Abstract
Poly(vinyl alcohol) (PVA)/poly(ethylene oxide) (PEO) hydrogel with various PVA/PEO compositions was prepared by freezing/thawing method. The effect of PEO content on the structure and property of the gel was investigated. The molecular chains of PEO with relatively high crystalline and molecular weight may limit the movement of PVA chains, resulting in the increase in glass transition temperature (
Keywords
Introduction
In recent years, hydrogels with high water content, mechanical strength and elasticity synthesized by aqueous solution of poly(vinyl alcohol) (PVA) have received increasing interest for their various pharmaceutical and biomedical applications. PVA hydrogels can be cross-linked by difunctional cross-linking agents, electron beam (EB) or γ-irradiation and freezing/thawing method, of which the freezing/thawing method addresses toxicity issues because it does not require the presence of a cross-linking agent, and frozen/thawed gels have demonstrated enhanced mechanical properties, particularly for biomedical applications. 1,2
It is now accepted that the freezing/thawing process involves phase separation and formation of ice crystals. The porous polymer network structure of frozen/thawed hydrogels comprises two separate phases, one with a low polymer concentration and the other with a high polymer concentration. The polymer-rich phase is segregated into closely interconnected regions and forms macropores, which are filled with polymer-poor phase. Moreover, the polymer-rich phase organized itself and consisted of polymer microcrystallites and amorphous domains. The porous polymer network structure is imprinted by the formation of ice crystals. During the freezing step, ice crystals expel amorphous polymer segments, increase the polymer concentration in the surrounding environment and induce the formation of polymer microcrystallites. Upon thawing the gel to room temperature, the ice crystals melt, but the gel structure does not collapse.
The blending of PVA with other polymers produces hydrogels with improved characteristics and properties. For example, interpenetrating polymer network hydrogels composed of PVA and poly(arylicacid) were proved to be pH sensitive. 3 PVA/poly(vinylpyrrolidone) hydrogels, exhibiting nonlinear stress–strain behavior, may be viable as nucleus pulposus implants. 4 Highly absorbent PVA/glycerin hydrogels can convert wound exudates to the hydrogel matrices which may create a moist and clean environment in the wound-healing process. 5 The system of PVA/poly(ethylene oxide) (PEO) polymer blend is also of great interest. The solution-casting films of PVA/PEO have been examined by many researchers. Inamura et al. reported the presence of a liquid–liquid phase separation in the PVA/PEO/water system. 6 Yoshii et al. investigated the behavior of EB cross-linked PEO/PVA hydrogel for application in wound dressing. 7
PEO is a water-soluble, biocompatible polymer, which has been widely applied in biomedical fields. In this work, PVA/PEO hydrogels with various PVA/PEO compositions were prepared by freezing/thawing method. The effect of the content of PEO on the structure and property of the gel was investigated.
Experimental
Materials
PVA (polymerization degree of 1700, hydrolysis degree of 99%) used in this work was a commercial product and supplied by Sichuan Vinylon Co., Ltd (China). Other chemical agents were all of analytical purity and used as received.
Preparation of PVA/PEO hydrogels
Aqueous solution of 16 wt% PVA/PEO was prepared by dissolving PVA and PEO in distilled water at 95°C. The PVA/PEO compositions were 100/0, 98/2, 96/4, 94/6, 92/8 and 90/10 by weight, respectively. The PVA aqueous solutions were poured into the mold, and hydrogel films of 2 mm thickness were formed after freezing at −20°C for 12 h and then thawing at room temperature for 4 h.
Measurements
Gel fraction
To measure the gel fraction, the preweighed dry samples of PVA hydrogel were immersed in deionized water to remove the uncross-linked part of the hydrogel. After 48 h, the washed hydrogel was dried in the vacuum oven at 50°C until the weight of the sample became stable. The gel fraction was calculated with the following formula:
where
Tensile property
The tensile properties of PVA/PEO hydrogel samples were measured with a 4302 material testing machine from Instron Co. (USA) according to ISO527/1-1993 (E). The sample was of dumbbell shape with a size of 150 × 10 × 4 mm3. The tensile speed and temperature were 50 mm/min and 23°C, respectively.
Swelling property
The preweighed dry samples were immersed in distilled water for different times at room temperature until an equilibrium state of absorption was achieved. After the excessive surface water was removed with filter paper, the weight of the swollen gel was measured at various time intervals. The procedure was repeated until there was no further weight increase. The swelling ratio can be determined as a function of time.
8

where
DSC analysis
The state of water in swollen PVA/PEO hydrogel was measured with a Netzsch 204 Phoenix differential scanning calorimeter (DSC; Germany) under N2 atmosphere. Samples of about 5–10 mg were heated from −40 to 50°C at a constant rate of 3°C/min.
The
SEM analysis
The fractured surface morphology of PVA/PEO hydrogel was observed with a JEOL JSM-5900LV scanning electron microscope (SEM; Japan) with an acceleration voltage of 20 kV. The PVA/PEO hydrogel was freeze dried and cryogenically fractured in liquid nitrogen. Then the samples were sputter coated with gold.
Results and discussion
Preparation of PVA/PEO hydrogel films
Freeze–thawing of PVA/PEO solution leads to the formation of insoluble hydrogel. The gel fraction of PVA/PEO hydrogel films was shown in Table 1. It can be seen that all samples had a gel fraction of more than 90%, suggesting that PVA/PEO solution was almost completely cross-linked.
