Abstract
Isotactic poly(4-methyl-1-pentene) (PMP) is a semicrystalline polyolefin with many unique properties. The crystallization theory of pure PMP had been well studied based on its molecular structure. In this study, the crystallization behavior and compatibility of PMP/polypropylene (PP) blends were investigated by differential scanning calorimetry, x-ray diffraction, scanning electron microscopy (SEM), and mechanical testing. It was found that the PP crystallization behavior was strongly affected by the presence of PMP in the blend: first, PMP crystallized and then acted as nucleating agent during the crystallization process of PP at lower temperature. In contrast, the influence of PP on the crystallization of PMP was relatively small due to the wide disparity of the melting temperatures of PMP and PP. The fracture morphology observed by SEM showed that there was no obvious phase separation structure in the blends; but the mechanical properties of PP/PMP blends are not good as expected for an ideal homogenous mixture.
Keywords
Introduction
Isotactic poly(4-methyl-1-pentene) (PMP) is a semicrystalline polyolefin with a bulky side group (CH2-CH(CH3)2), which endows PMP with a number of unique properties such as high transparency, low density, high chemical stability, high melting temperature, low dielectric constant, and high permeability. 1 –5 PMP is widely used in many industrial and medical applications such as automotive parts, separating membrane, and sterile containers. 6 However, its mechanical properties deteriorate easily even under mild condition (e.g. slightly above room temperature), and this has limited its application. 7
Until now, the crystallization behavior and theory of PMP have been well studied. For example, it was found that PMP had five different crystalline forms depending on the crystallization condition including pressure, solvent and thermal history, and so on. 8 The helix conformations with different spiral degrees make the chain packing in different unit cells, such as tetragonal unit cells of forms I and III, a monoclinic unit cell of form II, and a hexagonal unit cell of form IV. 9 –11 Forms II, III, and V spontaneously transform into the stable form I with increasing temperature. Thus, form I is the most stable and ordinary crystalline form, which exists in melt-crystallized samples and extruded fibers. It is characterized by chains of 7/2 helical conformation. 12 The results of Flory’s studies indicated that the physical properties and the molecular arrangement within the material depended on crystallization conditions. 13 Silvestre et al. studied the morphology development of isotactic PMP during melt crystallization and showed that different populations of lamellae could be obtained by changing the crystallization conditions. 14
In contrast, only very few studies have been carried out regarding the crystallization of PMP mixture with other polymers, 15 –17 but some interested crystallization behavior has been reported. For example, recently, Lee et al. reported that in electrospun poly-1-butene (PB)/PMP blends, the crystallization of PB was greatly restricted, while the behavior of PMP was similar as that of pure PMP regardless of the mixture composition. 18
In this study, we investigated the crystallization behavior, micromorphology, and mechanical properties of PMP/polypropylene (PP) blends. PP and PMP are both alpha polyolefins with similar molecular structures; so they should have very good compatibility. In addition, PP has very good bending fatigue resistance, while PMP does not. So PP and PMP mixtures might have both the good bending fatigue resistance due to the presence of PP as well as the robust physical properties contributed from both PP and PMP.
Experimental
Materials and sample preparation
The isotactic PMP (Grade RT-18) and PP (Grade F401) were purchased from Mitsui Petrochemical Industries (Tokyo, Japan) and Sinopec Yangzi Petrochemical Company (Beijing, China), respectively, without further processing. RT-18 is a commercial product and may contain some copolymer to enhance its physical or mechanical properties. 19 PMP/PP blends of various mass fraction of PP (5, 10, 20, 50, and 80 wt%) were prepared by melt-blending in a twin-screw extruder with a screw of 21.7 mm in diameter and lift-to-drag (L/D) ratio of 40. The extrusion temperature of the feeding zone/transporting zone/melting zone were set as 180/245/245°C. The rotor speed is 100 r/mim. The extrudates were subsequently pelletized.
Characterizations
Differential scanning calorimeter
The crystallization behaviors and miscibility of the blends were measured with a differential scanning calorimetry (DSC; Q20 DSC, TA Instruments, New Castle, Pennsylvania, USA) equipped with a liquid nitrogen cooling system. About 5–10 mg of each sample was encapsulated in aluminum pans and heated up to 270°C for 20 min. For non-isothermal crystallization, the melt were then cooled to 30°C with a cooling rate of 10°C/min for crystallization behavior study; the second heating were set to 270°C with a heating rate of 10°C/min to study the melting behavior. The melting transition temperature (T m) and the crystallization temperature (T c) were determined as the maximum of the corresponding transition peaks.
For isothermal crystallization of PMP, each sample was jumped to the desired T c (i.e. 223, 222, 221, or 220°C) and then held at this temperature until the heat flow returned to the initial baseline value. At this time, the transformation was considered as complete.
For isothermal crystallization of PP, all samples were first cooled with a rate of 20°C/min to 180°C and held there for 10 min for the whole crystallization of PMP in the blends. Each sample was then jumped to the desired T c, that is, 125, 124, 123, or 122°C, and held at this temperature until heat flow returned to the initial baseline value.
All measurements were carried out in a nitrogen atmosphere with an intercooler connected to keep stable control.
Scanning electron microscopy
The blend morphology was characterized with a model 505 scanning electron microscopy (SEM; Philips, Holland). The samples were freeze-fractured in liquid nitrogen. The fractured surfaces were coated with gold–palladium (Au-Pd) alloy before observation.
X-Ray diffraction
X-Ray diffraction (XRD) study was carried out using PW 1840 x-ray diffractometer with Cu-Kα targets at 2 mm slits at a scanning rate of 2° per min, range 4°–32°, operated at 36 kV, 20 mA, to get an explicit idea of the relative crystallinity of the composites. The area under the x-ray diffractogram was determined in arbitrary units. All the samples for XRD were prepared by heating up to 260°C and held there for 5 min and then cooled to room temperature at the same rate.
