Abstract
The thermal degradation of the polyamide 6,6 (abbreviated henceforth as PA 6,6) reinforced with different concentrations of carbon nanotubes (CNTs) was investigated by means of thermal analysis. In this study, the nanostructured composites were produced using 0.1, 0.5 and 1.0 wt% of CNT. X-ray diffraction analyses were performed in order to evaluate the crystallographic properties of nanostructured composite. The degradation kinetics of PA 6,6/CNT nanostructured composites were measured by thermogravimetric analysis at different heating rates under nitrogen flow. TGA experiments were performed to elucidate the thermal behavior and supply the data that characterize the degradation kinetic. The degradation parameter kinetics was determined using the Ozawa–Wall–Flynn (O-W-F) methods, which do not require knowledge of the reaction mechanism. In this work, the results show that the addition of CNT up to the amount of 0.5 wt% increases the thermal stability of PA 6,6.
Introduction
The study of thermal degradation of polymers is of major interest and it is very important in determining the maximum operating temperature for a material. A thorough understanding of degradation phenomena would allow prediction of the extent and nature of degradation under various process conditions and would facilitate reduction and/or prevention of degradation. Several researchers have employed various approaches and analytical techniques in attempts to elucidate the nature and kinetics of the degradation reactions under a variety of conditions. 1 –4
Specifically for polyamide 6,6 (PA 6,6), the thermal degradation does not lead to production of starting monomers, not occurring depolymerization. In contrast, it is known that the process is complex, involving hydrolysis, decarboxylation, deamination and dehydration, with the liberation of H2O, NH3, CO2, cyclopentanone, adipic acid, hexadiamine, nitrile and olefin in general. 5 –10
Fabrication and characterization of CNTs/PA composites have received increasing interest in these last years. In the polymeric matrices, the addition of CNTs in small amounts (between 0.1 and 2%) usually results in an improvement in physical and electrical properties of the material, since the dispersion of CNTs in polymer matrix is carried out effectively. 11 –17 In PA/CNT nanostructured composites, the moisture absorption may also be affected by the presence of CNTs, resulting in the reduced water absorption of nanostructured composites, compared to neat PA. Probably, the CNT blocks the penetration of water molecules, decreasing the moisture absorption. 18
According to the literature, 18,19 the addition of functionalized CNTs in quantities not more than 0.5 wt%, usually leads to inadequate dispersion in PA 6,6, when used in the mixing process. This fact can be explained by nonpolar nature of the nonfunctionalized CNTs, creating an incompatibility in PA 6,6, which has a polar character. The effect of an inadequate dispersion affects the degradation and activation energy of the nanostructured composites. 19 The literature 19 also report that during the degradation process of PA 6,6, the activation energy decreases when the CNTs are added in quantities exceeding 0.5 wt%.
The kinetic analysis of a decomposing material can be done using two different techniques: the dynamic and the isothermal thermogravimetric studies. The dynamic thermogravimetric data can be analyzed using two different methods, the isoconversional and the discrimination methods. The isoconversional methods use the data from a series of thermogravimetric (thermogravimetric analysis (TGA)) curves obtained at different heating rates to calculate the energy of activation (E a). 20,21 This method was used in the present work.
The present study focuses on the study of the effect of addition of the different amounts of carbon nanotubes (0.1, 0.5 and 1.0 wt% CNTs) on the degradation kinetics of the PA 6,6 polymer matrix. The nonoxidative thermal degradation kinetic of nanostructured composites was performed using the TGA in nonisothermal conditions.
Theoretical considerations
In the thermal decomposition of polymer, some kinetic parameters should be obtained to better understand the degradation process. The polymer conversion degree (α) can be calculated using TGA, which will result in obtaining important parameters for the kinetic process, such as activation energy (E
a) and order of reaction (n).
22
The reaction rate in TGA studies can be defined as the variation in the degree of conversion (a) with time or temperature and the conversion is typically calculated as follows
23
where w 0, w t and w f are, respectively, weight at the beginning of the degradation step, actual weight at each point of the curve and the final weight measured after the specific degradation process considered.
All kinetic studies assume that the isothermal rate of conversion, dα/dt, is a linear function of a temperature-dependent rate constant, k(T), and a temperature-independent function of the conversion, f(α), that is
24
where f(α) depends on the mechanism of the degradation reaction.
According to the Arrhenius equation, the constant k can be calculated as
where A is the preexponential factor (min−1); E a is the activation energy; R is the universal gas constant (8.314 J mol−1 K−1) and T is the absolute temperature (K). From this equation, it is observed that the reaction rate increases exponentially with increasing temperature.
Combining equations (2) and (3), equation (4) can be obtained
for nonisothermal conditions, at constant heating rate (β), β = dT/dt and equation (4) can be rewritten as
In the degradation of polymeric matrices, the conversion rate is proportional to the concentration that will react or decompose
24
Combining f(α) in equation (5) gives
From equation (7) several methods have been developed for the study of the thermal degradation kinetics for polymeric materials, and these methods allow obtaining parameters that can describe this process. 24
Ozawa–Wall–Flynn model
The Ozawa–Wall–Flynn (O-W-F) model is relatively simple and allows determining the activation energy from nonisothermal thermogravimetric data, obtained at different heating rates, without a previous knowledge of the fraction of mass conversion.
25
In this case, equation (4) can be rearranged and written as follows
Considering
Applying the logarithm, the follow equation can be obtained
For
Finally, the O-W-F model is defined by mathematically combining equation (10) with equation (11) to result in equation (12), as follows
For different heating rates (β), the activation energy can be determined from the plot of ln β versus 1/T, to be generated by a straight line. From the inclination of this line,
Finally, knowing the activation energy involved in the process, the half-life time for a fixed conversion degree in relation to temperature, can be determined by equation (13)
where t f is the lifetime of the material for a temperature T f and for a fraction of material decomposed, α is a tabulated value and dependent on Eα and T f and β is the heating rate near to the central heating rates. 27,28
Experimental
Materials
In this study, the polymer matrix used for obtaining nanostructured composite was PA 6,6 provided by the Rhodia Company (Brazil). The multiwalled CNT (Baytubes, C150 P—MWCNTs) used in this investigation have been supplied by Bayer Company (Germany). They are characterized by an average diameter of 13–16 nm, number of walls 3–15 and bulk density of 140–160 kg/m3. The manufacturer indicates that the CNTs contain less than 5% impurities, including residual catalyst. For dissolving the PA 6,6 and subsequent dispersion of CNTs, formic acid (CH2O2; 85%) was used as the solvent supplied by VETEC Química Fina LTDA company in order to obtain the PA 6,6/CNT nanostructured composites.
