Abstract
A new kind of polyamide-6,6/mobile crystalline material (PA66/MCM-41) hybrid composites were prepared by in situ polymerization. The crystal structural and physical properties of these composites were investigated by Fourier transform infrared and nitrogen adsorption/desorption measurements. The nonisothermal crystallization behavior of PA66/MCM-41 was studied through differential scanning calorimetry measurements. Ozawa, Mo, and Kissinger models were used to analyze the kinetics of the nonisothermal crystallization process. It was found that MCM-41 acting as a nucleating agent in composites accelerated the crystallization rate; the Ozawa method failed to describe the nonisothermal crystallization behavior of PA66/MCM-41 composites. However, the Mo model was able to describe the nonisothermal crystallization process fairly well.
Keywords
Introduction
Polyamide 6,6 (PA66) when compared with other polymers is the extensively used engineering plastic due to its versatile advantage, easy to mold, excellent mechanical properties even at elevated temperatures, extreme durability, good chemical and heat resistance. Therefore, PA66 polymers are widely produced and employed in various fields of engineering, such as aircraft, textiles, automotives, machines, electronic, accessories, and household tools. Polymer/inorganic hybrid composites have attracted a great interest during the last few years due to the potentially advanced properties compared to conventional composite materials. Most of the studies have shown that a very low concentration of inorganic addition can lead to a significant improvement in its performance. The development of PA66/inorganic composites could further enhance its competitiveness in various properties, such as mechanical properties, reduced liquid and gas permeability, electricity, thermal stability, flame retardancy, wear/friction, and catalytic performance. 1–5
Till date, the main methods used for fabricating the PA66/inorganic composites include solution mixing, 6 melt compounding, 7 electrospinning, 8,9 and reactive extrusion 10,11 . Nevertheless, PA66 inorganic hybrid composites have been scarcely studied prepared by in situ polymerization, apart from the electrical and mechanical properties of PA66/single-walled carbon nanotubes (SWNT) composites produced via in situ polymerization are briefly discussed. 12 And the incorporation of inorganic composites includes various kinds of materials, such as long/short glass fiber, 13–19 carbon fiber, 20,21 silica (SiO2), 22 montmorillonite, 23 nanoclay, 24 multi-walled CNTs (MWCNTs), 25–28 SWNTs, 29 tin(IV) phosphate, 30 kaolin, 31 mesoporous molecular sieve, 32 hydroxyapatite, 33 and apatite 34 . Most of studies reported the effect of addition of inorganic compounds on the crystallization behavior, thermal stability and flammability, mechanical properties, morphology, and molecular modeling of PA66/inorganic composites. However, the major challenges in achieving high performances are the strong interfacial adhesion between inorganic filler and PA66.
The mobile crystalline material (MCM-41) belongs to a group of mesoporous molecular sieves which was first reported by researchers Kresge et al. 35 and Beck et al. 36 of the Mobil Research and Development Corporation in 1992. Mesoporous molecular sieves have large pore dimensions and narrow pore size distributions. Several researchers’ published findings are aimed at controlling the penetration of macromolecules to the mesopore. 37–41 Yin et al. 42 reported that the mesoporous silica SBA-15 particles can be well incorporated with polytrimethylene terephthalate (PTT) by in situ polymerization, and the presence of SBA-15 particles has large influence on the crystallization of PTT. Poly(methyl methacrylate)/mesoporous molecular sieve 43 composite materials were prepared by in situ polymerization, and its morphological, rheological, thermal, and dynamic mechanical properties were investigated. Because the polymer molecular chain is confined within the nanochannel, the composites show higher glass transition temperature, thermal stability, tensile strength and modulus, storage modulus, shear storage modulus, and viscosity with increasing mesoporous molecular sieve contents. In these articles, polymer incorporated directly into porous materials have been examined to understand their interaction and to control physical or chemical properties of the resulting composites.
Moreover, Kojima et al. 32 prepared PA66/mesoporous molecular sieve composites by pushing the molten polymer molecules into the mesoporous molecular sieve pores above 2.0 MPa and 300°C. The results indicated that the heat of PA66 fusion crystallite in the mesoporous molecular sieve pores was low compared with that of PA66. In this article, the PA66/MCM-41 hybrid composites are prepared by in situ polymerization. Because the pore diameter of MCM-41 particles varied in the range of < 1.5–3.2 nm, which is larger than the van der Waals diameter of the polymer chains, the polymer can partially penetrate the particle mesopores and bind to polymer matrices. Due to the interpenetrating nature of organic polymer chains with the inorganic scaffold, the interfacial interaction between the surface of inorganic particles and the polymer resin was improved. In addition, the effects of adding MCM-41 on the nonisothermal crystallization kinetics of the PA66 composites were also investigated.
Experimental
Materials
PA66 salt used in this study was commercially supplied by Jiangsu Huayang Nylon Co. Ltd (China). Silica MCM-41 mesoporous molecular sieve was purchased from Nankai University catalyst Co. Ltd (China). Formic acid and n-dodecanoic acid (chemical pure grade) were obtained from Tianjin Kermel Chemical Reagent Co. Ltd (China).
Sample preparation
PA66 salts and MCM-41 with the specific surface area of 1000 m2 g−1 was used in the experiment. Polymer hybrid composite was performed by adding inorganic nanoparticles during in situ polymerization. First, MCM-41 particles and PA66 salts were outgassed at 120°C under vacuum for more than 5 h. Second, various contents of mesoporous nanoparticles, 7.0 g of PA66 salts and 0.16 g of n-dodecanoic acid were introduced into a steel pan with a magnetic stirring bar. Third, when the pan was heated to 200°C, stirring was started; continued to heat up, and was degassed by vacuum pump every 5 min; then, the polymerization was carried out at 260°C in a thermal cell for 1 h under vigorous stirring. Finally, all the molten samples was poured out onto the glass and quenched at room temperature and then then cut into pellet. The pellets were dried in a vacuum oven at 140°C for 6 h before use in differential scanning calorimetric analyses. Corresponding to the weight percentage of MCM-41, the products were named as M1 (MCM-41 = 0%), M2 (MCM-41 = 1 wt%), M3 (MCM-41 = 2 wt%), M4 (MCM-41 = 4 wt%), and M5 (MCM-41 = 8 wt%) as shown in Table 1.
The composition of PA66/MCM-41 composites.
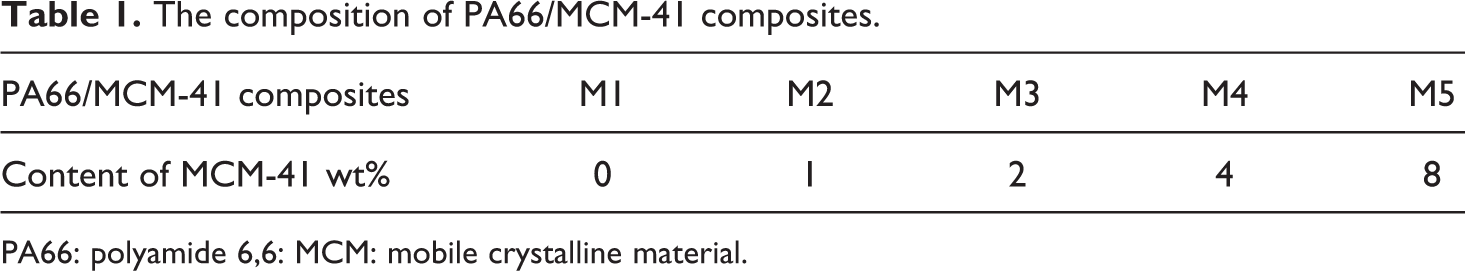
PA66: polyamide 6,6: MCM: mobile crystalline material.
