Abstract
Organic hybrid composites based on carboxylated nitrile rubber and nylon-12 reinforced with mercerized and diisocyanated lignocellulose residue (LCR) was prepared. The influence of the LCR on the viscoelastic properties of these organic hybrids was investigated by dynamic mechanical analysis and thermal analysis (differential scanning calorimetry (DSC)). It is found that either the position of the damping peak was shifted to higher values or the intensity of the damping peak was significantly increased with LCR. These results could imply that the LCR enhanced the damping properties of the composites. The thermal stability of the composites was evaluated with the mean values obtained using thermogravimetrical analysis. The decomposition rate was investigated using differential thermal gravimetry. The crystallization behavior of the prepared composites was checked by DSC.
Introduction
Polyamide blends have attracted interest as engineering thermoplastics due to their excellent strength properties, chemical resistance and wear resistance with high melting points. They are ideally suited for use in various critical applications. The presence of the reactive –CONH– group within polyamide materials renders nylon-based materials superior properties such as high melting temperature and good toughness. 1 Thermoplastic vulcanizates based on polyamides fabricated by melt-mixing technique with suitable rubbers like nitrile butadiene rubber (NBR) can also exhibit oil resistances due to the polarity of both components. 1 Compared with NBR, the carboxylated nitrile rubber (XNBR) is a reactive rubber with more polarity, and it shows good compatibility with polar or nonpolar resins and higher degree of interaction with fillers. 2,3 It is also reported that XNBR has improved damping properties, showing a great potential as matrix polymers. 4 –7 In the light of the aforementioned information on XNBR and nylon, XNBR/nylon-12 was chosen in the current report as the polar matrix system to accommodate chemically modified biomass material instead of using it as landfill material. The new XNBR-based organic hybrids are prepared by adding a polar organic biomass compounds, which is an industrial agro-polymer obtained during the olive oil extraction from olive seeds. The aim of this combination is to develop a new organic hybrid system and to extend the application field of XNBR into organic hybrid systems containing biomass materials. Thus, the three components are capable to get hybridized due to their capability to form reversible hydrogen bonding as a result of proton donor/proton acceptor action between –CONH–, –COOH– and –C–OH– groups within the components of the system. 8 –10 The reversibility of the intermolecular hydrogen bonding leads to the dissipation of a great amount of mechanical energy as heat formation will improve the damping properties of the system. Therefore, in the current investigation, the effects of the lignocellulose residue (LCR) on the damping properties of XNBR/nylon-12 were first investigated. Subsequently, the thermal stability of the prepared composites as a function of LCR modification was investigated to obtain some ideas on the capability of the biomass to thermally stabilize the system.
Experimental
Materials and formulation
The formulation used to produce the composites is presented in Table 1. Filler treatments and the technique followed to produce the XNBR/nylon-12 composites reinforced with modified and unmodified LCR are reported recently. 8 Three formulations were prepared in this study. The first is XNBR/nylon-12, which is set as the control. The second is XNBR/nylon-12 reinforced with mercerized LCR and is abbreviated as XNBR/nylon-12-M. The third is XNBR/nylon-12 reinforced with diisocyanated LCR and is abbreviated as XNBR/nylon-12-D. 8
Recipe used to produce the XNBR/nylon-12 thermoplastic composites filled various types of LCF (php).a
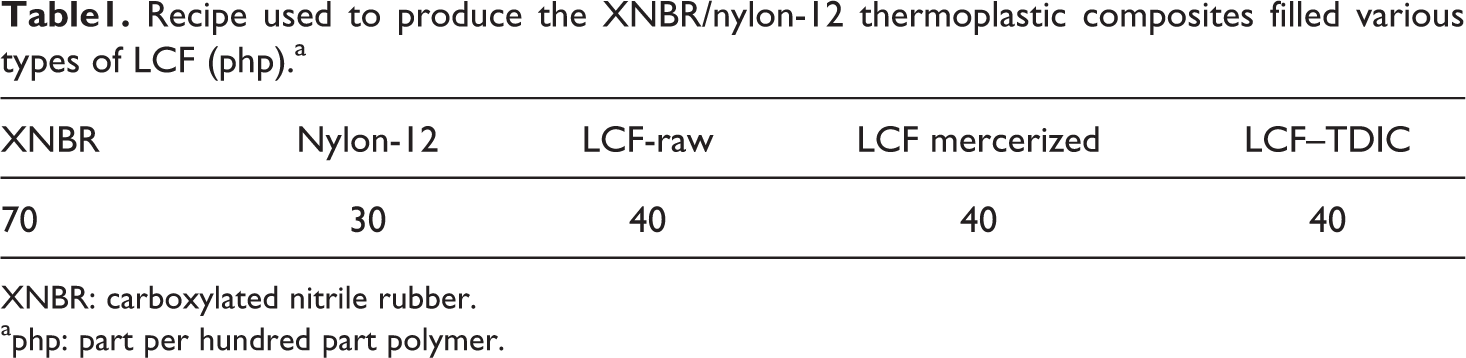
XNBR: carboxylated nitrile rubber.
aphp: part per hundred part polymer.
Thermal behavior
Dynamic mechanical parameters such as storage modulus (E′) and the mechanical loss factor (tan δ) as a function of temperature were measured with a rheometric scientific thermal analysis system in tension mode on rectangular specimens. The samples were scanned as a function of temperature from −100 to 100°C at a heating rate of 3°C/min in a dual cantilever mode and constant frequency of 1 Hz. The static load strain was 1% and dynamic load strain was 0.5%. The thermal stability is determined by thermogravimetric analysis (TGA) with a high resolution TGA (Q500, TA Instruments, USA) at a heating rate of 10 K/min. The samples were scanned from room temperature (RT) to 700°C under nitrogen atmosphere. Degradation temperatures are determined by differential thermal gravimetric (DTG) scans, at the peak maximum.
Differential scanning calorimeter
Differential scanning calorimeter (Perkin-Elmer DSC-6) is used to investigate crystallization behavior. Samples (between 10 and 15 mg) are sealed in aluminum pans. The heating and cooling rates were 10 K/min. A nitrogen flow is maintained throughout the test. For all materials, the first scan is used for eliminating the thermal history of the material. Each sample is heated to 250°C then cooled to RT before a second heating and cooling cycle. The crystallization temperature as well as the area under the crystallization peak set as the enthalpy of crystallization (▵H c) was recorded.
