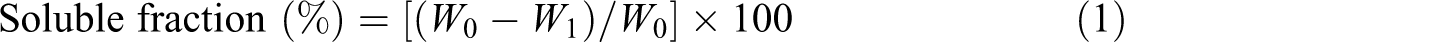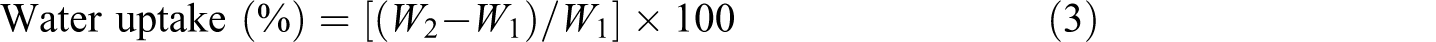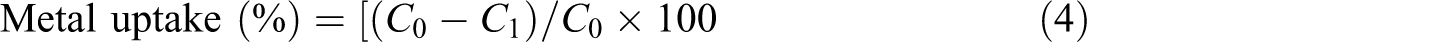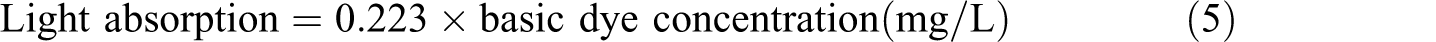Abstract
In this work, poly(vinyl alcohol) (PVA)/clay hydrogel based on various ratios of clay (20%, 50%, and 60%) were prepared using freezing and thawing techniques followed by electron beam (EB) irradiation at different irradiation doses (20, 25, and 30 kGy). The physicochemical property of such hydrogel in terms of gel fraction (%) and water uptake (%) was investigated. The structure and property of the hydrogel were investigated by x-ray diffraction (XRD) and scanning electron microscope (SEM). Thermal property was investigated using differential scanning calorimetry and thermogravimetric analysis. The results indicated that the gel fraction (%) increases on increasing the clay content in the hydrogel at all irradiation doses. The results obtained by XRD and SEM show an intercalation and exfoliation between PVA and clay. The occurrence of one T g and T m of the hydrogel indicates the presence of complete miscibility between PVA and clay. Also, the prepared hydrogel exhibit high thermal stability by increasing clay content in the hydrogel. It was found that the prepared PVA/clay hydrogel has a high water uptake, making it acceptable for the use in treatment of wastewater from heavy and toxic metal ions and dye wastes.
Introduction
Hydrogels are three-dimensional hydrophilic polymer networks capable of imbibing large amounts of water, which have been used widely in the field of industrial applications. 1 Numerous applications of polymer hydrogels have been described recently. 2
Synthetic polymers such as poly(vinyl alcohol) (PVA), polyvinyl pyrrolidone (PVP), and polyethylene oxide (PEO) hydrogels have been studied widespread, but their properties need to be improved further for special applications.
PVA has wide commercial applications due to its unique chemical and physical properties. It is a nontoxic, highly crystalline, water-soluble polymer having good film-forming and high hydrophilic properties. However, PVA as a soluble polymer cannot be used in wide applications. Thus, it has to be converted to completely insoluble materials with high mechanical properties. 3
Modification of the barrier and physical properties of polymers using nanoscale filler particles with a high surface area was investigated. Montmorillonite (MMT) is the most commonly used layered silicate because it is environmentally friendly and readily available in large quantities at low cost. Therefore, it is possible to improve the properties of polymer by the addition of small amount of MMT.4–6
The effect of organo clay quantity on the structural, swelling, physical, and mechanical properties of PVA nanocomposite hydrogel wound dressing was investigated. 7 The results indicated that the quantity of the clay added to the nanocomposite hydrogel is the key factor in obtaining suitable properties required for wound dressing such as relatively good swelling, appreciated vapor transmission rate, excellent barrier properties against microbe penetration, and mechanical properties.
Strawhecker and Manias 8 prepared the PVA/clay nanocomposites by casting the water suspension of PVA and MMT. They studied the structure of the nanocomposite and found a coexistence of exfoliated and intercalated MMT layers, especially for low and moderate silicate loadings. They also reported that the mechanical, thermal, and water vapor transmission properties of the nanocomposites are superior to that of the pure PVA and its conventionally filled composites.
This work concerned with the preparation of PVA/clay hydrogel with different clay concentrations. The effect of different doses of electron beam (EB) was studied. The dispersion of clay into the PVA matrix and the effect of its addition on the structure and properties of PVA/clay hydrogels were investigated by different techniques such as differential scanning calorimetry (DSC), x-ray diffraction (XRD) analysis, and scanning electron microscope (SEM). Moreover, thermal properties of the prepared hydrogels were investigated by thermogravimetric analysis (TGA). The efficiency of PVA/clay hydrogel for uptaking metal ions such as Cu(II) and Cr(III) and dye sorption was measured using ultraviolet (UV)–visible (vis) spectrophotometer.
Materials and experimental techniques
Materials
PVA, laboratory grade, in the form of powder, having an average molecular weight of 125,000 is obtained from the Laboratory Rasayan S. D. Fine-chem Ltd. (Boisar). Clay montomorillonite (MMT) was supplied by Zhejiang Fenghong clay chemicals Co. Ltd (People’s Republic of China). Two dyestuffs belonging to different classes were used throughout this work. These dyes are sandocryl blue (basic dye), which was kindly supplied by Hockest (Germany), and Remazol Golden (reactive dye), which was kindly supplied by Sandoz (Switzerland). The treatments of industrial wastewater were carried out on a dye waste taken from the draining released from Hosni Dyers and Finishers Co. (Cairo). The dye waste contains different reactive dyes: Ramazol black, Remacryl Red2 BS, and Ramazol blue RSP at different concentrations according to the factory recipe.
The reactive dye Remazol, a product of Hockest, contains different colors and the usual Glauber salt, sodium hydroxide, and detergents were added. The metal salts (CuSO4, NiSO4, and CoSO4) used were of pure and AR grades.
Experimental techniques
Preparation of PVA–clay hydrogel
PVA-clay hydrogel was prepared by dissolving different ratios of PVA and clay 20%, 50%, 60%, and 80% in a known volume of distilled water on hot plate stirrer at 40°C for 4 hours. After complete solubility, the solution was poured into a Petri dish followed by freezing at −20°C for 18 hours and thawing at a room temperature for 6 hours; for three consecutive cycles then exposed to different doses of 20, 25, and 30 kGy at EB irradiation then dried at ambient air to obtain hydrogel.
Analysis and measurements
Gel fraction
A known weight of the sample (W
o) was extracted in a refluxing system by boiling in bidistilled water for 2 hours. The samples were then removed and dried in ambient air to get rid of excess water to reach a constant weight (W
1). Finally, the soluble fraction was calculated according to the following equation
where W
0 is initial weight of the dry sample and W
1 is the weight dry insoluble gelled part after extraction
Percentage of water uptake
A known weight of the sample was soaked in bidistilled water for different time intervals at room temperature; the sample was then removed and blotted on filter paper to remove the excess water on the surface. The percentage of water uptake was calculated using the following equation
where W 1 is initial weight of the sample and W 2 is final weight of swollen sample.
