Abstract
The effect of processing conditions, nucleating agent and compatibilizer on the crystallinity, mechanical property and oxygen permeability of polypropylene was studied using differential scanning calorimetry, x-ray diffraction, thermogravimetric analysis and universal tensile testing. The as received polymer had much lower extent of crystallinity, which was enhanced to 58% by the processing and molding using either slow cooling or fast cooling conditions. Although the overall crystallinity was not affected by the rate of cooling, the crystallization behavior was, however, affected as indicated by different onset and peak crystallization temperatures as well as different crystallite sizes indicated by the differences in oxygen permeation and mechanical performance of the polymer. Nucleating agent further enhanced the extent of crystallinity (crystal plane peaks were shifted and broadened) and tensile modulus, but the polymer was observed to become brittle and had higher gas permeation through it. The compatibilizer, on the other hand, reduced the tensile modulus, but enhanced the yield strain owing to matrix plasticization and retained the similar oxygen permeation as the polymer. The temperature used during processing also affected the oxygen permeation and mechanical property as the high temperature may induce partial polymer degradation resulting in the reduction of molecular weight and deteriorated properties.
Keywords
Introduction
Polypropylene (PP) is a linear hydrocarbon polymer containing little or no unsaturation. Owing to the presence of a methyl group attached to alternate carbon atoms on the chain backbone, the properties of PP can be affected in a number of ways.1–4 A slight stiffening of the chain occurs in the structure owing to the pendant alternate methyl group and it also interferes with the molecular symmetry. An increase in the crystalline melting point is observed owing to the first effect, whereas the interference with molecular symmetry tends to depress it. The morphological structure of PP is rather complex and at least four different types of spherulite structures have been observed. The properties of the polymer depend on the size and type of crystal structure formed, which, in turn, is dependent on the relative rates of nucleation to crystal growth. The polymer has a glass transition temperature (T g) below room temperature, but is still a rigid solid at the room temperature owing to crystallinity, which defines the useful properties of PP. Thus, the effect of processes or additives, which can affect the crystallinity of polymer, thereby, affecting the performance, is required to be understood.
The polymers undergo a number of processing steps before being formed into a useful product. Processing of polymers at high temperature is also common, which can induce certain degradation of the polymer, thus, affecting the mechanical performance as well as crystal structure and the capability of the polymer to crystallize. 5 Control of rate of cooling of the polymer during the last processing step is also vital to define the final crystal structure and extent of crystallinity of the polymer, thus, also final product properties. 6
To control the crystal structure of the semicrystalline polymers like polyethylene and PP, addition of nucleating agents is also a common route.7–10 Marco et al. 7 studied the isotactic PP nucleated with the sorbitol derivatives 1,3:2,4-bis(4-methyldibenzylidene)sorbitol and 1,3:2,4-bis(3,4-dimethylbenzylidene)sorbitol. The isothermal crystallization temperature and rate were observed to increase with increasing nucleating agent concentration. Similarly, Zhang et al. 8 used a nucleating agent based on sorbitol and toughness, and stiffness of PP was reported to increase. Thus, initiation of crystallization can be controlled by the addition of nucleating agents leading to the appearance of a substantial increase in heterogeneous nucleation and a reduction in the size of crystalline structures. Apart from various inorganic and organic nucleating agents, mineral fillers added to the polymers to enhance the mechanical properties have also been reported to have nucleation action. 11 However, such an action also depends on the processing conditions as well as the state of the filler surface. Organically modified fillers were observed to have no nucleation action on the polymer when the surface of the filler was optimally covered with the organic modifications.12,13
Low-molecular-weight compatibilizers (e.g. PP grafted with maleic anhydride (PP-g-MA)) are also commonly added to the polyolefin matrices during the generation of polymer nanocomposites to induce positive interactions between the filler and polymer phase.14–19 Compatibilizers or amphiphilic surfactants owing to their polar and nonpolar constituents can bridge the polymer and filler phases together allowing them to well mix. However, these compatibilizers are low-molecular-weight polymers, which can affect the mechanical properties and crystallinity of the matrix polymer negatively.
The goal of the current study was to study the effect of processing conditions (e.g. processing temperature, rate of cooling, etc.), organic nucleating agent and PP-g-MA compatibilizer on crystallinity, mechanical property and oxygen permeability of PP. The amount of the nucleating agent and the compatibilizer was fixed at 1 and 2 wt%, which is also similar to the amounts generally used in reality. Thermal properties were characterized using thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC), whereas crystal structure was analyzed with wide angle x-ray diffraction (WAXRD).
Experimental
Materials
PP used was the homopolymer grade H733-07 from Dow (Dow Plastics, Horgen, Switzerland). It has a melt flow index of 7.5 g/10 min (230°C at 2.16 kg load) and a density of 0.91 g/cm3. Nucleating agent based on sodium benzoate and PP-g-MA compatibilizer was procured from Aldrich (Buchs, Switzerland).
Polymer compounding and compression molding
The pure polymer and additives (nucleating agent and compatibilizer) were compounded in a twin-blade kneader ‘Plasti-Corder W 50 EH’ (Brabender, Duisburg, Germany). The mixer was equipped with a 60-cm3 cavity and counter-rotation of the rotors was used for compounding process. The polymer (either pure PP or PP dry mixed with nucleating agent and compatibilizer pellets) was molten at 170 or 190°C, sheared for 10 min at 50 r/min followed by kneading for further 10 min at 60 r/min. At the end, the compounds were quickly taken from the cavity in order to avoid polymer degradation because of prolonged contact of polymer with the hot surface of the mixer. The compounds were then compression molded to 1.5-mm-thick plaques in a brass frame between two thin aluminum plates at 220°C. Reduced pressure (0.01 mbar) was maintained to avoid insertion of air. The mold was either transferred to a cold press and quickly cooled with cold water or was left to cool slowly. To measure oxygen permeation, pieces of the plaques were pressed similarly to generate 100-µm-thick films between poly(ethylene terephthalate) foils.
Characterization techniques
The calorimetric behavior of the molded polymer plaques was studied using DSC (DSC 7, Perkin-Elmer, Norwalk, Connecticut, USA) under nitrogen at a heating rate of 10°C/min. The earlier reported procedure for measuring heat capacities was used. 20 The temperature-dependent instrumental deviations were corrected using aluminum pans (sample and reference) of the same weight; the calorimeter base line was also scanned and subtracted. A sapphire standard sample was used to calibrate the response of the calorimeter and the samples weight was kept constant (about 6 mg). Same temperature range (100–180°C) was used to measure the melting enthalpy (ΔH m) of samples and the enthalpy was adjusted to the weight of PP in the sample. Two heating and one cooling cycles were performed.
Tensile tests on the dumbbell shaped samples (type 5B) stamped out of the compression molded plates were carried out at room temperature following the standard ISO 527-2. Zwick Z020 universal testing machine with testXpert 9.01 software (Zwick, Ulm, Germany) coupled with a Video-Extensometer V4.19.02 (Messphysik, Furstenfeld, Austria) for accurate measurement was used. The drawing speed of 0.1 mm/min was used for the measurement of elastic modulus determined in the range of 0.05–0.25% strain. A speed of 6 mm/min was used for the measurement of other tensile properties and an average of five measurements was taken. WAXRD, oxygen permeation (23°C, 0% relative humidity) for the PP films and high resolution TGA on the plaque samples were performed as reported earlier.21,22 For scanning electron microscopy (SEM), the block face of the sample was sputter coated with 5 nm platinum and the morphology was observed in Hitachi S-900 field emission SEM at an accelerating voltage of 15 kV.