The solution mixing technique was used for the preparation of PA 6,6/CNT nanostructured composites, which is currently considered as the most used processing when CNTs are aggregated loads of very small dimensions in polymer matrices. 29,30 The process of obtaining nanostructured composites, using the solution mixing, consists of three steps: (a) dispersion of nanotubes in formic acid (PA 6,6 solvent); (b) mixture of this dispersion in a solution of PA 6,6 dissolved in formic acid (room temperature); and (c) recovery of nanostructured composite by solvent evaporation (casting) resulting in a PA 6,6/CNT film. The CNT dispersion in formic acid was performed with the assistance of an ultrasonic probe (Sonics & Materials, Model VC 750, USA). Using this methodology, the following films were produced in the CNT concentrations of 0.1, 0.5 and 1.0 wt%.
Morphological analysis
Wide angle X-ray diffraction (WAXD) was performed using Philips Xpert PRO 3060 at 40 kV and 45 mA. X-rays diffraction analyses were performed in order to determine the crystallographic properties of the PA 6,6/CNT nanostructured composite.
Thermal analysis
The TGA was performed with TG/DTA 6200, model EXSTAR6000 of SII Nanotechnology operated in the dynamic mode at different heating rates. In the thermal decomposition analysis of the nanostructured composites about 6.0 mg platinum pan and alumina were used as the reference materials. The samples were heated at the rates of 2.5, 5.0, 10.0, 15.0 and 20.0°C min−1 under constant nitrogen flow of 100 ml min−1 and temperature range between 25 and 800°C. For each sample, three tests were carried out under the same heating rate and the temperatures were reproducible at ±3.0°C. The values of maximum decomposition rate temperature (T max) and mean activation energy (E) reported in the following tables are the average of three values. In this study, the O-W-F method was used and the following conversion values 5, 7.5, 10, 12.5 and 15% were adopted to solve the O-W-F equation.
Results and discussion
Morphological analysis—WAXD
The WAXD technique is nondestructive and it is used to obtain information about distance between interlayer, structural tensile and sample purity. However, such as the CNTs standard diffraction mainly characteristics are very close of graphite, X-rays diffraction profile is not useful to distinguish microstructurals details among CNT and graphite structures. But the WAXD technique is useful to verify whether the nanotubes were incorporated in polymeric matrix by the increase in peak amplitude (0 0 2).
The standard X-ray diffraction of the nanotubes is similar to that of graphite, the peak (0 0 2) and distance of interlayers can be obtained by its position using the Bragg’s law. The intensity and the width of this peak (0 0 2) are related to the number of layers, change in the distance of interlayer, content of nanotubes, distortion in the network and nanotubes orientation. Besides the peak (0 0 2), a family of peaks (h k 0), due to honeycomb network of leaf of graphene, is found in the nanotubes diffraction standard. These peaks (h k 0) show an asymmetric shape due to the balance of nanotubes and only the reflections (h k l) are found in the diffraction standards with normal layers. 31
The X-ray diffractogram obtained for the PA 6,6/CNT nanostructured composites is shown in Figure.1. In this curve, two distinct peaks can be observed at 2θ = 20° and 2θ = 25°, which are consistent with the crystal planes (0 0 2) and (1 0 0, 1 0 1), respectively. The increase in the concentration of CNTs in the samples increases the peak amplitude (0 0 2). This fact suggests the CNTs were incorporated in progressive way into PA 6,6 matrix, that is the increase in percentage (w/w) of CNT favors a proportional increase in the peak amplitude at 20° ((0 0 2) planes).
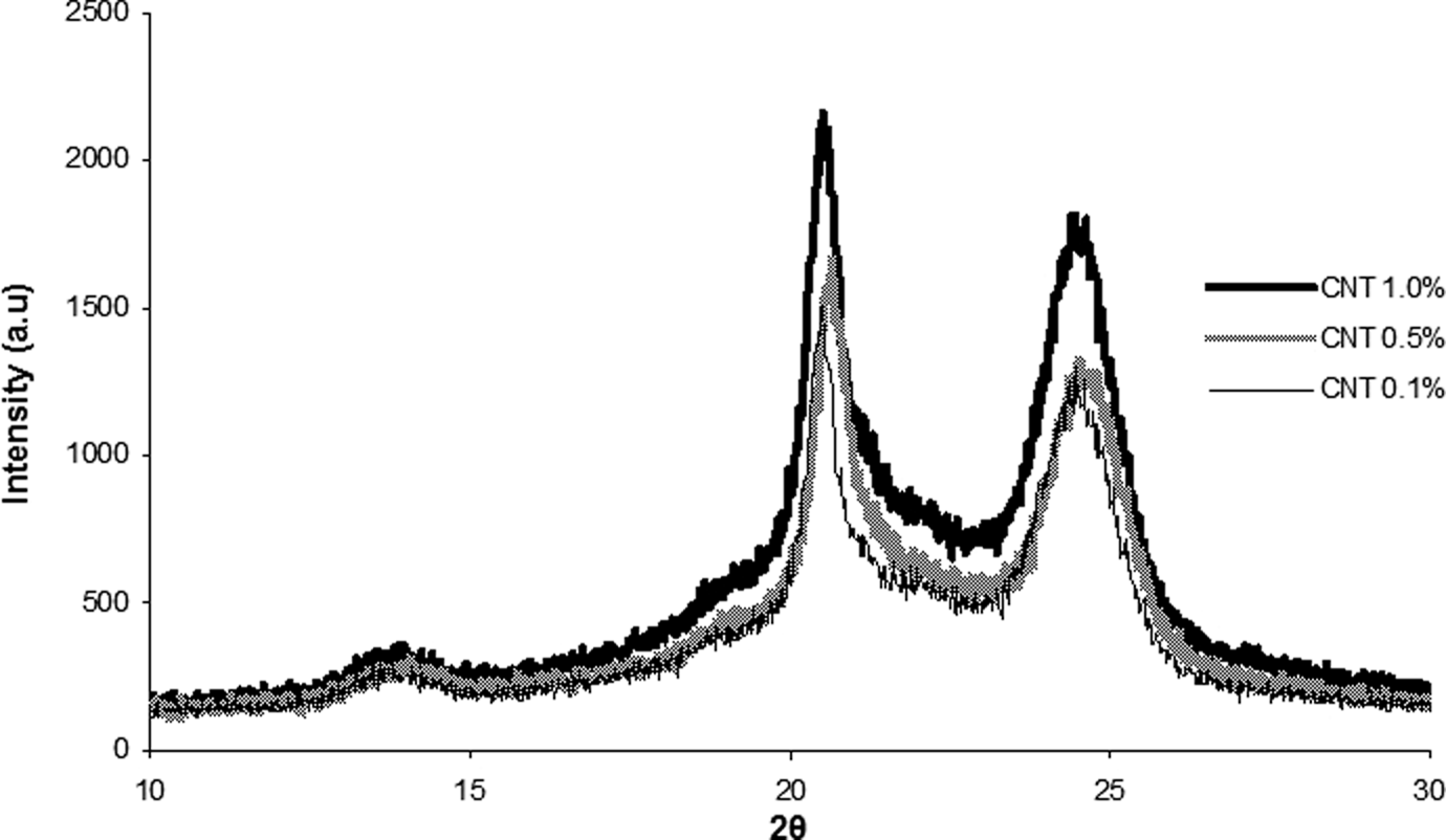
Diffractograms of PA 6.6/CNT nanostructured composites. CNT: carbon nanotube; PA: polyamide.
