Abstract
Zinc oxide (ZnO) is a very important compound used in several industries. We synthesized ZnO with different particle sizes and it was incorporated in high-density polyethylene (HDPE) matrix. The resulting nanocomposites were characterized using thermal, physical, and morphological techniques. The mechanical properties of the nanocomposites were also assessed. The nanocomposites were studied for their ultraviolet (UV) absorption properties and were shown to have very good spectral as well as mechanical properties which make them a perfect candidate for applications where UV absorption is essential for example food packing, UV shields, and many other applications.
Introduction
Zinc oxide (ZnO) powder has long been utilized as anticorrosive coatings for various metals. They are especially effective for galvanized iron. ZnO paints, however, retain their flexibility and adherence on such surfaces for many years. Various plastics, such as polyethylene naphthalate (PEN), can be protected by applying ZnO coating. The coating reduces the diffusion of oxygen with PEN. ZnO layers can also be used on polycarbonate (PC) in outdoor applications. The coating protects PC from solar radiation and decreases the oxidation rate and photo-yellowing of PC. One of the most interesting properties of ZnO is its extraordinary absorption of light in the UV region.
Polyethylene (PE) as an example is a widely used material in outdoor applications, so its lifetime is determined by various environmental factors such as solar radiation, temperature, thermal cycling, humidity, weather, and pollutants but most importantly by ultraviolet (UV) radiation. 1 The most damaging region of wavelengths is between 280 nm and 315 nm. 2 The UV degradation is due to combined effects of photolysis and oxidative reactions. High-density polyethylene (HDPE) is the most crystalline form of the commercial PEs. Its reasonable cost and low energy demand for processing makes it ideal for many applications as packaging, bottles, films, pipes manufacture, and so on. Also HDPE distributes well-balanced mechanical properties making it ideal for industrial applications. Its highly crystalline structure, in comparison to other kinds of PEs, results in very high tensile strength. HDPE offers a good barrier for humidity and resistance to abrasion and corrosion, and it presents inertness to most of the chemicals. Additionally, it is light in weight and has low thermal conductivity.
PE is the simplest hydrocarbon polymer (–(CH2–CH2)n–) composed of a repeat unit of two carbon atoms, where n is the number of monomer units. HDPE is 90% crystalline and possesses density as high as 0.965 g cc−1. It has a melting point in the range of 130–150°C. 3 . Whenever high tensile strength and stiffness are required, HDPE finds better use because its nanocomposites have enhanced mechanical properties (tensile modulus and impact strength) compared with the neat polymer. 4,5
Polymer nanocomposites (PNs) consist of a polymeric material such as thermoplastics, thermosets, or elastomers and a reinforcing nanoscale material. Nanocomposites are a special class of materials having unique physical properties and wide application potential in diverse areas. 6 –9
During the past years, many works have been carried out on the elaboration of nanocomposites system by embedding of inorganic particles into polymeric matrix, 10 –16 affording a new class of polymeric materials which combine the properties of inorganic particles with the processability and flexibility of organic polymer matrix. Novel properties of nanocomposites can be obtained by successfully imparting the characteristics of parent constituents to a single material. These materials differ from both the pure polymers and the inorganic nanoparticles in some physical and chemical properties.
Encapsulation of inorganic nanoparticles inside the shell of polymers is the most popular and interesting approach to synthesize nanocomposites. Accordingly, a number of different metal and metal oxide particles have so far been encapsulated into the shell of polymers giving rise to a host of nanocomposites. 17 –20
Li et al. 21 prepared ZnO/epoxy nanocomposites with high-UV shielding efficiency. Optical properties of ZnO/epoxy nanocomposites, namely visible light transparency and UV light shielding efficiency, were studied using an ultraviolet–visible (UV-Vis) spectrophotometer. The optical properties of the obtained nanocomposites were shown to depend on ZnO particle size and content. The nanocomposite containing a very low content (0.07% in weight) of ZnO nanoparticles with an average particle size of 26.7 nm after calcination at 350°C possessed the most optimal optical properties. Hong et al. 22 synthesized Polymethyl methacrylate (PMMA)/poly styrene (PS)-grafted ZnO nanoparticles. The ZnO/PS nanocomposite was prepared by adding PMMA-grafted ZnO into styrene monomer, followed by radical grafting polymerization. The resulted nanocomposite was characterized by thermogravimetric (TG), differential scanning calorimetry (DSC), and X-ray diffraction (XRD) analyses, and the testing results indicated that thermal behaviors of PS were improved. Wacharawichanant et al. 23 investigated the effects of particle size of ZnO on mechanical, thermal, and morphological properties of pure polyoxymethylene (POM) and POM/ZnO nanocomposites. Peng et al. 24 prepared ZnO/glass fiber unsaturated polyester composites. On exposure to the metal halide lamp, their resistance to UV degradation was evaluated. Tang et al. 25 found from scanning electron microscopy (SEM) that ZnO nanoparticles can be homogeneously dispersed in the polyvinyl chloride (PVC) matrix. The absorbance spectrum of the nan composite polymer suggested that increasing the amount of nano-ZnO in composite particles could enhance the UV-shielding properties of the polymers. Similar conclusions were confirmed by other authors 26 and confirmed by SEM and transmission electron microscopic (TEM) analyses. Grigoriadou et al. 5 prepared different series of HDPE nanocomposites by melt mixing on, containing 2.5 wt% of different nanoparticles. Nanocomposites in the form of thin films were exposed to UV irradiation and constant relative humidity (50%) for several times. From tensile strength and Young’s modulus measurements, a high increase with initial UV irradiation times (till 100 h) and a slight reduction thereafter were determined. The increase was higher in nanocomposites compared with neat HDPE. Untreated nanoparticles have also a small influencing effect, while HDPE nanocomposites containing treated nanoparticles have the highest UV stability.
