification. Lastly, treated fibers were thoroughly washed with water to remove residual chemicals until a pH of 7 was obtained and then dried in an oven at 80°C for 48 h. The reactions in this treatment can be illustrated as follows:
5–6
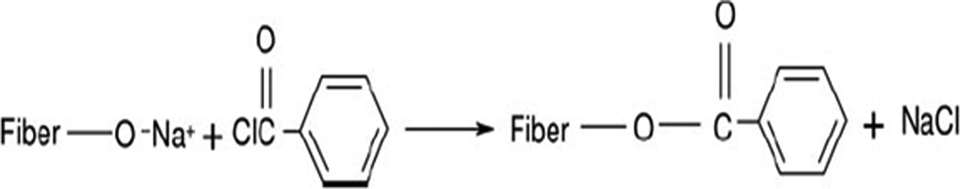
Benzoylation
The wood fibers pretreated with NaOH solution were suspended in 10 wt% NaOH solution and mixed well with 3 wt% (weight percent compared with the fiber) benzoyl chloride for 30 min. Excess amount of water was used to wash out NaOH solution, and the benzoylated fibers were immersed in ethanol solution for 1 h to remove unreacted benzoyl chloride. Finally, the fibers were washed out with water and dried in oven at 80°C for 24 h. The reactions in this treatment can be illustrated as follows:
5–6
Composite preparation
Before preparation of samples, WF was dried in an oven at 65 ± 2°C for 24 h. Composite profiles consisting of PP and WF at 50% weight ratios, with various amounts of organo-MMT (0, 3, and 6 per hundred compounds (phc)) produced. The amount of coupling agent was fixed at 2 phc for all formulations. The mixing was carried out by a Haake internal mixer (HBI System 90, Dearborn, Michigan, USA). First, the PP was fed to the mixing chamber; after melting of high-density polyethylene (HDPE), coupling agent and MMT were added. At the 5th min, the WF was fed, and the total mixing time was 13 min. The compounded materials were then ground using a pilot-scale grinder (WGLS 200/200 model, WIESER, Germany). The resulted granules were dried at 105°C for 4 h. Test specimens were prepared by injection molding (Eman machine, Iran). Finally, specimens were conditioned at a temperature of 23°C and relative humidity of 50% for at least 40 h according to ASTM D 618 standards prior to testing.
Measurements
The flexural and tensile modulus tests were measured according to the ASTM D790 and D638 standards, using an Instron machine (model 4486, England); the tests were performed at a crosshead speed of 5 mm min−1. A Zwick impact tester (model SIT 20 D, Iran) was used for the Izod impact test. All the samples were notched on the center of one longitudinal side according to ASTM D 256 standard. For each treatment level, five replicate samples were tested.
Water uptake tests were carried out according to ASTM D 7031 specification. Five specimens of each formulation were selected and dried in an oven for 24 h at 102 ± 3°C. The weight and thickness of dried specimens were measured to a precision of 0.001 g and 0.001 mm, respectively. The specimens were then placed in distilled water and kept at room temperature (720 h). For each measurement, specimens were removed from the water, and the surface water was wiped off using a blotting paper. Weight and thicknesses of the specimens were measured after 720 h. The values of the water absorption (WA) in percentage were calculated using the following equation:
Also the values of the thickness swelling (TS) in percentage were calculated using equation (2):
Wide-angle X-ray diffraction (XRD) analysis was carried out with a Seifert-3003 PTS (Germany) with copper Kα radiation (λ = 1.54 nm, 50 kV, 50 mA) at room temperature; the scanning rate was 1° min−1. The morphology structure of the nanocomposites was investigated by a Philips (Model EM 208, Netherlands) transmission electron microscope (TEM) with an acceleration voltage of 100 kV. The ultrathin slides were obtained with a Leica Ultracut UCT device (Germany).
Fourier transform infrared (FTIR) spectroscopy measurements were performed using a Digilab FTIR spectrometer (model SP-1100; Marlborough, Massachusetts, USA) equipped with a deuterated triglycine sulfate (DTGS) detector. A total of five scans were taken for each sample with a resolution of 4 cm–1. Grounded dried fiber and potassium bromide (KBr; 2 mg fiber per 150 mg KBr) were pressed into a disk for FTIR measurement.
The statistical analysis was conducted using SPSS programming (Version 13) method in conjunction with the analysis of variance techniques. Duncan’s multiply range test was used to test the statistical significance at α = 0.05 level.
Results and discussion
Figure 1(a) shows FTIR spectra of the untreated WF. The strong peak at 3444.2 cm–1 is characteristic of the hydrogen-bonded hydroxyl group (OH) stretching vibration. The peak at 3008.4 cm–1 was considered due to C–H stretching vibration of all hydrocarbon constituents of the fibers and the shoulder band observed at 1741.4 cm–1 was due to the absorption of carbonyl (C=O) stretching of hemicellulose present in the fibers. The peak at 1658.5 cm–1 due to CH is related to absorbed water and the peak at 1511.9 cm–1 was considered due to the benzene ring. The peak appearing at 1461.8 cm–1 was due to CH2 bending in lignin, and the peak at 1322 cm–1 was due to O–H in plane bending. The band at 1249.6 cm–1 was considered due to C–O stretching of acetyl in lignin. A small sharp band at 892 cm–1 appears to have arisen from β-glucosidic linkages between the sugar units in hemicellulose and cellulose.
