Abstract
Poly(butylene adipate-co-terephthalate) (PBAT)/nano-alumina (NA) composite films were prepared using extrusion and compression moulding processes. The aim of this work is to improve the performance property of PBAT. Concentration of NA varied from 0.1 to 5 wt% in PBAT. The prepared films were characterised for mechanical (tensile strength (TS), tensile modulus (TM) and elongation at break (PEB)), thermal (melting temperature (Tm) and enthalpy of melting (Hm)), rheological (viscosity vs. shear rate), morphological (SEM), barrier (water vapour permeability (WVP) and oxygen permeability(OP)) and crystallinity (x-ray diffraction) properties. PEB and crystallinity decreased, whereas, TS and Tm remained unaffected on addition of NA in PBAT, as the addition of NA decreased the crystallinity of PBAT. Nevertheless, significant reduction in WVP and OP of 59 and 33% were obtained for 3 wt% addition of NA in PBAT. Hm and viscosity of PBAT decreased with increase in concentration of NA, due to the thermally conducting nature of NA. Considering the obtained properties, PBAT containing 3 wt% NA can be a potential material for food and pharmaceutical packaging applications.
Keywords
Introduction
Development of biodegradable polymers obtained from renewable resources has received considerable research interest in the recent years, with a primary aim to reduce the utilization of petroleum-based polymers in various applications. Biodegradable polymers are degraded upon exposure to bioactive environments, by organisms such as bacteria, algae, fungi, etc., or by hydrolysis in buffer solutions or sea water. Poly(lactic acid) (PLA), poly(butylene adipate-co-terephthalate) (PBAT) and other linear aliphatic biodegradable polyester are derived from biomass through bioconversion and polymerization. They have become potential candidates for various large-scale industrial applications in areas like packaging, biomedical, pharmaceutical, etc., 1,2 Growth and development of biodegradable polymer for short-range applications have become important to reduce the impact on the environment. However, high cost and poor performance characteristics have been the major limitations for penetration of these materials into markets for various end user applications. 3
Reinforcing these polymers with nano-fillers would be a cost effective way to improve their properties. Nanoscale fillers can significantly enhance the mechanical, thermal, dimensional and barrier performance in the biodegradable polymers. 4 Biodegradable nanocomposites can be prepared using various methods such as solution intercalation, melt intercalation, in situ intercalative polymerization and template synthesis. 5 However, polymer melt intercalation has proved to be a versatile technique in the fabrication of biodegradable nanocomposites because of its environmental friendly characteristics. 6 Several attempts have already been made in the fabrication of PLA, 7 –10 poly caprolactone (PCL), 11 –16 poly butylene succinate (PBS) 17 –20 based nanocomposites using melt blending technique.
PBAT is aliphatic-aromatic copolyester and the chemical structure of PBAT is shown in Figure 1. It is a fully biodegradable water-resistant polymer. It exhibits excellent compatibility to other biodegradable aliphatic polyesters or starch compounds. PBAT is designed to process like low density polyethylene into films or bags, having shelf life of about 1 year. 21 PBAT, an aromatic aliphatic biodegradable copolyester, is a flexible material with a higher elongation than PLA and PBS, suitable for various packaging and agricultural applications. 22 In order to improve the performance property of PBAT, PBAT based nanocomposites were developed by many researchers. Someya et al. prepared PBAT/octadecylamine modified montmorillonite nanocomposites, showing improved mechanical and crystallinity at 5 wt% loading. 23 Chivrac et al. studied the non-isothermal crystallization behaviour of PBAT and micro- and nano-sized layered silicate-based composites. They showed that layered silicates had negative effect on the crystalline growth process. 24 Mohanty and Nayak investigated the effect of sodium montmorillonite (SMMT) on the structure and properties of PBAT. Mechanical, thermal and degradation rate of PBAT increased with increase in concentration of SMMT. 25,26 Chen et al. prepared PBAT/ montmorillonite (MMT) by solution blending and found very little improvement in mechanical properties, but a substantial improvement in the cooling crystallization temperature. 27 Yang and Qui prepared PBAT/cloisite30A nanocomposites by solution casting process, and determined exfoliated morphology at 8 wt% loading, showing improvement in mechanical and enhanced crystallization. 28 Rao et al. investigated the combined effect of nanoclay and alumina on the properties of polypropylene based nanocomposites. They determined substantial improvement in the storage modulus, thermal stability and load bearing capacity of polypropylene on incorporation of coupling agent in the nanocomposite. 29 Ngu et al. determined improved the thermal stability of low-density of polyethylene by addition of surfactant-treated nano-alumina (NA). 30 Tadayyon et al. investigated the effect of addition of NA and ball milling time on the crystallinity of polyethylene. They determined decrease in crystallinity of polyethylene on addition of NA or increased ball milling time. 31
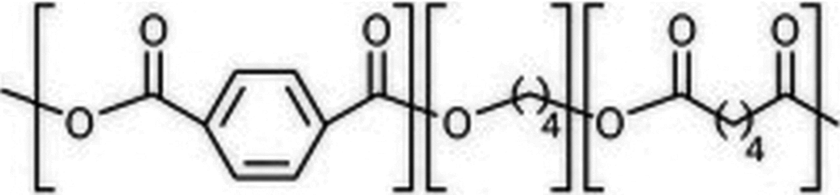
Chemical structure of poly(butylene adipate-co-terephthalate).
In this research work, an attempt is made to improve the performance properties of PBAT by the use of NA. Concentration of NA varied from 0.1 to 5 wt% in PBAT. PBAT/NA composites were prepared by extrusion melt blending. Prepared composites were characterized for mechanical, crystallinity, thermal, rheological, barrier and morphological properties.
