Abstract
The structure of polysodium acrylate (PSA) hydrogel was modified using
Introduction
Polymeric materials play an important role in modern life and gels are one kind of these materials. In a gel, long polymer chains are cross-linked producing a three-dimensional (3D) network, which is able to retain liquid. In many applications, gels hold big amounts of water inside the microscopic network. These materials are known as hydrogels. They can swell in water to the great extent such as to equilibrium volume by preserving their shape and transparency. The nature of polymeric network bonding and functional groups along the chains can be modified by the use of chemical agents, temperature and pH changes, amongst other factors. In general, microscopic sponges or systems retains and delivers diverse chemical substances. 1 Polymeric hydrogels have many applications and are currently used in hygienic products, paints, food, agriculture, electronic devices, molecular filters and drug delivery systems. 2 –7 These multistructured hydrogels are hydrophilic, elastic, water insoluble, soft polymers with an anisotropic optical response. The analysis of thermal and structural properties of hydrogels provides an insight on their macroscopic behavior and final applications.
Although hydrogels undergo substantial volume or mass change due to the change in total water content, the state of water in the hydrated polymeric networks is more crucial because it governs the interaction between the polymer and other substances, which is vital when the hydrogel is used as bioactive carriers. These materials have attracted much attention in the medical and mechanical engineering fields. The state of water in the hydrogel can also influence the biocompatibility when the hydrogel is used as an implant material. It determines the reactivity and bioavailability, when used as an encapsulation material for enzyme or cell immobilization. 8 –10
According to its molecular nature, the water in hydrogels can exist as free water, bound water, freezable water and nonfreezable water. The existence and quantification of free water, bound water, freezable water and nonfreezable water have been determined through the use of differential scanning calorimetry (DSC). It is well known that polar groups in a polymer molecule interact more strongly than nonpolar groups with water molecules, in the form of ion–dipole, dipole–dipole or hydrogen bonding. Functional groups of different polarity bind with water in varying degrees, thus affecting the distribution of bound to free water. The presence of a cross-linking agent is the key to holding a hydrogel system together. Many hydrogel properties are affected by the number of cross-linkages (or cross-linking density). It is well documented that increasing the density of cross-linkages causes a decrease in water content. 11
The physiochemical, mechanical and biological characteristics of these materials were investigated by different techniques. In particular, the mechanical behavior was analyzed by dynamic mechanical analysis (DMA). The investigation of the dynamic mechanical behavior of polymeric materials is of great interest and importance for many reasons. Mechanical damping is often the most sensitive indicator of all kinds of molecular motions, which are going on in a material, and these motions, apart from their scientific interest, are of great practical importance in determining the mechanical behavior of polymeric materials. 12
To overcome the weakness and limitations of polymer hydrogels, they are reinforced with fillers to improve the mechanical properties and overall performance. Various researchers have attempted to modify acrylic-based hydrogels with minerals such as mica, kaoline and clays such as attapulgite, bentonite and montmorrilonite to enhance their absorbency, gel strength, absorption rate and to improve thermal, mechanical and physical properties. Cellulose microfibrils (CMFs) are commonly the employed fillers that are obtained from banana fibers. As opposed to the fillers mentioned above, CMFs form physical networks at low-volume fractions. 13,14 The effect of CMF on the water state and viscoelastic properties of polysodium acrylate (PSA) hydrogel is another interesting issue to be discussed here.
Our present work aims to investigate the water state and dynamic mechanical behavior of PSA/CMF hydrogels cross-linked with
Experimental
Materials
Acrylic acid, MBA, ammoniumpersulfate (APS),
Synthesis of PSA hydrogel
Acrylic acid of 30 ml was dissolved in 20 ml of distilled water. NaOH of 17 g was dissolved in 40 ml of distilled water. Acrylic acid solution was neutralized by adding NaOH solution. This solution is called as stock solution (monomer stock solution). To get hydrogels with various percentage of cross-linker, different amounts (20, 40, 80 and 100 mg) of MBA were dissolved in 10 ml of stock solution. About 4 mg of APS as an initiator and 0.5 ml of TEMED were added as an accelerator to each of the above cross-linker solutions. Within 5–6 min hydrogel was formed, which was thick, transparent and water insoluble.
