Abstract
Recycled carbon fiber (RCF) was employed as a reinforcing material to prepare polyoxymethylene (POM)-based composites through a simple melting extrusion. An effective approach was developed to clean and modify the surface of the as-received RCF with nitric acid and then with a silane coupling agent. The mechanical evaluation demonstrated that a significant reinforcement was achieved for POM/RCF composites due to the improved interfacial adhesion between the fibers and the matrix. The thermal stabilities of the composites were also improved in the presence of RCF. The morphological observation of impact fracture surfaces indicated that the RCF gained a homogeneous dispersion in POM matrix due to good interfacial boding between fibers and matrix. The studies on nonisothermal and isothermal crystallization behaviors showed that RCF acted as a nucleation agent for the crystallization of POM domain in composites; therefore, the crystallization rate and nucleation density increased remarkably due to the heterogeneous nucleating effect of RCF. These crystallization features may be advantageous for the enhancement of mechanical performance and processability of POM-based composites.
Introduction
Carbon fiber-reinforced thermoplastic composites are a family of high-performance composite materials and can offer many desirable properties such as the high rigidity and high strength to weight ratio in comparison with metals, easy processability to fabricate any complex three-dimensional shapes, reduced coefficient of thermal expansion, low mold shrinkage, increased thermal conductivity, improved creep resistance, superior wear resistance, and outstanding impact toughness. 1 –4 Especially, considering of the reinforcement effect and the physical property retention of composites in humid environments, carbon fiber exhibits a great superiority over glass fiber or the other inorganic fibers. 5,6 Therefore, the development of carbon fiber-based composites has brought a great competitor to traditional organic–inorganic composites and has created many structural materials. 7 –10 Meanwhile, demand for carbon fiber-based composites continues to grow as a result of increasing applications in the areas of aerospace, military, motor vehicles, and sports appliance. The aircraft industry is an impressive example, with the new Boeing 787 and Airbus A350 having up to 50% of their weight in the carbon fiber-reinforced composites and military aircraft showing a similar trend. 11,12
According to statistics, the worldwide demand for carbon fiber reached approximately 35,000 tons in 2008, and this number is expected to double by 2014, suggesting a growth rate of over 12% per year. 13,14 Recently, the worldwide capacity growth of carbon fiber in thermoplastic composites has been limited by availability of carbon fiber due to its high cost resulting from the high-energy consumption in manufacture. 15
Moreover, there is a concern about the potential tonnage of waste from manufacturing processes and end-of-life products. The wastes related to carbon fiber products will quickly reach a significant level to become an important environmental issue since such products are not biodegradable. 16,17 In this case, the recycling of carbon fiber will contribute to the sustainable development of industrial processes. In recent years, there is an increasing interest in recycling and reuse of carbon fiber from waste composite materials. Several methods have been developed for carbon fiber recycling from the carbon fiber-reinforced thermoplastic and thermosetting wastes, and a recycling technology for these composite materials has been developed based upon a fluidized bed process. 18,19 Such a technology can provide the clean fibers with a high quality as well as a good mechanical retention. 20,21 In the meantime, a survey of literature also indicated that the recycled carbon fiber (RCF) could well serve reinforcement to most of the thermoplastics. 22 –24 McNally et al. 25 reported the preparation of polyethylene/RCF composites via melt compounding and found that both the Young’s modulus and ultimate tensile stress increased with increasing RCF content. Akonde et al. 26 investigated the reinforcing effect of RCF on polypropylene and found the average values obtained from tensile and flexural strength were 160 and 154 MPa, respectively, for composite specimens containing 27.7 vol% of RCF. Chen et al. 27 reported an effective reinforcement of RCF on poly(butylene terephthalate) (PBT) and also studied the effect of RCF on the isothermal crystallization kinetics of PBT. Yan et al. 28 developed a type of thermoplastic composite based on polycarbonate/acrylonitrile–butadiene–styrene copolymer alloys and RCF, and these composites exhibited considerably high mechanical strength and lightweight characteristics.
Moreover, Han et al. 29 –31 explored the possibility of reinforcement of RCF on a series of biodegradable thermoplastics like poly(lactic acid), poly(butylene succinate), and polyhydroxybutyrate. In summary, with a significant improvement in mechanical properties, the thermoplastic composites derived from RCF could be used as a class of high-performance and low-cost materials for many nonstructural applications.
Polyoxymethylene (POM) is an engineering semicrystalline thermoplastic with excellent surface lubrication, outstanding antifatigue performance, high electrical insulation, and good chemical and weathering resistance. 32 It can also be widely used in injection-molded, extruded, and blow molded parts and products for automotive, mechanical, and electronic applications. With a rapid growth of POM usage in the worldwide plastic marketplace, the development of high-performance POM-based composites and compounds has received a great attention. 33,34 In this work, we reported a study on mechanical enhancement and morphologies of POM/RCF composites, and the effect of RCF on crystallization behaviors of POM was also described in light of nonisothermal and isothermal process. Considering of the high cost of both raw carbon fiber and engineering thermoplastic POM, it is prospective that the recovery and reuse of RCF toward the POM reinforcement can offer sound economic benefits. This work aims to develop an application of RCF in reinforced POM composites via a simple melt compounding using twin-screw extruder.