In order to carry out the nitrogen (N2) adsorption/desorption and Fourier transform infrared spectrometry (FTIR) measurements, the M5 hybrid composite was resolved in a large volume of formic acid by stirring for 4 h, then centrifugal classified, washed for several times to remove the polymer from the external surface of the MCM-41 particles, and the residual product named as PA66/MCM-41 was dried in vacuum at 140°C for 2 h before testing.
Characterizations
Differential scanning calorimetry (DSC, Netzsch DSC 204F1 Phoenix, Germany) was used to analyze the dynamic crystallization process. All the runs were performed under N2 atmosphere to prevent extensive oxidation. The samples were heated from room temperature (25°C) to 280°C at a heating rate of 10°C/min and kept at that temperature for 3 min. Then the temperature was reduced to 25°C at cooling rates of 4, 8, 16, and 32°C min−1, respectively. The samples were kept at 25°C for another 3 min, then heated again to 280°C at a heating rate of 10°C min−1, and then the second heating scan was recorded.
N2 adsorption/desorption measurements were performed by an automatic instrument (TriStarII3020, Micromeritics Instrument Co., Norcross, Georgia, USA). The sample was outgassed for 4 h at 120°C and 0.015 kPa before measurement.
FTIR measurement was carried out by VERTEX 70 FTIR spectrometers (Bruker Co., Germany). The PA66 samples were cut into pieces and then clamped on the sample stage for FTIR spectrometer. The PA66/MCM-41 and MCM-41 was characterized on potassium bromide pellets. The data were recorded in the wave number ranging from 600 cm−1 to 4200 cm−1. Baseline-corrected infrared spectra were obtained for all samples at room temperature.
Results and discussion
N2 adsorption/desorption and FTIR of PA66/MCM-41
As shown in Figure 1, the polycondensation reaction of PA66 salt occurred in the presence of MCM-41, so the PA66 macromolecular chains penetrated into the hexagonal pores. Run et al. 43 studied hybrid material properties of the poly(methyl methacrylate)/mesoporous molecular sieve and proposed a structure of “interpenetrating organic–inorganic network”. In other words, the PA66 chains that extend to the outside of the MCM-41, and then the entangled polymers, were formed around the inorganic particles. Thus, the compatibility between the inorganic particles and polymer resin may be improved significantly.
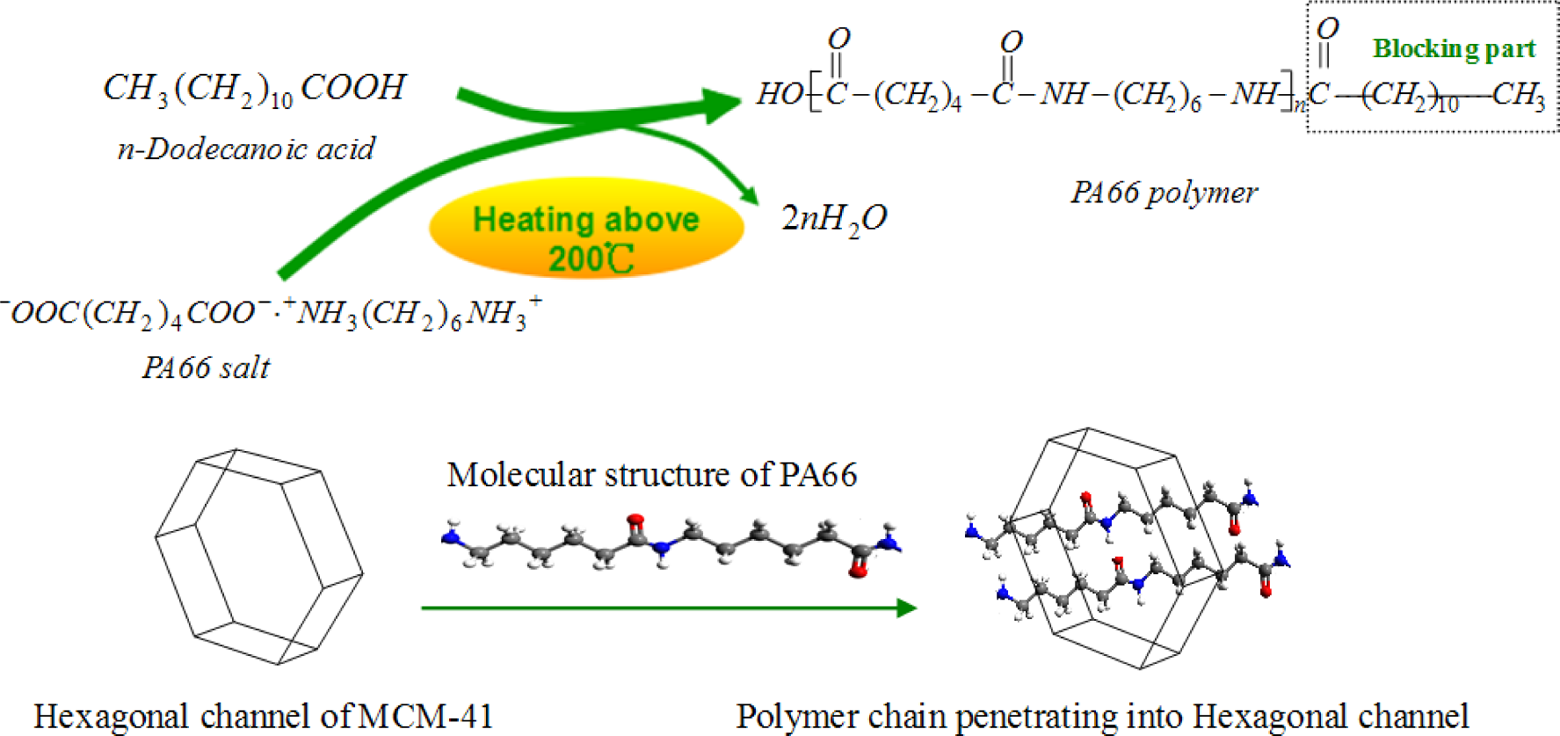
Above: PA66 salts polymerization reaction process; below: the structure model of PA66/MCM-41 composites; atoms are represented as spheres and are color-coded: carbon (grey), hydrogen (white), nitrogen (blue), and oxygen (red). PA66: polyamide 6,6: MCM: mobile crystalline material.
As shown in Figure 2(a), the N2 adsorption–desorption isotherm of MCM-41 (M1) exhibited both a reversible type IV isotherm and a sharp pore-filling step at p/p 0 0.3–0.4 which were characteristic of uniform pore. However, that of PA66/MCM-41 composite showed nearly no adsorption step, which implied that PA66 macromolecules had been indeed inserted into the channels of MCM-41 and occupied most channel space of the MCM-41. It also indicated that the MCM-41 had a narrow distribution of pore diameter centered at 2.8 nm as shown in Figure 2(b).
