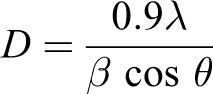Abstract
In the present investigation, novel optically active poly(amid–imide) (PAI)/organoclay (OC)/titanium dioxide (TiO2) bionanocomposite (BNC) films were prepared by solution intercalation method. These BNCs were synthesized by blending the chiral OC and modified TiO2 nanoparticles (NPs) with the PAI solution. An optically active PAI containing
Introduction
In the area of nanotechnology, polymer-based nanocomposites (NCs) represent one of the most active fields in the polymer industry because of the unique properties that can be achieved with the polymer. 1,2 For this purpose, many different types of fillers including materials such as organoclay (OC) and titanium dioxide (TiO2) nanoparticles (NPs) have been added to polymers to improve the properties of the final product. 3 –5 Bionanocomposites (BNCs) are another type of NCs formed by the combination of polymers coming from natural resources (biopolymers) or synthetic biofunctional polymers and inorganic/organic solids at nanometric scale. 6,7 The properties of polymer NCs have been extensively studied and many properties performances such as flame retardation, enhanced barrier properties, ablation resistance, gas permeability and so on were enhanced. 8 –12
Clay minerals are an important class of nanosized materials used as fillers in polymer NCs. 13,14 Smectite clays (e.g. montmorillonite (MMT)) belongs to the family structure that is called 2:1 phyllosilicates. This type of clay minerals is very important because they have special properties such as cation-exchange capacity and swelling ability. 15,16 For the most polymers, owing to their hydrophobic character, the MMT must be modified with a surfactant (e.g. quaternary ammonium salts) in order to make the MMT hydrophobic to permit it to interact with the polymer. 17 Organo-MMT are typically synthesized by ion-exchange reactions involving the exchange of organic cationic surfactants for the interlayer cations (Na+ or Ca2+), which also causes an increase in the interlayer space. 18,19 The mechanical properties and thermal stability of the NCs depend on the ratio of intercalated/exfoliated nanostructures. 20 –22 In fact, NCs containing a combination of partly intercalated and mainly exfoliated nanostructures exhibit higher thermal stability as compared to mainly exfoliated NCs and the exfoliated systems give better mechanical properties than intercalated ones. 23,24
In inorganic NPs, TiO2 is one of the most potential materials in research and application fields because it is nontoxic, chemically inert, low cost, corrosion resistant and has high refractive index, ultraviolet filtration capacity and high hardness. 25,26 TiO2 NP is widely applied as nanofiller to polymers due to their improved physical and mechanical properties without loss of ease processing and often the ductile nature of neat polymer. 27
Poly(amide–imide) (PAI) is one of the most successful classes of copolyimides that contains both amide and heterocyclic imide structures along the main chain of the polymer backbone and thus possesses excellent thermal stability balanced with processability. 28,29 PAIs bring the higher mechanical properties associated with amide groups and the high thermal stability determined by imide rings together. 30 Generally, PAIs are a good matrix candidate for hybrid materials. 31 In the synthesis of amino acids-based PAIs, the resulting polymer is optically active and polymers with chiral structures are biologically important. 32
In recent years, ionic liquids have been considered as environmentally friendly solvents because they have negligible vapor pressure and other properties, such as non-flammability and high thermostability. 33 They can be used as a solvent in polymerization processes instead of organic solvent. In this method, the reaction is safe and green. 34,35
In the present investigation, the synergetic effect of the surface-modified TiO2 (M-TiO2) and chiral OC on PAI matrix for the preparation of PAI/OC/M-TiO2 bionanocomposite (POTBNC) was investigated. These BNC films were prepared by the solution intercalation method through blending of OC and M-TiO2 with the PAI solution for full dispersion of these NPs throughout the matrix. Chiral OC was synthesized by a cation-exchange reaction of Cloisite Na+ and ammonium salt of
Experimental
Materials
Pyromellitic dianhydride (PMDA), TBAB and
Equipments
A Jasco-680 Fourier transform infrared (FT-IR) spectroscope (Tokyo, Japan) was employed to examine the chemical bonds on the polymer and POTBNCs. FT-IR spectra of the hybrid films and polymer were recorded at a resolution of 4 cm−1, and they were scanned at a wave number ranging from 400–4000 cm−1. Vibration bands were reported as wave number (per centimeter). FT-IR spectra of Cloisite Na+, OC and M-TiO2 were obtained as potassium bromide as pellets. Inherent viscosities were measured by a standard procedure using a Cannon-Fenske routine viscometer (Mainz, Germany) at the concentration of 0.5 g dL−1 at 25°C. Specific rotations were measured by a Jasco Polarimeter (Tokyo, Japan). Thermogravimetric analysis (TGA) was performed with a STA503 win TA instrument (Hüllhorst, Germany) in a nitrogen (N2) atmosphere at a rate of 20°C min−1. X-Ray diffraction (XRD) patterns were recorded using CuKα radiation on a Bruker D8 Advace, (Karlsruhe, Germany) diffractometer operating at a current of 100 mA and a voltage of 45 kV. The diffractograms were measured for 2θ, in the range of 1.3–80°, using CuKα incident beam (λ = 1.51418 Å). Bragg’s law nλ = 2d sin θ was used to compute the d-spacing. The dispersion morphology of the NPs on PAI matrix was observed using field emission scanning electron microscopy (FE-SEM; HITACHI S-4160) (Tokyo, Japan). Transmission electron microscopic (TEM) images were obtained using a Philips CM120 (Eindhoven, The Netherland) microscope with an accelerating voltage of 100 kV.