Experimental
Materials
The POM resin used in this work is purchased from Asahi Kasei Chemicals Co. (Japan). It is a polyacetal copolymer (commercial grade Tenac–C 4520) with a number-average molecular weight of 25,000, melt flow index of 9.0 g/10 min, and specific gravity of 1.41. RCF was kindly supplied by Weiyuan Chemical Fiber Co. Ltd (China). The length (
Preparation of composites
The RCF were first immersed in a concentrated solution of nitric acid for 2 h to clean and activate the fiber surfaces. After washing to neutrality with deionized water, the activated RCF was further surface treated with a solution of γ-glycidoxypropyltrimethoxysilane in acetone at a concentration of 5 wt% for 4 h. Then, the RCF with the saturated absorption of coupling agent was baked in a vacuum at 90°C for 2 h.
The raw POM resin was dried at 80°C in a vacuum oven for 12 h to ensure that the moisture content is sufficiently low to prevent viscosity degradation. The POM/RCF composites with various weight percentage of surface-treated RCF were blended through melt extrusion using a corotating twin-screw extruder (
Characterization
FTIR spectroscopy
The Fourier transform infrared (FTIR) spectra of RCF samples were obtained using a Bruker Tensor-27 FTIR spectrometer with a scanning number of 48. A finely ground, approximately 1% mixture of a solid specimen in potassium bromide powders was fused into a transparent disk for FTIR measurement using a hydraulic press.
Measurement of mechanical properties
All of the pelletized samples were dried at 80°C in a vacuum oven for 8 h prior to injection molding and then were injection molded into the test bars with the different shapes required for mechanical and heat-resistant measurements. The tensile and flexible properties were measured with a CMT–4104 universal testing instrument (SANS Group, China) using a 10,000-N load transducer according to ISO 527 and ISO 178 standards, respectively.
Notched Izod impact strength was measured with a SANS ZBC–1400A impact tester according to ISO 180 standard. The impact test bars were notched with a depth of 2 ± 0.2 mm, and the impact energy was set to 2.75 J. All the measurements were performed at a constant temperature of 23°C, and the reported values reflected an average from five tests.
Scanning electron microscopy
Scanning electron microscopic (SEM) images for the morphologies of RCF surface and fractography of POM/RCF composites were taken on a Hitachi H–4700 scanning electron microscope. (Japan) The fracture surfaces obtained from the impact test bars after impact measurement were made electrically conductive by sputter coating with a thin layer of gold–palladium alloy. The images were taken in high-vacuum mode with 20 kV acceleration voltage and a medium spot size.
Differential scanning calorimetry
The crystallization kinetics of POM/RCF composites was studied in terms of both nonisothermal and isothermal crystallization behaviors using a Q20 differential scanning calorimeter (TA Instruments, New Castle, Delaware, USA) equipped with a Universal Analysis 2000 data station. All operations were performed under nitrogen flow of 50 mL min−1 with a sample weight around 5–7 mg. All the samples were first heated to 180°C and held at this temperature for 5 min to eliminate the effect of the thermal and processing history. For nonisothermal crystallization, the samples were heated to 180°C at a rate of 20°C min−1, and then, they were cooled at a given scanning rate to 40°C to observe the crystallization, which is marked as a cycle. In this study, four cycles were carried out with the cooling rates 5, 10, 15, and 20°C min−1. Isothermal crystallization was observed with the procedures where the samples were heated to 180°C at a rate of 10°C min−1 and rapidly cooled to a desired crystallization temperature, and then, they were kept at the same temperature until the crystallization finished. The crystallization peak and melting temperatures were directly read from the cooling and heating thermograms, respectively. The curve integral was run to calculate the enthalpies of overall crystallization behavior.
Polarized optical microscopy
Polarized optical microscopy was performed to observe the crystalline morphology of POM and its composites on an Olympus BX51 (Japan) polarizing microscope equipped with a Linkam THMS 600 temperature controller and a Sony CCD–IRIS digital camera. The samples were heated to 260°C on the hot stage, held at this temperature for 3 min, and then cooled to a temperature of 198°C, where the growing of spherulites started. The samples were held at this temperature for 1 h so as to perform the isothermal crystallization.
Dynamic mechanical analysis
The dynamic mechanical behaviors of POM and its composites were measured on a TA Q800 dynamic mechanical analyzer under a dual cantilever mode. The test was run during the temperature range from –80°C to 60°C with a heating rate of 5°C min−1 and a strain amplitude of 10 μm at a frequency of 1 Hz.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was performed under nitrogen atmosphere using a TA Instrument Q50 thermal gravimetric analyzer.
