Abstract
A new type of N-isopropyl acrylamide/high-substituted hydroxypropyl cellulose/graphite carbon nitride (NIPAAm/HHPC/g-C3N4) smart hydrogel-based photocatalyst with thermally driven characteristic was successfully prepared by electron beam pre-radiation polymerization and radiation cross-linking methods. The agglomeration and loss of g-C3N4 nanosheets can be avoided effectively, and ensured high photocatalytic activity under visible light, once the g-C3N4 nanosheets are uniformly dispersed into the skeleton of a thermosensitive NIPAAm/HHPC hydrogel. NIPAAm/HHPC/g-C3N4 (NHC) hydrogel was characterized by nuclear magnetic resonance spectroscopy, Fourier-transform infrared spectroscopy, differential scanning calorimetry, thermogravimetric analysis, X-ray diffraction, X-ray photoelectron spectroscopy, and diffuse reflectance spectroscopy. The microstructure of NHC was further characterized by scanning electron microscopy, transmission electron microscopy, and Brunauer–Emmett–Teller. The adsorption–photocatalytic removal rate of rhodamine B reached 71.4% at the mass ratio of g-C3N4 of 0.8% (NHC-0.8%) hydrogel in an aqueous medium under visible light. The thermal shrinkage ratio can reach 90.6% at 60°C after 5 min and could effectively achieve the function of recycling-free in a portable photocatalytic reaction device under the optimal conditions. Possible mechanism of adsorption–photocatalysis and thermally driven recycling-free on NHC hydrogel was also obtained. These thermally driven recycling-free characteristic and highly photocatalytic properties of the hybrid hydrogel-based photocatalyst show that it can be used as a promising new material with extensive applications in wastewater treatment.
Introduction
One of the most common smart hydrogels is a thermosensitive hydrogel, which can easily produce a stimulus response behavior with a change in external temperature.1–3 A low critical solution temperature (LCST) is one of the most representative properties of thermo-responsive polymers, 4 especially the LCST can be adjusted by varying the content ratio of hydrophilic and hydrophobic comonomer/polymer,5,6 which has been applied widely in the fields of controlled drug release, 7 drug delivery, 8 tissue engineering, 9 wound dressing, 10 sensor and actuator, 11 and so on. Our group 12 has also reported recently the thermosensitive discoloration hydrogel based on N-isopropyl acrylamide/high-substituted hydroxypropyl cellulose (NIPAAm/HHPC) has been fabricated via two steps of electron beam radiation. Small nanomaterials such as GO and Fe2O3 were also added severally into the system of hydrogel as chromogenic agent. Different colors can be displayed in nanocomposite hydrogel system at the low, medium, and high temperature, respectively. The LCST of hydrogel system can be also adjusted precisely by the changes of irradiation dose of the prepolymer and composition ratio. Experimental results showed that the hydrogel can be changed quickly itself color in its near LCST. The novel NIPAAm/HHPC-based nanocomposite hydrogel is suitable especially to indicate different fever temperatures for fever patients and can be expected to as a new type of temperature-sensitive sensor and replace the traditional clinical thermometer. Another, the hydrogel is a flexible material, its structure can be repeatedly expanded and collapsed, many activated sites for loaded catalyst can be provided by the high specific surface area. 13 Therefore, the thermosensitive hydrogel can be used as a carrier of functional (photo)catalysts. 14 However, as far as we know, there are few reports on thermosensitive nanocomposite hydrogels with the thermally driven recycling-free and highly photocatalytic properties.
Graphite carbon nitride (g-C3N4), as a novel “metal-free” semiconductor, has been considered as one of the star photocatalysts in view of its adjustable electronic structure and outstanding thermal stability, physicochemical stability, and photocatalytic activity. 15 Up to date, g-C3N4 is mainly used as a powder for photocatalytic reactions as photodegradation of pollutants and for solar fuel production.16–19 However, g-C3N4 nanosheets are restricted in practical applications because of the difficulties in separating and recycling them after photocatalytic reactions, also can possibly cause secondary pollution. Fortunately, the immobilization of g-C3N4 nanosheets in hydrogel-based polymer materials with an appropriate size allows the facile separation of the hydrogel-based photocatalysts after use and can be reutilized after convenient recycling processes.
Electron beam radiation is a novel preparation technique of hydrogel-based photocatalysts,12,20,21 which has many significant advantages compared with conventional chemical methods, such as high efficiency, no initiator, requiring no complex process to control the experimental conditions, and having no limit for the shape of the substrate.22,23 Therefore, it has become more necessary to develop green synthetic methods of photocatalysts/hydrogel-based nanocomposite functional materials for industrial processes in the future. 24
In this work, a new type of NIPAAm/HHPC/g-C3N4 (NHC) smart hydrogel-based photocatalyst was successfully prepared by an electron beam preradiation method. g-C3N4 nanosheets were uniformly dispersed onto the skeleton of a thermosensitive NIPAAm/HHPC hydrogel. Not only is the temperature sensitivity of the original hydrogel can not be affected by this method, but the agglomeration and loss of g-C3N4 nanosheets is effectively avoided. NHC hydrogel was characterized by nuclear magnetic resonance (NMR) spectroscopy, Fourier-transform infrared (FTIR) spectroscopy, differential scanning calorimetry (DSC), thermogravimetric analysis (TG), X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), and diffuse reflectance spectroscopy (DRS). The microstructure of NHC was further elucidated by scanning electron microscopy (SEM), transmission electron microscopy (TEM), and Brunauer–Emmett–Teller (BET). The photocatalytic activity of NHC was tested by the degradation of rhodamine B (RhB) in aqueous medium under visible light. The thermally driven recycling-free property of NHC photocatalyst is further verified by the portable photocatalytic reaction device under the optimal conditions. Possible mechanism of photocatalytic and thermally driven recycling-free are also revealed. These unprecedented properties of hybrid hydrogel-based photocatalysts can lead to new and promising materials for water purification.
Experimental section
Materials
HHPC (viscosity of a 2% solution being 6–10 mPa·s) was purchased from Maya Reagent Co., Ltd (China). NIPAAm was purchased from Sahn Chemical Technology Co., Ltd (China). Urea powder was purchased from Guoyao Co., Ltd (China). Other reagents were purchased from Wuhan Shenshi Chemical Co., Ltd (China). All the chemicals were analytical grade and used as purchased without further purification.
Preparation of g-C3N4
The synthetic process for g-C3N4 was as follows: 10 g urea powder was added into an alumina crucible, and the crucible covered. This was heated in muffle furnace (Sx2-4010) from room temperature to 550°C with a heating rate of 5°C min−1, then maintained at a constant temperature of 550°C for 3 h. After this reaction time, the resulting yellow powder was collected and washed with 0.1 mol L−1 HNO3 and ultrapure water three times. Finally, the g-C3N4 sample was obtained by drying at 60°C for 24 h.
Preparation of thermosensitive NIPAAm/HHPC hydrogel
NIPAAm/HHPC hydrogel was prepared by the radiation cross-linking method. As shown in Figure 1, 10.0 g of NIPAAm was dissolved in 100.0 g ultrapure water, then magnetically stirred under nitrogen for 0.5 h until completely dissolved. Finally, it was injected into a polyethylene bag (200 × 150 mm2) with a syringe under anaerobic conditions and sealed. At room temperature, the monomer solution was radiated at a dose of 25 kGy and at a dose rate of 5 kGy pass−1 by a 1 MeV electron accelerator (Wasik Associates, Erie, USA), and the prepolymer (solution A) was obtained after radiation. After that, 10.0 g HHPC was dissolved in 100.0 g deionized water to obtain a colorless, transparent 10% HHPC aqueous solution (solution B). Then, solution A was added to solution B to form a mixture with mass ratio 1:1, and to a blank the same amount of water was added. After the solution was fully mixed and added to a 90 mm culture dish, a 3 mm thickness of film was produced (the thickness of the hydrogel film can be adjusted according to the loading volume). Finally, the samples were irradiated by electron beam at room temperature (50 kGy, 5 kGy pass−1). The samples were soaked in ultrapure water for 48 h and washed in excess ultrapure water to remove the unreacted components. Finally, the washed product was dried in a freeze dryer (SCIENTZ-10N, China) for 24 h to obtain the NIPAAm/HHPC thermosensitive hydrogel.
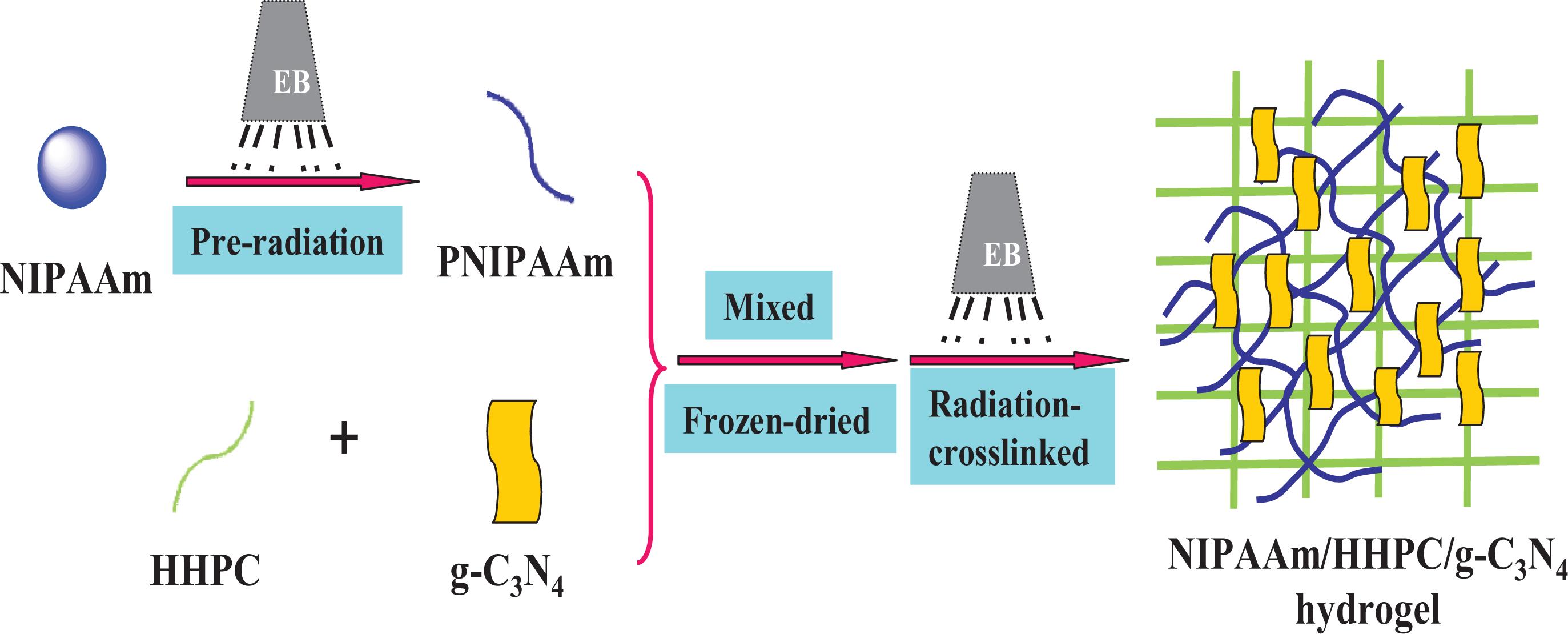
Schematic preparation of NHC smart hydrogel-based photocatalyst.
