Abstract
Poly(vinylidene chloride-co-acrylonitrile)-based gel polymer electrolytes are obtained using solution casting technique. The ionic conductivity and thermal behavior of polymer electrolytes have been investigated as a function of wt% of ethylene carbonate (EC). The thermal decompositions of polymer electrolytes have been determined by thermogravimetry and differential thermal analysis in the temperature range between 30 and 800°C. All the polymer complexes can be operated upto 170°C. This range well includes the operating temperature range of the lithium polymer batteries incorporating these materials. Bulk resistance of the electrolyte from alternating current (AC) impedance analysis gives the optimal EC ratio as 80 wt% for maximum ionic conductivity.
Introduction
In recent years, the development of polymer electrolytes have become more widely used in solid state generators with high energy density. Most of the processes for preparing gel polymer electrolytes require a moisture-free environment because of the higher water sensitivity of the lithium salt. Furthermore, their mechanical properties are often very poor. 1 The addition of vinylidene chloride unit to polyacrylonitrile (PAN) gives high mechanical stability. Changsuk et al. 2 polymerized mixtures of poly(ethylene oxide) (PEO)-oligomers with poly(vinylidene chloride-co-acrylonitrile) (PVdC-co-AN) and yielded mechanically stable polymer electrolytes with an ionic conductivity of 10−4 to 10−5 S cm−1. Mucha and Kryszewski 3 reported that polymer blends of PVdC and PAN begin to undergo destruction at lower temperatures than homopolymers PVdC and PAN. O’shea et al. 4 recorded the Fourier transform infrared spectra of chars produced by heating pure PVdC and the copolymer Saran over the 100–690°C range. In order to provide a detailed description about the effect of organic solvent on the thermal stability of PVdC-co-AN, four different formulations of PVdC-co-AN electrolyte films have been prepared and studied by including ionic conductivity measurement at different temperatures and thermal properties. Lithium tetraflouroborate (LiBF4) has been chosen as ionic salt in the present study, since it has low lattice energy. The researchers who initiated the study on LiBF4 mentioned the multiple advantages of LiBF4 as compared to other lithium salts (e.g. less toxicity than lithium hexafluoroarsenate (LiAsF6) and higher safety than lithium perchlorate (LiClO4)).
Experimental
Preparation of solid polymer electrolyte
PVdC-co-AN (Average Mw ∼150,000, Sigma Aldrich, USA), electrolyte salt LiBF4 (Sigma Aldrich, USA), and plasticizer ethylene carbonate (EC; Alfa Aesar, Hyderbad, India) were used as received. All the solid polymer electrolyte films were prepared by solution casting technique. Appropriate weight of polymer and salt was dissolved in the given organic solvent tetrahydrofuran (THF; Merck, Mumbai, India) separately at room temperature. After the complete dissolution of polymer and salt, they were mixed together and the plasticizer was added. The mixture was allowed to stir continuously for 24 h with the help of magnetic stirrer. The solvent in the mixture was allowed to evaporate slowly until a homogeneous solution was obtained and it was degassed to remove air bubbles. The thus obtained highly viscous solutions were cast onto clean glass plates to obtain the desired thin film membranes. The films were further dried for 5 h in vacuum to remove the residual solvent THF.
Sample characterization
The ionic conductivity of polymer electrolytes was carried out with the help of stainless steel blocking electrodes using a computer controlled µ-Autolab Type-III Potentiostat/Galvanostat in the frequency range of 40 Hz to 300 kHz with the signal amplitude of 0.25 V. The measurements were taken between 303 and 363 K. The value of bulk resistance R b was found to be the intercept on the x-axis of the impedance plot. The conductivity (σ) was calculated using the formula
where l is the thickness of the polymer electrolyte film, R b is the bulk resistance, and A is the surface area of the polymer electrolyte film. 5
All the polymer complexes were subjected to thermal analysis, which was carried out using Perkin Elmer STA-6000 thermogravimetry/differential thermal analysis (TG/DTA) analyzer from STIC Cochin. The thermogravimetric analysis (TGA) and DTA curves were recorded in nitrogen atmosphere from room temperature to 800°C. The heating rate was 10°C min−1. From TG and differential thermogravimetric (DTG) plots, temperature of maximum process rate, temperature at the end of decomposition, and weight loss were determined. The recorded DTA curves allow comparing the endothermal effect in polymer complexes.