Results and discussions
Thermogravimetrical analysis
The temperature–weight loss diagram of the prepared composites is shown in Figure 1. The thermogram presents three main events over the scanned temperature range for the three formulations studied in this work. Regarding the control sample, the first stage lasts up to 300°C in which the total weight loss was less than 11%. The weight loss up to this point is not linked to pyrolysis, instead this is related to loss of the moisture uptake of the matrix, since it is well known that nylon is hygroscopic material. The second main event is related to pyrolysis by minor decomposition of the polymer chain for unfilled sample, this stage starts at 300°C and elapses at 377°C. The 20% weight loss that occurs within this stage was recorded at 336°C. This stage is most likely related to the degradation of the polymer matrix, that is, nylon and XNBR. The major decomposition was proceeded at 377°C to 422°C after that charring was occurred from 422°C to 700°C. Note that the total weight loss of the unfilled XNBR/nylon-12 was 86% of the original sample. The effect of the mercerized lignocellulose on the thermal stability of XNBR/nylon-12 is shown in Figure 1 as well. Note that the weight loss in the first stage is slightly higher than the control sample as reflected by the zoom-in part of Figure 1. The rationale behind this is the higher moisture content of the prepared composite in the presence of LCR. The total moisture content in this case is the sum of the water uptake from the matrix mentioned earlier and LCR. The second step of pyrolysis by minor decomposition shows that the decomposition temperature was shifted to the right indicating that the incorporation of the mercerized LCR increased the pyrolysis resistance of the XNBR/nylon-12 composite. Note that the temperature for 20% weight loss was 370°C, which means the temperature increase was 34°C compared with the control. This is most likely due to the high lignin content of the LCR due to the extraction of the hemicellulose via mercerization, consequently the phenolic content of the lignin is responsible for the enhanced thermal stability of the composites. The last stage was the charring stage, where the total weight loss of the sample is 82%, which is lower the total weight loss of the control by 4%. The influence of diisocyanate treated powder on the thermal stability of the XNBR/nylon-12 is presented in Figure 1 as such. Note that the moisture content is less than the other two formulas. This is due to the formation of carbamate derivative that imparts hydrophobic qualities to the LCR material, consequently this characteristic was conferred to the whole sample. 8 It is observable that pyrolysis resistance of the carbamated sample has been improved compared with the unfilled sample. The temperature for 20% weight loss was 346°C, that is, 10°C increment when compared with the control. Finally, the total weight loss of the sample was 88%. This could be due to the consumption of the –OH phenolic groups cited as thermal stability aid 9 –11 by the formation of urethane linkage reported earlier.
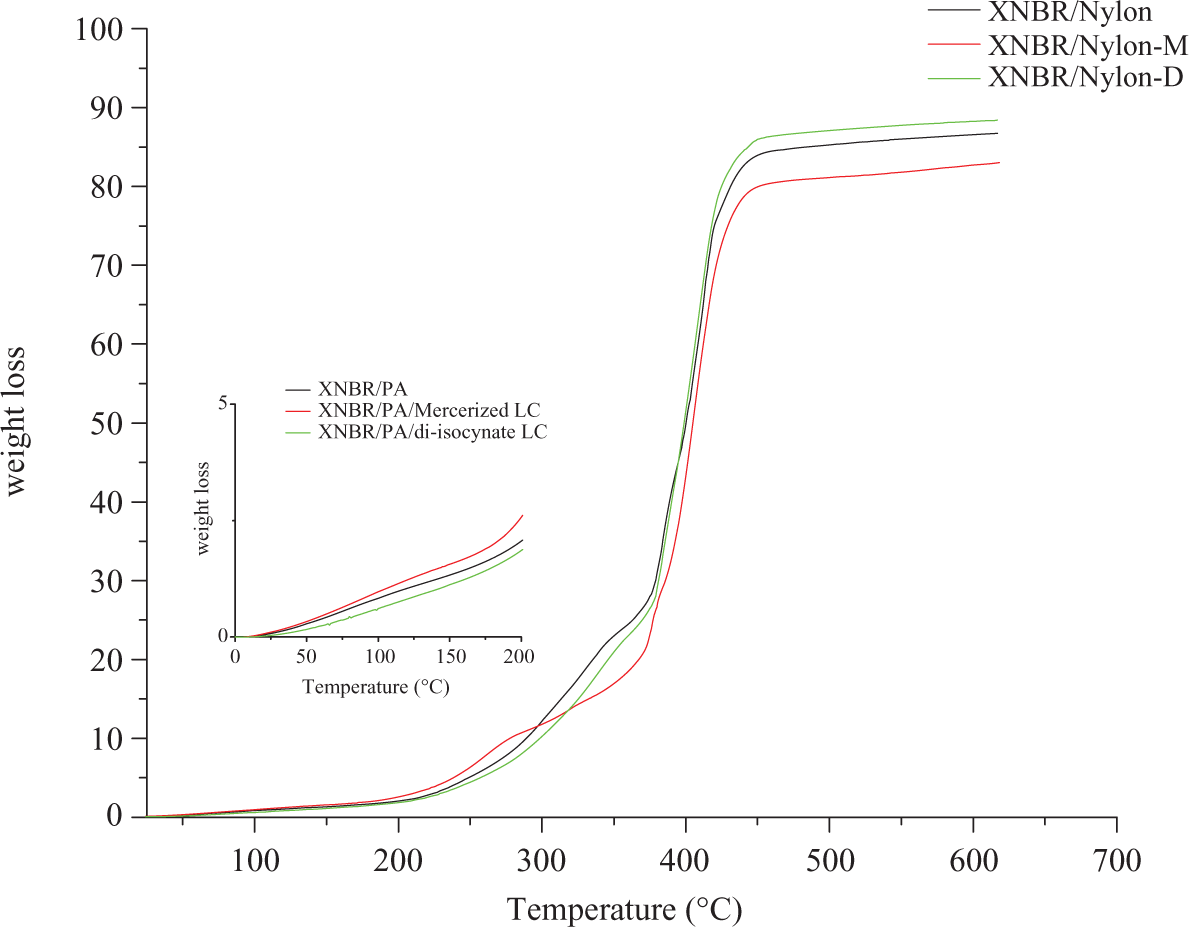
TGA curves of XNBR/nylon-12 composites reinforced various chemically treated LCR. TGA: thermogravimetric analysis; XNBR: carboxylated nitrile rubber; LCR: lignocellulose residue.
