Abstract
The crystallization behavior of polypropylene (PP)-polyamide 6 (PA6) composites prepared by melt blending was analyzed by differential scanning calorimetry. Nonisothermal experiments were used to evaluate the effect of lithium chloride (LiCl) on the crystallization process of PP from molten state. The addition of LiCl dramatically modified the nonisothermal crystallization behavior of the PP matrix, the crystallization temperature first increased and then decreased with the increase of LiCl, and crystallization rate and effective activation energy decreased compared with PP-PA composite. The influence of LiCl on the thermal stability of PP-PA6 composites was investigated by thermogravimetry. The results showed the addition of LiCl increased the temperatures at the maximum degradation rate and improved the thermal stability of PP-PA6 composites. In addition, the hydrophilicity of PP was largely improved due to the introduction of hygroscopic PA6 and LiCl. The water contact angle of PP-PA 2.25 decreased from 103° to 65° compared with pure PP.
Introduction
Polypropylene (PP) is one of the most versatile common thermoplastic polymers owing to its outstanding performance including low cost and great chemical resistance, but it has some disadvantages such as the impact strength, and so on. 1 –6 To improve effectively the performance of PP, it is important to blend PP with other polymers. 7 –10 However, isotactic PP is a polycrystalline material, which may be in α-, β-, or γ-crystal forms and the crystal forms can be changed in the presence of the second component. 11 –14 Moreover, β-nucleated PP is generally attractive because of its good thermal and mechanical properties. Toughness and heat distortion temperature of β-PP are much larger than those of α-PP. These two characteristics are very important from an industrial point of view. However, β-PP has less stability than α-PP and large contents of β-crystal can only be obtained in special crystallization conditions. But it has been observed that β-PP blends can be prepared without any difficulty if the PP is compounded with an amorphous material.
It has been reported that polyamide 6 (PA6) and PP-g-MAH have some α-crystal-nucleating ability on PP. 15,16 However, in our previous study, it was observed that LiCl could react with the amide groups of PA6 by complexing, which changed its crystallization behavior and an amorphous PA6 was obtained in the presence of more LiCl. 17 Therefore, based on the above results, LiCl will be added in the process of preparing PP-PA6 composites with excellent properties besides maleic anhydride-grafted polypropylene (PP-g-MAH) as a compatibilizer. It is expected to find out how LiCl will affect the crystallization behavior of PP by its effect on PA6 phase, about which there was no report. Moreover, majority of the practical manufacturing techniques of polymer composites involve crystallization conditions that are analogous to nonisothermal crystallization. Thus, understanding the crystallization kinetics of polymer composites under nonisothermal conditions is very crucial from scientific prospective. Therefore, in this study, the effects of LiCl on the thermal properties and crystallization kinetics of PP-PA6 composites will be also investigated. Besides, the hydrophilicity of PP might be largely improved due to the introduction of hygroscopic PA and LiCl, which will develop a new application for it in membranes applied in wastewater treatments.
Experimental
Materials
Linear PP (T30 S PP) with 3.5 g/10 min of melt flow index was supplied by Maoming Petrochemical Corporation (China). 1013B PA6 was a commercially available product from Ube (Japan). PP-g-MAH, with the grafting degree of 0.8%, was purchased from Shanghai Sunny (China). Anhydrous LiCl was supplied by Tianjin Kermel Chemical Reagents (China).
Sample preparation
Before blending, all the materials were adequately dried in a vacuum oven at 80°C for 24 h. The weight ratio of PP, PP-g-MAH to PA6 was kept constant at 65/10/25 and the amount of LiCl was varied at 0, 0.25, 0.75, 1.25, 1.75, and 2.25 parts by weight, respectively. Then the blends were extruded on a SHJ-50 twin-screw extruder, Nanjing Jieya Extrusion Equipment Co., Ltd. (China) (L/D = 48, D = 42 mm) at 230°C with the screw rotational speed of 160 r/min and resultant samples were denoted as PP-PA, PP-PA 0.25, PP-PA 0.75, PP-PA 1.25, PP-PA 1.75, and PP-PA 2.25, respectively. Finally, all pellet samples from extruder was dried for 12 h at 80°C and then was used to prepare all testing specimens by injection molding. The temperature profile was 220, 215, and 210°C.
Characterizations
Differential scanning calorimetry
Differential scanning calorimetry (DSC) measurements were made on a DSC TA-Q10 instrument (TA Company, USA)under a nitrogen atmosphere with various cooling and heating rates (Φ) of 5, 10, and 15°C min−1. Samples of about 5–8 mg were used for all tests. Samples were quickly heated to 250°C and held at 250°C for 5 min to erase thermal history, and then cooled to 40°C and held at 40°C for 1 min. Finally, the samples were heated to 250°C again. The crystallization curve and the second melting curve were recorded.
X-ray diffraction
The X-ray diffraction (XRD) analysis was used to investigate the crystal form change of PP/PA6 composites. The measurements were performed on sheets with a D/Max-RA X-ray diffractometer at room temperature. The copper-K α radiation source was operated at 35 kV and 25 mA, and the patterns were recorded by monitoring the diffractions from 2° to 45° at the scanning speed of 3° min−1.
Thermogravimetry
The thermal degradation of PP/PA6 composites (sample mass 10 ± 0.5 mg) was performed on a TA Instruments Q50 (TA Company, New Castle, Delaware, USA) in the temperature range 50–650°C at the heating rates 10°C min−1. The nitrogen flow rate was 60 cm3 min−1.
Contact angle measurements
Both sides of the samples were tested on the DSA25 S Optical Contact Angle Tester (Germany) to measure the water contact angle (WCA) for evaluating the hydrophobicity of the composites. All WCA tests were performed with 3 μL deionized liquid water.
Results and discussion
Nonisothermal melt crystallization behavior
The crystallization and melting curves of PP-PA6 composites at various cooling and heating rates are illustrated in Figures 1 and 2, and the related data are listed in Table 1. As can be seen from Figure 1, an increase in cooling rate shifted exothermic peaks of PP and PA6 phases toward lower temperature as expected and also led to broadening of the peak. This indicates that when the samples were cooled slowly, crystallization began at higher temperature. This observation is attributed to the fact that at low cooling rates, polymer chains have sufficient time to overcome nucleation energy barrier and initiate crystallization at higher temperature. But at high cooling rates, motion of polymer chains cannot follow the cooling rate and they have less time to align themselves and hence more undercooling is required to initiate nucleation. However, the endothermic peaks of PP and PA6 hardly changed with the increase of heating rates (see Figure 2). In addition, it is also observed that with the increase of LiCl, the exothermic and endothermic peaks of PA6 phase gradually became weak, shifted toward to lower temperature, and then disappeared at 1.25 wt% of LiCl. According to our previous study, this is probably due to the reaction of LiCl with the amide groups of PA6 by complexing, which prevented PA6 from crystallizing.
