Abstract
In this study, an epoxy resin was dynamically cured in a polypropylene (PP)/maleic anhydride–grafted polypropylene (MAH-g-PP)/calcium carbonate (CaCO3) matrix to prepare dynamically cured PP/MAH-g-PP/CaCO3/epoxy composites. The torque measurement shows that the addition of epoxy resin into the PP/MAH-g-PP/CaCO3 composites results in a slight increase in the torque at equilibrium, and epoxy resin in the composites has been cured by 2-ethylene-4-methane-imidazole (EMI-2,4). Scanning electron microscopic analysis shows that dual compatibilizers composed of MAH-g-PP and an epoxy resin were demonstrated to effectively compatibilize the immiscible PP/CaCO3 composites. MAH-g-PP with a low MAH content is miscible with PP to make it quasi-functionalized, while the epoxy resin can react with MAH-g-PP and hydroxyl groups at the CaCO3 surface. Thus the formed MAH-g-PP-co-epoxy-co-CaCO3 copolymer at the interface is able to anchor along the interface and serve as an efficient compatibilizer. Dynamic cure of the epoxy resin can further increase the interaction adhesion in the PP/CaCO3 composites. The epoxy resin does not show compatibilization effects for the PP/CaCO3 composites without the presence of MAH-g-PP. The shift of crystallization peaks to the higher temperature suggests that the CaCO3 filler in the composites can act as a nucleating agent, accelerating the crystallization of PP component, and the PP/CaCO3 composites compatibilized by MAH-g-PP and the epoxy resin have higher crystallization peaks. This shows that an increase in the interaction adhesion between the CaCO3 and PP matrix, improving the nucleating activity of the filler and promoting crystallization of PP. Thermogravimetric analysis shows that the incorporation of the CaCO3 into the PP could improve the thermal stability of PP, and the addition of MAH-g-PP and the epoxy resin into the composites obviously could further improve the thermal stability. The mechanical properties of PP/MAH-g-PP/CaCO3 and PP/MAH-g-PP/CaCO3/epoxy composites are better than that of the PP/CaCO3 composites. The PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 composites have the best mechanical properties in all the PP/CaCO3 composites, which is attributed to the better interaction adhesion. The suitable content of epoxy resin is about 5 wt% in the presence of MAH-g-PP in the PP/CaCO3 composites, while an excess epoxy resin of above 5 wt% acts as an organic filler for PP.
Introduction
The addition of fillers to polymers is a fast and cheap method to modify the properties of the base materials. 1 For this reason, particulate-filled polymers have been, and are, a subject of increasing interest in both industry and research. In the last decade, polypropylene (PP) and its blends have been found to be interesting matrices for filled materials, due to their combination of remarkable properties and low cost. 2,3 Among the fillers for PP, mica, 3 –6 calcium carbonate (CaCO3) 2,3,7 and talc 8 –10 are the most often used. Each filler brings its own characteristics to the composites, but it may also influence the crystallization of PP and, as a consequence, the properties of the composites.
The properties of the composites strongly depend on the interfacial adhesion between a filler and the polymer matrix. A good interfacial adhesion may improve the mechanical properties of the composites, while a poor adhesion may decrease the mechanical properties. 10 To enhance the mechanical properties of the composites, coupling agents were used to modify the surface of the fillers. The ordinary coupling agents are silane and titanate coupling agents, and other small molecular coupling agents. Another method is the modification of the matrix. Grafting of different chemical moieties to a polyolefin chain is a frequently applied method to improve the compatibility of the immiscible polymer blends and the interfacial adhesion in the composites. PP, a nonpolar polymer, is usually grafted with polar groups like acrylic acid, maleic anhydride (MAH), acrylic esters and so on. Functionalized polyolefins were used as a coupling agent in filler/polyolefin composites. 11 –18 The interaction between matrix and filler was improved, and the mechanical properties of the composites were enhanced. Low-molecular-weight coupling agents including silanes, titanium esters, phosphate, aluminate, and zirconate, cannot form a strong adhesion bond between nonpolar polyolefin and the filler. Although they can create a relatively strong bond with the filler surface, do not form a chemical bond with either the nonpolar polyolefin molecules and physical entanglements causing an improvement in polyolefin cohesive strength. 10 Functionalized polyolefins can create strong bonds with the filler surface and form physical entanglements with the matrix in the bulk. The effect of functionalized polyolefins on PP composites filled with mica, 11 –13 CaCO3 14 –16 and reinforced with glass fiber 17 –19 has been investigated.
At present, a series of investigations on in situ compatibilization of various blends using epoxy resins as compatibilizers have been reported. 20 –22 In this study, a combination of the MAH-grafted polypropylene (MAH-g-PP) and an epoxy resin was used as dual compatibilizers for the PP/CaCO3 composites. The MAH-g-PP with a low MAH content is totally miscible with PP to make the PP phase quasi-functionalized, so that the epoxy resin has the chance to contact and react with hydroxyl groups at the CaCO3 surface and MAH-g-PP, simultaneously to form MAH-g-PP-co-epoxy-co-CaCO3 copolymers at the interface. These desired copolymers are able to anchor along the interface and are expected to serve as efficient compatibilizers. The addition of the curing agent (EMI-2,4) can further increase the interaction adhesion between the CaCO3 and PP matrix. We intend to report their specific compatibility and correlation with their resultant morphological, thermal and mechanical properties.
Experimental
Raw materials
PP, F401, was supplied by Yangzi Petrochemical Co., Ltd (China), with a melt flow index of 1.9 dg/min (230°C, 2.16 kg). Mechanical properties of PP are shown in Table 1. Diglycidyl ether of bisphenol-A resin (M w: 1120 g/mol) was supplied by Shanghai Resin Co., Ltd (China). The curing agent, 2-ethylene-4-methane-imidazole (EMI-2,4), was purchased from Shanghai Chemical Agent Company (China). MAH-g-PP with a MAH content of 1% was supplied by Shanghai Sunny New Technology Development Co., Ltd (China). CaCO3 nanoparticles were supplied by Shanghai PLT Composite Materials Co., Ltd (China).