Synthesis of chiral OC
The OC was prepared by a cation-exchange method, according to our previous study. 36
Surface modification of TiO2 NPs
The surface of TiO2 NPs was functionalized by treatment with KH550, according to the reported procedure. 37
Synthesis of chiral diacid
N,N′-(Pyromellitoyl)-bis-
Synthesis of PAI and amine end-capped PAI
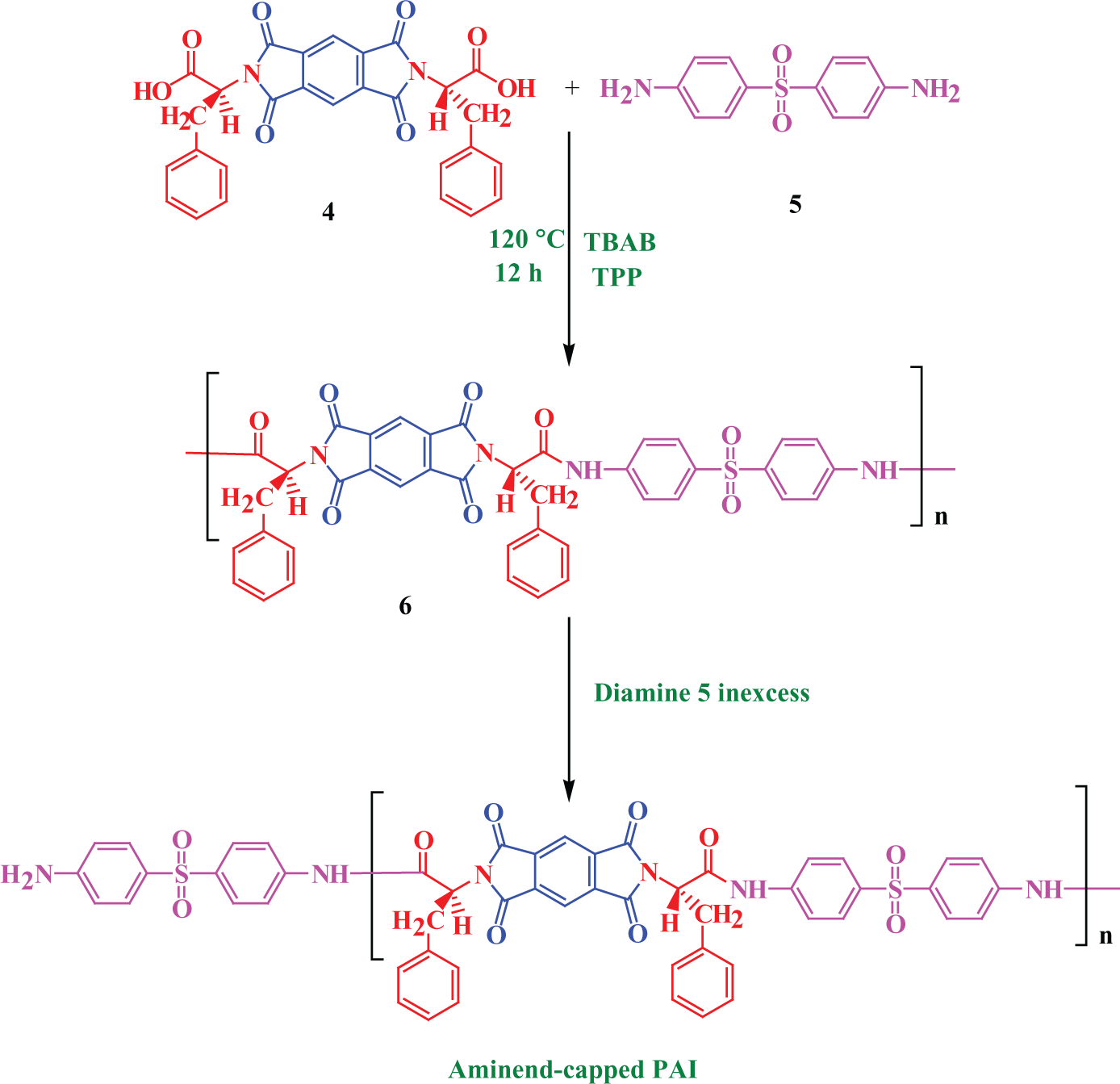
An optically active PAI was prepared by the following procedure: a mixture of 0.50 g (9.76 × 10−4 mol) of diacid 4 and 1.26 g (3.91 × 10−3 mol) of TBAB was ground until a powder was formed and then it was transferred into a 25-mL round-bottom flask and 0.24 g (9.66 × 10−4 mol) of diamine 5 was added to the mixture. It was heated until a homogeneous solution was formed and then, triphenyl phosphite (TPP; 0.49 mL, 3.89 × 10−3 mol) was added. After that the solution was stirred for 12 h at 120°C to obtain a viscous solution. The viscous solution was poured into 20 mL of methanol and the precipitated solid was filtered off and washed with hot water and then dried at 80°C for 6 h under vacuum to yield 0.66 g (89%) of the solid PAI6, and the inherent viscosity of the PAI was 0.31 dL g−1 and also the specific rotation was measured (
An amine end-capped PAI chain was produced by adding excess (1%) amount of diamine 5 near the end of the polymerization reaction.