Results and discussion
Processing parameters, nucleating agent and compatibilizer affect the microstructure of the polymer that has direct influence on its mechanical, thermal and barrier properties. The compatibilizer used in the study had a Mn of 3900 (Mw of 9100) and had 4 wt% of MA content that corresponded to 3.7 MA units per polymer chain, thus, making it a high MA compatibilizer. Acid number of the compatibilizer was 47 mg KOH/g of PP-g-MA. The density of both the compatibilizers was 0.934 g/cm3, which was very near to that of PP.
Calorimetric properties
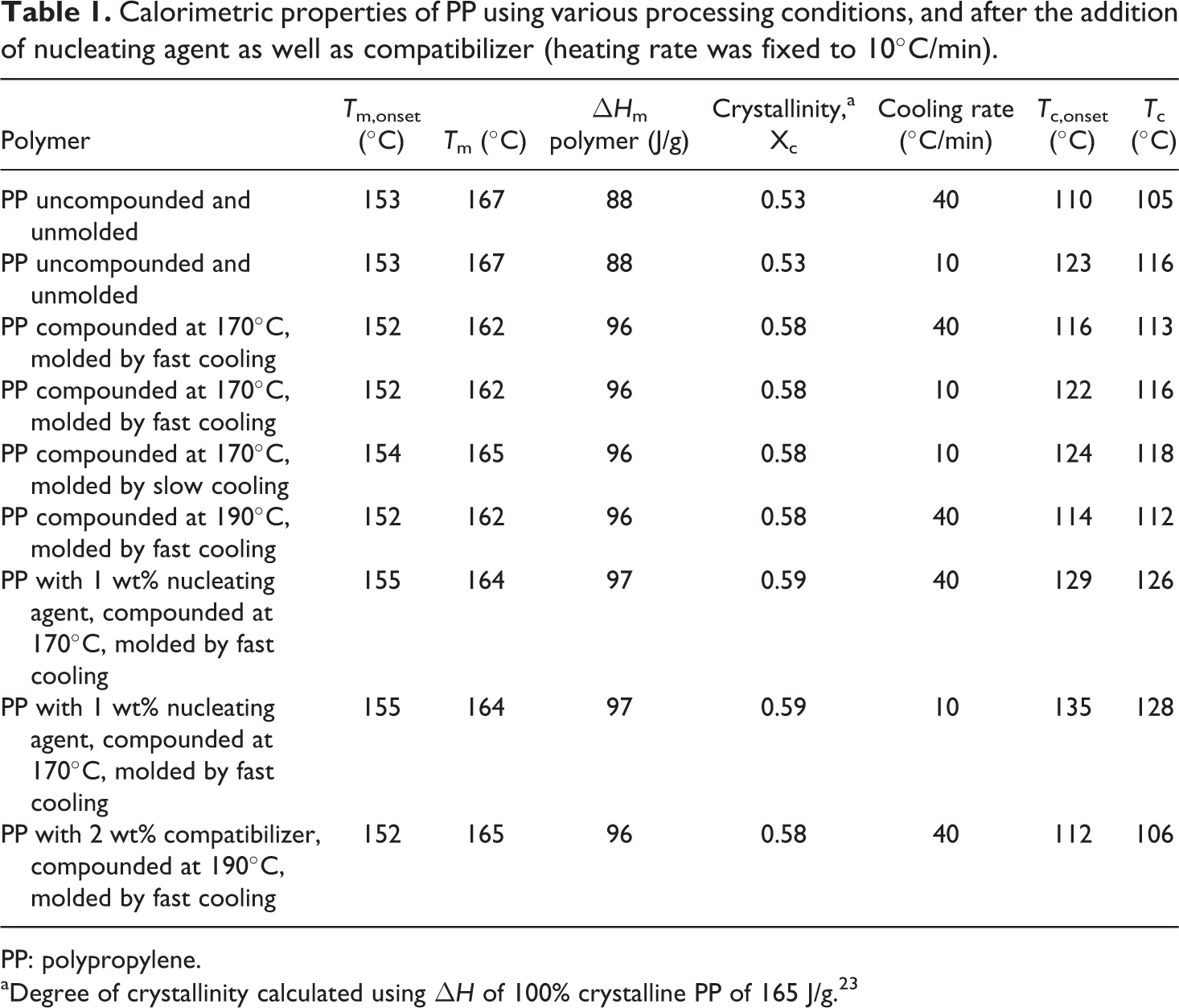
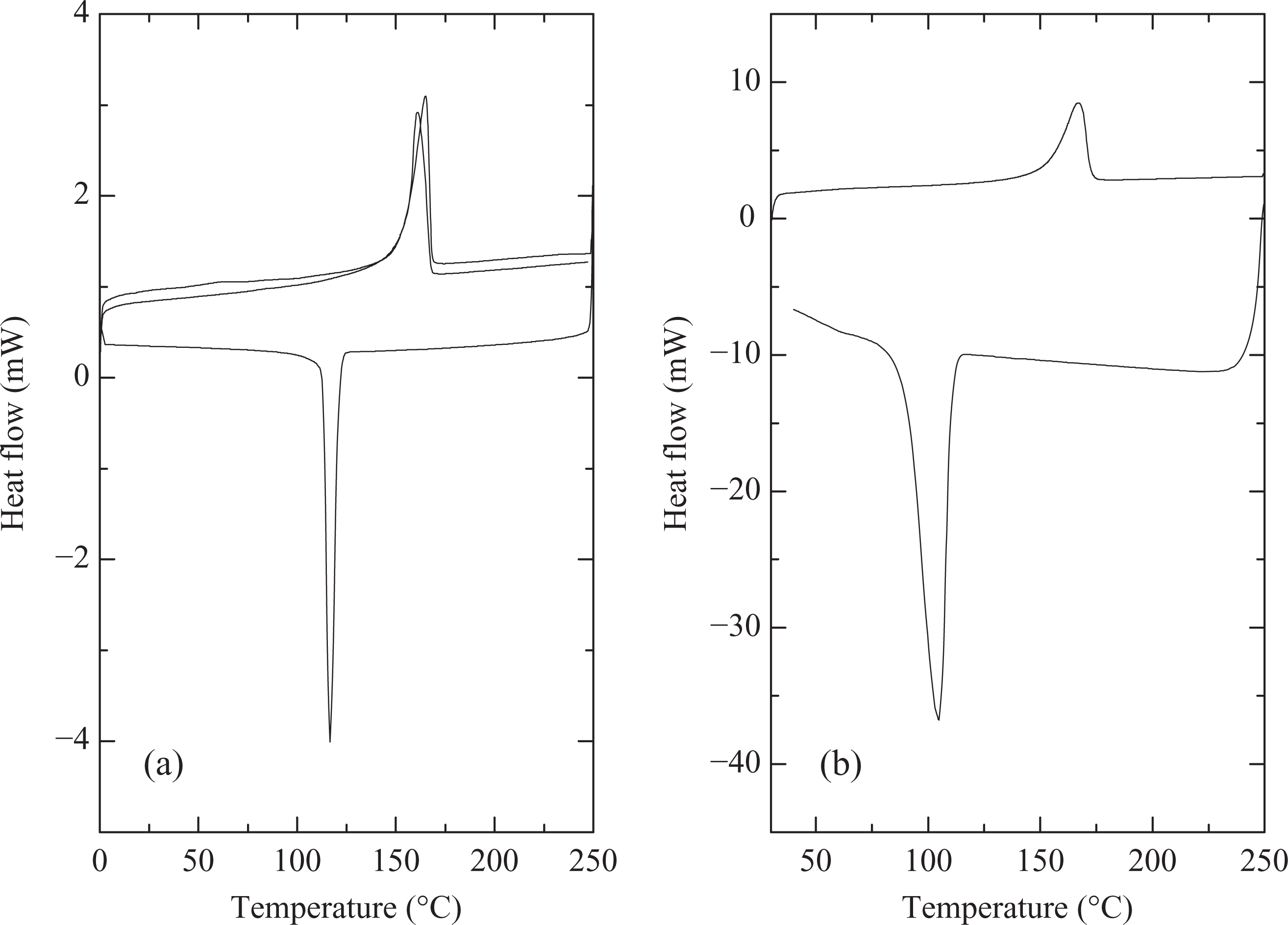
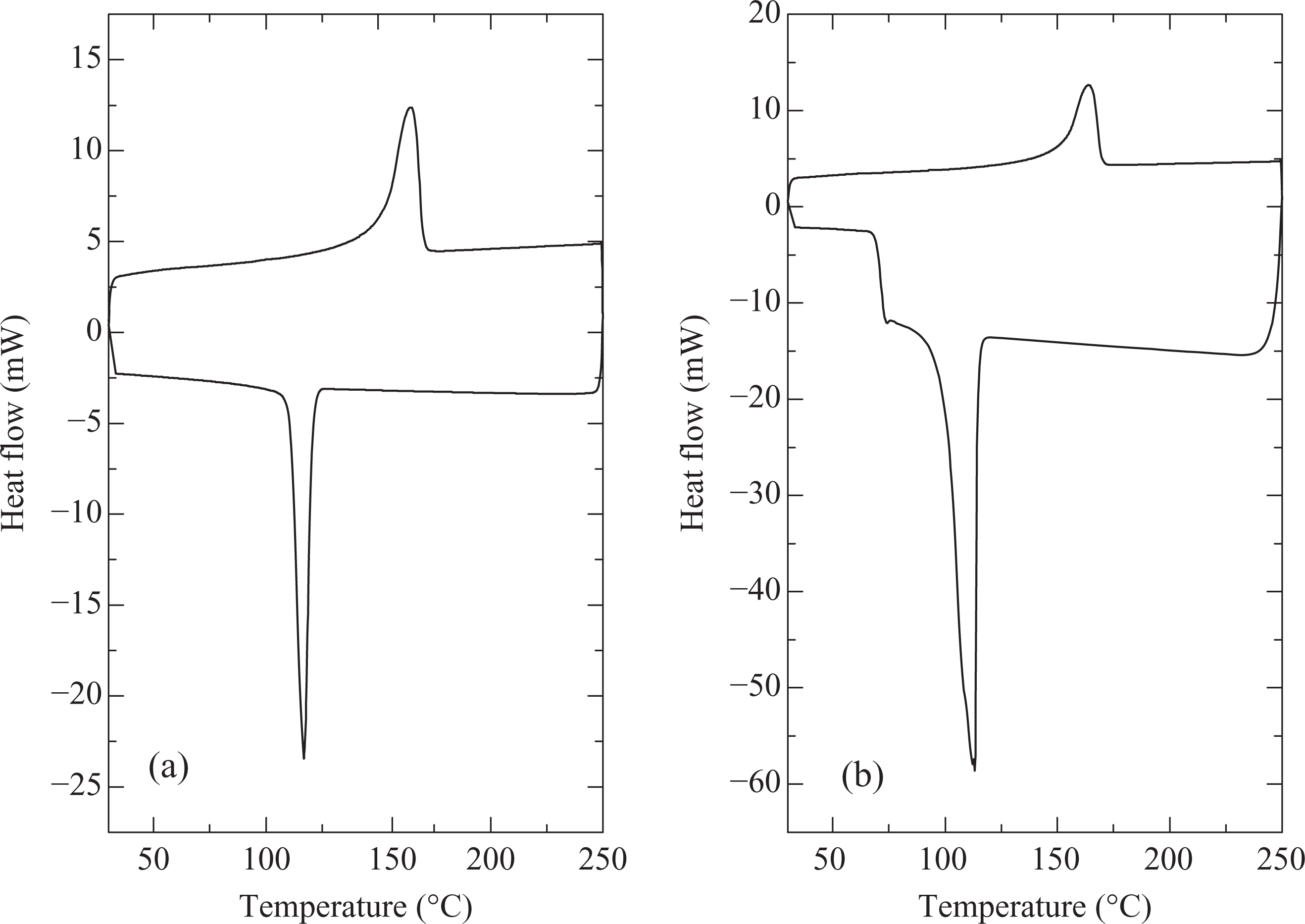
Table 1 details the calorimetric properties of PP subjected to various processing conditions or compounded with nucleating agent and compatibilizer. The heating rate was always fixed to 10°C/min, whereas cooling rates of 10 or 40°C/min were used. In Table 1, the onset of melting temperature, peak melting temperature, enthalpy and degree of crystallinity corresponding to first heating cycle of the calorimetric analysis are detailed. Uncompounded and unmolded PP (tested as received from the supplier) had a relatively lower melt enthalpy (hence degree of crystallinity calculated using ΔH of 100% crystalline PP of 165 J/g23) when compared with other processed polymers indicating the different heat history of the samples. The sample would have been cooled very fast, which did not allow the polymer chains to extensively crystallize. Cooling at two different rates in DSC (40°C/min represents fast cooling and 10°C/min represents slow cooling) showed differences in the crystallization behavior of the polymer. Onset of crystallization was significantly faster in the case of slow cooling conditions (123°C when compared with 110°C for the fast cooling). Peak crystallization temperature was also observed to be 116°C for slow cooling, when compared with 105°C for fast cooling conditions (Figure 1). Figure 1 also shows the DSC thermograms for the uncompounded and unmolded PP with two different cooling rates. Also, as shown in Figure 1, the second heating cycle for the PP cooled at 10°C/min (Figure 1(a)) showed a slightly higher heat flow (a measure of melt enthalpy) signal when compared with first cycle owing to higher extent of crystallization. Apart from that, the crystallization transition