Thermal analysis
The thermal decomposition study of PA 6,6 and its nanostructured composites is in general complex, which can be evaluated using a kinetic model. Nowadays, there are several models that explain the thermal decomposition of solids based on different theoretical concepts and empirical studies. In this study, the integral model of O-W-F was chosen, because it is a widely used method in the thermal decomposition of polymeric materials and do not require the knowledge of the reaction mechanism. 24 –26
According to the literature, 31 the thermal degradation of PA 6,6 involves a β-C-H transfer reaction mechanism. The primary products for the unsubstituted PAs are ketoamides, which undergo further decomposition to form nitriles (Figure.2). This mechanism results in the degradation of dimethyl substituted PAs. The primary products are ketoamides via the same hydrogen transfer mechanism. No nitriles were isolated from the substituted PAs because they are precluded from forming nitriles due to the methyl substitutes on the nitrogen.
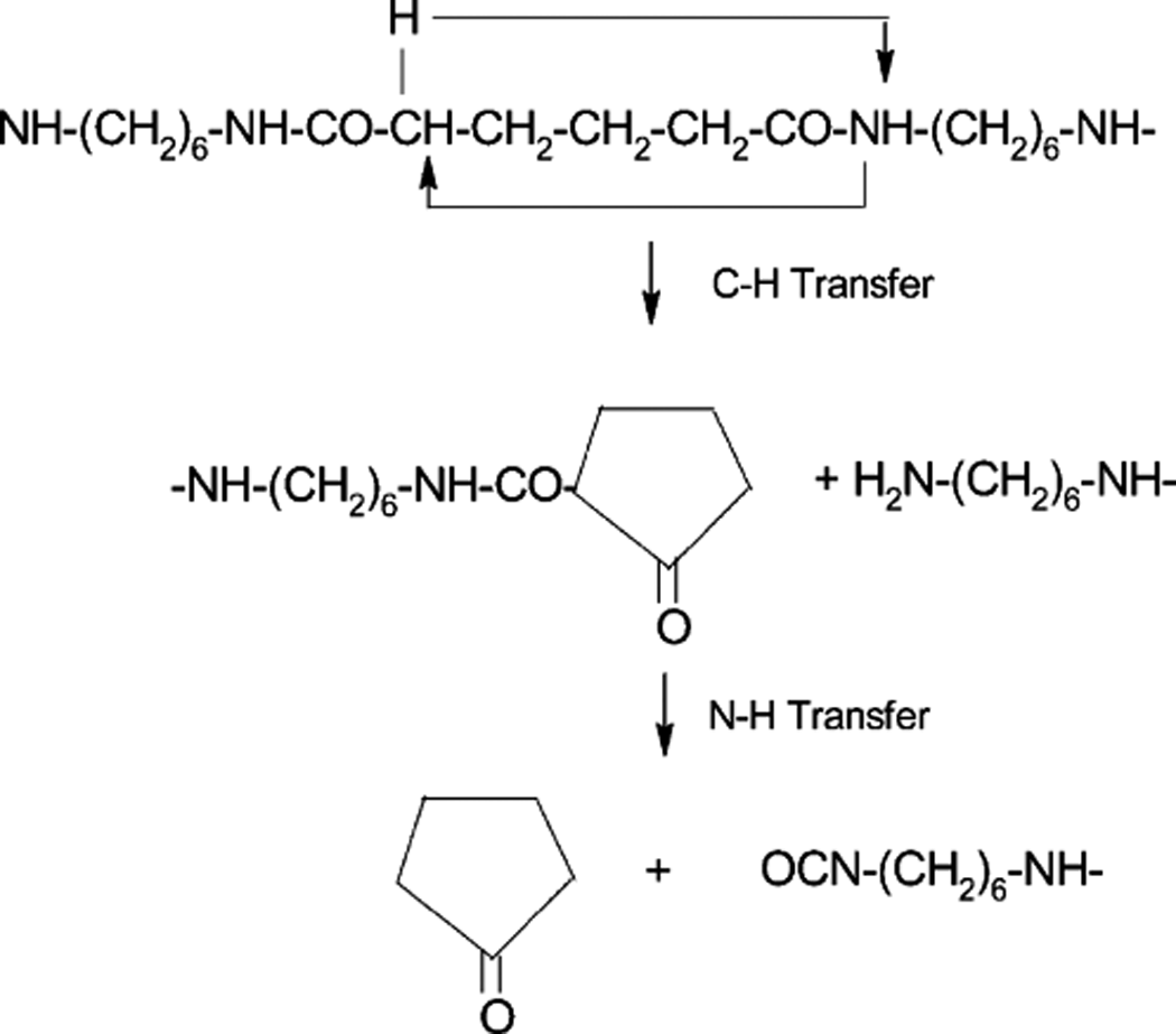
Thermal degradation mechanism of polyamide 6.6 (PA 6,6). 31
The TGA decomposition curves for neat PA 6,6 and their nanostructure composites are presented in Figure 3. The neat PA 6,6 TGA curve corresponds to a single-stage degradation with well-defined initial and final degradation temperatures and might have been a result of a random chain scission process. PA 66 eliminates in the form of cyclopentanone, the main organic product, and also in the form of hydrocarbons, nitriles and vinyl groups. 20
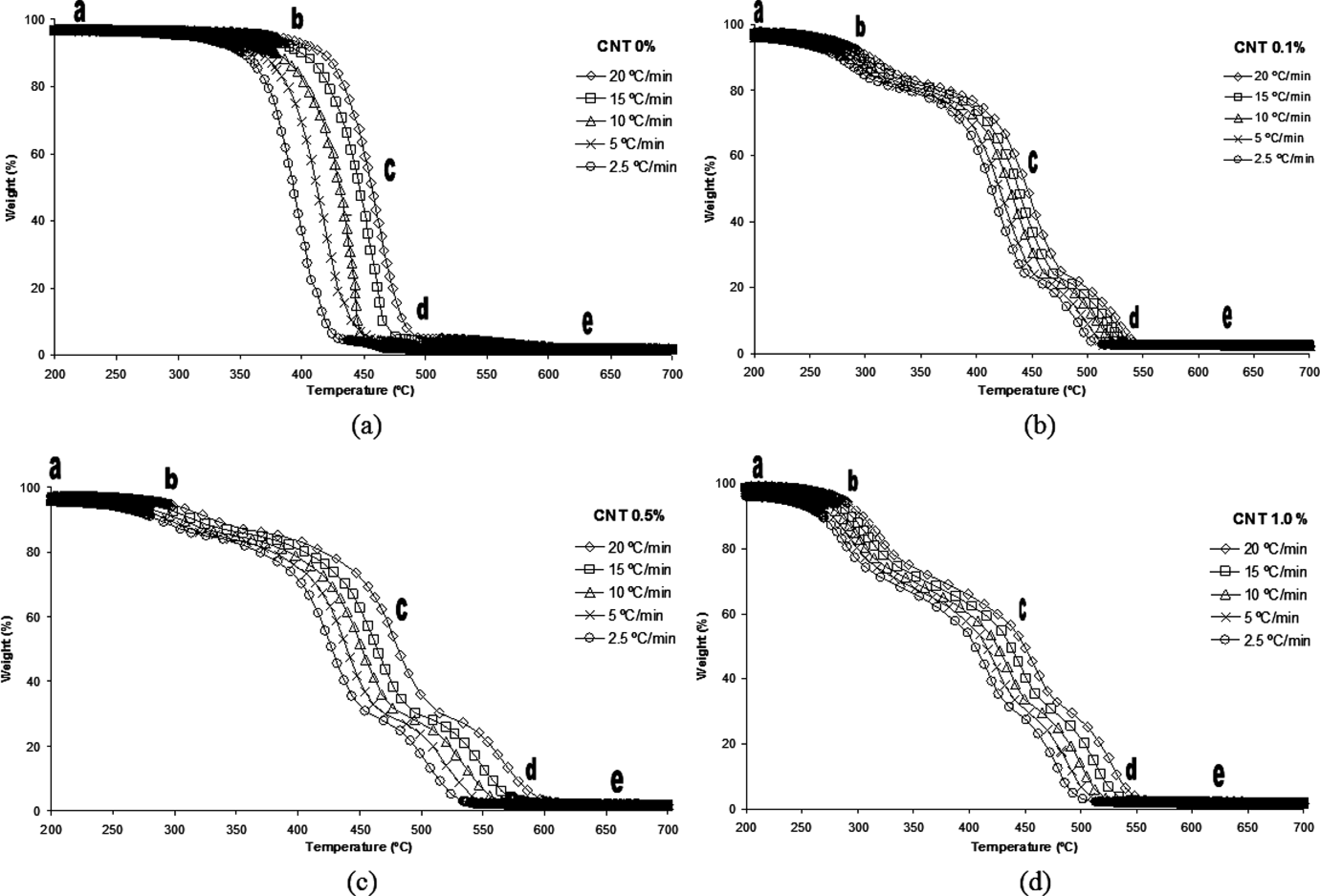
Thermogravimetric analysis (TGA) curves for polyamide 6.6 (PA 6.6) and their nanostructured composites: (a) carbon nanotube (CNT) 0%; (b) CNT 0.1%; (c) CNT 0.5% and (d) CNT 1.0%.
TGA curves for nanostructured composites have a similar behavior with reference to mass variation, as shown in Figure.3(a–d). There are at least five stages of decomposition for the nanostructured composites from the TGA curves. In these curves, the step (a) shows the losses from volatile products of low molar mass (water or solvent), in which the degradation process has not yet started. The region designated as (b) corresponds to the beginning of the degradation process. The step (c) is associated with the inflection of the degradation rate, in which some reactions can maintain their maximum rate for a significant period of time. 20
Variations in the maximum rate present at point (c) at each heating rate lead to a variety of kinetic behavior, where the maximum reaction rate determines the conversion degree (α). This step is associated with the release of low-molecular-weight fraction components such as water and/or organic solvents. This is the most complex stage in the thermal degradation study for polymeric composites.