Results and discussion
Thermal studies
As a qualitative tool TG can be used to identify the thermal stability and decomposition of polymers and its constituents based on their volatility. Volatile components such as water and organic solvents evaporate at low temperatures (say 100–120°C); at intermediate temperatures, plasticizers are removed and at high temperatures, only nonvolatile components such as inorganic fillers remain as residue. 6,7
TG and DTA curves of polymer complexes are presented in Figure 1.
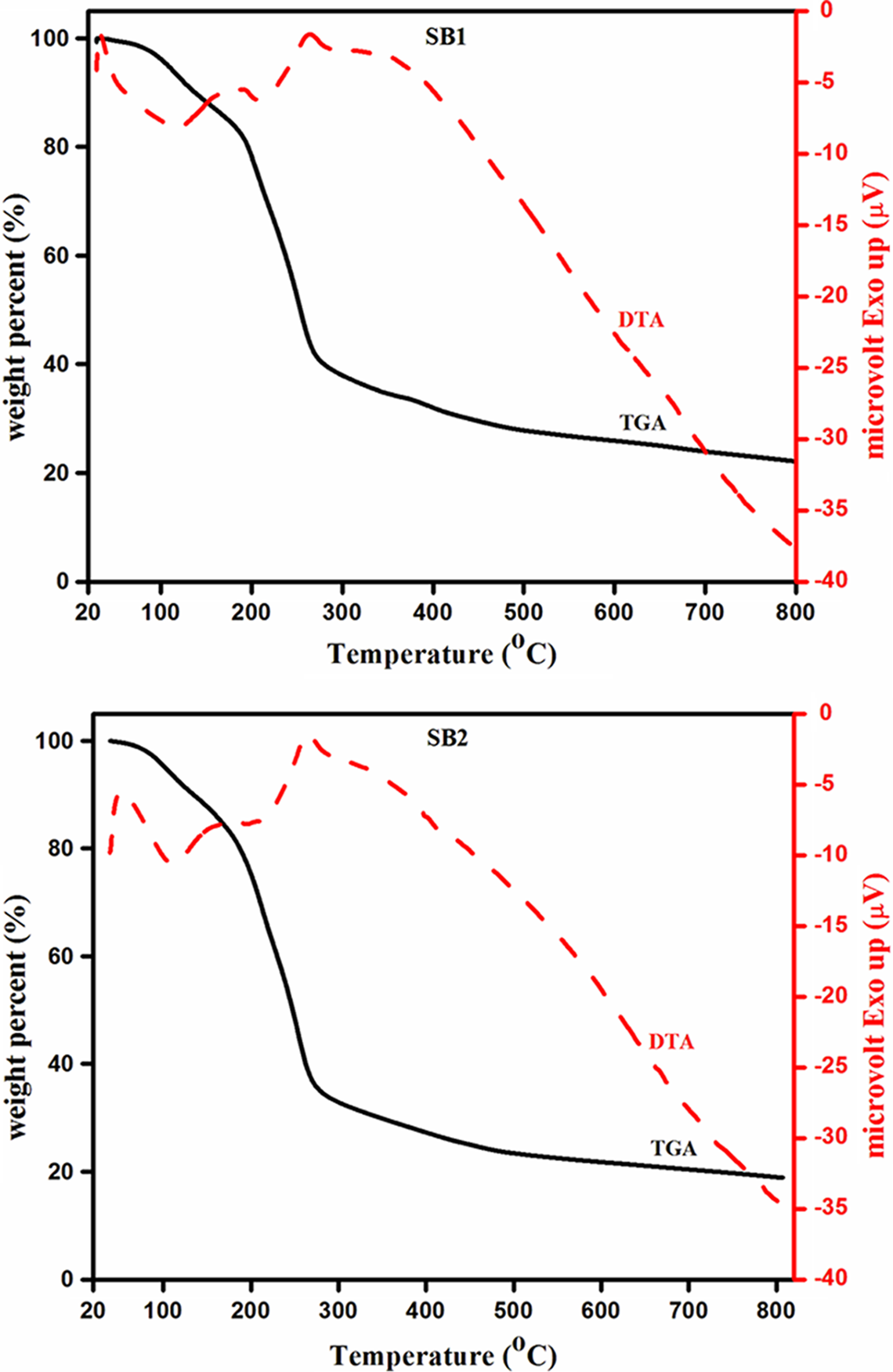
TG/DTA curves of polymer complex with different EC concentrations in wt% (SB1(20), SB2(40), SB3(60), and SB4(80)). TG: thermogravimetry; DTA: differential thermal analysis; EC: ethylene carbonate.