Preparation of thermosensitive NHC hydrogel
The preparation of NHC hydrogel was similar to that of NIPAAm/HHPC hydrogel. The only difference was that a certain amount of g-C3N4 was added to the mixed solution of HHPC and NIPAAm prepolymer. The prepared samples were denoted as NHC-X%, where X% corresponds to the mass ratio of g-C3N4 to NIPAAm/HHPC. In our experiments, the hydrogels were randomly cut into different sizes of microspheres and graded using a standard test sieve (diameter range 75–150 μm, Shangyu, GB6003.1-1997, China).
Characterization
Thermal stability and the g-C3N4 content in the composite hydrogels were determined by a NETZSCH thermogravimetric analyzer (TG209F3, Germany). The dried samples were cut into powder in a crucible and heated from 30°C to 800°C at a heating rate of 10°C min−1 under nitrogen. The melting temperature of the dried samples was determined using a DSC200F3 (NETZSCH, Germany) under a nitrogen atmosphere, with a heating rate of 5°C min−1 from 30°C to 50°C. NMR spectra were recorded on an ACF400 spectrometer (Bruker, 400 MHz, Germany). The crystal structures of the hydrogels were characterized by X-ray powder diffraction (LabX XRD-6100, Shimadzu, Japan). The patterns with Cu-Kα radiation (λ = 0.15406 nm) at 40 kV and 20 mA were recorded in the 2θ range of 5–80° (2θ) with a scan speed of 4° min−1. The morphology and microstructure were characterized by field emission SEM (ZEISS Merlin Compact VP, Germany) and SEM (VEGA-3 SBH, Tescan, Czech Republic). Before examination, the hydrogels were freeze-dried. Then, the dried hydrogels were cut open to expose their inner structures, and the cross-sections were examined. High-resolution TEM images were obtained by field emission transmission electron microscopy (JEOL, JEM-2010F, Japan). FTIR spectra were recorded on a NICOLET 5700 spectrometer (Thermo Fisher, Waltham, Massachusett, USA) in the range of 4000–400 cm−1. Ultra violet–visible (UV-vis) absorption spectra were obtained at room temperature by a UV-vis spectrophotometer (Shimadzu, UV-2600). BET specific surface area of the aerogels was determined at 77K (NOVA TOUCH LX1, Quantachrome, Florida, USA). Prior to the measurements, the samples were heated under vacuum at 423K for 5 h. XPS measurements were performed using Perkin-Elmer PHI 5000C XPS (Waltham, Massachusett, USA).
Measurement of photocatalytic activity of NHC
The photocatalytic activity of NHC-X% was evaluated at room temperature (25 ± 2°C) by measuring the removal rate of RhB from solution in a portable photocatalytic system. First, 50 mg photocatalyst was dispersed into 50 mL RhB solution (10 mg L−1) and allowed to equilibrate in the dark for 1 h to ensure the adsorption–desorption equilibrium. Then, the mixed solution containing NHC-X% hydrogel-based photocatalyst was irradiated with a 300 W xenon lamp (UV cutoff filter, λ > 400 nm) at the distance of solution 12 cm. After each experiment, there was no elution, but the adsorbed substrate (5 min at 60°C) was squeezed out directly by thermal switching and fresh RhB solution was added again to continue the next cycle of photocatalytic experiments. Repeatability and photostability of NHC were studied by repeated experiments, where the initial concentration of RhB was always maintained at 10 mg L−1 throughout the application experiment. The concentration of RhB was monitored by UV-vis spectrophotometer (UV1901, Youke, Shanghai, China) at a wavelength of 553.5 nm.
Results and discussions
Process of preparation of NHC hydrogel
The preparation of photoactive thermosensitive NHC hybrid hydrogel involves a two-step electron beam radiation method. As shown in Figure 1, firstly, it is very important to obtain the NIPAAm preploymer (PNIPAAm) prepolymer with NIPAAm monomers by electron beam pre-radiation polymerization, and secondly, a certain amount of g-C3N4 is added to the mixed solution of HHPC and PNIPAAm prepolymer, so NHC hydrogel was prepared by an electron beam radiation cross-linking method. The advantage of a two-step radiation method is to ensure that the chain length of the PNIPAAm prepolymer reaches a certain length and cross-links with HHPC to form an interpenetrating network of hydrogel. Some hydrogel wafers with differing content of g-C3N4 are shown in Figure S1. The pure NIPAAm/HHPC is colorless and transparent. After the g-C3N4 is introduced, the color of NHC becomes deepened from transparency to light yellow, and deepens continuously with an increasing amount of g-C3N4.
1H NMR and 13C NMR analysis
The 1H NMR of NIPAAm prepolymer (25 kGy) with CDCl3 as solvent at 25°C is shown in Figure 2(a). The shape and width of the peaks indicate that the NIPAAm prepolymer has the characteristics of a macromolecular polymer. Specifically, the peak at 1.13 ppm (a) is attributed to the characteristic peak of the hydrogen atom of an isopropyl methyl group. The double peaks at 1.64 ppm (b) and at 2.12 ppm (c) are characteristic peaks of hydrogen atoms in methylene and methine, respectively. The peak at 4.00 ppm (d) corresponds to the characteristic peak of the hydrogen atom of an N-isopropyl group, but the characteristic peak (e) of the hydrogen atom of the secondary amine group is not found due to the occlusion of the characteristic peak of the hydrogen atom (4.00 ppm) on an N-isopropyl group. 25 Figure 2(b) showed the solid 13C NMR of NIPAAm prepolymer. The three main peaks near 24.5 ppm, 45 ppm, and 175 ppm are attributed to methyl carbon, methylene carbon, and carboxylic carbon in the main chain of the polymer, respectively. The above results confirmed further that the NIPAAm prepolymer was successfully prepared by electron beam irradiation polymerization.
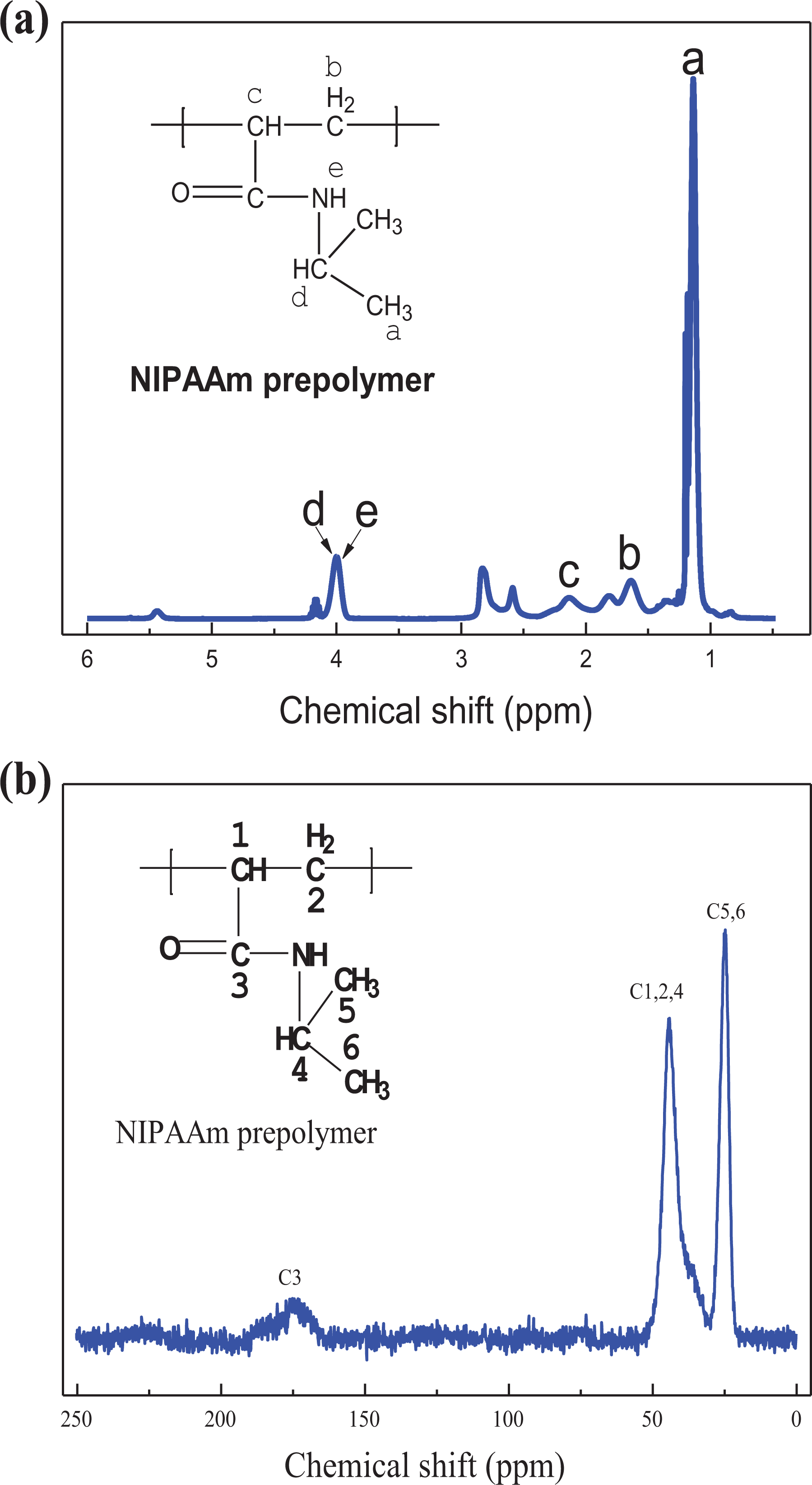
NMR spectra analysis of PNIPAAm prepolymer. (a) 1H NMR spectra of NIPAAm prepolymer at 25°C with CDCl3 solvent: peak a = hydrogen atom of isopropyl methyl group; peak b = hydrogen atoms of methylene; peak c = hydrogen atoms of methine; peak d = hydrogen atom of N-isopropyl group; peak e = hydrogen atom of secondary amine group. (b) 13C NMR spectra of NIPAAm prepolymer.