Mechanical analysis
Tensile, flexural, and impact bars were molded at 250°C using a reciprocating screw injection-molding machine (SHJ-20; Nanjing Jieya Extusion Equipment Co Ltd, Nanjing, China). Tensile properties were measured with a Universal Testing Machine (Zwick/Roell Z005, Zwick Roell Testing Machines Pvt Ltd, Tamil Nadu, India) according to ASTM-D638 at a crosshead speed of 100 mm/min. Standard flexural tests (ASTM-D790) were carried out at a crosshead speed of 2.0 mm/min and with a span length of 64 mm. Notched Izod impact strength was measured with a Universal Impact Testing Machine (ZBC-50, China Shenzhen SANS Testing Machine Co., Ltd, Shenzhen, China). The thickness of the Izod impact specimens was 4 mm, and the impact energy was 5.5 J. All tests were carried out at room temperature. Six measurements were performed for each data point in all mechanical property tests.
Results and discussion
Nonisothermal crystallization behavior of PMP/PP blends
The melting temperatures of the PP and PMP are 112°C of PP and 212°C, respectively. When the PMP/PP blends crystallize from the molten state, it is possible that PMP will crystallize first in the presence of melted PP chains and then the molten PP crystallizes in the presence of PMP crystals because of the wide disparity of the melting temperatures of the two components. Figure 1 showed the thermograms of pure PMP, PP, and their blends obtained during crystallization. When the amount of PP was 50 wt% or lower, the T c of PMP (T c, PMP) in the blends are similar to that of pure PMP. This implied that the addition of PP had no significant influence on the crystallization of PMP under this condition. But when the PP amount was increased to 80 wt%, the T c, PMP moved to a lower temperature, and the crystallization peak also became very small. The reason for this impeded crystallization of PMP might be PMP became dispersed phase in this blend, in which large amount of PP at the melting state make PMP molecular chains hard to arrange regularly. Comparing to PMP, the crystallization behaviors of PP were much more obviously influenced in the blends. When the amount of PP in the blends was very low (e.g. 5, 10, and 20 wt%), there was no crystallization peak at about 112°C as in pure PP. Instead, the T c of PP (T c, PP ) decreased to as low as 50 to 85°C, as indicated that the crystallization process of PP was greatly postponed. A possible explanation for this phenomenon is that due to the presence of high concentration of PMP and also PMP’s much higher T m than that of PP, a large amount of PMP crystals already formed before the start of PP crystallization. This greatly prevented the movement of PP chains during the cooling process and accordingly postponed the crystallization process of PP to a much lower temperature. In contrast, blends with medium to high level of PP showed much higher T c, PP (e.g. ∼115 and 117°C, respectively, for blends with 50 and 80 wt% of PP) and their T c, pp was even higher than that of pure PP (i.e. 112°C). This indicated that the crystallization process of PP has been accelerated or advanced due to the presence of a small to a medium amount of PMP. It is suspected that in this case, a small amount of PMP crystals first formed at higher temperature, and then they acted as nucleating agents and facilitated the PP crystallization process. The PP nucleation therefore occurred earlier during the cooling process and at higher temperature.
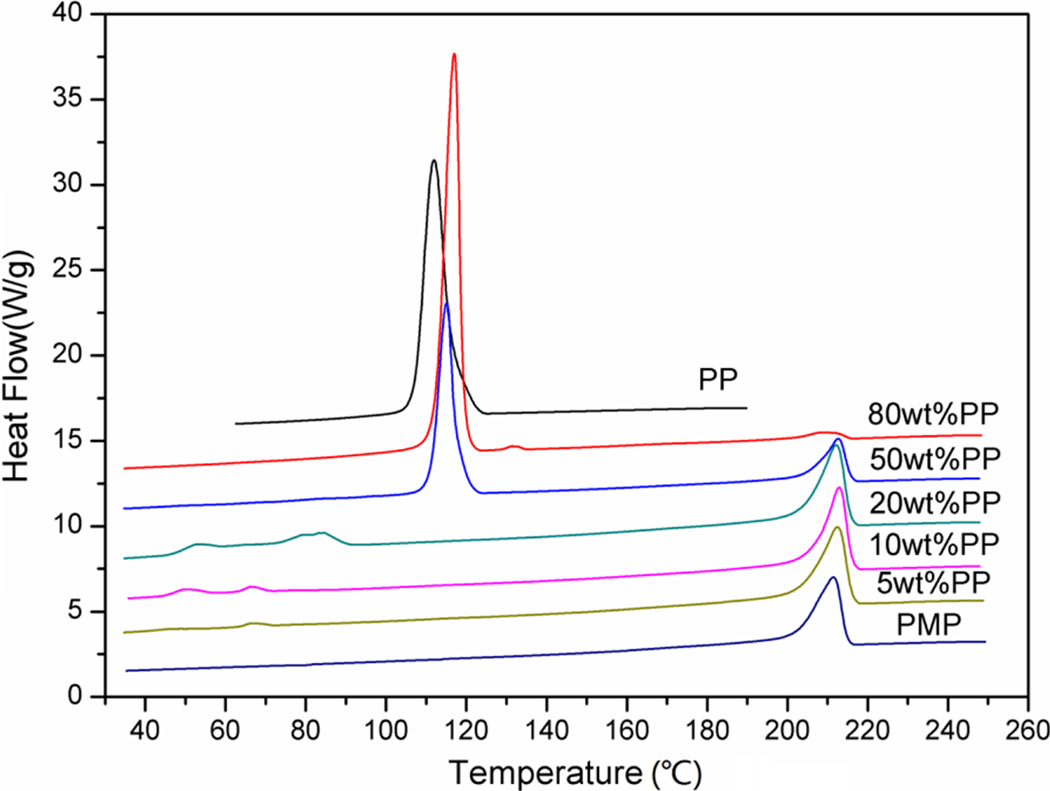
Crystallization curves of pure PMP, PP, and PMP/PP blends. PMP: poly(4-methyl-1-pentene); PP: polypropylene.