Gel fraction of PVA/PEO hydrogel films with various compositions.
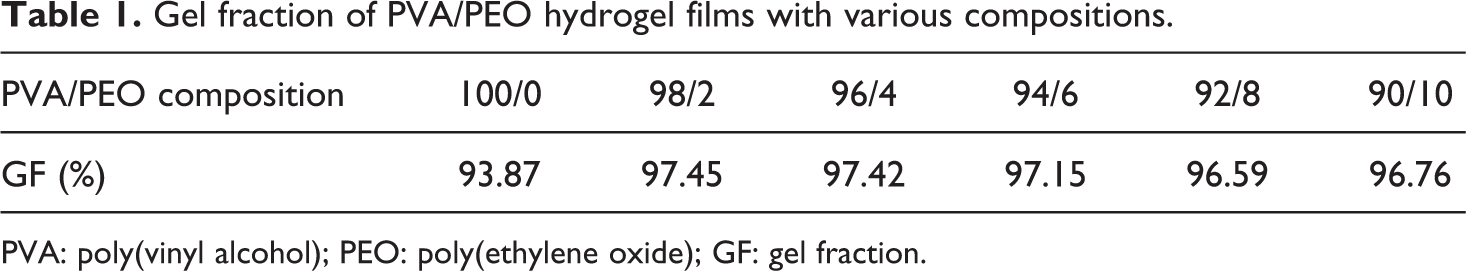
PVA: poly(vinyl alcohol); PEO: poly(ethylene oxide); GF: gel fraction.
The thermal behavior of dried PVA/PEO hydrogel films was investigated by DSC analysis. Figure 1 was DSC thermograms of pure PEO and PVA/PEO hydrogels with various compositions. It can be seen that the melting temperature of PEO ranged from 50 to 75°C. The
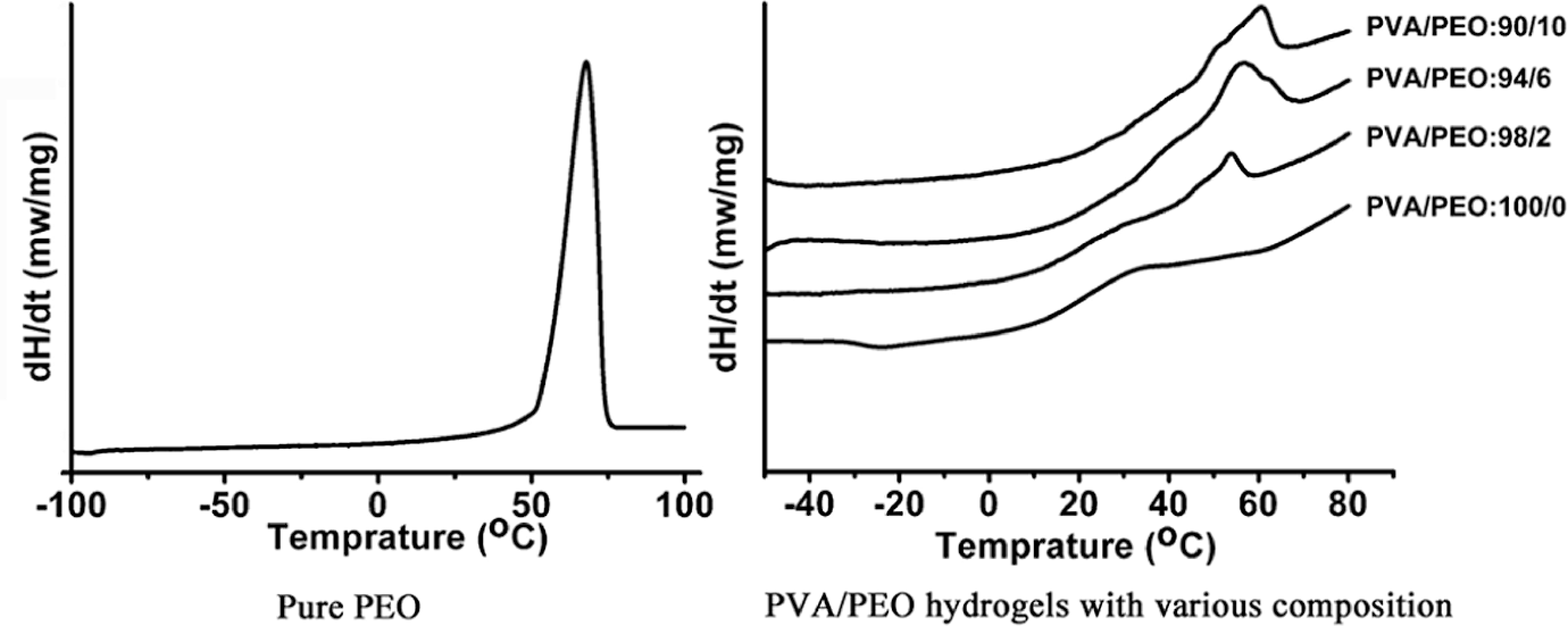
DSC thermograms of PEO and PVA/PEO hydrogels with various compositions. PVA: poly(vinyl alcohol); PEO: poly(ethylene oxide); DSC: differential scanning calorimeter.
