Abstract
Poly(ethylene glycol) (PEG) was mixed at various weight percentage with biodegradable poly(lactic acid) (PLA) via direct melt compounding method. Nanocomposites of PLA/PEG, with two types of organoclay namely, cloisite 93A (C93A) and cloisite 30B (C30B) were also prepared by melt blending method. The crystallization behaviors of PLA/PEG and PLA/PEG/organoclay were studied using differential scanning calorimetry (DSC). The isothermal crystallization kinetics has been investigated using the Avrami’s equation. In PLA/PEG, the PLA crystallized first followed by crystallization of PEG during the cooling. The viscoelastic behavior of the PLA/PEG and PLA/PEG/organoclay were investigated by dynamic mechanical analysis where the nanocomposites showed higher magnitude of storage modulus. The plasticizing effect of PEG acts predominantly in the mechanical performance of PLA/PEG, which shows increased percentage elongation and impact strength in the blend. The sample with 20 wt% PEG showed optimum impact strength. Again, the nanocomposite with 3 wt% C30B exhibited better tensile strength and modulus than the sample with C93A. The characterization of fractured surface of the matrix polymer and the blend were compared through scanning electron microscopy. The rheological and compostability study of the system were further investigated.
Introduction
Nowadays, the consumption of biodegradable materials increased as a new group of products. Biodegradable materials are used in specialist industry (tissue engineering), in mass production (packaging), and also have been applied as modifiers accelerating decomposition of synthetic polymers in natural environment. 1,2 Among the biodegradable polymers, aliphatic polyester is one of the most promising biodegradable materials because they are readily susceptible to biological attack. 3 –5
Poly (lactic acid) (PLA), biodegradable aliphatic polyester, produced from renewal resources has received much attention in the research of alternative biodegradable polymers. 6,7 PLA shows good biocompatibility and physical properties, such as high mechanical strength, thermoplasticity, and fabricability. 8 However, their relatively stiff, brittle, slow crystallization, and hydrolysis properties are restrict their use in many applications. The limitations can be improved through several approaches including copolymerization, 9,10 blending, 11 –13 plasticization, 14 –16 and incorporation of filler materials. As compared to copolymer synthesis, the blending technique is a simple and more economic way. Among the organic and inorganic additives, the blends of PLA with low-molecular-weight polymer poly(ethylene glycol) (PEG), which act as a biodegradable plasticizer has proved its efficiency to strongly increase the elongation at break and the impact resistance. 17 This combination of PLA/PEG will be in line to compete with more flexible and ductile polymer such as polyethylene or polypropylene. 17 Jacobsen and Fritz 15 investigated the properties of plasticized PLA, with 2.5–10 wt% of PEG (molecular weight (Mw) = 1.5 × 103gmol−1) and observed decreased tensile strength and modulus with an increase in percentage of elongation at break. Also, increased impact resistance to about five times more as compared to that of a pure PLA observed with the addition of 10 wt% of PEG. Kulinski and Piorkowska 18 reported that incorporation of 10 wt% of PEG, within amorphous plasticized PLA results in considerable deformation to about 550%, while semicrystalline PLA exhibits nonuniform plasticization of the amorphous phase and showed less ability to the plastic deformation. Further due to the difference in crystallization temperature of PLA matrix and PEG plasticizer, the PLA/PEG blends are miscible in the amorphous phase. The mechanical properties of PLA/PEG blends are becoming lost over time which results in crystallization and phase separation. 19
Further several authors revealed that PEG act as an effective polymeric plasticizer to facilitate the crystallization rates of the PLA.
18,20
–23
With the addition of PEG, the cold crystallization temperatures of PLA molecules decreased remarkably as the PEG accelerates the crystallization ability of PLA.
21
Further study on isothermal crystallization of PLA/PEG blend at 120°C revealed that the spherulite growth rates and nucleation densities of PLA molecules increased and decreased, respectively.
21
–23
The study of crystallization behavior and miscibility in the polymer blends are very interesting and important to evaluate the properties. The miscibility and crystallization effect of poly(ethylene oxide) (PEO)/
The addition of PEG reduced the glass transition temperature (Tg) and the melting temperature (Tm) of the virgin polymer. 14,16,26 The lower Tg value of PLA/PEG may affect the processing and molding of final products. However, the results of thermal properties as well as stability and gas-barrier properties of PLA/PEG materials still need to be improved for many packaging applications.
Thus, incorporation of organoclays to PLA/PEG has received considerable importance to improve the properties. Paul et al. 17 studied thermal and morphological properties of PLA/PEG/organoclay nanocomposites prepared by melt blending technique. Shuichi et al. 27 also studied various properties of the PLA/PEG/organoclay nanocomposites using two grades of PEG with different molecular weights along with the organoclay by melt blending technique.
This article investigates the preparation and properties of PLA/PEG and PLA/PEG/organoclay obtained by melt intercalation method. The optimized PLA/PEG ratios were investigated through the study of mechanical properties. The thermal, morphological, and dynamic mechanical properties were studied for PLA/PEG, PLA/PEG/C93A, and PLA/PEG/C30B. Further, the rheological and compostability studied were made for the optimized PLA/PEG/organoclay. The rheological study plays an important role in polymer processing, and the compostability test is a prime concern of environmental issues. Differential calorimetric analysis was also performed to investigate the thermal and crystallization behavior of these combinations.
Experimental
Materials and melt blending
PLA (NatureWork 4042D) with 1.24 g cm−3 as density and average molecular weight of 74,000 g mol−1 was purchased from M/s Cargill Dow (Blair, Nebraska, USA) and consists of 92%
Prior to processing, PLA was dried at 50°C for 6–8 h, PEG was dried at 40°C for 3–4 h, and the nanoclay was dried at 80°C for 8–10 h, respectively. PLA matrix, PLA/PEG, and PLA/PEG/organoclay were prepared in a micro 15 cc twin-screw compounder (DSM Xplore, Netherlands) using melt blending technique with extruder barrel temperature of 170, 175, and 180°C at screw speed of 40 r min−1 and acceleration rate of 50 r min−1. Subsequently, the melt mixtures were transferred to the specimen molds by the microinjection molding machine with process parameters of 180°C temperature, 7–7.5 bar injection pressure, and cycle time of 10 s. The specimens were prepared as per the ASTM D standard. The details of the composition of the melt-blended specimens prepared in this study are summarized in Table 1.
Details of the compositions of the melt-blended specimens.
PLA: poly(lactic acid); PEG: poly(ethylene glycol); C93A: cloisite 93A; C30B: cloisite 30B.
Measurements
Tensile and impact characterization of specimens
The tensile specimens of dimension 167 × 12.7 × 3 mm3 were subjected to tensile test, using universal testing machine (UTM, Instron 3386, UK) as per ASTMD638 standard, at crosshead speed of 5 mm min−1 and gauge length of 50 mm. The izod impact test specimens of dimensions 63.5 × 12.7 × 3 mm3 has been carried out as per ASTM D-256 standard, and the samples were notched with a ‘V’ notch at depth of 2.54 mm and notch angle of 45°, using Tinius Olsen Model 899 specimen notcher (Horsham, Pennsylvania, USA). The impact test was carried out using Tinius Olsen Model IT504 Plastic Impact (USA).
DSC analysis
Test samples of ≤5 mg were heated from 30 to 200°C at a rate of 5°C min−1 under a constant flow rate of 20 ml min−1 nitrogen gas purging through the differential scanning calorimetry (DSC) (Perkin-Elmer Diamond DSC, Waltham, Massachusetts, USA) to study the Tg, crystallization temperature (Tc), and Tm. The sample was kept at 200°C for 1 min and subsequently cooled to 30°C and then heated up to 200°C with the same heating rate of 5°C min−1. The second heating cycle was followed to erase previous thermal history in the samples. Corresponding Tg and Tmwere represented from the second heating curve.