Differential thermal gravimetry
The DTG courses of the prepared samples are displayed in Figure 2. The degradation behavior conform the results obtained from the TGA curves. The control sample displayed a single pyrolysis peak at 427° at which the maximal weight loss occurred at this point was set as T max. The sample with the mercerized LCR showed T max at 433°C. The sample with isocyanated LCR as shown in Figure 2 displayed T max at 438°C. Based on the aforementioned results, it can be concluded that the incorporation of the mercerized LCR has enhanced the thermal stability of XNBR/nylon-12 composites due to the phenol of the lignin of the LCR. Such phenolic compounds are expected to improve the stability of the composite by the absorption of some of the heat that will prolong the ability of the matrix to resist the heat prior to pyrolysis and the formation of the polar–polar interactions that requires extra heat ahead to secession. The same reasons hold to explain the observation in the case of the sample with diisocyanated filler provided the formation of the urethane linkage due to the chemical reaction between the isocyanated filler and the matrix. 8 To sum-up, we can say that—judging from the peak height at T max—it can be concluded that the control sample has the highest peak height and the lowest T max compared with the two other composites. The implication is that it has the highest decomposition rate and the lowest thermal stability. Note that the incorporation of the LCR either mercerized or isocyanated into the composites has curbed the rate of decomposition by lowering the peak height and enhanced the thermal stability by increasing T max. To confirm this result, the data obtained from TGA curves were analyzed by the differential method in which ln W o − W 1/Wt − W 1 is plotted against time t, where W 0 is the initial weight, W t is the weight after time t and W 1 is the weight of the ash remaining after heating. 12,13 The slope of the obtained line is the rate constant for the thermal decomposition. 13,14 From Figure 3, it is clear that the relationship between ln(W o − W 1/Wt − W1) against t under thermal conditions gave two part lines. The first part, which occurs over the initial 30 min, is due to the loss of moisture. The second part is due to thermal decomposition of samples. This indicates that the loss in weight due to thermal decomposition is first order reaction. The rate constants of the weight loss due to thermal decomposition are shown in the Figure 3. It is clear that the control sample has the highest decomposition rate constant compared with the other two formulas. Again, it can be inferred that the LCR acts as a pyrolysis retardant due to the lignin content.
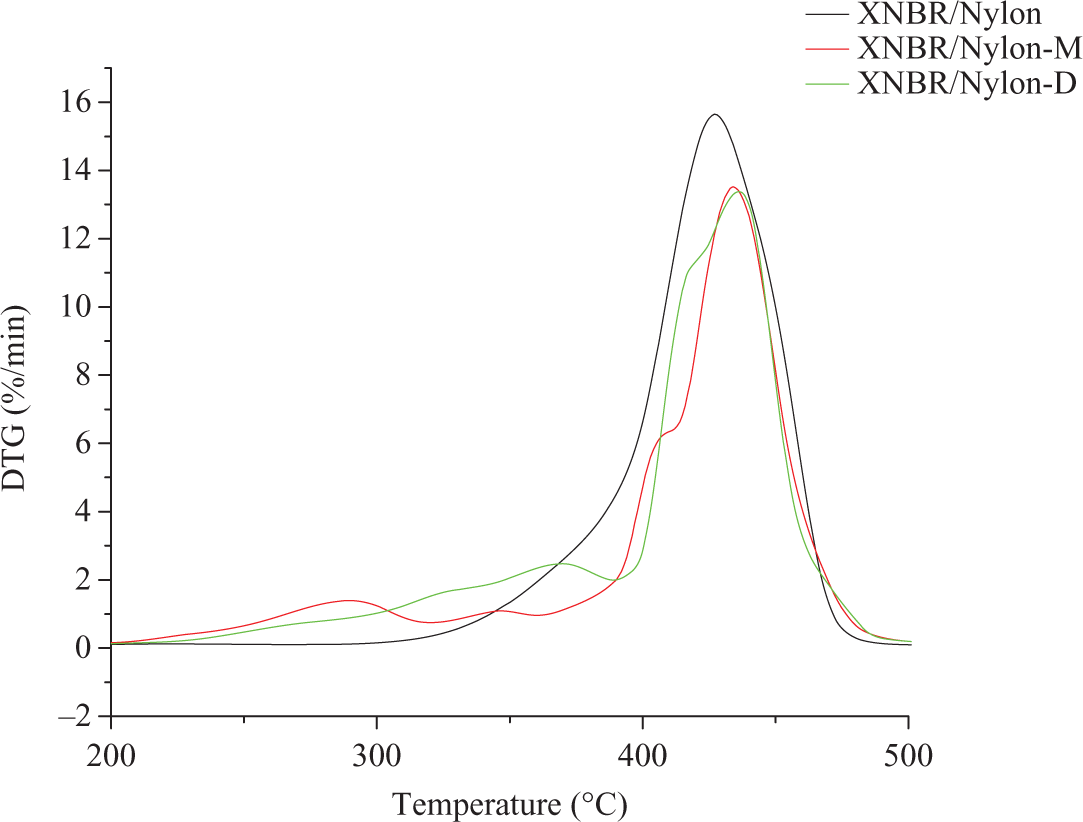
Differential thermal gravimetric curves of XNBR/nylon-12 composites reinforced various chemically treated LCR. XNBR: carboxylated nitrile rubber; LCR: lignocellulose residue.