Several other studies focused on nanocomposites of ZnO with PVC, 27 poly (ethylene terephthalate), 28 low-density PE, 29 –31 and polypropylene 32 studying the optical and mechanical properties and also using molecular simulation 33 to study the effect of incorporating ZnO nanoparticles in polymer matrix. All results confirm that ZnO nanoparticles play an important role for UV shielding effect which is desirable for many applications. 34 –36 Hybrid inorganic–organic materials are promising systems for a variety of applications due to their extraordinary properties based on the combination of the different building blocks. The combination of nanoscale inorganic particles with organic polymers has a high potential for future applications and has therefore attracted a lot of attention during the last years. There are large numbers of methodologies to combine them in one material. This article is written with the intention to shed light on nanocomposites of ZnO nanoparticles and HDPE and illustrate the possibility of their usage in UV absorption applications. It focuses on the chemical aspects of the incorporation of inorganic nanoparticles into an organic polymeric matrix by melt mixing process forming nanocomposites with optimum mechanical properties and high UV-shielding efficiency and required for packaging materials.
Experimental
High density polyethylene
HDPE was supplied by oriental petrochemicals company (Egypt; in the form of homopolymer pellets HDPE-6070UA). It has density of 0.9 g cm−3, Standard Deviation of 0.06 and melt flow index of 8.1 g/10 min.
Preparation and modification of ZnO nanoparticles
ZnO nanoparticles were prepared by a precipitation method similar to that described by Wang et al. 37 and detailed in our publication. 38 The precipitation method makes possible the production of large quantities of ZnO nanoparticles in a reproducible way. Moreover, this is a relatively cheap synthesis method.
Preparation of ZnO/HDPE nanocomposite
The preparation of ZnO/PE nanocomposite was preceded by melt mixing, in an appropriate ratios, the molten HDPE pellets and treated nano-ZnO filler using a twin-screw extruder (Type: DSM Xplore) (model 600) with roller blades and a mixing head with a volumetric capacity of 69 cm3 at a rotor formulations speed of 30 r min−1. The melt-blended extrudates were then injection molded into plates with thickness of 4 mm. Different mix formulations are given in Table 1. Then different ZnO nanoparticles filler contents were used from sample Z 4 as given in Table 2.
Mix formulations of HDPE mixes with different particle size of treated ZnO nanoparticles.
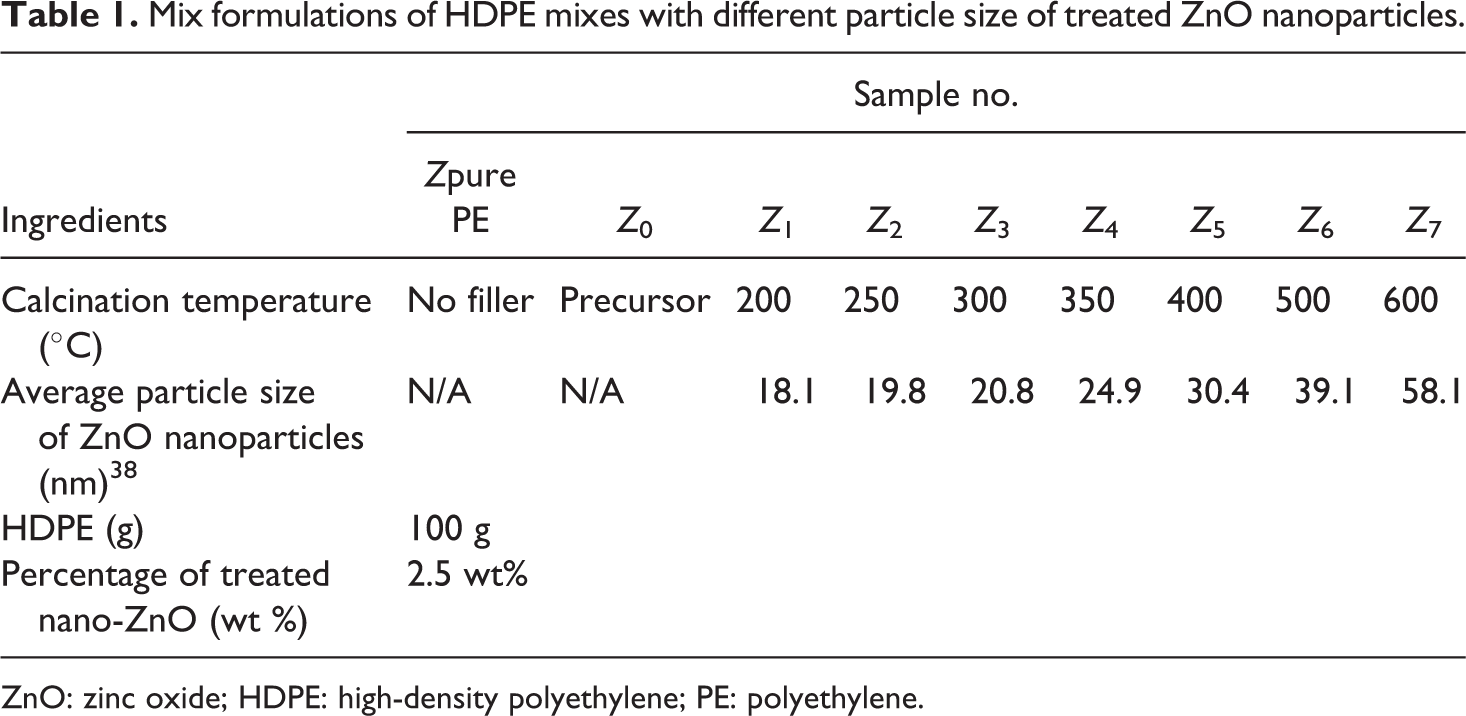
ZnO: zinc oxide; HDPE: high-density polyethylene; PE: polyethylene.