Metal ion uptake
An attempt was made to use the modified polymers for the removal of some heavy metals such as Cu2+, Ni2+, and Co2+ from wastewater. Different metal salt solutions (CuSO4, NiSO4, and CoSO4) of known concentration (500 ppm) were first prepared. A sample of known weight (0.5 g) of the modified polymer was then immersed in these solutions for different time intervals ranging from 1 hour to 24 hours. The metal uptake (%) was determined according to Nizam and Ibrahim
9
(2000) using the following equation
where C 0 and C 1 are the concentrations of the metal solution before and after sorption, respectively.
Dye sorption measurements
The dye sorption of basic and reactive dyes by modified polymers was carried out by a general procedure based on the measurements of the absorption of the dye solution. Standard curves were first constructed representing a relationship between different known concentrations from basic and reactive dye and the corresponding absorption, which was measured at the wavelengths of 660 and 384 nm, respectively.
The different expressions governing these relationships were calculated as follows
In this procedure, the dye solution was prepared as follows. A certain concentration from each dye under investigation (20 mg/L) was first dissolved in the boiled water. Thereafter, a constant weight of the sample was then immersed in different dye solutions and the dye uptake was determined by measuring the light absorption of the residual dye solution. The dye sorption by samples was determined according to the following equation
Instrumentation
X-ray diffraction
XRD patterns of the synthesized samples of clay were determined using x-ray diffractometer (PW1390) from Philips (Holland).
Scanning electron microscope
The surface morphology of the samples was investigated by SEM technique. The micrographs were taken with JSM-5400 instrument of Jeol (Japan).
Differential scanning calorimeter
DSC measurements were performed using a Perkin-Elmer DSC-7 calorimeter (Japan). A heating rate of 10°C/min was utilized under nitrogen atmosphere. The recorded glass transition temperature (T g) was taken as the temperature at which one half of the change in heat capacity ΔC p has occurred.
Thermogravimetric analysis
The TGA studies were carried out on a Shimadzu-30 (TGA-30, Kyoto, Japan) at a heating rate of 10°C/min in air over a temperature range from room temperature up to 500°C.
Ultraviolet spectroscopy
Occasionally, a UV-vis Pye Unicam Spectrophotometer Type Sp 8-200 (England) was used to determine C 1 and C 0. All spectrophotometric measurements were carried out at the room temperature.
Results and discussions
A series of experiments were carried out with the objective of obtaining the optimum concentration of clay and PVA, which gives a homogeneous hydrogel. Hence, hydrogel containing 80%, 70%, 60%, 50%, and 20% clay were prepared. The PVA/clay composites were irradiated with different EB irradiation doses (20, 25, and 30 kGy) after being subjected to three cycles of freezing and thawing. The effect of irradiation doses on the properties of this composite as gel fraction was investigated.
Gel fraction
The dependence of gel fraction (%) on the irradiation dose for hydrogel composed of different clay composition is given in Figure 1. From these results, it can be observed that there is no marked effect of irradiation dose on the gel fraction (%) for all the hydrogel compositions under investigation.
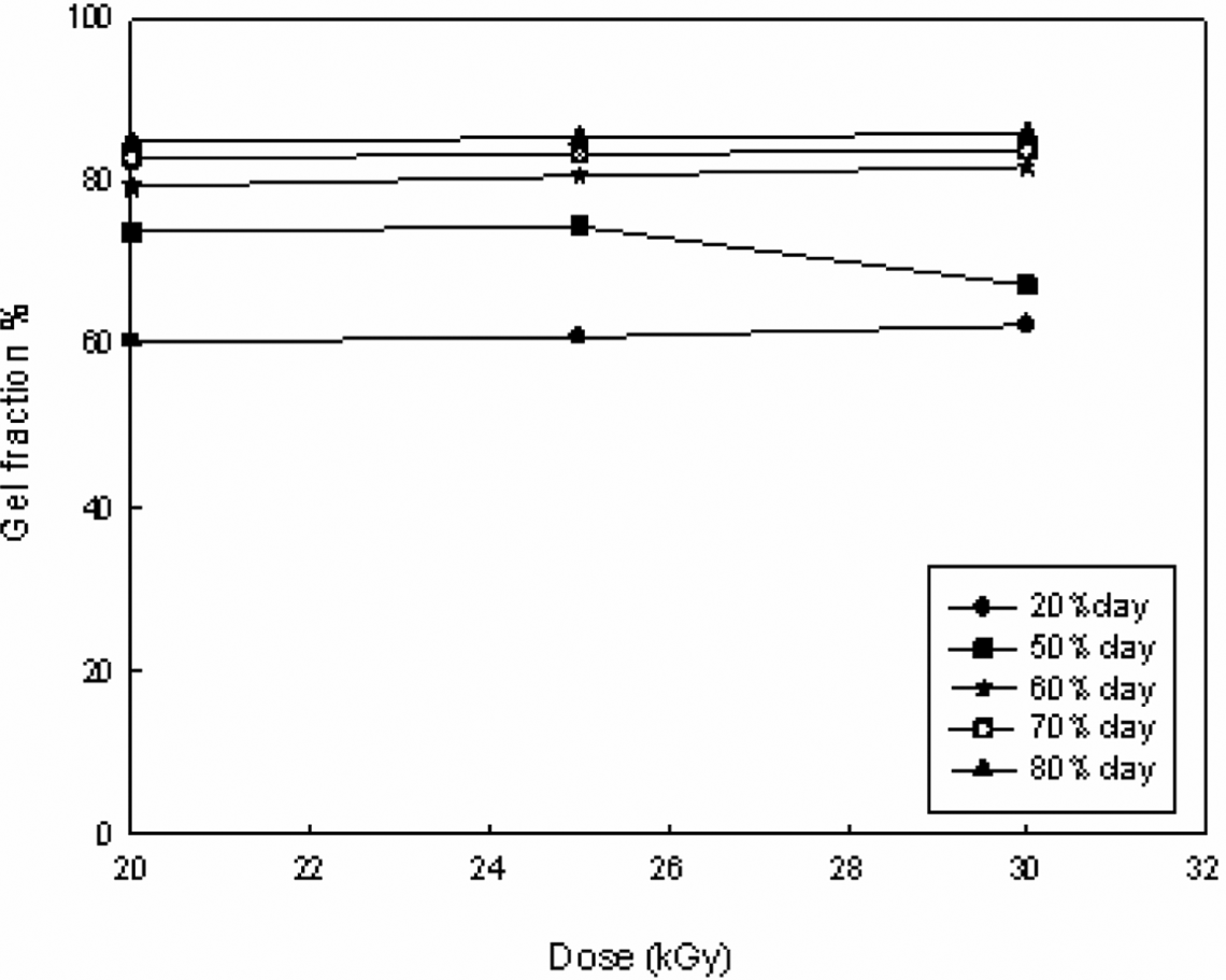
Effect of electron beam irradiation dose on gel fraction (%) of different hydrogel compositions.
