Abstract
In this work, novel optically active poly(amide-imide)/zinc oxide bionanocomposites (PAI/ZnO BNCs) containing
Introduction
In recent years, polymer bionanocomposites (BNCs) have attracted wide research interest. BNCs have a number of potential applications depending upon their synthesis techniques and the nature of the inorganic materials. The ratio between the metal oxide and the pure polymer in the composite controls the properties of these BNCs. 1 –5 Among the inorganic fillers, zinc oxide (ZnO) NPs have a special place because of good stability, high refractive index, and ultraviolet (UV) resistance and its remarkable application potential in photocatalysts, 6 solar cells, 7 chemical sensors, 8,9 piezoelectric transducers, 10 and bioimaging. 11–13 ZnO nanoparticles (NPs) were combined with polystyrene, 14 polyamide, 15 poly(amide-imide) (PAI), 16 polyacrylonitrile, 17 polyacrylate, 18 and so on. The yielded composite materials present a lot of excellent performance when compared with the pure polymers. 19
PAIs are a class of polymers with good compromise between thermal stability and processability.
20,21
They possess desirable characteristic for a variety of applications as they retain good mechanical properties at high temperature when compared with the properties of aromatic polyamides and polyimides.
22
PAIs containing
There are a variety of factors affecting the properties of BNCs, such as the particle size, distribution of particle, and filler content. The bond strength between inorganic particles and pure polymer should be improved, which is usually repaired by the type of dispersion aid or coupling agent used. 29 –32 There are different processing methods, sol–gel and molecular capping, that have been employed to form finely dispersed ZnO NPs in either organic or inorganic matrix. Ultrasonic irradiation is a moderately new but quite well-established method, which has been commonly used in preparing nanocomposites. 33 –35 Ultrasonic wave scattered modified nanostructure ZnO particles in the polymer matrix.
In the present investigation, we wish to report the synthesis and characterization of optically active and potentially biodegradable PAI, which was prepared by polycondensation reaction of N,N′-(pyromellitoyl)-bis-valine diacid with 3,5-diamino-N-(thiazol-2-yl)benzamide. In order to prevent agglomeration of NPs and improve the dispersion of ZnO NPs, the surface of NPs was treated with coupling agent of γ-aminopropyltriethoxysilane (KH550) to introduce organic functional groups on the surface of ZnO. Then novel optically active PAI/ZnO BNCs were synthesized under ultrasonic irradiation conditions. The resulting novel BNCs are characterized by numerous techniques.
Experimental
Materials
All chemicals used were of analytical grade purchased from Fluka Chemical Co. (Buchs, Switzerland), Aldrich Chemical Co. (Milwaukee, Wisconsin, USA), Riedel-deHaen AG (Seelze, Germany), and Merck (Darmstadt, Germany) unless otherwise stated. 2-Aminothiazole, 3,5-dinitrobenzoylchloride, acetone, hydrazine monohydrate, ferric chloride, and propylene oxide from Merck were used for the synthesis of mediators. The silane coupling agent (KH550) was obtained from Merck Chemical. Nanosized ZnO powder was purchased from Neutrino (Tehran, Iran) with an average particle size of 25–30 nm.
Measurements
Proton nuclear magnetic resonance ( 1 H NMR; 400 MHz) and carbon-nuclear magnetic resonance ( 13 C NMR; 125 MHz) spectra were recorded in deuterated dimethyl sulfoxide (DMSO-d 6) solution using a Bruker Avance 400 instrument (Rheinstetten, Germany). Proton resonances are designated as singlet (s), doublet (d), and multiplet (m). Fourier transform infrared (FT-IR) spectra were recorded on a 400D IR spectrophotometer (Tokyo, Japan). The vibrational transition frequencies are reported in wave numbers (cm−1). Band intensities are assigned as weak (w), medium (m), and strong (s). Thermogravimetric analysis (TGA) data were taken on PerkinElmer TGA analyzer in a nitrogen atmosphere at a heating rate of 20°C min−1. The x-ray diffraction (XRD) patterns were recorded by employing a Philips X’PERT MPD diffractometer (Eindhoven, Netherland) (Cu Kα radiation: k = 0.154056 nm at 40 kV and 30 mA) over the 2 h range of 20–80° at a scan rate of 0.05 min- −1.The dispersion morphology of the BNCs on PAI matrix was observed using field emission scanning electron microscopy (FE-SEM, Hitachi (S-4160) (Tokyo, Japan). Transmission electron microscopy (TEM) images were recorded using a Philips CM120. UV-Visible (Vis) absorbance spectra were obtained from a Perkin UV-Vis lambda 850 spectrometer (Tokyo, Japan).
