Abstract
The present study aimed at exploiting the unfavorable changes of thermally modified shellac with the idea of developing starch/shellac-based composites intended for food contact applications. On baking shellac samples at 200°C for different time intervals (0–20 min), functional groups and linkages were drastically modified, especially anhydride formation, polyesterification, and ether/acetyl linkage occurrences. These processes caused a marked increase in the glass transition temperatures (from about 33 to 60°C) and melting temperatures (from 175 to about 230°C) but a decreased solubility in ethanol (from 95 to 9%). The addition of shellac into a starch-based formulation for producing shaped bodies in combination with heat could significantly strengthen the composite materials and drastically reduce the water uptake after soaking in both cold water and hot water. Furthermore, application of a food-grade biodegradable coating showed remarkable improvement in terms of water resistance and mechanical strength of the composites. The first attempt at making chopsticks was successful and showed promising results. Nevertheless, forming processes and mold designs of such articles—long narrow tubes—need further study.
Introduction
Shellac is the purified product of lac, a natural resin secreted by Kerria lacca (Kerr), a tiny insect that is parasitic on certain trees and is native to Thailand, India, Myanmar, and Laos. The resin exhibits many useful properties, including excellent film-forming ability, adhesion, and a glossy finish. These properties have been utilized for the production of wooden varnishes, adhesives, paints, and coatings. 1,2 Moreover, shellac is biodegradable, self-sustaining, and nontoxic. The latter is evident by the Generally Recognized as Safe status approved by the U.S. Food and Drug Administration. 2,3 Therefore, shellac has been widely used as coatings for foods, confectioneries, fresh fruits and vegetables, and solid pharmaceutical dosage forms. 4
The chemical constituents of shellac are composed of mono- and polyesters of hydroxy fatty acids (mainly aleuritic acid) and sesquiterpenoid acids (mainly jalaric and laccijalaric acids), as shown in Figure 1. 1,5 These constituents significantly affect the mechanical and thermal properties of shellac. In particular, the functional groups governing the properties of shellac include carboxylic acid, hydroxyl moieties, aldehydic groups, unsaturated points, and ester linkages. 3,6,7 Shellac generally shows gradual changes in color (darkening) and a decreased solubility during storage. On heating, shellac starts to soften at temperatures above its glass transition temperature (T g; around 60–70°C) and gradually turns rubbery and finally into a viscous mass as the temperature increases up to 125–150°C. 3 After cooling, the solidified shellac becomes relatively insoluble and infusible. Bhatia et al. 7 reported the improved scratch resistance and adhesion ability of shellac baked at 100–200°C. The substantial changes in thermal behavior and solubility of shellac on baking (and aging as well) are usually considered to be unfavorable circumstances, and new technologies have been applied to overcome these drawbacks; for instance, modifying functional groups 8 or grafting with new monomers. 9
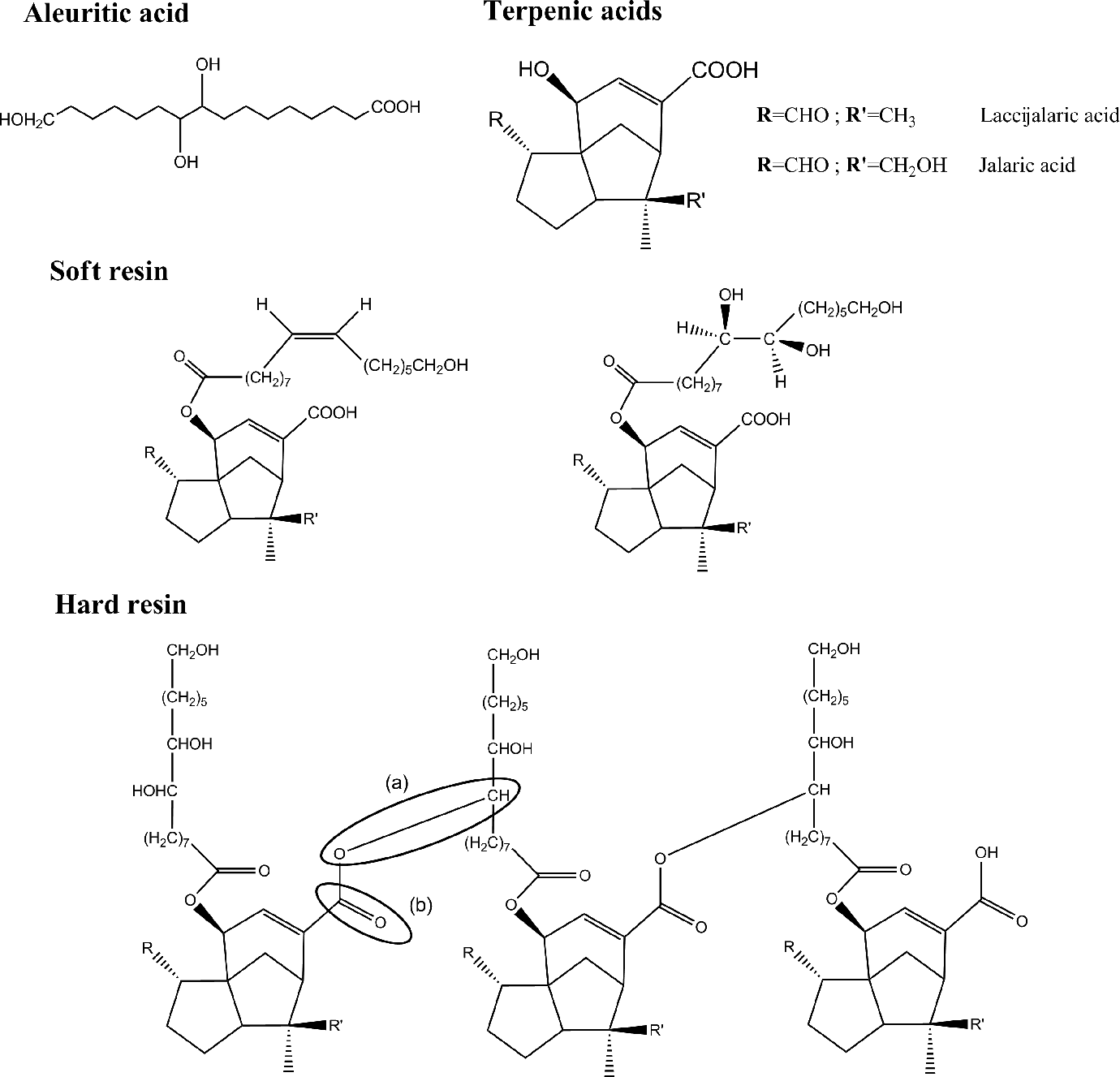
Major chemical components of shellac.
