Abstract
An epoxy macromolecular chain extender, polymethyl methacrylate-co-glycidyl methacrylate (PMMA-co-GMA), was synthesized by continuous random free radical polymerization, which was introduced into poly(ethylene terephthalate) (PET) and PET blends. The modified PET blends exhibited higher torque with increasing the content and the polymerization degree of PMMA-co-GMA. The rise of torque after initial melting period was observed, which indicated the occurrence of reaction between the epoxy group of PMMA-co-GMA and hydroxyl or carboxyl end groups of PET. The melting temperature, crystallization temperature, and crystallization degree of PET blends decreased. The chain extender was introduced in PET/ethylene–propylene–diene monomer (EPDM)-graft-GMA (EPDM-g-GMA) blends with the composition of 85/15, which exhibited brittle fracture behavior. The blends, in which the chain extender content in PET was over 8 wt%, exhibited ductile fracture behavior. EPDM-g-GMA was used to toughen PET with 8 wt% chain extender. The brittle–ductile transition took place between the rubber content of 10 wt% and 15 wt%. The transition was in advance when compared with PET/EPDM-g-GMA blends. The content of chain extender in PET showed little influence on the compatibility of PET blends. The macromolecular chain extender was an effective extender for PET. The introduction of macromolecular chain extender showed contribution for the toughening PET.
Keywords
Introduction
Poly(ethylene terephthalate) (PET) has been widely used in applications such as bottles, fibers, films, and engineering products due to its good comprehensive properties, such as transparency, chemical resistance, thermal resistance, and barrier properties. 1 –5 However, during melt processing the molecular weight, mechanical performance, and thermal properties of PET may be easily reduced when subjected to thermal oxidation and hydrolytic degradation, 6 which limited its processability such as blow molding and extrusion foaming. 7
A general method to increase the molecular weight of PET is chain extension reaction, which is rather simple, less expensive, and can be conducted in ordinary processing equipments like extruder and injection molding machine. 8 –13 In this process, a polyfunctional low-molecular-weight compound reacts with PET end groups to rejoin the polymer chain broken during processing and use. 14 –18 There are many publications reporting the success achieved with the chain extension process using PET with various types of chemical compounds, 19 and also, some work was carried out in the industrial stage such as injection blow molding and extrusion. 20,21 Some chain extenders used in these investigations include bis-2-oxazolines, diepoxides, triphenyl phosphite, and bisphenol-A dicyanatedianhydrides. 22 –26
In this work, an epoxide group was introduced as a reactive functional group onto polymethyl methacrylate (PMMA) with an expectation to enhance molecular weight, mechanical performance, and thermal properties of PET. The epoxide group has good reactivity toward hydroxyl and carboxyl groups of PET. The modification reaction was carried out in an internal mixer using different concentrations of chain extender and various polymerization degrees. Torque changes were a direct representation of the modifications induced in the molecular structure during the chain extension and thermal degradation in the system. In order to investigate the effect of chain extender PMMA-co-GMA on toughening PET, the morphological and mechanical properties of the PET/ethylene–propylene–diene monomer (EPDM)-grafted GMA (EPDM-g-GMA)/PMMA-co-GMA blends were discussed. The brittle–ductile transition in PET/EPDM-g-GMA/PMMA-co-GMA blends was in advance in comparison with PET/EPDM-g-GMA blends. The amount of rubber and the production cost were reduced.
Experiment
Materials
Granules of virgin PET under the trade name of J155, with the intrinsic viscosity of 0.9 dl g−1, were provided by Sanjing (Bangkok, Thailand). It is a grade specifically designed for the production of carbonated soft drink bottles in the injection blow molding process. It was used after drying in vacuum at 100°C for 24 h to remove sorbed water completely. EPDM elastomer (KEP 570P, ethylene content 67 wt%, 5-ethylidene-2-norbornene content 4.8 wt %,), with the Mooney viscosity of 53 at 125°C, was supplied by Kumho Polychem Co.(Seoul, Korea). Dicumyl peroxide (DCP, Akzochem, Shanghai, China), a radical initiator was used as received. Methyl methacrylate (MMA) and GMA were supplied by Suzhou Amway Reagent Factory (Suzhou, China). Toluene, di-tert-butyl peroxide, and dodecyl mercaptan were provided by BASF (Shanghai, China).
Preparation of chain extender
The copolymer extender was synthesized by random free radical polymerization with continuous bulk copolymerization in our laboratory. The schematic diagram for the polymerization equipment is shown in Figure 1. The components were fed into the premix container according to the polymerization ingredient shown in Table 1 and then stirred. The monomer mixture was fed into the reactor with a capacity of 5000 ml by the feeding pump. The reactor was then swept by nitrogen gas to deaerate oxygen. The reactor was heated to 130°C. Polymerization took place, and the exothermic reaction raise the system temperature to about 140°C. The system became ropy with the rising of the percent conversion. The monomer mixture was fed into the reactor continuously by the feeding pump at the speed that the monomer mixture shown in Table 1 was fed within 2 h. The melt pump was run simultaneously at the same speed to transfer the mixture from the reactor to the twin-screw extruder. The melt was purified by devolatilization in the twin-screw extruder at the temperature of 210°C in vacuum with the pressure of about 10 kPa and then pelletized. The system reached a balance after five batches of monomer mixture was fed. The pellets were collected for use. Polymerization was stopped when the pellets were enough for use. The yield rate of 60 wt% was calculated by gravimetric analysis. The copolymers were marked as PG0, PG4, PG8, and PG12 in this work. The molecular weight of PMMA–GMA copolymer was estimated by gel permeation chromatography (Waters410, Milford, Massachusetts, USA), and the results are listed in Table 2. The analysis was calibrated with polystyrene standards. The rheological properties of virgin PET and copolymer extenders at 240°C are shown in Figure 2. It revealed that the balance torque value of virgin PET was little higher than that of chain extenders. The balance torque value of chain extenders showed no difference, which were approximated to that of virgin PET.
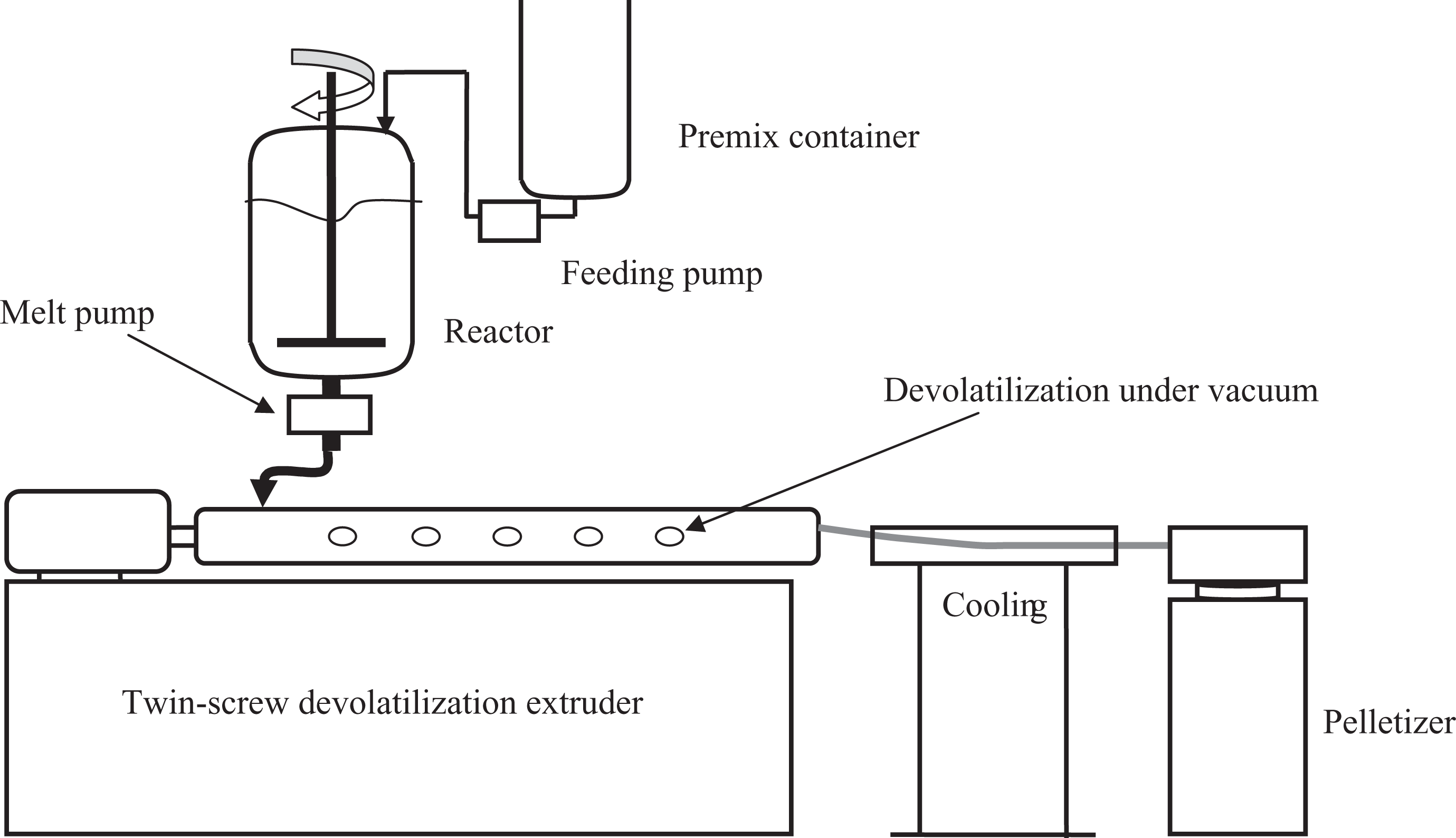
Schematic diagram for the polymerization equipment.
