Abstract
Compared with the traditional composites, the incorporation of carbon nanotubes into polymeric matrices can generate materials with superior properties, especially thermal, electrical and tribological properties. The aim of this study was to study the polyamide 6.6/carbon nanotubes (PA 6.6/CNT) nanostructured composites crystallization kinetics. The solution mixing technique was used to obtain the nanostructured composites studied in this work. PA 6.6 films were produced with amounts of 0.1, 0.5, and 1.0 wt% (weight/weight) CNT. X-ray diffraction analyses were performed in order to determine the crystallographic properties of nanostructured composite. The nanostructured composites crystallization kinetic study was performed using the differential scanning calorimetry under isothermal and nonisothermal (dynamic) conditions. The results have shown addition of CNTs in the PA 6.6 reduces the Avrami exponent, affecting the crystallization process of the composite.
Introduction
The nanostructured materials have gained great prominence in the scientific community due to their inclusion in various scientific and technological fields, such as catalysis, biological sensors, optoelectronic materials and polymeric composites. Nowadays, the search demands to find methods of preparation, characterization and manipulation of nanostructures for a specific use, as in the case of nanotechnology related to composite materials. Recently, one of the most used materials in combination with polymeric matrices are carbon nanotubes (CNTs), forming the nanostructured composites. 1
CNTs show a unique combination of mechanical, electrical and thermal properties that make them excellent candidates to replace or supplement conventional loads used in the preparation of polymeric nanostructured composites. Single-wall carbon nanotubes (SWCNTs) may present metallic or semiconducting behavior, with Young’s modulus from 300 to 1000 GPa and tensile bond strength from 150 to 180 GPa. 2–4 Multiwall carbon nanotubes (MWCNTs) show electrical features between metal and semiconductor, with Young’s modulus from 270 to 950 GPa, tensile bond strength from 11 to 63 GPa. 5–7 Another important property, which is worth mentioning, is their high thermal conductivity along the longitudinal axis of the nanotubes. In this case, it is estimated that the values of around 6000 W m−1 K−1can be reached, which is much higher than the values found for copper (an average of 385 W m−1 K−1). When the CNTs are added into polymeric matrices, an increase in their thermal conductivity up to 125% can be observed. 8–11
The CNTs incorporation into polymers can result in materials with higher mechanical strength, 12 better thermal stability 13 or better optical and electrical properties. 2,14 Currently, a wide type of thermoplastic and thermoset matrices have been used for this purpose, however, few studies report the CNTs effect on the crystallization kinetics of the thermoplastic matrices with structural application.
Among the major thermoplastic matrices, polyamide 6.6 (PA 6.6) has been highlighted in several applications due to their good mechanical properties, high melting temperature (263°C), high impact and fatigue strength. When reinforced, the PA 6.6 can be used in a wide range of applications, especially in different sectors of industry including the automobile, electronics, aerospace, light and precision mechanical engineering. 15–18
Polymeric materials with similar chemical compositions may exhibit different properties, like toughness, elasticity, permeability and transparency. 19 These properties depend on the microstructure of the material which is determined by its thermal cycle processing. In the case of semicrystalline polymers, the solidification process is followed by crystallization in isothermal and nonisothermal conditions. The final product will depend on important factors such as physical dimensions (its crystal size distribution, its median crystal size, its purity and its crystal shape) and the cooling rates (the properties of the liquid and solid phases) which will be submitted to the polymer and the crystallization kinetic parameters (geometry and type of the crystallizer). 19–21
In the present work, the effect of adding different amounts of CNTs (0.1, 0.5 and 1.0 wt% CNT) on the crystallization kinetics of the PA 6.6 polymer matrix was studied. The nanostructured composites crystallization kinetic was performed using differential scanning calorimetry (DSC) in nonisothermal and isothermal conditions.
Theoretical considerations
Isothermal crystallization kinetics
The isothermal crystallization kinetics of polymeric materials is accomplished by the Avrami equation,
22–27
which estimates the crystalline fraction,

where
The Avrami exponent
Nonisothermal crystallization kinetics
For the study of nonisothermal crystallization kinetics, it should be assumed that with increasing temperature constant, the reaction rate constant, ‘

where

Equation (4) can be described from the derivative plot of the crystallized fraction, (d
Experimental
Materials
The polymer matrix used, for obtaining nanostructured composites, was PA 6.6, provided by Rhodia company (Brazil). MWCNTs used in this work were produced by chemical vapor deposition (CVD) technique and supplied by Bayer (Brazil), coded as 150 C Baytubes P. PA 6.6 was dissolved in a dispersion of CNTs and formic acid (CH2O2) 85%. The formic acid was supplied by the VETEC Química Fina LTDA (Brazil).
Morphological analyses
X-ray diffraction was performed in order to determine the crystallographic properties of the PA 6.6/CNT nanostructured composites. The tests were performed in a Philips Xpert 3060 PRO with 40 kV and 45 mA.
Thermal analysis
The crystallization kinetics study under isothermal and nonisothermal conditions was carried out using DSC PerkinElmer equipment model Pyris1 with cooling system called Intracooler 2. For nonisothermal conditions, the samples with approximately 6.0 mg were encapsulated in an aluminum standard sample pans and were initially heated from 160 to 290°C at 10°C min−1, leaving the samples for 1 min at this temperature to allow the complete melting of all PA 6.6 crystals, eliminating the remaining crystals that may act as germs during crystallization. Afterward, the samples were cooled at the cooling rates of 2.5, 5.0, 10, 20 and 50°C min−1 until 160°C, aiming the formation of exothermic peaks for determining the enthalpy crystallization.
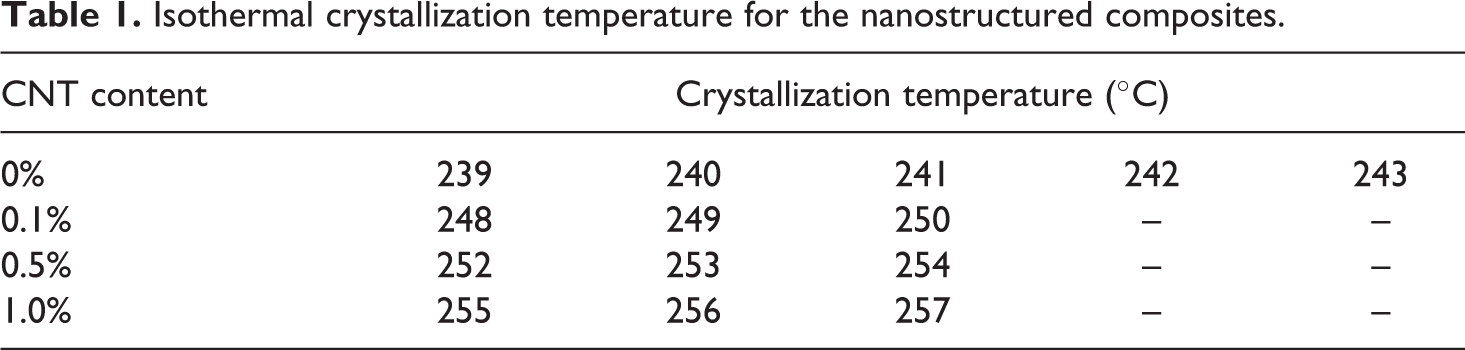
For isothermal conditions, initially, a dynamic analysis between 160 and 290°C at 10°C min−1 was performed to determine the melting and crystallization temperatures of the samples. The samples were heated again at 10°C min−1 from 160 to 290°C. However, in this second heating, the samples remained for 1 min at 290°C to allow complete melting of all crystals, eliminating its thermal history and avoiding the presence of the small crystals. Afterward, the samples were cooled at 100°C min−1 until the desired crystallization temperature isotherms (Table 1) and kept in isotherm condition until the formation of crystallization exothermic peak.
Isothermal crystallization temperature for the nanostructured composites.