DSC and UV-vis transmission spectra experiments
LCST is an important temperature point which in temperature-sensitive polymers produces thermal deformation. Therefore, the LCST values and the phase transition behaviors for a number of different samples were measured by DSC. Figure 3 shows the DSC curves for different samples. It is obvious that pure HHPC does not possess the characteristics of thermosensitive polymers (no LCST) in the range from 30°C to 50°C, but NIPAAm/HHPC hydrogel does have the characteristic of thermosensitive polymers and demonstrates a fast response rate with an LCST of 35.5°C when the NIPAAm prepolymer is introduced. NHC still maintains the characteristics of a thermosensitive polymer after loading with g-C3N4. Compared with the NIPAAm/HHPC hydrogel carrier, the LCST of NHC is almost unchanged, however, the response rate is slightly slower. The response temperature of 35.5°C is very suitable for free diffusion of dye molecules within the hydrogel carrier, and for photocatalytic reactions the low LCST of NHC requires only a low heat to deform the carrier and ensure full utilization and recovery.
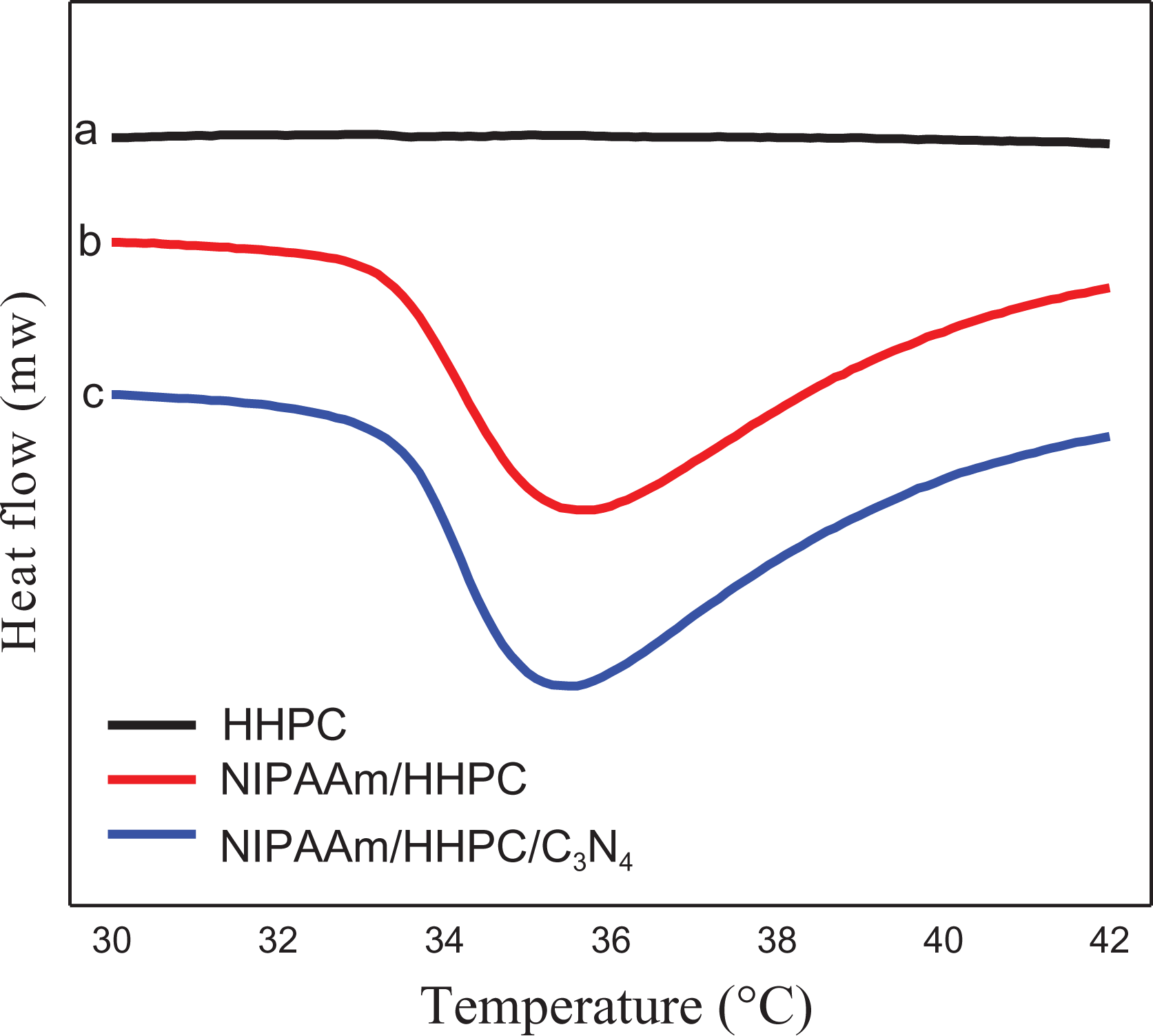
DSC curves of NHC hydrogel: (a) HHPC, (b) NIPAAm/HHPC, and (c) NHC.
Figure S2 shows the UV-vis light transmission spectra of pure NIPAAm/HHPC hydrogel film. It is clear that the film exhibits high permeability across the whole UV and visible region. This indicates that the NIPAAm/HHPC matrix does not affect the transport of light, making it an ideal carrier for photocatalysts, and affording the possibility for greater photocatalytic performance of NHC.
XRD characterization
XRD characterization for the different samples is shown in Figure 4 including the XRD spectrum of pure g-C3N4. The strong peak at 27.4° stems from the strong response due to aromatic interlayer stacking of g-C3N4 (in Figure 4), which can be indexed to the crystal plane (002) of C3N4, while the characteristic peak at 13.3°, corresponding to the (100) crystal plane, is the intra-surface structure peak of C3N4. Figure 4 also shows the XRD patterns of NHC-X% composite hydrogels with different loading amounts of g-C3N4. Except for broad and weak semicrystalline peak which appears near 8.5° in the diffraction zone of HHPC, the sharp diffraction peak around values of 20.0° indicates that the HHPC is partially crystallized, which is beneficial for the formation of more active free radicals and may increase the reaction rate of subsequent radiation cross-linking reactions when HHPC has been irradiated by the electron beam. There are no new characteristic diffraction peaks to be found after the cross-linking reaction between HHPC and NIPAAm prepolymer, but it is possible that the characteristic diffraction peak of the NIPAAm prepolymer (2θ = 19.7°) is obscured by the characteristic peak of HHPC. In addition, the strength of all the original peaks is weakened and the width of the half peak also is increased, indicating that the original crystallinity of HHPC has been damaged after the introduction of PNIPAAm by radiation cross-linked method. With the increase of g-C3N4, the weak characteristic peak of g-C3N4 appears on the NHC-X% hybrid hydrogel spectrum, indicating that g-C3N4 was uniformly dispersed on the framework of NIPAAm/HHPC hydrogel without agglomeration. The results showed that g-C3N4 nanosheets were successfully introduced into NIPAAm/HHPC hydrogel to ensure the photocatalytic activity of the NHC photocatalyst.
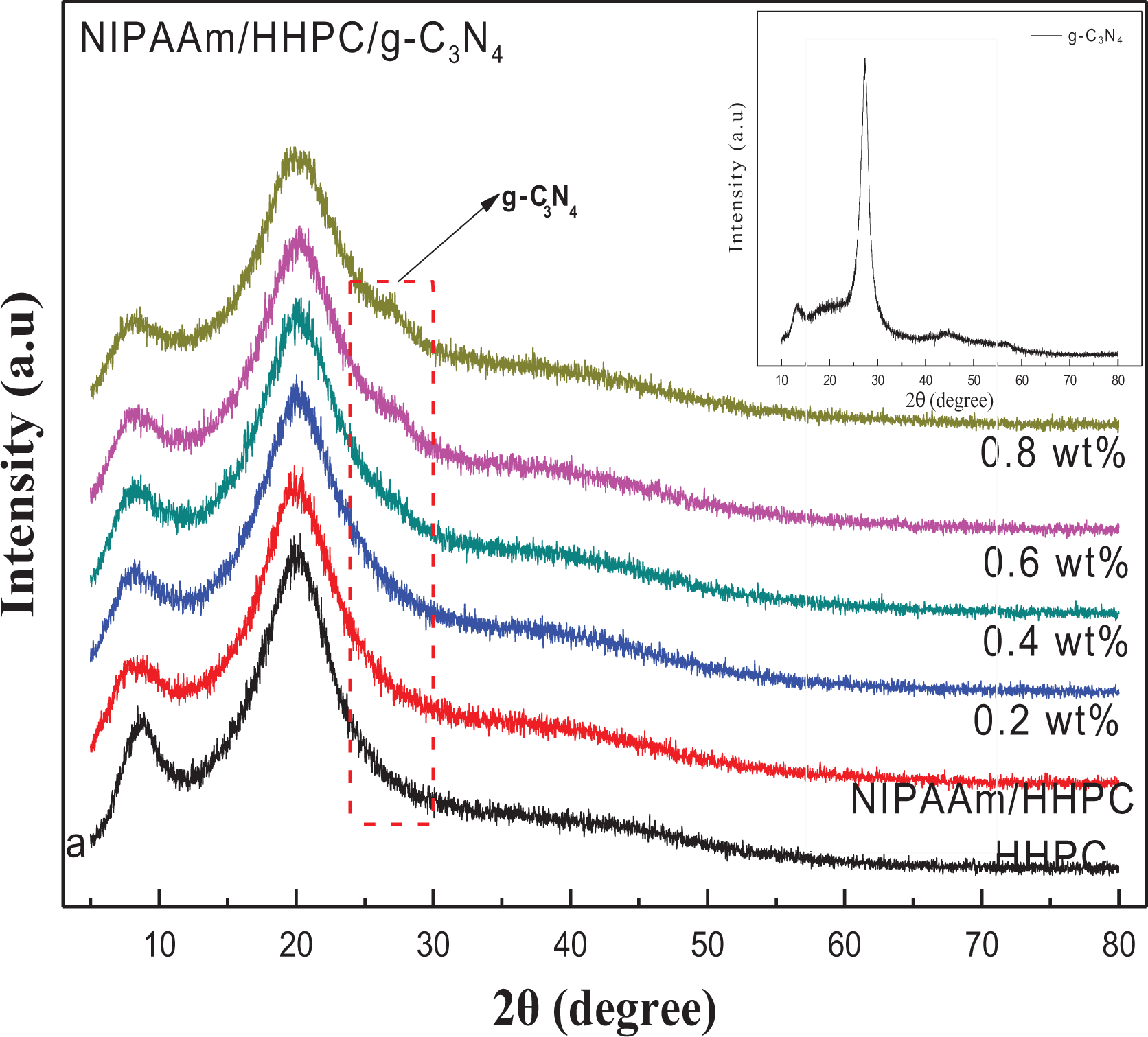
XRD patterns of different contents of g-C3N4 on NHC-X% hydrogels. XRD curves of different samples; inserted figure on XRD peak of g-C3N4.