Polymerization ingredient for the copolymer extender.
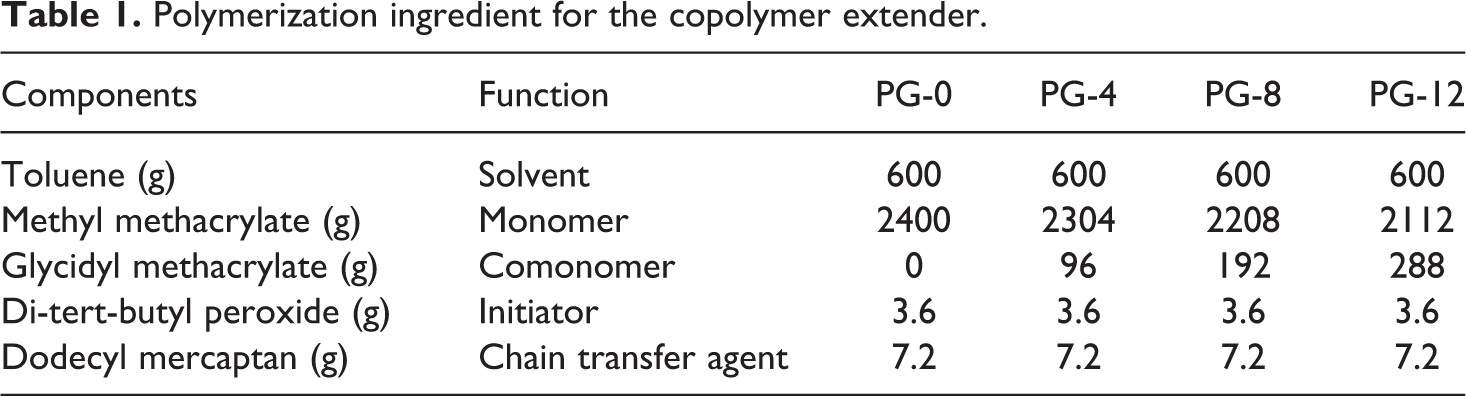
Molecular weight of copolymer extender.
Mn: number-average molecular weight; Mw: weight-average molecular weight.
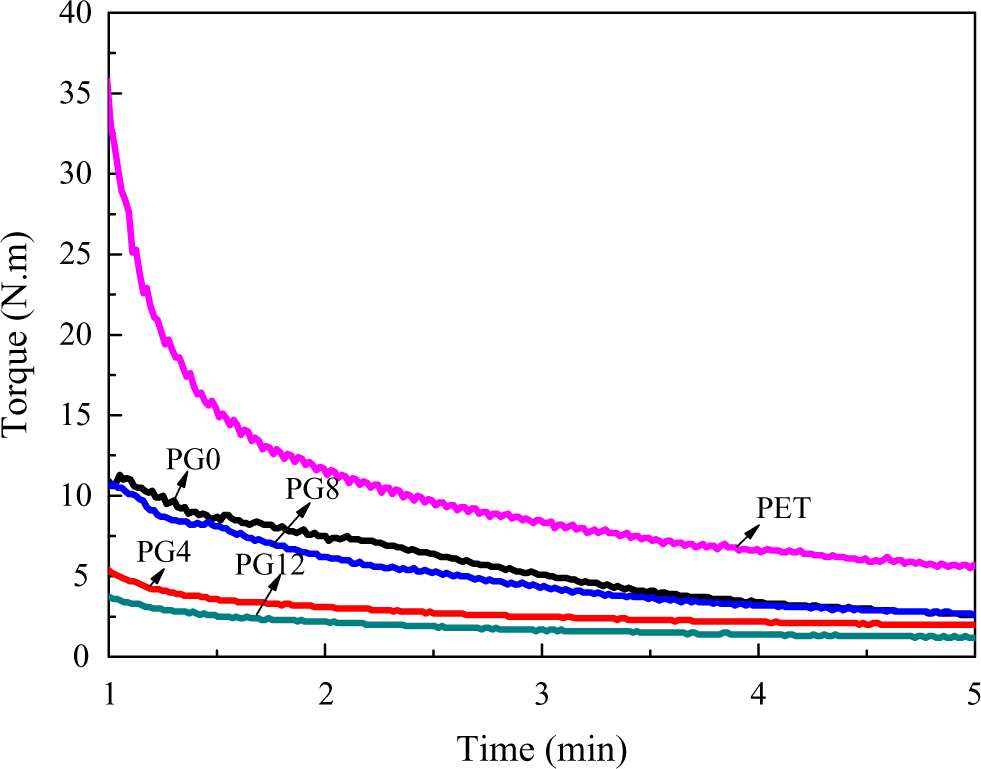
Rheological properties of PET and copolymer extenders. PET: poly(ethylene terephthalate).
Graft copolymerization of GMA onto EPDM
Grafting of GMA onto EPDM, with DCP as an initiator, was carried out in a twin-screw extruder. Styrene was introduced to increase the graft ratio of GMA onto EPDM. EPDM (2000 g) was added with GMA (60.0 g)/DCP (10.0 g)/styrene (60.0 g) solution, the temperature along the extruder were 170, 170, 180, 180, 180, 180, and 180°C, and the rotation speed was 100 r min−1. The blends were cooled in a water bath and then pelletized.
Blend preparation
Chain extension reaction was carried out in an internal mixer (Haake, Rheomix 600, Thermo Electron (Karlsruhe) GmbH, Karlsruhe, Germany). The temperature inside the mixing chamber was 240°C, and the roller speed was 60 r min−1. The mixing continued for about 30 min to ensure the completion of the reaction. The chain extender PG12 was also introduced in PET/EPDM-g-GMA blends; the concentration of chain extender in PET was varied from 0.0 wt% to 20.0 wt%. The total weight was 65 g. The content of EPDM-g-GMA was kept at 15 wt%. The blending ingredient for PET/PG12/EPDM-g-GMA blends was illustrated in Table 3.
Blending ingredient for PET/PG12/EPDM-g-GMA blends.
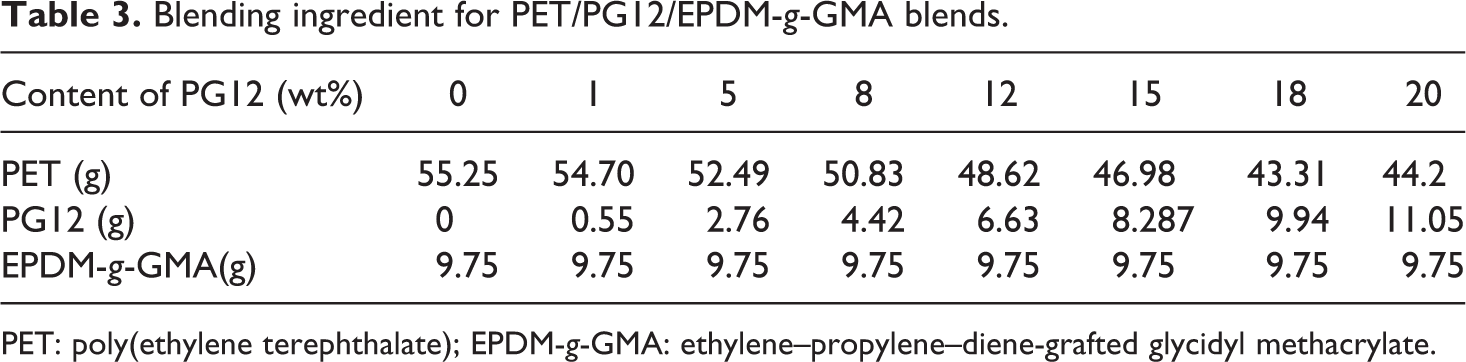
PET: poly(ethylene terephthalate); EPDM-g-GMA: ethylene–propylene–diene-grafted glycidyl methacrylate.
