Abstract
Thin films of poly(ethylene oxide) (PEO) doped with sodium iodide (NaI) were prepared by casting method. The films have an average thickness of 70 µm and contain different NaI concentrations, that is 1, 2, 4, 6, 8, 10, and 15% by weight (wt%). The dielectric constant, alternating current (AC)-conductivity, and thermal conductivity of the electrolytic composites were studied as a function of applied frequency, temperature, and NaI concentration. The AC conductivity and dielectric constants were determined from the impedance data, and the thermal conductivity was measured using the transient electric pulse method. It was found that the dielectric constants and AC conductivity increase with increasing temperature and NaI concentration. The percolation threshold of the observed AC conductivity occurs at about 1 wt% NaI dispersed in the PEO matrix. The charge transfer in the electrolytic films is dominated by ions transport and creation of more localized energy states due to high amorphous phase in the solid electrolyte. The formed iodide complexes also contribute in increasing the AC quantities. It was found that the thermal conductivity of the polymer electrolyte films increases with both temperature and NaI concentration. Empirical models were used to describe the dependence of electrical and thermal conductivities on the dopant content, frequency, and temperature.
Keywords
Introduction
Solid polymer electrolytes have significant interest due to their technological and commercial applications. They are successfully used in high energy rechargeable batteries, full cells, integrated optical devices, electrochemical sensors, and electrochromic displays. To get polymetric electrolytes with favorable electrical, optical, mechanical, and thermal properties, a clever choice of polymer and the metal electrolyte as dispersed component in the polymeric matrix are needed. It has been reported that the choice of polymer host (as matrix) depends mainly on the existence of polar groups to form electron/cation coordination and a low hindrance to polymer segmental motion. For example of these polymers that be used as polymer host is poly (ethylene oxide) (PEO). They have good ability to dissolve concentrations of metallic ions and good electrochemical stability compared with those other polymetric matrices. PEO complexes with sodium iodide (NaI) have high ionic conductivity, which gave it a recognized characteristic as a favorable solid electrolyte to be used in solid state batteries. 1 –3
Studying the electrical and thermal properties of the PEO/NaI polymer electrolyte helps greatly to characterize and interpret the electrical and conduction mechanism taking place in the composite bulk. Space charge limited current can contribute to explain the charge transfer in addition to ionic conduction dominated by the amorphous phase in the polymeric matrix. 4 –6
The physical characterization of thin films of polymer electrolyte can be carried out using a variety of analytical techniques, such as alternating current (AC) impedance, x-ray diffraction, and ultraviolet (UV)-spectroscopy, in addition to direct current (DC) measurements. 7 –11 For example, Mohan et al. 12 reported about the ionic conductivity of some PEO/salt electrolytes; Faleh et al. 13 studied the UV–optical absorption of doped PEO with and with out NaI; and Abu-Jamous and Zihlif 14 studied the AC conductivity of doped PEO with iodine at low-doping concentrations. At the same time, we feel that the study combines the electrical and thermal conductivities, which are very useful to exhibit physical correlation and comparison between the thermal and electrical conduction mechanisms that take place in solid polymer electrolytes.
