Abstract
In this article, an attempt is made to improve the filler-loading capacity of fly ash (FA) by its surface modification with zinc hydroxide and calcium hydroxide. In the modification reaction, FA to calcium hydroxide weight ratio was maintained constant at 5:1, while that of FA to zinc hydroxide was varied as 5:0.4, 5:0.6, and 5:0.8 and the optimized composition was utilized as filler in nylon 6. Modified FA was found to have formed hemimorphite on to its surface, giving rise to spine-structured surface morphology. FA surface modified with 5:0.6 weight ratio of FA to zinc hydroxide (FaZn6Ca) provided FA with the highest level of spine-structured surface morphology and thus was determined to the optimized composition to be utilized as filler in nylon 6. Concentration of FaZn6Ca was varied up to 40 phr in nylon 6. Prepared composites were characterized for mechanical, thermal, rheological, crystallinity, and morphological properties. Melting and crystallization temperatures remained approximately constant; tensile strength, flexural strength, crystallinity, tensile modulus, flexural modulus, enthalpy of melting (Hm), and enthalpy of crystallization increased, whereas elongation at break and impact strength decreased with increase in FaZn6Ca concentration in nylon 6. Spine-structured surface morphology formed on the surface of FA increased the filler-loading capacity of FaZn6Ca in nylon 6; while the improvement in the properties were caused due to better interactions happening between nylon 6 and FaZn6Ca due to the hydrophilic nature of both the materials. Since FA is obtained as waste from thermal power plant, the use of this material as a filler in nylon 6 would help ease the solid waste disposal problem which will benefit the environment.
Introduction
Fly ash (FA) is defined (Cement and Concrete Terminology, ACI Committee 116) as the “finely divided residue resulting from the combustion of ground or powdered coal, which is transported from the fire box through the boiler by flue gases”. In India, 30–40% of the coal used in the coal-based thermal power plants is converted into ash. 1 Coal with ash content of 40% is predominantly used in India. 2 Whereas thermal power plants based on coal make about 65% of the total thermal power plants in India, generating over 120 million tons of FA. Large quantity of this generated FA is used as soil stabilizer, as metal releaser, and as a construction material. 3 However, this utilization is only about 38% compared to 85 and 100% in Germany and Netherlands, respectively. 4,5
Recently, FA has been utilized as a filler material in varied type of plastics. FA has about 90% of good spherical-shaped particles having low density and good dispersibility, which enhances its suitability as a polymer-filling material. 6,7 It had been used as a filler in varied polymers like acrylonitrile butadiene styrene copolymer, 8 high-density polyethylene, 9 vinyl ester resin, 10 polyaniline, 11 polyurethane-modified epoxy, 12 polyvinyl alcohol, 13 polypropylene (PP)/poly(methyl methacrylate) blend, 14 hytrel, 15 nylon 6, 16,17 and so on. However, they all reported improvement in thermal properties; nevertheless, mechanical properties reduced with increased addition of FA in the polymer matrix. The reduction in mechanical properties is attributed to the weak interfacial bonding between FA and polymer and low friction at the polymer-FA interface. 18,19 Also, the low whiteness value of FA (giving undesirable color to the product, which even makes addition of any external color difficult) makes the prepared polymer composite aesthetically inferior. Thus, surface treatment of FA becomes very important, not only to improve its whiteness but also to make the surface of FA particle rough, which can provide higher interacting surface with the polymer chains and also would increase the friction at the polymer-FA interface. This would not only help in improvement of thermal and mechanical properties of the composite but would also increase the filler-loading capacity of FA. Bose and Mahanwar investigated the effect of coupling agent like tetra isopropyl titanate on nylon 6/FA composite and determined improvement in mechanical as well as thermal properties compared with nylon 6/FA composite without the use of coupling agent. 20 Khan et al. utilized acid-functionalized oil-based FA (a by-product of oil fuel power plants) as filler in low-density polyethylene (LDPE). The concentration of acid-functionalized oil FA was found to have no effect on the mechanical property of LDPE. 21 Recently, Nath et al. utilized FA, which was chemically modified by activation with sodium hydroxide, as filler in poly (vinyl alcohol). Prepared composite films containing 20% of the modified FA showed three times improvement in mechanical properties compared with pristine poly (vinyl alcohol) films. 22 Yang et al. reported surface-modified FA with calcium hydroxide using isothermal heating method and blended it with PP. PP/modified FA composite showed noticeable change in properties compared with PP/FA composite. 23 Recently, Yeole et al. investigated the surface modification of FA using nanosized zinc oxide as potential material to improve the anticorrosion property of polymer-based organic coatings. 24
The aims of this article are to (i) develop a morphologically suitable FA (FaZnCa) by its surface modification with calcium hydroxide and zinc hydroxide and (ii) prepare nylon 6/FaZnCa composite using the optimized FaZnCa composition and characterize its mechanical, thermal, crystallinity, rheological, and morphological properties. Since FA is obtained as waste from thermal power plant, the use of this material as a filler in nylon 6 would help ease the solid waste disposal problem which will benefit the environment.
Materials and methods
Materials
FA (composition: 57.13 wt% silicon dioxide (SiO2), 34.24 wt% aluminum oxide (Al2O3), 2.84 wt% calcium oxide, 0.91 wt% magnesium oxide, 2.78 wt% iron oxide, 0.65 wt% potassium oxide, and 0.91 wt% titanium dioxide; specific gravity: 2.20 g cm−3. Unburned carbon determined by Loss on Ignition ((LOI): 2.90%) was obtained from Nashik Thermal Power Plant (Nashik, Maharashtra, India). Zinc chloride, calcium hydroxide, and sodium hydroxide were obtained from SD Fine Chemicals Pvt. Ltd (Mumbai, Maharashtra, India). Nylon 6 (NXE-01 NC) was obtained from Next Polymers Ltd (Mumbai, Maharashtra, India). Finalux G3 (wetting agent) was obtained from Fine Organics Pvt. Ltd (Mumbai, Maharashtra, India). All chemical materials were used as obtained without any modification.
Methods
Size separation
FA was sieved to get particles in the size ranging between 45 µm and 75 µm. Sieving was first done with 45 µm sieve. Collected oversized particles were then sieved with 75 µm sieve. FA particles of size in between 45 and 75 µm were thus obtained.
Purification
FA collected from thermal power plants contains unburned carbon and ferrous particles, showing its presence as dark black particles. These particles increase the darkness of FA; removing which would make the FA become little light in shade. The magnetic separator removed ferrous particles; whereas the unburned carbon particles were removed by froth floatation method as mentioned by Altun et al. 25
Surface modification of FA and optimization of zinc hydroxide concentration
Surface modification of purified FA was carried out in two stages.
In first stage (stage 1), FA was surface modified with varied concentrations of zinc hydroxide. Ratio of FA to zinc hydroxide (FaZn) was varied as 5:0.4 (FaZn4), 5:0.6 (FaZn6), and 5:0.8 (FaZn8) (weight ratio).
Zinc chloride and sodium hydroxide were used to produce zinc hydroxide. Initially, in a reaction kettle, zinc chloride was dissolved in 25 ml distilled water. After about 10 min, sodium hydroxide was added to this solution. Zinc chloride and sodium hydroxide were reacted in 1:1 molar ratio. The reaction mixture was stirred for 20 min or until a white precipitation of zinc hydroxide was produced. Then, stirring was stopped, and the mixture was allowed to settle down; and clear water was pumped out. To this settled mixture excess of distilled water was added and stirred again for few minutes. Stirring was stopped, and the mixture was allowed to settle; after settling, the clear water was again pumped out. This procedure was repeated one more time to remove sodium chloride (by-product) and any unreacted zinc chloride or sodium hydroxide. Required amount of purified FA was then added to the purified white precipitate and stirring was continued for 1 h at 30°C and 150 r min−1. The reaction between silicate present on the surface of FA and zinc hydroxide led to the formation of zinc silicate on to the surface of FA (FaZn—FaZn4, FaZn6, and FaZn8). All calculations were made taking 250 g FA as the basis.
In second stage (stage 2), calcium hydroxide was added to the FaZn mixtures obtained from stage 1. The FaZn (FaZn4, FaZn6, and FaZn8)/calcium hydroxide ratio was maintained constant at 5:1 (weight ratio), as suggested by Yang et al.
23
Distilled water was added to the mixture to maintain the water/solids weight ratio as 5:1. The slurry was continuously agitated at 150 r min−1, fleetly heated to 90°C, and maintained therein for 5 h. Temperature was maintained constant using a temperature regulator. The reaction of calcium hydroxide with the silicate present on the surface of FA led to the formation of calcium silicate on to the surface of FA. Thus, the FA obtained from stage 2 had its surface coated with both zinc silicate and calcium silicate (FaZnCa). Depending on the weight ratio of FA to zinc hydroxide in stage 1 and its subsequent modification in stage 2 provided us with three types of surface-modified FA:
FaZn4Ca: wherein FA:zinc hydroxide weight ratio was maintained constant at 5:0.4 and the obtained zinc silicate surface modified FA was further treated with calcium hydroxide by maintaining their weight ratios as 5:1; FaZn6Ca: wherein FA:zinc hydroxide weight ratio was maintained constant at 5:0.6 and the obtained zinc silicate surface modified FA was further treated with calcium hydroxide by maintaining their weight ratios as 5:1; and FaZn8Ca: wherein FA:zinc hydroxide weight ratio was maintained constant at 5:0.8 and the obtained zinc silicate surface modified FA was further treated with calcium hydroxide by maintaining their weight ratios as 5:1.
