Abstract
Nanocomposites prepared by the dispersion of unmodified and organically modified montmorillonite (MMT) clay into poly(2,6-dimethyl phenylene oxide)/polystyrene miscible blend in the range of 2–10 wt% clay were investigated by wide-angle x-ray diffraction, transmission electron microscopy, differential scanning calorimetry, thermogravimetric analysis and tensile mechanical tests. The systems based on unmodified sodium MMT (Na+MMT) as well as Cloisite 20A, Cloisite 30B and Cloisite 10A organically modified clays showed polymer intercalation. The glass transition temperature (
Introduction
In the past two decades, polymer–clay nanocomposites containing low-weight fraction of expandable layered silicates have gained importance due to the enhancements these materials show for bulk properties such as glass transition temperature (
Intercalated and exfoliated nanocomposites show enhancement of polymer matrix properties. 1,2,4,5 Melt-processing or melt-mixing method is a viable and versatile method for nanocomposite preparation. The advantage of this method being the small preparation time and the formation of mostly intercalated and partially exfoliated nanocomposites without the requirement of solvent or other chemicals. The melt-mixing method to clay/silicate-based polymer nanocomposite has been used for a variety of engineering thermoplastics such as poly(methyl methacrylate) (PMMA), 7 polystyrene (PS), 8,9 nylon 6 and 10 polycarbonate. 11
In the field of polymer–clay nanocomposites, relatively few studies exist especially pertaining to polymer–polymer blends.
12
–36
The poly(ethylene oxide) (PEO)/polymethyl-methacrylate (PMMA) organoclay nanocomposites prepared by the solvent casting have shown improved tensile mechanical modulus when compared with the polymer blend.
12
The study on the immiscible PMMA/PS blend system describes a partial level of compatibilization by organoclays as facilitated by a reduction in the size of the microdomains.
15
A substantial enhancement in the dispersion of organoclay in the polyvinylidene fluoride (PVDF) matrix was realized using PMMA as an interfacial agent at, as low as, 5 wt% in this miscible blend.
15
The dispersion of clay in this matrix resulted in an increase in shear viscosity and storage modulus. Melt-mixed PA6/PP nanocomposites prepared in the presence of maleic anhydride-
Clay nanocomposites with miscible polymer blend, as compared to a single-polymer matrix phase, have not received significant attention so far in the literature.
15,28,31,34
Recently, a study of styrene-acrylonitrile (SAN)/PMMA blend nanocomposite showed that intercalated nanocomposites are formed with commercially available Cloisite 25A MMT clay and with pristine (unmodified) MMT.
28
An insignificant increase in
It would be interesting to consider opportunities that miscible blends offer in terms of understanding the behavior of nanocomposites and toward the design of appropriate systems with tailored properties. Recently, clay nanocomposites of miscible blend PPO/PS prepared using Cloisite 10A organoclay having phenyl groups on the quaternary cationic surfactant modifier presented sufficient clay dispersion with an improved tensile modulus. 31 In case of PPO/PS blend, both the polymers PPO and PS are amorphous and molecularly miscible as observed in various characterization techniques such as optical microscopy, calorimetry and electron microscopy. 37 –39 The van der Waals and dipolar interactions between phenyl groups of PPO and PS give miscibility at the molecular level in this blend. 40 PS is brittle, while PPO is a ductile plastic. Hence, the blend exhibits a characteristic property from brittle material to ductile material and composition-dependent mechanical properties. PPO along with PS exhibits a broad range of outstanding properties for applications in computers and business equipment, in automotive industry, for electrical insulation, in telecommunications, electronics and many other industries. 41 The mechanical strength, thermal stability and flame retardancy are desirable properties for machine and appliance housings.
The objective of the present study is to investigate the effect of organic modification versus unmodified MMT clay for nanocomposite formation, glass transition (
Experimental
Materials
Na-MMT and organically modified clays were supplied by Southern Clay Products Inc. (Gonzales, TX, USA) in powder form. Cloisite® Na (MMT), a natural Na-MMT with cation exchange capacity (CEC) 92.6 mequiv/100 g was used as is without further modification. The organically modified clays used included Cloisite 10A (C10A) having dimethyl benzyl hydrogenated tallow quaternary ammonium as an organic modifier with a CEC value of 125 meq/100 g, Cloisite 30B (C30B) having methyl tallow bis-2-hydroxyethyl quaternary ammonium with a CEC value of 90 meq/100 g and Cloisite 20A having dimethyl ditallow quaternary ammonium with a CEC value of 90 meq/100 g. Here, HT or MT refers to hydrogenated tallow or tallow as be the source; however, the aliphatic tail length is a mixture of C14, C16 and C18 (65% by mol.) consistently in all three types of modifier molecules. For upto a difference of ±5 meq/100 g in the CEC value, the number of organic modifier cations per lattice unit cell of MMT clay would not differ by an integer, and therefore, the density of the modifiers on the ion-exchanged MMT surface of Cloisite 20A and Cloisite 30B or on the unmodified Cloisite Na-MMT would be the same. Cloisite 10A presents an overexchanged organoclay with the density of the modifier molecules being higher than that of the exchangeable Na+ ions. Therefore, in this clay, there is propensity for the organic modifiers to diffuse out of the interlayer space into the polymer matrix and act as diluents in the course of nanocomposite preparation at temperatures used in this study. Given the much lower density of organic modifier compared with silicate MMT, the differences in the molecular weights and CEC across the systems would not present a significant difference in the weight fraction of silicate across these organoclays for a fixed value of the organoclay loading. Therefore, mechanical properties were assessed on the basis of actual organoclay loading instead of silicate (MMT clay) loading. The exception being Cloisite 10A systems, wherein the excess organic surfactant (given 125 meq/100 g overexchanged concentration compared with 90 meq/100 g CEC value of the original unmodified Na+MMT) would leach out of the interlayer spacing during the processing of the hybrids and results in a reduction in the mechanical properties of the matrix phase of the nanocomposite (effectively, for the nanocomposite itself). The PPO/PS blend commercial sample grade Noryl 701 supplied by General Electric was obtained in the form of pellets and used as is without further modification.
Nanocomposite preparation
The blend samples and clays were dried at 110°C and 80°C, respectively, in an air circulatory oven for a time period of 8 h prior to mixing. The blend and required amount of organoclay (2, 4, 6, 8 and 10 wt%) were physically mixed in a glass beaker and further melt-blended in a Haake Rheocord® batch mixer (Waltham, MA, USA) (50 g scale) originally at 220°C, as test condition, which is a typical processing temperature for PPO/PS blend.
31
However, for the purpose of investigating a suitable lower processing temperature, in order to avoid degradation of organic modifier moieties, the samples were processed at 180°C successfully.
31
WAXD data for the organoclay systems in the present study as well as earlier study revealed the degradation of organic modifier at the higher processing temperature.
31
Therefore, lower processing temperature of 180°C was used for conducting the further study. The mixing speed and processing time were fixed at 60 r/min and 15 min, respectively, as these conditions are optimized from earlier studies on PMMA and PS nanocomposites processed in the range of 20–75 r/min.
7,31,42,43
The d001 spacing of the clay layers in the nanocomposites were found to be constant beyond 15 min of mixing time. The maximum change ▵d001 spacing was observed for the hybrids prepared at 60 r/min.
31
The approximate composition of the blend was determined using Flory-Fox equation and the measured
Characterization
Wide angle x-ray diffraction
The intercalation of PPO/PS molecularly compatible (miscible) blend in between the clay layers was confirmed using WAXD. The WAXD patterns were recorded on a Rigaku (Japan) diffractometer with CuKα radiation (wavelength
Transmission electron microscopy
Ultrathin sections of approximately 50–70 nm were cut with a diamond knife at room temperature with a Leica Ultracut UCT® microtome (Vienna, Austria). Sections were collected on 300 mesh carbon-coated copper grids and dried on filter paper overnight. TEM imaging was performed using a JEOL 1200EX electron microscope (Tokyo, Japan) operating at an accelerated voltage of 80 kV. Images were captured using charged couple detector camera and images were viewed using Gatan® digital micrograph software. The procedures followed were per the standard ones in the literature, which are similar to earlier investigations. 31,42,43
Thermal characterization
The onset of degradation as the temperature at 1 wt% loss (
Mechanical characterization and testing
Tensile properties of the nanocomposites were determined using rectangular strips prepared by compression molding, per procedure used earlier. 31 The PPO/PS nanocomposites could not be injection molded using known conditions for typical thermoplastics and this blend, due to their significantly high melt viscosity. Hence, rectangular strips were cut from the compression molded nanocomposite sheet. Films were prepared by compression at 185°C for 5 min using electrically heated Carver® press, and the hot plates were cooled using water circulation. The dimensions of the strips were 150 × 12 × 0.8 mm3. Compression molding parameters were optimized to get uniform sheets without any defects. The rectangular strips were obtained from the compression molded sheet using metal sheet cutter machine, and samples with smooth edges and without defects were selected for test.