Experimental
Materials
PBAT (trade name: ECOFLEX®, F, Blend C1200, melt flow index (MFI): 3.5 g 10 min−1, density: 1.25 g cm−3, biodegradable according to US standard ASTM D6400) was supplied by BASF, Mumbai, India. NA (99.8% purity, specific surface area 70 m2 g−1, α-phase, white colour, spherical shaped, 200–300 nm in diameter) was procured from Reinste Nano Ventures, New Delhi, India. Finalux G3 (wetting agent) was obtained from Fine Organics Pvt. Ltd, Mumbai, India. All chemicals were used as obtained without any purification or modification.
Preparation of PBAT/NA composite films
Dry blending
NA powder and PBAT granules were predried at 60°C in an air-circulating oven for 12 h to remove any traces of absorbed or adsorbed moisture, before processing. NA was premixed in different loadings: 0.1, 0.5, 1, 2, 3, 4 and 5 wt% in PBAT. Dry blending of NA and PBAT was performed in a tumbler mixer for 10–15 min, using finalux G3 as the wetting agent.
Melt blending
The dry blended PBAT/NA compositions were melt blended in a co-rotating twin-screw extruder (Model MP 19 PC, APV Baker, UK) having length/diameter (L/D) ratio of 25:1. Temperature profile from zone 1 to 4 was maintained as: 70°C, 110°C, 140°C, and 160°C respectively, and die temperature at 180°C. Rotation speed of screw was kept constant at 80 r min−1. Vacuum was applied in the vent of the extruder to facilitate easy removal of moisture (if present any). Extruded strands were water cooled (30°C) and pelletized.
Compression moulding
PBAT/NA pellets were again dried in an air-circulating oven at 60°C. Pellets were then transformed into films of 100 ± 10 µm thickness, by compression moulding. Compression moulding was performed in a hot-plate hydraulic press at 180°C and 15 MPa pressure for 4 min. 32 –35 The polymer films were eventually allowed to cool to room temperature under pressure. Prepared composite films were characterized for mechanical, thermal, rheological, barrier, crystallinity and morphological properties. Prepared formulations and their nomenclature are shown in Table 1.
Prepared PBAT/NA compositions with their nomenclature.a
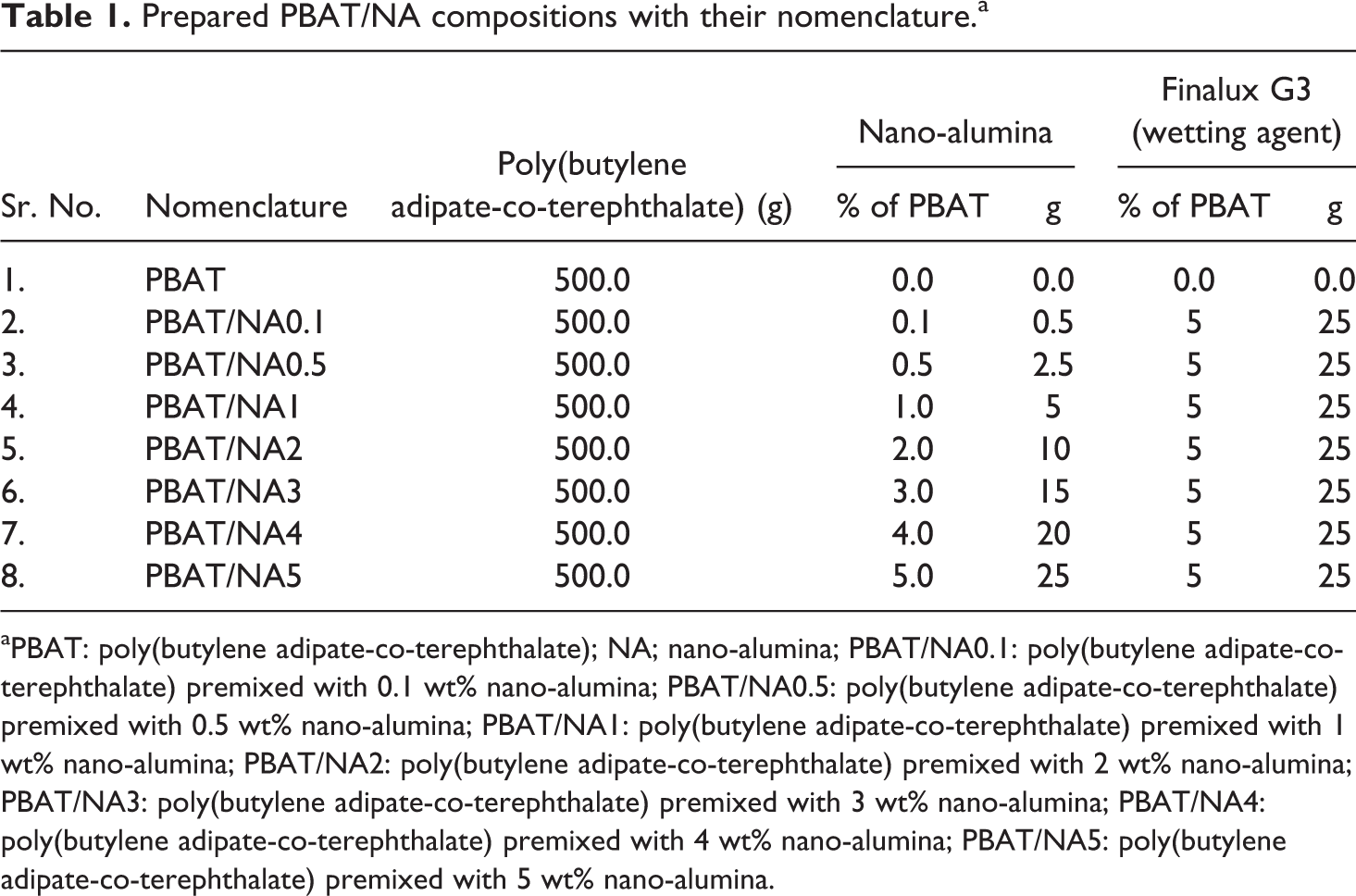
aPBAT: poly(butylene adipate-co-terephthalate); NA; nano-alumina; PBAT/NA0.1: poly(butylene adipate-co-terephthalate) premixed with 0.1 wt% nano-alumina; PBAT/NA0.5: poly(butylene adipate-co-terephthalate) premixed with 0.5 wt% nano-alumina; PBAT/NA1: poly(butylene adipate-co-terephthalate) premixed with 1 wt% nano-alumina; PBAT/NA2: poly(butylene adipate-co-terephthalate) premixed with 2 wt% nano-alumina; PBAT/NA3: poly(butylene adipate-co-terephthalate) premixed with 3 wt% nano-alumina; PBAT/NA4: poly(butylene adipate-co-terephthalate) premixed with 4 wt% nano-alumina; PBAT/NA5: poly(butylene adipate-co-terephthalate) premixed with 5 wt% nano-alumina.