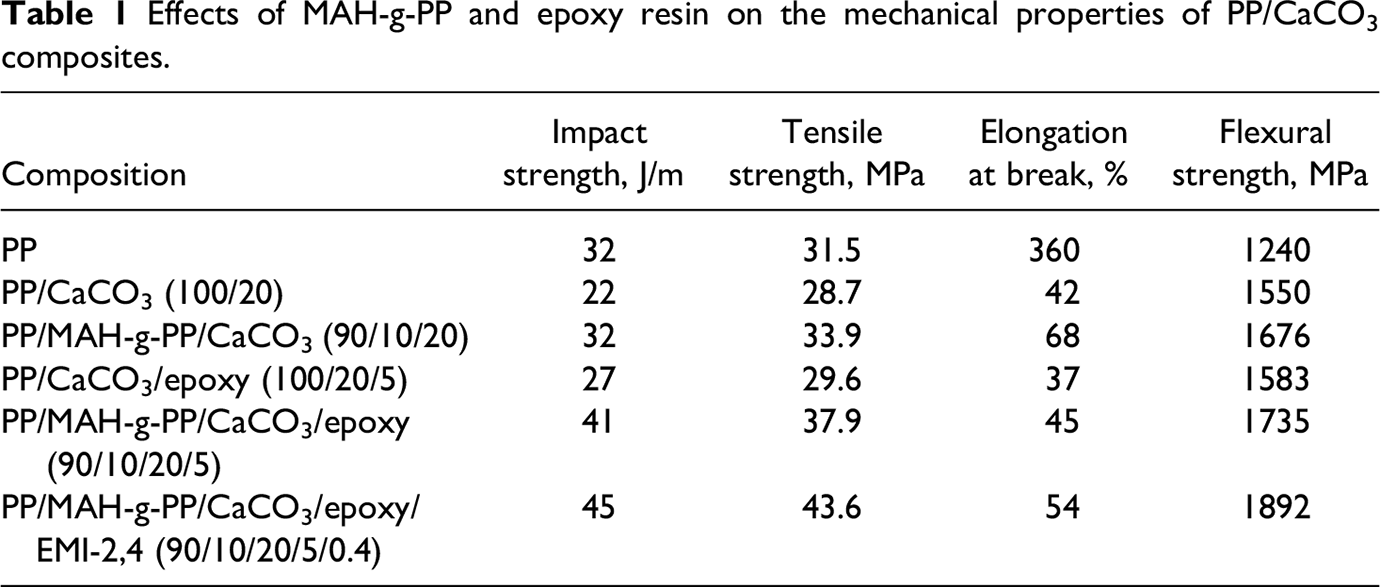
Effects of MAH-g-PP and epoxy resin on the mechanical properties of PP/CaCO3 composites.
Sample preparation
Before mixing, MAH-g-PP, the CaCO3 and epoxy resin were dried at 80°C under vacuum for about 8 h. The PP/CaCO3 composites were prepared in the mixing chamber of a Haake Rheometer RC90 at 190°C and 50 r/min. MAH-g-PP and PP were first mixed for 2 min, then the epoxy resin and CaCO3 were added. Two minutes later, the curing agent (EMI-2,4) was added to the mixture. The total mixing process lasted for about 6 min. The composites were moved out and compression molded in a press at 195°C for 5 min, then cold pressed to give samples for testing.
Measurements of mechanical properties
The tensile properties were measured using a Tensile Tester (Instron 4465, USA) at a crosshead speed of 20 mm/min according to American Society for Testing and Materials (ASTM) D638. Notched izod impact strength was measured using a Ray-Ran Universal Pendulum Impact Tester (UK) at an impacting pendulum speed of 3.5 m/s according to ASTM D256. Flexural properties were measured using the Instron 4465 Tester and a three-point-loading rig, and the central head was loaded on the specimen at a speed of 1.7 mm/min according to ASTM D790.
Scanning electron microscopy
The samples were fractured under liquid nitrogen coated with the gold and observed using a Hitachi S-2150 scanning electron microscope (SEM).
Differential scanning calorimetry analysis
Crystallization behavior was analyzed using a Perkin-Elmer differential scanning calorimetry (DSC; Paris 1, USA) in a nitrogen atmosphere. For each test, 4–5 mg of sample was first heated to 200°C at 50°C/min and then maintained at this temperature for 5 min in order to eliminate any previous thermal history. The specimen was subsequently cooled down to 40°C at a cooling rate of 10°C/min and then heated to 200°C at 10°C/min for data collection.
Thermogravimetric analysis
The thermogravimetric analysis (TGA) was carried out in a Perkin–Elmer TGA7 (USA). The samples were scanned from 30 to 800°C at a heating rate of 20°C/min in nitrogen atmosphere.
Results and discussion
Torque versus time
Torque measurements can be used to give qualitative information concerning the chemical reactivity and the extent of reaction in the polymer blends. 23 Figure 1 shows the relationship between torque versus time for the PP/CaCO3 composites at 190°C. The torque at equilibrium of the PP/MAH-g-PP/CaCO3 (90/10/20) composite is slightly higher than that of the PP/CaCO3 (100/20) composite, and the addition of 5 wt% epoxy resin into PP/MAH-g-PP/CaCO3 (90/10/20) composite gives rise to an increase in the torque at equilibrium. The PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 (90/10/20/5/0.2) composite has the highest torque at equilibrium in all the PP/CaCO3 composites. As MAH-g-PP has active maleic anhydride (MAH) groups, MAH-g-PP in the PP/MAH-g-PP/CaCO3 (90/10/20) composite can create with hydroxyl groups at the surface of CaCO3 and gives rise to an increase in the viscosity compared with the PP/CaCO3 (100/20) composite. For the PP/MAH-g-PP/CaCO3/epoxy (90/10/20/5) composite, an increase in the torque at equilibrium due to the presence of the epoxy resin indicates that the epoxy resin in the composite is involved in the interaction adhesion between functionalized PP and CaCO3. We thought that epoxy resin can react with MAH groups of MAH-g-PP and hydroxyl groups at the CaCO3 filler surface, and leads to a higher viscosity in the PP/MAH-g-PP/CaCO3/epoxy (90/10/20/5) composite. Shieh et al. 24 have reported that MAH-g-PP and a multifunctional epoxy resin were used as dual reactive compatibilizers for the PP/poly(butylene terephthalate) (PBT) blends. It can be seen that the addition of the curing agent (EMI-2,4) at the mixing time of 4 min leads to an obvious rise of torque. This shows that the epoxy resin has been cured by EMI-2,4 during its mixing with the molten PP. It can be seen that the addition of the curing agent (EMI-2,4) can further increase the torque at equilibrium, which may be attributed to the higher viscosity of cured epoxy resin and the better interaction adhesion between PP and the CaCO3.