Moreover, it can be deduced from Figure 1 that the suitable dose used for this study is 20 kGy, since there is no change in gel fraction (%) on increasing the irradiation dose up to 20 kGy, as other workers. 10 Afterward, the relationship between gel fraction (%) and hydrogel composition was focused at 20 kGy. The results obtained are shown in Figure 2. From these results, it was found that on increasing the clay content especially from 20% to 60%, there is an increase in the gel fraction (%). Further increase in clay content in the hydrogel is accompanied by a leveling off in the gel fraction (%). As for example, the gel fraction (%) is equal to 60%, 74%, 81%, 83%, and 84% at 20%, 50%, 60%, 70%, and 80% clay, respectively.
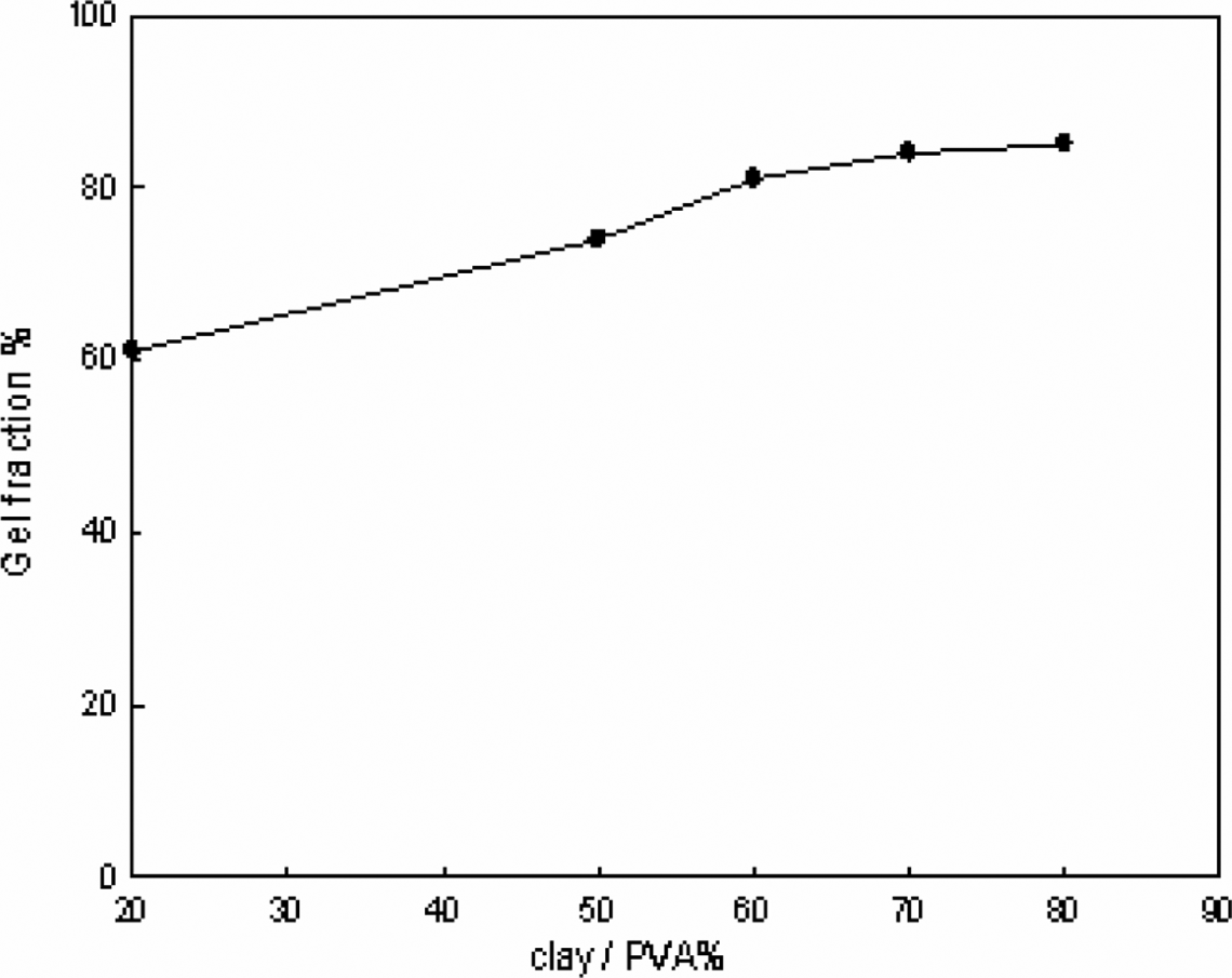
Effect of different clay/poly(vinyl alcohol) percentage on gel fraction (%) at a constant irradiation dose (20 kGy).
Moreover, the results in Figure 2 reveal that the presence of clay, which acts as filler, with the three-dimensional networks of hydrogel that causes an increase in cross-linking, creating more entangling structure. Here, it is possible to mention that when clay is added to hydrogel, strong interactions are developed between functional groups of clay and polymer chain. Repeated cyclic freezing and thawing have an important role in the increasing gel fraction (%) summarized as follows.
Additional interaction between PVA and clay.
The bonds exist between long neighboring chains of PVA induced crystallization and hence cause an increase in gel fraction (%). 7
From the practical point of view, the hydrogel should exhibit suitable hydrophilic properties to be used in removing metal ions and waste dyes from wastewater. Hydrophilic property of the hydrogel is investigated by measuring the percentage of water uptake.
Percentage of water uptake
Water content is a very important factor for the applicability of synthetic absorbents in the field of removal of pollutant from wastewater.
Hence, the effect of immersing time on percentage of water uptake of different hydrogel compositions of 20%, 50%, and 60% clay at constant irradiation dose (20 kGy) was studied. The results obtained are shown in Figure 3, which indicates that the water uptake increases with increasing immersing time up to 5 hours, then there is a leveling off for the three hydrogel compositions under investigation. The affinity of PVA/clay hydrogel for water may be explained by the presence of resting places that facilitate the diffusion of water.
