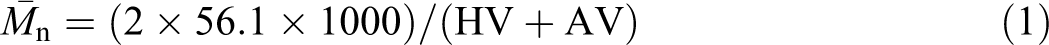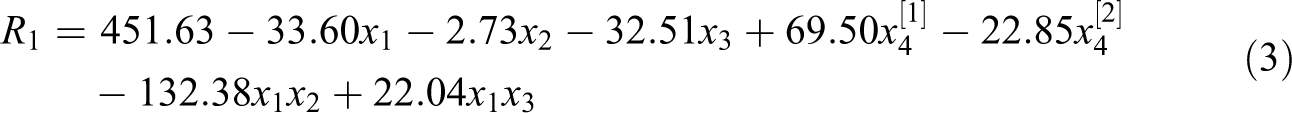Abstract
Poly(ethylene terephthalate) (PET) from off grades of industrial manufacturer was partially and thoroughly depolymerized using excess glycol to synthesize PET oligomers and bis(hydroxyethyl) terephthalate (BHET), respectively. Design of experiments and analysis of variance were applied for optimization of samples. Effects of reaction time, volume of glycol, catalyst concentrations and particle size of off grade PET on the yield of products were investigated based on the design of experiments. Thermal analysis of glycolyzed products is performed by differential scanning calorimetry. The optimum samples were also well characterized by Fourier-transform infrared spectroscopy and nuclear magnetic resonance spectroscopy (proton nuclear magnetic resonance spectroscopy and carbon nuclear magnetic resonance spectroscopy). The optimal conditions to synthesize PET oligomers were glycol/PET molar ratio of 2 with no catalyst using granule-shape PET for a 120-min glycolysis reaction time. Desired oligomer repeating units were 3–8. On the other hand, a reaction time of 180 min, weight ratio (catalyst to PET) of 0.25 wt% and glycol/PET molar ratio of 5 were obtained as the suitable conditions of BHET production. Then, end capped PET oligomers, as a compatiblizer for preparing PET nanocomposites, were produced via reaction between maleic anhydride and phthalic anhydride (MA/PhA) composition. The combination of reaction time of 106 min and PhA/MA molar ratio of 0.85 produced the best results based on d-spacing and peak intensity of nanocomposite samples. Moreover, the reaction of MA and BHET from glycolyzation of PET was successfully performed at 160°C and 190°C for 8 h.
Keywords
Introduction
Poly(ethylene terephthalate) (PET) is the most important member of aromatic polyesters widely used in textile and packaging, especially for soft drink bottles, fibers and films. Because of a large annual consumption, 1 efficient use of PET waste is an important research field for environmental protection. Consequently, processes for PET recycling were carried out almost parallel to the production of PET on a market scale. In recent years, there is an increasing interest in the use of PET waste for production of featured yields such as unsaturated and saturated polyester resins, 2 –8 polyurethanes, 9 –12 coating substances 13,14 and additives. 15,16
Among all different recycling techniques including chemical, mechanical and thermal recycling, the most suitable method based on ‘sustainable development’ is the chemical recycling, simply because it yields raw materials and other value-added crops from polymer waste. 17 The chemical degradation processes of PET waste are frequently demonstrated as follows: hydrolysis, 18 glycolysis, 19 methanolysis, 20 aminolysis, 21 ammonolysis 22 and other processes. 23,24 Glycolysis and methanolysis are commercially used; however, the most interesting one is the glycolysis method that yields related monomers or raw materials that could be reused for plastic production or other advanced materials. 25 Glycolysis method allows the use of very small amounts of reactants and to apply lower pressures and temperatures in comparison to other techniques such as thermal degradation 26 and supercritical methanol, 27 whereas acidic or basic hydrolysis method may corrode equipment and pollute environment. 28 Glycolysis of PET waste with ethylene glycol (EG) can lead to the production of bis(hydroxyethyl) terephthalate (BHET) monomer and PET oligomers. BHET has been extensively used in the production of polymeric substances such as unsaturated polyester resins (UPR), 7 rigid or flexible polyurethanes 9 –12 and polymer nanocomposites. 29 Vaidya and Nadkarni 30,31 studied the formation of UPR from PET glycolysis. Later on, they also polyesterified glycolyzed PET waste for the formation of polyurethanes. 32 Baliga and Wong 33 carried out the glycolysis of PET with EG using various catalysts. The products of glycolysis are mainly composed of monomer, dimer and trimer with hydroxyl end groups. The extent of depolymerization increased with increasing glycol/PET molar ratio. 34 Chen et al. 35 found that the glycolysis conversion is near to 100% at 190°C, 1.5 h glycolysis time and 0.002 mol cobalt acetate as a catalyst. However, the depolymerization reaction into monomer was incomplete. Xi et al. 36 showed the yield of BHET from postconsumer PET is 85.6% under the conditions of 3-h reaction time, EG/PET molar ratio of 5 and catalyst/PET weight ratio of 1%. Pardal and Tersac 37 described the kinetics of PET glycolysis by solvolytic reagents of different chemical structures with or without catalyst. They showed the glycolysis reaction rate is extremely based on the operating conditions such as reaction time, amounts of catalyst and glycol/PET molar ratio. In previous works, a simple glycolysis system was set up in the presence of zinc acetate, as an efficient catalyst, and the glycolyzed products of off grade PET were identified. 7,38
In recent years, there has also been a raised notice in fabrication of midway oligomers from PET waste in chemical industry. 39 –41 Partial PET glycolysis can be carried out under suitable preselected conditions, leading to low-molecular-weight oligomers.
In this study, optimization of glycolysis is presented. Therefore, a series of experiments is conducted particularly to determine the effects of reaction conditions on the depolymerization degree. In the first step, to continue a previous work, 7 BHET separation from glycolysis products was optimized based on Taguchi’s design of experiments. Then, the suitable sample, that is the purest one, was taken to react with maleic anhydride (MA) in order to synthesize UPR. In the next step, oligomers with 3–8 repeating units were produced. Hence, a lower efficient catalyst, manganese acetate, was used. Effects of reaction time, EG/PET molar ratio, catalyst concentrations and particle size of off grade PET on yield of partial glycolysis reaction were investigated based on Box–Behnken's design of experiments. The optimal conditions in order to synthesize suitable PET oligomers were obtained. Then, end capped PET oligomer as a compatiblizer for preparing PET nanocomposites was produced via reaction between MA/phthalic anhydride (PhA) compositions and optimized PET oligomers. Central composite design of experiments was applied. The analysis of variance (ANOVA) was also used for studying both glycolyzed products and functionalized PET oligomers.
Experimental
Materials
Amorphous off grade PET was obtained from Shahid Tondguyan Petrochemical Complex, Mahshar, Iran. The intrinsic viscosity [η] of PET was measured in a 60/40 (w/w) phenol/1,1,2,2-tetrachloroethane solution at 30°C. The [η] value of 0.5 dl g−1 that corresponds to viscosity average molecular weight
Measurements
Samples thermal behavior were studied with a Mettler Toledo 822 DSC (Greifense, Switzerland) equipped with a control and programming unit (microprocessor TC 10) and a calorimetric cell DSC 30 that allows a temperature scan from −170 to 600°C to analyze the glycolyzed products. Proton nuclear magnetic resonance spectroscopy (1HNMR) and carbon nuclear magnetic resonance spectroscopy (13CNMR) of samples were recorded with a Brucker DPX-500 (CT, USA) advance spectrometer operating at 500 MHz. NMR solvents were deuterated dimethyl sulfoxide and deuterated chloroform. Fourier-transform infrared spectroscopy (FTIR) spectra were obtained with a NEXUS 670 instrument from Nikolet (Madison, WI, USA). In addition, hydroxyl values (HV) were evaluated according to ASTM D2849 Method A. Furthermore, molecular weights of samples were measured by vapor pressure osmometry (VPO) on a Knaur K7000 (Berlin, Germany).
The products were analyzed by HV according to ASTM D2849 Method A. The acid values (AV) were determined by titration of 0.5 g sample dissolved in 25 ml pyridine with 0.5 N KOH solution using phenolphthalein as an indicator.
The number-average molecular weights of glycolysis products could then be calculated using equation (1).
42
Synthesis of poly(bis(2-hydroxyl ethyl) terephthalate-MA)
The reaction of BHET formation, along with other side products, is shown in Figure 1. PET waste flakes, totaling 100–200 g, equivalent to 0.52–1.04 mol repeating unit (mol. wt. 192), were added to 242.46–258.62 g EG (mol. wt. 68.11), dependent on the EG/PET molar ratio. The mixture was charged into a home-made reactor that was fitted with stirrer, reflux condenser, nitrogen inlet and pt-100 thermometer (Figure 2). The reactor was heated with an electric coil, and temperature was brought up to 200°C close to the boiling point of EG (198°C) and controlled by fuzzy logic method. Fifty gram of the homogenous final contents of the reaction were taken and allowed to cool to room temperature and 500 ml of distilled water was added while stirring vigorously. Water would dissolve any unreacted EG and the catalyst. After the first filtration using a suction filter funnel, the residual was transferred to a beaker containing 500 ml of distilled water and the suspension was boiled with stirring. This was intended to extract the BHET, which is known to be quite soluble in boiling water. After prolonged boiling, while still hot, it was quickly filtered (second filtration). Upon immersing the filtrate in an ice bath, the crystalline needles were precipitated and its melting point, after filtration and drying in a vacuum oven, was in the range of 108–111°C according to DSC graphs. After obtaining the suitable glycolyzed product, polyesterification of MA with glycolyzed product was performed in bulk in a four-necked flask, with a condenser, a nitrogen inlet, a mechanical stirrer and a thermometer. The reaction mixture was kept under nitrogen at 160°C for 2 h and then the temperature was increased to 190°C and kept for 6 h. The stirrer speed was 100 r min−1. Then the reaction was allowed to proceed under nitrogen atmosphere.
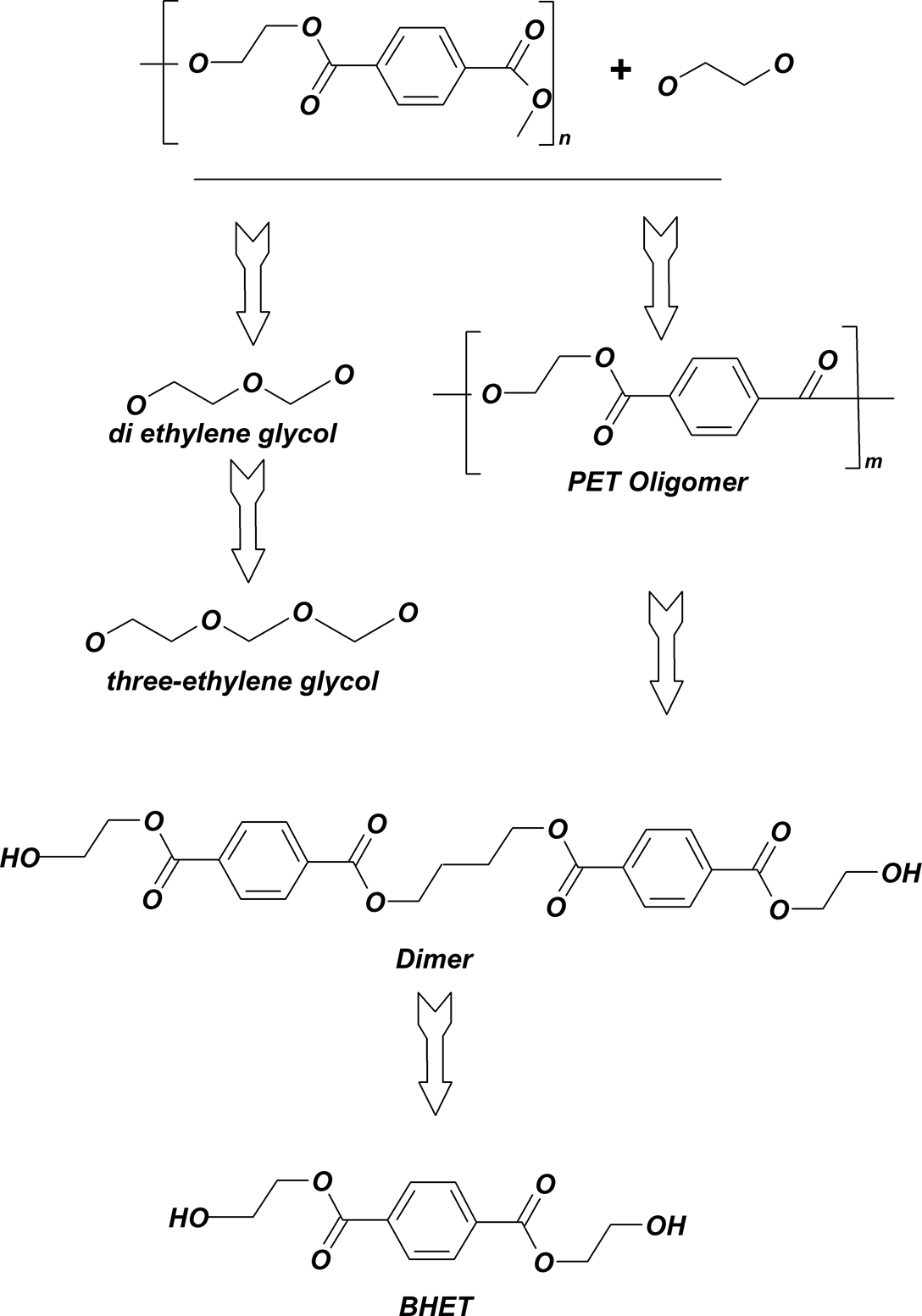
Reaction scheme for the glycolysis depolymerization of PET. PET: Poly(ethylene terephthalate).