Thermogravimetric analysis
It is observed from Figure 1 that all four polymer complexes in TGA exhibit the one-step degradation in which almost complete decay is observed (weight loss is about 80% at the end of process i.e. at 800°C). The samples SB1, SB2, SB3, and SB4 show weight loss of about 2, 3, 2, and 4%, respectively, at 90°C and this weight loss is mainly due to the moisture or by the evaporation of residual solvent which is used in the sample preparation.
Thermal decomposition in polymer electrolyte SB1 starts at about 192°C, while in compounds SB2, SB3, and SB4 at significantly lower temperature (186, 176, and 170°C respectively). Though vinylidene chloride polymers are highly resistant to oxidation, permeation of small molecules, and biodegradation, they are extremely durable under most usage conditions. These materials are thermally unstable and, when heated above 120°C, undergo degradative dehydrochlorination. 8 About 170°C, it is observed a significant change in the polymer’s original, highly ordered structure, which indicates the breakage of CH2 bonds in the polymer chain and the expansion of the carbon–halogen bonds (C–Cl), which leads to the elimination of the halogen atoms at sites along the polymer chain. 4 All the prepared samples show appreciable weight losses of about 16, 18, 15, and 16%, respectively, at 180°C. The loss of approximately half of the chlorine atoms from the polymer chain also results in significant changes in the stability of polymer electrolyte. Acrylonitrile copolymers release a small amount of ammonia and hydrogen cyanide 9,10 as well as hydrogen chloride above, 11 since degradation apparently begins in vinylidene chloride units adjacent to comonomer units. The weight loss observed in the temperature range of 180–270°C is mainly due to the degradation of polymer, lithium salt, and plasticizer. It is found that there is no appreciable weight loss for all the polymer electrolytes beyond 270°C.
TG/DTA parameters of the polymer electrolyte prepared in the present study are summarized in Table 1. The results show that the gel polymer electrolyte with 20 wt% EC (SB1) exhibits higher thermal stability. All polymer electrolytes incorporating different ratios of EC exhibit maximum thermal stability of about 170°C. The results obtained show that the degradation process of the polymer electrolyte complexes depends not only on their composition but also on the chemical structure of the components, whereby the components at various concentrations exhibit mutual interactions. 12
Thermal parameters of polymer complexes.
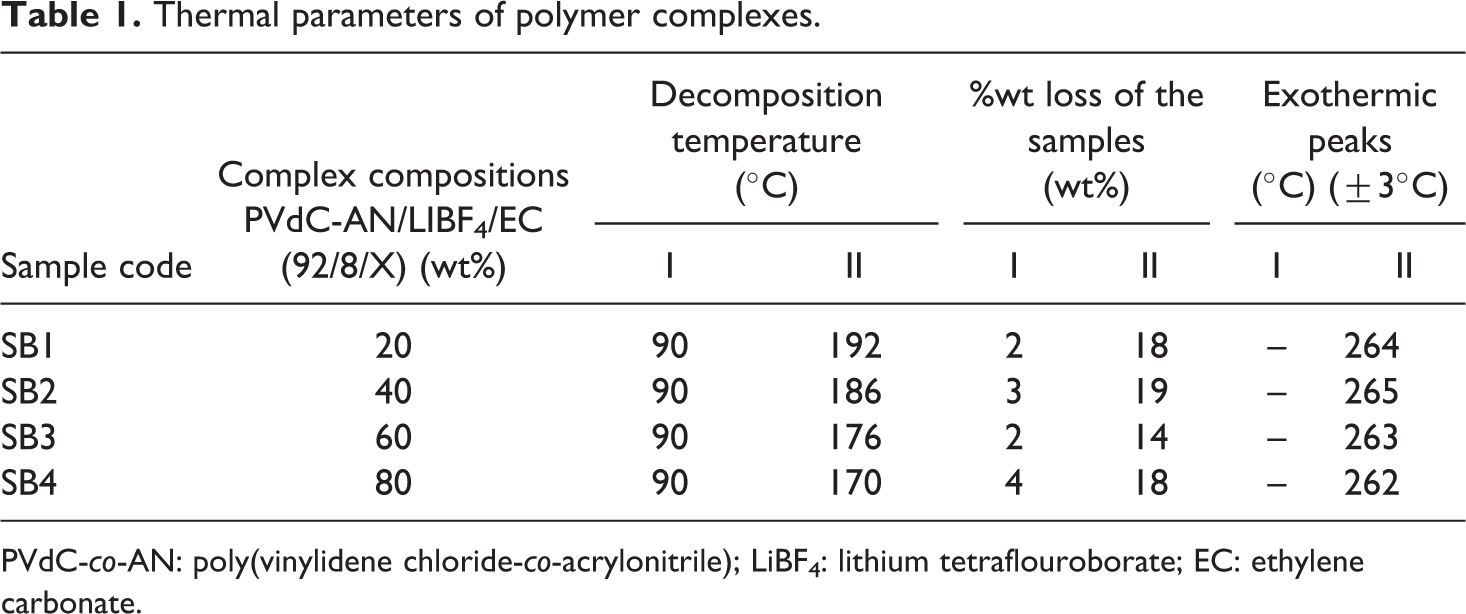
PVdC-co-AN: poly(vinylidene chloride-co-acrylonitrile); LiBF4: lithium tetraflouroborate; EC: ethylene carbonate.