In the present study, thin films of PEO doped with NaI were prepared using casting method. The AC electrical and thermal conductivities of those electrolyte films will be determined under different NaI concentration, temperature, and applied field frequency.
Experimental
Composites films preparation
PEO with average molecular weight of 300,000 g/mol was used to prepare electrolyte films by casting from solution. PEO powder and solid NaI were mixed together and dissolved in methanol as a suitable solvent. The solution was then stirred continuously by a rotary magnet at room temperature for few hours. The stirring process lasted until the mixture reached a homogenous viscous molten state. The mixture was immediately poured to thin films on a glass plate, and the methanol was allowed to evaporate completely at room temperature and atmospheric pressure for few days. The composite films were dried in an oven at 40°C for few days. Thus, we think that the water content is very negligible and has no effect on the measured physical quantities. The films obtained have thickness of about 70 µm and different NaI concentration 1, 2, 4, 6, 8, 10, and 15% by weight.
Impedance measurements
Impedance measurements were carried out through the standard ring method at different temperatures and applied field frequency range 30 kHz–5 MHz using LF impedance analyzer (HP model 4192). Disk-shaped specimens of 2-cm-diameter were cut from the prepared sheets. The test specimens were placed firmly in a cell between two copper electrodes connected through cables to the impedance analyzer. The cell was put in an oven and the temperature is measured by thermocouple wires. A period of about 20 min was maintained between successive impedance measurements to allow a steady state of temperature to be reached. The impedance analyzer reads values of impedance and phase angle of the specimen by varying the applied frequency. The mean values and SDs were estimated with an average error of about 2–4%. Impedance measurements were performed in the temperature range of 27°C–60°C; no higher temperature measurements were taken since the melting temperature for PEO polymer is about 65°C. 8
The real and imaginary components of the complex impedance 

where
The dielectric constant 

where 
where
The AC conductivity of the composite samples is calculated from the equation
and the thermal activation energy (
Here
Thermal conductivity measurements
Measurements of thermal conductivity were carried out through the electrical pulse method using a thermocouple amplifier (model AD 594 instrumentation amplifier) calibrated as (10 mV/C). The sample holder consists of two small disks of 5 cm in diameter. Two composite specimens each were sandwiched between hot and cold plates. A heater made of chrome and a power of 100 W is placed between the test specimens. Two sheets of mica are used as insulators. There are four thermocouples, each sample has two faces (upper and lower), and additional thermocouple is inserted in an oven that reads a temperature up to 200°C. The pulsed voltage (
Results and discussion
Electrical conductivity results
Experiments were conducted to measure the AC impedance and phase angle using impedance analyzer. The phase angle takes negative values ranging between −89.63 for pure PEO and −77.3 for 15 wt% NaI electrolyte system; the negative values of the phase angle indicate that the films can be represented by parallel RC circuits connected in series. Impedance values decrease with increasing the NaI concentration. From impedance data, values of the dielectric constant (
It can be seen from Figure 3 that the empirical power law gives a good fit for the observed values of dielectric constant (
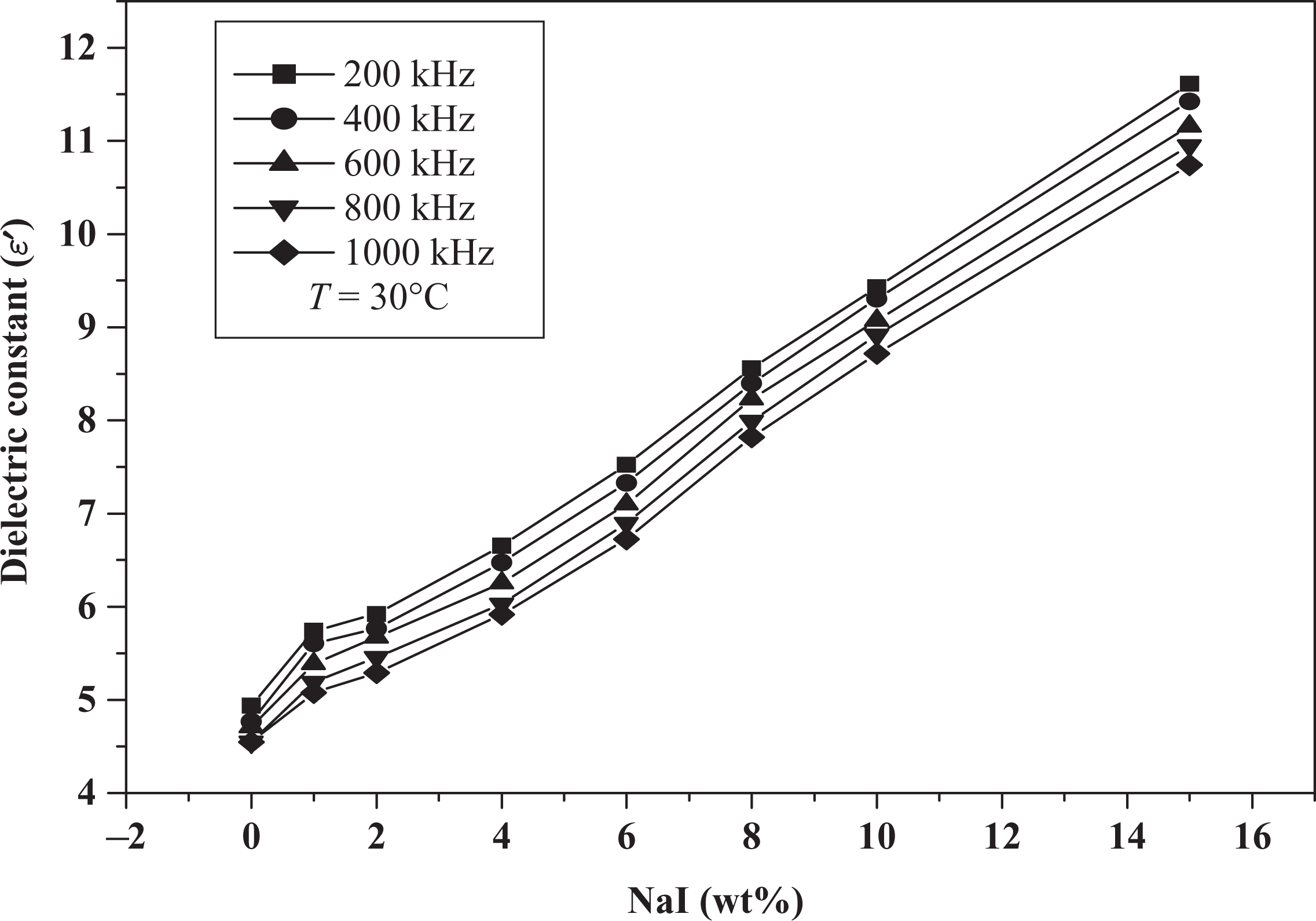
Variation in dielectric constant with NaI concentrations. NaI: sodium iodide.
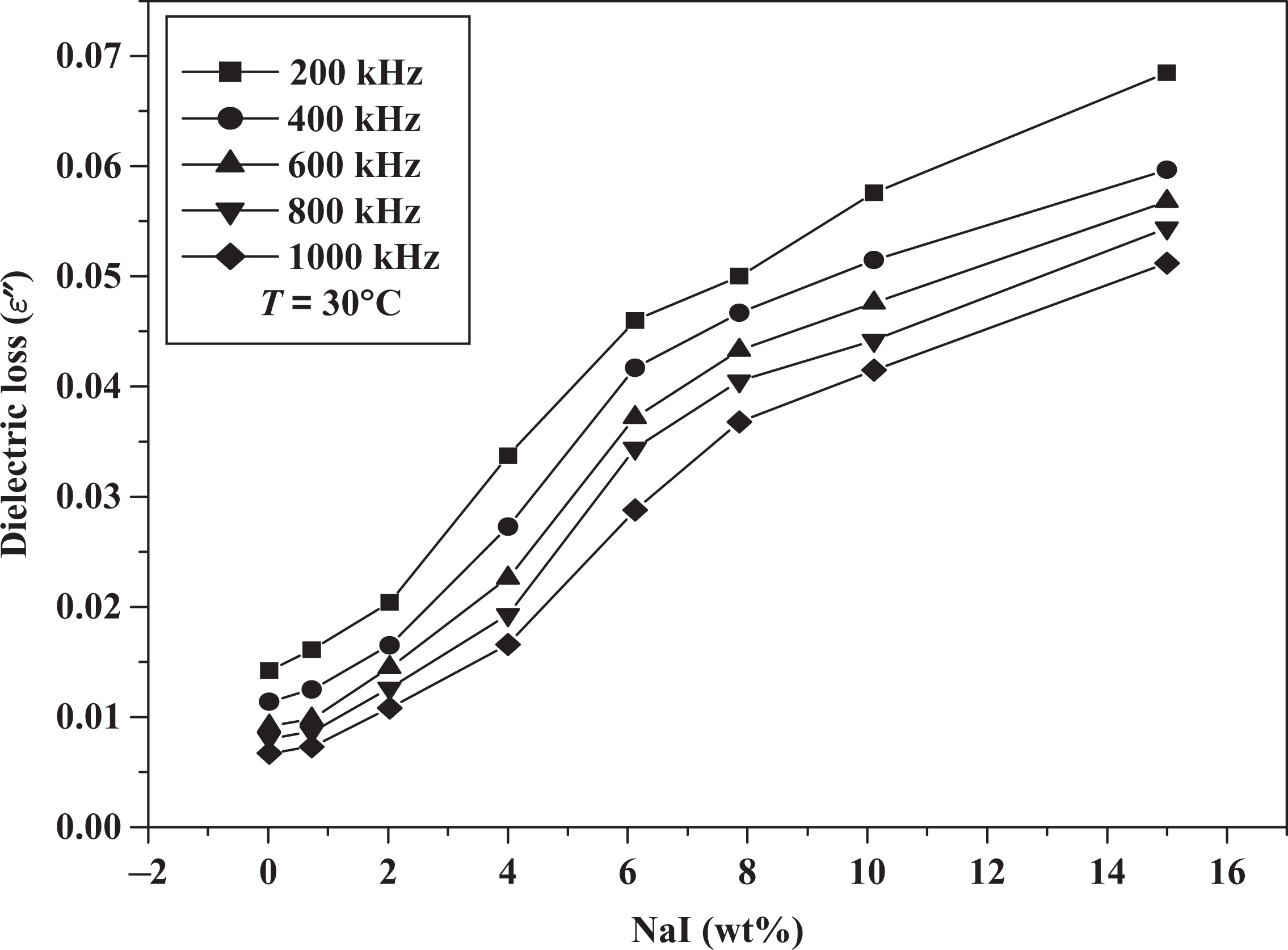
Variation in dielectric loss with NaI concentrations. NaI: sodium iodide.