Point (d) represents the decay of the degradation reaction rate. At this stage, there is a slight slope in the final stage of the curve, that it is a consequence of the gas liberation process resulting from the polymer chain decomposition. The point (e) corresponds to the end of the reaction for nanostructured composites degradation. From Figure 3, it has been observed that there is a slight difference in the slope of curves among the steps (b) and (d). The increase in the heating rate causes an increase in the degradation temperature. This is explained by the accommodation of the molecules and the thermal inertia caused by the high heating rates.
The derivative thermogravimetric (DTG) plot of neat PA 6,6 is shown in Figure.4, in which two peaks of decomposition are observed, and there are several multiple peaks associated probably with overlapping decomposition reactions. It is observed that the overlapped peaks are separated by the addition of CNTs in polymer matrix. For the nanostructured composite with 1.0 wt% CNT, the DTG curve is best solved. This evidence suggests the existence of a good interaction (dispersion) between the CNTs and the polymer matrix. The DTG peak curve gives the rate of mass variation as a function of temperature, at which the highest reaction rate occurs. This rate is represented by point (c) (Figure.4).
By the addition of CNT into PA 6,6, two thermal events becomes more evident in the DTG curves: (a) the formation of a new phase represented by point (f), occurring at around 350°C and (h) the degradation process of nanostructured composites occurs between 550 and 650°C.
The maximum temperature degradation rate for the PA 6,6 and their nanostructured composites are presented in Table.1. The presence of CNTs modified the thermal properties of nanostructured composites when these are compared with the behavior exhibited by neat PA 6,6. In those cases, an increase in the temperatures of maximum degradation rate is observed increasing of CNTs amount. This behavior can be explained as a consequence of an increase in the thermal stability of the composites caused by the presence of CNTs in PA 6,6. The trend in thermal stability is explained on the basis of increasing content of CNTs as well as agglomeration tendency of the in situ generated CNTs. Therefore, at very low loading level (within a theoretical CNT content of 0.5 wt%), the CNT improves the thermal stability of the PA 6,6/CNT composites significantly.
Maximum temperature degradation rate for the PA 6,6 and its nanostructured composites as function of the heating rate.