Figure 2 shows the melting curves obtained during second heating of pure PP, PMP, and PMP/PP blends. It could be found that the positions of endothermal peaks of both PMP and PP in blends (data shown in Table 1) are similar to their pure ones. This implied that the crystal types of PMP and PP in blends are probably the same as those in the pure polymer samples. However, the peak areas of these samples are very different, which means that the crystallinity of PP or PMP in each sample is different. The normalized crystallinities of pure PMP and PP and their blends are calculated with the DSC curves by using the following formula 20
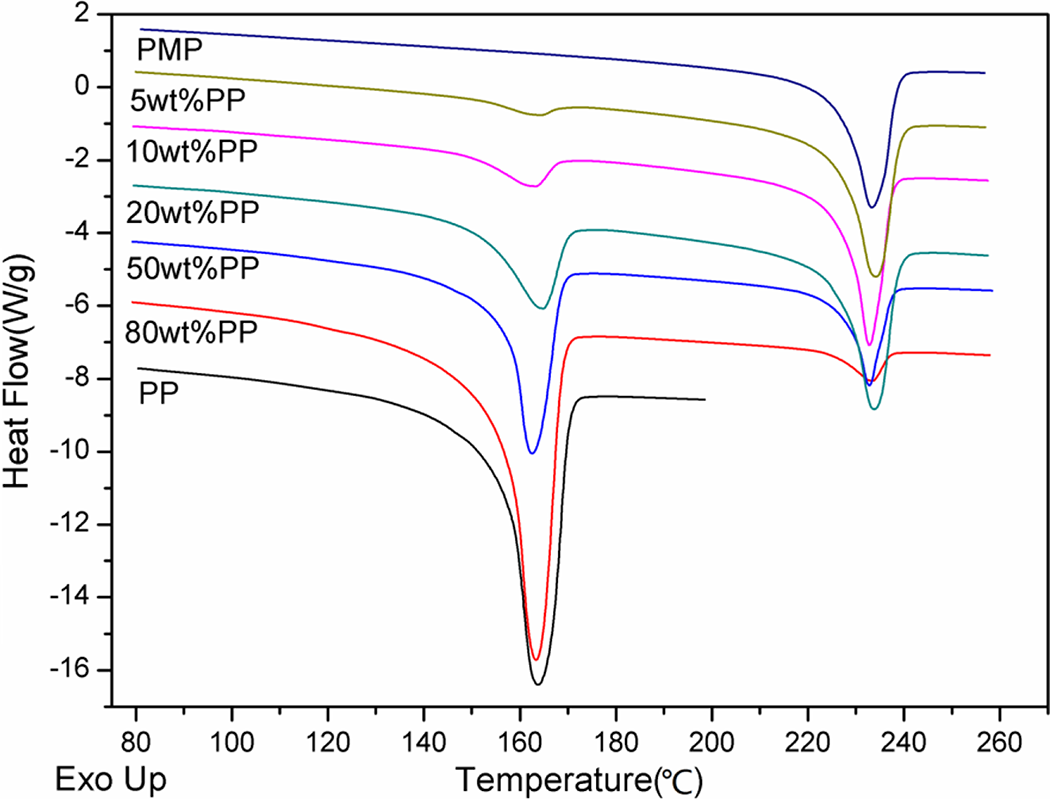
Melting curves of pure PMP, PP, and PMP/PP blends. PMP: poly(4-methyl-1-pentene); PP: polypropylene.
Melting characteristics of PMP, PP, and PMP/PP blends.a
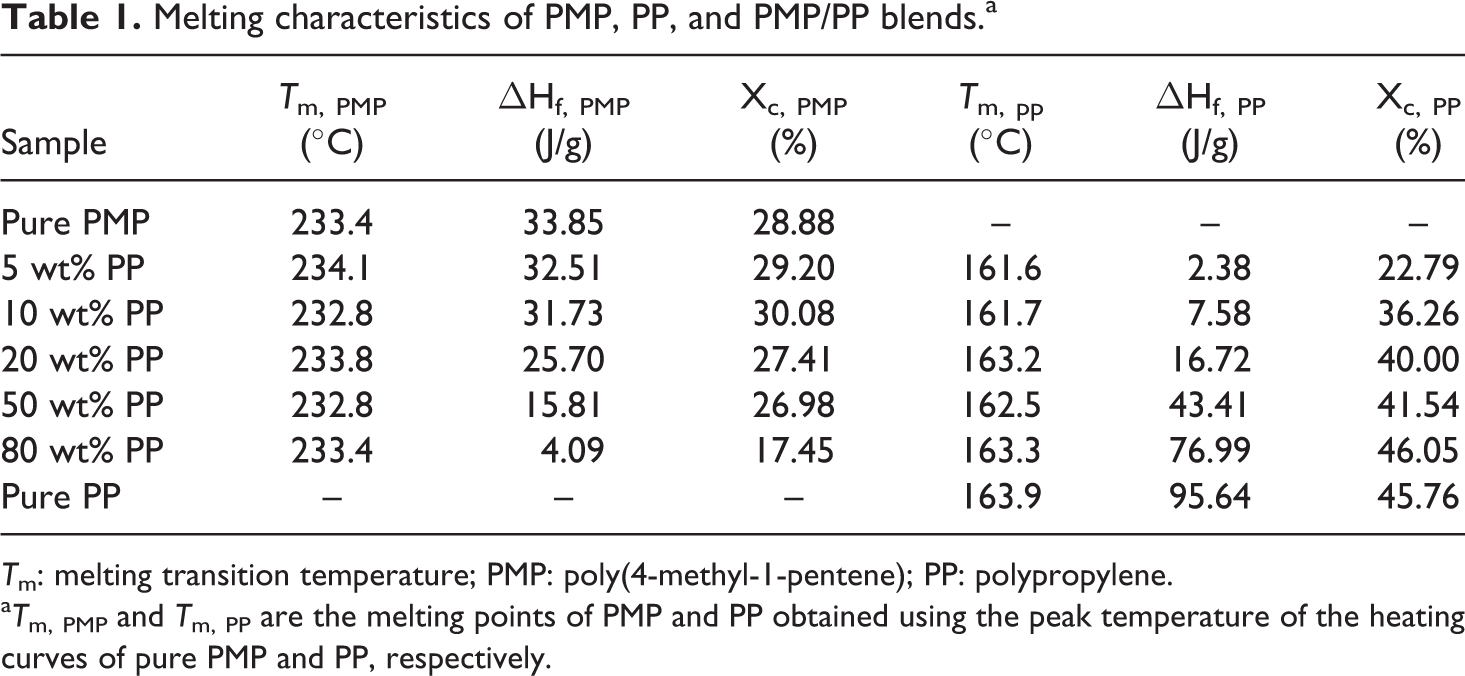
T m: melting transition temperature; PMP: poly(4-methyl-1-pentene); PP: polypropylene.
a T m, PMP and T m, PP are the melting points of PMP and PP obtained using the peak temperature of the heating curves of pure PMP and PP, respectively.