Results and discussion
Morphological analyses
The X-ray diffraction is a method that has been widely used to obtain information about the interlayer spacing, structural strain and purity of the sample. Figure 1 shows the diffractograms obtained from the PA 6.6/CNT nanostructured composites. In this graphic, three distinct peaks can be observed at 2θ from 15° to 25° which are consistent with the crystal planes (002), (100) and (101), respectively. The presence of these peaks suggests the CNTs have been satisfactorily incorporated into the polymer matrix. Moreover, it was observed that (Figure 1) the increase in the CNT concentration in polymer matrix may enhance the intensity on the X-rays bands, leading to the gradual incorporation of CNTs in polymer matrix.
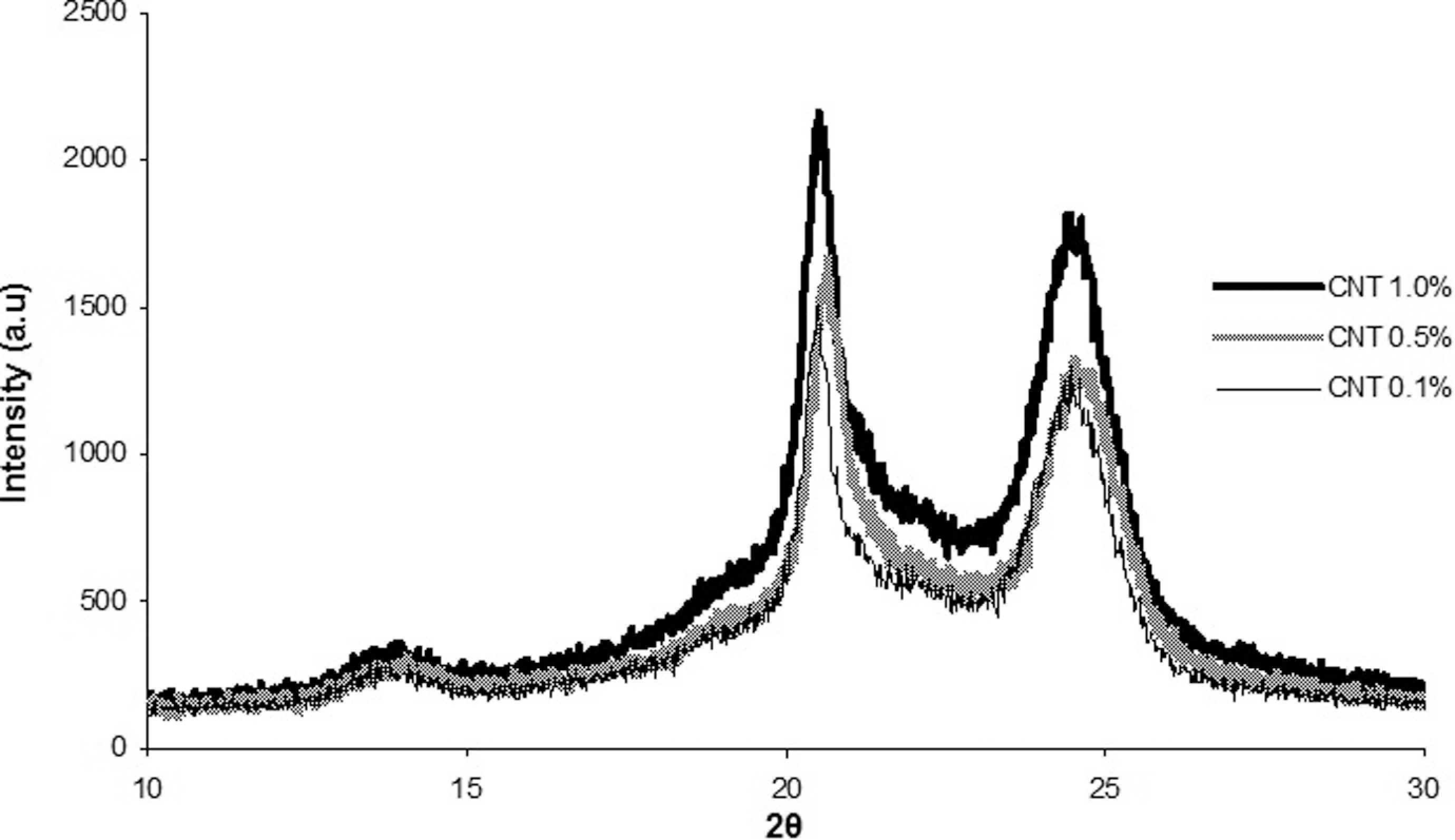
Diffractograms of polyamide 6.6/carbon nanotube (PA 6.6/CNT) nanostructured composites.
Thermal analysis
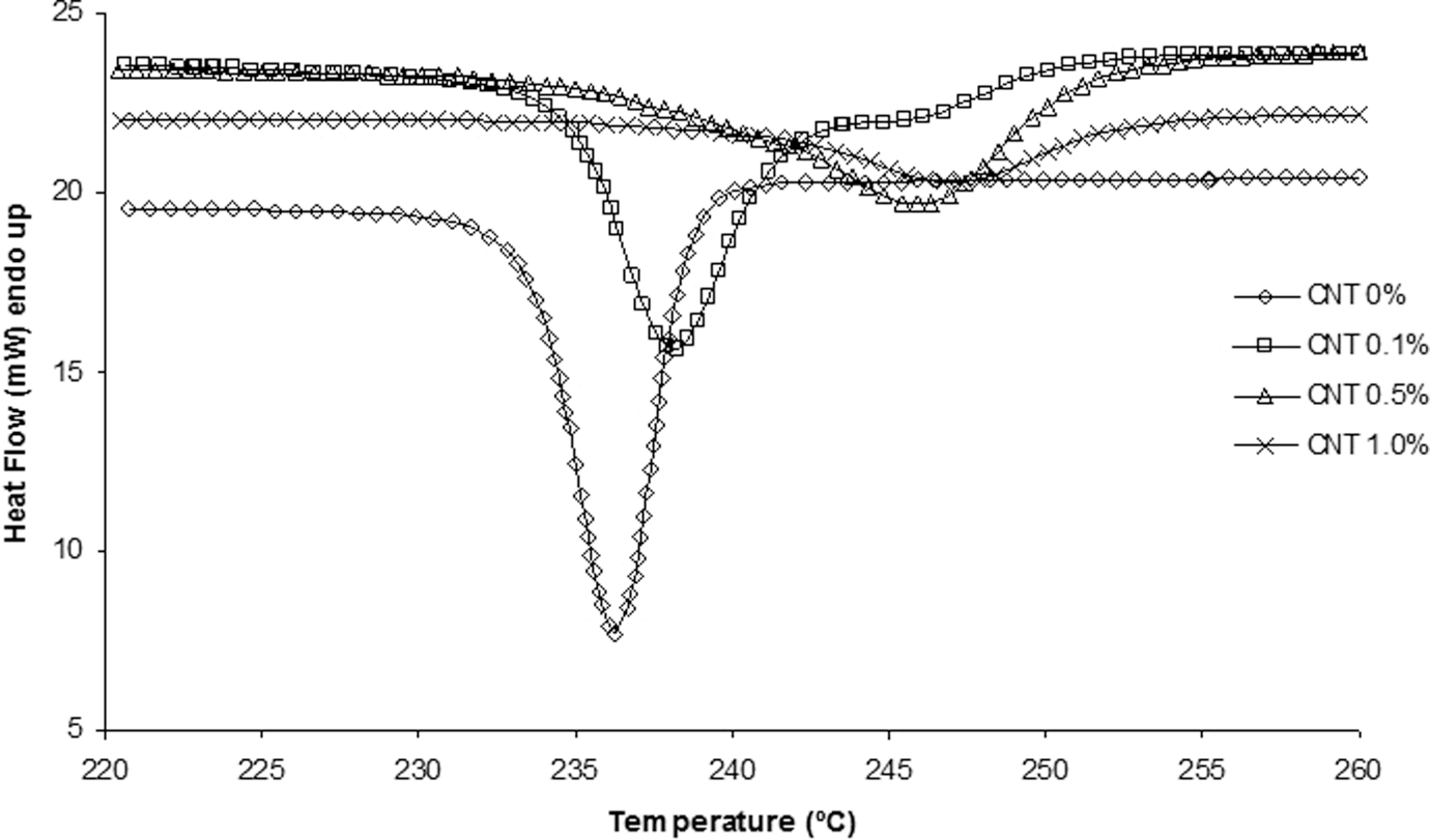
Isothermal crystallization curves for the neat polyamide 6.6 (PA 6.6) and its nanostructured composites.