The obtained zinc silicate and calcium silicate-coated FA (FaZn4Ca, FaZn6Ca, and FaZn8Ca)—water mixture was dried in an air-circulating oven at 70°C for 8 h. Prepared FaZnCa (FaZn4Ca, FaZn6Ca, and FaZn8Ca) were characterized using scanning electron microscopy (SEM), specific gravity, Fourier transform infrared spectroscopy (FTIR), and X-ray diffraction (XRD) properties. Optimized composition of FaZnCa was then utilized as filler in nylon 6.
Modification mechanism
FA is chemically active at certain reaction conditions and can react with other suitable reactants. FA contains about 90% of silicate and aluminate which are chemically active in the modification process. Experimental study carried out separately by Esquevin 26 and Roy and coworkers 27,28 favored the synthesis of zinc silicate (hemimorphite) when zinc hydroxide and silicate undergo reaction. Zinc hydroxide is highly unstable and reactive. It undergoes quick reaction with silicate (present on the surface of FA) giving zinc silicate (hemimorphite), forming the coating of zinc silicate on the surface of FA, providing surface roughness. The conceivable chemical reactions happening during modification with zinc hydroxide are shown in Figure 1(a).
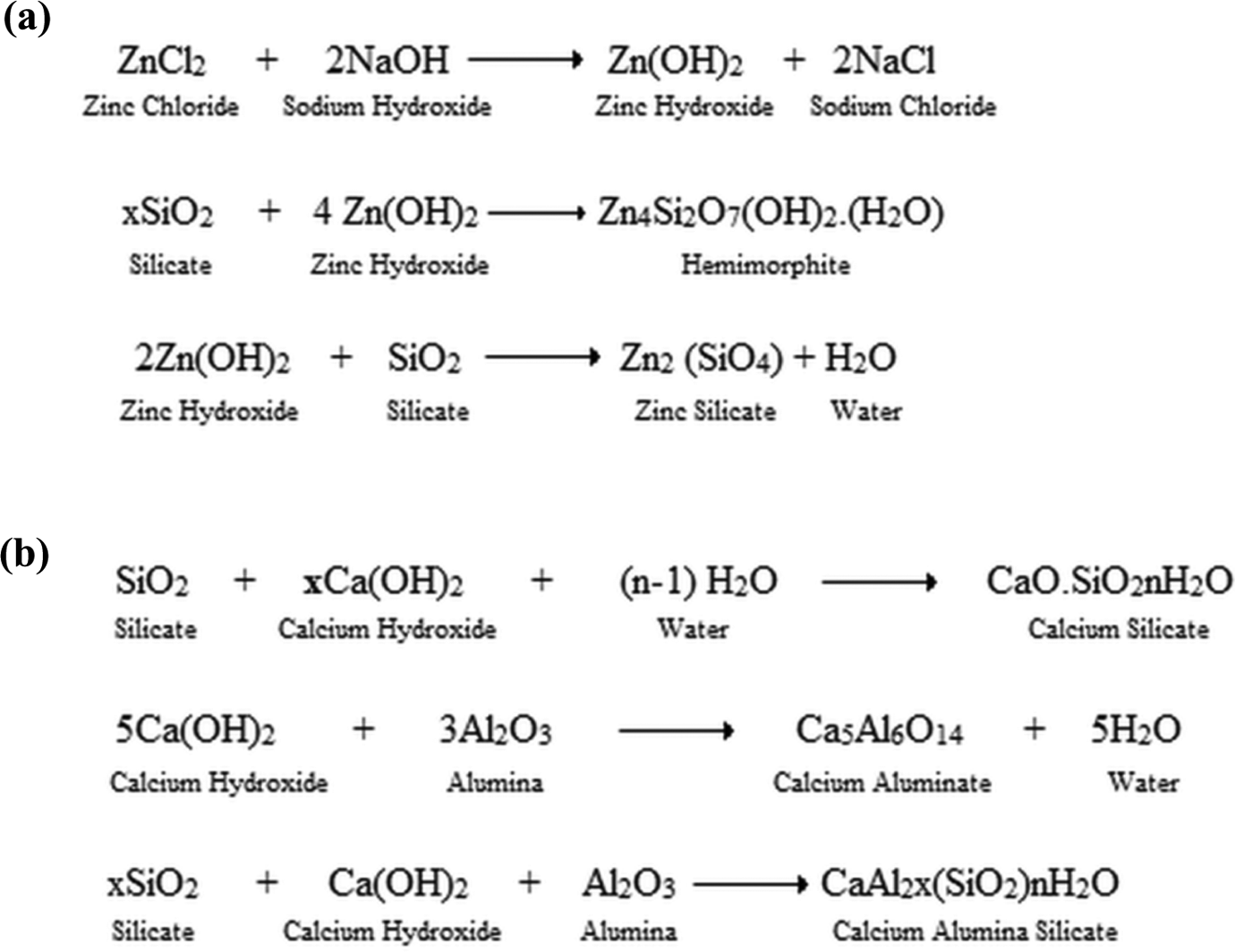
Conceivable chemical reactions happening during the surface modification of FA with zinc hydroxide and calcium hydroxide. FA: fly ash.
Lu et al. 29 explained the reaction mechanism of calcium hydroxide and silicate present on the surface of FA. Calcium hydroxide being stable at room temperature required harsh condition (90°C) to undergo reaction with the silicate of FA. Calcium hydroxide undergoes reaction with silicate giving calcium silicate. Calcium silicate also produced roughness on the surface of FA. The conceivable chemical reactions happening during modification with calcium hydroxide are shown in Figure 1(b).
From the analysis, FaZn6Ca (mentioned previously) was determined to be the optimized composition to be utilized as filler in nylon 6
Preparation of nylon 6/FaZn6Ca composite
Dry blending
FaZn6Ca and nylon 6 were dried in an air-circulating oven at 80°C for 12 h to remove the adsorbed and absorbed moisture. Dry blending of FaZn6Ca and nylon 6 was performed in a tumbler mixer for 10–15 min, using Finalux G3 as the wetting agent.
Melt blending
Dry-blended nylon 6/FaZn6Ca mix was melt blended in a corotating twin-screw extruder (Lab Tech. Engineering Co. Ltd, Thailand) having length/diameter ratio of 32:1, and the temperature profile from the hopper to the die as 165, 180, 190, 205, 215, 225, 235, and 250°C. The feeder screw rotation speed was maintained at 10 r min−1; whereas the twin-screw rotation speed was kept constant at 200 r min−1. Vacuum was applied in the vent of the extruder to facilitate easy removal of moisture (if any present). Extruded strands were water cooled (30°C) and pelletized.
Injection molding
Pellets were dried in an oven at 80°C for 10 h to remove the adsorbed and absorbed moisture. Injection molding (Boolani Machineries India Ltd, Mumbai, Maharashtra, India) was performed by maintaining the temperature profile as 210, 230, and 250°C from the hopper to the ejection nozzle. Standard ASTM samples for tensile (D 638), flexural (D 790), and impact (D 256) testing were obtained from injection molding. Injection pressure, packing pressure, and cooling time were maintained constant at 115 MPa, 50 MPa, and 1 min, respectively.
Samples for impact testing were notch cut before testing. Prepared formulations and their nomenclature are shown in the Table 1. Concentration of FaZn6Ca was varied from 0 phr to 40 phr of nylon 6, whereas the concentration of wetting agent (Finalux G3) was maintained constant at 2 phr of nylon 6.
Prepared nylon 6/FaZn6Ca compositions and their nomenclature.
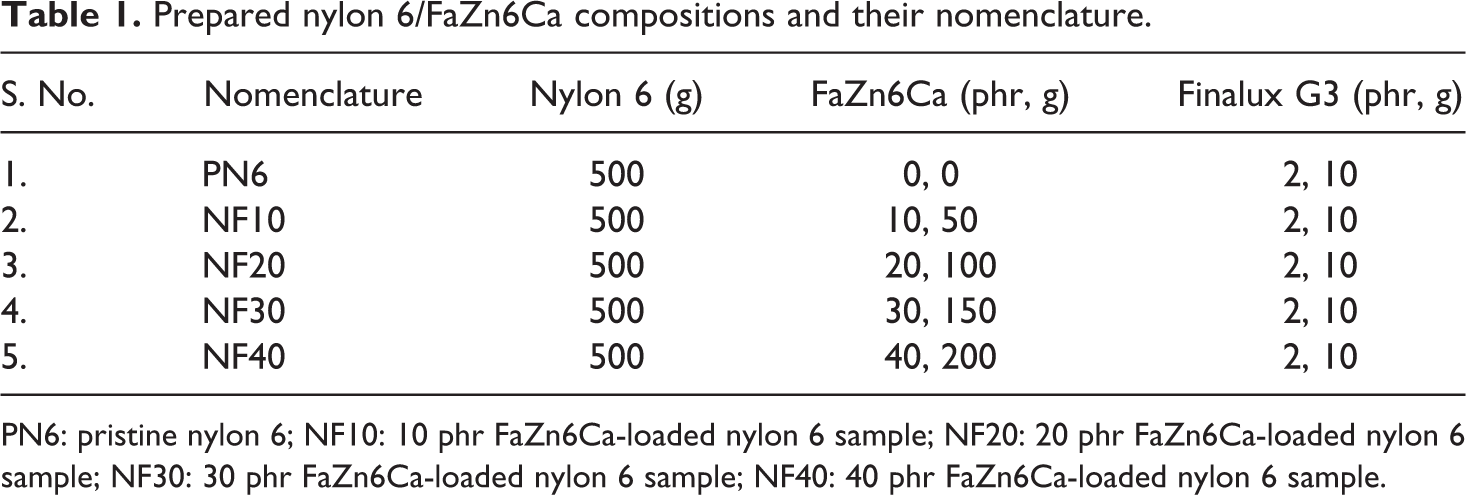
PN6: pristine nylon 6; NF10: 10 phr FaZn6Ca-loaded nylon 6 sample; NF20: 20 phr FaZn6Ca-loaded nylon 6 sample; NF30: 30 phr FaZn6Ca-loaded nylon 6 sample; NF40: 40 phr FaZn6Ca-loaded nylon 6 sample.