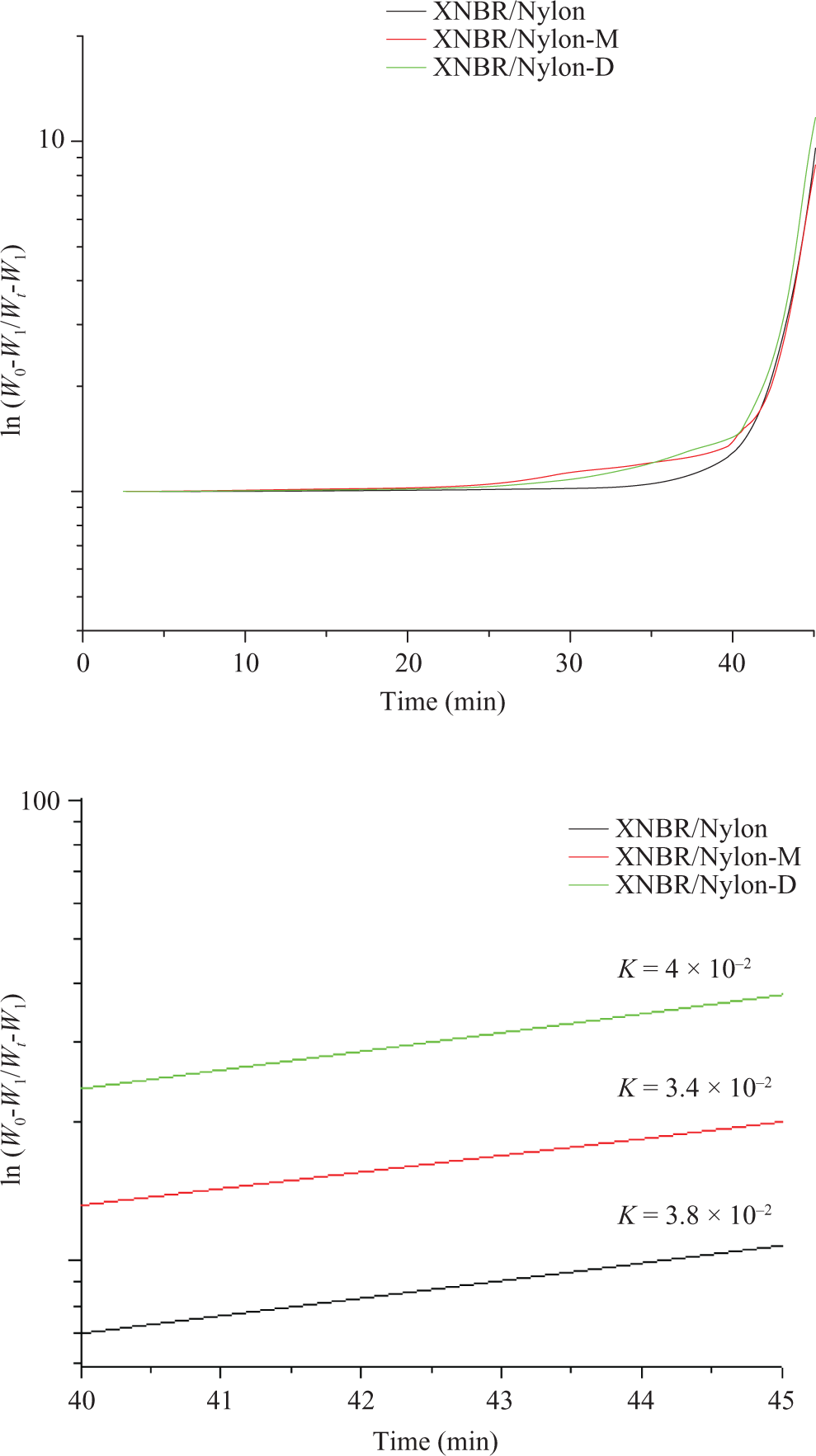
(a) ln (W o − W1/Wt − W1) against t for XNBR/nylon-12 composites reinforced various chemically treated LCR. (b) Decomposition rate composition of XNBR/nylon-12 composites reinforced various chemically treated LCR. XNBR: carboxylated nitrile rubber; LCR: lignocellulose residue.
Differential scanning calorimetry
The effect of the LCR on the crystallization behavior as obtained from the differential scanning calorimetry (DSC) for the prepared composites are shown in Figure 4. Note that the crystallization temperature (T c) of the nylon-12 within the composite as read off from Figure 4 is 140°C for the control, 142°C in the case of mercerized LCR and 139°C in the case of diisocyanated LCR. It is clear that the modified LCR has a slight influence on the value of T c of the nylon-12 within the composite; however, the peak intensity was clearly influenced by the addition of the LCR as shown in Figure 4. This was reflected from the value of enthalpy of crystallization shown in Figure 4. The decrease of ▵H c with LCR incorporation is due to the fact that the LCR acts as diluting agent for the nylon within the matrix. In other words, the volume fraction of the nylon-12 was reduced. Additionally, the LCR was not acting as nucleating agent for the nylon-12. The observed trend concords other authors who have noticed similar results in the case of nylon/wood composites. 15,16 The DSC crystallization behavior of PA-12 (Poly amide-12) within the composites concords the crystallization behavior extracted from the storage modulus of the DMA as reflected in Figure 5. In this regard, it can be seen that after the glass transition temperature (T g), the modulus curve starts to increase, which is a result of the recrystallization process of the PA-12. Note that the extent of crystallization of PA-12 of the composites is influenced by the presence of the unmodified and modified LCR in the sense that the stiffer the compound, the stronger the crystalline peek.