Synthesis of CMF
Cellulose fibers are generally lignocellulosic consisting of helically wound cellulose microfibrils in an amorphous matrix of lignin and hemicellulose. So cellulose fibers were treated to remove the lignin content in it. Sisal fibers were cut into small pieces of 2 mm length. Sisal fibers of 1 g was taken in a beaker and dispersed in 0.5 M, 10 ml NaOH solution. The mixture was stirred vigorously using a magnetic stirrer at 80°C for 2 h. The sample obtained was vacuum filtered and residue was dispersed in 0.5 M NaOH solution (10 ml) for 15 min at room temperature. Cellulosic residue was washed with distilled water using centrifuge for several hours until constant pH was reached and subjected to drying at 60°C.
Sample obtained was added to 0.59 ml of 0.7% sodium chlorite in 0.2 M sodium acetate buffer solution (pH = 4.9) and volume of the solution was increased to 10 ml. It was then vigorously stirred using magnetic stirrer at 70°C for 2 h. The sample thus obtained was vacuum filtered and dried at 60°C. Subsequently, it was dispersed in 10 ml of 20 wt% of H2SO4. Finally, it was stirred using a magnetic stirrer at 40°C for 35 min. The resulting suspension was vacuum filtered so that most of the acid was removed and then washed with water using a centrifuge until neutrality was reached. Aqueous solution of the above sample (prepared by adding 10 ml of distilled water) was subjected to sonication for 15 min. The length of the microfibrils was ranging from 800 to 1100 μm and a diameter from 10 to 40 μm with an aspect ratio ranging from 20 to 100. The residue obtained was dried in the oven at 60°C and then it was analyzed using XRD for the confirmation of CMF formation.
Synthesis of PSA/CMF hydrogel
Different percentages of filler content varying from 0.4 to 2 wt% were added to the monomer solution, which was sonicated and the same procedure of gel preparation as discussed above was followed.
Characterization
The synthesized cross-linked gel, raw banana fiber and CMF were analyzed by the Fourier transform infrared (FTIR) spectroscopy (NICOLET 6700, Thermo Scientific, USA, www.thermoscientific.com). The synthesized CMF was characterized using XRD (Shimadzu D-115 diffractometer, Japan) with monochromatized CuKα radiation (
Determination of hydrogels total water content
The total water content of the hydrogel was determined gravimetrically. Initial weight of the 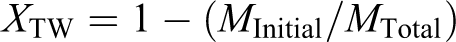
Determination of free and bound water content in hydrogels by DSC
The bound water content of the swollen gel was determined using a differential scanning calorimeter (DSC Q20, TA Instruments, USA) by the standard test method ASTM D3418. Hydrogel specimens (5–10 mg) were cut from the pre-equilibrated gels, placed in the appropriate aluminum pan, sealed and cooled to −40°C at a rate of 5°C min−1 and then reheated at the same rate up to 20°C. The phase transition of water in the hydrogel during heating was recorded as the endothermic peak. Fraction of the freezable (free water) water and unfreezable (bound water) water in the samples was determined using the following equation, assuming that the heat of fusion of free water in the hydrogel is the same as that for ice. 12
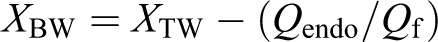
where,
Dynamic mechanical analysis
Dynamic mechanical properties of both PSA and PSA/CMF hydrogels were characterized using dynamic mechanical analyzer (DMA Q800, TA Instruments, USA). The samples having dimensions of 60 × 12 × 3 mm3 were fixed in the dual cantilever fixture and subjected to 1% strain and a frequency of 1 Hz over a temperature range of 35–160°C at 10°C min−1.
Results and discussion
FTIR analysis
Figure 1(a) shows the FTIR spectra of untreated banana fiber (raw banana fiber) and CMF. The absorption peaks at 3350 and 1040 cm−1 correspond to the stretching vibrations of O–H in cellulose and C–O in hemicelluloses and cellulose. The peak at 1645 cm−1 in all the samples is the indicative of C=O bond of hemicellulose. The absorbance at 1516 cm−1 is associated with aromatic skeletal vibration in lignin. Disappearance of this peak after treatment indicates complete removal of lignin in CMF. Peaks observed at 1236 and 1158 cm−1 also show the presence of hemicelluloses.
(a) FTIR spectra of CMF and untreated banana fiber. (b) FTIR spectra of monomer solution and PSA gel. FTIR: Fourier transform infrared; CMF: cellulose microfibrils; PSA: polysodium acrylate.