Characterization and testing
X-ray diffractometer
X-ray diffraction (XRD) analysis was undertaken to determine the effect of NA addition on the crystallinity of PBAT. XRD patterns were obtained on a Rigaku Miniflex X-Ray Diffractometer using a copper target, having wavelength of 1.54 Å. The normal focus copper x-ray tube was operated at 30 kV and 15 mA. The PBAT and PBAT/NA composite film samples were placed in a sample holder and the analysis was carried out in a static position, and detector moving through angle 2 to 60°. Sample scanning was done from 10 to 60° at a rate of 3° min−1. The data processing was done using Jade 6.0 software.
Morphological properties
SEM analysis was carried out with the JEOL® 6380 LA (Japan) scanning electron microscope. Samples were fractured under liquid nitrogen to avoid any disturbance to the molecular structure and the specimens were coated with gold (thickness 30 Å) before imaging and observed with an accelerating voltage of 15 kV.
Thermal properties
PBAT and PBAT/NA composite films were characterized by differential scanning calorimeter (DSC), to determine their melting temperature (Tm) and enthalpy of melting (Hm). The test was performed with a Q100 DSC (TA Instruments, Delaware, USA) equipment, fitted with a nitrogen-based cooling system. All the measurements were performed in the temperature range from −50 to 200°C, at a heating rate of 10°C min−1. The reported values were recorded from the second heating scan. Tm was measured from the maximum of the endothermic peak; whereas, Hm was measured from the area of the endothermic peak. The degree of crystallinity (Dc) was estimated according to the following equation: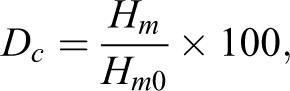
where, Dc is the degree of crystallinity (%), Hm is the enthalpy of melting determined from the DSC heating scan, Hm0 is the theoretical enthalpy of melting for 100% crystalline PBAT Hm0 for PBAT is 114 J g−1. 32
Mechanical properties
In mechanical properties, prepared PBAT and PBAT/NA film samples were analysed for tensile strength (TS) and percentage elongation at break (PEB). TS and PEB of the films was determined with a Universal Testing Machine (LR-50 K, Lloyd instrument, Sussex, UK), using a 500 N load cell and at the crosshead speed of 50 mm min−1. This test was performed in accordance to ASTM standard D 882. An average value of six replicates for each sample was taken.
Barrier properties
Water vapor permeability
Water vapour permeability (WVP, gm.mm cm−2 day−1) of the PBAT and PBAT/NA composite films was determined gravimetrically according to ASTM E96-water method. Each test film was sealed on the top of the permeation cell with melted paraffin, containing distilled water. The permeation cells were placed in a desiccator maintained at 0% RH. 0% RH was maintained using anhydrous calcium chloride. The desiccant absorbs the water transferring through the film, which is determined from the weight of the permeation cell. Each permeation cell was weighed at an interval of 24 h. An average value of three replicates for each sample was taken.
Oxygen permeability (OP)
Oxygen permeability (OP) of the films was determined using a gas permeability tester machine (Labthink BTY-B1). After placing the film in the cell, oxygen was introduced on one side of the film and the OP was measured. Relative humidity was maintained at 50% and temperature at 30°C. OP (cm3.cm cm−2 s−1 cm−1Hg) was calculated by multiplying oxygen transmission rate by the film thickness (µm), and dividing the oxygen gradient in the cell of the testing machine (0.1 Pa). An average value of three replicates for each sample was taken.
Rheological properties
Measurements of viscosity (η) were made at 150°C, using a Rheometer (MCR 101, Anton Paar) employing the Cone and Plate geometry having 35 mm diameter. PBAT and PBAT/NA composites films were measured over a shear rate range of 0.01–100s-1.
Results and discussion
X-ray diffractometer (XRD)
XRD patterns obtained for PBAT and PBAT/NA composite films are illustrated in Figure 2. Characteristic PBAT crystal structure peaks were observed in the 2θ region range from 15 to 30°. To be precise, 5 peaks were at 16.5, 17.2, 20.7, 22.8 and 24.8° respectively. These peculiar peaks of PBAT are also observed in the diffraction patterns of all PBAT/NA composite films, at the same 2θ values. It can be said that, there is no important trans-crystallinity happening at the NA/PBAT interface. Thus, there is no change in the crystal structure of PBAT on addition of NA. Intensity of the five characteristic PBAT peaks decreased with increase in NA concentration. However, the decrease in peak intensity is not appreciable. Thus it can be said that NA is playing no role in affecting the crystallinity of PBAT.