Samples were placed in a platinum crucible and ramped from room temperature to 750°C at a heating rate of 10°C min−1 while the flow of nitrogen was maintained at 50 mL min−1.
Results and discussion
Surface treatment of RCF
The surface morphologies of the as-received RCF and the surface-treated one were observed by SEM. Figure 1 shows the SEM micrographs of these local specimen surfaces. As shown by Figure 1(a), some small knurls are distinctly observed on the surface of the as-received RCF. These small knurls may be attributed to the carbonaceous deposition as a result of thermo-oxidative decomposition during the recycling process of carbon fiber. The presence of these carbonaceous deposits evidently spoils the interfacial adhesion between fibers and new resin and, thus, reduces the capability of interfacial bonding of RCF with polymeric matrix. Therefore, it is essential to clean and modify the surface of the as-received RCF in order to achieve a good reinforcement effect.

SEM micrographs of (a) as-received RCF and (b) surface-treated RCF. SEM: scanning electron microscopic; RCF: recycled carbon fiber.
As a feasible pathway, the oxidation of nitric acid toward the as-received RCF can not only clean the RCF but can also activate its surface. 35,36 Furthermore, considering the inert and smooth RCF surface, the chemical treatment toward the surface is also necessary to enhance the interfacial adhesion between fibers and POM. In this work, a silane coupling agent was used to treat the RCF surface in terms of the aforementioned method described in experimental section. Figure 1(b) depicts a clean and smooth surface of RCF after cleaning with concentrated nitric acid and then treated with the coupling agent. The fiber surface shows no carbonaceous deposits and no visible flaws, indicating a well recovery from the as-received RCF. The FTIR spectrum also confirms the surface-treating effect on the as-received RCF as presented in Figure 2. A absorption peak is observed at 1627 cm−1 on the spectrum of the RCF etched by nitric acid, which is attributed to the C=O stretching vibration of carboxyl group. Meanwhile, another broad absorption band at 3435 cm−1 is assigned to the presence of hydroxyl group. This indicates that the as-received RCF was activated. As for the FTIR spectrum of the RCF treated with silane coupling agent, besides hydroxyl absorption band at 3427 cm−1, the several new bands at 2937, 2871, and 1105 cm−1 are found to represent the –CH3, –CH2–, and Si–O stretching vibrations. In addition, an important feature of the spectrum, three characteristic bands corresponding to oxiranic C–O–C stretching vibration are observed at 1199, 907, and 850 cm−1. These characteristic bands confirm the presence of the silane coupling agent on the RCF surface.
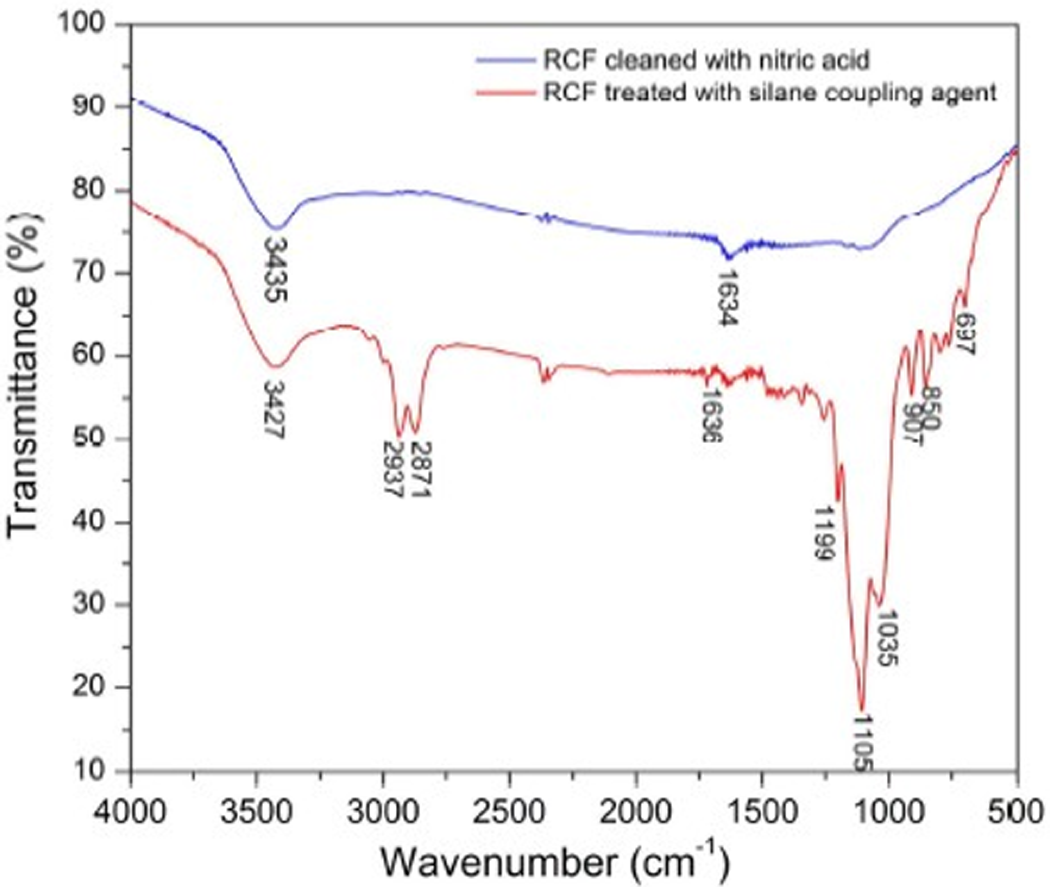
FTIR spectra of the as-received RCF and surface-treated RCF. FTIR: Fourier transform infrared; RCF: recycled carbon fiber.