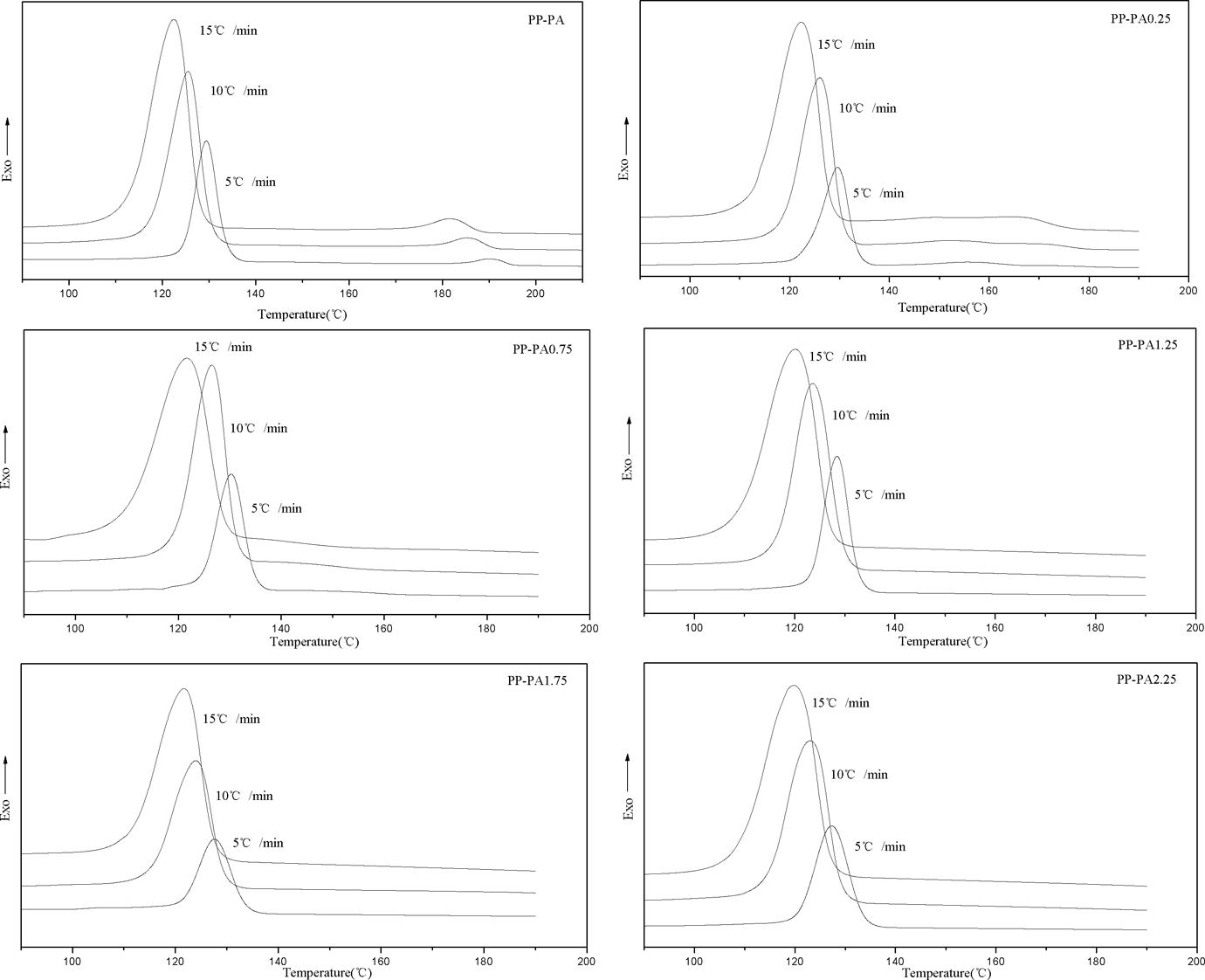
Crystallization exotherms of PP-PA6 composites at various cooling rates. PP: polyamide; PA6: polyamide 6.
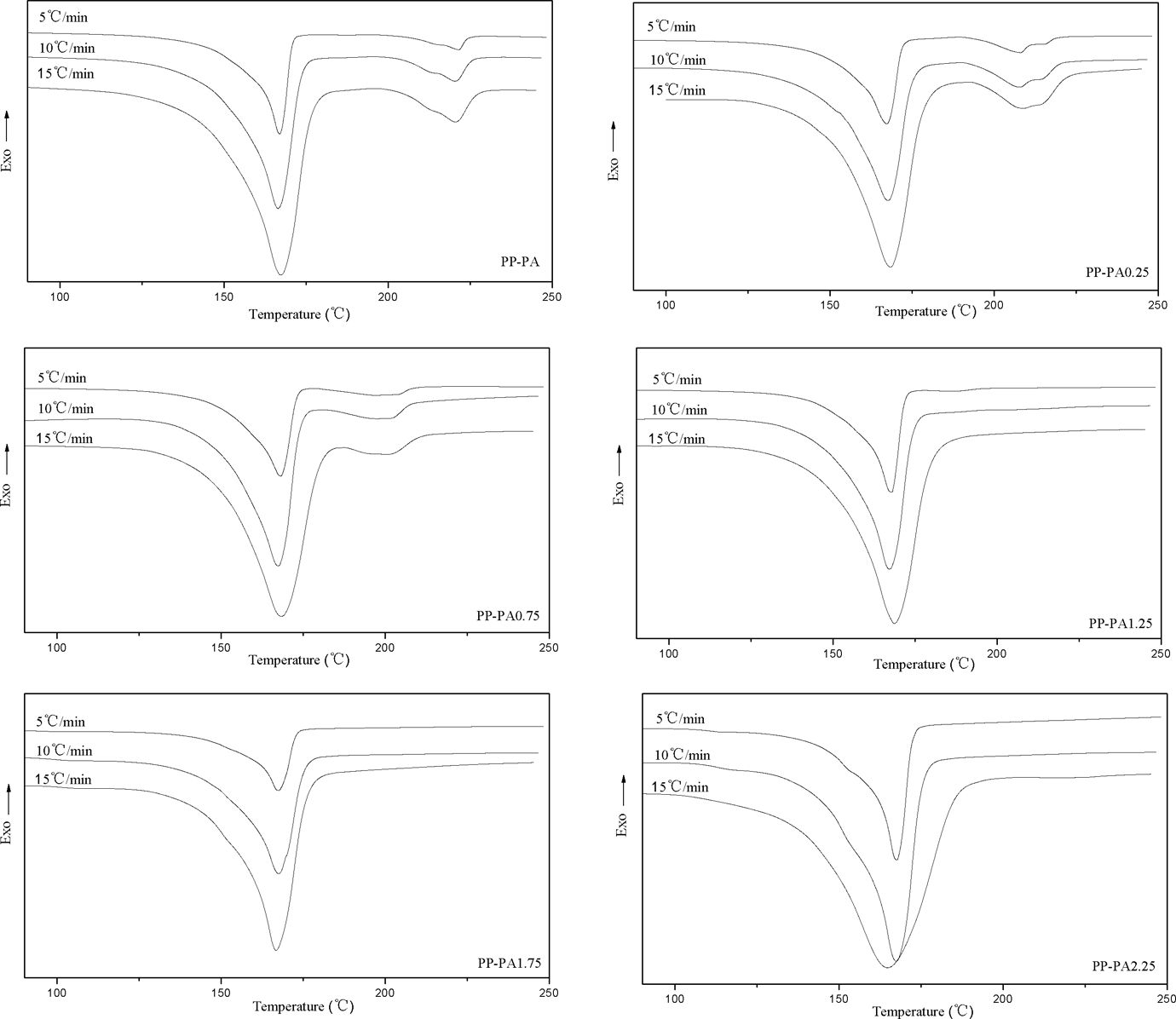
Melt endothermic curves of PP-PA6 composites at various heating rates. PP: polyamide; PA6: polyamide 6.