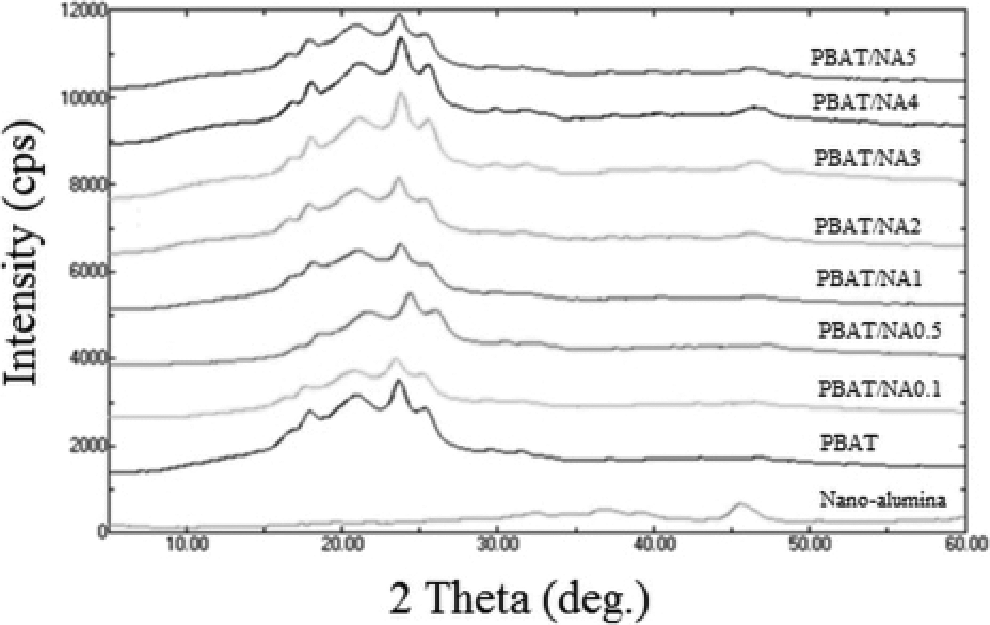
X-ray diffraction patterns obtained for PBAT and PBAT/NA composite films. PBAT: poly(butylene adipate-co-terephthalate), PBAT/NA: poly(butylene adipate-co-terephthalate)/nano-alumina.
NA shows its characteristic peak at 2θ of about 46.2°. This peak is completely absent in PBAT. However, on addition of NA in PBAT, its characteristic peak becomes visible. Intensity of this increased with increase in concentration of NA in PBAT, indicating increased concentration of NA in PBAT.
Morphological properties
SEM obtained for PBAT/NA1, PBAT/NA3 and PBAT/NA4 are shown in Figure 3(a), (b) and (c) respectively. Alumina (Al2O3) is composed of much heavier elements (Al, O) compared to the surrounding PBAT matrix (C, H and O). This makes alumina distinctly visible in the PBAT matrix. Uniform dispersion of NA was seen in PBAT/NA1 and PBAT/NA3 than in PBAT/NA4. Aggregates were observed in PBAT/NA3 as compared to PBAT/NA1, but the number and size of aggregates are far too less than PBAT/NA4. 4 wt% concentration of NA in PBAT becomes too high to maintain uniform dispersion.

SEM images obtained for (a) PBAT/NA1, (b) PBAT/NA3 and (c) PBAT/NA4. PBAT/NA1: poly(butylene adipate-co-terephthalate) premixed with 1 wt% nano-alumina; PBAT/NA3: poly(butylene adipate-co-terephthalate) premixed with 3 wt% nano-alumina; PBAT/NA4: poly(butylene adipate-co-terephthalate) premixed with 4 wt% nano-alumina.
Thermal properties
Tm and Hm determined for PBAT and PBAT/NA composites is summarized in Table 2; whereas, their melting curves are shown in Figure 4. PBAT was found to have Tm of 120.69°C, Hm of 12.1 J g−1 and percentage crystallinity equal to 10.6%. Addition of NA had no effect on the Tm of PBAT. All the prepared PBAT/NA composites had melting temperature of near about 120°C. Thus, addition of NA does not affect the crystal organization of PBAT. However, Hm values are affected by the addition of NA. Hm decreased with increase in concentration of NA in PBAT; however, the decrease is not very significant. This result is in harmony with the XRD analysis, indicating that NA hinders the PBAT crystalline growth.