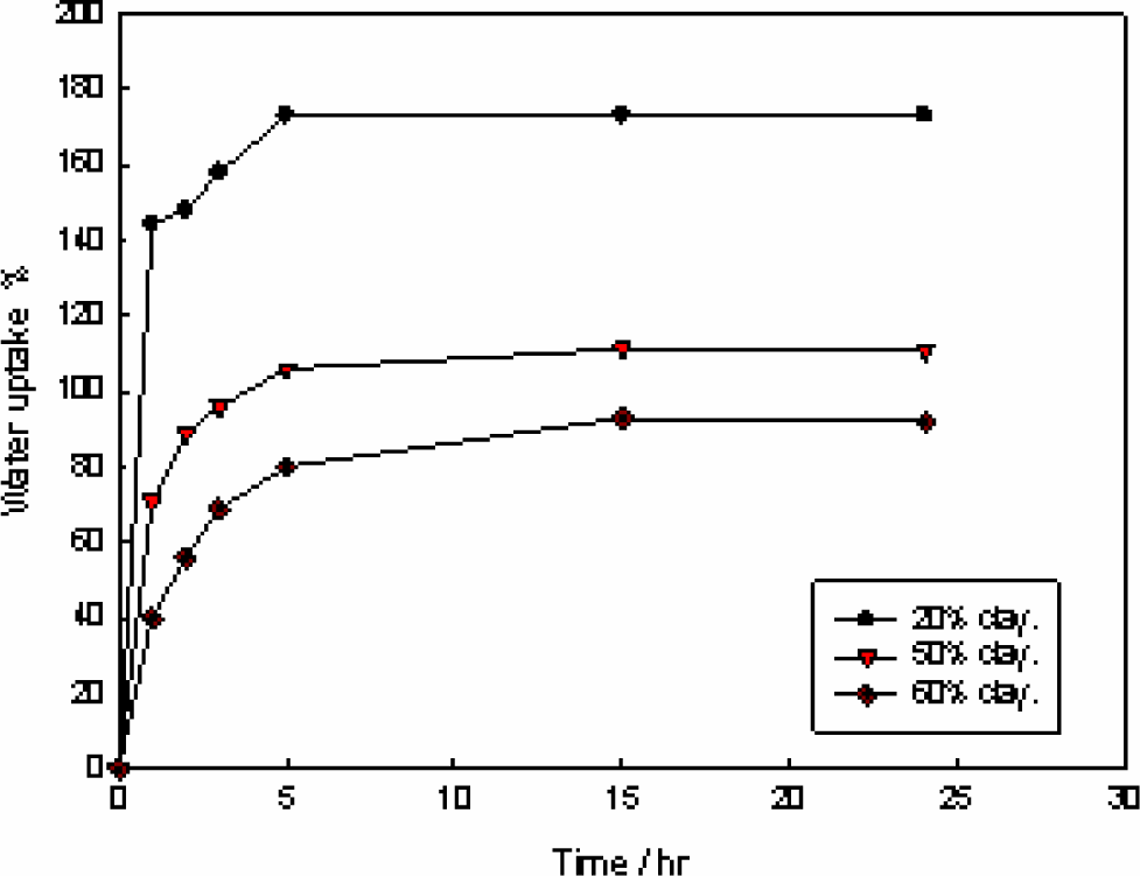
Effect of immersing time on water uptake (%) for clay/poly(vinyl alcohol) hydrogel prepared at 20 kGy.
Moreover, it was found that the higher clay content will be the lower percentage of water uptake because higher clay shows a higher percentage of gel, which is inversely proportional to water uptake (%). A higher clay concentration results in the generation of more cross-linking density of nanocomposite and leaves less space for water to enter; in addition, clay itself has lower water absorbancy than PVA; therefore, the low value of water absorbancy of clay reduce the water uptake (%) of the hydrogel. 11
Structural property of PVA/clay hydrogel
The structural properties of PVA/clay hydrogels were investigated by XRD and SEM XRD analyses. XRD analysis is a conventional technique generally used to obtain the first information on nanocomposite and to identify intercalated structures in these nanocomposites. The XRD profile of pure clay and PVA/clay hydrogel containing 20%, 50%, and 60% clay composition is shown in Figure 4. It was seen from the results obtained that the characteristic diffraction peak for pure clay is observed at 6.6°, whereas it disappears for the hydrogel containing 20%, 50%, and 60% clay composition. The absence of this clay peak reflects the dispersion and intercalation between the clay and the PVA.
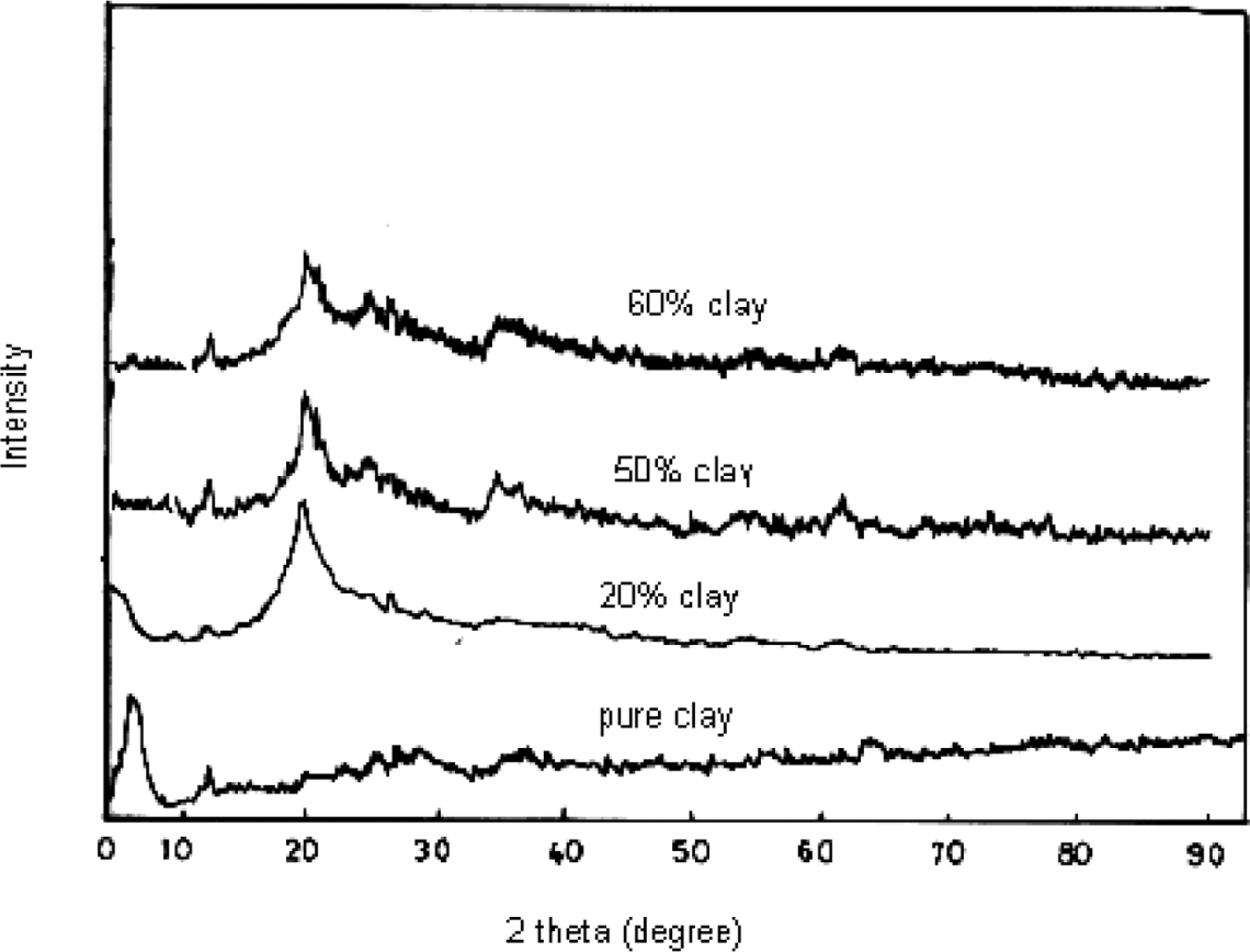
XRD patterns of pure clay and different ratios of PVA/clay. PVA: poly(vinyl alcohol); XRD: x-ray diffraction.