Surface functionalization of ZnO NPs
The surface of the ZnO NP was modified with KH550 through ultrasonic irradiation. Typical steps carried out are as follows: nano-ZnO was dried at 110°C in an oven for 24 h to remove the adsorbed water. Dried nano-ZnO of 0.10 g with absolute ethanol was sonicated (through an ultrasonic instrument MISONIX (Raleigh, North Carolina, USA), 100 W) in an ice bath for 15 min, then 0.04 g of KH550 was added to this mixture and sonicated for 20 min. The mixture was filtered and dried at 60°C for more than 24 h 16, 18
Monomer synthesis
N,N'-(Pyromellitoyl)-bis-
Synthesis of polymer
The PAI was prepared by the following general procedure: a mixture of aromatic diacid N,N′-(pyromellitoyl)-bis-valine (0.200 g, 4.8 × 10−4 mol), 3,5-diamino-N-(thiazol-2-yl)benzamide (0.112 g, 4.8 × 10−4 mol), and TBAB (0.58 g, 1.8 × 10−3 mol) was ground until a powder was formed. After the mixture was completely ground, it was transferred into a 25-mL, round-bottom flask and then 0.28 mL (8.9 × 10−4 mol) of triphenyl phosphate was added to the mixture and was heated until a homogeneous solution was formed. Then, the solution was stirred for 12 h at 120°C, and the viscous solution was precipitated in 10 mL of methanol. The white solid was filtered off and dried to give 0.26 g (89%) of PAI.
FT-IR (KBr, cm−1): 3500 (m, br), 3105 (w), 2910 (w), 1780 (m), 1723 (s), 1620 (m), 1522 (s), 1455 (s), 1382 (s), 1348 (s), 1317 (s), 980 (s), 965 (m), 804 (m), 756 (m), 720 (m), 680 (m), 550 (m). 1 H NMR (400 MHz, DMSO-d 6, ppm): 1.03 (d, 12H, CH3, distorted), 2.80 (m, 2H, CH), 4.64–4.65 (d, 2H, CH, J = 3.6 Hz), 6.94 (s, 1H, Ar-H), 7.49 (s, 2H, Ar–H), 7.89–7.91 (d, 1H, Ar–H, J = 6.4 Hz), 8.09–8.11 (d, 1H, Ar–H, J = 8.4), 8.34 (s, 2H, Ar–H), 10.27 (s, 2H, NH), 12.67 (s, 1H, NH). Elemental analysis calculated for C30H26N6O7S (614.60 g mol-1): C, 58.40; H, 4.33; N, 13.77; S, 5.22%; Found C, 58.42; H, 4.36; N, 13.78; S, 5.24%.
Preparation of the PAI/ZnO BNCs
The PAI/ZnO BNCs were synthesized via mixing of the 0.1 g PAI with different amounts of modified ZnO NPs (4, 8, and 12 wt%) in 20 mL of absolute ethanol followed by irradiation with high-intensity ultrasonic wave for 4 h. After irradiation, the solvent was removed and the obtained solid was dried in a vacuum at 80°C for 2 h.
Results and discussion
Surface modification of ZnO NPs
The surface of ZnO NPs was treated by a KH550 silane coupling agent. The reaction mechanism is illustrated in Figure 1. Due to the presence of OH groups on the residual surface in NPs, it is easy for ZnO NPs to significantly agglomerate in the polymer matrix. KH550 hydrolyzes to trihydroxyl compounds. The hydroxyl group on the surface of ZnO will replace the OCH3 of the KH550 to bond to it. 38–40 Si–OH of coupling agent reacts with the hydroxyl groups of ZnO surface and with Si–OH of other coupling agent and forms a covalent bond or can be in a free state and form hydrogen bond.
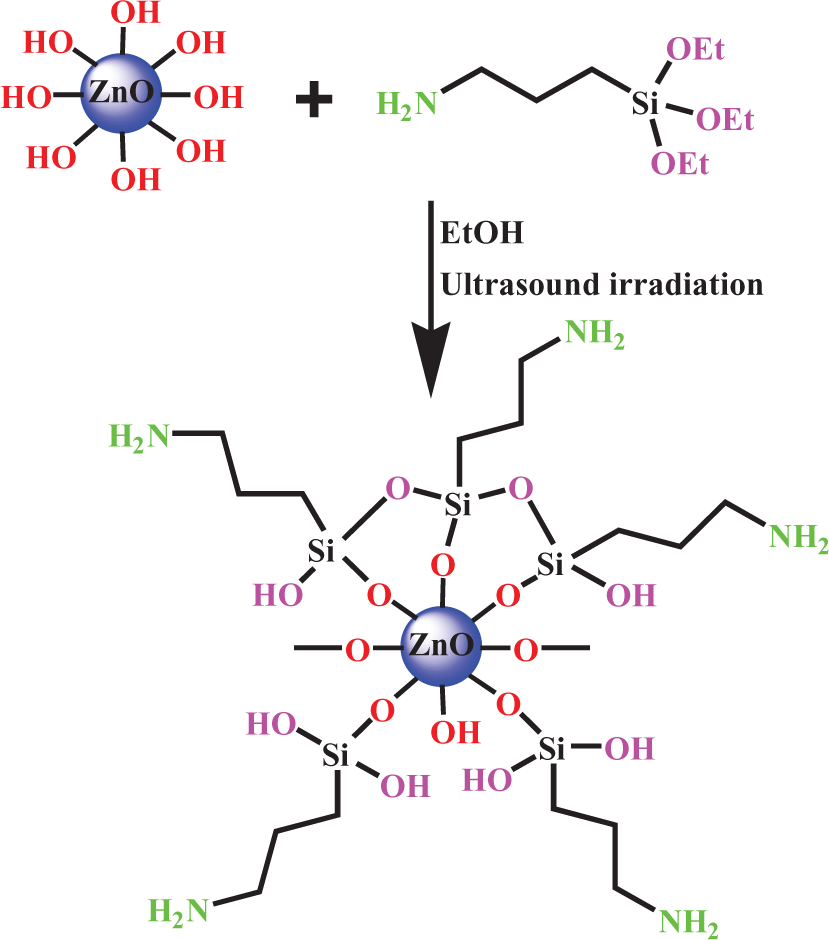
Reaction of ZnO NPs with KH550. ZnO: zinc oxide; NPs: nanoparticles; KH550: γ-aminopropyltriethoxysilane.