was also sharp at the slow cooling conditions. Similarly, Figure 2 shows the DSC thermograms of the polymer compounded at 170°C and molded by fast cooling. Peak melting temperature of 162°C was observed and the polymer had 58% crystallinity. Cooling the polymer in DSC using two different rates similarly showed higher onset of crystallization and peak crystallization temperatures for the slow cooled sample (116 and 122°C, respectively) when compared with the fast cooling conditions (113 and 116°C, respectively). The crystallization transition was sharp in case of slow cooling conditions, as observed earlier. Figure 3(a) also shows the DSC thermograms of the polymer compounded at 170°C and molded by slow cooling, when heating and cooling rates of 10°C/min were used. Slightly higher onset of crystallization and peak crystallization temperatures were observed; however, the melt enthalpy was similar to the case when the polymer was molded at fast cooling conditions. Although PP molded by slow or fast cooling had no difference in melt enthalpy, the crystallite size can still be expected to be different, thereby, affecting other properties of the polymer. The reproduction of the similar heating curve in the second cooling cycle indicated that the polymer was optimally crystallized using the cooling rate of 10°C/min. PP compounded at 190°C and molded by slow cooling had similar calorimetric behavior as the polymer treated at 170°C indicating that the processing temperature at the slowly cooled molding conditions did not affect the polymer crystallization behavior. For the fast cooled molding conditions, there were differences in the polymer calorimetric behavior depending on the processing temperature.
Calorimetric properties of PP using various processing conditions, and after the addition of nucleating agent as well as compatibilizer (heating rate was fixed to 10°C/min).
PP: polypropylene.
aDegree of crystallinity calculated using ΔH of 100% crystalline PP of 165 J/g. 23
DSC thermograms of uncompounded and unmolded PP using heating rate of 10°C/min and cooling rate of (a) 10°C/min and (b) 40°C/min. DSC: differential scanning calorimetry; PP: polypropylene.
DSC thermograms of PP compounded at 170°C and molded by fast cooling using heating rate of 10°C/min and cooling rate of (a) 10°C/min and (b) 40°C/min. DSC: differential scanning calorimetry; PP: polypropylene.
