Abstract
To study the dynamic mechanical kinetics of exfoliated nanocomposites using dynamic mechanical analysis, polyvinyl acetate (PVAc)–montmorillonite (MMT)–dioctadecyl dimethyl ammonium bromide (DOAB) was synthesized through five different processes. Different synthesis processes and MMT-DOAB had little effect on the structure of PVAc-MMT-DOAB. MMT-DOAB lowered the glass transition temperature of PVAc, hence improving its low temperature resistance greatly. PVAc and PVAc-MMT-DOAB were homogeneous amorphous linear polymers and they appeared cold crystallized, and their glass transition and cold crystallization kinetics were analyzed using Agrawal integral equation.
Introduction
Dynamic mechanical property can reflect the internal structure and law of molecular motion of materials by measuring their change with temperature under a certain frequency or their change with frequency or deformation at a certain temperature. This is always done by dynamic mechanical analysis (DMA). The elastic modulus, loss modulus, loss tangent, and other parameters are available on DMA spectra, and they can reflect the damping properties of materials intuitively. He and coworkers 1 –4 once used dynamic torsional vibration methods and Avrami theory to study the curing kinetics of some thermosetting resins. However, in the article, Agrawal integral equation was used to study the dynamic mechanical kinetics of polyvinyl acetate (PVAc)–montmorillonite (MMT)–dioctadecyl dimethyl ammonium bromide (DOAB). With more and more applications of MMT in polymers, 5–6 such as polyacrylate ester, 7,8 poly(methyl methacrylate), 9,10 polyurethane, 11,12 epoxy, 13,14 polycarbonate, 15,16 polyethylene, 17 polypropylene, 18 coatings, 6,19 and so on, to improve their chemical, physical, and mechanical properties, PVAc should also be in them. In our previous work, PVAc, 20 –23 organic MMT, 24,25 and exfoliated nanocomposites of PVAc-MMT, 26 PVAc-MMT-octadecyl trimethyl ammonium bromide (STAB), 27 –29 and PVAc-MMT-DOAB 30,31 were prepared. PVAc-MMT in Ref. 26, PVAc-MMT-STAB in Refs. 27 and 28, and PVAc-MMT-DOAB in Ref. 30 were all synthesized through a single process, in which the polymerization time being 6 h and the major water in the synthesis being 250 g were fixed as controls; the amount of MMT, MMT-STAB, and MMT-DOAB used in the synthesis varied from 0.5% to 5% of VAc, and at 2%, the properties of these exfoliated nanocomposites, including rheology, apparent viscosity, molecular weight, static tensile, and so on, were the best, and no better results on these properties could be obtained by exceeding this point, so the 2% was optimized and chosen. In Refs. 29 and 31, PVAc-MMT-STAB and PVAc-MMT-DOAB were synthesized through five different processes; the 2% amount of MMT-STAB and MMT-DOAB was fixed as the control, while the polymerization time being 6 h, 5 h, 4 h, 3 h, and 2 h and the major water in the synthesis being 250 g, 200 g, 150 g, 100 g, and 50 g were the variables, and others, including polymerization temperature, violent stirring, and additives, were same as Ref. 26–28, and 30. In the article, PVAc-MMT-DOAB was also synthesized through these same five different processes, MMT-DOAB was 2% of VAc and, in fact, some of its dynamic mechanical properties were simply introduced in Ref. 31, but never involved the dynamic mechanical kinetics, especially the glass transition and cold crystallization kinetics analyzed using Agrawal integral equation, which was the point of this article. Therefore, in the article, PVAc and PVAc-MMT-DOAB were synthesized through five different processes; they all were homogeneous amorphous linear polymers and their structure was investigated mainly by x-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), transmission electron microscopy (TEM), and dispersion in water, and DMA tests for all PVAc-MMT-DOAB obtained from these five different synthesis processes were carried out, and their glass transition and cold crystallization were studied, and using Agrawal integral equation to follow their kinetics was also introduced. In addition, by studying the dynamic mechanical kinetics, some hints or inspirations might be given, some methods to improve PVAc and PVAc-MMT-DOAB were provided, and also help in their processing, forming, and application.
Experimental
Materials
Materials used in the synthesis included VAc, DOAB, polyvinyl alcohol, alkylphenol polyoxyethylene ether, ammonium persulfate, sodium lauryl sulfate, ethanol, sodium benzoate, sodium bicarbonate, di-n-butyl phthalate, and water. All these chemical reagents were from Sigma-Aldrich, Shanghai, China. MMT-DOAB used in the synthesis was prepared by MMT and DOAB as follows: 500 ml of water and 10 g of MMT were mixed together and stirred strongly for 5 h at room temperature; when the mixture became a suspension liquid, temperature rose to 70–80°C; a certain amount of DOAB was dissolved in the mixture of water and hydrochloric acid and then this were added into the suspension liquid in 0.5–1 h; after strongly stirring the mixture for 2–3 h, MMT-DOAB colloid was obtained, and after washing, filtering, drying, and grinding, MMT-DOAB solid samples were prepared.
Samples
As shown in Table 1, 1 g of MMT-DOAB was immersed into 25 g of VAc for 24 h. They were then mixed with 70 g of 10% polyvinyl alcohol solution, 0.50 g of alkylphenol polyoxyethylene ether, 3.75 g of 10% ammonium persulfate solution, 6.25 g of sodium lauryl sulfate, and 250 g (200 g, 150 g, 100 g, and 50 g) of water while stirring vigorously for 8 h. When the mixture became a homogeneous emulsion, temperature rose to 70°C. While stirring vigorously, 3.75 g of 10% ammonium persulfate solution and 25 g of VAc were added into the homogeneous emulsion gradually in 6 h (5 h, 4 h, 3 h, and 2 h) for polymerization. Subsequently, temperature rose to 85–90°C and the emulsion further polymerized for 0.5–1 h. After polymerization, temperature dropped to below 50°C, 4 g of ethanol, 3 g of water, 0.30 g of sodium benzoate, 0.18 g of sodium bicarbonate, and 6 g of di-n-butyl phthalate were added into the emulsion. Finally, PVAc-MMT-DOAB-A (PVAc-MMT-DOAB-B, PVAc-MMT-DOAB-C, PVAc-MMT-DOAB-D, and PVAc-MMT-DOAB-E) was obtained. Here, A, B, C, D, and E were used to, respectively, correspond different synthesis processes with different polymerization time and different proportion of water. PVAc-A, PVAc-B, PVAc-C, PVAc-D, and PVAc-E were also synthesized by the same processes as PVAc-MMT-DOAB but without MMT-DOAB. In the synthesis, polyvinyl alcohol was the protective colloid, alkylphenol polyoxyethylene ether was the emulsifier, ammonium persulfate was the initiator, sodium lauryl sulfate was the anionic surfactant, di-n-butyl phthalate was the plasticizer, and the mixture of ethanol, water, sodium benzoate, and sodium bicarbonate was the pH regulator.