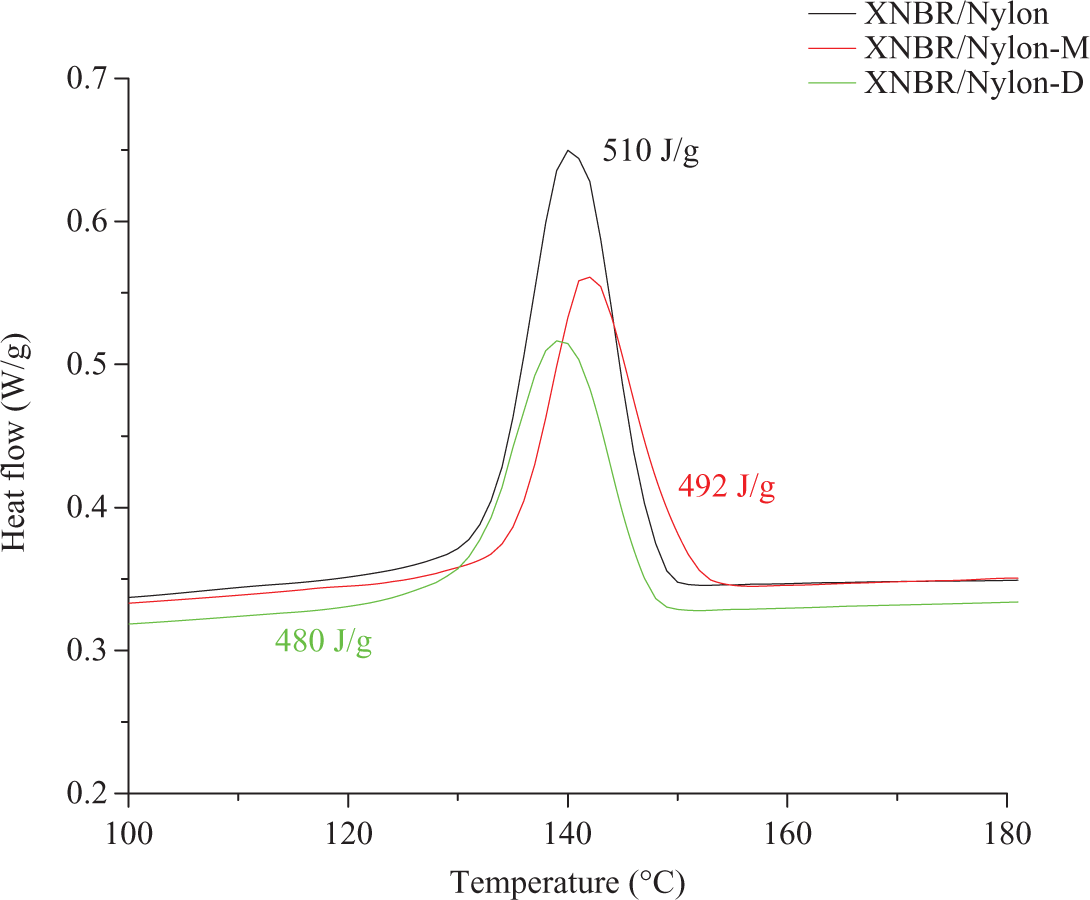
DSC trace of XNBR/nylon-12 composites reinforced various chemically treated LCR. DSC: differential scanning calorimetry; XNBR: carboxylated nitrile rubber; LCR: lignocellulose residue.
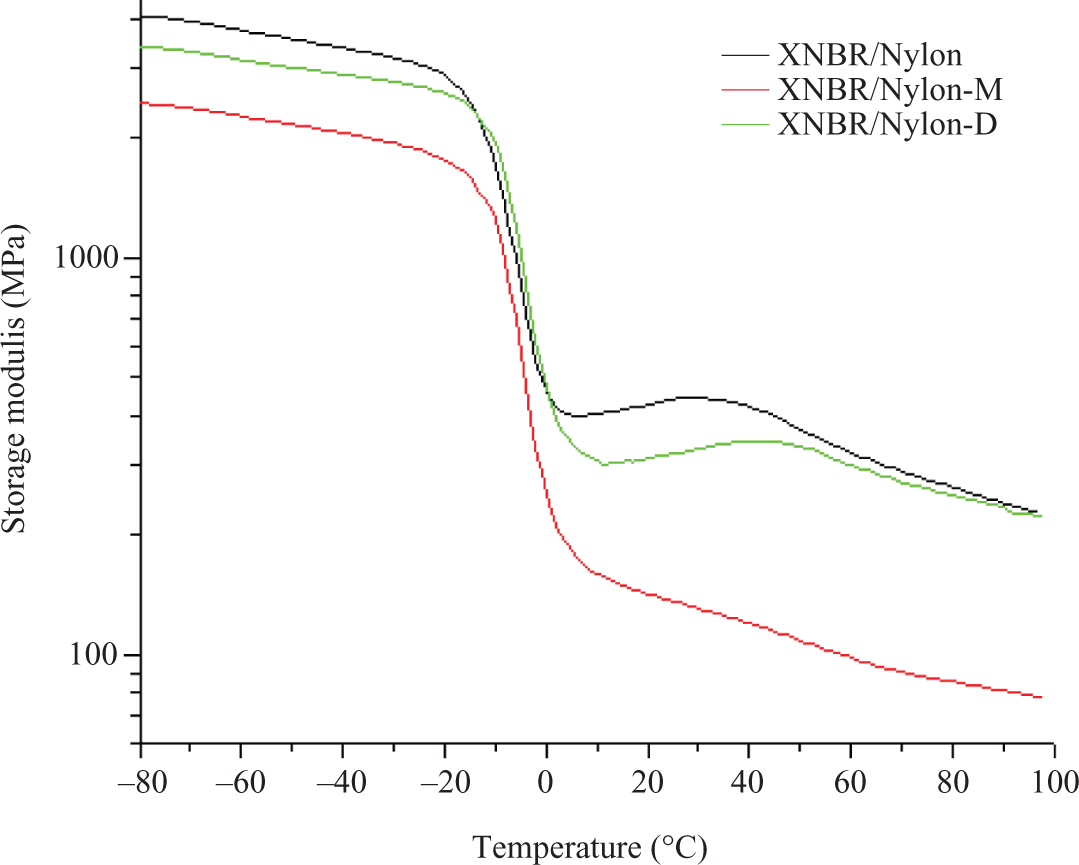
Temperature dependence of storage modulus of XNBR/Nylon-12 composites reinforced various chemically treated LCR.
