Abstract
This work is focused on the modification of montmorillonite by hexadecyl trimethyl ammonium bromide (HDTMA) as a surfactant by cation exchange reaction. HDTMA-modified layered silicate (HDTMA-MLS) was compounded with polylactide (PLA) by melt extrusion technique for making nanocomposites. These nanocomposites were subjected to morphological, rheological, thermal and mechanical analyses. Variations in mechanical properties, plasticity and thermal stability with the addition of modified nanoclays in PLA were investigated. The presence of HDTMA in clay minerals was confirmed by Fourier-transform infrared (FTIR) analysis that shows two absorption bands at 2927 cm−1 and 2860 cm−1 that corresponds to the asymmetric and symmetric stretching vibrations of C–CH2 of alkyl chain, respectively, and the band at about 1470 cm−1 was assigned to the vibration of trimethyl ammonium quaternary group C–N(CH3)3. Melt rheology of virgin PLA and nanocomposites performed by small amplitude oscillation shear measurement. Incorporation of HDTMA-MLS (1–7 wt%) into PLA matrix yields significant improvements in the elastic modulus (
Introduction
Clay finds a wide range of applications due to its natural abundance and a propensity to be chemically and physically modified to suit the technological needs.
1
Montmorillonite (MMT) is a dioctahedral smectite having plate-shaped particles with an average diameter of approximately 1 nm. Chemically modified MMT is a hydrated sodium calcium aluminium magnesium silicate hydroxide ((Na,Ca)0.33(Al,Mg)2(SiO10)(OH)2·
The objective of our work is to investigate the intercalation/exfoliation efficiency of MMT by modification with hexadecyl trimethyl ammonium bromide (HDTMA) and reinforcement of HDTMA-MLS with PLA to form nanocomposites and to analyse the rheological, thermal and mechanical characters of the nanocomposites. Incorporation of organoclay platelets into the PLA polymer matrix should improve the barrier, mechanical and thermal properties of the resulting nanocomposites to the extent that they meet the diverse needs of the packaging and other industries, while providing an environmentally friendly material that is commercially producible through extrusion and other conventional moulding process.
Experimental
Materials
PLA (92% leavo (
Modification of Na+ montmorillonite (Na+ MMT) using organic surfactant
Ion exchange method is an effective and easy method for modifying the clay surface and making it more compatible with polymer matrix.
Unmodified Na+ MMT clays of 10 g were dispersed in 300 ml of distilled water and placed in a ultrasonicator for an hour at 60°C, parallely 10 g of hexadecyl trimethyl ammonium bromide was dispersed in 300 ml of distilled water and placed in a ultrasonicator for 2 h at 60°C. After 2 h, about 1 ml of concentrated hydrochloric acid was added with aqueous dispersion of surfactant. Both dispersion of Na+ MMT and the surfactant were mixed together and subjected to vigorous stirring at room temperature for 2 h.
A dense precipitate formed were filtered by centrifugation and washed about four times with distilled water until no chloride was detected with 0.1 ml of silver nitrate solution. The precipitate was dried in an oven at 60°C for 72 h. The dried organoclays were grounded into powder form for further testing and characterization and used for making nanocomposites with PLA.
Preparation of PLA/HDTMA-MLS nanocomposite
PLA and HDTMA-MLS were melt mixed and the samples fabricated by injection moulding process for different testing analysis in a Xplore, 15 ml (a microcompounder cum injection moulding machine; DSM, RD Geleen Netherlands). The batch-type twin-screw extruder exerting axial forces up to 800 N operated at the screw speed of 30 r/min, torque of 6 N m−1, melt temperature of 185°C, injection pressure of 10 bar and mould temperature of 30°C with a batch size of 20 g. Prior to mixing and moulding, both clays and PLA were dried for 24 h at 80°C in an oven. Samples were moulded with 1 wt%, 3 wt%, 5 wt% and 7 wt%, loading of nanoclay for testing and characterization analysis.
It is significant to note that as expected, the above injection moulded specimens had excellent surface finish without any moulding defects with very close tolerance of specimen dimension which is a prerequisite to yield correct test results.
Characterization and testing analysis
Morphological analysis
Fourier-transform infrared spectroscopy
Infrared (IR) spectra of the sample (0.1 g) of HDTMA-MLS were obtained by the potassium bromide (KBr) method using a Nicolet-6700 (Thermo Fisher Scientific, Waltham, USA) Fourier-Transform Spectrometer in the region of 500–4000 cm−1. The spectrometer were equipped with a global IR source, KBr beam splitter and deuterated triglycine sulfate detector. For each spectrum, 64 scans were obtained with the resolution of 2 cm−1.
X-Ray diffraction
X-Ray diffraction (XRD) analysis was performed to determine the degree of intercalation and/or exfoliation of different types of powders/compositions, viz. Na+ MMT, HDTMA-MLS and PLA nanocomposites using Shimadzu XRD-700L diffractometer (scanning range 1–10°, graphite monochromator and a Cu Kα radiation source operated at 40 kV and 30 mA). Samples were scanned in continuous mode at a speed of 2° min−1. The basal spacing or
Transmission electron microscopy
Ultrathin sections of the PLA nanocomposites with a thickness of about 50–70 nm were microtomed at room temperature, using a Leica Ultracut UCT microtome (San Marcos, CA) with diamond knife (M/s Leica, UK), and then investigated by a transmission electron microscope (JEOL-1200 EX, JEOL Ltd, Akishima, Tokyo, Japan), using an acceleration voltage of 100 kV without any chemical staining.
Rheological measurement
Rheological measurements of molten composites were performed in a parallel plate rheometer (Mars III, Thermofisher Scientific, Walldorf Germany) with parallel plate geometry of 25 mm diameter. For each test, the PLA and nanocomposites were placed at the centre of the rheometer plate for 5 min for complete melting and temperature equilibrium before the actual measurements. The measurement was carried out within linear viscoelastic range by applying small-amplitude oscillation shear (SAOS) after temperature equilibrium to 190°C over a frequency range between 0.01 Hz and 10 Hz which enables the material to retain the structure. Rheological parameters, such as elastic or storage modulus,
Thermal analysis
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was performed using TA instruments (New Castle, DE USA) (TGA Q 50). Samples of 5 mg were heated from room temperature to 800°C at a rate of 10°C min−1 under a nitrogen (N2) atmosphere. TGA was performed on unmodified Na+ MMT, HDTMA-MLS and PLA nanocomposites.
Differential scanning calorimetry
The differential scanning calorimetry (DSC) measurement was conducted on a TA instruments (New Castle, DE, USA) (DSC Q 20). Samples of (10 mg) virgin PLA and nanocomposites were run at a scanning rate of 10°C min−1 in a N2 atmosphere. Samples of indium and cyclohexane were used to calibrate the DSC. Samples were heated from 20°C to 250°C at 10°C min−1.
