Abstract
In this study, the thermal behavior of modified urea–formaldehyde (UF) resin with nanosilica (nano-SiO2), wood flour (WF), and their mixture of SiO2/WF was investigated. Five modified UF hybrid composite materials with 0.8 F/U ratio with different filler were synthesized using the same procedure. The thermal behavior of materials was studied using nonisothermal thermogravimetric analysis, differential thermal gravimetry (DTG), and differential thermal analysis and supported by data from infrared spectroscopy. The shift of DTG peaks to a high temperature indicates the increase in thermal stability of modified UF resin with hybrid SiO2/WF fillers, which is confirmed by the data obtained from the Fourier transform infrared spectroscopic study. It was estimated that the UF/WF samples based on nano-SiO2 have better thermal stability.
Introduction
Wood flour plastic composites (WPCs) attracted a great interest in recent years owing to their relatively good properties and the lower costs. WPCs are composite materials containing both wood (in various forms) and thermoplastic or thermosetting materials. These materials are a relatively new family of composite materials in which a natural fiber and/or filler (such as wood flour (WF)/fiber, hemp, and sisal) is mixed with a thermoplastic such as polyethylene (PE), polypropylene (PP), and poly(vinyl chloride) (PVC). Today, attention has been focused on the nanocomposites, especially those obtained from layered silicates in thermoplastic or thermosetting matrices since they demonstrated a noticeable improvement in thermal and mechanical properties with respect to microcomposite such as WF. The growing interest in environment-friendly materials has motivated academic and industrial research in the development and use of biopolymers for applications in which synthetic polymers or mineral fillers are traditionally used. 1 WPC covers a wide range of area in composite field. 2 These are eco-friendly, low-cost consuming, biodegradable, and renewable. The consumption of plastic materials has increased enormously due to their various advantages. In the polymer composites, different types of fillers are used for improving the thermal, mechanical as well as other properties.
Urea–formaldehyde (UF) resin is a polymeric condensation product of the chemical reaction of F with U and a kind of amino resin. UF resins offer unique potential technical advantages in a variety of applications in abundance unmatched in the competing product. Among those advantages are the above-mentioned low price, the nontoxicity of resin and resin products, and the environmental compatibility of resin bonded products such as particle board. As a typical amino resin, UF resin adhesive possesses some advantages such as fast curing, good performance in the panel, water solubility, and lower price. 3
Filler has a modifying effect on the properties of UF resin. But the formerly used fillers are all particles with sizes above micron grade, which have only small modifying effect. Nanocomposites are a class of composite materials in which the dimensions of the dispersed phase are nanometric. In some cases, nanocomposites exhibit greatly improved properties with respect to conventional microcomposites loaded with the same amount of reinforcing phase, thus allowing for a potential weight-saving design for many plastic products. Today, attention has been focused on the nanocomposites, especially those obtained from layered silicates 4,5 in thermoplastic or thermosetting matrices 6 since they demonstrated a noticeable improvement in thermal and mechanical properties with respect to microcomposites. 7 These new materials, called nanocomposites or organic–inorganic hybrids, afford to combine the advantages of both organic materials such as lightweight, flexibility, and good moldability and inorganic materials such as high strength, heat stable, and chemical resistance. 8,9 Hybrid organic–inorganic (nanocomposite) materials offer a variety of advantageous properties in various applications. These materials have been fabricated into membranes, sheets, and fibers, and they are usually applied in various applications such as optical, mechanical, electronic, and biomedical. 10,11 Silica (SiO2) can enhance the mechanical as well as thermal properties of the composite. Polymer-SiO2 composites are of technological importance due to their potential applications in electrochromic windows, fuel cells, chemical separation, electrochemical sensing, and water treatment.
The use of WF as a filler is a choice offering an economical solution for the increasing costs of wooden products and construction materials. There is a considerable commercial interest in thermoplastic composites filled with WF due to the potential opportunities of combining the attractive characteristics and properties of both components. 12 WF is derived from a renewable resource that does not have a large energy requirement to process and is biodegradable. 13
Hybrid fillers (combination of two or more different types of fillers) are sometimes very useful because they possess different properties that cannot be obtained with a single type of filler. Most of the previous research on filler reinforcement composites has been focused on the hybridization of mineral and mineral fillers for reinforcing composites. However, only a few studies have explored the effect of mineral and lignocellulosic materials such as WF and fibers on their physical and thermal properties. 2,14,15
Siimer et al. 16 used thermogravimetric analysis (TGA)–differential thermal analysis (DTA) to study the curing behavior of UF adhesive resins in the presence of a wood substrate. Deka and Maji 17,18 used transmission electron microscopy, scanning electron microscopy, x-ray diffractrometry, and Fourier transform infrared (FTIR) spectroscopy to examine the influence of wood, montmorillonite, glycidyl methacrylate, clay, and SiO2 on the properties of PE, PP, and PVC. The effect of nano-SiO2 content with coupling agent on SiO2/UF resin was discussed by Lin et al. 4 Also the mechanism of the strengthening effects of nano-SiO2 on UF resin by means of infrared spectrum analysis and x-ray photoelectronic spectrum was analyzed.
The goal of this work was to determine the thermal stability of synthesized modified urea-formaldehyde (UF composite materials with nano-SiO2 and WF and their mixture based on their thermal behavior. The thermal behavior of five types of modified UF composite materials was investigated using nonisothermal TGA, differential thermal gravimetry (DTG), and DTA supported by data from IR spectroscopy.