FT-IR spectra of the ZnO NPs, pure KH550, and ZnO–KH550 NPs are shown in Figure 2. The FT-IR spectra of ZnO (Figure 2(a)) show main absorption bands at 3450, 1580, and 1375 cm−1, which correspond to the O–H mode, and peak at 437 cm−1 is the characteristic absorption of the Zn–O bond. In the KH550 spectrum (Figure 2(b)), the stretching and bending modes of the –NH2 group were viewed at 3435 and 1575 cm−1, respectively. The peak at 920 cm−1 originates from the Si–O stretching of Si–OH groups due to the hydrolysis of KH550. It is also found that the infrared spectrum of the coupling agent (KH550)-treated ZnO NPs exhibits absorptions at 3422 (–OH and–NH2), 2973 (Si–OH), 940 (Zn–O–Si), and 850 cm−1 (Si–O–Si). Such results and appeared new bands at 2885–2958 cm−1in the FT-IR spectrum of modified ZnO NPs indicate that the silane molecules have been successfully grafted onto the surface of ZnO NPs.
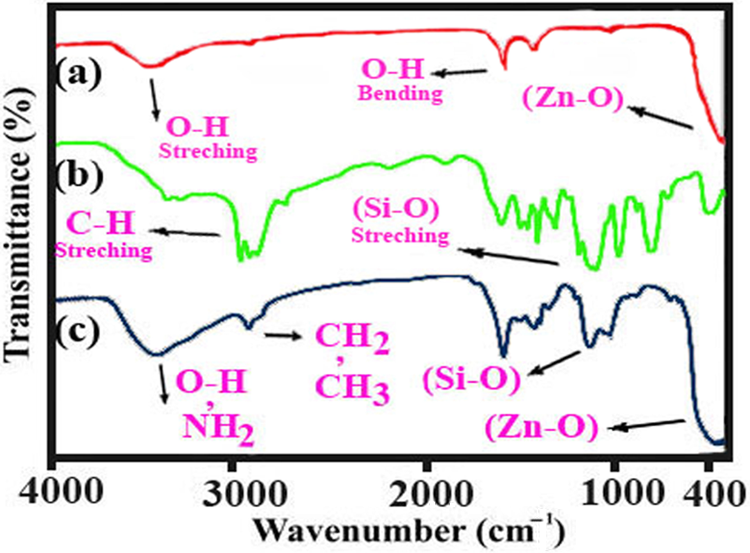
Fourier transform infrared spectra of ZnO nanoparticle and modified ZnO nanoparticle. ZnO: zinc oxide.
Polymer characterization
FT-IR study
The structures of these polymers were confirmed as PAI by means of FT-IR spectroscopy. The polymer was characterized by absorption peak for the amide N–H groups at around 3300 cm−1. Absorption of amide N–H bonds appeared at around 3320–3330 cm−1 (hydrogen band) and the peaks at 1760 and 1710 cm−1 (C=O asymmetric and symmetric, respectively, imide group) and 1620 cm−1 (C=O, amide) confirm the presence of different carbonyl groups in the polymer chain. The absorption bands that appeared around 3015–3110 and 2800–2950 cm−1 are related to the corresponding aromatic and aliphatic C–H stretching vibration, respectively. Strong absorptions at 1384 and 718–725 cm−1 show the presence of the imide heterocyclic ring in this polymer.
1 H NMR study
In the 1 H NMR spectrum of the PAI, appearance of the N–H protons of amide groups at 12.77 and 10.30 as two singlet peaks, respectively, indicates the presence of amide groups in the polymer’s side chain as well as main chain (Figure 3). The resonance of aromatic protons appeared in the range of 6.94–8.92 ppm. The proton of the chiral center appeared as a doublet at 4.82 ppm. The resonance of the CH protons of PAI appeared as multiples at 1.89 ppm.