Dynamic mechanical analysis
Dynamic mechanical behaviour of virgin PLA and its nanocomposites was measured using a TA instruments (New Castle, DE, USA) (dynamic mechanical analysis (DMA) Q 800) in the tension–torsion mode. The temperature dependence of dynamic storage modulus (
Mechanical properties
Test for the determination of mechanical properties was carried out in a standard temperature of 23 ± 2°C and 50 ± 2% relative humidity. The data reported are from the average of five specimens for each test.
Heat distortion temperature
HDT of virgin PLA and nanocomposites was tested in a Go Tech HV 2000-C3 (Gotech Testing Machines Inc., Taichung City, Taiwan) equipment at 66 psi. The samples (80.5 × 3 × 14 mm) were subjected to a deflection at 2 ± 0.2°C min−1 in a refrigerant liquid circulation.
Results and discussion
Morphological analysis
Fourier-transform infrared analysis
Figure 1 shows the Fourier-transform infrared (FTIR) spectra of unmodified Na+ MMT and HDTMA-MLS in the region of 4000–500 cm−1. The intensity of two bands shown in Figure 1 at 2927 cm−1 and 2860 cm−1, corresponding to the antisymmetric and symmetric CH2 stretching modes of amine, respectively, increases gradually with increase in the packing density of amine chain within the MMT galleries. It is believed that the intensity of the bands strongly depends on the packing density of amine chains within the MMT galleries. A broad band at 3627.4 cm−1 indicates OH stretching vibration of the structural OH groups due to sorbed water independent of the concentrations of the surfactant. The peak between 1470 cm−1 and 1516 cm−1 is due to the methylene (–CH2) scissioning mode at higher surfactant concentration.
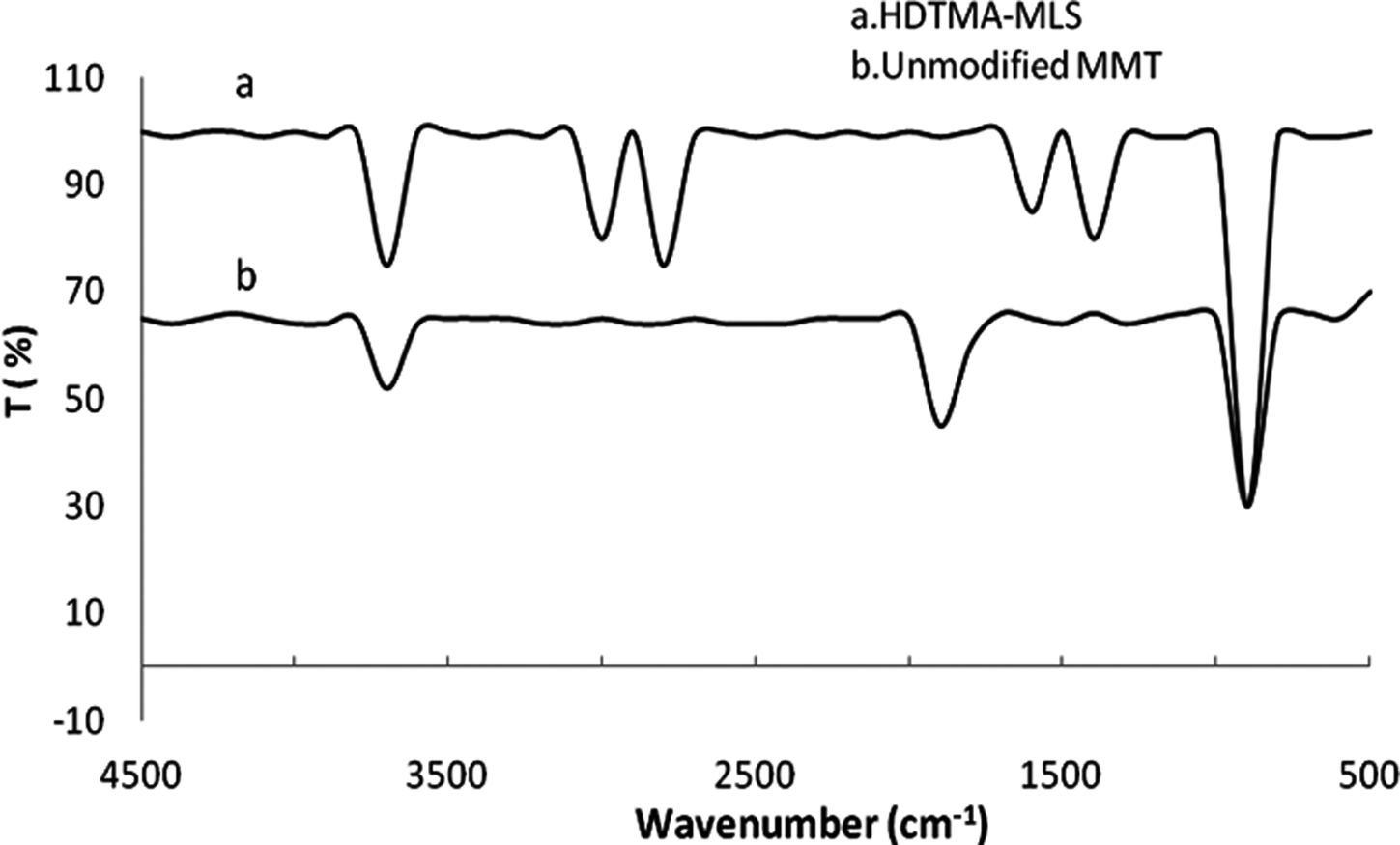
FTIR spectra of unmodified MMT and HDTMA-MLS powder. MMT: montmorillonite; FTIR: Fourier-transform infrared; HDTMA-MLS: hexadecyl trimethyl ammonium bromide-modified layered silicate.