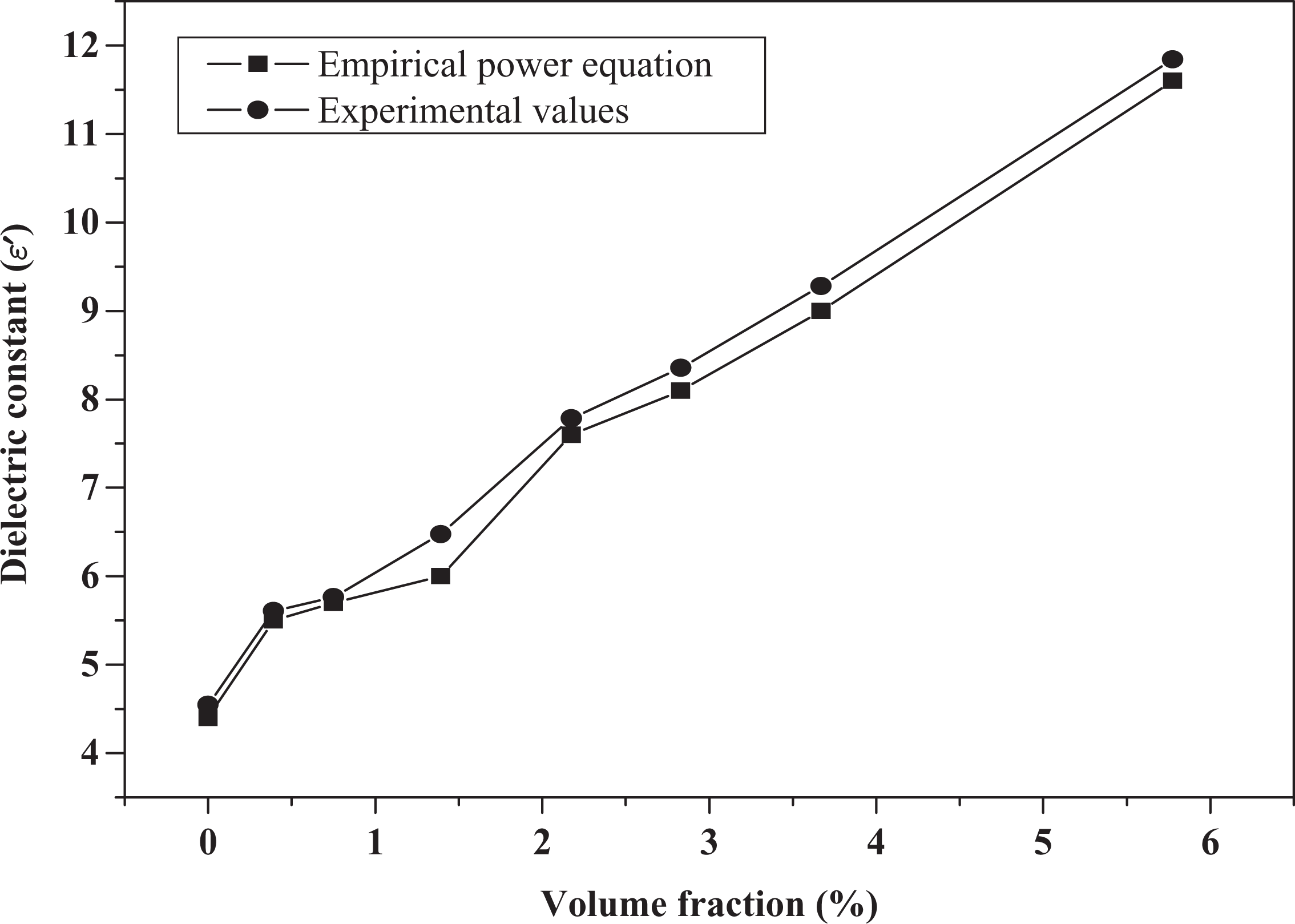
Fitting the observed dielectric constant data of PEO/NaI composites. PEO: poly(ethylene oxide); NaI: sodium iodide.
Figures 4 and 5 show the variation in dielectric constant (
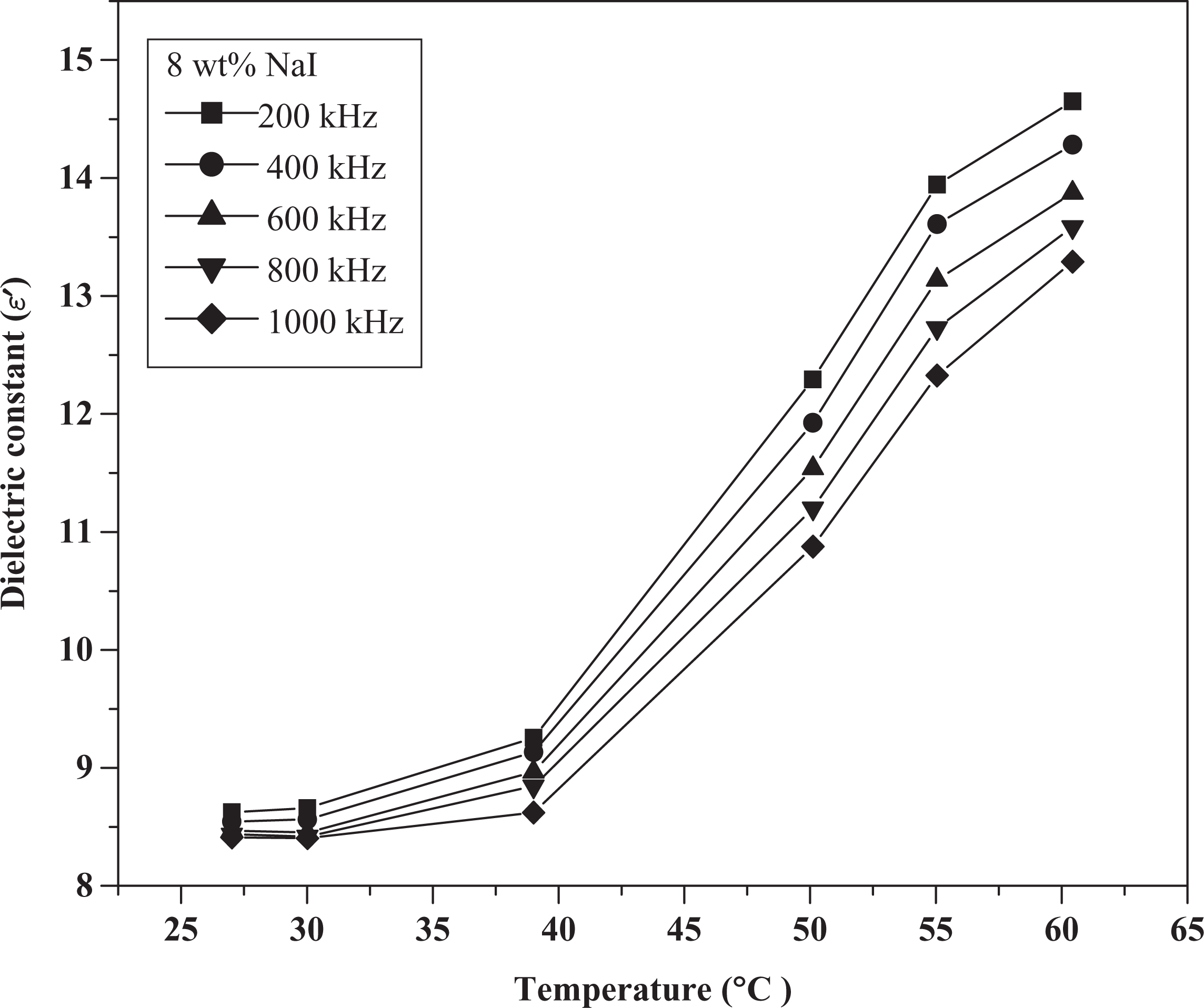
Dielectric constant versus temperature for 8 wt% NaI composite. NaI: sodium iodide.
