Abstract
In this study, the filler–matrix interactions are assessed in two nanocomposites having different antibacterial activity. The two polymers used as matrix are poly(amide) 6 (PA6) and low-density poly(ethylene) (LDPE). The filler, zinc oxide (ZnO) nanoparticles, with a content as low as 1 w/w% in the polymers showed great antibacterial activity against Escherichia coli and Staphylococcus aureus. However, the bacterial slaying capability of composites was found better when ZnO was dispersed in PA6, where the efficiency is similar to pure ZnO particles. The dispersion of ZnO and its interactions with the matrix have been investigated by means of scanning electron microscope, rheology, thermogravimetric analysis and differential scanning calorimeter. This study shows stronger interactions of ZnO particles with LDPE, which could have an effect on final antibacterial properties.
Introduction
The interest in polymer composites exhibiting antimicrobial activity is continuously increasing due to the growing demand for healthy living. When antimicrobial agents are incorporated into a polymer, the material limits or prevents microbial growth. Potential fields of application include, for example, textile, 1,2 packaging 3 or medical devices to prevent nosocomial infections. 4 Antimicrobial packaging materials have to extend the lag period and reduce the growth rate of microorganisms to extend shelf life of packaged food, not only in the form of films but also as containers or utensils.
Antimicrobial activity of a polymer is commonly achieved by adding metal particles, 5 –7 and also organic compounds. 8,9 Zinc oxide (ZnO) is well known as antibacterial agent both against gram-positive and gram-negative bacteria. 10 –14 However, very few studies report success to preserve antibacterial activity when blended with polymer. 14,15
The antibacterial mechanism seems complex, 16 and the exact mechanism of cell membrane damage involved with ZnO is not fully put into evidence. 13 The hydrogen peroxide (H2O2) is identified as one of the main factor concerned in antimicrobial activity. 10,17 Indeed, as a powerful oxidizing agent, it is well known that H2O2 is harmful to the cells of living organisms. Another possible mechanism is the release of zinc (Zn2+) ions. 13
In any case, direct contact with ZnO surface is not recognized as a possible mechanism that is coherent since particles functionalized do not affect the antibacterial properties. 13
The concentration, size, morphology and the surface area of ZnO are the factors that may contribute to H2O2 or Zn2+ ions release. Therefore, when blended in a polymer phase, amount of filler, particle dispersion, filler–matrix interactions and processing conditions influence the antibacterial activity of ZnO.
This study is performed with the same samples made in a previous work. 14 Here, the aim is to complete the analysis through the investigation of the filler–matrix interactions in order to establish a relationship with antibacterial efficiency that was described in our previous work, 14 where the poly(amide) 6 (PA6)/ZnO nanocomposites were in any case more effective than the low-density poly(ethylene) (LDPE)/ZnO ones. Moreover, the amount of filler had an impact onto the antibacterial activity, which increases when the filler content increases. To quantify the different factors that could influence the ZnO antibacterial mechanism, when dispersed in thermoplastic polymer, our approach is to relate the efficiency of direct contact of filler with bacterial medium, an aqueous solution, that leads to H2O2 or Zn2+ ions release. Here, the filler–matrix interactions are investigated through the studies of crystal phase changes, thermal stability and rheological behaviour when dispersed in a PA6 or a LDPE.
Materials and techniques
Materials
The two matrices were chosen both for their different polarity and their common use in packaging. 3 LDPE (Lupolen 1840 H from Basell, The Netherlands) exhibits a hydrophobic behaviour. While PA6 (Dinalon PA6 from Repol, Spain) is more hydrophilic. The main properties of polymers are presented in the Table 1.
Polymers properties.
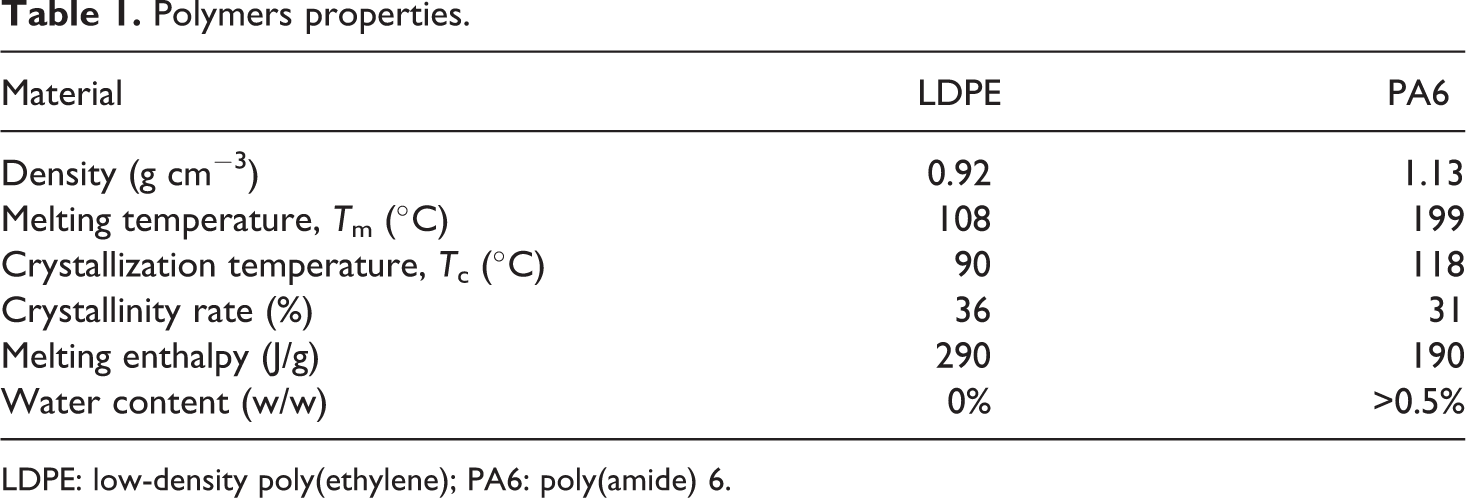
LDPE: low-density poly(ethylene); PA6: poly(amide) 6.
Synthesis
The ZnO nanoparticles are obtained at 600°C in a pilot scale platform by spray pyrolysis. 18 During the process, droplets of a precursor solution are dried and decomposed to the required compound. The presence of an additional soluble flux in the precursor solution permits to obtain agglomerate–free nanoparticles after washing of the product. Therefore, pure ZnO nanoparticles were synthesized by adding sodium nitrates to the initial zinc nitrate solution. 18
Sample elaboration was made by blending polymer matrix (5 cm3) with ZnO in a mini extruder (Minilab from Thermo Haake, Germany). Compounds were blended for 15 min at 200°C for LDPE and 250°C for PA6 in two conical contra-rotative screws with a mixing speed of 150 r min−1. Different composite formulations were obtained with filler content ranging from 0 to 5 w/w% for antimicrobial testing.
Characterization
The morphologies of powder and composites were observed by a field-emission scanning electron microscope (SEM; Jeol JSM 6700F, Japan). Prior observation, the polymer composites were fractured in liquid nitrogen.