The standard ASTM D882 test method typically used for thin plastic sheets with the thickness of <1 mm was used. The width/thickness ratio in this test method should be at least 8 and is ∼15 in the present cases. The Young’s modulus, the break stress (tensile strength at break) and the break strain (%) were determined using INSTRON® 4204 universal testing machine (Norwood, MA, USA) with 1 kN load cell. Grip separation was used as the means by which the strain was measured. The initial grip distance was 120 mm and the crosshead speed was maintained at 5 mm/min. All tests were performed at room temperature and 50% relative humidity. The data reported in all the cases for all the samples are the average of 10 independent measurements with standard deviations. Proper care was taken in selecting samples for the test, as in some cases, especially for Cloisite 10A-based systems, samples had to be prepared at least twice in order to obtain visually clear and macroscopically homogeneous molded sheet.
Results and discussion
Nanocomposite structure
The hybrids prepared at 220°C (the usual processing temperature for PPO/PS blend) clearly showed degradation of the organic modifier, similar to the observation made in the earlier study. 31 Under these mixing conditions, the depreciation (reduction) in the d001 spacing (data not shown specifically here) for different chemical systems was in the range of 0.3–0.5 nm over 2–8 wt% organoclay loading. By lowering the processing temperature to 180°C, the melt was easily formed within few minutes at 60 rpm, followed by a mixing time of 10 min. WAXD results (d001) for PPO/PS composites processed at 180°C are given in Table 1. The lower processing temperature results in intercalated nanocomposites without the degradation of the organic modifiers. This was confirmed by weight measurements via TGA (Table 2).
d-Spacing values (nm) for PPO/PS blend nanocomposite samples processed and prepared at 180°C.
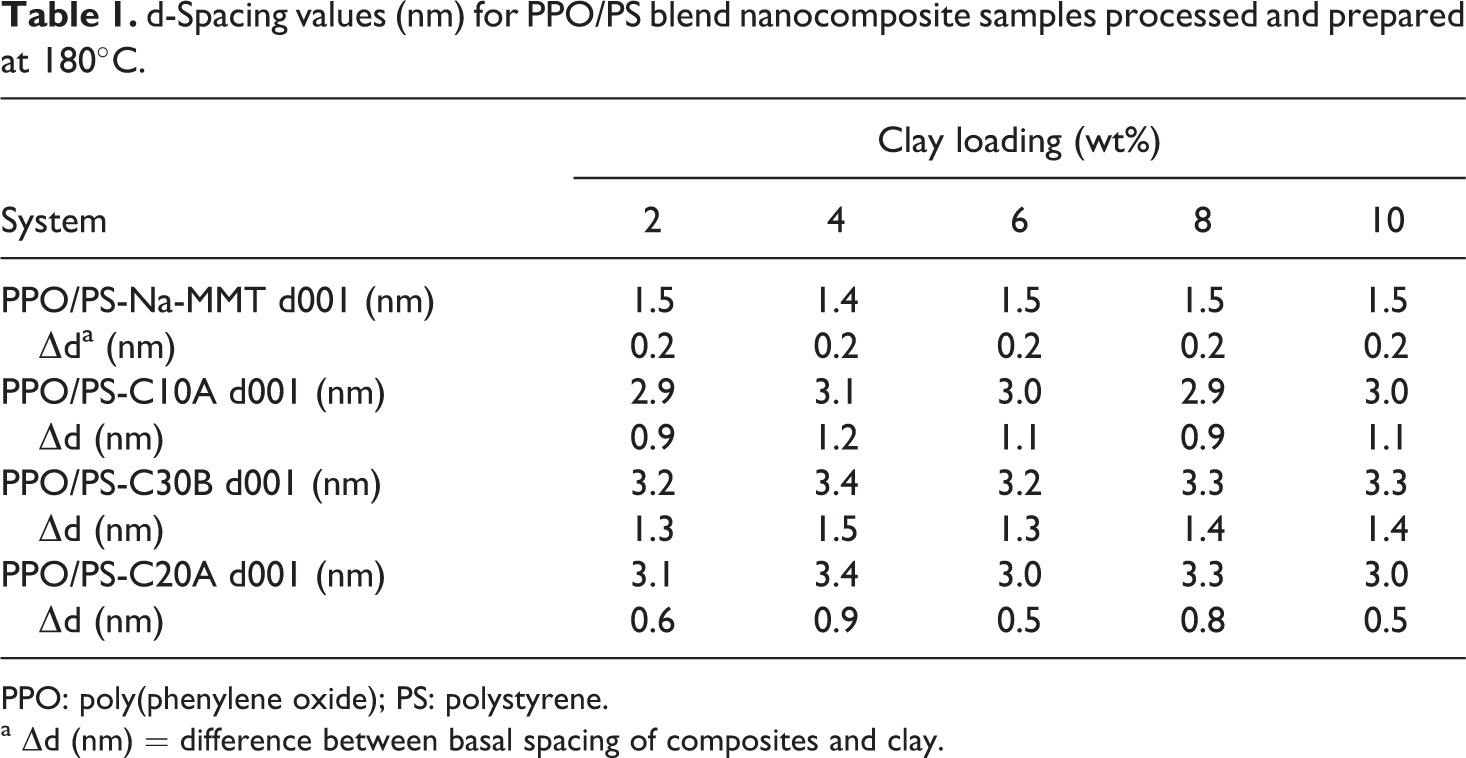
PPO: poly(phenylene oxide); PS: polystyrene.
a ▵d (nm) = difference between basal spacing of composites and clay.
Comparison of thermal degradation properties.
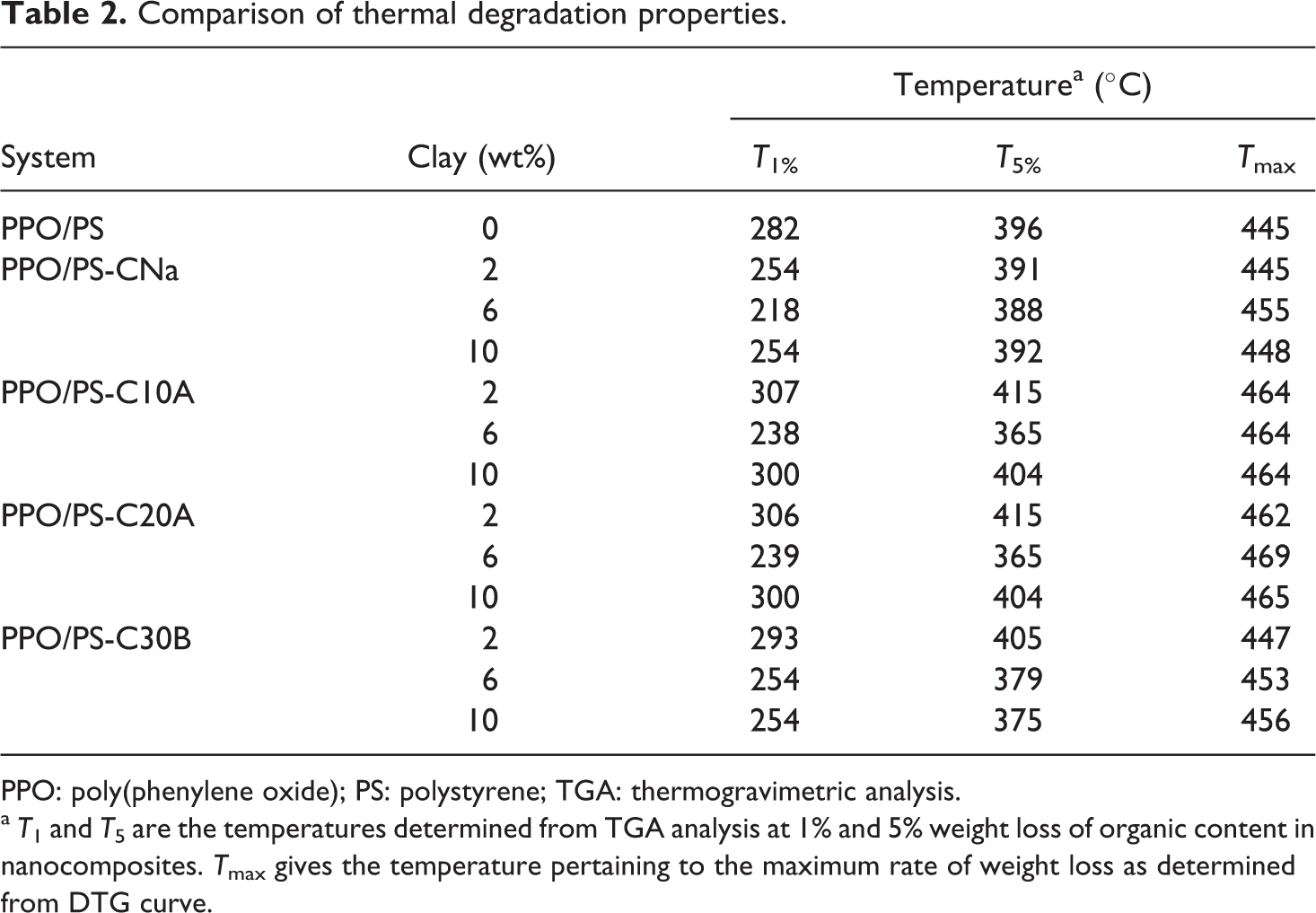
PPO: poly(phenylene oxide); PS: polystyrene; TGA: thermogravimetric analysis.