T c, T m, t 1/2, and CRP of PP phase in PP-PA6 composites.
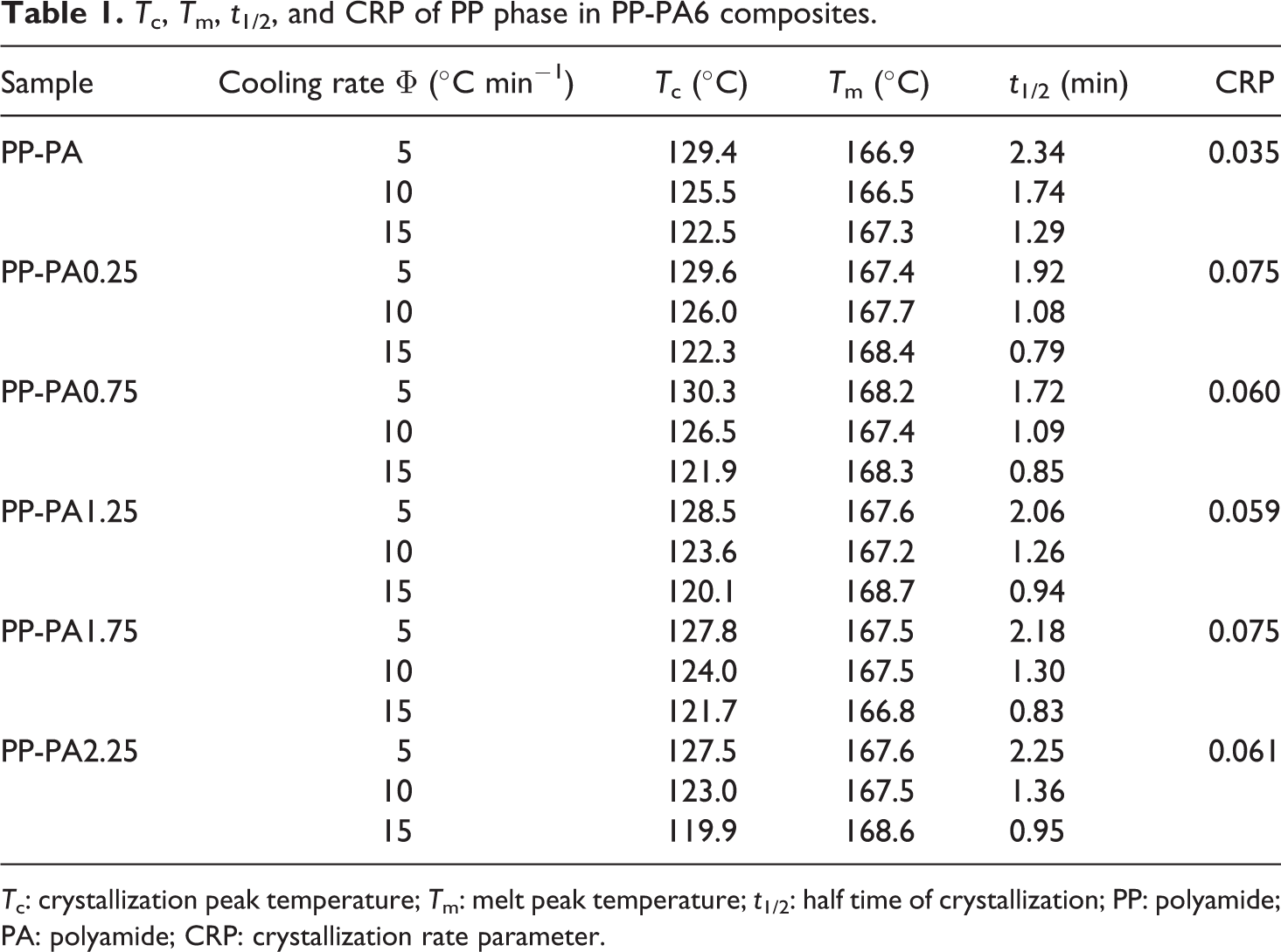
T c: crystallization peak temperature; T m: melt peak temperature; t 1/2: half time of crystallization; PP: polyamide; PA: polyamide; CRP: crystallization rate parameter.
Peak temperatures (T c), which correspond to the maximum rate of crystallization, are summarized in Table 1. Generally, T c values of PP phase in composites with LiCl were higher than that of PP-PA when the content of LiCl was less than 1.25 wt%, but the situation was just the opposite when the content of LiCl was more than 1.25 wt%, indicating that the crystallization process of PP in composites was affected by LiCl. In other words, the crystallinity and crystal forms of PP in composites might change due to the effect of LiCl on the crystallization of PA6.
XRD study of PP-PA6 composites
PP is a kind of polymorphic material with three known potential crystals, namely α-, β-, and γ-crystals. 18 Recently, more attention has been paid to isotactic PP β-crystal because of its excellent thermal and mechanical properties. In order to study the crystal form of PP in composites in detail, XRD analysis was used to investigate the crystal form change of PP-PA6 composites. As shown in Figure 3, for PP-PA, the reflection appeared at 2θ = 14.0°, 16.8°, 18.4°, 21.0°, 21.7°, and 16.1° corresponding to the planes (110), (040), (130), (111), and (131) of α-crystal and the plane (300) of β-crystal, respectively. It has been reported that PA6 and PP-g-MA have some α-crystal-nucleating ability on PP. But based on the above results, it was observed that there was also β-crystal in the PP-PA. This cause might be that during the melting blending, the terminal amino groups of PA6 and anhydride groups in PP-g-MAH reacted and formed PP-g-PA6 graft copolymer, which mainly existed at the interface of PP and PA6 phase and was wrapped in PA6 phase. In other words, PA6 phase contained the restricted PP chains. Perhaps the limited availability of PP chains hindered the crystalline transformation process from β-crystal to α-crystal and a few β-crystals were preserved.