Mechanical properties
The enhancement effect of RCF on POM was evaluated by tensile and flexural experiments, and the obtained results are presented in Figures 3 and 4. As was anticipated, all of the mechanical parameters achieved a remarkable improvement with incorporating RCF. The tensile and flexural strength is found to increase by 76% and 72%, respectively, with addition of only 5 wt% RCF. Meanwhile, the tensile and flexural moduli also present an increment by 78% and 161%, respectively. Such an increasing trend was well maintained with an increase of RCF content. In this case, the incorporation of 20 wt% RCF into POM led to an improvement by 180% in tensile strength, while the composite achieved the 310% increment in flexural strength compared to pure POM. Nevertheless, the composite-containing 20 wt% RCF shows an increase in tensile and flexural moduli by 240% and 460%, respectively, indicating a more remarkably increasing trend in these two mechanical parameters for POM/RCF composites. These mechanical results confirm a significant reinforcing effect of RCF on POM. Such an enhancement in mechanical strength of POM/RCF composites is mainly attributed to the improvement in interfacial shear strength between fibers and polymer matrix. 37 The excellent interfacial adhesion between two phases as well as the high strength and modulus of RCF may synergistically allow an effective stress transfer from fibers to matrix and, thus, can enable the composites to withstand more test stress.
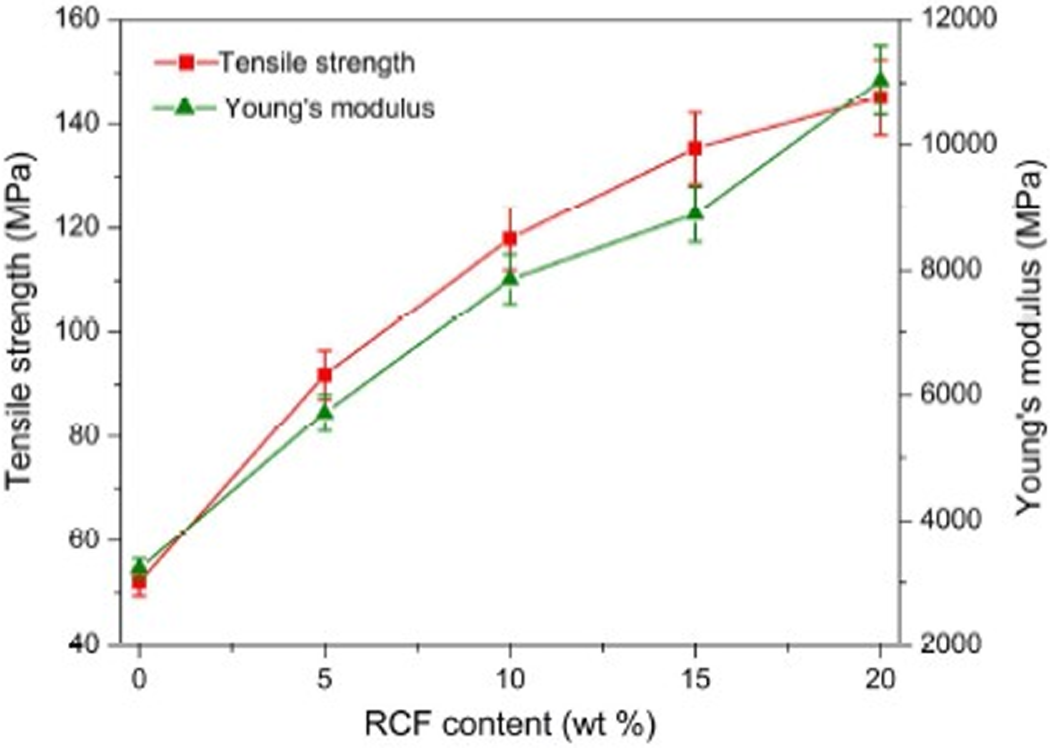
Tensile strength and modulus of POM/RCF composites as a function of RCF content. POM: Polyoxymethylene; RCF: recycled carbon fiber.
