Abstract
Monodisperse polystyrene (PS) nanoparticles with a size of 190 nm were first synthesized by emulsion polymerization. Then PS/titanium dioxide (TiO2; core/shell) nanocomposite particles were prepared by coating TiO2 nanoparticles on the surface of the PS nanoparticles using aqua ammonia/triethanolamine as the positive/negative catalyst pair. Roughness degree of the surface of the shell of the PS/TiO2 nanocomposite particles increased with increasing amounts of aqua ammonia or triethanolamine. Morphology of the surface of the TiO2 shell can be regulated by changing the amounts of aqua ammonia and triethanolamine synchronously. The PS/TiO2 nanocomposite particles obtained at 2.00 g/0.10 g, 3.00 g/0.20 g and 5.00g/0.40 g of aqua ammonia/triethanolamine showed good morphology of the surface of the TiO2 shell. Roughness on the surface of the TiO2 shell was remarkably heightened when the reaction temperature of the hydrolysis and condensation process was increased from 50°C to 70°C.
Introduction
Inorganic hollow particles in nanometer scale have recently been the subject of extensive research in chemistry and materials science. In this family, the hollow TiO2 spheres have attracted considerable interest owing to their applications in catalysis, photochemical solar cells, controlled release, chemical sensors and so on. 1 –4 The polystyrene/titanium dioxide (PS/TiO2) core shell spheres are prepared in two main approaches: layer-by-layer self-assembly 5 and sol-gel nanocoating. 6 –9 The sol-gel nanocoating is widely used due to its simplicity and high efficiency. But the TiO2 shells obtained in the sol-gel nanocoating are amorphous, and they could transform from amorphism to anatase or rutile after calcining at different temperatures. Anatase or rutiles TiO2 have been extensively investigated because of its notable functions for photocatalysis and photoelectron transfer. 10 –12 However, the widespread technological uses of TiO2 have been impaired by its wide band gap, which require ultraviolet (UV) irradiation for photocatalytic activation. Because the UV light accounts for only a small fraction (5%) of the sun’s energy compared with the visible light (45%), any shift in the optical response of TiO2 from the UV to the visible spectral range would have a profound positive effect on the photocatalytic efficiency of this material. 13 The widely used approaches that shifted the optical response of TiO2 from the UV to the visible spectral range are carried out by dye sensitization, metal ion doping, nonmetal doping, etc. 14 –17
In this study, monodisperse PS nanoparticles were first synthesized, and then TiO2 nanoparticles were coated on the surface of the PS nanoparticles by sol-gel reaction resulting in the formation of TiO2 shell, namely PS/TiO2 (core /shell) nanocomposite particles. Effects of ammonia and triethanolamine content and reaction temperature on the morphology of the surface of the TiO2 shell were investigated.
Experiment
Materials
Styrene (St), polyvinylpyrrolidone (PVP) and triethanolamine were purchased from Shanghai Chemical Reagent Company (China). St was treated with 10 wt % NaOH aqueous solutions to remove the inhibitor and distilled under reduced pressure prior to polymerization. Absolute ethanol, tetrabutyl titanate (TBT) and aqua ammonia (25 wt %) were purchased from Nanjing Chemical Reagent Company (China). Potassium persulfate (KPS) was purchased from Tianjin Chemical Reagent Company (China) and purified by recrystallization from water. Distilled water was applied for all polymerization and treatment processes.
Synthesis of PS nanoparticles
PS nanoparticles were synthesized by emulsion polymerization as follows: 10.0 g of St, 1.5 g of PVP and 90.0 g of H2O were added into the flask equipped. The solution was stirred and deoxygenated by bubbling N2 and heated to 70°C, and 10.0 g of KPS aqueous solution (1 wt%) were added and allowed to reacted for 12 h.
Synthesis of PS/TiO2 nanocomposite particles
In a typical procedure, 3.00 g of aqua ammonia, 5.00 g of PS emulsion and 95.00 g of ethanol were mixed and heated to 50°C, at a stirring rate of 150 r/min, and 10.20 g of TBT/ triethanolamine/ethanol (1.00 g/0.20 g/9.00 g) was added dropwise to the mixture at a rate of 4 ml/h and then allowed to react for 4 h after the dripping.
Figure 1 revealed the scheme of synthesis of PS/TiO2 nanocomposite particles.

Scheme synthesis of polystyrene/titanium dioxide (PS/TiO2) nanocomposite particles.