Experimental
Materials
In this study, (NH2)2CO (Alkaloid-Skopje, FYR of Macedonia), 35% CH2O (Unis-Goražde, Bosnia and Herzegovina), SiO2 nanoparticles (Degussa Co., Germany) were used as nanofillers. The average diameter, density, and specific surface of the SiO2 nanoparticles were 12 nm, 0.37 g cm−3, and 200 ± 25 m2 g−1, respectively. WF (Pinus silvestris L.) with particle size of 250–300 μm supplied by Tigar Corporation (Serbia). All the other materials and solvents used for analytical methods were of analytical grade.
Synthesis of modified UF composites
Five samples of modified UF hybrid composite materials with 0.8 F/U ratio with different types and loading of filler were synthesized using the same procedure. Synthesis procedure was as follows: 60 cm3 of distilled water and 0.1 mol of U are mixed into reaction vessel with a magnetic stirrer. Other components such as different ratio, type of fillers (7.25 g SiO2, 3.625 g SiO2, and 3.625 g SiO2:3.625 g WF, 3.625 g WF, and 7.25 g WF for composites marked as UF/SiO2, UF/1/2 SiO2, UF/SiO2/WF, UF/1/2 WF, and UF/WF, respectively), 0.12 mol 35% F, and 0.6 cm3 of concentrated sulfuric acid were added into the reaction mixture according to the following order. The pH value was lower than 6. Reaction mixture is mixed for 3 h; 0.22 mol of sodium hydroxide was dissolved in 6 cm3 of distilled water and added to reaction mixture before the stirring was done. The modified UF resin was cured at 110°C for 2 h in a convective drying oven. Table 1 presents the characteristics of resins determined by standard analysis methods.
Characteristics of used UF composites.
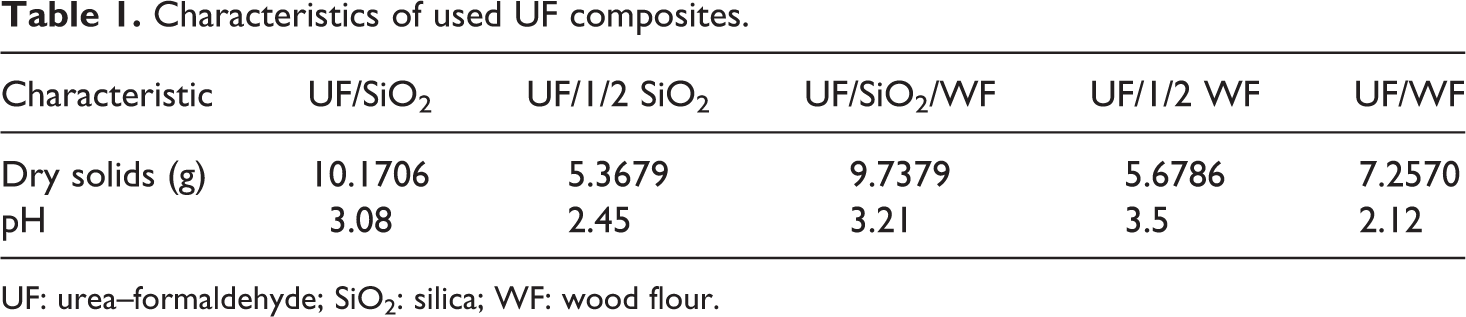
UF: urea–formaldehyde; SiO2: silica; WF: wood flour.
Composite characterization
Thermal analysis
The thermal stability was investigated using nonisothermal TGA and DTA (1750 instrument; Setaram Setsys Evolution, Caluire, France).
Samples (6 ± 0.2 mg) were placed in alumina crucibles. An empty alumina crucible was used as a reference. The samples were heated from 30 to 600°C in a 20 cm3 min−1 flow of argon atmosphere with a heating rate of 10°C min−1. The temperatures at maximum decomposition rate were determined from the peak maxima of the DTG curves.
FTIR spectroscopy
The FTIR spectroscopy in transmittance mode was used for the characterization of the functional group of the resin. The potassium bromide (KBr) pellets of samples were prepared by mixing finely grounded 1.5 mg of samples with 200 mg KBr (FTIR grade) in a vibratory ball mixer for 20 s. The 13 mm KBr pellets were prepared under vacuum in a standard device under a pressure of 75 kN cm−2 for 3 min. FTIR spectra of samples were recorded at room temperature in the range of 4000–400 cm−1 wave number on a Nicolet spectrophotometer (model 380, Thermo Nicolet Corporation, Madison, Wisconsin, USA), with 64 scans for each spectrum and spectral resolution of 4 cm−1.
Results and discussion
During UF resin manufacturing, the final reaction products between U and F can range from the simple monomethylolurea to very complicated cross-linked structures. The formation of linear condensation products in cure process begins at lower temperature if the resin contains greater amount of reactive methylol groups. Depending on different synthesis conditions and technology, generally we used two-step reaction of U and F produces resins with a broad variety of both linear and branched chains. 16 Bonding between organic molecules and surface of SiO2 resulted in the replacement of most of the strongly hydrophilic silanol functionality with a material exhibiting modified and usually predictable new or improved properties. 19
Thermal analysis
Polymers are often exposed to high temperatures during processing and/or use. Therefore, the thermal stability is the most important properties of polymers for a wide range of applications. TGA is one of the methods of thermal analyses which can be used to measure the mass change, thermal decomposition, and thermal stability of composite materials.