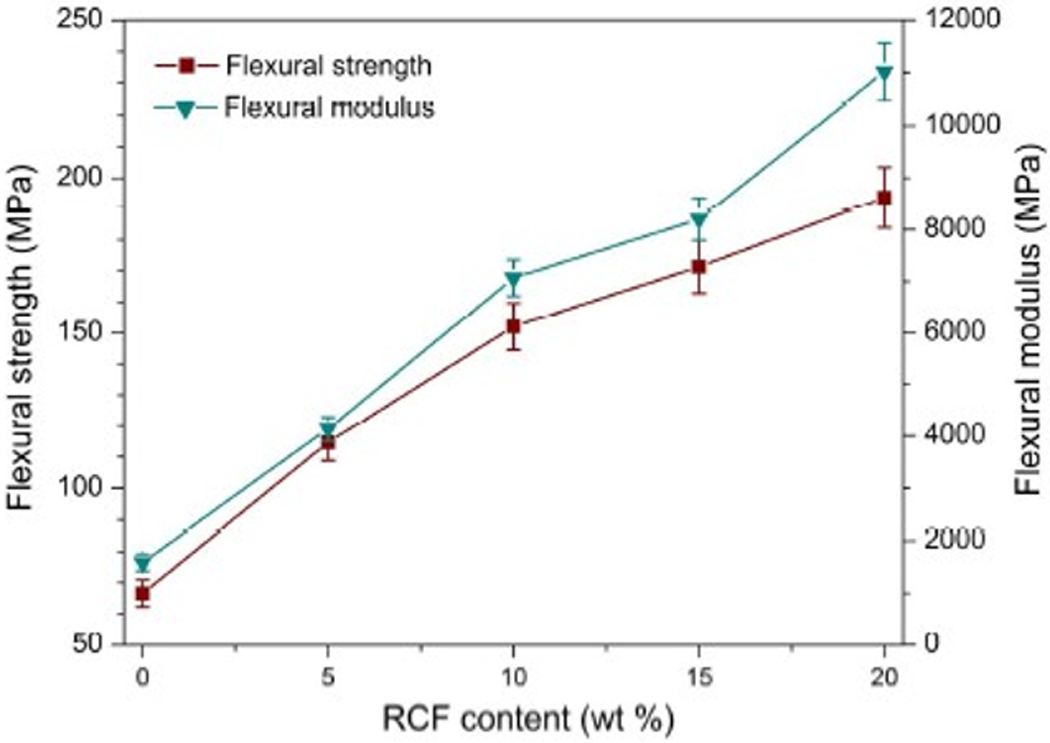
Flexible strength and modulus of POM/RCF composites as a function of RCF content. POM: Polyoxymethylene; RCF: recycled carbon fiber.
The effect of RCF on impact toughness of POM was also investigated according to the notched Izod impact tests. From the test results shown in Figure 5, it is interesting to note that the POM/RCF composites exhibit a slight increase in notched Izod impact strength with incorporating over 10 wt% of RCF, though the introduction of 5 wt% RCF results in a deterioration of impact toughness. However, the impact strength is found to reach a maximum value only at a level of 10 wt% RCF. The toughness enhancement of POM composites with respect to RCF content may be attributed to the fracture-transformation mechanism. It was reported that the impact toughness was generally dominated by two factors for most of the fiber-reinforced thermoplastic composites. 38 The first one is that new stress concentrations are formed around the fiber ends, area of poor adhesion, and region of fiber aggregation. The second one is that the fibers can enhance the impact resistance through reducing the crack propagation rate by forcing cracks around the fibers. The practical effects of fibers on impact toughness of composites depend on the competition of these two factors. In the case of the POM/RCF system, the reducing effect of RCF on crack propagation rate of composites sometimes dominates the crack initiation through forming new stress concentration, and the fibers can restrict the crack propagation rate so as to alleviate the fracture of the composites. The energy required to produce a new fracture surface increases with fiber volume fraction leading to an increase in impact strength. Moreover, the fiber pull out and fracture, interfacial debonding, and matrix deformation also contribute to the improvement in impact strength of POM/RCF composites. However, the aggregation of fibers may occur for the composites with high content of RCF and, thus, leads to locally destroy the continuity of matrix. This directly leads to a decrease of crack propagation capability and a severe matrix deformation. Furthermore, the agglomeration of RCF also causes the stress concentration in polymer matrix and prevents efficient load transfer to the matrix. The investigation on fractography will supply an evidence of microscopic images in the latter section.