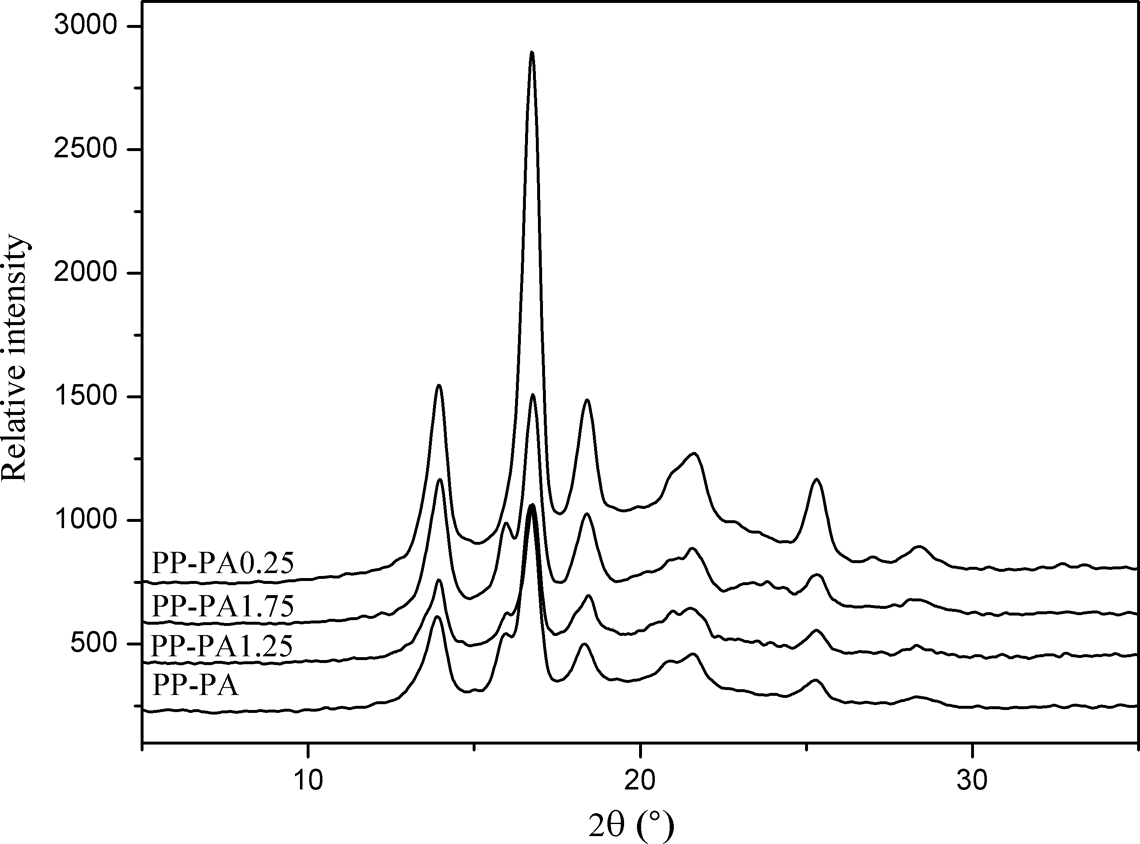
XRD spectra of PP-PA6 composites with various contents of LiCl. XRD: X-ray diffraction; PP: polyamide; PA6: polyamide 6; LiCl: lithium chloride.
Furthermore, when 0.25 wt% of LiCl was added in composites, the reflection appearing at 16.0° disappeared and the reflection at 18.4° strengthened, corresponding to the plane (300) of β-crystal and the plane (130) of α-crystal respectively, indicating the crystal form of PP in composites was affected by crystallinity of PA6. It has been observed that β-PP blends can be prepared without any difficulty if the PP is compounded with an amorphous. Based on the above DSC results, with the increase of LiCl, the PA6 phase in composites gradually transformed from semicrystalline to amorphous state. Accordingly, there might be β-crystal for PP with the increase of LiCl. As shown in Figure 3, there was β-crystal for PP in the presence of 1.25 wt% and 1.75 wt% of LiCl and reflection strength gradually increased. This was in agreement with the above inference.
Nonisothermal melt crystallization kinetics
Nonisothermal crystallization behavior of the PP-PA6 composites was investigated at different cooling rates by DSC. From the DSC curves, the values of the relative crystallinity at various cooling rates can be calculated. The relative crystallinity as a function of temperature can be defined as:
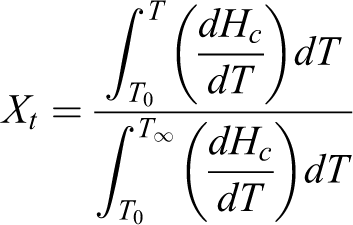
where T 0 and T ∞ are crystallization onset and end temperature, respectively. T is an arbitrary temperature and dH c/dT is heat flow rate. The horizontal crystallization temperature scale can be transformed to time axis as follows:
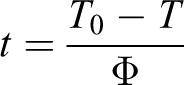
where T is the temperature at time t, T 0 is the temperature at which crystallization begins (t = 0), and Φ is the cooling rate. The development of Xt with crystallization time at different cooling rates for PP-PA6 composites is shown in Figure 4. As can be seen from Figure 4, the higher the cooling rate, the shorter time required for completion of crystallization process. An important parameter that can be readily estimated from these curves is the crystallization half time (t 1/2), which is defined as the time required for the relative crystallinity to reach 50%. The t 1/2 values of PP-PA6 composites are listed in Table 1. It is obviously observed from Table 1 that t 1/2 values of all PP-PA6 composites decreased with the increase of cooling rate, indicating that crystallization proceeded at a faster rate with increasing cooling rate. At any cooling rate, the t 1/2 values of PP-PA6 composites with LiCl were lower than those of PP-PA composite. However, for the PP-PA6 composites with LiCl, the t 1/2 values increased generally with the increase of LiCl. The t 1/2 value is a direct indication of the rate of crystallization process.