Tensile property of PVA/PEO hydrogel
The tensile properties of PVA/PEO hydrogel films as a function of the PVA/PEO composition were shown in Figure 2. It can be seen that the tensile strength of PVA/PEO hydrogel films increased with increasing PEO content. The elongation at the break of the PVA/PEO hydrogel films first increased remarkably at low PEO content. However, it decreased slightly thereafter with the increase in PEO content. PEO is a high crystalline polymer with high molecular weight, resulting in the enhancing effect on the mechanical properties of PVA hydrogel, which was consistent with the increasing
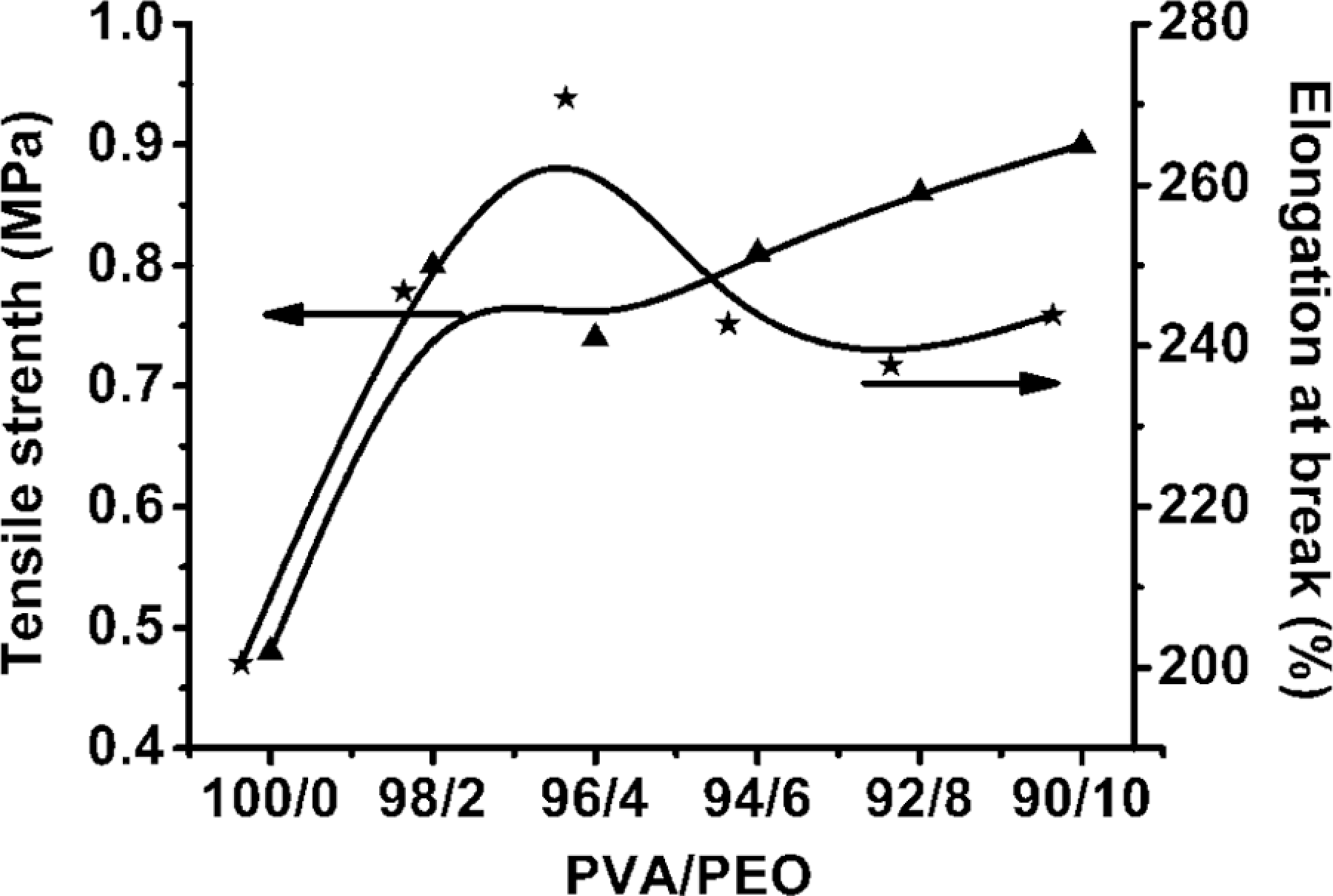
Tensile properties of PVA/PEO hydrogels with various compositions. PVA: poly(vinyl alcohol); PEO: poly(ethylene oxide).