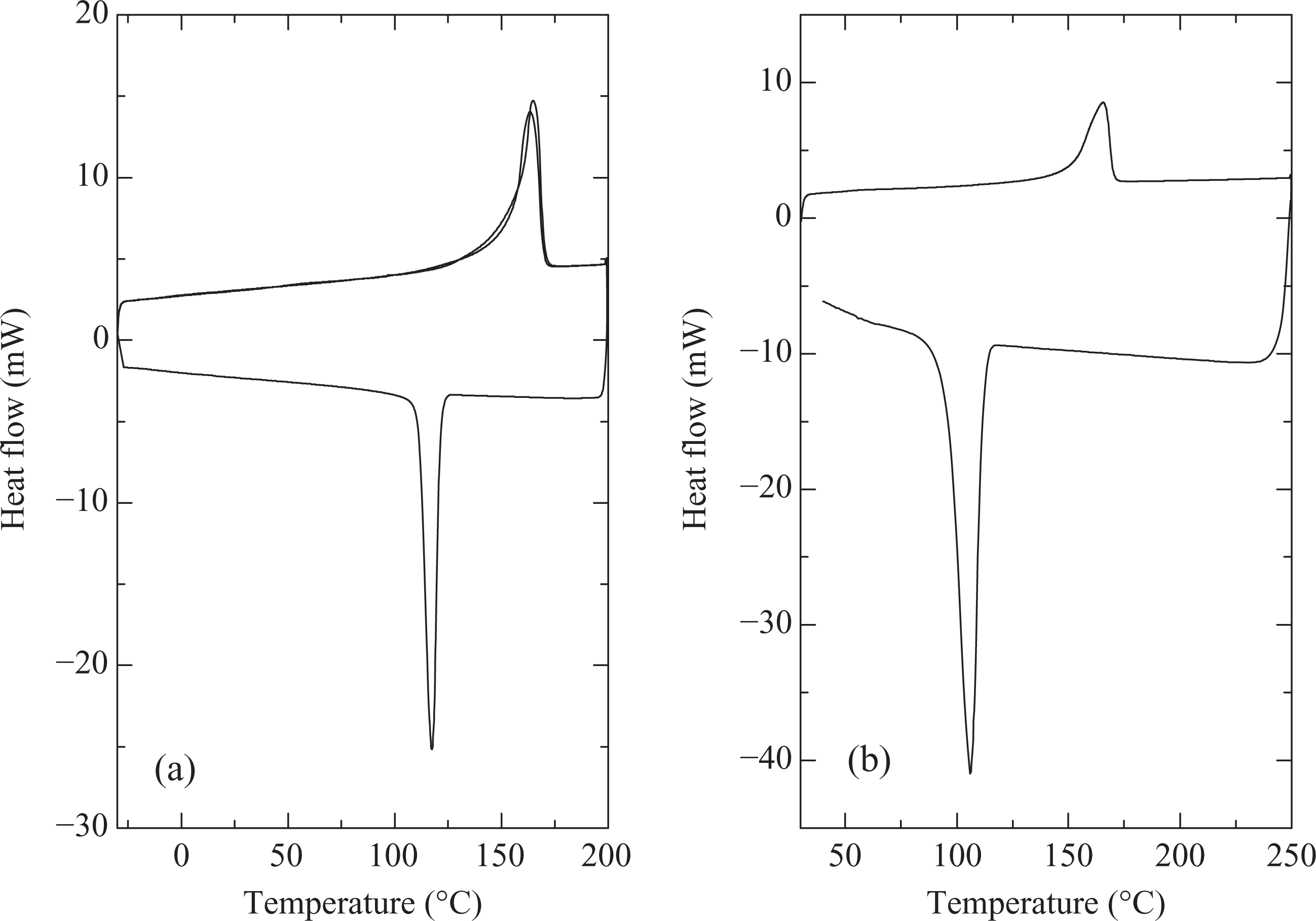
Figure 3(b) shows the DSC thermograms of polymer compounded with 2 wt% compatibilizer at 190°C and compression molded by fast cooling. Although the overall melt enthalpy and degree of crystallinity were not affected when compared with the pure polymer processed at 170 or 190°C, the peak crystallization temperature was significantly reduced to 106°C. Figure 4 also shows DSC thermograms of PP with 1 wt% of nucleating agent (compounding at 170°C, molding by fast cooling). Onset of melting was observed at 155°C and peak melting temperature of 164°C was measured. The melt enthalpy and hence extent of crystallinity was slightly higher than the polymer processed without nucleating agent at similar conditions. The sample with slow cooling showed onset and peak crystallization temperatures of 128 and 135°C respectively, whereas these temperatures were observed to be 126 and 129°C at fast cooling cycle of the sample. Thus, in both the cases, faster nucleation when compared with polymer without nucleating agent was observed. The second heating cycle also overlapped exactly with the first heating in the case of slow cooling of the sample, confirming the earlier findings of optimum crystallization at the rate of 10°C/min. Figure 5 also demonstrates the uniform formation of polymer crystals on the addition of 1 wt% nucleating agent. These findings coincide with the results reported earlier by Avella et al. 6 for sodium benzoate nucleated isotactic PP. The nucleating agent influenced only the size of spherulites and not the final crystallinity, which indicated that the nucleating agent promoted rapid crystallization of the material at low temperatures. Marco et al. 7 also reported the maximum increase in the isothermal crystallization temperature and rate at 0.3 wt% of the sorbitol-based nucleating agents. Very fine and uniform spherulites were similarly observed by Zhang et al. 8 on the addition of 0.2 wt% of sorbitol derivative as nucleating agent to PP/poly(ethylene-octene) blends.
DSC thermograms of PP: (a) compounding at 170°C, molding by slow cooling, heating and cooling rates of 10°C/min and (b) with 2 wt% compatibilizer, compounding at 190°C, molding by fast cooling, heating rate of 10°C/min, cooling rate of 40°C/min. DSC: differential scanning calorimetry; PP: polypropylene.
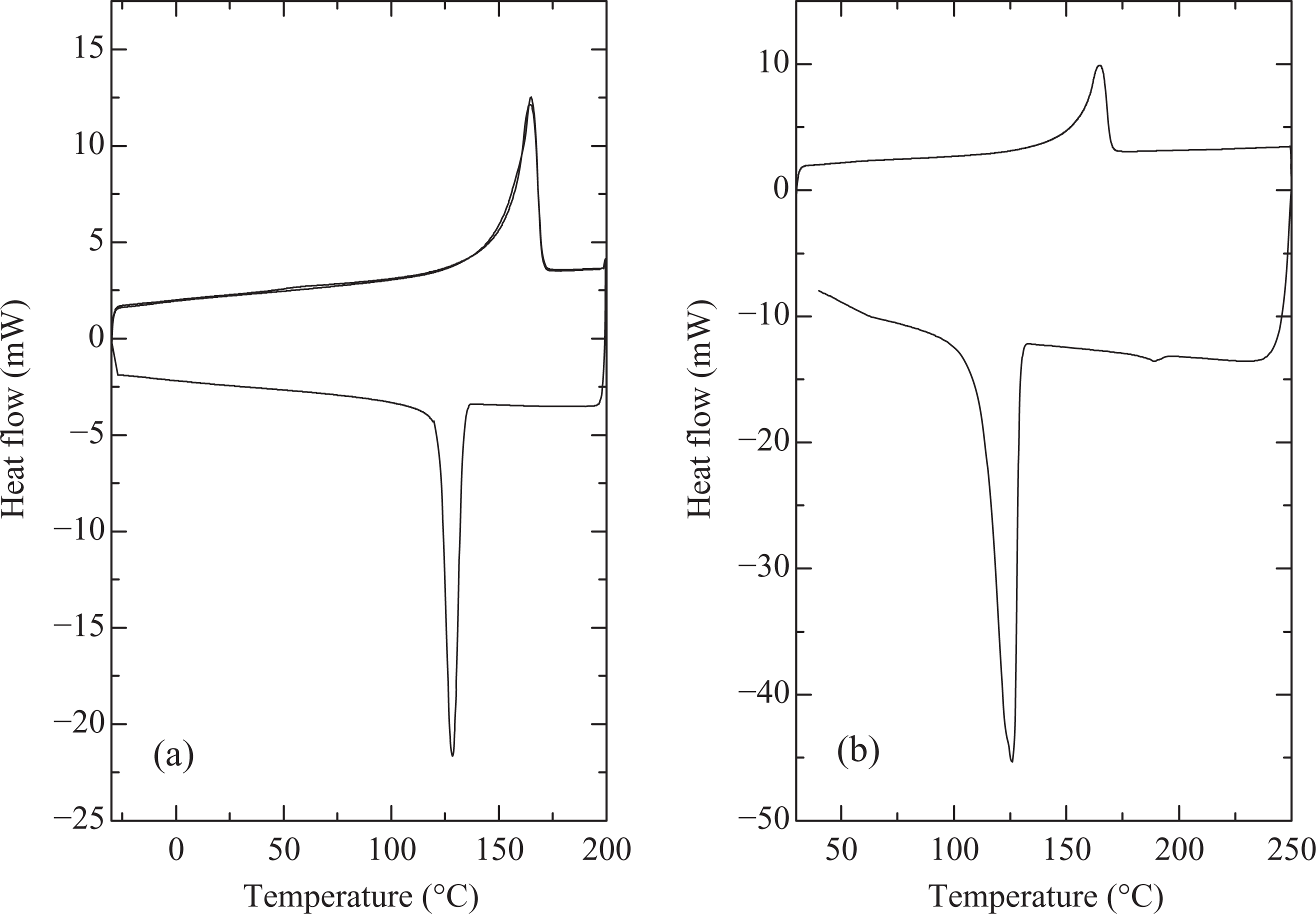
DSC thermograms of PP with 1 wt% of nucleating agent (compounding at 170°C, molding by fast cooling) using a heating rate of 10°C/min and cooling rate of (a) 10°C/min and (b) 40°C/min. DSC: differential scanning calorimetry; PP: polypropylene.