The disappearance of the characteristic peak of clay in addition to the increases in interlamellar space of clay as shown in Figure 4 was in agreement with the results obtained in other researches,8,12 which confirm the intercalation and exfoliation between clay and PVA.
Structure morphology
The strength of interfacial interaction between polymer matrix and layered clay was investigated by examining the fracture surfaces using SEM. The results obtained are shown in Figure 5. The SEM micrograph of pure PVA is characterized with a smooth surface. On the other hand, the SEM micrographs of PVA/clay hydrogel containing 20%, 50%, and 60% clay Figure 5(b) to (d) was examined. These figures showed a different morphology in which the surface is affected by the intercalation with clay.

Scanning electron micrographs of PVA/clay hydrogel with different ratios. (a) pure PVA, (b) pure clay, (c) 20% clay/PVA, (d) 50% clay/PVA, and (e) 60% clay/PVA. PVA: poly(vinyl alcohol).
It was found that the surface morphology of clay/PVA with 20% and 50% as shown in figure 5(b) and (c) show a denser dispersion of layers of clay in PVA, while hydrogel containing 60% clay is characterized by the corn flakes texture and is also characterized by the presence of cavities and holes as shown in Figure 5(d).
Thermal property of PVA/clay hydrogel
Thermal properties such as DSC and TGA were measured.
Differential scanning calorimetry
DSC technique was used to study the effect of addition of different concentrations of clay to PVA on T g and melting transition temperature (T m) of the hydrogel. Thus, the T g and T m and T m of pure PVA and PVA-clay hydrogel with different ratios of 20%, 50%, and 60% at 20 kGy are shown in Table 1. Pure PVA hydrogel has a T g at ∼83°C and a melting point at ∼190°C. On the other hand, clay is an organometallic compound; so it seems that its T g is weak or too broad to measure. 13 From the results obtained in Table 1. It was found that T g of the PVA/clay hydrogel begin to have a definite value after adding 20%, 50%, and 60% clay equal to 90°C, 93°C, and 106°C, respectively. It was found that the presence of clay causes an increase in T g and T m. These results are evident since clay acts as filler and increases the cross-linking density. Also the presence of one T g and T m of the hydrogel indicate the complete compatibility and miscibility between clay and PVA.
Glass transition and melting temperatures of PVA/clay hydrogel containing 20%, 50%, and 60% clay composition.

PVA: poly(vinyl alcohol).
In another work, 8 a systematic study of DSC for PVA/MMT with increased MMT content (1%–5%) does not detect any T g between room temperature and 120°C, which is due to the very small content of MMT with respect to the PVA matrix.
This work was carried out using a ratio of clay equal to 20%, 50%, and 60%, which permits the appearance of T g at a precise value. In addition, the high content of clay with its inorganic layers affects all the polymer morphology and hence affects the T g and Tm of the hydrogel.
Thermogravimetric analysis
TGA results of pure clay, pure PVA, and clay/PVA hydrogel with its entire ratio in the temperature range from 0°C to 500°C are shown in Figure 6 and the percentage weight loss at different decomposition temperatures for the same hydrogels is shown in Table 2. The TGA of pure clay shows two degradation steps attributable to water loss (first step) and to clay dehydroxylation at ∼725°C (second step). 14 Within the temperature range from 0°C to 280°C, there is nearly no difference in the thermal stability between PVA bulk polymer and PVA/clay hydrogel with all its compositions. Pure PVA shows weight loss at 300°C equal to 40%, while pure clay shows a weight loss at the same temperature equal to 0%. Hydrogel containing 20%, 50%, and 60% shows weight loss equal to 12%, 10%, and 6%, respectively, at the same temperature. The increase in temperature from 300°C to 500°C does not affect the thermal stability of the clay, hence weight loss is approximately 10%, while the weight loss of PVA is equal to 86% at the same temperature range. On the other hand, the hydrogel nanocomposites containing 20%, 50%, and 60% clay show a weight loss equal to 63%, 29%, and 22%, respectively, at the same temperature range. It can be concluded from all the above results that the PVA nanocomposites loaded with 20%, 50%, and 60% clay possesses higher thermal stability than that of neat PVA polymer.
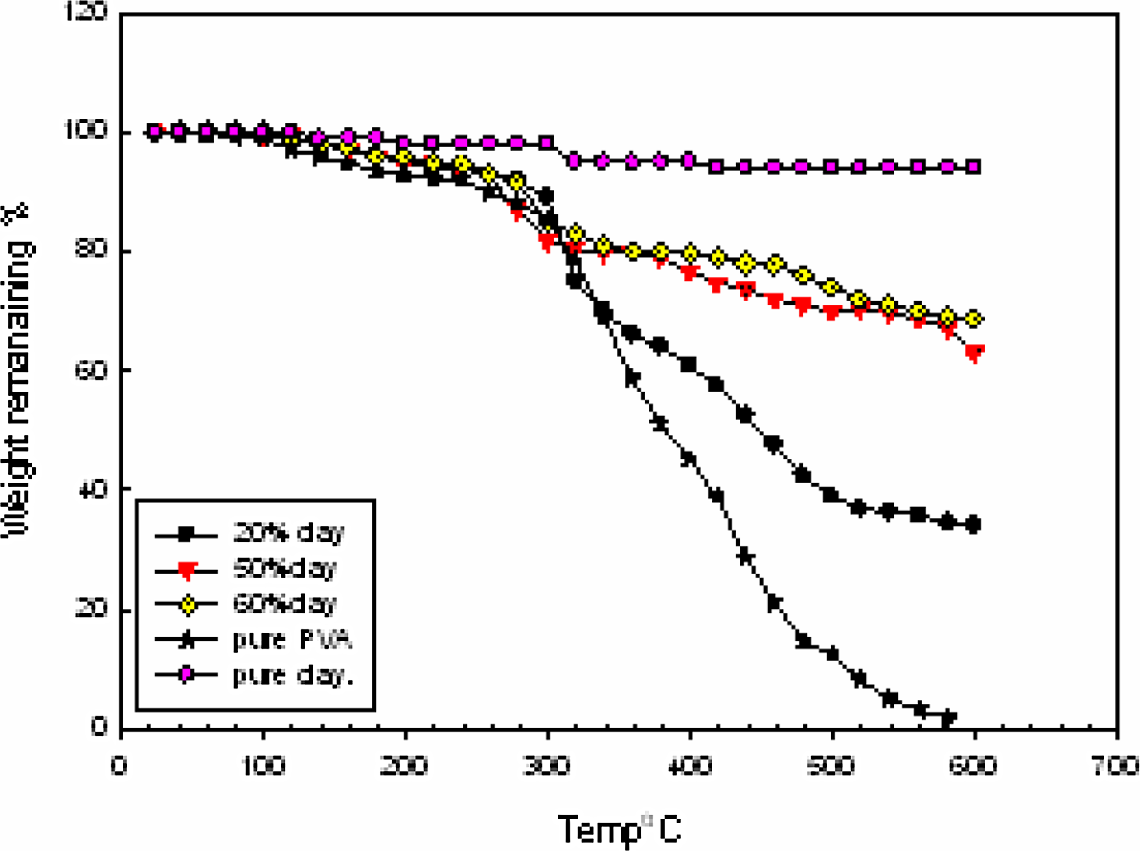
Thermogravimetric analysis thermograph of poly(vinyl alcohol)/clay with different ratios.