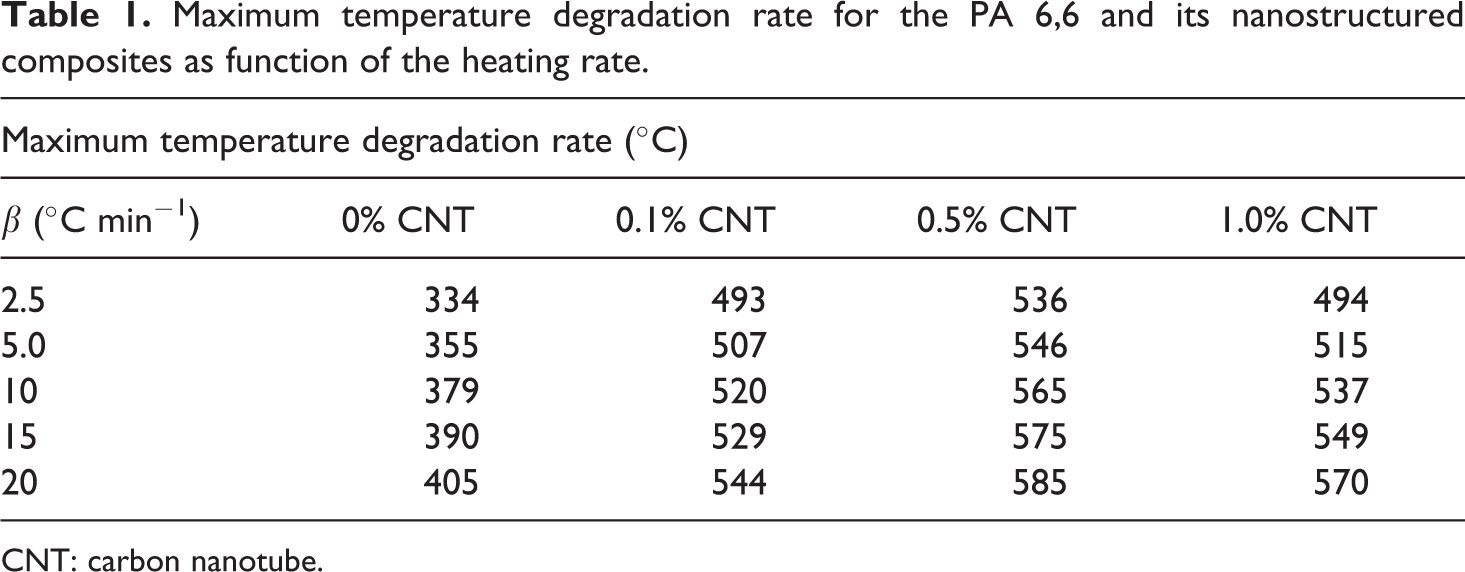
CNT: carbon nanotube.
The thermal degradation kinetic study in nonisothermal conditions of nanostructured composites was performed using the O-W-F method, which provides additional results about the characteristics inherent to the process of thermal degradation, allowing a more complete description of this phenomenon.
The behavior of the curves obtained from the O-W-F method for PA 6,6 and their nanostructured composites are shown in Figure 4, in which the conversion degrees (α) between 5 and 15%, in intervals of 2.5% have been used. The straight lines observed in Figure 5 should be parallels having the same distance from each other. Therefore, there is an irregular spacing between the straight lines. This behavior is characterized by the degradation processes with breaks in covalent bonds at different energy levels.
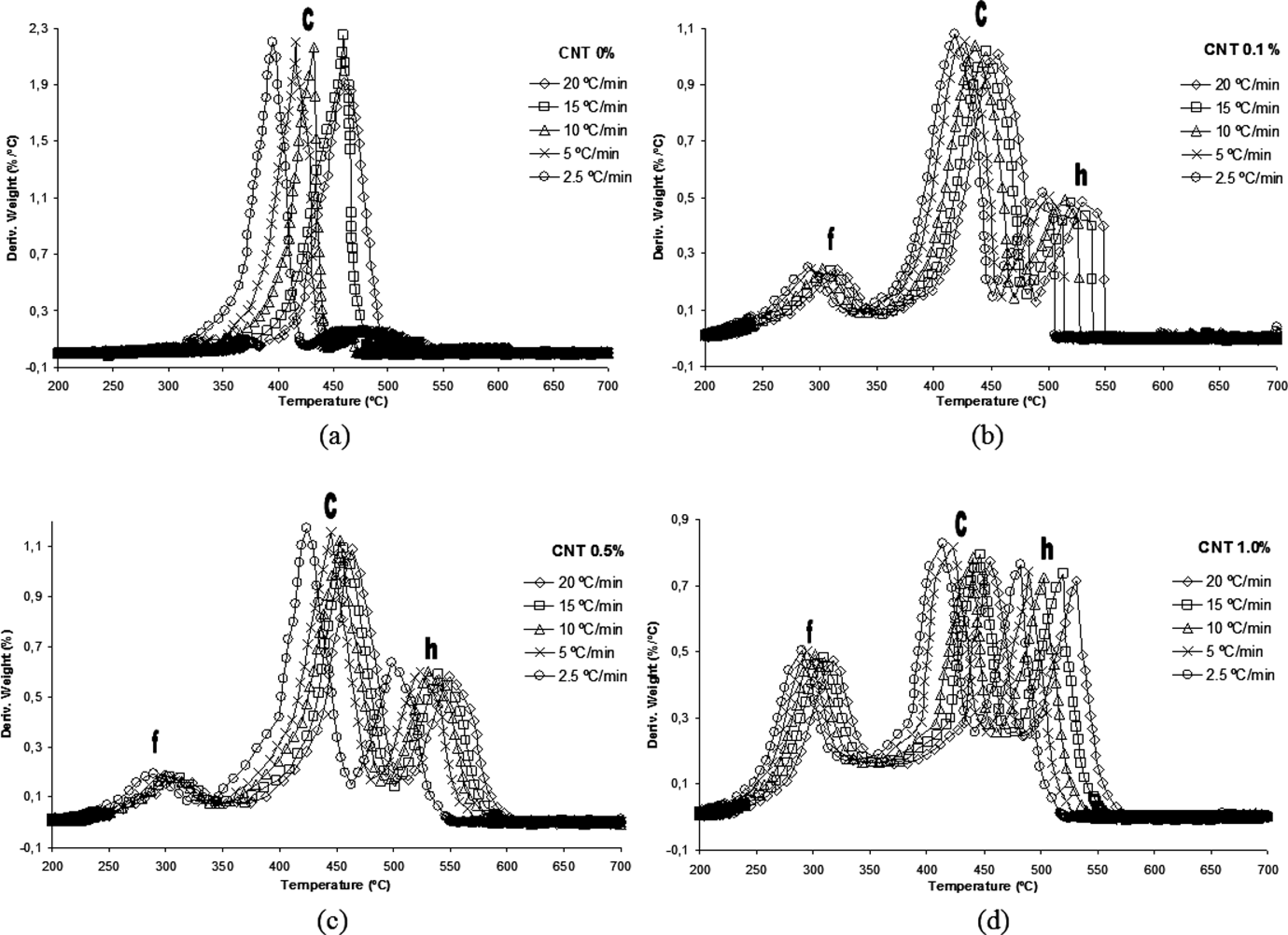
Derivative thermogravimetric (DTG) curves for polyamide 6.6 (PA 6.6) and their nanostructured composites: (a) carbon nanotube (CNT) 0%; (b) CNT 0.1 %; (c) CNT 0.5% and (d) CNT 1.0%.