The increasing demand for materials, which are safe for food contact application and also environmentally friendly, presents a challenge to researchers and business. Starch is a naturally occurring biopolymer, consisting principally of amylose and amylopectin. It is of interest as a biodegradable material because of its availability as agricultural surplus material, its low cost, and its thermoprocessibility. Starch-based materials in various forms have been successfully developed to replace synthetic plastic articles, as reported in the literature. 10 These composite materials are mainly composed of cassava starch, reinforcing fibrous filler, and processing aid agents. However, these products still have limitations in terms of water resistance, which dramatically affects the mechanical strength of materials. Thus, the present study was initiated to exploit the unfavorable changes of thermally modified shellac with the idea of developing starch/shellac-based composites intended for food contact applications.
Experimental
Materials
Bleached shellac was supplied from Asia Shellac Ltd (Bangkok, Thailand). Native cassava flour and modified starch (commercial grade) were obtained from, a local producer, Sanguan Wongse Industries Co. Ltd, Thailand; virgin pulp was prepared in the author’s laboratory using disk refining. Water-based coatings for food contact papers were supplied by NuCoat Inc., Plymouth, MN, USA. Analytical-grade ethanol (95%) was used as received (manufactured by Merck, Germany) without additional purification.
Preparation of bleached shellac films
Bleached shellac was finely ground and sieved (40 mesh) before being dissolved in 95% ethanol at a concentration of 25% (weight in volume; w/v). The mixture was gently heated to 50°C while stirring thoroughly until a clear mixture was obtained. The mixture was then filtered and cast onto a polished aluminum plate to a thickness of 1 mm. After drying at 50°C for 1 h, shellac films were then peeled off and cut into pieces of 1 × 1 cm2. Film samples were divided into five sets; each set was subjected to thermal treatment (baking) in an oven at 200°C for 0, 5, 10, 15, and 20 min, respectively. Baked films were kept in desiccators at room temperature prior to further investigation.
Preparation of starch/shellac-based composites
Cassava starch dough was prepared by the method of Poovarodom and Praditdoung. 10 Bleached shellac solution at 20% w/v using 95% ethanol was added to the starch dough at a weight ratio of dough–solution = 1:1.25. This formulation was obtained from the preliminary study (data not shown). The mixture was kneaded using a bakery dough mixer until a uniform starch/shellac mixture was obtained. Samples of the mixture (11 g each) were pressed into individual aluminum molds, 1.5 × 7.0 × 0.7 cm3, and dried at 50°C overnight. These rectangular specimens were divided into four groups; each sample group was then subjected to baking in an oven at 200°C for 0, 10, 15, and 20 min, respectively. Samples were conditioned at 27°C, 65% relative humidity for at least 24 h before further testing. Control specimens were prepared using the same procedure as the studied specimens, except with no shellac solution added.
Coating of starch/shellac-based composites
Specimens of starch/shellac-based composites were prepared from the same formulation as previously described. After being dried at 50°C overnight, the specimens were divided into two groups: group 1 was baked in an oven at 200°C for 10 or 15 min and then coated; while group 2 underwent coating before baking at 200°C for 10 or 15 min. To obtain a uniform coating and enhance impregnation of coatings into the starch matrix, specimens were immersed in the coating solution and then placed in a rotor at 800 rpm for 30 s.
Sample characterization and material testing
Analysis of chemical components of shellac samples
Baked film was pulverized, mixed with potassium bromide, and then pressed at a pressure of 100 bar into small tablets of 1 mm thickness. Transmission infrared spectra of each sample set were recorded using a Fourier transform infrared (FTIR) spectrophotometer (Bruker Tensor 27, Germany), operating in the region 4000–400 cm−1.
Determination of T g and T m
Differential scanning calorimetry (DSC) was performed on baked film samples to determine their T gs and melting temperatures (T ms) using a Mettler-Toledo DSC 1 STARe system (Switzerland). Baked film sample was pulverized, then accurately weighed (5 mg), and hermetically sealed into an aluminum pan. The first scan was run from 25 to 110°C at a heating rate of 10°C min−1 with nitrogen purge and followed by cooling to 25°C at the same rate. This is to eliminate previous history of samples, which could affect the measurement of T g. 11 The second scan was then operated, reheating the sample to 250°C at the same heating rate. DSC data recorded from the second scan were used to determine the T g and T m of the sample. In general, the DSC operating temperature of shellac does not exceed 250°C in order to preclude substantial alteration and decomposition of the sample. 12,13 In addition, the results obtained from the preliminary thermogravimetric analysis (data not shown) using a Thermogravimetric Analyzer Linseis STA PT-1000 system (Linseis International, Germany) indicated the decomposition temperature of shellac samples of 263°C.
Determination of solubility
Ground and sieved (40 mesh) baked film sample was accurately weighed (1 g) and placed into each of the five flasks containing 50 ml of 95% ethanol. The mixtures were constantly stirred at 800 r min−1 for 1 h at room temperature. At the end of the mixing time, the mixtures were left for 24 h to allow insoluble mass to precipitate out. The soluble mass was determined by evaporative gravimetric method. The clear mixture (25 ml) were transferred to a vial and evaporated to dryness under nitrogen gas. The residue was weighed and subtracted by the solid content of the solvent; the solubility of shellac was then calculated and expressed as weight per volume percentage.