Synthesis design.
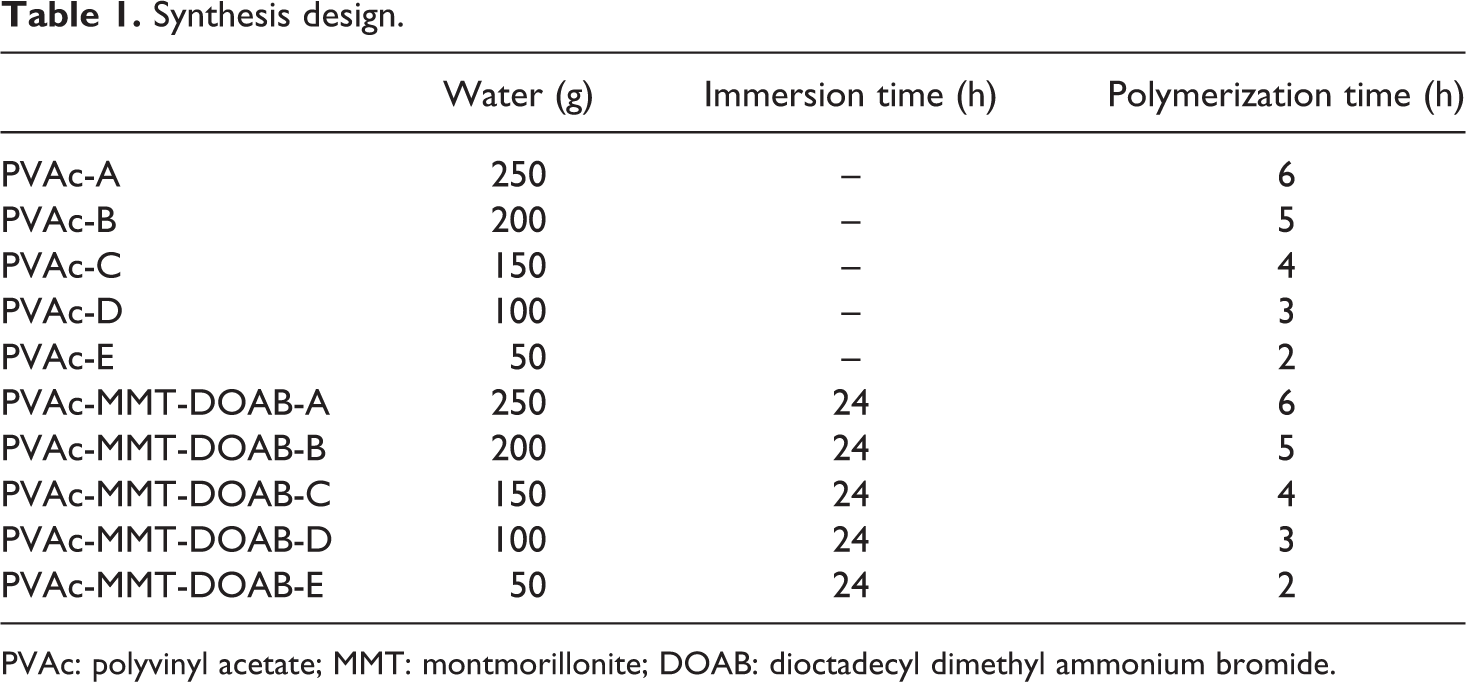
PVAc: polyvinyl acetate; MMT: montmorillonite; DOAB: dioctadecyl dimethyl ammonium bromide.
X-Ray diffraction
To prepare the tested solid samples, PVAc and PVAc-MMT-DOAB were dried to constant weight at room temperature and then were stored under −20°C for about 2 h. These frozen PVAc and PVAc-MMT-DOAB were crushed, grounded, and made into powder samples. After their weight getting constant at room temperature, these solid powder samples were tested by DX-2000 XRD (Dandong Fangyuan, China) under Cu Kα of radiating, 40 kV of tube voltage, 30 mA of tube current, scanning from 0.5° to 15° at a rate of 0.02°s−1, and λ = 1.54184 Å of wavelength. The large value of d (001) for MMT or MMT-DOAB is calculated by Bragg’s law of λ = 2dsinθ, where λ is the wavelength representing the intensity of X-ray, d is the distance between two layers of MMT, and θ is the diffraction angle.
Fourier transform infrared spectroscopy
The tested solid samples were prepared by the same method as XRD. They were then tested by NICOLET 380 FTIR (Nicolet, USA).
Dispersion
Liquid samples of 3 to 5 drops were added to 20 ml of water in a glass dish (diameter 90 mm). When they became homogeneous, the dispersion was observed with water as reference.
Transmission electron microscopy
The liquid samples were observed by JEM-3100F TEM (Jeol, Japan) with an amplification of 20,000 times.
Dynamic mechanical analysis
PVAc and PVAc-MMT-DOAB were made into circular films, and their diameter was 10 mm, their thickness was 0.5 mm. After drying them to constant weight at room temperature, they were tested by NETZSCH DMA 242C (Netzsch, Germany) through compression model under 10 Hz of frequency, rising the temperature from 0°C to 200°C at a rate of 5°C min−1, 120 μm of the maximum amplitude, 8 N of the maximum dynamic force, and 2 N of the minimum static force.
Results and discussion
Structure
Now, determining a material to be an intercalated or exfoliated nanocomposite is generally based on its diffraction peak position, shape, and intensity of XRD patterns. 32,33 If one or more diffraction peaks corresponding to d (001) for layered silicate crystal layers appear on XRD patterns and the diffraction peak position has a certain migration to the small angle when compared with layered silicate powder, this indicates that the layered silicate was opened, and it is called the intercalated nanocomposite. For the exfoliated nanocomposite, its XRD patterns seem to be linear; no diffraction peak appears within the diffraction angle range of 0.5°–5°, indicating that the clay layers and their ordered structure were destroyed and exfoliated completely.