Thermal properties of PBAT and PBAT/NA composites measured using DSC.a
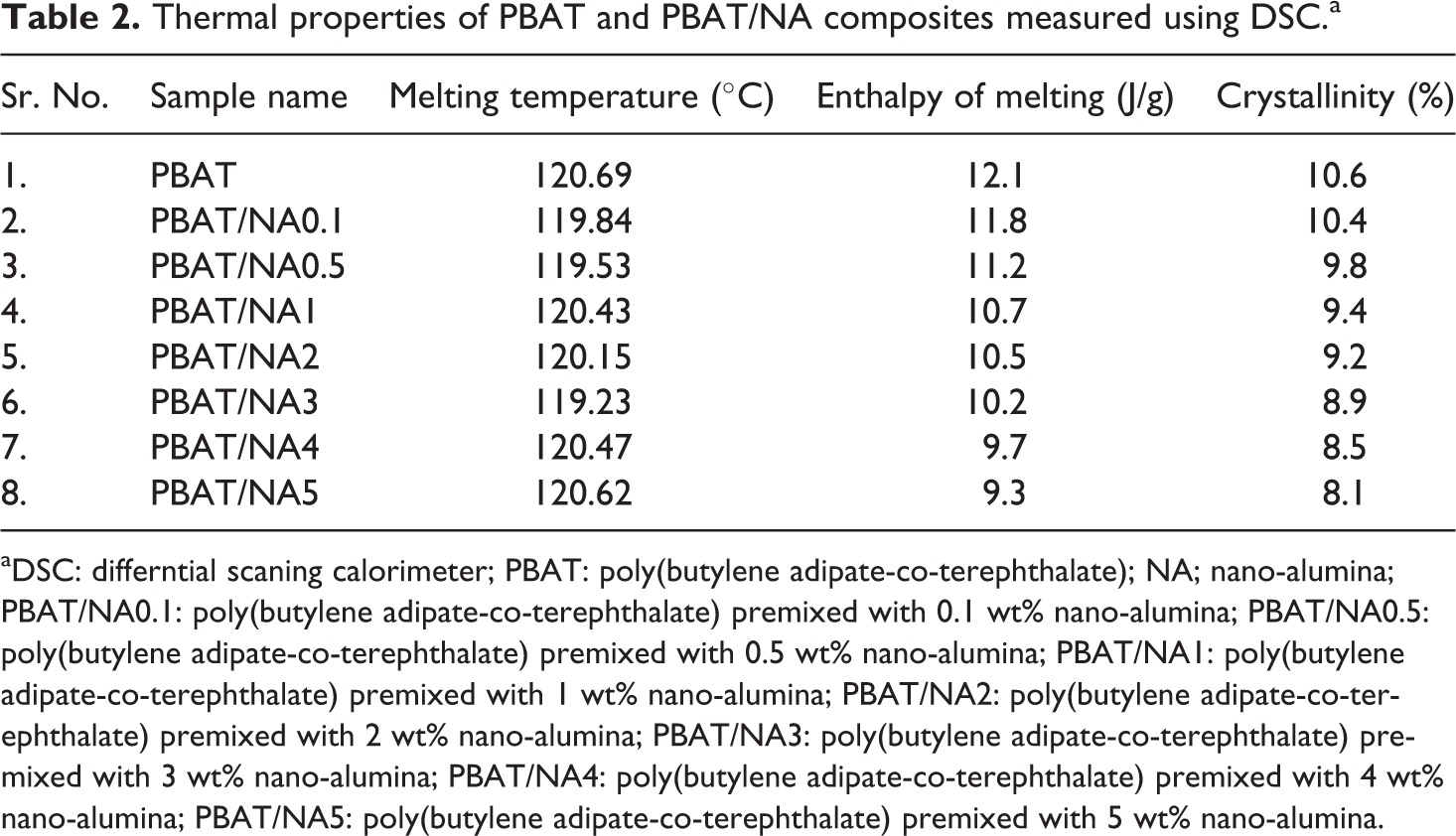
aDSC: differntial scaning calorimeter; PBAT: poly(butylene adipate-co-terephthalate); NA; nano-alumina; PBAT/NA0.1: poly(butylene adipate-co-terephthalate) premixed with 0.1 wt% nano-alumina; PBAT/NA0.5: poly(butylene adipate-co-terephthalate) premixed with 0.5 wt% nano-alumina; PBAT/NA1: poly(butylene adipate-co-terephthalate) premixed with 1 wt% nano-alumina; PBAT/NA2: poly(butylene adipate-co-terephthalate) premixed with 2 wt% nano-alumina; PBAT/NA3: poly(butylene adipate-co-terephthalate) premixed with 3 wt% nano-alumina; PBAT/NA4: poly(butylene adipate-co-terephthalate) premixed with 4 wt% nano-alumina; PBAT/NA5: poly(butylene adipate-co-terephthalate) premixed with 5 wt% nano-alumina.
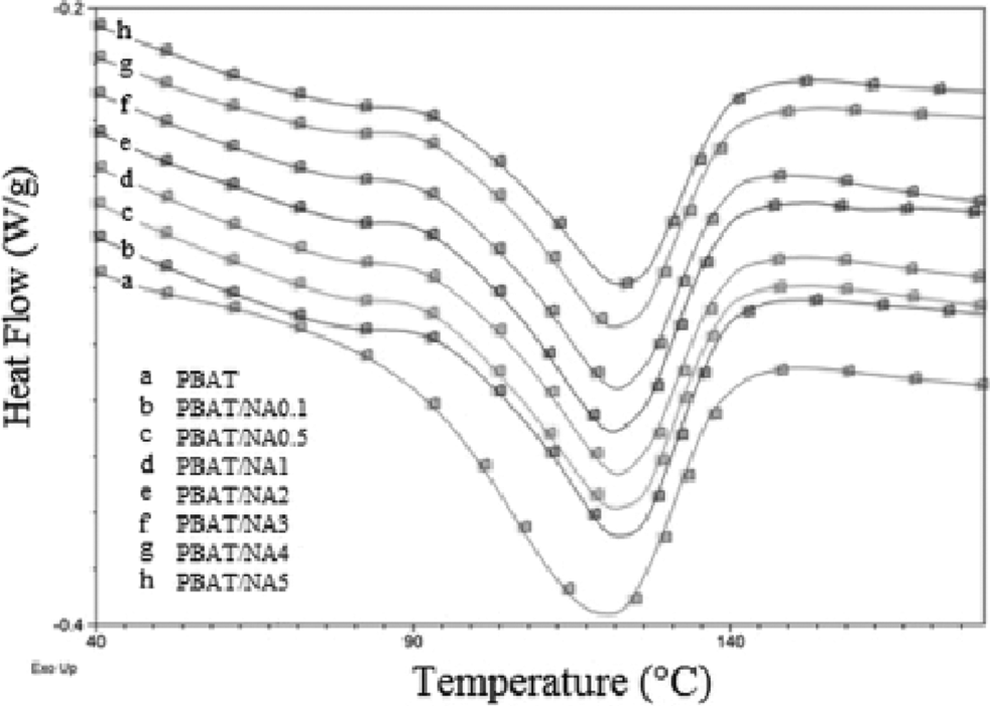
DSC heating scan curves obtained for PBAT and PBAT/NA nanocomposites. PBAT: poly(butylene adipate-co-terephthalate); PBAT/NA: poly(butylene adipate-co-terephthalate)/nano-alumina.