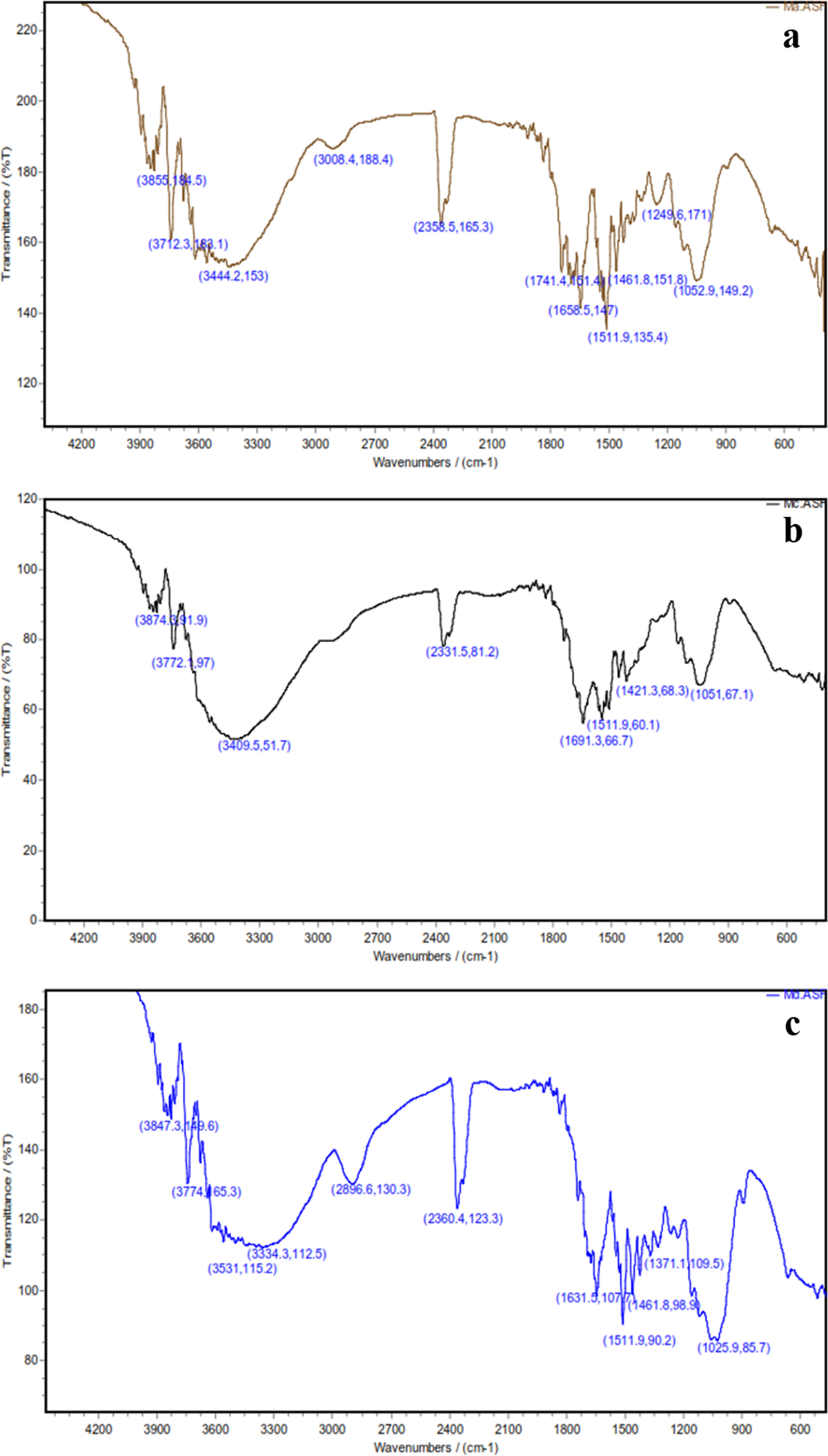
FTIR spectra of the chemical-treated WFs: (a) untreated, (b) mercerization, (c) acetylation, and (d) benzoylation. FTIR: Fourier transform infrared; WF: wood flour.
FTIR spectra of alkali treatment in Figure 1(b) show that the peak at 1741.4 cm–1 disappeared, which may be to be due to the removal of non-celluloses including fats, wax, pectin, and hemicellulose present in the fibers. 5 –9,21–22 Also, as can be seen in Figure 1(b), the O–H bond at 3444.2 cm–1 is affected by alkali treatment and its intensity is decreased. The main reason for this behavior is that by alkali treatment an amorphous region is developed in cellulose by the removal of OH groups from its network. Figure 1(c) shows that the acetylation of fibers with acetic anhydride led to the appearance of a new peak at 1631.5 cm–1 attributed to the C–O stretching of the ester carboxyl group. The lowering in intensity of the OH stretching band at 3334.3 cm–1 in the spectra also provided further evidence of successful acetylation. Figure 1(d) shows FTIR spectra of benzoylation of fibers. The intensity of the bond at 1744.1 cm–1 is decreased due to the formation of ester bonds from the reaction between O–H groups and benzoyl chloride (Figure 1(d)), while the OH band at 3444.2 cm–1 intensity tends to decrease due to its consumption in this reaction.