Characterization and testing
Mechanical properties
Tensile properties (tensile strength, tensile modulus, and percentage elongation at break) and flexural properties (flexural strength and flexural modulus) were measured at ambient condition using a universal testing machine (LR-50K, Lloyds Instrument, UK), according to ASTM procedures D 638 and D 790, at a crosshead speed of 50 and 2.8 mm min−1, respectively. The notch for impact test was made using a motorized notch-cutting machine (Polytest model 1, Ray Ran, UK). Notched Izod impact strength was determined at ambient condition according to ASTM D 256 standard using an impact tester (Avery Denison, UK) having striking velocity of 3.46 m s−1, employing a 2.7-J striker.
Differential scanning calorimetry
Differential scanning calorimetry (DSC; Q 100 DSC, TA instruments Ltd, Bengaluru, Karnataka, India) characterization was done to investigate the crystallization and melting behavior of the prepared composites. Two consecutive heating scans were performed to minimize the influence of any possible residual stresses in the material, due to any specific thermal history. Scanning rate of 10°C min−1 was maintained constant for both the heating and cooling cycles; whereas nitrogen gas purge rate was maintained at 50 ml min−1. Melting temperature (Tm) and Hm were determined from the second heating scan, whereas the crystallization temperature (Tc) and enthalpy of crystallization (Hc) from the only cooling scan.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) of the prepared nylon 6/FaZn6Ca composite samples was performed on a DSC Q-100 instrument in the temperature range of 100–600°C at a heating rate of 10°C min−1 under nitrogen atmosphere.
Rheological properties
The melt viscosity was measured using a rotational rheometer (MCR101, Anton Paar, Gurgaon, Haryana, India) with parallel-plate assembly, having diameter of 35 mm. Samples were predried before analysis. Viscosity was determined for shear rates from 0.1 s−1 to 1000 s−1 at a constant temperature of 250°C. Damping factor (tan δ) was determined for angular frequencies from 0.5 s−1 to 500 s−1 at a constant temperature of 250°C.
Morphological properties
SEM analysis was performed with a JEOL 6380 LA (Japan) instrument. Samples were fractured under liquid nitrogen to avoid any disturbance to the molecular structure and then coated with gold before imaging.
XRD analysis
The XRD analysis was used to determine the chemical characteristics of FaZnCa samples and also to determine the percentage crystallinity of the prepared composites (nylon 6/FaZn6Ca). A normal focus copper X-ray tube was operated at 30 kV and 15 mA. Sample scanning was done from 10° to 60° at a rate of 3° min−1. The data processing was done using Jade 6.0 software.
FTIR analysis
The FA and FaZn6Ca were characterized using a Perkin Elmer Paragon 1000 (Waltham, Massachusetts, USA) FTIR spectrophotometer. The samples were vacuum-dried at room temperature for 2–3 days before characterizations. The samples were taken at random, and the data were collected over 16 scans with a resolution of 4 cm−1 at room temperature in the scan range of 450–4000 cm−1.
Specific gravity
The specific gravity of FA and FaZnCa compositions was measured according to ASTM D-153 standard.
Results and discussion
Characterization of FaZnCa and optimization of zinc hydroxide concentration
Morphological analysis
SEM micrographs obtained for unmodified FA (a) and zinc hydroxide and calcium hydroxide-modified FA compositions (FaZnCa—FaZn4Ca (b), FaZn6Ca (c), and FaZn8Ca (d)) are shown in Figure 2. FA particles were found to be completely spherical in shape with very smooth surface area. On the other hand, FaZnCa compositions were seen to have very rough spine-structured surface as compared to unmodified FA. Surface modification of FA with zinc hydroxide and calcium hydroxide leading to the formation of zinc silicate (hemimorphite) and calcium silicate on to its surface was the prime reason for getting this morphology. All particles of FA can be seen to have completely coated with zinc silicate and calcium silicate (reaction product formed by reaction of zinc hydroxide and calcium hydroxide with silicate present on the surface of FA). The degree and characteristic of roughness is similar on all FaZnCa particles of a particular composition. This roughness is due to the hemimorphite formation on the surface of FA by its treatment with zinc hydroxide.

Scanning electron micrographs obtained for FA (a), FaZn4Ca (b), FaZn6Ca (c), and FaZn8Ca (d). FA: fly ash.
From Figure 2(b) to (d), it can be seen that the highest level of rough spine-structured surface was obtained for FaZn6Ca. Rough-structured surface is also formed in the case of FaZn4Ca, but it is not spine shaped, which is attributed to the less zinc silicate (hemimorphite) formation as it was treated with lesser concentration of zinc hydroxide. In FaZn8Ca, the amount of zinc hydroxide exceeds to such an extent that the spine-structured surface is converted to globular structure, thereby decreasing the effective surface roughness. Thus, FaZn6Ca provides optimized improvement in surface area to interact with the polymer molecules in which it would be added. Therefore, we have utilized FaZn6Ca as a filler material in nylon 6 in order to understand the effect of its concentration on the performance properties of nylon 6.
The smooth surface of FA will have limited interaction with the polymer chains. In addition, there will not be proper gripping between polymer chains and FA surface. The roughness of the prepared FaZn6Ca can prove helpful in increasing the effective surface area for interacting with the polymer chains and there would be proper gripping, in which it would be incorporated as filler. Similar results have been obtained by Yang et al. 23 and Lu et al. 29 on calcium hydroxide treatment of FA. However, the degree of roughness obtained by us is appreciably high compared to that obtained by Yang et al. and Zhai et al. on just treatment by calcium hydroxide. Incorporation of zinc hydroxide in addition to calcium hydroxide helps improve the surface roughness of FA by formation of the spine-structured surface morphology. Thus, the spherical and smooth surface of unmodified FA has been converted into rough and spine shaped on its surface treatment with zinc hydroxide and calcium hydroxide.
Specific gravity
Specific gravity values obtained for FA and FaZnCa compositions (FaZn4Ca, FaZn6Ca, and FaZn8Ca) are listed in Table 2. Specific gravity of FA increased with increased concentration of zinc hydroxide, used for its surface modification. Zinc silicate and calcium silicate were formed on the surface of FA by its surface modification with zinc hydroxide and calcium hydroxide. The addition of this led to increase in weight of FA, with not so appreciable change in volume of the particles. This led to increase in density of FaZnCa compositions and thus their specific gravity. Density of zinc silicate is about 3.44 g cm−3 while that of calcium silicate is 2.9 g cm−3, which is much higher than the density of FA (2.22 g cm−3). Thus, the formation of zinc silicate and calcium silicate on to the surface of FA increases its density and thus specific gravity. This increase in the values of specific gravity of FA with increased concentration of zinc hydroxide to surface modification indicates increased formation of zinc silicate on to its surface, which is also corroborated by SEM analysis.
Effect of zinc hydroxide and calcium hydroxide surface modification on specific gravity of FA.

FA: fly ash.
XRD and FTIR analysis were performed on unmodified FA and FaZn6Ca, as all the modified FA samples were prepared in a similar way giving similar type of modification, with the only difference in the ratio of FA to zinc hydroxide, which would only change the thickness and morphology of zinc silicate deposition on the surface of FA. Thus, as a representative of all the samples, FaZn6Ca was analyzed and compared with unmodified FA for XRD and FTIR analyses.
XRD analysis
XRD was employed to analyze the chemical composition and crystal structure of the prepared FaZnCa compositions. Chemical composition of FA depends on the chemical composition of the coal used in the thermal power plant (FA is the waste obtained from thermal power plant). Since it varies from region to region, chemical composition of FA varies slightly. Phase identifications were made by comparing the diffraction pattern of the mixture to a database of pure phase reference patterns of diffraction data. In FA sample, key crystalline compounds identified by X-ray powder diffraction include quartz (card no. 650466; 3.353, 4.26, and 1.844 Å) and mullite (card no. 150776; 3.397, 3.429, 2.210, and 5.395 Å). The XRD pattern of FaZn6Ca (Figure 2(b)) shows the existence of mullite (card no. 150776; 3.397, 3.429, 2.210, and 5.395 Å), quartz (card no. 650466; 3.353, 4.26, and 1.844 Å), and significant amount of hemimorphite (card no. 241259; 3.104, 6.600, and 1.541 Å). The silicate and aluminate components such as Zn4Si2O7(OH)2·(H2O), ZnSiO4 Ca3Si3O9·H20, CaAl2(SiO3)4·2H2O, and Ca5Al6O14 are present in FaZn6Ca which would be the reaction products of Zn(OH)2, Ca(OH)2, and active SiO2 or Al2O3. These products adhere on to the surface of FA particle altering its surface morphology. It can be seen from Figure 3 that on addition of Zn(OH)2, the characteristic peak of hemimorphite were determined, whereas the characteristic peaks of mullite become weaker, indicating the consumption of silicate. Data published by Hill et al. 30 conformed the formation of hemimorphite in FaZn6Ca, having spine-structured surface morphology. This peculiar morphological development is the reason for the obtained surface roughness of FaZn6Ca. Hemimorphite is a zinc silicate mineral. Both FA and FaZn6Ca produced broad patterns in the XRD spectra indicating the presence of amorphousness.