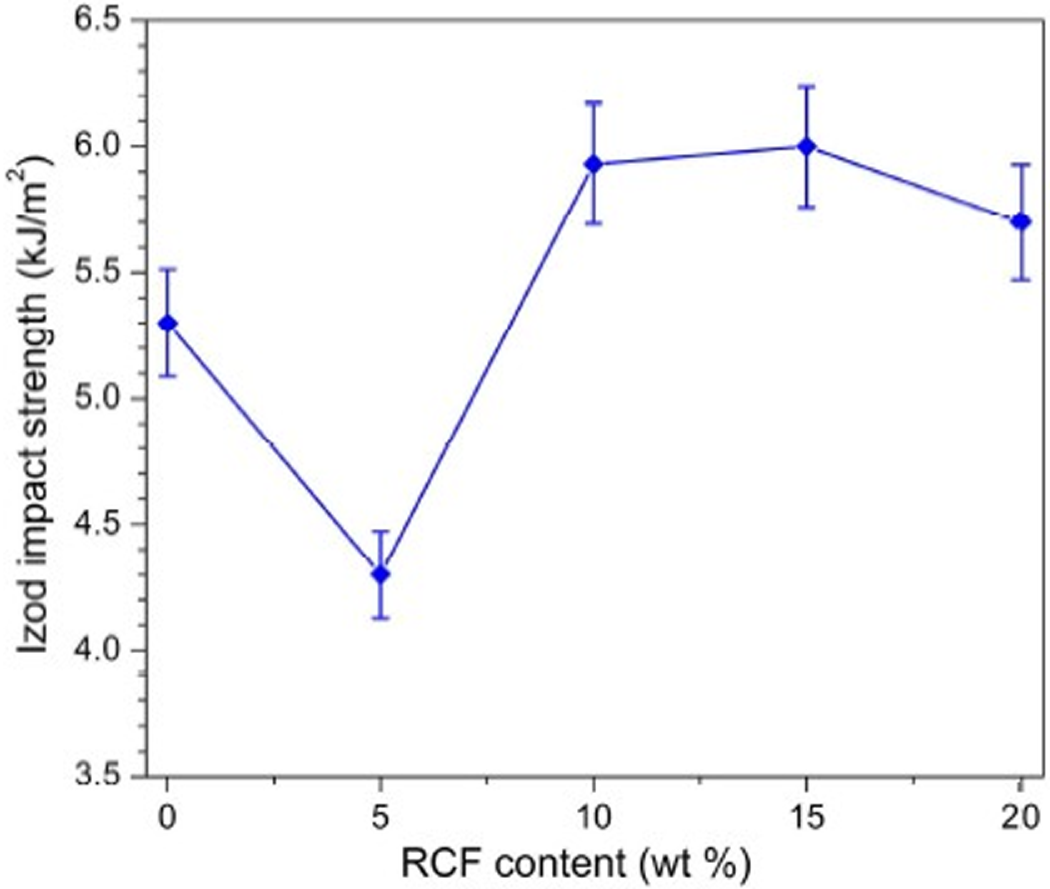
Notched Izod impact strength of POM/RCF composites as a function of RCF content. POM: Polyoxymethylene; RCF: recycled carbon fiber.
Morphology of fracture surface
The impact fracture surfaces of POM and its composites with RCF were observed with SEM, and the relevant micrographs are demonstrated in Figure 6. The pure POM presents a brittle fracture behavior due to its slightly deformed fracture surface as shown in Figure 6(a). Meanwhile, it is found from Figure 6(b) to (f) that the impact fracture surfaces of POM/RCF composites exhibit a feature of major fiber breakage with a few fibers pulled out.

SEM micrographs of impact fracture surfaces for (a) pure POM and its composites containing (b) 5 wt%, (c,d) 10 wt%, (e) 15 wt%, and (f) 20 wt% of RCF. SEM: scanning electron microscopic; POM: polyoxymethylene; RCF: recycled carbon fiber.
The fibers are homogeneously distributed in the matrix (see Figure 6(c)) and also maintain a considerable length. Even if 20 wt% of RCF is incorporated into POM, these long fibers are still imbedded in the matrix when debonding occurs. Furthermore, it is noted from Figure 7(a) that a few fibers were clung with some POM resin, indicating a good interfacial adhesion between the matrix and RCF. These phenomena suggest that a mechanical interlocking has been established between the fibers and the matrix, and thus, better stress transfer can be gained. However, from a magnified SEM micrograph of the composite containing 20 wt% of RCF shown by Figure 7(b), it is distinguished that some fibers aggregated due to the high loading of RCF in the matrix. This result is consistent with the impact text data. On the basis of the SEM observation, it can be confirmed that the surface treatment of RCF with the silane coupling agent efficiently enhanced the interface adhesion and compatibility between RCF and POM matrix. Such an improved interfacial adhesion leads to a significant increase in mechanical properties. Moreover, it is noteworthy from the SEM micrographs of Figure 6 that most of the fibers on fracture surfaces were orientated in the flowing direction of injection molding, indicating a higher degree of fiber orientation for the composites. This can induce a higher fiber efficiency factor and, hence, results in better reinforcement effect. Additionally, the impact energy was efficiently dissipated via fiber pulling out, interface debonding, and matrix deforming.