where ▵H f* is the enthalpy of fusion of the perfect PMP or PP crystal, ▵H f is the enthalpy of fusion of PMP or PP crystal measured in DSC, and w% is the mass fraction of PMP or PP in blends. The values of ▵H f* for PMP and PP are 117.2 and 209.00 J/g, respectively. 21,22 The calculated data are listed in Table 1. It showed that the crystallinity of PMP (X c, PMP) did not show significant variation with the increase in the amount of PP in the blends until the amount of PP of the blend reached 80 wt%. This crystallinity result is in consistent with the crystallization behavior as described and discussed above. The crystallinity of PP (X c, PP) in the blends obviously increased with the increase in the amount of PP in the blends. In addition, the crystallinity of PP in the blend with 80 wt% PP was 46.05%, which was even higher than that of pure PP. This also clearly showed that a small amount of PMP could improve the degree of crystallization of PP. The above crystallinity results also indicated the addition of PP had slightly influence on the crystallization of PMP, but using PMP greatly affected the crystallization of PP.
The wide-angle x-ray diffraction (WAXD) profiles of pure PMP, PP, and PMP/PP blends are presented in Figure 3. In pure PMP, tetragonal crystal (crystal form I) formed as confirmed by the reflections at 2θ = 9.8, 17.0, 18.7, 20.9, and 21.9°. As for pure PP, the characteristic reflections at 2θ = 14.05, 16.94, 18.48, 21.09, and 21.84°indicated the presence of the typical crystal form of α-PP. In the blends with 5 and 10 wt% PP, there was no peaks corresponding to PP crystals, so it is suspected that in these two samples, the crystallinity of PP might be too low to be detected. The blend with 20 wt% PP showed reflection peaks at 14.05 and 21.84° belonging to PP crystals. This showed that monoclinic α-PP crystal stilled formed in the blends as in the pure PP, even thought the T c of the blends was much lower than that of the pure PP (i.e. 50–85°C vs. 120°C). Figure 3 also showed that with the increase in the amount of PP in the blends, the characteristic peaks of the PP crystal became larger, while that of the PMP crystal became smaller. But even in the blend with 80 wt% PP, the peak at 9.8° belonging to PMP was still very clear. This peak confirmed the formation of tetragonal PMP crystal (i.e. crystal form I) in the blends as in the pure PMP even thought the crystallization of PMP was greatly postponed in the blends due to the presence of large amount of PP as mentioned earlier. From the above information, one can see that even though some PMP/PP blends showed different crystallization behavior from pure PP or PMP materials, the same types of PP or PMP crystals were formed in the blends as in the pure PP or PMP.
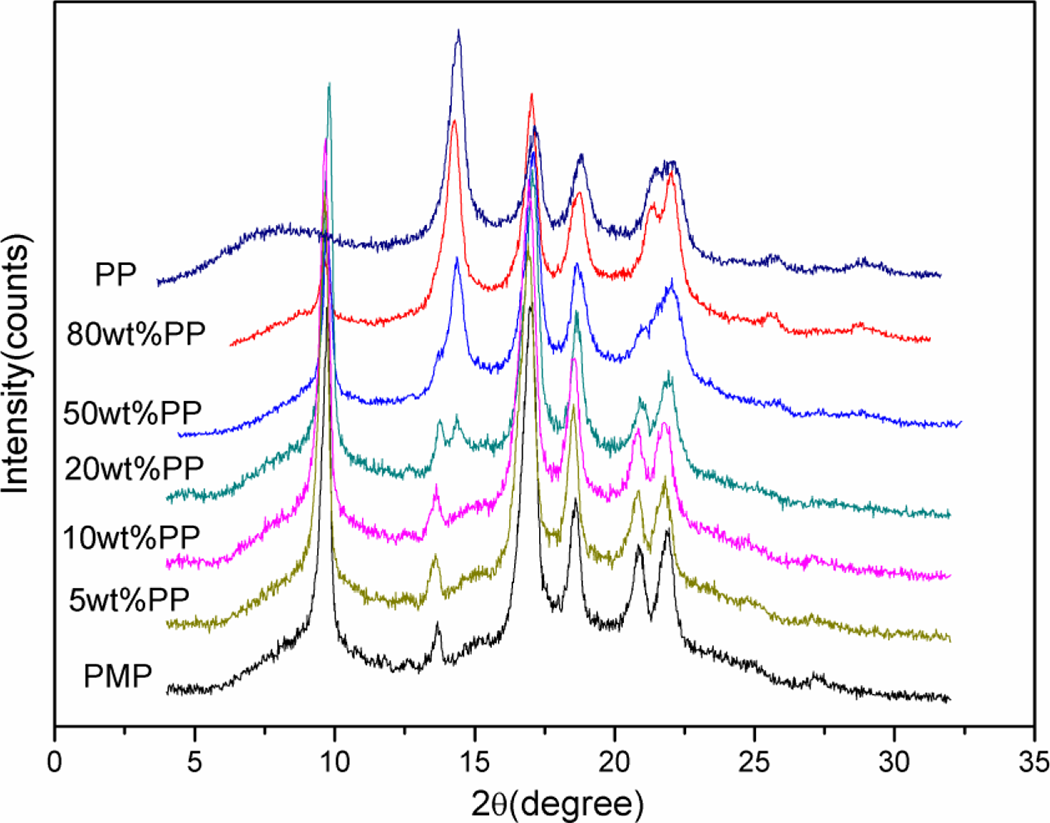
WAXD profiles of pure PMP, PP, and PMP/PP blends. PMP: poly(4-methyl-1-pentene); PP: polypropylene; WAXD: wide-angle x-ray diffraction.