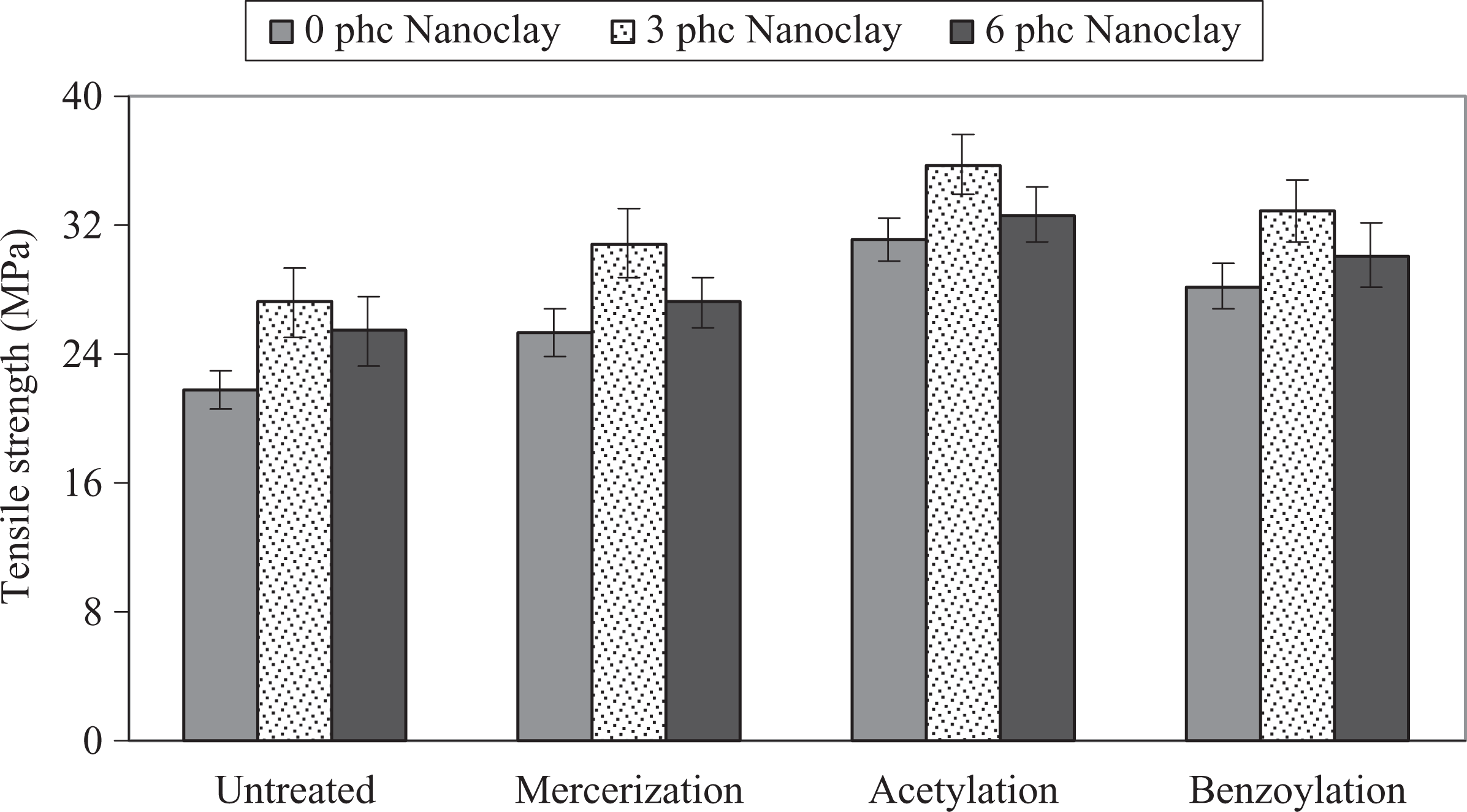
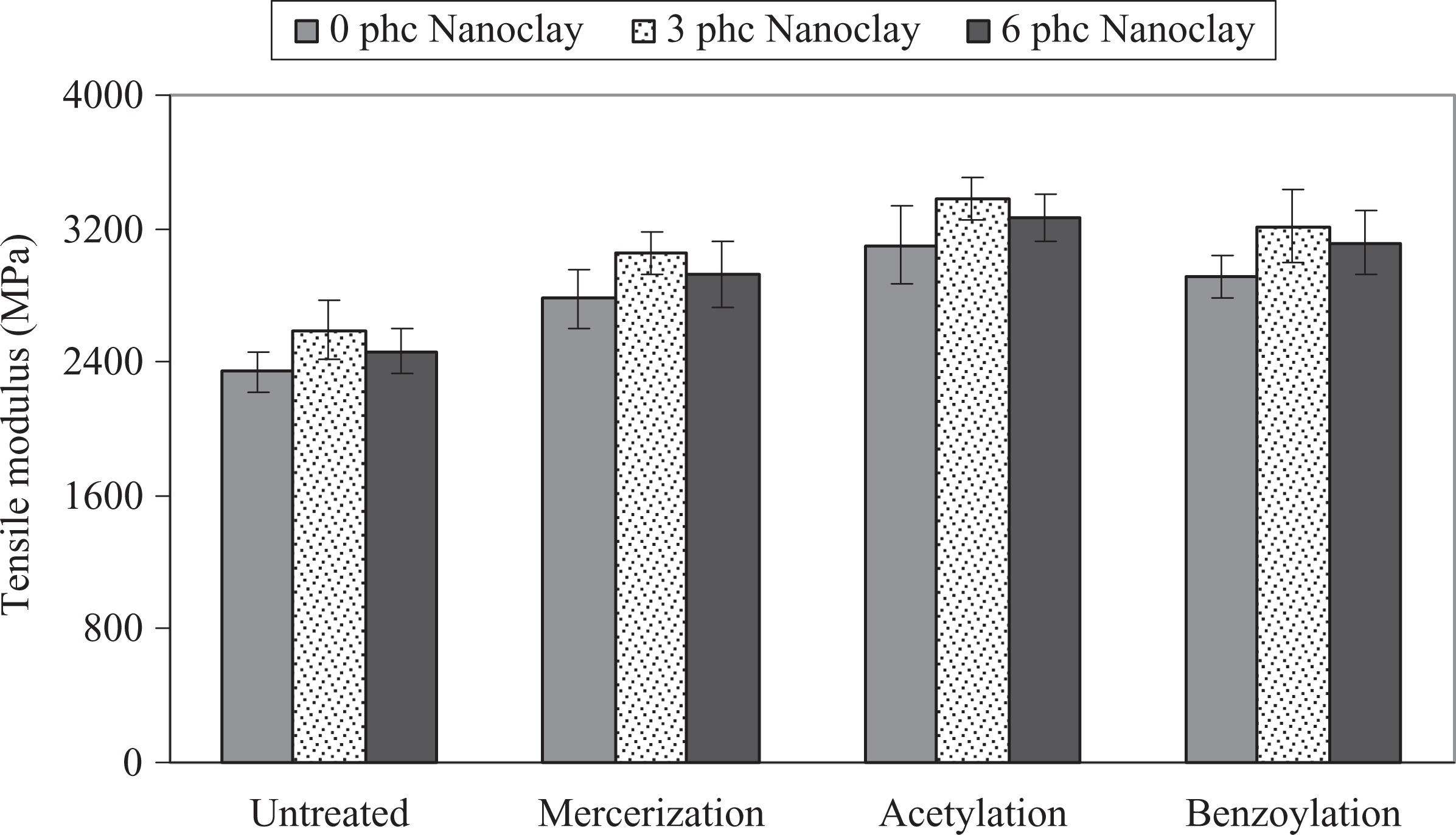
The influence of chemical modification of WF on the tensile modulus and strength of PP/WF/nanoclay hybrid composites is shown in Figures 2 and 3. As can be seen, the chemical modification increased the tensile modulus and strength of composites. The order of average tensile modulus and strength is acetic anhydride treatment > benzyl chloride treatment > alkali treatment > untreated. It is well known that the chemical modification promoted good wettability and better fiber–matrix interface adhesion, allowing efficient stress transfer between the WF and PP, which consequently results in higher tensile modulus and strength. 5 –9,21 –29 Moreover, as reported by Matuana et al., 22 chemical modification reduces fiber diameter and thereby increases the aspect ratio, improving mechanical characteristics of the composites.
Influence of chemical modification of WF on the tensile modulus of PP/WF/nanoclay hybrid composites. PP: polypropylene; WF: wood flour.
Influence of chemical modification of WF on the tensile strength of PP/WF/nanoclay hybrid composites. PP: polypropylene; WF: wood flour.
Also, Figures 2 and 3 show that the tensile modulus and strength increased with the increase of nanoclay up to 3 phc and then decreased. It is well known that the nanoclay particles with very high aspect ratio can improve the tensile modulus and strength of the PP. 14 –16 The increment of the modulus depends on the morphology of nanocomposites. 17 –19 The reinforcing efficiency of the nanofiller is balanced by two opposite phenomena. A negative effect is attributed to the migration of nanoparticles into the wood–plastic interface causing decreased performance. At 6 phc of nanoclay, agglomeration of nanoparticles could decrease the reinforcement of clay. Dispersion of nanoclay, as a positive effect, could enhance the modulus and strength; therefore, it can be concluded that at a level of 3 phc of nanoclay in the hybrid composite, the former phenomenon was dominant and the tensile modulus and strength increased.