3-point bending flexural test
Specimens (prepared using the methods mentioned in “Preparation of starch/shellac-based composites” and “Coating of starch/shellac-based composites” sections) were immersed in water either at room temperature (for 3 h) or hot water at 85°C (for 30 min) prior to mechanical tests. In comparison, unsoaked specimens were also subjected to the same test.
A 3-point bending flexural test was performed in accordance with ASTM D 790-07, 14 using a universal testing machine (Instron model 5965, Grove City, PA, USA) equipped with a load cell of 5 kN and supporting span of 50 mm. The load was applied to the center of each specimen at a crosshead speed of 1.3 mm min−1 and was stopped when the specimen broke. Flexural strength was determined and reported as an average value of six replications.
Determination of water absorption
Water resistances of all specimens in each group were determined by a gravimetric method. Accurately weighed specimens were immersed either in water at room temperature for 3 h or in hot water at 85°C for 30 min. After removing from water, the specimens were patted dry with tissue paper and weighed. Water absorption was then expressed as the increase in weight percentage. Six replications were tested for each treatment.
Results and discussion
Characterization of thermally modified shellac samples
Analysis of chemical components
A comparison of the FTIR spectra of shellac samples subjected to baking at 200°C for different time intervals, as depicted in Figure 2, shows significant chemical changes in terms of functional groups and linkages. The intensity of –OH groups (peak at 3460 cm−1) decreases with increased baking time. This indicates the decrease in intermolecular hydrogen bonds and loss of –OH groups. On the contrary, the peak indicative of the carbonyl group
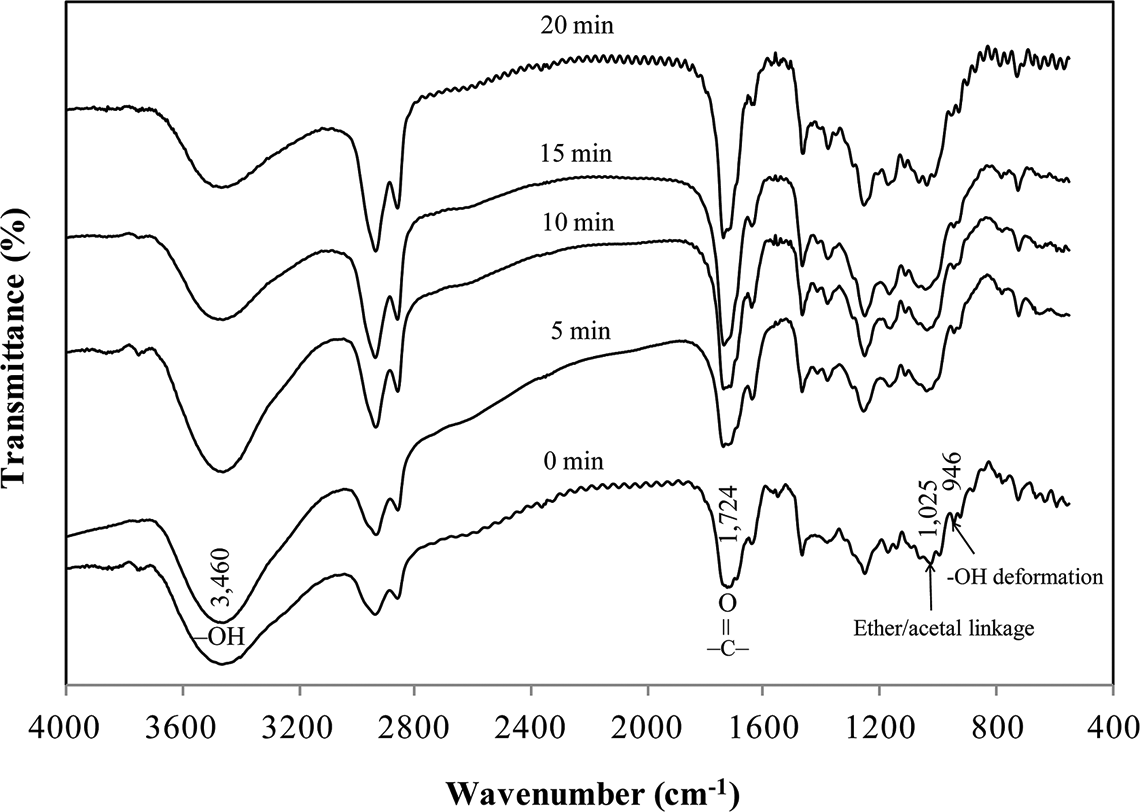
A comparison of the spectra of shellac films subjected to baking at 200°C for different time intervals.