There could be one more reason for the observed reduction in Hm of PBAT with increase in concentration of NA. NA is a thermally conducting material. As the concentration of NA increased in PBAT, number of NA particles available for thermal conduction increases. This makes PBAT polymer chains more prone to heat absorption, decreasing the energy requirement for melting and thus decreasing Hm.
Mechanical properties
Tensile test was performed in mechanical properties. TS and PEB values obtained for PBAT and PBAT/NA composite films are illustrated in Figures 5 and 6 respectively. PBAT was found to have TS and PEB of 23 MPa and 784% respectively. Addition of NA increased TS and decreased PEB. However, the improvement in TS is insignificant. TS of PBAT/NA5 increased by just 18% compared to PBAT. Addition of NA does not affect the crystallinity of PBAT (as is known through XRD and DSC analysis). This is the reason for the insignificant improvement in TS. Thus, NA is not acting as a nucleating agent in PBAT. PEB decreased significantly with increase in concentration of NA. NA does not have very good interaction with PBAT. So, the particles of NA acts as point of rupture in PBAT on elongation, decreasing the PEB.
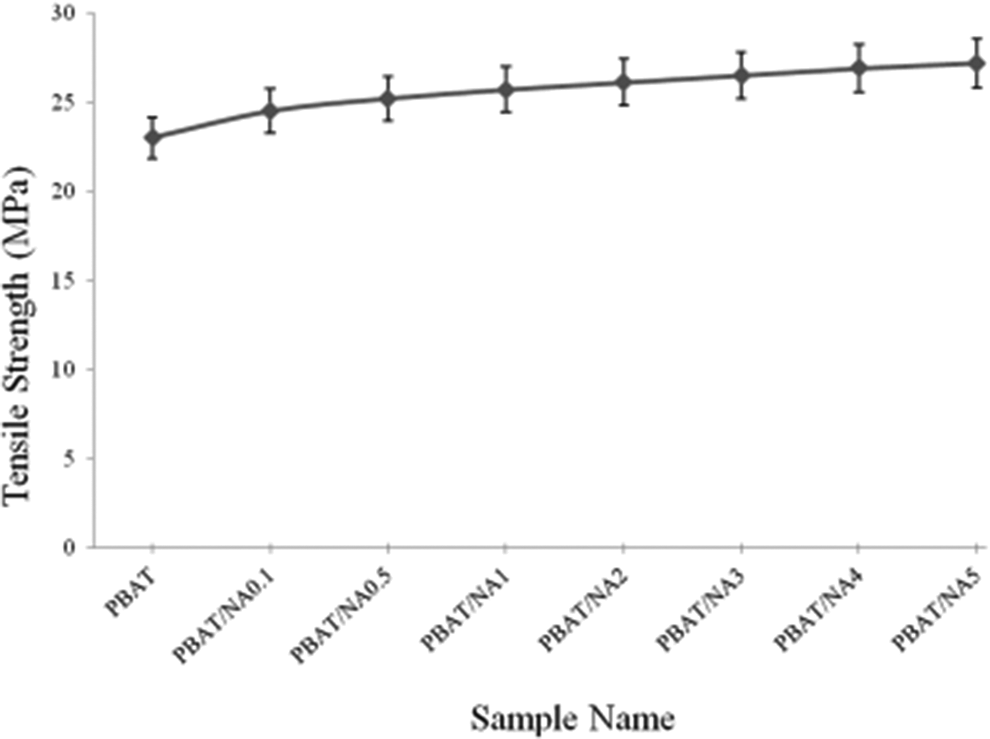
Effect of NA concentration on the tensile strength of PBAT. PBAT: poly(butylene adipate-co-terephthalate); NA; nano-alumina.
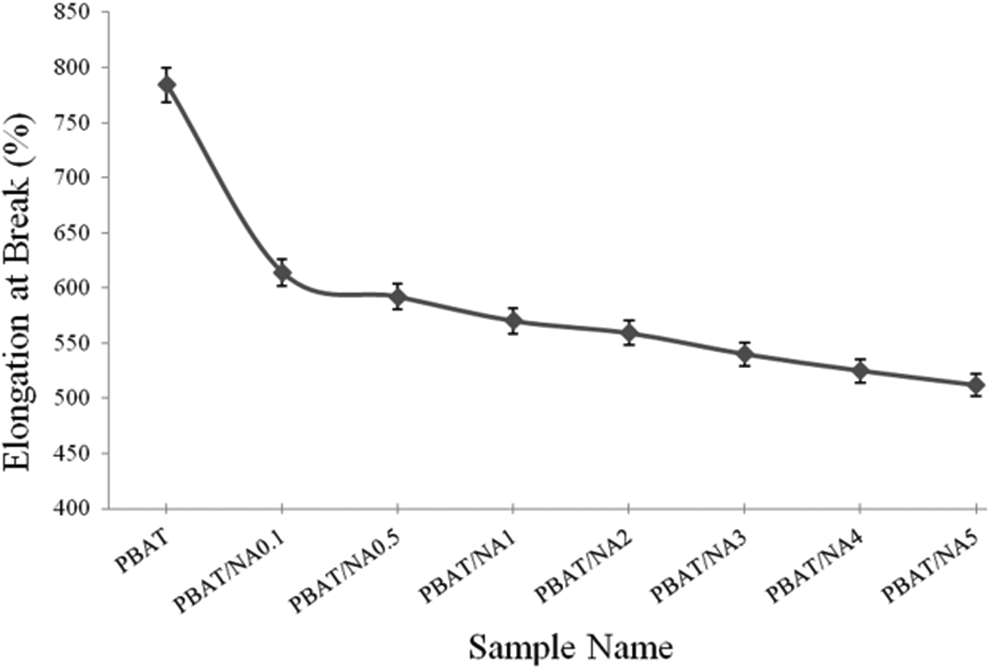
Effect of NA concentration on the percentage elongation at break property of PBAT. PBAT: poly(butylene adipate-co-terephthalate); NA; nano-alumina.