The final powders are characterized by their x-ray diffractions (XRDs) recorded on a (Seifert C3000, Germany) diffractometer. The crystallite size of ZnO particles is calculated from the half-width of diffraction peaks using Scherrer’s equation.
An Ultrasonic probe (Fisher Bioblock Scientific SAS, Vibracell-75043, France) is used during 1 h at 40% of amplitude to prevent ZnO aggregates in order to compare the size distribution with the composite one.
The particle size distribution of ZnO powder was measured by laser diffraction (Malvern Mastersizer S, Malvern Instruments Ltd, UK). A concentration of approximately 30 mg L−1 of powder was poured in distilled water. The accuracy is ±1% on the D(v, 50), being the equivalent volume diameters at 50% cumulative volume.
The surface area was estimated by the Brunauer–Emmett–Teller (BET) method using a Belsorp mini 22473-9 from BEL (Japan). The data were extracted from the adsorption curves in the range 0 to 0.35 p/p0. The filler content was measured by carbonization of few grams of sample in a furnace at 600°C during 5 h.
The thermal stability was measured in air by thermogravimetric analysis (TGA) using a (Mettler Toledo TGA 50, Switzerland). Dynamic measurements were performed from 25 to 600°C at a heating rate of 20°C min−1. Sample weight was approximately 20 mg. The measurement precision was estimated to be less than 0.5%.
The rheological properties of the LDPE composites were measured at 200 and 250°C for PA6, corresponding to the processing temperature. The measuring system was a Rheotron Brabender viscometer with cone–plate geometry of 20 mm in diameter. To characterize the rheological behaviour of our systems, we prefer to work in oscillating shear to prevent sample fracture. The study in transient state of dynamic viscosity (η*), storage modulus (G′) and loss modulus (G′′) was performed in a pulse range between 0.1 and 100 r m−1. All the measurements were carried out in the linear viscoelastic range determined for each matrix by an amplitude sweep with a frequency of 1 Hz between 0.01 and 1 rad.
Our approach was to describe the flow curves of composites by power law equations, Cross and Carreau-Yasuda models
19,20
and use a superposition principle to account for the system characteristics. The power law is given by
where n is the flow behaviour index and K being the flow consistency index.
For a large range of shear rate, the viscosity follows two Newtonian limits (at low and high shear rates). These different behaviours can be taken into consideration by the following Cross model
where η
0 and η
∞ represent the Newtonian limit at zero and infinite shear rates, respectively. The parameter,
The Carreau-Yasuda model allows a fitting of the transition at the critical shear rate by the use of the parameter a
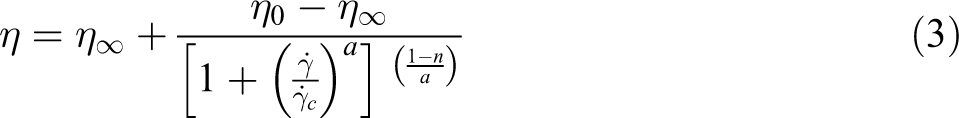
This model is the most flexible since it is corresponding to Cross model when a = 1 and to the former Carreau model for a = 2.
Calorimetric measurements were performed with a Mettler Toledo differential scanning calorimeter (DSC) 30. The calibration was done with indium and zinc. Aluminium pans with holes were used and the sample mass was found to be approximately 10 mg. All the samples were first heated up to 250°C during 5 min to get rid of thermal history. Nonisothermal crystallization and melting temperatures were T c,n and T m, respectively, and were determined from the peak extrema in experiments at ±10°C min−1 heating and cooling rates. To prevent any sample degradation, the experiments were carried out under nitrogen flow. All the temperatures measured from a peak extrema (T c and T m) were determined at less than ±0.5°C.
The sample preparation and the methodology for antimicrobial testing were fully described by Droval et al. 14 Briefly, the applied dynamic method was based on European Pharmacopeia (2001) and ASTME 2149-01 to determine the antimicrobial activity of immobilized antimicrobial agents under dynamic contact conditions.
The microorganisms used to test antimicrobial efficiency were Staphylococcus aureus ATCC 6538, Escherichia coli ATCC 8739 and Aspergillus niger ATCC 16404. All the organisms were maintained according to good microbiological practice and checked for purity, by making streak plates and observing for a single species characteristic type of colonies.
Results and discussion
The filler content in the formulations tested was very low. Moreover, occasional extra powder loss in the vessel and the hopper can lead to a lower experimental value. To check these low percentages, the actual filler content in weight was measured by carbonization technique. The results are given in Table 2 in weight and volume. The calculation in volume gives us a direct comparison between the two composites. As we can see, the filler amounts involved were very low considering they do not exceed 1 v/v%.
Carbonization measurement of ZnO content in LDPE and PA6 composites.
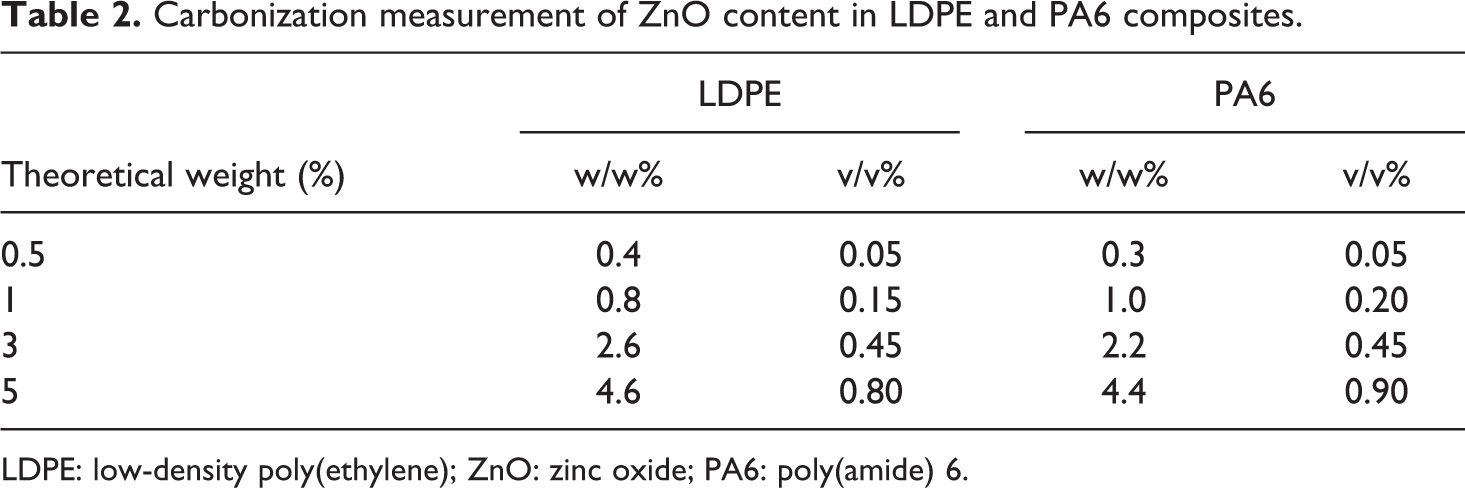
LDPE: low-density poly(ethylene); ZnO: zinc oxide; PA6: poly(amide) 6.