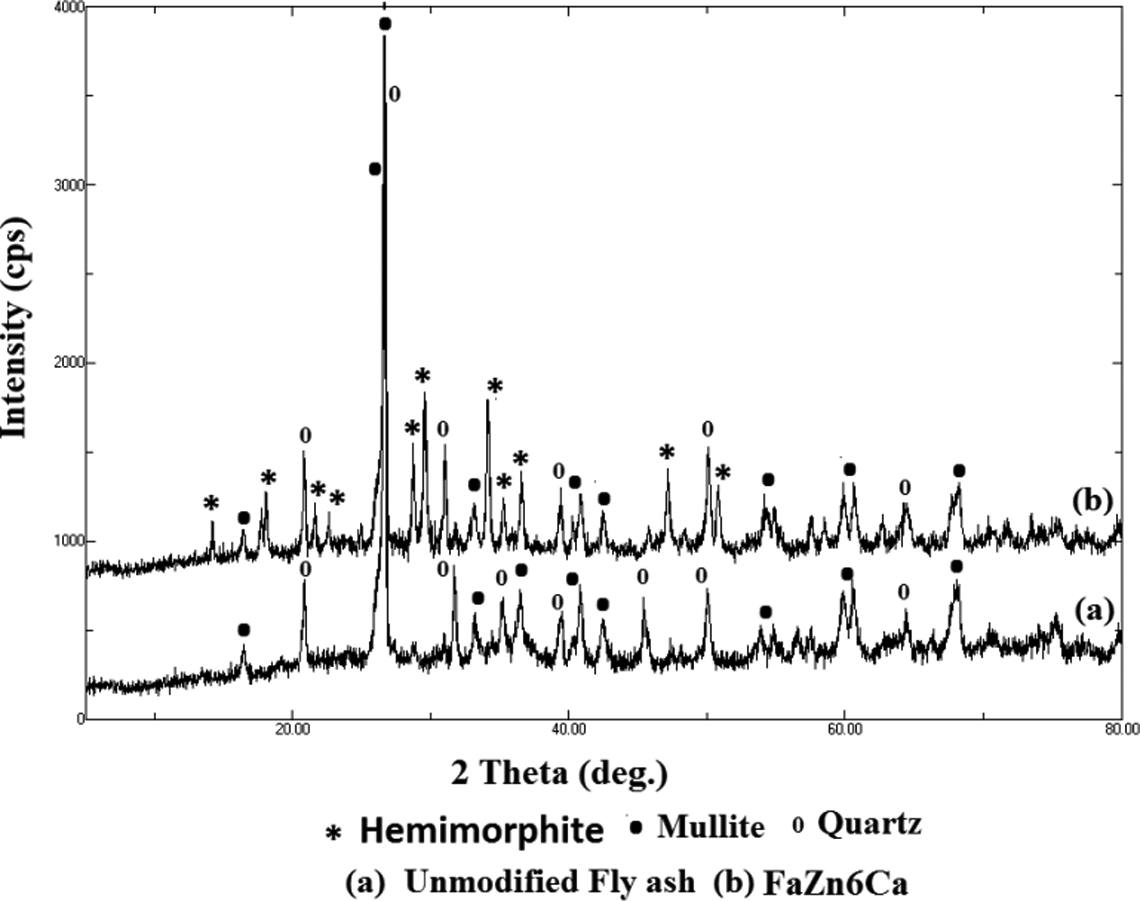
X-ray diffractograms obtained for FA and FaZn6Ca. FA: Fly ash.
FaZn4Ca and FaZn8Ca were also prepared in a similar way with the only difference in the ratio of FA to zinc hydroxide, giving variation in the formation of zinc silicate (hemimorphite) on to the surface of FA. So, similar peaks at similar locations can be expected in them as that observed in FaZn6Ca and so their XRD characterizations were not performed.
FTIR spectroscopy
FTIR analysis was used to examine FaZn6Ca and compare it with unmodified FA. Absorption in the IR region takes places because of the rotational and vibrational movements of the molecular groups and chemical bond of a molecule. The two fundamental vibrations are stretching (where the atoms stay in the same bound axis but the distance between atoms increases or decreases) and deformation (where the positions of the atoms change relative to the original bound axis). FTIR spectra of FA, before and after the modifications, are presented in Figure 4.
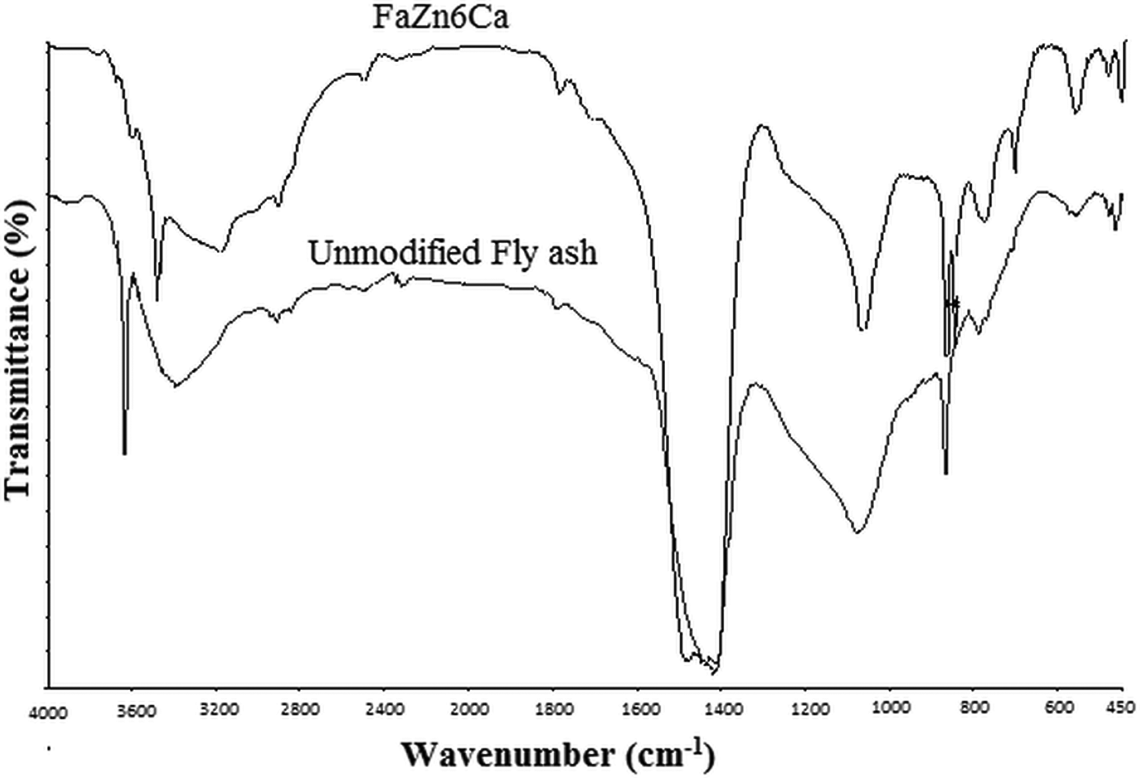
FTIR spectra obtained for FA and FaZn6Ca. FTIR: Fourier transform infrared; FA: fly ash.
As can be seen from Figure 4, the most characteristic difference observed between the peaks of FA and FaZn6Ca is the broad bands that appeared in the region of 3650 and 2600 cm−1 in FaZn6Ca and are assigned to stretching (–OH) and bending (H–O–H) vibrations of bound water molecules, which are crystal lattice water. Broadness of this peak increased due to component of zinc silicate and calcium silicate formed on to the surface of FA. Both these materials are hydrophilic in nature. Thus, they increased the water-holding ability of the FA, which led to the increase in the broadness of the particular peaks.
Peak at 1085 cm−1 in unmodified FA is attributed to the Si–O–Si chain structure of the internal SiO4 tetra hedra. Whereas the peak at 1070 cm−1 in FaZn6Ca is attributed to the cyclic Si–O–Si structure, which is an indication of the conversion of the random glass network structure of the unmodified FA to zeolites-like structure on its surface modification with zinc and calcium hydroxide. 21
Thus, FTIR suggests the surface modification of FA by its treatment with zinc hydroxide and calcium hydroxide through the formation of zinc silicate and calcium silicate. FaZn4Ca and FaZn8Ca were also prepared in a similar way as that of FaZn6Ca, thus suggesting the formation of zinc silicate and calcium silicate on to the surface of FA.
Characterization of nylon 6/FaZn6Ca composite
Prepared optimized FaZn6Ca was used as filler in nylon 6. It was expected that the spine structure formed on to the surface of FA on treatment with zinc hydroxide and calcium hydroxide would provide extensive surface area for interaction with nylon 6, helping to improve its performance property.
Mechanical properties
Mechanical properties like tensile strength, tensile modulus, percentage elongation at break, flexural strength, flexural modulus, and impact strength obtained for nylon 6 and the prepared nylon 6/FaZn6Ca composites are listed in Table 3.
Mechanical properties obtained for the nylon 6 and nylon 6/FaZn6Ca composites.