TGA–DTG analysis performed showed differences in thermal behavior of composite materials considered in the study. The thermogravimetric diagrams evidence an initial mass loss below 100°C resulted from the gradual evaporation of absorbed moisture, and another mass loss stage in the temperature domain from 170°C to 465°C occurring through decomposition processes of the major constituents present in the WF (cellulose, hemicelluloses, and lignin). Thermal decomposition of WPC depends on wood species, amount of wood, particle size, moisture content, quantity and type of polymers, coupling agents, lubricants, and other additives. Hemicelluloses, cellulose, and lignin primarily decompose between 150–350°C and 250–500°C. 20
The thermal behavior of modified UF resins occurs generally in two main stages for all modified UF resins but in some peaks is overlapping (Figures 1 and 2). The rate of the thermal decomposition reaction shows more than one maximum rate with increase in temperature. This behavior indicated that thermal decomposition of these resins passed through multiple stages, depending on the state of decomposition and not on the components. 21,22 Table 2 summarizes the percentage of total mass loss for all modified UF resins. Figure 3 outlines the major routes of thermal degradation of the modified UF composites.
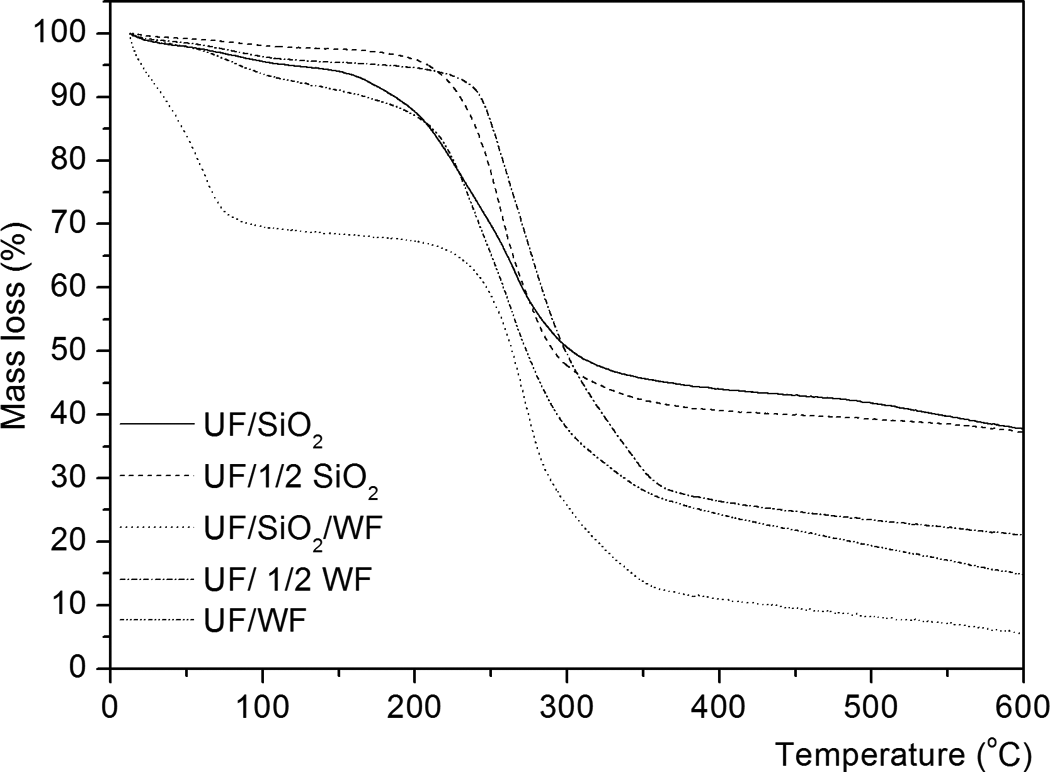
TGA curves of modified UF composite with nano-SiO2 and WF. TGA: thermogravimetric analysis; UF: urea–formaldehyde; nano-SiO2: nanosilica; WF: wood flour.
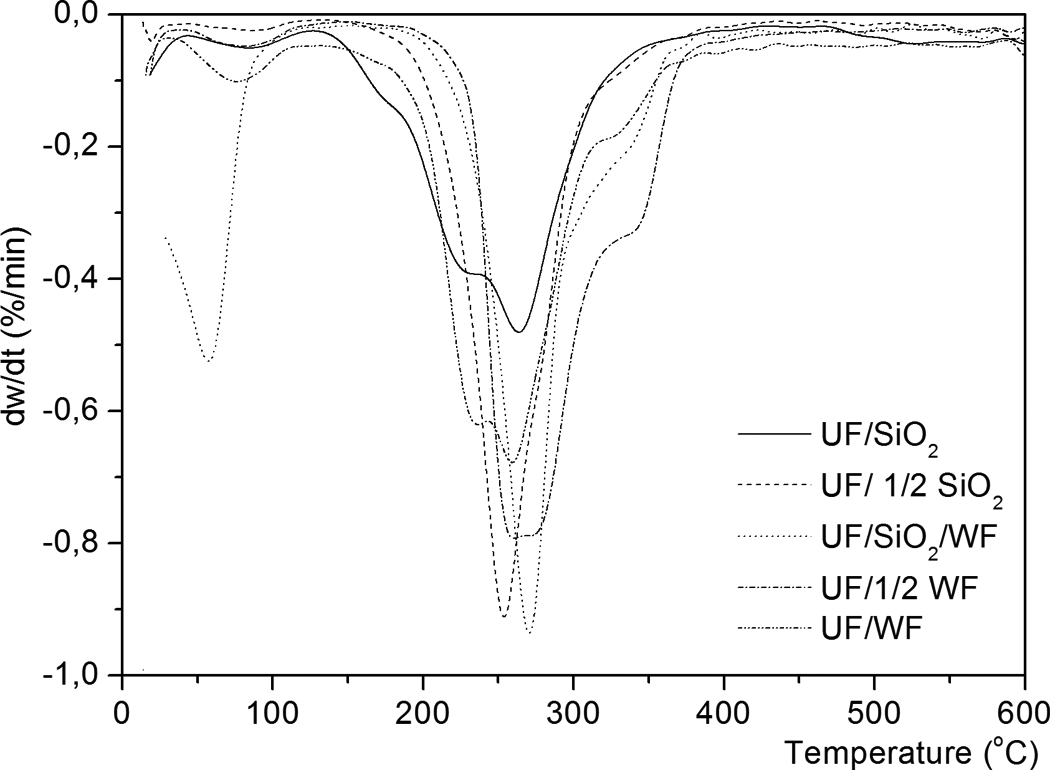
DTG curves of modified UF composite with nano-SiO2 and WF. DTG: differential thermal gravimetry; UF: urea–formaldehyde; nano-SiO2: nanosilica; WF: wood flour.