For convenience, the labelling of composites will remain in weight in this article. For Morphological, rheological and thermal measurements, only the composites with 0.5 and 1 w/w% were tested.
Properties of ZnO
A good knowledge of our synthesized ZnO is essential for direct comparison with literature. The zincite phase has been identified by XRD. 14 The average crystallite size from the diffraction peaks is about 35 nm. The size of the lattice of hexagonal structure given by JCPDS-ICDD 2003 database is a 0 of 3.25 Å and c 0 of 5.205 Å. The variation of some hundredth of Å along c 0 axis may have an effect on antibacterial activity by H2O2 generation as shown by Yamamoto et al. 21 The specific surface area may also be an influent factor in antibacterial activity by increasing the surface contact area between particles and the aqueous bacterial media. Calculated by BET method, it is estimated at 15 m2 g−1, that is to say three times larger than the particles used in the study by Yamamoto et al. 21 However, this value is coherent with Cho et al, 22 where the specific surface area is about 20 m2 g−1. The synthesized ZnO is a good antibacterial since in our previous work, 14 1 w/w% of ZnO showed a reduction in the death rate after 48 h of 99.99% of Bacteria (Table 3).
Results of antibacterial activity in CFU ml− 1 after an incubation time of 48 h.
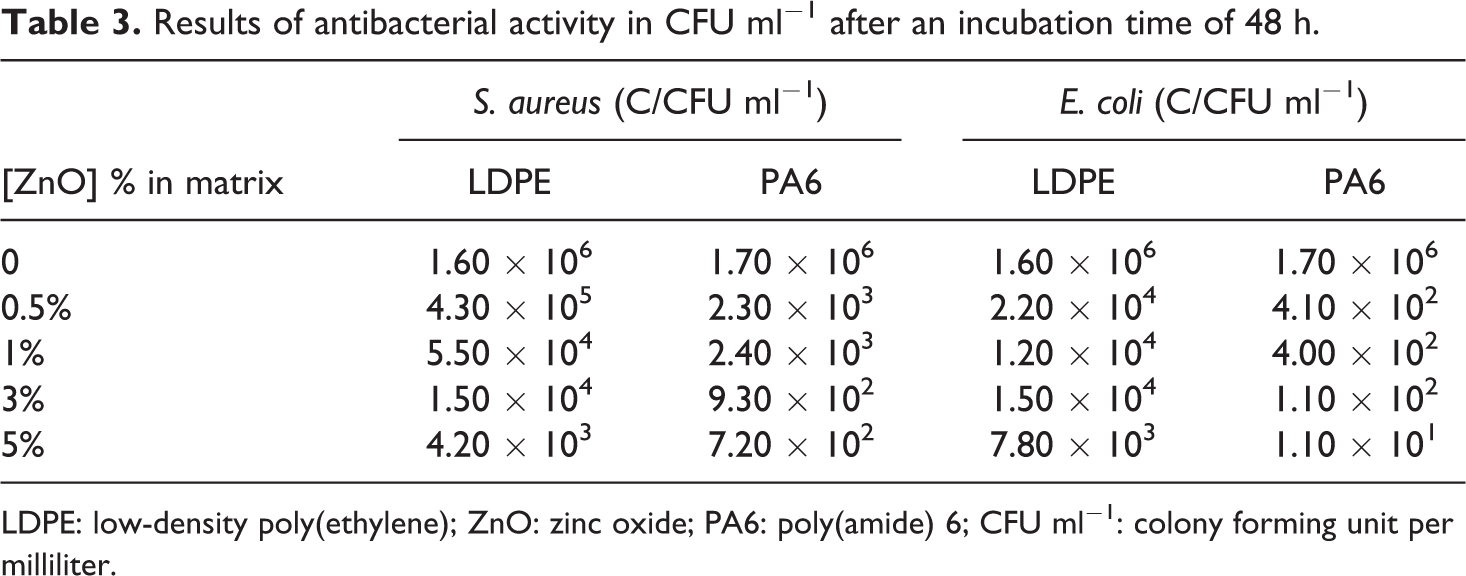
LDPE: low-density poly(ethylene); ZnO: zinc oxide; PA6: poly(amide) 6; CFU ml−1: colony forming unit per milliliter.
In order to evaluate the level of dispersion of filler in the matrix, the size distribution of raw particles is studied. As we can see in Figure 1, the particle size is relatively large with a trimodal distribution. It is well known that the nanoparticles tend to aggregate themselves. It may correspond to individual particles, aggregates or even aggregates of aggregates hence an apparent micrometric size. The mean size is about 5 µm with a standard deviation of 2 µm.
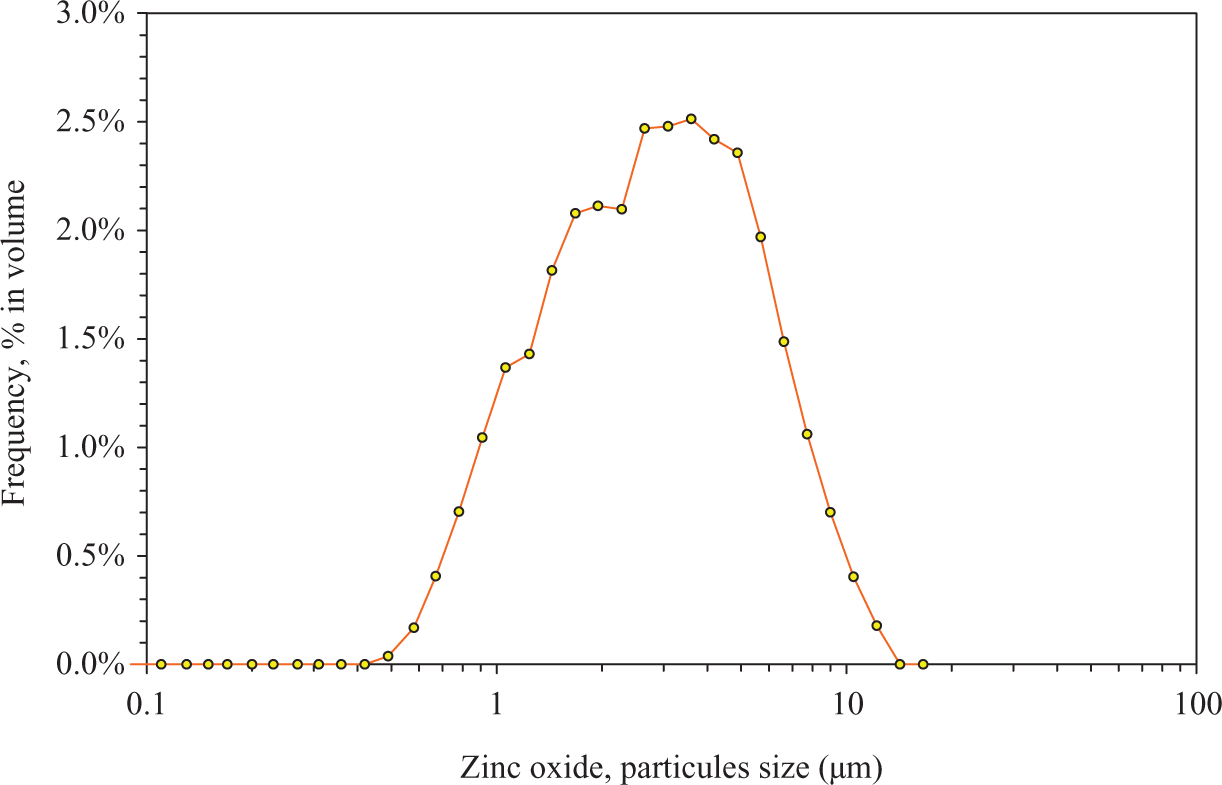
Size distribution of zinc oxide particles.