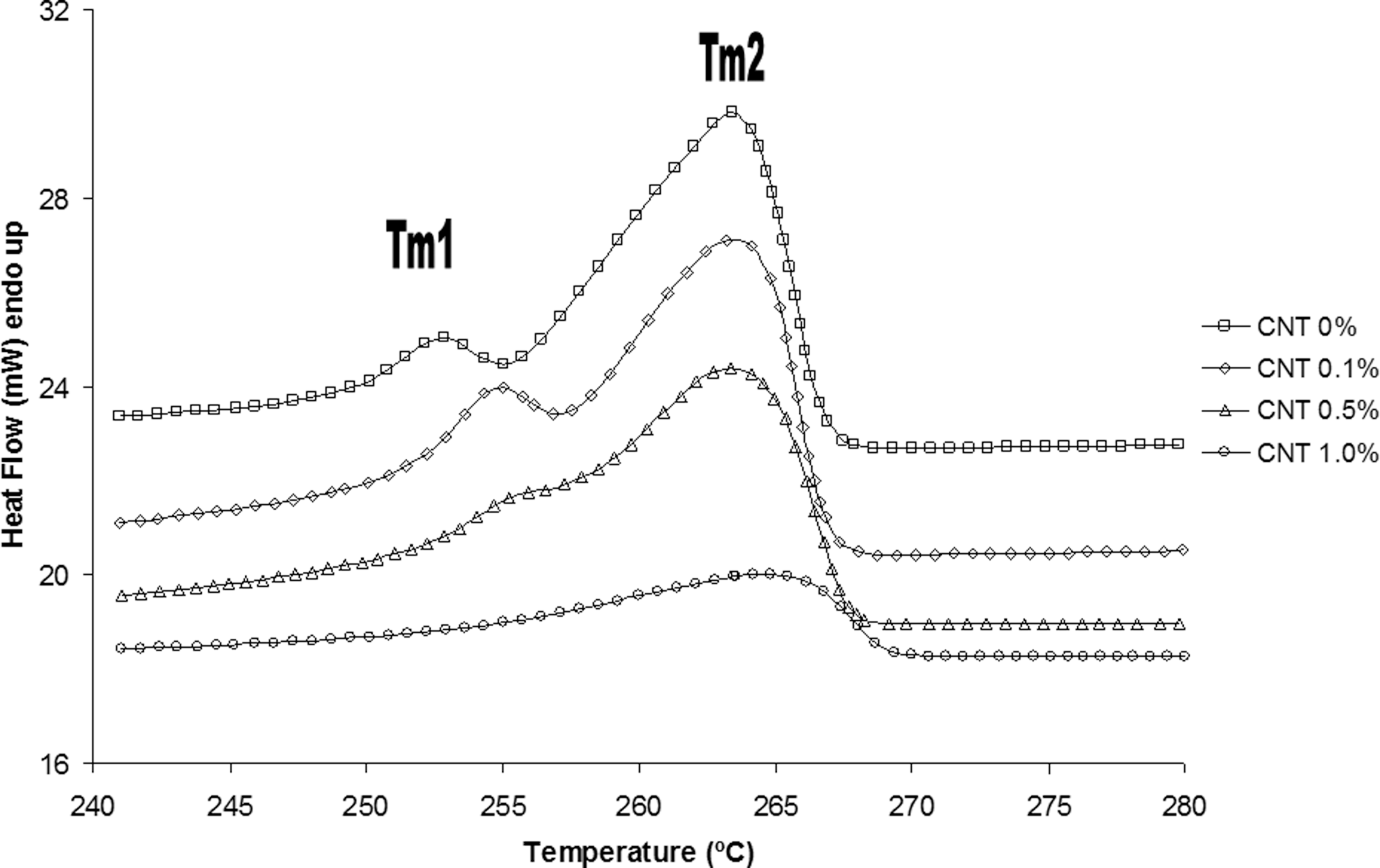
Nonisothermal melting curves for the neat polyamide 6.6 (PA 6.6) and its nanostructured composites.
It can be observed, from Figure 2, the crystallization peak temperature is 236°C for neat PA 6.6. The CNT addition in amounts from 0.1 to 1.0 wt% resulted in an increase in the crystallization peak temperature, but reducing its intensity (decrease the peak amplitude). This fact might be explained due to the presence of CNTs in PA 6.6, which generates active nucleus (germs) in the polymer matrix, advancing the crystallization process and, in turn, increasing the crystallization peak temperature and decreasing the crystallization peak amplitude, due to the disorder in the crystallization rate.
Figure 3 shows the results from DSC curve corresponding to the second heating for PA 6.6, indicating the polymeric matrix melting temperature occurs at 263°C (considering as the main peak of the higher intensity). According to the literature, 34 the PA 6.6 melting temperature is around 255–265°C. So, the temperature value found in the present work is within the range provided by the literature. 34 Besides that, the CNT addition does not change significantly the melting temperature of nanostructured composites, since the main melting peak remains within the range described in the literature. 34
Although the more representative melting peak of PA 6.6 occurs at 263°C (Tm2), the presence of two melting peaks called here Tm1 and Tm2 was observed (Figure 3). Tm1 was found at 253°C. According to the literature, 18,35 it is known that the PA 6.6 has different polymorphic phases, taking the dominant phase (α phase) melting temperature around 265°C and γ phase melting temperature in approximately 255°C. Thus, particular cooling rates lead to the formation of a variety of perfect or imperfect crystals. This effect of polymorphism might be seen on heating as the multiple melting peaks (Tm1 and Tm2 transitions) and, in this case, the last peak, more intense (higher amplitude), reflects the predominant crystal structure generated by the heat treatment applied. 36
Similar behavior is observed in the nanostructured composites when amounts up to 0.5 wt% of CNT were added into the polymeric matrix. Again, the presence of two melting peaks (Tm1 and Tm2) in the DSC curves may be observed for the nanostructured composites. However, the amount of CNT added in the nanostructured composite changes the form and intensity of the first melting peak (Tm1). The addition of 1.0 wt% of CNT in the PA 6.6 results in the extinction of this first melting peak (Tm1). Li and coworkers 34 also studied and discussed the behavior of multiple melting peaks for PA 6.6 and its nanostructure. According to the literature, 34 the occurrence of multiple peaks during the melting of nanostructure composites is due probably to the rearrangement of the crystalline lamellae PA 6.6. The first melting peak is also attributed to the thin lamellae formation that arises during the crystallization process, and Tm2 refers to the melting of crystalline structure achieved during the crystallization process. With the addition of CNT, a reduction in the melting peak area and, consequently, the melting enthalpy value were observed. In the present work, Tm1 disappears when amounts equal to or above 1.0 wt% CNT were used.
Figure 4 shows the curves of neat PA 6.6 and its nanostructured composites isothermal crystallization. These curves were used to study the PA 6.6 and its nanostructured composites isothermal kinetics. The CNT addition in polymer matrix, as previously discussed, increases the crystallization peak temperature, making it impossible to obtain isothermal temperatures equal for all nanostructured composites used in this study.