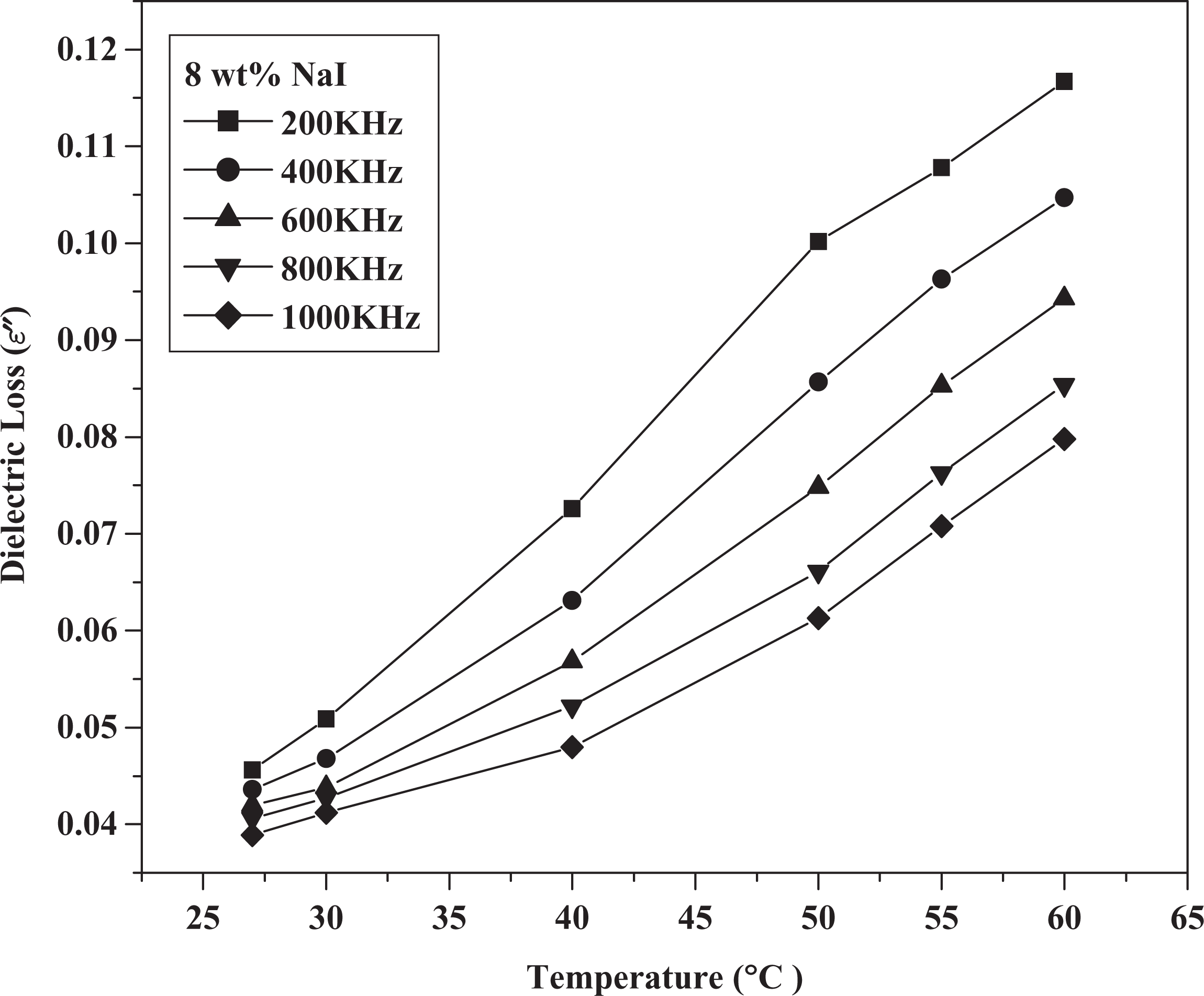
Dielectric loss versus temperature at different frequencies for 8 wt% NaI. NaI: sodium iodide.
An important technique for the analysis of the dielectric data is the Cole and Cole plots of
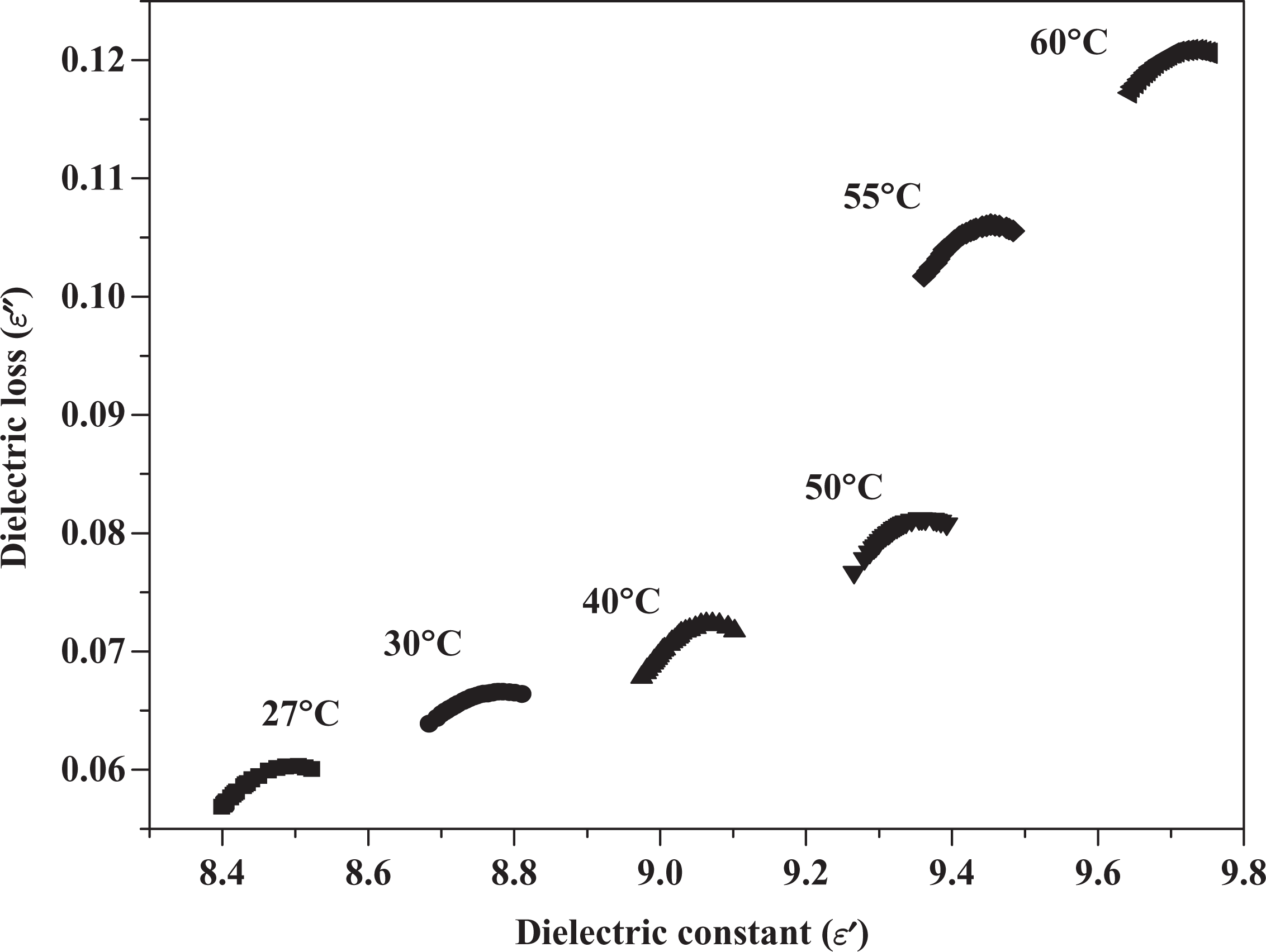
Cole–Cole plots for 8 wt% PEO/NaI composite at different temperature. PEO: poly(ethylene oxide); NaI: sodium iodide.