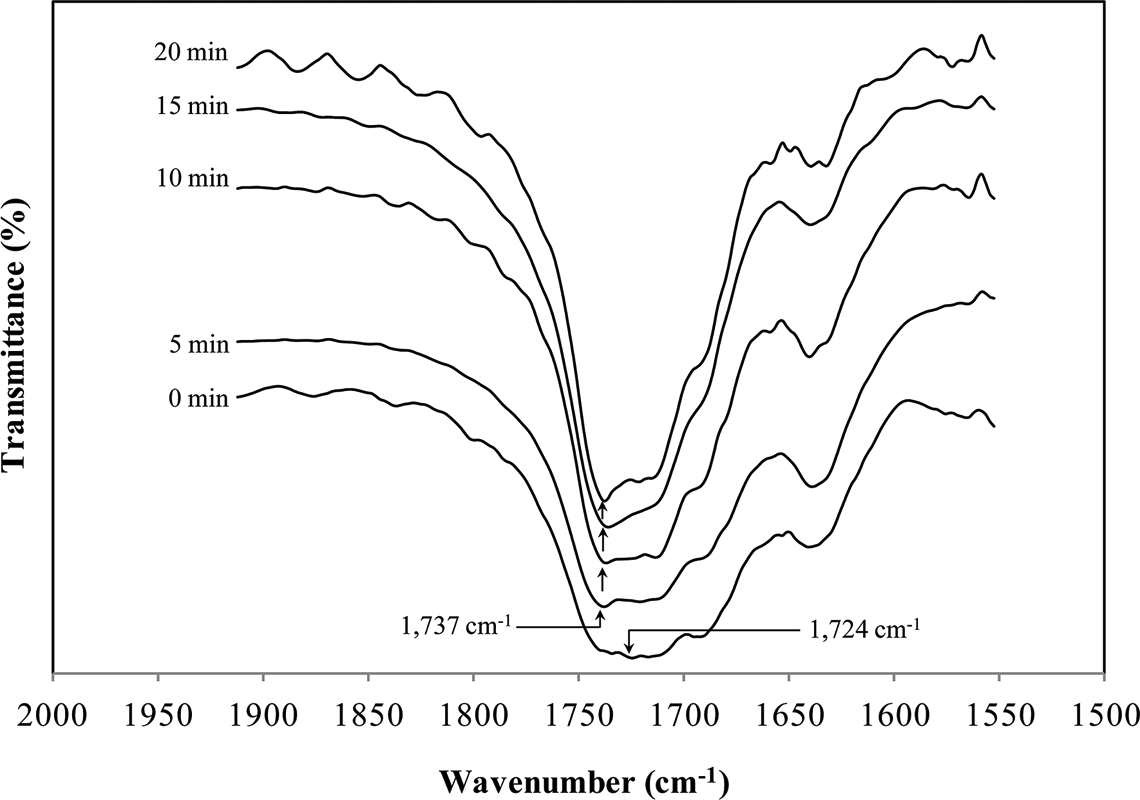
Expanded spectra, in
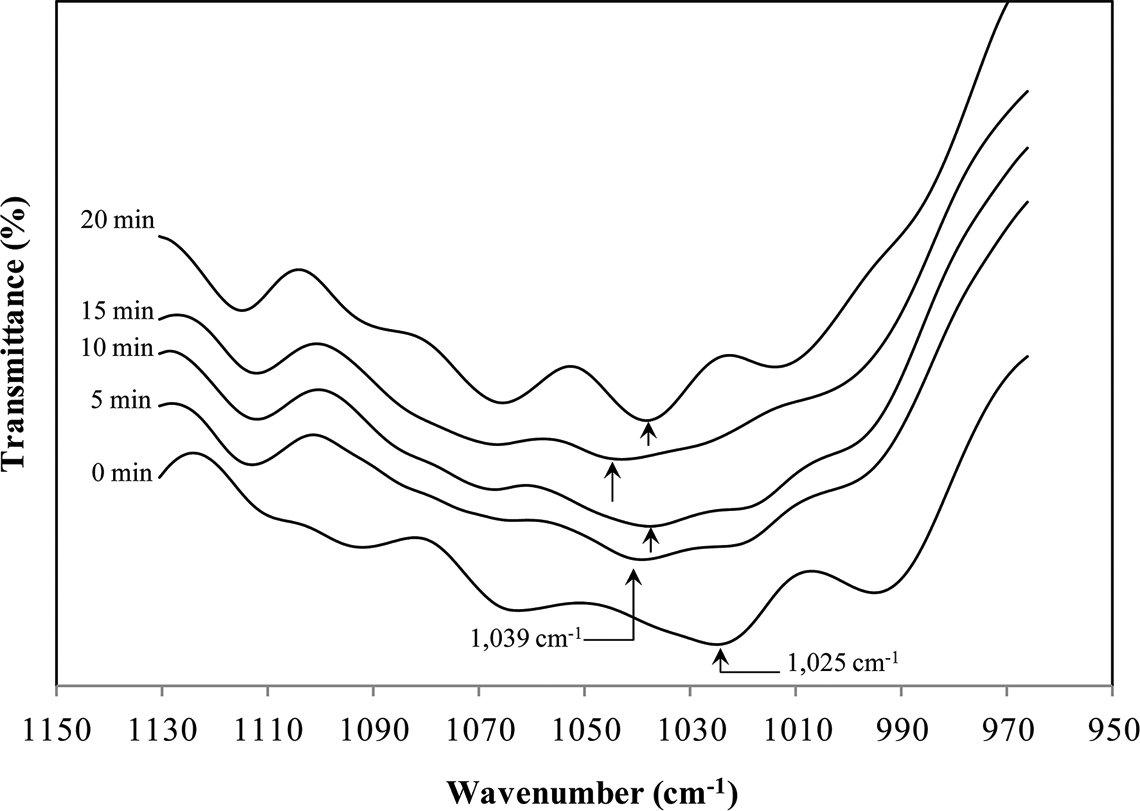
Expanded spectra, in
Furthermore, the peak at 946 cm−1 diminishes in intensity on baking at 200°C and shifts to a higher wave number, 954 cm−1 after baking for 20 min. This phenomenon highlights the loss of –OH groups in dimeric carboxylic acids, 7 which in turn results in the formation of anhydride groups. Also, the shifting of peaks in the case of a baking time of 20 min can suggest the onset of –C–O of anhydride deformation. It is worth noting that these functional groups and linkages are the major determinants of shellac properties, particularly solubility and thermal properties.
Determination of solubility and thermal properties
The solubility of shellac samples subjected to baking at 200°C for different time periods was measured to determine the effect of baking on resin properties. Table 1 clearly shows that the solubility of baked shellac in ethanol was reduced dramatically with increased baking time, from about 95% for unbaked to 27% for 5 min, 15% for 10 min, 12% for 15 min, and 9% for 20 min, for baking. This confirms the loss of –OH groups, which causes lower polarity of shellac and consequently reduces its solubility in ethanol. Besides, the formation of less polar linkages, that is, ester, ester/anhydride and ether/acetyl, and polyesterification accentuate the transformation of shellac into an insoluble material upon heating.