a
As seen in Figure 1 for unmodified Na-MMT-based hybrids, the nanocomposites reach the peak at 1.5 nm when compared with 0.9 nm for dehydrated Na+MMT and 1.3 nm for hydrated-Na+MMT. This shows polymer intercalation between the silicate layers. It can be inferred that PPO chains have intercalated into Na+MMT layers. It is known that PS does not intercalate into unmodified Na-MMT, while alkyl ammonium quaternary ion–modified MMT does promote intercalation. 8,47 This was also observed by our independent experiments on the PS-MMT system not reported here.
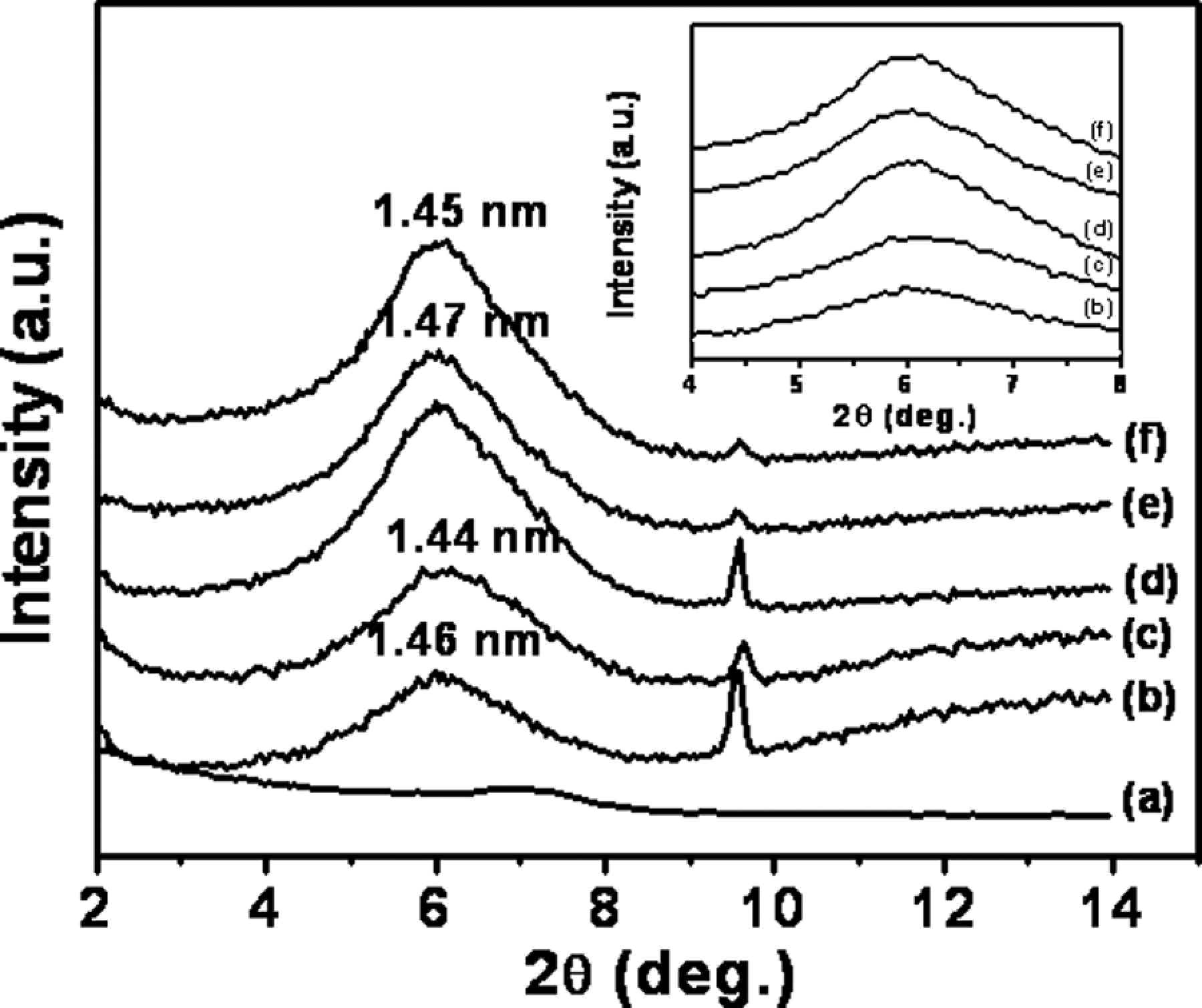
WAXD curves of PPO/PS-Na-MMT nanocomposites as a function of organoclay loading. (a) CloisiteNa clay, (b) 2 wt%, (c) 4 wt%, (d) 6 wt%, (e) 8 wt% and (f) 10 wt%. The d001 peak for montmorillonite CloisiteNa clay is not sharp as portrayed here, given the rescaling of intensity to relative measure for the purpose of plotting all systems on a common graph. Value of d001 peak for Na+MMT clay occurs at 2
As shown in Figure 2 for Cloisite 30B-based nanocomposites, the shifts of the x-ray diffraction (XRD) peaks toward lower 2
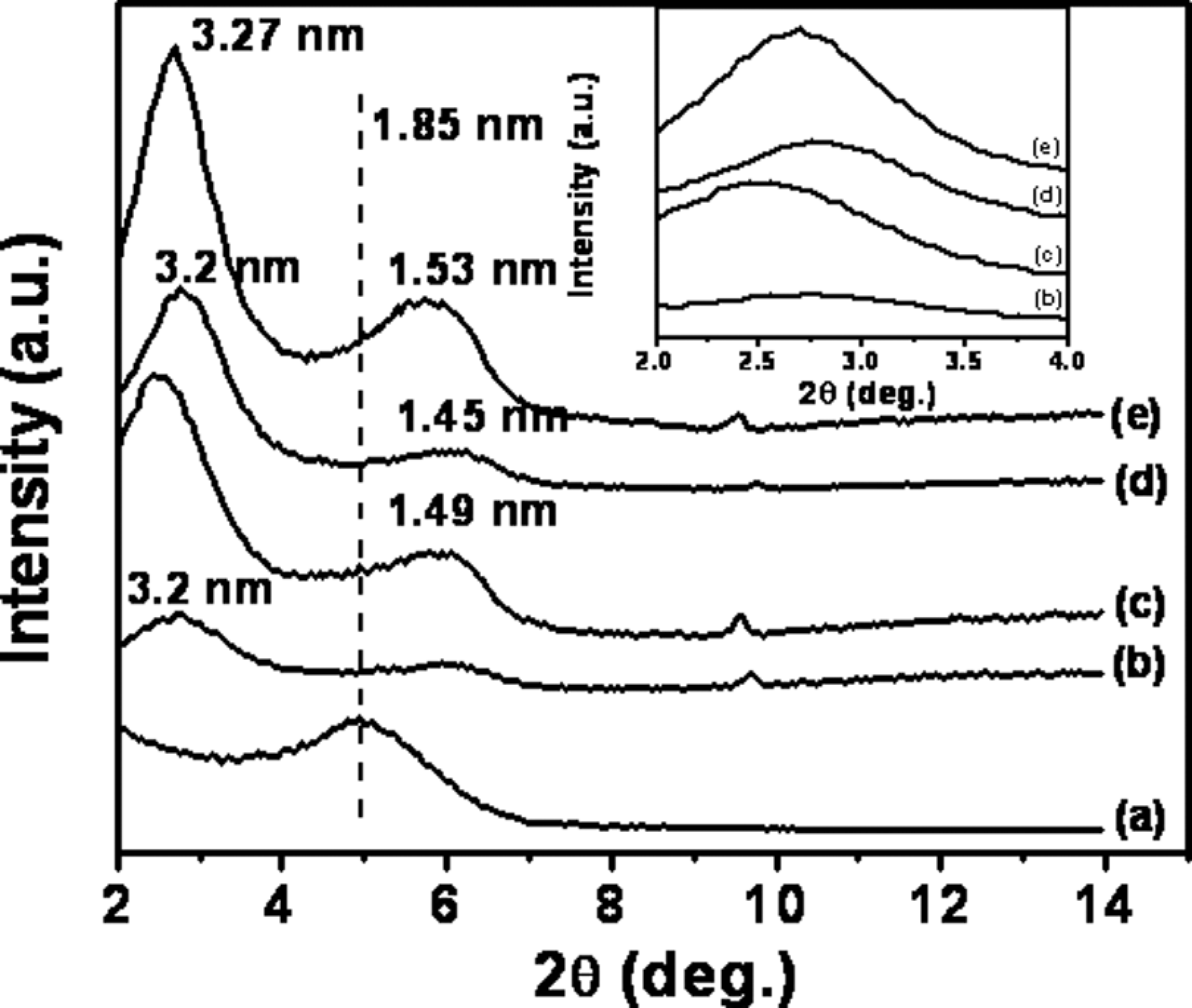
WAXD curves of PPO/PS-Cloisite 30B nanocomposites as a function of organoclay loading. (a) Cloisite 30B organoclay, (b) 2 wt%, (c) 4 wt%, (d) 6 wt%, (e) 8 wt% and (f) 10 wt%. Dashed vertical line shows the location of the organoclay d001 peak. WAXD: wide-angle x-ray diffraction; PPO: poly(phenylene oxide); PS: polystyrene.