Relaxation time for 8 wt% NaI composites as a function of temperature.

NaI: sodium iodide.
Figure 7 shows the variation in the AC conductivity (
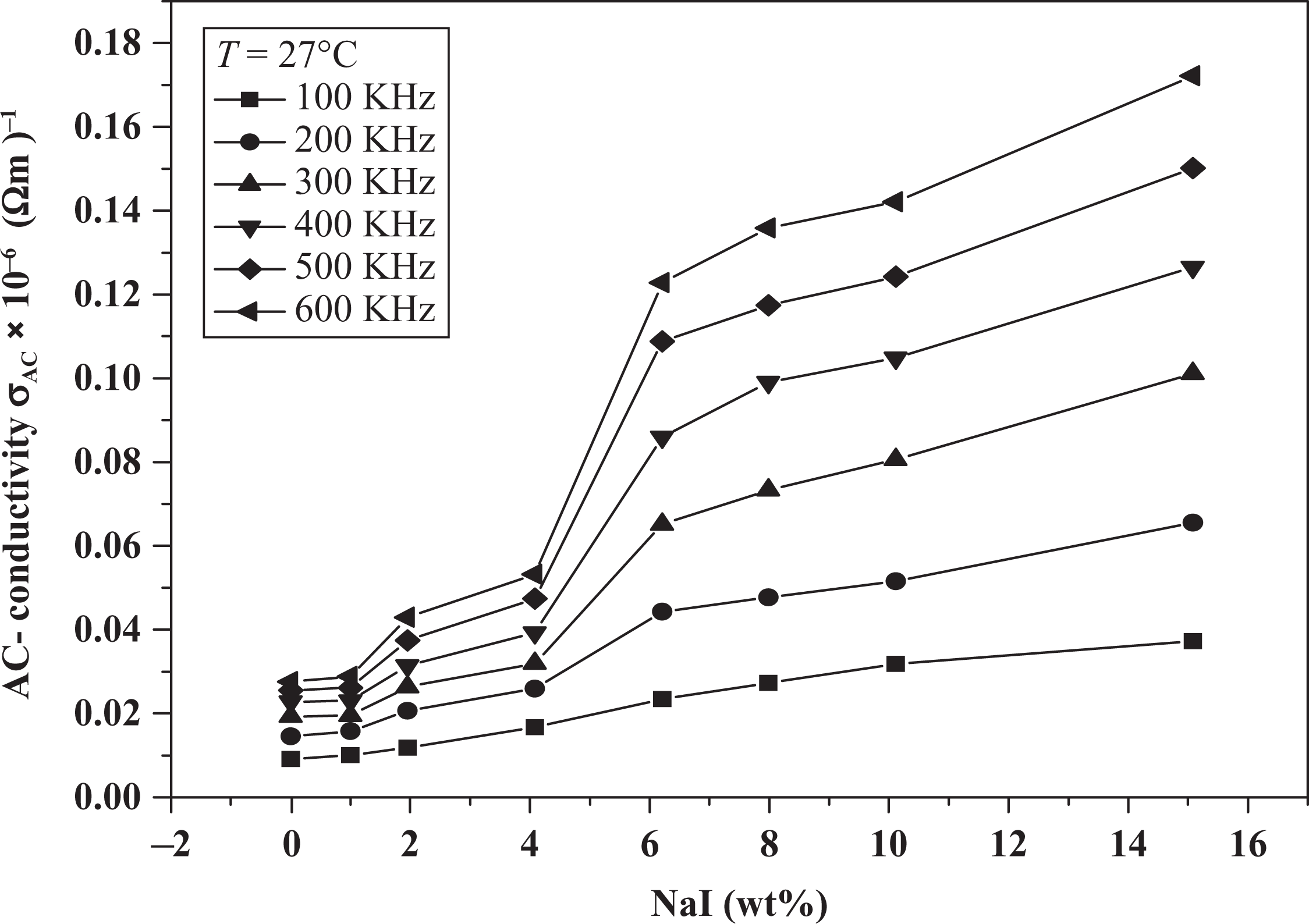
Variation in AC conductivity with NaI concentration. AC: alternating current; NaI: sodium iodide.
It is a general thought that the AC conductivity is related to frequency as reported in the empirical Jonschers universal law model,
19
which is given by the equation
where B and 
From this figure, the coefficients
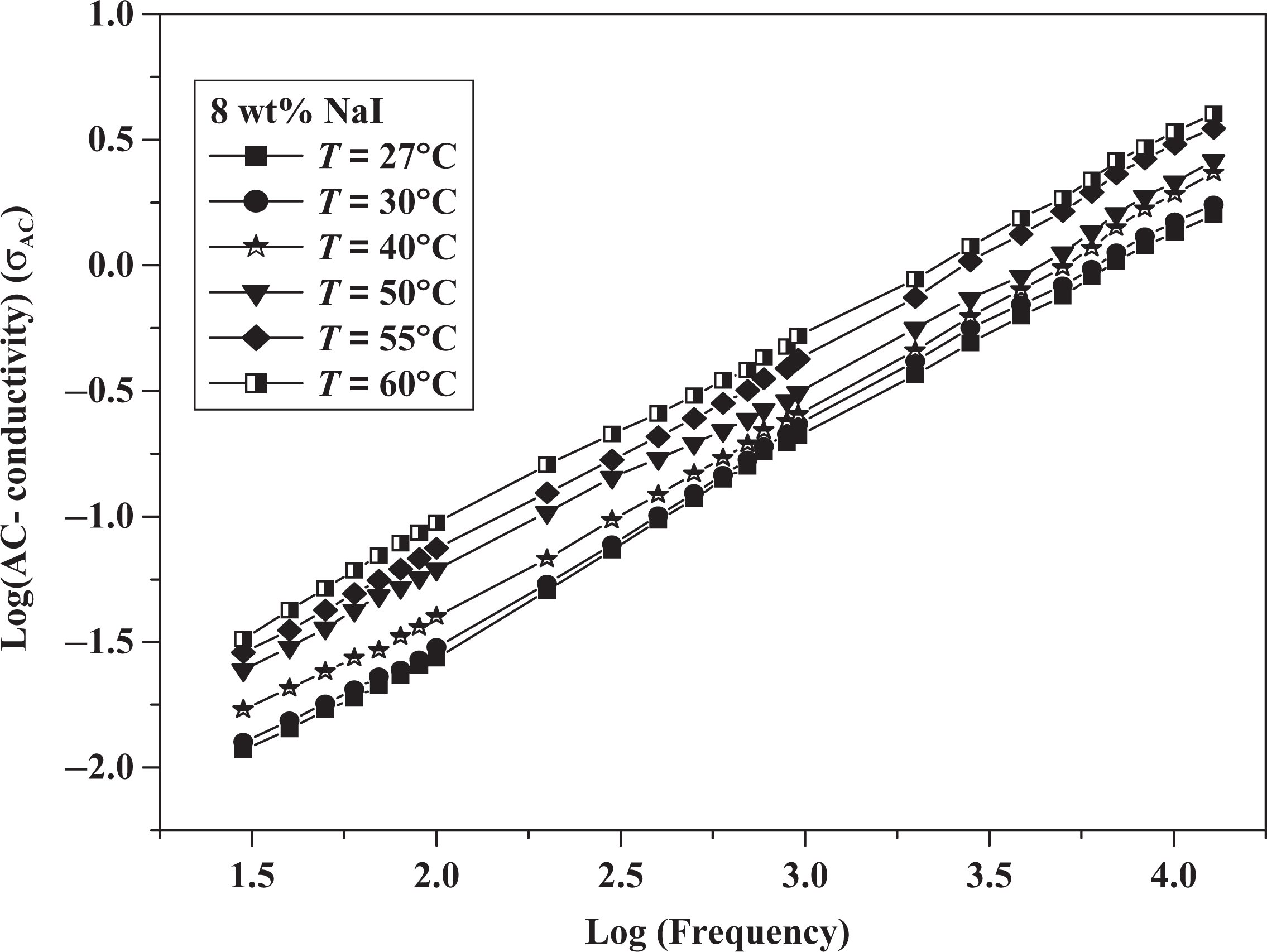
Variation in log (AC conductivity) versus log (frequency) for 8 wt% NaI composite. AC: alternating current; NaI: sodium iodide.