Data of DTG and DTA peak values and mass loss from UF composites.
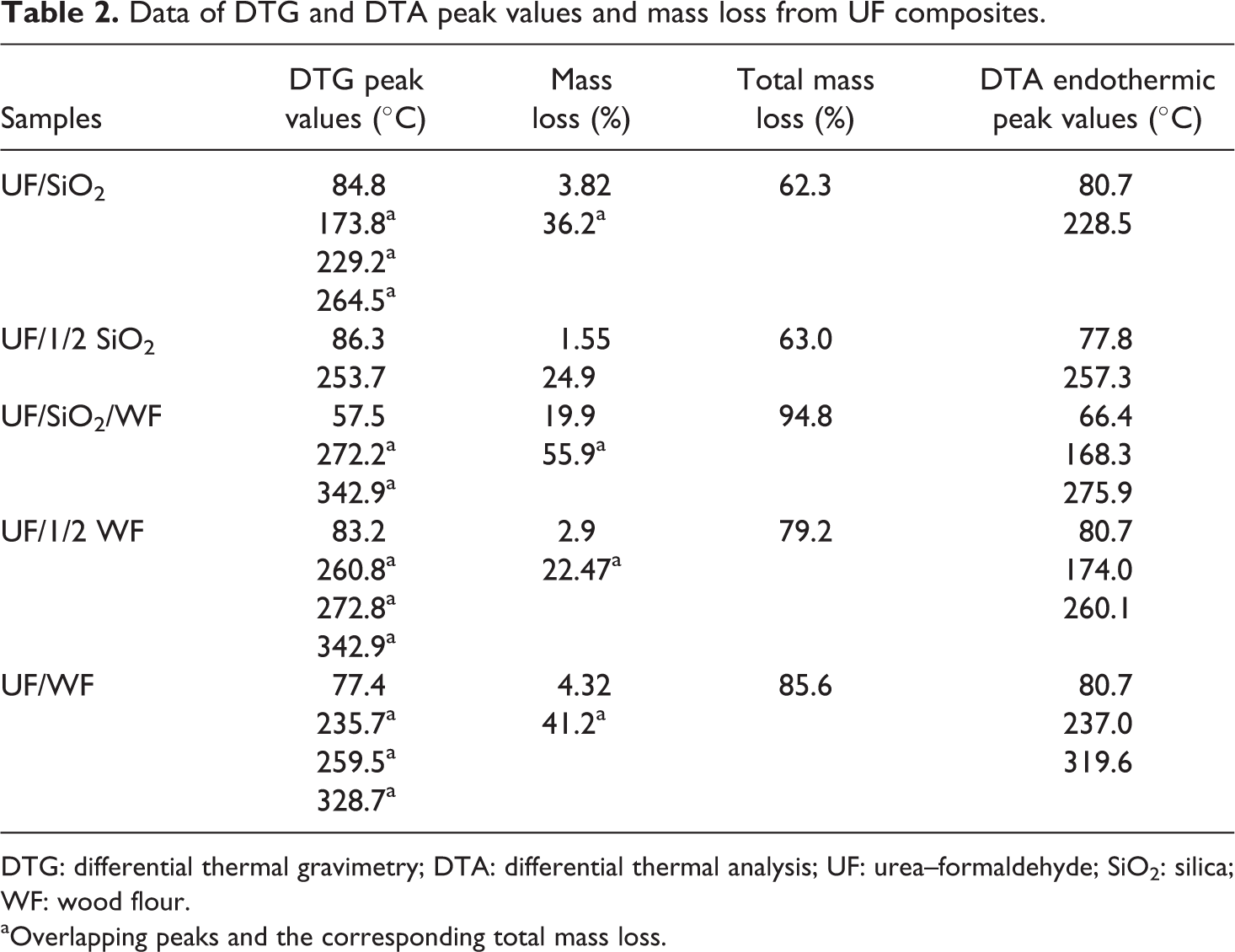
DTG: differential thermal gravimetry; DTA: differential thermal analysis; UF: urea–formaldehyde; SiO2: silica; WF: wood flour.
aOverlapping peaks and the corresponding total mass loss.