Wide angle X-ray diffraction
Intercalation of HDTMA increases the interlayer spacing of the MMT. Similarly, intercalation of polymer chain into the silicate galleries usually increases the interlayer spacing of the HDTMA-MLS, leading to the shift in the XRD peak towards the lower value of 2
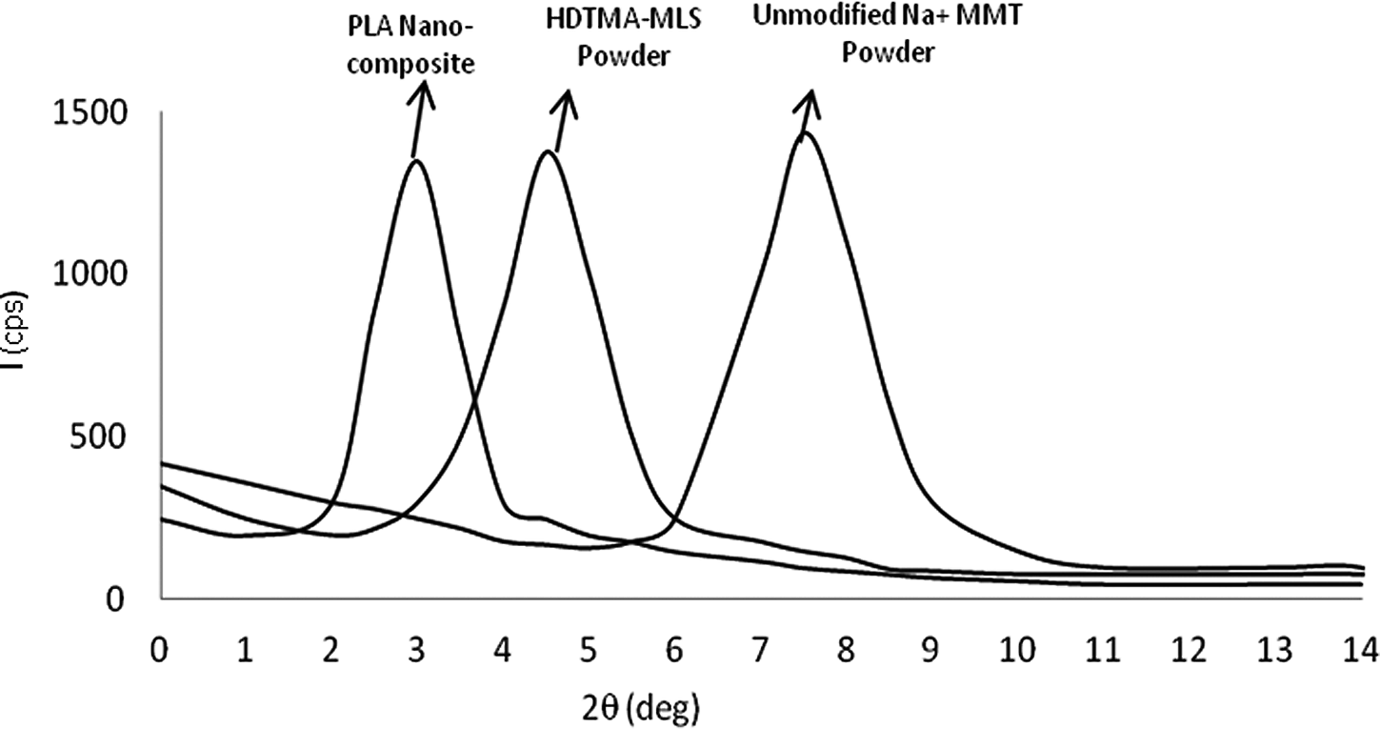
XRD pattern of unmodified MMT, HDTMA-MLS and PLA nanocomposites. MMT: montmorillonite; PLA: polylactide; XRD: x-ray diffraction; HDTMA-MLS: hexadecyl trimethyl ammonium bromide-modified layered silicate.
Diffraction angle and
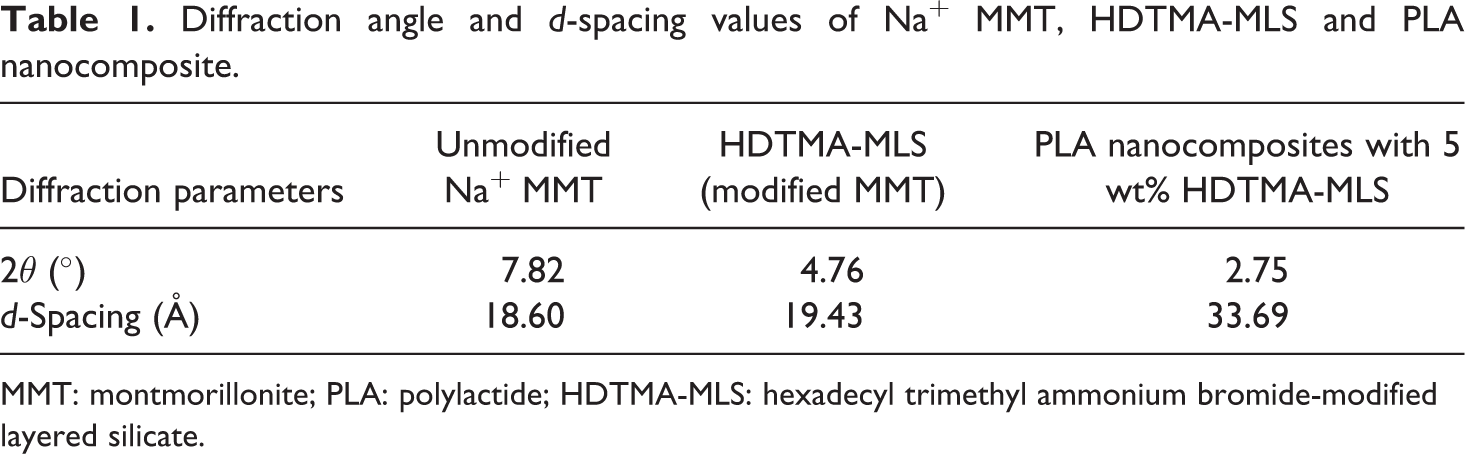
MMT: montmorillonite; PLA: polylactide; HDTMA-MLS: hexadecyl trimethyl ammonium bromide-modified layered silicate.
From Figure 2 it is clear that the XRD peaks for HDTMA-MLS and PLA nanocomposites with 5 wt% HDTMA-MLS are shifted to lower diffraction angle compared to the Na+ MMT and the corresponding
Transmission electron microscopy
Transmission electron microscopy (TEM) image of PLA/HDTMA-MLS nanocomposite is shown in Figure 3(a) and (b).