Derivative of thermogravimetric analysis
Since it is difficult to read the temperature of maximum rate of decomposition from the inflection point on TG in the case of partially overlapped degradation steps, differential curve of TG (i.e., DTG) is used. The DTG plot is given in Figure 2, which shows that the slope observed in TGA represents two steps of thermal decomposition. This two-step decomposition of polymer complex mainly is due to the decomposition of nitrile group and chloride group in PVdC-co-AN. In our study, the degradation steps of all polymer complexes start at (192, 236), (186, 242), (176, 242), and (170, 240)°C, respectively. Also the weight loss in second step was smaller compared to first step decomposition. It can be explained by the higher energy of dissociation of C≡N compared to C–Cl bond. The residual mass of about 20% at 800°C can be assigned to the carbonaceous residue formed at both previous stages. 13 It has been concluded that the thermal stability decreased with increasing content of EC.
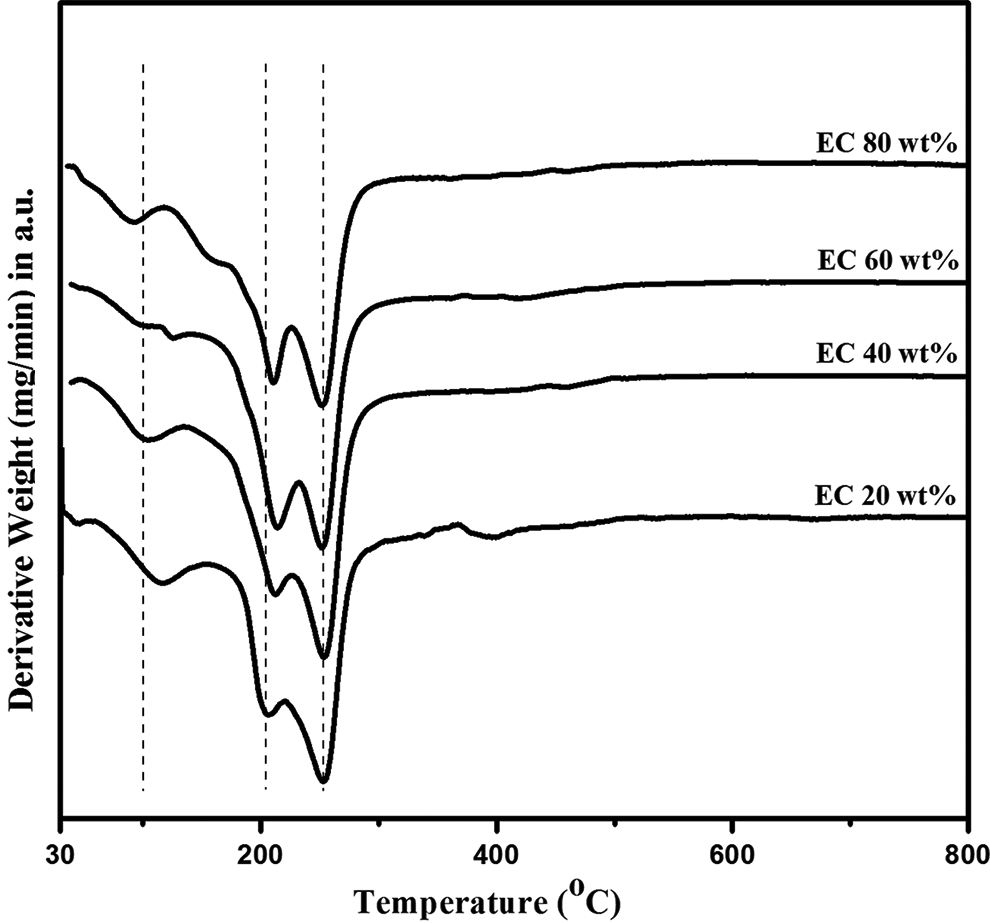
Derivative of thermogravimetry curves of PVdC-co-AN/LiBF4/EC complexes. PVdC-co-AN: poly(vinylidene chloride-co-acrylonitrile); LiBF4: lithium tetraflouroborate; EC: ethylene carbonate.