As seen from Figure 3 for PPO/PS–Cloisite 10A hybrids (lower loadings given in earlier work 31 ), sufficient polymer intercalation is observed even at higher loadings (8 and 10 wt%). The intercalation change ▵d001 is found to vary in the range of 0.9–1.5 nm. Cloisite 10A contains dimethyl benzyl hydrogenated tallow quaternary ammonium modifier. The presence of the benzyl ring (due to dipolar interactions) in this modifier improves the thermodynamic compatibility between the organoclay and PS chains. 48,49 The ▵d001 value reported in the literature for PS–Cloisite 10A intercalated nanocomposite is in the range of 4.0–4.9 nm depending on the PS molecular weight and clay loading, as obtained by the use of higher shear in the specific twin-screw extruder configuration used. 49 If the excess organic modifier was not present (i.e. washed off chemically prior to the use of organoclay in melt-mixing with the PPO/PS blend), then a larger change in ▵d001 value would have been observed for the same final value of the nanocomposite d001. This confirms the earlier observation made on PS homopolymer system intercalated into cationic phosphonium and quaternary ammonium based (containing polar-substituted aromatic ring) on excess quantities that lessened the tendency of PS chains to penetrate the organoclay during melt processing, 50 likely due to blockage presented by the excess modifier between successive clay platelet layers.
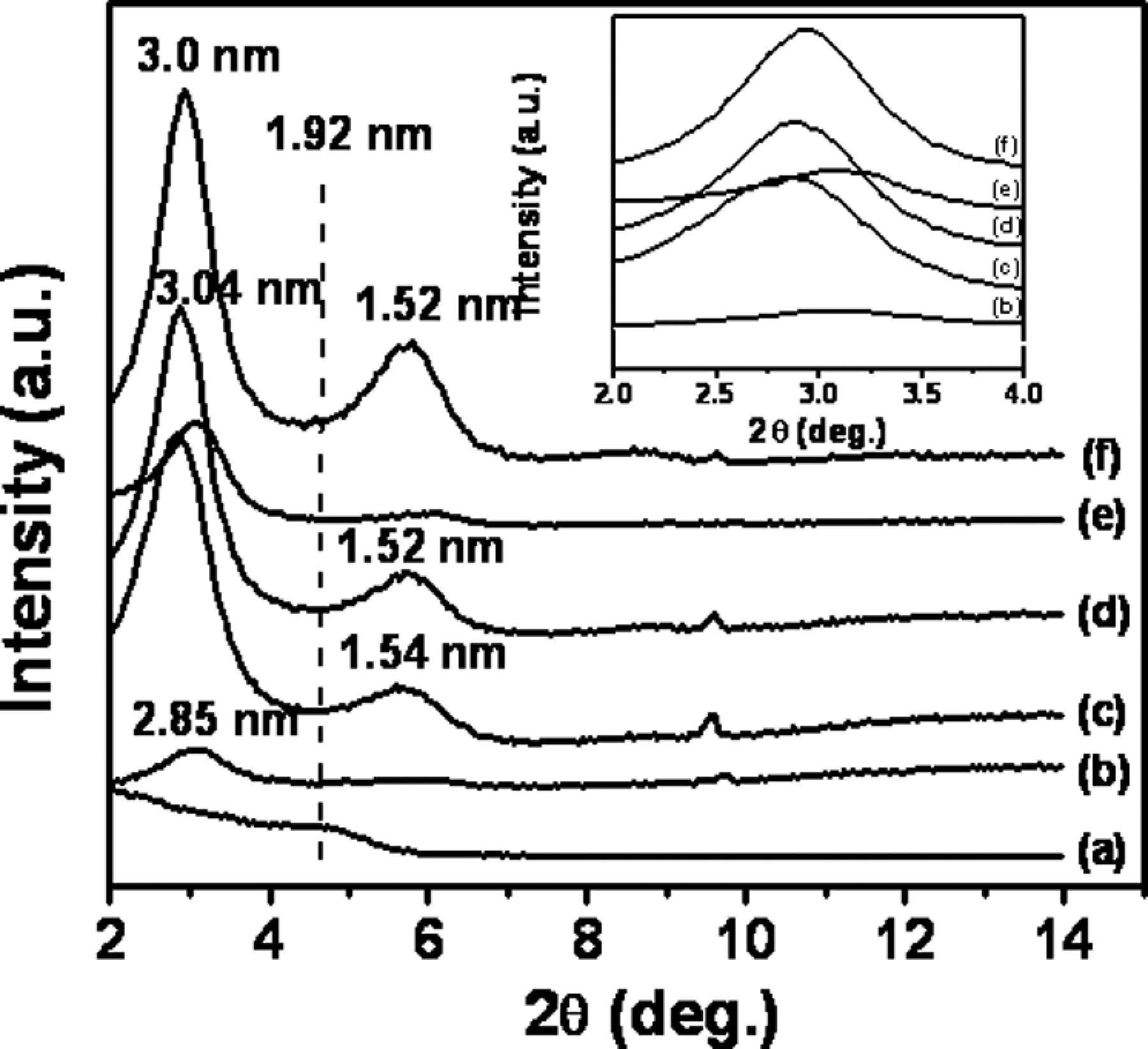
WAXD curves of PPO/PS-Cloisite 10A nanocomposites as a function of organoclay loading. (a) Cloisite 10A organoclay, (b) 2 wt%, (c) 4 wt%, (d) 6 wt%, (e) 8 wt% and (f) 10 wt%. Dashed vertical line shows the location of the organoclay d001 peak. Data for systems (a) to (c) are reproduced from Wiley, 2008. 31 WAXD: wide-angle x-ray diffraction; PPO: poly(phenylene oxide); PS: polystyrene.
Figure 4 shows the WAXD patterns for PPO/PS–Cloisite 20A nanocomposites. The value of ▵d001 change obtained due to polymer intercalation is lower than that seen in the nanocomposites with the other two organoclays. This is likely due to the very hydrophobic nature of the organic modifier in this organoclay. The largest value of ▵d001 in the Cloisite 20A nanocomposite occurs at the same clay loading (4 wt%) at which the best improvement in tensile modulus is observed, and this shows the percolation threshold observed in this system.