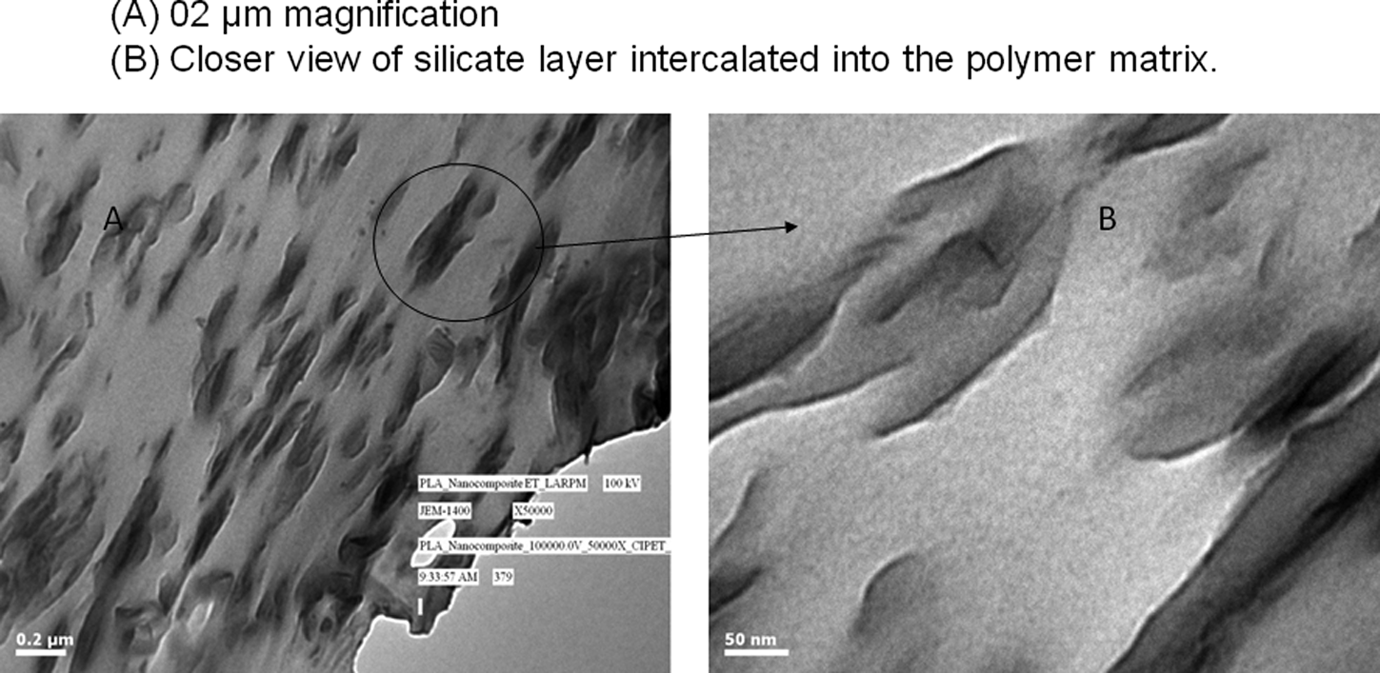
Transmission electron micrographs of PLA nanocomposites. (a) 02 µm magnification and (b) closer view of silicate layer intercalated into the polymer matrix. PLA: polylactide.
Figure 3 reveals that the intercalated clay galleries as well as stacks of agglomerated clays galleries were noticed within the PLA/HDTMA-MLS nanocomposites. Clearly, the large anisotropies of the stacked and agglomerated silicate layers are randomly distributed in the PLA matrix. The dark lines represent the thickness of the individual clay layers or agglomerates. Thick darker lines represent the stacked silicate layers due to clustering or agglomeration. Figure 3(b) represents the closer view of the silicate layers, which is strongly intercalated into the PLA matrix due to good compatibility, which leads to very good dispersion of silicate layers into the PLA matrix. Thus, PLA/HDTMA-MLS is a typical intercalated nanocomposite, which is in agreement with the results of XRD.
Rheological study
Oscillatory measurement is the most common dynamic method to study the viscoelastic behaviour of any material. Figure 4(a) and (b) shows the storage modulus (
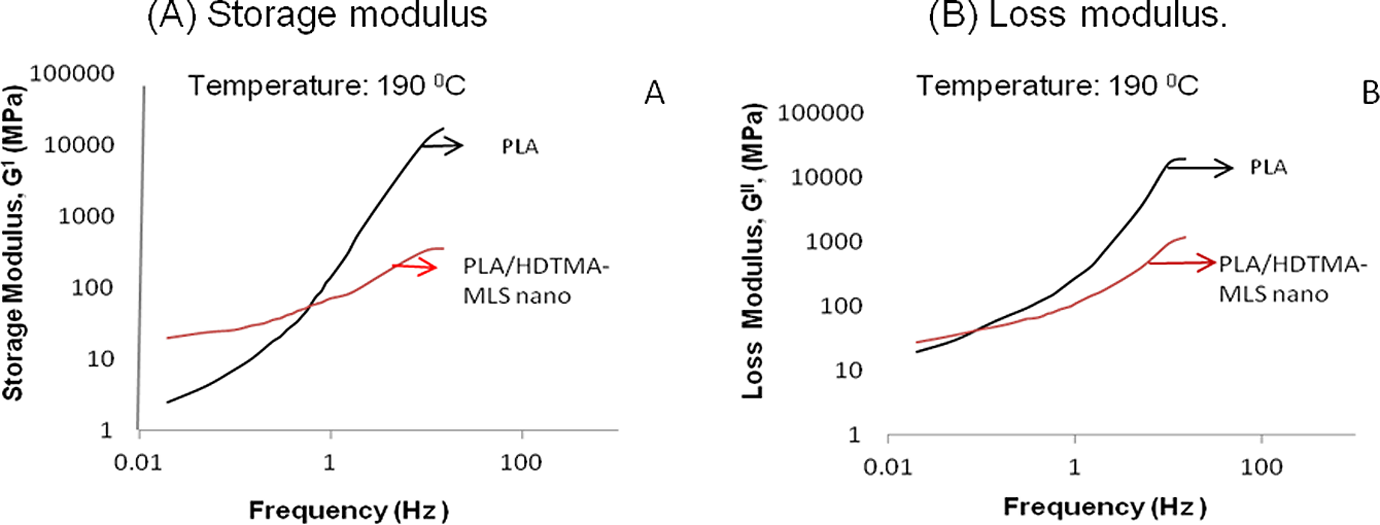
Dynamic mechanical behaviour of PLA and nanocomposite. (a) Storage modulus and (b) loss modulus. PLA: polylactide.
The
Figure 5 shows the complex viscosity (
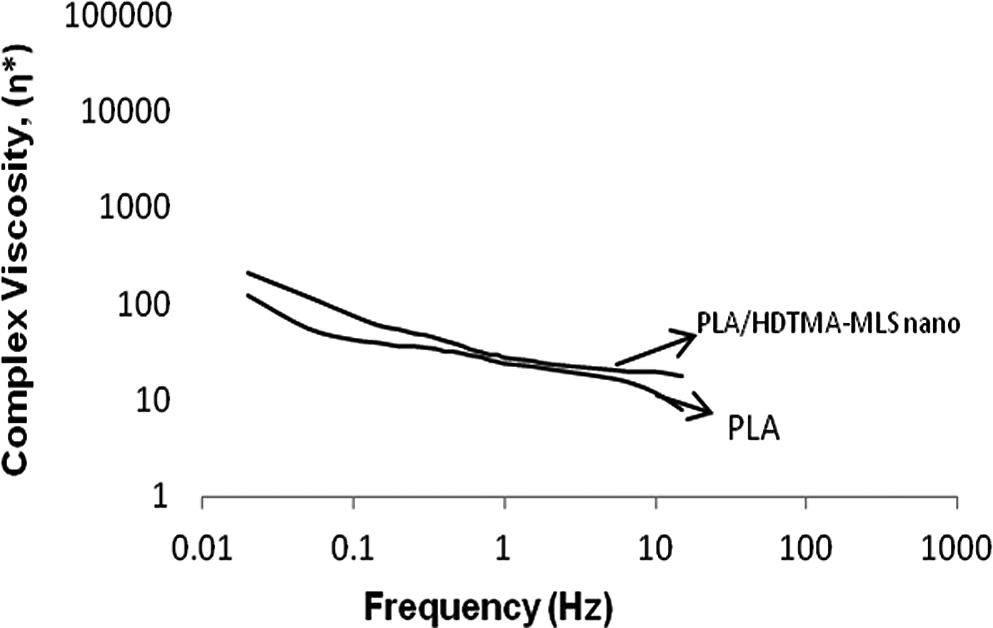
Complex viscosity (
The complex viscosity (
Thermal analysis
Thermogravimetric analysis
TGA thermograms of unmodified MMT, HDTMA-MLS and PLA nanocomposite are shown in Figure 6.