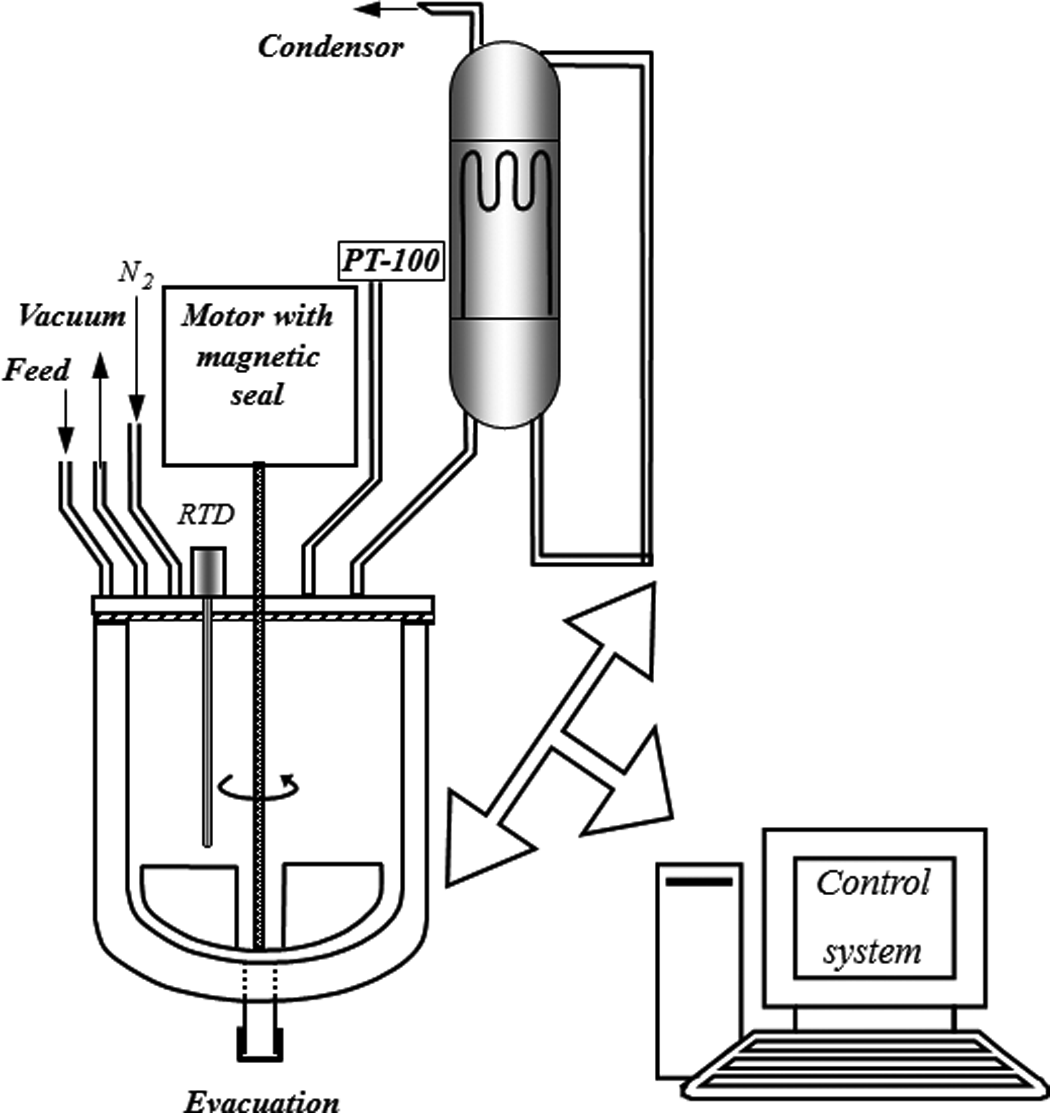
Schematic representation of the setup.
Synthesis of PET oligomers end capped with PhA/MA
Synthesis of PET oligomers was performed by partial glycolysis (alcoholysis) of PET by EG in the presence of catalyst (Figure 1). PET waste flakes, 200–250 g, equivalent to 1.04–1.3 mol based on repeating unit (mol. wt. 192), were added to 62–322.92 g EG (mol. wt. 62.11), with reference to EG/PET molar ratio (1, 2 and 4) in the presence of 0–0.5 wt% catalyst, based on the weight of PET. The mixture was charged into the reactor. At the end, all products were discharged and cooled to room temperature and distilled water was added while stirring vigorously. Water dissolved any unreacted EG and catalyst. After filtration with a suction filter funnel, residuals were transferred to a 70°C vacuum oven for 2 h. The mixture of PET oligomers and PhA/MA composition was reacted. The reactor temperature was set at 180°C nearby the melting point of oligomers. The stirrer speed was 100 r min−1. Then the reaction was allowed to proceed under nitrogen atmosphere.
Design of experiments
In this study, for synthesizing poly(bis(2-hydroxyl ethyl) terephthalate-MA) (UPR), four factors were used including x 1: reaction time (1, 2 and 3 h), x 2: EG/PET molar ratio (4,5 and 8), x 3: amount of catalyst (0, 0.25 and 0.5 wt%) and x 4: off grade PET particle size (fine, medium and granule). Therefore, three levels were also defined for each independent variable. In this section, R 1, based on the molecular weight of PET residue, was selected as the response of glycolyzed products.
In the case of synthesizing of PET oligomers, y
1: particle size of off grade PET (fine, medium and granule), y
2: EG/PET molar ratio (1, 2.5 and 4) and y
3: amount of catalyst (0, 0.25 and 0.5 wt%) were selected as the most effective factors. Then, in the partial glycolysis reaction, three levels were determined for each factor. It was determined that the full factorial method needed 33 test runs for each reaction time (60 and 120 min). Therefore, this number of tests would be too costly and time consuming. Hence, the Box–Behnken’s design of response surface method was applied to indicate necessary experiments. Two sets of experiments were performed for the glycolysis time equal to 60 and 120 min. Although different parameters may be selected as response variable such as reaction yield, in this research, R
2, based on the following equation, was selected as the response of glycolyzed products:
where w i is the peak area, T i is the melting point and T opt is the desired melting temperature which is corresponding to the suitable oligomers. In this study, T opt is chosen near to 180°C that is melting point of oligomers with 3–8 repeating units. These oligomers can be strongly recommended for nanocomposite materials based on PET/nanoparticle. 25 Therefore, this response is the weighted melting temperature difference. Then reaction time and PhA/MA molar ratio were selected as the most important factors of end capping of PET oligomers. Central composite method was applied to design the experiments. In end capping PET oligomers with PhA/MA, z 1: reaction time (43–106 min) and z 2: PhA/MA molar ratio (15–85%) were selected as the most effective factors. In this section, d-spacing, R 3, and peak intensity, R 4, of nanocomposite samples were selected as responses of products.
Results and discussion
Preliminary tests were conducted for 0.5 to 8 h. The results show that the yield reaches the highest value at 3 h of glycolysis. It seems that the equilibrium state is achieved at 3–5 h of glycolysis. DSC results confirm the production of BHET and dimer. Figure 3 shows DSC scans of the glycolyzed products. The melting onset temperature of glycolysates (110°C) agrees very well with the known melting point data (BHET 109–110°C) (Figure 3(a)). DSC scan of glycolyzed products was also taken before the second filtration (Figure 3(b)). This DSC result consists of two endothermic peaks: the first peak, located at around 110°C, can be associated with the melting point of BHET, whereas the second peak, located at around 142°C, is quite broad and is, subsequently, not readily identifiable with the melting of BHET dimer (Figure 3(c)). If the glycolyzed product can be assumed to consist predominantly of BHET and dimer, as indicated by the melting points and HV, then its DSC scan seems to suggest some interactions between the monomer and dimer at elevated temperatures. On the other hand, the results of thermal analysis and number-average molecular weight of oligomers calculated by HV suggest that glycolysis of PET includes at least three steps (oligomers, dimer and monomer). Oligomers in different glycolysis conditions show multiple melting regions. The filtrate fraction after two-step filtration was labeled as the ‘B’ fraction that is BHET rich fraction. The rest glycolysates henceforth was labeled the ‘O’ fraction that is dimer and other oligomers rich fraction.
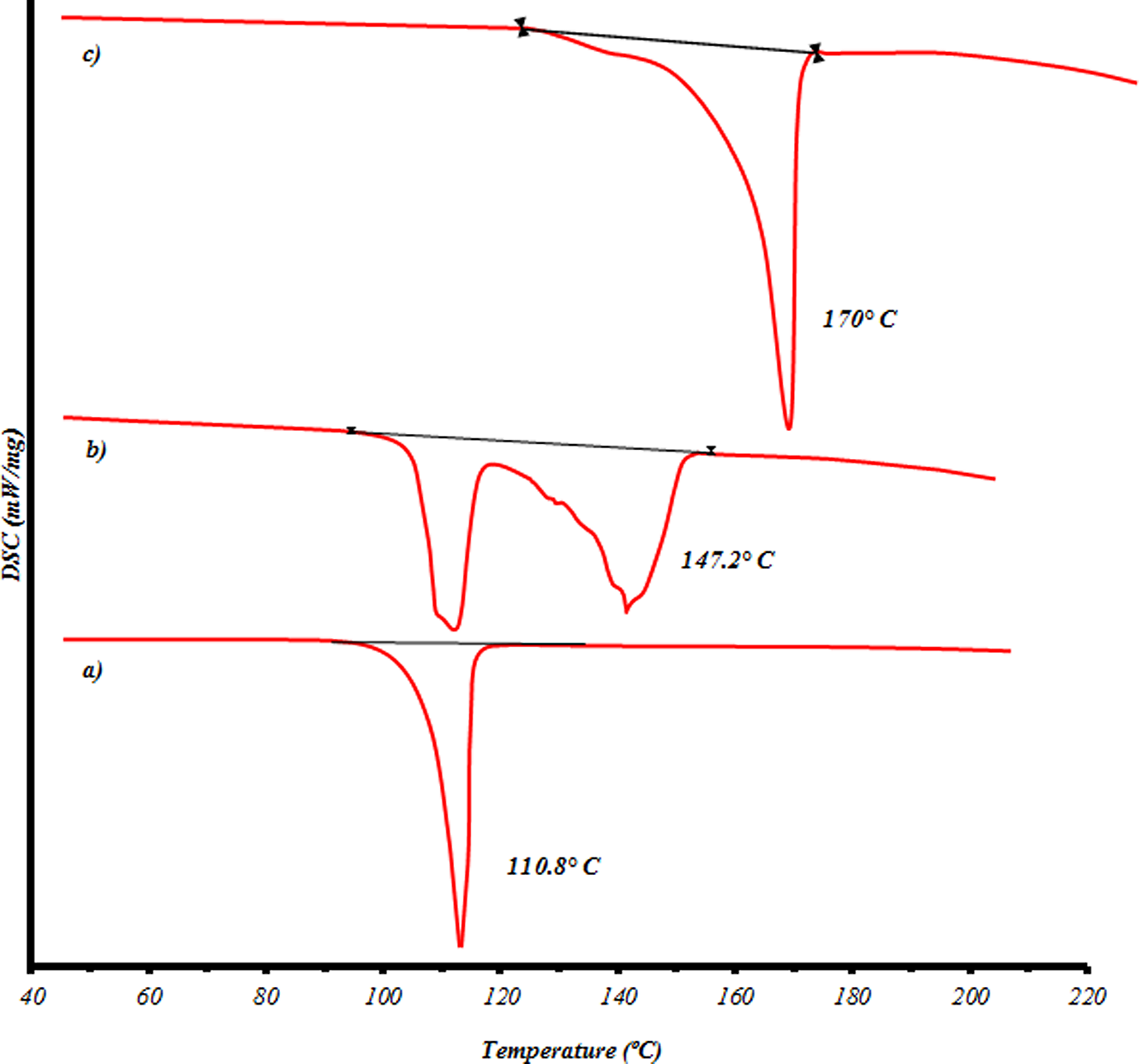
Differential scanning calorimetric scans of glycolyzed products: (a) after two main filtrations (BHET); (b) before the second filtration (BHET and dimer) and (c) dimer. BHET: bis(hydroxyethyl) terephthalate.