Mechanical tests
Samples with dimensions 63.5 × 12.7 × 3.14 mm3 were milled from thermal molded plate. The notch was milled with a depth of 2.54 mm, an angle of 45°, and a notch radius of 0.25 mm. Notched Izod impact tests were performed at 23 ± 2°C according to ASTM D256 standard on an XJU-22 apparatus (Chengde COTS Scientific Instruments Co. Ltd., Chengde, China). The tensile test bars were cut from thermal molded plate with the thickness of 1 mm. The tensile tests were carried out with an AGS-H tensile tester (Shimadzu Corporation, Kyoto, Japan) at a crosshead speed of 50 mm min−1 at room temperature according to ASTM D638 standard.
Scanning electron microscopy
For comparison of the morphology of each blend, the cryogenically fractured surfaces were investigated using scanning electron microscopy (JSM-6500, JEOL Japan Electronics Co. Ltd, Tokyo, Japan). The samples were fractured after dipping in liquid nitrogen and then the fractured surfaces were etched in cyclohexane at least for 24 h to remove the rubber phase on the surface, dried in the oven, and then sprayed with a thin layer of gold before scanning.
Thermal analysis
Thermal analysis was carried out using a differential scanning calorimeter (DSC-7; Perkin-Elmer, Shanghai, China) to study the influence of variety concentration of chain extender on thermal behavior of PET/PG blends. First, calefactive scan was abandoned to erase the thermal history, then the temperature was raised from 40°C to 350°C at a rate of 10°C min−1 and then it was cooled to 50°C at 10°C min−1. All measurements were performed under nitrogen atmosphere.
Dynamic mechanical properties
Dynamic mechanical properties of the blends were studied with Perkin–Elmer 7 dynamic mechanical analysis (DMA) equipment (Shanghai, China) with the aim to evaluate the possible changes in glass transition temperatures (Tgs) induced by compatibilization. The temperature range studied was from −80°C to 200°C at a heating rate of 3°C min−1, and the frequency was 1 Hz.
Results and discussion
Chain extension reaction
As well known, torque changes were a direct representation of the modifications induced in the molecular structure during the chain extension and thermal degradation in the system. The torque evaluation of PET blends was investigated. The effect of PG12 content on torque values during melt processing of PET was shown in Figure 3. The torque values were measured as a function of time for the virgin PET and the PET with 5, 10, 15, and 20 wt % of PG12. As expected, after an initial melting period, the virgin PET shows a continuous decrease in the torque values, as a result of chain disentanglements and orientation in the flow direction. On the contrary, for other samples with chain extender, a peak was observed in the torque curves in initial melting period. With the extension of reaction time, PET/PG12 blends showed a drastic increase in torque values, which were observed after 5 min. The torque maximum value of 8, 14, 20.5, and 23.4 N m occurred at 17, 15, 12, and 8.5 min for PET blends, respectively. The more the chain extender in the blends, the earlier and higher the maximum torque value was observed. After reaching a maximum, the torque values started to decrease steadily and leveled off a certain value higher than virgin PET. This decrease was an indication that thermal degradation was taking place during processing. The increment of torque value after initial melting period was the direct representation of the chain extension reaction.
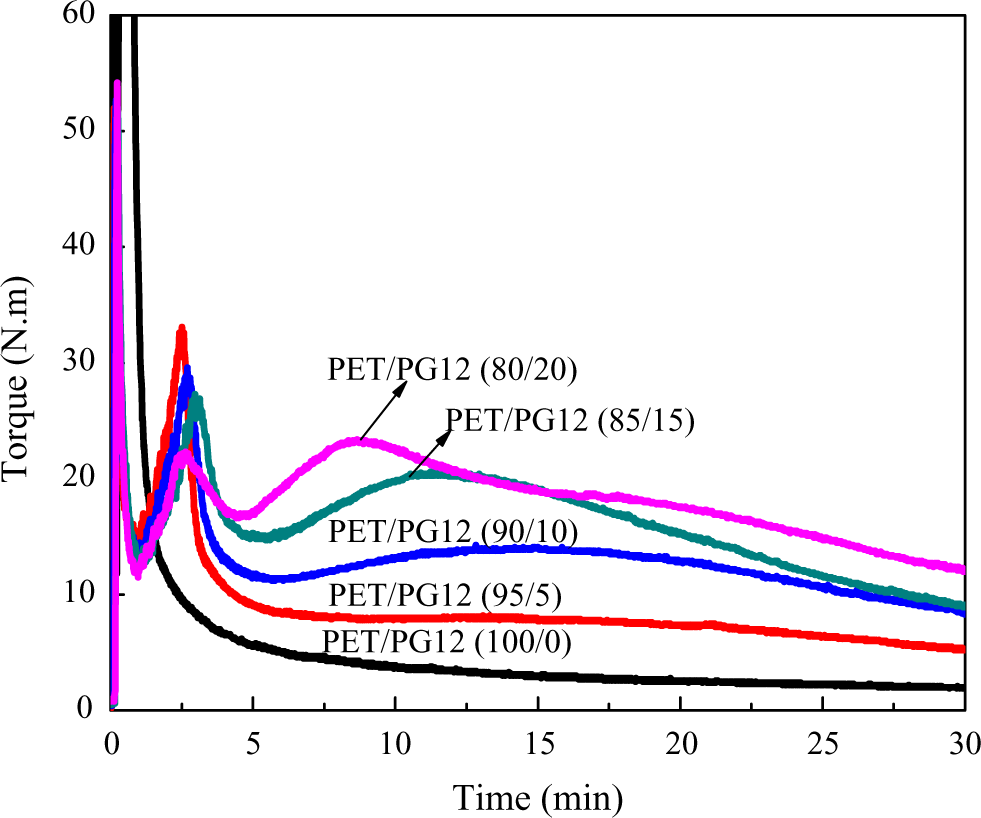
The effect of PG12 content on the torque of PET blends. PET: poly(ethylene terephthalate).
The blends of PET and chain extender with different polymerization degree showed similar behavior. The content of chain extender was kept at 20 wt%. The more the polymerization degree of the chain extender, the higher and the earlier the maximum torque exhibited, as shown in Figure 4. However, it was worth to note that the balance torque value of PET/PG0 was approximated to that of virgin PET, indicating that the processing property of PMMA was approximate to virgin PET and epoxy functional groups in PMMA-co-GMA played a crucial role. The proposed mechanism for the reactions leading to the chain extension of PET in the presence of PMMA-co-GMA is shown in Figure 5. The reaction took place between epoxy groups in PMMA-co-GMA and hydroxyl or carboxyl end groups in PET. These results suggested that PMMA-co-GMA was an effective modifier for PET.
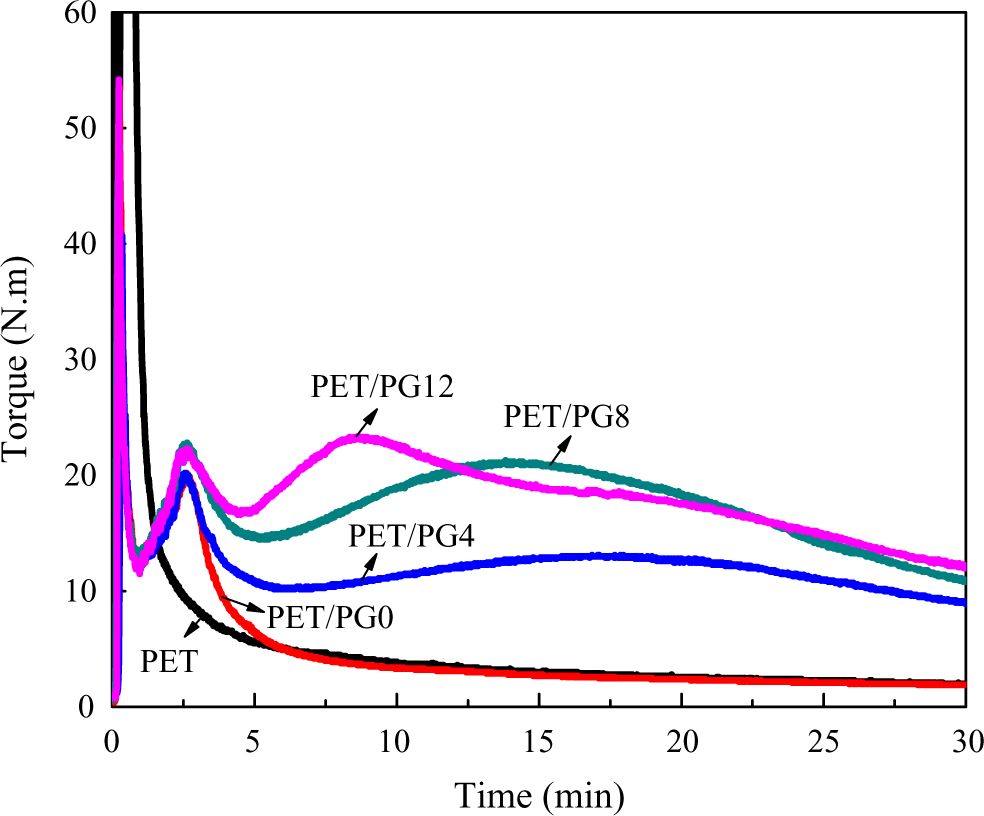
The effect of polymerization degree of PMMA-co-GMA on the torque of PET blends. PMMA-co-GMA: polymethyl methacrylate-co-glycidyl methacrylate; PET: poly(ethylene terephthalate).