Figure 4 shows the effect of chemical modification of WF on the impact strength of PP/WF/nanoclay hybrid composites. As can be seen, the chemical modification increased the impact strength of composites. The order of average impact strength is acetic anhydride treatment > benzyl chloride treatment > alkali treatment > untreated. It seems that by applying chemical treatment, the hydrophilic nature of fiber is reduced for better dispersion of the fiber into the polymer matrix. This phenomenon leads to higher impact resistance in comparison with the samples containing untreated fiber. 9,26 –29
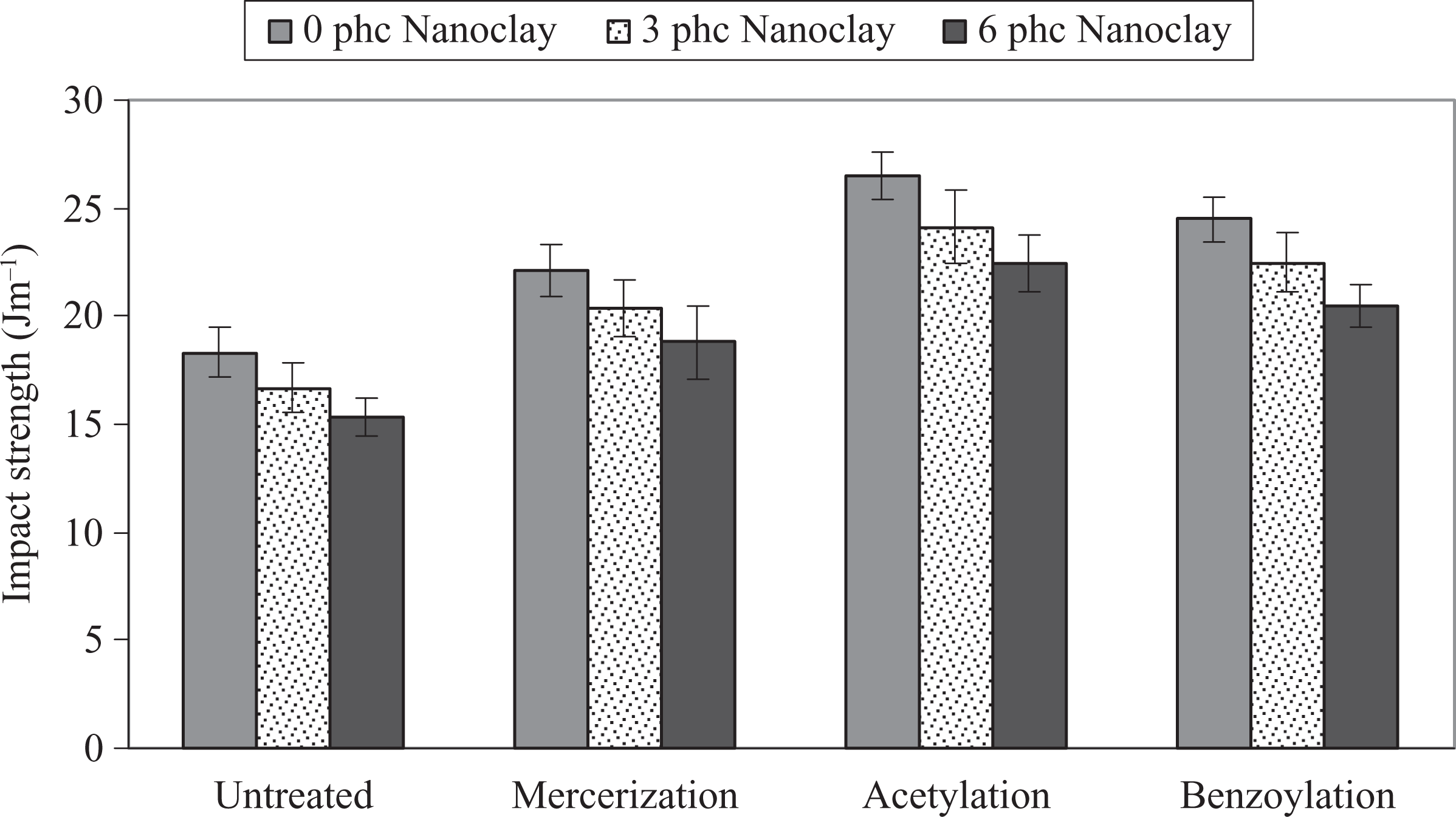
Influence of chemical modification of WF on the impact strength of PP/WF/nanoclay hybrid composites. PP: polypropylene; WF: wood flour.
Also, Figure 4 shows significant reduction in the impact strength at 6 phc nanoclay loading. The decrease in impact strength at higher clay content levels is probably due to the formation of clay agglomerates and the presence of unexfoliated aggregates and voids. 14 –19 So, the composites with 3 phc nancolay had higher impact strength compared with those composites with 6 phc nanoclay. This may be attributed to a greater extent of intercalation and better dispersion of clay.