Synthesis of poly(bis(2-hydroxyl ethyl) terephthalate-MA)
Experiments based on the Taguchi's design, and their results are described in Tables 1 and 2. HV, and molecular weights based on equation (1) and VPO results are given, too. HV of the ‘B’ fraction were between 342.31 and 439.8 mg KOH g−1. These results are close to the HV of BHET; therefore, the ‘B’ fraction mainly consists of BHET. HV of the ‘O’ fraction were 121–253 corresponds to HV of dimer and higher. This implies that the ‘O’ fraction was a mixture of dimer and other oligomers. Based on the ANOVA, a reduced two-factor interaction model, with R
2 = 0.9939, was derived. This model in terms of coded factors is:
Results of Taguchi’s experimental design based on ‘B’ fraction.
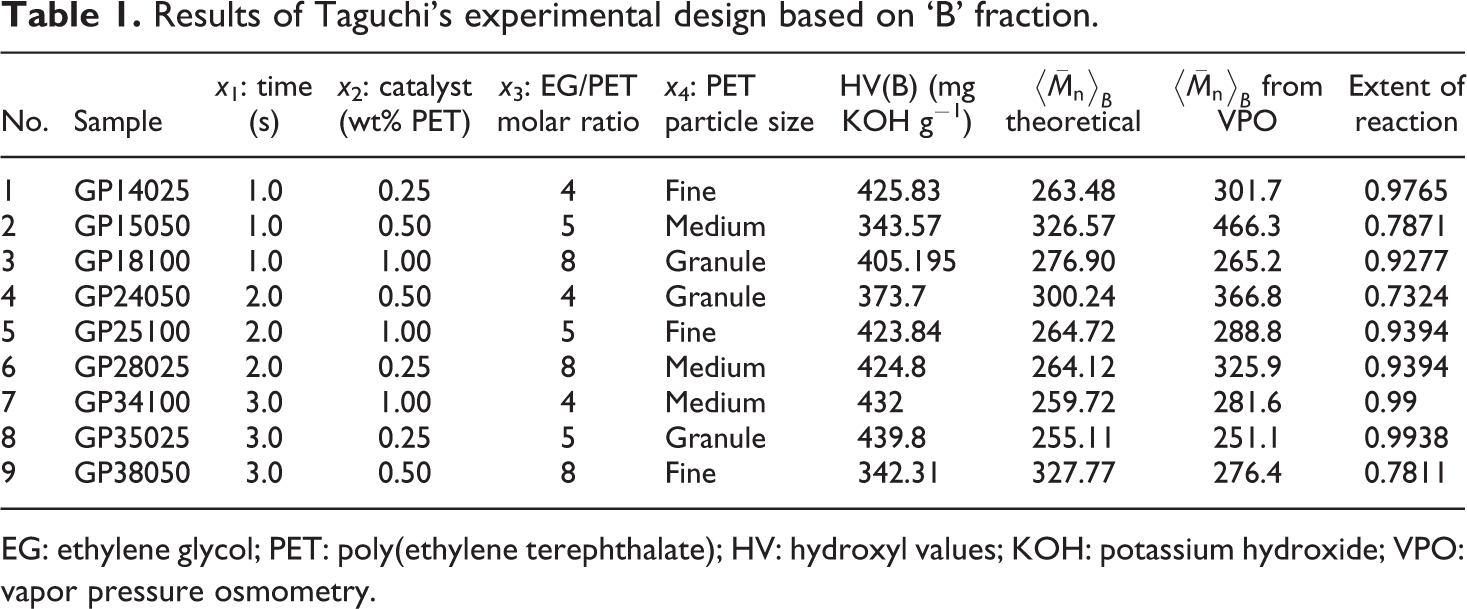
EG: ethylene glycol; PET: poly(ethylene terephthalate); HV: hydroxyl values; KOH: potassium hydroxide; VPO: vapor pressure osmometry.
Results of Taguchi’s experimental design based on ‘O’ fraction.
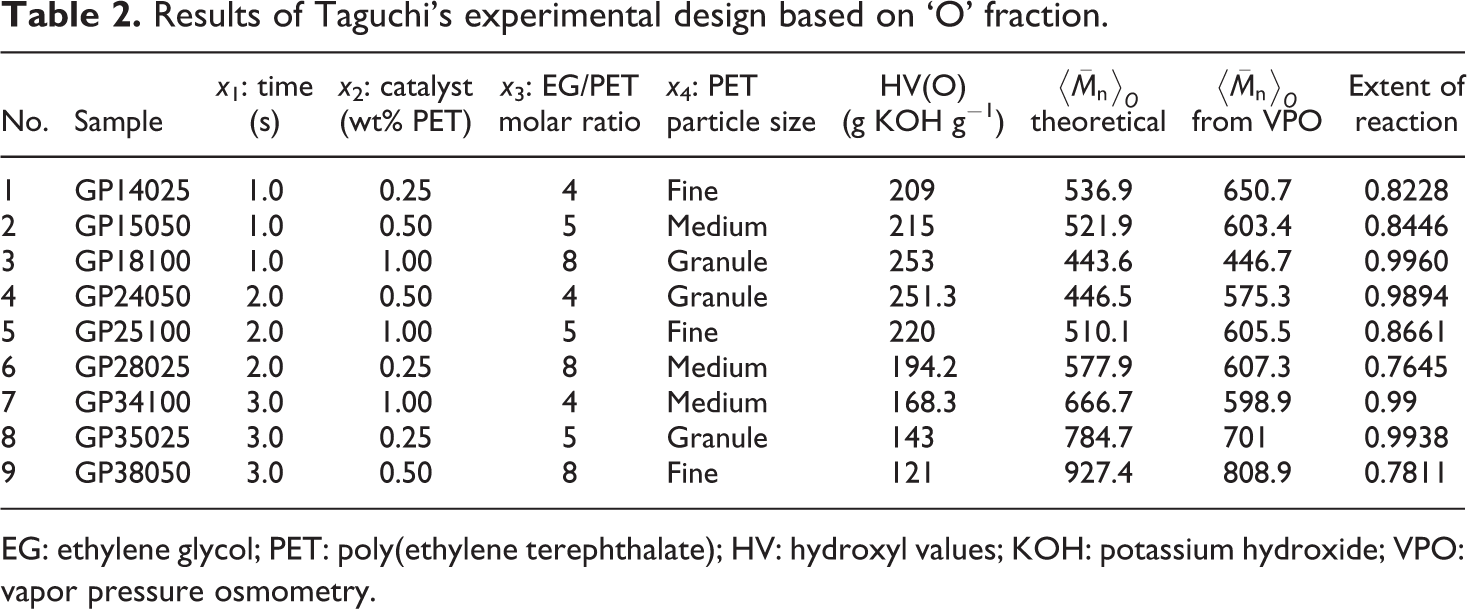
EG: ethylene glycol; PET: poly(ethylene terephthalate); HV: hydroxyl values; KOH: potassium hydroxide; VPO: vapor pressure osmometry.
Moreover, final equations in terms of actual factors are: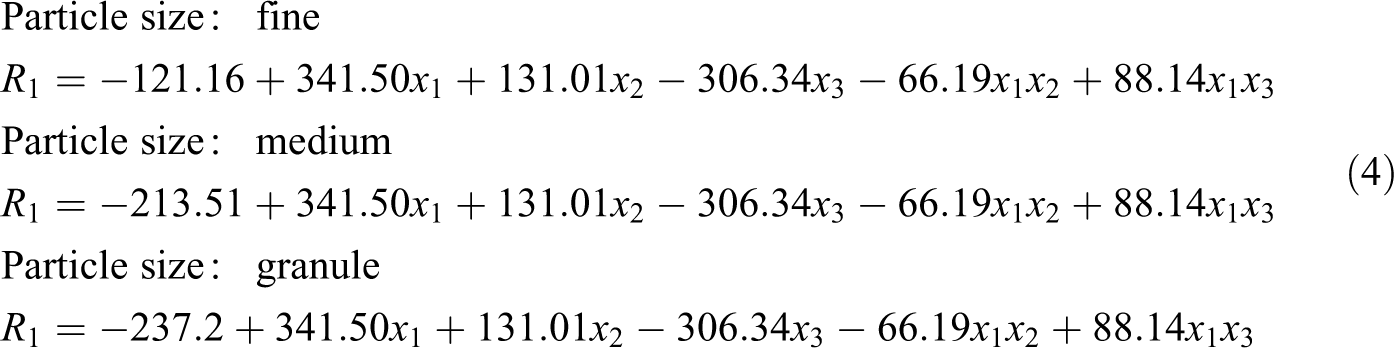
where x
1, x
2, x
3 and x
4 are reaction time, EG/PET molar ratio, catalyst concentration and particle size of PET, respectively. A term with a p-value less than 0.05 is significant. In this case, all terms are significant, except x
1 and x
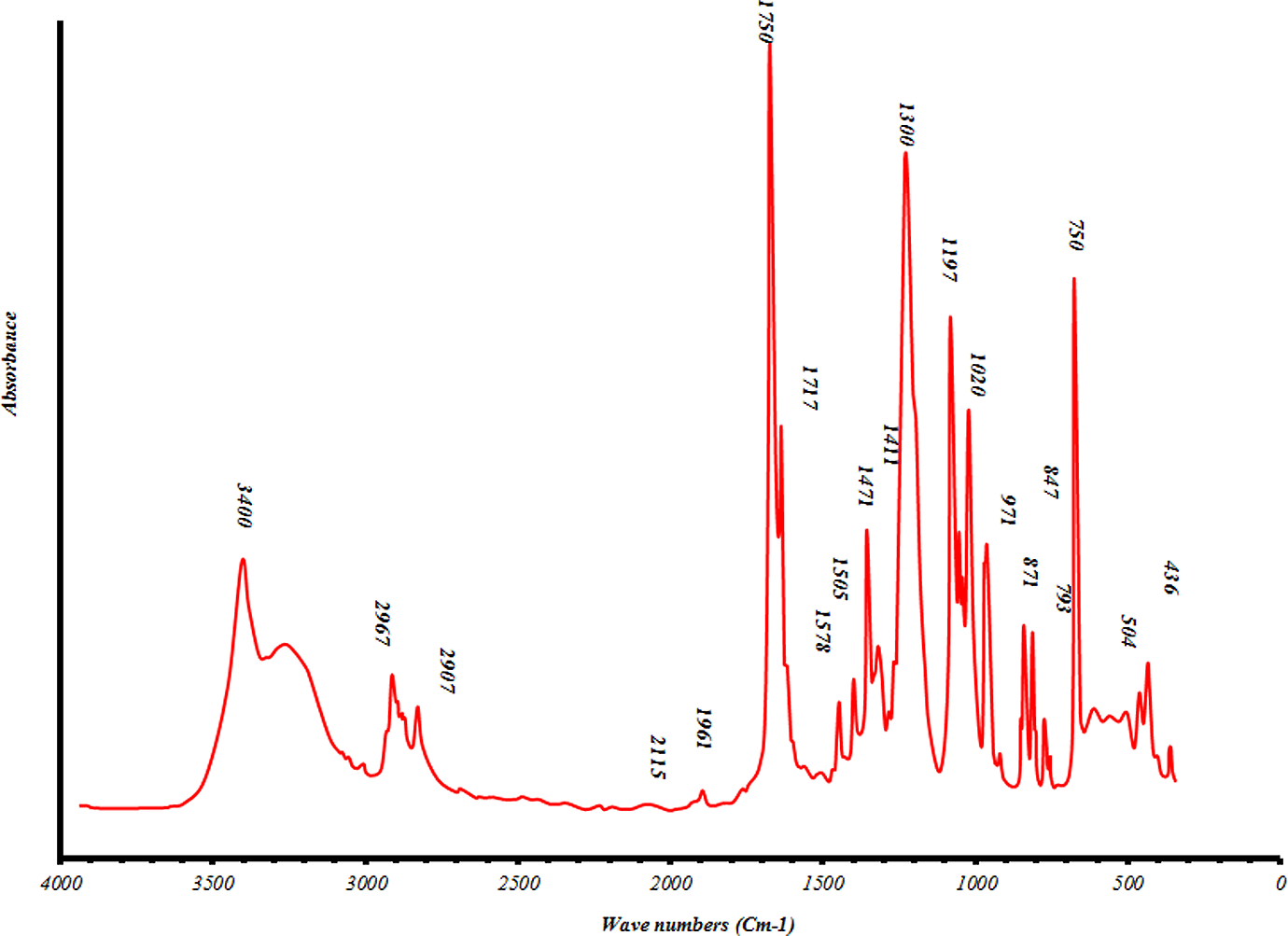
2. The values between 0.05 and 0.10 are considered marginally significant. Values greater than 0.1 indicate the model terms are not significant. Moreover, it is seen that there is no significant interaction between three factors, but there are some dual interactions between parameters. Based on the data in Table 1, glycolysis reaction rate increases significantly with glycolysis time, the amount of catalyst and EG/PET molar ratio. However, particle size does not have strong effect. BHET was achieved by the suggested sample GP35025, with the reaction