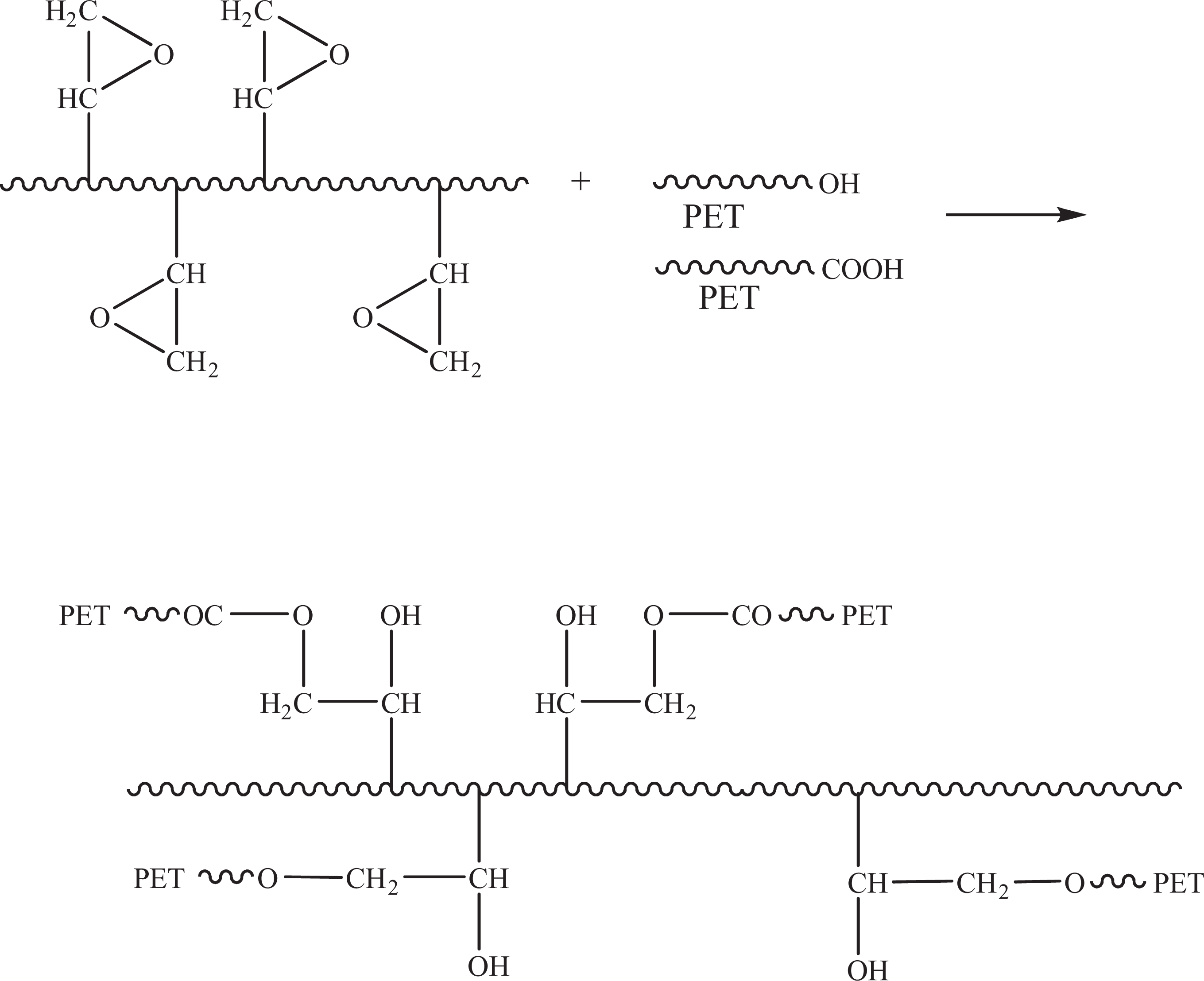
Schematic representation of reaction mechanism.
Thermal properties
Although PET has high symmetrical structure in the macromolecular chain, it is a well-known semicrystallization polymer with a low rate of crystallization nature. As well known, the crystallization temperature (Tc) and melting temperature (Tm) are always used to characterize the symmetry of macromolecular chain and the density of branch point. In this study, the crystallization behaviors of virgin PET and modified PET during heating and cooling processes were investigated using DSC. The influence of concentration of chain extender on the thermal properties of PET blends is shown in Figure 6. It revealed that both Tc and Tm of modified PET shifted to lower temperature, and the peak area reduced by increasing the concentration of PG12 compared with the virgin PET. The influence of polymerization degree of chain extender on the thermal properties of PET blends is shown in Figure 7. The content of chain extender was kept at 20 wt%. The blends of PET and chain extender with different polymerization degree showed similar behavior to the blends of different contents of PG12. The values of Tm and Tc are illustrated in Tables 4 and 5. The area under melting curve was used to calculate the degree of crystallinity (Xc) using the following equation: Xc(%) = (ΔHm/ΔHm0) × 100; where ΔHm0 is the melting enthalpy of 100% crystalline PET (ΔHm0 = 122.6J g−1) 27 and ΔHm is the melting enthalpy of the samples. The results are illustrated in Tables 4 and 5 to compare the effect of different PET blends on crystallization degree.
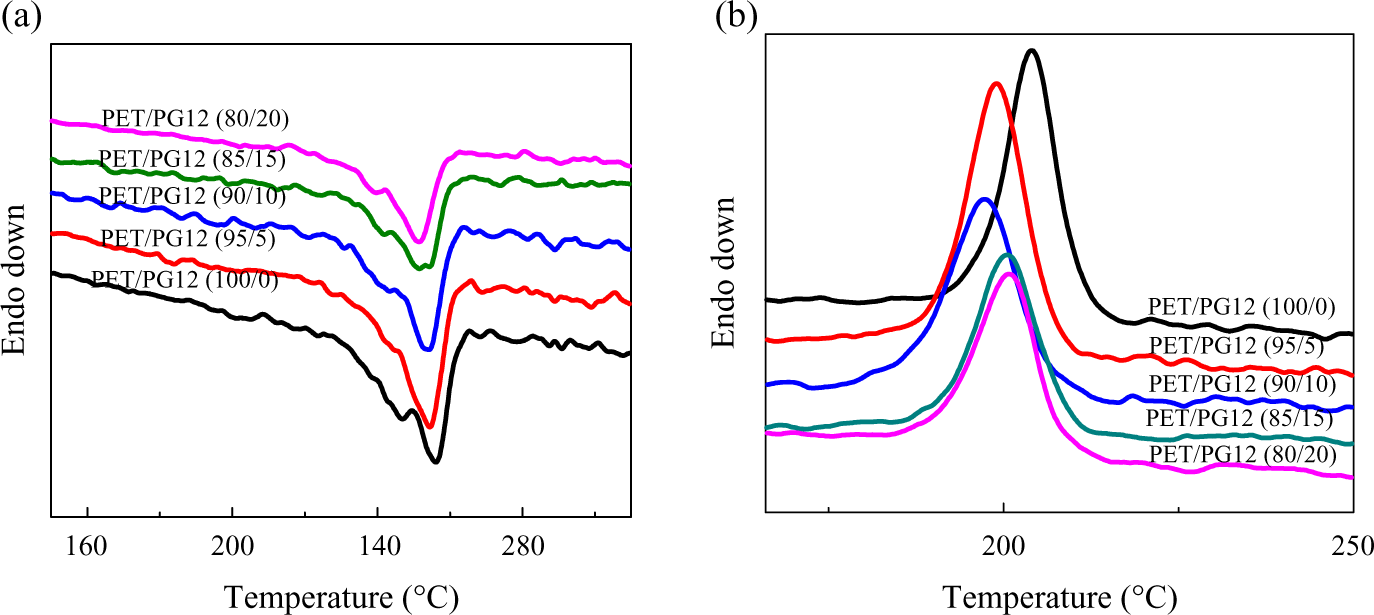
DSC heating scans (a) and cooling scans (b) of PET blends with different content of PG12 at the rate of 10°C min−1. DSC: differential scanning calorimetry; PET: poly(ethylene terephthalate).