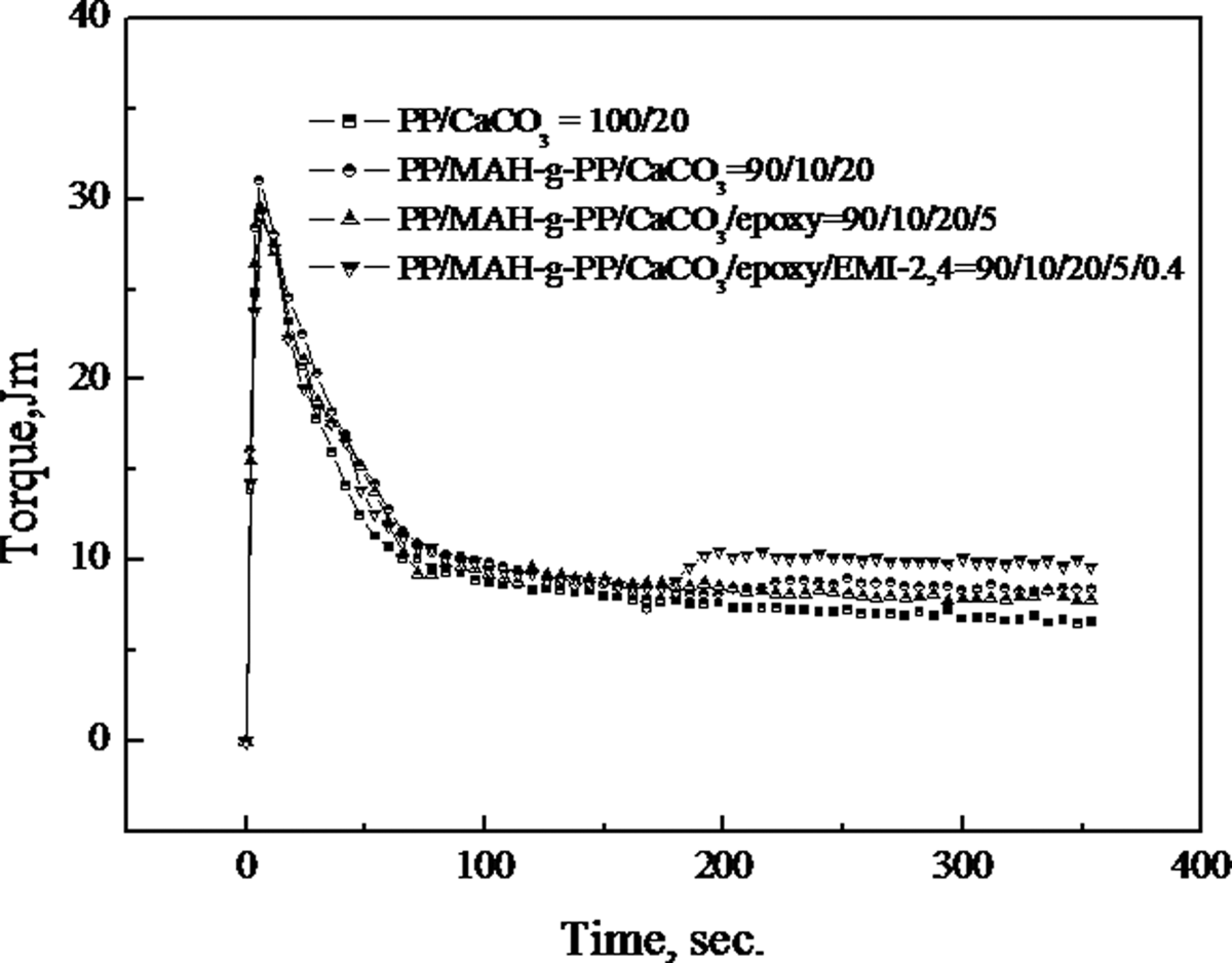
Plots of torque versus time for the PP/CaCO3 composites at 190°C. CaCO3: calcium carbonate; PP: polypropylene.
SEM analysis
Figure 2 shows SEM micrographs of the fracture morphology of the PP/CaCO3 composites. For the PP/CaCO3 (100/20) composite (Figure 2(a)), the CaCO3 is difficult to disperse in the PP matrix and some CaCO3 gather with a size of 0.3–0.8 μm in the composite, and the boundaries between the CaCO3 and the PP matrix are distinct, it shows that CaCO3 and PP are immiscible. While for the PP/MAH-g-PP/CaCO3 (90/10/20) composite (Figure 2(b)), the CaCO3 is able to disperse in the PP matrix, the granule of CaCO3 in the composite cannot be seen. It can be concluded that MAH-g-PP can improve the interaction between PP and the CaCO3. Figure 2((c) and (d)) shows the fracture morphology of PP/MAH-g-PP/CaCO3/epoxy (90/10/20/5) and PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 (90/10/20/5/0.4) composites. The boundaries between the CaCO3 and PP become more obscure, some of CaCO3 in the composites cannot be seen. As MAH-g-PP contains active MAH groups, the functional groups can not only react with the hydroxyl or epoxy groups of the epoxy resin but also react with hydroxyl groups at the surface of CaCO3. We thought that the reactions taken place in the PP/MAH-g-PP/CaCO3/epoxy composites and form an anchor for promoting the interfacial adhesion between the CaCO3 and PP matrix. Shieh et al. 24 reported using MAH-g-PP and a multifunctional epoxy resin as dual reactive compatibilizers for PP/PBT blends. Dynamic cure of epoxy resin can further increase the interfacial adhesion. The fracture morphology of the PP/CaCO3/epoxy (100/20/5) composite (Figure 2(e)) is similar to that of the PP/CaCO3 (100/20) composite, it shows that the epoxy resin do not act in the absence of MAH-g-PP, which is attributed to the different polarity of the epoxy resin and PP.