SEM micrographs as a close-up view for the impact fracture surface of POM composites containing (a) 10 wt% and (b) 20 wt% of RCF. SEM: scanning electron microscopic; POM: polyoxymethylene; RCF: recycled carbon fiber.
Dynamic mechanical property
Dynamic mechanical analysis (DMA) was performed to evaluate the dynamic mechanical properties of POM and its composites with RCF, and the temperature dependences of storage modulus and loss factor (tan
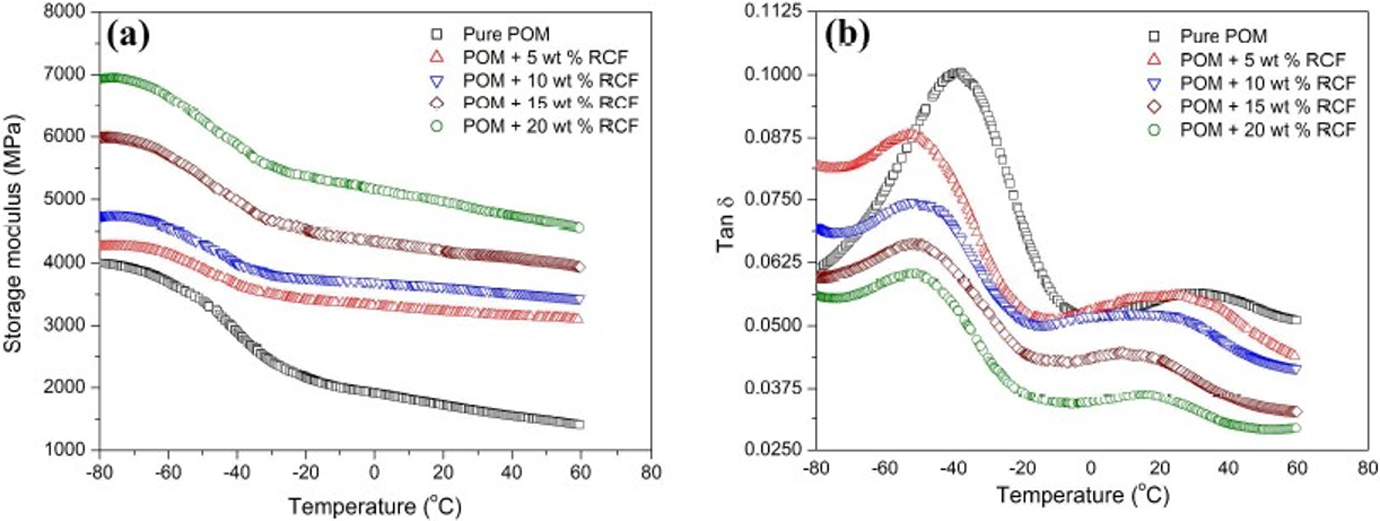
Temperature dependence of (a) storage modulus and (b) loss factor (tan
Thermogravimetric analysis
TGA was performed to investigate the thermal stabilities of POM and its composites with RCF. Figure 9 demonstrates the obtained TGA thermograms, which reflect the thermal degradation behaviors of POM/RCF composites under nitrogen atmosphere. These mass loss profiles indicate that both POM and its composites with RCF underwent a typical one-step degradation during pyrolysis, which may be ascribed to the chain scission of the POM backbone as the prevailing decomposition reaction. As shown in Figure 9, pure POM is found to encounter an initial decomposition with 3 wt% of mass loss at a temperature of approximately 327°C and then a rapid decomposition with a maximum weight loss at 383°C due to the random scission of C–O–C bonds in POM molecular chains. Such a thermally induced chain rupture can result in a complete unzipping of the damaged chain and, thus, almost leaves no residual char. As for the POM/RCF composites, the presence of RCF seems not to influence the thermal degradation behavior of POM significantly. However, it should be noted that the characteristic temperatures at the maximum mass loss rate were considerably improved with increasing the RCF content. It was reported that the incorporation of carbon materials in polymeric matrix could reduce the heat release rate, which plays a key role in retarding the decomposition temperature. 38
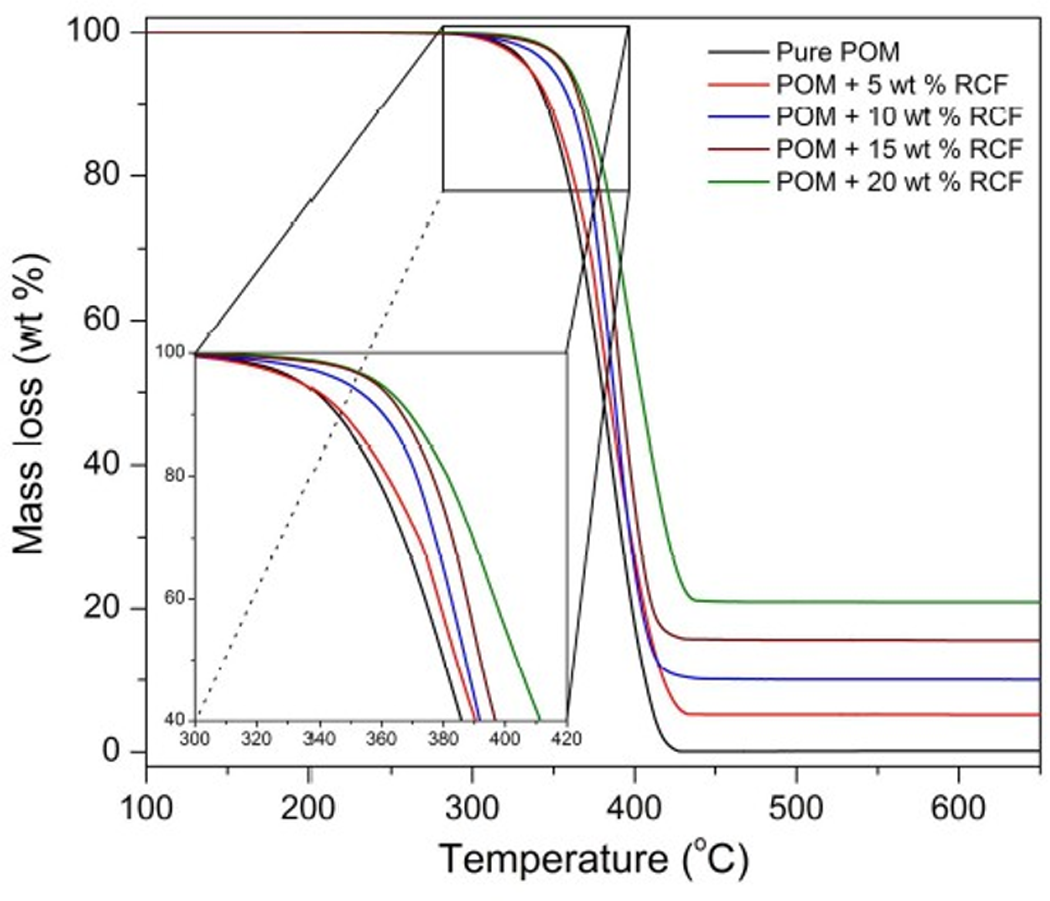
TGA thermograms of pure POM and its composites with RCF. TGA: thermogravimetric analysis; POM: polyoxymethylene; RCF: recycled carbon fiber.