Preparation of the new optically active POTBNCs
The novel BNCs were prepared by mixing the appropriate amounts of the PAI and OC and M-TiO2 in DMAc as a solvent in a flask for a particular concentration. At first, 0.2 g (0.2 mmol) of PAI was dissolved in 5 mL of DMAc in a 25-mL round-bottomed flask. Then 0.01 g (5 wt%) of OC was added to the solution and the mixture was agitated to high-speed stirring at 80°C for 2 h, 60°C for 2 h and then at 40°C for 20 h for uniform dispersion of OC platelets in the polymer matrix. In the next step, different percentages of M-TiO2 (5, 10 and 15 wt%) were added to the solution in an analogous procedure. Finally, the resulting mixtures were cast in glass petri dishes and kept at room temperature until the thin POTBNC films of uniform thickness were obtained.
Results and discussion
Synthesis of optically active aromatic PAI
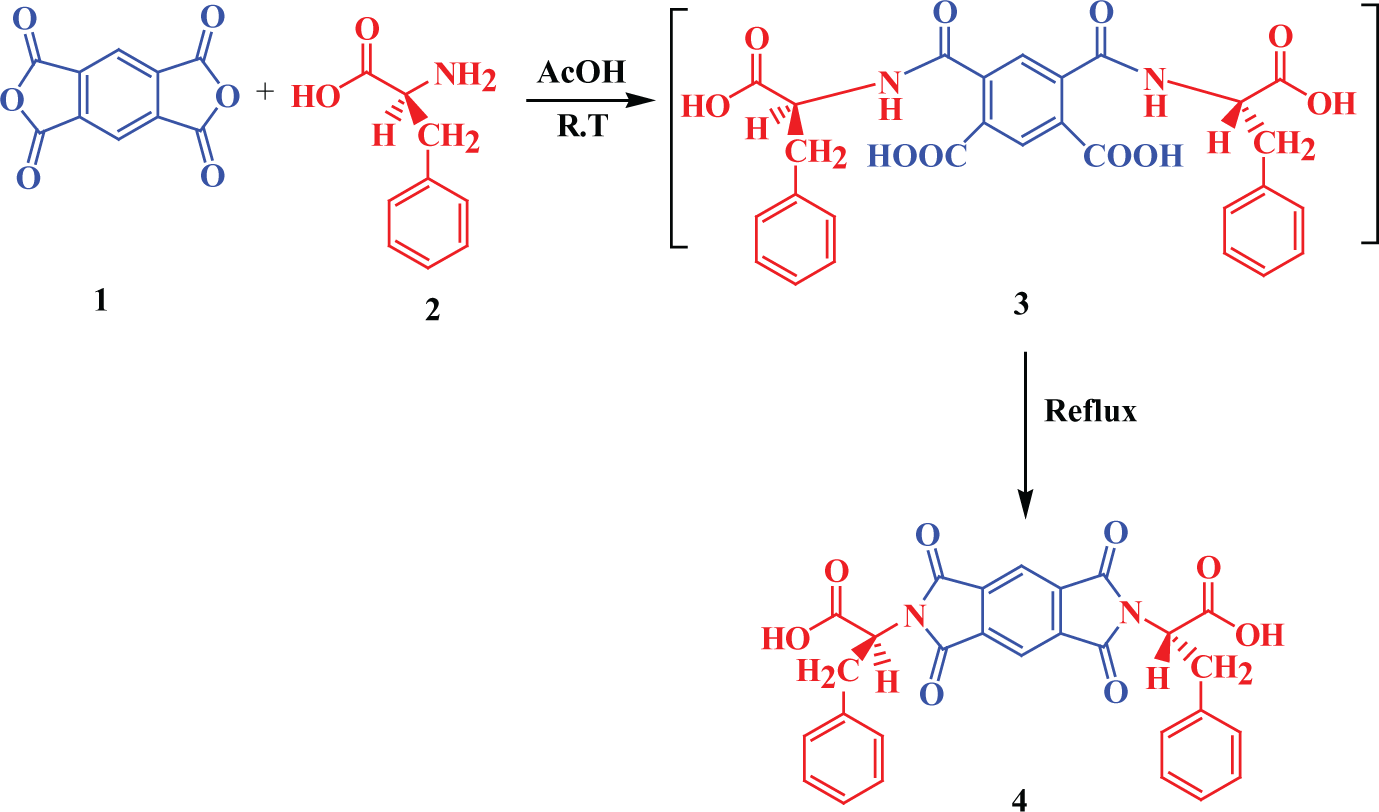
Molten TBAB was used as a green solvent for the formation of optically active PAI by the direct polymerization reaction of chiral diacid 4 with diamine 5. An optically active diacid was synthesized according to the reported procedure as shown in Figure 1. In this study, the polymerization reaction was performed under conventional heating in the presence of molten TBAB and TPP and it took 12 h at 120°C (Figure 2). The inherent viscosity of the PAI under optimized condensations was 0.31 dL g−1 and the yield was 89%. The incorporation of a chiral unit into the polymer backbone was provided by measuring the specific rotations of this polymer, and the results show that this PAI is optically active with a specific rotation of
Synthesis of chiral diacid.
Preparation of optically active amine end-capped PAI. PAI: poly(amide–imide).
Preparation of POTBNCs
Positively charged safe
Preparation of the POTBNCs. POTBNC: PAI/OC/M-TiO2 bionanocomposite.