Solubility and thermal properties of shellac subjected to baking at 200°C at different time intervals.a
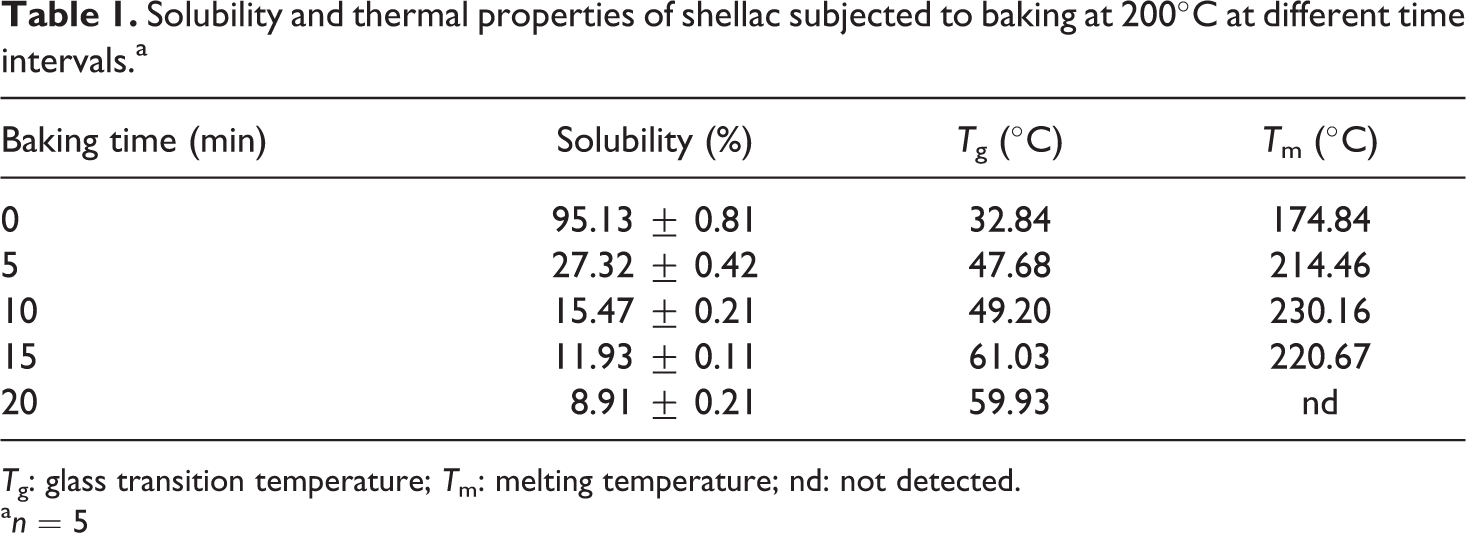
T g: glass transition temperature; T m: melting temperature; nd: not detected.
a n = 5
The effect of baking at 200°C on the thermal behavior of shellac was investigated using DSC technique. Table 1 shows a T g of 32.84°C for unbaked shellac, which is lower than T g values (39.5–52°C) reported in the literature. 13 The difference is mainly due to the different history of samples. However, low T g value broadly indicates the presence of low-molecular-weight compounds in the studied shellac. T g value increases with increase in baking time, that is, 47.68°C for 5 min, 49.20°C for 10 min, 61.03°C for 15 min, and 59.93°C for 20 min. The rise of T g suggests the presence of high-molecular-weight components in shellac, which indicates the occurrence of polyesterification and the formation of ester, ester/anhydride and ether/acetyl linkages, as observed from FTIR spectra.
Similarly, the T m values of shellac rose sharply on baking for 5 min (Table 1); afterward, the increase in T m became less prominent. This indicates that polyesterifi-cation and the formation of ester, ester/anhydride and ether/acetyl linkages, as observed from FTIR spectra, would occur to a large extent shortly after baking (within 5 min) and slow down on further baking (for 10 and 15 min). It is important to note that no T m value was detected in the case of 20 min baking time. In addition, shellac decomposition and burns were observed when heating at a temperature of 250°C or above. 12 These suggest that the largest extent of the occurrence of polymerization, compared to the cases of shorter baking time, and the unique formation of anhydride, as observed from FTIR spectra, could result in the transformation of shellac into an infusible material upon baking at 200°C for 20 min.
These findings should encourage further study on the development of starch/shellac-based composites for food contact applications. Disposable items intended to come into contact with liquid and hot foods, such as chopsticks and cutlery, are particularly challenging.
Development of starch/shellac-based composites
Cassava-based materials were invented 10 as a potential replacement for disposable plastic food containers because of the anticipated environmental benefits. However, the water resistance of the materials requires improvement in order to allow their effective use in diverse applications. The prominent properties of thermally modified shellac observed in this study, especially solubility and thermal behaviors, confirm the potential of including shellac solution in starch formulations for making shaped bodies formed at high temperatures.
Figure 5 clearly shows that the addition of shellac in combination with heat treatment could drastically reduce water uptake of the specimens in both cold water and hot water. In addition, control specimens (no shellac, no heat treatment) were almost instantly damaged after soaking in water, consequently the water uptake could not be determined. The formation of anhydride groups and polyesterification—together with the loss of –OH groups and the formation of functional groups and linkages, that is, ester, ester/anhydride and ether/acetyl—collectively make shellac less polar; hence, the water resistance of specimens was improved. Moreover, the extent to which shellac becomes less polar and more resistant to water increased with increased baking time, as observed from FTIR spectra. Nevertheless, soaking in hot water yielded higher water absorption, which in turn was markedly reduced with increased baking time. This may result from the transition of shellac into a rubbery stage while soaking in hot water at a temperature (85°C) that exceeded its T g.