Estimated
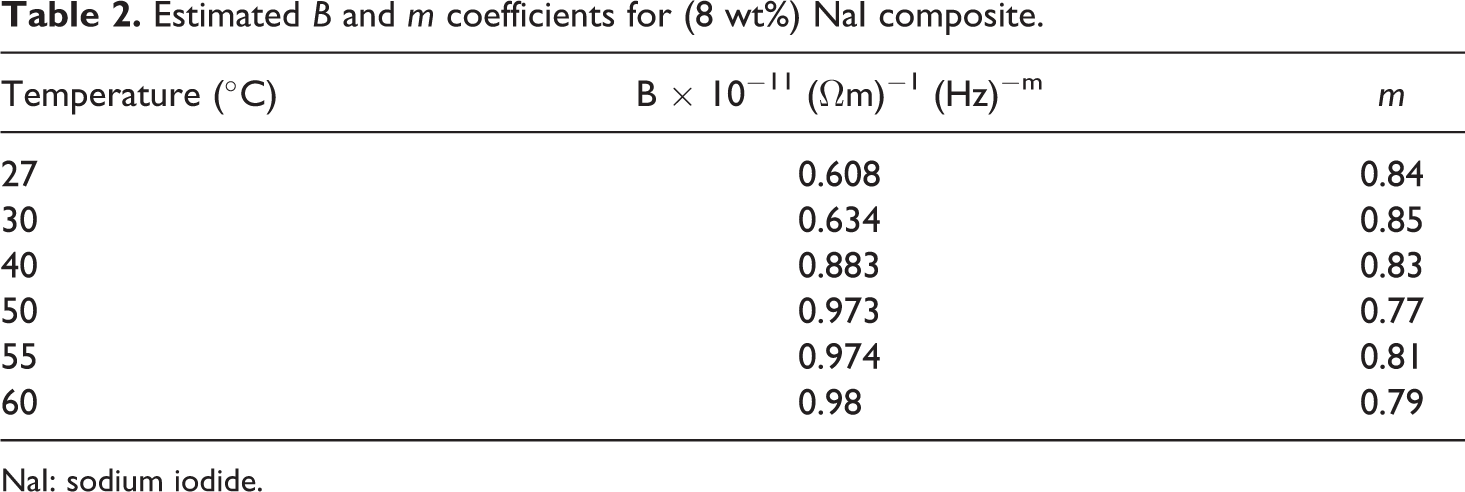
NaI: sodium iodide.
Figure 9 shows the dependence of the AC conductivity on temperature and NaI concentration. The
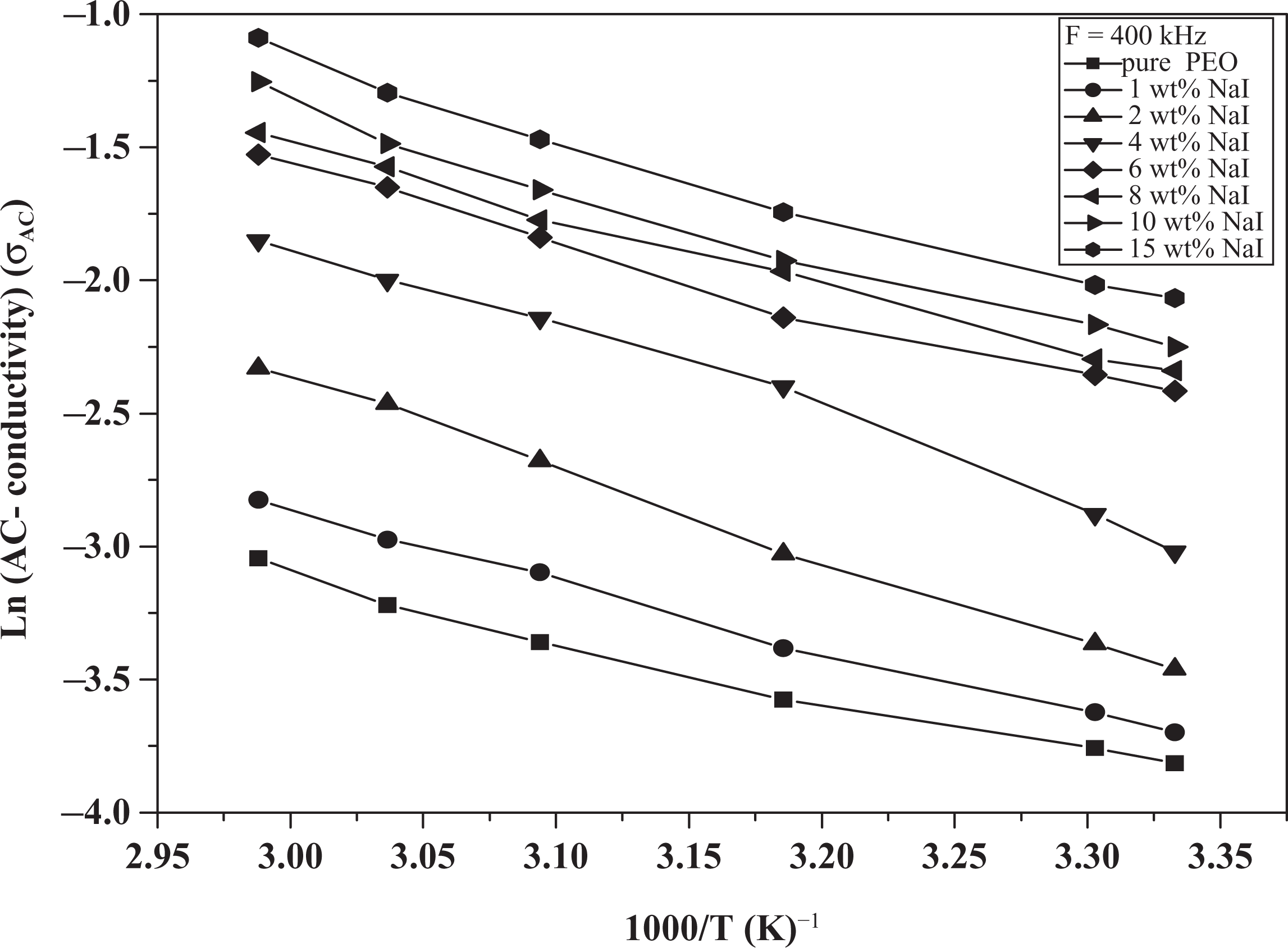
Ln(
The activation energy values for all samples at (

PEO: poly(ethylene oxide); NaI: sodium iodide.