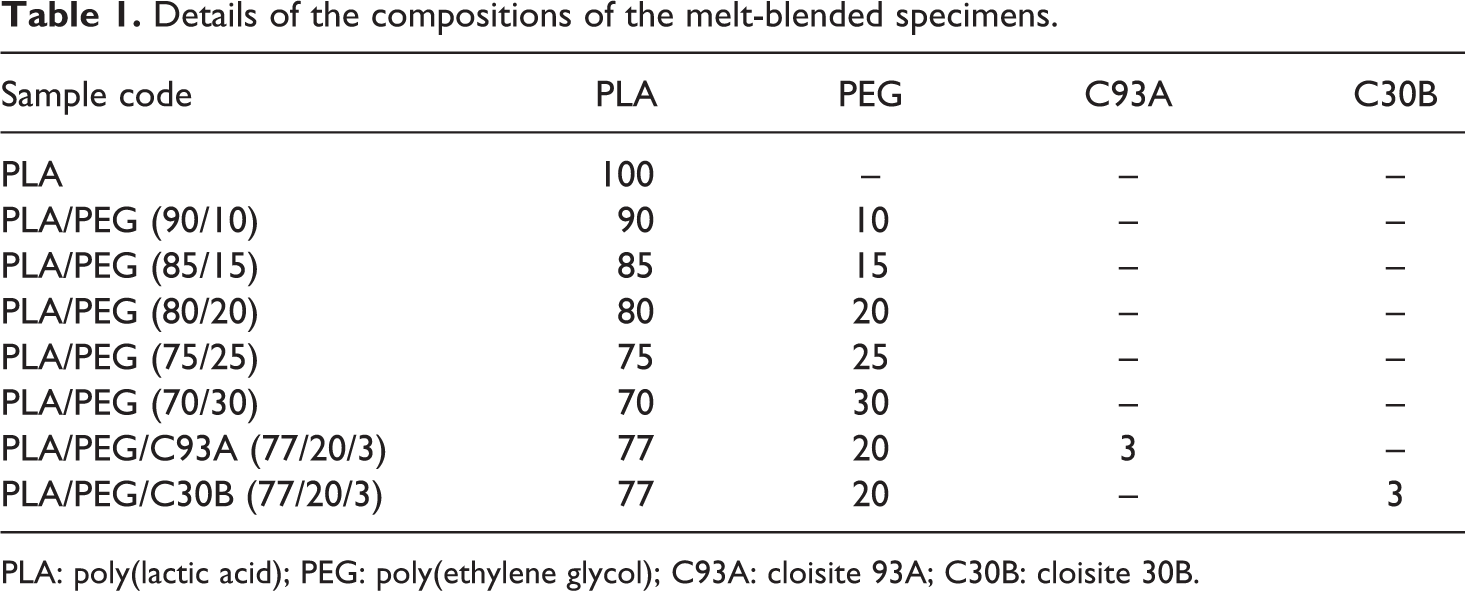
The degree of crystallinity (Xc) can be determined using the following equation:
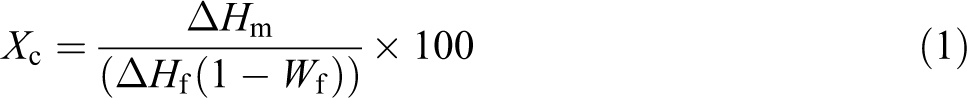
Fractured surface characterization
The scanning electron microscopy (SEM) analysis of impact-fractured blend specimen was carried out using JEOL-JSM T330A (Japan).The samples were sputter coated with platinum and were dried for 0.5 h at 70°C in vacuum, prior to analysis.
Nanomorphology characterization
Wide-angle x-ray diffraction (WAXD) analysis was carried out using XRD-7000 (M/s Shimadzu, Tokyo, Japan), which has a monochromatic and a copper radiation source of wave length 1.54 Å operating at 40 kV and 30 mA. The basal spacing or d-spacing (d001) reflection of the PLA/PEG blend nanocomposites was calculated using Bragg’s law equation (i.e. nλ = 2d sin θ) by monitoring the diffraction angle 2
Dynamic mechanical characterization
The dynamic mechanical analysis was performed with a dynamic mechanical analyzer (DMA Q 800, M/s TA Instruments, USA). The experiments were carried out at a fixed frequency of 1 Hz and at a heating rate of 3°C min−1. The tests were conducted in three-point bending mode with a temperature range of 30 to 120°C using a specimen dimension of 40 × 10 × 3 mm3.
Rheological characterization
Rheological measurements of molten composites were performed in a parallel plate rheometer (Mars III, Thermo Fisher Scientific, Germany) with parallel plate geometry of 25 mm diameter. For each test, the PLA and nanocomposites were placed at the center of the rheometer plate for 5 min for complete melting and temperature equilibrium before the actual measurements. The measurement was carried out within linear viscoelastic range by applying small amplitude oscillation shear after temperature equilibrium to 190°C over a frequency range between 0.01 and 10 Hz that enables the material to retain the structure. Rheological parameters such as elastic or storage modulus (G′), viscous or loss modulus (G″), and complex viscosity (η*) were mainly derived from the analysis at a strain rate of 10%.
Compostability
The test method determines the degree and rate of biodegradation of plastic materials on exposure to controlled composting environment under controlled laboratory conditions. The samples were exposed to inoculums that are derived from compost from municipal solid waste. The aerobic composting takes place in an environment where temperature, aeration, and humidity are closely monitored and controlled. The percentage of biodegradability is obtained by determining the percentage of carbon in the test sample that is converted into carbon dioxide (CO2) during the duration of the test. The test method determines the degree of aerobic biodegradation by measuring evolved CO2 as function of time that the plastic is exposed to soil. Soil is an extreme species—rich source of inoculums for evaluation of biodegradability of plastics in the environment, when maintained appropriately with regard to moisture content and oxygen availability.
Extruded samples as well as the PLA pellets compression molded to films with thickness of around 0.3 mm 100 T compression molding machine (M/s Delta Malikson, Mumbai, Maharashtra, India) at 190°C and 80 kg cm−2 pressure. Furthermore, the prepared films were cut in to 2 × 2 cm2 pieces and conditioned at room temperature for 24 h.
Compostability under controlled compost conditions has been conducted in the laboratory using glass desiccators of 2 L capacity according to the standard test method ASTM D 5988-03 by vermicompost. Filter paper (cellulose) has been used as a positive control and high-density polyethylene (HDPE) film as negative control.
A mixture of vermicompost (250 g by dry weight) and sample (10 g by dry weight) were introduced into the glass desiccators and incubated at room temperature. In this study, in total, six reactors have been utilized for testing, one blank, two controls (positive and negative), and three sample vessels containing PLA, PLA/PEG, and PLA/PEG/C30B nanocomposite. CO2 evolved as a result of biodegradation was trapped in each vessel by means of 50 ml of 0.05 N potassium hydroxide (KOH) solutions taken in a 100 ml beaker, kept in the test vessels. The absorbing solutions were back titrated with 0.1 N hydrochloric acid (HCl) within regular 5 days interval up to 90 days. Also, 50 ml distilled water was kept as reference solution in each reactor.
The percentage of degradation has been calculated by the CO2 trapping method using 0.5 N KOH solutions which was kept inside the reacting chamber. CO2 has been liberated as end product of biodegradation process. The liberated CO2 has been estimated as per the mechanism given below:
The carbon content from the polymer backbone converts to CO2 as a result of bioassimilation process. The liberated CO2 reacts with KOH solution to form potassium carbonate (K2CO3) and remaining KOH solution titrates blank using 0.05 N HCl solutions. Using the titrate value, corresponding liberated CO2 and extent of degradation in percentage also estimated as mentioned below.
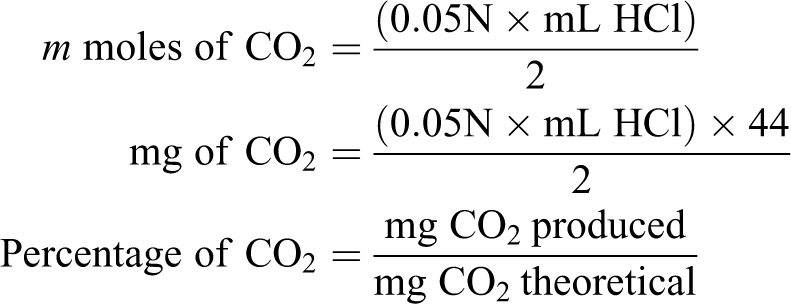
Results and discussion
Tensile and impact characterization of specimens
Figure 1(a) to (d) represents the tensile and impact properties of PLA, PLA/PEG, and PLA/PEG/clay nanocomposites as a function of PEG loading and clay type. From Figure 1(a) and (b), it was observed that due to the plasticizing effect of PEG the tensile strength and modulus decreased with the increase in PEG content to the tune of 10 to 30 wt% into the PLA matrix. However, addition of PEG as a plasticizer to PLA had successfully increased the elongation at break as represented in Figure 1(c). Similar observation was also reported by Hu et al. 30 with the preparation of plasticized PLA using PEG 8000 g mol−1. The impact strength of PLA significantly increased from 25.7 J m−1 in the virgin matrix to 831.88 J m−1 in the case of PLA/PEG at 80:20 blend ratio as shown in Figure 1(d). It is clearly seen from the figure that there was a decrease in impact strength beyond 20 wt% of PEG content, possibly attributed to the lack of cohesion between the matrix polymer and PEG. Hence, the optimum impact strength observed at 20 wt% loading of PEG with the virgin PLA matrix. Sungsanit et al. 31 also observed similar behavior of PLA/PEG blend with variable concentration of PEG at the molecular weight of 1000 g mol−1. As shown in Figure 1(a) and (b), increased modulus and strength was observed for nanocomposite at 3 wt% of clay loading. The tensile modulus increased from 415 to 1208 and 841 MPa, and the tensile strength increased from 23.24 to 27.70 and 23.60 MPa in case of PLA/PEG/C30B and PLA/PEG/C93A nanocomposites, respectively. From the figures it was observed that PLA/PEG/C30B showed better tensile and impact properties as compared to PLA/PEG/C93A which proved strong interaction between –OH group in the surfactant of C30B nanoclay with the carbonyl groups of PLA and –OH groups in PEG at the interface. Again, the swelling agents of organoclays affect the tensile properties of nanocomposites.