Particles synthesized by spray-pyrolysis were in a nonequilibrium state and thus their different physical and thermal history can lead to morphologies from compact to burst spheres (platelets). Figure 2 shows the types of aggregates morphologies encountered: relatively spherical aggregates of spheres and/or platelets between 6 and 4 µm. The single spheres encountered are between 1.4 µm and 750 nm and platelets about 300 nm long until 40 nm thick.

Scanning electron microscope observations of raw zinc oxide particles.
Morphology of composites
Figure 3 presents a picture of the formulation PA 0.5% ZnO. All particles of about 400 nm are isolated. Moreover, the interaction between filler and matrix appears very weak as particles are not well surrounded by the matrix, which is not the case with LDPE composites, where Figure 4 (LDPE 0.5% ZnO) shows a particle size between 300 nm and 1 µm with aggregates about 2 µm and a better covering of the LDPE phase around fillers.

Scanning electron microscope observations of 0.5 w/w% zinc oxide particles dispersed in PA6.

SEM observations of 0.5 w/w% zinc oxide particles dispersed in LDPE. SEM: Scanning electron microscope; LDPE: low-density poly(ethylene).
Compared with raw particles (Figure 2), the particle size is smaller in composites. When dispersed in polymer, ZnO aggregates are broken due to high shear rate of about 100 s−1, involved during extrusion process. An equivalent effect is obtained directly with raw particles by the use of ultrasonic bath, which provides additional mechanical energy, prior to laser diffraction measurement. As seen in the figure, after 1 h in ultrasound, the particle size distribution leads to a normal distribution, where the mean size is reduced to 1 µm and the standard deviation to 0.5 µm. This size distribution obtained after ultrasonic bath is coherent with the particles size observed in the composites (Figure 5). In composites, the size distribution is obtained by particle size sampling in SEM pictures. The mean size is drastically reduced in PA 6, equivalent to 390 nm with a standard deviation of 90 nm, where in LDPE, the mean size is 770 nm with a standard deviation of 120 nm. This shows that the use of ultrasonic bath prior blending is not necessary. The difference of particle size between LDPE and PA 6 can be explained be processing condition, mainly temperature that can weaken the particle joint (200°C in LDPE against 250°C with PA 6). This change in particle size also influences the antibacterial activity by increasing the surface area in contact with bacteria medium.
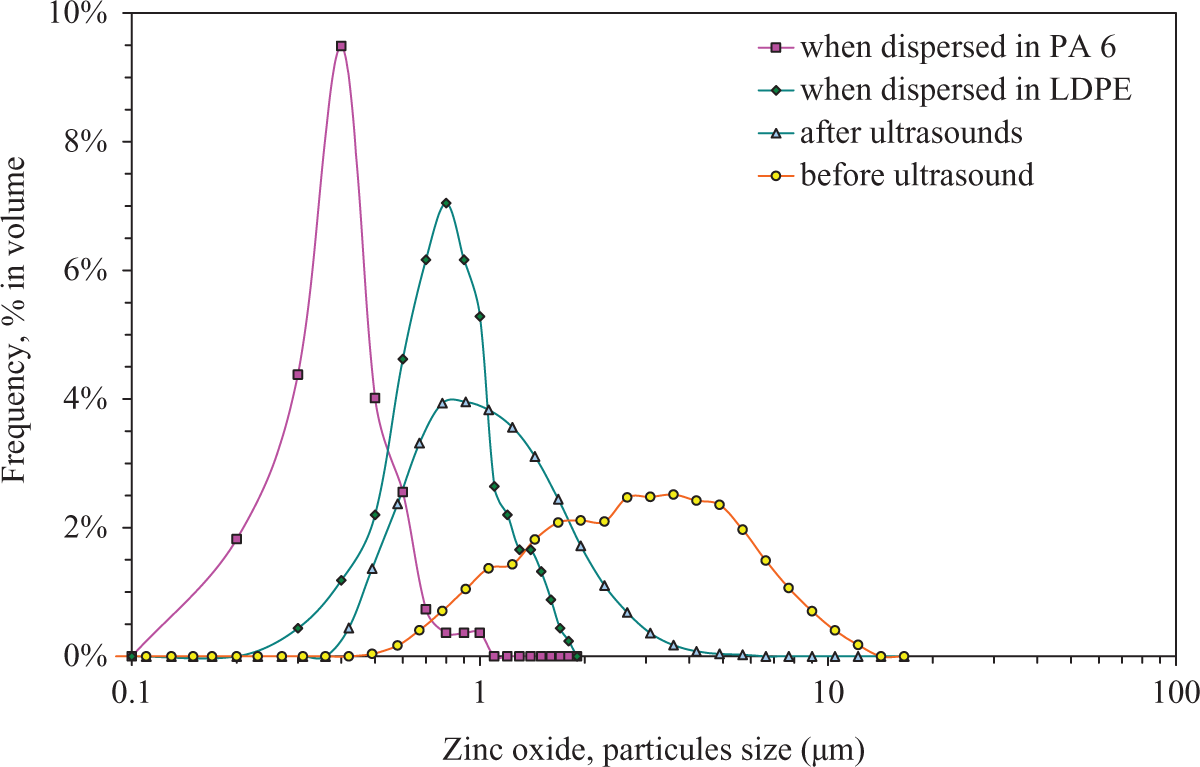
Size distribution of zinc oxide particles depending on dispersing medium.
In the following part, the interactions of the polymer chains with the ZnO particles are investigated by rheological measurements to assess different behaviours of the matrix with ZnO.
Rheology
Due to the very low filler content studied, we considered our systems as homogeneous. The viscosity in steady shear η (γ) is thus supposed equivalent to the complex viscosity in dynamic shear η*(ω) referring to the experimental Cox-Merz law [η (γ) = η*(ω)]. 23
In Figure 6, only the pure LDPE exhibits Newtonian flow behaviour at low frequency. When the frequency is increasing, beyond the critical shear rate, the polymer shows a shear thinning behaviour as well as the composites.