Characterization methods
FT-IR spectra
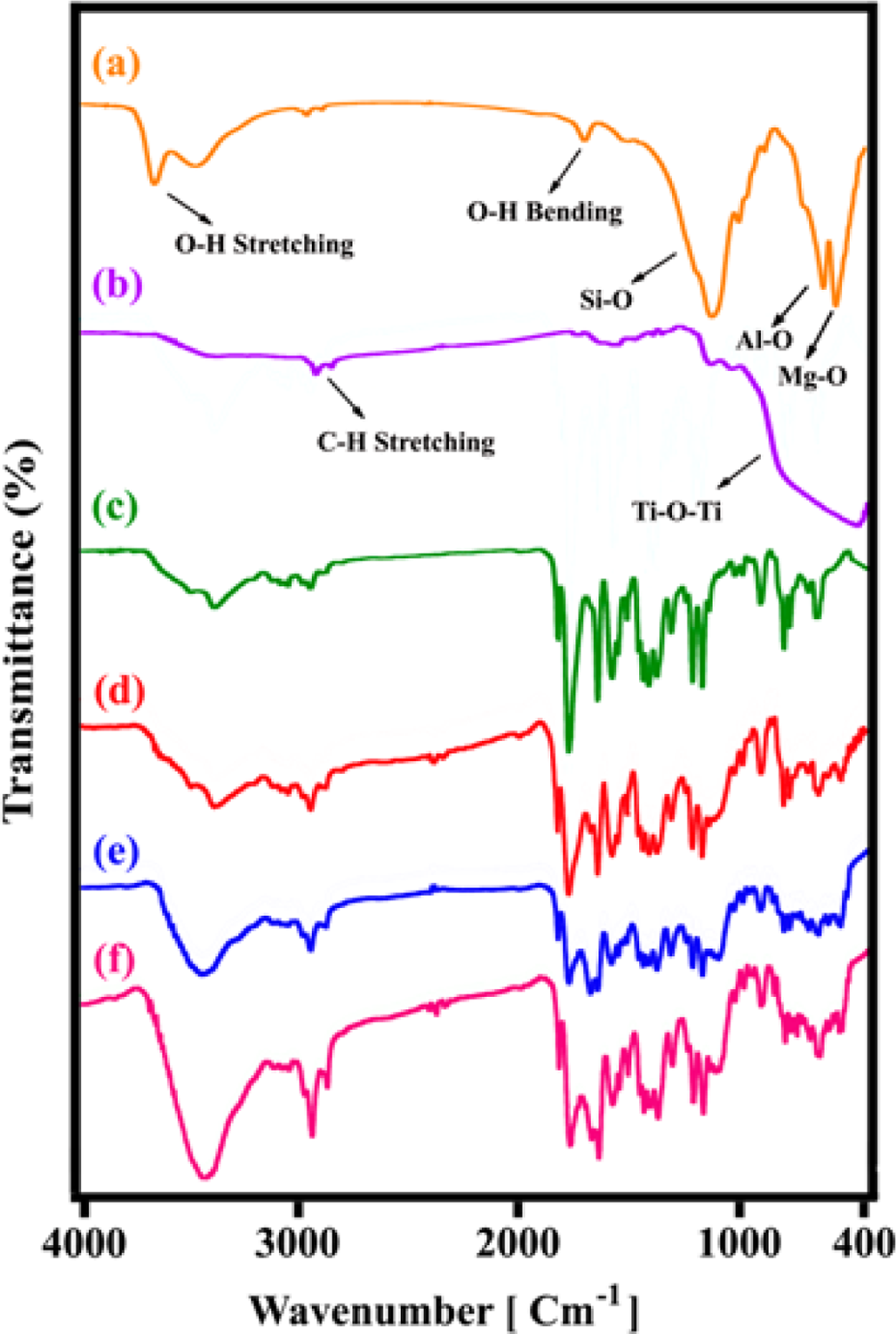
The FT-IR spectra of OC, M-TiO2, pure polymer and different percentages of POTBNCs are shown in Figure 4. In the spectrum of the OC, the absorption band at 3631 cm−1 is due to –OH stretching of Al–OH and Si–OH. Bands in the 2500–3500 cm−1 region of

FT-IR spectra of (a) OC, (b) M-TiO2, (c) PAI, (d) POTBNC 5%, (e) POTBNC 10% and (f) POTBNC 15%. PAI: poly(amide–imide); M-TiO2: surface-modified titanium dioxide; OC: organoclay; POTBNC: PAI/OC/M-TiO2 bionanocomposite; FT-IR: Fourier transform infrared.
In the FT-IR spectra of M-TiO2 (Figure 4(b)), the peak at 2853–2922 cm–1 is attributed to a CH stretching band of KH550. The band at 1116 and 1024 cm–1 can be assigned to the stretch vibration of Si–O–Si and Ti–O–Si bonds, respectively. The peak at 1725 cm−1 indicates the presence of bending vibration of the N–H bond. A broad absorption peak at 450–800 cm−1 is assigned to the Ti–O–Ti stretching band.
The FT-IR spectrum of pure PAI (Figure 4(c)) showed absorptions at around 3370 cm–1 (N–H), 3100 cm–1 (aromatic C–H stretching), 2923 cm–1 and 2876 cm–1 (CH2 stretching) and two overlapped carbonyl (amide and imide’s C=O) absorptions at 1775, 1725 and 1668 cm–1. Absorption at 1383 and 726 cm–1 shows the presence of the imide heterocycle in this polymer. Absorptions at 1249 and 1150 cm–1 were due to the sulfone moiety (SO2 stretching). These FT-IR data confirm the formation of the optically active PAI.
Figure 4(d) to (f) also shows the FT-IR spectra of POTBNCs containing 5% of OC and 5, 10 and 15 wt% of M-TiO2, respectively. As the loading of OC and M-TiO2 is increased, the intensities of NPs bands become more pronounced. From these data, it can be concluded that the POTBNCs not only have the characteristic neat PAI bands but also have a characteristic peak for OC and M-TiO2. These data, in sum, confirm the formation of the POTBNCs.