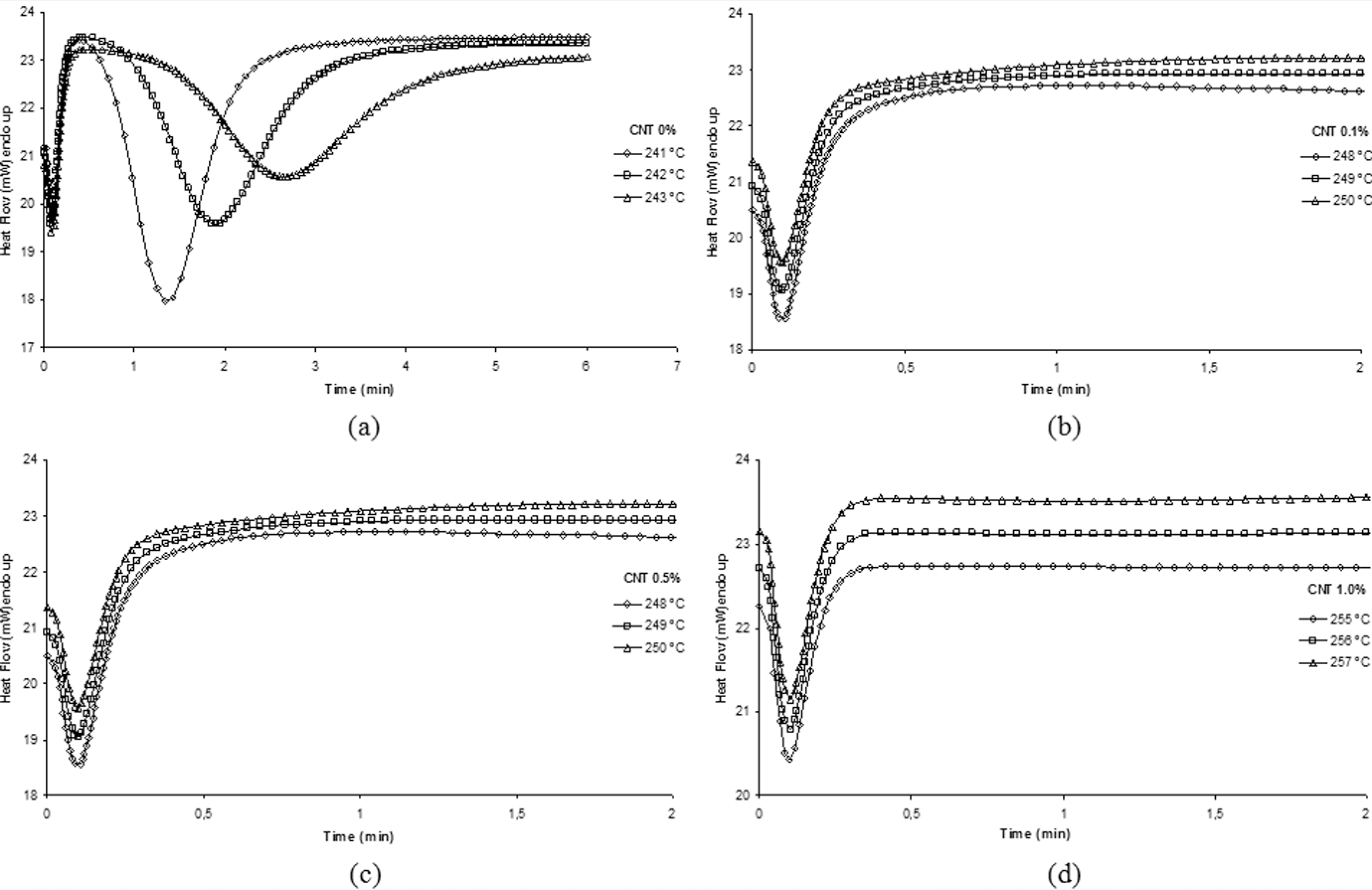
Isothermal crystallization curves for the neat polyamide 6.6 (PA 6.6) and its nanostructured composites: (a) 0 wt%, (b) 0.1 wt%, (c) 0.5 wt% and (d) 1.0 wt% carbon nanotube (CNT).
Figure 5 shows the curves of relative crystallinity (
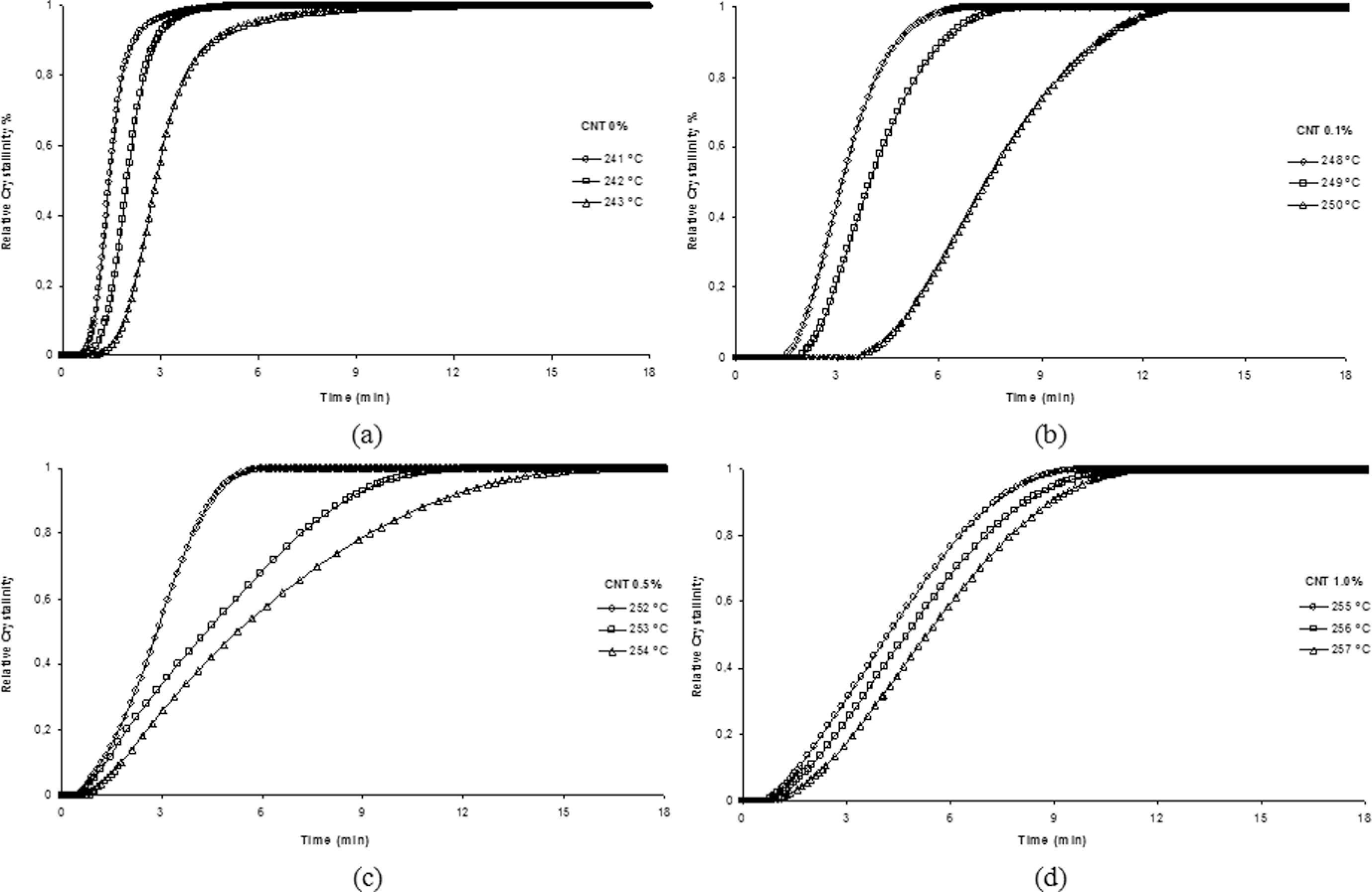
Relative crystallinity versus time at different isothermal crystallization for the neat polyamide 6.6 (PA 6.6) and its nanostructured composites: (a) 0%, (b) 0.1%, (c) 0.5% and (d) 1.0% carbon nanotube (CNT).