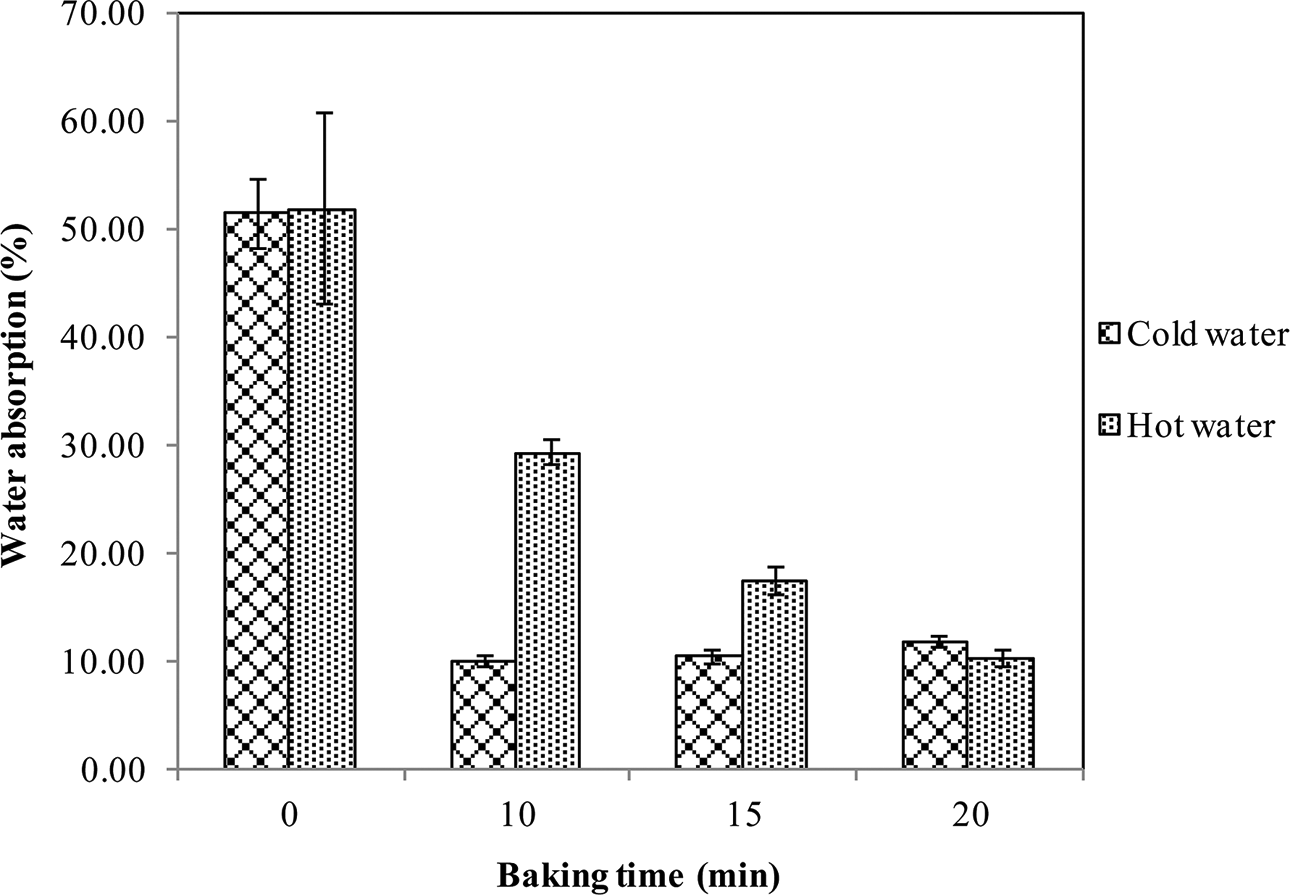
Water absorption of starch/shellac-based composites subjected to baking at 200°C for different time intervals.
Correspondingly, a significant improvement in mechanical strength, 3-point bending flexural strength, of specimens was observed (Figure 6). The findings agree well with the results of FTIR analysis—that the formation of anhydride groups and polyesterification on baking, which are responsible for improvement in mechanical properties of shellac, 3,7 increased with increase in baking time. In addition, control and unbaked specimens were damaged a few minutes after soaking in water. This accentuates the beneficial effects of shellac in combination with heat on the properties of starch/shellac-based composites.