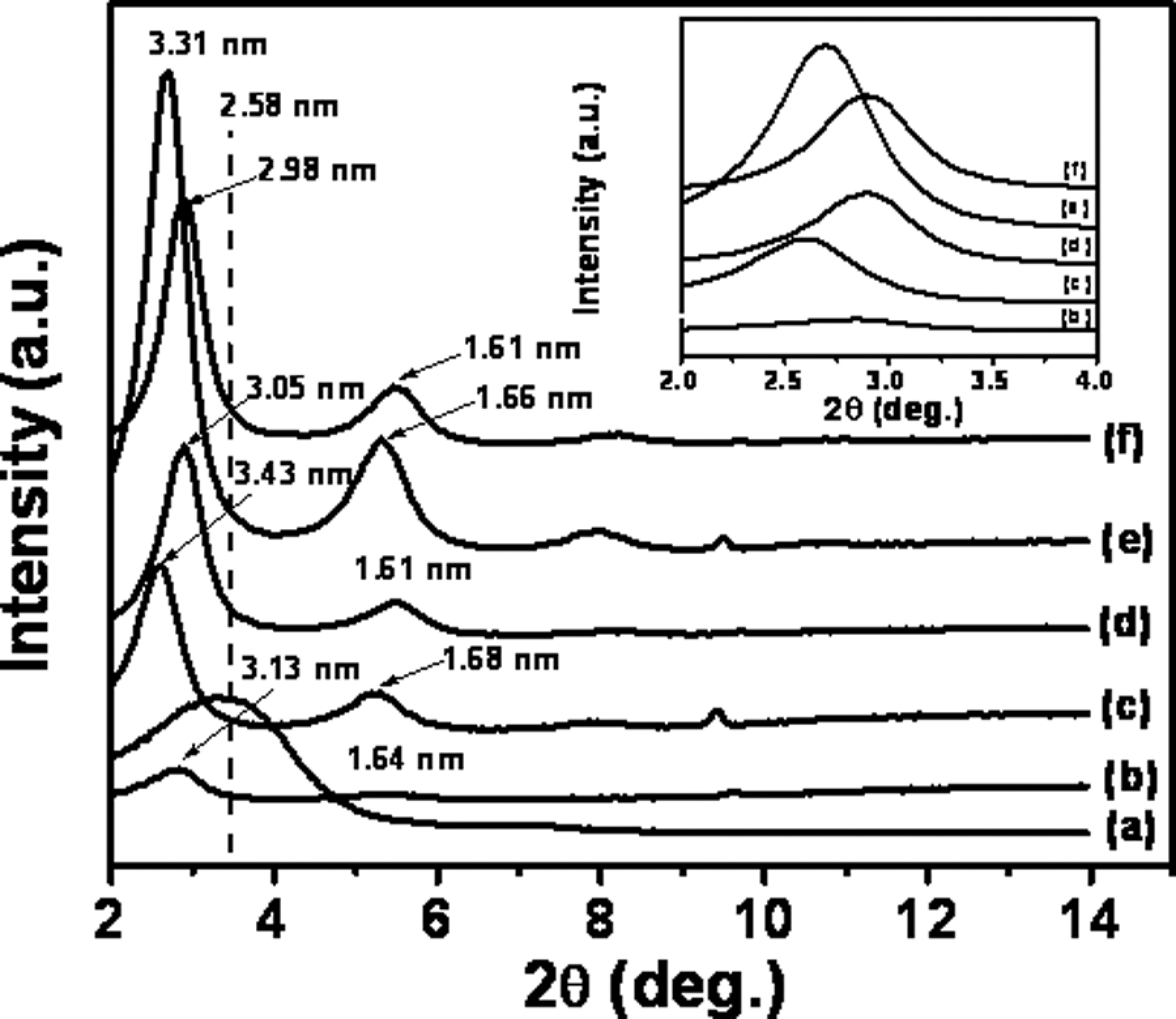
WAXD curves of PPO/PS-Cloisite 20A nanocomposites as a function of organoclay loading. (a) Cloisite 20A organoclay, (b) 2 wt%, (c) 4 wt%, (d) 6 wt%, (e) 8 wt% and (f) 10 wt%. Dashed vertical line shows the location of the organoclay d001 peak. WAXD: wide-angle x-ray diffraction; PPO: poly(phenylene oxide); PS: polystyrene.
The difference seen in ▵d001 between Cloisite 10A and Cloisite 30B systems, in going from organoclay to the polymer-intercalated nanocomposite, even though these contain organic modifier molecules of similar size and not too dissimilar polarity, can be rationalized by the fact that Cloisite 10A contains excess modifier, which if were washed away prior to nanocomposite formation should give similar d-spacing change as Cloisite 30B. Similarly, it should be noted that interlayer spacing increases in a step-wise manner in alkyl ammonium–modified MMTs as more longer alkyl substituents are added to the modifier molecule, consistent with the formation of a modifier monolayer, then a bilayer, and so on. Cloisite 30B and Cloisite 10A do not contain quite enough alkyl chains in the interlayer galleries to initiate the formation of the next monolayer whereas Cloisite 20A does. The addition of polymer would be expected to cause a large increase in d-spacing in the Cloisite 30B and Cloisite 10A cases than in Cloisite 20A case, even if the final d-spacings are similar in all cases.
TEM images are shown in Figure 5. As seen in Figure 5(a) clay tactoids are visible in the unmodified MMT system, suggesting the formation of a dispersed microcomposite rather than a nanocomposite. As seen in Figure 5(b) to (d) for intercalated nanocomposites, the dispersion of clay layers appears to be slightly better at 2 wt% (low loading) supported by the WAXD data, wherein low intensity and slightly broader peaks are observed for systems at 2 wt% when compared with those at 10 wt%. The TEM images at higher loadings show the presence of clay stacks and tactoids.
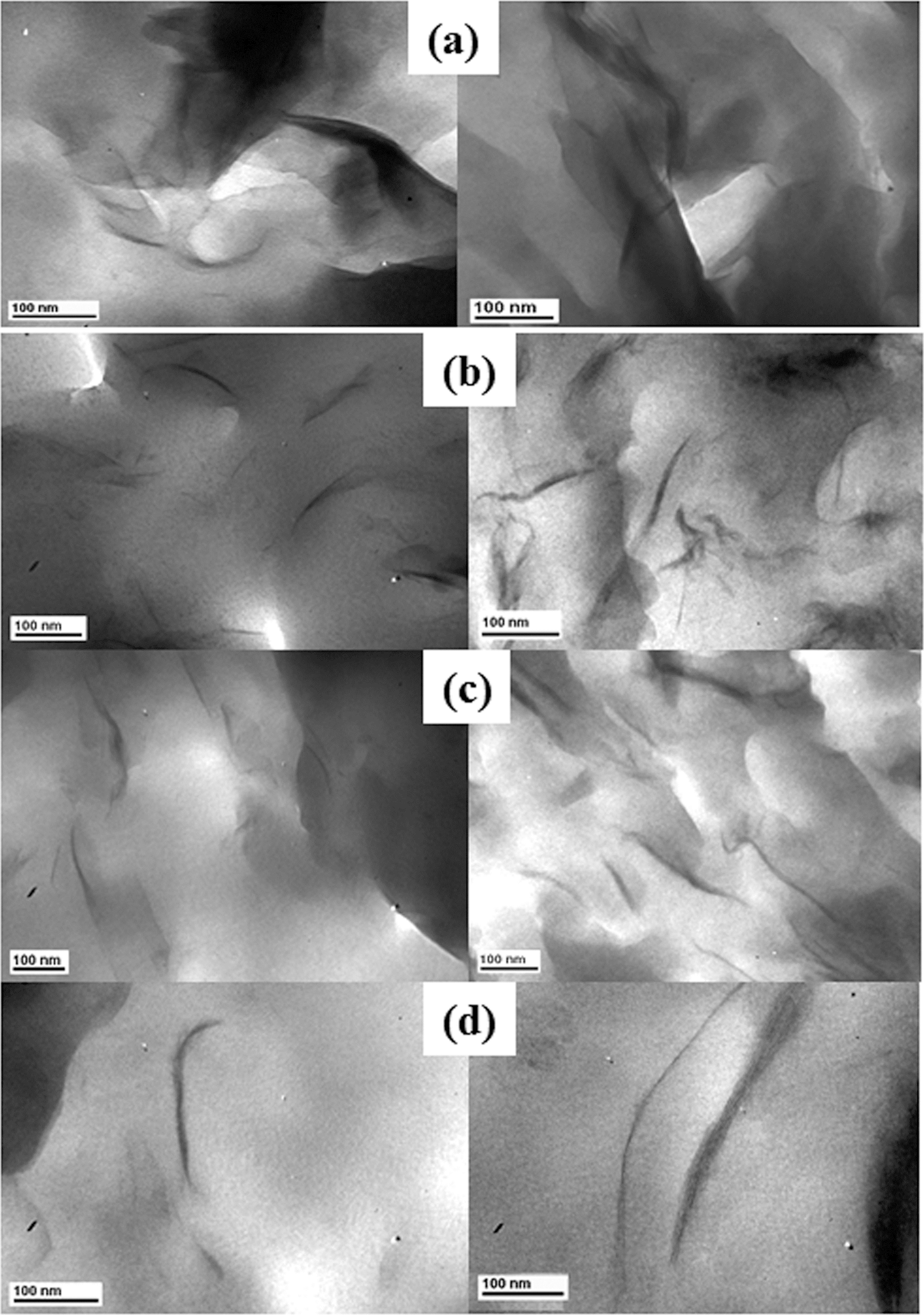
High-magnification TEM images of PPO-PS blend clay nanocomposites. Left-side images are for 2 wt% and right-side images are for 10 wt% clay except for C20A (4 wt%). (a) Na-MMT-based nanocomposite, (b) Cloisite 10A-based nanocomposite (image for 2 wt% loading taken from Wiley, 2008, 31 (c) Cloisite 20A-based nanocomposite and (d) Cloisite 30B-based nanocomposite. TEM: transmission electron microscopy; PPO: poly(phenylene oxide); PS: polystyrene; Na-MMT: sodium montmorillonite.