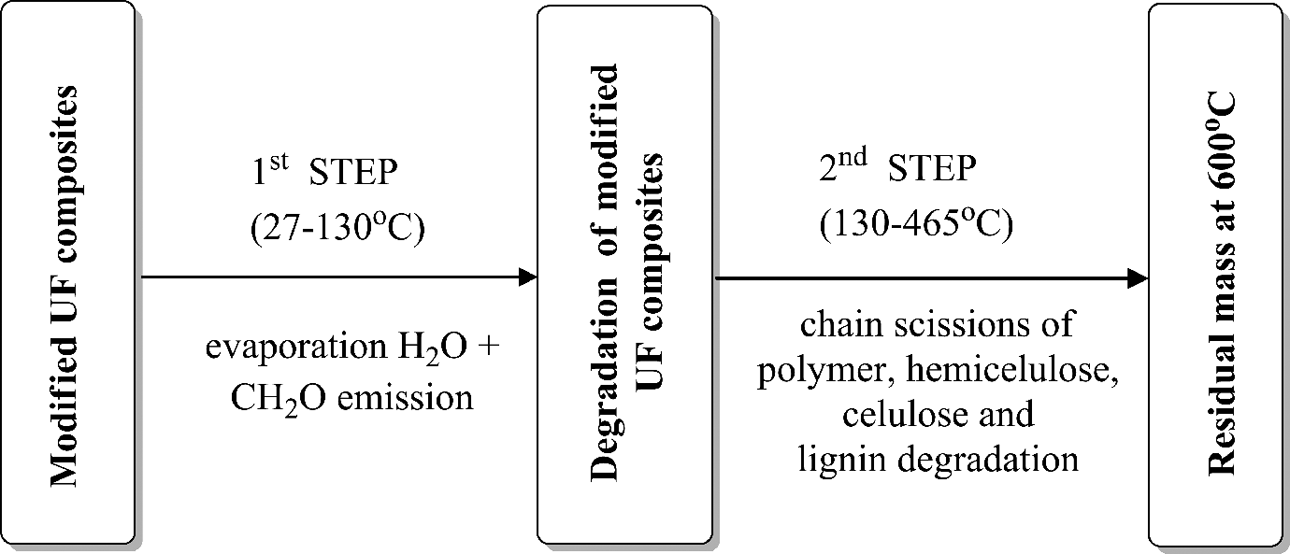
Major degradation routes of modified UF composites. UF: urea–formaldehyde.
The thermal degradation of samples up to 400°C with nano-SiO2 (UF/SiO2 and UF/1/2 SiO2), WF (UF/1/2 WF and UF/WF), and their mixture (UF/SiO2/WF) (Figures 1 and 2) occurs in four and three successive steps but can be noted that the second, third, and fourth degradation regions are overlapped. In each step, partial volatilization takes place while the polymer undergoes chemical modification to give progressively more stable structures. 23
From the TGA, DTG, and DTA curves shown in Figures 1, 2, and 4, the first mass loss occurs below 200°C and can be attributed to the water evaporation and the slow free F emission, accompanied by a small and wide endothermic peak temperature below 100°C (Figure 4). Above 200°C, the main degradation step is initiated when chain scissions begin and the radicals formed induce the formation of cyclic structures in the polymer chain. This process results in the extensive polymer fragmentation. The type of bond between the U molecules depends on the reaction conditions: low temperatures and only slightly acidic pH favor the formation of methylene ether bridges (–CH2–O–CH2), while higher temperatures and lower pH led to the formation of stable methylene (–CH2–) bonds. Degradation of cured resins begins with release of F from dimethylene ether groups 22 and the maximum degradation rate happens when the stable methylene ether linkages deconstruct. 24
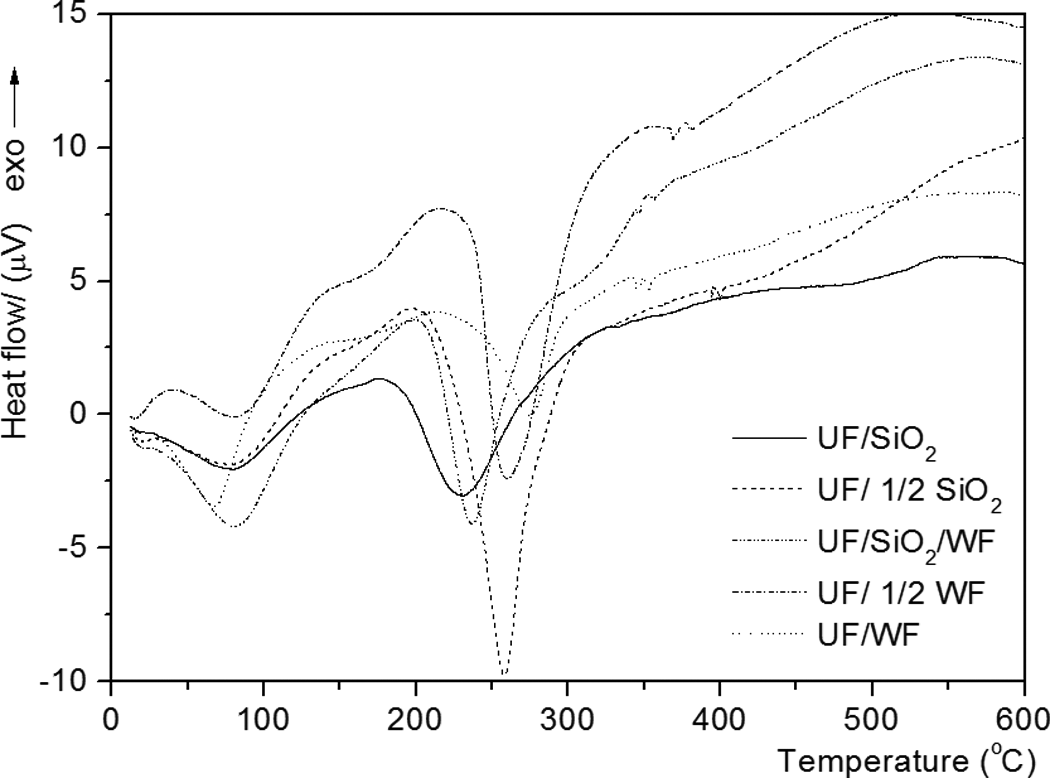
DTA curves of modified UF composite with nano-SiO2 and WF. DTA: differential thermal analysis; UF: urea–formaldehyde; nano-SiO2: nanosilica; WF: wood flour.