Swelling property of PVA/PEO hydrogel
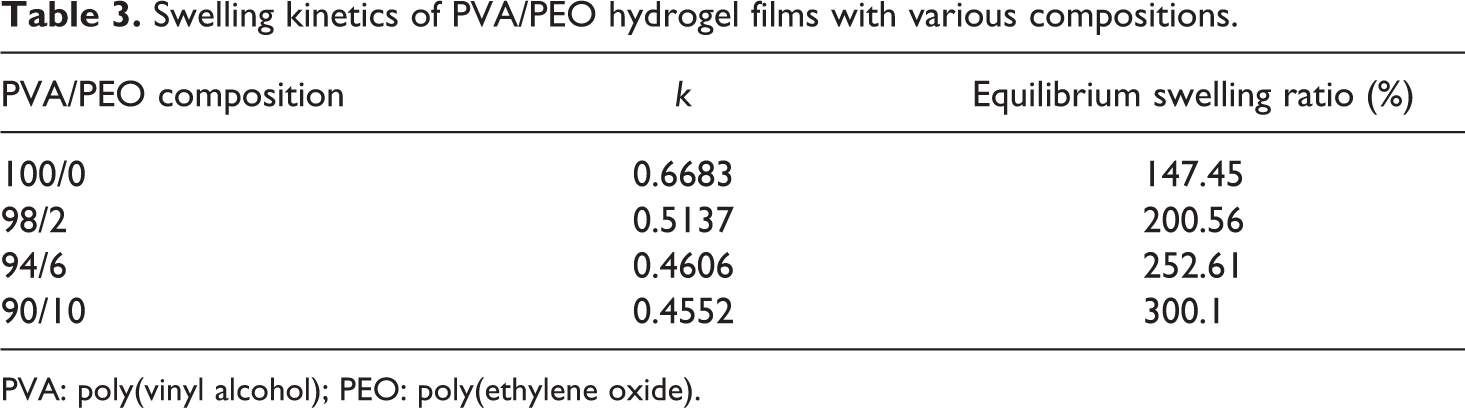
The swelling ratio of PVA/PEO hydrogel films with various compositions as a function of swelling time was shown in Figure 3. It can be seen that in the initial swelling stage, all the samples of hydrogel films absorbed water rapidly, and the swelling ratio increased dramatically with time. The swelling ratio was significantly improved by the addition of PEO, and the equilibrium swelling ratio of PVA hydrogel films increased from 147% in the absence of PEO to 300% when the gel fraction of PVA/PEO was 90/10, indicating that more content of PEO could enhance the capillary water absorption capacity of PVA/PEO hydrogel.
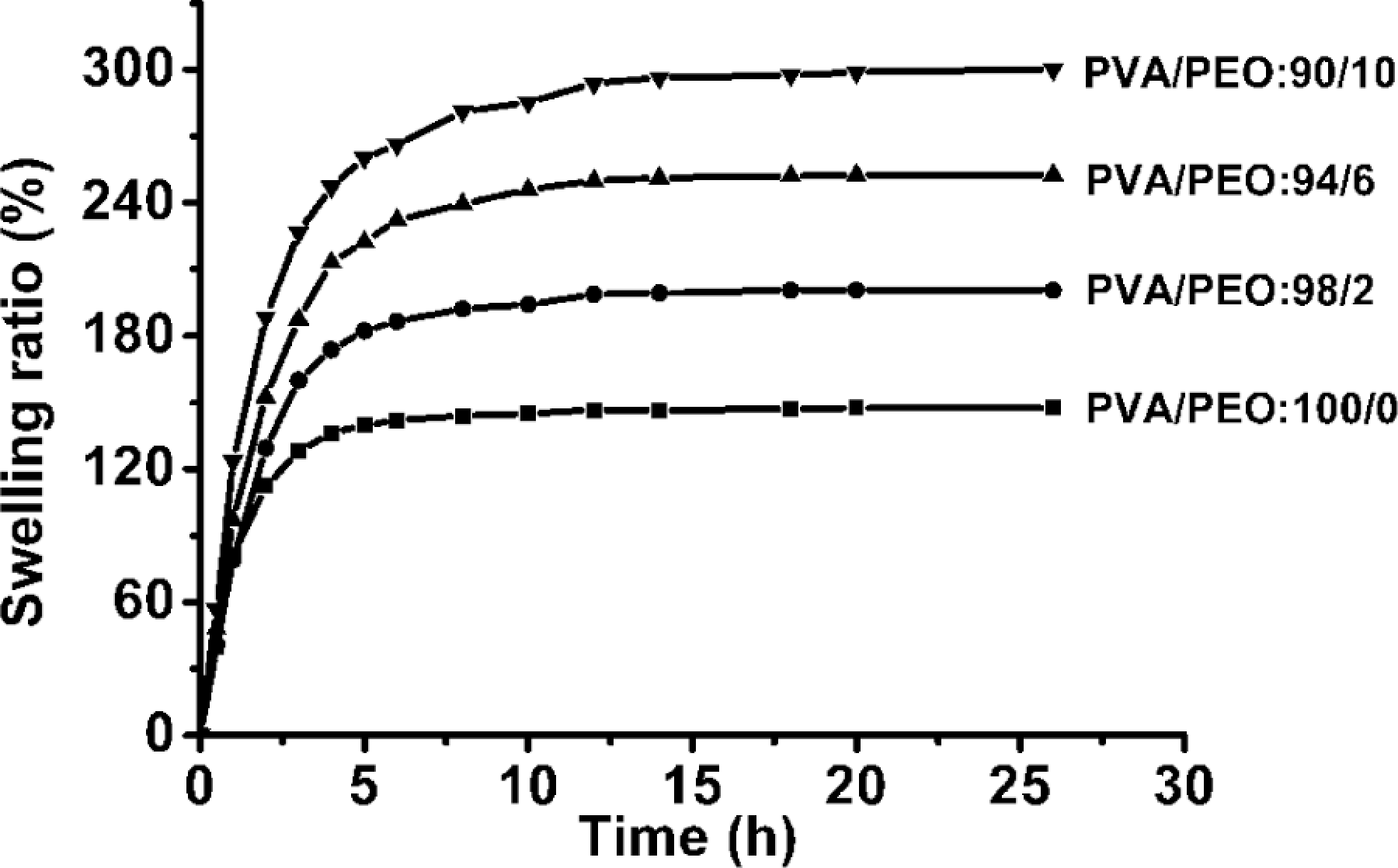
Swelling ratio curve of PVA/PEO hydrogel films with various compositions. PVA: poly(vinyl alcohol); PEO: poly(ethylene oxide).