It is worth mentioning here that at present no consistent behavioral trend in particular can be ascribed to the role of the excess organic modifier toward the development of the nanocomposite structure, as observed from literature restricted to melt-processed nanocomposites. A more open (separated) organoclay structure due to the excess surfactant could enhance exfoliation during nanocomposite synthesis
51,52
or could improve mechanical properties of a poly(ethylene-
Glass transition and thermal degradation behavior
DSC data revealed that hybrids for all systems showed glass transition, but clay loading and confinement due to intercalation did not have a significant effect on the transition temperature. The CloisiteNa composites revealed approximately 5°C change in
TGA results are presented in Figure 6. It is known that
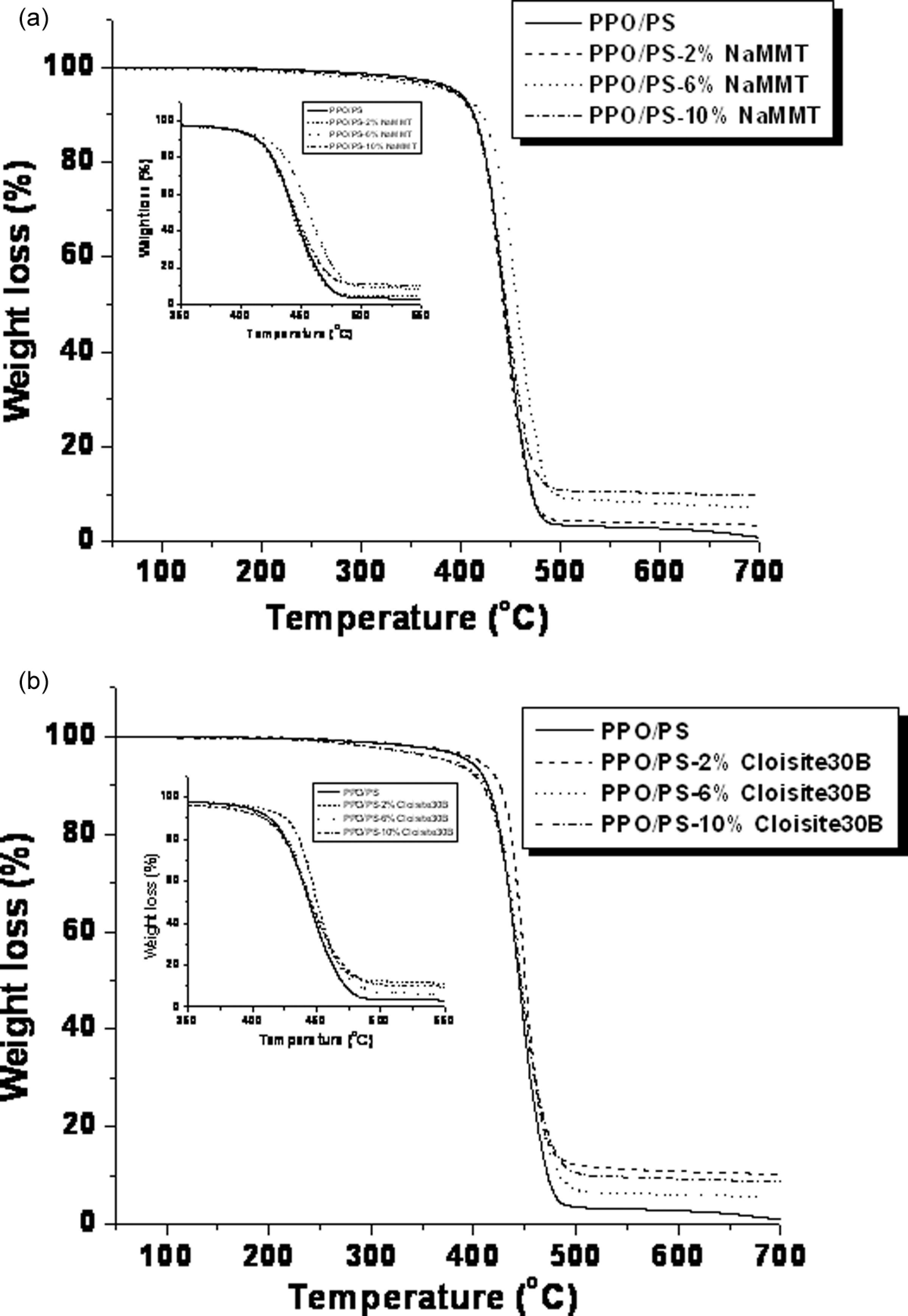
TGA thermographs; (a) PPO/PS-Na-MMT and (b) PPO/PS-Cloisite 30B. TGA: thermogravimetric analysis; PPO: poly(phenylene oxide); PS: polystyrene; Na-MMT: sodium montmorillonite.
Tensile properties
The Young’s modulus, tensile strength and tensile break strain data are presented in Tables 3 to 5 and Figure 7. Depending on the type of organoclay, the improvement in the modulus is 11–31%, with the lowest value pertaining to that for the weakly intercalated PPO/PS–Na-MMT hybrid and the highest value is obtained for the Cloisite 30B nanocomposite beyond 6 wt%. Such increase in modulus has been previously observed for melt-processed thermoplastic nanocomposites of 3–69% for PMMA, 42,43 4–24% for PS 9 and 25–66% for SAN 60 over 2–10 wt% MMT filler.