time of 3 h, an EG/PET molar ratio of 5, and amount of catalyst, 0.25 wt% based on PET weight. FTIR spectrum was obtained for ‘B’ fraction of sample GP35025 (Figure 4). The peaks at 3400, 3000–2800, 1750 and 1300 cm−1 are corresponding to OH, C–H, C=O and C–O stretching, respectively. A sharp peak at 750 cm−1 in FTIR spectrum is related to the para-substituted aromatic ring. In order to characterize the ‘B’ fraction of GP35025, 1HNMR and 13CNMR spectra were also performed. 1HNMR and 13CNMR spectra of this fraction are presented in Figures 5 and 6, respectively. The signal at δ 8.15 ppm indicates the presence of the four aromatic protons of terephthalic residue. The two triplets between δ 4.4 and 3.9 ppm (8H) are characteristic of the methylene protons CH2–OH of EG and COO–CH2, respectively. Number of terephthalic base units in oligomers is calculated by area under related signal: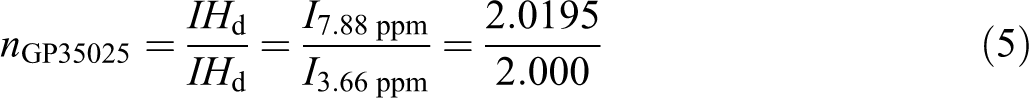
FTIR spectrum of sample GP35025. FTIR: Fourier-transform infrared spectroscopy.
1HNMR spectra of ‘B’ fraction of sample GP35025. 1HNMR: proton nuclear magnetic resonance spectroscopy.
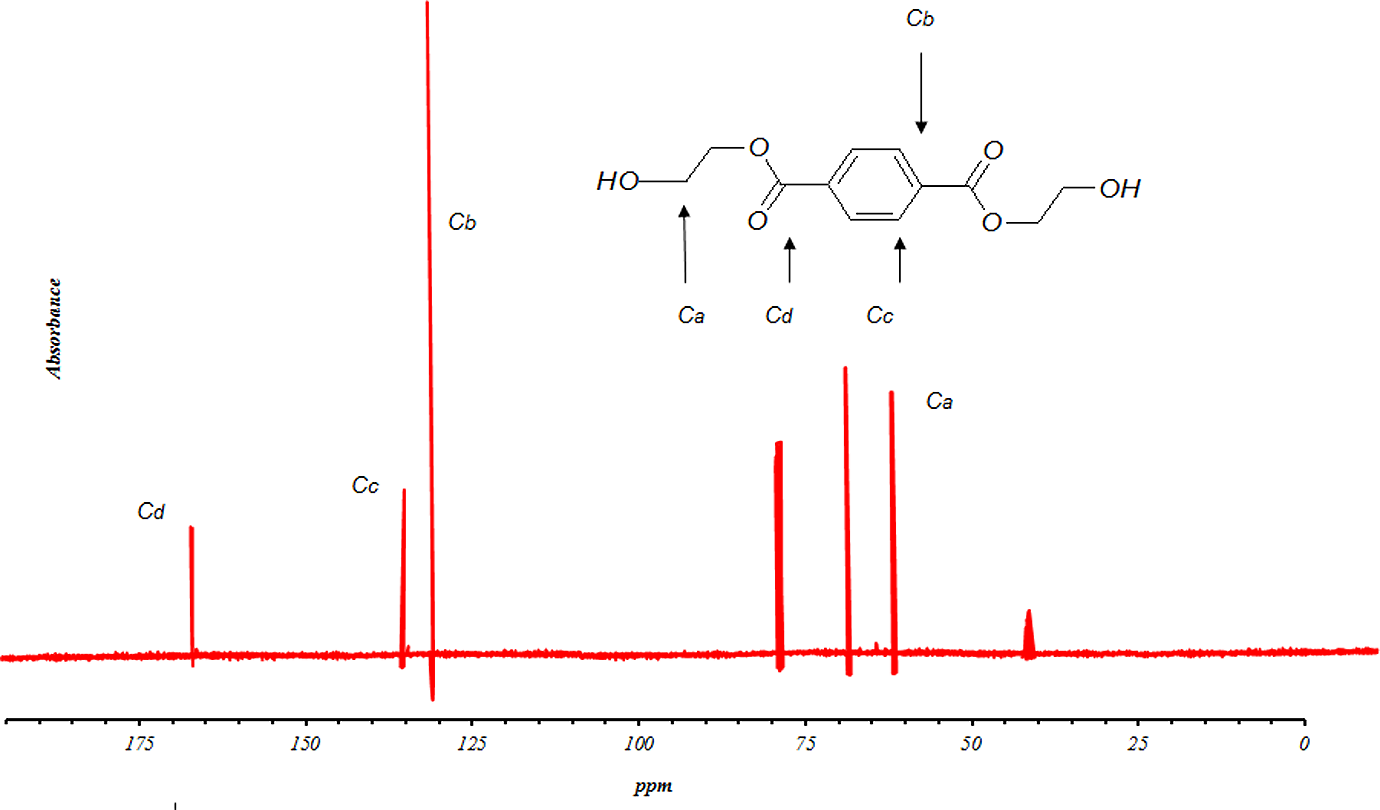
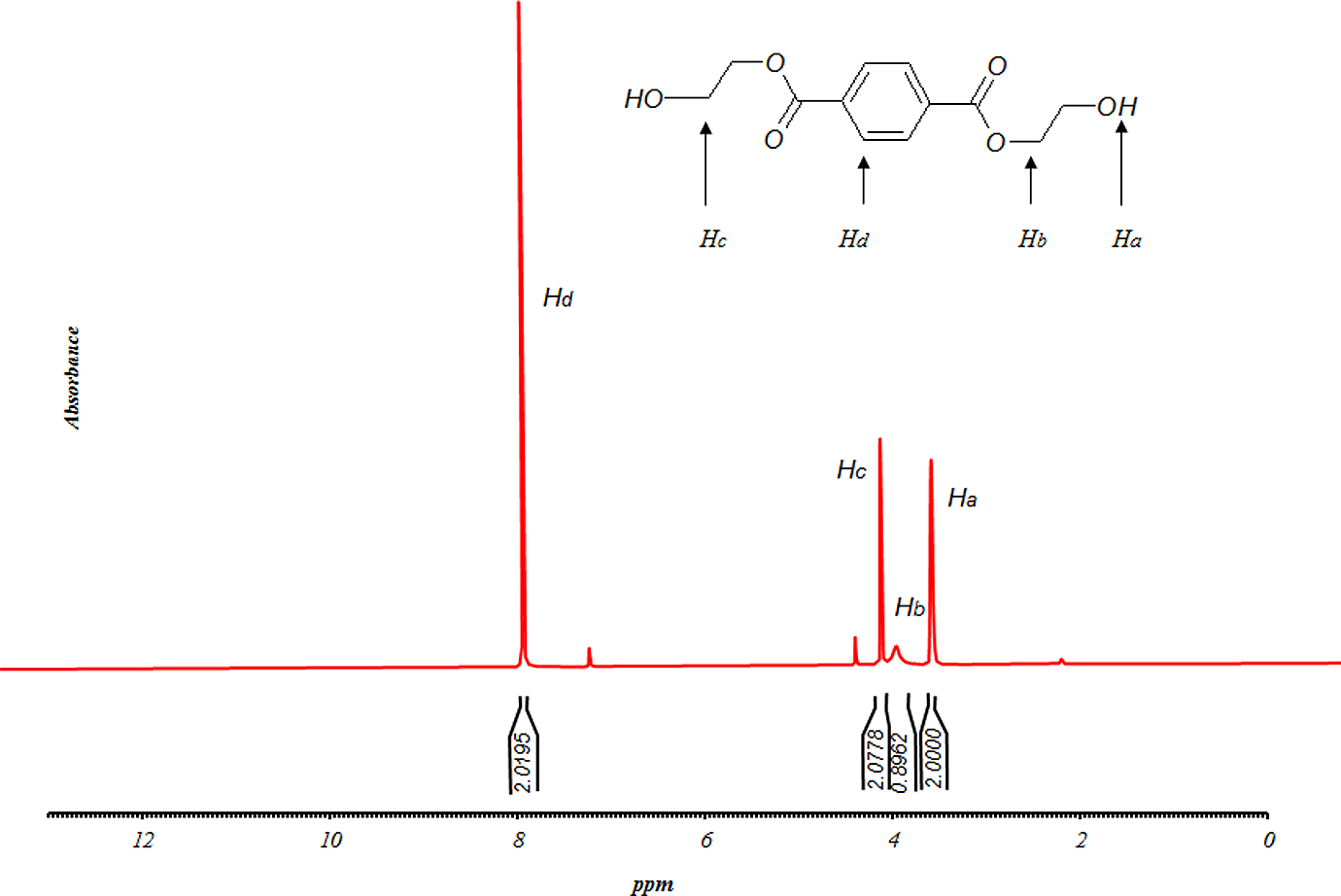
13CNMR spectra of ‘B’ fraction of sample GP35025. 13CNMR: carbon nuclear magnetic resonance spectroscopy.
where H d represents the integration value of the signals of aromatic protons, H a represents the integration value of the signals of methylenic protons in an α of an hydroxyl end group and I x ppm represents the integration value of the peak centered at x ppm. So, it is possible to determine the number-average molecular weight of the sample. The number-average molecular weight of sample GP35025 was determined to be 254 g mol−1. As seen in Figure 6, the 13CNMR result has been obtained as follows: δ (ppm) = 63.38 (CH2 bonds); δ (ppm) = 129.96 (CH in benzene loop); δ (ppm) = 134.1 (fourth type carbon in benzene loop); δ (ppm) = 164.65 (esteric group). The other peaks are related to the carbons in the solvent. The signals of 13CNMR were in accordance with those predicted in 1HNMR.