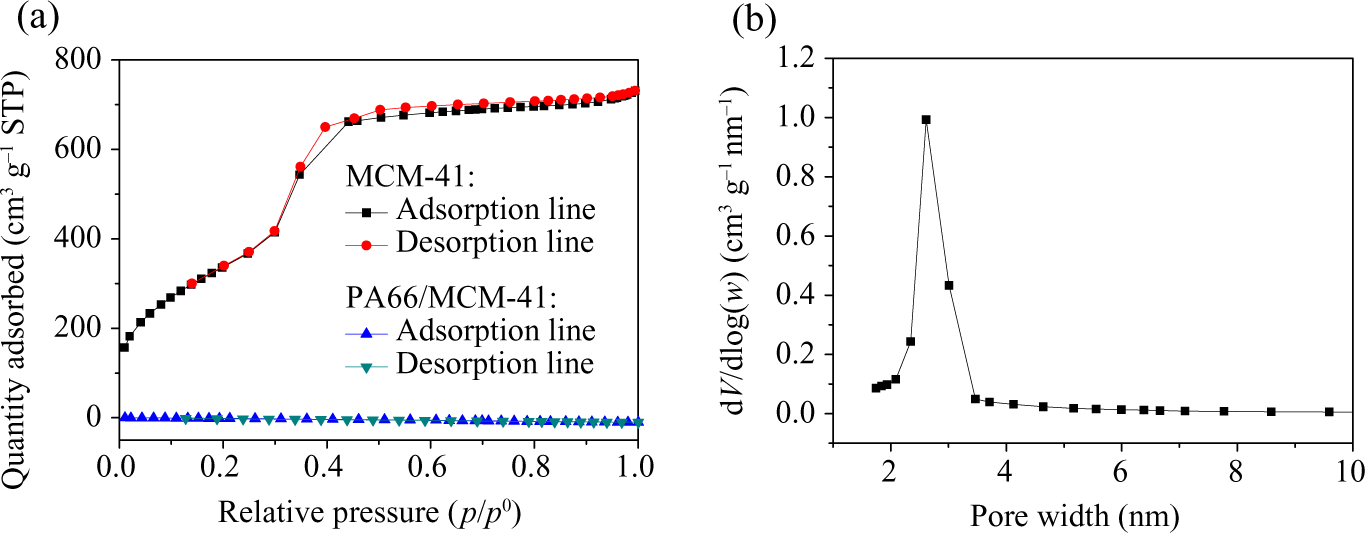
Nitrogen sorption isotherms of MCM-41 and PA66/MCM-41 (a) and pore diameter distribution of MCM-41 (b). PA66: polyamide 6,6: MCM: mobile crystalline material.
FTIR spectra of PA66 polymer, MCM-41, and PA66/MCM-41 are shown in Figure 3. First, the band at 3450 cm−1 was ascribed to the stretching mode of adsorbed water in MCM-41. The disappearance of water adsorbed peak in PA66/MCM-41, which implied the PA66 molecule chain penetrated into the pores of MCM-41. The spectrum observed at 1082 and 1636 cm−1 was characteristic of internal asymmetric Si–O stretching modes and SiO2, respectively. 44,45 Second, the FTIR of PA66/MCM-41 composite indicated many characteristic absorption bands not only of PA66 but also of MCM-41; and the typical polyamide peaks were at 3302 cm−1 (N–H stretching), 2934 cm−1 (C–H stretching vibration), and 1541 cm−1 (N–H bending, amide II). 46 In addition, the strong absorption peaks of MCM-41 (1082 and 1636 cm−1) were also seen in the spectrum of PA66/MCM-41, although some of them were overlapped by the absorption bands of PA66. These results indicated that Nylon salt molecule in the pores of MCM-41 had been polymerized into PA66, which was consisted with the result of N2 sorption measurements. Finally, the peak of 686 cm−1 was assigned to amide V in α phase of PA66. Further evidence can also be found at about 1541, 1419, and 933 cm−1, which was attributed to α phase of PA66 crystal structure. 47 Therefore, the PA66 macromolecule seemed to occupy the pore room of MCM-41 but do not transform the crystal phase structure of PA66.
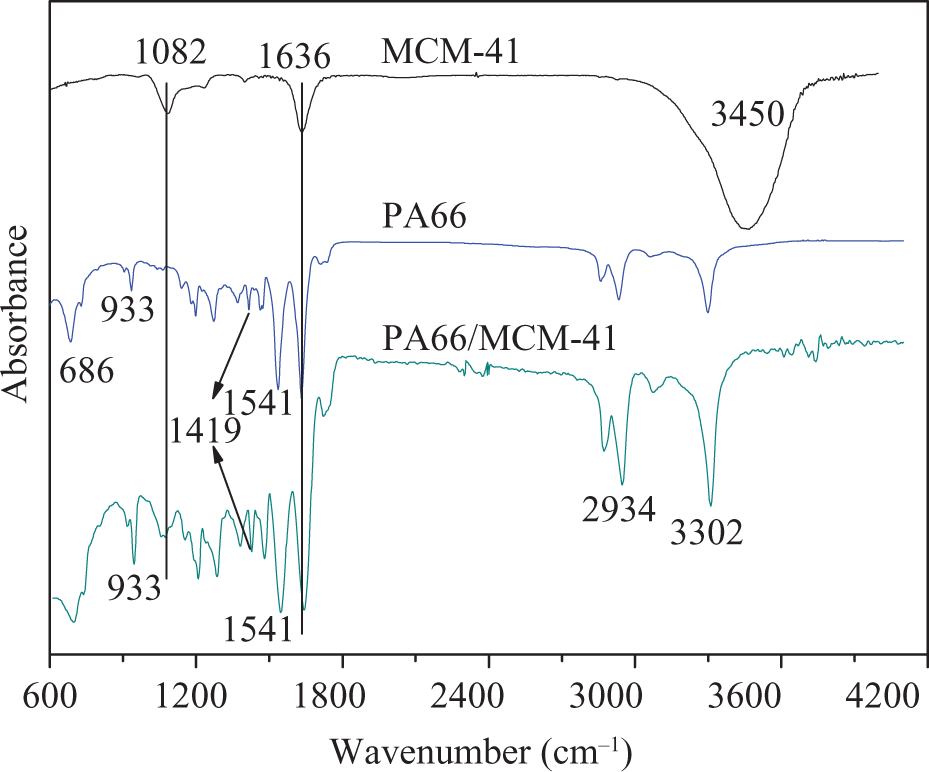
The FTIR spectra of MCM-41, PA66, and PA66/MCM-41 composites. FTIR: Fourier transform infrared; PA66: polyamide 6,6: MCM: mobile crystalline material.
Nonisothermal crystallization behavior of PA66/MCM-41 composites
The nonisothermal melt crystallization exotherms for the PA66/MCM-41 composites with 0–8% MCM-41 at different cooling rates are shown in Figure 4. With increasing cooling rate, the crystallization peaks shifted toward lower temperature, indicating that PA66 took a shorter time to crystallize.