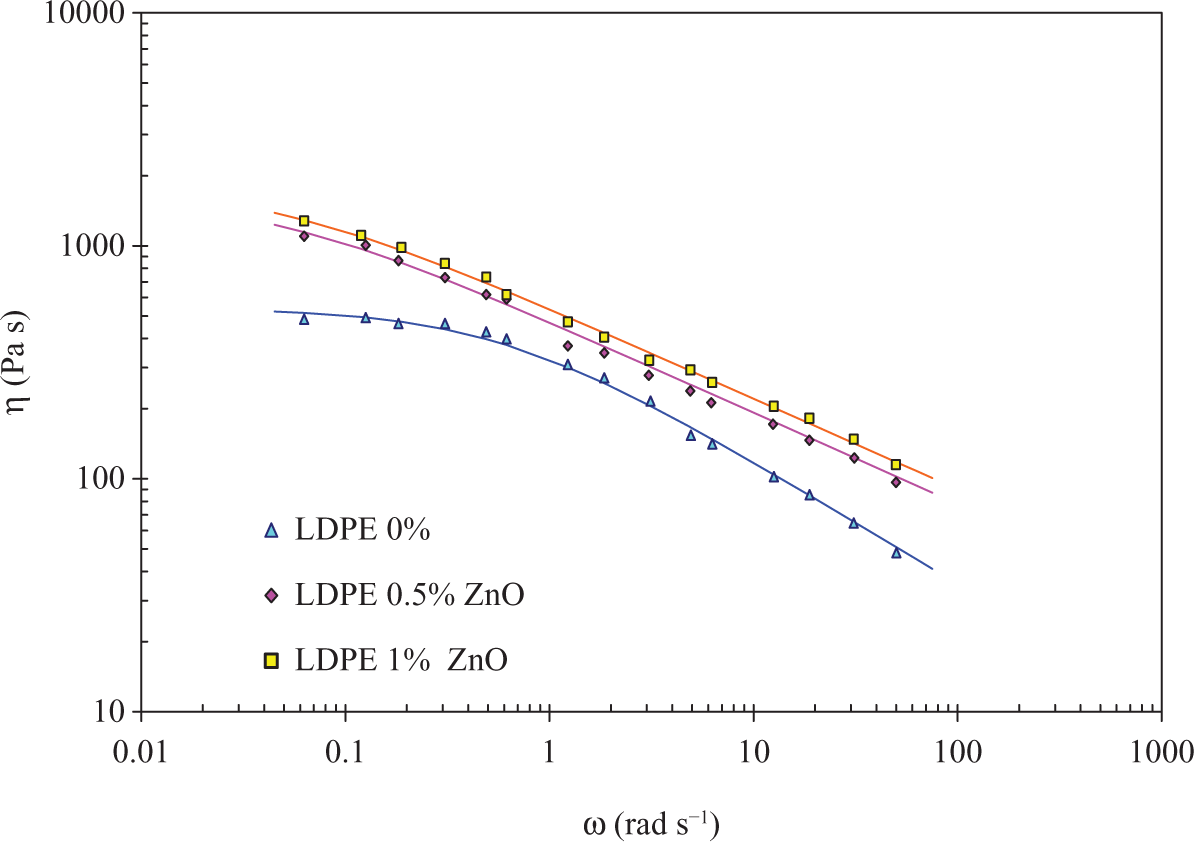
Dynamic viscosity of LDPE/ZnO as a function of frequency at 200°C. LDPE: low-density poly(ethylene); ZnO: zinc oxide.
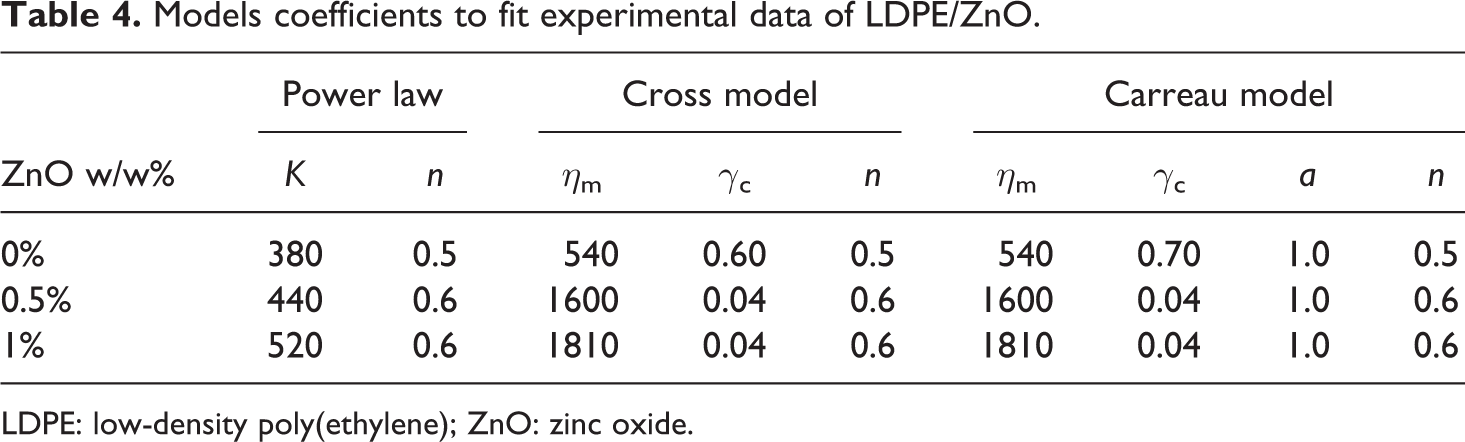
To accurately determine the influence of ZnO on the LDPE phase, the experimental data are fitted with models (plain line in Figure 6, corresponding to the Carreau model). The coefficient for the three models: power law (equation (1)), Cross model (equation (2)) and Carreau model (equation (3)), are given in Table 4, where the infinite viscosity η ∞ is set to zero.
Models coefficients to fit experimental data of LDPE/ZnO.
LDPE: low-density poly(ethylene); ZnO: zinc oxide.
For polymer (shear thinning flow), the pseudoplastic index n is included between 0 (very shear shinning flow behaviour) and 1 (Newtonian flow). We can see in this table that the power law index n slightly increases from 0.5 for pristine LDPE to 0.6 for both LDPE filled with 0.5 and 1% of ZnO. At this low percentage, particles do not have significant effect on the shear shinning flow. On the other hand, the fluid consistency is continuously increasing with filler content. This is also observed with Cross model, where the Newtonian viscosity η 0 is increasing from 540 to 1810 with the filler content. In this model, the power law index n is kept the same as the power law model one. The rise of η 0 while the critical shear rate decreases show that a very low amount of ZnO is enough to vanish the Newtonian plateau as already noticed with another system. 24 However, here the filler content is too low to directly influence the viscosity through a cooperative motion in a particle network. This increase in viscosity can be explained by an increase in the reticulation level in LDPE macromolecules due to filler presence.