Swelling kinetics of PVA hydrogel beads were analyzed by assuming that the swelling process met the first-order kinetic equation:
where 
Another form of the equation can be written as:
By plotting the graph of ln[(
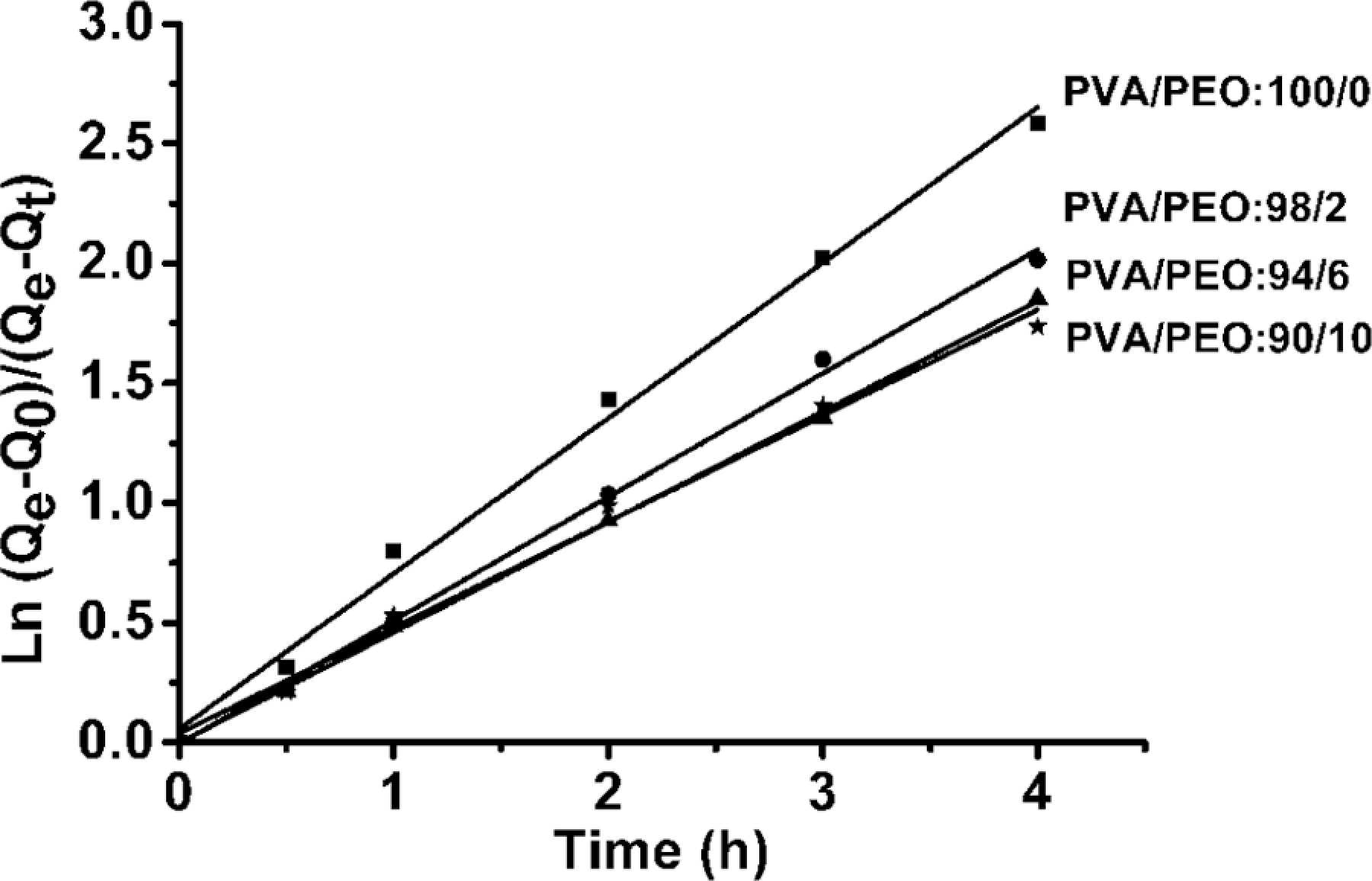
Swelling kinetics of PVA/PEO hydrogel films. PVA: poly(vinyl alcohol); PEO: poly(ethylene oxide)

PVA: poly(vinyl alcohol); PEO: poly(ethylene oxide);
Swelling kinetics of PVA/PEO hydrogel films with various compositions.
PVA: poly(vinyl alcohol); PEO: poly(ethylene oxide).