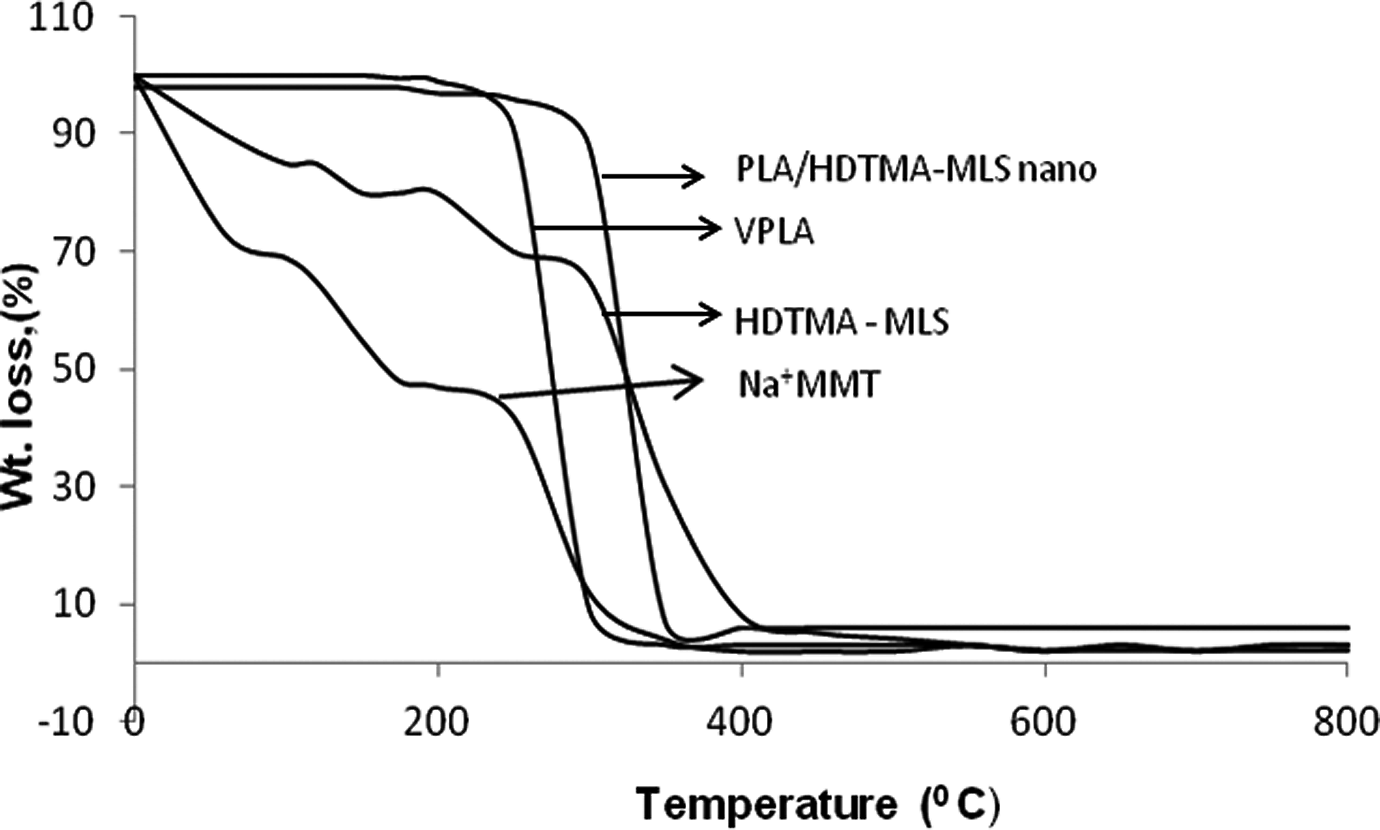
TGA thermograms of unmodified MMT, HDTMA-MLS, virgin PLA and nanocomposite. MMT: montmorillonite; PLA: polylactide; TGA: thermogravimetric analysis; HDTMA-MLS: hexadecyl trimethyl ammonium bromide-modified layered silicate.
It is observed that TGA thermogram of unmodified MMT shows three mass loss steps that occur between ambient to 103°C, 129°C and at 362°C. These mass loss steps are attributed to the desorption of water from the clay, dehydration of the hydrated cation in the interlayer and dehydration of MMT, respectively. Weight loss in the temperature range of 0–200°C for unmodified MMT was higher than that of HDTMA-MLS. This is due to the fact that most of the mass loss in this range was adsorbed water whose amount was very high in the unmodified Na+ MMT, whereas four steps mass loss were observed for the organoclays.
The first mass loss occurs between ambient to 103°C associated with desorption of water, the second one is at around 230°C, which is believed to be due to the loss of hydration water from the Na+, the third one is at around 400°C, attributed to the removal of the surfactant. The fourth weight loss is found at around 510°C, and this is assigned to the loss of structural hydroxyl groups within the clay, which indicates the thermal stability of the modified clays. The first two stages of mass loss are important for utility of such organically modified MMT in polymer-based nanocomposite material prepared via melt state processing. On the basis of the above TGA results, it is clear that modification of MMT distinctly shifted the onset degradation temperature of modified organoclays by about 10% towards higher value compared to that of unmodified clay. Hence, HDTMA-MLS can be used for making organic–inorganic hybrids by melt processing with thermoplastics, such as polyethylene, polypropylene, PLA, poly(hydroxybutyrate), etc. 28 Similar effect was reflected in the case of PLA nanocomposite reinforced with 5 wt% HDTMA-MLS, where the degradation temperature of nanocomposite appeared at 360°C compared to vinyl-terminated polylactide.