Table 2 provides the half-time life (
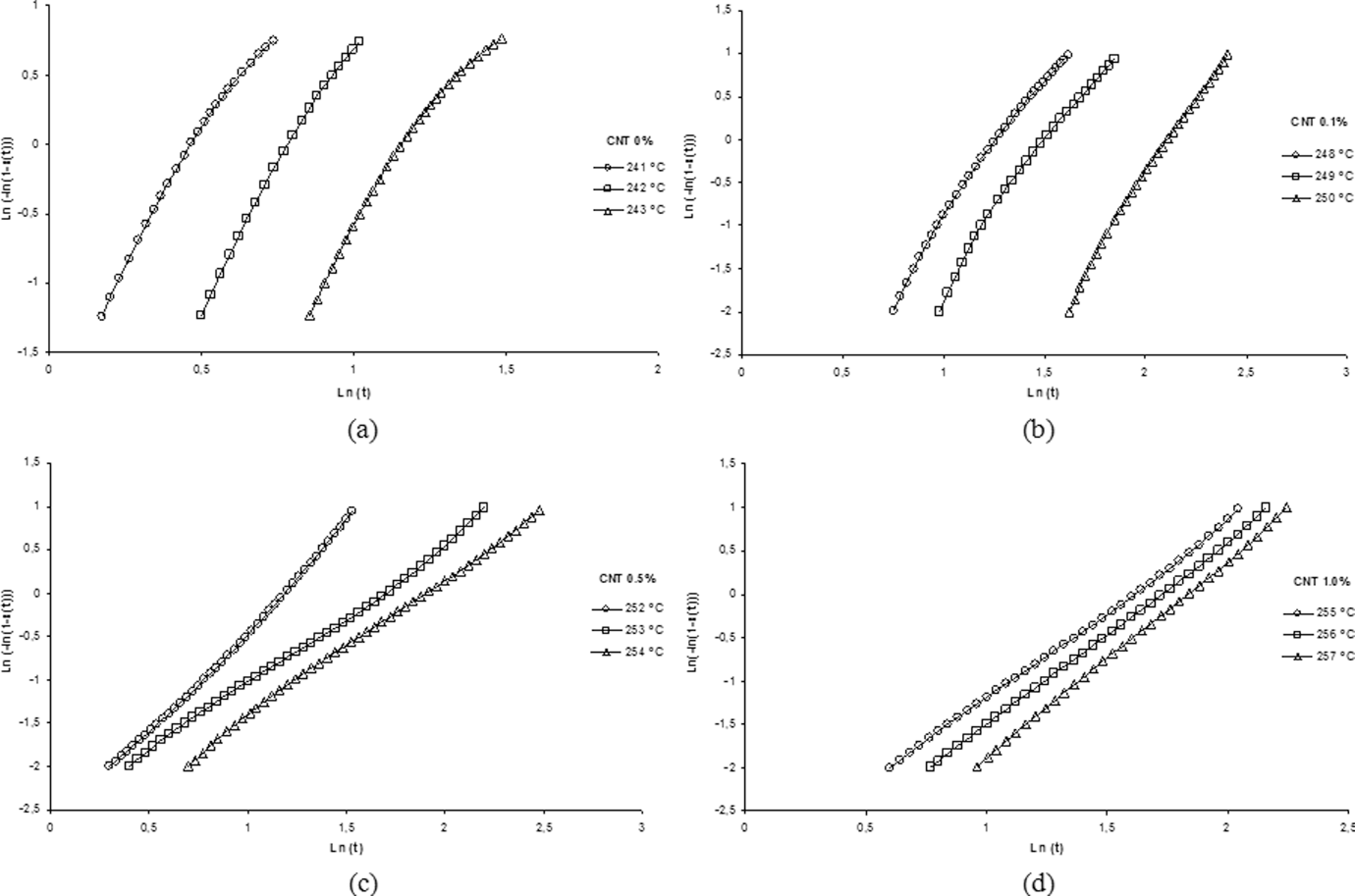
Avrami curves for isothermal crystallization of the neat polyamide 6.6 (PA 6.6) and its nanostructured composites: (a) 0%, (b) 0.1%, (c) 0.5% and (d) 1.0% carbon nanotube (CNT), with indicated temperatures.
Kinetic parameters for the neat PA 6.6 and its nanostructured composites at different temperatures of isothermal crystallization.
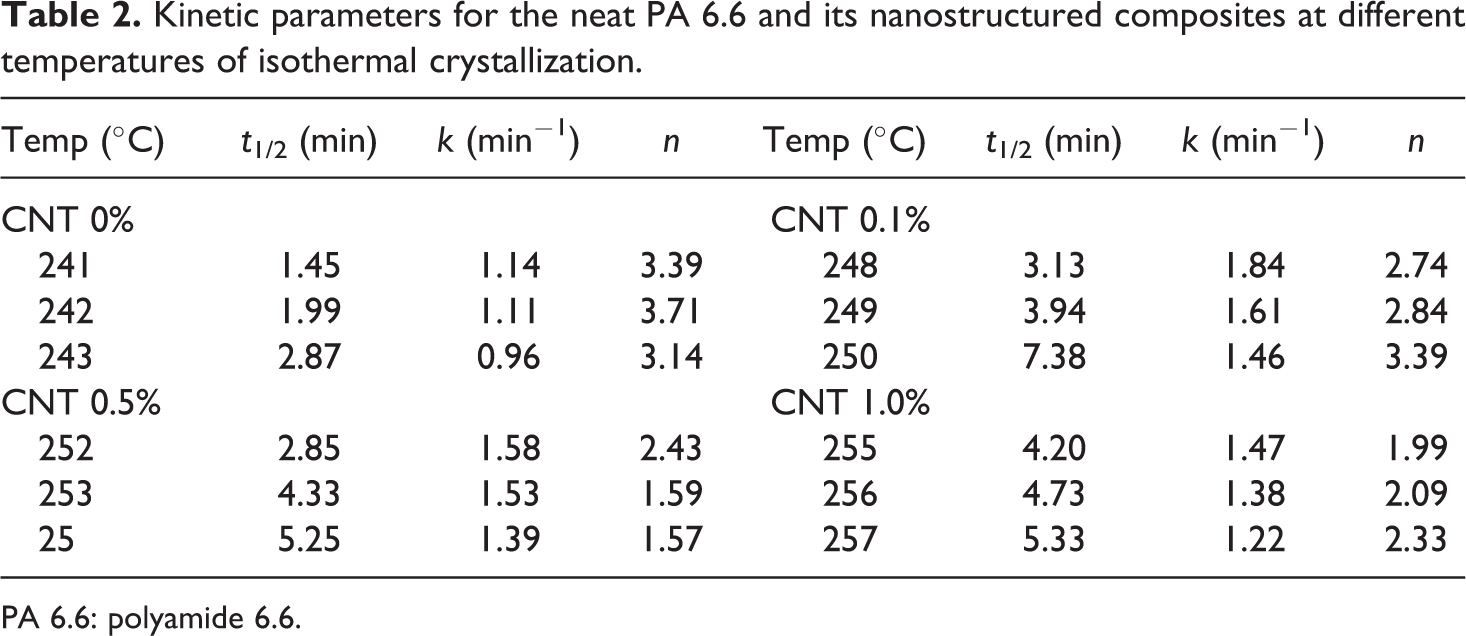
PA 6.6: polyamide 6.6.
Although the isothermal crystallization temperatures are different for each PA 6.6/CNT composite, it is observed (Table 2) that a tendency of
From Table 2 and Figure 6, it can be observed the Avrami exponent (
Nonisothermal crystallization kinetics
In this study, PA 6.6 and its nanostructured composites were analyzed by DSC using different cooling rates, in order to investigate the crystallization kinetics under nonisothermal conditions. During this study, the crystallization kinetics of nonisothermal samples was performed using the Avrami kinetic model.