State of water in PVA/PEO hydrogel
Generally, the state of water in the polymer hydrogel includes free water, which cannot form hydrogen bonding with polymer molecules; freezing bound water, which forms weak interaction with polymer molecules through Van der Waals force; and nonfreezing bound water, which forms hydrogen bonding with polymer molecules. 9,10
DSC curves of PVA/PEO hydrogel films with various compositions are shown in Figure 5. It can be seen that with increasing temperature, two obvious endothermic peaks appeared between −20 and 5°C. Because the phase transition temperature of freezing bound water was lower than that of free water, the left peak was ascribed to the freezing bound water, and the right one was ascribed to free water. The nonfreezing bound water exhibited no endothermic peak in DSC curve. By calculating the area of the two endothermic peaks, the total amount of free water and freezing bound water ( DSC curves of swollen PVA/PEO hydrogel films with various compositions. PVA: poly(vinyl alcohol); PEO: poly(ethylene oxide); DSC: differential scanning calorimeter.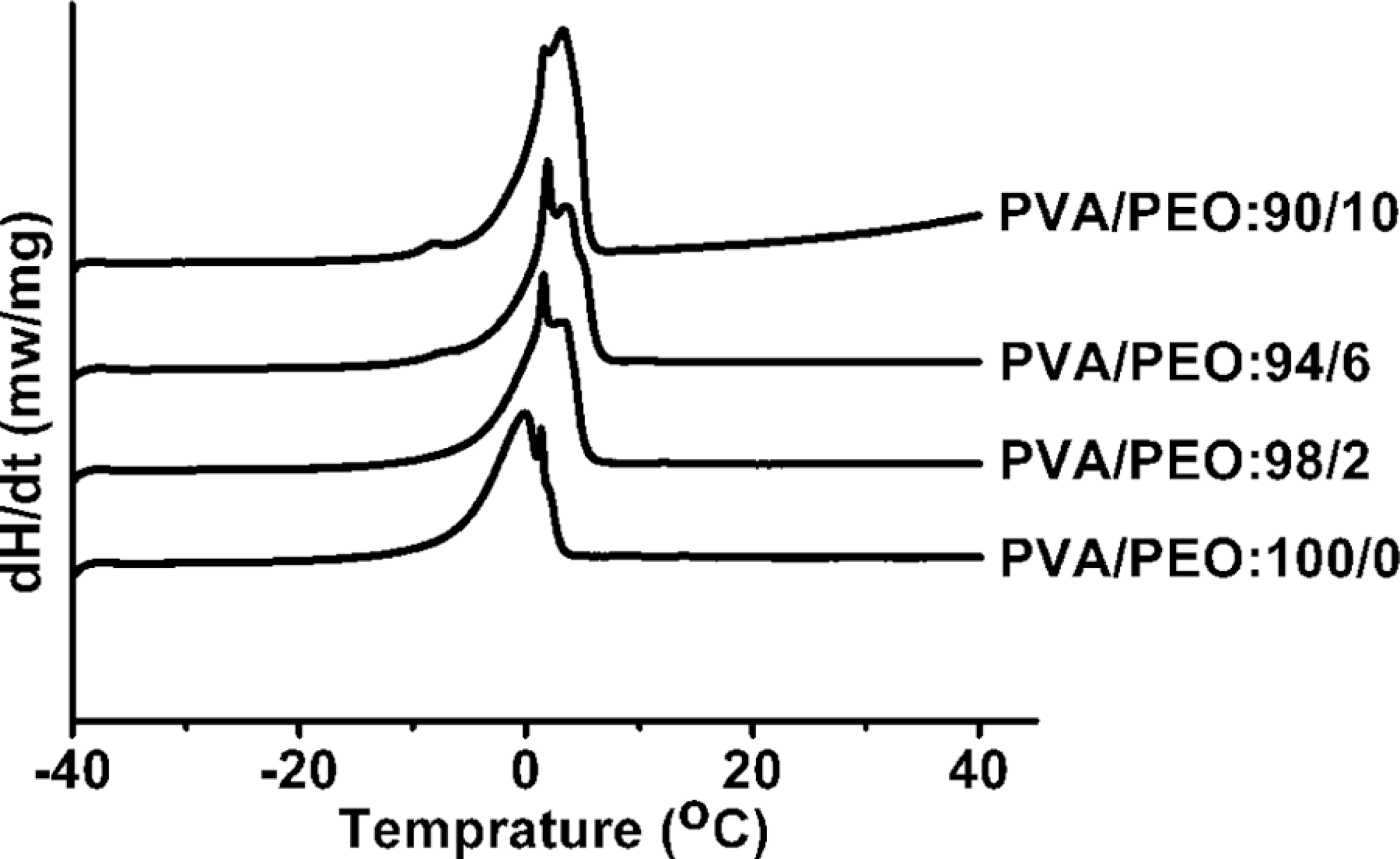

where
The amount of free water and freezing bound water can be calculated by the two peak area ratios, then the amount of nonfreezing bound water can be obtained by the difference in the total water content of PVA/PEO hydrogel and the above two states of water, while the total water content was the equilibrium water content (EWC), which can be calculated as follows