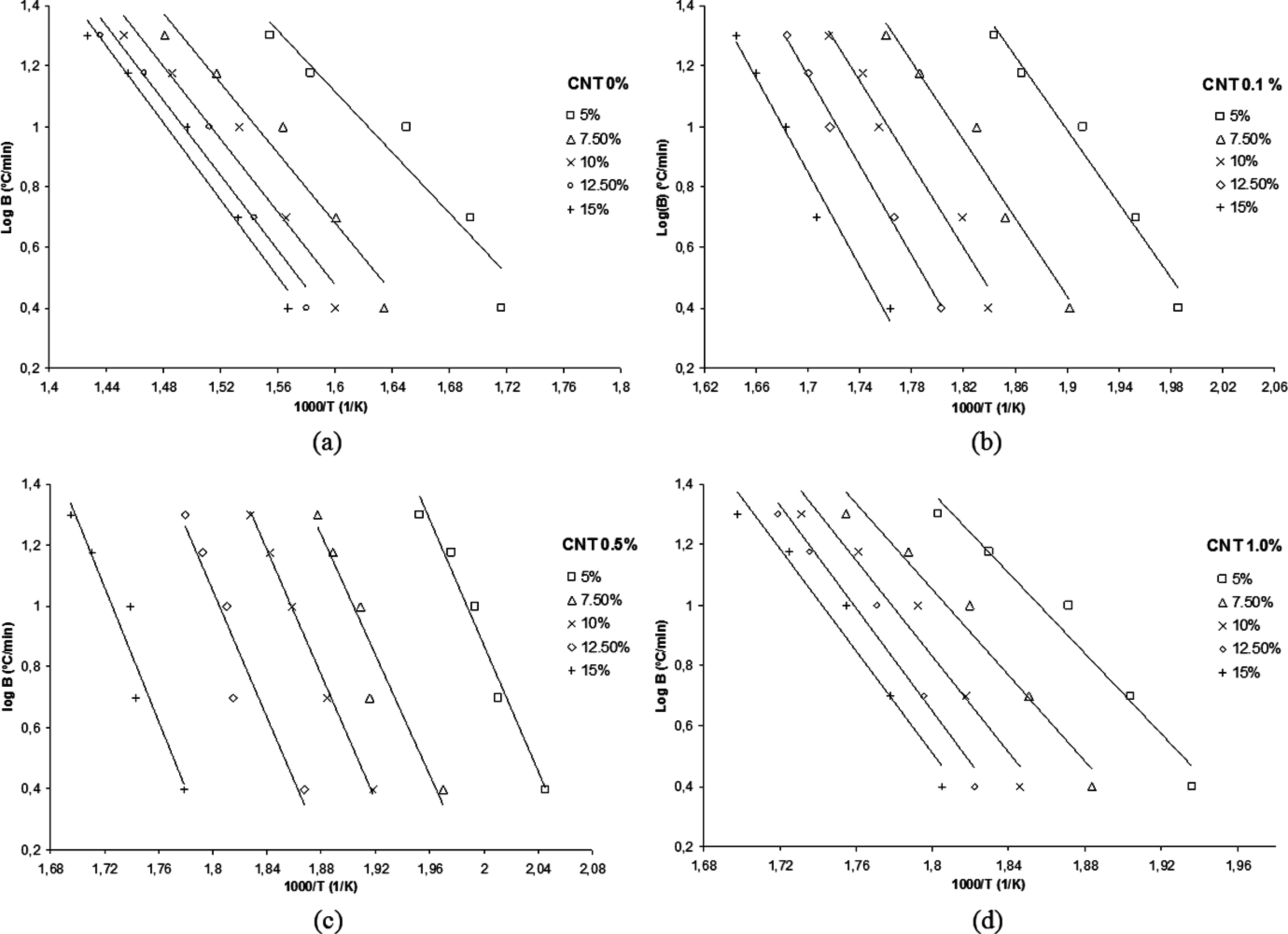
Isoconversional curves obtained by Ozawa–Wall–Flynn (O-W-F) model for the polyamide 6.6 (PA 6.6) and their nanostructured composites: (a) CNT 0%; (b) CNT 0.1%; (c) CNT 0.5% and (d) CNT 1.0%.
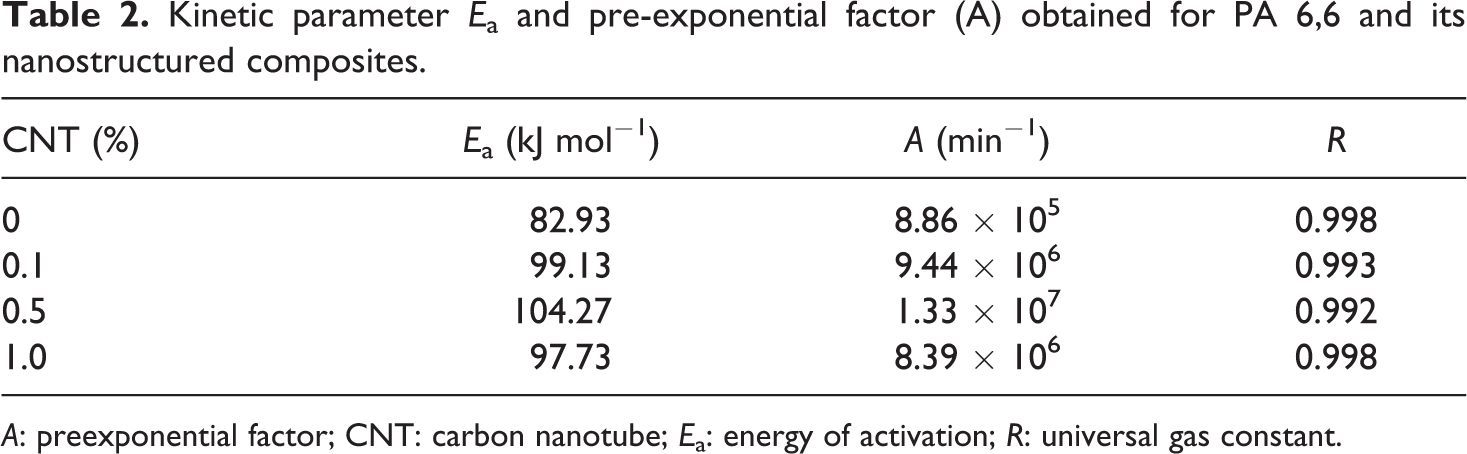
The behavior of activation energy (E a) of thermal degradation as function of the degree of conversion (α) is shown in Figure.6. The kinetic parameters of nanostructured composites and their correlation coefficients (r) calculated from the O-W-F method are depicted in Table 2. In this study, the heating rate 10°C min−1 was used to study the thermal decomposition of the nanocomposites, which corresponds to the midpoint of the experimental heating rate. So, all the considerations about the kinetic parameters obtained in this work are based on this heating rate.