Finally, polyesterifications of the glycolyzed product with MA were produced under different conditions. It is apparent from these data that the extent of polyesterification increases by time. Figure 7 shows molecular weight increases with reaction time; however, HV and AV decrease continuously. Products were further characterized by FTIR, 1HNMR and 13CNMR (Figures 8 to 10, respectively). Based on the FTIR spectra for the sample UP190800, the peaks at 3400, 2968, 1715, 1620–1680 and 727 cm−1 are related to OH, C–H, C=O, C=C and =CH stretching, respectively. The higher intensity and broadening of the C–O stretching around 1000–1300 cm−1 in the polymer compared with sample GP35025 indicate that more ester groups are present and confirm that polyesterification has occurred. Furthermore, the sharp peak around 750 cm−1, related to para-substituted aromatic ring, is characteristic of PET. On the other hand, the NMR results were obtained as follows: 1HNMR: δ (ppm) = 2.3 (CH2 in a carboxyl group and methylenic carbon); δ (ppm) = 2.65 (CH2 between an ester and a carboxylic function); δ (ppm) = 3.6 (CH2 between two ester functions); δ (ppm) = 6.8 (vinylinic protons) and δ (ppm) = 7.8–8.07 (CH on benzene ring). Also the 13CNMR results were obtained as follows: δ (ppm) = 28.86–30.6 (CH3 in acetone solvent); δ (ppm) = 63.38 (CH2 bonds); δ (ppm) = 129.86–130.96 (CH in benzene loop); δ (ppm) = 133.49–134.38 (fourth type carbon in benzene loop); δ (ppm) = 164.56–166.2 (esteric group) and δ (ppm) = 205.63–206.84 (carbonyl group in acetone solvent).
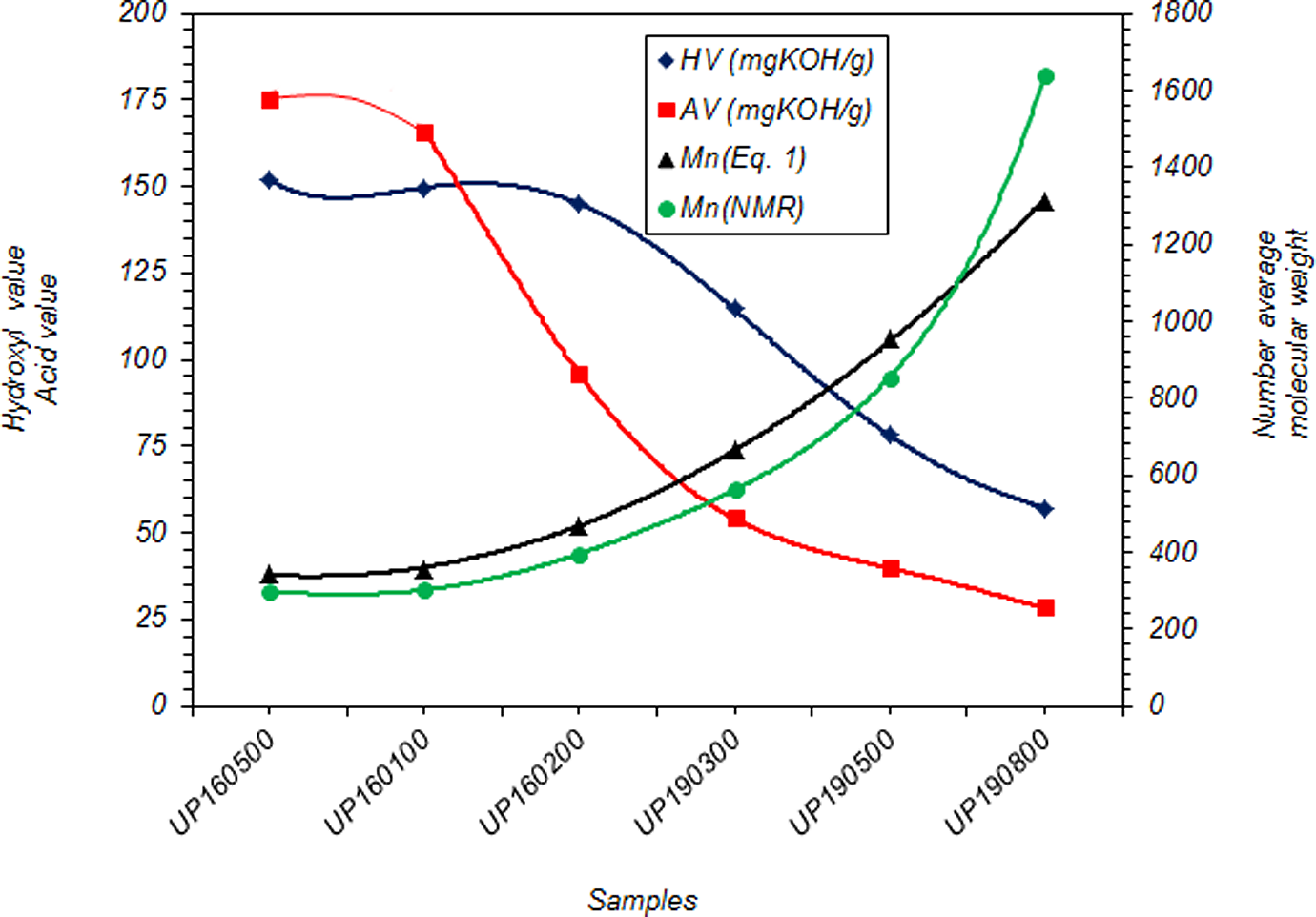
HV, AV and Mn of UPR products as a function of glycolysis time (UP16010 and UP160200 are UPR products yielded at 160°C during 1 and 2 h, respectively. UP190300, UP190500 and UP190800 are UPR products yielded at 190°C during 3, 5 and 8 h, respectively). HV: hydroxyl value; AV: acid value; Mn: number-average molecular weight; UPR: unsaturated polyester resins.
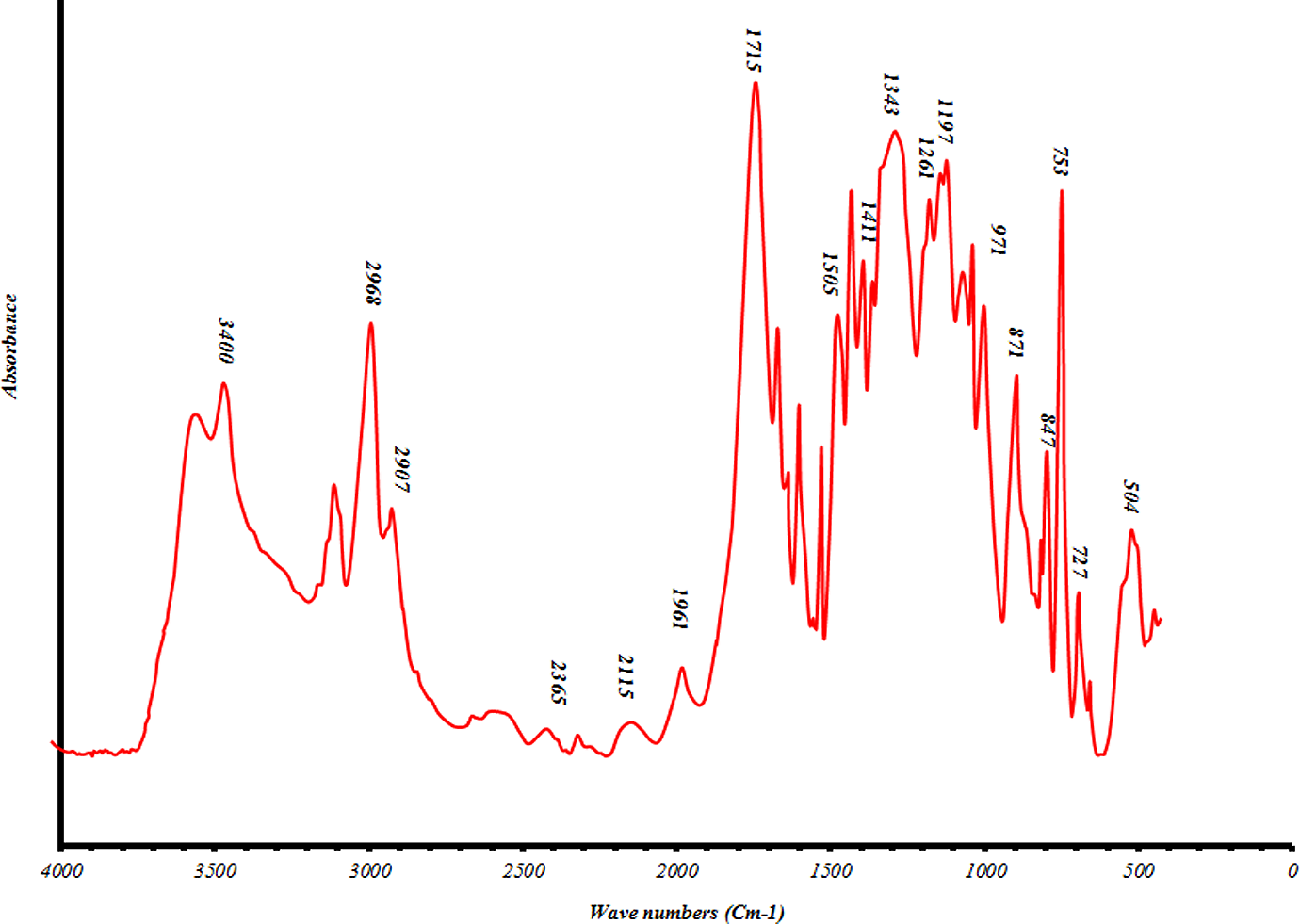
FTIR of sample UP190800. FTIR: Fourier-transform infrared spectroscopy.
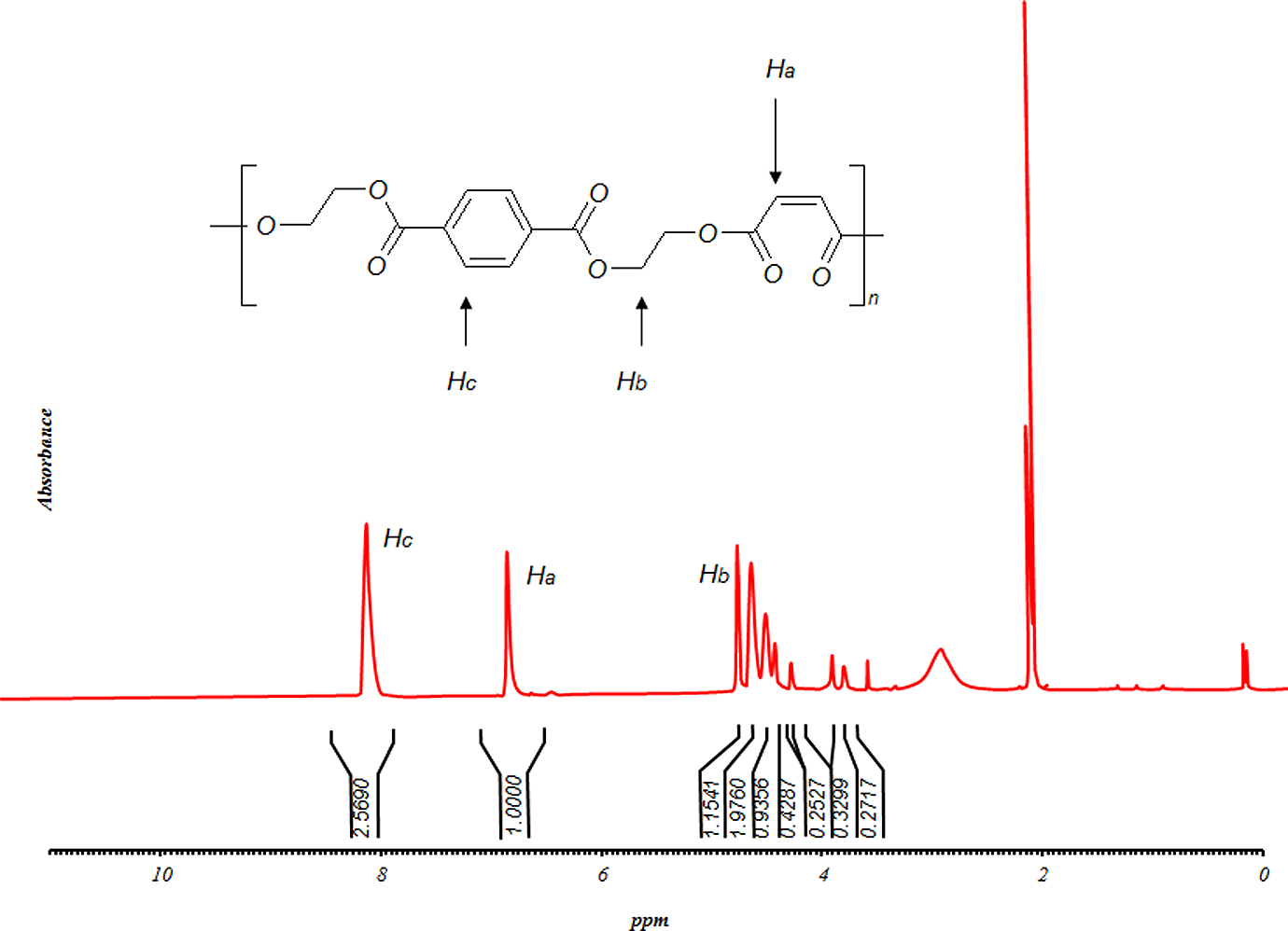
1HNMR spectrum of sample UP190800. 1HNMR: proton nuclear magnetic resonance spectroscopy.