FTIR spectra
The FTIR spectra of NHC hydrogel are shown in Figure 5. The main absorption band of g-C3N4 located at around 1000–1700 cm−1 corresponds to C–N and C=N stretching vibration peaks. Furthermore, it was confirmed that the appearance of a g-C3N4 peak at 800 cm−1 is a characteristic of outer plane bending vibration of the triazine ring. In the spectral curve of HHPC, the characteristic peak at 3453 cm−1 is attributed to the stretching vibration peak of –OH, but the weak characteristic peak at 1415 cm−1 belongs to –OH stretching vibration on the ring, and the aliphatic –CH3 deformation peak is visible at 1378 cm−1. It can be confirmed by the new peak at 1552 cm−1, corresponding to N–H bending vibration, and a C–N stretching vibration peak (amide II band) that NIPAAm and HHPC have been radiation cross-linked.26,27 In addition, the peak at 1661 cm−1 is indexed to the carbonyl stretching vibration peak of NIPAAm (C=O, amide I band). 28 It is obvious that the –OH stretching vibration peak and aliphatic –CH3 deformation peak on the ring have disappeared after the introduction of NIPAAm. The successful synthesis of NIPAAm/HHPC hydrogel can be confirmed by the formation of new chemical bonds and the retention of original groups.
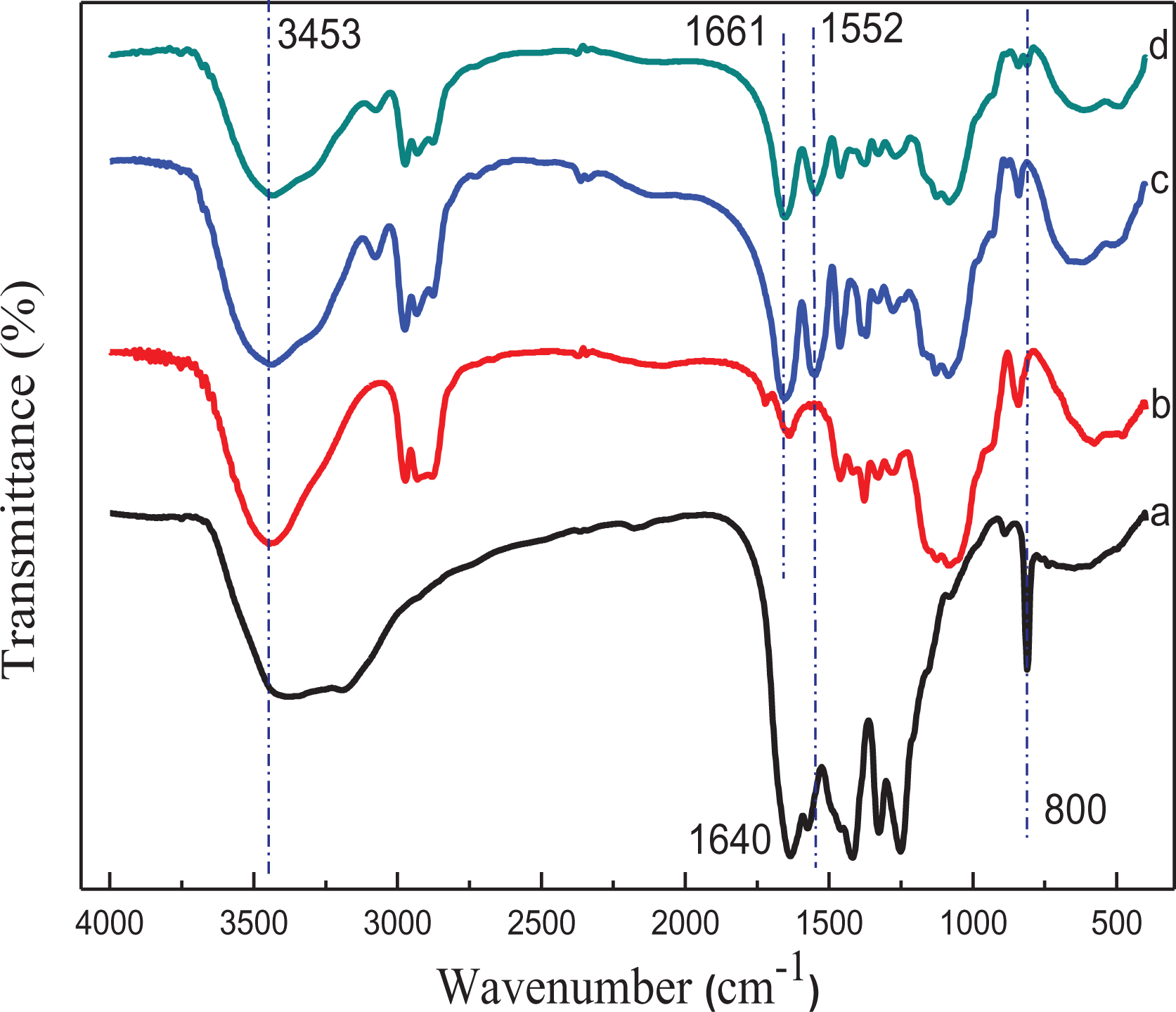
FTIR spectra of different hydrogels: (a) g-C3N4, (b) HHPC, (c) NIPAAm/HHPC, and (d) NHC.
TG analysis
Figure 6 shows the thermal analysis of dry composite hydrogel microspheres. The total weight loss is about 3% when the sample is slow heating from room temperature to 300°C, which is attributed to the evaporation of adsorbed water in the sample. However, the weight loss rate increased quickly between the range of 300°C and 420°C, indicating that the skeleton of the cross-linked NIPAAm/HHPC hydrogel was decomposed quickly and this accelerated with increasing temperature. The results show that the thermal stability of NHC hydrogel is slightly higher than that of single HHPC hydrogel, which may be due to the formation of a 3D (three-dimensional) interpenetrating network structure so as to raise the initial decomposition temperature of NHC hydrogel above that of pure HHPC hydrogel. The highly thermal stability of NHC hydrogel is very beneficial for thermally derived recycling and photocatalytic behaviors.
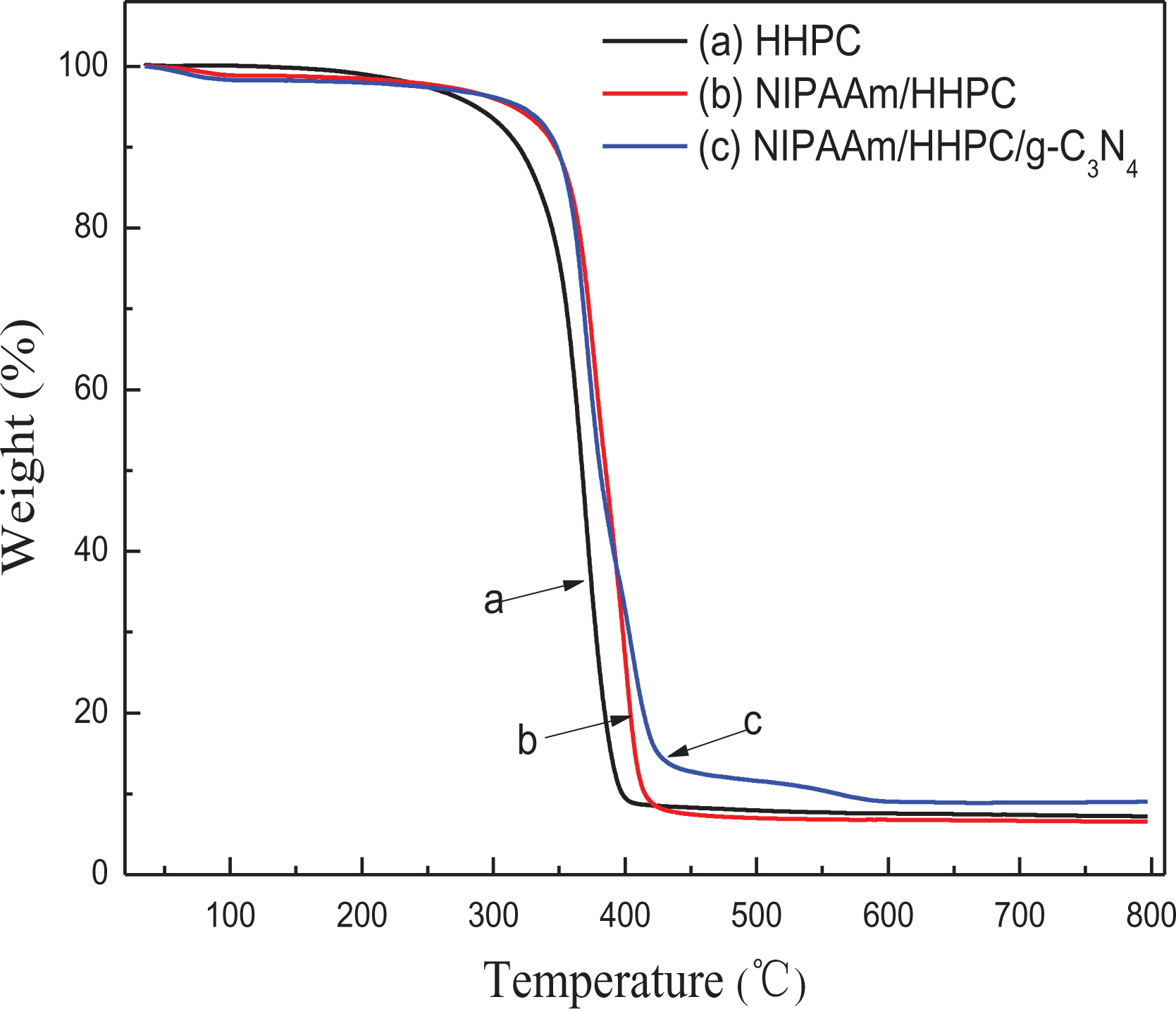
TG curves of different samples: (a) HHPC, (b) NIPAAm/HHPC, and (c) NHC.