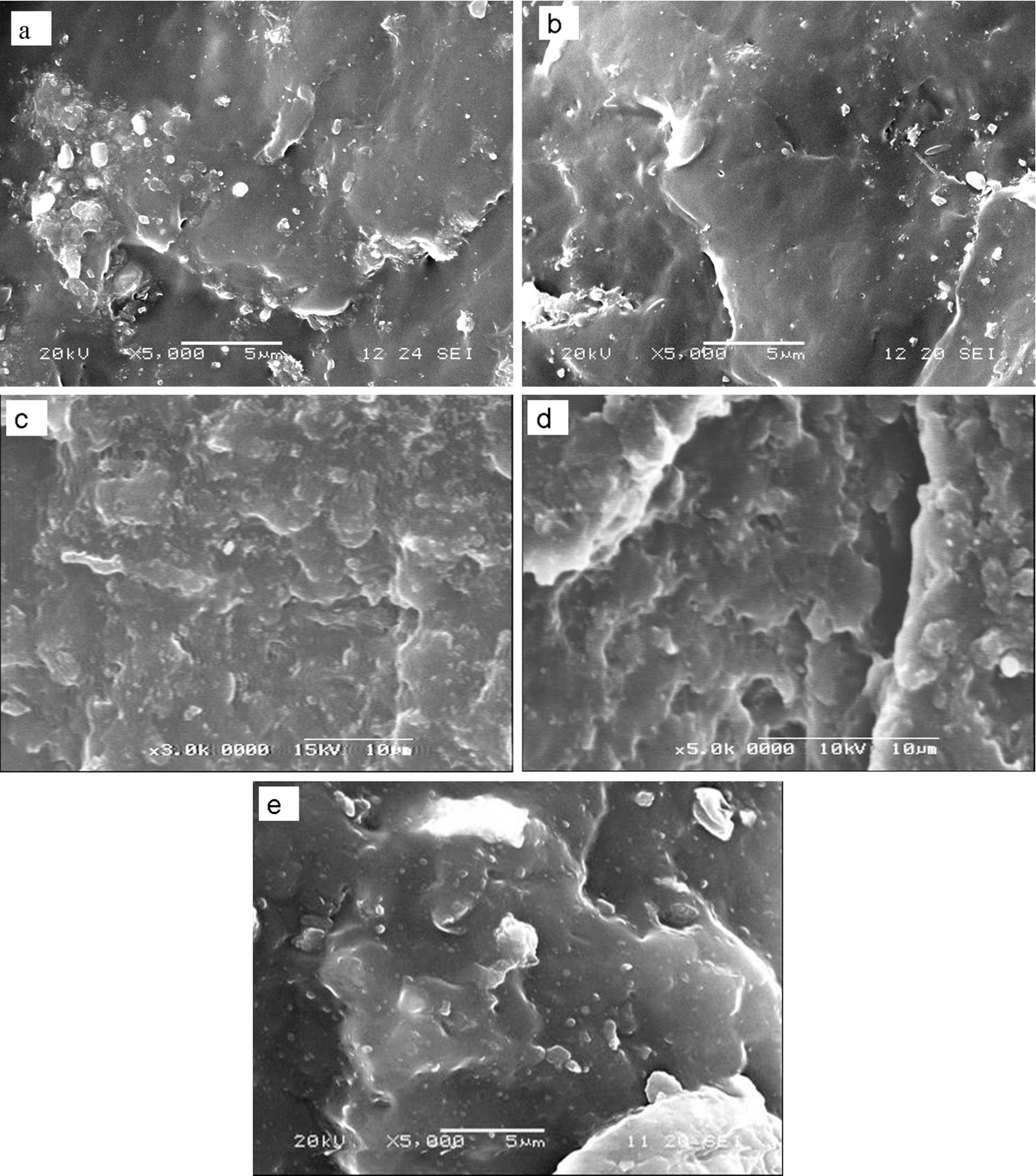
Scanning electron microscopy (SEM) micrographs of PP/epoxy composites. PP: polypropylene.
Crystallization and melting behaviors
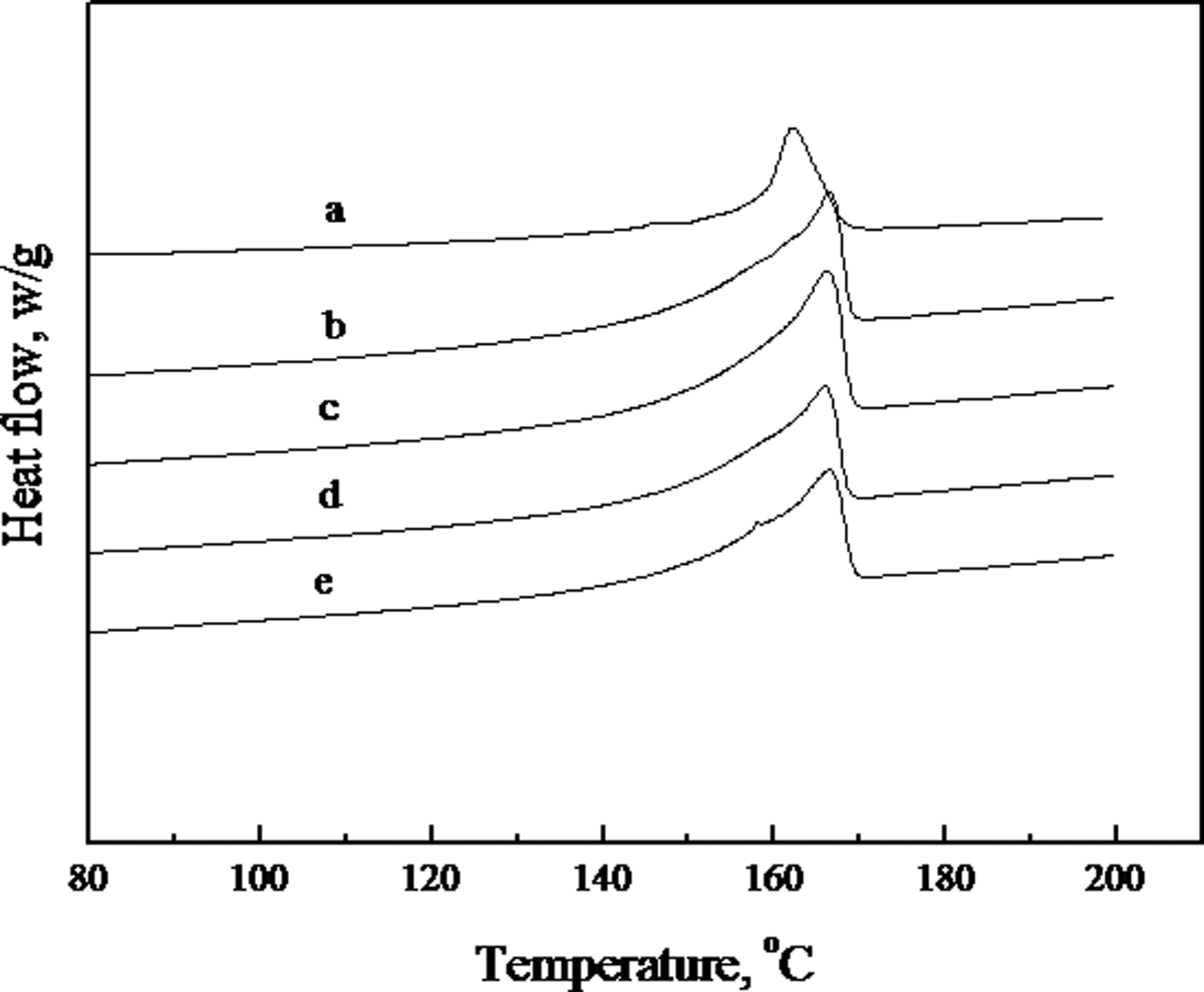
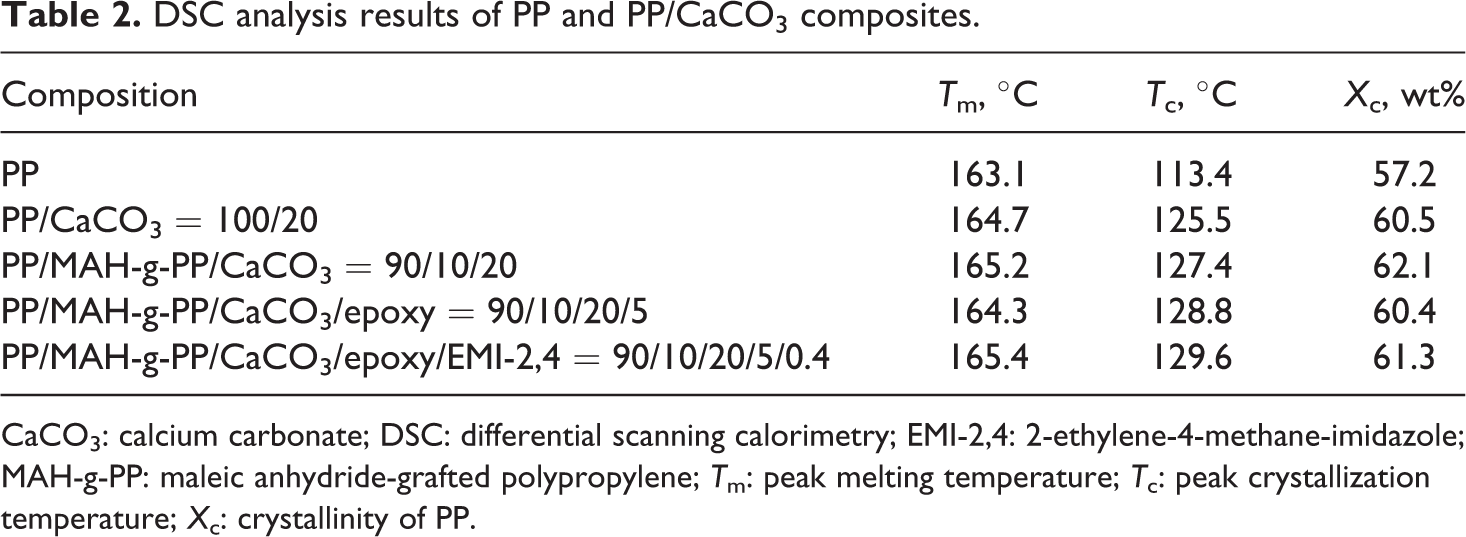
PP is a highly crystalline polymer that can crystallize rapidly even under quenched condition. Figures 3 and 4 show the first cooling and second heating DSC scans of PP and different PP/CaCO3 composites, and Table 2 gives a summary of the crystallization and melting data from DSC thermograms. The crystallinity of the PP component in the composites was calculated using the following equation
Differential scanning calorimetry (DSC) thermograms at first cooling of (a) PP; (b) PP/CaCO3 = 100/20; (c) PP/MAH-g-PP/CaCO3 = 90/10/20; (d) PP/MAH-g-PP/CaCO3/epoxy = 90/10/20/5; (e) PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 = 90/10/20/5/0.4. CaCO3: calcium carbonate; MAH-g-PP: maleic anhydride-grafted polypropylene; PP: polypropylene. Differential scanning calorimetry (DSC) thermograms at second heating of (a) PP; (b) PP/CaCO3 = 100/20; (c) PP/MAH-g-PP/CaCO3 = 90/10/20; (d) PP/MAH-g-PP/CaCO3/epoxy = 90/10/20/5; (e) PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 = 90/10/20/5/0.4. CaCO3: calcium carbonate; MAH-g-PP: maleic anhydride-grafted polypropylene; PP: polypropylene. DSC analysis results of PP and PP/CaCO3 composites. CaCO3: calcium carbonate; DSC: differential scanning calorimetry; EMI-2,4: 2-ethylene-4-methane-imidazole; MAH-g-PP: maleic anhydride-grafted polypropylene; T
m: peak melting temperature; T
c: peak crystallization temperature; X
c: crystallinity of PP.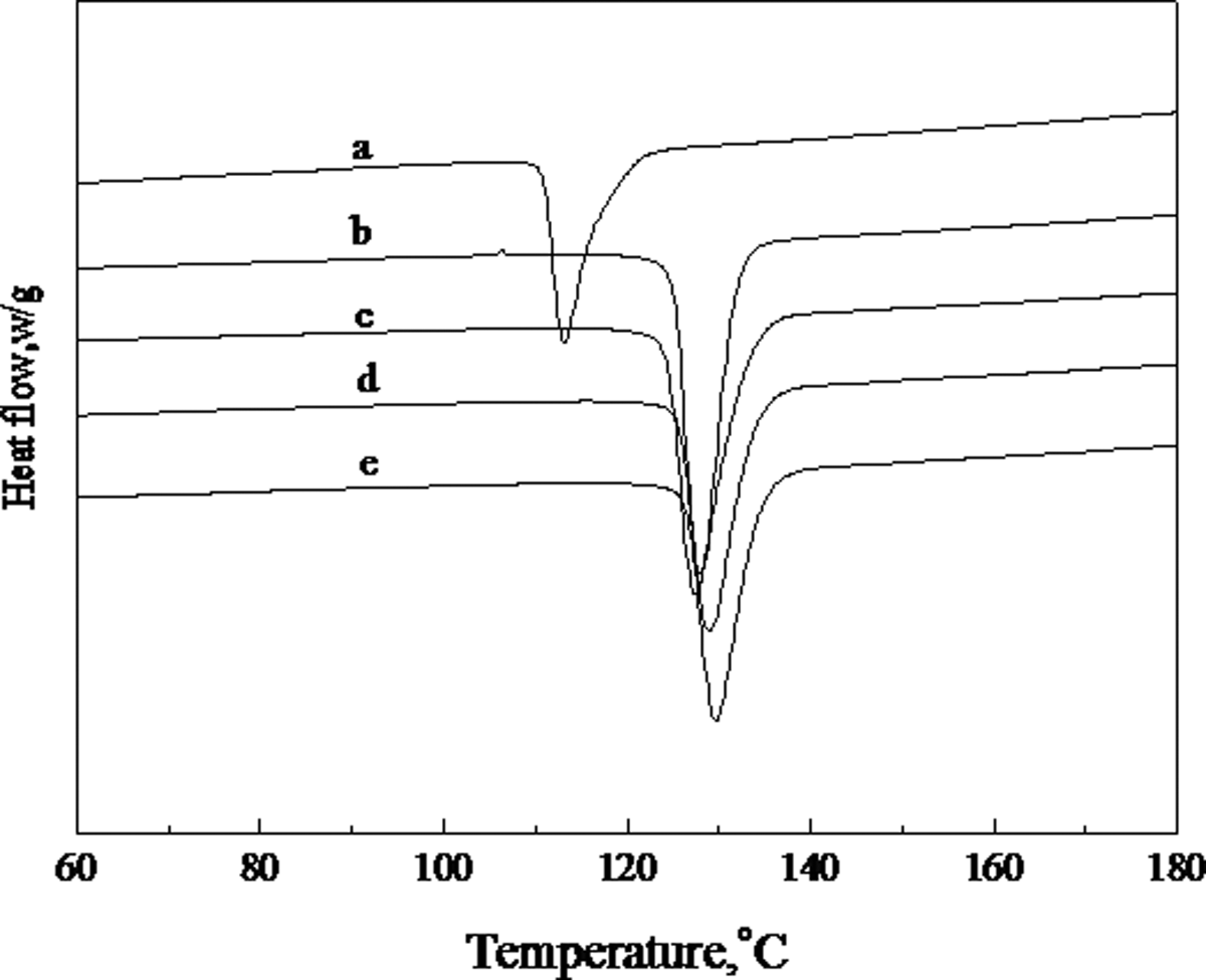

where X c is the crystallinity of PP, ▵H is the measured heat of crystallization of PP in the PP/CaCO3 composites, ▵H 0 (=170 J/g) is the heat of crystallization of 100% crystalline isotactic PP 25 and x is the mass fraction of PP in different PP/CaCO3 composites.