DMA studies
Figure 6 shows the influence of LCR on the loss factors (tanδ) of XNBR/nylon-12 composites. Note that the pristine nylon-12 shows two peaks, the first one at −45°C related to the β transition, while the second at 75°C related to the α-transition (T g). Concerning the prepared composites, it can be seen that the control sample presents two distinct relaxation peaks. The first one located at −2.3°C is assigned to the XNBR of the composite; the second one should be assigned to T g of the nylon-12. However, the intensity of this peak is much lower than that of the pure nylon-12 as appeared in the same figure. Furthermore, the location of this peak has shifted from 75°C for the pristine nylon-12 to 62°C in the composites. Simultaneously, the β-transition peak of the nylon at −45°C disappeared after mixing. Therefore, it can be inferred that the change in position and intensity of the nylon-12 tanδ peak are signs on the partial miscibiliity of the two polymers due to the polarity of both polymers. The incorporation of the mercerized LCR has increased the intensity of the XNBR tanδ max. This is a hint that the damping properties of the composites have improved as a result of the LCR addition. The improved damping properties should be due to the hydrogen bond formation between –CONH–, –COOH– and –C–OH– groups within the system components. The formation of H-bond in this system was reported by us for this composite. 8 The reversibility of the intermolecular hydrogen bonding leads to the dissipation of a great amount of mechanical energy as heat formation, thus will improve the damping properties of the system. Furthermore, the respective T g of the blend components, that is, Nylon and XNBR were moved closer to each other after the incorporation of the mercerized LCR indicating improved miscibility. The incorporation of diisocyanated LCR has further increased the intensity of the tanδ max. This is a further evidence on the ability of the LCR to enhance the damping of the composites, which is again attributed to the H-bond formation, simultaneously; the second peak at 62°C assigned to the nylon-12 in the blend had almost diminished as shown by the zoom-in part of Figure 6. This is attributed to the consumption of the nylon-12 by the formation of the urethane linkage between the diisocyanated LCR and the blend components indicating better miscibility compared with the control and the mercerized LCR filled formulation. The viscous moduli as a function of temperature for the prepared composites are shown in Figure 7. Similar behavior to that of the tanδ is detected, wherein the intensity of the damping peak for the mixture with LCR is much higher; the highest was recorded for the composite with diisocyanated counterpart. The storage modulus as a function of temperature of the unfilled and filled composites is presented in Figure 5. Note that below T g, the plain composites has higher modulus with reference to the filled composites, whereas above T g, the modulus of the composites with diisocyanated LCR is more or less the same as that of the control. The observed scenario is an indication on the fact the diisocyanated LCR has better reinforcement efficiency than the mercerized LCR. This is due to the chemical and physical interactions via the formation of the urethane linkage and the H-bond reported earlier. 8,16,17 On the other hand, the capability of the mercerized one is limited to the physical forces between the filler and the matrix.
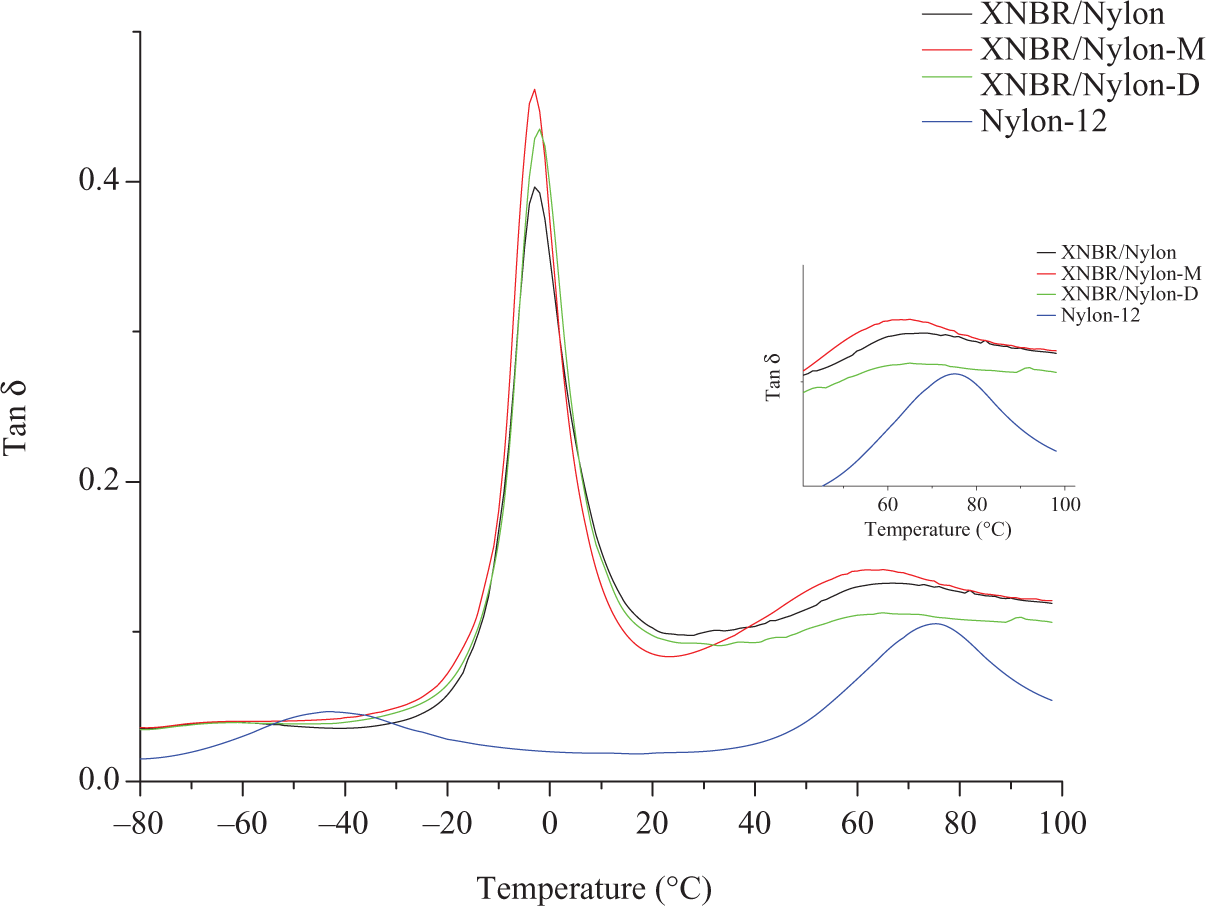
Temperature dependence of tan δ of XNBR/Nylon-12 composites reinforced various chemically treated LCR.