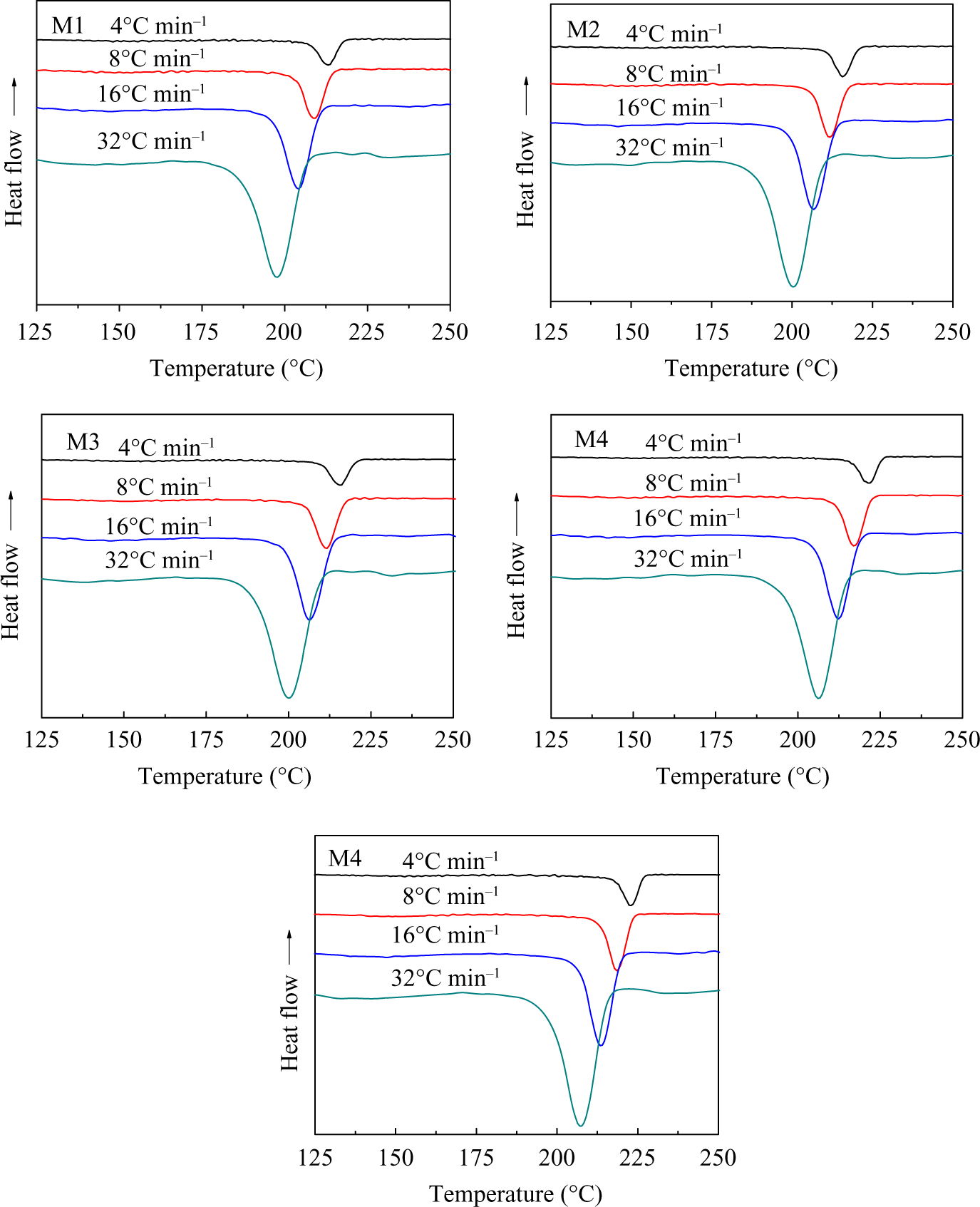
DSC curves of PA66/MCM-41 composites during nonisothermal crystallization at different cooling rates. DSC: differential scanning calorimetry; PA66: polyamide 6,6: MCM: mobile crystalline material.
In order to quantify the nonisothermal crystallization data of the PA66/MCM-41 composites, T p, which is the temperature of crystallization peak, values are listed in Table 2.
The crystallization temperature of PA66/MCM-41 at different cooling rates.
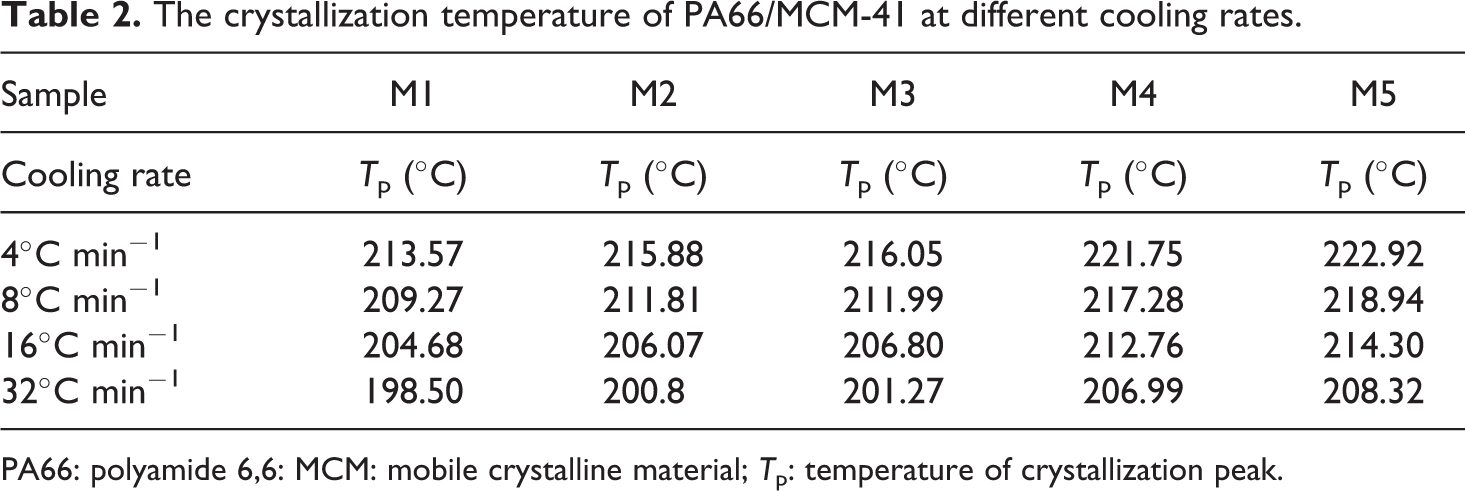
PA66: polyamide 6,6: MCM: mobile crystalline material; T p: temperature of crystallization peak.
For a given sample, it was found that all the values of T p shifted to lower values with increasing cooling rate, implying that the higher the cooling rate, the later the crystallization process began and ended. In addition, at the same cooling rate, the T p of all composites were increased with increasing MCM-41 content, indicating the crystallization of PA66/MCM-41 composites became easier. All the results showed that, when containing small amounts of MCM-41, the crystallization rate of the polymer composites decreased; the crystallization rate of the polymer composites (M5) with 8 wt% MCM-41 was the fastest, while the crystallization rate of M1 was the slowest.
Nonisothermal crystallization dynamics
In order to study the nonisothermal crystallization dynamics, the relative degree of crystallinity as a function of temperature X(T) can be formulated as
where T 0 and T represent the onset and an arbitrary temperature, respectively, dH C is the enthalpy of crystallization released during infinitesimal temperature range dT, and ΔH C is the total enthalpy of crystallization for a specific cooling condition. Based on equation (1), in order to analyze nonisothermal crystallization data obtained by DSC, the functional diagrams of X(T) was determined for the PA66/MCM-41 hybrid materials, as shown in Figure 5.