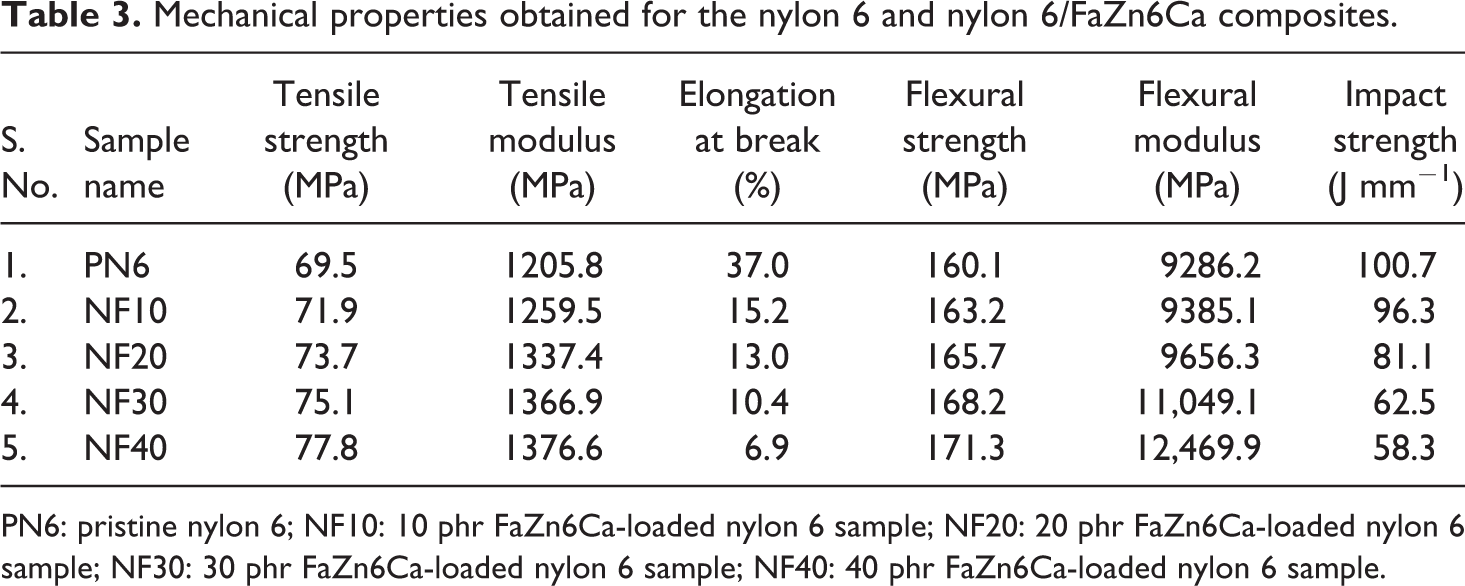
PN6: pristine nylon 6; NF10: 10 phr FaZn6Ca-loaded nylon 6 sample; NF20: 20 phr FaZn6Ca-loaded nylon 6 sample; NF30: 30 phr FaZn6Ca-loaded nylon 6 sample; NF40: 40 phr FaZn6Ca-loaded nylon 6 sample.
Tensile strength, tensile modulus, flexural strength, and flexural modulus increased, while percentage elongation at break and impact strength decreased with increased concentration of FaZn6Ca in nylon 6. Reaction of zinc hydroxide and calcium hydroxide with silicate present on the surface of FA led to the formation of hemimorphite (zinc silicate) on the surface of FA. Nylon 6 is widely known to be a hydrophilic polymer. Thus, FaZn6Ca and nylon 6 have better compatibility with each other. Zinc silicate and calcium silicate formed on the surface of FA giving spine-structured morphology is also hydrophilic in nature and is also conformed through FTIR analysis. In addition, the spine-structured morphology formed on the surface of FaZn6Ca increased the surface area for interaction with nylon 6. Thus, better compatibility and increased surface area increased the level of interactions happening between nylon 6 and FaZn6Ca. This caused polymer molecules to orient themselves about FaZn6Ca, increasing crystallinity (see XRD analysis). As the concentration of FaZnCa increased, the effective surface area for interaction between FaZn6Ca and nylon 6 increased, increasing the crystallinity. Thus, even at higher loading of FaZn6Ca (40 phr), tensile strength, flexural strength, tensile modulus, and flexural modulus increased with increase in FaZn6Ca concentration. Increased concentration of FaZn6Ca increased the stiffness of the composite, as the interaction between FaZn6Ca and nylon 6 increased, increasing the crystallinity. Percentage elongation at break and impact strength decreased appreciably on addition of FaZn6Ca in nylon 6. This was due to the increase in stiffness, induced by better interaction between FaZn6Ca and nylon 6. Thus, surface modification of FA with zinc and calcium hydroxide improved its filling capacity, which is mainly due to better compatibility and increased surface area. Similar results are reported by Yang et al., 23 Khan et al., 21 and Nath et al. 22
Bose and Mahanwar 31 had reported the effect of addition of unmodified FA on the mechanical properties of nylon 6. They determined decrease in the values of tensile strength, percentage elongation at break, and impact strength, while the values of flexural strength remained near about constant. However, the values of tensile and flexural modulus increased on 40 wt% addition of FA in nylon 6. Compared to their work, our investigation demonstrates improved mechanical properties with continuous improvement in the values of tensile strength, tensile modulus, flexural strength, and flexural modulus, which is attributed to the altered surface morphology of FA. Thus, surface modification of FA with zinc hydroxide and calcium hydroxide proves beneficial in improving the mechanical properties of nylon 6 as compared to unmodified FA.
Differential scanning calorimetry
DSC analysis was undertaken to understand the effect of FaZn6Ca addition on the melting and cooling characteristics of nylon 6. Melting and cooling curves obtained for the prepared composites are presented in Figures 5 and 6, whereas the values obtained for Tm, Hm, Tc and Hc are listed in Table 4.
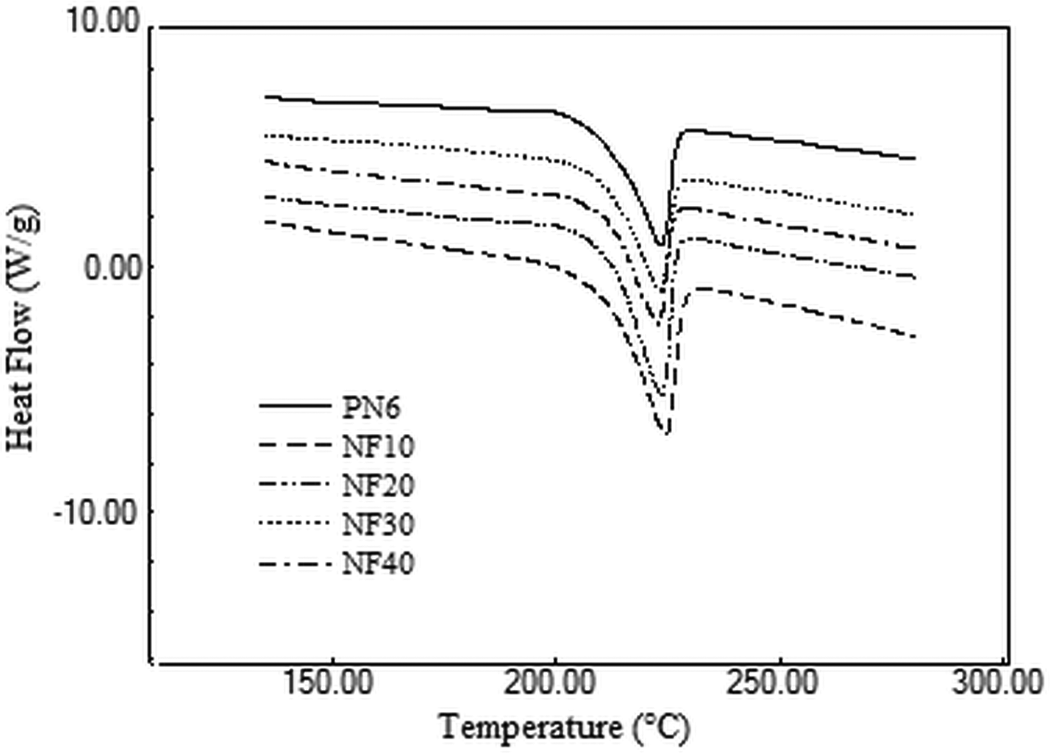
DSC heating scans obtained for nylon 6 and the prepared nylon 6/FaZnCa composites. DSC: differential scanning calorimetric.
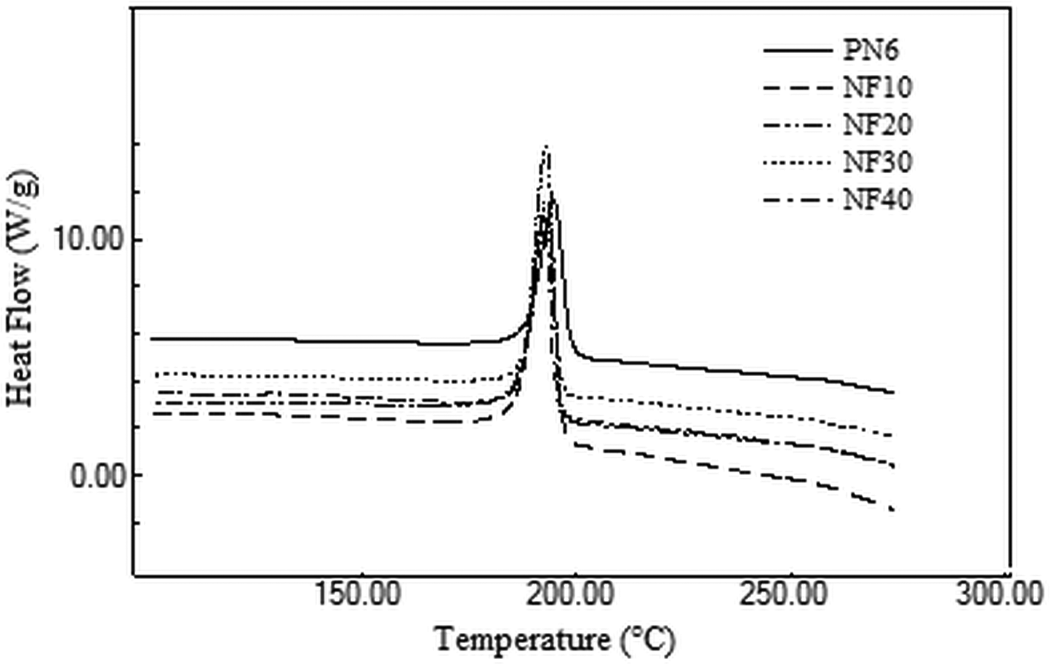
DSC cooling scans obtained for nylon 6 and the prepared nylon 6/FaZnCa composites. DSC: differential scanning calorimetric.
Thermal characteristics of nylon 6 and the prepared nylon 6/FaZn6Ca composites.