Kinetic parameter E a and pre-exponential factor (A) obtained for PA 6,6 and its nanostructured composites.
A: preexponential factor; CNT: carbon nanotube; E a: energy of activation; R: universal gas constant.
E a values above 100 kJ mol−1 suggest a degradation mechanism associated with the scission of strong bonds (random breaks in the chain), reflecting the existence of multiple reactions competing in the degradation process. According to literature, 22 the reaction order equals zero indicates mass loss by the splitting of the polymer chain and/or division of smaller molecules of the lateral chain. On the other hand, reactions of the random scission of main chain are indicated for first-order reactions, while intermolecular transfer reactions exhibit second-order reaction. In the present study, nanostructured composites of first-order kinetics are considered.
Neat PA 6,6 has a lower activation energy, E a, as compared with the nanostructured composites produced, indicating the pure polymer has a lower thermal stability. The addition of CNT showed a synergistic effect because the values of E a were higher than those determined for the neat PA 6,6, suggesting an increase in thermal stability of polymer matrix by the addition of CNTs. All curves from Figure 6 present the same profile indicating that as the CNTs content increases, the samples become more stable, requiring more energy for bond breaking.
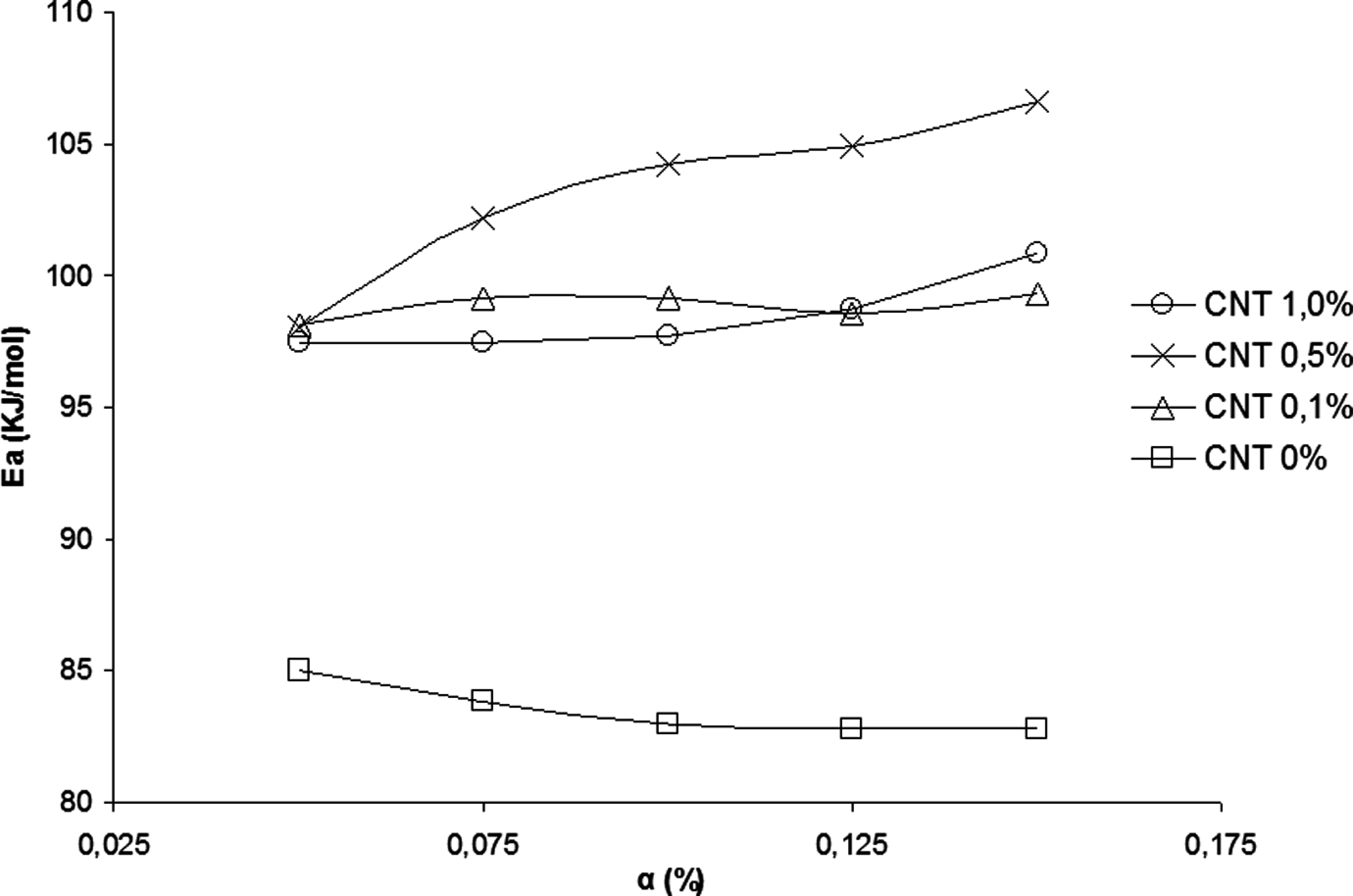
Activation energy (E a) as function of the conversion degree (α) for the polyamide 6.6 (PA 6.6) and their nanostructured composites.