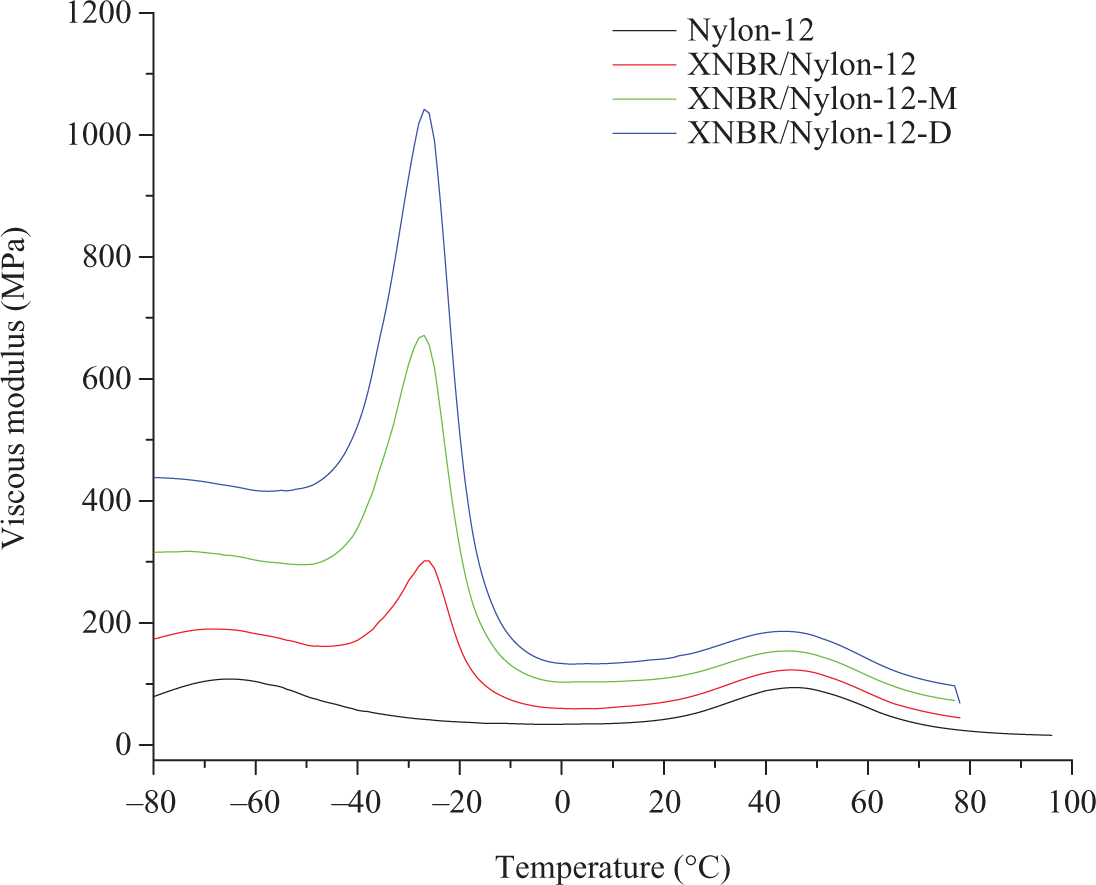
Temperature dependence of viscous modulus of XNBR/Nylon-12 composites reinforced various chemically treated LCR.
Tensile properties
Figure 8 shows tensile strength and modulus of composites with and without biomass. In the first place, it can be seen that the tensile strength at break of the composites is low. The possible reason for the reduction in tensile strength at break could be that the unreacted isocyanate may act as a plasticizer thereby reducing the strength of the composite. On the other side, the absence of sulfur curing system could be cited as a second reason for the observed trend. Figure 9 shows the percentage elongation at break (%EB) of the samples as a function of filler type. Interestingly, it is observed that the %EB of the sample with diisocyanated filler increased tremendously when compared with the control sample. This again is due to the formation of the urethane linkage between the treated filler and the XNBR/nylon-12 matrix. 8 This is a sign that the formula with diisocyanate treated sample will possess improved compression properties.
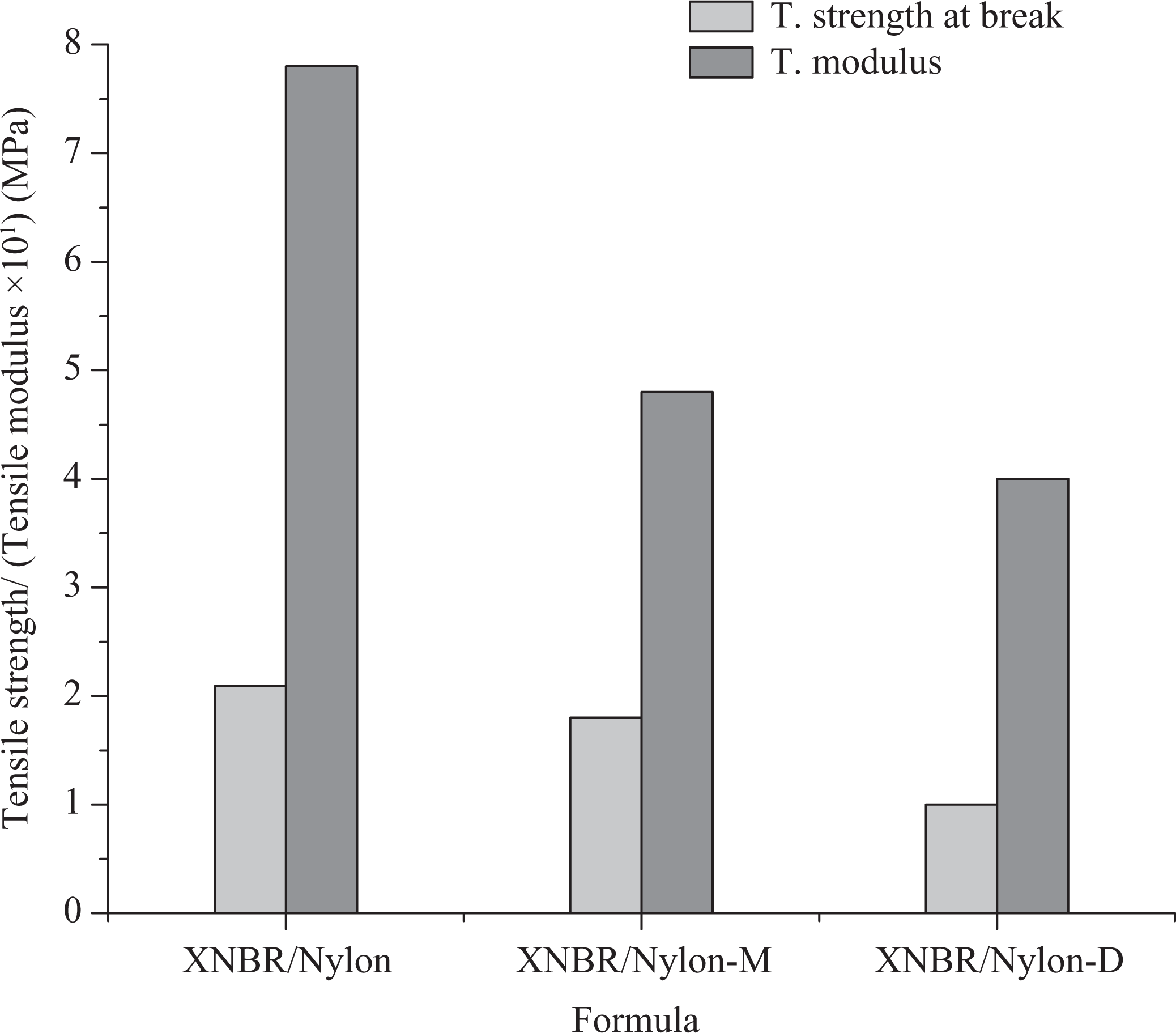
Tensile strength at break and tensile modulus of XNBR/nylon-12 composites reinforced various chemically treated LCR. XNBR: carboxylated nitrile rubber; LCR: lignocellulose residue.