The first step in degradation occurs in the temperature range of 43.2–121.2, 47.5–116.1, 27.2–98.0, 41.7–129.6, and 35.9–126.7°C for UF resins with SiO2, 1/2 SiO2, SiO2/WF, 1/2 WF, and WF fillers, respectively, and the mass loss is 3.81, 1.6, 19.9, 2.9, and 4.3%, indicating the evaporation of both water and F. The increased mass loss in samples that contain WF filler is understandable because it is hydroscopic. The small and wide endothermic peak minimum temperatures (Figure 4) at 80.7°C, 77.8°C, 66.4°C, 80.7°C, and 80.7°C for examined UF resins, respectively, indicating water evaporation from the initial water’s resin.
The second step in degradation starts at different temperatures of 130.8°C, 145.7°C, 182.3°C, 175.4°C, and 140.2°C and ends at different temperatures of 418.2°C, 417.4°C, 385.9°C, 463.6°C, and 409.8°C for modified UF resin with different ratios and type of fillers. Table 1 shows the corresponding DTG peaks. In the second step of degradation, the percentage of mass loss at 36.2, 24.9, 55.9, 22.8 and 41.2%, respectively. The second stage corresponds to the main decomposition process and proceeds at a high rate. It can be attributed to the thermal degradation of hemicelluloses, cellulose, and polymer degradation. Above 200°C, radicals formed by chain scission induce the formation of cyclic structures in the polymer, which undergoes extensive fragmentation above 300°C. 23
In samples containing WF as a filler, the corresponding DTG overlapped peaks it at around 260 and 340°C. The endothermic peak minimum temperatures at 228.5, 257.3, 275.9, 260.1, and 319.1°C are attributed by many authors to the degradation of methylene ether bridges into methylene bridges and branching and cross-linking reactions in the resins network. 24 The endothermic peak temperatures at 168.3 and 174.0°C refer to the different states of water from resin synthesis and condensation water arising during postcure. 25 The water comes up either by the quantity added during the synthesis of the resin or results from the condensation reaction. 26
The second major mass loss in samples with WF might be attributed to the depolymerization of hemicelluloses, glycosidic linkages of cellulose, and thermal decomposition of cellulose. 17 Figure 4 shows that the minimum endothermic peak attributed to water evaporation, which leads to lower temperatures for UF/SiO2/WF than other modified UF resins.
The presence of WF in nano-SiO2 modified UF resin (UF/SiO2/WF) increases the DTG peak temperature from 253.7 and 264.5 to 272.2°C (Table 2). The presence of WF retards the decomposition of nano-SiO2-based UF resin (DTG peak temperatures about 8–18.5°C higher than that of nano-SiO2-based UF resin), probably as a result of WF initially absorbing most of the heat energy.
As the filler loading increased in WF-modified UF resin (UF/1/2 WF and UF/WF), the thermal stability of the composites slightly decreased, whereas the total mass loss increased (Table 2). These results show that the thermal stability of the composites decreased as the lignocellulosic filler content increased, which is a logical consequence of the lower thermal stability of the lignocellulosic filler. In contrary, increasing the nano-SiO2 content in the UF resin increases its thermal stability and the total mass loss is similar. Nano-SiO2 can react with active groups of UF. Hydrogen bonding is believed to be involved in the interaction between UF resin and nano-SiO2. 4,27,28 In the presence of both fillers, percentage of total mass loss might be due to the increase in interaction of hydroxyl and carbonyl groups of WF with the hydroxyl groups present in SiO2, as a result reduced interaction with the polymer matrix.
Thus, the three components are capable to get hybridized due to their reversible hydrogen bonding as a result of proton–donor/proton–acceptor action between –CONH–, –COOH–, and –Si–OH– groups within the components of the system. The presence of hydroxyl and other polar groups in various constituents of the WF resulted in poor compatibility between hydrophilic WF and hydrophobic plastic, which increased the water absorption. With the increase in wood content, there are more water residence sites thus more water was absorbed. Water absorption in composites is mainly due to the presence of lumens, fine pores, and hydrogen bonding sites in the WF, the gaps and flaws at the interfaces, and the microcracks in the matrix formed during the compounding process. 29
Relating to previous observations, it can be assumed that the incorporation of the WF has enhanced the thermal stability of UF/SiO2 composites due to the phenolic lignin of the WF. Such phenolic compounds are expected to improve the stability of the composite by the absorption of some heat will prolong the ability of the matrix to resist the heat prior to pyrolysis and the formation of the polar–polar interactions that requires extra heat ahead to secession. 30
To sum-up, it can be concluded that the UF/SiO2/WF sample has the highest DTG peak temperature compared with the other composites.