From Figure 1(b) it is noticed that when sodium acrylate (monomer) is polymerized to PSA gel, the intensity of the absorption peak at 1645 cm−1 is reduced suggesting that C=C bond is converted to C–C bond.
XRD analysis
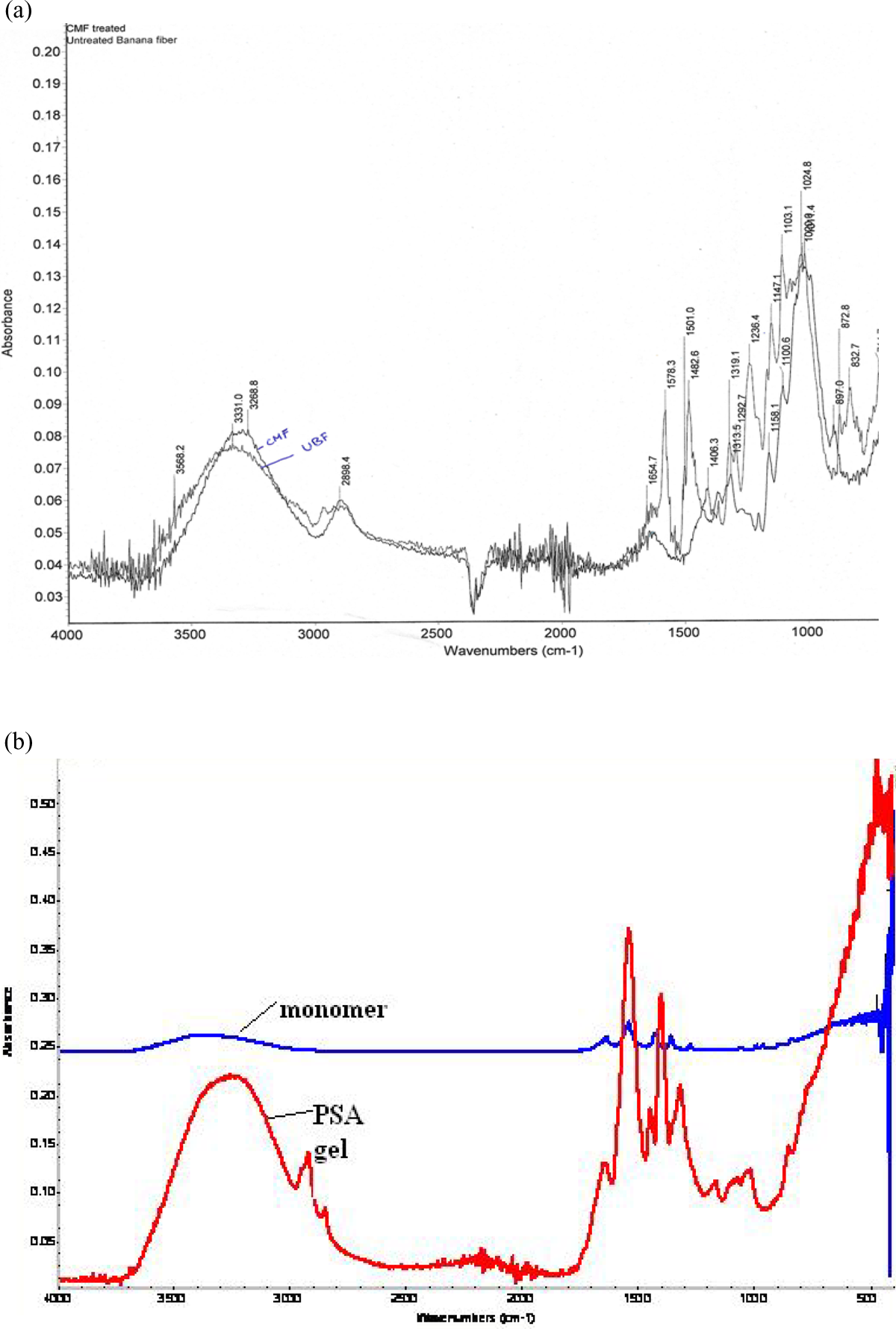
Figure 2 shows the XRD spectra of the CMF obtained from banana fiber. Characteristic diffraction peak observed at 2
XRD spectra of the CMF obtained from banana fiber. CMF: cellulose microfibrils; XRD: x-ray diffraction.