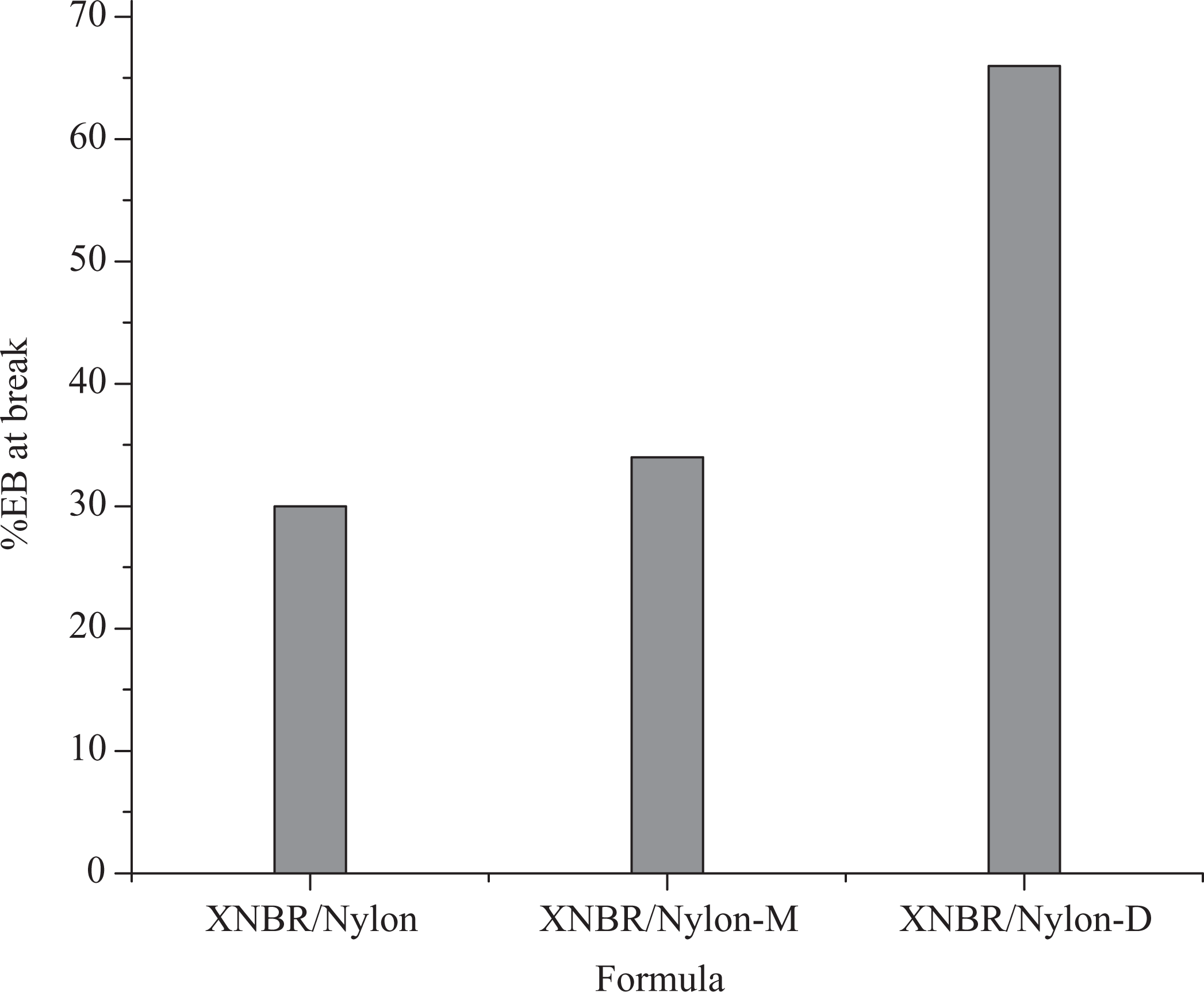
Percentage elongation at break of XNBR/nylon-12 composites reinforced various chemically treated LCR. XNBR: carboxylated nitrile rubber; LCR: lignocellulose residue.
Conclusions
The following conclusions can be drawn from the study: The LCR displayed a profound effect on the damping behavior of the XNBR/nylon-12 composites. This was related to the reversibility of the hydrogen bonds. The H-bonds originated from the proton acceptor/proton donor of the various functional groups within the system. The LCR acted as pyrolysis retardant for the XNBR/nylon-12 composites. This was evidenced by the increase in the temperature for 20% weight loss to happen after the addition of the LCR. The residue of the pyrolysis process was higher after the incorporation of the LCR. This was due to the phenols of the lignin content. The incorporation of the LCR has not influenced the crystallization temperature; instead, it slightly decreased the enthalpy of crystallization. This happened because the LCR dilutes the nylon content, while the LCR did act as nucleating agent. The incorporation of diisocyanated LCR has improved the %EB of the composite, whereas the tensile strength and modulus were slightly decreased when compared with the control.
Footnotes
Acknowledgements
A Mousa thanks the administration and the technical staff of the Leibniz-Institut für Polymerforschung Dresden e.V., Dresden (Germany) for making all the facilities available to carry out this project.
Funding
This work was supported by Alexander von Humboldt Foundation through a research scholarship (to A Mousa).