Components analysis by XPS
XPS spectra were obtained after argon sputtering (i.e. only the bulk of the film was considered, and surface-related changes were avoided). The XPS analysis of NHC-0.8% hydrogel powder was performed for the composition analysis of the samples. The peak positions of different atoms were determined by internally referencing the adventitious carbon at a binding energy of 284.6 eV. The general scan spectrum of XPS (in Figure 7(a)) of annealed NHC-0.8% hydrogel at 100 eV for 30 min over a large energy range at low resolution shows sharp XPS peaks for. The characteristic peaks from C, N, and O atoms were apparent. The three main peaks with binding energy of 284.87 eV, 399.4 eV, and 532.2 eV were corresponded to the characteristic regions of C1s, N1s, and O1s, respectively. At the same time, the precise scanning spectra of C1s, N1s, and O1s on NHC-0.8% hydrogel are presented in Figure 7(b) to (d). Measured atomic concentrations, determined using XPS, of the annealed sample are shown in Table S1. As observed from Table S1, the binding energy peaks of C1s, N1s, and O1s on NHC are smaller than NIPAAm/HHPC (in Figure S3) in core level XPS spectrum, but the atomic contents of C1s and O1s on NHC are more higher than NIPAAm/HHPC. This result showed that g-C3N4 may be introduced into the NIPAAm/HHPC hydrogel.
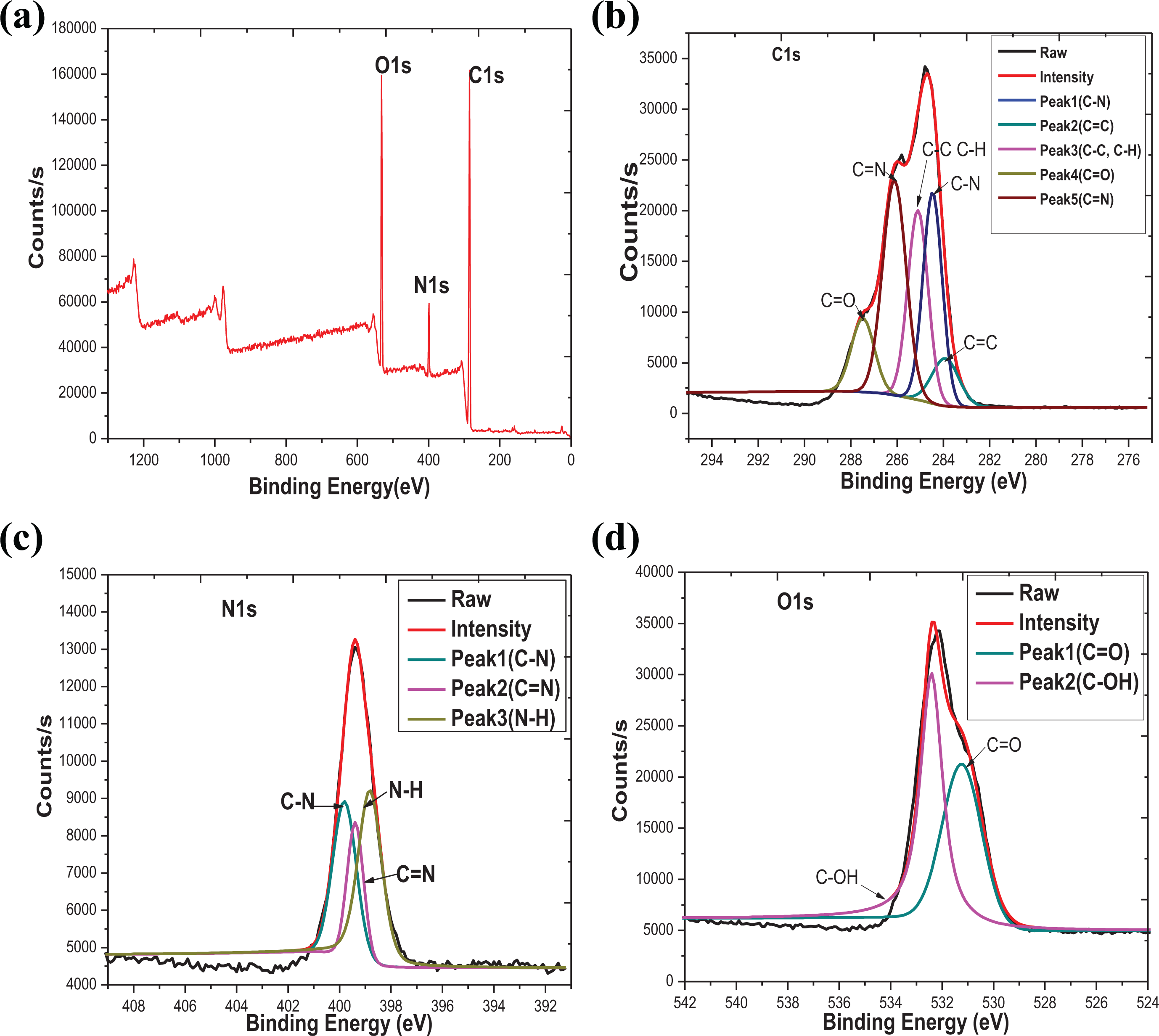
XPS spectra of NHC hydrogel: (a) XPS general spectra of NHC-08% hydrogel, (b) C1s, (c) N1s, and (d) O1s.
BET determination
The adsorption–desorption isotherms for NHC-0.8% hydrogel were determined by the BET gas adsorption method. As shown in Figure S4, the adsorption–desorption isotherm is an IV type isotherm of the H3 narrow stagflation ring, which indicates that the NHC-0.8% hydrogel has porous microstructures with open holes. This result is consistent with the SEM morphology analysis in Figure 9 below. In addition, the specific surface area of NHC-0.8% hydrogel was determined to be 15.8 m2 g−1, which is very beneficial for the subsequent adsorption and photocatalytic reactions.
UV-vis diffuse reflectance spectra of different samples.

HRTEM and FESEM images of different samples: (a) HRTEM image of g-C3N4 and (b to d) FESEM images of HHPC, NIPAAm/HHPC, and NHC.
Absorbance of NHC by diffuse reflectance spectrum
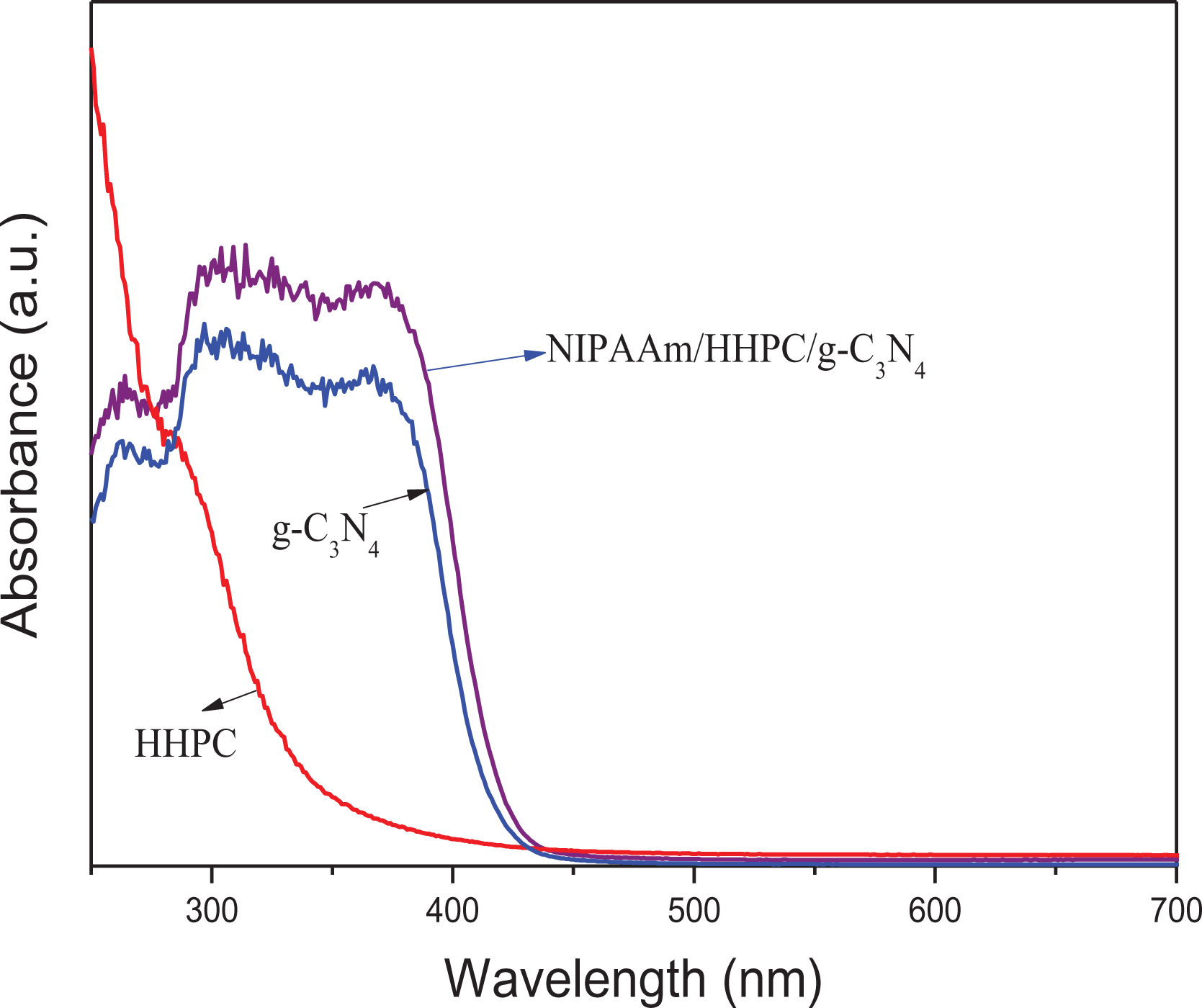
Figure 8 shows the UV-vis diffuse reflectance spectrum for various samples. Though the strong absorption of g-C3N4 obviously exhibits itself across the whole UV and visible regions, the optical response range of NHC-0.8% hydrogel is enlarged compared with pure HHPC and g-C3N4, the reason being the introduction of g-C3N4, and additionally the light absorption has been further improved by multiple light scattering on the special 3D structure of the NHC hydrogel. In short, the expansion of the range of light absorption and the increase of light absorption intensity mean that the highly photocatalytic activity of the NHC photocatalyst in a broader light responsive region can be achieved.