Mix formulations of HDPE mixes with treated ZnO nanoparticles of sample Z 4 with different filler contents.
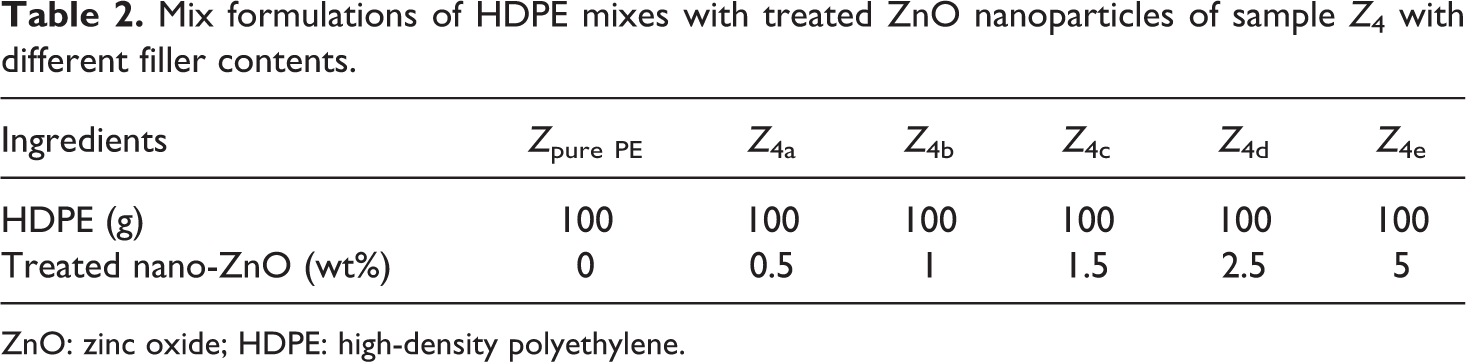
ZnO: zinc oxide; HDPE: high-density polyethylene.
Characterization of ZnO/HDPE nanocomposite
Scanning electron microscope
The morphologies of ZnO sample were studied using SEM (model XL 30; Philips, Germany) with an accelerating voltage of 30 kV and magnification 10× up to 400,000× and resolution of 3.5 nm. This analysis shows the change in the particle morphology after the calcination process. Also the fracture surface morphology of composites was studied. Prior to the measurement, the specimens were coated with gold.
Thermogravimetric analysis
TGA was performed on nano-ZnO and PE/ZnO nanocomposite on an analyzer (model TA-50, Shimadzu, Japan) by using 5 mg sample, at a heating rate of 10°C min−1. TGA was conducted with the compounds placed under high-quality nitrogen atmosphere in order to avoid unwanted oxidation.
DSC analysis
DSC analysis was performed on nano-ZnO and PE/ZnO nanocomposites on a DSC-50 (Shimadzu, Japan) under nitrogen atmosphere at a heating rate 10°C min−1 and sample weight was about 10 mg.
Physicomechanical properties
Sheets were cut out to five individual strips specimens by a steel die of constant width (0.4 cm). The thickness of the test specimen was determined by a dial gauge. A bench mark of 25 mm length was marked on the working part of each specimen under test. Mechanical tests including tensile strength, elongation at break, and elastic modulus were performed at room temperature (23 ± 2°C) using a tensile testing machine (model Z 010, Zwick, Germany) at a crosshead speed of 50 mm min−1, according to ASTM D-638 standards.
Tensile strength
It is defined as the applied force per unit area of the original cross-sectional area at the time of rupture of the specimen. Tensile strength is measured in mega pascal units (1MPa = 10 kg cm−2) and it is calculated according to the method described by Beecroft and Ober. 7
Ultimate elongation (elongation at break)
It is defined as the extension between two bench marks produced by tensile force applied to a specimen at the moment of rupture and is expressed as percentage of the original distance between the marks.
Hardness test
This test is based on measurement of the penetration of a specified type of indenter when forced into the specimen under specified conditions. The test was performed according to ASTM D 2240 standards using hardness tester (model 3150, Zwick, Germany). The samples used for this test shall be at least 6 mm in thickness, and the indenter must be hold at least 12 mm from any edge of the specimen. The average of five different measurements distributed over the specimen was taken.
UV-Vis spectrophotometry
UV-Vis spectrophotometry was carried out using a Shimadzu UV-2101PC instrument. Absorbance and transmittance spectra were determined in the 250–800 nm wavelength range. The specimens with thickness nearly 2 mm of the composites were measured.