Barrier properties
Water vapour permeability (WVP)
WVP values obtained for PBAT and PBAT/NA composite films are illustrated in Figure 7. WVP for PBAT was determined to be 9.99 x 10-10 gm.mm cm−2 day−1. WVP decreased with increase in concentration of NA in PBAT. Lowest WVP was obtained for PBAT/NA3, which was 4.1 x 10-10 gm.mm cm−2 day−1. However, for higher concentration of NA (above 3 wt%) WVP started increasing. For PBAT/NA3, WVP decreased by 59%, that is, for just 3 wt% loading of NA in PBAT The increase is highly appreciable. From SEM analysis it was found that NA particles were uniformly dispersed in PBAT at 3 wt% concentration and aggregates were formed at 4 wt% concentration. At 3 wt% concentration, uniformly distributed NA particles acted as barrier for the water molecules transferring through it. Crystallinity of PBAT is not affected by the addition of NA is proven by XRD and DSC analysis. Thus, it is only the presence of NA particles, which decreased the WVP of the PBAT films. The formation of aggregates at 4 wt% and above decreased the effective surface area of NA particles, decreasing the resistance of PBAT films to WVP. Thus, uniformly dispersed NA particles upto 3 wt% increased the tortuosity of the path for the transfer of water molecules, decreasing WVP. PBAT/NA3 had the lowest possible WVP compared to PBAT.
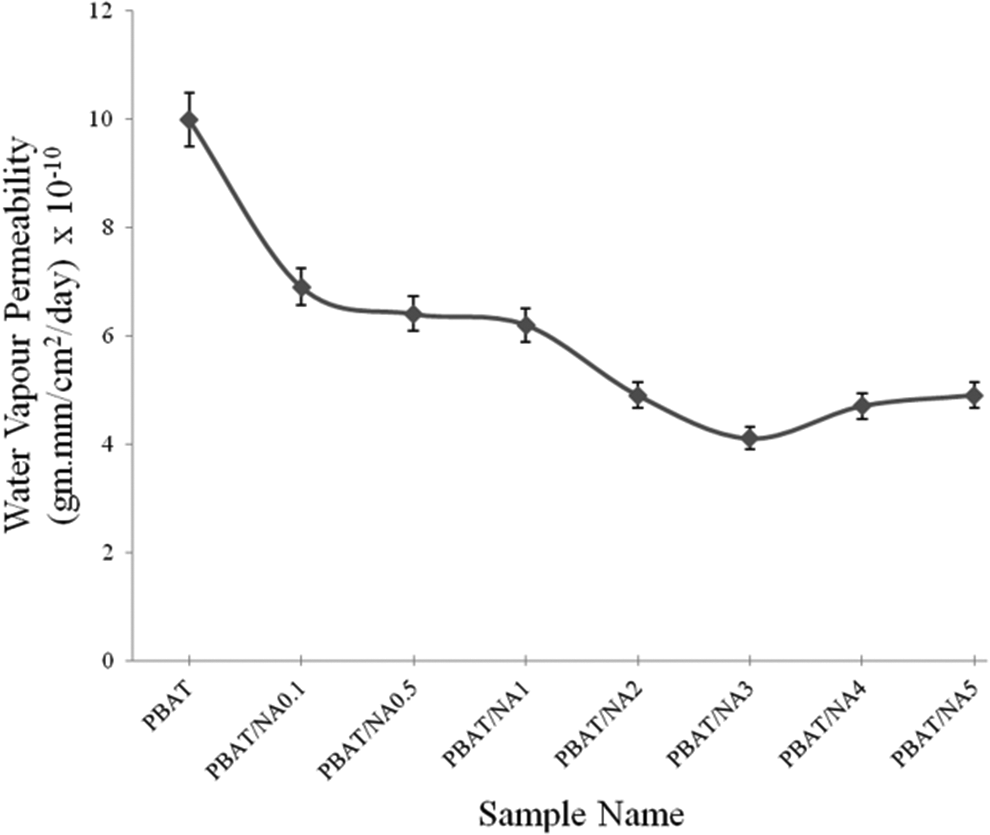
Effect of NA concentration on the WVP of PBAT. PBAT: poly(butylene adipate-co-terephthalate); NA; nano-alumina; WVP: water vapour permeability.
Oxygen permeability (OP)
OP values obtained for PBAT and PBAT/NA composite films are illustrated in Figure 8. OP for PBAT was determined to be 11.6 × 10−11 cm3.cm cm−2 s−1 cm−1Hg. OP decreased with increase in concentration of NA in PBAT. Lowest OP was obtained for PBAT/NA3, which was 7.8 × 10−11 cm3.cm cm−2 s−1 cm−1Hg. However, for higher concentration of NA (above 3 wt%) started increasing. OP decreased by about 33% for 3 wt% NA loaded PBAT. Reasons are same as that explained for WVP. NA increased the tortuosity of the path for the transfer of oxygen molecules, decreasing OP. However, at higher concentration, NA started forming agglomerates (as evident from SEM analysis), decreasing the effective surface area for resisting the flow of oxygen molecules.