For PP and all PP/CaCO3 composites, only a single crystallization peak can be seen. All the crystallization peak temperatures of the PP/CaCO3 composites are higher than the crystallization peak of PP (113.4°C). The crystallization peak temperature of PP in the PP/CaCO3 (100/20) composite is increased by approximately 12°C compared to the pure PP. A shift in the crystallization peak to a higher temperature suggests that CaCO3 can act as an effective nucleating agent, accelerating the crystallization of PP in the composites. Liu et al. 26 have reported that the silicate layers can act as nucleating agents and accelerate the crystallization of PP in the PP/clay composites. The temperature of crystallization peaks of PP in the PP/MAH-g-PP/CaCO3 (90/10/20), PP/MAH-g-PP/CaCO3/epoxy (90/10/20/5) and PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 (90/10/20/5/0.4) composites are slightly higher than that of PP in the PP/CaCO3 (100/20) composite. This may be due to an increase in the interactions between the CaCO3 and the PP matrix, improving the nucleating activity of the filler and promoting crystallization during the cooling process. A similar result was reported by Wang et al. 14 For all PP/CaCO3 composites, the crystallinity of PP in the composites is higher than that of the pure PP, and the crystallinity of PP in the PP/CaCO3 (100/20) is 60.5% and increases by 3% compared with pure PP. The melting temperature of PP in all the PP/CaCO3 composites is slightly higher than that of pure PP (163.1°C). An increase in the melting temperature of PP in the composites is directly related to the size of the PP crystals. 27
TGA analysis