SEM micrograph of PP with 1 wt% nucleating agent depicting the uniform formation of crystals. SEM: scanning electron microscopy; PP: polypropylene.
Oxygen permeation properties
Table 2 demonstrates the oxygen permeation property of PP films. Oxygen permeation coefficient of 84.5 cc µm/(m2 d mmHg) was observed for PP compounded at 170°C and molded by fast cooling. The oxygen permeation, however, increased to 90.8 cc µm/(m2 d mmHg) for PP compounded at 170°C and molded by slow cooling. Although the extent of crystallinity was observed to be similar in both the cases, differences in crystallite size and number can be expected (also owing to earlier onset of crystallization in slow cooling conditions). The generation of larger-sized crystals during slow cooling can result in brittleness of the structure leading to voids in the films, which increases the oxygen permeation through the polymer. Similar observations were also made for the polymer compounded at 190°C. Oxygen permeation coefficient of 88.9 cc µm/(m2 d mmHg) was observed for PP compounded at 190°C and molded by fast cooling, whereas PP compounded at 190°C and molded by slow cooling exhibited an oxygen permeation coefficient of 97.9 cc µm/(m2 d mmHg). It is also to be noted that the polymer processed at 190°C showed higher oxygen permeation than the polymer processed at 170°C. It indicates that the high temperature processing may have thermally degraded the polymer. PP with 1 wt% nucleating agent showed an oxygen permeation coefficient of 92.7 cc µm/(m2 d mmHg) indicating that the increased crystallinity deteriorated the oxygen permeation resistance of the polymer. However, the observed deterioration cannot be assigned to the crystalline structure itself, but to the defects associated with it like crystallite boundary mismatch with other crystallites and brittleness. The addition of a small amount of compatibilizer surprisingly enhanced the barrier performance of the polymer as an oxygen permeation coefficient of 82.8 cc µm/(m2 d mmHg) was measured through it. A much lower peak crystallization temperature was observed in this case possibly owing to lower crystallite size, which again underlines the importance of controlling the crystallite size for better barrier performance of the polymer.
Oxygen permeation properties of PP with various processing conditions, nucleating agent as well as compatibilizer.

PP: polypropylene.
Thermogravimetric analysis
In order to analyze the effect of additives on the thermal performance of polymer, high-resolution TGA of the polymer compounds was carried out. Figure 6 shows the TGA thermograms of PP, PP compound with nucleating agent and PP compound with compatibilizer. However, it was obvious that the addition of small amount of additives though affected other properties of the polymer, the thermal performance of the polymer was not affected by their presence. The onset of degradation as well as peak degradation of polymer followed the same mechanism in the presence or absence of nucleating agent and compatibilizer. Only after most of the polymer had degraded (around 70%), the behaviors showed slight differences. The polymer compound with compatibilizer had further sharp degradation, whereas the pure polymer and polymer compound with nucleating agent showed slightly better thermal stability.
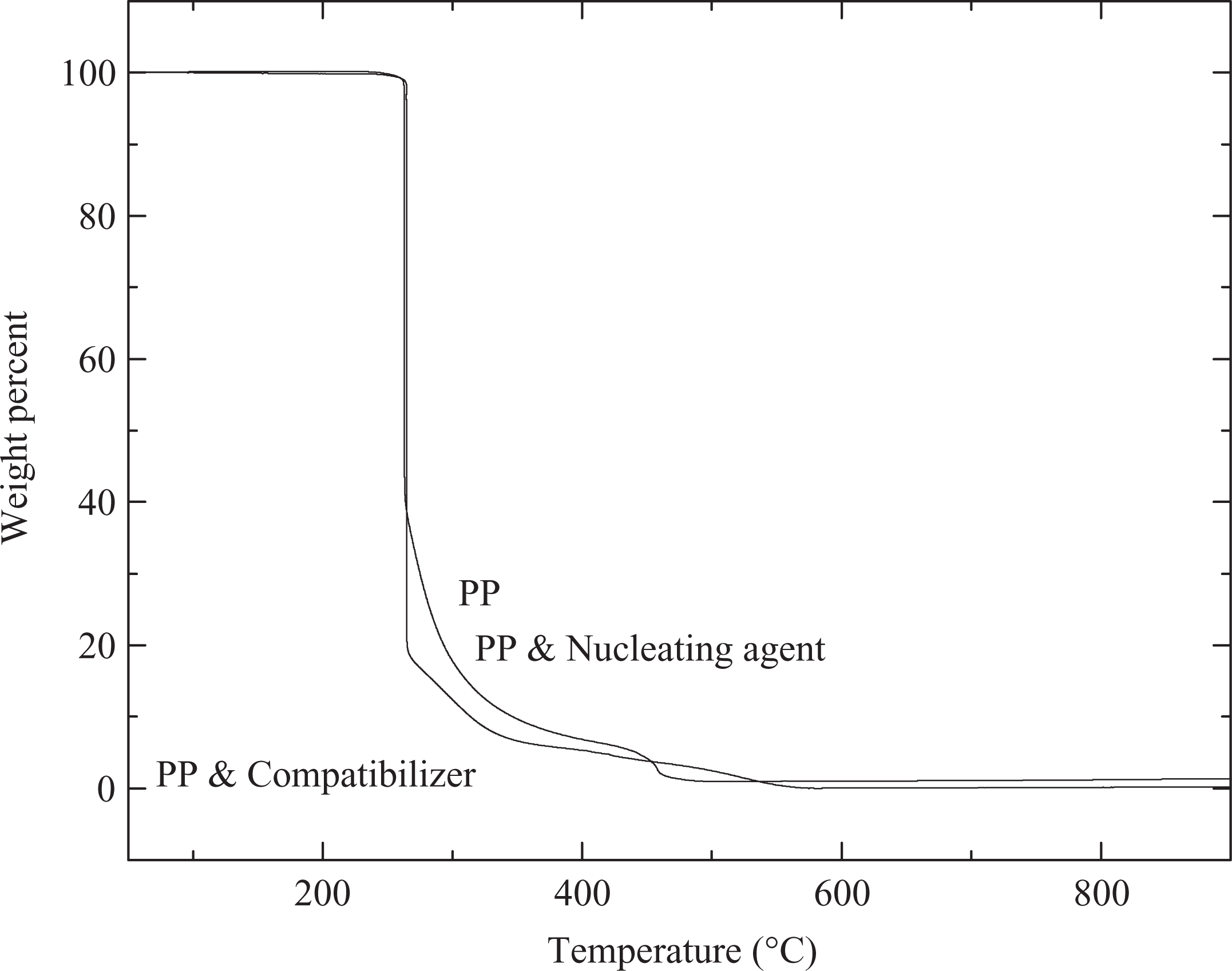
TGA thermograms of PP, PP compound with nucleating agent and PP compound with compatibilizer. PP: polypropylene; TGA: thermogravimetric analysis.