Isothermal crystallization kinetics analysis of PMP in PMP/PP blends
The isothermal crystallization process is analyzed by Avrami equation as follows


where n is the Avrami exponent, and it is associated with nucleation mechanism and the crystal growth dimensions; K is the isothermal crystallization rate parameter; X(t), the normalized crystallinity at time t, is obtained with the following equation

where Q(t) is the heat flow rate obtained directly from the area of the exothermic DSC peak at time t. The value of the final crystallinity
Figure 4 shows the plots obtained regarding the isothermal crystallization of pure PMP and also PMP in the blend with 20 wt% PP at various T cs of 223, 222, 221, and 220°C: the DSC exotherm plots, the normalized crystalinity X(t) against time t plots, and the log{−ln[1 − X(t)]} versus log t plots. The plots for other blends samples were provided as supporting information (see Supplement Figure S1). The corresponding n, K, and t 1/2 are listed in Table 2.
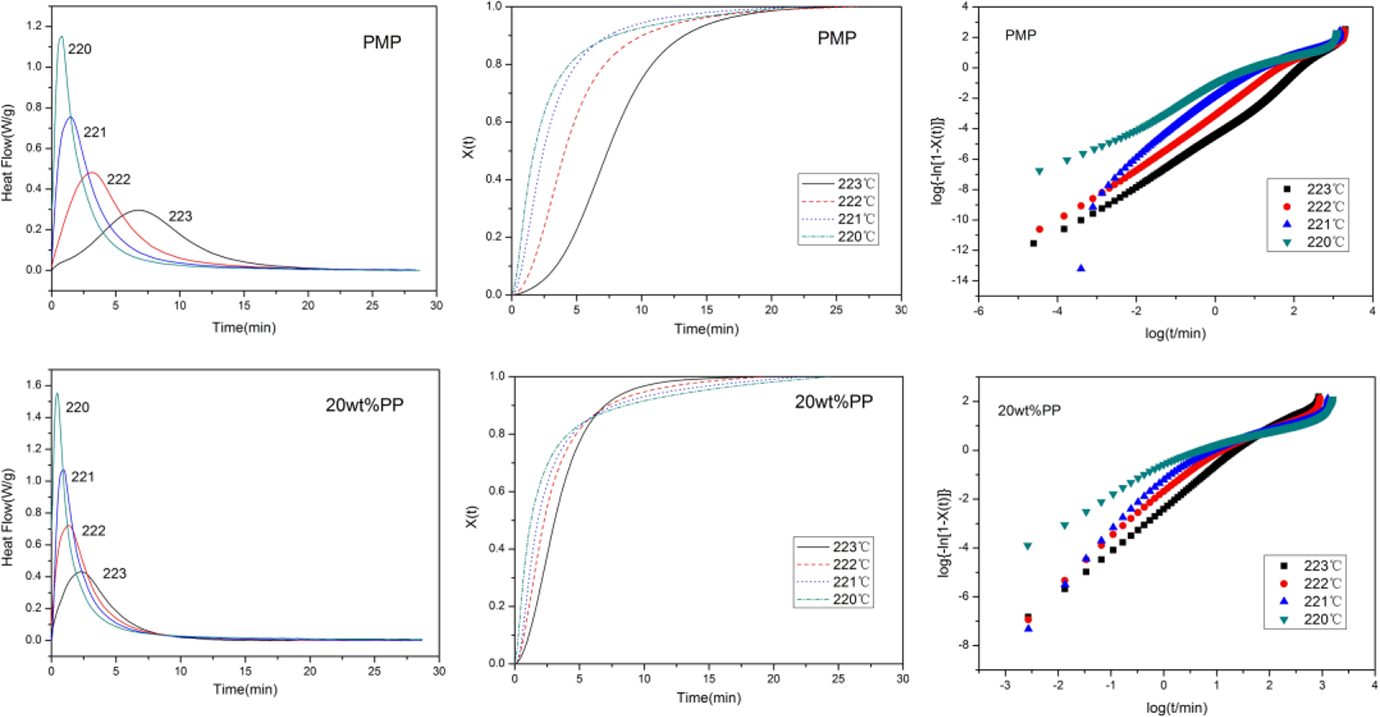
The DSC exotherm graphs, the X(t) versus time t plots, and the log{−ln[1 − X(t)]} versus log t plots for the isothermal crystallization of pure PMP and also PMP in the PP/PMP blend with 20 wt% PP obtained at various crystallization temperatures. DSC: differential scanning calorimetry; PMP: poly(4-methyl-1-pentene); PP: polypropylene.
Values of crystallization half-time t 1/2, Avrami exponent n, and crystallization rate parameter K at different crystallization temperatures T c of isothermal crystallization of PMP in pure PMP and PMP/PP blends.
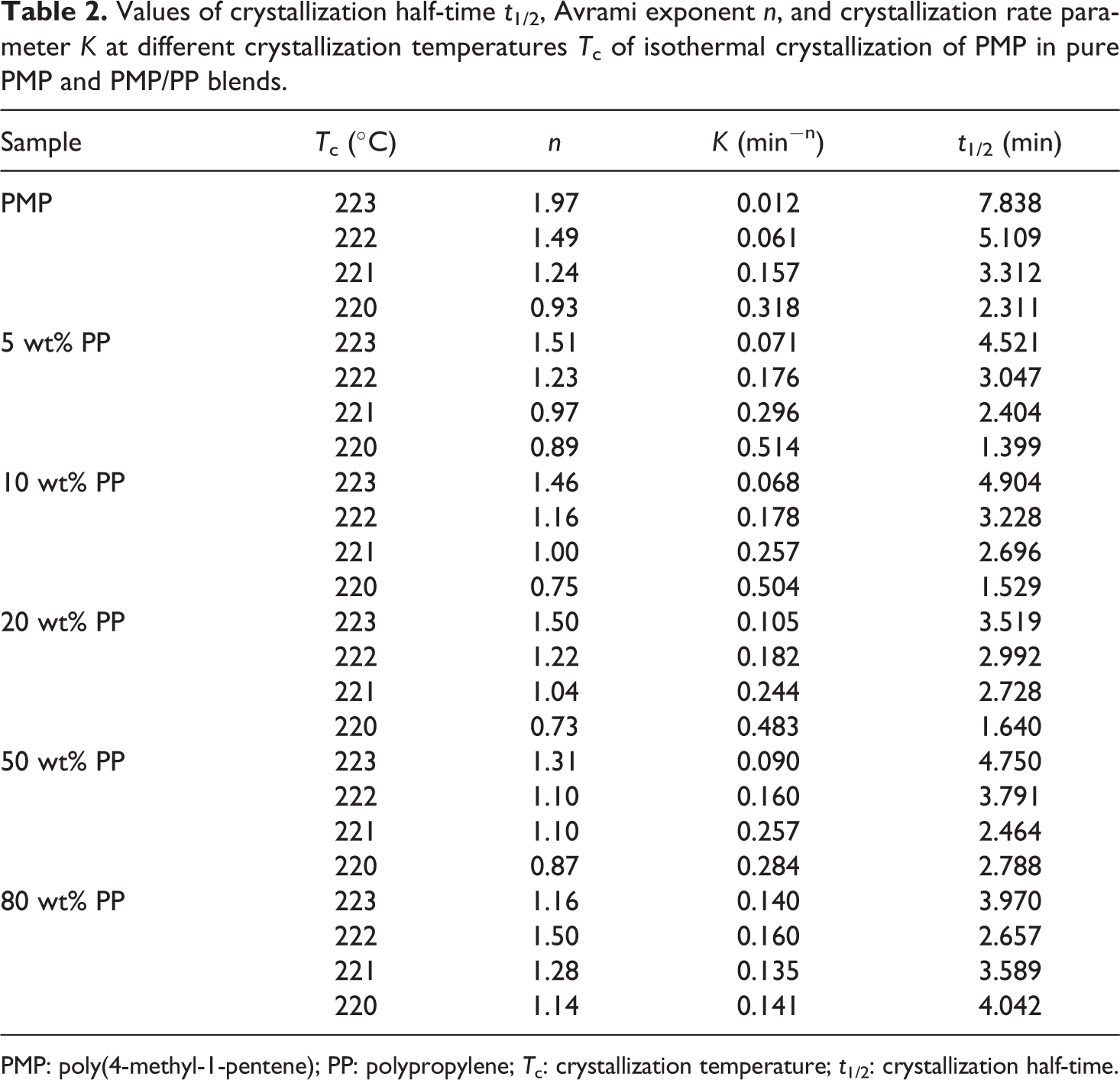