11,12:
where
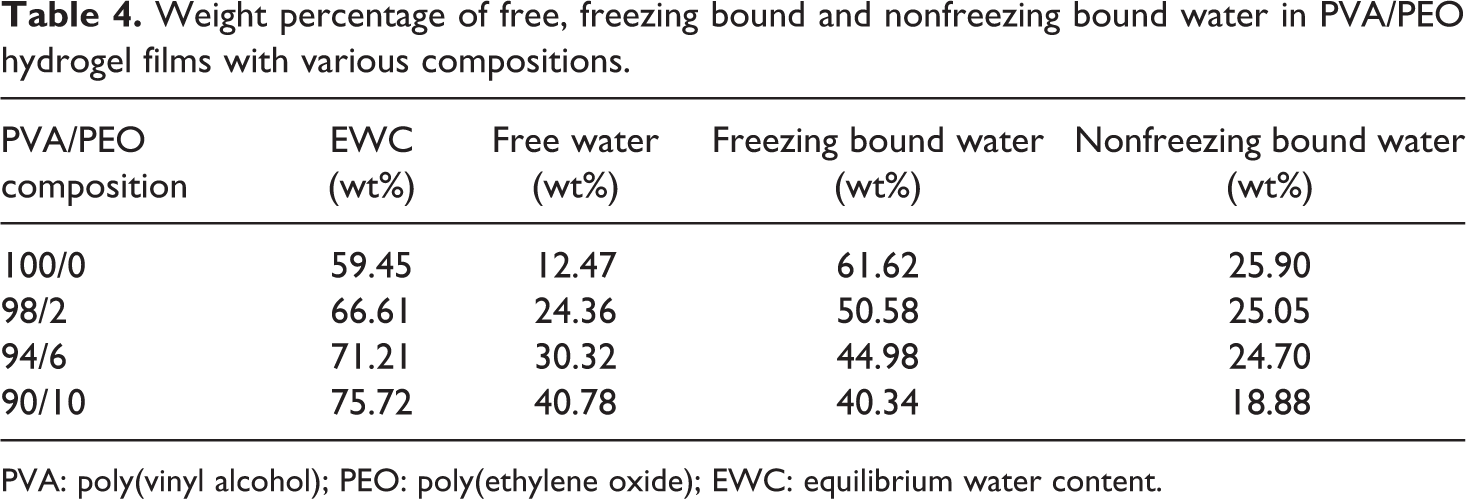
The water content for the three states of PVA/PEO hydrogel at equilibrium swelling was listed in Table 4. It can be seen that most of the water in PVA hydrogel was in the form of free water and freezing bound water, and relatively less water was in the form of nonfreezing bound water, because during freezing–thawing process, more hydrogen bonds are formed between PVA molecules, and fewer hydroxyl groups of PVA formed hydrogen bonds with water.
Weight percentage of free, freezing bound and nonfreezing bound water in PVA/PEO hydrogel films with various compositions.
PVA: poly(vinyl alcohol); PEO: poly(ethylene oxide); EWC: equilibrium water content.
Addition of PEO improved the EWC of PVA hydrogel, and with increasing content of PEO, the free water content was remarkably increased due to the formation of relatively large pores in gels, while both freezing bound water and nonfreezing bound water content decreased.
Water absorbent property of PVA/PEO hydrogel films with various compositions.
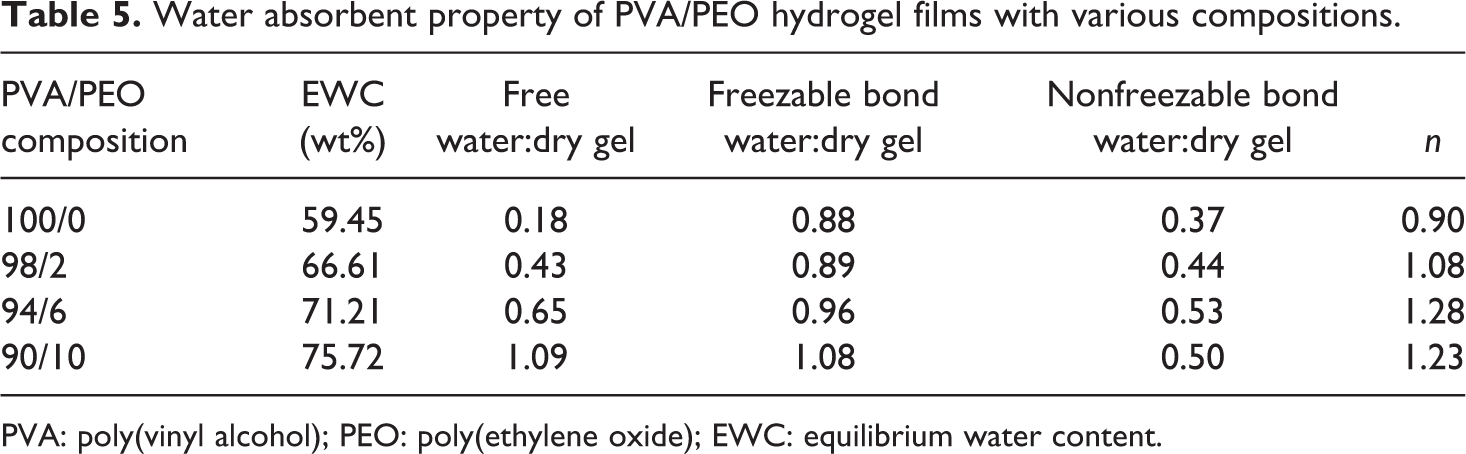
PVA: poly(vinyl alcohol); PEO: poly(ethylene oxide); EWC: equilibrium water content.