Weight loss (%) at different decomposition temperatures for pure PVA, and pure clay and hydrogel with different ratios.
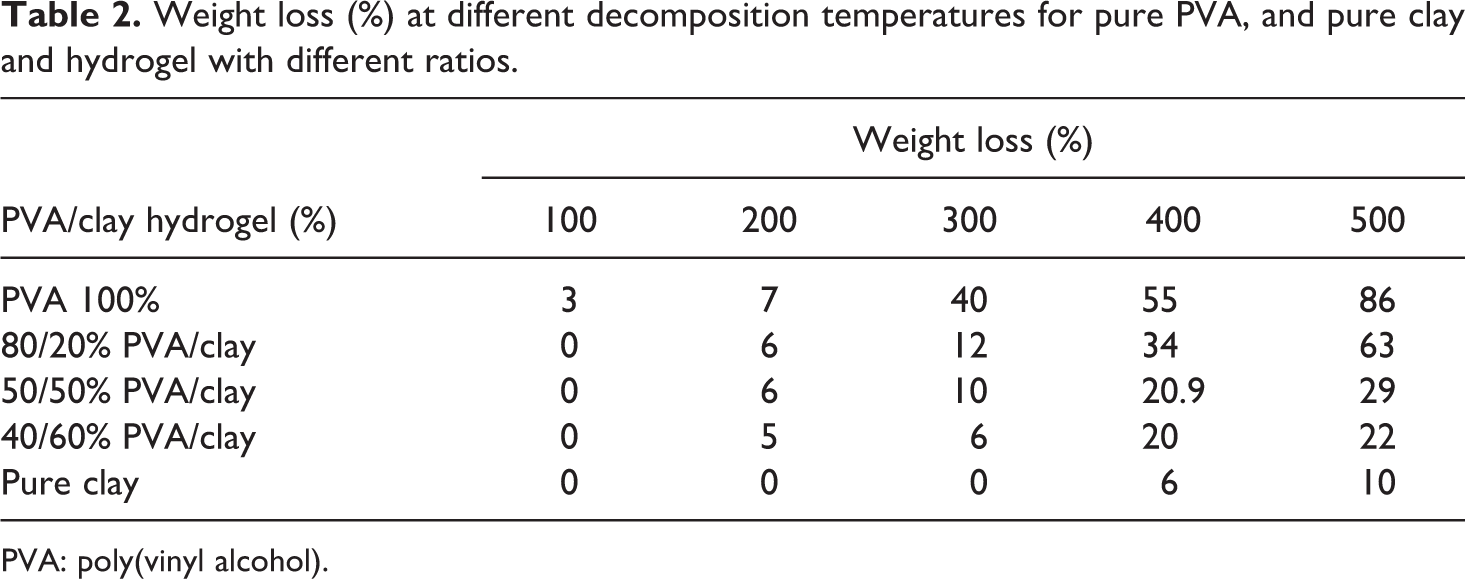
PVA: poly(vinyl alcohol).
Treatment of wastewater by the prepared hydrogel
From the practical point of view, it is valuable to use some hydrogels for minimizing the environmental pollution. In this respect, hydrogel of PVA/clay containing various functional groups acts as chelating agents and is used for the purpose of wastewater treatment from some heavy metal ions and dye wastes.
Uptake of metal ions
The percentage of uptake of metal ions was determined by UV/vis spectrophotometric analysis based on the absorption of different colored solution for different metal ions before and after metal uptake process.
UV/vis spectrophotometric analysis
Figure 7(a) and (b) shows the relationship between percentage of uptake of metal ions and immersing time of PVA/clay hydrogel of 50% and 60% clay for different concentrations of heavy metal ions. From the obtained results, it was found that the hydrogel containing 50% clay shows maximum percentage of uptake of metal ions was obtained after 15 hours for all metal ions under investigation and then up to 15 hours there is a leveling off. In case of hydrogel containing 60% clay, there is a continuous increase in the percentage of uptake of metal ions for Co2+ and Ni2+ with immersing time, while there is a leveling off after 5 hours in case of Cu2+. The result shows that clay/PVA hydrogel have a tendency toward metal ions according to the following order

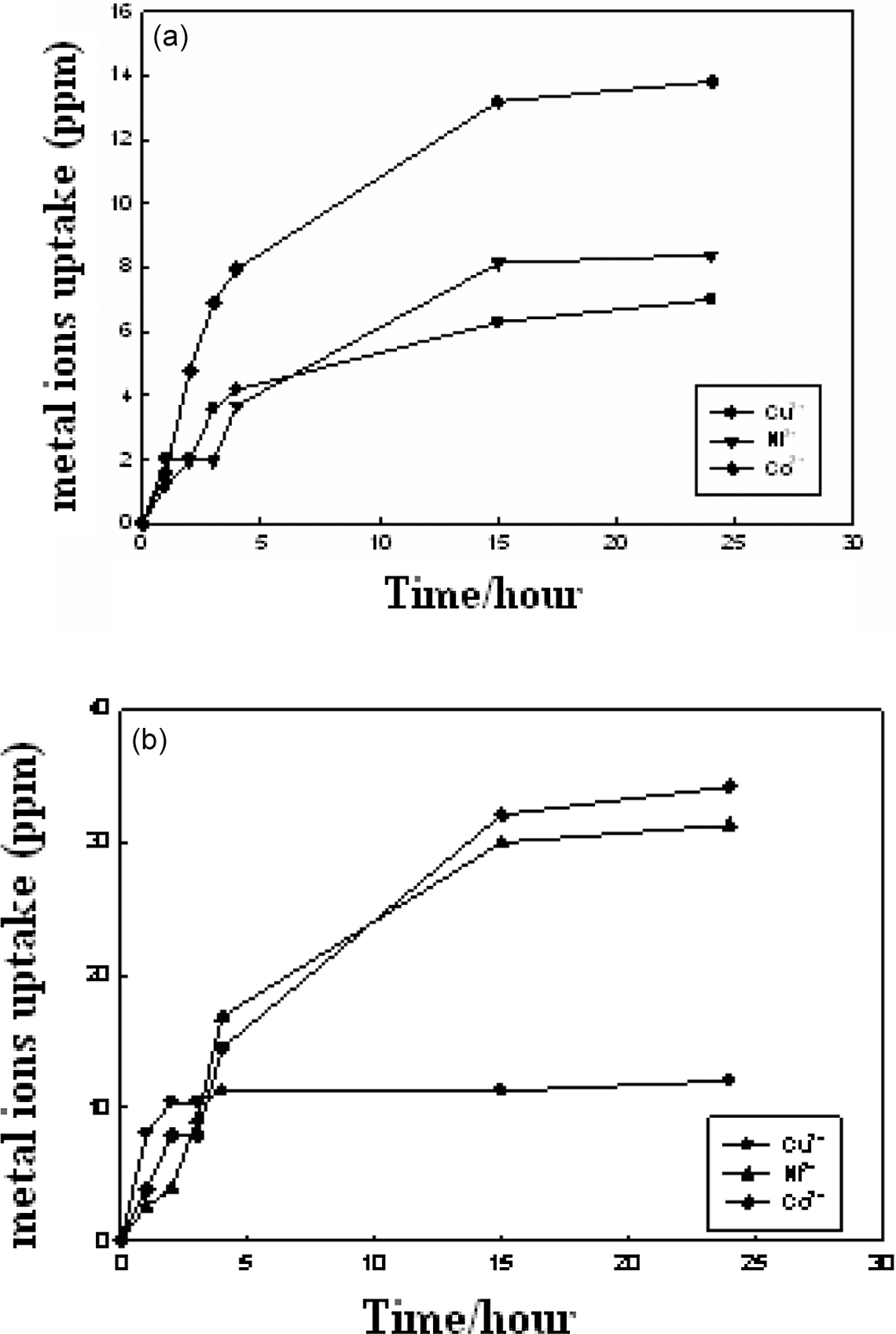
Effect of immersion time on the uptake of metal ions of hydrogel containing (a) 50% clay and (b) 60% clay.
Moreover, the results obtained show that generally the hydrogel having a concentration of 60% clay has a high tendency toward the metal ion absorption than that which contain 50% independent of the type of metal ions. These results can be explained by taking into consideration that alkali metal ions