PMP: poly(4-methyl-1-pentene); PP: polypropylene; T c: crystallization temperature; t 1/2: crystallization half-time.
Avrami exponent n is associated with the nucleation type and the crystal growth dimension. For homogeneous nucleation, n = 4 for three-dimensional linear crystal growth, and n = 3 and n = 2 for two- and one-dimensional growth, respectively. For heterogeneous nucleation, n represents the dimensionality of the crystal growth. From the log{−ln[1 − X(t)]} versus log t plots, one can see that with the increase in log t, log{−ln[1 − X(t)]} first increased linearly until a certain point and then it leveled off. We used the slope of the initial linear part of the plots to calculate the Avrami exponent n. Table 3 showed that the values of n are less than 2 for all the samples, as seems inconsistent with normal value of n. But the data obtained in this research is similar as that reported by Silvestre et al. 23 and Fatou. 24 In addition, it is known that fractional values of n could be obtained especially at long crystallization times, due to the method used for analyzing the experimental data. 25 Anyway, in our Avrami analysis, all the n values of PMP in the blends were smaller than that of the pure PMP. This indicated that in the presence of PP, PMP had a stronger tendency to undergo heterogeneous nucleation.
Values of crystallization half-time t1/2, Avrami exponent n and crystallization rate parameter K of isothermal crystallization of PP in PMP/PP blends at different T c.
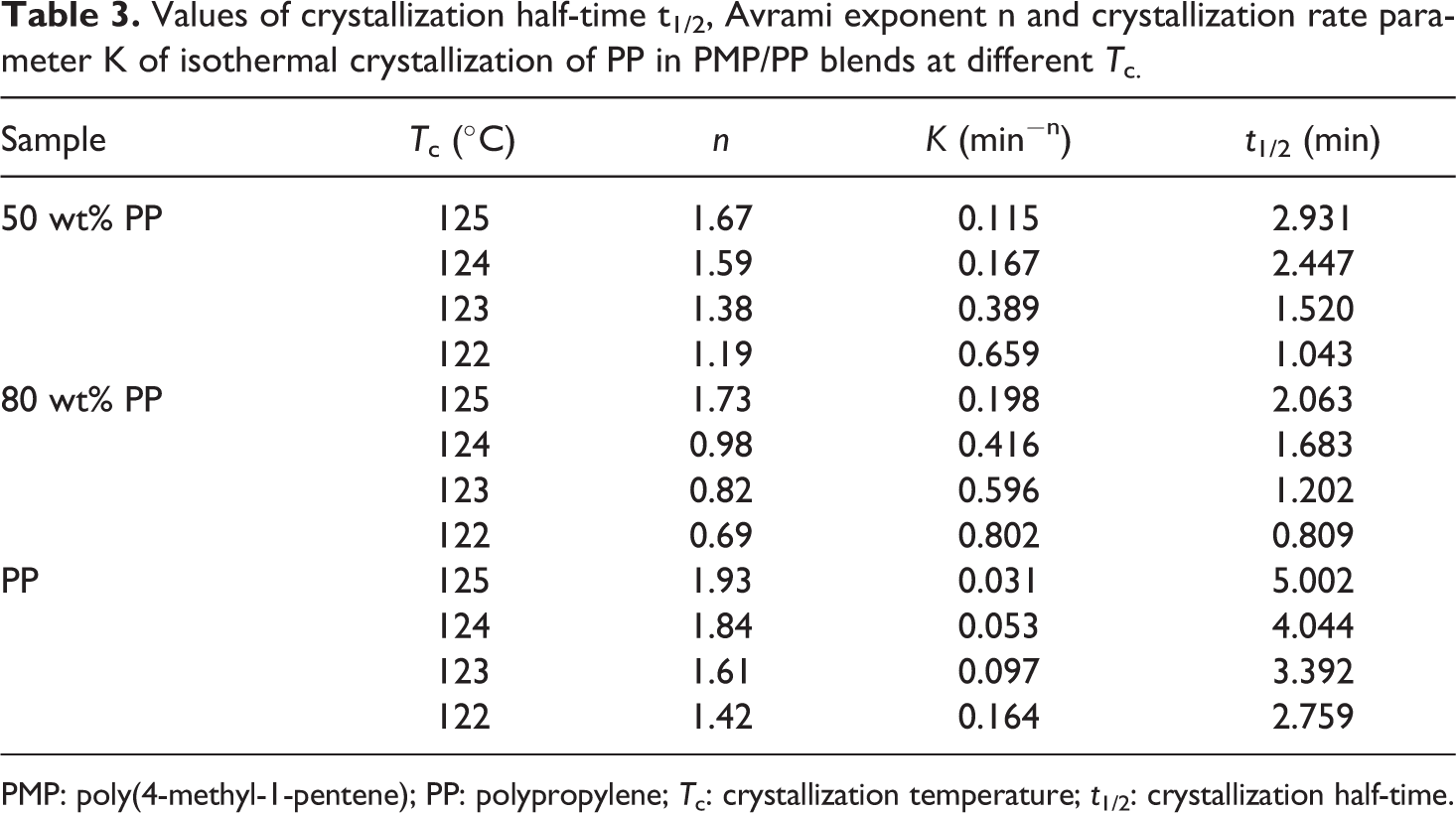
PMP: poly(4-methyl-1-pentene); PP: polypropylene; T c: crystallization temperature; t 1/2: crystallization half-time.