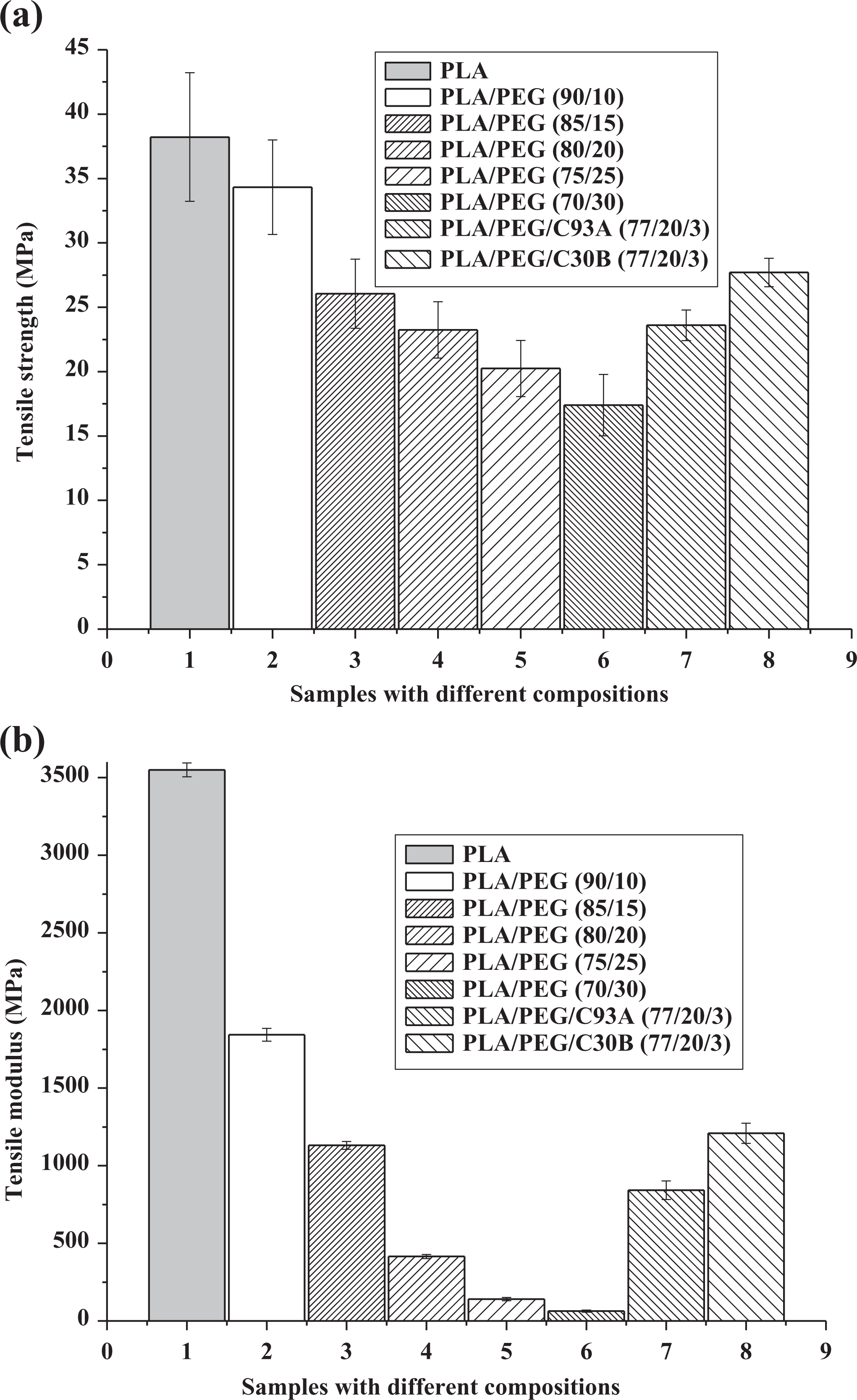
(a) Tensile strength of PLA, PLA/PEG, and PLA/PEG/organoclay; (b) tensile modulus of PLA, PLA/PEG, and PLA/PEG/organoclay; (c) percentage elongation of PLA, PLA/PEG, and PLA/PEG/organoclay; and (d) impact strength of PLA, PLA/PEG, and PLA/PEG/organoclay.
Differential calorimetry analysis
From DSC thermograms, the values of Tg, Tm, and Tc of PLA and PEG were at 57.38, 152.55, and 105.06°C and– 29.3, 62.25 and 35.27, respectively. For better clarity, the DSC thermograms of PEG are represented in values. The depression in the value of Tg, Tm, and Tc of PLA matrix were observed for PLA/PEG blend at 80:20 ratio as evident from Figure 2(a) to (c), which is primarily due to the plasticizing effect of PEG. With the incorporation of nanoclays into PLA/PEG, the Tg value was slightly recovered which may be attributed to the segmental immobilization of the matrix chains due to the presence of nanoscaled-layered silicates as given in Table 2. However, decreased Tc value in case of PLA/PEG/organoclay indicates that the organoclay acts as a nucleating agent for PLA crystallization. 32 Similar observations have also been reported by Ozkoc and Kemaloglu 33 for Tg and Tc in their study of PLA/ PEG and PLA/PEG/C30B. Furthermore, the depression in melting point in case of PLA/PEG is an indication of a miscible system. 21 But, no significant change was observed in the value of Tm with the addition of nanoclays into the PLA/PEG. As represented in table, the value of Xc increased as the value of ΔHm of PLA/PEG and PLA/PEG/organoclay increased as compared to PLA. Sungsanit et al. 31 also observed increased crystallinity in the preparation of PLA/PEG blend. Hence, the ductility characteristics of PLA increased due to the plasticizing effect of PEG which in turn increasing the mobility of PLA molecule. 34
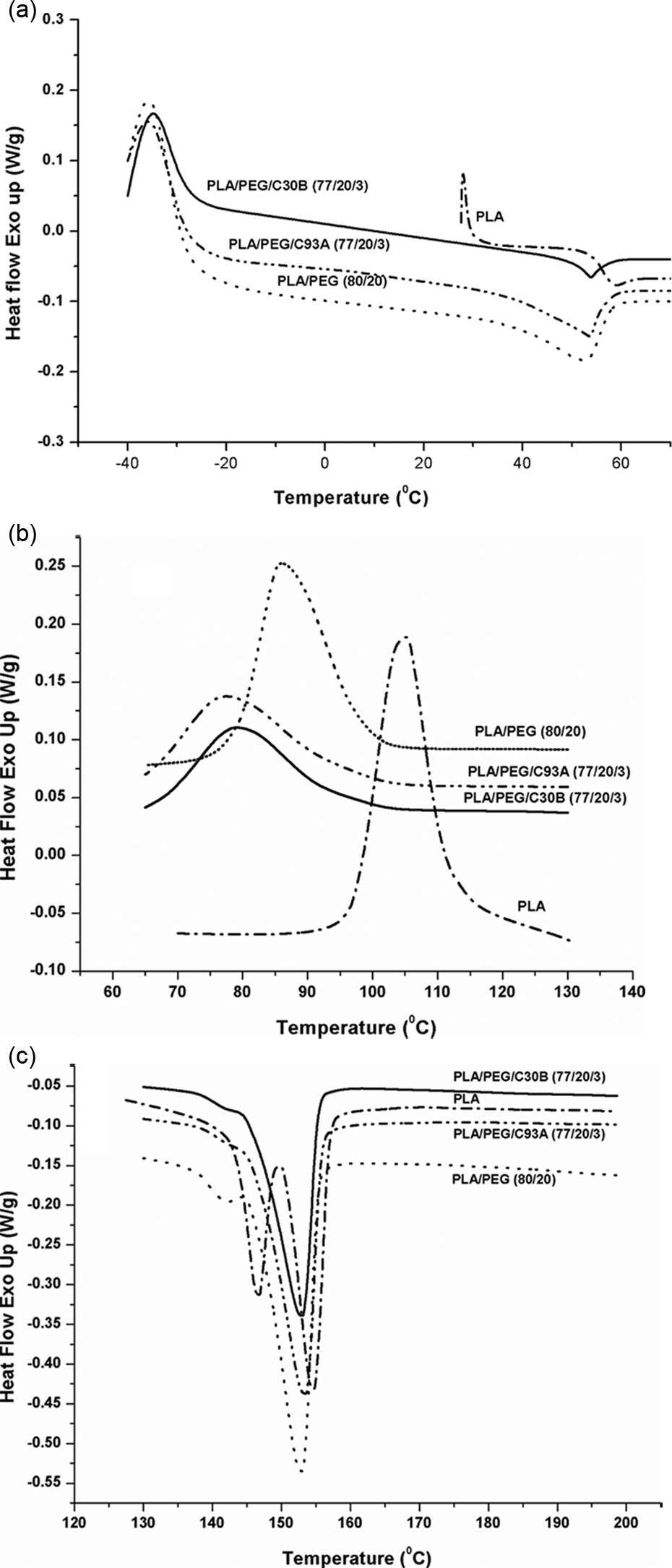
(a) Tg thermogram of PLA, PLA/PEG, and PLA/PEG/organoclay; (b) Tc thermogram of PLA, PLA/PEG, and PLA/PEG/organoclay; and (c) Tm thermogram of PLA, PLA/PEG, and PLA/PEG/organoclay.