Characterization
Transmission electron microscopy (TEM) analysis was done using a Hitachi H-7650 microscopy (Japan) operating at 80 kV. The sample was diluted with ethanol and ultrasonicated for 10 min and then dried onto carbon-coated copper grids before testing.
Dynamic light scattering (DLS) analysis: was performed using a Brookhaven BI-200SM photon correlation spectrometer (USA).
Energy-dispersive X-ray (EDX) analysis was done using a JEOL JEM-2100 spectrometer (Japan). The individual PS/TiO2 nanocomposite particle was exposed to electron beam of the X-ray at different positions.
Results and discussion
PS nanoparticle analysis
The TEM image of PS nanoparticles is shown in Figure 2. It could be seen that the size of the PS nanoparticles was symmetrical, and its diameter was 190 nm. Figure 3 showed the size distribution of the PS nanoparticles. The results of DLS proved that the PS nanoparticles were monodisperse with the dispersion coefficient of 0.005.

Transmission electron microscopy (TEM) image of polystyrene (PS) nanoparticles.
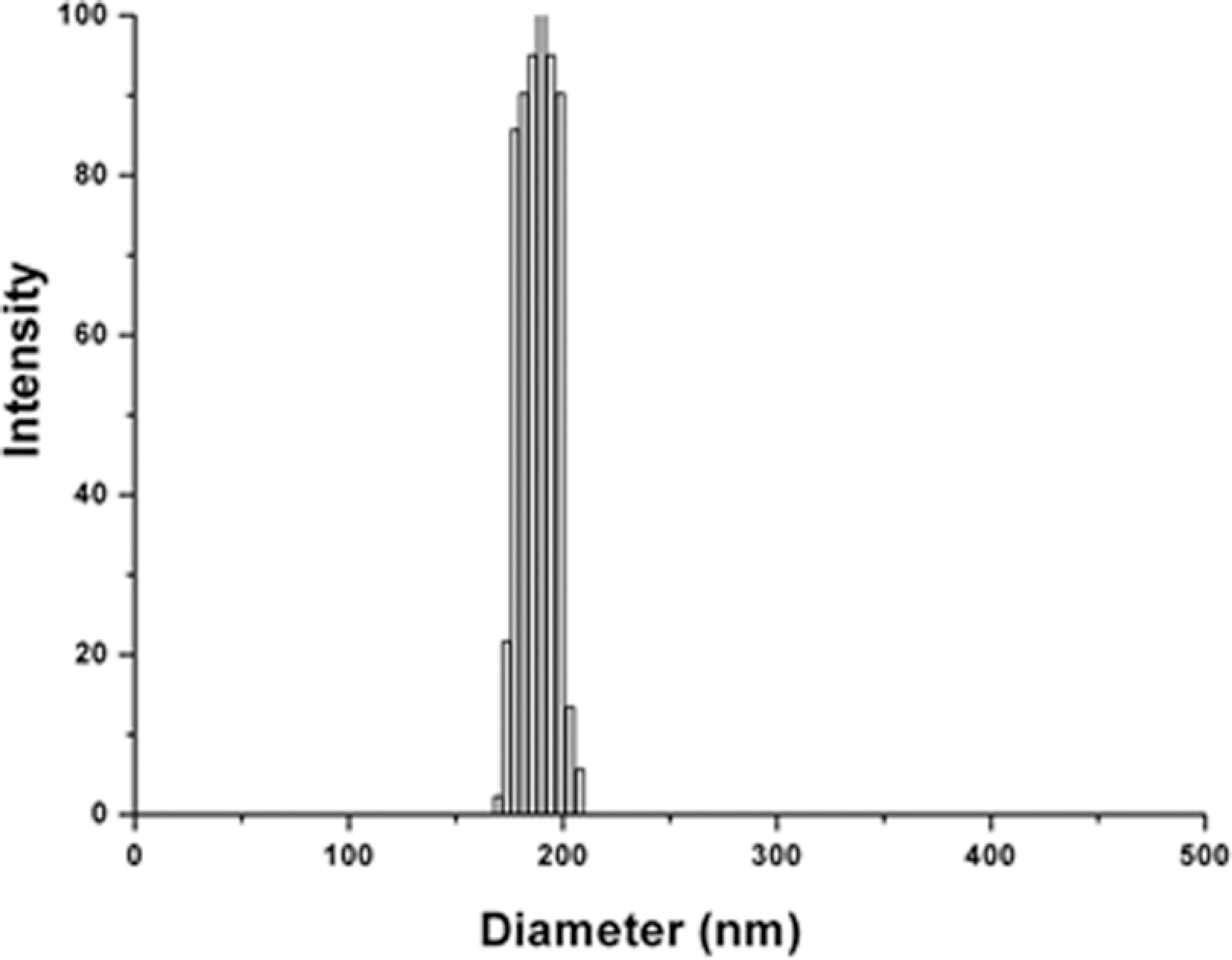
Size distribution of polystyrene (PS) nanoprticles.