Results and discussion
Effect of particle size of ZnO on thermal properties of nanocomposites
The thermal properties of pure HDPE and HDPE/Z 0-Z 7 nanocomposites with filler contents up to 2.5 wt% are shown in Figure 1(a) and (b). The thermal stability and onset of degradation for polymers are usually improved by the incorporation of nanoparticles. 39 TGA measurement results for HDPE/ZnO nanocomposites are shown in Figure 1(a). It is obvious that thermal decomposition for the different systems is all one-step process except that of pure HDPE. This might be due to the existence of two different forms in pure HDPE that degrades at two different temperatures as observed earlier. 40 It can be seen that the TGA curves for pure HDPE and the ZnO/HDPE nanocomposites are nearly overlapping each other and at the same onset of degradation, also the change in weight loss is nearly negligible. The thermal degradation curves under nitrogen flow did not show significant differences in behavior between the nanocomposites and blank HDPE. However, the slight increase of the onset degradation temperature (15–20°C) of nanocomposites compared to the blank HDPE sample is due to the increased stability of the polymer as a result of the incorporation of the nanoparticles. 39
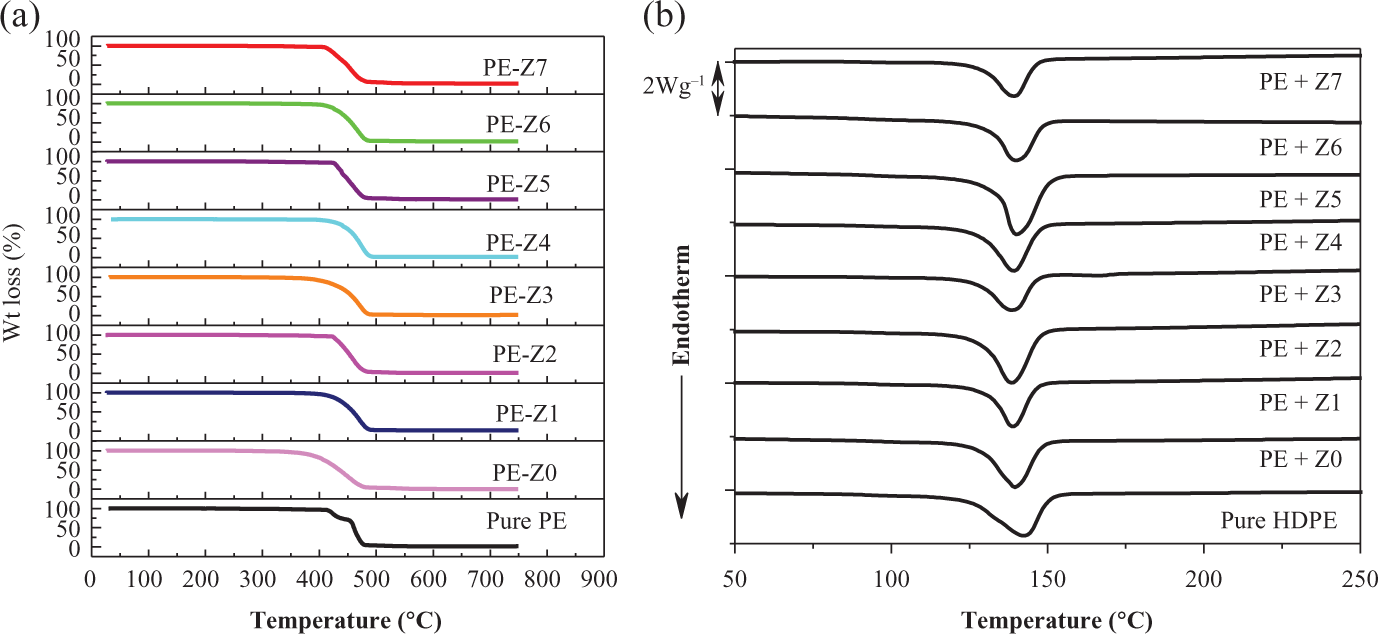
(a) TGA patterns of nanocomposites containing HDPE and ZnO nanoparticles calcinated at different temperatures. (b) DSC patterns of nanocomposites containing HDPE and ZnO nanoparticles calcinated at different temperatures. TGA: thermogravimetric analysis; HDPE: high-density polyethylene; ZnO: zinc oxide; DSC: differential scanning calorimetry.
The DSC curves of unfilled HDPE and HDPE/ZnO nanocomposites are shown in Figure 1(b). The peak nearly around 132°C corresponds to the melting temperature (T m) of HDPE. DSC data show that the melting behavior of HDPE was not affected by increasing size of ZnO nanoparticles. 41
The crystallinity values of the nanocomposites were determined by using equation (1) and values listed in Table (3).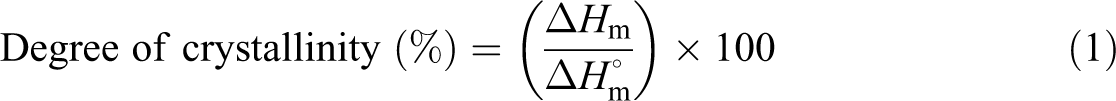
where
From Table 3, the crystallization of HDPE/ZnO nanocomposites was judged via the heat of fusion
Degree of crystallinity from DSC curves.