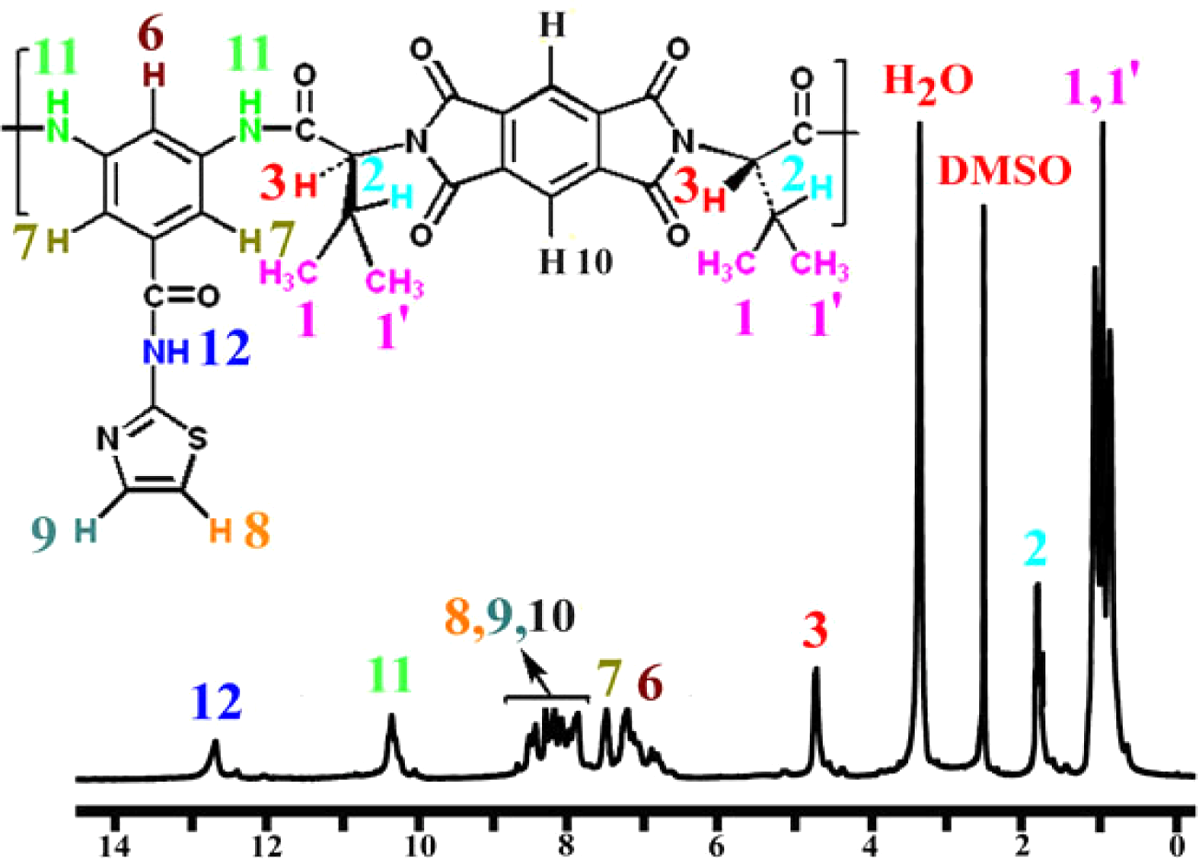
Proton nuclear magnetic resonance (400 MHz) spectrum of PAI in DMSO-d 6 at RT. RT: room temperature; PAI: poly(amide-imide); DMSO-d 6: deuterated dimethyl sulfoxide-d 6.
Characterization of PAI/ZnO BNCs
FT-IR analysis
The procedure for the preparation of PAI/ZnO BNCs is shown in Figure 4. The FT-IR spectra of the PAI/ZnO–KH550 NPs clearly exhibit the characteristic absorption peaks corresponding to polymeric groups. FT-IR spectrum of PAI showed the characteristic absorptions of amide and imide groups occurring around 3335, 1765, 1733, and 1683 cm−1, which are peculiar to N–H stretching, C=O stretching of imide and amide. The incorporation of nano-ZnO into PAI caused the slight changes in the intensities of absorption bands to 1727 and 1665 cm−1 as well as the formation of new absorption bands in the range of 400–700 cm−1. The peak at around 400–700 cm−1 is attributed to the zinc–oxygen stretching of ZnO. This corroborated the presence of ZnO NPs present in the PAI matrix. This indicates that there is no chemical linkage between PAI and ZnO NPs. Therefore, the comparatively weak interaction is thought to be a hydrogen bond, and also short-ranged steric and electrical interaction among active sites of ZnO and various functional groups of PAI. FT-IR spectra of PAI and BNC polymers with different amounts of ZnO NPs (4, 8, and 12 wt%) (Figure 5) show that the intensity of the Zn–O stretching band raises with an increase in the ZnO-NPs content in PAI.