Polymer degradation is generally an undesirable process involving a deterioration of their properties. One of the most accepted methods for studying the thermal properties of polymeric materials is thermogravimetry which by means of integral and derivative thermogravimetric curves provide information about the nature and extent of degradation of the polymeric materials. 28 Thermograms of the PP and PP/CaCO3 composites are shown in Figure 5. The degradation of PP started at 352°C and finished at 420°C. The weight loss in this temperature region, 352–420°C, corresponds to the formation of volatile products which arise from the random chain scission and intermolecular transfer involving tertiary hydrogen abstractions from the polymer by the primary radical. 29 The CaCO3 does not degrade in the range of the measured temperature. In the case of the PP/CaCO3 (100/20) composite, the incorporation of 20 wt% CaCO3 increases the initial degradation temperature (T 0) from 352 to 362°C, T 0 of the PP/MAH-g-PP/CaCO3 (90/10/20) composites is slightly higher than that of the PP/CaCO3 (100/20) composite, and similar to that of the PP/MAH-g-PP/CaCO3/epoxy (90/10/20/5) composite. The PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 (90/10/20/5/0.4) composite has the highest initial degradation temperature (T 0) in all the PP/CaCO3 composites (shown in Table 3). All the degradation of PP/CaCO3 composites was in one step similar to PP. The result shows that the incorporation of the CaCO3 into the PP could improve the thermal stability of PP, and the addition of MAH-g-PP and epoxy resin in the PP/CaCO3 composites obviously could further improve the thermal stability. George et al. 28 suggested the thermal degradation of the polymer blends depends not only on the thermal stability of components but also on the morphology and extent of interaction between the phases. The increase in T 0 of PP/MAH-g-PP/CaCO3 (90/10/20) composite is attributed to the better interaction compared with the PP/CaCO3 (100/20) composite. The better interaction between the CaCO3 and PP gives rise to an increase in T 0s of PP/MAH-g-PP/CaCO3/epoxy (90/10/20/5) and PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 (90/10/20/5/0.4) composites.