Morphologies
In order to detect the morphologies and microstructures of the g-C3N4 and NHC photocatalyst, several images of the selected location were acquired by TEM and SEM in Figure 9. Due to the production of NH3 and CO2 gases during the pyrolysis of urea, the TEM image of g-C3N4 displays a nanosheet structure which is thin and porous (shown in Figure 9(a)). At the same time, the SEM images of HHPC, NIPAAm/HHPC, and NHC are shown in Figure 9(b) to (d). It can be seen that the pore size of HHPC hydrogel is largest of the three hydrogels, with a porous network structure. NIPAAm/HHPC hydrogels show smaller and more regular pores than the NHC-X% hydrogel. After introducing g-C3N4, the large holes were arranged by disorderly stacking, which caused changes to the microstructure on NHC. Possible agglomerates of g-C3N4 nanosheets in NHC have also been discovered in Figure 9(d). This result is consistent with the above analysis of XRD, FTIR, and XPS.
Mechanical property testing
Generally, it is necessary for the catalyst carrier to have a certain mechanical strength. Therefore, the mechanical properties of the swelling equilibrium wet composite hydrogel were evaluated by simple compression tests. As shown in Figure S5, the NIPAAm/HHPC hydrogel demonstrated rupture phenomena when loaded with less than 300 g weight (in Figure S5(a)). However, the mechanical properties of the NHC hydrogel were significantly enhanced with the introduction of g-C3N4, because the structure of NHC remained intact under the same pressure from a 300 g weight (shown in Figure S5(b)). The above results indicated that g-C3N4 in NHC photocatalyst not only can act as the active sites of NHC but can also promote the cross-linked degrees and the stability of NHC as a cross-linking agent. 29
In order to study the effect loading the hydrogel with g-C3N4 on the physical and chemical properties of NHC hydrogel, the water content, swelling degree, and gel fraction of NHC hydrogel were determined by conventional weighing methods. As shown in Table 1, the water content and swelling degree were decreased slowly and the gel fraction increased slightly with increasing g-C3N4 content. In addition, it was found that different contents of g-C3N4 on NHC hydrogels display an excellent swelling degree and allow the free migration of dye molecules within the 3D network skeleton of the NHC hydrogel, which is beneficial to the mass transfer and the adsorption–degradation of water-soluble organic pollution in the hydrogel-based photocatalyst system.
Water content, swelling ratio, and gel fraction of different contents of g-C3N4 on NHC-X% hydrogel.
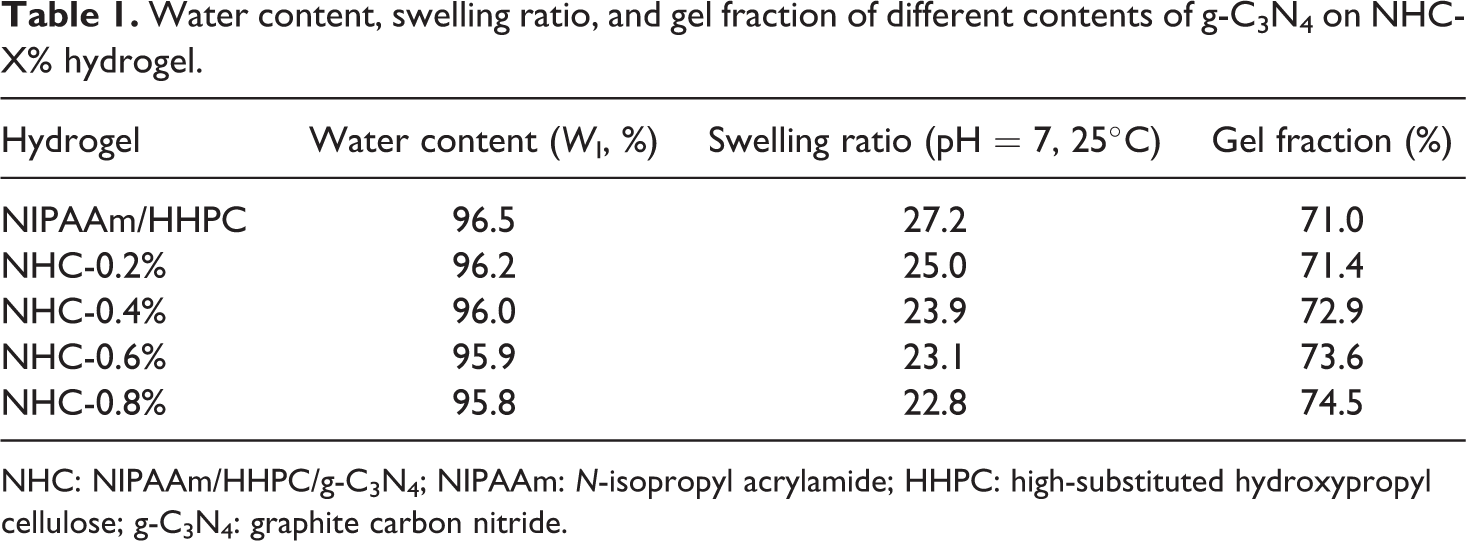
NHC: NIPAAm/HHPC/g-C3N4; NIPAAm: N-isopropyl acrylamide; HHPC: high-substituted hydroxypropyl cellulose; g-C3N4: graphite carbon nitride.
Thermally driven recycling-free experiment
The properties of thermally driven free-recycling of NHC smart hydrogel-based photocatalysts were measured by the thermal shrinkage of NHC-0.8% hydrogel films at different temperatures and different times. As shown in Figure 10 and Table 2, the circular hydrogel films shrank at a particular ratio after the heat treatment temperature was raised above the LCST of NHC hydrogel. At the same time, as the temperature of heat treatment is increased, the shrinkage ratio becomes greater. The thermal shrinkage ratio of a circular NHC hydrogel film was only 53.1% when treated for 5 min at low temperature (40°C). However, the thermal shrinkage ratio was increased greatly and reached 90.6% at 60°C for 5 min. The circular NHC hydrogel film was placed into a blast dryer at 80°C for 12 h, and the limited thermal shrinkage ratio was determined to be 96.4%. The results showed that the NHC-0.8% hydrogel can be shrunk rapidly at temperatures higher than its LCST with the water bath treatment, and the thermal shrinkage ratio is relatively similar at 60°C for differing times. Considering the adsorbed substrate can be squeezed from the 3D network of NHC hydrogel very economically, the thermal switching treatment temperature was set at 60°C for 6 min in the subsequent photocatalytic cycling experiments.

Photographs of thermal shrinkage characteristic on NHC-0.8% hydrogel at different temperatures and times: (a) NHC-08% hydrogel at room temperature, (b) NHC-0.8% hydrogel at 40°C for 5 min in water bath, (c) NHC-0.8% hydrogel at 60°C for 5 min in water bath, and (d) NHC-0.8% hydrogel at 60°C for 6 h in air dry oven.
The thermal shrinkage rate of NHC-0.8% hydrogel at different temperatures and times.
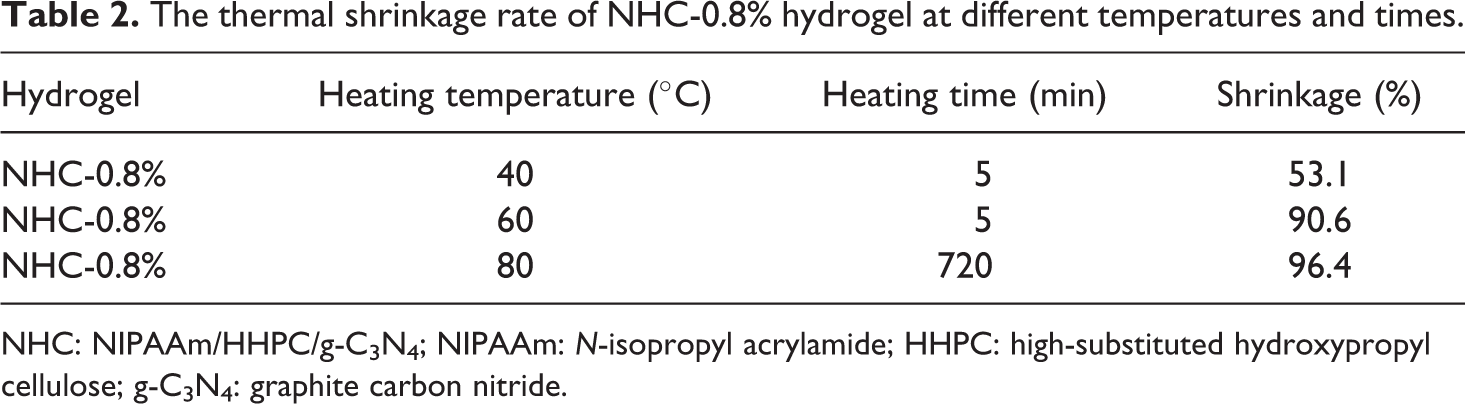
NHC: NIPAAm/HHPC/g-C3N4; NIPAAm: N-isopropyl acrylamide; HHPC: high-substituted hydroxypropyl cellulose; g-C3N4: graphite carbon nitride.
Adsorption–photocatalytic behaviors of NHC hydrogel
The effect of various loading amounts of g-C3N4 on the NHC hydrogel for adsorption–photocatalytic degradation experiments of RhB was studied at room temperature (25 ± 2°C). Figure 11(a) shows the removal efficiency of adsorption–degradation of RhB with differing contents of g-C3N4 on the NHC hydrogel while under visible light irradiation. It can be seen that RhB is difficult to self-degrade in NIPAAm/HHPC hydrogel without g-C3N4, and that the photocatalytic activity was improved with increasing content of g-C3N4, as shown from different contents of g-C3N4 on NHC hydrogel, the total removal rate of RhB dyes was reached at 71.4% loading on NHC-0.8% hydrogel.