XRD patterns
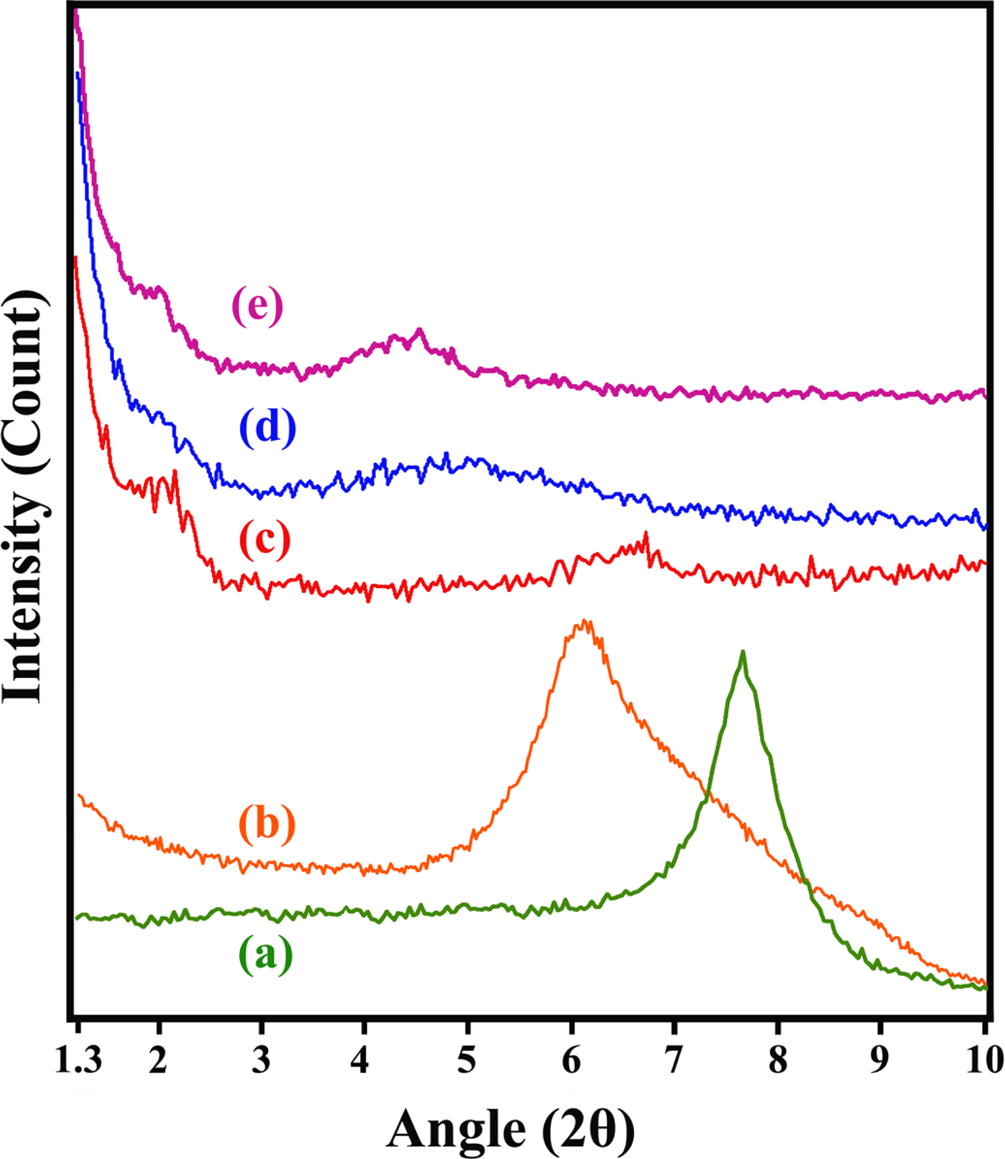
XRD data of the modified Cloisite Na+ and different POTBNC samples are presented in Figure 5. Also, XRD data of pure polymer and POTBNC 5% in the range of 10–80° are showed in Figure 6. The d-spacing of pristine Cloisite Na+ is 1.17 nm calculated from the peak position 2θ = 7.56° using Bragg’s equation. After the ion-exchange reaction with ammonium salt of
XRD patterns of (a) Cloisite Na+, (b) OC, (c) POTBNC 5%, (d) POTBNC 10% and (e) POTBNC 15%. OC: organoclay; POTBNC: PAI/OC/M-TiO2 bionanocomposite; XRD: x-ray diffraction.