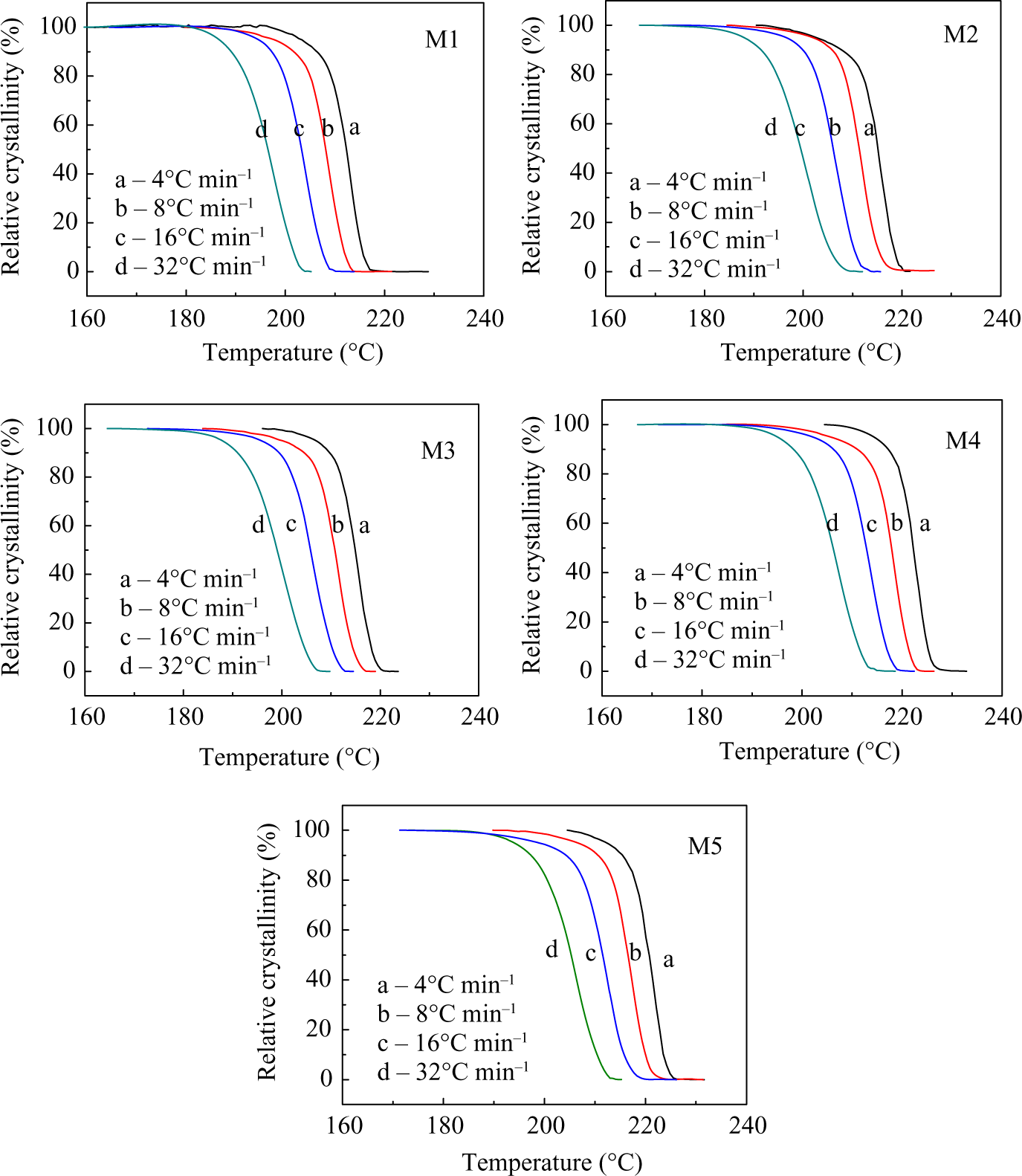
Relative degree of crystallinity as a function of temperature X(T) for PA66/MCM-41 at the cooling rates of 4, 8, 16, and 32°C min−1, respectively. PA66: polyamide 6,6: MCM: mobile crystalline material.
In nonisothermal crystallization process, the relationship of the crystallization time t to temperature T can be formulated as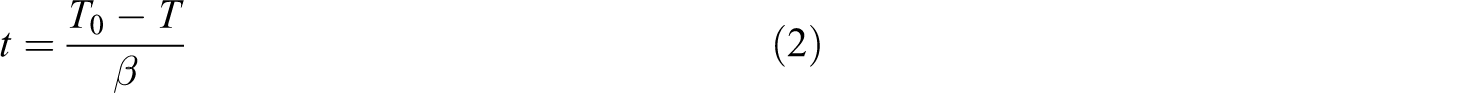
where T 0 is the temperature at which the crystallization begins (t = 0), T is the temperature at time t, and β is the cooling rate. The development of the relative crystallinity with the time for PA66/MCM-41 blends is shown in Figure 6.
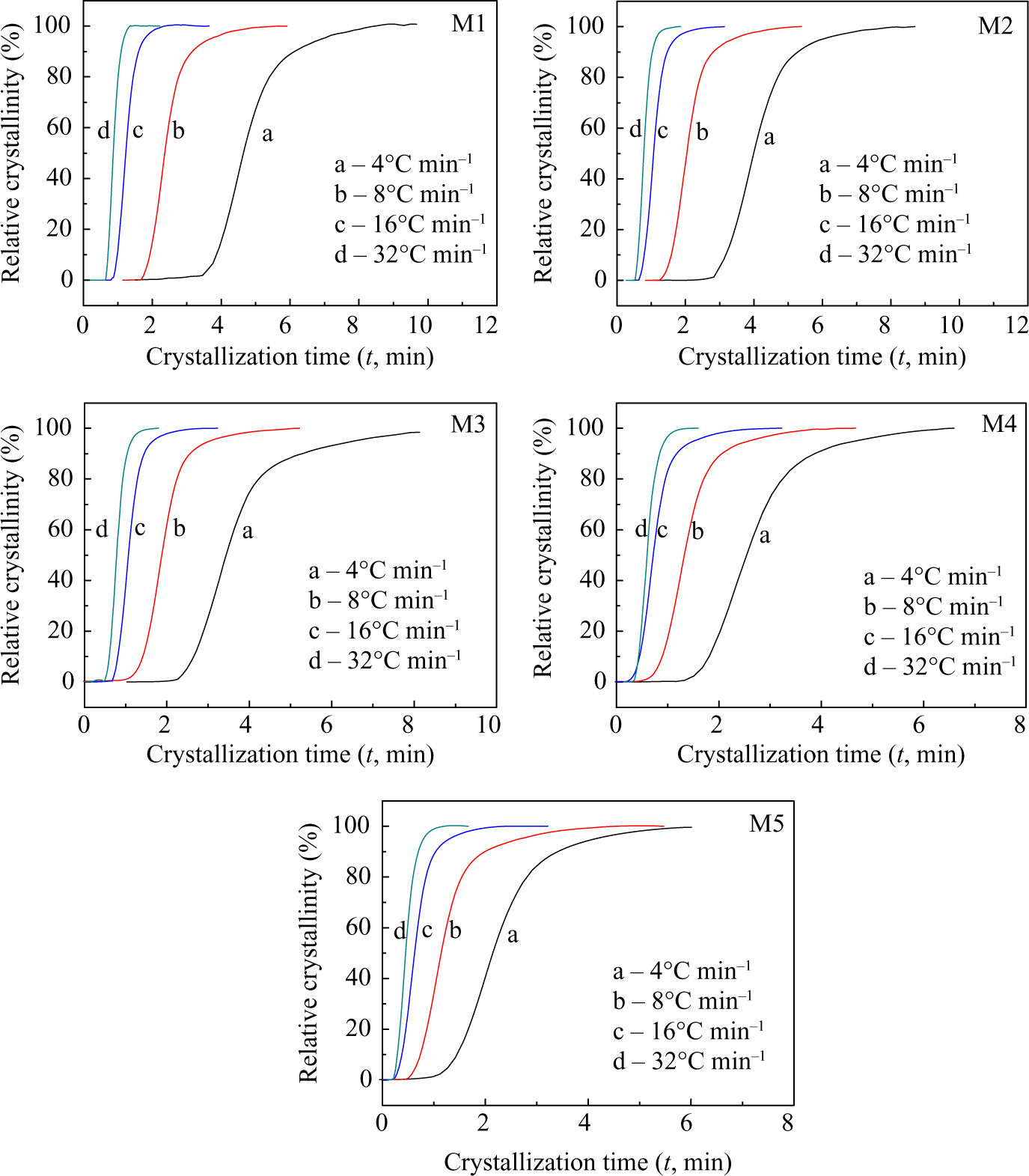
Relative degree of crystallinity as a function of time X(t) for the PA66/MCM-41 composites at the cooling rates of 4, 8, 16, and 32°C min−1, respectively. PA66: polyamide 6,6: MCM: mobile crystalline material.