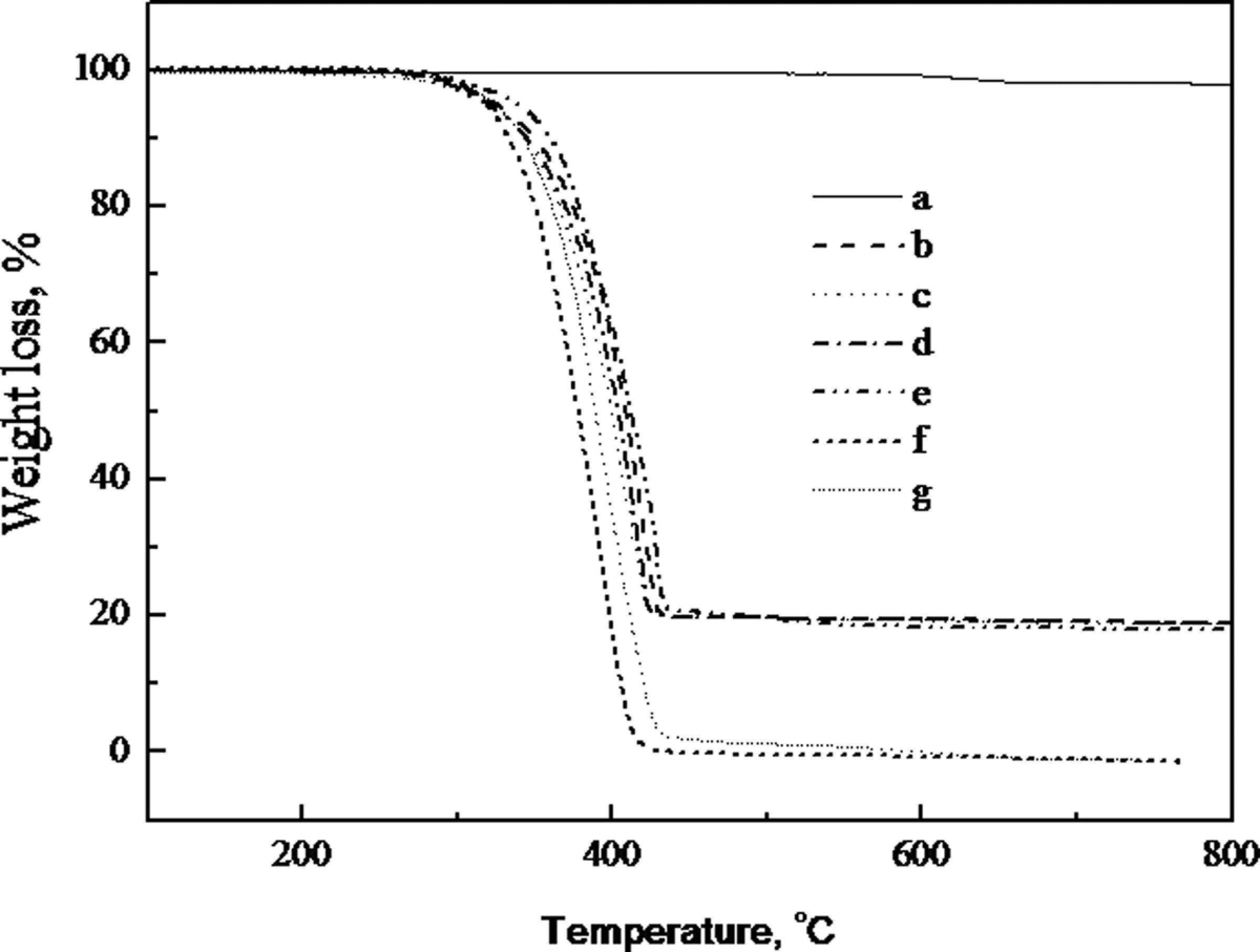
Thermograms of the PP and the PP/CaCO3 composites. (a) CaCO3; (b) PP; (c) PP/CaCO3 = 100/20; (d) PP/MAH-g-PP/CaCO3 = 90/10/20; (e) PP/MAH-g-PP/CaCO3/epoxy = 90/10/20/5; (f) PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 = 90/10/20/5/0.4; (g) PP/MAH-g-PP/epoxy = 90/10/5. CaCO3: calcium carbonate; MAH-g-PP: maleic anhydride-grafted polypropylene; PP: polypropylene.
TGA analysis results of the PP and PP/CaCO3 composites.

CaCO3: calcium carbonate; EMI-2,4: 2-ethylene-4-methane-imidazole; MAH-g-PP: maleic anhydride-grafted polypropylene; TGA: thermogravimetric analysis; T 0: initial degradation temperature; T f: final degradation temperature.
Mechanical properties
Mechanical properties including notched izod impact strength, tensile properties and flexural modulus of different PP/CaCO3 composites are summarized in Table 1. It can be seen that the addition of 20 wt% CaCO3 in the PP matrix gives rise to improved flexural modulus but decrease the impact strength, tensile strength and elongation at break due to the poor interaction between PP and the CaCO3. By adding 10 wt% MAH-g-PP, the PP/MAH-g-PP/CaCO3 (90/10/20) composite has the higher flexural modulus and tensile strength, but lower elongation at break compared with the PP. The PP/MAH-g-PP/CaCO3/epoxy (90/10/20/5) composite has better mechanical properties than that of the PP/MAH-g-PP/CaCO3 (90/10/20) composite, and the PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 (90/10/20/5/0.4) composite has the highest flexural modulus (1892 MPa) and impact strength (45 J/m) in all the PP/CaCO3 composites, but the elongation at break is almost the same as that of the PP/MAH-g-PP/CaCO3/epoxy (90/10/20/5) composite. We thought that the improvement in the mechanical properties of PP/MAH-g-PP/CaCO3/epoxy composite is due to the higher interaction between PP and CaCO3 compared with PP/MAH-g-PP/CaCO3, and dynamical cure of the epoxy resin in the PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 composite can further increase the interaction between the CaCO3 and PP matrix, and also increase the strength and modulus of composite. The mechanical properties of the PP/CaCO3/epoxy (100/20/5) is similar to that of the PP/CaCO3 (100/20), and it shows that epoxy resin cannot improve the interaction between PP and the CaCO3 in the absence of MAH-g-PP. Figure 6 shows that the effect of epoxy resin content on the mechanical properties of PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 composites. The flexural modulus increase with increasing epoxy resin content, the impact strength reach a maximum\ of 45 J/m at an epoxy resin content of 5 wt%, and the tensile strength has a best value of 47 MPa at an epoxy resin content of 8 wt%. But the elongation at break decreases with increasing epoxy resin content. The result show that the suitable content of epoxy resin in the presence of MAH-g-PP in the PP/CaCO3 composites is about 5 wt%, while excess epoxy resin of above 5 wt% acts as a organic filler for PP.