Thermogram data of PLA, PLA/PEG, and PLA/PEG/organoclay.
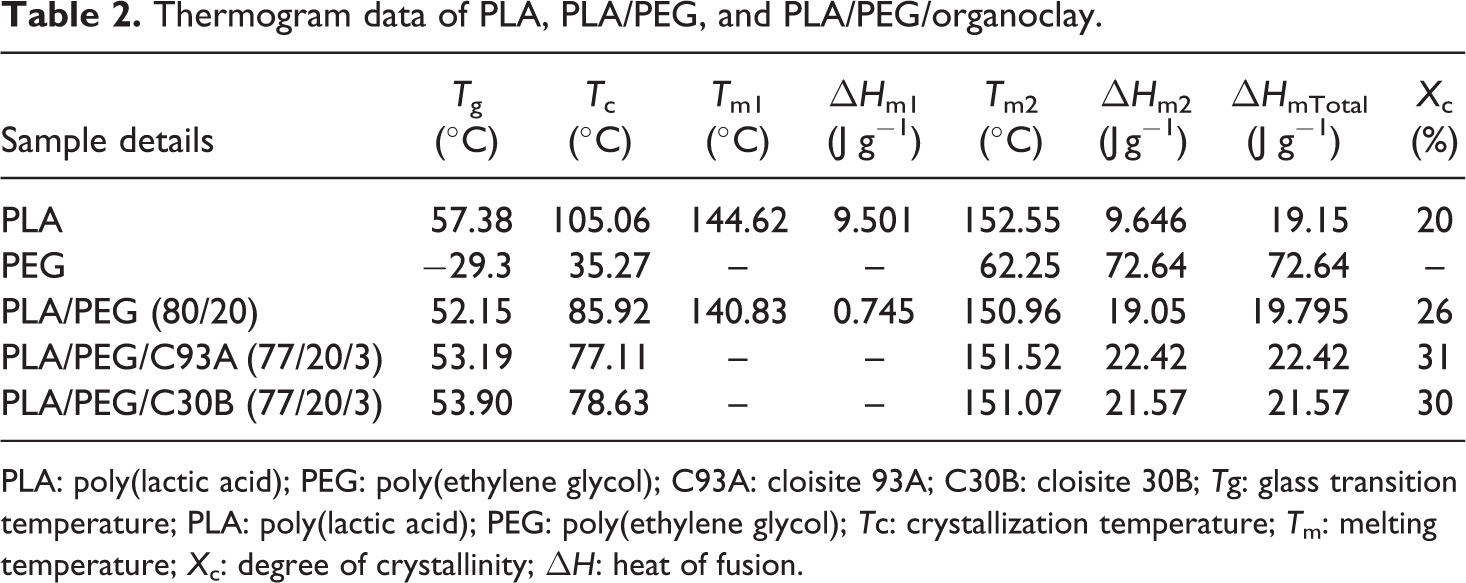
PLA: poly(lactic acid); PEG: poly(ethylene glycol); C93A: cloisite 93A; C30B: cloisite 30B; Tg: glass transition temperature; PLA: poly(lactic acid); PEG: poly(ethylene glycol); Tc: crystallization temperature; Tm: melting temperature; Xc: degree of crystallinity; ΔH: heat of fusion.
Isothermal crystallization kinetics
Isothermal crystallization kinetics has been studied by evaluating the degree of crystalline conversion, as a function of time at a constant temperature for virgin PLA, PLA/PEG, and PLA/PEG/C30B nanocomposite. The relative crystallinity at different crystallization time (Xt) can be calculated according to the equation:
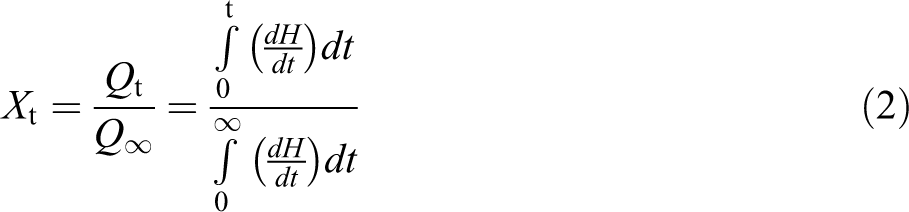
The theory of Avrami is used to analyze the increase of relative crystallinity with time using the crystallization rate constant (K) and Avrami exponent (n) whose value depends on the mechanism of nucleation and on the form of crystal growth. The values of n and K can be obtained, respectively, from the slope and intercept plot of 
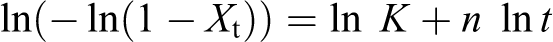
The rate of crystallization of PLA decreases with the increase in temperature; which subsequently increase in the presence of nanoclays. The nanoclays act as nucleating agents within PLA matrix that contributes in improving crystallinity of the later. However, in the case of the PLA/PEG as well as PLA/PEG/C30B, the kinetics of crystallization involves evaluation of two different conditions as reported by Laia et al., 21 the first is the crystallization of PLA prior to the formation of PEG crystals and the second is crystallization of PEG after the formation of PLA crystals.
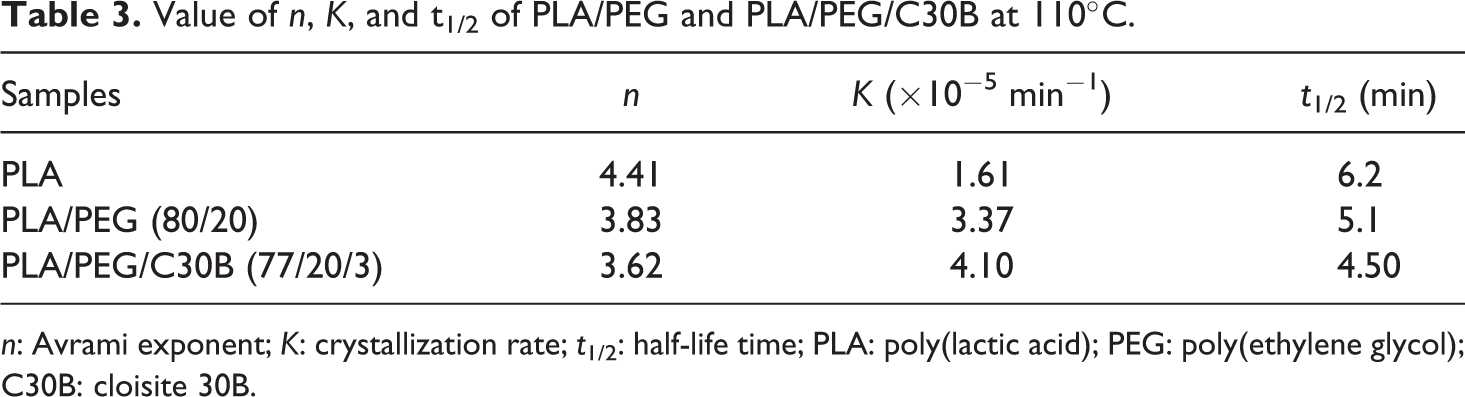
The kinetics of isothermal crystallization has been analyzed using Avrami’s equation in double logarithm form. The corresponding values of kinetic parameters are depicted in Tables 3 and 4, respectively. In both cases of the PLA/PEG and PLA/PEG/C30B, the analysis has been carried out at 110°C, wherein only PLA matrix can crystallize. Comparing the half-life time (t1/2) value of the virgin polymer with the blend, it is proven that incorporation of PEG reduces the value of t1/2 with increasing the K value. As reported by Laia et al., 21 in case of two miscible systems, the value of K, n, and t1/2 in the Avrami equation are different. Similar facts are substantiated in our experimental findings also. The K and t1/2 values change continuously with incorporation of PEG as well as C30B nanoclay within the PLA matrix.