The rheological behaviour of PA6 is presented in Figure 7. All formulations tested exhibit Newtonian flow behaviour at a low frequency. The level of viscosity is roughly similar as seen in Table 5, where the model coefficients to fit the experimental data are given.
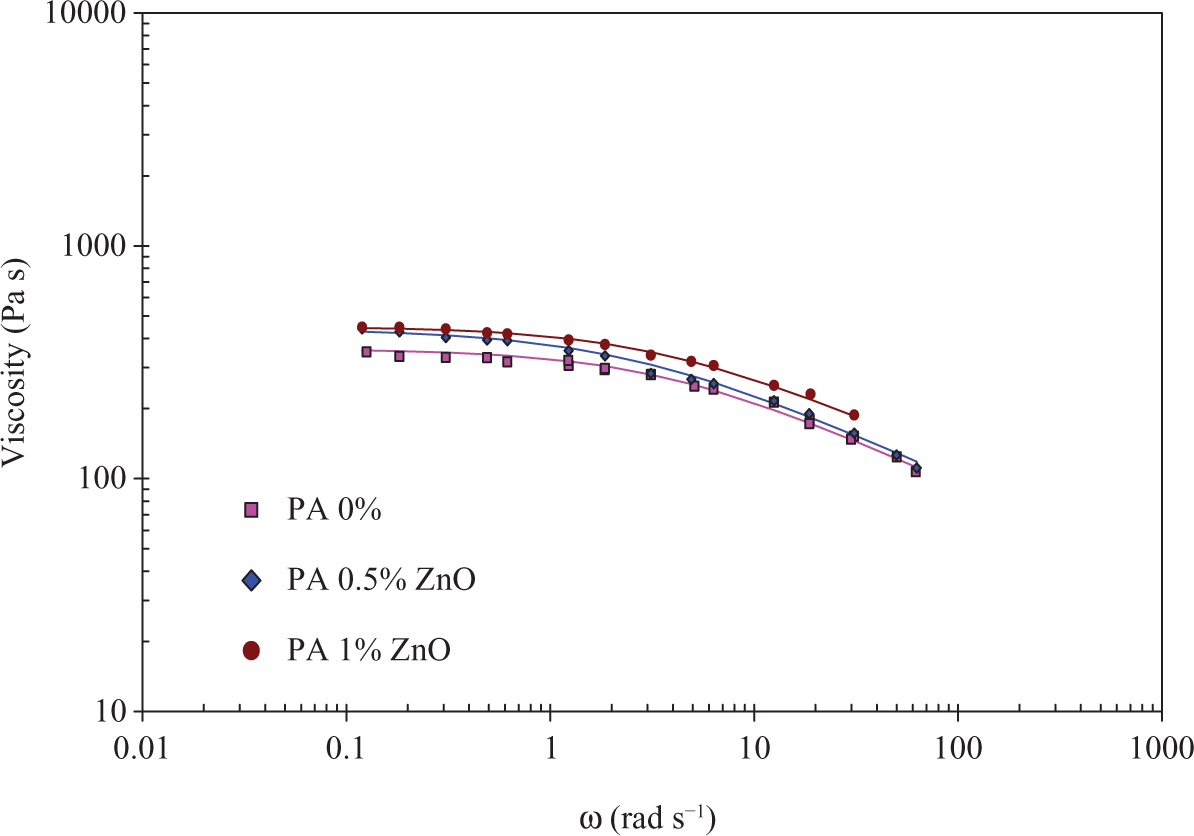
Dynamic viscosity of PA6/ZnO as a function of frequency at 250°C. ZnO: zinc oxide; PA6: poly(amide) 6.
Models coefficients to fit experimental data of PA6/ZnO.
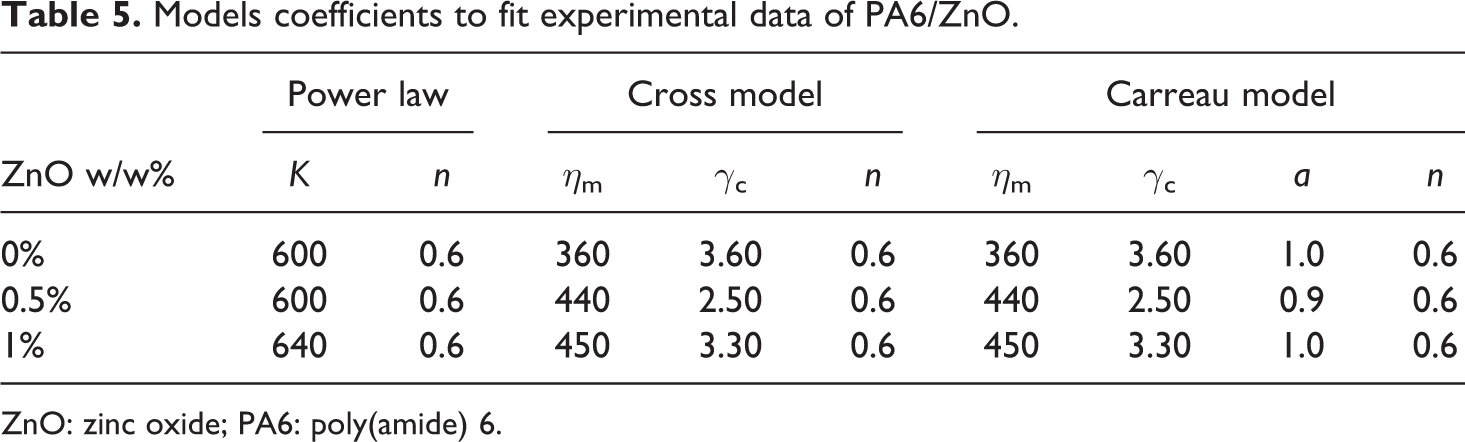
ZnO: zinc oxide; PA6: poly(amide) 6.
The power law index n remains about 0.6 and the Newtonian viscosity slightly increases from 360 for pristine PA6 to 450 when filled with 1% of ZnO. At these percentages, particles have no significant effect on the matrix flow.
The ZnO particles have thus a different impact on the flow behaviour depending on the matrix. The LDPE is reinforced and no apparent effect is put into evidence with the PA6 phase.