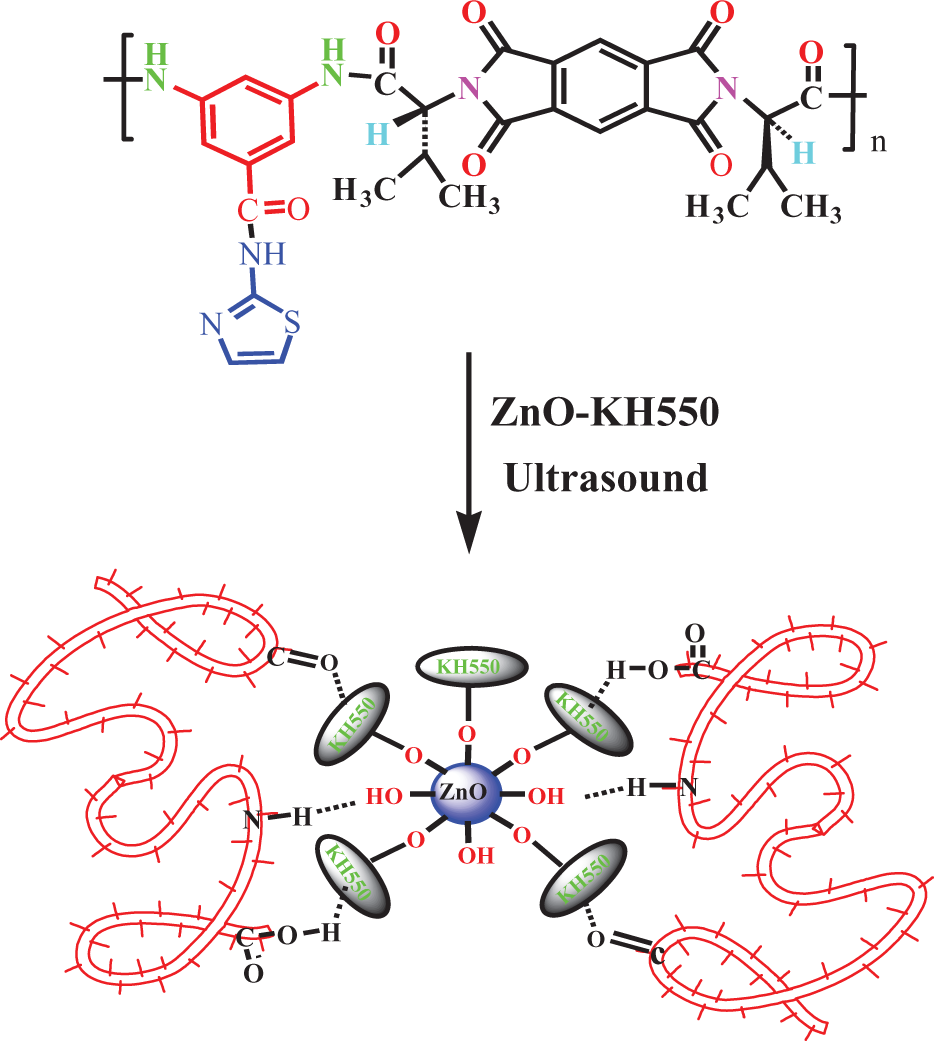
Preparation of PAI/ZnO BNCs. PAI/ZnO BNCs: poly(amide-imide)/zinc oxide bionanocomposites.
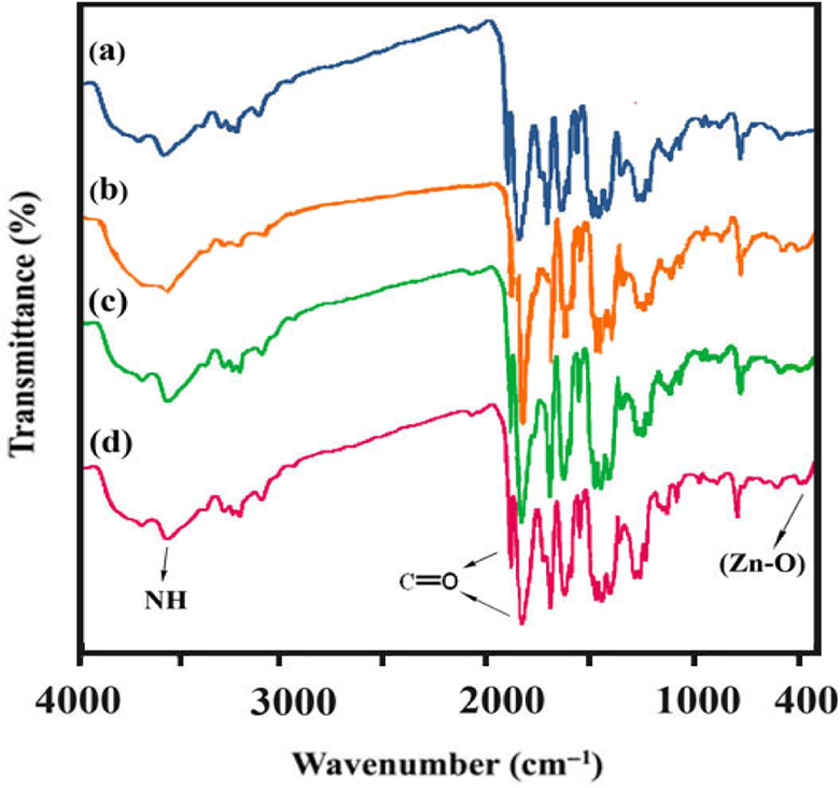
Fourier transform infrared spectrum of the PAI/ZnO-KH550 NCs: (a) PAI3, (b) PAI/ZnO (4%), (c) PAI/ZnO (8%), and (d) PAI/ZnO (12%). ZnO: zinc oxide; PAI: poly(amide-imide); KH550: γ-aminopropyltriethoxysilane; NCs: nanocomposites.