Effect of aqua ammonia content
Aqua ammonia acted as a positive catalyst in TiO2-coating process. Positively charged PS spheres were obtained by the absorption of ammonium ion which also played a role in promoting the dissociation of TBT. Figure 4 showed TEM images of PS/TiO2 nanocomposite particles synthesized at different amounts of aqua ammonia. By increasing the content of aqua ammonia, the TiO2-coating rate was enhanced, and the degree of roughness on the surface of the TiO2 shell was large. The chelate complex of triethanolamine-TBT undissociated at 0.75 g of aqua ammonia did not produce TiO2, and did not form TiO2 shell. When the amount of aqua ammonia was 1.50 g, it resulted in TiO2 coating on the surface of the PS nanoparticles. When the amounts of aqua ammonia were increased to 12.0 g, the generating rate of the TiO2 was quick, and some TiO2 particles aggregated and led to the formation of rougher surface than that of the nanocomposite particles synthesized using 1.50 g of aqua ammonia.

Transmission electron microscopy (TEM) images of polystyrene/titanium dioxide (PS/TiO2) nanocomposite particles synthesized at 0.75 g (a), 1.50 g (b) and 12.0 g (c) of aqua ammonia.
Effect of triethanolamine content
Triethanolamine acted as a negative catalyst in TiO2-coating process. Triethanolamine and TBT can form a stable chelate and generate countercharged precursor of TiO2. Effect of triethanolamine on the surface morphology of the TiO2 shell was investigated. TEM images of PS/TiO2 nanocomposite particles obtained at different amounts of triethanolamine are shown in Figure 5. When the amount of triethanolamine was 0.20 g, the TiO2 shell revealed a slippery surface. When the amount of triethanolamine was enhanced (0.40 g), the TiO2 shell displayed a rougher surface, and the degree of roughness augmented with further increase in the amount of triethanolamine. By increasing the triethanolamine content, the dissociation of triethanolamine-TBT chelate became difficult and affected the succeeding nucleation process due to surface inducement and led to rougher surface of the TiO2 shell.
Transmission electron microscopy (TEM) images of polystyrene/titanium dioxide (PS/TiO2) nanocomposite particles synthesized at 0.20 g (a), 0.40 g (b) and 0.80 g (c) of triethanolamine.
Effect of aqua ammonia/triethanolamine content