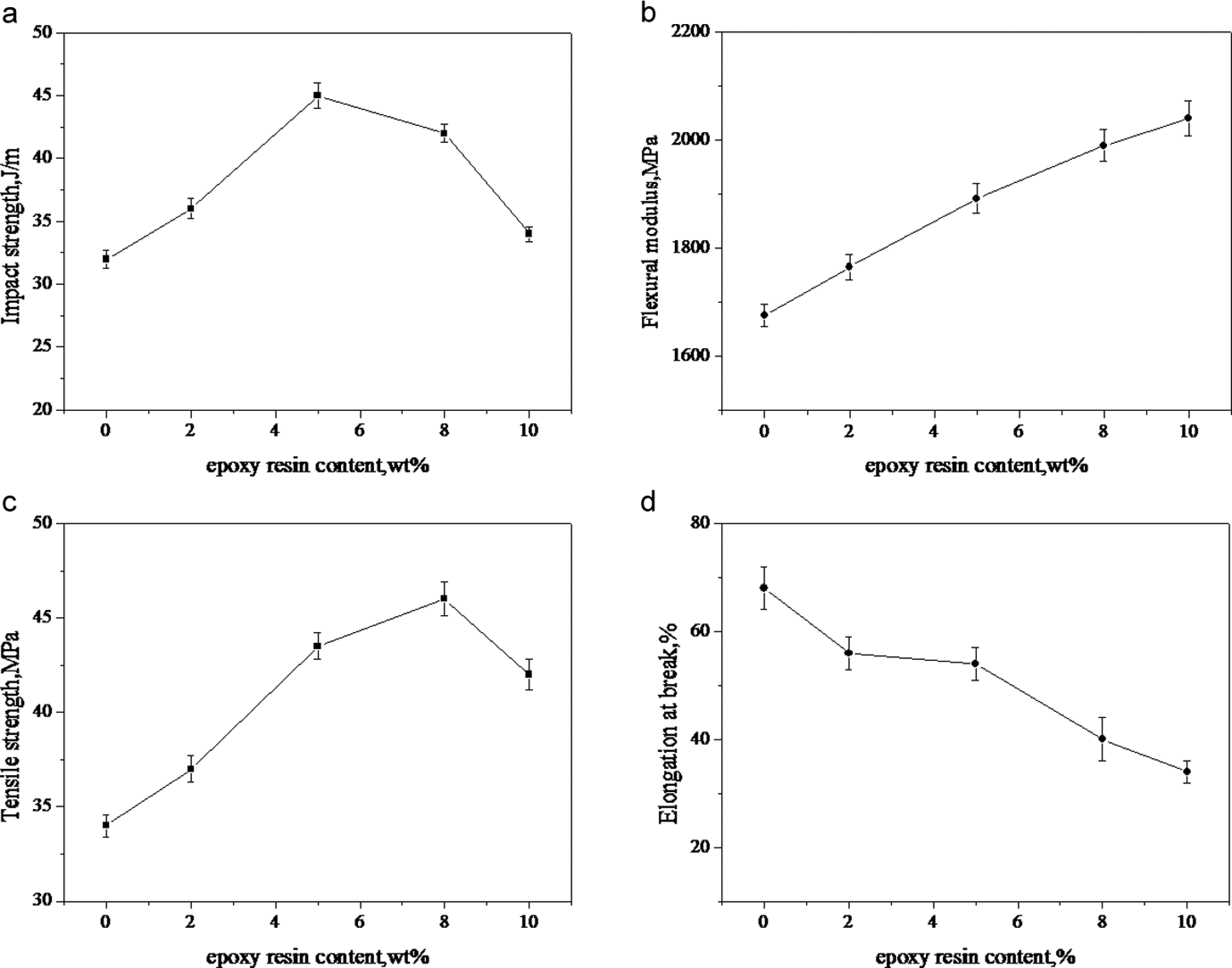
Effect of epoxy resin content on the mechanical properties of the PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 composites. CaCO3: calcium carbonate; EMI-2,4: 2-ethylene-4-methane-imidazole; MAH-g-PP: maleic anhydride-grafted polypropylene; PP: polypropylene.
Conclusions
The mixture of MAH-g-PP and an epoxy resin were demonstrated to effectively increase the interaction between PP and the CaCO3 in the PP/CaCO3 composites. The torque at equilibrium of the PP/MAH-g-PP/CaCO3/epoxy (90/10/20/5) composite is higher than that of the PP/MAH-g-PP/CaCO3 (90/10/20) composite, and it shows that the epoxy resin has the chance to react with MAH-g-PP and hydroxyl groups at the CaCO3 surface and MAH-g-PP. The formed anchor at the interface increases the interaction between PP and CaCO3. Dynamic cure epoxy resin can further increase the interaction adhesion. SEM analysis shows that the epoxy resin does not show compatibilization effects for the PP/CaCO3 blends without the presence of MAH-g-PP. DSC analysis shows that the CaCO3 in the PP/CaCO3 composites can act as nucleating agents, accelerating the crystallization of PP component, and the PP/CaCO3 composites containing MAH-g-PP and the epoxy resin have higher crystallization peaks. TGA shows that the incorporation of the CaCO3 into the PP could improve the thermal stability of PP, and the addition of MAH-g-PP and the epoxy resin further improves thermal stability of the PP/CaCO3 composites. The PP/MAH-g-PP/CaCO3/epoxy composites have better mechanical properties than that of PP/MAH-g-PP/CaCO3 composites, and the PP/MAH-g-PP/CaCO3/epoxy/EMI-2,4 composites have best mechanical properties in all the PP/CaCO3 composites, which is attributed to dynamical cure; epoxy resin can further increase the interaction adhesion. The suitable content of epoxy resin in the presence of MAH-g-PP in the PP/CaCO3 composites is about 5 wt%, when epoxy resin content is above 5 wt%, the excess epoxy resin acts as a filler in composites.
Footnotes
Acknowledgments
The authors would like to thank the support from the projects for the Excellent Youth Scholars of Educational Commission of Hubei Province of China (Grant No. Q20101505) and Hubei Provincial Natural Science Foundation of China (Grant No. 2011CDB220).
Funding
This research received the specific grant from Graduate Innovative Fund of Wuhan Institute of Technology.