X-Ray diffraction
Figure 7 shows the x-ray diffractograms of PP compounded at 170°C and molded by slow cooling, PP compounded at 170°C and molded by fast cooling, PP compounded with nucleating agent at 170°C and molded by fast cooling and PP compounded with compatibilizer at 190°C and molded by fast cooling. The diffractograms seem similar, thus, indicating that no extensive changes occurred in the crystal structure, but the x-ray diffraction curve corresponding to the (041) plane (peak between 20° and 22° 2θ) was observed to be broad in case of polymer compound with nucleating agent. It signified the variation in the size of crystal grain. The intensity of this diffraction peak also reduced. 9 Apart from that, the peak maximum of the (110) plane with peak between 12° and 16° 2θ and (130) plane with peak between 18° and 20° 2θ also shifted to higher angles in the case of polymer compound with nucleating agent further signifying changes in the crystal structure the properties of which are reported in Table 1.
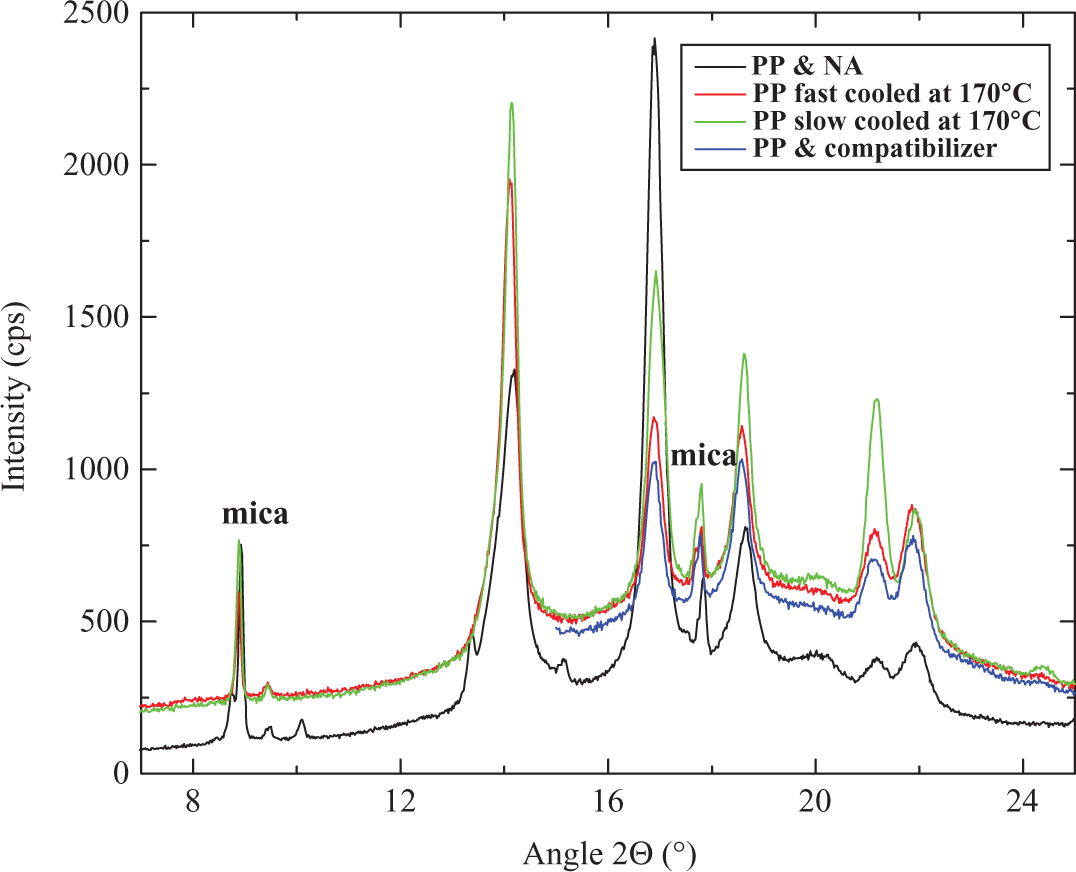
X-Ray diffractograms of (1) PP compounded at 170°C and molded by slow cooling, (2) PP compounded at 170°C and molded by fast cooling, (3) PP compound with nucleating agent made at 170°C and molded by fast cooling and (4) PP compound with compatibilizer made at 190°C and molded by fast cooling. PP: polypropylene.
Tensile properties
To further analyze the effect of processing conditions and additives on the mechanical performance, tensile testing of the samples was carried out. Table 3 details the mechanical properties of PP using various processing conditions, nucleating agent and compatibilizer. The tensile modulus for PP compounded at 170°C and molded by fast cooling was observed to be 1631 MPa, which was higher than 1510 MPa measured for the polymer compounded at 190°C and molded by fast cooling. It also coincides with the earlier finding of better oxygen permeation resistance for the polymer processed at lower temperature owing to probable partial damage to the polymer structure when processed at higher temperatures. The yield stress and yield strain were also higher for the polymer processed at lower temperature indicating that the polymer processed at higher temperature also became slightly brittle. Figure 8 also shows the representative stress–strain correlations. Lower strain of the polymer compounded at 190°C and molded by fast cooling when compared with PP compounded at 170°C and molded by fast cooling is evident.