In the emulsion polymerization, polymerization time and water proportion are very important, 20 –22 so five different processes were designed, A, B, C, D, and E: their polymerization time was 6 h, 5 h, 4 h, 3 h, and 2 h and their major water was 250 g, 200 g, 150 g, 100 g, and 50 g, respectively. PVAc-MMT-DOAB were prepared through these processes, and they all were exfoliated nanocomposites (Figure 1). The diffraction peaks of MMT and MMT-DOAB appeared, their 2θ were 7.00° and 2.22° and their d (001) were 1.263 nm and 3.980 nm, respectively. After the organic modification, MMT-DOAB was an intercalated nanocomposite, its XRD pattern still had the diffraction peak. For PVAc-MMT-DOAB, including PVAc-MMT-DOAB-A, PVAc-MMT-DOAB-B, PVAc-MMT-DOAB-C, PVAc-MMT-DOAB-D, and PVAc-MMT-DOAB-E, no diffraction peak appeared within 0.5°–5° of θ on their XRD patterns. In the emulsion polymerization, on the basis of the first intercalation of DOAB into MMT, linear macromolecular chains of PVAc were formed in MMT-DOAB layers, and these chains formed made the second intercalation, so the distance between MMT layers was increased greatly and the original ordered crystal layers were destroyed completely. MMT-DOAB layers were exfoliated completely and dispersed independently in the emulsion with nanometer-thick layers. No chemical bond was found between MMT-DOAB and PVAc in PVAc-MMT-DOAB, but only physical absorptions or secondary bonds (Figure 2) were found. The absorption bands of PVAc-MMT-DOAB were stacked by those of MMT and PVAc, and neither new absorption bands formed nor did existing absorption bands disappear. Different synthesis processes had little effect on the interaction between MMT-DOAB and PVAc: FTIR spectra of PVAc-A to PVAc-E were same and those of PVAc-MMT-DOAB were also same.
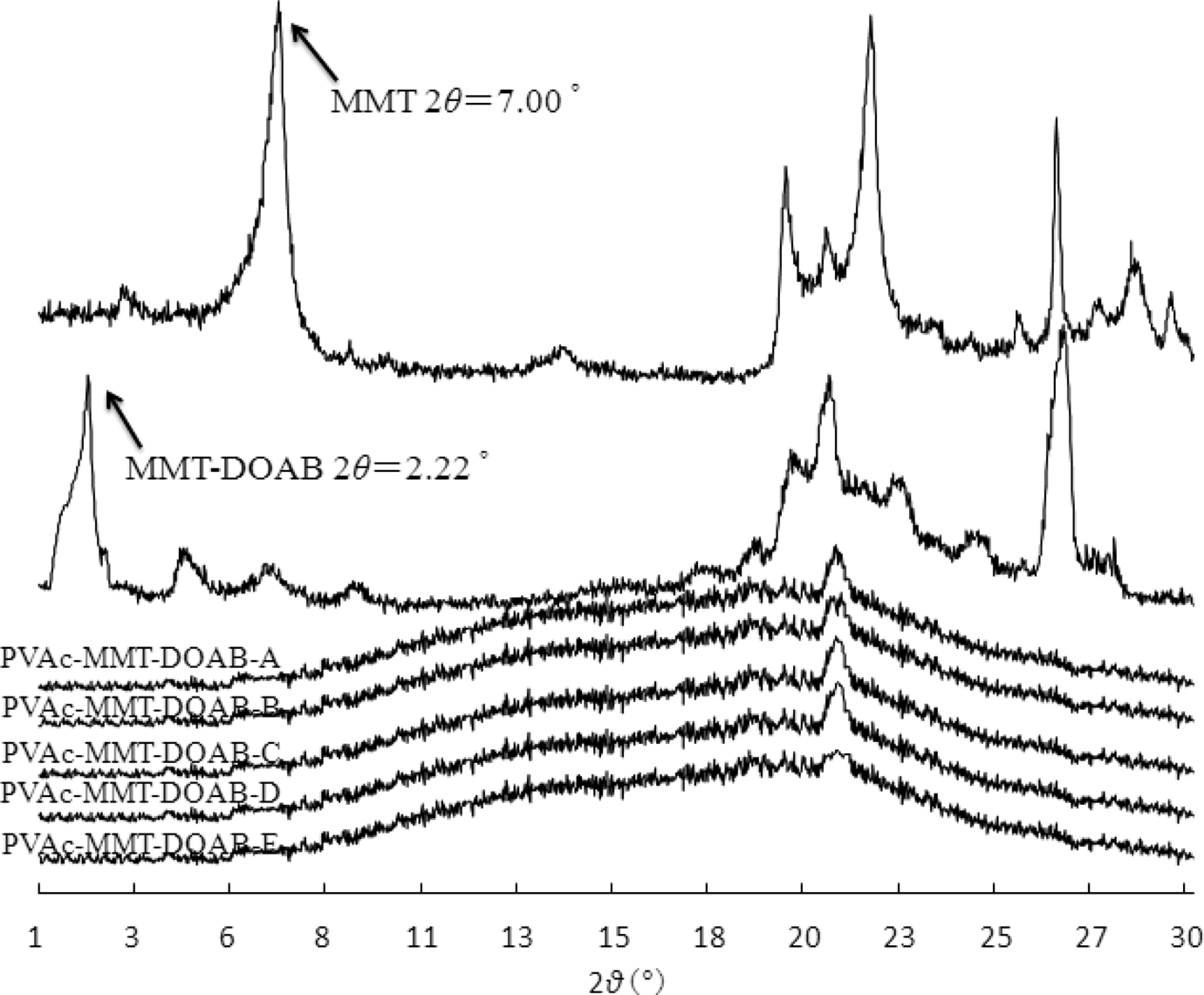
X-ray diffraction patterns.