Thermal properties
The thermal stability measurements were carried out in the air. The ZnO induces a thermal stability improvement in LDPE as shown in Figure 8(a), where a less mobility of macromolecules delay the beginning of degradation of about 25°C. This could be explained by the nanoparticle size. 25 This can also be attributed to higher branch content. 26
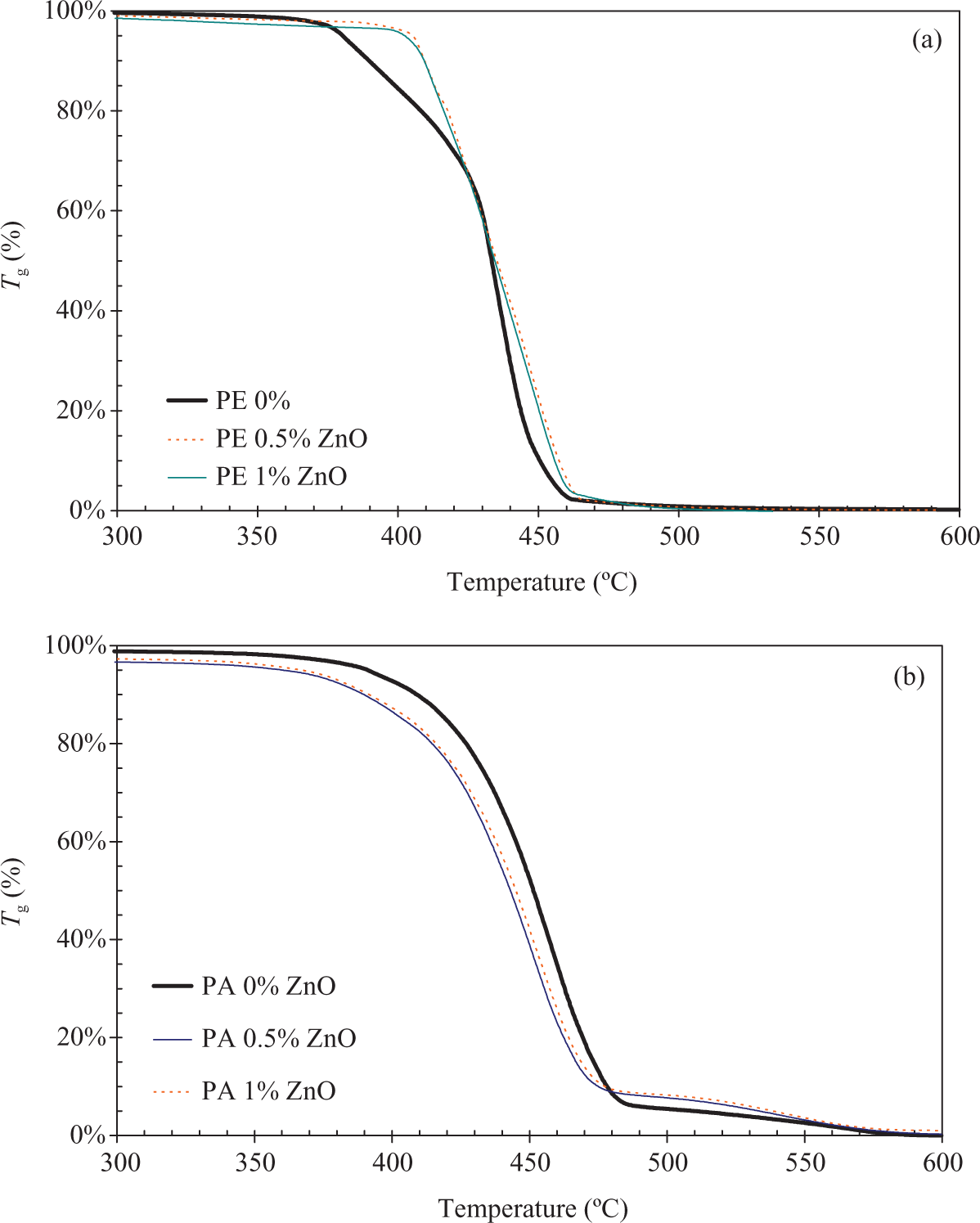
Thermal degradation in air of composites filled with ZnO at 20°C min−1: (a) LDPE and (b) PA6. LDPE: low-density poly(ethylene); ZnO: zinc oxide; PA6: poly(amide) 6.
In the case of PA6, shown in Figure 8(b), the change in thermal stability due to the presence of ZnO particles is low (about 5°C). Temperature phase changes are sensitive to increase in macromolecules mobility induces by ZnO particles in PA6. The slight speed up of degradation can also be explained by shear rate during processing, which can reduce the molecular weight in accordance to ZnO presence. This chain scission has been shown by viscosity measurement in a study of polyamide blends. 27
Following Ribes-Greus et al, 26 the effect of filler on the thermal stability can also have an equivalent effect on the cristallinity rate. The crystallization temperature will depend on the nuclei speed that can be improved by nucleation effect of the filler (Figure 9). 28,29 The melting temperature is generally related to the crystallite thickness 29,30 and the function of the macromolecules mobility and their thermal stability will be less. 26
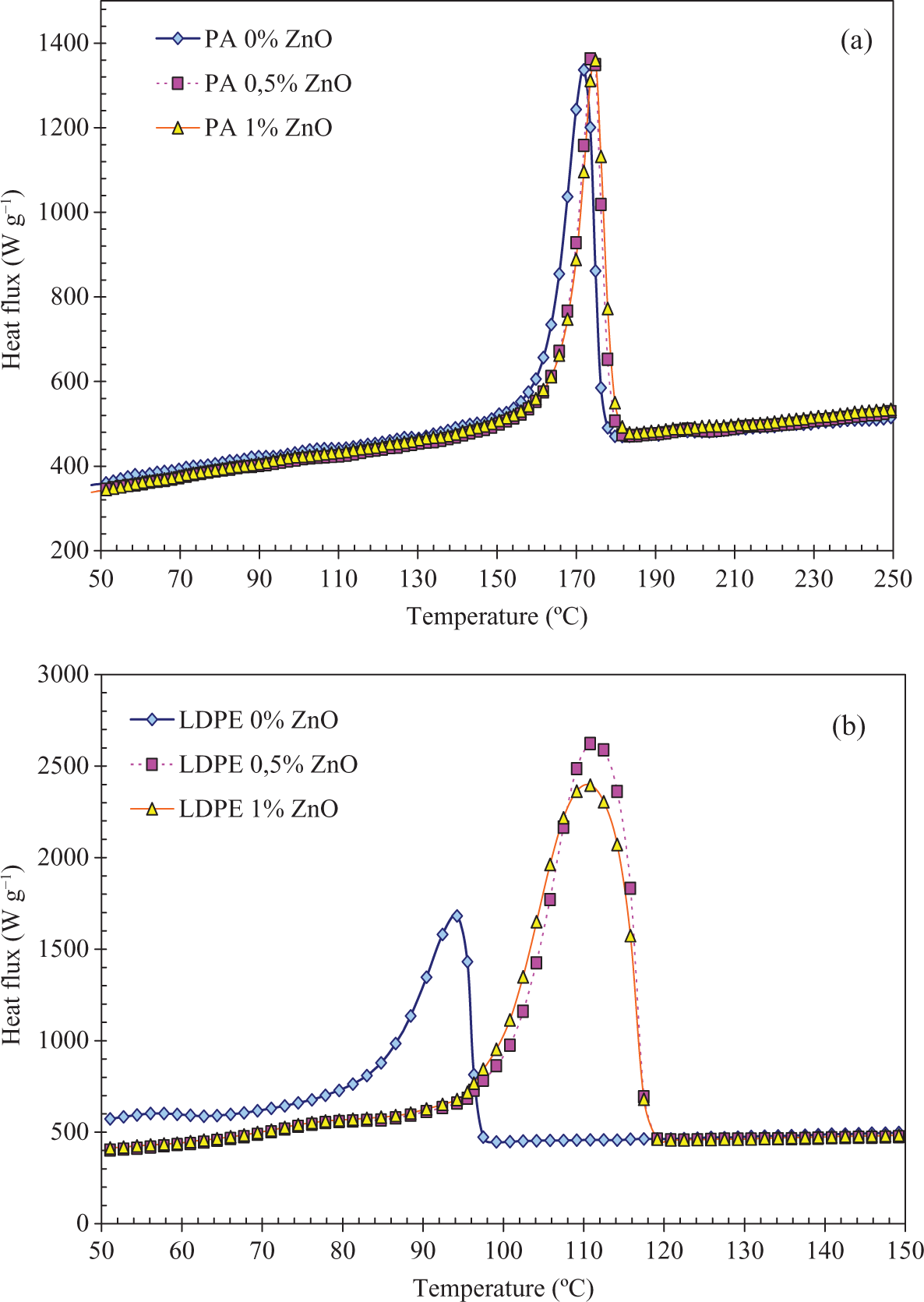
Crystallization of composites filled with ZnO at 10°C min−1: (a) PA6 and (b) LDPE. LDPE: low-density poly(ethylene); ZnO: zinc oxide; PA6: poly(amide) 6.