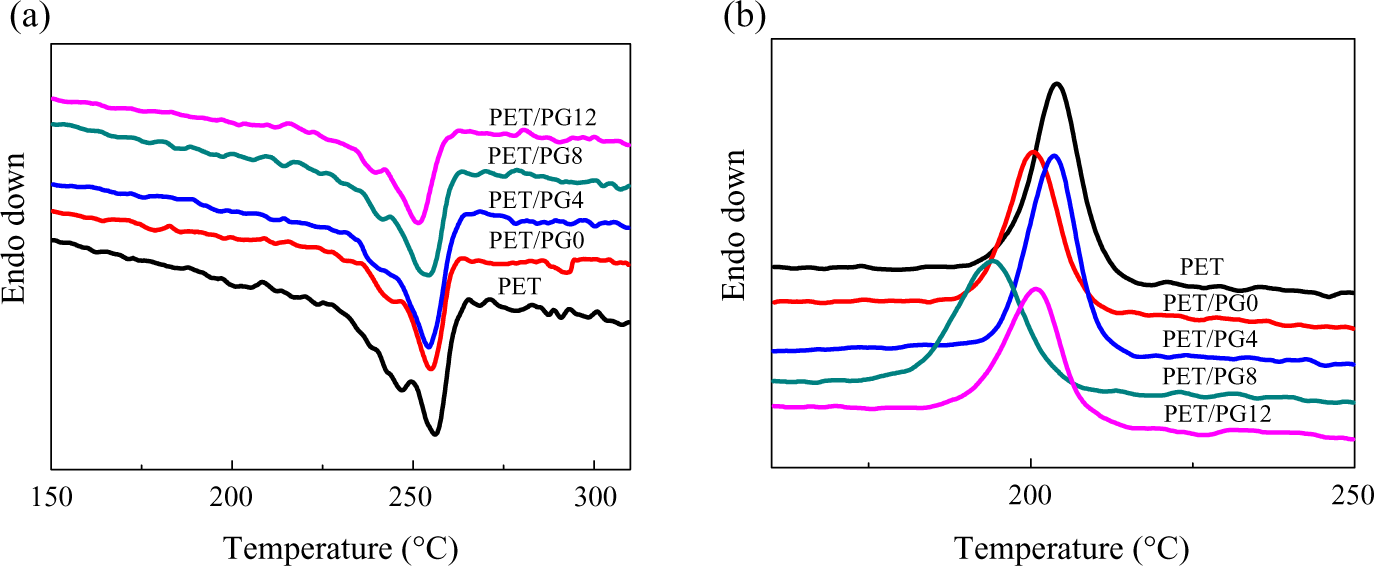
DSC heating (a) and cooling (b) thermograms of PET blends with different polymerization degree of PG at the rate of 10°C min−1. DSC: differential scanning calorimetry; PET: poly(ethylene terephthalate).
DSC data of PET blends with different content of PG12.
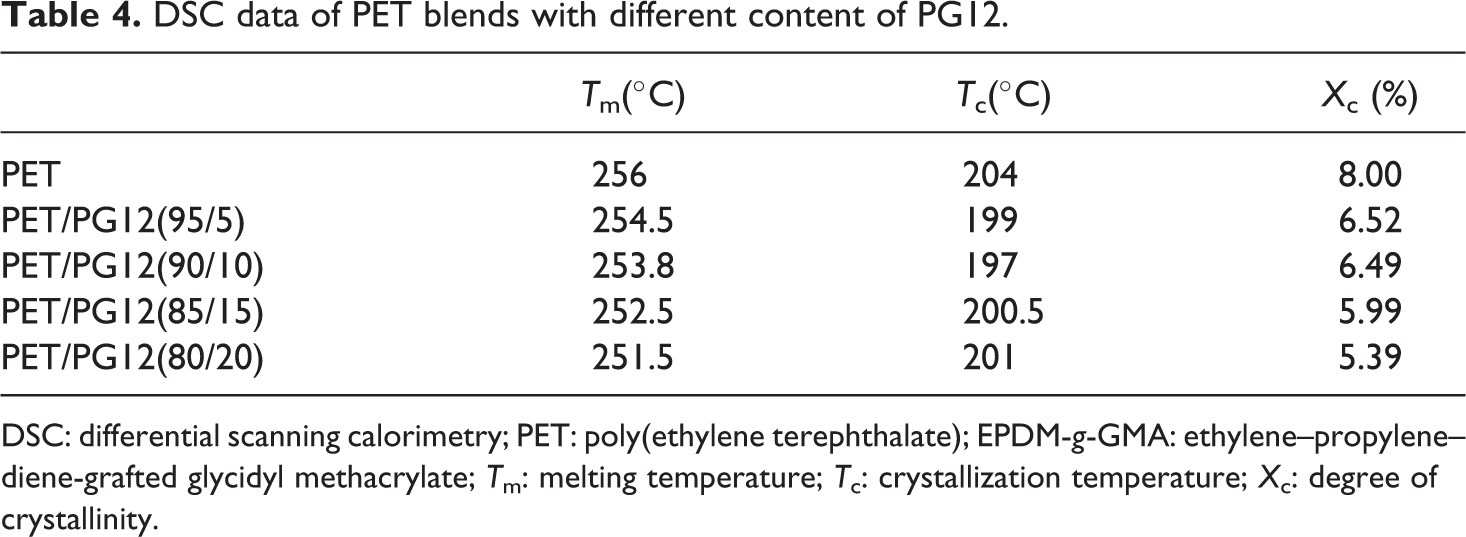
DSC: differential scanning calorimetry; PET: poly(ethylene terephthalate); EPDM-g-GMA: ethylene–propylene–diene-grafted glycidyl methacrylate; Tm: melting temperature; Tc: crystallization temperature; Xc: degree of crystallinity.
DSC data of PET blends with different polymerization degree of PG.
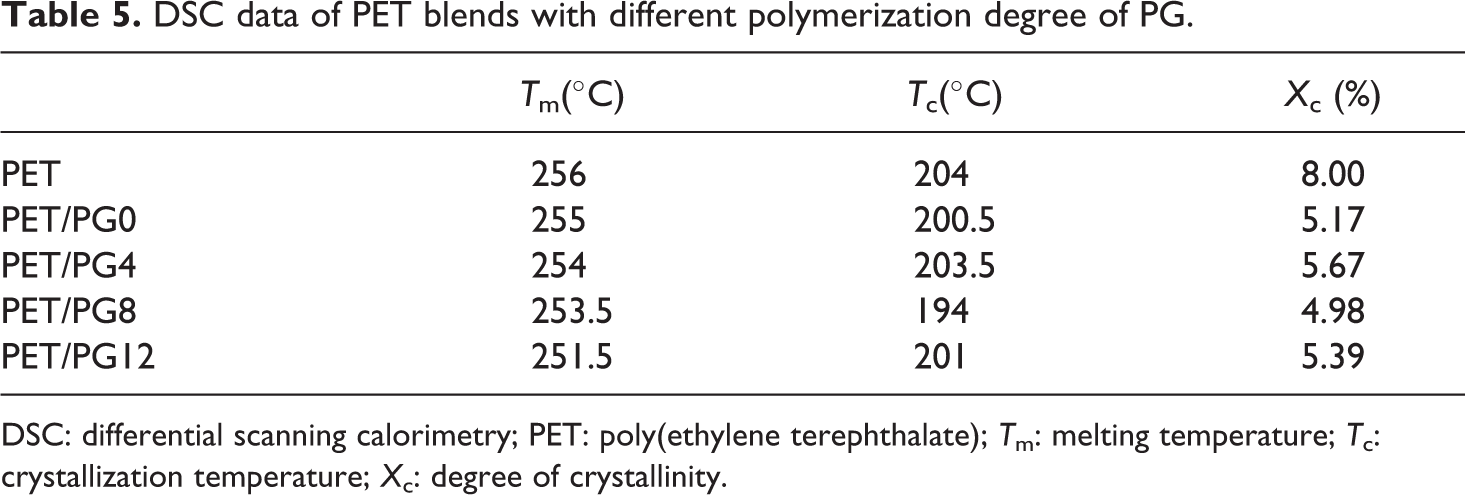
DSC: differential scanning calorimetry; PET: poly(ethylene terephthalate); Tm: melting temperature; Tc: crystallization temperature; Xc: degree of crystallinity.