The half-time of the crystallization, t 1/2, can be calculated from the equation for X(t), being the required time for crystallization to be half of the total crystallinity; the smaller the t 1/2, the faster the crystallization rate. As shown in Figure 7, t 1/2 decreased with increasing cooling rate, indicating the crystallization rate of the PA66/MCM-41 composites becoming faster with increasing cooling rate. At the same cooling rate, the t 1/2 values of PA66/MCM-41 composites with 8 wt% MCM-41 were the smallest, and the values of t 1/2 of 0% MCM-41 were the largest. All results showed that the crystallization rate of the PA66/MCM-41 composites increased with increasing MCM-41. The crystallization rates are the combined results of the two competing effects of nucleation and restriction of molecular mobility. The increase of MCM-41 in the blends enhanced the crystal nucleation, and the nucleating effect of polymer was the most significant, especially the polymer molecule chain prealignment in pore of the MCM-41. Therefore, increasing the MCM-41 can result in the reduction of t 1/2. Meanwhile, increasing the cooling rate could also raise the nucleation ability and decrease t 1/2, as shown in Figure 7.
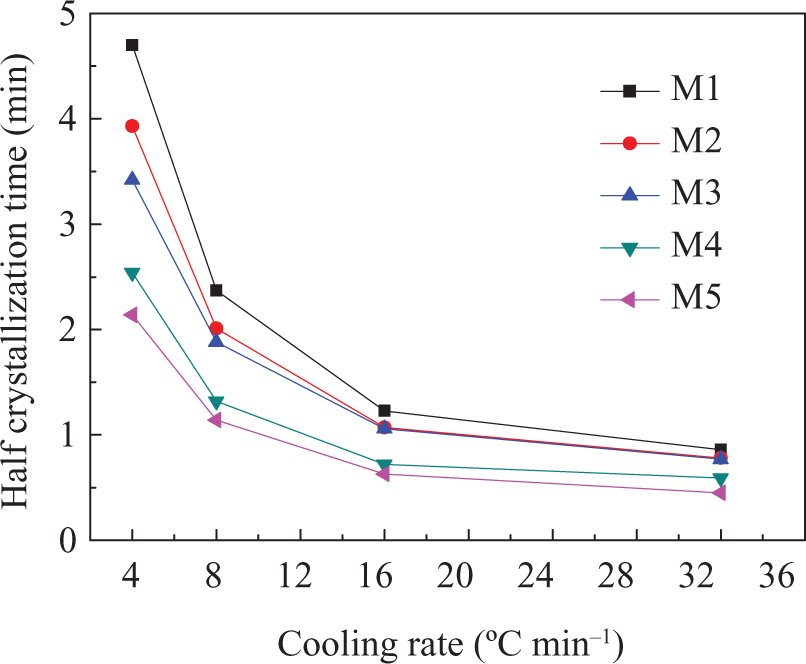
Half crystallization time as a function of cooling rates for the PA66/MCM-41 composites. PA66: polyamide 6,6: MCM: mobile crystalline material.
Ozawa analysis
Numerous theories and models can be used for the nonisothermal crystallization kinetics by modification of Avrami equation, including the Gupta,
48
Ozawa,
49
and Ziabicki models.
50
Ozawa modified the Avrami equation by assuming that a sample was cooled at constant rate. According to the Ozawa theory, X(T) at temperature T and cooling rate β is given by
where K(T) is the cooling function of nonisothermal crystallization at temperature T and m is the Ozawa exponent, depending on the nucleation mechanism and the growth dimension. Equation (3) can be rewritten as follows: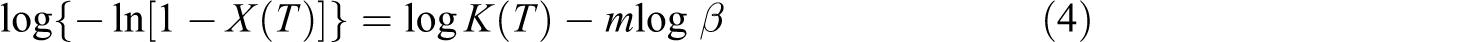
Where m and log K(T) can be easily determined from their respective slopes and intercepts on the basis of the linear relationship between
The Ozawa plots of
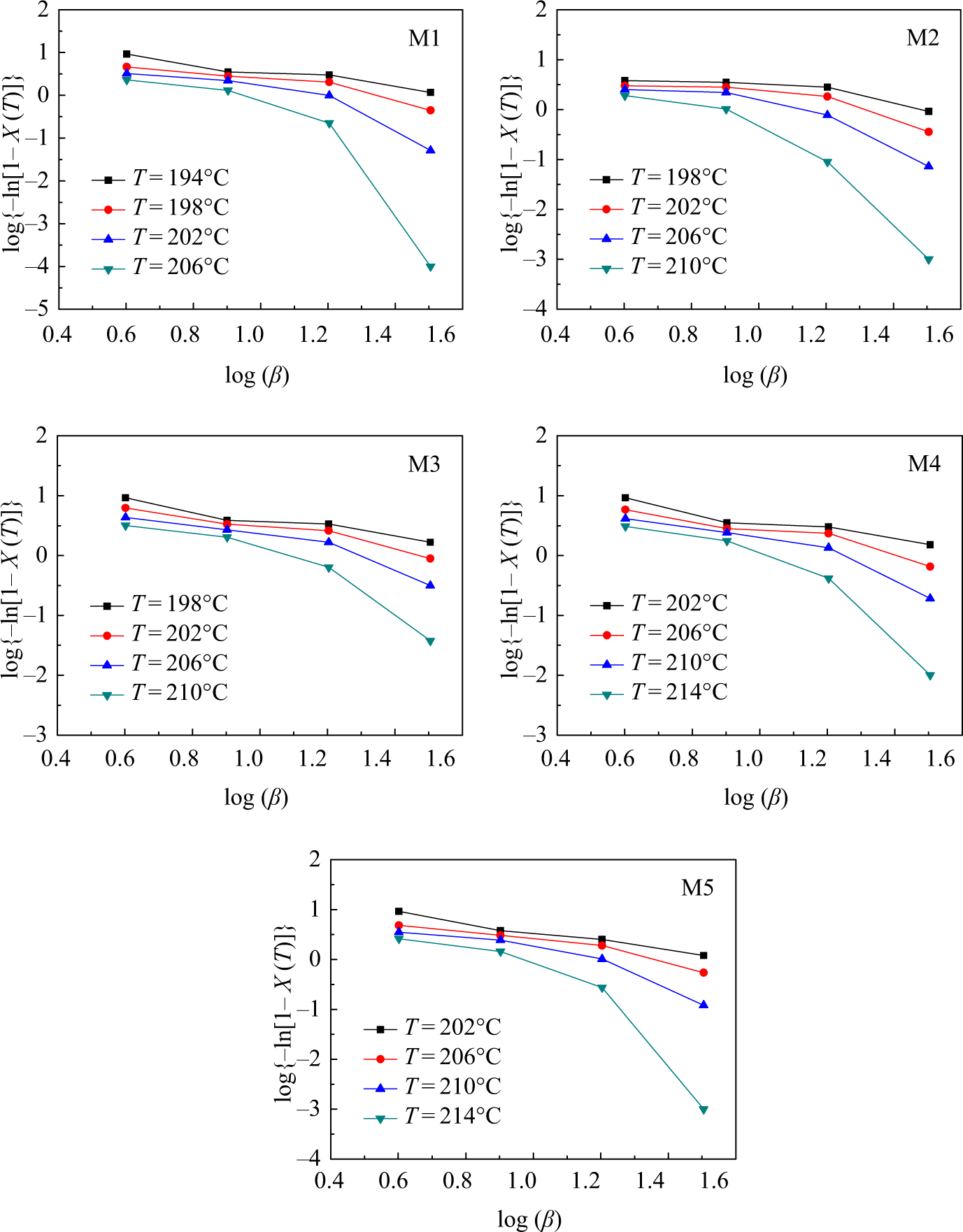
Plots of
Mo analysis (combined Avrami and Ozawa method)
Another method, developed by Mo and coworkers,
53
was used to study the nonisothermal crystallization process. Mo et al. proposed a kinetic equation (5), by combining the Avrami and Ozawa equations, which involves the relationship between cooling rate β and crystallization time t.