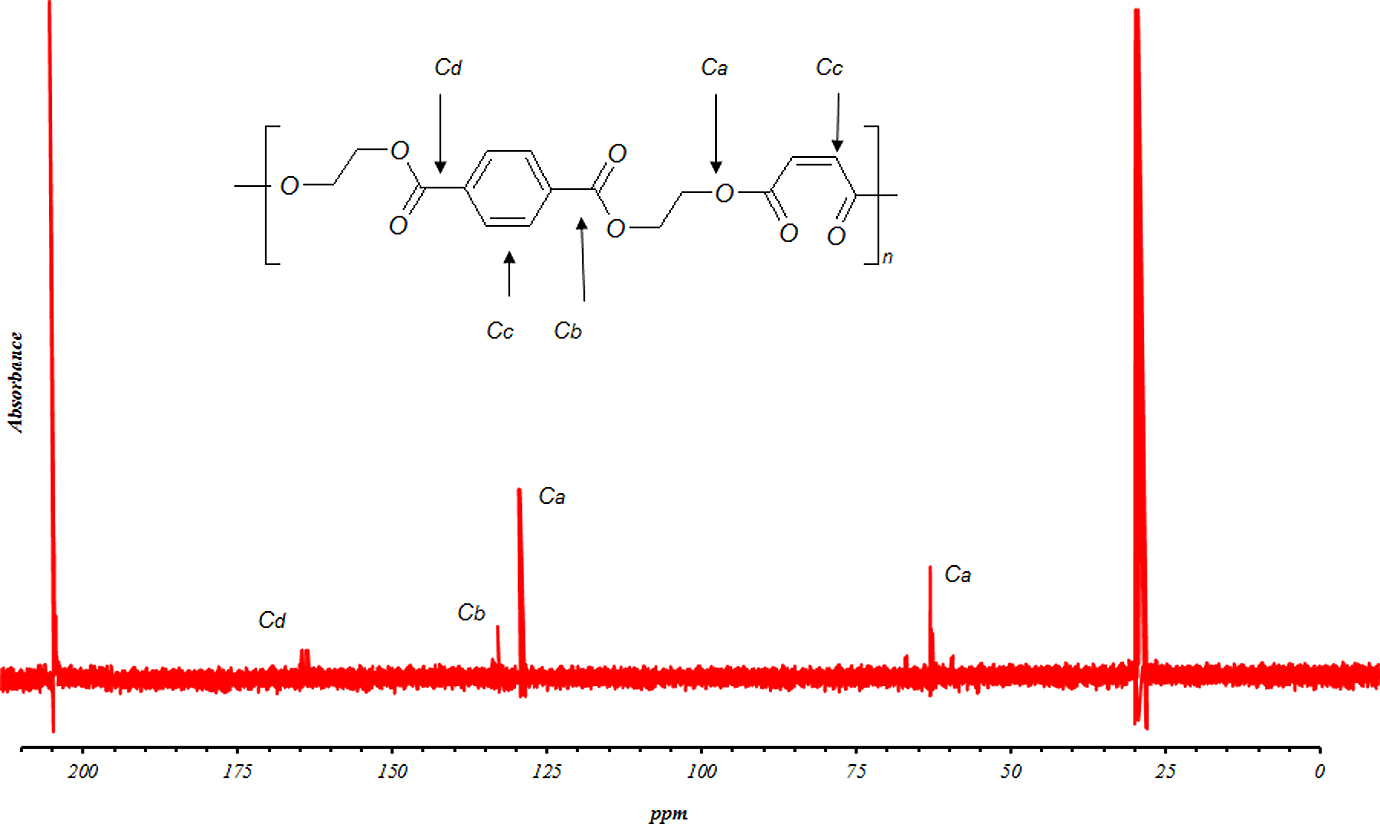
13CNMR spectrum of sample UP190800. 13CNMR: carbon nuclear magnetic resonance spectroscopy.
Synthesis of PET oligomers end capped with PhA/MA
Table 3 gives the list of experiments based on the Box–Behnken design. By performing experiments, the measured responses after 60 and 120 min are given in Table 3. Based on the ANOVA, a quadratic model, with R
2 = 0.9949 and R
2 = 0.9980, is derived for 60- and 120-min glycolysis reaction time, respectively. For 60-min glycolysis reaction time, the final equation in terms of coded factors: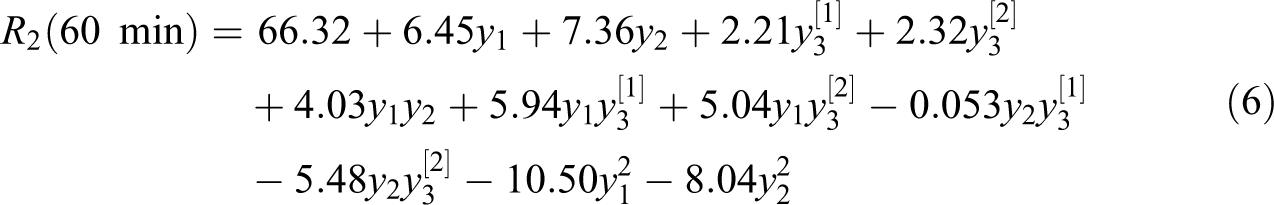
List of experiments based on the Box–Behnken’s design for 60 and 120 min glycolysis reaction time.
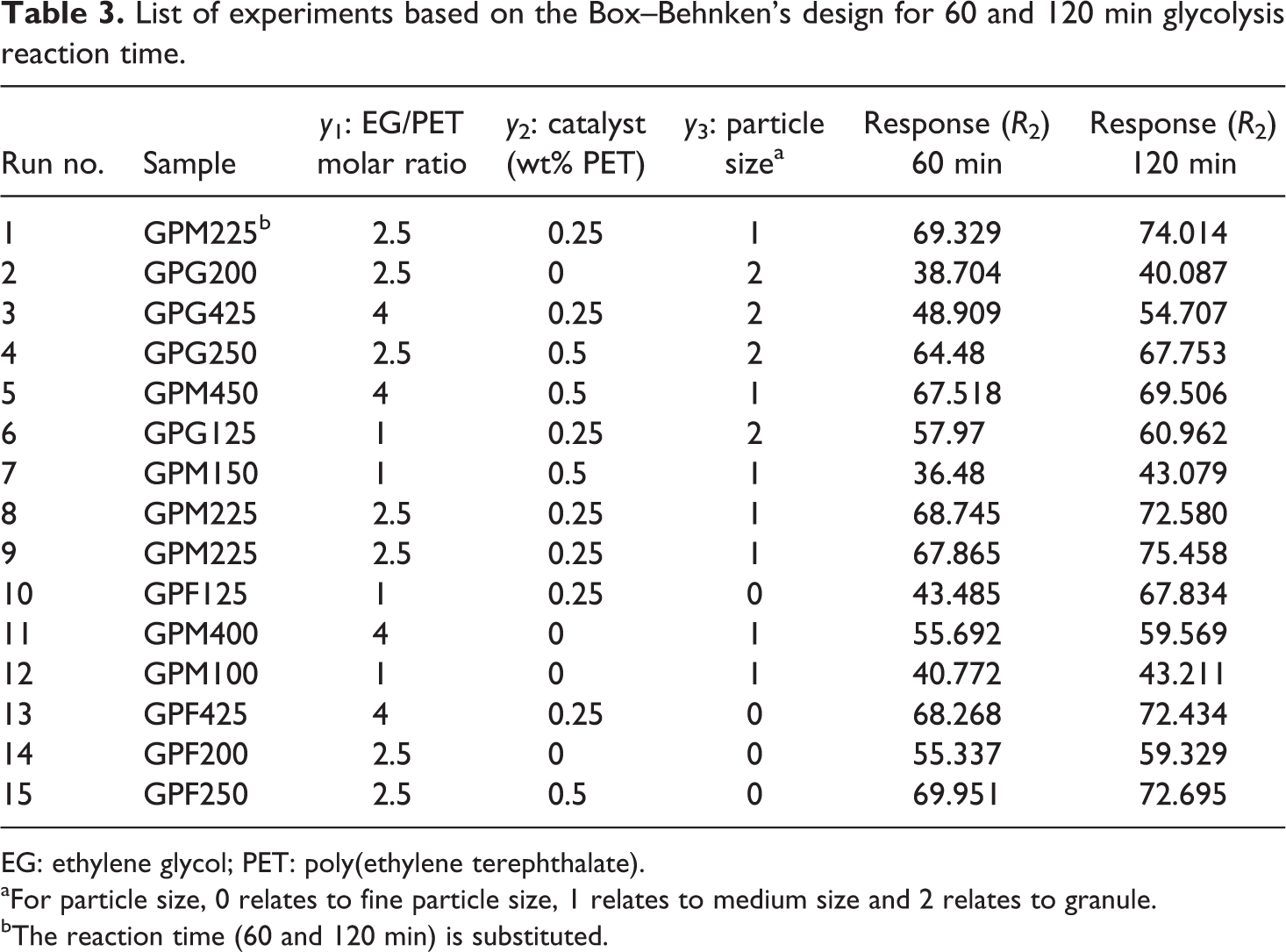
EG: ethylene glycol; PET: poly(ethylene terephthalate).
aFor particle size, 0 relates to fine particle size, 1 relates to medium size and 2 relates to granule.
bThe reaction time (60 and 120 min) is substituted.