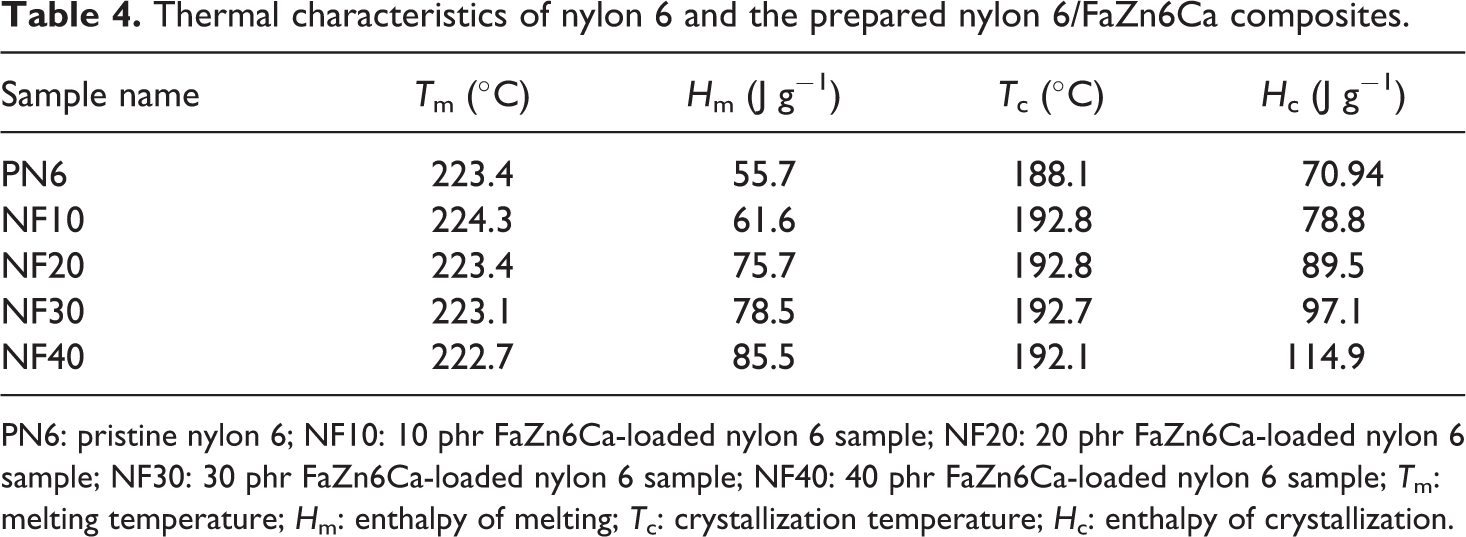
PN6: pristine nylon 6; NF10: 10 phr FaZn6Ca-loaded nylon 6 sample; NF20: 20 phr FaZn6Ca-loaded nylon 6 sample; NF30: 30 phr FaZn6Ca-loaded nylon 6 sample; NF40: 40 phr FaZn6Ca-loaded nylon 6 sample; Tm: melting temperature; Hm: enthalpy of melting; Tc: crystallization temperature; Hc: enthalpy of crystallization.
Addition of FaZn6Ca had no appreciable effect on Tm and Tc of nylon 6. However, Hm and Hc increased with increase in concentration of FaZn6Ca. FaZn6Ca and nylon 6, both being hydrophilic in nature, had better interactions with each other. This led to better orientation of nylon 6 polymer chains about FaZn6Ca particles. In addition, the spine-structured morphology of the surface of FaZn6Ca due to the surface modification of FA with zinc and calcium hydroxide increased the effective surface area of FA to interact with nylon 6 polymer chains. Both better compatibility and increased surface interaction area led to increase in crystallinity of the composite (see XRD analysis of composite ahead). Crystallinity increased with increase in concentration of FaZn6Ca. Higher the concentration of FaZn6Ca, higher is the effective surface area provided for interaction with nylon 6 polymer chains. Increase in interaction and crystallinity with increase in FaZn6Ca concentration, increased the heat required for melting the composite structure. FA is a thermally nonconducting material. Even zinc and calcium silicates adhered to the structure of FaZn6Ca are thermally insulating materials. FaZn6Ca must also have induced this property into nylon 6, increasing the resistance to flow of heat toward nylon 6 polymer chains for melting. Hm increased by 50%, while Hc increased by 100% on 40% addition of FaZn6Ca in nylon 6.
Thus, FaZn6Ca is not only useful is increasing the mechanical property but also is helpful in improving the thermal stability of nylon 6. Better compatibility and increased surface area made it an ideal material to be added as a filler in nylon 6.
Thermogravimetric analysis
TGA curves obtained for pristine nylon 6 (PN6), 20 phr FaZn6Ca-loaded nylon 6 sample (NF20) and 40 phr FaZn6Ca-loaded nylon 6 sample (NF40) are illustrated in Figure 7, and the data are used to understand the effect of FaZn6Ca addition on the degradation characteristics of nylon 6. Temperature at which samples were decomposed by 5 wt% are marked in Figure 7. It can be seen that the 5 wt% decomposition temperature of nylon 6 increased with increased addition of FaZn6Ca. Nylon 6 used by us in the study was determined to have 5 wt% decomposition temperature of about 314.8°C and increased to 324.6°C on addition of 40 phr of FaZn6Ca in it, which is an increase of about 10°C. Thus, FaZn6Ca induces thermal stability in to nylon 6, which is attributed to the increased level of interaction happening between FaZn6Ca and nylon 6 polymer chains due to the hydrophilic nature of both the materials and the increased surface area provided by the spine-structured morphology developed by zinc silicate and calcium silicate on to the surface of FA. Additionally, FaZn6Ca might also have induced its thermal insulation property in to nylon 6, making it difficult for the heat to get easily transferred into the composite structure. Both these factors led to the increase in the thermal stability of the nylon 6.
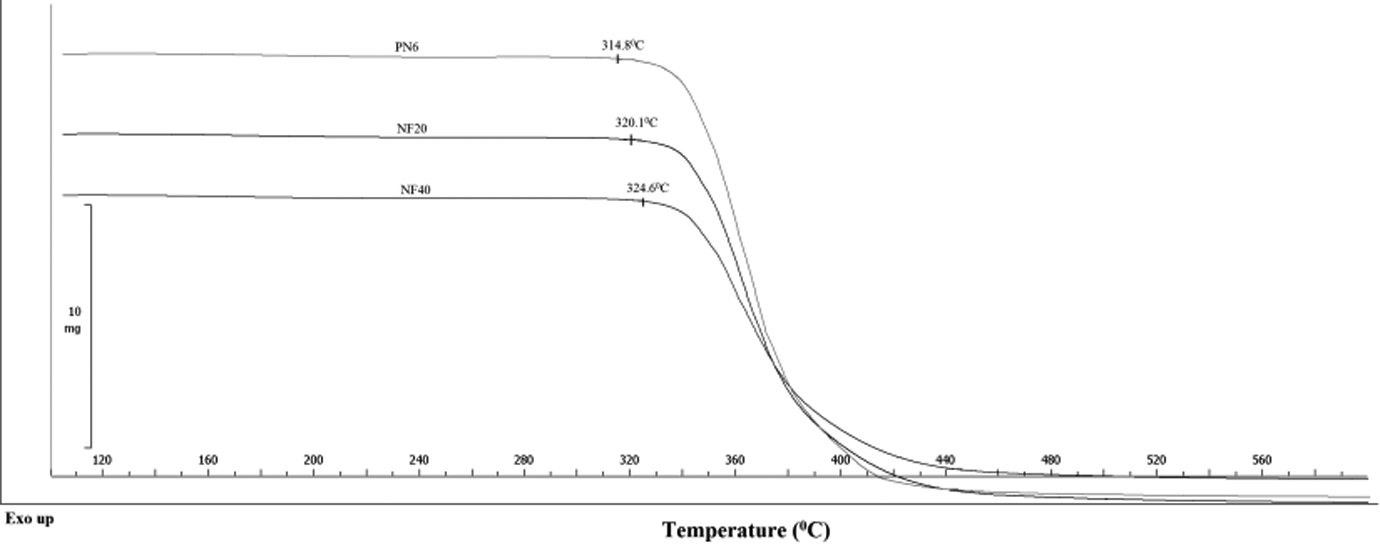
TGA curves obtained for PN6, NF20, and NF40. TGA: thermogravimetric analysis; PN6: pristine nylon 6; NF20: 20 phr FaZn6Ca-loaded nylon 6 sample; NF40: 40 phr FaZn6Ca-loaded nylon 6 sample.
XRD analysis
Figure 8 demonstrates the X-ray diffractograms obtained for nylon 6 and nylon 6/FaZn6Ca composite samples, whereas the crystallinity values are listed in Table 5. Nylon 6 was found to have crystallinity of about 31.6%, while it showed its characteristic peaks at and 2θ = 21.4° and 23.7° in its X-ray diffractograms. The reflection peak at approximately 2θ = 23.7° belongs to nylon 6 crystal plane of α002/202 of α-form, while the reflection peak at approximately 2θ = 21.4° belongs to the crystal plane γ001 of γ-form of nylon 6. 32 While in case of FaZn6Ca characteristic hemimorphite peaks (2θ = 17.3°, 28.3°, and 33.6°) were observed in the nylon 6/FaZn6Ca composite samples, showing the presence of FaZn6Ca in nylon 6.