The influence of chemical modification of WF on the WA and TS of PP/WF/nanoclay hybrid composites is shown in Figures 5 and 6. As can be seen, the chemical modification reduced the WA and TS. The order of average WA and TS is untreated > alkali treatment > benzyl chloride treatment > acetic anhydride treatment. It is well known that with better adhesion and wettability between the matrix and fibers, the moisture diffusional processes could be much slower because there are fewer gaps in the interfacial region. 23 –25
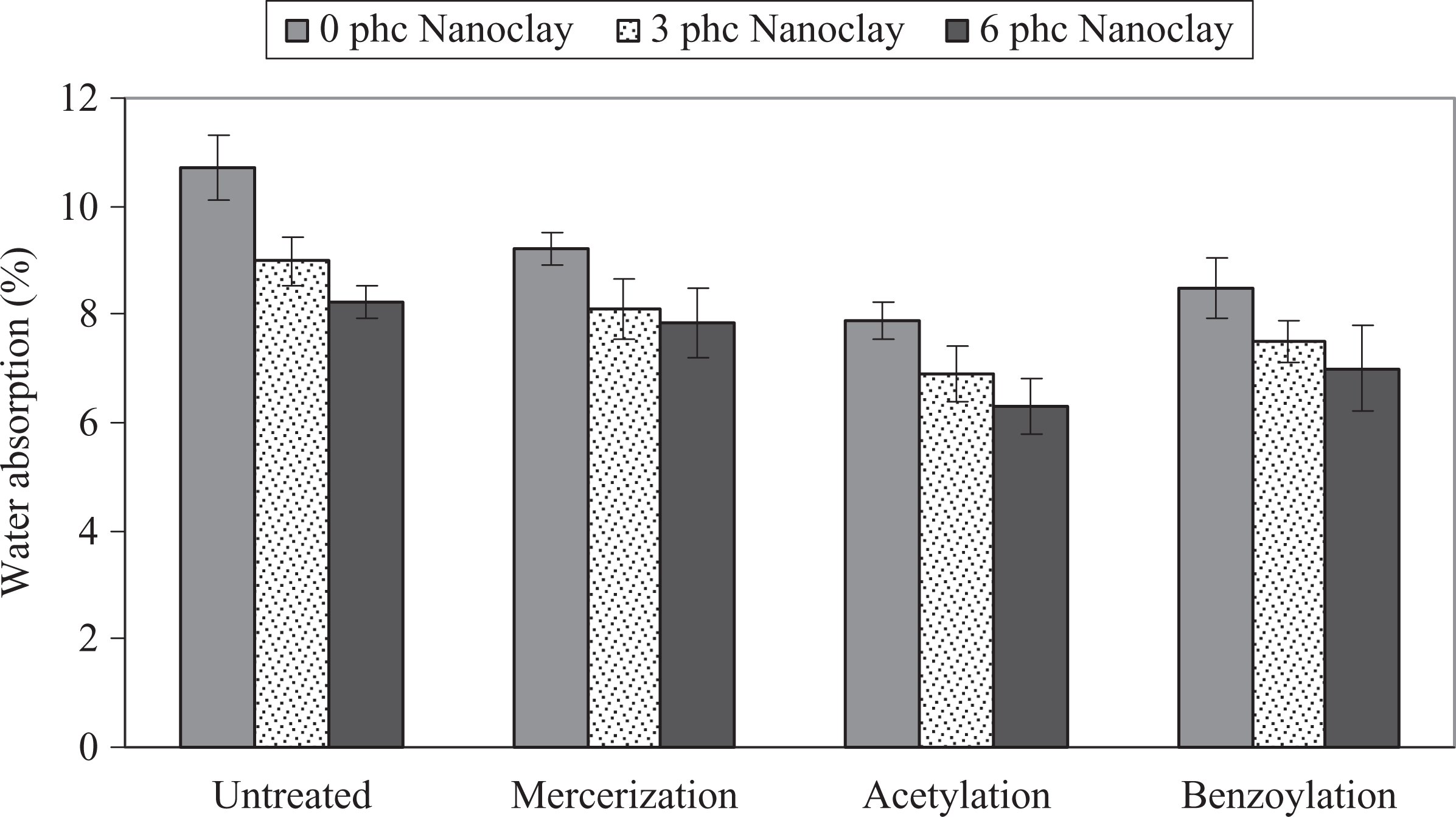
Influence of chemical modification of WF on the WA of PP/WF/nanoclay hybrid composites. PP: polypropylene; WA: water absorption; WF: wood flour.