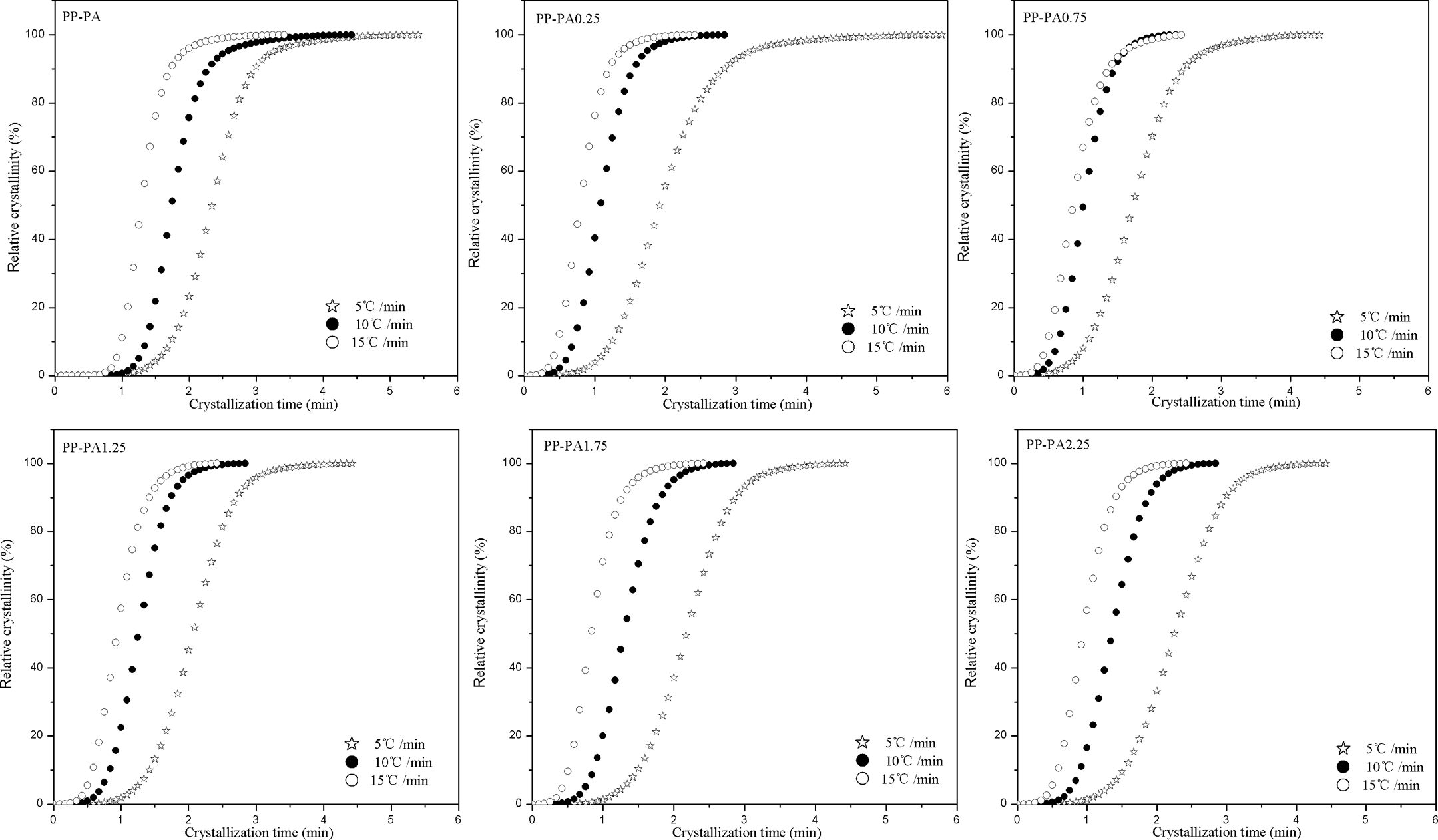
Development of relative crystallinity with time plots at different cooling rates for PP-PA6 composites. PP: polyamide; PA6: polyamide 6.
In order to quantitatively compare the nonisothermal crystallization rates of PP-PA6 composites, crystallization rate parameter (CRP) was evaluated from the slope of line by plotting the reciprocal of crystallization half time versus cooling rate. 19 –21 The higher the slope of line, the faster the crystallization rate. The 1/t 1/2 versus cooling rate plot for PP-PA6 composites is shown in Figure 5 and the corresponding CRP values are given in Table 1. As can be seen, the CRP value of PP-PA composite is 0.35 but those of PP-PA6 composites with LiCl are 0.075, 0.060, 0.059, 0.075, and 0.061, respectively. It is very obvious that the CRP values of PP-PA6 composites with LiCl are higher than that of PP-PA composite. This suggested that crystallization rate of PP phase in composites was enhanced by the addition of LiCl but hardly affected by its content.
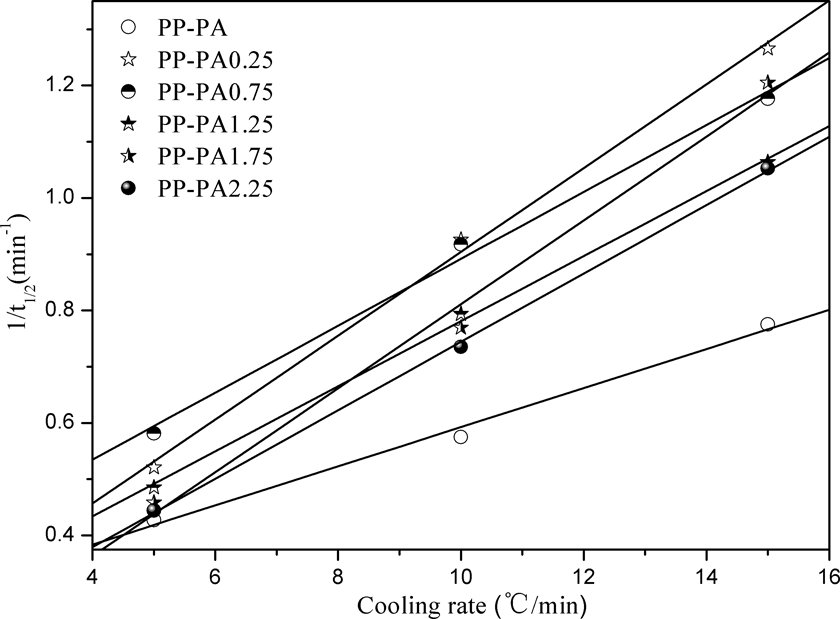
Reciprocal half time of crystallization versus cooling rate plot of PP-PA6 composites. PP: polyamide; PA6: polyamide 6.