Table 2 showed that the isothermal crystallization rate K was affected by both the T c as well as the blends composition. With the decrease in T c, all the samples showed increase in K. This was expected, because lower temperature facilitated the crystallization of PMP. Meanwhile, the crystallization rates of PMP in all the PMP/PP blends were higher than that in pure PMP, especially at higher temperature zones. This showed that the adding of PP could promote the crystallization of PMP, although this effect was not obvious in Figure 1. The value of crystallization half-time t 1/2 is another important parameter to characterize the crystallization rate. The greater the value of t 1/2, the lower the crystallization rate. From Table 2, one can see that all the t 1/2 for PMP/PP blends were smaller than that of pure PMP. This also confirmed the positive effect of adding PP on the crystallization of PMP.
Isothermal crystallization kinetics analysis of PP in PMP/PP blends
Pure PP and PP/PMP blends with various ratios have been employed to study the isothermal crystallization of PP at various T cs of 122, 123, 124, and 125°C. However, only pure PP and blends with 50 and 80 wt% PP showed observable PP crystallization, so the results were reported below for these samples. The analysis of the isothermal crystallization kinetics analysis for PP was the same as what was used for PMP shown earlier. In Figure 5, the DSC exothermic curves, the plots of X(t) versus time t, and the plots of log{−ln[1 − X(t)]} against log t were shown for the PP isothermal crystallization under four temperatures. The calculated data of K, n, and t 1/2 are shown in Table 3.
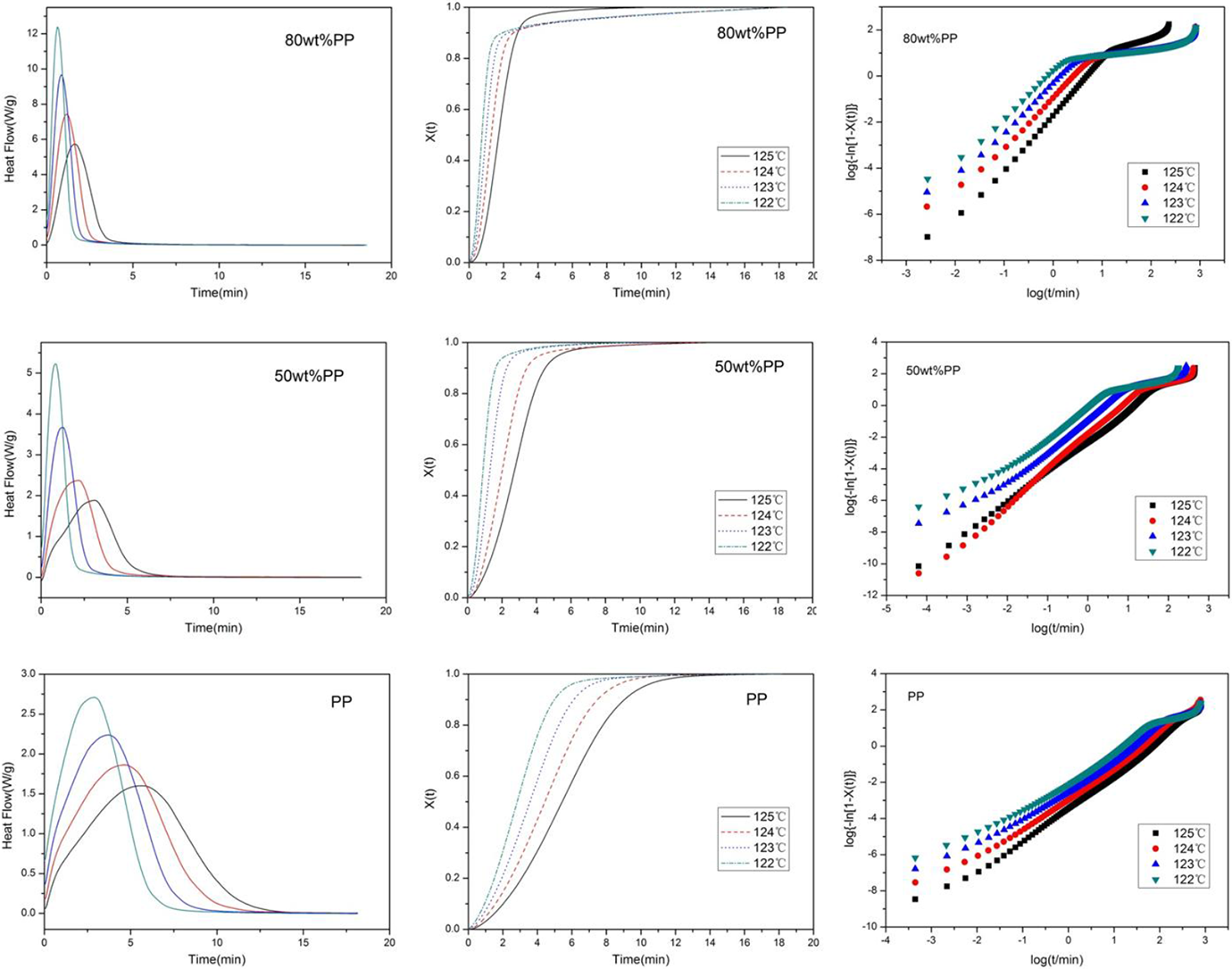
The DSC exothermic curves, the plots of X(t) against time t, and the plots of log{−ln[1 − X(t)]} against log t for the isothermally crystallization of PP in the 50 wt% PP, 80 wt% PP blends, and pure PP at various crystallization temperatures. DSC: differential scanning calorimetry; PP: polypropylene.