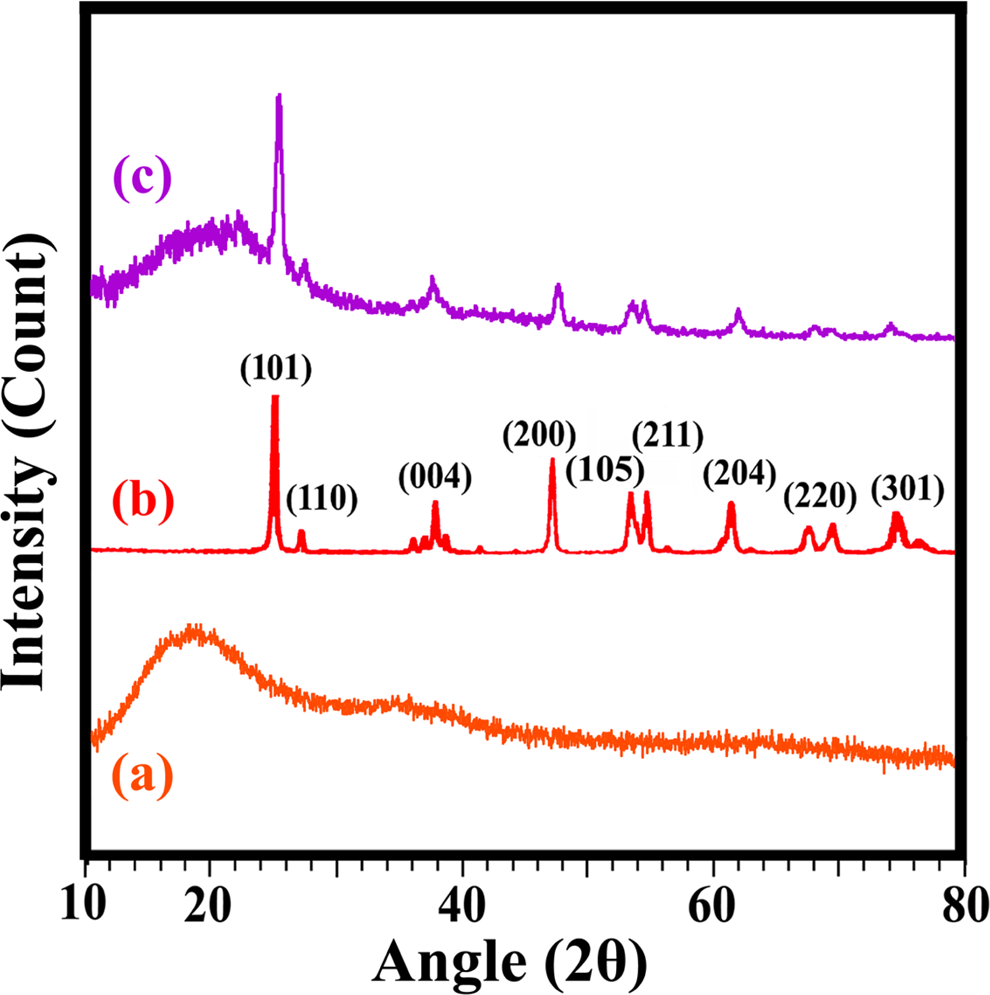
XRD patterns of (a) PAI, (b) M-TiO2 and (c) POTBNC 5%. PAI: poly(amide–imide); M-TiO2: surface-modified titanium dioxide; POTBNC: PAI/OC/M-TiO2 bionanocomposite; XRD: x-ray diffraction.
The XRD patterns of POTBNC 5% in the range of 10–80° show characteristic peaks of M-TiO2 and PAI, indicating that the morphology of TiO2 NPs has not been disturbed during the process.
The average crystalline size of TiO2 NPs, which has been determined from the half-width of the diffraction using the Debyee–Scherrer equation, is approximately 35–40 nm. The Sherrer’s equation is as follows
where D is the crystallite size, λ is the wavelength of the radiation, θ is the Bragg’s angle and β is the full width at half maximum.
Field-emission scanning electron microscopy
The surface morphology of BNCs was investigated by FE-SEM as shown in Figure 7. From FE-SEM images, the white spots are TiO2 NPs and any aggregation or phase separation has not been observed. This shows that the method used in this study for the preparation of POTBNCs could make the NPs well dispersed into polymer matrix and gave uniform and homogeneous distribution of both OC and M-TiO2 in these POTBNCs.
FE-SEMs of (a and b) POTBNC 5%, (c and d) POTBNC 10% and (e and f) POTBNC 15%. POTBNC: PAI/OC/M-TiO2 bionanocomposite; FE-SEM: field-emission scanning electron micrographs.
Transmission electron microscopy
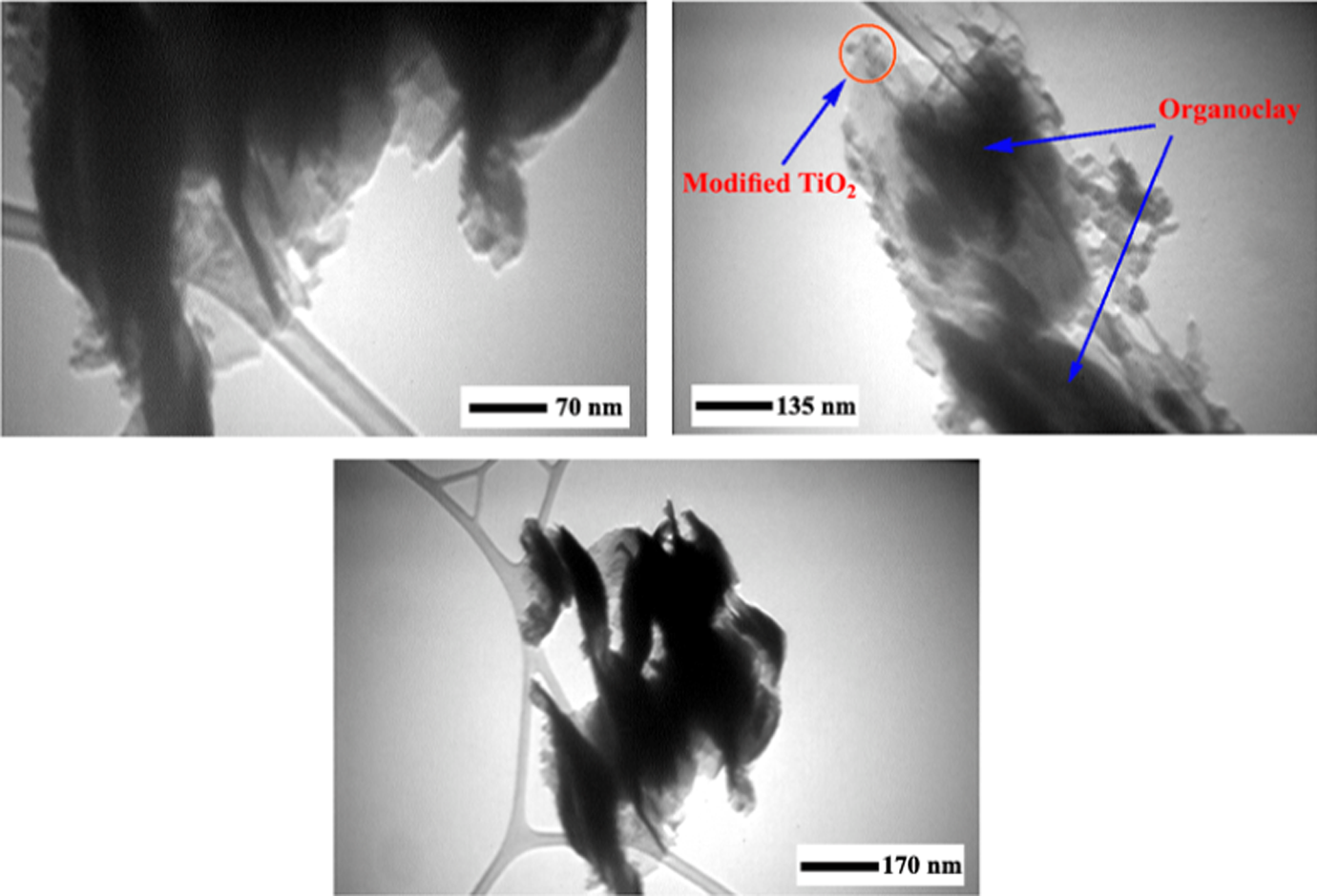
The additional confirmation of this nanometer-scale dispersion of intercalated silicate layers and M-TiO2 in PAIs was investigated by TEM. The morphology of BNCs was studied using TEM, and the images of POTBNC 5% are given in Figure 8. In the TEM photograph of the POTBNC structure, the brighter region represents the polymer matrix while the dark lines indicate OC. This sample structure showed a coexistence of exfoliated and intercalated OC. Also, the central black spot showing M-TiO2 indicated that the TiO2 NPs were well dispersed in the polymer matrix.