Jeziorny-modified Avrami 22,23 and Ozawa 24 analyses were usually used to study the nonisothermal crystallization kinetics. However, based on literature survey, a model proposed by Liu and Mo can be used to describe the nonisothermal crystallization process more effectively, which is deduced by combining Avrami equation and Ozawa equation as follows:
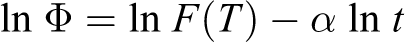
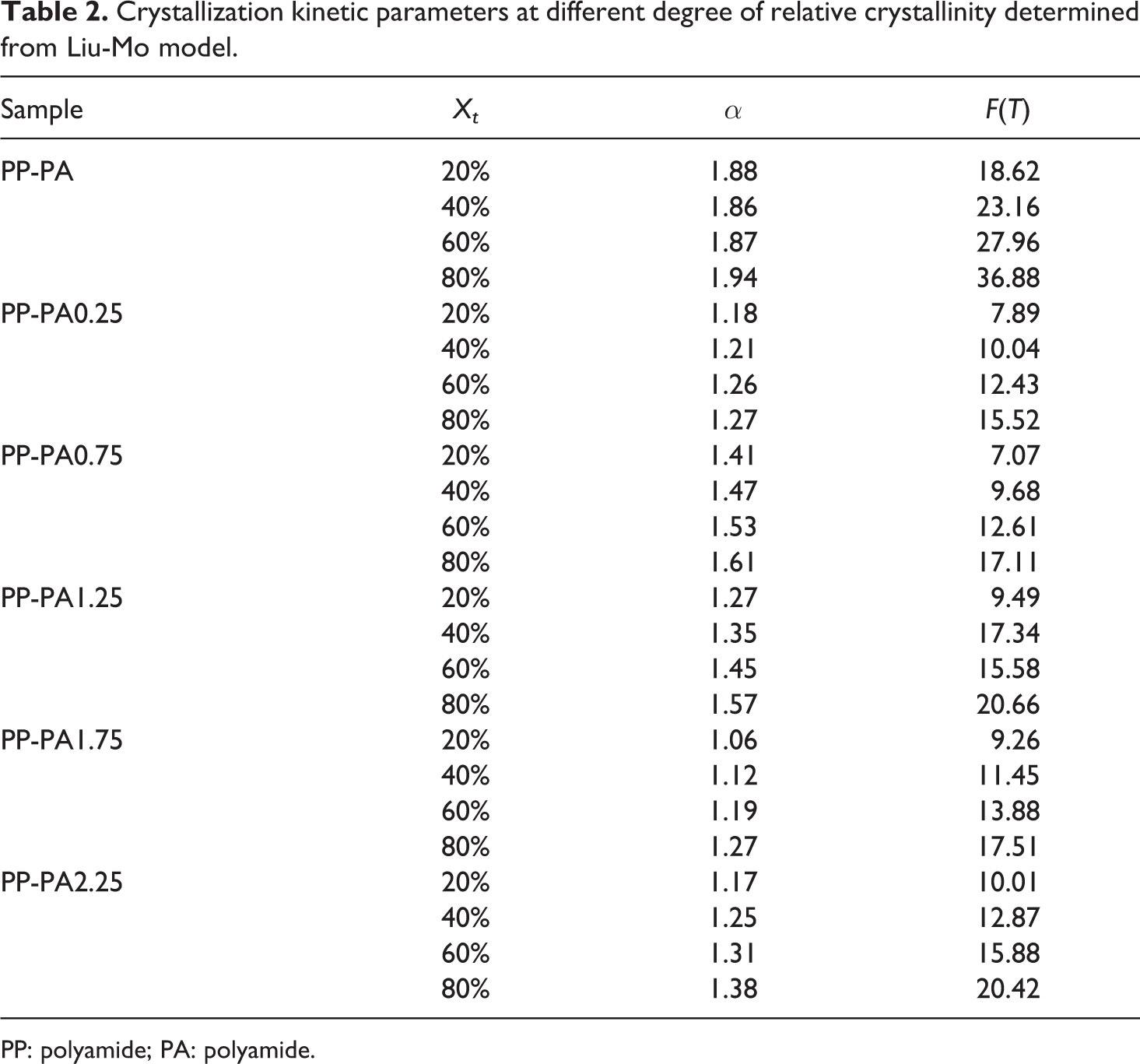
where F(T) refers to a value of cooling rate that has to be chosen at a unit crystallization time when the system has to reach a certain degree of crystallinity, and α is the ratio of Avrami exponent to Ozawa exponent. F(T) value has a definite physical and practical meaning and the smaller the value, the higher the crystallization rate. The plot of lnΦ against ln t at a given relative crystallinity should give a straight-line from which kinetic parameter F(T) and α can be determined from intercept and slope, respectively. The values of F(T) and α calculated from the lnΦ versus lnt plots at different relative crystallinity for PP-PA6 are given in Table 2. From these values, it can be observed that all samples exhibit a linear relationship and the variation in α values is in a small range indicating that this method is suitable for describing the nonisothermal crystallization process.
Crystallization kinetic parameters at different degree of relative crystallinity determined from Liu-Mo model.
PP: polyamide; PA: polyamide.
It is found that the F(T) values for all the samples increased steadily with an increase in relative degree of crystallinity indicating that at unit crystallization time, higher degree of relative crystallinity can be obtained with a higher cooling rate. Additionally, at a given degree of relative crystallinity, comparison of F(T) values of different samples revealed that the values of PP-PA6 composites with LiCl were lower than that of PP-PA. For a given degree of relative crystallinity at a given cooling rate, the higher the F(T) values, the longer the crystallization time needed to reach that crystallinity, indicating the difficulty of polymer crystallization. This result is in good accordance with the values obtained from CRP analysis.
Effective activation energy of nonisothermal crystallization
Apart from the nonisothermal crystallization kinetics study of PP-PA6 composites, the effective activation energy barrier (ΔE) associated with nonisothermal crystallization process has also been investigated as it determines the rate of crystallization. In the current study, the isoconversional method of Friedman 25 has been used to evaluate effective activation energy. In the view of literatures, this method is most appropriate since it evaluates effective activation energy for every degree of crystallinity by using the Friedman equation expressed as follows:
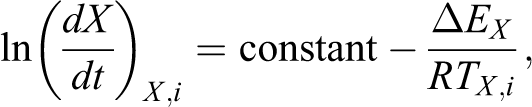
where dX/dt is the instantaneous crystallization rate as a function of time at a given degree of crystallinity (X), ΔEx is the effective activation energy at a given degree of conversion, Tx,i is a set of temperatures related to X at different cooling rates, and subscript i refers to different cooling rates.
According to this method, initially the instantaneous crystallization rate (dX/dt) is calculated by differentiating Xt with respect to time. Then by selecting various degree of crystallinity from 5% to 95%, the appropriate values of dX/dt at that specific Xt are correlated to corresponding crystallization temperature Tx . Subsequently plotting left side of equation against 1/Tx at different relative crystallinity gives straight-lines with slope equal to ΔEx /R.