Value of n, K, and t1/2 of PLA/PEG and PLA/PEG/C30B at 110°C.
n: Avrami exponent; K: crystallization rate; t1/2: half-life time; PLA: poly(lactic acid); PEG: poly(ethylene glycol); C30B: cloisite 30B.
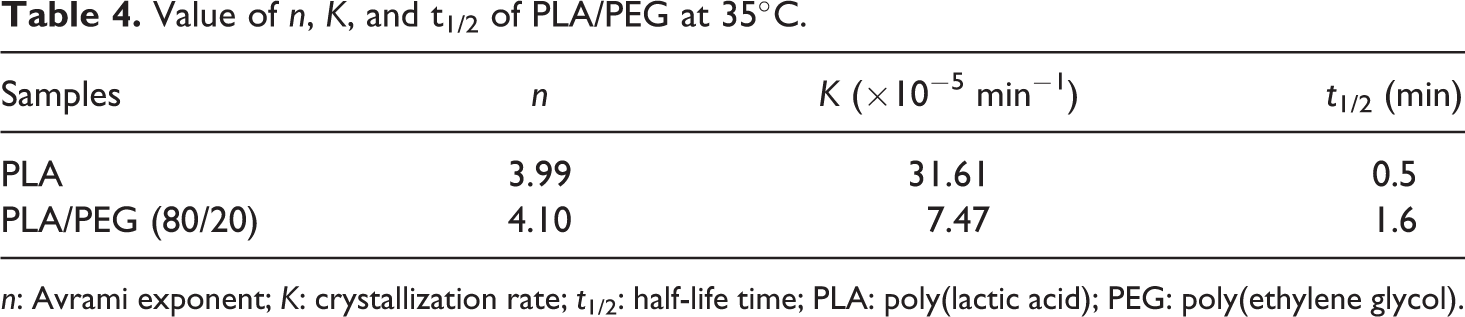
Value of n, K, and t1/2 of PLA/PEG at 35°C.
n: Avrami exponent; K: crystallization rate; t1/2: half-life time; PLA: poly(lactic acid); PEG: poly(ethylene glycol).
Further, it is also observed that isothermal crystallization rate of PLA increases from 1.61 × 10−5 to 3.37 × 10−5 min−1 in the case of PLA/PEG which additionally increases to 4.10 × 10−5 min−1 in the PLA/PEG/C30B. This confirms the nucleating effect of the nanoclay.
The isothermal crystallization rate of PEG after PLA crystals are formed is also depicted in Table 4. For better clarity, the values corresponding to virgin PEG is also represented in Table 2. The samples were initially heated to 180°C, held for 3 min, and then cooled to 110°C until PLA crystals are formed completely and subsequently quenched to 35°C to evaluate the Tc of PEG. It is observed that both t1/2 as well as the crystallization rate decreases in the blend, which is possibly because addition of PLA results in segmental immobilization of PEG, which thereby increases the Tg. Also, the formation of PLA crystal hinders the crystallization rate of PEG.
Fractured surface characterization
As observed from the SEM micrographs of Figure 3(a), the impact-fractured surface of PLA is extremely flat and smooth indicating brittle fracture of PLA under impact loading.

SEM micrographs of (a) PLA and (b) PLA/PEG (80/20).
The white round particles as represented in Figure 3(b) of SEM micrograph indicate the PEG phases distributed within the PLA matrix. Due to the presence of PEG into PLA, the impact-fractured surfaces show more evidences of ductile fractures and longer fibrils which attributes proper dispersion of PEG within PLA matrix. This fibril formation appeared to be related to the increase of the temperature in the crack tip region above the Tg due to heat generation during shock loading. 35 The presence of thread-like structure observed in the PLA/PEG reveals the plasticizing effect that has already been corroborated with the increased percentage of elongation at break.
Nanomorphology characterization
In WAXD analysis, the d001 spacing was calculated from the peak positions using Bragg’s law. Figure 4 represents the WAXD patterns of the PLA/PEG/organoclay. The primary silicate reflection at 2θ = 4.8° (d-spacing ≈ 1.76 nm) and 3.8° (d-spacing ≈ 2.32 nm) were recorded for the C30B and C93A nanoclay from the data sheet. From Figure 4, it has been demonstrated that the diffraction peaks of PLA/PEG/C30B and PLA/PEG/C93A have been shifted to 2θ = 2.65° (d001-spacing ≈ 3.33 nm) and 2θ = 2.84° (d001-spacing ≈ 3.11 nm), respectively. The increase in interlaminar spacing indicates that most of the organoclays are intercalated or partially exfoliated by the help of shearing action during processing and diffusion of polymer chains through the clay galleries. The d-spacing is larger in PLA/PEG/C30B which indicates effective interaction of polar C30B nanoclay within the PLA/PEG matrix at the interface.
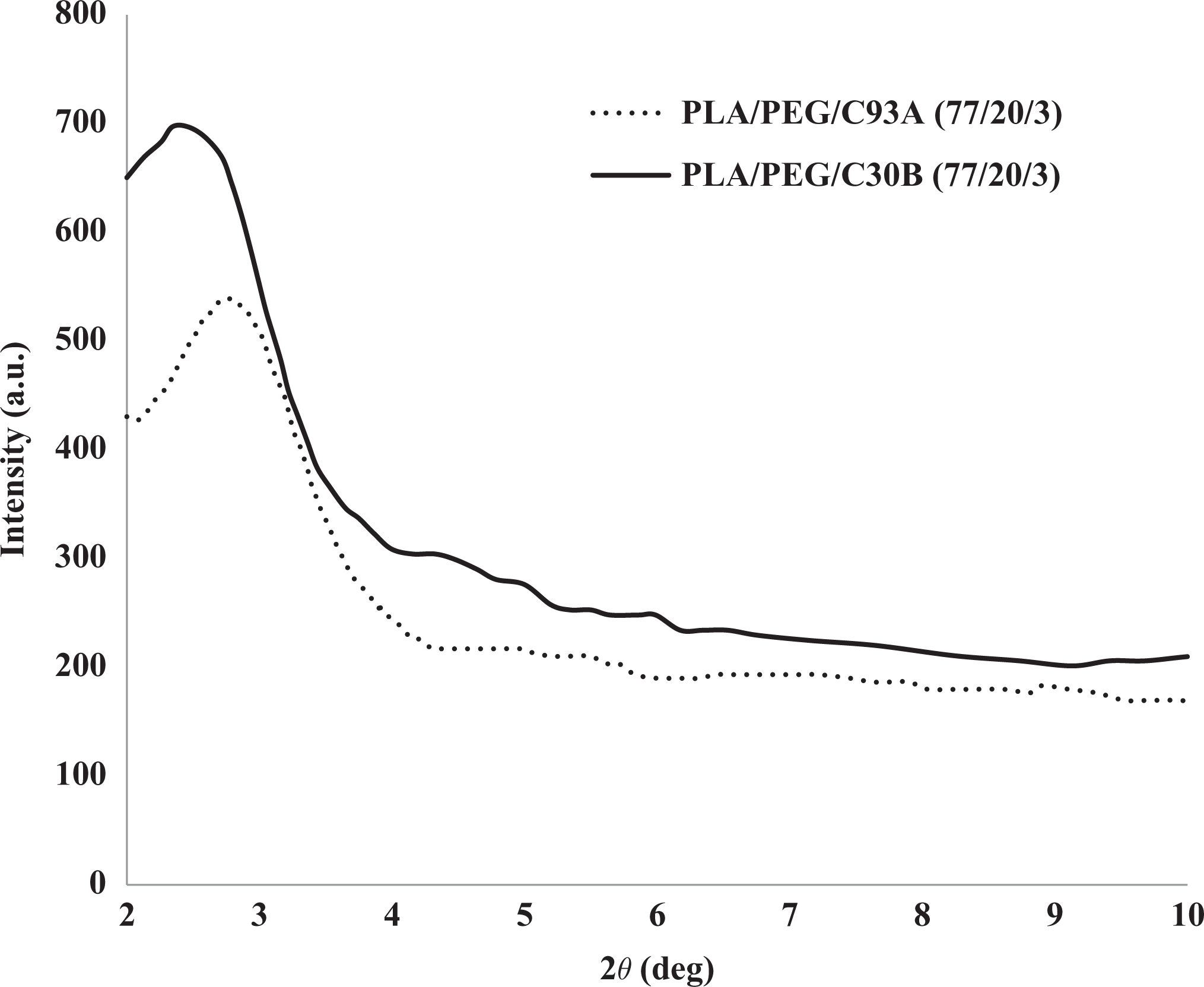
WAXD of PLA/PEG/organoclay.