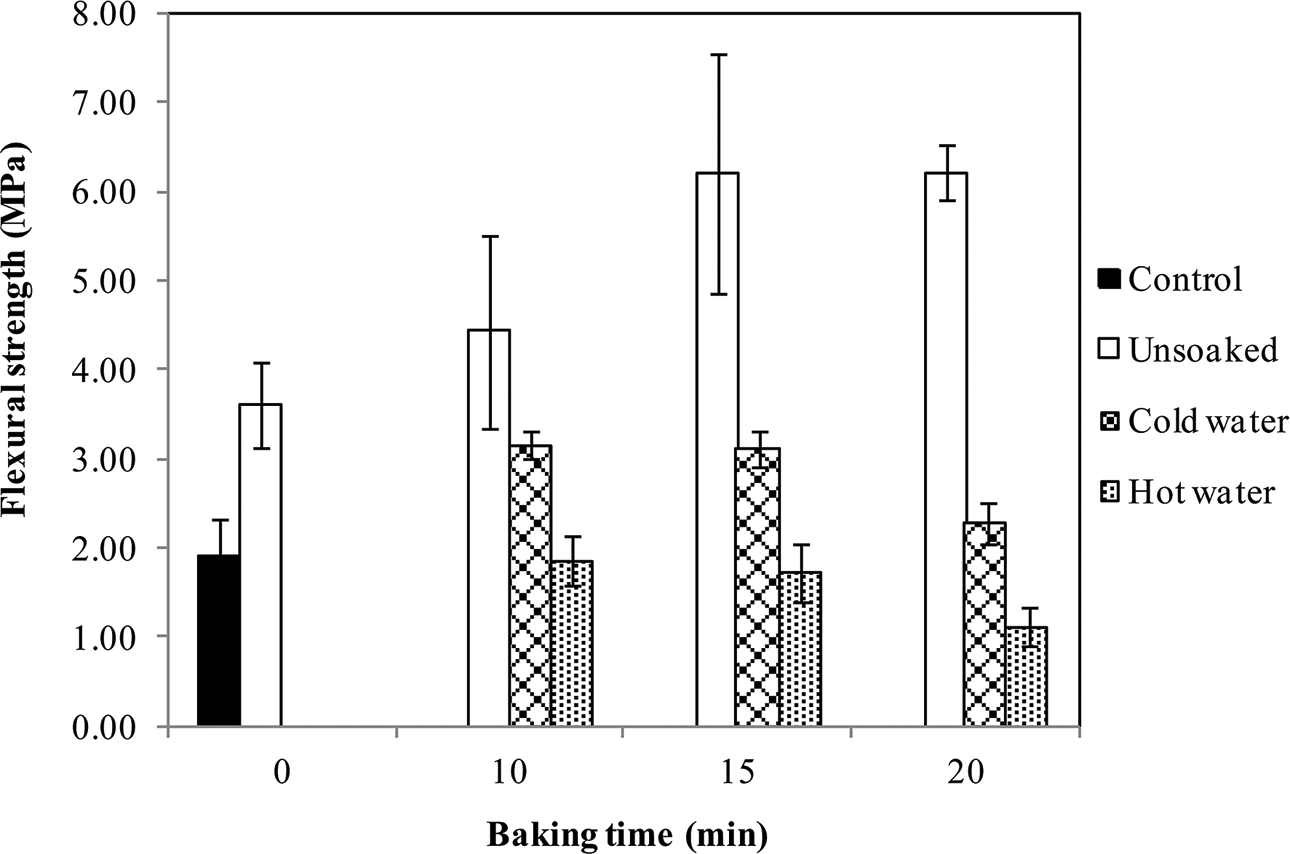
Mechanical strength of starch/shellac-based composites subjected to baking at 200°C for different time intervals, compared with control (specimens without shellac).
Effects of thermal treatment and coating
The findings initiated the idea of producing food contact articles, in particular, disposable items, which generally come in contact with hot liquid foods during customary use. Items such as chopsticks and cutlery are especially challenging. However, the retention of mechanical strength after soaking in water should be improved to allow their effective use under unforeseen circumstances. Therefore, a food-grade biodegradable coating, which is commercially available, and, generally, used in the paper industry was selected for further study.
It was found that the addition of this coating significantly improved the water resistance and retention of mechanical strength of the specimens. Moreover, the seque-nce of operations during production played an important role in product improvement (Figures 7 and 8). Since thermal treatment could make the starch/shellac matrix less polar, consequently the dispersion and impregnation of coating liquid into the matrix was reduced. Therefore, coating before baking had advantages over the reverse sequence. In addition, baking times of 10 and 15 min produced relatively similar effects on the hot water resistance properties of specimens. It was also observed that specimens of 10 min baking time, after soaking in hot water, could retain more strength than those of 15 min baking time and were as strong as unsoaked specimens, as well. These results together with energy consumption benefits suggest the preferable conditions—coating before baking at 200°C for 10 min—for producing starch/shellac-based composites for food contact applications, especially hot liquid foods.
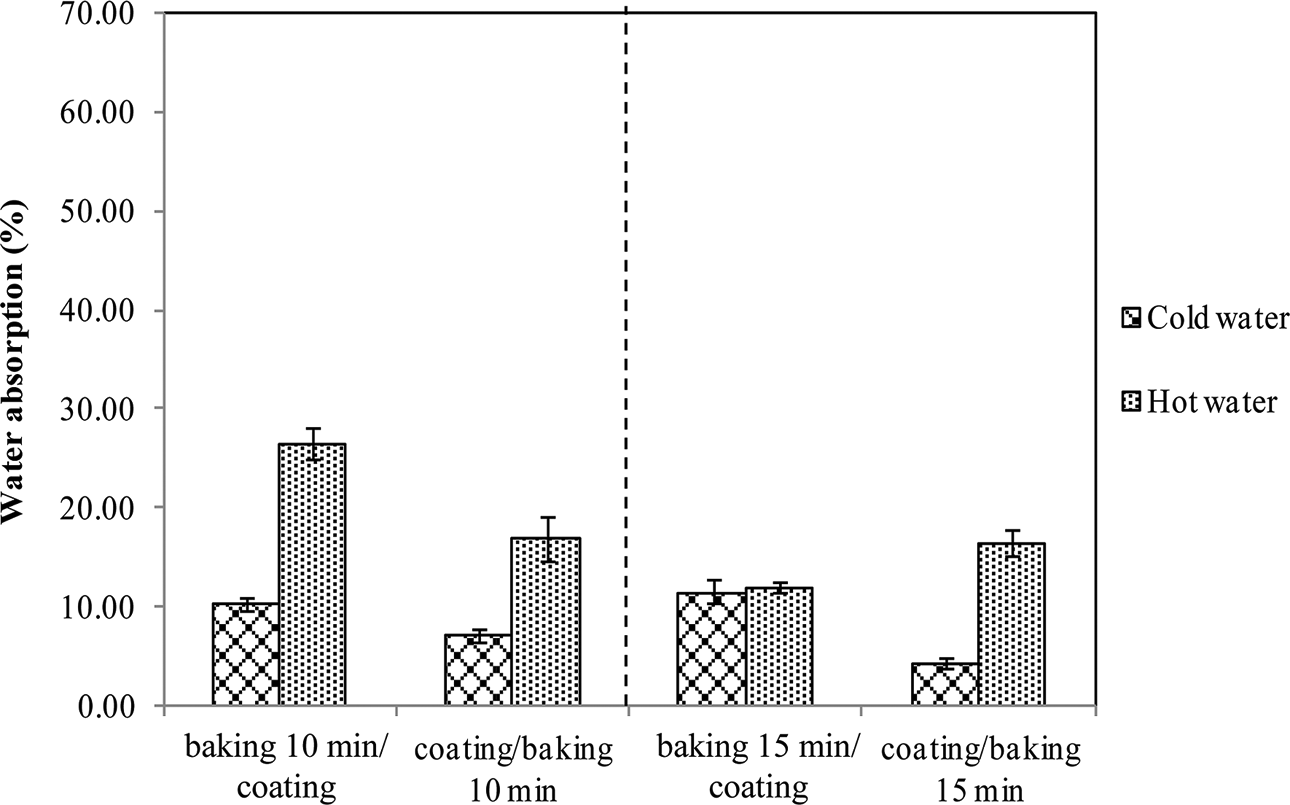
Water absorption of starch/shellac-based composites coated and subjected to baking at 200°C for 10 and 15 min.