Figure 7 shows the DSC cooling curves obtained for the neat PA 6.6 and its nanostructured composites at several cooling rates from 2.5 to 50°C min−1. From the curves shown in this figure, the following can be obtained: the peak temperature (
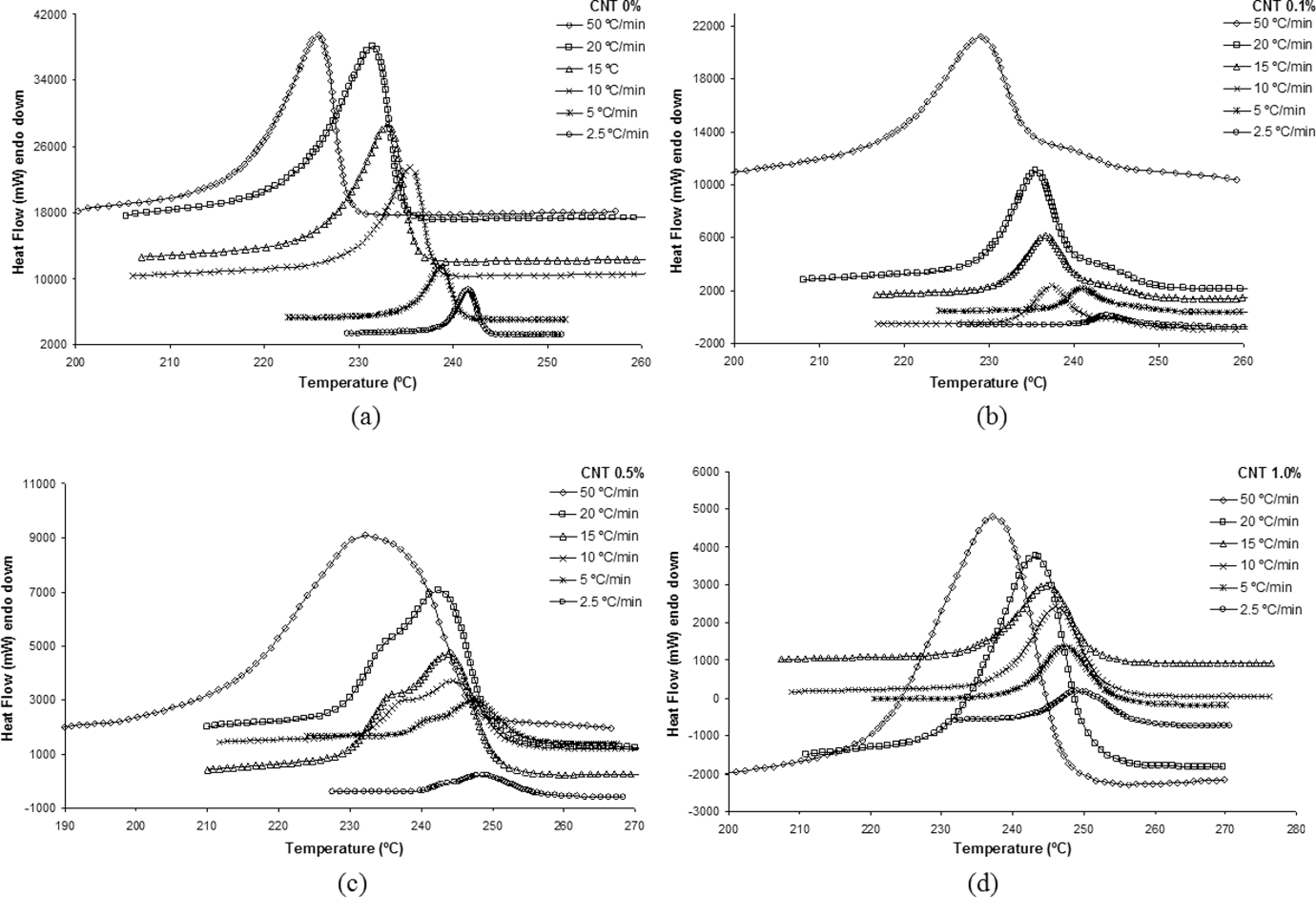
Differential scanning calorimetry (DSC) curves for the neat polyamide 6.6 (PA 6.6) and its nanostructured composites at different cooling rates: (a) 0%, (b) 0.1%, (c) 0.5% and (d) 1.0% carbon nanotube (CNT).
Kinetic data for neat PA 6.6 and its nanostructured composites during the nonisothermal crystallization.
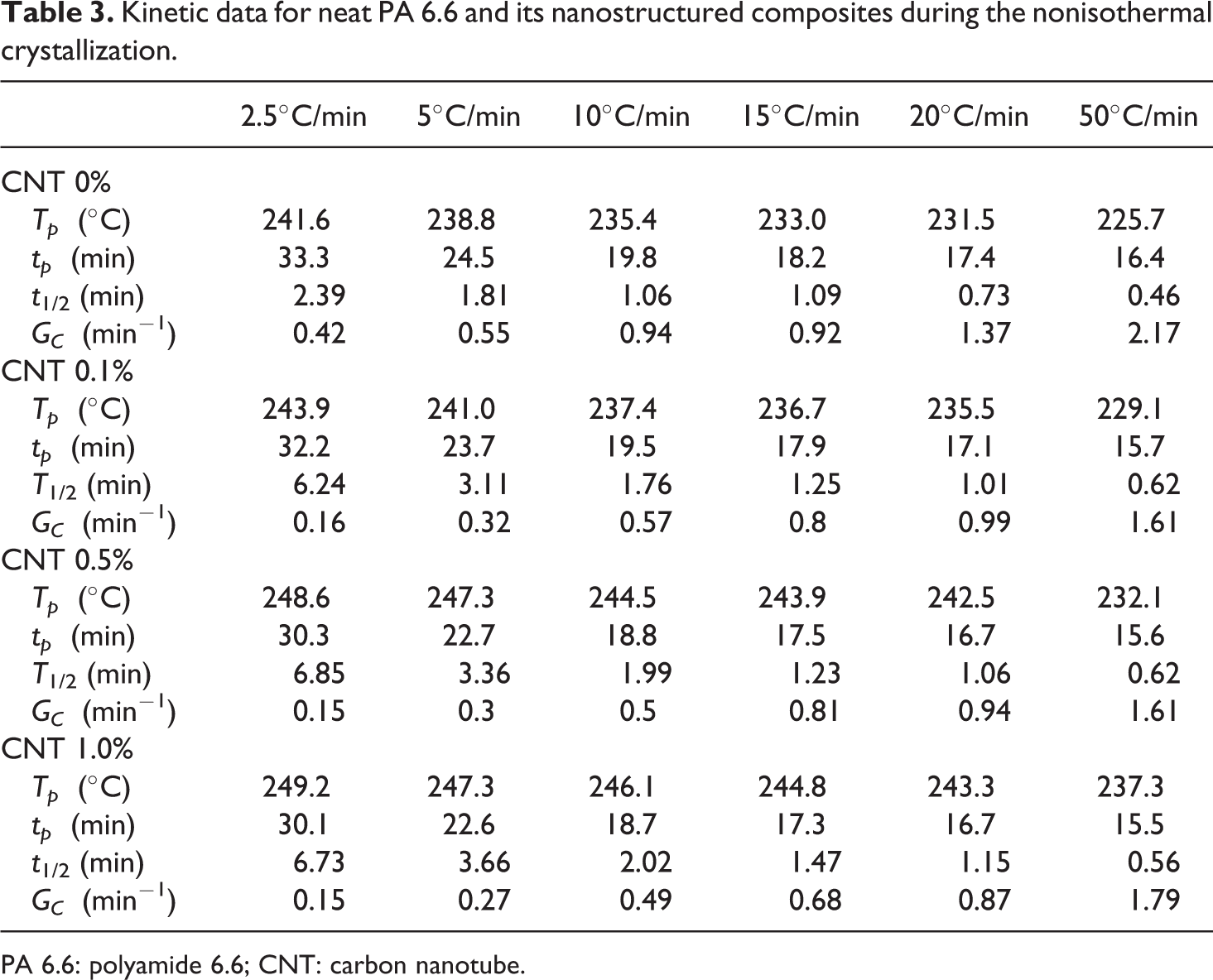
PA 6.6: polyamide 6.6; CNT: carbon nanotube.