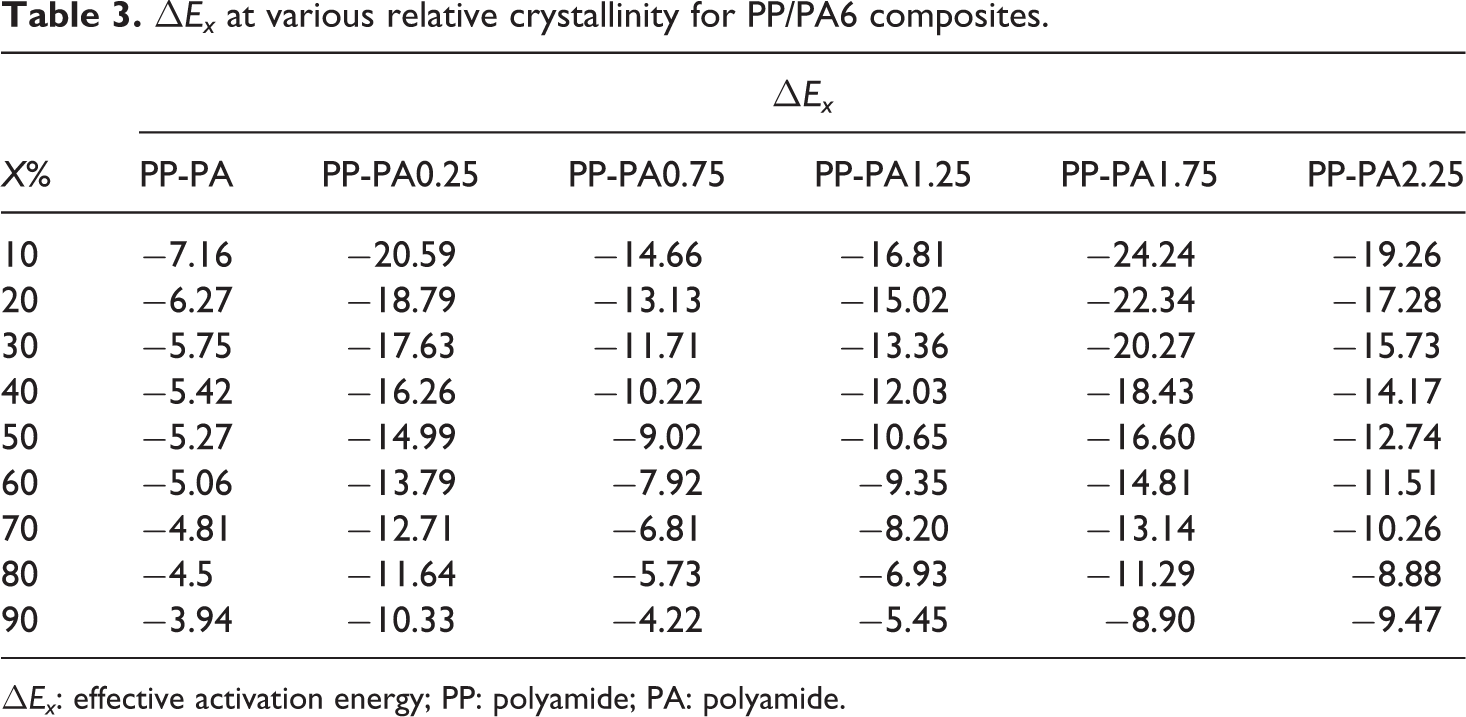
The effective activation energy thus obtained for PP-PA6 composites is listed in Table 3 and plotted as a function of relative crystallinity as shown in Figure 6. It should be noted that the lower the ΔE values, the higher the crystallization tendency of polymer. From Figure 6 and Table 3, it can be seen that the effective activation energy for all PP-PA6 composites went on increasing gradually with an increase in Xt , indicating that crystallization of polymer became difficult with an increase in crystallinity. As crystallization proceeded, diffusion of polymer molecular segments from the melt to the growth front will be deterred by the rejected molecular segments.
ΔEx at various relative crystallinity for PP/PA6 composites.
ΔEx : effective activation energy; PP: polyamide; PA: polyamide.
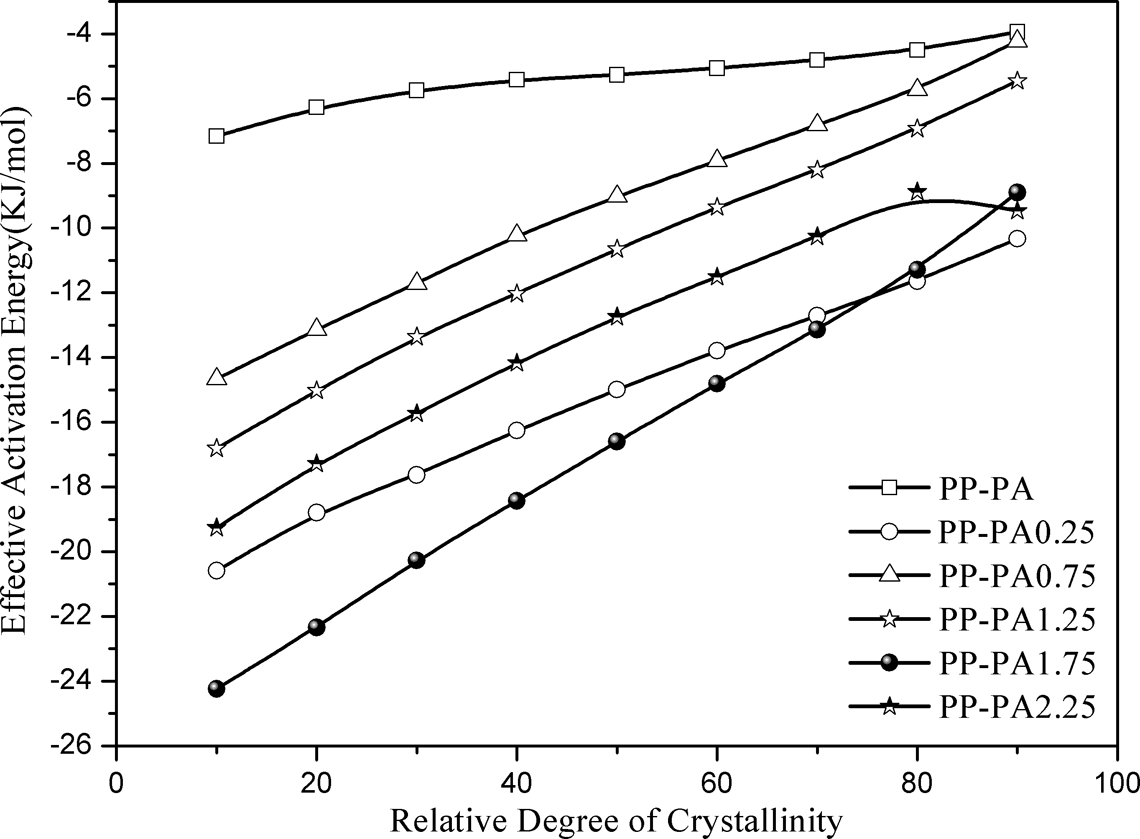
Effective activation energy as a function of relative crystallinity for PP-PA6 composites. PP: polyamide; PA6: polyamide 6.