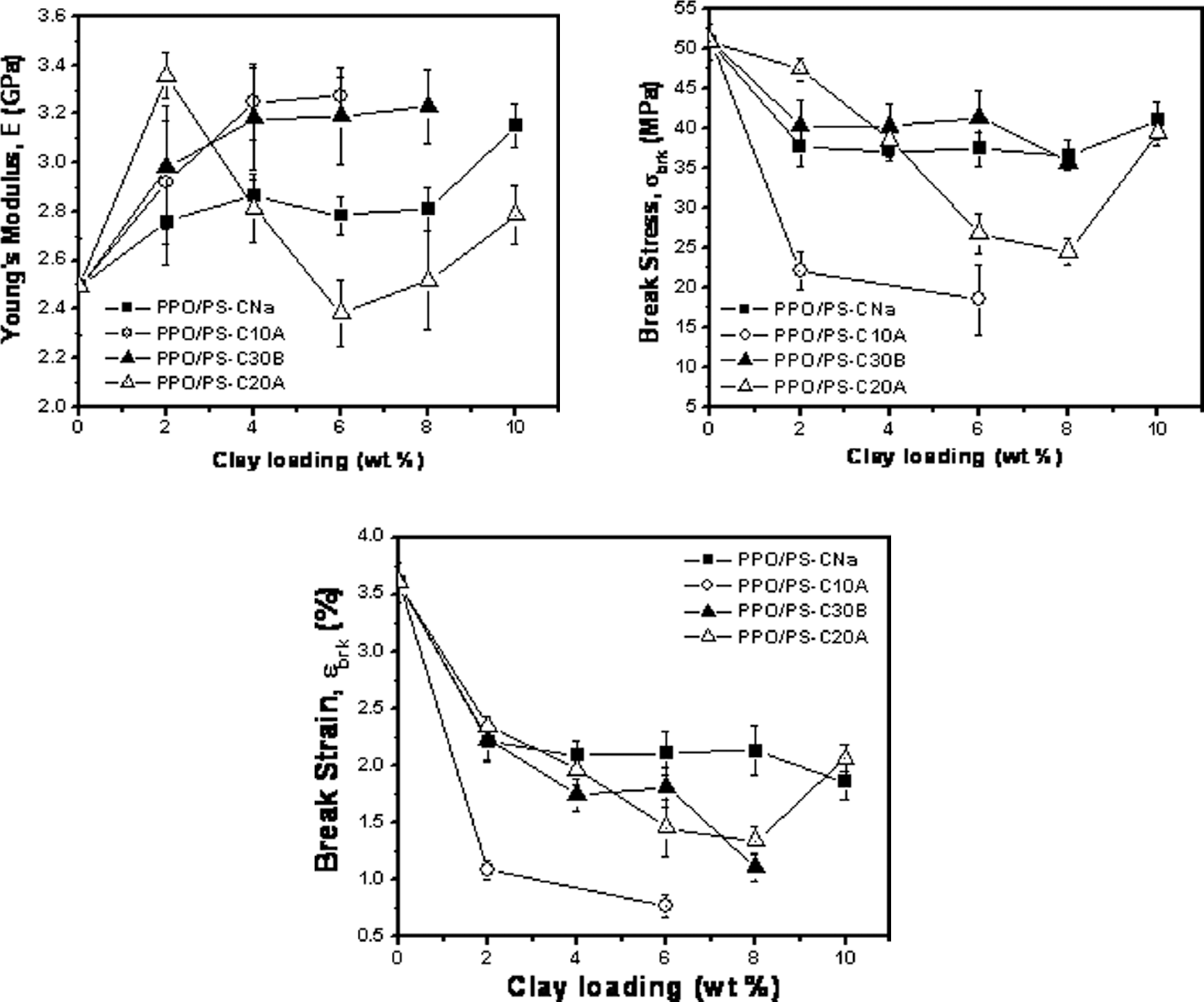
Variation in the mechanical properties of PPO/PS blend nanocomposite. PPO: poly(phenylene oxide); PS: polystyrene.
Young’s modulus (
PPO: poly(phenylene oxide); PS: polystyrene.
a Modulus of unfilled blend is 2.5 ± 0.2 GPa.
Break stress (
PPO: poly(phenylene oxide); PS: polystyrene.
a Break stress of unfilled blend is 50.8 ± 2.2 MPa.
Break strain (∊brk; %) of PPO/PS blend and nanocomposites.
PPO: poly(phenylene oxide); PS: polystyrene.
a Break strain of unfilled blend is 3.6 ± 0.17.
The intercalated nanocomposites and the composites show modulus enhancement as compared to the blend (modulus 2.49 GPa) at the lowest loading value (i.e. 2 wt%). All three organoclays give sufficient enhancement in the modulus. The Cloisite 20A-based systems showed significant loss of modulus at higher loading values; a behavior that is seen to be unusual and the understanding of which is beyond the scope of the present work. It is likely that in this particular system, clay loadings beyond the percolation threshold (seen at 4 wt% organoclay) lead to poorly structured composites. Cloisite 10A-based nanocomposites at 8 and 10 wt% organoclay loading were very brittle to be tested on the INSTRON.
The Young’s modulus of nanocomposite was calculated from Halpin-Tsai theory. The aspect ratio of clay stack were calculated using stack thickness as determined from Scherrer equation and stack length from TEM image using Gatan digital micrograph software (Warrendale, PA, USA). The variation in aspect ratio with clay loading was not significant so as to affect the modulus significantly. The Halpin-Tsai theoretical model was used to predict the stiffness of these nanocomposites as a function of the aspect ratio of the clay filler particles within the polymer blend matrix. The results of the calculation are given in Table 4 using structural parameters listed in Table 3 as obtained from the experiments discussed in this study. The number of clay platelets per stack in the nanocomposite samples was determined using the Scherrer equation with the data on d001 (
Tensile modulus and parameters for PPO/PS nanocomposites.
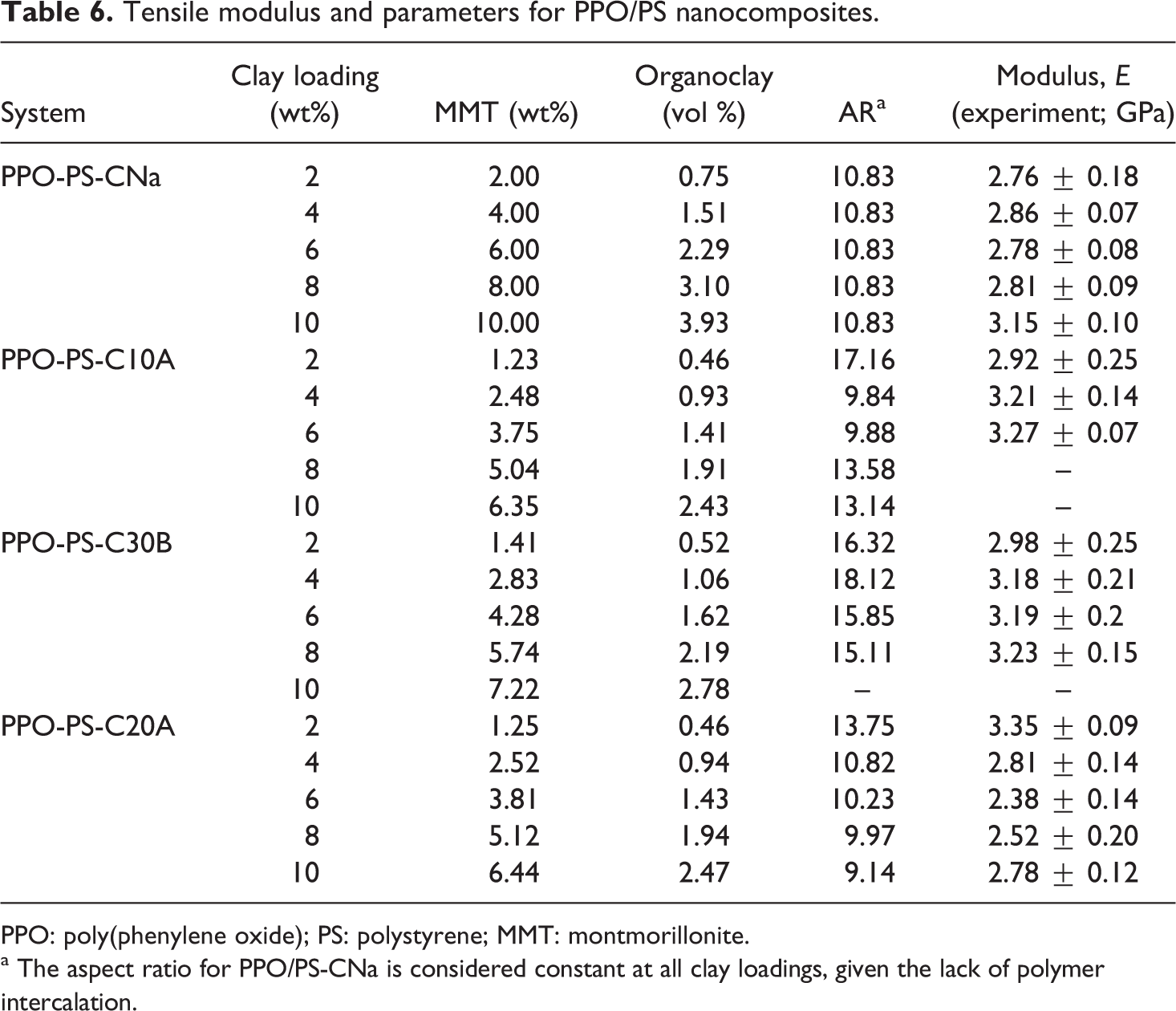
PPO: poly(phenylene oxide); PS: polystyrene; MMT: montmorillonite.
a The aspect ratio for PPO/PS-CNa is considered constant at all clay loadings, given the lack of polymer intercalation.