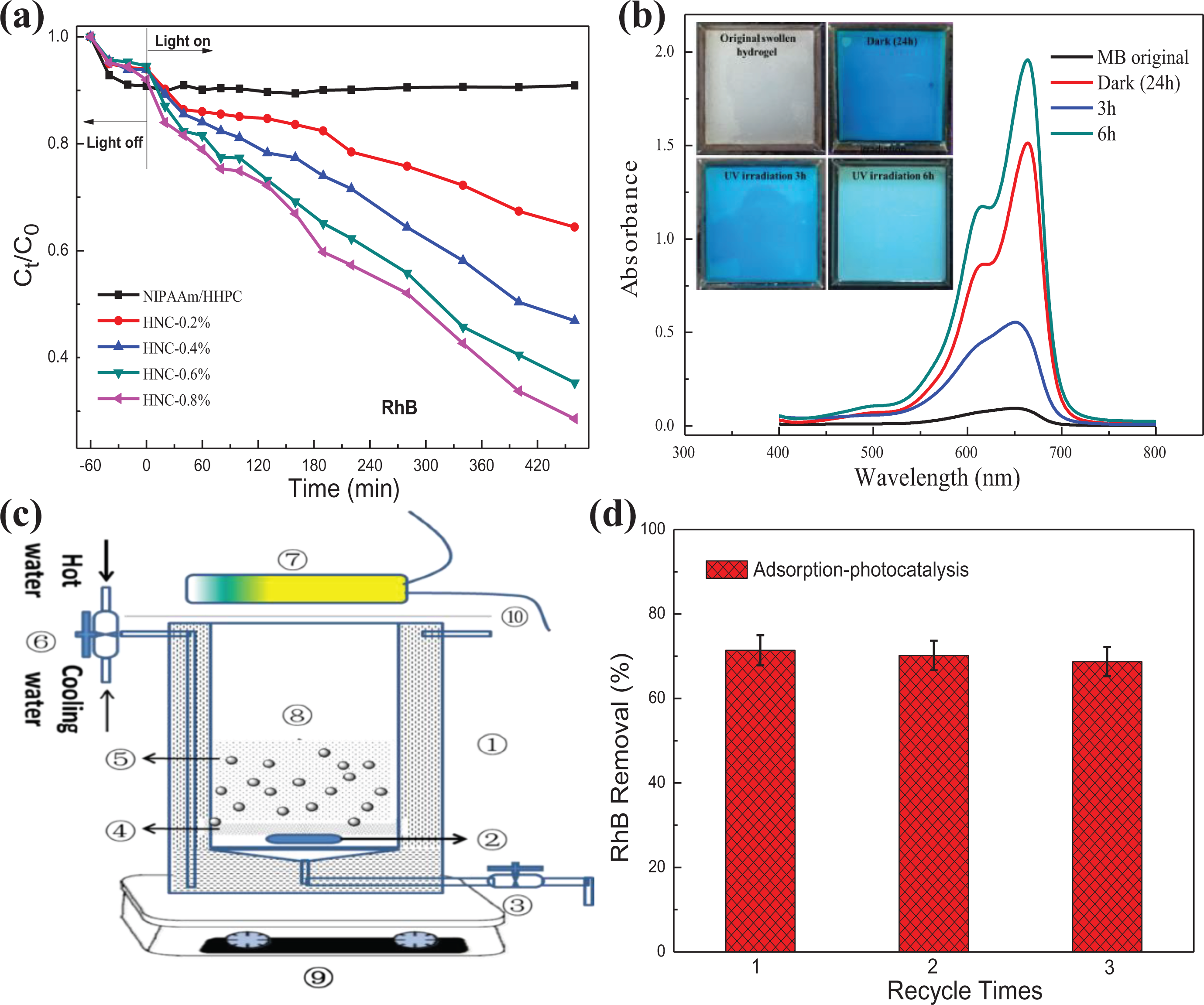
Adsorption–photocatalytic behaviors of NHC hydrogel: (a) adsorption–photocatalytic kinetic curves of RhB with different contents of g-C3N4 on NHC-X% hydrogel; (b) absorbance of adsorption–photocatalytic degradation of MB with NHC-0.8% hydrogel; (c) diagram of portable photocatalytic reactor (① sandwich quartz, ② magnet, ③ straight way valve, ④ isolation grid, ⑤ NHC-0.8% hydrogel, ⑥ three-way valve, ⑦ light source, ⑧ reaction solution, ⑨ magnetic stirring system); and (d) thermal driven recycling-free property of adsorption–photocatalytic degradation of RhB with NHC-0.8% hydrogel.
The properties of adsorption–photocatalytic degradation of NHC-0.8% hydrogel on different dyes are explored by UV-vis spectra in Figure 11(b). The experimental procedure was as follows: 100 mL methylene blue (MB) (10 mg L−1) was injected into a square NHC hydrogel film (8.5 × 8.5 × 0.3 cm3), MB was kept in a non-photostatic state for 24 h in order to ensure the adsorption–desorption equilibrium, and the color of NHC-0.8% hydrogel turned blue after desorption equilibrium. Here, the color of NHC-0.8% hydrogel is changed to light blue after 100 W UV lamp irradiation for 3 and 6 h. Specifically, the characteristic peak value of MB decreased gradually with increasing irradiation time.
Considering the thermal shrinkage characteristics of NHC-0.8% hydrogel catalyst, a portable photocatalytic reactor was designed to verify further the properties of thermally driven free-recycling on NHC. As shown in Figure 11(c), a grid (200 mesh) is used to prevent collision between the magnets and NHC hydrogel catalysts, and avoid mass loss of NHC hydrogel catalyst. After each experiment, the adsorbed matrix of NHC hydrogel can be extruded by the behavior of heating shrinkage when the recycling water is switched through a three-way valve and the conditions maintained for 5 min at 60°C. Then, the reaction liquid and NHC hydrogel catalyst were separated automatically by the action of potential energy, and then fresh reaction solution is added, and the next cycle of the catalytic reaction is carried out. Although the adsorption–photocatalytic removal rate of RhB on NHC-0.8% hydrogel was decreased slightly from 71.4% to 68.7% after the third cycle (in Figure 11(d)), the removal capacity can still be maintained at a high level. This result further supports the conclusion that thermally driven recycling of NHC hydrogel is highly feasible.
Photostability of NHC hydrogel
The photostability of NHC-0.8% hydrogel is investigated and the results are shown in Figure 12. The experimental verification steps are as follows: 0.2 g NHC-0.8% xerogel film (diameter 1 cm) was placed in a quartz tube containing 50 mL ultrapure water and then irradiated with UV light (mercury lamp, GGZ100) after the film had completed adsorption and swelled up for 24 h. Immediately, the hydrogel film was removed, dried, and weighed at given intervals. The photostability of NHC-0.8% hydrogel can be evaluated by the weight loss rate of hydrogel before and after irradiation and the UV-vis absorption spectra of the reaction solution. As shown in Figure 12(a), compared with the weight loss rate of NIPAAm/HHPC hydrogel, the weight loss rate of NHC-0.8% hydrogel is very small. Rapid weight loss may be mainly due to the overflow of the unbonded HHPC and NIPAAm prepolymer on the surface of the hydrogel skeleton within the first 20 h, instead of hydrogels lost by photocatalytic decomposition. Although NHC-0.8% hydrogel can also be degraded by UV light, the degradation rate is very low, with no obvious degradation product of NHC-0.8% hydrogel being observed after UV irradiation for 60 h (shown in Figure 12(b)). The above results indicated that NHC-0.8% hydrogel can be used as the carrier of visible light photocatalysts (such as g-C3N4) and is expected to be applicable in wastewater treatment.
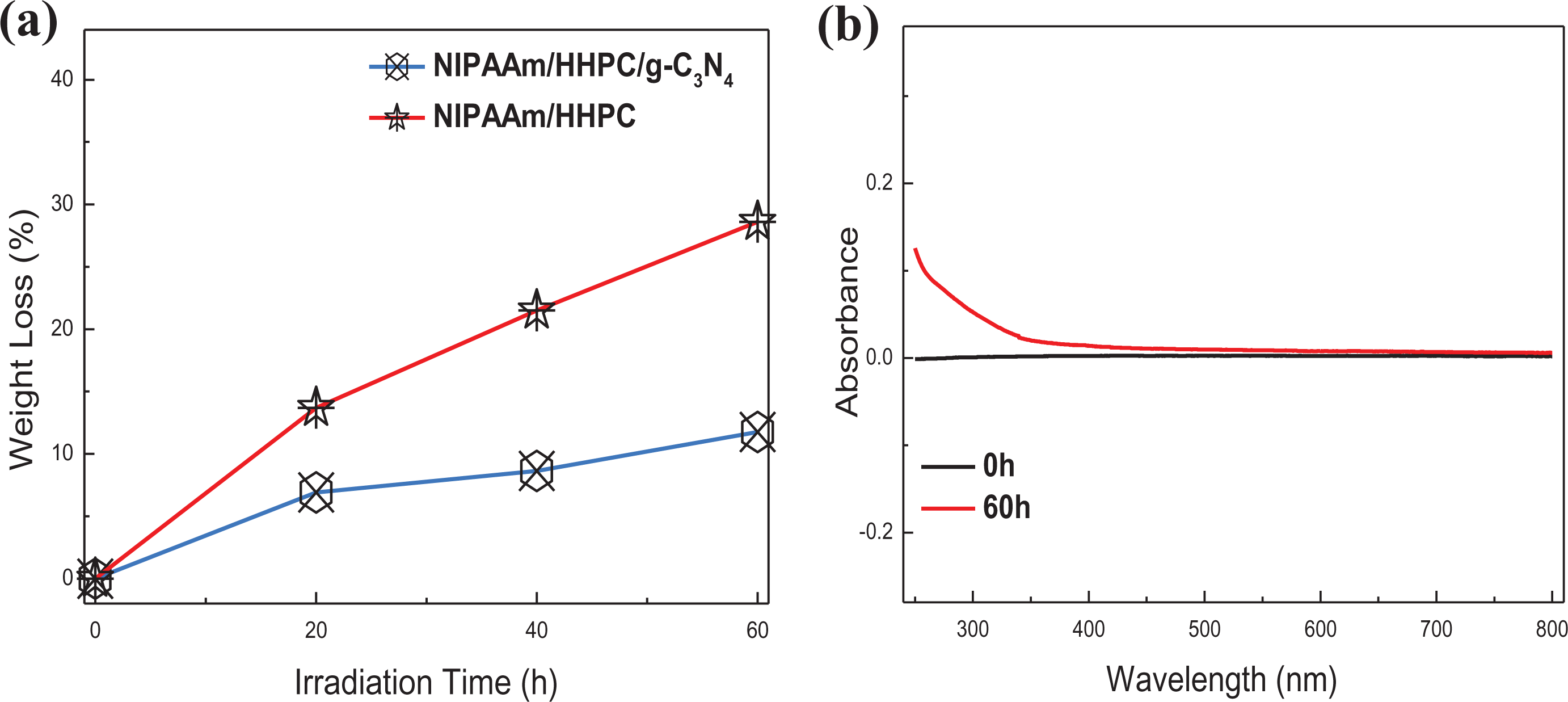
Photostability of NHC hydrogel: (a) the weight loss rate of different hydrogels under 100 W UV lamp irradiation and (b) absorbance of degradation product of NHC-0.8% hydrogel under 100 W UV lamp irradiation for 60 h.