Differential scanning calorimetry
The DSC thermograms of PLA and its nanocomposites with 5 wt% HDTMA-MLS are shown in Figure 7.
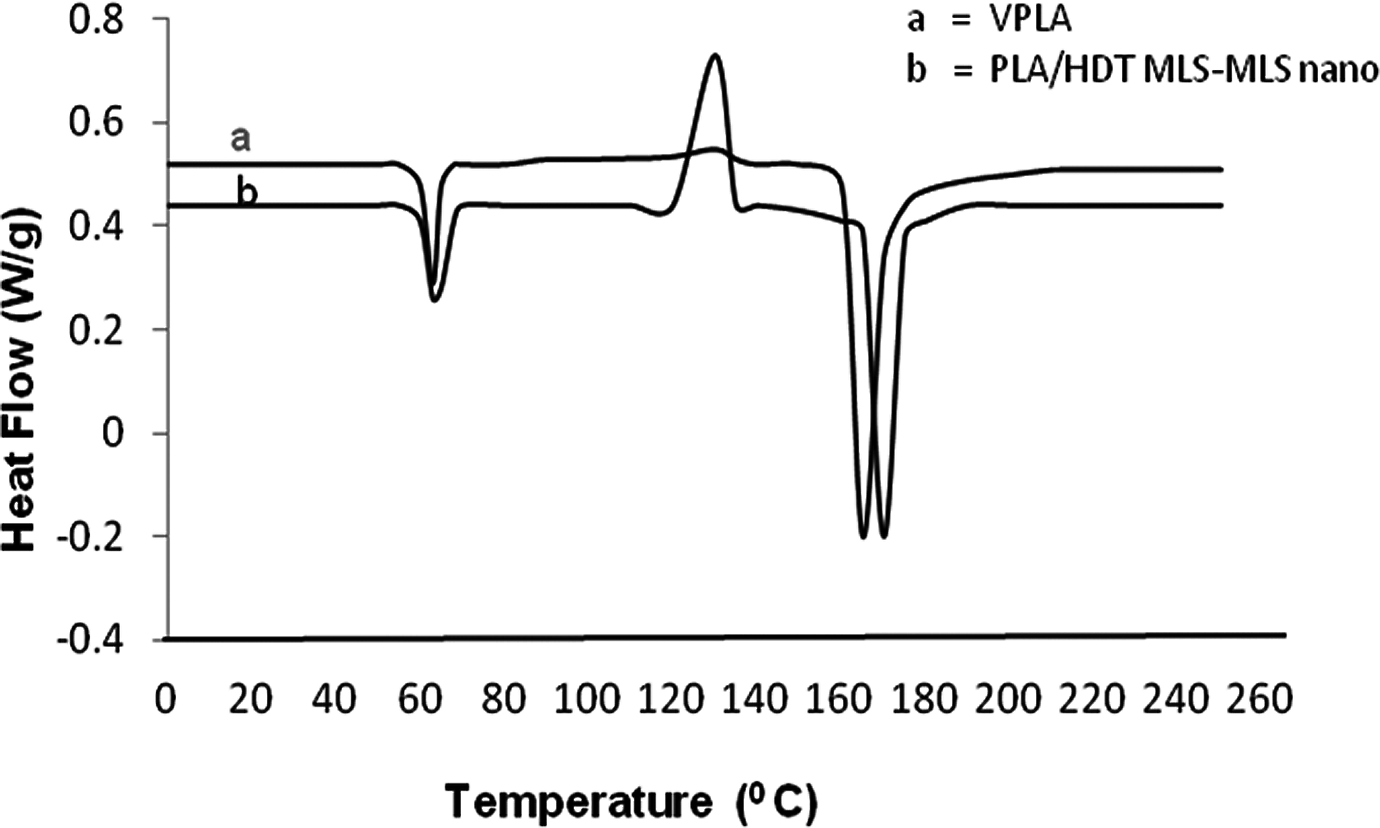
DSC thermograms of virgin PLA and nanocomposite. PLA: polylactide; DSC: differential scanning calorimetry.
An enthalpy change (▵
Dynamic mechanical analysis
Figures 8 and 9 show the temperature dependence of the dynamic mechanical properties of PLA and nanocomposites with 5 wt% HDTMA-MLS, and the results are presented in Table 2.
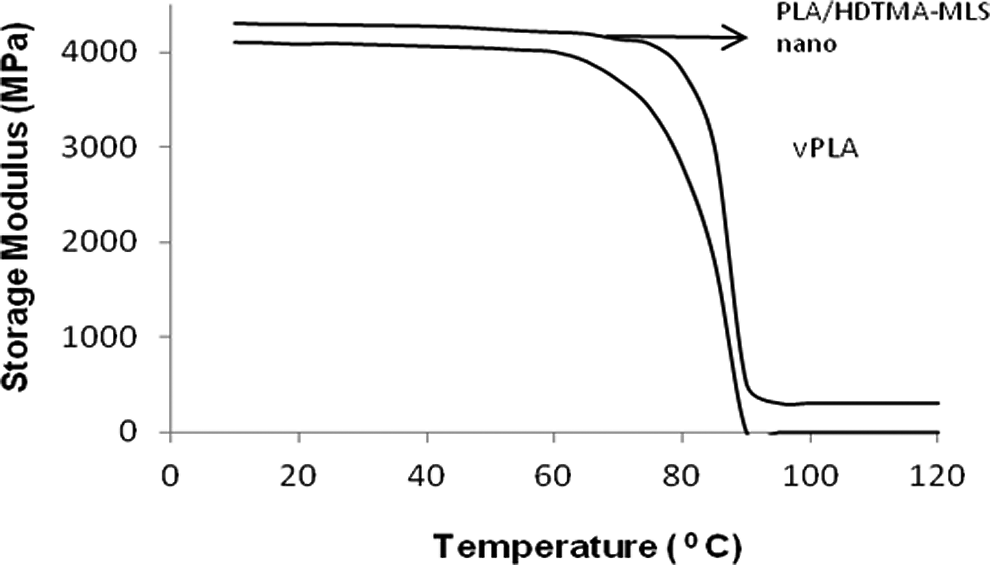
Storage modulus of PLA and nanocomposites with 5% HDTMA-MLS. PLA: polylactide; HDTMA-MLS: hexadecyl trimethyl ammonium bromide-modified layered silicate.