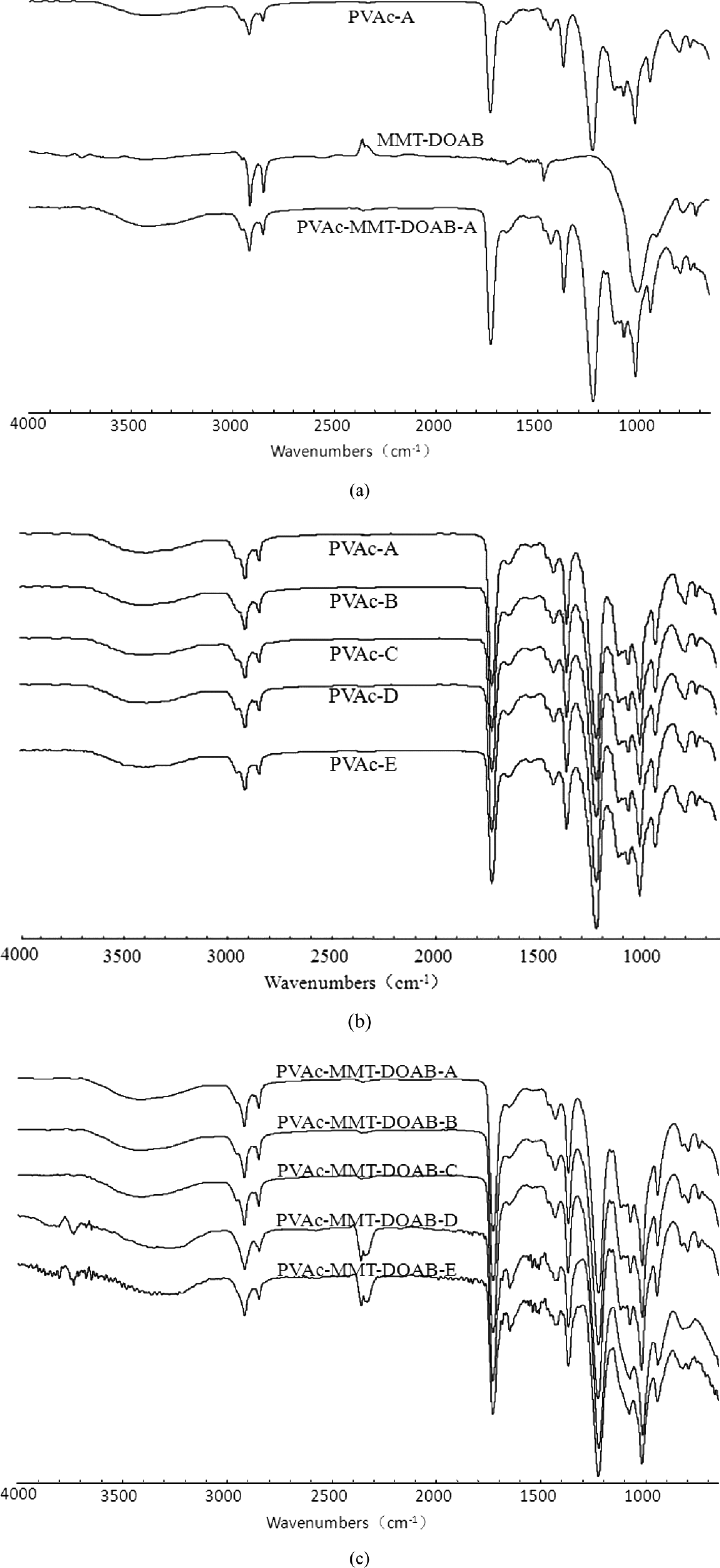
Fourier transform infrared spectra.
From the visual, all the PVAcs, including PVAc-A, PVAc-B, PVAc-C, PVAc-D, and PVAc-E, and some PVAc-MMT-DOABs, including PVAc-MMT-DOAB-A, PVAc-MMT-DOAB-B, and PVAc-MMT-DOAB-C, were same, were viscous, milk-white, homogeneous, fine emulsions, and all had no coarse particles, no foreign bodies, and no delamination. PVAc-MMT-DOAB-D and PVAc-MMT-DOAB-E were different, appeared delaminated, and PVAc-MMT-DOAB-Es were worse. Actually, PVAc-MMT-DOAB-D also was a viscous, milk-white, homogeneous, fine emulsion but had many large or small coarse particles that were deposited at the bottom and formed white precipitate. PVAc-MMT-DOAB-Es were very clear: the top part was colorless, transparent liquid, like water, with no viscous; the bottom part was viscous, milk-white floccus. Moreover, when some were dropped in water, their dispersion was also different (Figure 3). Comparing with water, no coarse particle was found in the dispersion of all PVAc, PVAc-MMT-DOAB-A, PVAc-MMT-DOAB-B, and PVAc-MMT-DOAB-C, and their dispersion was better. The dispersion of PVAc-MMT-DOAB-D and PVAc-MMT-DOAB-E was not so good. The top sample of PVAc-MMT-DOAB dispersing in water appeared to be more viscous, the color was deeper, like milk, and its bottom sample dispersing in water showed some coarse particles, some were small and some were large, and other part liquid was also like milk. PVAc-MMT-DOAB-E was an exception and was completely different from all the others; its dispersion with top sample was colorless, transparent liquid, like water, and there were white floccus and coarse particles in the dispersion with bottom sample. Under TEM observation, their particle morphology was clear (Figure 4); the larger particles were PVAcs with a diameter of 250–500 nm and the smaller particles were MMT-DOAB with a diameter of 25–100 nm. TEM also showed that MMT-DOAB particles dispersed with PVAc particles randomly; these particles were round or almost round and dispersed together randomly, as platelets in a flat form but not uniformly dispersed as described by Chien et al. 34,35 By physical absorptions or secondary bonds, they formed various, interesting structures, such as “strawberry” structure, 36,37 “core-shell” structure, 38,39 “tactoids” structure, 40,41 and so on.

Dispersion.
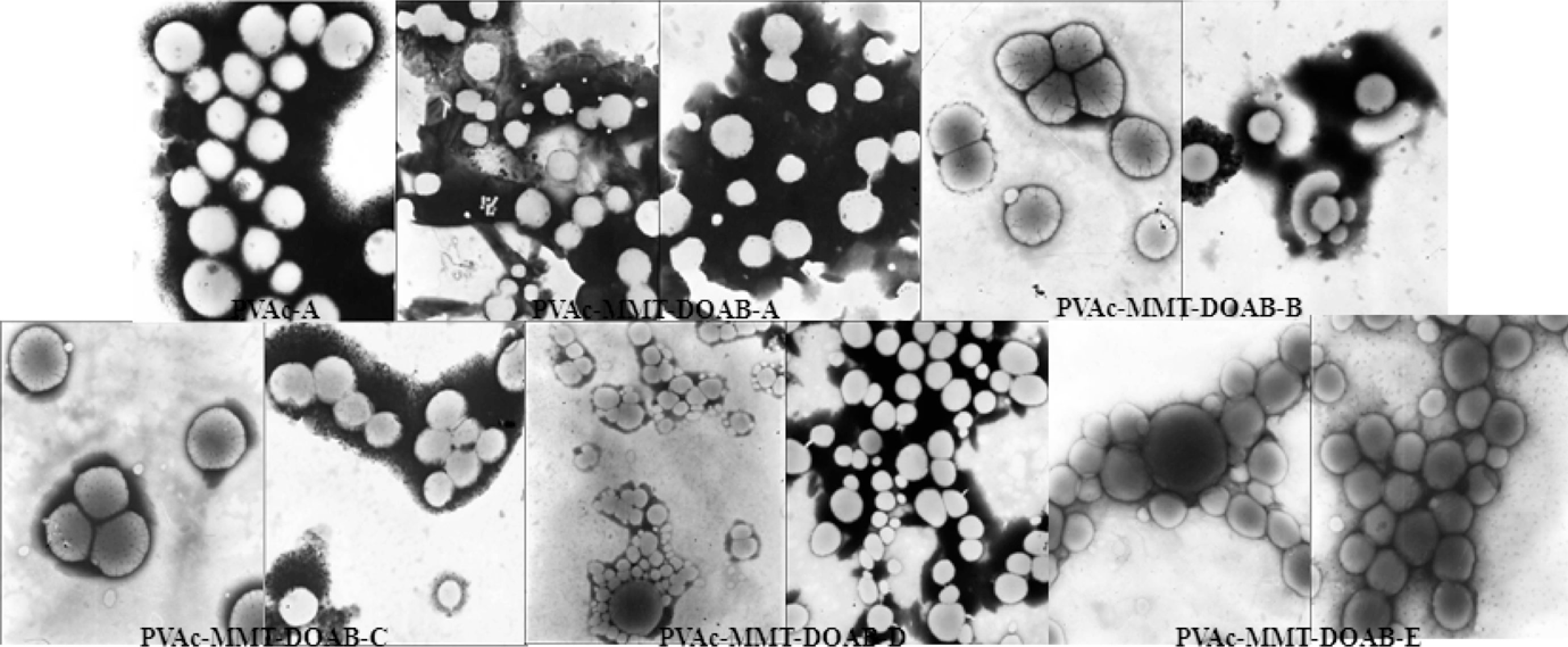
TEM images of PVAc and PVAc-MMT-DOAB. TEM: transmission electron microscopy; PVAc: polyvinyl acetate; MMT: montmorillonite; DOAB: dioctadecyl dimethyl ammonium bromide.