Dynamic mechanical characterization
Figures 5 and 6 show the dynamic storage modulus (E′) and tan δ of PLA, PLA/PEG, and PLA/PEG/organoclay over a temperature range of 30–120°C. In the case of PLA/PEG, decreased E′ was observed, thus indicating the reduction in the elastic properties of PLA matrix. Below Tg, the entire samples lose its stiffness. As shown in figure, within the investigated range of temperature the PLA/PEG shows a single Tg at 54.30°C, which is predominantly the Tg of the PLA phase and referred to the miscibility of the both polymers. The magnitude of E′ enhanced in case of PLA/PEG/organoclay due to the stress transfer from the nanofiller to the blend matrix below Tg.
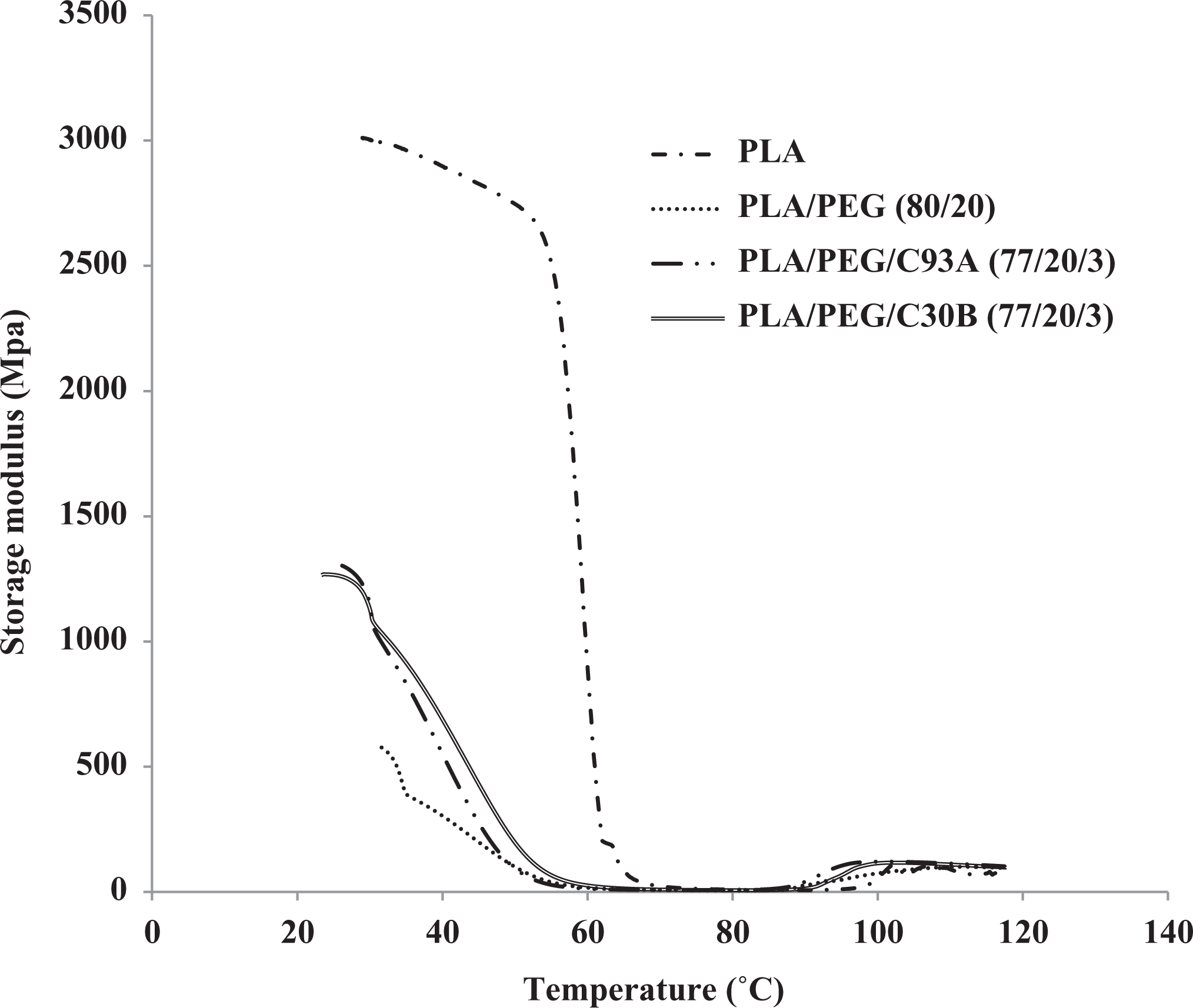
Storage modulus of PLA, PLA/PEG, and PLA/PEG/organoclay.
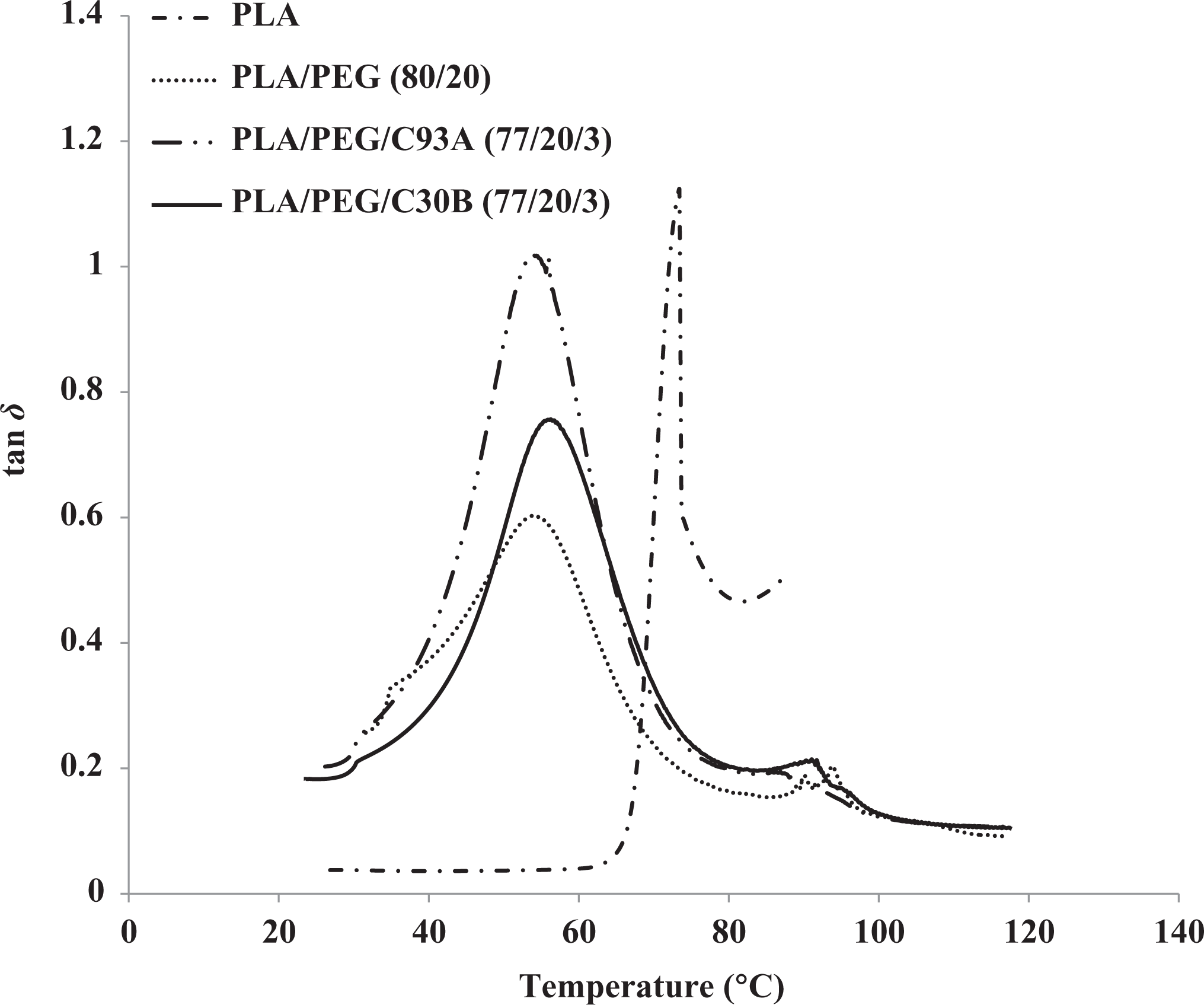
Tan δ of PLA, PLA/PEG, and PLA/PEG/organoclay.