Water state characterization of PSA hydrogel network by DSC
In this study, free and bound water contents of PSA hydrogels have been measured for various cross-linker (MBA) and CMF concentrations in the distilled water. From Figure 3(a) and (b) it is observed that water in the bulk liquid phase has an ice melting point at 0°C and melting enthalpy of 333.93 J g−1. A DSC endothermic peak cannot be observed when the amount of water absorbed in the hydrogel is very low. The peaks become more apparent with increasing water swelling ratio. If the water swelling ratio further increases, the endothermic peaks at about 0°C become so strong that they hide other endothermic peaks merge progressively to become broader at high swelling ratio.
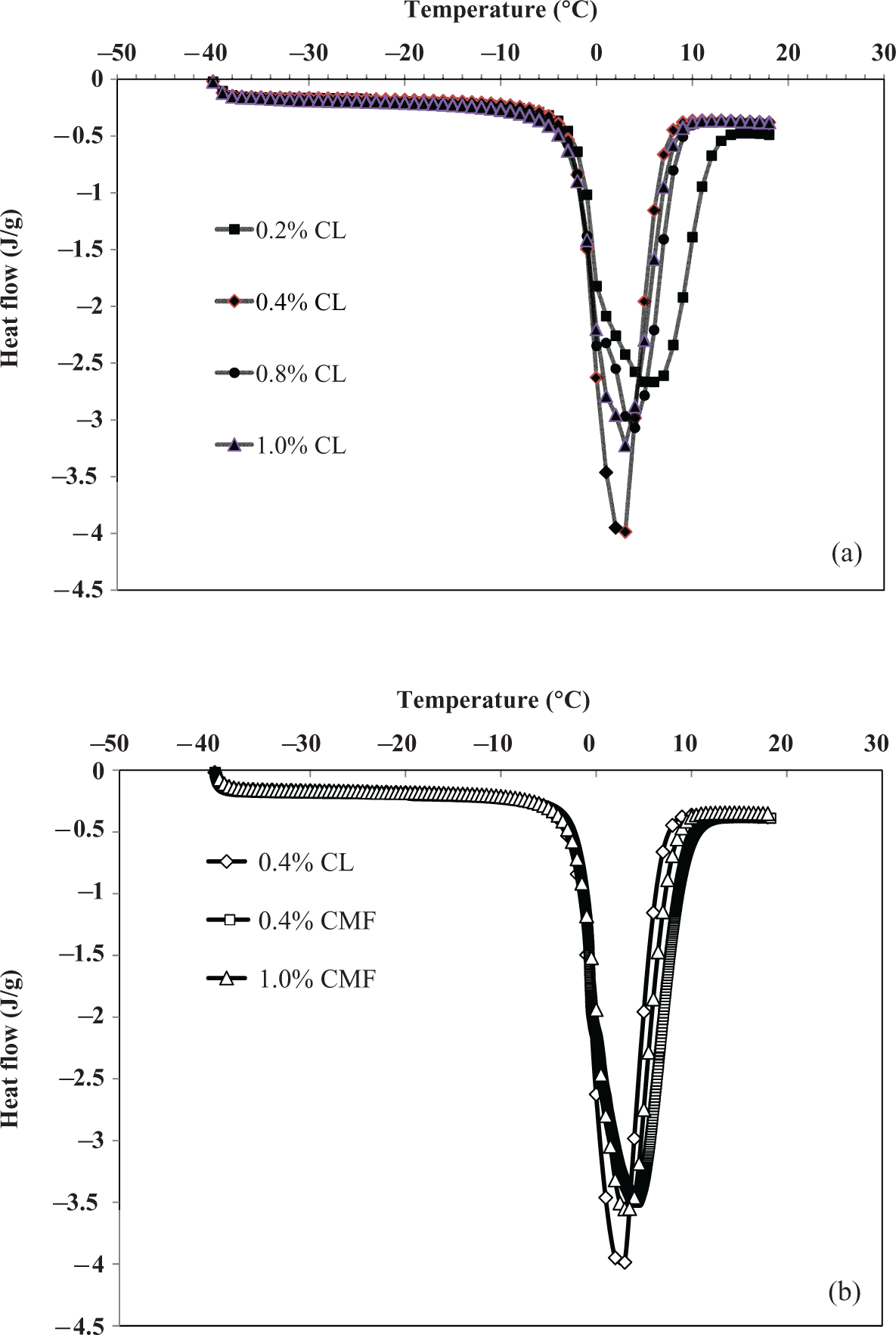
(a) DSC thermograms of PSA hydrogels at equilibrium swollen state in the distilled water at room temperature for different cross-linker (MBA) concentrations. (b) DSC thermograms of 0.4% cross-linked PSA hydrogels at equilibrium swollen state in the distilled water at room temperature for different CMF concentrations. DSC: differential scanning calorimetry; PSA: polysodium acrylate; CMF: cellulose microfibrils; MBA:
The water molecules which melt at a temperature close to 0°C are assigned to free water (mostly, water molecule clusters), and those that melt at lower temperature are assigned to freezable-bound water (mostly, second layer water molecules).