Dynamic mechanical properties
Different synthesis processes had little effect on the structure of PVAc and PVAc-MMT-DOAB, but the dispersions of PVAc-MMT-DOAB-C and PVAc-MMT-DOAB-D, therefore in this part, PVAc-A and all PVAc-MMT-DOABs, including PVAc-MMT-DOAB-A, PVAc-MMT-DOAB-B, PVAc-MMT-DOAB-C, PVAc-MMT-DOAB-D, and PVAc-MMT-DOAB-E, were tested by DMA (Figure 5), and their elastic modulus (E′, MPa), loss modulus (E′′, MPa), loss tangent (tanδ), and glass transition temperature (T
g, °C) were obtained. T
g and tanδ are two important indexes for damping materials, tanδ determines their quality, and T
g determines their applied temperature and frequency range. On a DMA spectrum, T
g is always defined as the temperature corresponding to the maximum loss tangent. Moreover, for damping materials, their effective damping temperature zone (Z
0, °C) can be calculated by the following equation Dynamic mechanical analysis spectra.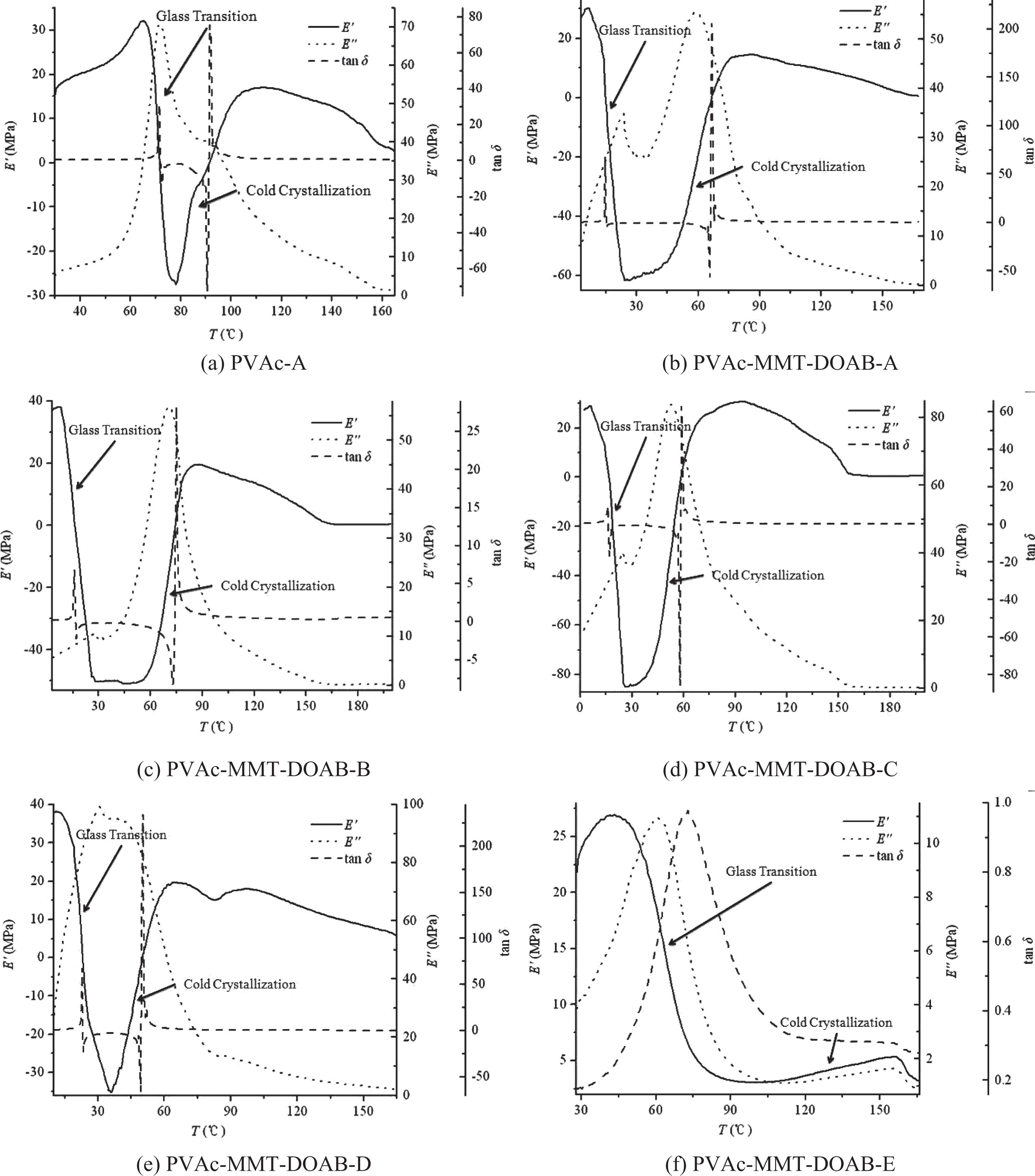

In DMA test, PVAc and PVAc-MMT-DOAB samples were circular films. In these films, polymer chains entered into MMT-DOAB layers, while their movement was restricted by the limited space, their molecular chains interacted with MMT or the organics in the layers, which could bind the polymer chains and increase their resistance of movement, the glass relaxation movement, a noncooperative movement related to the glass transition; hence, for these nanocomposites, glass transition often happened at a slightly higher temperature. 42,43 When glass transition was ongoing, at the interface between MMT-DOAB and PVAc matrix, the interaction between them limited the movement of polymer chains near the interface. In thin layers or under sandwiched restriction, the transformation of these macromolecules differed from their ontological status, shifted their behavior, and demonstrated the properties of both polymer and ceramics. 