Mechanism of photocatalytic and thermal driven recycling-free on NHC hydrogel
From the above characterizations, the possible mechanism can be deduced and shown in Figure 13. Under visible light irradiation, both RhB dye and g-C3N4 can be excited (corresponding to formulas (1) and (2), respectively), excitation of the adsorbed RhB dye takes place by light to the appropriate singlet or triplet states, the generated electrons in RhB are then immigrated to the conduction band (CB) of g-C3N4, while the dye is converted into a cationic RhB+ dye radical (formula (3)).
30
This transferring process is thermodynamically favorable because both the CB and valence band (VB) of RhB dye lie above that of g-C3N4. The lifetime of the excited electrons (e−) and holes (h+) is prolonged in the transfer process, inducing higher quantum efficiency. Meanwhile, the generated electrons of g-C3N4 probably react with dissolved oxygen molecules and produce oxygen peroxide radical
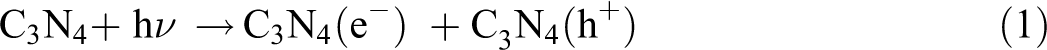

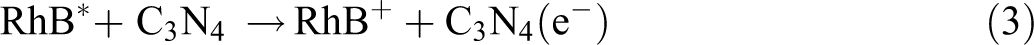
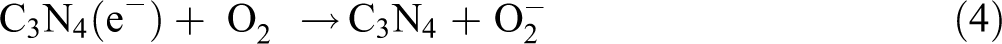
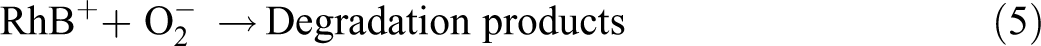
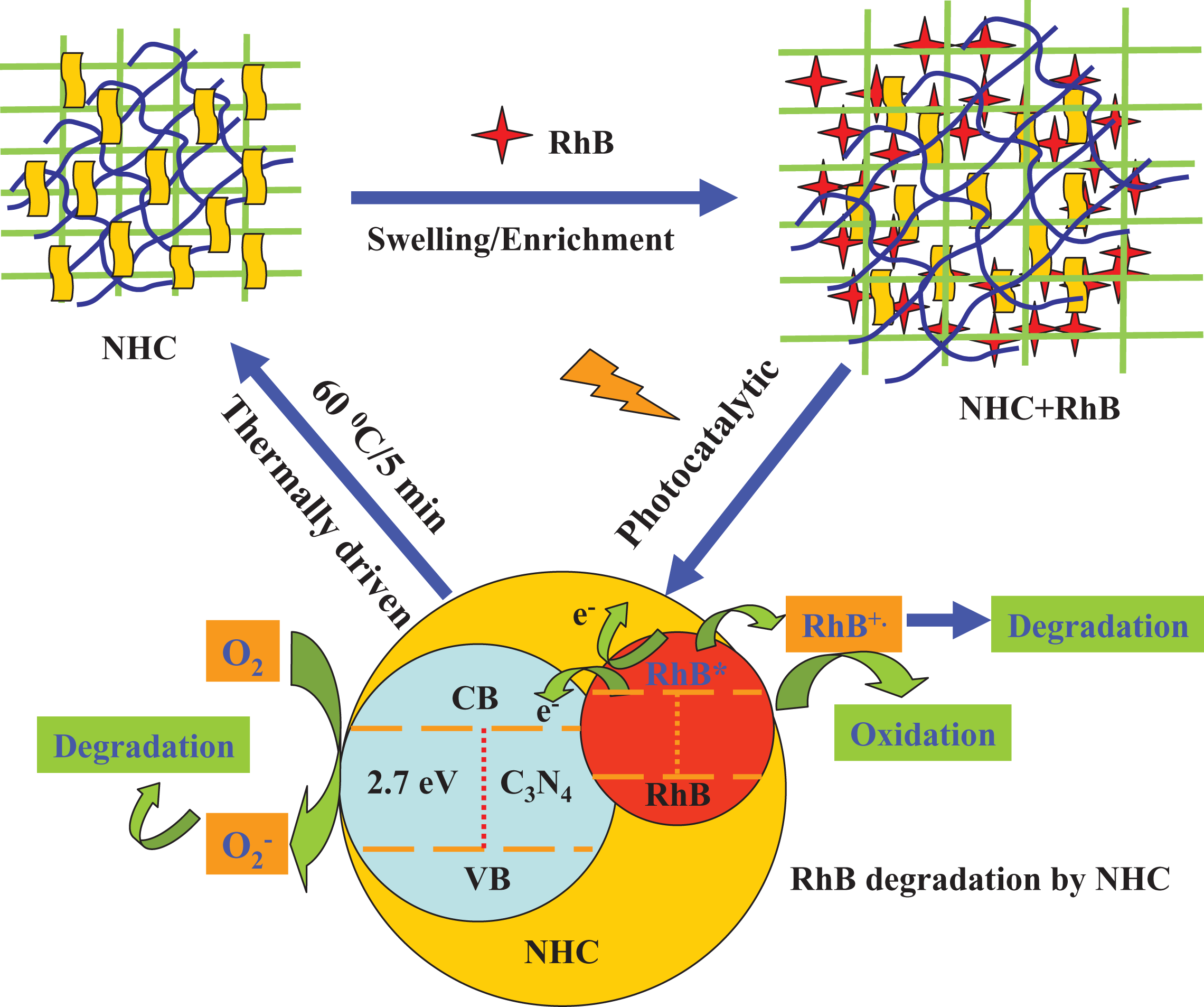
Mechanism of thermally driven recycling-free and photocatalytic performances on NHC hydrogel.
In a word, dye ions have played two functions: first, RhB dye ions were adsorbed and enriched onto the surface of the g-C3N4 nanoparticles in the 3D network nanostructure of NHC hydrogel, owing to the electrostatic interaction, and additionally the high specific surface area and porosity enhanced the contact area with RhB dye. On the other hand, RhB dyes were removed effectively by this NHC hydrogel portable photocatalyst system and realized the synergistic effect of adsorption and photocatalysis. 33
Because NHC hydrogel has excellent swelling degrees and thermal shrinkage rate, which is beneficial to the mass transfer and the adsorption–photocatalytic degradation of water-soluble organic pollution in the hydrogel-based photocatalysts system, 34 so as to the photogenerated holes on the VB of g-C3N4 could be transferred out through the 3D network nanostructure of NHC hydrogel, which improves the separation efficiency of electrons and holes and enhances the photocatalytic activity. 35 In addition, the excellent adsorption–photocatalytic activity and 3D network structure of NHC hydrogel is convenient for separation and recycling, which is also beneficial to water purification in the flow systems.36,37
Conclusions
NHC smart hydrogel-based photocatalyst with thermally driven and free-recycling properties was prepared successfully by electron beam pre-radiation polymerization and radiation cross-linking methods. g-C3N4 nanosheets were uniformly dispersed into the skeleton of the thermosensitive NIPAAm/HHPC hydrogel, and ensured the high photoactivity of the NHC hydrogel. The composition and physical and chemical properties of NHC hydrogel were characterized by NMR, FTIR, DSC, TG, XRD, XPS, and DRS. The microstructure of NHC hydrogel was characterized further by SEM, TEM, and BET. The adsorption–photocatalytic removal rate of RhB dyes on NHC-0.8% hydrogel was observed to reach 71.4% in aqueous medium under visible light. The thermal shrinkage ratio was observed to reach 90.6% at 60°C for 5 min, and effectively achieved free-recycling behavior in the portable photocatalytic reaction device under optimal conditions. The separation efficiency of electrons and holes can be improved and the photocatalytic activity of NHC hydrogel can be enhanced. In addition, the excellent adsorption–photocatalytic activity and 3D network structure of NHC hydrogel is helpful for separation and recycling-free. These thermally driven free-recycling characteristics, and the highly photocatalytic properties of the hybrid hydrogel-based photocatalyst, show that it can be used as a promising new material with extensive applications in wastewater treatment.
Supplemental material
Supplemental Material, Supporting_Information - Thermally driven characteristic and highly photocatalytic activity based on N-isopropyl acrylamide/high-substituted hydroxypropyl cellulose/g-C3N4 hydrogel by electron beam pre-radiation method
Supplemental Material, Supporting_Information for Thermally driven characteristic and highly photocatalytic activity based on N-isopropyl acrylamide/high-substituted hydroxypropyl cellulose/g-C3N4 hydrogel by electron beam pre-radiation method by Guo Liu, Ting-Ting Li, Xiao-Fang Song, Jin-Yu Yang, Jiang-Tao Qin, Fang-Fang Zhang, Zheng-Xi Wang, Hong-Guo Chen, Ming-Hu Wu and Yue-Sheng Li in Journal of Thermoplastic Composite Materials
Footnotes
Authors’ note
Guo Liu and Ting-Ting Li are the co-first authors.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (no. 11405050 and 21302150), the Project of the Science and Technology Department of Hubei Province and Xianning City (no. 2018AFB150 and 2019kj02), the Project of the Education Department of Hubei Province (no. S201910927021), the Science Development Foundation of Hubei University of Science & Technology (no. 2018-19KZ05 and 2020TD01), and the Nuclear Technology Key Subject Project of Hubei University of Science & Technology (no. 2019-20KZ01 and H2019005).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