According to literature 19 studies on the thermal degradation of PA and their nanostructured composites observed that the dispersion of nonfunctionalized CNTs in the PA 6.6, exceeding 0.5 wt% was not adequate. This fact is explained by the nonpolar characteristic of the CNTs, creating an incompatibility with the polymer matrix of polar nature. The dispersion of inadequate amounts of CNTs, exceeding 0.5 wt%, produced a decrease in activation energy (E a) and consequently the preexponential factor (A). This same behavior is observed in Figure 6, in which the nanostructured composite with 1.0 wt% of CNT presents the E a lower than the others (0.1 and 0.5 wt% CNT).
In this study, the half-life time estimated for the nanostructured composite was chosen as one in which there are approximately 10% of mass loss at different temperatures, as shown in Figure 7. The addition of nanostructured material increases the thermal stability of the PA 6.6. Among all the studied nanostructured composites, the system with 0.5 wt% CNT presents the higher thermal stability. As discussed above, the nanostructured composites with content of CNT higher than 0.5 wt% have an inadequate dispersion in the polymer matrix. This fact is evident, because both the activation energy (E a) and the half-life time (t 1/2) decrease when larger quantities of CNTs are added in polymeric matrix. Therefore, the thermal stability of nanostructured composites is impaired when a quantity exceeding 0.5 wt% CNT are dispersed into the PA 6.6 matrix.
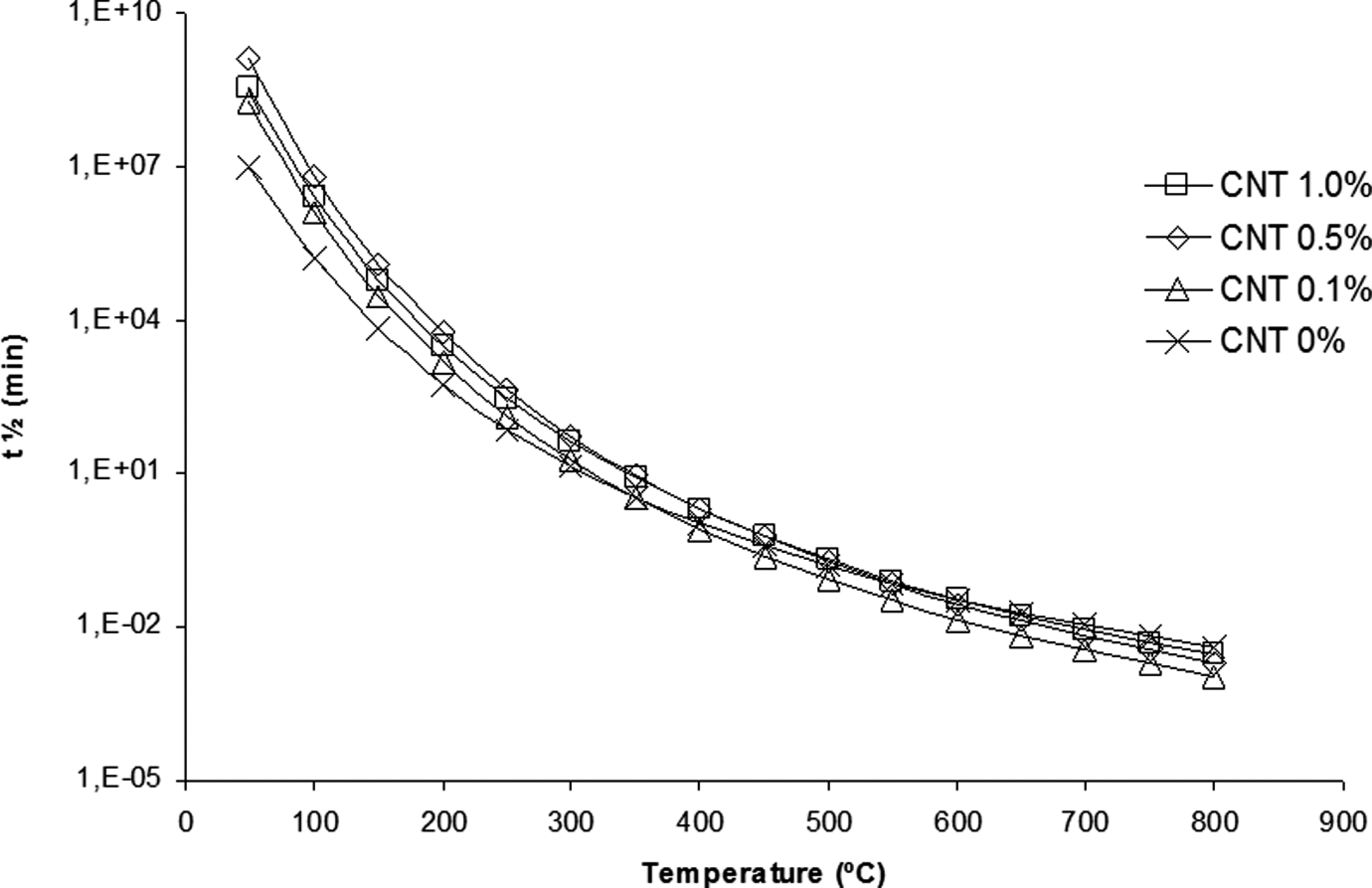
Half-life time as function of the temperature for the polyamide 6.6 (PA 6.6) and their nanostructured composites.
Conclusion
TGA was used to compare the thermal stability in dynamic nitrogen environment of neat PA 6.6; 0.1 wt% CNT/PA 6.6; 0.5 wt% CNT/PA 6.6 and 1.0 wt% CNT/PA 6.6 composites. The neat PA 6.6 decomposes apparently in one step in nitrogen and at least five steps are identified for the nanostructured composites studied in the present work. Based on the dynamic thermogravimetric curves, the kinetic parameters E a, A and the maximum degradation temperature rate were calculated by O-W-F model, and it was shown that the presence of CNT in the PA 6.6 matrix affects their thermal properties. An increase in the maximum degradation temperature rate and in the E a with an increase in the amount of CNTs is observed in the nanostructured composites. A similar trend in the thermal stability of the samples is reflected from O-W-F method. The trend in the thermal stability is explained on the basis of increasing CNT content as well as agglomeration tendency of CNT in the PA 6.6. Therefore, at very low loading level (within a theoretical CNT content of 0.5 wt%), the CNT improves the thermal stability of the PA 6.6/nanostructured composites significantly. An exception was found for the formulation with 1.0 wt% CNT which is the nonpolar characteristic of the CNTs, creating an incompatibility with the polymer matrix of polar nature, by reducing its thermal stability.
Footnotes
Funding
The authors acknowledge financial support received from FAPESP (under grants 05/54358-7 and 08/00171-1) and CNPq under grants 306053/2006-3.