Besides this it is observed that ΔE values for PP-PA6 composites with LiCl were lower than that of PP-PA, which is in accordance with the aforementioned crystallization kinetics study but increased generally with the increase of LiCl. According to the above DSC and XRD analyses, this indicated that the crystallinity and crystal forms of PA6 phase had a pronounced effect on the effective activation energy. In addition, it is found that ΔE values for PP-PA6 composites with LiCl approached the value of PP-PA at the end of crystallization. This is probably due to the nucleating efficiency of PA6 phase to induce heterogeneous nucleation and reduce the energy barrier to nucleation.
Thermal properties of PP-PA6 composites
Figure 7 shows thermogravimetric analysis curves of all samples. It is found that PP-PA6 composites are thermally stable in the applied temperature region, and degradation temperature (T 0) corresponding to the mass loss (approximately 5 wt%) decreased with the increase of LiCl, which is attributed to their dehydration due to the hygroscopic property of LiCl. The residual mass (mf ) increased gradually with the addition of LiCl due to its thermal stability in this temperature range. Additionally, in order to establish the influence of the LiCl content on the thermal stability of PP-PA6 composites, the degradation temperatures (T 0) and the temperatures at the maximum degradation rate (T max) are given in Table 4. In general, the T max values of PP-PA6 composites with LiCl are higher than that of PP-PA, showing that the introduction of inorganic components into organic materials improved their thermal stabilities due to the physical protective barrier and delayed volatilization of degradation products. 26
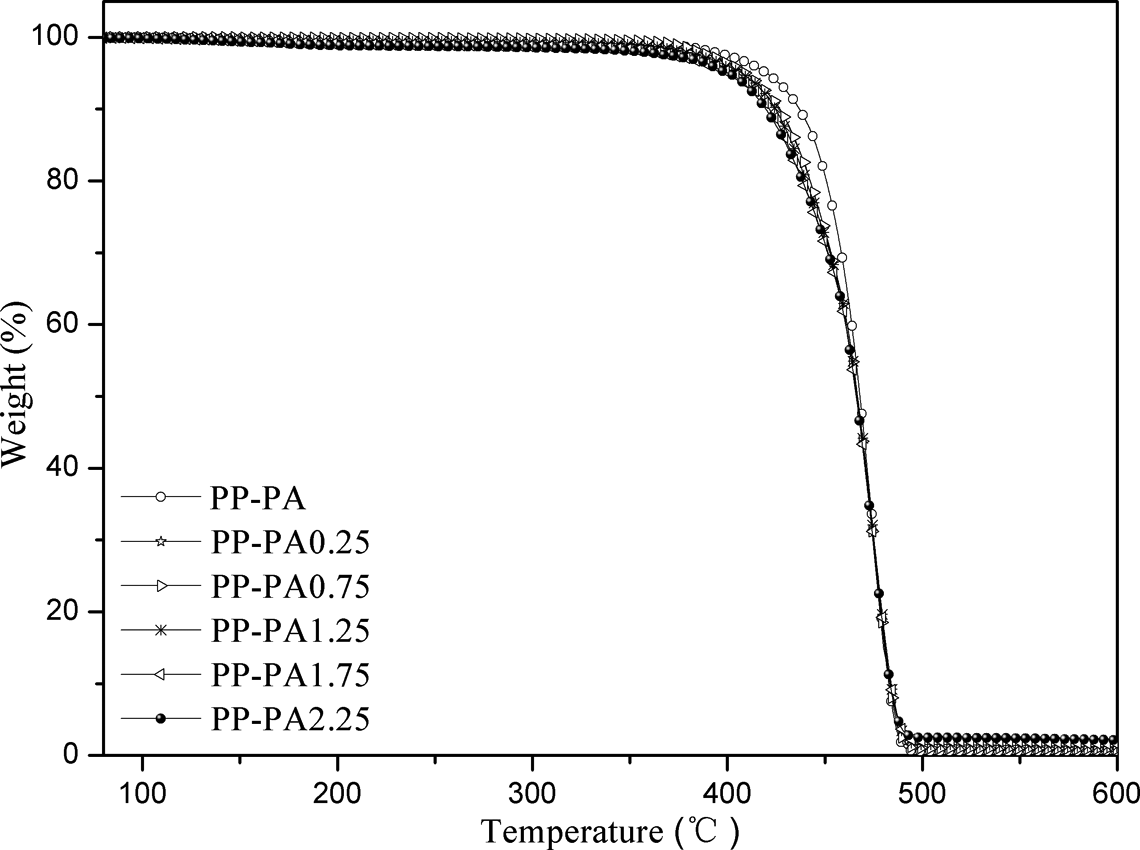
TGA curves of PP-PA6 composites. TGA: thermogravimetric analysis; PP: polyamide; PA6: polyamide 6.
Effect of the LiCl content on the thermal characteristics of PP/PA6 composites.

LiCl: lithium chloride; PP: polyamide; PA: polyamide; T max: maximum degradation rate.
The hydrophilicity of PP-PA6 composites and its application in membrane materials
Polymer membranes are increasingly employed for separation processes. They are relatively economic and cover a broad range of applications from microfiltration to reverse osmosis. But biofouling is a major problem of membrane processes in bioseparation, artificial organs, or water and wastewater treatments. However, it is generally acknowledged that hydrophilic membranes have lower fouling potential than hydrophobic ones. Therefore, biofouling of polymer membranes was improved by developing hydrophilic materials. The WCA data of PP and PP-PA6 blends are listed in Table 5. Figure 8 shows the shapes of water drop on pure PP and PP-PA2.25, respectively. As shown in Table 5 and Figure 8, WCA of PP-PA6 blends decreased from 103° to 65° and their hydrophilicity was greatly improved compared with pure PP. Thus, PP-PA blends may be new materials for polymer membranes.
WCAs of PP and PP/PA blends.

WCA: water contact angle; PP: polyamide; PA: polyamide.

Shapes of water drop on pure PP and PP-PA2.25: (a) pure PP and (b) PP-PA2.25. PP: polyamide; PA: polyamide.
Conclusions
The nonisothermal crystallization behavior of PP-PA6 composites has been investigated in this study. The crystallization temperature and crystal forms of PP phase were influenced by the crystallization behavior of PA6 phase owing to the addition of LiCl. It is found by nonisothermal crystallization kinetics study that at any cooling rate, the t 1/2 values of PP-PA6 composites with LiCl are lower than those of PP-PA composite, F(T) values lower, and CRP parameters higher. This revealed that the addition of LiCl enhanced the crystallization rate of PP phase in composites. These results are also supported by the effective activation energy calculations by Friedman’s method, which showed that ΔE values for PP-PA6 composites with LiCl were lower than that of PP-PA. In addition, the thermal stability of PP-PA6 composites was slightly improved due to the introduction of LiCl, and PP-PA6 composites as a new material may be applied in wastewater treatment due to the improvement of their hydrophilicity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support from Development Program of China (863 Program) (Grant No. 2012AA03A601).