According to Figure 7 and Table 3, the crystallization process is affected by the cooling rate used and the CNTs concentration present in the polymeric matrix (especially when considered 0.5% wt).
Similar to other parameters, the crystallization time (
The
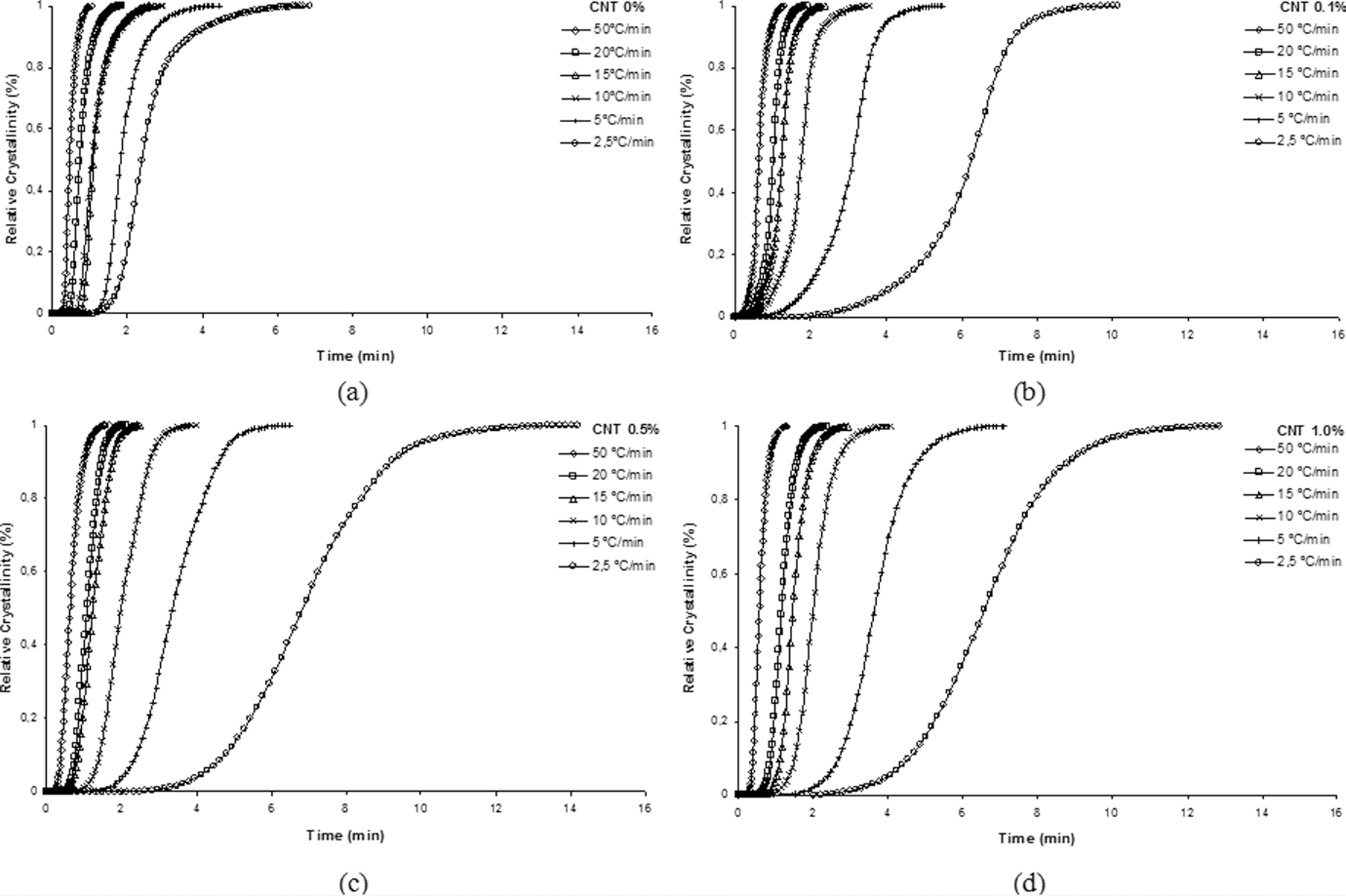
Crystallized fraction as function of time for the neat polyamide 6.6 and its nanostructured composites at different cooling rates: (a) 0%, (b) 0.1%, (c) 0.5% and (d) 1.0% carbon nanotube (CNT).
As can be seen from Figure 8, PA 6.6 and its nanostructured composites, when subjected to high cooling rates, crystallize in shorter times, so, the crystallization process becomes faster. This fact can be easily identified from the curves slope, where the increase in the slope means increasing the crystallization process rate. With increasing CNT concentration, it is observed that the crystallization process becomes slower, resulting in increased
Figure 9 shows the curves from the crystallized fraction rate (d
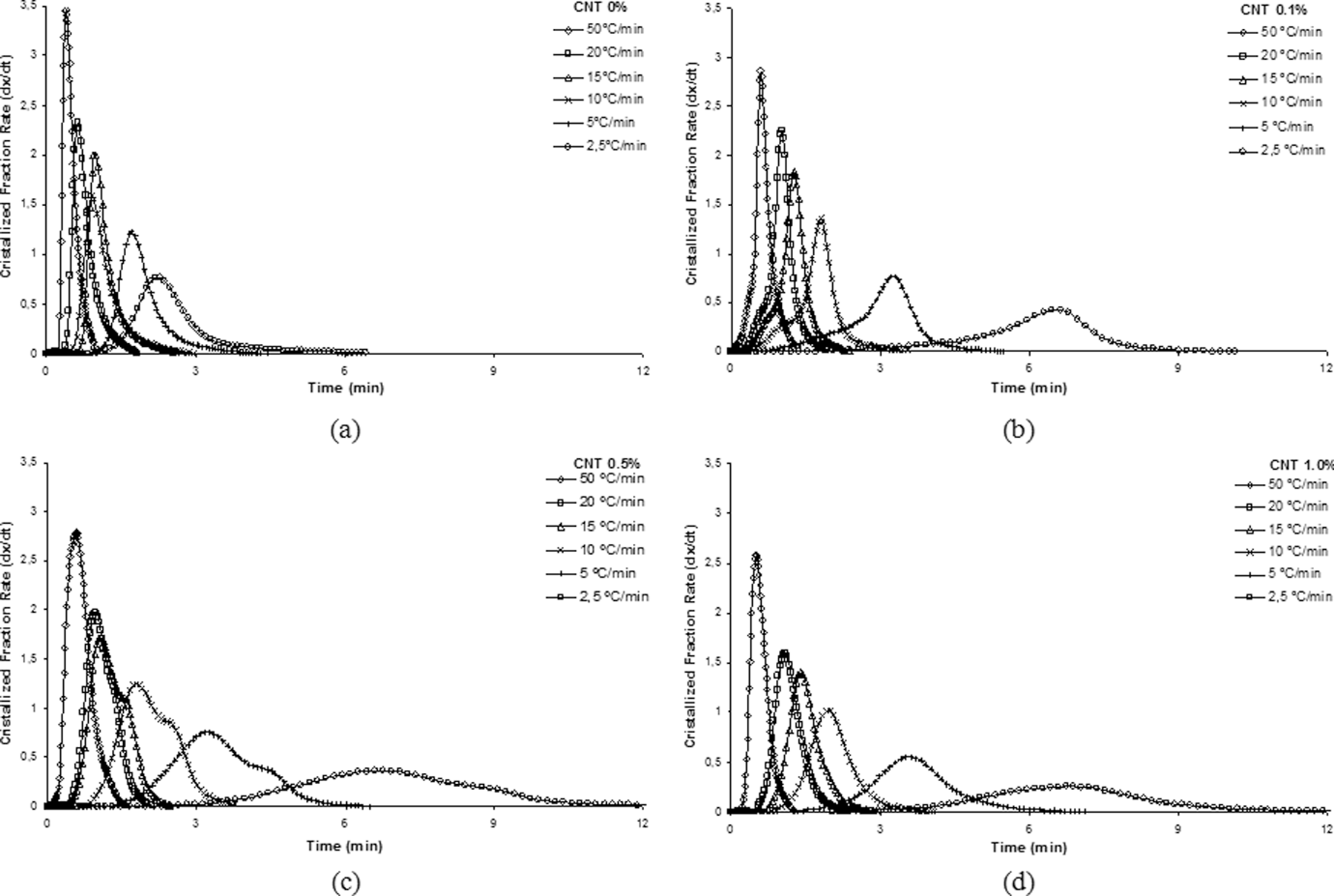
Crystallized fraction rate as function of time for polyamide 6.6 and its nanostructured composites at different cooling rates: (a) 0%, (b) 0.1%, (c) 0.5% and (d) 1.0% carbon nanotube (CNT).