Increasing the content of chain extender or enhancing the polymerization degree of chain extender induced to lower crystallization rate for modified PET. This can be attributed to the chemical reaction took place between epoxy functional groups in PMMA-co-GMA and carboxyl or hydroxyl end groups of PET. The increment of branches by high extender concentration and polymerization degree destroyed the symmetrical structure of PET molecular and increased the amount of amorphous phase. These made the crystallization of PET chain difficult. So, Tc and Xc shifted to lower value.
Mechanical properties
EPDM and EPDM-g-GMA were introduced as impact modifier of PET. The impact strengths of PET/EPDM blends and PET/EPDM-g-GMA blends are compared in Figure 8. Within the range of rubber content investigated, the impact strengths of PET/EPDM-g-GMA blends were higher than those of PET/EPDM blends. The notched impact strength of pure PET was about 17 J m−1, while the impact strength of PET/EPDM blends was about 25 J m−1. All PET/EPDM blends fractured in brittle mode. EPDM is not an effective impact modifier for PET because of the poor compatibility. However, the impact strengths of PET/EPDM-g-GMA blends improved significantly with the increment of EPDM-g-GMA. The impact strength value of 840 J m−1 was achieved at the rubber content of 30 wt %, which displayed super toughness. The results showed that brittle–ductile transition took place between the rubber content of 15 and 20 wt%. On the contrary, the impact strength of PET/EPDM-g-GMA blends decreased slightly with the increment of EPDM-g-GMA content over 30 wt %. The slight decrease in impact strength can be related to the destruction of excessive rubber on the structure of matrix. EPDM-g-GMA is an effective impact modifier for PET. The introduction of GMA onto EPDM improved the compatibility between PET and EPDM.
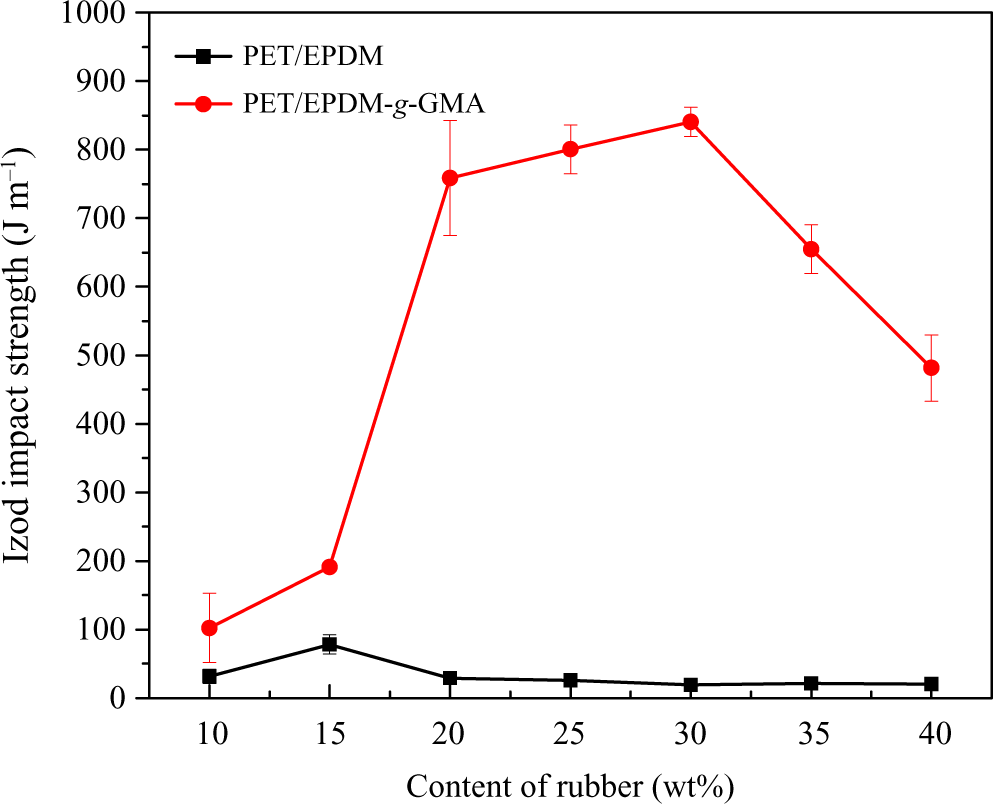
Effect of rubber content on Izod impact strength of PET blends. PET: poly(ethylene terephthalate).
The effect of rubber content on tensile strength of PET blends is shown in Figure 9. The tensile strength values of PET/EPDM-g-GMA blends gradually decreased with the increasing EPDM-g-GMA content. This is due to the introduction of excessive elastomeric nature of rubber with lower tensile strength.
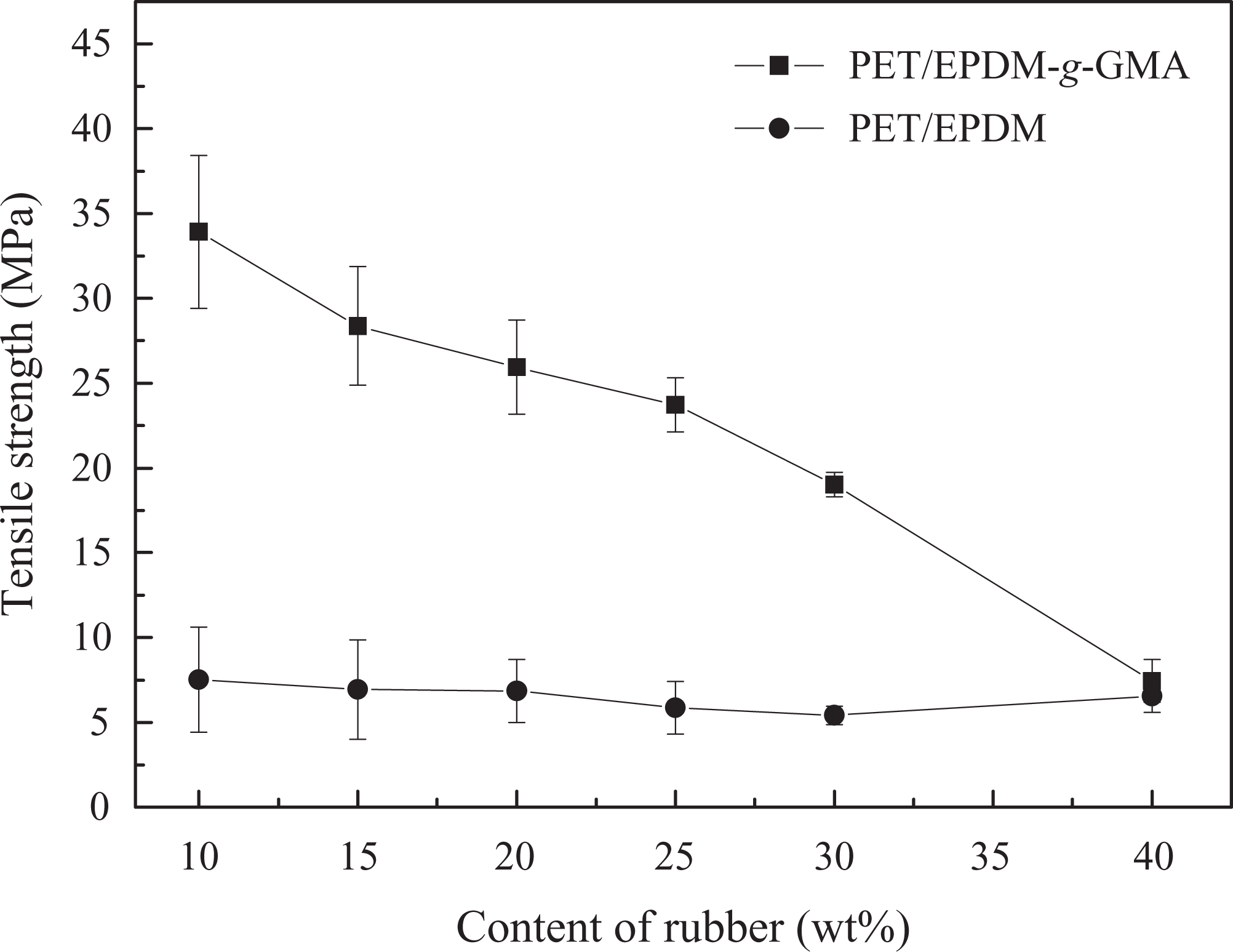
Effect of rubber content on tensile strength of PET blends. PET: poly(ethylene terephthalate).