42,44 From above statements, MMT or organic MMT should increase the T g of some systems. For PVAc-MMT-DOAB, its T g was not the case (Table 2). It was affected by different synthesis processes and MMT-DOAB. This unusual phenomenon about the T g may be because the polymer chains were of great rigidity and volume, as MMT-DOAB were also rigid flat chips, and these two molecules with very different shape and rigidity and with considerable size, packed together, made the free volume of the system to increase and then decreased T g. 45
DMA data
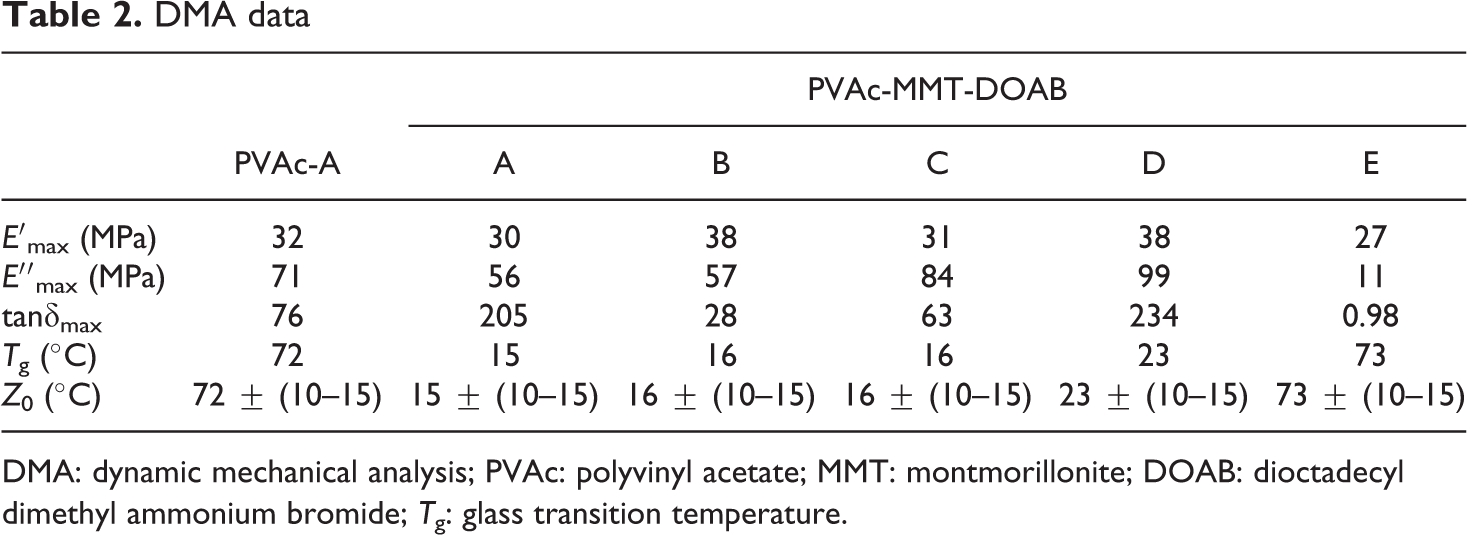
DMA: dynamic mechanical analysis; PVAc: polyvinyl acetate; MMT: montmorillonite; DOAB: dioctadecyl dimethyl ammonium bromide; T g: glass transition temperature.
PVAc and PVAc-MMT-DOAB were all homogeneous amorphous linear polymers. Under different temperatures, these polymers generally experience three different mechanical states: glass state, high elastic state, and viscous flow state; but for some amorphous polymers which possess the crystallization ability but cannot crystalline at the beginning because of inappropriate conditions, they are more likely to appear cold crystallization above T g when temperature is rising; as a result, E′ will recover and increase after falling around the end of glass transition until the crystallization phase is melt, and then it will fall sharply (Figure 6). Obviously, PVAc and PVAc-MMT-DOAB appeared the cold crystallization, which had very important practical significance and would make the material’s structure to be controlled easily in processing and molding. 46,47
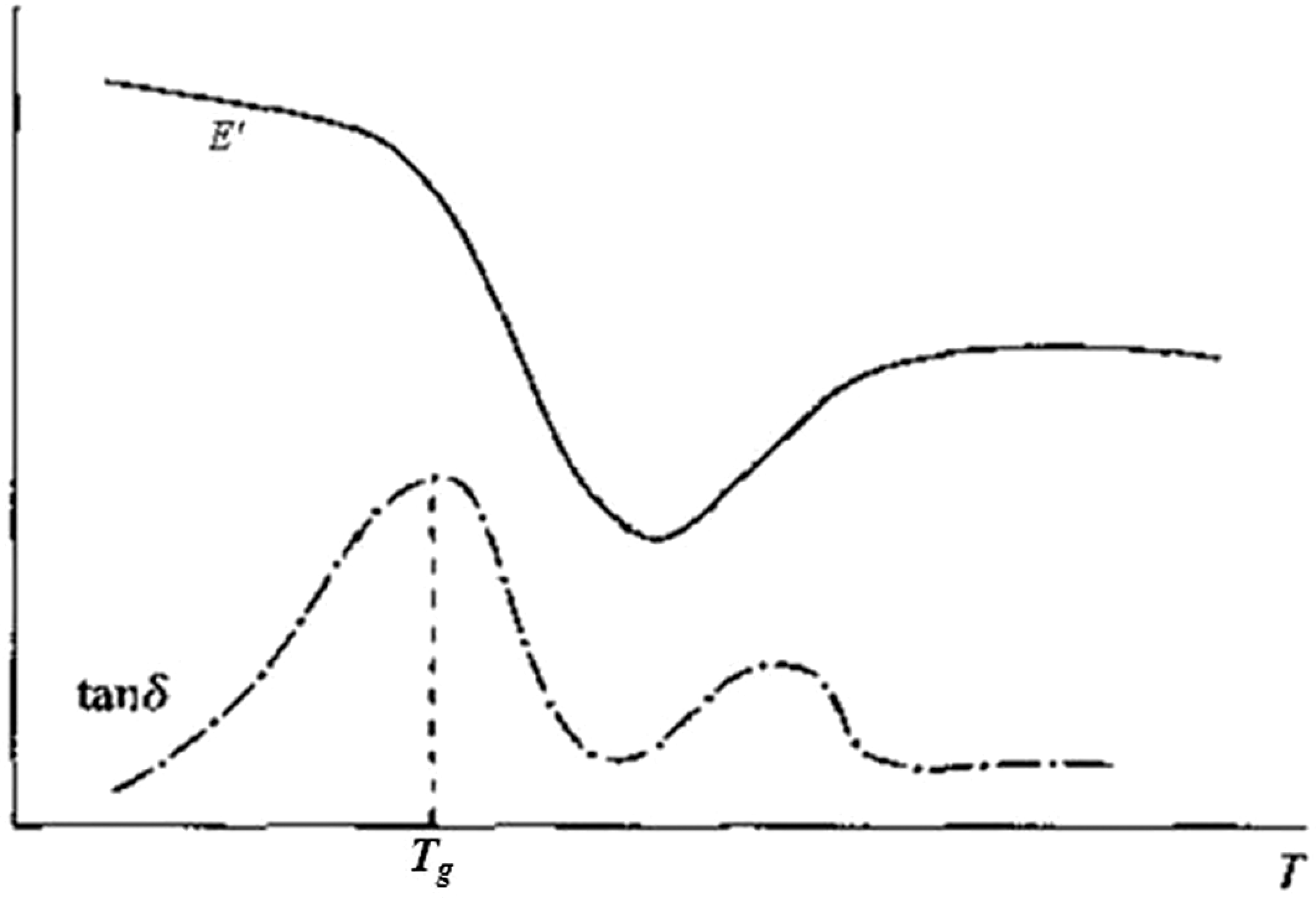
Cold crystallization.