X-Ray diffraction
Figure 6 displays the XRD patterns of PAI (a), PAI/ZnO (8%) (b), and PAI/ZnO (12%) (c). The XRD pattern of BNCs shows the characteristic peaks of PAI and ZnO indicating that the crystallinity of ZnO NPs was not changed during the preparation process and the intensity of diffraction peaks increases with increasing ZnO content. The average particle size of NPs was estimated based on the Scherrer correlation of particle diameter (D), D = Kλ/β cos θ, where K is the Scherrer constant, λ the x-ray wavelength, β the peak width at half-maximum, and θ is the Bragg diffraction angle. The average crystallite size of the ZnO calculated from the width of the diffraction peak according to the Scherrer equation is approximately less than 40 nm. This is in agreement with the size of the used ZnO NPs. Pure PAI was totally amorphous in nature, which did not show any sharp diffraction peaks.
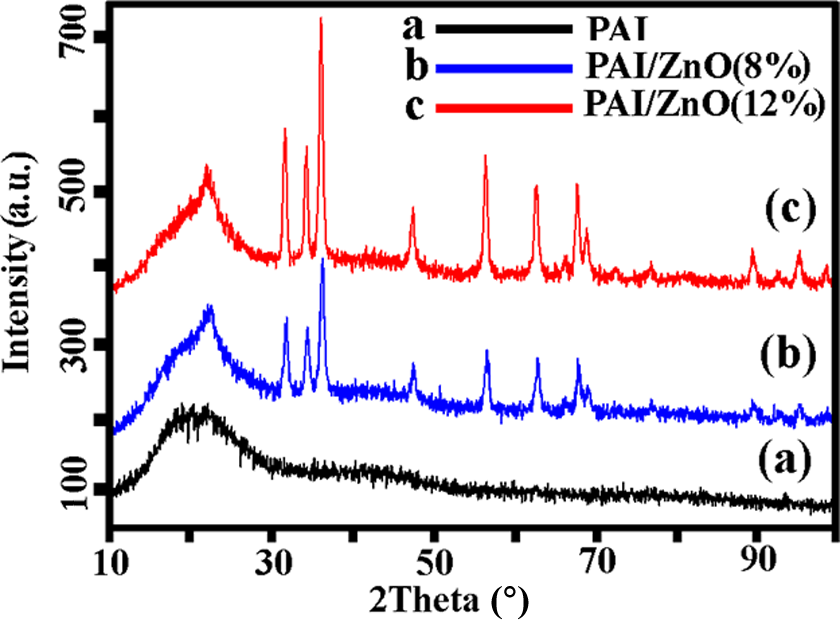
X-Ray diffraction patterns of (a) pure PAI, (b) PAI/ZnO (8%), and (c) PAI/ZnO (12%). ZnO: zinc oxide; PAI: poly(amide-imide).
UV-vis spectra
The optical properties of pure PAI, ZnO-KH550, and PAI/ZnO BNCs are shown in Figure 7. UV-vis absorption spectra of PAI/ZnO BNCs showed a maximum absorption at 376 nm. The UV shielding ability relates with the scattering and/or absorbance of ZnO NPs. The scattering property plays a major role in shielding UV irradiation. Thus, these BNCs have potential to be used as a coating to block the UV radiation, particularly between 350 and 400 nm.
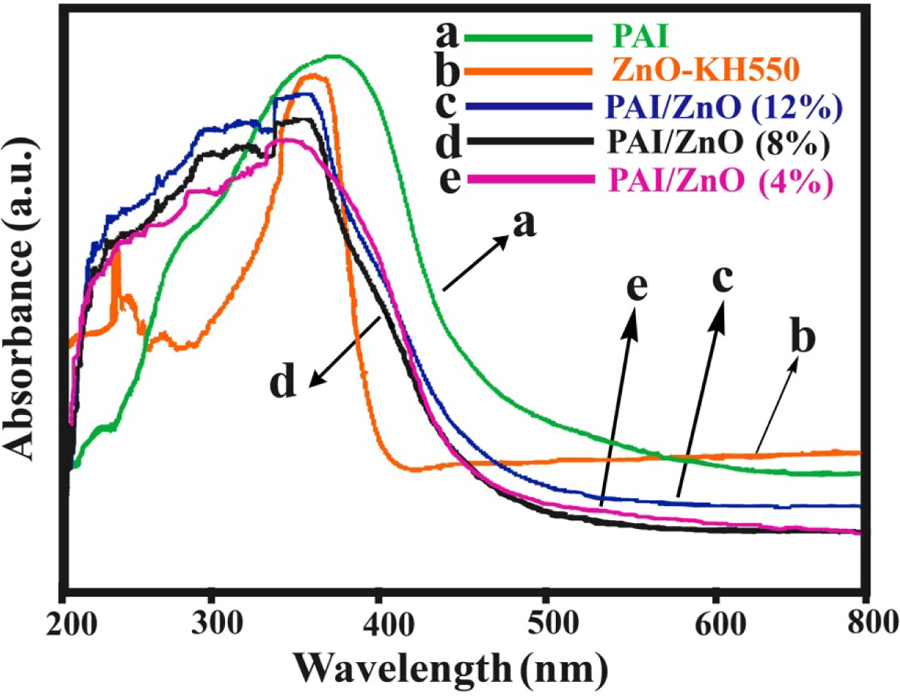
Ultraviolet–visible absorption spectra of ZnO–KH550, PAI, and PAI/ZnO BNCs. PAI/ZnO BNCs: poly(amide-imide)/zinc oxide bionanocomposites; KH550: γ-aminopropyltriethoxysilane.