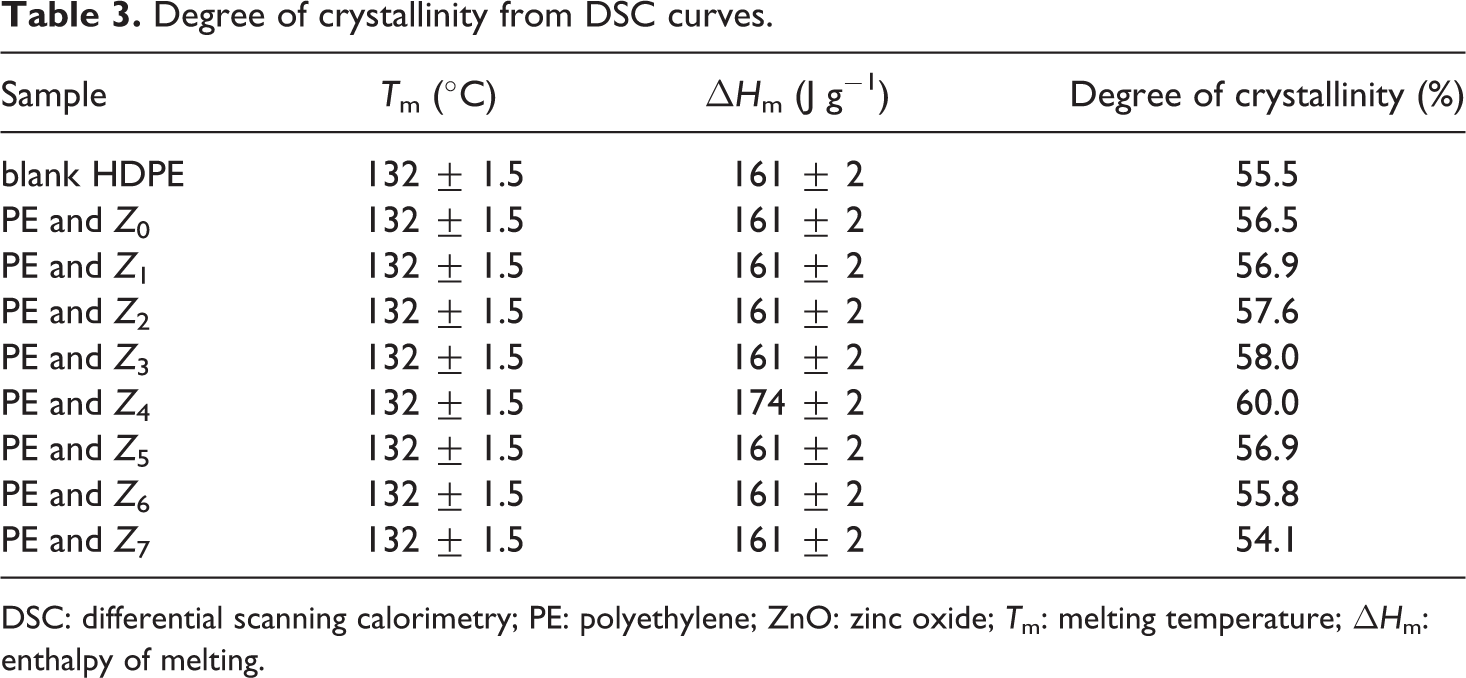
DSC: differential scanning calorimetry; PE: polyethylene; ZnO: zinc oxide; T m: melting temperature; ΔH m: enthalpy of melting.
Morphological properties of ZnO/HDPE nanocomposites
SEM analysis was used to study the morphology of fracture surfaces of impact specimens of the nanocomposites with different ZnO particle sizes as shown in Figure 2. SEM micrographs of nanocomposites containing 2.5 wt% of ZnO (different particle size Z 0, Z 4, and Z 7) show that they are homogeneously dispersed in the HDPE matrix with white spots of ZnO nanoparticles 26 and a few agglomerates for large particle size as in Figure 3(d).
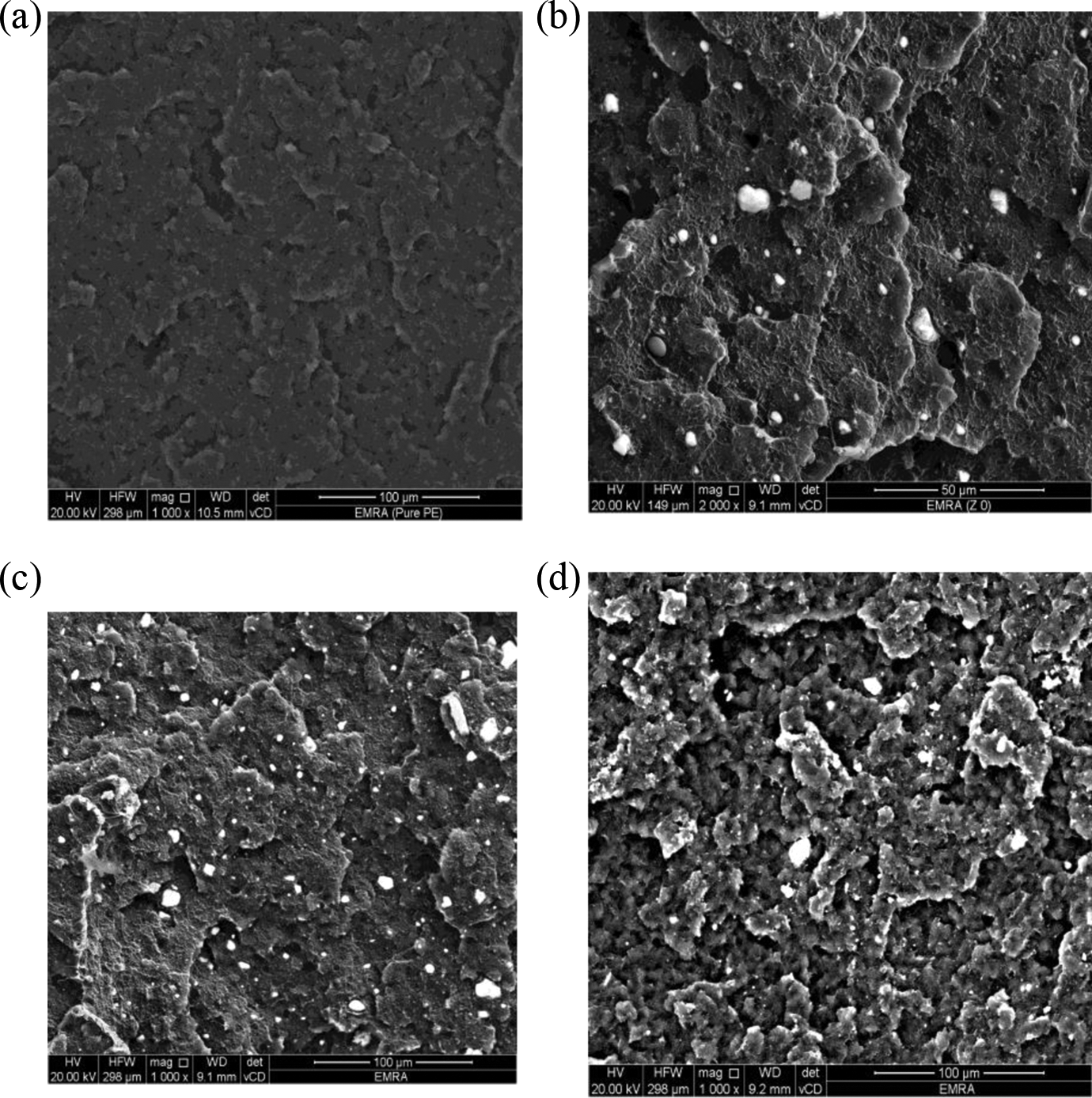
SEM morphology of nanocomposites at different ZnO calcination temperatures of (a) pure PE, (b) PE/Z 0, (c) PE/Z 4, and (d) PE/Z 7; all with concentration 2.5 wt% but with different ZnO particle size. SEM: scanning electron microscopy; ZnO: zinc oxide; PE: polyethylene.