From the above results and analysis, we know that with the rising temperature, PVAc and PVAc-MMT-DOAB would go through the glass transition, the cold crystallization, and then melt into the viscous flow. During these phases, E′ was varying all the time, and from its variation, their kinetics can be learnt, including the glass transition kinetics and the cold crystallization kinetics. The dynamic mechanical kinetics can be expressed by the simplified Agrawal integral equation 48

where a is the relative elastic modulus calculated by

where, in a phase,
The relationship between
The glass transition and cold crystallization kinetics.
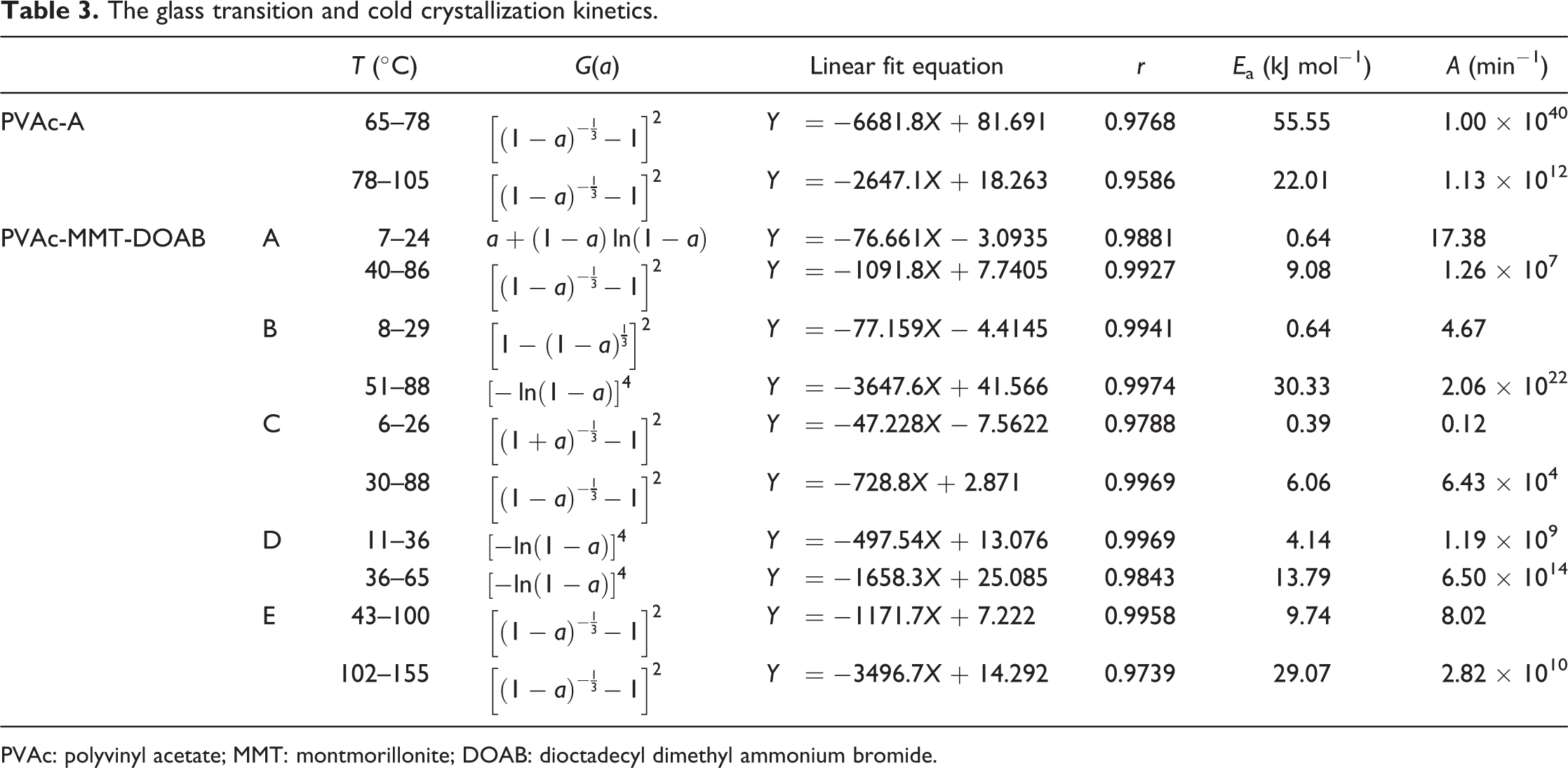
PVAc: polyvinyl acetate; MMT: montmorillonite; DOAB: dioctadecyl dimethyl ammonium bromide.