FTIR spectroscopy
Figures 5 and 6 show the FTIR spectra of WF, nano-SiO2, UF/WF, UF/SiO2, and UF/SiO2/WF hybrid resin composites. Due to the complex structure of polymer, the absorption frequencies are broad for resin spectra. Broadening may also be observed due to the presence of by-products in the resin such as water and excess F, which allow hydrogen bonding with the reactive functional groups like CH2OH, NH2, and NH. 31,32
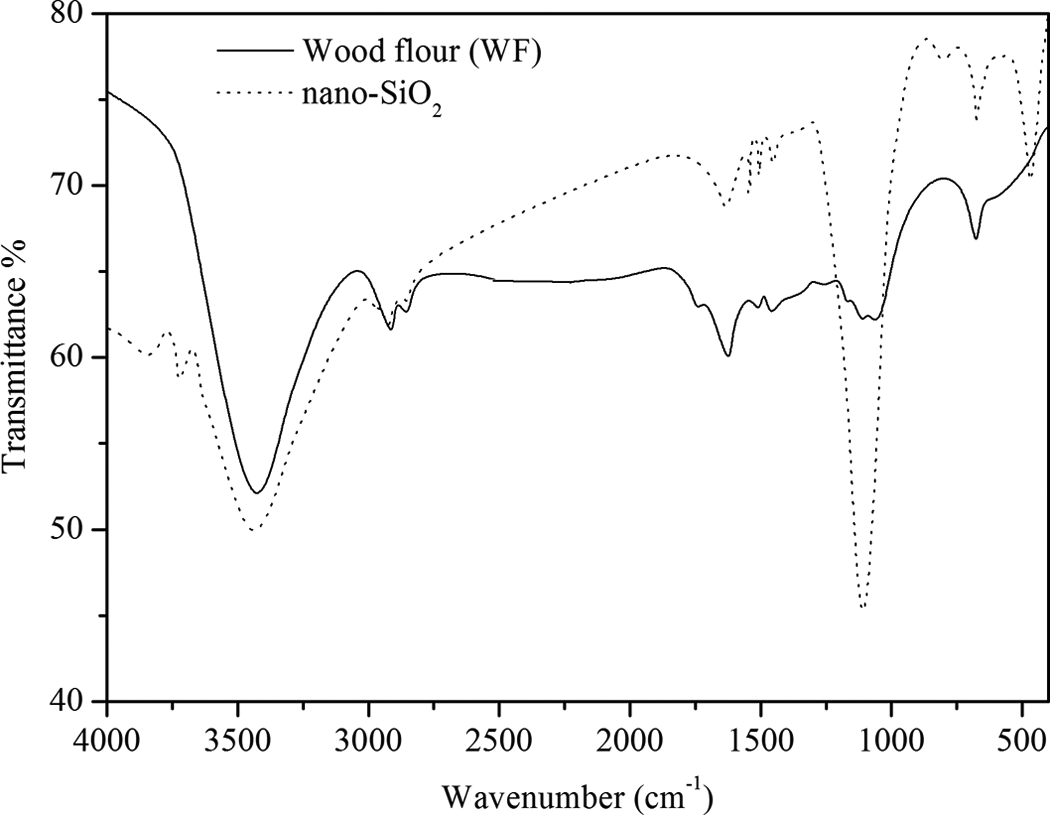
FTIR spectra of nano-SiO2 and WF. FTIR: Fourier transform infrared spectroscopy; nano-SiO2: nanosilica; WF: wood flour.
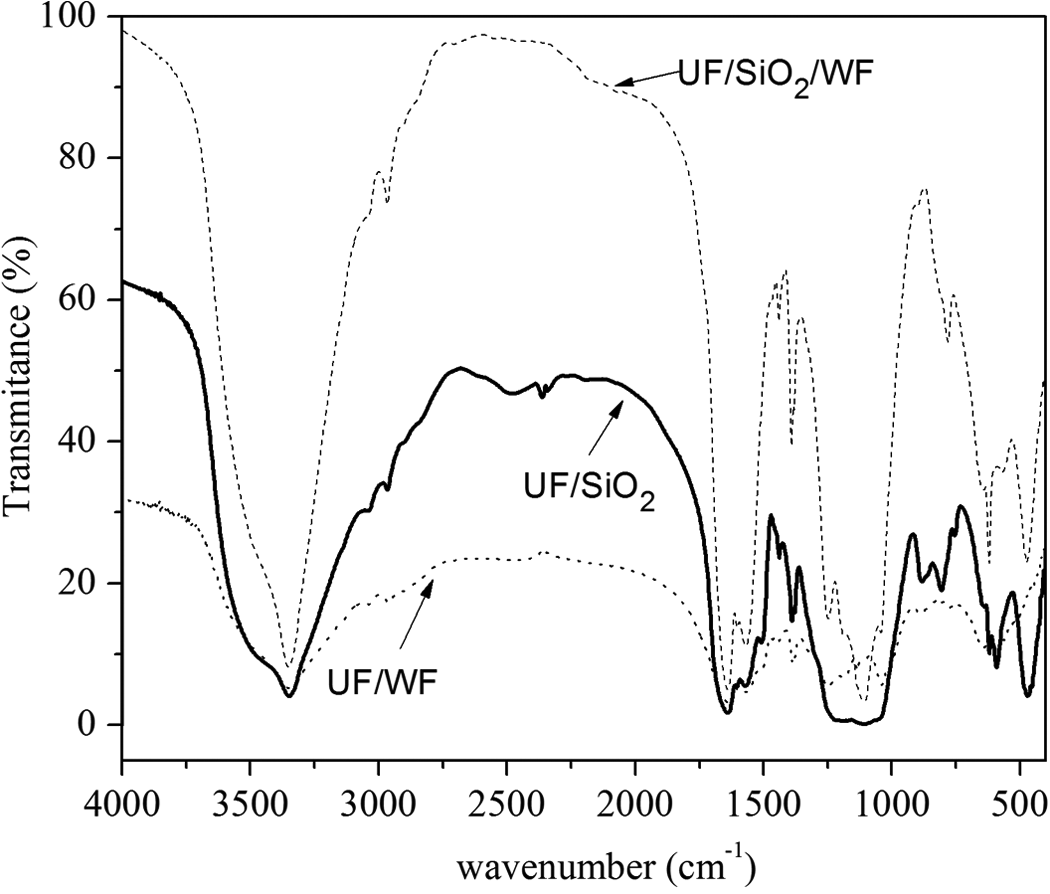
FTIR spectra of modified UF composites with nano-SiO2 and WF. FTIR: Fourier transform infrared spectroscopy; UF: urea–formaldehyde; nano-SiO2: nanosilica; WF: wood flour.