Electron microscope characterization
The morphology before and after ZnO deposition on the surface of PAI polymer was confirmed by the FE-SEM. Figures 8(a) to (c) show the FE-SEM micrographs of PAI. Pure PAI have relatively spheroid nanostructured morphology, and FE-SEM pictures (Figures 8(a) to (c)) show that the polymer particles are in nanoscale and almost uniform in size. The average mean diameter of pure PAI polymer is between 70 and 88 nm. Figure 8(d) to (f) shows the FE-SEM micrographs of PAI/ZnO BNCs (12 wt%). The FE-SEM images of PAI/ZnO BNC (12%) reveal that the ZnO NPs were homogeneously dispersed in the polymer matrix and the average particle size of the NPs was in the range of 30–50 nm. The compatibility of ZnO filler with PAI matrix can be improved by treating ZnO filler with modifying agents such as KH550 and ultrasound irradiation that shows an effect on the distribution and particle size of the NPs.

Field emission scanning electron microscopic images of pure PAI (a–c) and PAI/ZnO (12%) (d–f). ZnO: zinc oxide; PAI: poly(amide-imide).
The TEM micrograph of the PAI/ZnO 12 wt% BNC shows that ZnO NPs were homogeneously dispersed in polymer matrix (Figure 9). The modified NPs might be dispersed absolutely and will combine with PAI via the H-bonding of NH2 coupling agent with –NH and C=O groups in PAI. In addition, –OH groups on the surface of ZnO NP can bond to the amide group (C=O) of PAI through interchange hydrogen bonding. The average size of the nanoscale ZnO particles is about 33 nm. The obtained results indicate that the surface modification plays a major role in dispersion of NPs.

Transmission electron micrograph of PAI/ZnO (12%). ZnO: zinc oxide; PAI: poly(amide-imide).
Thermal stability of BNCs
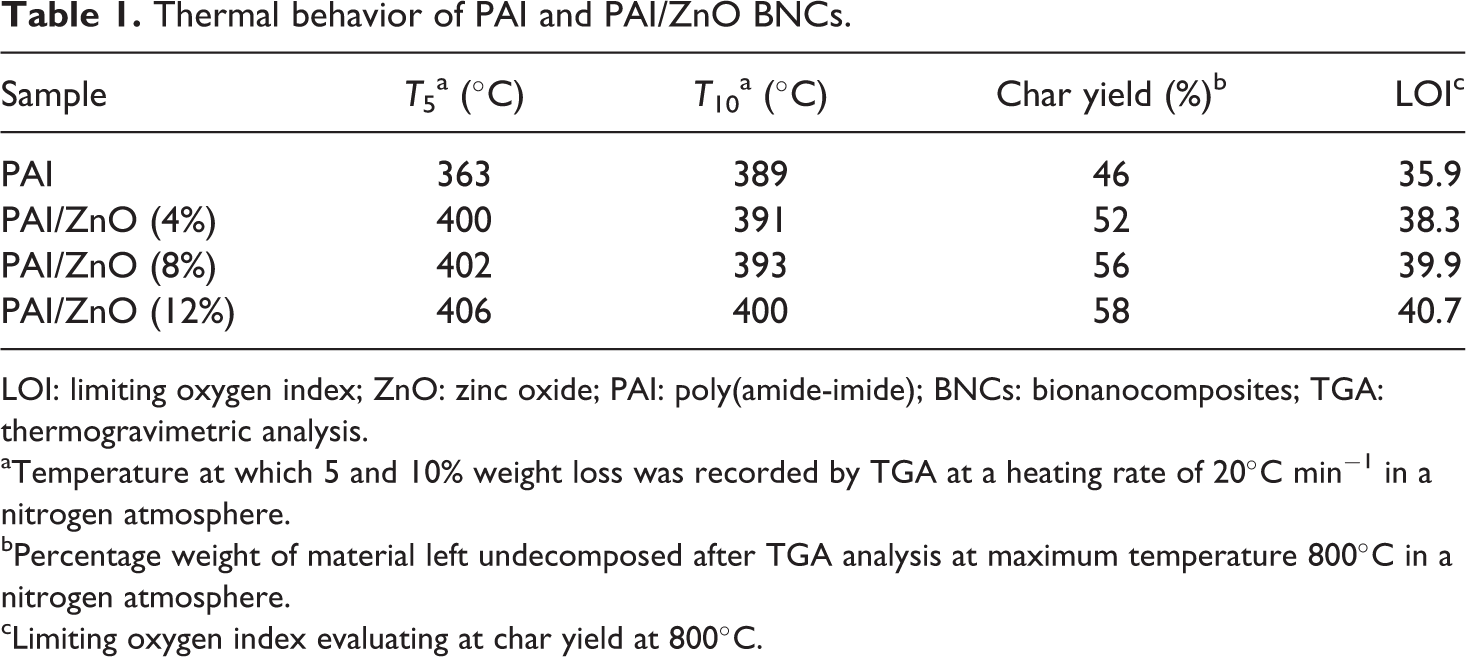
The thermal properties of the PAI and PAI/ZnO BNCs were evaluated by means of TGA in a nitrogen atmosphere at a heating rate of 20°C min−1. Table 1 shows the data for the thermal degradation of the PAI and PAI/ZnOBNCs, including the temperature at which 5% (T 5) and 10% degradation occurs (T 10), limiting oxygen index (LOI), and char yield (CR) at 800°C. LOI values were calculated based on the Van Krevelen and Hoftyzer equation. 41 LOI = 17.5 + 0.4 CR. As shown in Figure 10, the initial decomposition temperatures of the pure PAI and PAI/ZnO BNCs are about 390°C. The CR values of BNCs have higher thermal stability than that of pure PAI at 800°C. Increase in the thermal stability in BNCs is attributed to the high heat resistance exerted by the ZnO, because the ZnO NPs have high thermal stability; so coupling of ZnO NPs can improve the thermal stability of the BNCs.