Moreover, final equations in terms of actual factors are:
For 120 min glycolysis reaction time final equation in terms of coded factors: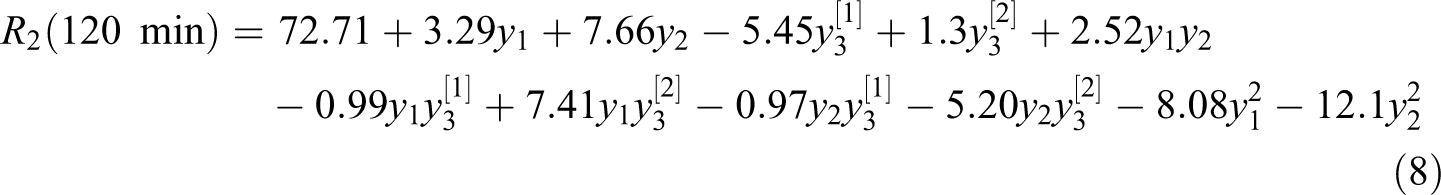
Moreover, the final equations in terms of actual factors are: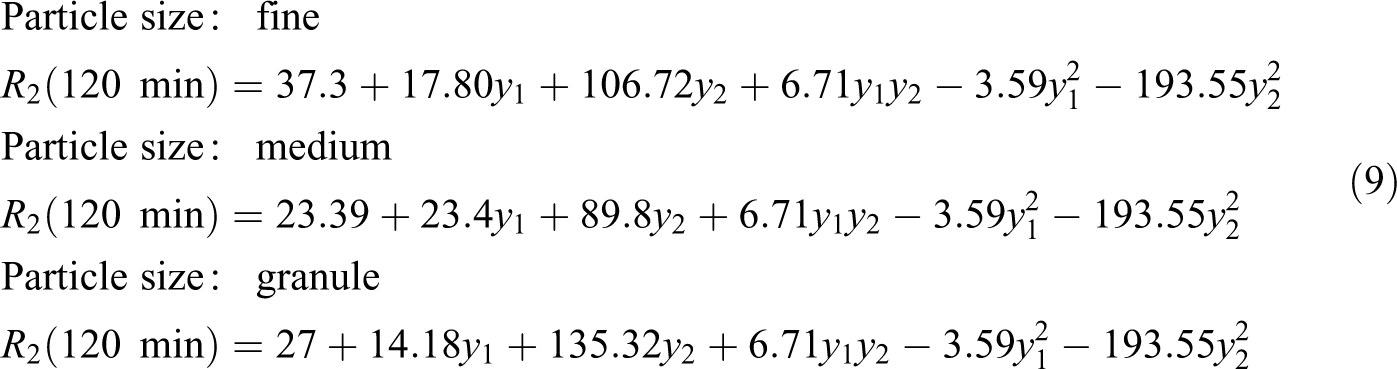
where y
1, y
2 and y
3 are EG/PET molar ratio, catalyst concentration and particle size of PET, respectively. In this case, the terms y
3, y
1 × y
2 and y
2 × y
3 are insignificant for samples produced in 60 min. While all terms for samples produced in 120 min are significant. Moreover, it is seen that there is no significant interaction between three factors, but there are some dual interactions between parameters. Based on the data in Table 3, glycolysis reaction rate increases significantly with glycolysis time, the amount of catalyst and EG/PET molar ratio. However, particle size does not have strong effect. PET oligomers near the melting point of 180°C was achieved by the sample GPG200120, with the reaction time of 120 min, EG/PET molar ratio of 1, the amount of catalyst, 0.5 wt% and granule particle size of PET. For 60-min reactions, sample GPM150060 has a similar response to GPG200120. This sample has an EG/PET molar ratio of 1 without catalyst using average particle size of PET. Therefore, samples GPG200120 and GPM150060 are named as optimum samples. FTIR of optimum samples were obtained. Figure 11 shows spectra of these samples. The peaks at 3430, 3000–2800, 1717 and 1343 cm−1 are corresponding to OH, C–H, C=O and C–O stretching bonds, respectively. A sharp peak at 726 cm−1 is related to the para-substituted aromatic rings. Figures 12 and 13, respectively, show 1HNMR and 13CNMR spectra of GPM150060 and GPG200120 samples. The methylenic protons adjacent to the hydroxyl end functions permit the determination of molecular weight of oligomers by 1HNMR spectroscopy; the four peaks of the 1HNMR spectrum (Figure 12) correspond to the signals of PET oligomer’s protons. Ratio of peak area of aromatic protons (H
d) to peak area of methylenic protons (H
a) allows calculation of the number of terephthalic base units in oligomers by the following equations: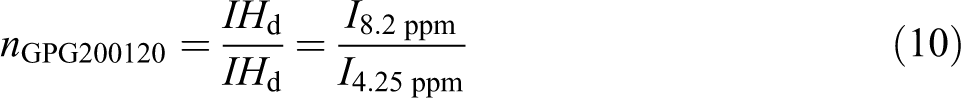
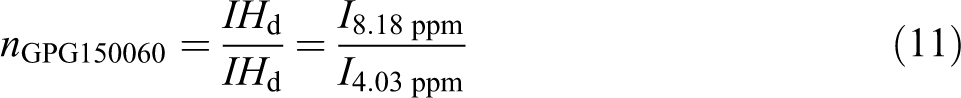
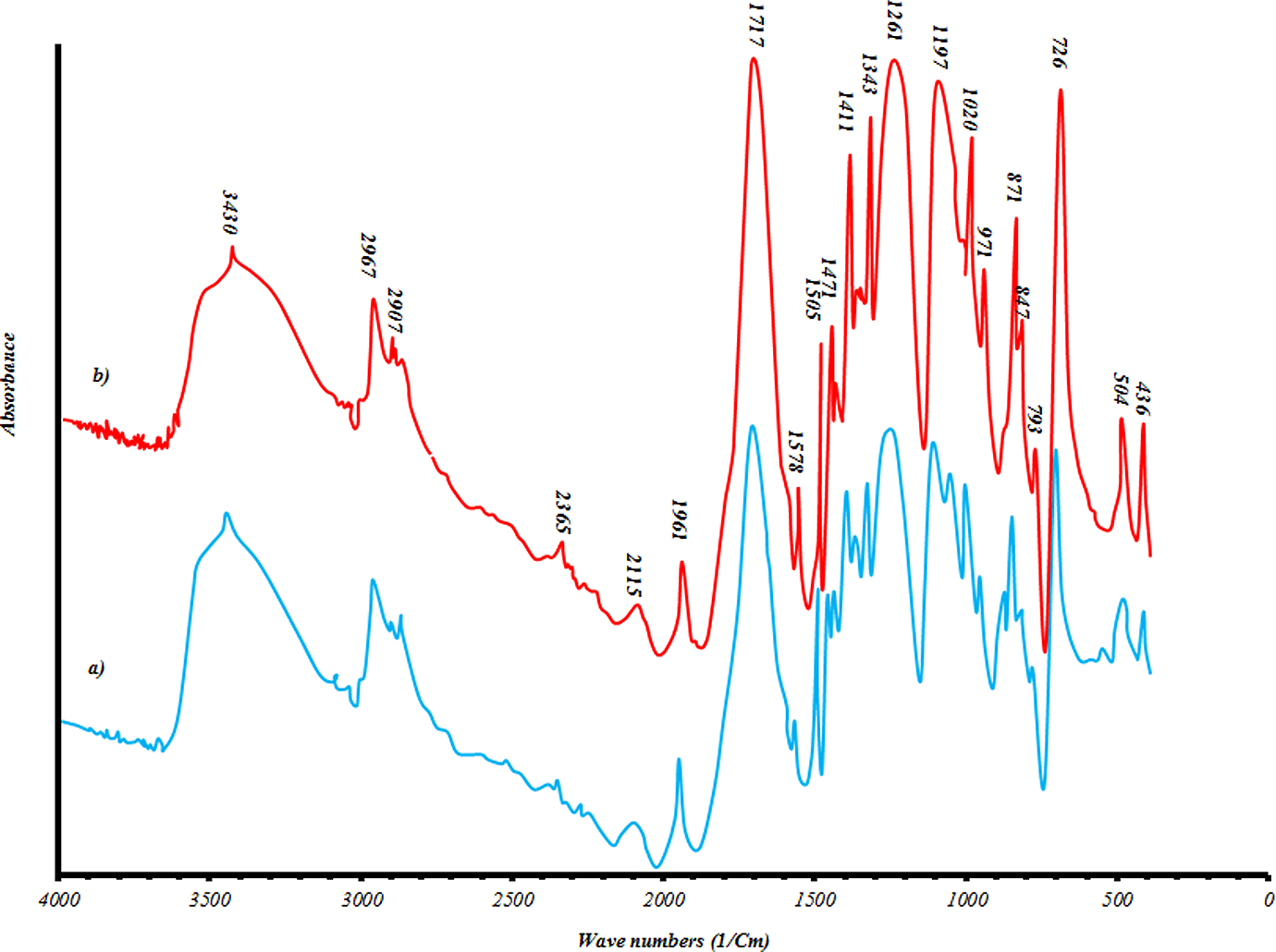
FTIR spectra of (a) GPM150060 and (b) GPG200120. FTIR: Fourier-transform infrared spectroscopy.
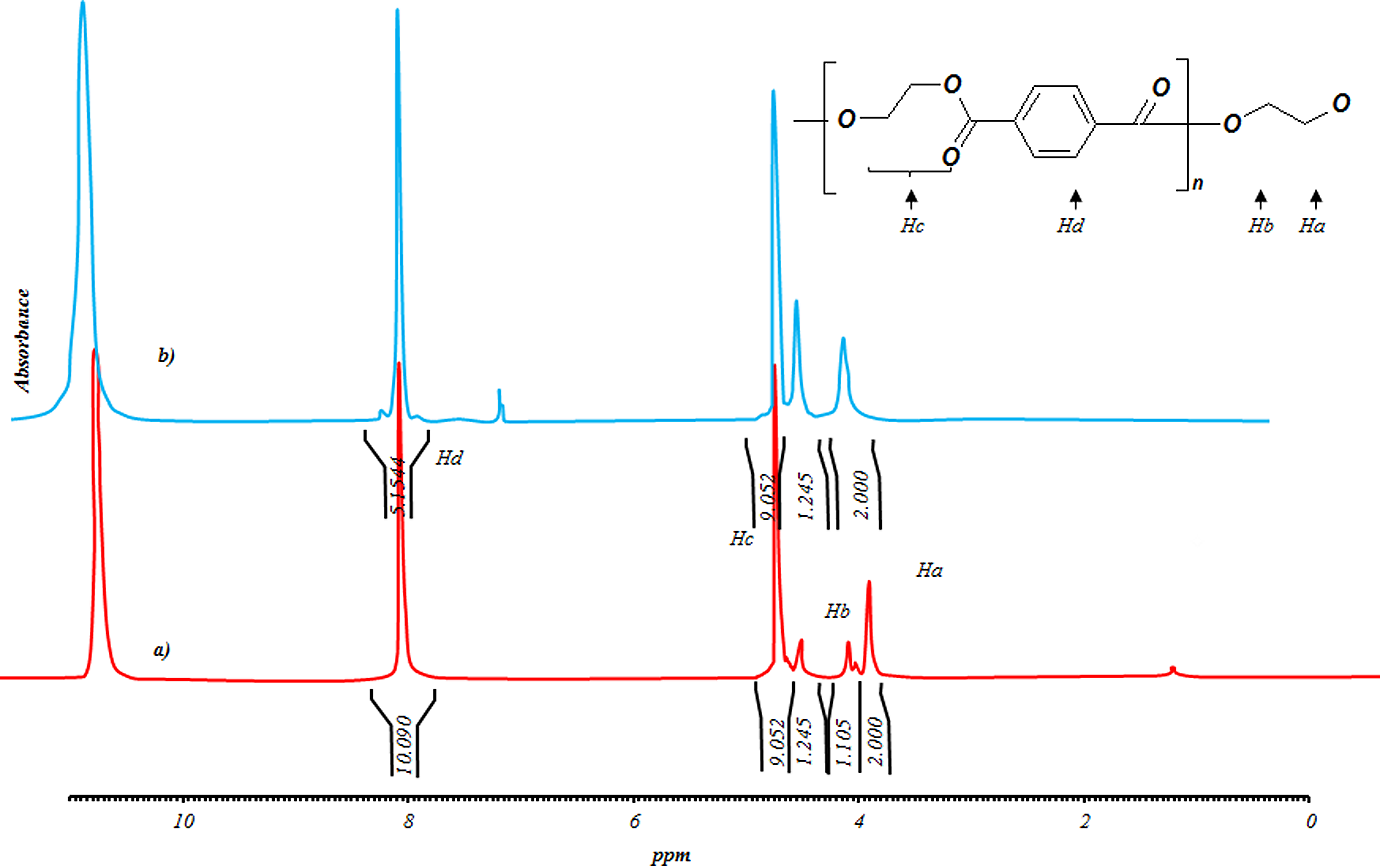
1HNMR spectra of (a) GPM150060 and (b) GPG200120. 1HNMR: proton nuclear magnetic resonance spectroscopy.