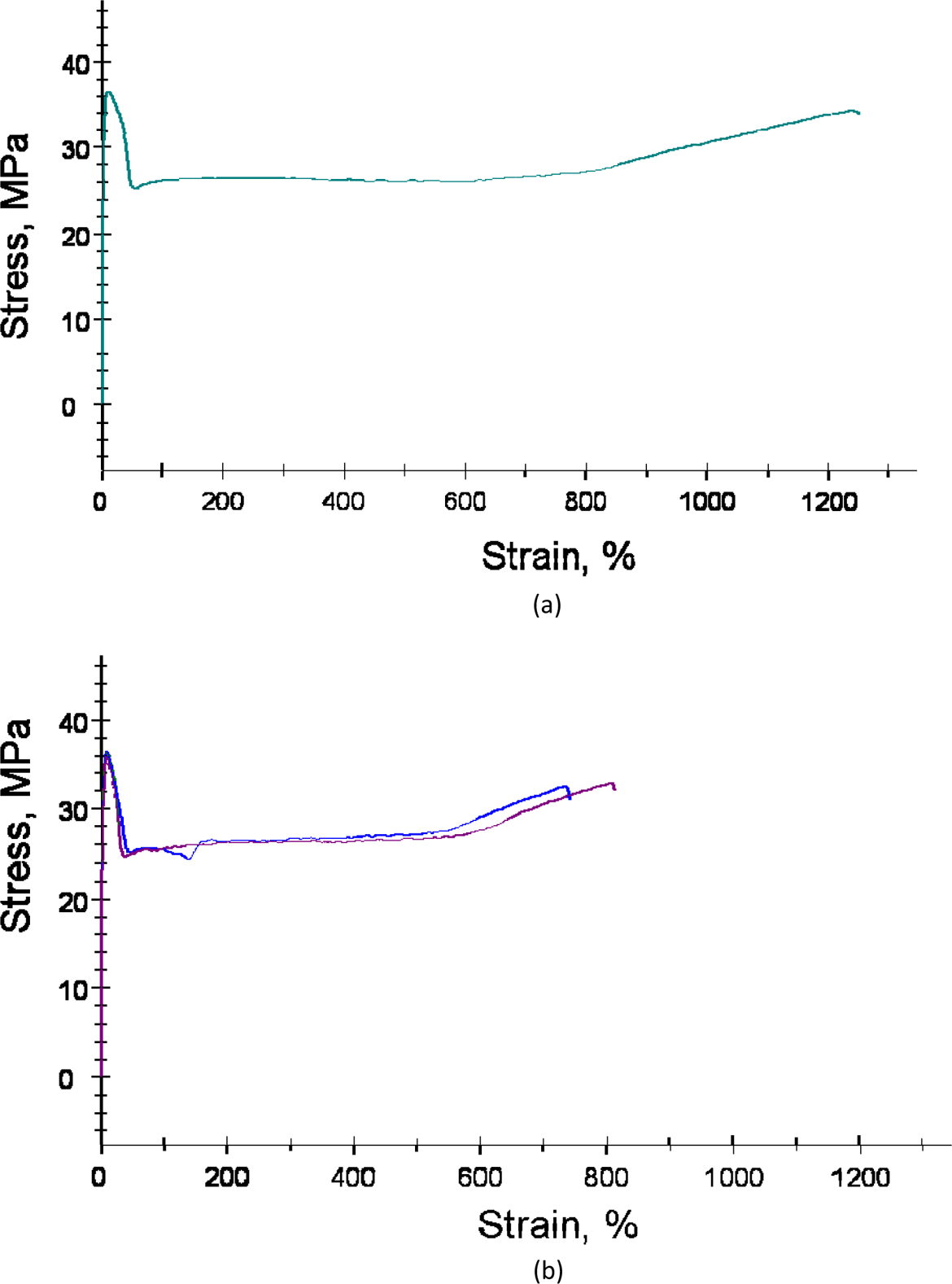
Stress–strain correlations for (a) PP compounded at 170°C, molded by fast cooling and (b) PP compounded at 190°C, molded by fast cooling. PP: polypropylene.
Mechanical properties of PP using various processing conditions, nucleating agent as well as compatibilizer.
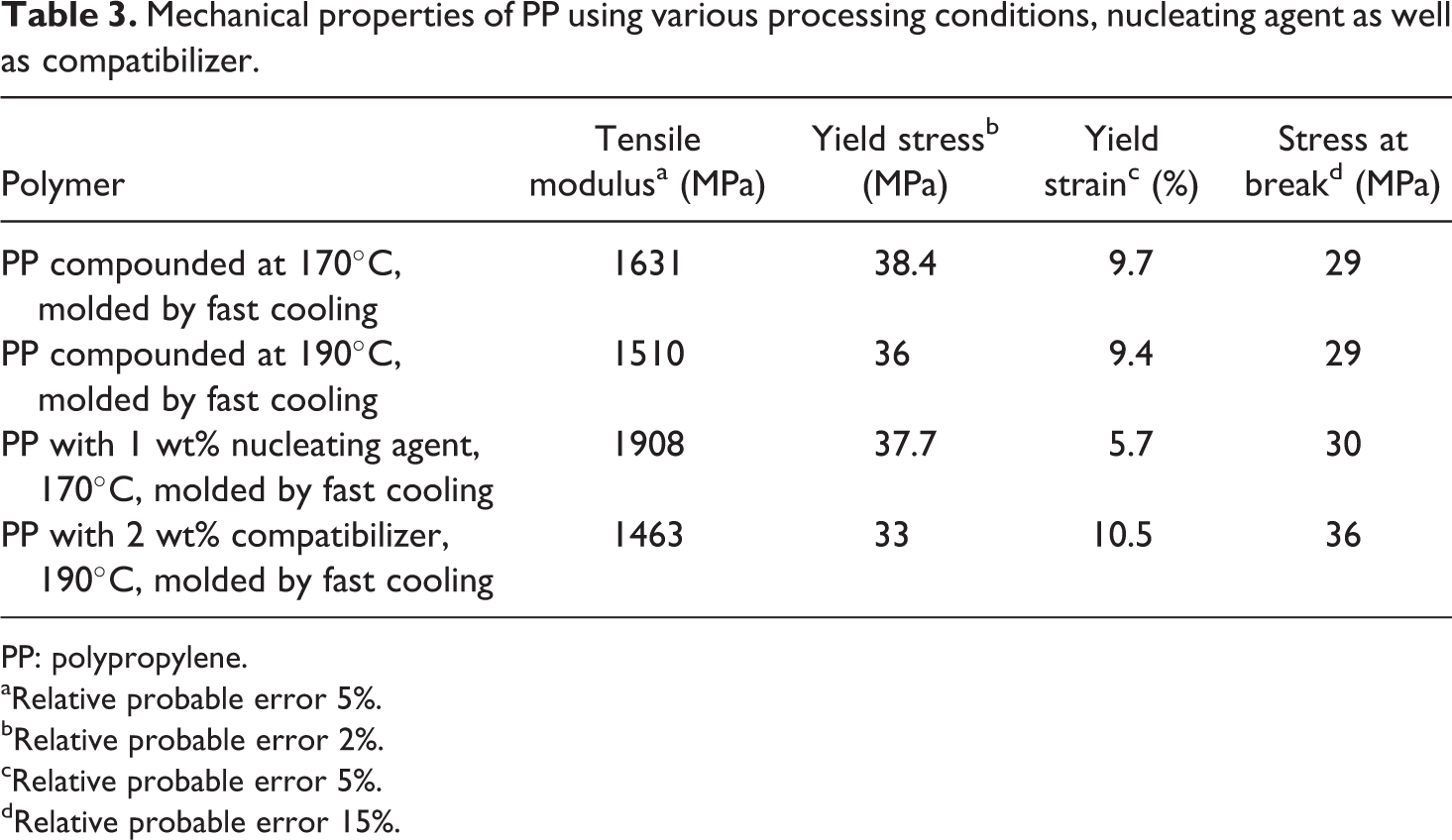
PP: polypropylene.
aRelative probable error 5%.
bRelative probable error 2%.
cRelative probable error 5%.
dRelative probable error 15%.
Much higher enhancement in tensile modulus was observed for PP with 1 wt% nucleating agent when compounded at 170°C and molded by fast cooling (Figure 9(a)). When compared with similarly processed polymer without nucleating agent, the modulus increased by nearly 20%. The yield strain in the nucleated sample also decreased significantly confirming that the polymer became brittle on the addition of nucleating agent. Zhang et al. 8 also reported similar findings for the nucleated PP/poly(ethylene-octene) blends. At 0.2 wt% of the nucleating agent, the impact strength and flexural modulus of the blends enhanced to >300 J/m and 1000 MPa when compared with <100 J/m and approximately 850 MPa for non-nucleated blends. The reduction in the elongation in the present case could be a result of much higher content of nucleating agent, which also correspondingly led to much higher increase in the tensile modulus.