SEM morphology for nanocomposites containing Z 4 and different ZnO concentrations: (a) pure HDPE, (b, c, d, e, f) HDPE/Z 4 with contents 0.5, 1, 1.5, 2.5, and 5 wt%, respectively. SEM: scanning electron microscopy; ZnO: zinc oxide; HDPE: high-density polyethylene.
It has been established that the nature of nanoparticle surfaces is a key factor to promote the dispersion of the nanoparticles in the matrix. Therefore, the presence of the organic surface treatment on the ZnO particles, which changes the surface properties from inorganic into organic, reduces the particle surface tension. This is essential to promote the uniform dispersion of the ZnO nanoparticles in the PE matrix. 32 From Figure 4(c) we can presume an internal particle homogenous distribution. This kind of particle dispersion in the polymer matrix indicates that the lower attraction of the particles, the enthalpy gain, caused by that the adsorption of polymer segments and the particle surface, is high enough to hinder aggregation of particles to a great extent. Actually, larger ZnO particles are less homogeneously dispersed in HDPE (Figure 3(d)) than smaller ones. This can be ascribed to the higher attraction between larger particles due to lower surface potential, causing an aggregation of ZnO nanoparticles in polymer matrix and this is not preferable for nanocomposites so sample Z 4 indicating the best particle size distribution.
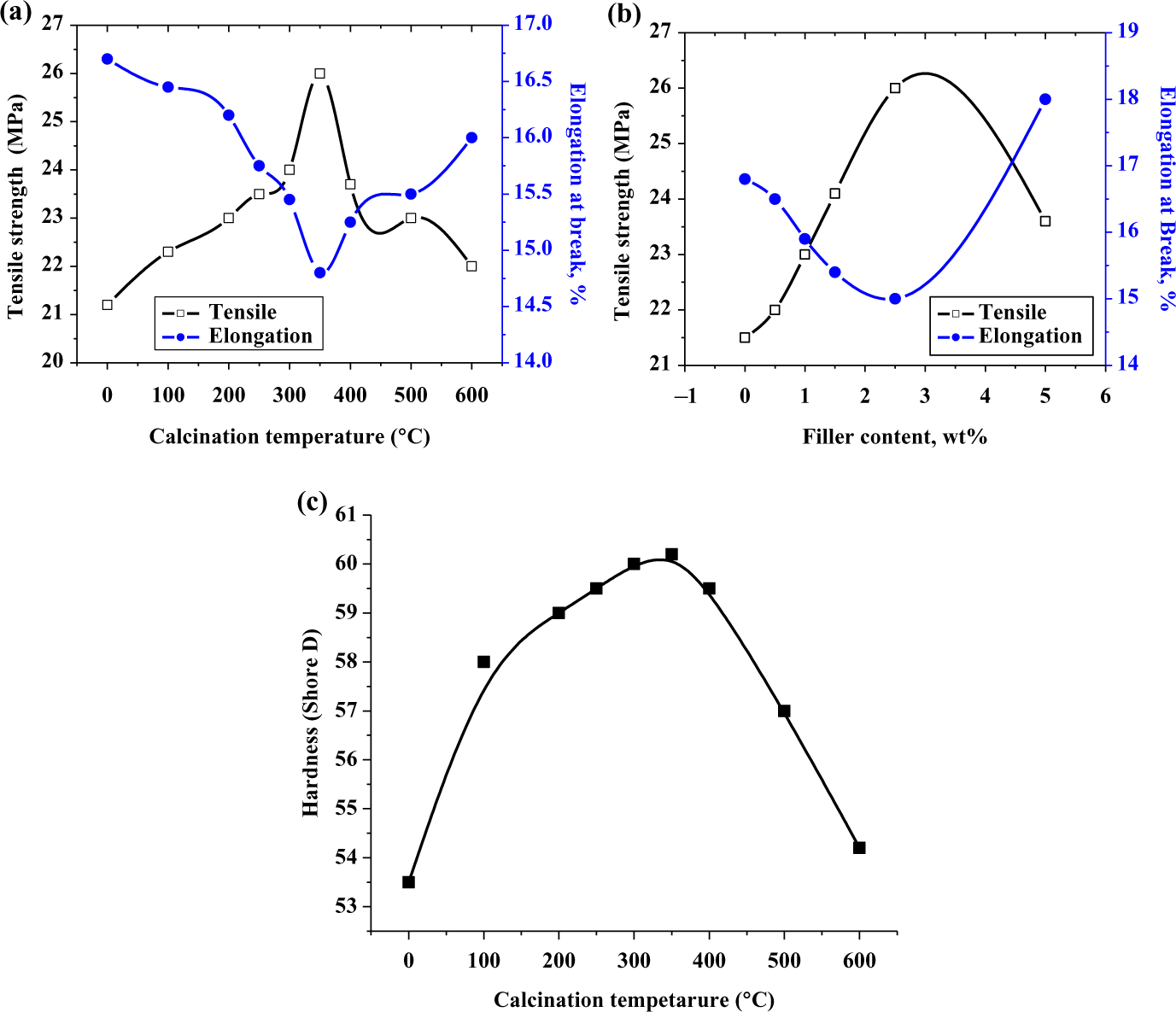
(a) The tensile strengths and elongation of the composites with different particle size of nano ZnO. (b) The tensile strengths and elongation of the composites made of different filler content of ZnO nanoparticles. (c) Hardness of the composites made of different particle size of ZnO nanoparticles. ZnO: zinc oxide.