It can be seen that addition of PEO resulted in a remarkable increase in the weight ratio of free water content to dry gel weight by nearly 6 times, and the free water permeated into the hydrogel by capillary pores, indicating that the PEO greatly increased the total pore volume of PVA gel. It can also be observed that the weight ratio of freezing and nonfreezing bound water to dry gel weight increased with increasing PEO content, and
Morphology of PVA/PEO hydrogel
The PVA/PEO composition affects the morphology of the hydrogel. As shown in Figure 6, the PVA/PEO hydrogel film represented a porous structure and the size of the pores increased with increasing PEO content. As Kondo et al. 13 reported, the –OH in PVA, which is a secondary –OH, does not interact with the oxygen of PEO. As a result, the addition of PEO perhaps diluted the density of –OH of PVA, which resulted in loose network with larger pores.
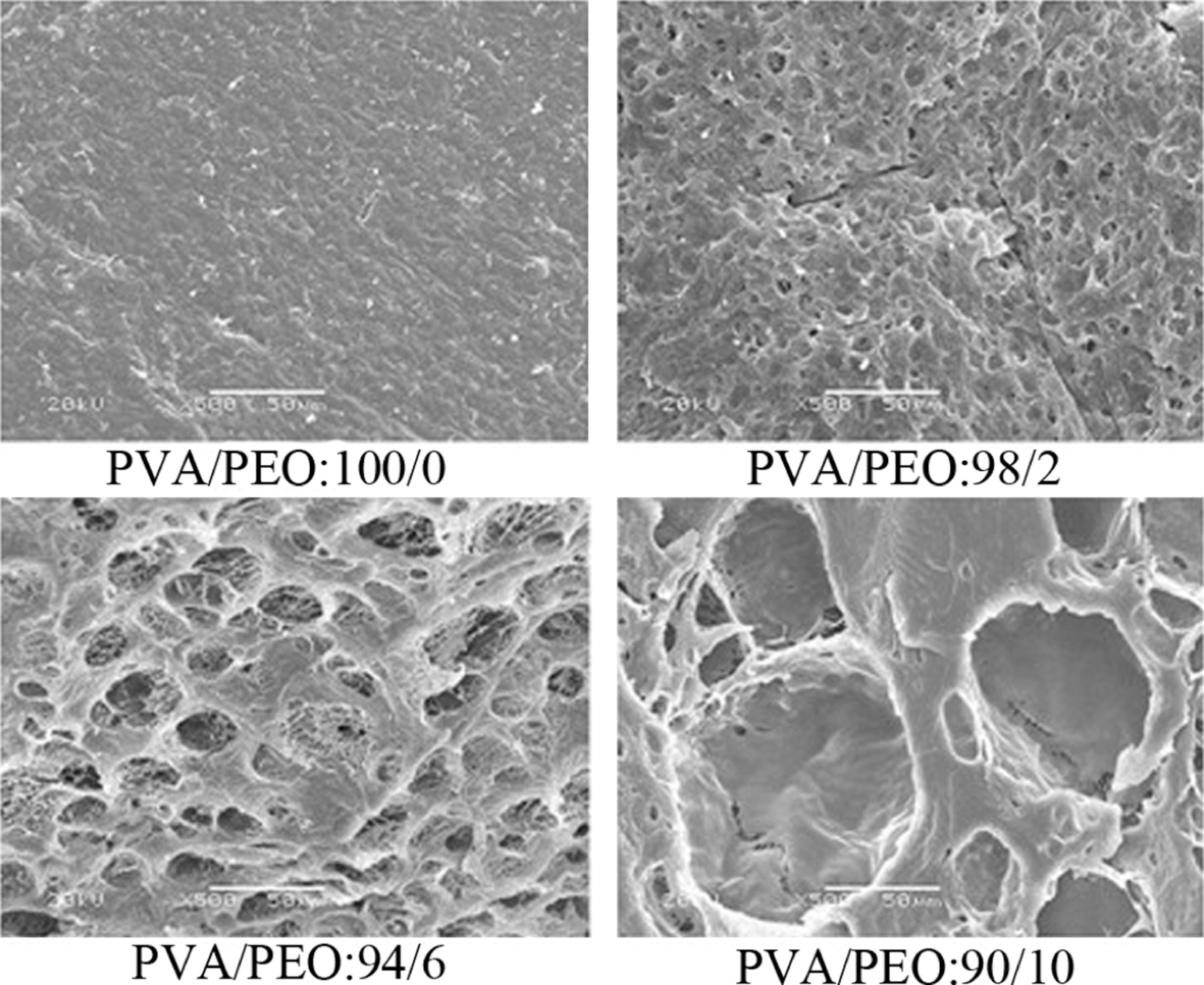
SEM of PVA/PEO hydrogel films with various compositions. PVA: poly(vinyl alcohol); PEO: poly(ethylene oxide); SEM: scanning electron microscopy.
Conclusion
In this work, the effect of PEO content on the structure and property of the frozen/thawed PVA/PEO hydrogel was investigated. The molecular chains of PEO with relatively high crystalline and molecular weight may limit the movement of PVA chains, resulting in the increase of
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