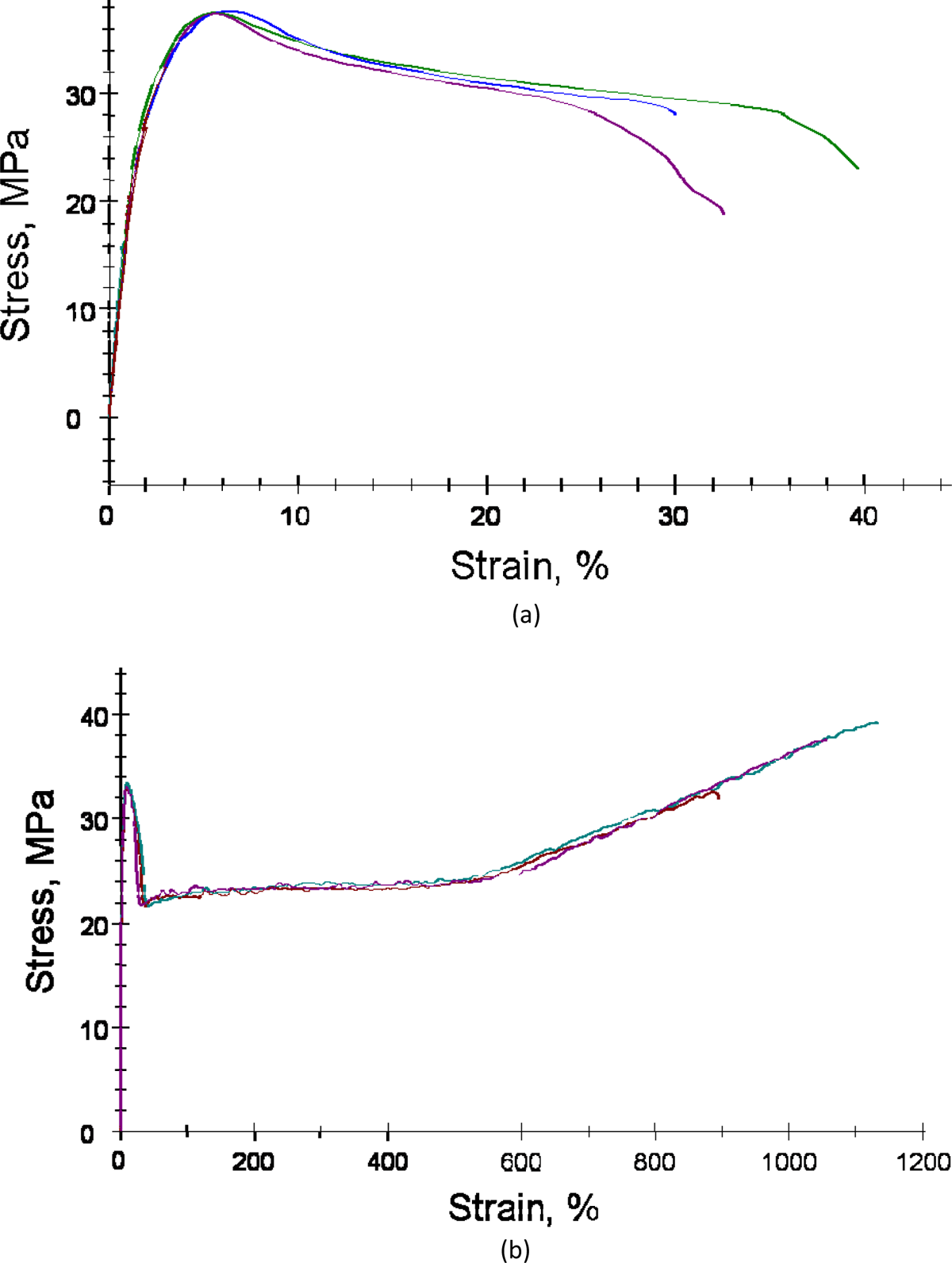
Stress–strain correlations for (a) PP compound with nucleating agent made at 170°C, molded by fast cooling and (b) PP compound with compatibilizer made at 190°C, molded by fast cooling. PP: polypropylene.
The addition of compatibilizer led to a decrease in the tensile modulus of the polymer to 1463 MPa. The yield strain was observed to increase signifying the plasticization of the matrix on the incorporation of low molecular weight compatibilizer (Figure 9(b)). The interactions of the compatibilizer with the polymer chains and the nature (extent of MA grafting, molecular weight, etc.) of the compatibilizer impact the mechanical properties of such hybrids. Therefore, no single trends in the mechanical behaviors of the compatibilized polymers have been reported. Hasegawa et al. 19 reported similar findings as the current study for PP-g-MA compatibilized PP matrix. The tensile strength of the polymer decreased from 780 MPa to 714 and 760 MPa, when the amount of compatibilizer was increased from 7 to 22%. The tensile strength was unimpacted, whereas the strain decreased on increasing compatibilizer content. Reichert et al. 17 also reported an enhancement in the modulus of PP from 1490 MPa for pure polymer to 1680 and 1690 MPa for two different compatibilizers at 20 wt% contents. The tensile strength was not affected, but the strain was significantly reduced (from 321 to 8–9%).
Strain at break followed the similar trend as the yield strain. For PP compounded at 170°C and molded by fast cooling, strain at break values were in the range of 1200%. The values reduced to approximately 700–800% in the case of PP compounded at 190°C and molded by fast cooling. Addition of 1% nucleating agent led to significant reduction in the strain at break to 30–40% due to increased brittleness. On the other hand, PP compound with compatibilizer retained the strain at break at original levels owing to plasticization.
These findings indicate that the addition of small amounts of nucleating agent and compatibilizer to the polymer can significantly impact its application. For example, nucleating agent deteriorated the oxygen permeation resistance and tensile strain, but significantly improved the tensile modulus due to controlled and faster crystal growth. An opposite effect was observed for the compatibilizer. Thus, for the applications of polymer in packaging industry, the use of nucleating agent may not be advisable and compatibilizer can be preferred. 12 On the other hand, applications of the polymer in automotive applications would benefit from the addition of nucleating agent rather than compatibilizer. It should also be noted that when the polymer was added with 1 vol% of the layered silicate filler, 22 the modulus was observed to be 1786 MPa, which was significantly lower than value of 1908 MPa observed by adding only 1 wt% of the nucleating agent. It required roughly 2 vol% filler to reach the similar modulus value indicating that nucleating agent is a good alternative to enhance the mechanical performance of the polymer. On the other hand, the oxygen permeation of the polymer significantly reduced to 77 cc µm/(m2 d mmHg) on the addition of 1 vol% of the filler, 21 whereas the permeation increased to 92.7 cc µm/(m2 d mmHg) when nucleating agent was added to the polymer. Thus, in order to achieve resistance against gas permeation, inorganic fillers and compatibilizers have better performance than the nucleating agent.
Conclusions
In the current study, isotactic PP was subjected to different processing temperatures and cooling rates. It was also compounded with nucleating agent (1 wt%) and low molecular weight compatibilizer (2 wt%). The processing of the polymer at higher temperature affected the oxygen permeation as well as mechanical properties as it was suspected to partially degrade the polymer structure. The slow or fast cooling also changed the crystallization behavior as the onset and peak crystallization temperatures were significantly affected. The slow cooling conditions also led to increase in the oxygen permeation through the polymer owing to brittleness and defects at boundaries of larger sized crystals. Although the addition of compatibilizer did not negatively impact the oxygen permeation, matrix plasticization was observed leading to the decrease in tensile modulus and increase in yield strain. The peak crystallization temperature was also significantly reduced. The addition of nucleating agent showed changes in the crystal structure of the polymer as x-ray diffraction peaks corresponding to certain crystallographic planes were either shifted slightly or were broadened. The oxygen permeation through the nucleated polymer was also worse than the polymer without the nucleating agent owing to similar reasons of brittleness and crystal boundary voids. The yield strain was also decreased significantly owing to brittleness. The tensile modulus of the nucleated polymer increased by roughly 20% when compared with non-nucleated polymer, an increase which otherwise was achieved by adding 2 vol% of the layered silicate filler. The thermal behavior of the polymer with and without compatibilizer or nucleating agent was similar.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