In the glass transition, the kinetics mechanism function of PVAc-A and PVAc-MMT-DOAB-E was Zhuralev–Lesokin–Tempelman equation, its mechanism was three-dimensional (3D) diffusion; the kinetics mechanism function of PVAc-MMT-DOAB-As was Valensi equation, its mechanism was two-dimensional (2D) diffusion, cylindrical symmetry, D2, deceleration a–t curve; PVAc-MMT-DOAB-B’s was Jander equation, its mechanism was 3D diffusion, spherical symmetry, D3, deceleration a–t curve, the reaction order (n) was 2; PVAc-MMT-DOAB-C’s was anti-Jander equation, its mechanism was 3D diffusion; and PVAc-MMT-DOAB-D’s was Avrami-Erofeev equation, its mechanism was random nucleation and subsequent growth, n was 4. In the cold crystallization, the kinetics mechanism functions of PVAc-A, PVAc-MMT-DOAB-A, PVAc-MMT-DOAB-C, and PVAc-MMT-DOAB-E were Zhuralev–Lesokin–Tempelman equation, their mechanism was 3D diffusion; and that of PVAc-MMT-DOAB-B’s and PVAc-MMT-DOAB-D’s were Avrami-Erofeev equation, their mechanism was random nucleation and subsequent growth, n was 4. These all showed that the glass transition and cold crystallization of PVAc-A and all PVAc-MMT-DOABs were similar.
In addition, the linear relationship between lnA and E a, named as kinetic compensation effect, explains that A partly compensates to the change of E a 49

where K and Q are the kinetic compensation effect parameters that can be calculated by linear fit between E a and A. Then, the kinetic compensation effects of the glass transition and the cold crystallization for PVAc-A and PVAc-MMT-DOAB were also obtained (Table 4). Because the kinetic compensation effect parameters have nothing to do with the experimental factors, the kinetic compensation effect equations could clearly explain the properties of glass transition and cold crystallization of PVAc-A and PVAc-MMT-DOAB and also were their theoretical expressions.
Kinetic compensation effect equations.
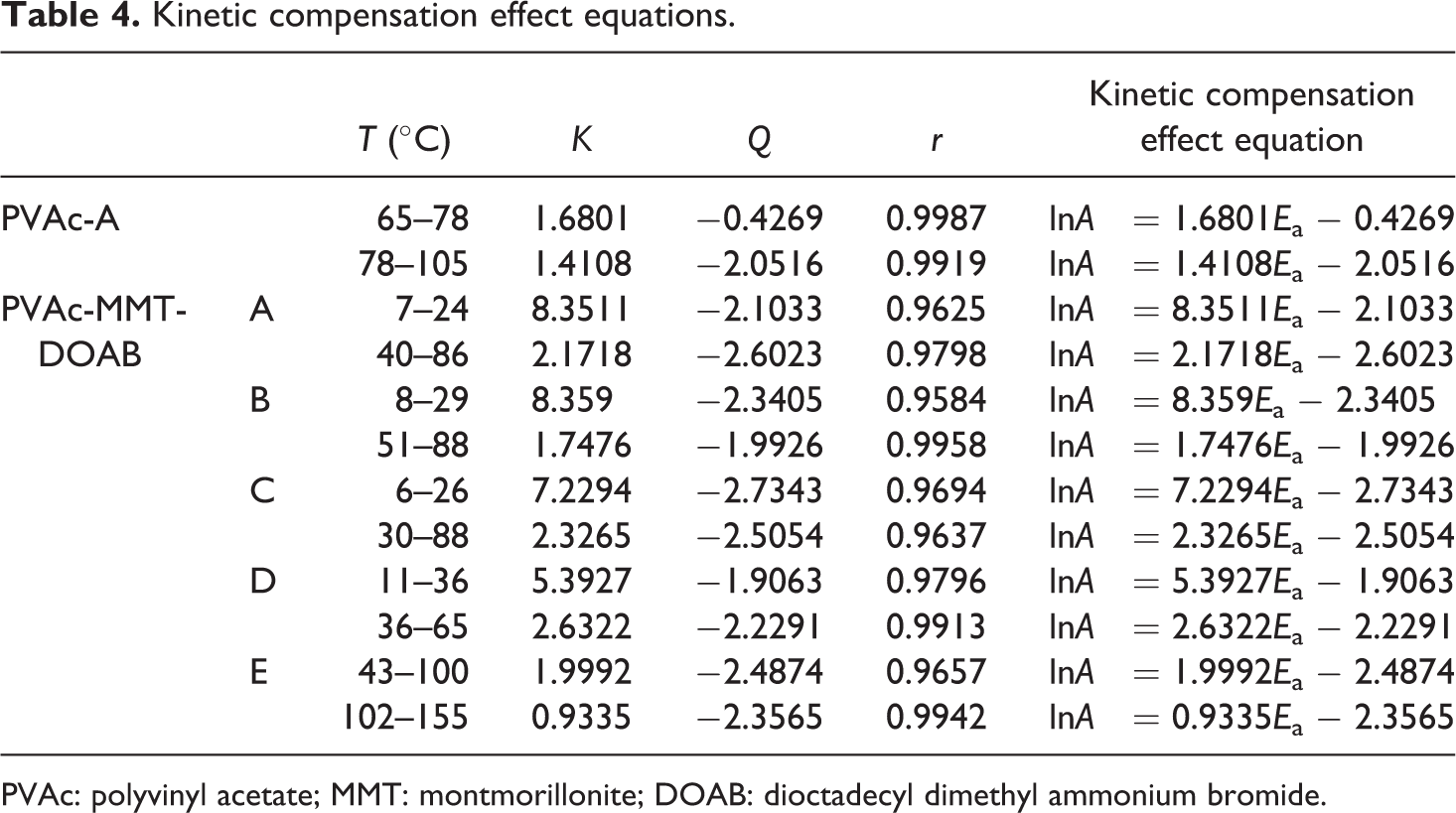
PVAc: polyvinyl acetate; MMT: montmorillonite; DOAB: dioctadecyl dimethyl ammonium bromide.
Conclusions
In the polymerization, linear macromolecular chains of PVAc were formed in MMT-DOAB layers, PVAc-MMT-DOAB was obtained through five different synthesis processes. No chemical bond existed between MMT-DOAB and PVAc but only physical effects existed. MMT-DOAB was exfoliated into the layers or sheets of nanoparticles, dispersed in PVAc matrix randomly. MMT-DOAB lowered the T g of PVAc and greatly improved its low temperature resistance. Both PVAc and PVAc-MMT-DOAB were all homogeneous amorphous linear polymers, and they all appeared cold crystallized. In addition, Agrawal integral equation was used to analyze the glass transition and cold crystallization kinetics of PVAc-MMT-DOAB; the kinetic parameters, including G(a), linear fit equation, r, E a, A, mechanism, n, and kinetic compensation effect equation, were obtained.
Footnotes
Funding
The authors are grateful for the financial support of the National Nature Science Fundation of China (30930074).