The effect of chain extender concentration on 15 wt% EPDM-g-GMA toughened PET was investigated. The chain extender and PET formed homogeneous matrix phase when PET, PG12, and EPDM-g-GMA were blended together. The content of EPDM-g-GMA was kept at 15 wt%. The influence of concentrations of PG12 in matrix on the impact strength of PET/PG12/EPDM-g-GMA blends is shown in Figure 10. It illustrated that the impact strength of PET blends showed no difference with the incremental of the PG12 content in matrix from 0 wt% to 5 wt%. However, the impact strength of PET/EPDM-g-GMA blends increased significantly with the increment of the PG12 content from 5 wt% to 8 wt%. The notched impact strength of PET/EPDM-g-GMA 85/15 was about 190 J m−1. The impact strength value of 570 J m−1 was achieved at the chain extender content of 8 wt%. The addition of 8 wt% chain extender in matrix induced the brittle–ductile transition in PET/EPDM-g-GMA 85/15 blends. The content of chain extender in matrix showed little effect on the impact strength of PET blends when the PG12 content was over 12 wt%. It indicated that the introduction of macromolecular chain extender was beneficial for the toughening of PET. It was attributed to the increasing of amorphous phase induced by the chemical reaction, as shown in Figure6(b).
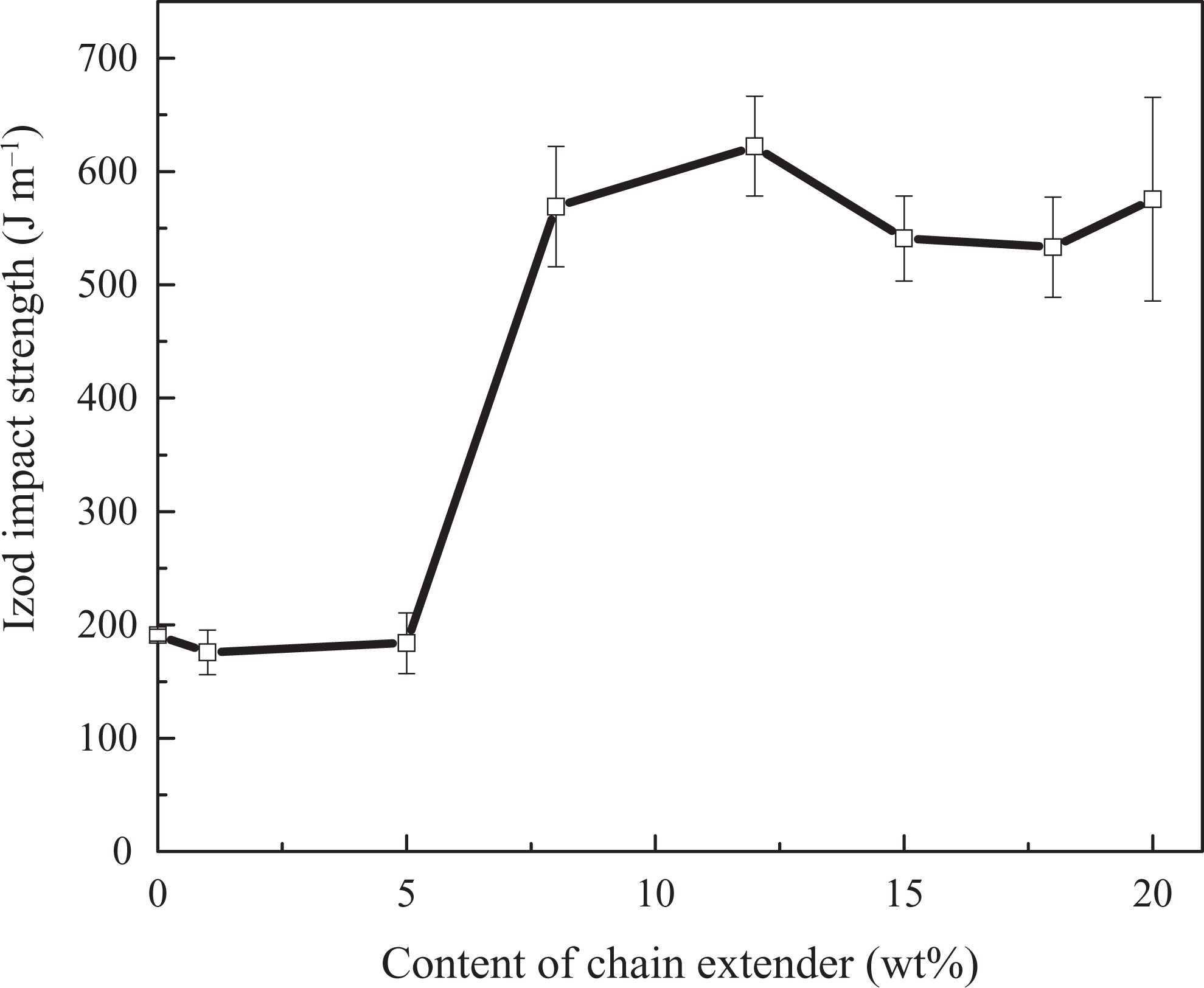
Effect of chain extender content in PET on Izod impact strength of PET/PG12/EPDM-g-GMA blends. PET: poly(ethylene terephthalate); EPDM-g-GMA: ethylene–propylene–diene monomer-grafted glycidyl methacrylate.
The effect of EPDM-g-GMA content on impact strength of different PET blends is shown in Figure 11. The content of PG12 was kept at 8 wt% in PET matrix. The brittle–ductile transition took place between the rubber content of 15 wt% and 20 wt% in PET/EPDM-g-GMA blends. However, the brittle–ductile transition took place between the EPDM-g-GMA content of 10 wt% and 15 wt% in the presence of 8 wt% chain extender in matrix. Compared with PET/EPDM-g-GMA blends, the brittle–ductile transition was in advance with the introduction of chain extender.
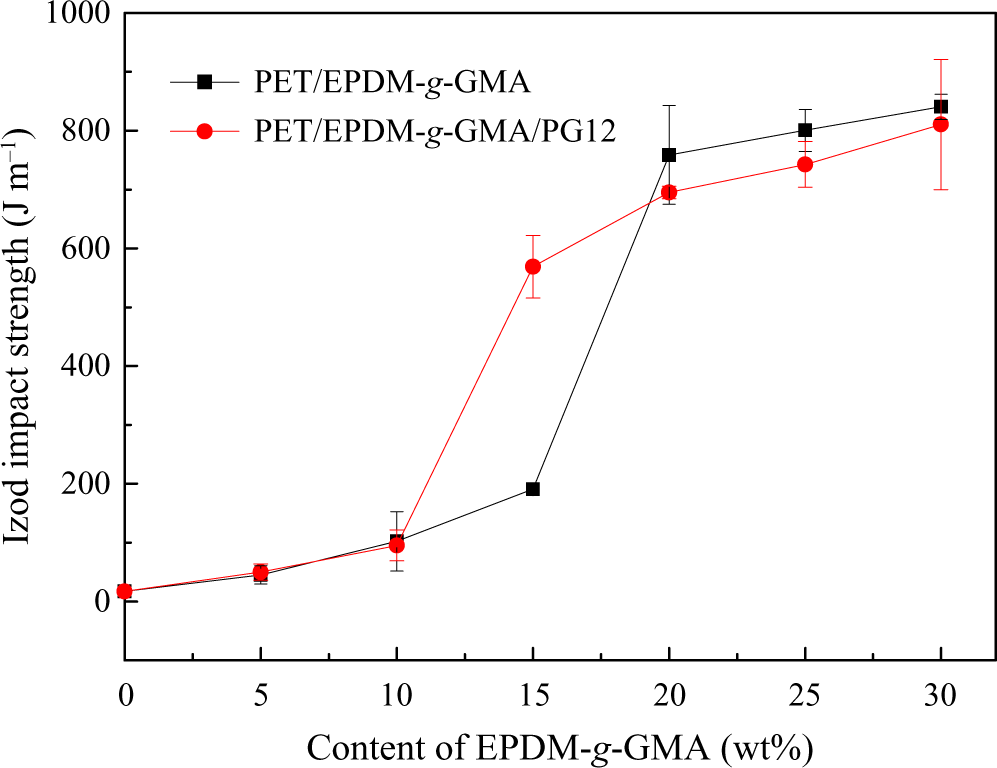
Effect of EPDM-g-GMA content on Izod impact strength of PET blends. PET: poly(ethylene terephthalate); EPDM-g-GMA: ethylene–propylene–diene monomer-grafted glycidyl methacrylate.