The FTIR spectra of nano-SiO2 (Figure 5) show absorption peaks at 3421 and 1631 cm−1 originated from O–H stretching and O–H bending vibrations of hydroxyl group adsorbed on the particle surface. The other peaks originated from Si–O–Si group appeared in the range of 1111–473 cm−1.
The FTIR spectra of the WF (Figure 5) show absorption bands originated from O–H stretching vibrations at 3435 cm−1, –CH stretching vibrations at 2924 and 2855 cm−1, C=O stretching vibrations at 1736 cm−1, –OH bending vibration at 1656 cm−1, C–O stretching vibrations at 1112 and 1043 cm−1, and C–H bending vibration at 1000–669 cm−1 (out of plane). Similar values for these absorption bands were found by Deka et al. 33 The absorption band at 1656 cm−1 corresponds to the C=C stretching of aromatic skeletal in lignin. 34
However, FTIR spectra of cured resins showed characteristic absorption bands in this region. The FTIR spectra of the UF/SiO2 and UF/SiO2/WF (Figure 6) show a strong absorption band at 3351 cm−1. For UF/SiO2 and UF/SiO2/WF, this band is sharp and typical hydrogen bond between N–H and OH. Medium absorption bands for UF/WF resin composites were recorded in the same region at 3351 cm−1, which is the characteristic absorption band of the NH stretching vibration originated from NH2 group. The sharpness of these bands indicated a reduction in the extent of hydrogen bond interaction, which is expected as the structure becomes more cross-linked due to methylenization reaction. 26 A medium absorption band in all resin composites spectra appears in the range of 2967–2962 cm−1, which is ascribed to the symmetrical C–H stretching vibrations in CH2 group of ether, CH2OH group, and N–CH2 group. A very strong multiple absorption band and some overlapped bands are observed at 1637 cm−1 in all spectra of modified UF resin composites, which may be assigned to the C=O stretching vibration (amide I) of CONH2 group and amide II as well as the –N–H scissors of amide I. The strong and overlapped bands at 1560–1570 cm−1 are attributed to –N–H bending vibrations of amide II. The cross-linking between two methylol groups provides ether linkages (–CH2–O–CH2) in which –NH is attached to both sides. The weak multiple peaks at 1436 cm−1 may be attributed to C–H bending vibrations of –CH2O and –CH2–N groups, while the small peaks at the area of 1350–1460 cm−1 can be assigned to C–N stretching vibrations of amide I and II. The weak absorption band around 1384–1393 cm−1 for all polymer samples may be ascribed to C–H bending mode in –CH2/–CH2OH/N–CH2–N group.
The medium absorption band in the region of 1110–1120 cm−1 assigned to –N–CH2–N– asymmetric stretching vibrations, ν(C–O–C) of ether linkage, and ν(Si–O–C), ν as(Si–O–Si) vibrations of siloxane or silicone. 4,10,35 These peaks (–N–CH2–N– and –C–O–C–) show the cross-linking in the resin via N atoms. Moreover, –C–O–C– peaks in the both resins show polyether product. 36 From Figure 6, it was observed that the intensity of –OH stretching vibration in UF/SiO2/WF resin composites shifted from 3434 to 3350 cm− 1 and lower than UF/WF and 3421 cm− 1 for UF/SiO2, respectively. This confirmed the bond formation between polymer and hydroxyl groups from both WF and nano-SiO2. Moreover, the intensity of –CH stretching vibrations at 2967 cm− 1 for modified UF resins was found to be higher. The peak at 1110–464 cm− 1 originated from metal oxide bonds of nano-SiO2 decreased to a considerable extent. All these are suggested a strong interaction between wood, nano-SiO2, and polymer matrix.
Cross-linking between organic molecules and surface of SiO2 results in the replacement of the most strongly hydrophilic silanol functionality with a material exhibiting modification. The most used attachment process of an organic moiety to SiO2 surface involves the formation of Si–O–Si or Si–O–C bonds, which confirms the existence of absorption band in this region. The band at approximately 780–891 cm−1 may be assigned to asymmetric stretching vibrations of –C–O- in –CH2OH group, –N–H wagging vibrations in 1° and 2° amines, and valence vibrations of Si–O group at 474 cm−1, respectively. Bending vibrations of CH appeared at 620–640 cm−1.
Conclusion
In this work, thermal stability of nano-SiO2 and WF-modified UF composite were synthesized. Based on our research presented in this article, we can conclude the following aspects: As the filler loading of nano-SiO2 in the UF/SiO2 resin increased with an increase in thermal stability and the total mass loss is similar. Thermal stability of the UF/WF composites decreased as the lignocellulosic filler content increased, which is a logical consequence of the lower thermal stability of the lignocellulosic filler. UF/SiO2/WF composite has DTG peak, which is shifted to a high temperature indicating its higher thermal stability than all UF composites modified only with different contents of nano-SiO2 and WF. On the basis of the DTG peak-modified UF/WF composites indicated higher thermal stability than modified UF/SiO2 composites. The intensity of –OH stretching vibration in UF/SiO2/WF resin composites is shifted from 3434 to 3350 cm−1 and is lower than UF/WF and 3421 cm−1 for UF/SiO2, which confirmed the bond formation between polymer and hydroxyl groups of both WF and nano-SiO2. Higher intensity of –CH stretching vibrations at 2967 cm−1 and the lower peak at 1110–464 cm−1 originated from metal oxide bonds of nano-SiO2 are suggested a strong interaction between wood, nano-SiO2, and polymer matrix.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was granted by the Ministry of Science and Technological Development of the Republic of Serbia (projects numbers 45022 and 45020).