Rearranging equation (5), equation (6) can be obtained.
where
Plots of ln β versus ln t for various X(t) are shown in Figure 9. A linear relationship was obvious, indicating that the Mo method was satisfactory for dealing with the nonisothermal crystallization process of the PA66/MCM-41 composites. From the slope and intercept, α and F(T) can be found, respectively; the results are listed in Table 3. Table 3 showed that the F(T) values increased systematically with the relative degree of crystallinity for all the samples; within an equal time, a quicker cooling rate β was required to get a greater relative crystallinity for the same sample. The above-mentioned context indicated that the crystallization rate of the PA66/MCM-41 composites with 8 wt% MCM-41 was the fastest, which is also indicated by the previous results. The value α ranged from 1.21 to 1.66 and the variation for the same blend with different crystallinity was small, indicating that Mo’s method successfully described the nonisothermal crystallization process of the PA66/MCM-41 composites.
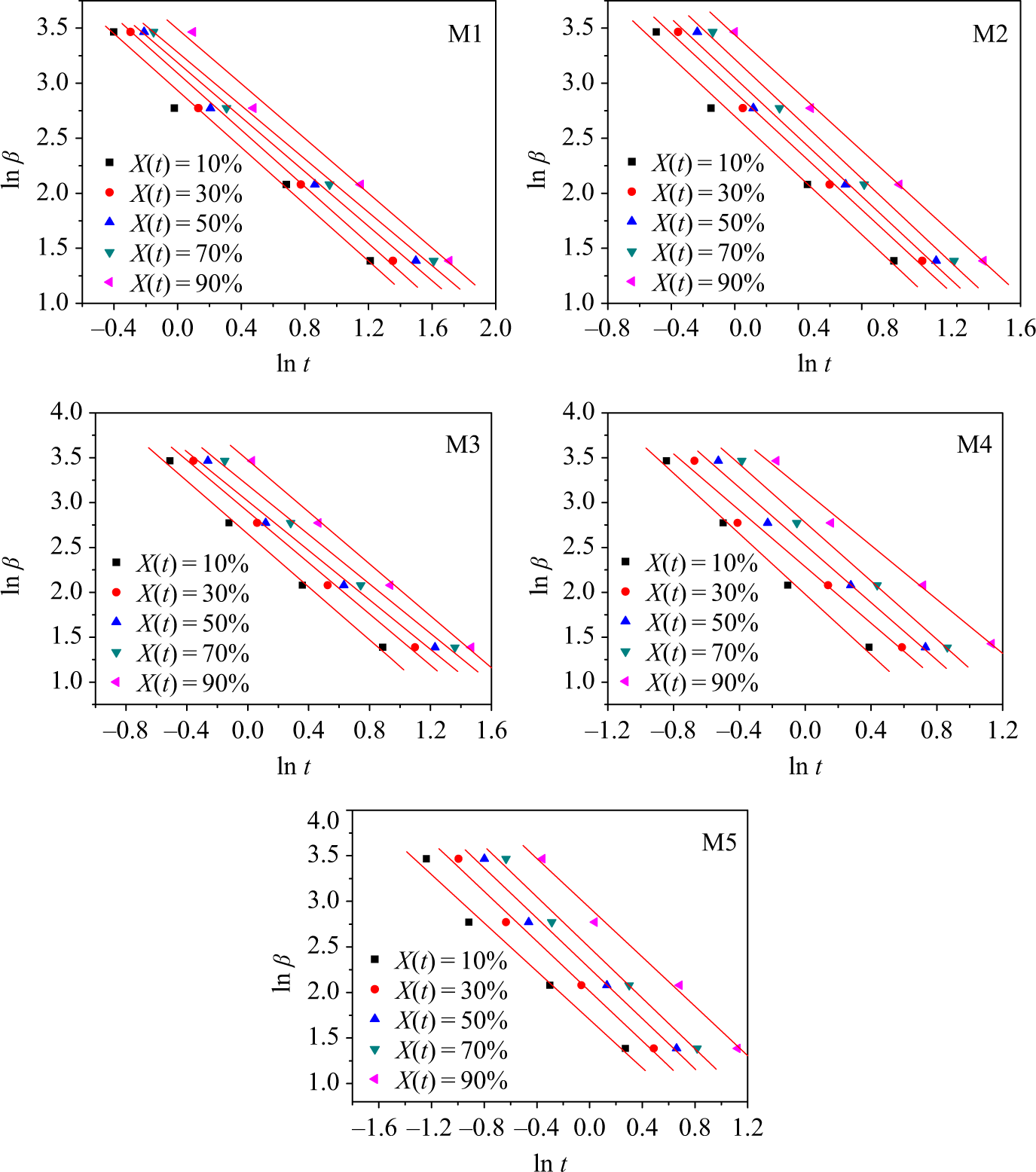
Plots of ln β as a function of ln t for PA66/MCM-41 composites at different relative crystallinity X(t). PA66: polyamide 6,6: MCM: mobile crystalline material.
Nonisothermal crystallization kinetic parameters of PA66/MCM-41 composites by Mo method.