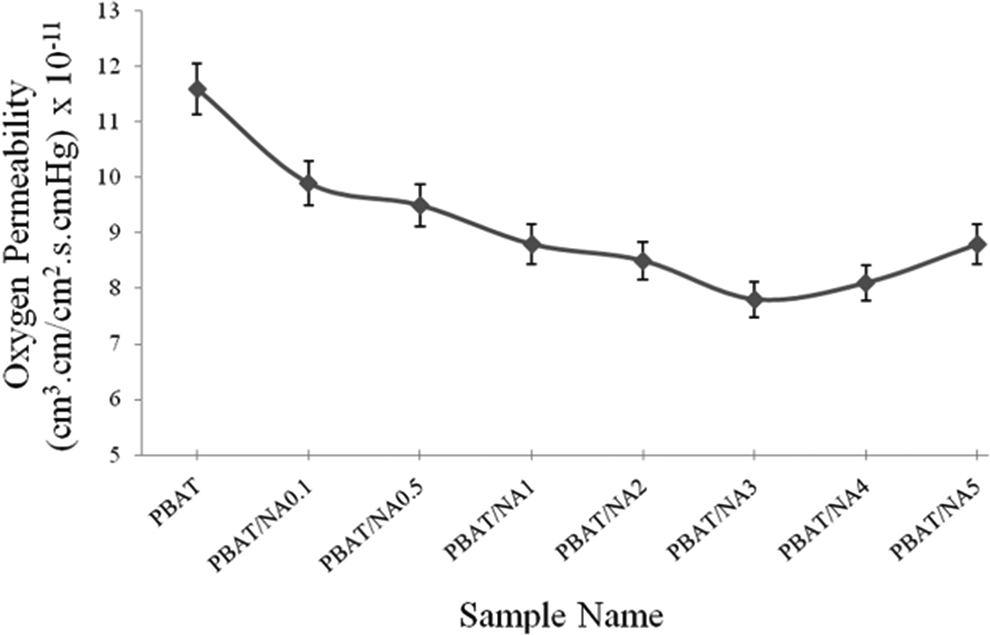
Effect of NA concentration on OP of PBAT. PBAT: poly(butylene adipate-co-terephthalate); NA; nano-alumina; OP: oxygen permeability.
Thus, PBAT/NA3 PBAT containing 3 wt% NA is the optimized concentration of NA to bring about maximum reduction in WVP and OP.
Rheological properties
Figure 9 plots the effect of NA concentration on the viscosity of PBAT. Viscosity decreased continuously with increase in shear indicating the shear thinning nature of PBAT and PBAT/NA composites. For PBAT, PBAT/NA 0.1 and PBAT/NA 0.5 viscosity decrease was sharp from shear rate of about 5 s-1; whereas, for PBAT containing higher concentration of NA this sharp decrease point of viscosity was seen at about 0.1 s−1 shear rate. Also, viscosity decreased with increase in concentration of NA in PBAT. NA being a thermally conducting material, as the concentration of it increased in PBAT, the heat absorption rate of PBAT/NA composite increased. Thus PBAT polymer chains had more exposure to high temperature. This led to breakage of the PBAT chain structure making its viscosity decrease more compared to PBAT. This would prove very beneficial in processing of this composite by extrusion or injection moulding or compression moulding process, as they would require less pressure.
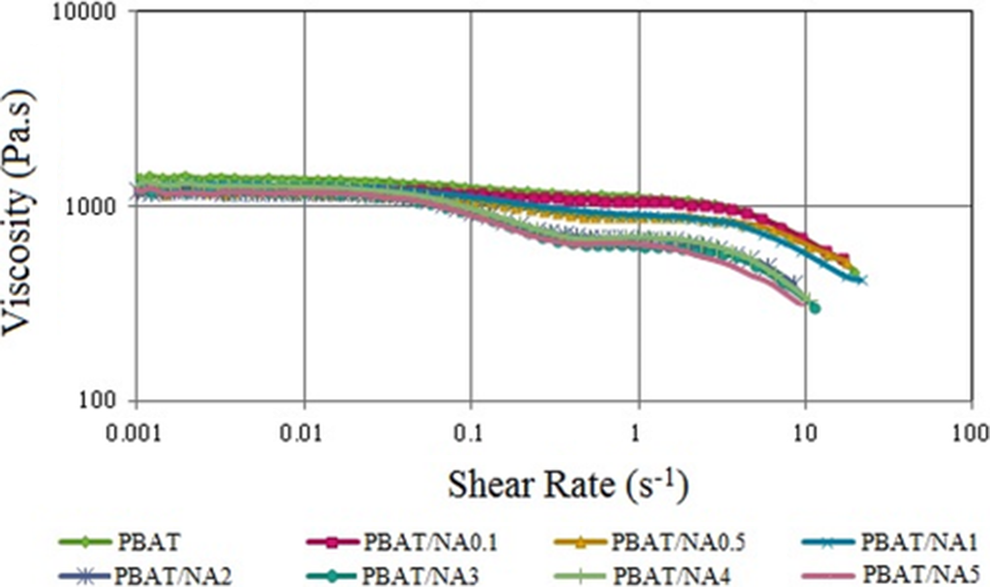
Effect of NA concentration on the viscosity of PBAT. PBAT: poly(butylene adipate-co-terephthalate); NA; nano-alumina.
Conclusion
PBAT/NA composite films were successfully prepared by extrusion processing and subsequent compression moulding. In PBAT concentration of NA varied from 0.1 to 5 wt%. PEB and crystallinity decreased with increase in concentration of NA, as NA decreased the crystalline structure formation capacity of PBAT. However, tensile of PBAT on addition of NA remained unaffected. WVP and OP decreased by 59 and 33% respectively for 3 wt% loading of NA in PBAT. Thus, this film can have potential application in food and pharmaceutical packaging. Viscosity and Hm also decreased with increase in NA concentration in PBAT, due to the thermally conducting nature of NA. This will be very much beneficial is processing the PBAT/NA composites at lower pressure in extrusion or injection moulding or compression moulding, decreasing the wear and tear of the equipment.
Footnotes
Acknowledgement
The authors are thankful to National Agricultural Innovation Project (NAIP) and Indian Council of Agricultural Research (ICAR) for the support through its sub-project entitled ‘Synthesis and characterization of CNW and its application in biodegradable polymer composites to enhance their performance’, code number ‘C2041’.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