Effect of cross-linker
From Table 1, it is observed that both swelling ratio and total water content decreases as the cross-linker concentration is increased. As the cross-linker concentration increases, the number of covalent cross-link points within the hydrogel also increases. This covalent cross-link points may prevent the diffusion of water molecules into hydrogel and hence decreases both swelling ratio and total water content. At the same time, it is also noticed that with increasing cross-linker concentration, bound water increases as the amount of free water decreases. Increasing cross-linker concentration causes hydrogel structure more rigid, which reduces the mobility of water molecules and hence decreases free water content. Normally, bound water interacts with polymer network chains through hydrogen bonds. As the cross-linker concentration increases, the interaction between water molecule and polymer network also increases. This causes increase in bound water content.
Free and bound water content of different PSA hydrogels calculated from DSC thermograms.
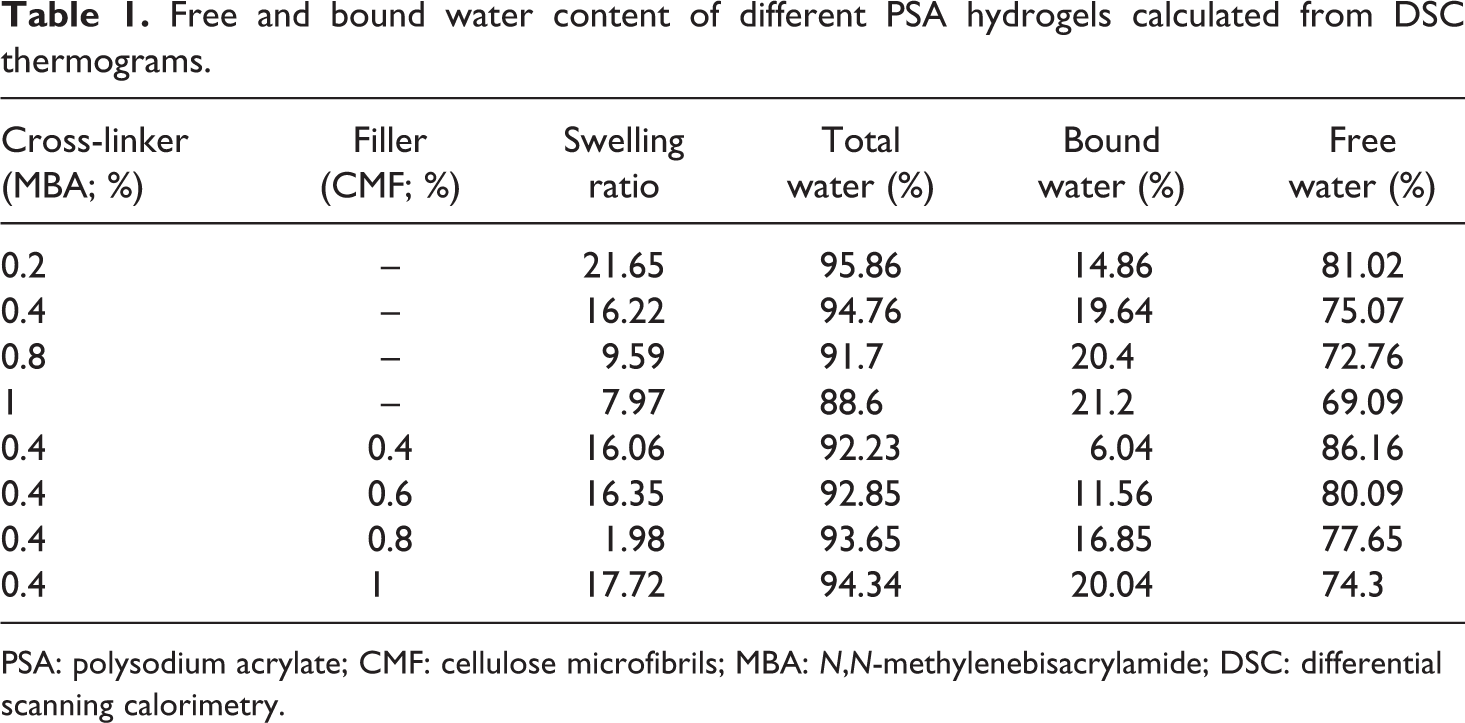
PSA: polysodium acrylate; CMF: cellulose microfibrils; MBA:
Effect of CMF concentration
Table 1 shows both swelling ratio and total water content increases with increasing CMF concentration. As the CMF concentration increases, number of hydrophillic groups also increases, which enhances the diffusion of water molecules inside the hydrogel resulting in the increase in both swelling ratio and total water content. Furthermore, it is also observed that bound water content is increased as free water