Differential thermal analysis
The recorded DTA parameters are listed in Table 1 in order to compare the endothermal effect of polymer complexes. The melting temperature (T m) of the gel polymer complexes can be found from the second endothermic peak in DTA curves. The comparison of TGA and DTA shows that the thermal decomposition of all complexes starts before the melting temperature. The endothermic heat flow observed around 210°C in DTA for the entire polymer complexes corresponds to the melting point of polymer PVdC-co-AN. It is confirmed that as plasticizer ratio increases from 20 to 100 wt%, the melting point of the complex decreases as observed in TGA and DTG.
The exothermic heat flow at 264, 264, 263, and 262°C, respectively, in the complexes in DTA represents the decomposition of EC, since its boiling point is 260°C. There is no heat flow above 300°C in all the complexes, which shows the complete decomposition of polymer, plasticizer, and salt. The weight loss observed in TGA curves is also confirmed from the DTA curves by the presence of endothermic peaks in all the four samples, and the DTA results are in close agreement with the TGA analysis.
Ionic conductivity studies
The typical impedance plot for PVdC-co-ANdF gel polymer electrolytes at room temperature is presented in Figure 3. The highest ionic conductivity has been observed at 80 wt% of plasticizer in the overall range of temperatures studied. The ionic conductivities of PVdC-co-AN/LiBF4 complexes with different ratios of EC are shown in Figure 4. Ionic conductivity as a function of plasticizer ratio can be generally explained by the decrease in glass transition temperature (T g) competing with the increase in the number of charge carriers. Figure 4 shows that the addition of EC results in an increase in conductivity values. As the plasticizer ratio increases, the ionic conductivity also increases, which is due to the increment in the mobility of charge carriers in the polymer–salt complexes. The observed maximum ionic conductivity value shows a balance between number of charge carriers and ion mobility. 14,15
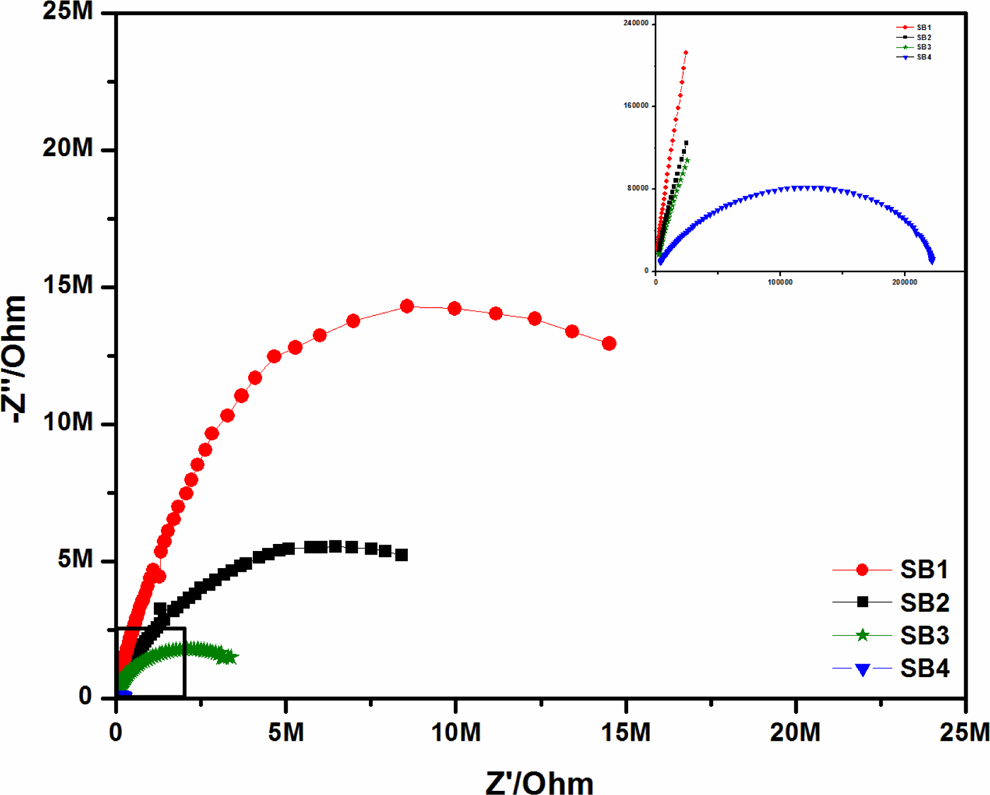
Impedance plots of PVdC-co-AN/LiBF4/EC complexes at room temperature. PVdC-co-AN: poly(vinylidene chloride-co-acrylonitrile); LiBF4: lithium tetraflouroborate; EC: ethylene carbonate.
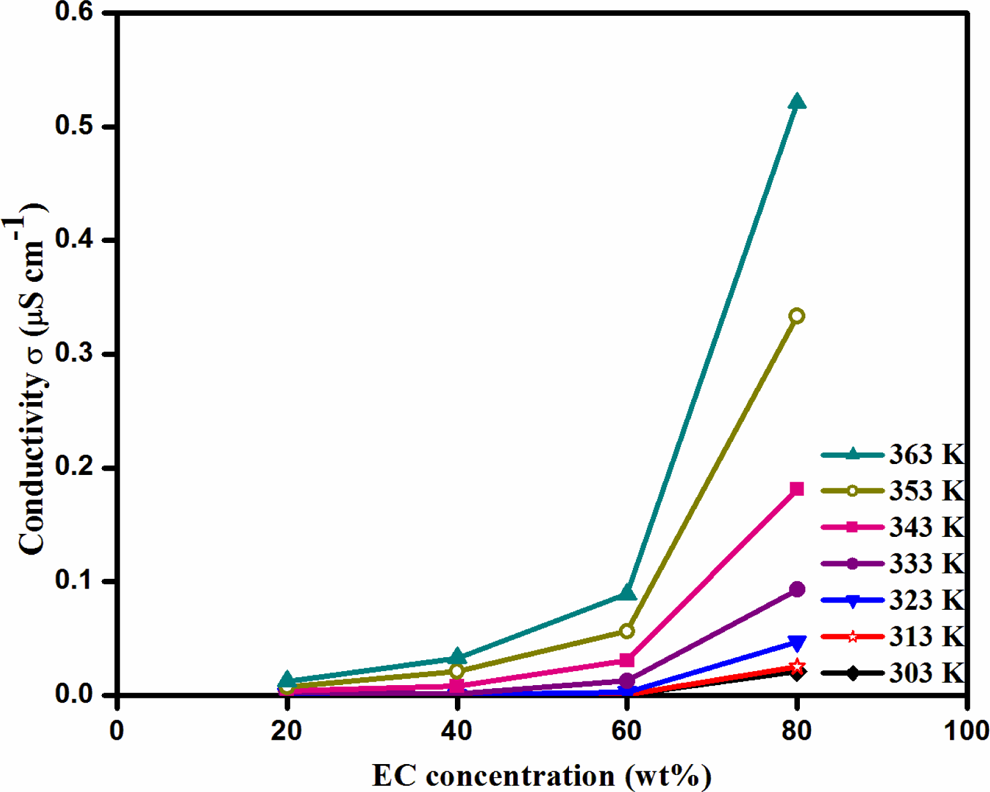
Dependence of ethylene carbonate concentration on the ionic conductivity of PVdC-co-AN/LiBF4/EC complexes at different temperatures. PVdC-co-AN: poly(vinylidene chloride-co-acrylonitrile); LiBF4: lithium tetraflouroborate; EC: ethylene carbonate.