present in the main structure of clay, due to the substitution of Si4+ ion by Al3+ ion in some layers, act as active sites in the absorption of clay minerals toward heavy metal ions. The diffusibility of such metals through the pores of the hydrogel and the stability of chelation of metal ions to polymer hydrogel depends on molecular size and atomic radii of metal ions. The affinity of clay/PVA hydrogel toward such metal ions may be attributed to the presence of free hydroxyl groups along its molecular structure which permit the occurrence of ion exchange reaction. These modified clays were successfully used in wastewater treatment. This is because the modified clays can absorb some heavy metal ions by coordination, formation of hydrogen bonding, or/and dipole association or through hydrogen bonding of water molecule.
Sorption of dye waste by clay–PVA hydrogel
In this part, aqueous solutions containing constant concentrations of the basic dye (sandocryl blue) and reactive dye (Remazol Golden) were prepared. The dye sorption of different dyes by clay–PVA hydrogel as a function of immersion time at room temperature was investigated using UV spectrophotometric analysis. The results obtained are shown in Figure 8. From these results, it can be seen that the value of percentage of dye sorption by the hydrogel differs from one dye to other. The dye sorption of the reactive dye was found to increase significantly after 3 hours and afterward tends to level off with increasing time of immersion up to 2 hours as shown in Figure 8. In case of basic dye, the sorption by the hydrogel was shown to increase substantially by increasing the time of immersion up to 24 hours. Moreover, the results show that the hydrogel that contain 50% clay has a higher tendency toward the two dyes under investigation than that which contains 60% clay.
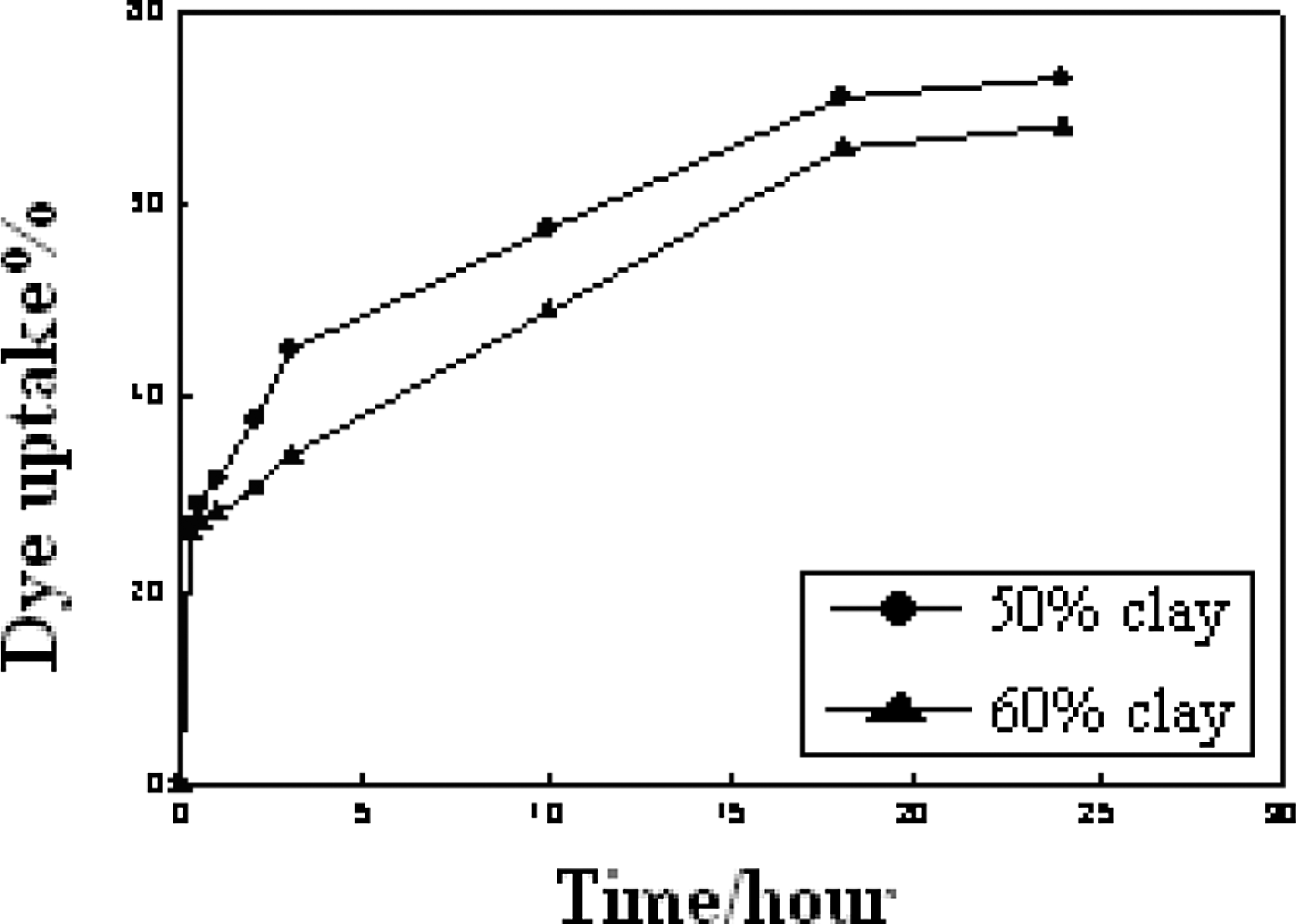
Dye uptake percentage of basic dye (sandocryl blue) by poly(vinyl alcohol)/clay hydrogel at different immersion times in room temperature.