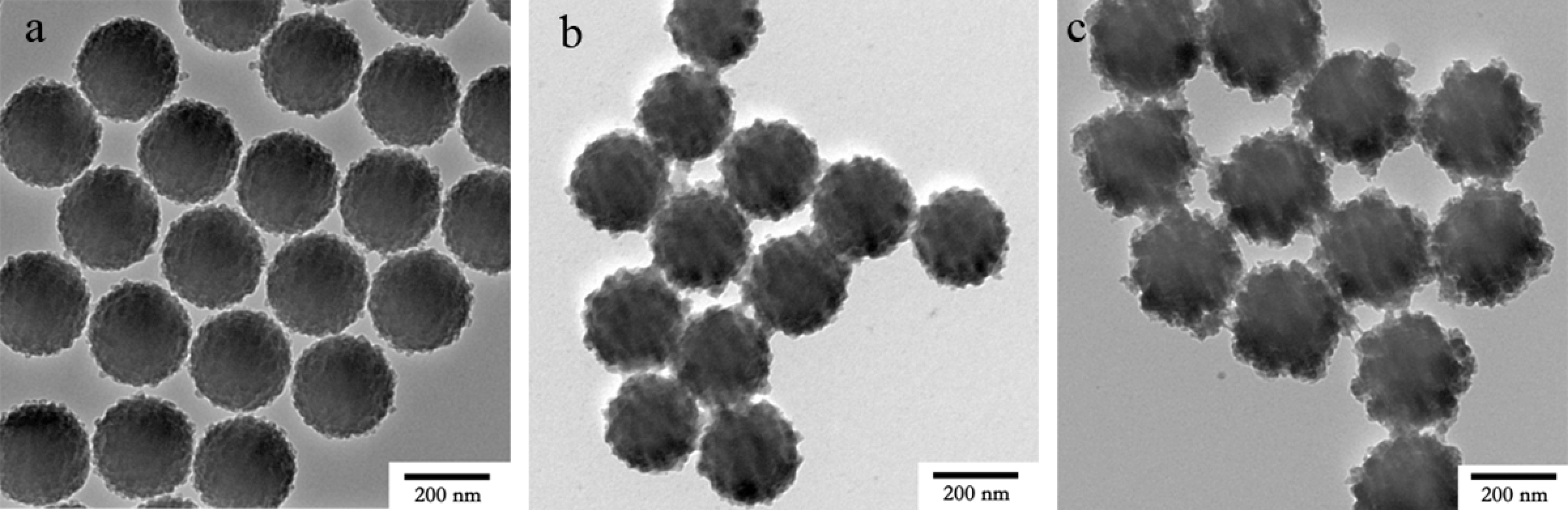
Effect of aqua ammonia and triethanolamine contents on the surface morphology of the TiO2 shell was investigated. Figure 6 showed the TEM images of PS/TiO2 nanocomposite particles synthesized at different amounts of aqua ammonia and triethanolamine. By changing the amounts of aqua ammonia and triethanolamine synchronously, the coating rate of the TiO2 nanoparticles can be regulated, and PS/TiO2 nanocomposite particles with a good surface morphology of TiO2 shell were obtained. The surface of the TiO2 shell was perfect when amount of aqua ammonia/triethanolamine was 2.00 g/0.10 g, 3.00 g/0.20 g and 5.00g/0.40 g.

Transmission electron microscopy (TEM) images of polystyrene/titanium dioxide (PS/TiO2) nanocomposite particles synthesized at 2.00 g/0.10 g (a), 3.00 g/0.20 g (b) and 5.00g/0.40 g (c) of aqua ammonia/triethanolamine.
Effect of reaction temperature
Effect of reaction temperature on the surface morphology of the TiO2 shell was indagated. TEM images of PS/TiO2 nanocomposite particles synthesized at different reaction temperatures are shown in Figure 7. Surface of the TiO2 shell became rougher by increasing the reaction temperature. Hydrolysis and condensation rate of the TBT was moderate at a reaction temperature of 50°C, and the surface morphology of TiO2 shell was thus gratifying. When the reaction temperatures were increased to 60°C and 70°C, the hydrolysis and condensation rate of the TBT quickened, and some TiO2 particles aggregated and subsequently coated, therby causing an increase in surface roughness of TiO2 shell.

Transmission electron microscopy (TEM) images of polystyrene/titanium dioxide (PS/TiO2) nanocomposite particles synthesized at 50°C (a), 60°C (b) and 70°C (c) of the reaction temperature.
Analysis of PS/TiO2 nanocomposite particles
TEM image of PS/TiO2 nanocomposite particles is shown in Figure 8, and its size distribution is shown in Figure 9. Size of the PS/TiO2 nanocomposite particles was 240 nm, indicating that the TiO2 nanoparticles coated on the surface of the PS core and the TiO2 shell thickness being 25 nm. The DLS analysis data showed monodisperse PS/TiO2 nanocomposite particles with a dispersion coefficient of 0.01.

Transmission electron microscopy (TEM) image of polystyrene/titanium dioxide (PS/TiO2) nanocomposite particles.