Figure 5 showed that the isothermal crystallization of PP was effected by the T c in the same trend as that of the crystallization of PMP reported previously. As for the values of Avrami exponent n, the PP in the blends was smaller compared to the pure PP. This showed that PP had a stronger tendency to undergo heterogeneous nucleation in the presence of PMP. The value of K and t 1/2 also showed that the crystallization rate of PP in blends is faster than in pure PP. The reason might be that a small amount of PMP crystal acted as nucleation sites that have promoted the heterogeneous nucleation of PP.
Fracture of PMP/PP blends
All of the above crystallization results showed that both PP and PMP affected the crystallization behavior of each other, as implied that they might have good compatibility. In order to further check the compatibility of PP and PMP, the blends were freeze-fractured in liquid nitrogen and the fracture surfaces were observed by SEM (see Figure 6). Except for the blend with 80 wt% PP, the fractures of the blends were smooth without obvious phase separation, and this means the compatibility of PP and PMP was good in these blends. The small white particles in the blend with 80 wt% PP were probably the PMP crystals, which acted as nucleating agents during the crystallization of PP as mentioned above.

SEM images of PMP/PP blends. PMP: poly(4-methyl-1-pentene); PP: polypropylene; SEM: scanning electron microscopy.
Mechanical properties of PMP/PP blends
The interfacial bond strength between the two phases in the blend or polymer compatibility not only affects their morphology but also their mechanical properties. 26,27 Polymer compatibility plays a key role in determining the final properties of the blends as well as their inherent properties. The mechanical properties of PMP/PP blends and pure polymers are given in Table 4. It is observed that the tensile strength, flexural strength, and flexural modulus of the blends increased with increasing PP content. When the PP content increased up to 80 wt%, these properties are comparable to those of pure PP polymer. However, the blends showed lower notched impact strength than both pure polymers. Moreover, the elongation at break presents instability for the blends. Apparently, these results did not show that PP/PMP blends had better mechanical properties, compared with the corresponding pure polymers. The detailed cause will be studied in a further work.
Mechanical properties of PMP/PP blends.
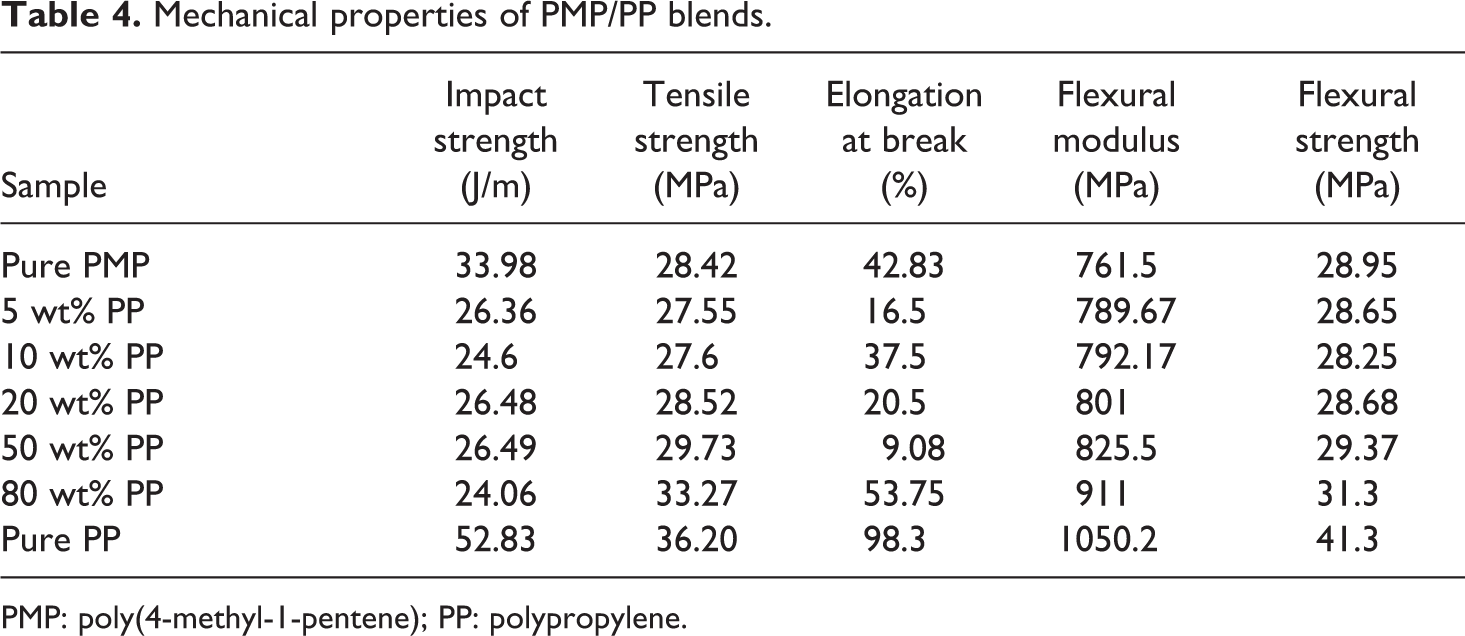
PMP: poly(4-methyl-1-pentene); PP: polypropylene.
Conclusions
The crystallization behavior and compatibility of PMP, PP and their blends were investigated by DSC, WAXD, SEM, and mechanical testing. Nonisothermal and isothermal crystallization showed that PMP would crystallize first upon cooling and act as nucleation agent for PP crystallization in the blends. When the PP content in the blends was low, PP could also facilitate the crystallization of PMP slightly. Both PMP and PP in the blends were found to remain the same crystal types as observed in pure sample. The smooth interphase boundary of the PMP and PP blends indicated a good compatibility, but the mechanical properties of the blends were not good as expected.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