The Tg in the PLA/PEG, PLA/PEG/C30B, and PLA/PEG/C93A nanocomposite reduces to 54.30, 56.19, and 54.30°C as shown to the Tg of PLA as seen from the tan δ peak, thus confirming the plasticizing effect of PEG. Further the Tgs of PLA/PEG and PLA/PEG/C93A are similar, whereas the Tg of PLA/PEG/C30B showed little increased value of about 3%. These indicated that PEG has dominant effect on Tg of whole system, as revealed in DSC results. This agrees well with the mechanical test results discussed in the earlier sections. This further reveals improved compatibility of C30B organoclay with the PLA/PEG matrix at the interface.
Rheological characterization
The melt rheological properties of PLA, PLA/PEG, and PLA/PEG/C30B are depicted in Figure 7(a) to (d). For better understanding, the rheogram of virgin PLA was also presented.
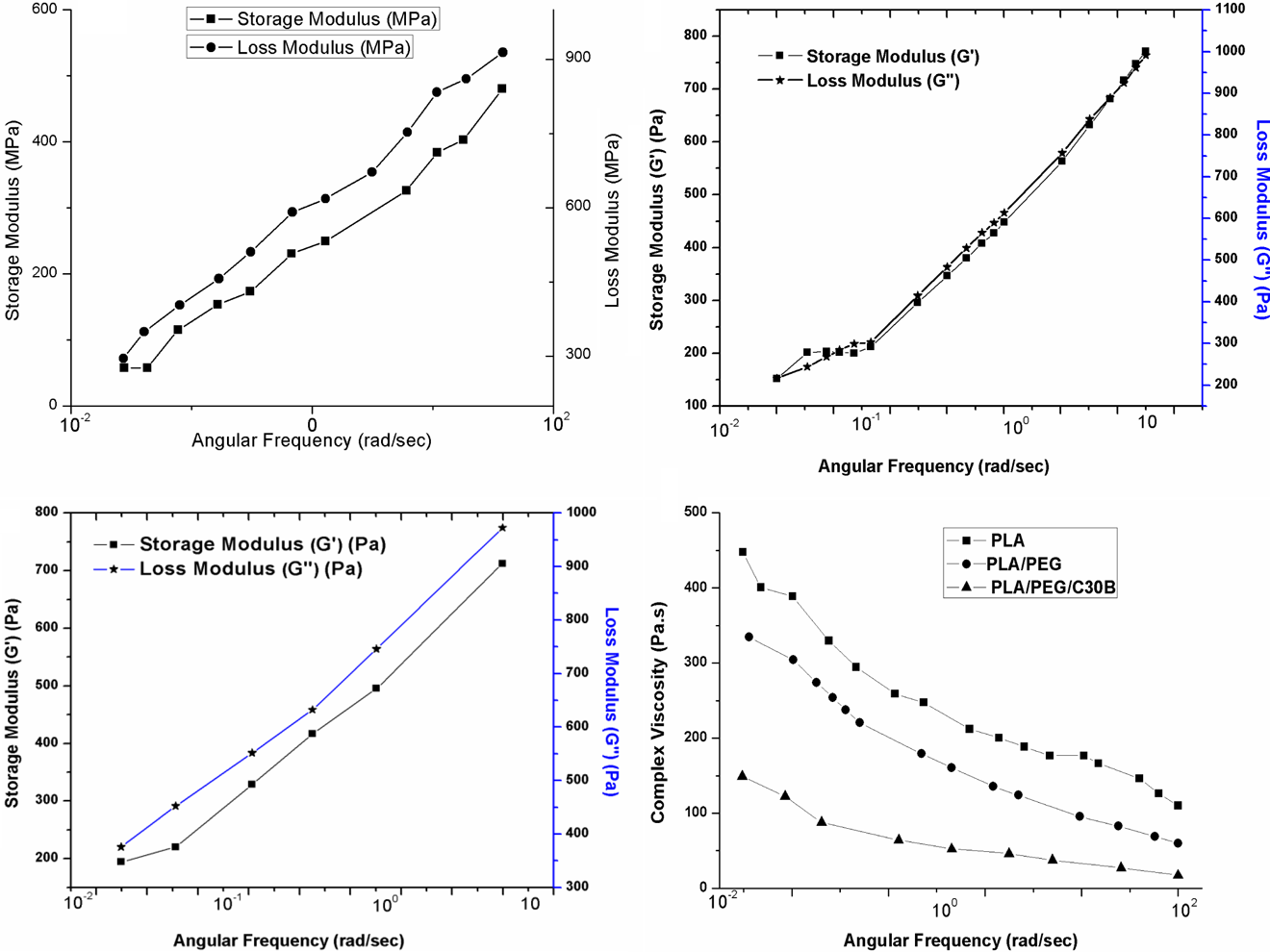
Variation of G′, G″ of (a) PLA, (b) PLA/PEG (80/20), (c) PLA/PEG/C30B (77/20/3), and (d) η* of PLA, PLA/PEG (80/20), and PLA/PEG/C30B (77/20/3) as a function of ω.
The degradation in PLA during processing in the presence of plasticizers with ester groups could also be due to a decrease of PLA molecular weights 36 which resulted in a decrease in PLA viscosity. The PLA/PEG showed a decreased value of G′ and G″ as compared to the matrix polymer due to the plasticizing effect of PEG. The PLA/PEG exhibited the rheological behavior of a typical polymer melt as characterized by a smaller G′ value than G″ value. Thus, PLA/PEG revealed liquid-like (G″ > G′) characteristics. However, the incorporation of the organoclays within the PLA/PEG resulted increased G′ and G″. In the case of PLA/PEG/C30B, the G″ also exhibited higher magnitude over G′ in the entire investigated frequency range thus confirming more liquid-like properties. The viscoelastic behavior of the PLA/PEG and PLA/PEG/C30B was calculated from the slopes of the linear regression of the power-type relationship of G′(ω) α ω 2 and G″(ω) α ω, which is commonly used to predict the solid-like characteristics in the polymers. The slopes in terminal zone of the PLA/PEG and PLA/PEG/C30B derived from the master curves for G′(ω) and G″(ω) at frequency levels are 0.96 and 0.88 and 0.92 and 0.84, respectively. The lower slope values and higher absolute values of dynamic moduli of PLA/PEG/C30B indicate the formation of entanglement structures in PLA/PEG melts. The blend represents a complex system wherein both plasticizing and antiplasticizing effects were attributed by the PEG and organoclays, respectively, which in turn is reflected in the rheological behavior. 37 The viscoelastic behavior of the system is assumed to be contributed by PEG and organoclays that individually has plasticizing and mechanical strengthening characteristics, respectively.
Variation in η* value as a function of angular frequency (ω) of PLA, PLA/PEG, and PLA/PEG/C30B is depicted in Figure 7(d). It is proven that the PLA/PEG depict non-Newtonian pseudoplastic characteristics with a decrease in η* value with increase in frequency. The η* value of PLA/PEG decreased within the PLA/PEG/C30B, thus revealing shear-thinning effect in the presence of organoclay.
Furthermore, it can also be seen from the figure that the PLA/PEG and PLA/PEG/C30B exhibited a liquid-like behavior due to plasticization effect of PEG as supported by other characterizations. The PLA/PEG/C30B exhibited slightly improved solid-like behavior with the addition of organoclays thus revealing an intermediate behavior. However, the liquid-like property is still affected predominantly. Lin et al. 38 also observed the similar trend for PLA/ Poly (ester amide) blend.
Compostability
Figure 8 represents the degradation pattern for filter paper (cellulose) and HDPE film as positive and negative controls, respectively. The cellulose film has been degraded completely (100% weight loss) within a time period of 30 days. As the initiation process in the early days leads to a slow rate of degradation up to 5 days and further the autocatalyzed propagation step end up with complete degradation of film by weight loss within 30 days of composting. On the other hand, the negative control of HDPE film did not show any considerable variation in weight loss in the composting medium during the 100 days time period of composting. This has been proved that the vermicompost used in the present investigation is an active system for polymer degradation.