Transmission electron micrograph of POTBNC 5%. POTBNC: PAI/OC/M-TiO2 bionanocomposite.
Thermal analysis
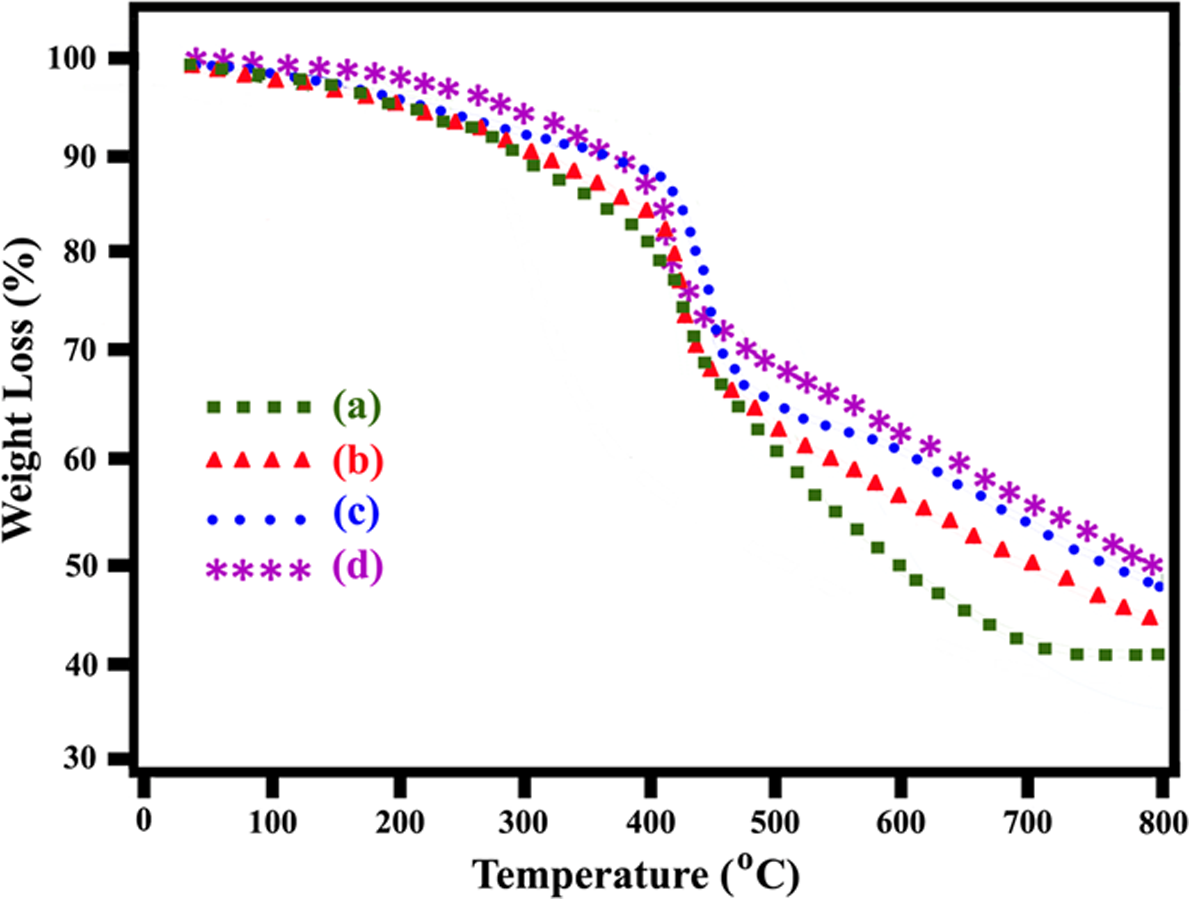
All samples were dried in vacuum before the TGA test. The TGA measurements of POTBNCs with different percentages of OC and M-TiO2 under a N2 atmosphere are shown in Figure 9, and the resulting TGA data are summarized in Table 1. These thermograms indicate that the decomposition temperatures of these materials are in the range of 400–490°C, which means that these POTBNCs are thermally stable and that the thermal stability slightly increases with the addition of OC and M-TiO2 content in the PAI matrix. The weight retained by these samples at 800°C is roughly proportional to the amount of OC and M-TiO2 in the POTBNCs. Char yield can be applied as a decisive factor for the estimated limiting oxygen index (LOI) of the polymers based on Van Krevelen and Hoftyzer equation
38
TGA thermograms of (a) PAI, (b) POTBNC 5%, (c) POTBNC 10% and (d) POTBNC 15%. PAI: poly(amide–imide); POTBNC: PAI/OC/M-TiO2 bionanocomposite; TGA: thermogravimetric analysis. Thermal properties of PAI and POTBNCs. PAI: poly(amide–imide); POTBNC: PAI/OC/M-TiO2 bionanocomposite; TGA: thermogravimetric analysis; LOI: limiting oxygen index; N2: nitrogen.
aTemperature at which 5 and 10% weight loss was recorded by TGA at a heating rate of 20°C min−1 in a N2 atmosphere.
bWeight percentage of the material left undecomposed after TGA at maximum temperature of 800°C in a N2 atmosphere.
cLOI evaluating at char yield at 800°C.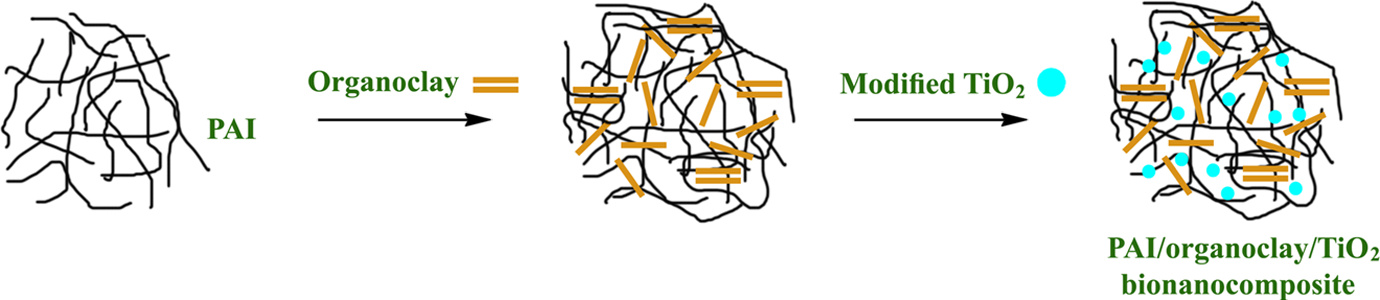

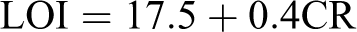
where CR is char yield.
The POTBNCs have LOI values around 37, which were calculated from their char yield at 800°C. On the basis of LOI values, all POTBNCs can be classified as self-extinguishing BNCs (Table 1). This enhancement in the char formation is attributed to the high heat resistance exerted by the OC and M-TiO2, because these NPs have high thermal stability, so the incorporation of these materials in hybrids can improve the thermal decomposition rate of the POTBNCs.