Morphology
The cryogenically fractured surfaces of PET/PG12/EPDM-g-GMA blends with various concentrations of PG12 was etched and observed using scanning electron microscopy (SEM). The SEM photographs are shown in Figure 12. The content of EPDM-g-GMA was kept at 15 wt%. The holes correspond to etched EPDM-g-GMA dispersed phase. EPDM-g-GMA phase in spherical shape dispersed in PET matrix homogeneously. The average size of the rubber phase was about 0.5–1 µm with small deviations. The size of the dispersed rubber phase showed little difference with the increasing content of the chain extender. It can be concluded that the increasing impact strength of PET blends was not due to the improvement of compatibility but was attributed to the chain reaction, which induced the changing of PET.

SEM images for the PET/PG12/EPDM-g-GMA blends with various concentration of PG12: (a) 0.0 wt%, (b) 5.0 wt%, (c) 8.0 wt%, (d) 12.0 wt%, and (e). 15.0 wt%. SEM: scanning electron microscopic; PET: poly(ethylene terephthalate); EPDM-g-GMA: ethylene–propylene–diene monomer-grafted glycidyl methacrylate.
Dynamic mechanical analysis
DMA was used to analyze the compatibility and study the possible shifts in Tgs of the PET and the EPDM-g-GMA phases. The DMA curves of tan δ versus temperature of PET blends with different concentrations of chain extender are shown in Figure 13. Two distinct peaks were observed at the temperature of −58 and 92°C, corresponding to Tg of EPDM-g-GMA phase and PET amorphous phase. With the increasing concentration of PG12, the peak corresponding to PET amorphous phase became higher. The value of Tg and tan δ are illustrated in Table 6. As shown in Figure 6, the introduction of chain extender made the crystallization of PET difficult, which induced the increment of amorphous PET content. The temperature corresponding to the peak shifted to higher temperature slightly, which indicated that the PET amorphous region was affected by the chain extender.
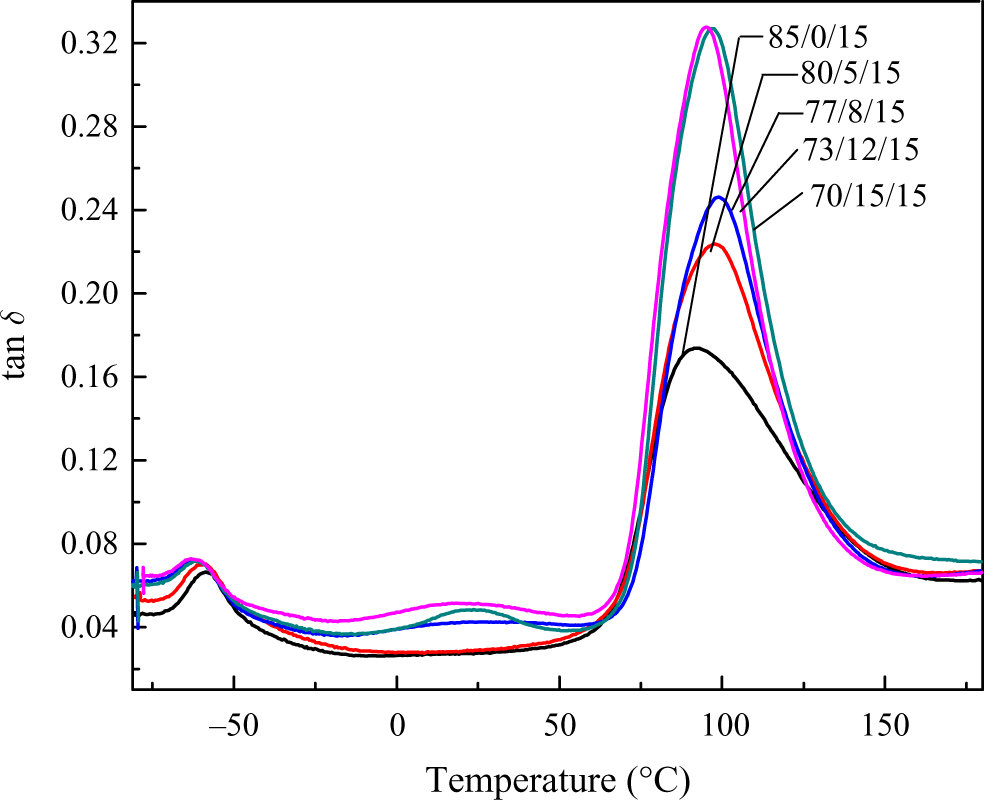
DMA curves for tan δ versus temperature of the PET/PG12/EPDM-g-GMA blends. DMA: dynamic mechanical analysis; PET: poly(ethylene terephthalate); EPDM-g-GMA: ethylene–propylene–diene monomer-grafted glycidyl methacrylate.
Glass transition data of PET blends with different content of PG12.
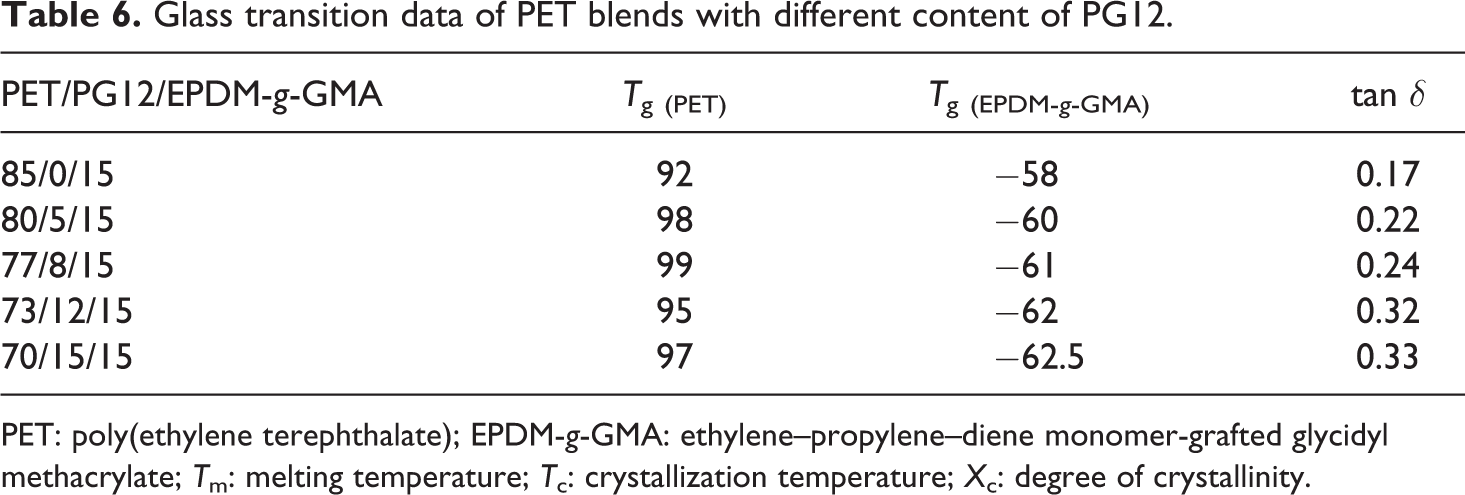
PET: poly(ethylene terephthalate); EPDM-g-GMA: ethylene–propylene–diene monomer-grafted glycidyl methacrylate; Tm: melting temperature; Tc: crystallization temperature; Xc: degree of crystallinity.
Conclusion
An epoxy macromolecular chain extender (PMMA-co-GMA) was synthesized by random free radical polymerization. The influence of concentration of chain extender and polymerization degree of GMA to PMMA on the molecular structure and thermal properties of PET were investigated. The rise of torque after initial melting period was observed, which indicated the occurrence of reaction between the epoxy group of PMMA-g-GMA and hydroxyl or carboxyl end groups of PET. DSC analysis indicated that Tm and Tc of the modified PET shifted to lower temperature, and the crystallization degree of PET blends decreased.
The macromolecular chain extender was introduced in PET/EPDM-g-GMA blends with the composition of 85/15 that exhibited brittle fracture behavior. The blends, in which the chain extender content in PET was over 8 wt%, exhibited ductile fracture behavior. EPDM-g-GMA was used to toughen PET with 8 wt% macromolecular chain extender. The brittle–ductile transition took place between the rubber concentration of 10 wt% and 15 wt%. The transition took place earlier than in PET/EPDM-g-GMA blends. In view of morphology, the addition of chain extender showed little influence on the compatibility of PET blends. The peak corresponding to PET amorphous phase became higher, with the introduction of PMMA-co-GMA. Tg of the PET amorphous phase shifted toward higher temperature. The introduction of chain extender prohibited the symmetrical arrangement of PET molecular and made the crystallization difficult.
The macromolecular chain extender was an effective extender for PET, which also showed contribution for toughening PET.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