The variations in log σ with inverse of absolute temperature for all the prepared polymer electrolyte films are shown in Figure 5. From the figure, it is observed that the increase in temperature increases the conductivity of polymer electrolytes over the studied temperature range. The nonlinear behavior of the temperature dependence on the ionic conductivity can be expressed by the Vogel–Tamman–Fulcher (VTF) expression,
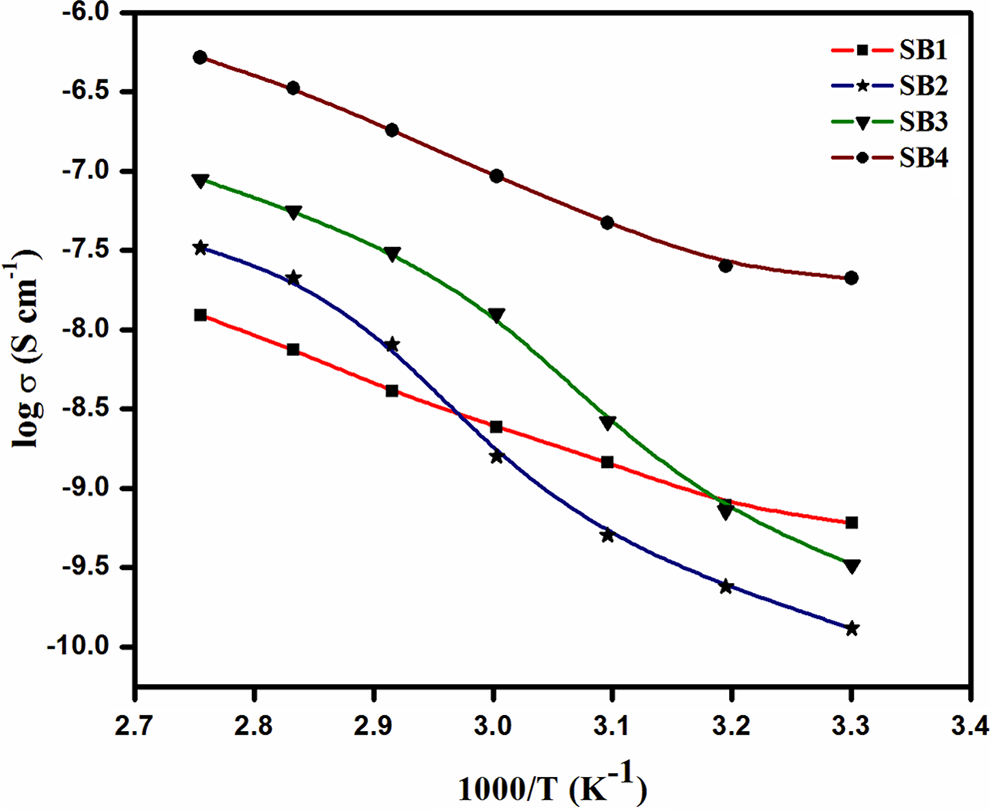
Temperature dependence plot of ionic conductivity for PVdC-co-AN/LiBF4/EC gel polymer complex containing various EC concentrations. PVdC-co-AN: poly(vinylidene chloride-co-acrylonitrile); LiBF4: lithium tetraflouroborate; EC: ethylene carbonate.
The increase in conductivity with respect to the increase in temperature can be explained by diffusion theory. The diffusion coefficient of small particles in such amorphous polymer electrolyte media can be expressed as
The ionic conductivity and diffusion coefficient can be related by Nernst–Einstein relation. Thus, a good fit of the conductivity data of plasticized PVdC-co-AN electrolytes to VTF relation suggests that the conductivity change with temperature is dominated by the change in the diffusion coefficient (ionic mobility), which can be explained by free volume mechanism. When the free volume large enough for their diffusive displacement (
Conclusion
The increase in temperature nonlinearly increases the ionic conductivity of polymer complex over the studied temperature range. The ionic conductivity of polymer complexes increases upto an optimum value of plasticizer ratio of 80 wt%. The thermal analysis leads to the conclusion that the addition of plasticizers does not produce positive results on their thermal stability. The results obtained show that the degradation process of gel polymer electrolyte depends not only on the chemical structure of the components but also on their composition, where the components at various ratios exhibit mutual interactions.
Footnotes
Funding
The author C.M.M. is financially supported by the Union Grants Commission (UGC), Government of India, India.