Thermal behavior of PAI and PAI/ZnO BNCs.
LOI: limiting oxygen index; ZnO: zinc oxide; PAI: poly(amide-imide); BNCs: bionanocomposites; TGA: thermogravimetric analysis.
aTemperature at which 5 and 10% weight loss was recorded by TGA at a heating rate of 20°C min−1 in a nitrogen atmosphere.
bPercentage weight of material left undecomposed after TGA analysis at maximum temperature 800°C in a nitrogen atmosphere.
cLimiting oxygen index evaluating at char yield at 800°C.
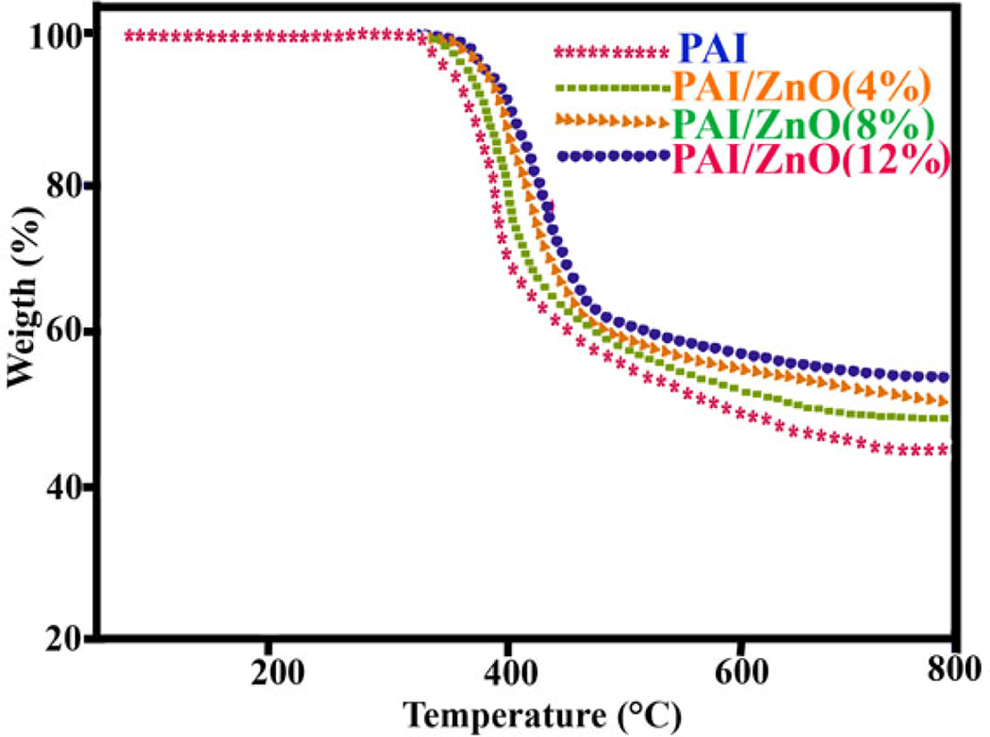
Thermogravimetric analysis thermograms of PAI and PAI/ZnO BNCs with different ZnO content. ZnO: zinc oxide; PAI: poly(amide-imide); BNCs: bionanocomposites.
Conclusions
PAI/ZnO BNCs were prepared by irradiation with high-intensity ultrasonic wave. FT-IR spectral measurements allow us to conclude that polymer nanocomposite has formed and also there is intermolecular interaction between the PAI and ZnO NPs. The thermal stability of the PAI/ZnO BNCs has increased compared with that of pure PAI. Both FE-SEM and TEM pictures show that the ZnO NPs were dispersed homogeneously in the polymer matrix in nanoscales. The UV-vis results indicate that the resulting hybrid BNCs could be potentially applied in a coating technology to shield against UV light.
Footnotes
Funding
This work was financially supported by the Research Affairs Division Isfahan University of Technology (IUT) and Center of Excellency in Sensors and Green Chemistry Research (IUT).