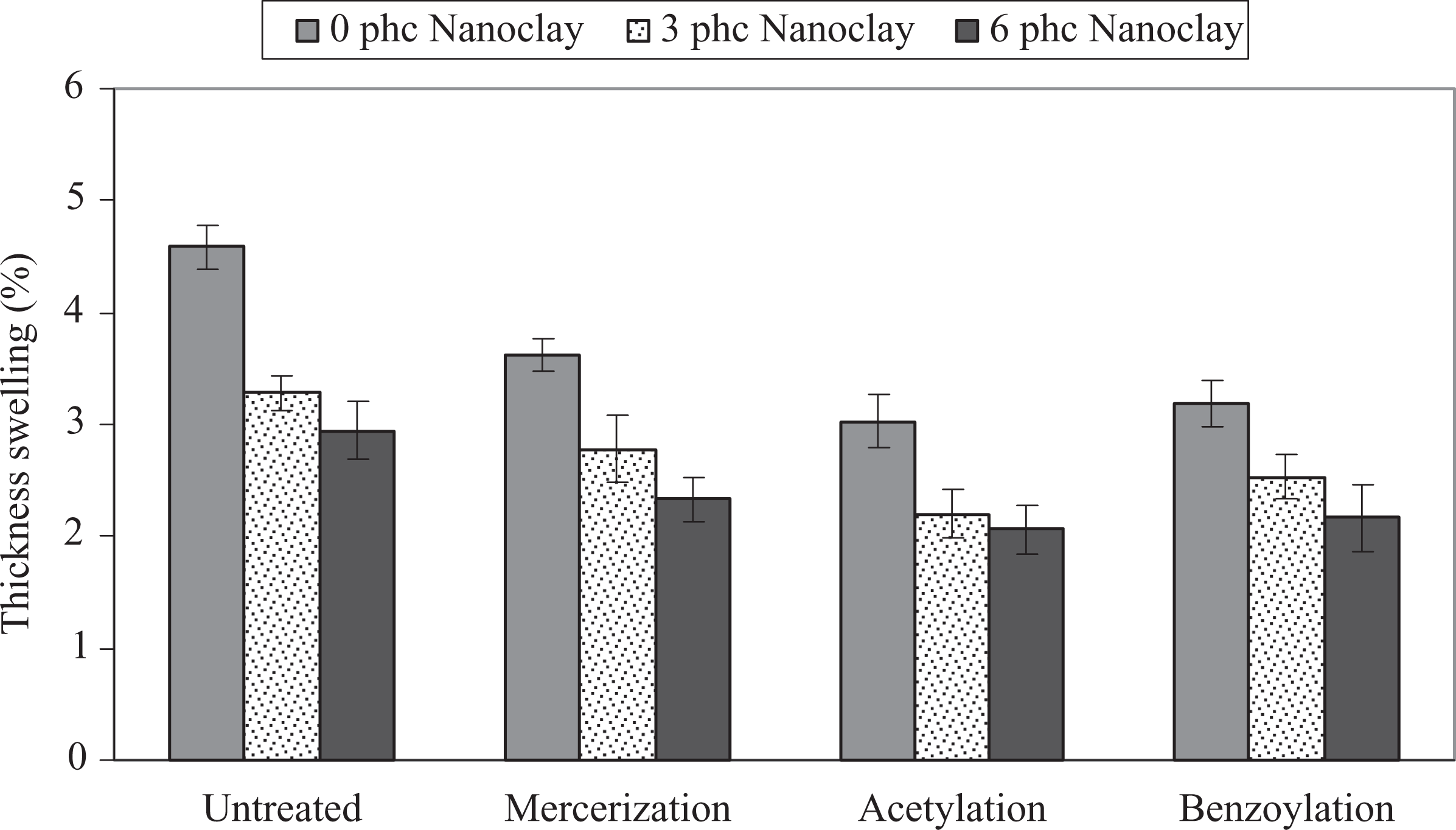
Influence of chemical modification of WF on the TS of PP/WF/nanoclay hybrid composites. PP: polypropylene; TS: thickness swelling; WF: wood flour.
Also as shown in Figures 5 and 6, the WA and TS decreased with increase of nanoclay loading. It seems that the barrier properties of nanoclay fillers inhibit the water permeation in the polymer matrix. Two mechanisms have been reported in order to account for this phenomenon. The first is based on the hydrophobic nature of the clay surface that tends to immobilize some of the moisture. 30 The second involves the ability of surfactant-covered clay platelets forming a tortuous path for water transport. 31 This barrier property hinders water from going into the inner part of the nanocomposite. It seems that both of the aforesaid mechanisms could be more efficient when the morphology is exfoliated. In other words, in an exfoliated state, there is more available surface area of organoclay (with hydrophilic nature) when using a surfactant (providing a more tortuous path), so the water transport goes down under severe conditions. Another reason for less WA and TS could be the change in crystallinity of WPCs by the effect of the nanoclay as a nucleating agent. 20,30,31
The X-ray scattering intensities for composites with different levels of nanoclay are demonstrated in Figure 7, where the quantity 2θ (equal to 4.76°) is related to neat clay with a basal spacing of 18.5 nm. In the sample with 3 phc concentration of nanoclay, the peak was shifted to a lower angle (2θ = 4.48Ω, d-spacing = 19.70 nm), which implies the formation of the intercalation morphology. The (001) peak of 6 phc nanoclay appeared at 2θ = 4.54°, with a d-spacing of 19.44 nm. These data show that the order of intercalation is higher for 3 phc of nanoclay than the 6 phc of nanoclay concentration.