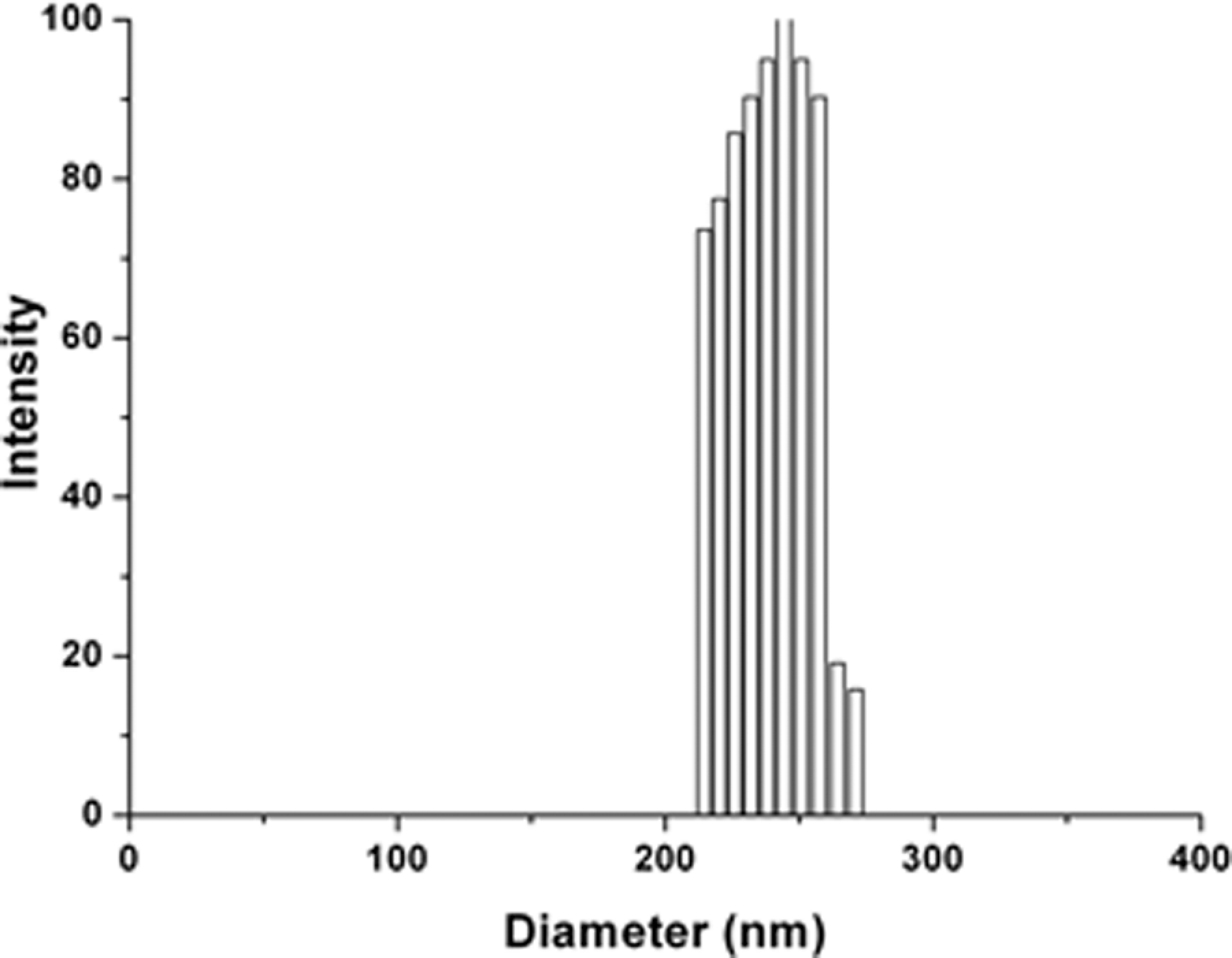
Size distribution of polystyrene/titanium dioxide (PS/TiO2) nanocomposite particles.
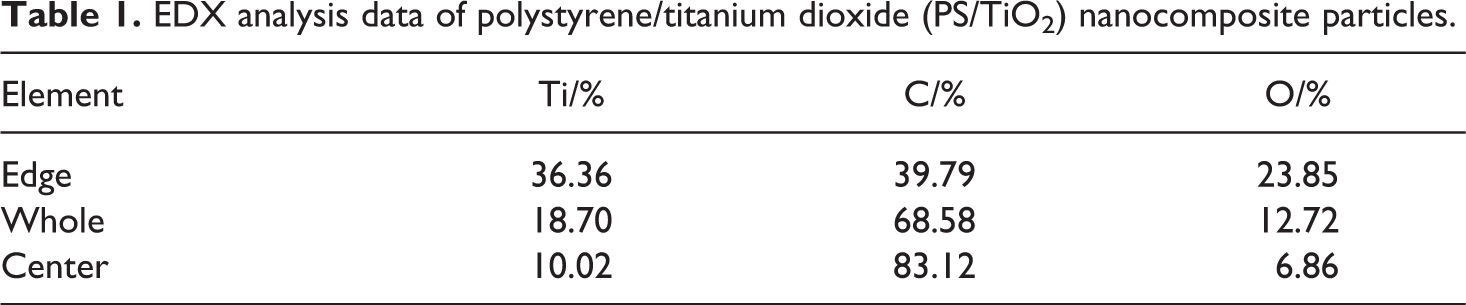
EDX spectra of PS/TiO2 nanocomposite particles are shown in Figure 10, and their analysis data listed in Table 1. Ti content in the edge area of the PS/TiO2 nanocomposite particles was higher than that in the whole and the center area of the nanocomposite particles, respectively, and the synchronous C content in the center area of the nanocomposite particles was higher than that in the whole and the edge area. This suggested that the PS/TiO2 nanocomposite particles were core/shell formation.