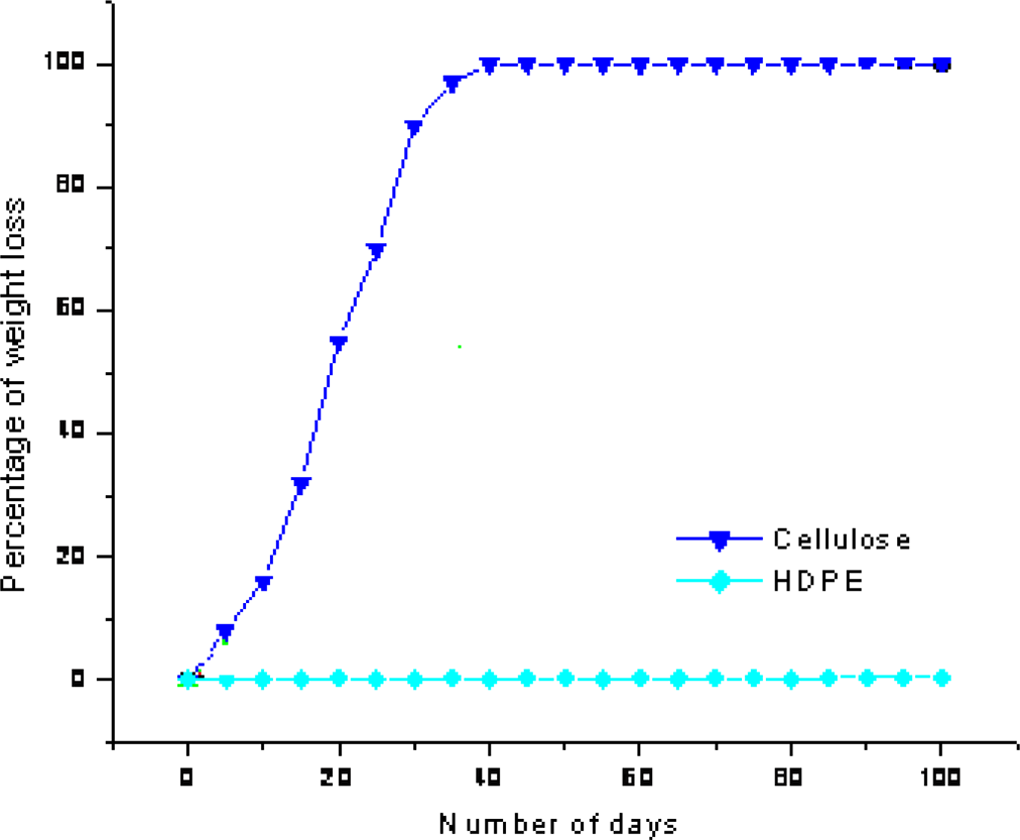
Percentage of degradation by weight loss in compost media of positive (cellulose) and negative (HDPE) control.
The observed percentage of degradation or weight loss of all system with time is depicted in Figure 9. The degradation behavior of polymer could be affected by molecular weight, chain configuration, hydrophilicity, and crystallinity. 39 The main effective factors in biodegradation of polymers are the molecular weight and the Xc. 40 The low-molecular-weight chains and amorphous phase of PLA exhibits higher biodegradation rate. PLA matrix has shown a slow rate of degradation in the beginning days where the initial deterioration was happening. After around 30 days, the rate of degradation increased rapidly where the enzymatic mechanisms have always shown the nature of autocatalytic reaction. The stable plateau of equilibrium has been approached after 90 days of composting where the PLA has been shown 100% degradation by weight loss. From the figure, it is observed that the order of increase in the rate of biodegradation is PLA > PLA/PEG > PLA/PEG/C30B. The addition of plasticizer and organoclay with the PLA attributed increased crystallinity for both cases of PLA/PEG and PLA/PEG/C30B films as compared to the matrix polymer. Again further higher value of Xc of PLA/PEG/C30B which may be due to the inclusion of organoclay into the PLA/PEG results in lower rate of biodegradation.
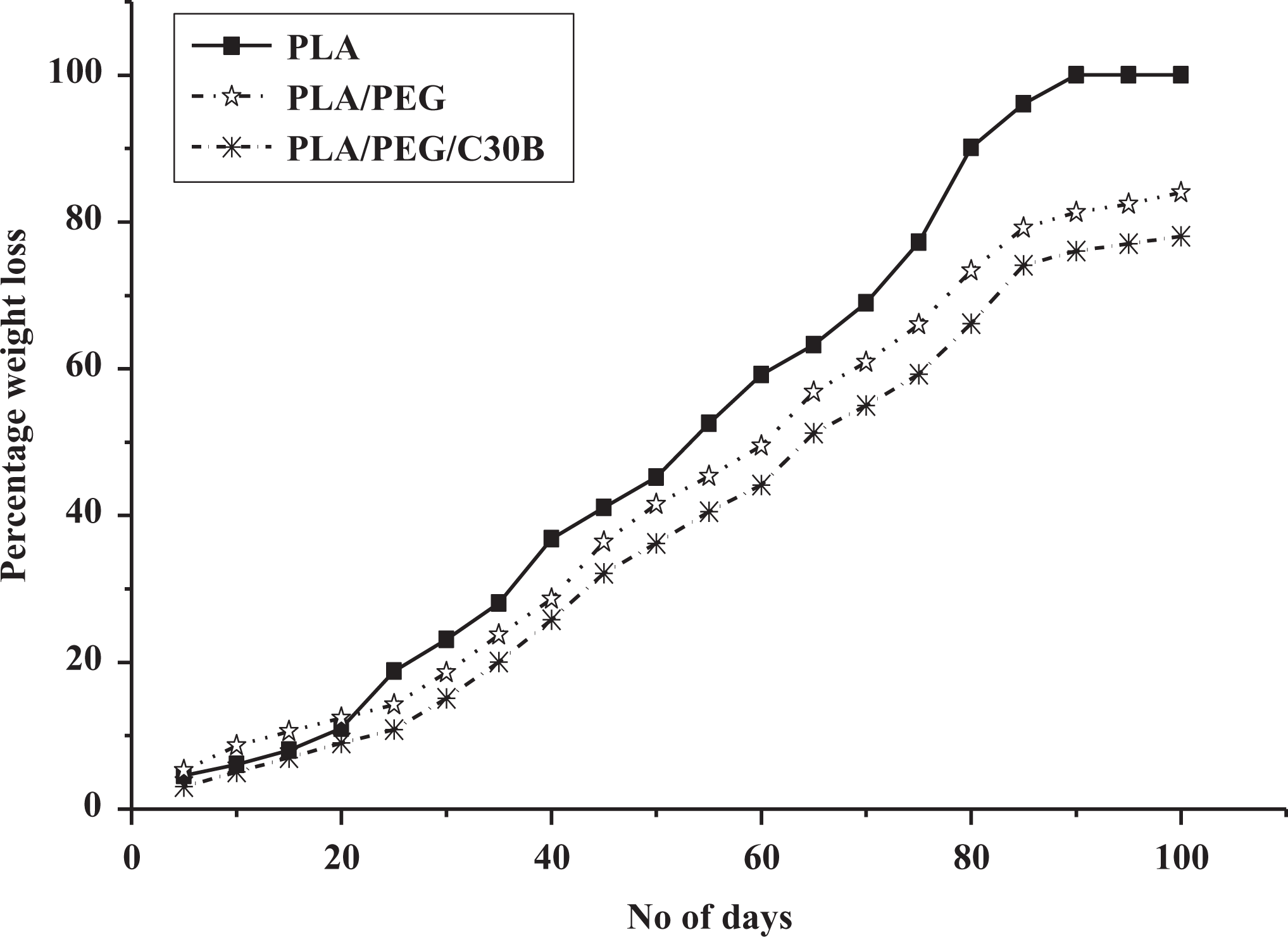
Percentage of degradation by weight loss in compost media of PLA, PLA/PEG (80/20), and PLA/PEG/C30B (77/20/3).
Conclusion
Incorporation of PEG within PLA matrix improves the ductility and impact properties of the blend, while reducing its tensile strength and tensile modulus. Addition of organoclays into PLA/PEG enhanced the tensile strength and modulus. PLA/PEG/C30B blend nanocomposites showed intercalated and partial exfoliation of nanoclays that has been proven from the WAXD analysis. The DMA analysis resulted lower storage modulus in case of PLA/PEG and subsequently higher in both cases of PLA/PEG/organoclays. However PLA/PEG/C30B showed increased G′ values as compared to PLA/PEG/C93A. The tan δ of the both PLA/PEG and PLA/PEG/organoclay decreased. The values of Tg, Tc, and Tm decreased with the addition of PEG into PLA matrix as proven by calorimetry analysis. The study of isothermal crystallization kinetics by Avrami theory revealed low crystallization rate of PLA matrix due to the plasticizing effect of PEG. Rheological measurements confirmed non-Newtonian psuedoplastic behavior in both cases of PLA/PEG and PLA/PEG/C30B. The compostability study of PLA, PLA/PEG, and PLA/PEG/C30B in the controlled compost provided a clear idea about the biodegradation stability of the systems. Thus, PLA/PEG and PLA/PEG/C30B can be used with optimal concentration of PEG and organoclays for packaging and food grade applications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