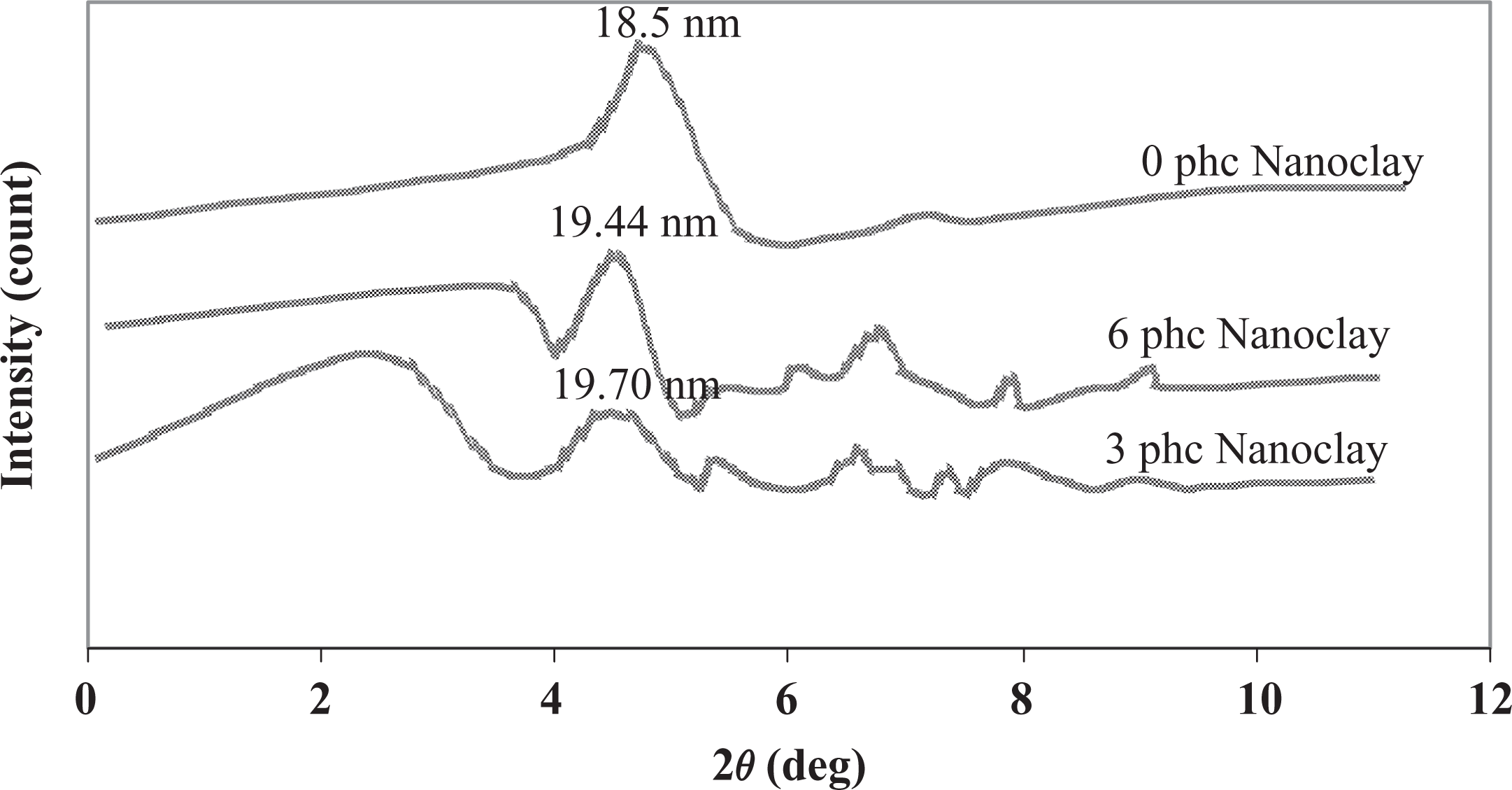
XRD patterns of composites with different levels of nanoclay. XRD: X-ray diffraction.
Figure 8 shows the dispersion state of nanoclay in the composites, as it was made evident by TEM. The dark line represents the intersection of the silicate layers, while the white background corresponds to PP matrix. When the loading level of nanoclay into the PP /WF composite was as low as 3 phc (Figure 8(a)), nanoclay exhibited better dispersion of the clay layers within the polymer matrix than at 6 phc of nanoclay content (Figure 8(b)). Increasing the level of nanoclay to 6 phc, the size of dispersed nanoclay became larger or even aggregated in part (as confirmed by decreased d-spacing from XRD in Figure 7).

Transmission electron micrographs of PP/WF/nanoclay composites: (a) 3 phc nanoclay and (b) 6 phc nanoclay. PP: polypropylene; WF: wood flour.
Conclusions
The following conclusions could be drawn from the results of the present study:
The tensile modulus, tensile strength, and impact strength of composites increased by applying chemical treatments.
Modified composites had lower WA and TS than unmodified ones.
FTIR spectra show that the intensity of O–H bond at 3444.2 cm–1 and formation of ester bond at 1741.4 cm–1 were indications of changes in the chemical structure of the fibers.
The tensile modulus and strength increased with increase of nanoclay up to 3 phc and then decreased. However, the impact strength and WA decreased with increased nanoclay loading.
The morphological findings showed that samples containing 3 phc of nanoclay had higher order of intercalation and better dispersion.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.