The relative sorption affinity of the hydrogel for the different dyestuffs may be explained on the basis of the following points:
The affinity of the hydrogel for absorbing different dyestuffs is essentially due to the highly hydrophilic groups present in the hydrogel. Also the sorption process goes through a chemical bonding between the active groups of the different dyes and those of the hydrogel and not through physical absorption.
The mechanism of dyeing with basic dyes implies the presence of an anionic part in the medium to react with the cationic part of the dye through ionic bond. Therefore, the sorption of the basic dye occurs through the reaction of hydroxyl groups of the PVA component of the hydrogel and the cation of the dye.
The rearrangement of the hydrogel structure during freezing and thawing before irradiation may also facilitate the absorption and diffusion of the dye into the hydrogel.
The chemical interaction between the reactive dye and hydrogel may be explained on the same basis of the dyeing of the cellulose fibers. 15 In this regard, the dyeing process goes through the OH groups to form a covalent bond with the reactive dye.
Moreover, the different color interceptions and color difference (ΔE) of hydrogel containing 50% clay for different dyestuffs were investigated. It should be noted that the color difference (ΔE) was measured after the sorption process had been completed or the sorption reaches equilibrium. The results obtained are shown in Table 3. From these results, it was found that the hydrogel containing 50% clay shows a great ability to basic dye than reactive dye.
Color–strength measurement of poly(vinyl alcohol)/50% clay hydrogel after the absorption of reactive and basic dyes at room temperature over night.
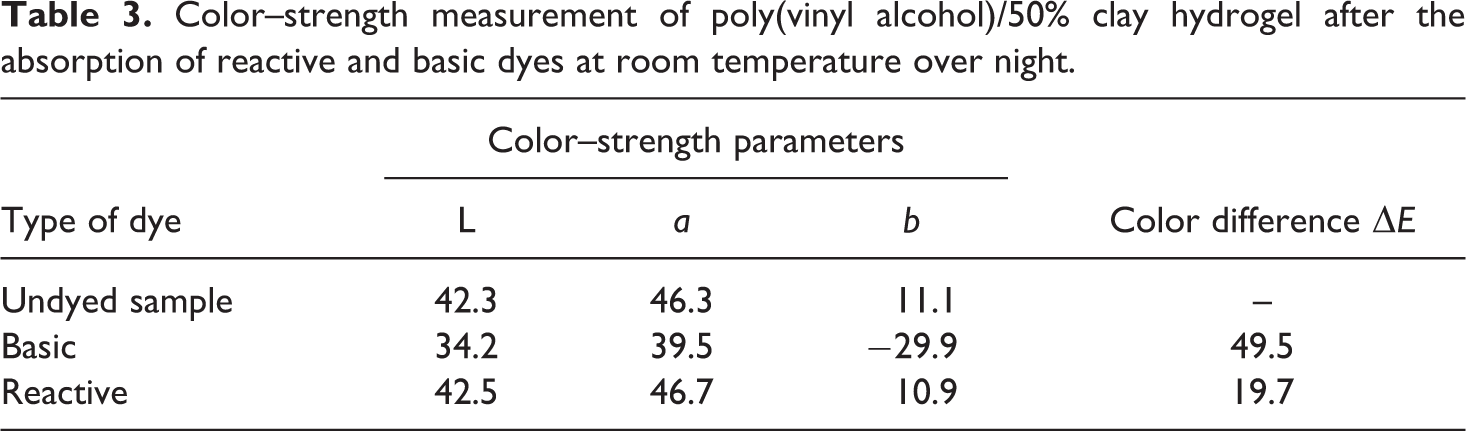
It is obvious that there is a good agreement between the data of the dye uptake measured spectrophotometrically and that obtained from the color difference value.
Treatment of industrial dye waste
In the present work, clay/PVA hydrogel was utilized in the treatment of industrial dye waste naturally released from some textile factors in Cairo. It should be noted that the dye waste sample was taken from the drain of dye bath at the end of the dyeing process. This dye bath contains, beside the reactive dyes, the usual additives used during dyeing such as salts, wetting agent, and alkalies. In practice, it is difficult to determine the amount of the absorbed dye by the hydrogel because the concentration of dyes in the waste drains is unknown. However, an attempt was made to determine approximately the dye sorption of the industrial dye waste by measuring the dye sorption of this class of dyestuff by measuring the color strength (ΔE) of hydrogel. The results obtained are shown in Table 4. It is obvious from this result that the hydrogel shows strong affinity toward the industrial dye waste (reactive dyes).
Color difference intensity (ΔE) of clay/poly(vinyl alcohol) immersed in industrial dye waste for different times.

Conclusion
PVA/clay nanocomposite hydrogel based on various ratios of 20%, 50%, and 60% of clay were exposed to freezing and thawing cycles (three times) followed by EB irradiation at doses of 20, 25, and 30 KGy, respectively. The effect of EB on the physicochemical property of such hydrogel was investigated. DSC confirmed the miscibility and compatibility of PVA/clay hydrogel. The occurrence of one T g and T m of the hydrogel indicate the presence of complete compatibility and miscibility between PVA and clay. The gel fraction increases on increasing the clay content in the hydrogel at all irradiation doses. XRD and SEM confirm the intercalated and exfoliated structure of the hydrogel. The prepared hydrogel has a high water uptake, making it acceptable for use in the treatment of wastewater from heavy and toxic metal ions and dye wastes. PVA/clay hydrogel rich in clay possess a high thermal stability.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.