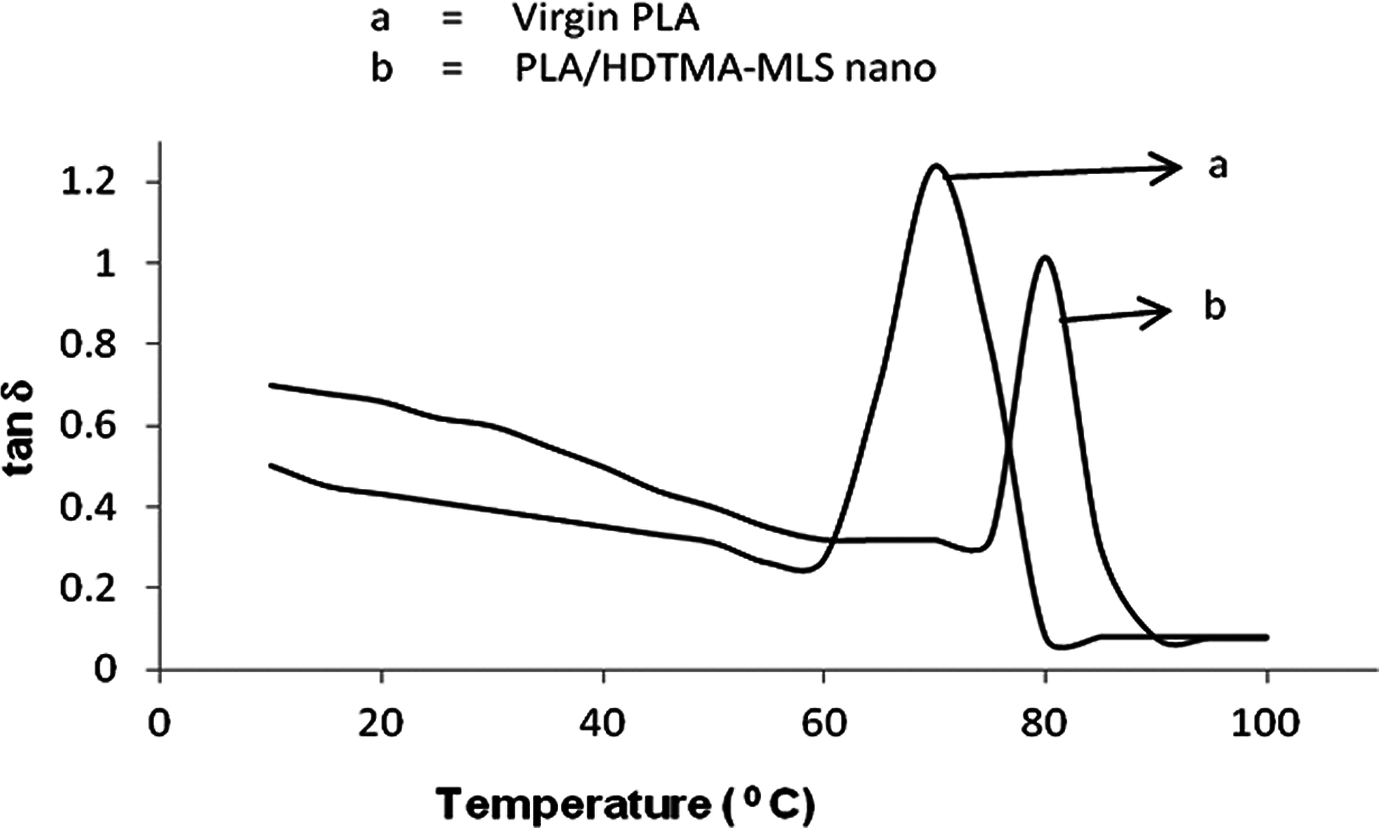
Damping factor of PLA and nanocomposites. PLA: polylactide.
Summarization of dynamic mechanical properties of PLA and nanocomposites with 5 wt% HDTMA-MLS.
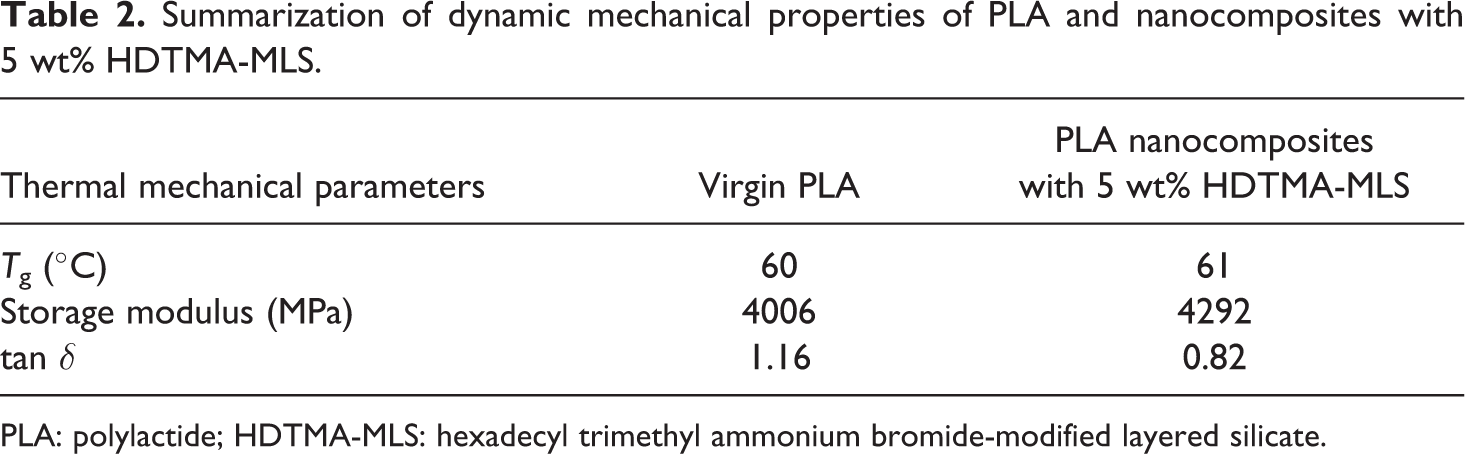
PLA: polylactide; HDTMA-MLS: hexadecyl trimethyl ammonium bromide-modified layered silicate.
From Figure 8 it is evident that significant enhancement in storage modulus,
The values given in Table 2 reveal no substantial change in the result of
Mechanical properties
Tensile properties
Organoclays can act as excellent reinforcing agents for polymer materials if dispersed uniformly in the polymer matrix. The effect of clay loading on the mechanical properties of PLA nanocomposites is presented in Table 3.