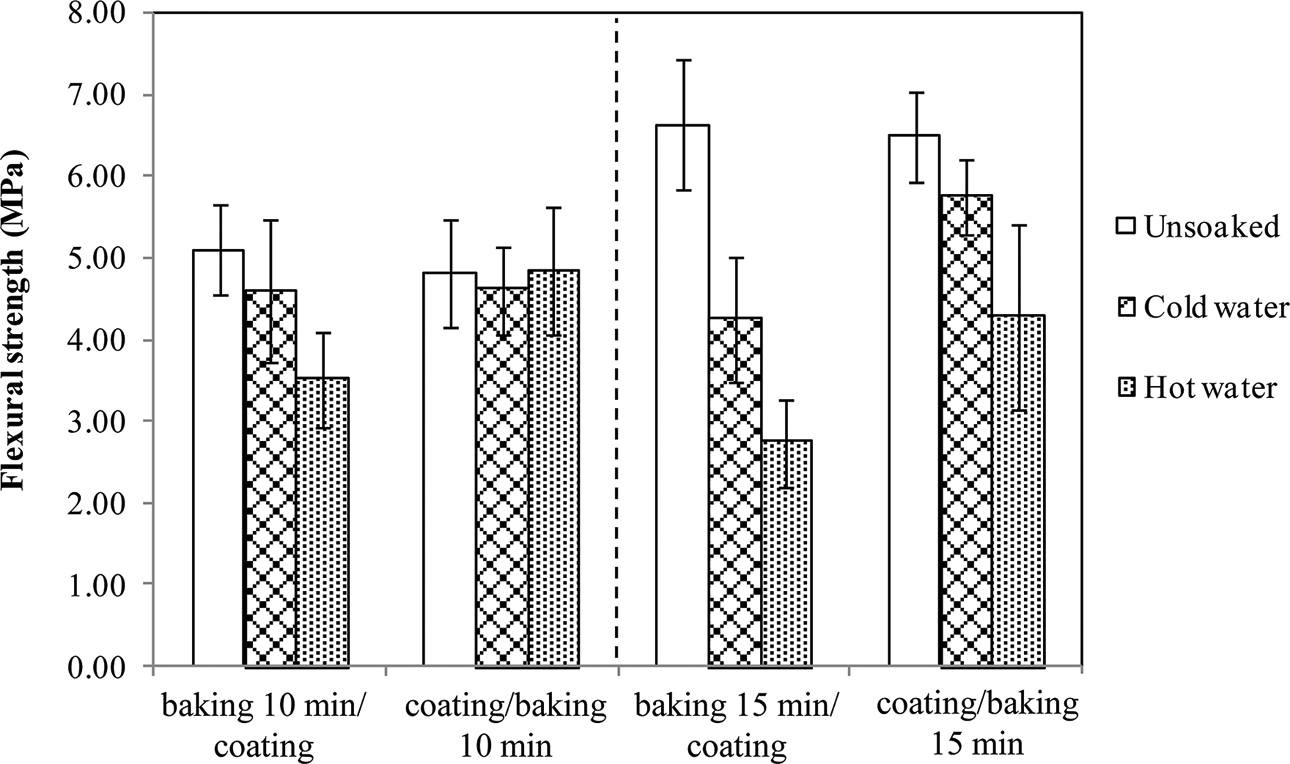
Mechanical strength of starch/shellac-based composites coated and subjected to baking at 200°C for 10 and 15 min.
Our first attempt at making chopsticks (diameter 1 cm and length 16 cm), using the same procedure as the studied specimens and baking at 200°C for 10 min after coating, achieved satisfactory results. Samples could retain strength after soaking in cold water and hot water (2.7 and 2.2 MPa, respectively) and could be used in actual practice (in-house trials and other applications, data not shown). Nevertheless, continued studies are needed to develop a production system, particularly involving forming steps and mold designs, to ensure uniformity of shaped articles.
Conclusions
On baking shellac films at 200°C, functional groups and linkages were modified, in particular, the occurrence of anhydride groups, polyesterification, and ester/anhydride and ether/acetyl linkages. These changes resulted in marked increase in T g and T m but decreased solubility in ethanol. The addition of shellac into a starch-based formulation for producing shaped bodies in combination with heat could significantly strengthen the composite materials and drastically reduce the water uptake after soaking in both cold and hot water. Furthermore, application of a food-grade coating for paper showed remarkable improvement in terms of water resistance and mechanical strength of the composites. The first attempt at making chopsticks was successful and showed promising results. Nevertheless, forming processes and mold designs of such articles—long narrow tubes—requires further study.
Footnotes
Acknowledgements
The authors would like to acknowledge all institutes for their support, both in kind and financially.
Funding
This study was a part of the project KU-GREEN, run by Kasetsart University, to improve the performance of cassava-based food contact articles. A part of this research was also funded by the Agricultural Research Development Agency and the Center for Advanced Studies in Agriculture and Food, Institute for Advanced Studies, Kasetsart University.