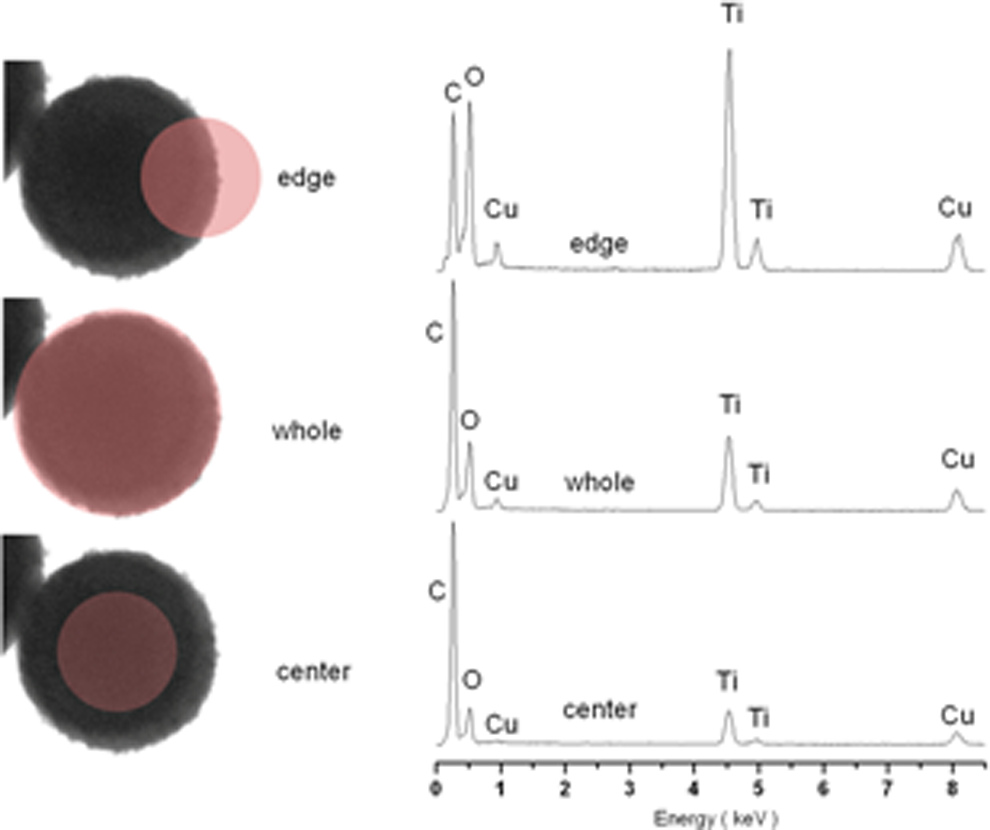
EDX spectra of polystyrene/titanium dioxide (PS/TiO2) nanocomposite particles (left transmission electron microscopy (TEM) images illustrated position of the electron beam).
EDX analysis data of polystyrene/titanium dioxide (PS/TiO2) nanocomposite particles.
Conclusions
Monodisperse PS nanoparticles were first synthesized, and then the PS/TiO2 nanocomposite particles were prepared by coating TiO2 on the surface of PS nanoparticles. Surface roughness of the TiO2 shell, respectively, was enhanced by increasing the content of aqua ammonia and triethanolamine. Surface morphology of the TiO2 shell can be regulated by changing the contents of aqua ammonia and triethanolamine synchronously. Increase in the reaction temperature during the TiO2coating process led to poor surface morphology of the TiO2 shell.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