The nucleation is of great importance in determining crystalline morphology and interface filler–matrix. Indeed, nucleating agent promotes the growth of smaller and more numerous spherulites by introducing a heterogeneous surface to the supercooled polymer melt, making crystallization thermodynamically more favourable. 24,31,32
The phase changes temperatures for each composite tested and the crystallinity rates are reported in Table 6. As we can see, the nucleating effect of ZnO in PA6 is very low (about 3°C). This value is compared with the one of PA6 processed in the same conditions (171.9°C; Table 6).
Crystallization and melting temperatures and crystallinity rate of PA6 and LDPE composites.
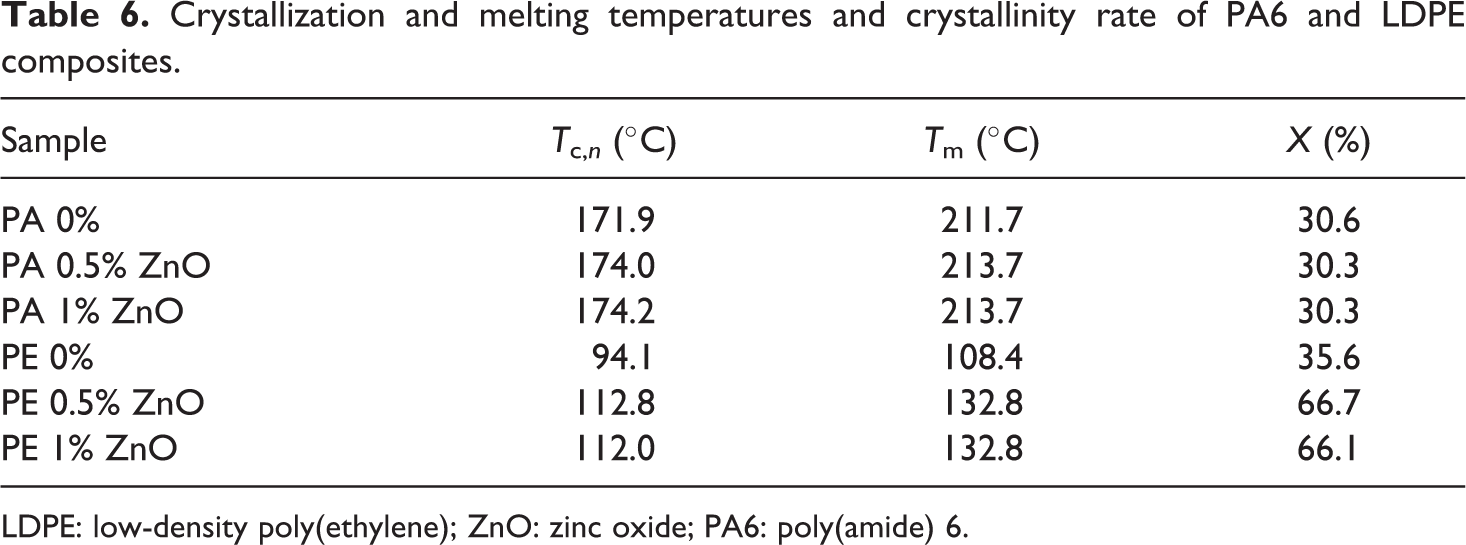
LDPE: low-density poly(ethylene); ZnO: zinc oxide; PA6: poly(amide) 6.
In LDPE, the increase in the crystallization temperature compared with the pristine LDPE processed in the same conditions is about 16.5°C (T c,n of LDPE being 94.3°C).
The melting temperature T m of PA is not or less affected by the filler. The crystalline structure or the crystals thickness is thus unchanged. On the other hand, the melting temperature of LDPE is shifted to about 25°C when filled with a few amount of ZnO. This shows thicker lamellae in LDPE. Moreover, the crystallinity rate also reported in Table 6 shows a high effect of ZnO particles onto the crystal content in LDPE compared with PA6, which remains unchanged.
Conclusions
We intend in this article to quantify the different factors that could influence the ZnO antibacterial mechanism when dispersed in thermoplastic polymer. However, the complete antibacterial mechanism of ZnO itself is not clearly identified. We supposed in this approach that the efficiency was related to direct contact of filler with bacterial medium, an aqueous solution, that lead to H2O2 or Zn2+ ions release.
The relationship between the antimicrobial activity and the filler–matrix interactions were investigated by means of SEM, rheology, TGA and DSC. The study of the composites morphology revealed both a better dispersion and less covering of ZnO nanoparticles when dispersed in the PA matrix. These observations are based by the rheological properties, which demonstrated a higher impact of ZnO particles on the effective viscosity of the LDPE composites. This was attributed to a reinforcement of the matrix by the filler, which is coherent with the thermogravimetric measurements, where strong interactions between ZnO–LDPE macromolecules were put into evidence.
Moreover, the nucleating effect of ZnO onto the LDPE phase was very significant with an important increase in the crystallinity rate. When particle serves as a nuclei site, it is more likely to be surrounded by crystalline phase. Hence, particles are less covered by polyamide phase.
As a consequence, at the material surface, the amount of ZnO particles in direct contact with bacterial media, since microorganisms evolve in aqueous solution, is drastically larger in PA 6 than in LDPE. Moreover, depending on the water diffusion in PA matrix, the particles in core could also take part in the overall antibacterial capability. However, this effect remains minor due to the low water content of PA6, 0.5w/w%.
Footnotes
Acknowledgements
The authors thank Dr C. Rossignol in providing us with ZnO powder.
Funding
This research work was financially supported by the European Union, Midi-Pyrenees Regional Council and the Department of Industry, Trade and Tourism of the Basque Government (SAIOTEK Program).