content decreases. Whenever hydrogel is filled with CMF, the hydrophilic groups increase the interaction of water molecules within the network through hydrogen bonding and hence increase the amount of bound water. At the same time, the increase in CMF content decreases the amount of free water inside the hydrogel. Addition of CMF leads to increase in rigidity of the hydrogel. As CMF content increases in cross-linked gels, it prevents the mobility of water molecules by holding them through hydrogen bonding.
Dynamic mechanical analysis of PSA hydrogels
Figure 4(a) to (c) shows the dynamic mechanical properties of PSA hydrogels with different cross-linker concentrations. Both storage and loss moduli decrease with increasing temperature. It suggests that irrespective of the cross-linker concentration, PSA gels become more and more flexible with increasing the temperature. As the cross-linker concentration increases, both the moduli increase. Addition of cross-linker concentration increases the number of covalent cross-link points within the hydrogel. Due to more number of covalent cross-link points, the hydrogel becomes stiffer and hence improving the moduli. The peak position in tanδ curve related to the glass transition temperature (
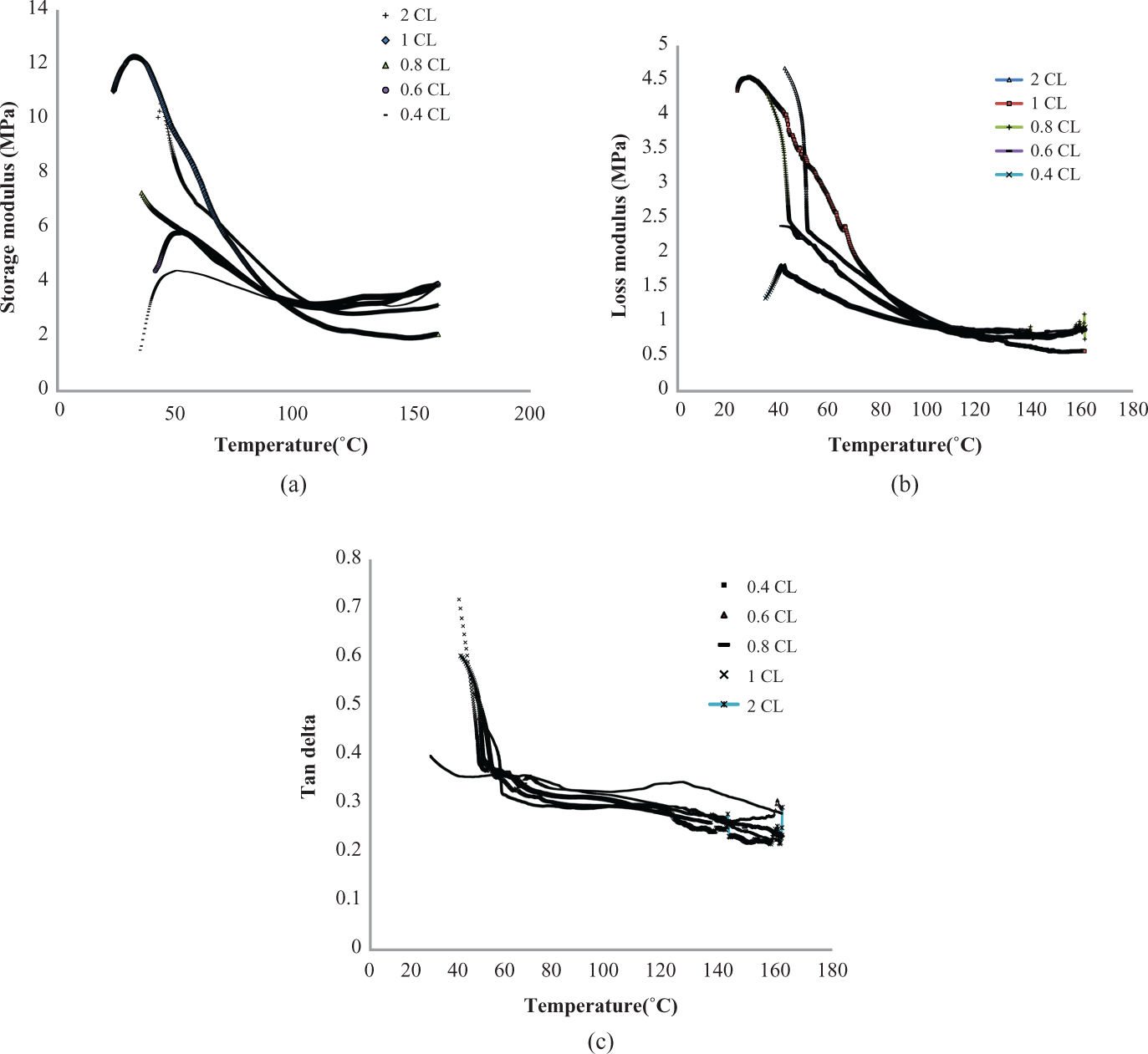
(a) Storage modulus of PSA gels with different cross-linker (MBA) concentrations. (b) Loss modulus of PSA hydrogels with different cross-linker (MBA) concentrations. (c) Damping factor of PSA hydrogels with different cross-linker concentrations. PSA: polysodium acrylate; MBA:
Figure 5(a) and (b) shows the temperature dependence modulus curves of PSA/CMF hydrogels. It is observed that an improvement in both the moduli has been observed when comparing with unfilled hydrogels. Furthermore, the moduli increase with CMF concentration. Addition of CMF as a filler reinforces the PSA hydrogels and hence improving the modulus. This significant improvement in the modulus with addition of CMF makes the week PSA hydrogels become stiff and strong and may be utilized in several biomedical applications. There is no significant difference in
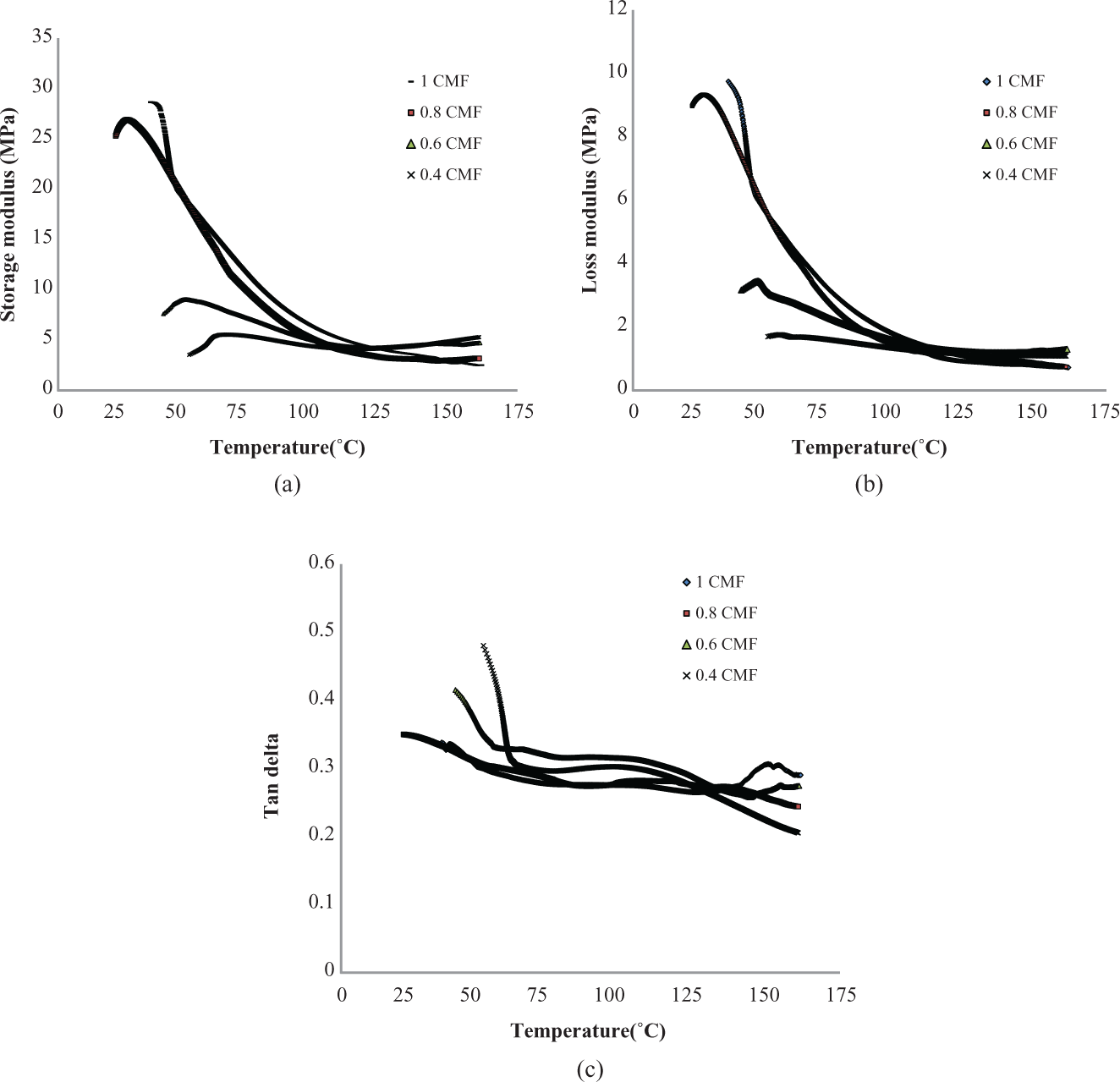
(a) Storage modulus of different PSA/CMF hydrogels as a function of temperature. (b) Loss modulus of different PSA/CMF hydrogels as a function of temperature. (c) Damping factor of different PSA/CMF hydrogels as a function of temperature. PSA: polysodium acrylate; CMF: cellulose microfibrils.
Morphology of PSA/CMF hydrogels
The scanning electron micrograph of different PSA/CMF hydrogels has been shown in Figures 6(a) to (f). CMF shows an entangled nature of fibrils as evident from the micrograph shown in the Figure 6(a). From Figure 6(b) to (d), a clear difference in the surface morphology of three samples (non-cross-linked, cross-linked and CMF filled) has been observed. In case of non-cross-linked gel, no patterns have been observed, whereas in cross-linked gel, there is an evidence of 3D polymer networks wherein water pockets are retained. On the contrary, in the swollen hydrogel (Figure 6(d)), the expansion of the network is clearly observed. The visible macropores observed at any magnification indicates that water molecules can be accommodated and easily diffused in