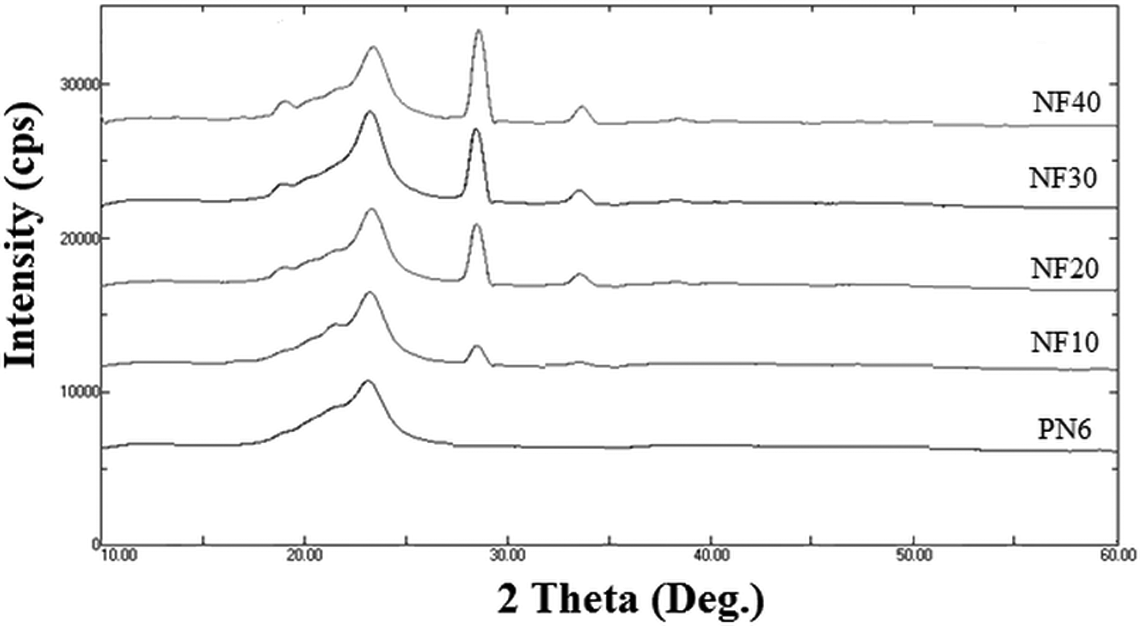
X-ray diffractograms obtained for nylon 6 and nylon 6/FaZn6Ca composites.
Crystallinity values obtained for the prepared nylon-6/FaZnCa composites.

PN6: pristine nylon 6; NF10: 10 phr FaZn6Ca-loaded nylon 6 sample; NF20: 20 phr FaZn6Ca-loaded nylon 6 sample; NF30: 30 phr FaZn6Ca-loaded nylon 6 sample; NF40: 40 phr FaZn6Ca-loaded nylon 6 sample.
It can be seen from Figure 8 that the intensity of characteristic hemimorphite peaks increased with increase in the concentration of FaZn6Ca in nylon 6, caused due to the increased presence of the hemimorphite phase on to the surface of FA. Also the crystallinity values of the composite increased with increased concentration of FaZn6Ca in nylon 6. NF40 was determined to have crystallinity of about 37.3%, which demonstrates an increase of about 18% as compared toPN6. The increase in crystallinity of nylon 6 with increased addition of FaZn6Ca is attributed to the better interactions happening between the two materials caused due to their hydrophilic nature, which led to molecular orientation of nylon 6 polymer chains about FaZn6Ca particles inducing nucleating effect and thus increasing crystallinity. This increase in crystallinity is the cause for the increase in the mechanical and thermal properties of nylon-6.
However, with increased addition of FaZn6Ca in nylon 6, the intensity of peak at approximately 2θ = 21.4° increased while that at approximately 2θ = 23.7° decreased, demonstrating the transformation of γ-form crystalline phase of nylon 6 to α-form. Interestingly, at higher loading of FaZn6Ca in nylon 6, high intensity peak is observed at 2θ = 21.4°, demonstrating the presence of very large proportion or dominance of α-form of nylon 6 crystalline phase over that of γ-form. The crystalline phase of nylon 6 is influenced by the thermal conditions, stress applied, moisture content, and type and concentration of additives. Usually, rapid cooling or low temperature crystallization promotes the γ-form of nylon 6, while higher crystallization temperatures or slow cooling ratio promotes its α-form. Thus, it can be said that addition of FaZn6Ca in nylon 6 promotes the α-form of nylon 6.
Rheological property
Rheological analysis was undertaken to understand the effect of FaZn6Ca addition on the flow behavior of nylon 6. Figures 9 and 10 are the plots for viscosity versus shear rate and damping factor versus angular frequency, respectively, obtained for the prepared nylon 6/FaZn6Ca composites.
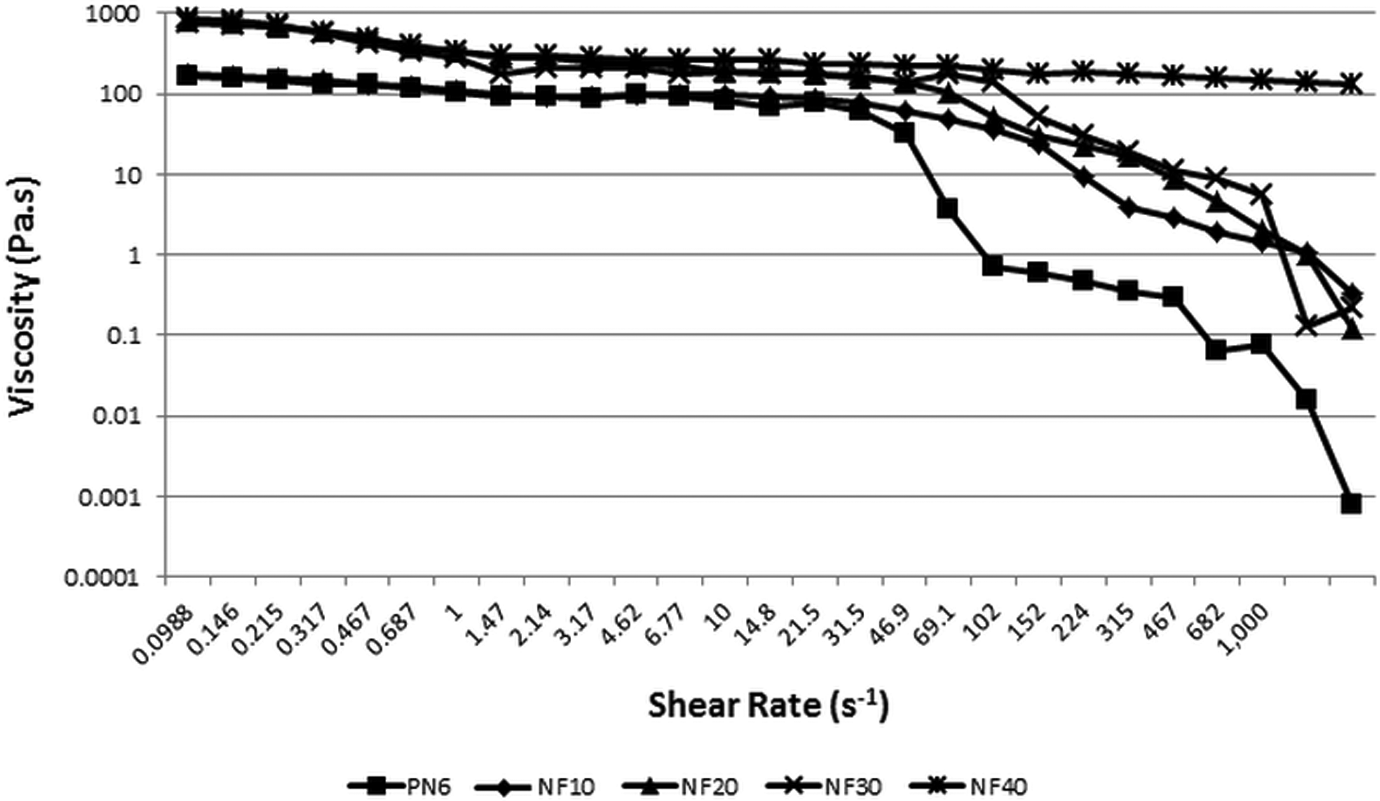
Plot of viscosity versus shear rate obtained for nylon 6 and nylon 6/FaZnCa composites.