The morphology of fracture surfaces of specimens of the nanocomposites containing different weight percentage of ZnO nanoparticles calcinated at 350°C was examined by SEM as shown in Figure 3. Figure 3(a) shows the micrographs of the surface of pure HDPE, while Figure 3(b) to (f) shows the micrographs of the fracture surfaces of PE/ZnO nanocomposites filled with 0.5, 1.0, 1.5, 2.5, and 5 wt% of ZnO nanoparticles, respectively. It is observed that the dispersion of ZnO on the polymer surface was relatively good for 2.5 wt% of ZnO but is nonuniformly dispersed forming local agglomeration of ZnO within the polymer for 5 wt% of ZnO, and the agglomeration of ZnO particles in the polymer matrix increased with extra increase in ZnO content. 42 This indicates that the optimum concentration of ZnO nanoparticles in the HDPE matrix that shows better dispersion due to better interaction between the polymer chains and ZnO nanoparticles is 2.5 wt% as supported by previous results. 5
Physicomechanical properties
Generally, the tensile properties of composites are markedly improved by adding fillers to a polymer matrix since fillers have much higher strength and stiffness values than those of the matrices. Also higher filler content is desired for the purpose of achieving high performance of filled polymer composites. The tensile strengths and elongation of 2.5 wt% ZnO nanoparticles/HDPE nanocomposites are shown in Figure 4(a) as a function of the particle size of ZnO nanoparticles.
It is observed that the presence of filler in the polymeric matrix raises the composite strength, and it can be seen that the tensile strengths of the composites increase while elongation decrease. 41 With increase in the calcination temperatures and increase in particle size of ZnO nanoparticles until critical particle size of sample containing ZnO is calcinated at 350°C (Z 4), the tensile strength of the composites slightly decrease. Elongation decrease due to the increase in the degree of crystallinity of ZnO with increasing particle size as estimated from XRD results 38 is due to the compatibility between HDPE and treated ZnO nanoparticles with uniform particle distribution but then the tensile strength decreases at higher particle size due to agglomeration of ZnO nanoparticles as shown from SEM graphs. This indicates that the proper particle size of nano-ZnO is Z 4 sample (ZnO which calcinated at 350°C) which gives maximum tensile strength.
The tensile strengths and elongation of the nanocomposites made of HDPE and different contents of ZnO nanoparticles are shown in Figure 4(b) as a function of the weight percentage of the ZnO nanoparticles. It is obvious from Figure 4(b) that the overall trend of the tensile strength increases with increasing ZnO nanoparticle content while elongation decreases. This result suggests that the interfacial interaction between ZnO nanoparticles and HDPE matrix is strong enough to delay the massive shear yielding along the tensile loading direction, 32,41 but as shown from SEM micrographs for ZnO content more than 2.5 wt%, the particles agglomerate resulting in reducing the interfacial interaction between the ZnO nanoparticles and HDPE matrix forming voids in the matrix thereby decreasing the tensile strength and increasing the elongation. Accordingly, the optimum concentration of ZnO nanoparticles in the HDPE matrix that shows better tensile strength and elongation due to better interaction between the polymer chains and ZnO nanoparticles is 2.5 wt%. Similar results are shown for the elastic modulus (not shown here); this suggests that nanocomposite Z 4 with particle size approximately 25 nm shows the best mechanical properties.
Hardness of nanocomposites
The hardness of a composite depends on the distribution of the filler into the matrix. Usually, the presence of a harder matrix causes the resultant composites to exhibit higher hardness. As shown from Figure 4(c), the variation in shore D hardness values for ZnO/HDPE nanocomposite increase gradually with increase the particle size of nano-ZnO. It can be concluded from the figure that the incorporation of nano-ZnO filler into the HDPE matrix reduced the flexibility of the polymer matrix resulting in more rigid composites. Hardness gradually increased with an increase in the particle size from Z 0 to Z 4 this due to better reinforcement and better interaction between ZnO and polymer matrix, until critical particle size of sample Z 4 then the hardness decrease from Z 5 to Z 7 as a result of agglomeration of filler in the matrix forming voids in the composites showing less hardness. This may be explained in terms of the homogeneity of dispersion between ZnO and HDPE; hardness decreases due to agglomeration of the particles that causes voids or weak points inside the composite. These results obtained were found to be similar with those for the tensile strength, that is, as the filler content increases, the hardness increases. Also, 2.5 wt% Z 4 gives the optimum hardness for HDPE/ZnO nanocomposite, indicating better reinforcement and better interaction between ZnO nanoparticles and HDPE matrix (results are not shown here).
UV-Vis spectrophotometry
One of the most interesting properties of ZnO is its extraordinary absorption of light in the UV region. 21 The UV-Vis absorbance and transmittance spectra of PE and nanocomposites (2.5 wt%) are shown in Figure 5(a) and (b). Figure 5(a) shows that UV light absorbance of the nanocomposites increases generally with increasing calcination temperature. This may be understood considering the fact that UV light absorption spectra consist of two contributions by ZnO, namely UV light absorption and light scattering. As the calcination temperature increases, the size of the ZnO nanoparticles increases, which is beneficial to UV absorption. Also, as light transmits in an inhomogeneous medium (like composites), the refractive index varies at different locations and light scattering will occur. 21 However, when calcinated at 400°C or above, particles grow and agglomerate meaning that the UV light scattering will play a larger role in shielding. Nanocomposites-containing ZnO nanoparticles calcinated at 400°C or above show high absorption of visible light which is not desirable for transparent packaging materials.