and out. After incorporation of CMF as filler, the pore size is reduced and shows an entanglement network structure of the gel. The entangled network has been seen in the filled nonswollen and swollen hydrogels, which are shown in the Figure 6(e) and (f), respectively.

(a) SEM of CMF showing entangled nature of fibrils. (b) SEM of non-cross-linked PSA hydrogel showing no patterns. (c) SEM of 0.4% cross-linked PSA hydrogel showing pattern. (d) SEM of 0.4% cross-linked PSA hydrogel showing the expansion of three-dimensional network in the swollen stage. (e) SEM of cross-linked PSA/CMF hydrogel showing the kind of entanglements of network filled with fibrils. (f) SEM of cross-linked PSA/CMF hydrogel in the swollen state showing the expansion of the network. PSA: polysodium acrylate; CMF: cellulose microfibrils; SEM: scanning electron micrograph.
Conclusion
The structure of PSA hydrogels has been modified using MBA and CMF as a cross-linker and filler, respectively. DSC investigation indicates that both cross-linker and filler alter the state of water, particularly bound and free water inside the hydrogel. Addition of hydrophilic CMF not only alters the state of water but also alters the dynamic mechanical properties. From DMA analysis, it is clear that the addition of CMF inside the PSA matrix leads to significant improvement in both storage and loss modulus. Balance between hydrophillicty and mechanical properties has been noticed by reinforcing CMF inside the PSA matrix. SEM analysis reveals the clear distinction between different types of hydrogels.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