Thermal conductivity results
The thermal conduction in solid polymer electrolytes is produced by phonons, ions transport (as major contributors), and also from electrons and impurities existing in their bulk structure. Measurements of the thermal conductivity of polymeric materials are difficult since its value is relatively small and in the range of 0.2–0.4 W/m °C. The thermal conductivity is so sensitive to the measuring conditions and physical and chemical structure of the polymer composite. Also, it is difficult to measure the thermal conductivity for the polymeric materials due to its smaller value. The growing needs for materials to be used in thermal applications lead to the design of new composite material with appropriate combination of selected polymeric matrices and suitable fillers. 20 –22
The thermal conductivity 
where
Figure 10 shows the variation in the thermal conductivity with temperature and NaI content. It can be seen that the conductivity increases with temperature and increases with the dopant concentration. This behavior of thermal conductivity is similar to that of the electrical conductivity concerning temperature and dopant content. In case of raising the temperature, the phonons, ions, electron, and impurities are activated and thus enhancement in the thermal conductivity is produced. Table 4 includes the variation in the thermal resistance (
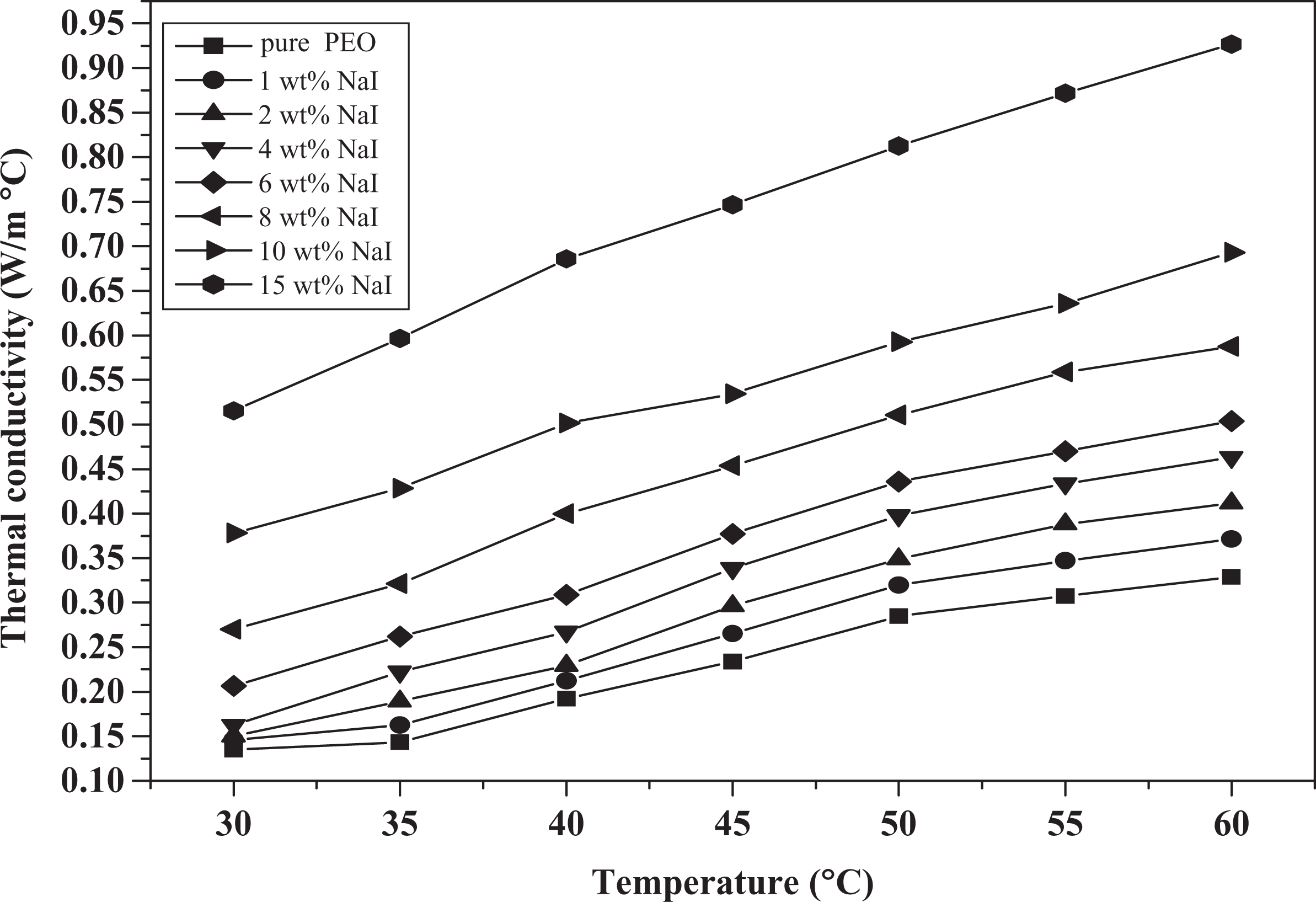
Variation in thermal conductivity with temperature for PEO/NaI composite. PEO: poly(ethylene oxide); NaI: sodium iodide.