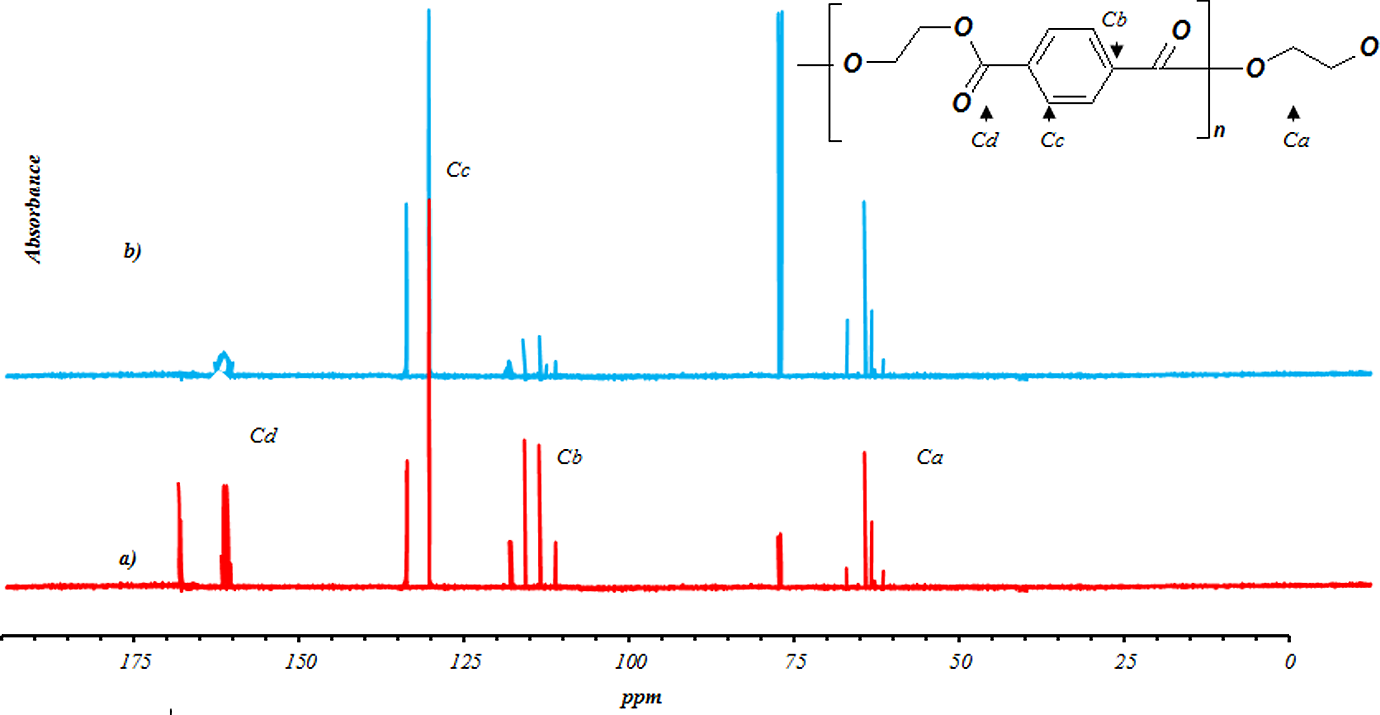
13CNMR spectra of (a) GPM150060 and (b) GPG200120. 13CNMR: carbon nuclear magnetic resonance spectroscopy.
Hence, it is easy to determine the number-average molecular weight of oligomers. Samples GPM150060 and GPG200120 have an average molecular weight
In the next step, optimum end capped PET oligomer sample (GPM150060) with the best response was applied to produce nanocomposites including 10 wt% of organoclay. This concentration is usually used for introducing masterbatch applied in final nanocomposite.
43
Necessary experiments were determined using central composite design (Table 4). d-spacing, R
3, and peak intensity of nanocomposite samples, R
4, were determined using x-ray diffraction (XRD) patterns. In this case, z
2
2 and z
2
2 × z
1 are significant terms for R
3. On the other hand, z
1
2 and z
2 are significant model terms for R
4. The following equations were fitted on responses, with R
2 = 0.9637 and R
2 = 0.9712, respectively: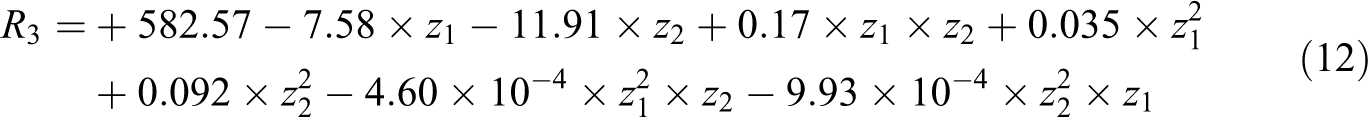
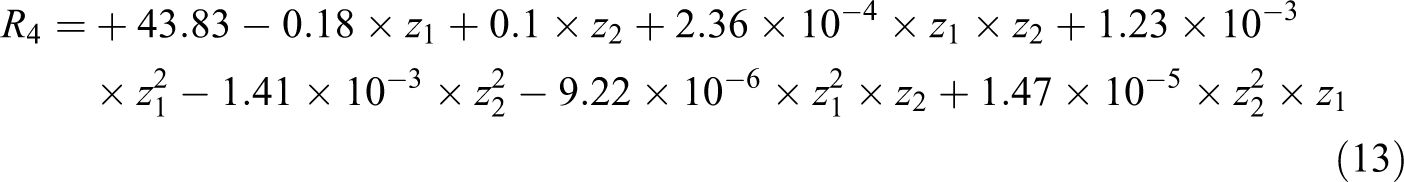
List of experiments based on the central composite design for PET functionalization.
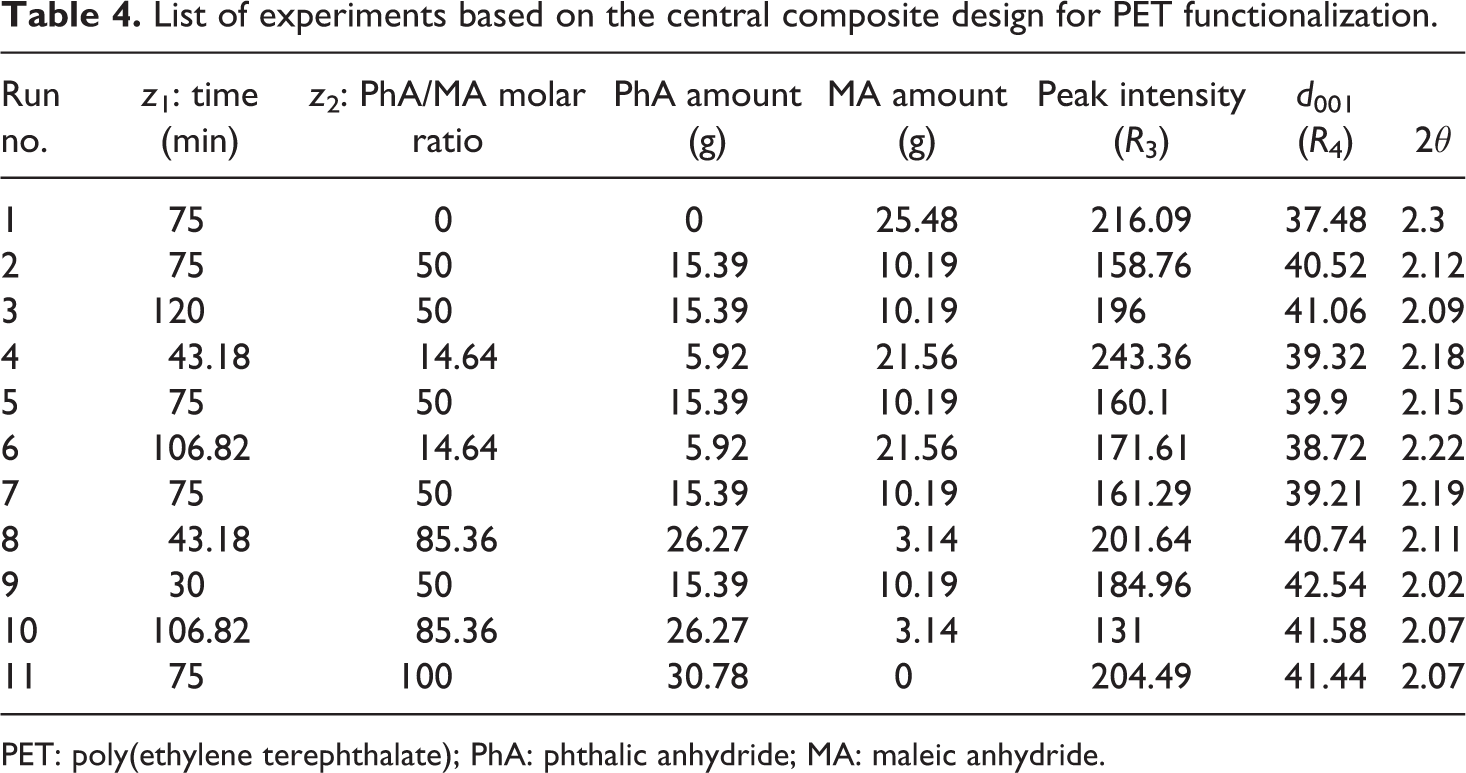
PET: poly(ethylene terephthalate); PhA: phthalic anhydride; MA: maleic anhydride.
where z 1 and z 2 are the reaction time and PhA/MA molar ratio, respectively. When response surface calculations were used, the combination of reaction time of 106 min and PhA/MA molar ratio of 0.85 gave the best results. The d 001 of organoclay is 18.5 Å (corresponding to 2θ = 4.8° according to Bragg’s law). Based on XRD results presented in Table 4, for almost all samples the 2θ degree was shifted to about 2.20°, which is corresponded to d 001 equal to 41 Å. Among all samples, the sample no. 10 has the minimum peak intensity, 131, while the sample No. 9 has the minimum 2θ value, 2.02. It means that the sample no. 10 causes clay layers to exfoliate more than other samples. Hence, the melt mixing of end capped PET oligomer with organoclay resulted in a good intercalation of layered silicate and good dispersion of clay in PET matrix.
Conclusions
Glycolysis conditions of off grade PET (melting point of 250–260°C) from a petrochemical company were investigated in order to reach its monomer, BHET at first. Yield of monomer increased with EG/PET molar ratio, the amount of catalyst and reaction time until the reaction reached equilibrium. Influences of glycolysis time, EG/PET molar ratio and the amount of catalyst (zinc acetate) were illustrated based on a response that is dependent on the melting peak area and melting point (equation (2)). According to the results, the optimal conditions are reaction time of 3 h, EG/PET molar ratio of 5, the amount of catalyst of 0.25%. The glycolyzed products were separated into monomer and oligomers. DSC and HV verified reaching BHET in order to obtain the number-average molecular weight. Moreover, the extent of depolymerization increased with glycolysis time, EG/PET molar ratio and the amount of zinc acetate (glycolysis catalyst). The suitable sample was also used to produce poly(bis(2-hydroxyl ethyl) terephthalate-MA) with MA. All the samples were also characterized by FTIR, 1HNMR and 13CNMR. These spectra confirmed that the desired polyester was successfully synthesized.
In the second process, partial glycolysis conditions of off grade PET were investigated in order to synthesize PET oligomers (melting point of about 180°C). The extent of depolymerization increased with an increase in the EG/PET molar ratio, the amount of catalyst and reaction time. Influences of glycolysis time, the EG/PET molar ratio and the amount of catalyst were studied. According to the results based on Box–Behnken’s method, the optimal conditions are the reaction time of 120 min, the EG/PET molar ratio of 2 and the weight ratio (catalyst to PET) of 0.5%. DSC scans were used for characterizing the glycolyzed products. For 60-min reactions, the optimum sample has an EG/PET molar ratio of 1 without catalyst using average particle size of PET. PET oligomers end capped with PhA/MA for preparing PET nanocomposites was produced via reaction between PhA/MA composition and PET oligomers of the optimum sample. Design of experiments based on central composite method was used to investigate the effects of reaction time and PhA/MA molar ratio to study the ability of end capped PET oligomer to intercalate organoclay layers. d-spacing and peak intensity of nanocomposite samples were selected as responses of functionalized products. Using response surface, the combination of reaction time of 106 min and PhA/MA molar ratio of 0.85 gave the best result. Again, all samples were also characterized by FTIR, 1HNMR and 13CNMR. These spectra confirmed that the desired functionalized PET oligomer was successfully synthesized.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.