Compilation of structural parameters and Young’s modulus determined experimentally and obtained by Halpin-Tsai model.a
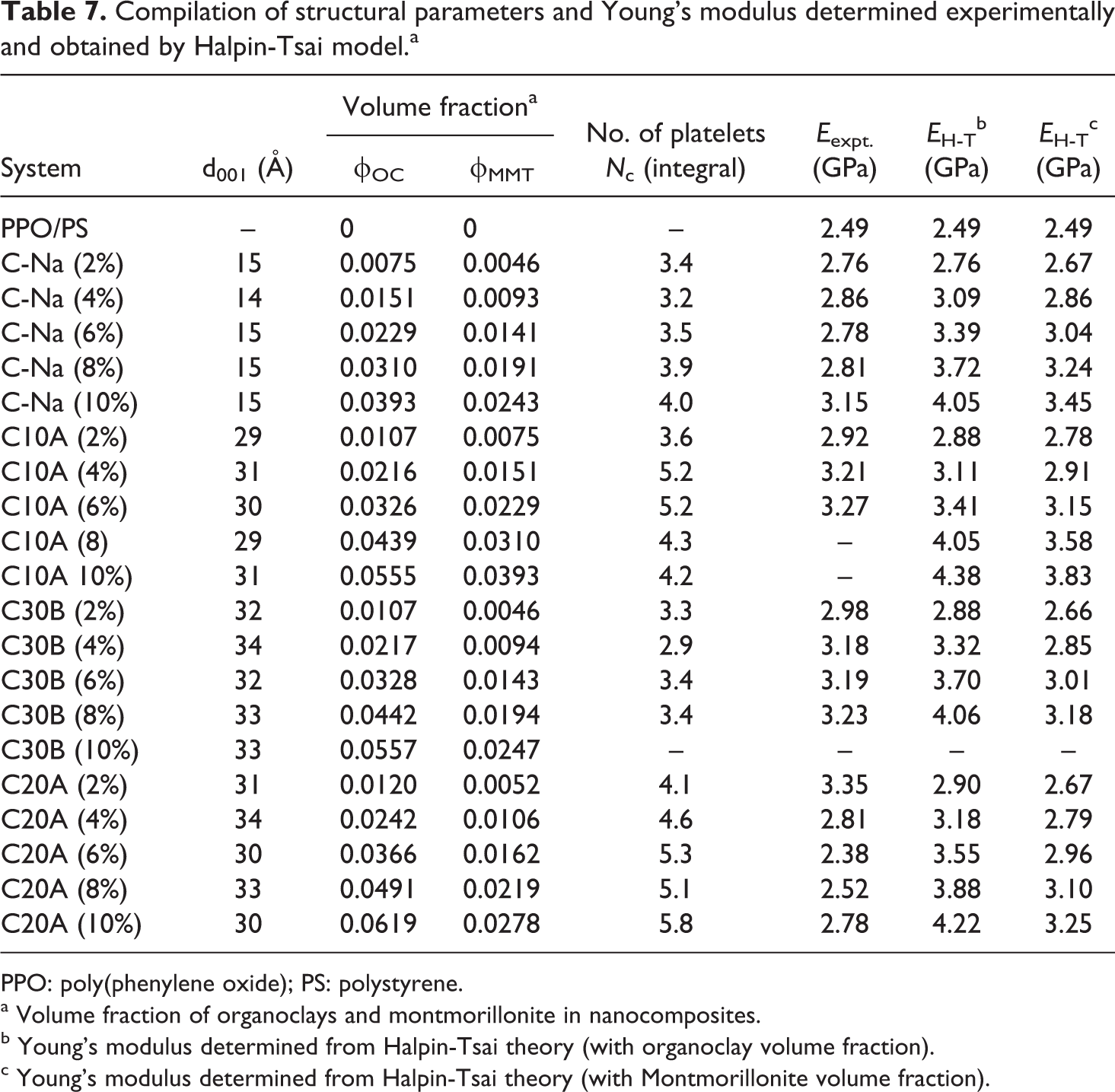
PPO: poly(phenylene oxide); PS: polystyrene.
a Volume fraction of organoclays and montmorillonite in nanocomposites.
b Young’s modulus determined from Halpin-Tsai theory (with organoclay volume fraction).
c Young’s modulus determined from Halpin-Tsai theory (with Montmorillonite volume fraction).
The depreciation in the break stress (as compared to neat blend matrix) of 19–64%, the lower value being for the Cloisite 30B nanocomposites as better than the Cloisite 10A nanocomposites investigated here and in earlier work. 31 The break strength for the systems based on CloisiteNa and Cloisite 30B does not depreciate significantly. In comparison, the Cloisite 10A nanocomposite system shows lower tensile strength likely due to the presence of excess surfactant (organic modifier). This is due to the presence of the excess organic modifier in Cloisite 10A over and above the stoichiometric amount. The minimum decrease in comparison with blend in percentage (loading in parenthesis) for each clay system was as follows: 26 (2 wt% for PPO/PS-CloisiteNa), 21 (4 wt% for PPO/PS-Cloisite 10A), 19 (6 wt% for PPO/PS-Cloisite 30B) and 6.7 (2 wt% Cloisite 20A). The effect of organic modifier and clay loading on break strength in PPO/PS blend nanocomposite corroborate with reported results for intercalated SAN nanocomposite, 60 except for the much lower value of break strength seen for Cloisite 10A system in the present study.
In the context of polymer-filler nanocomposite, in particular, it is known that the tensile strength for thermoplastic and thermoplastic blend nanocomposites reduces with clay loading. 9,31,42,43,61 The filler–filler interaction and inter-filler interaction causes clay particles to agglomerate, leading to a reduction in strength. 62 Bigg found increase in tensile strength for Noryl-talc composite, while tensile strength reduced for ABS-talc, with increase in talc composition, and this behavior was attributed to greater affinity of Noryl for talc arising from the presence of a coupling agent used with talc. 63 Leong et al. have shown either an increase or slight decrease in tensile strength of PP composites prepared with various inorganic fillers. The increase in the tensile strength was mainly due to the good filler–matrix interactions, which were largely due to the platy nature of talc and kaolin as compared to CaCO3. 64 The various factors such as matrix, crystallinity and filler type can have a significant effect on the tensile strength. More recent seminal literature on PP-clay nanocomposites 65,66 brings out the complex interplay between processing, microstructure (crystalline and noncrystalline fractions) and mechanical properties that needs to be understood in general for prediction and design of hybrid materials such as these for engineering applications.
The break strain (∊brk) of the nanocomposites studied in the present work is found to decrease with an increase in the clay loading. This qualitative trend is sufficiently general for brittle thermoplastic nanocomposites such as ABS, 61 PS 9 and PMMA 42,43 and other thermoplastic systems in the nanocomposite literature. The presence of dominating PS phase is responsible for the brittleness of the PPO/PS blend. Addition of clay to the blend matrix reduces the break strain by providing resistance to the elongation. The cloisite 10A organoclay leads to a significant reduction in the break strain, possibly due to the presence of the excess organic modifier.
Conclusions
PPO/PS blend nanocomposites were prepared by melt-mixing method with commercially available unmodified Na+-MMT and organically modified MMT clays Cloisite 10A, Cloisite 30B and Cloisite 20A. These were characterized for structure (WAXD and TEM), thermal behavior (DSC and TGA) and mechanical properties (tensile tests). XRD showed the formation of intercalated nanocomposites for the systems based on Na+MMT as well as organically modified clays. The ▵d001 change was in the range of 0.9–1.5 nm for the organically modified clays depending on the chemical nature of the organic modifier, while that observed for Na+MMT was only one molecular diameter of 0.2 nm. The
The PPO/PS systems prepared with Na-MMT and Cloisite 30B organoclay show a significant retention of tensile strength and break strain elongation. The composites showed improved modulus (upto 35% seen for Cloisite 20A-based system) as compared to the polymer matrix. The hybrids based on unmodified Na-MMT and Cloisite 30B (containing polar hydroxyl groups) showed good retention of tensile strength up to a significant clay loading level. The tensile strength (i.e. break-stress) is not depreciated significantly by the presence and distribution of clay platelets, in case of Cloisite 30B nanocomposites and the Na-MMT-based composites over 2–10 wt% clay loading range. Cloisite 10A organoclay containing excess organic modifier above the stoichiometric ratio in the MMT leads to a greater reduction in the tensile strength and the break strain of the nanocomposite, despite providing a similar level of increase in the tensile modulus as compared to the other organoclays. The observed behavior of modulus, strength and elongational break strain (which gives some measure of ductility) is found to be useful from a practical standpoint.
Footnotes
Acknowledgments
The authors thank the Council of Scientific and Industrial Research (CSIR), India, for providing a Graduate Senior Research Fellowship for unrestricted research (to RRT) and appreciate the availability of excellent experimental facilities in the Division of Polymer Science and Engineering and in the Center for Materials Characterization (CMC) at the National Chemical Laboratory, Pune; the authors also thank Southern Clay Products, Inc., Texas, and appreciate the provision of the clay samples.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