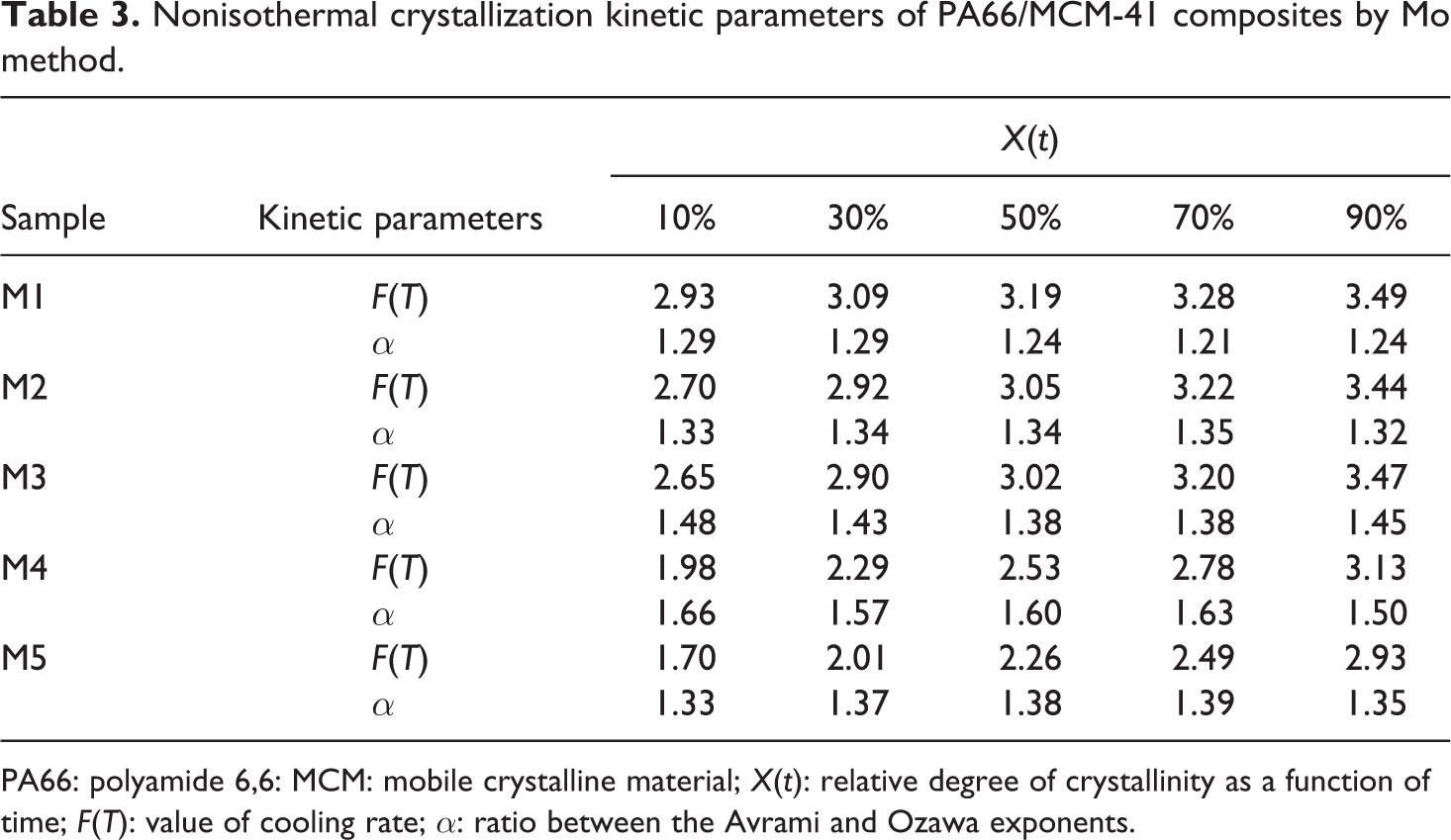
PA66: polyamide 6,6: MCM: mobile crystalline material; X(t): relative degree of crystallinity as a function of time; F(T): value of cooling rate; α: ratio between the Avrami and Ozawa exponents.
Crystallization activation energy
From the variation of T
p value with heating rate in the DSC, Kissinger
54
suggested that activation energy (ΔE) for nonisothermal crystallization can be derived from the combination of cooling rate and crystallization peak temperature. The Kissinger equation can be expressed by the following equation (7),
where β is the cooling rate, T p is the crystallization peak temperature with the cooling rate, R is the ideal gas constant, and ΔE is the crystallization activation energy.
Plots of
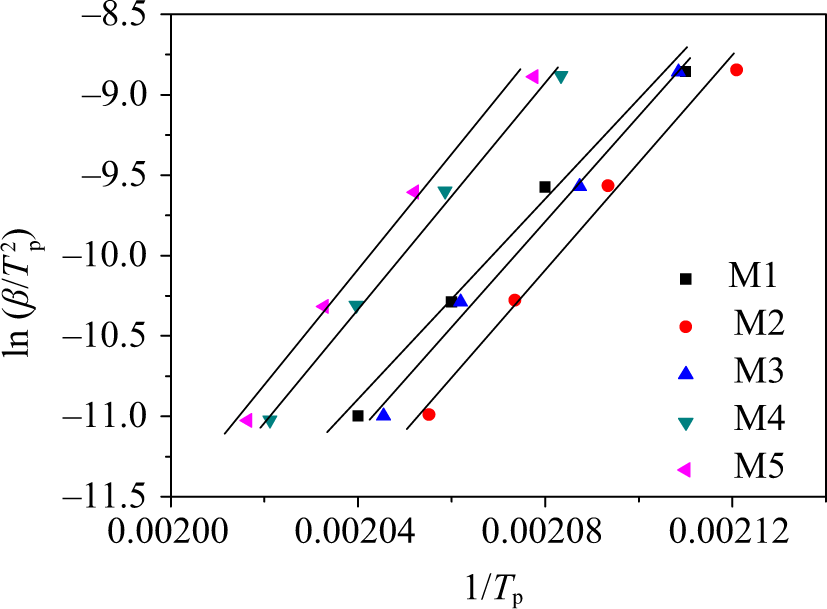
Plots of
ΔE of PA66/MCM-41.
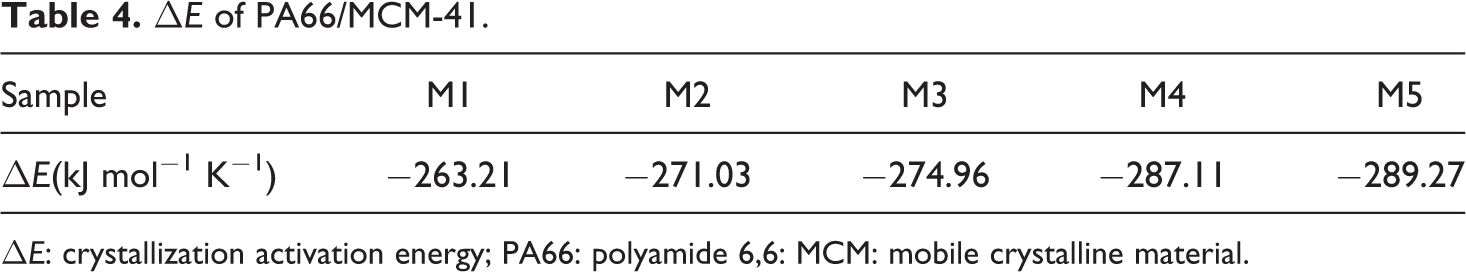
ΔE: crystallization activation energy; PA66: polyamide 6,6: MCM: mobile crystalline material.
Conclusions
PA66/MCM-41 composites are prepared by the in situ polymerization, and their nonisothermal crystallization kinetics behavior with five different ratios for four different cooling rates was investigated. The following conclusions can be obtained: The Ozawa method could not describe the nonisothermal crystallization behavior of PA66/MCM-41 composites, while the Mo’s method that combined Avrami and Ozawa equation successfully described the nonisothermal crystallization process of PA66/MCM-41 composites, and the F(T) and α were determined to be in the range of 1.70–3.49 and 1.21–1.66, respectively. In addition, the ΔE of hybrid composites was calculated with the Kissinger theory and indicated that increasing the MCM-41 content caused an increase in the crystallization ΔE. Therefore, the incorporation of MCM-41 promotes the PA66 crystallizing.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