Mechanical properties of PLA/HDTMA-MLS nanocomposite.a
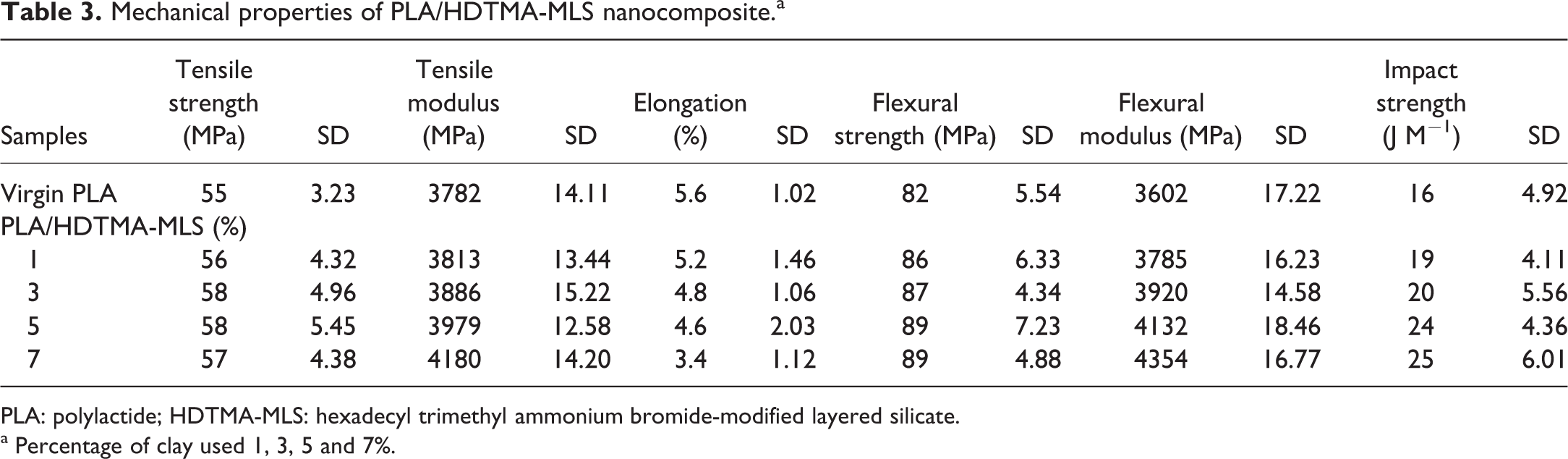
PLA: polylactide; HDTMA-MLS: hexadecyl trimethyl ammonium bromide-modified layered silicate.
a Percentage of clay used 1, 3, 5 and 7%.
Table 3 indicates a small increase in the tensile strength of PLA nanocomposites, however the tensile modulus of PLA nanocomposites was about 10% higher when compared to virgin PLA. It is believed that the enhancement in stiffness/tensile modulus of PLA nanocomposites may be due to the formation of supermolecular assemblies obtained by the presence of dispersed anisotropic-laminated nanoparticles, and also modification of MMT by functionalized organic cation can strongly interact with the matrix during curing. 26,29,30 Generally, incorporation of fillers/reinforcing agent into the polymer matrix will reduce elongation. As expected, the result shown in Table 3 indicates the decrease in the percentage elongation.
Flexural properties
While dealing with nanocomposites, many are generally interested in the tensile properties of the final materials. But there are very few reports concerning the flexural properties of PLA and nanocomposites. From Table 3, it is evident that incorporation of nanoclay into PLA matrix increases both flexural strength and modulus by about 9% and 21%, respectively, due to higher intercalation as supported by XRD analysis.
Impact properties
Impact values of PLA nanocomposites summarized in Table 3 reveal the enhancement in the value by about 56%. This may be due to the structural and morphological changes that promote chain mobility in the polymer nanocomposites and also the ability of large segments of PLA molecules to disentangle and respond rapidly to mechanical stress. 31
Investigation of the mechanical properties of PLA nanocomposites revealed that ultimate mechanical strength was obtained at 5 wt% loading of HDTMA-MLS; and hence, the materials was optimized at 5 wt%.
Heat distortion temperature
Figure 10 shows the effect of clay loading on HDT of PLA nanocomposites with 5 wt% of HDTMA-MLS.
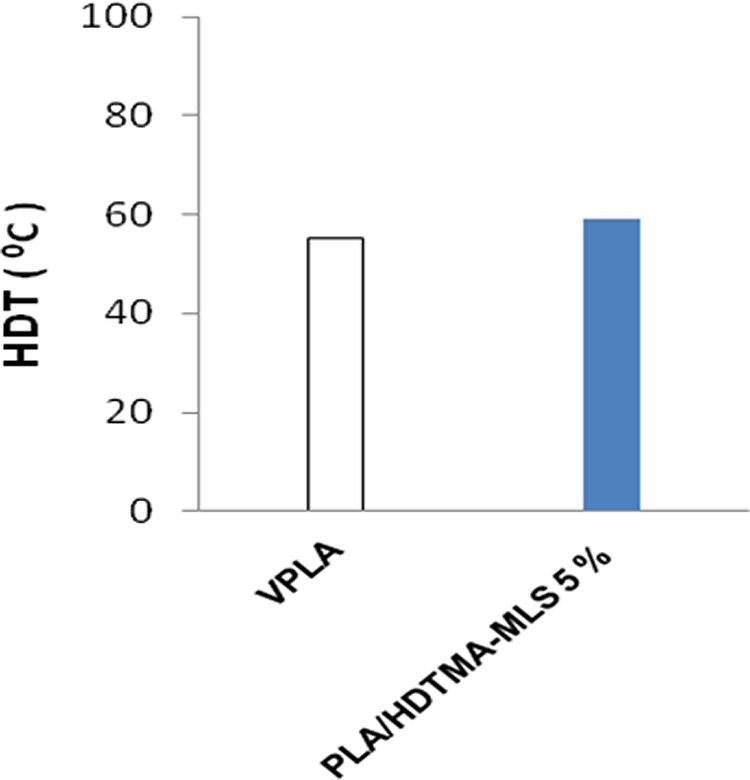
HDT of PLA matrix and nanocomposites with 5 wt% loading of HDTMA-MLS. PLA: polylactide; HDT: heat distortion temperature; HDTMA-MLS: hexadecyl trimethyl ammonium bromide-modified layered silicate.
The HDT values from Figure 10 reveal a little increase in the HDT of PLA nanocomposites with 5 wt% HDTMA-MLS (by 5 units) may be due to the formation of another crystal structure as supported by the XRD analysis where the peaks shifted to lower diffraction angle and due to some reinforcing effect of the clay.
Conclusion
MMT was modified using organic surfactant namely hexadecyl trimethyl ammonium bromide by ion exchange reaction and their intercalation morphology was investigated. Rheological, thermal, dynamic mechanical and morphological properties of the PLA/HDTMA-MLS nanocomposites have also been investigated. PLA/HDTMA-MLS nanocomposites were prepared using melt blending micromoulding. Tensile and flexural modulus and impact strength of nanocomposites show significant improvement (9%, 21% and 56%, respectively). Nanocomposites prepared at 5 wt% loading with HDTMA-MLS showed optimum mechanical performance. Rheological measurements revealed that PLA nanocomposites present a distinct solid-like behaviour as the clay loadings achieve 5 wt% with increase in melt elasticity (
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