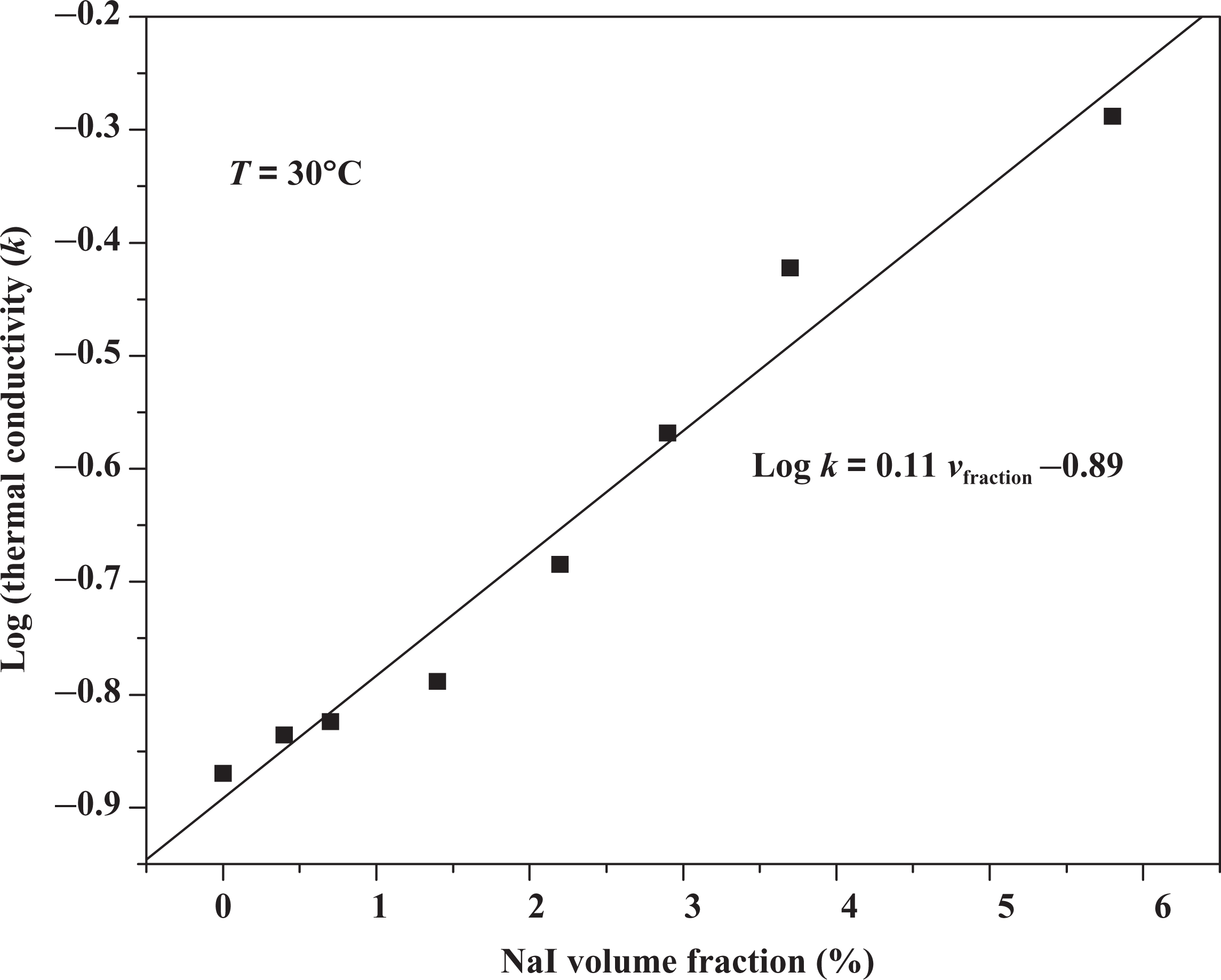
Agrawal model of thermal conductivity versus composites volume fraction.
Thermal and electrical conductivities results values (
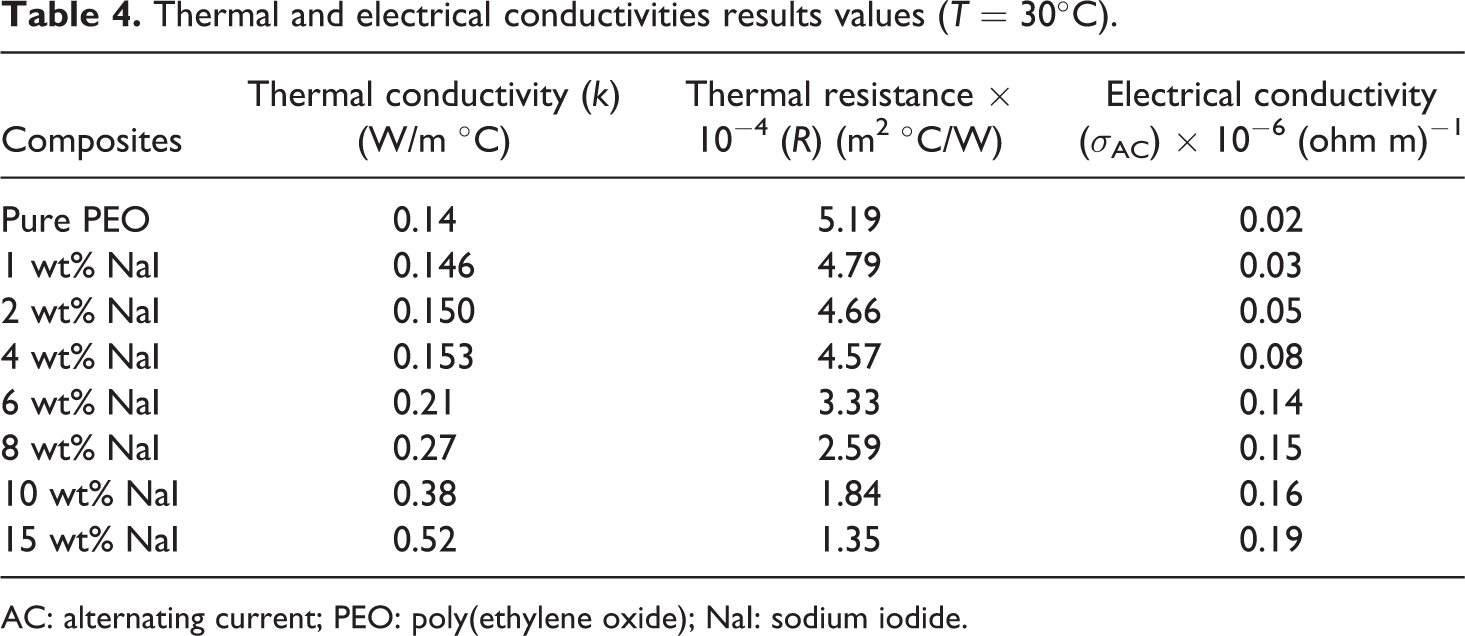
AC: alternating current; PEO: poly(ethylene oxide); NaI: sodium iodide.
Figure 12 shows the films morphology and dispersion state of the NaI dopant in the PEO matrix. The optical pictures of the two prepared films containing 4 and 10 wt% NaI reveal homogenous distribution of the dopant particles and complexes in the PEO matrix. Particle paths and contacts are clearly shown in the film background, a case which facilitates the transport mechanisms of the ions, electrons, charged complexes, and phonons, and thus leads to the enhancement of the measured AC quantities and thermal conductivity of the studied solid electrolytic composites.

Optical pictures: (a) 4 wt% NaI and (b) 10 wt% NaI.
Conclusion
In the present study, the AC electrical and thermal properties of PEO/NaI thin films were investigated as a function of applied field frequency, temperature, and NaI concentration: 1, 2, 4, 6, 8, 10, and 15 wt%. The AC quantities as dielectric constant, AC conductivity, activation energy, and relaxation time were determined from the analysis of impedance data. It was found that the dielectric constant and conductivity increase with applied frequency, temperature, and NaI concentration. The percolation threshold occurs at about 1 wt% NaI content. The charge transfer in the PEO/NaI electrolyte is dominated by ions transport which is enhanced by increasing the amorphous phase when temperature or NaI content become higher. The observed thermal conductivity increases with both NaI content and temperature. Some reported that the empirical equations are used to fit the measured AC quantities and thermal conductivity of the prepared PEO electrolyte films. Furthermore, correlation between the observed values of the electrical and thermal conductivities is presented.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