Conclusions
To briefly conclude, we have described the preparation of biodegradable and optically active PAI through direct polycondensation reaction in green media using TBAB, and then this polymer was end capped with amine end groups of diamine in the final polymerization process. The incompatibility between organophilic polymer and the hydrophilic clay was resolved through surface modification of the Cloisite Na+. Also, dispersion of NPs is a key property to obtain organo–inorgano hybrid composite, so by effective surface modification of TiO2 NPs using KH550, they were uniformly dispersed into PAI matrix without aggregation. The incorporation of different percentages of OC and M-TiO2 into optically active PAI, afforded an opportunity to prepare several novel POTBNCs via solution intercalation method. BNCs of PAI are characterized by FT-IR spectroscopy, XRD, TGA, FE-SEM and TEM. FT-IR data confirm that the NPs are dispersed in the PAI matrix. According to the results of XRD, FE-SEM and TEM, the formation of intercalated and exfoliated OC platelets is confirmed and also the M-TiO2 is well dispersed in the PAI matrix. Thermal stability of POTBNCs is also investigated by TGA. The incorporation of nanolayers of modified Cloisite Na+ and M-TiO2 in PAI matrix resulted in an increase in thermal decomposition temperature compared with neat PAI based on TGA. Since these POTBNCs have amino acid as a biodegradable segment in both OC and optically active PAI, it could be categorized under environmental NCs and could be composted with organic wastes and recycled to enrich the soil.
Footnotes
Funding
This work was financially supported by the Research Affairs Division Isfahan University of Technology (IUT), Isfahan; National Elite Foundation (NEF), Iran Nanotechnology Initiative Council (INIC) and Center of Excellency in Sensors and Green Chemistry Research (IUT).