Moreover, the RCF at a high loading level can effectively act as physical barriers to hinder the transport of volatile decomposed products out of POM composites during thermal decomposition. Therefore, the POM composite containing 20 wt% of RCF presents a much higher decomposition temperature than the other composites at the same percentage of mass loss. Additionally, the char yields of POM/RCF composites are found to slightly exceed the weight percentage of RCF in composites. This phenomenon is ascribed to the flame retardancy and carbonization of RCF toward the POM matrix. 39 Similarly, the RCF also has a good barrier effect on the thermal degradation process, resulting in the retardation of weight loss of thermal degradation products as well as the thermal insulation of POM matrix. As a result, much higher char yields were achieved at the end of the thermal decomposition for the composites.
Conclusions
RCF was surface treated with the silane coupling agent and, then, was used to prepared the POM-based composites through a simple melting extrusion. A significant reinforcement effect was achieved due to the enhancement of interfacial adhesion between the fibers and the POM matrix as a result of the surface treatment for the as-received RCF, thus leading to a remarkable improvement in mechanical properties and thermal stability. The SEM observation confirmed a homogeneous dispersion of RCF in the matrix as well as a good interfacial interaction between the fibers and the matrix. The incorporation of RCF into POM also caused an increase in storage moduli but a decrease in loss factors. The crystallization behaviors and kinetics of POM/RCF composites were intensively studied under both nonisothermal and isothermal conditions. On the basis of the results derived from the crystallization kinetic investigation, it was deduced that the RCF acted as a nucleation agent for the crystallization of POM domain. The crystallization rate and the nucleation density of POM domain in composites were improved remarkably as a result of the heterogeneous nucleation effect of RCF.
The presence of RCF enhanced the nucleation capability and crystallinity of POM domain. These crystallization features may be advantageous for the enhancement of the mechanical properties, heat resistance, and processability of POM-based composites. It is prospective that, owing to the use of recycled reinforcing materials as well as having good mechanical performance, the POM/RCF composites show a potential application in industrial and civil fields as low-cost and sustainable engineering materials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