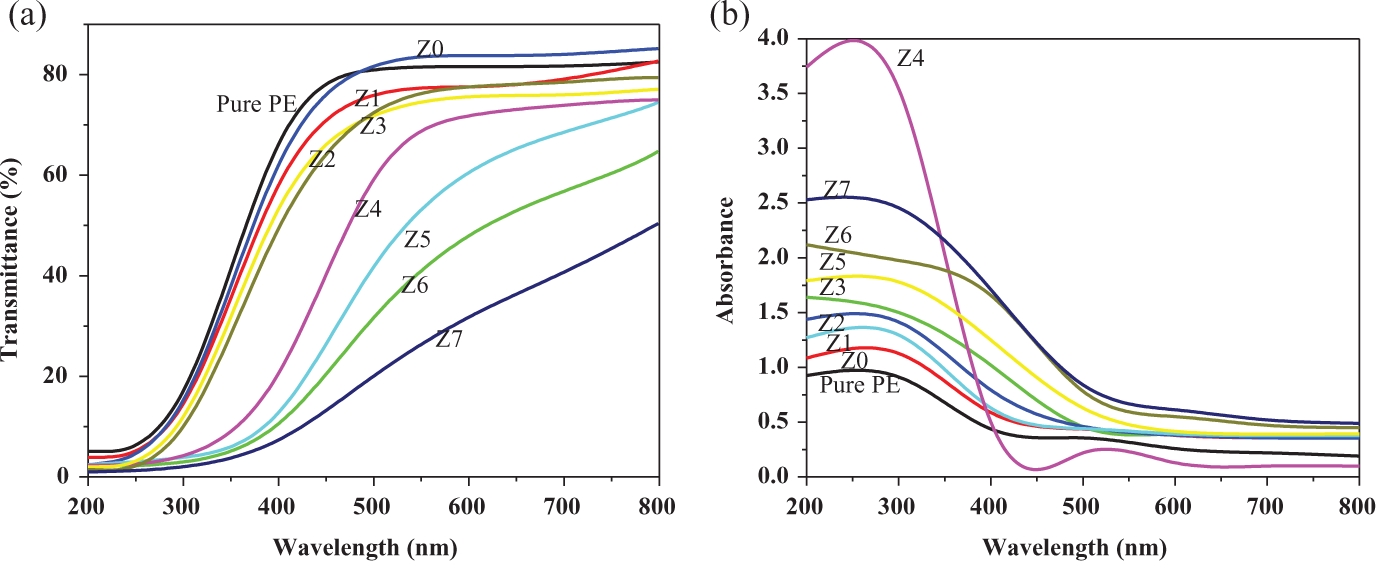
(a) UV-Vis Absorbance spectra of pure HDPE matrix and ZnO/HDPE nanocomposites with 2.5 wt% of ZnO nanoparticles. (b) UV-Vis transmittance spectra of pure PE and ZnO/HDPE nanocomposites containing 2.5 wt% ZnO nanoparticles. UV-Vis: ultraviolet–visible; ZnO: zinc oxide; HDPE: high-density polyethylene; PE: polyethylene.
However, the nanocomposite-containing nanoparticles with a mean particle size of approximately 25 nm and calcinated at 350°C exhibits high UV light absorption and very low absorption of visible light, hence high UV light shielding efficiency and high visible light transparency, both of which are preferred properties for transparent packaging materials.
The transmittance of UV light is in agreement with the absorbance results as shown in Figure 5(b). Although the samples calcinated at 400°C or above (Z 5-Z 7) exhibit almost high UV light-shielding efficiency, their transparencies are quite poor. It can also be seen from Figure 5(b) that the nanocomposite containing Z 4 nanoparticles calcinated at 350°C shows not only high UV light shielding but also high visible light transparency.
PE nanocomposites containing varying amount of ZnO nanoparticles calcinated at 350°C have also been studied for their UV–Vis absorbance and transmittance. The results showed that only the nanocomposite (Z 4c) containing 2.5 wt% ZnO nanoparticles corresponds to simultaneously high visible light transparency and high UV light-shielding efficiency. This is in agreement with the best mechanical and best dispersion properties of the nanocomposite suggesting the possibility of using this concentration in several applications requiring good mechanical properties and high UV shielding and visible light transmittance.
Conclusions
ZnO nanoparticles were chemically prepared in large quantities and with high purity suitable for use in semiindustrial and industrial processes as explained in our previous publication. 38 We are able to control the particle size of the synthesized ZnO in the nanometer range and incorporated the ZnO nanoparticles in HDPE. The mechanical, physical, and spectral properties of the nanocomposites were studied. Very good incorporation of the nanoparticles is observed for all calcinations temperatures especially at 350°C as observed by thermal and morphological analysis. Spectral analysis and UV-Vis measurements show that the nanocomposites can be used effectively and efficiently in UV absorption applications. Z 4 nanocomposites showed the best UV absorption properties and highest transparency in the visible region. Applications ranging from food packaging to transparent thin films in windshields are possible candidates for ZnO/HDPE nanocomposites.
Footnotes
Acknowledgment
Author EM expresses thanks to the Polymer Metrology and Technology Laboratory in the National Institute of Standards (NIS), Egypt, for continuous support and patience during the course of this work. Thanks are also due to Prof. I El-Sabbagh and Prof. M El-Fass for fruitful discussions during the course of this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded through the Academy of Scientific Research and Technology (ASRT), Egypt.