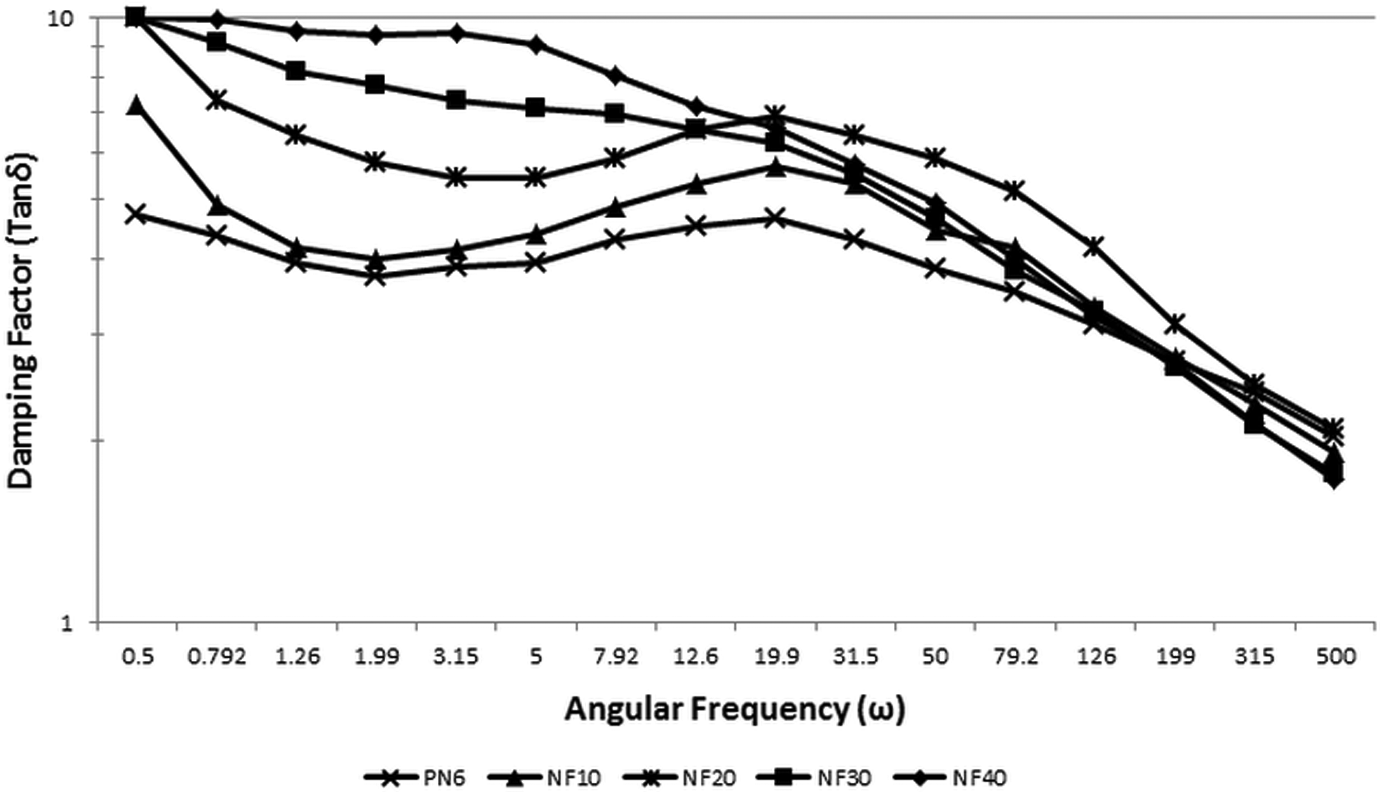
Plot of damping factor versus angular frequency obtained for nylon 6 and nylon 6/FaZnCa composites.
Figure 9 shows that the viscosity of nylon 6 increased with increase in concentration of FaZn6Ca. FaZn6Ca and nylon 6, both being hydrophilic, had better compatibility with each other, leading to better interaction. In addition, the spiny structure formed on the FaZn6Ca by the zinc hydroxide and calcium hydroxide modification increased the effective surface area for interaction with nylon 6 polymeric chains. This increase in the effective surface area and thus the interaction increased with increase in concentration of FaZn6Ca; which led to increase in resistance of this molten composite structure to the rotating spindle. This resistance increased with increase in FaZn6Ca concentration. Viscosity increased appreciably compared to the concentration of FaZn6Ca added. Thus, extrusion and injection molding processes would require higher force for processing the FaZn6Ca-filled nylon 6. NF40 would require the highest force. This, however, would prove beneficial in improving the melt viscosity and thus decreasing the drooling nature of nylon 6 at processing temperature.
Damping factor also increased with increase in concentration of FaZn6Ca in nylon 6. Damping factor is a ratio of loss modulus to storage modulus. Higher the damping factor, higher is the stiffness of the composite, which correlates strongly with the obtained mechanical properties. FaZn6Ca being a particulate filler increases the stiffness of the polymer rather than inducing flexibility. Better interaction between FaZn6Ca and nylon 6, caused by better interaction and increased surface area for interaction, and the induced crystallinity, increased the stiffness of the composite. This property is evident from the damping factor property.
Morphological property
SEM analysis was undertaken to understand the dispersion and interaction between FaZn6Ca filler particles and nylon 6. SEM images obtained for NF10 (a), NF20 (b), NF30 (c), and NF40 (d) are shown in Figure 11.
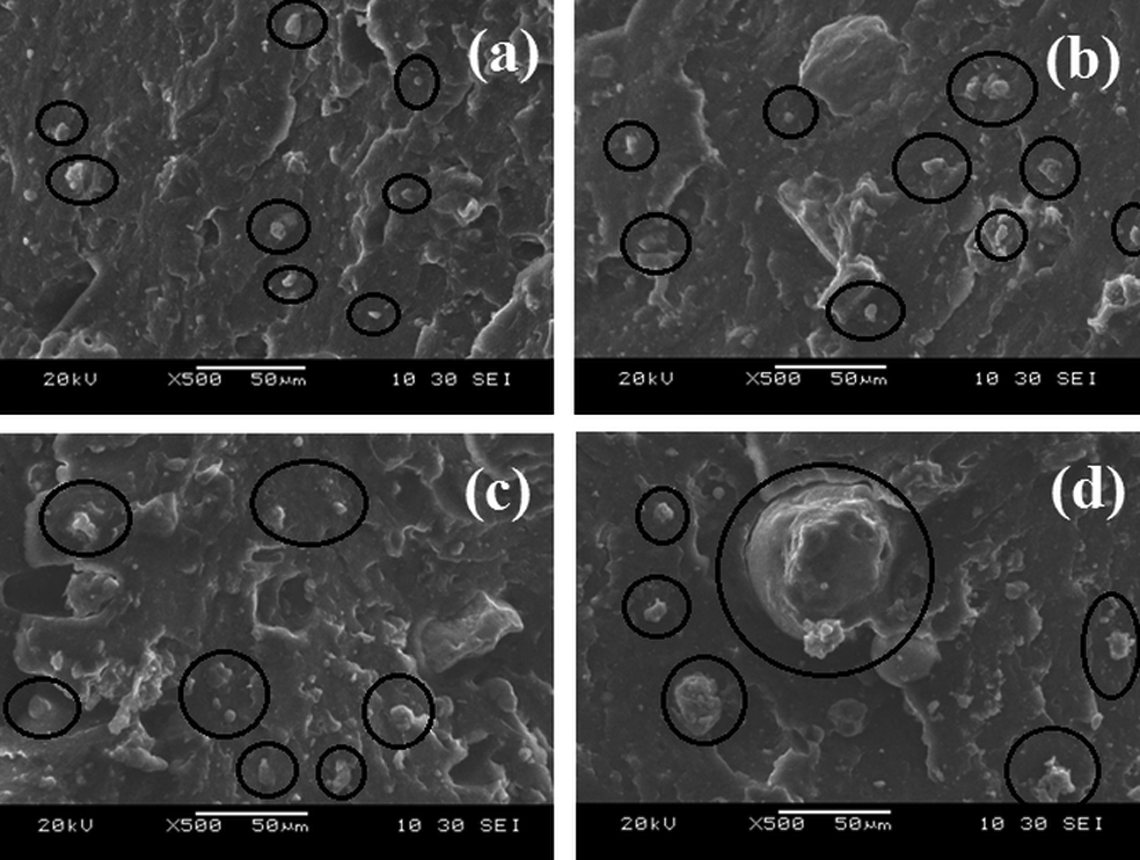
Scanning electron micrographs obtained for (a) NF10, (b) NF20, (c) NF30, and (d) NF40. NF10: 10 phr FaZn6Ca-loaded nylon 6 sample; NF20: 20 phr FaZn6Ca-loaded nylon 6 sample; NF30: 30 phr FaZn6Ca-loaded nylon 6 sample; NF40: 40 phr FaZn6Ca-loaded nylon 6 sample.
FaZn6Ca can be seen to have uniformly dispersed in the nylon 6 polymer matrix up to the concentration of 20 phr. However, it started forming aggregates at 30 phr concentration (NF30), while agglomerates can be seen in NF40. Size of agglomerate observed in NF40 is very large. Whereas the aggregate seen in NF30 is very small. With higher concentration of FaZn6Ca in nylon 6 (NF30 and NF40), the polymer matrix might prove insufficient to completely wet the surface area of the FaZn6Ca, making them form aggregates and agglomerates. Interestingly, even though aggregates and agglomerates were seen in the SEM images of the NF30 and NF40 composite samples, they still demonstrate improved mechanical properties compared to lower concentration FaZn6ca added nylon 6 composites. This is attributed to the interfacial friction occurring between the particles and the polymer matrix caused due to the spiny structure formed on the surface of FA due to the surface treatment of FA with zinc hydroxide and calcium hydroxide.
Second, FaZn6Ca particles can be seen to have very good adhesion with the nylon 6 polymer matrix, as the interface between the two is very smooth and uniform, without any presence of void structures. Thus, even though aggregates and agglomerates were formed by FaZn6Ca at higher concentrations, no regions of stress concentrates were generated, due to the proper interactions happening FaZn6Ca and nylon 6 as both the materials are hydrophilic in nature. Therefore, nylon 6 is able to accommodate up to 40 phr concentration of FaZn6Ca and demonstrate improved mechanical properties even though aggregates and agglomerates were formed by FaZn6Ca at higher concentrations.
Conclusions
Purified FA was successfully surface modified using zinc hydroxide and calcium hydroxide. Weight ratio of FA to zinc hydroxide was varied as 5:0.4, 5:0.6, and and 5:0.8 while that of FA to calcium hydroxide was maintained constant at 5:1. FA modified with FA to zinc hydroxide ratio of 5:0.6 and FA to calcium hydroxide ratio of 5:0.1 provided FA (FaZn6Ca) with highest spine-structured rough surface morphology; which has the ability to provide higher level of surface area to interact with the polymer chains as compared to unmodified FA and was utilized as filler in nylon 6. Concentration of FaZn6Ca was varied from 0 phr to 40 phr in nylon 6. Better compatibility and increased surface area caused by the spine-structured surface morphology of FaZn6Ca increased the effective surface area for interacting with nylon 6 polymeric chains improving mechanical, thermal, and rheological properties. FaZn6Ca formed agglomerates at higher concentration (30 and 40 phr); however, the spine-structured structure helped maintain improvement in properties mentioned above. Spin structure of FaZn6Ca makes it an effective filler for nylon 6.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support provided by The University Grant Commission (UGC) under the SAP-DRS scheme for the present work.