Figure 10 shows the results from the curve
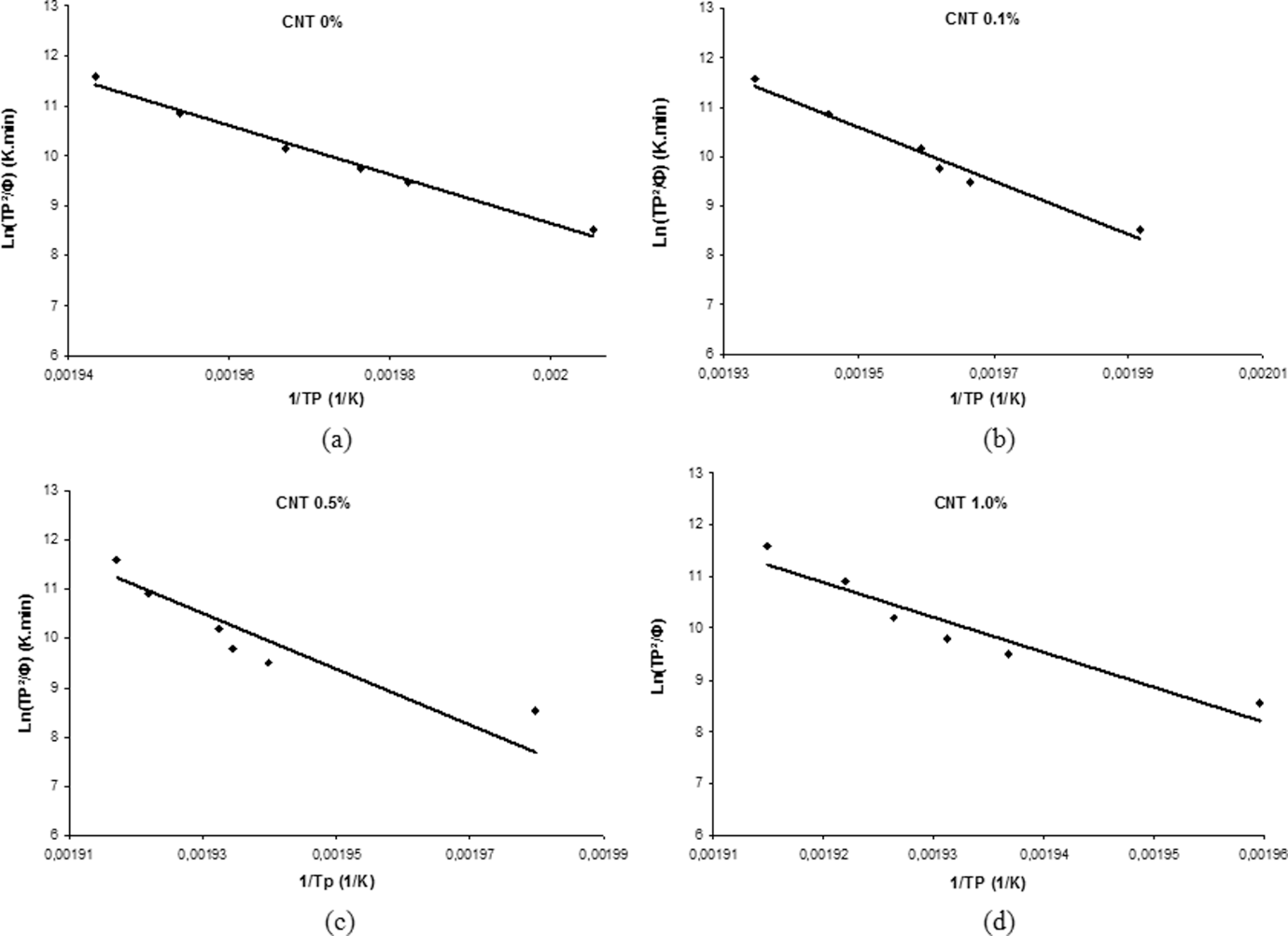
Kissinger plot for the neat polyamide 6.6 and its nanostructured composites: (a) 0%, (b) 0.1%, (c) 0.5% and (d) 1.0% carbon nanotube (CNT).
Activation energy for the neat PA 6.6 and its nanostructured composites.
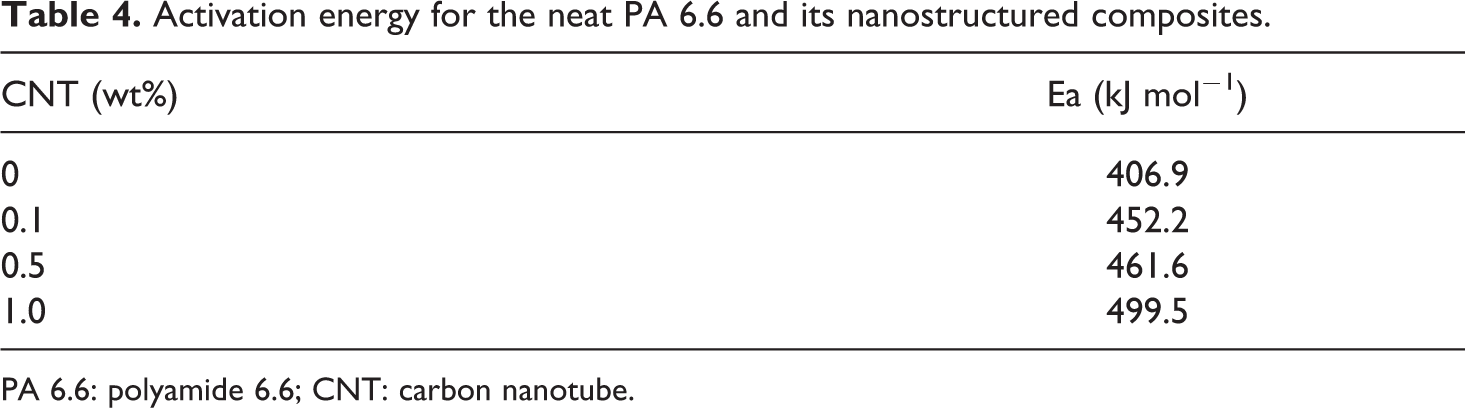
PA 6.6: polyamide 6.6; CNT: carbon nanotube.
According to the literature 32 and from the kinetic point of view, the activation energy is related to the crystallization rate. The increase in the activation energy makes crystallization difficult, resulting in the drop in crystallization rate. This result should be a consequence of the CNT dual effect on crystallization, as reported by Li and coworkers. 34 On one hand, CNT can serve as heterogeneous nucleation points and encourage the growth of crystallization at the molecular interface of the nanostructured composites. On the other hand, CNTs can hinder the migration of macromolecular polymeric segments melting on its surface, which is the region where the crystal growth occurs. This occurs due to the weak interaction between CNT and the polymeric segments, leading to an increase in activation energy and, consequently, reducing the crystallization rate.
From equation (4) and the kinetic parameters obtained in this work, the Avrami exponent
Kinetic parameters of the isothermal crystallization process for the neat polyamide 6.6 and its nanostructured composites.
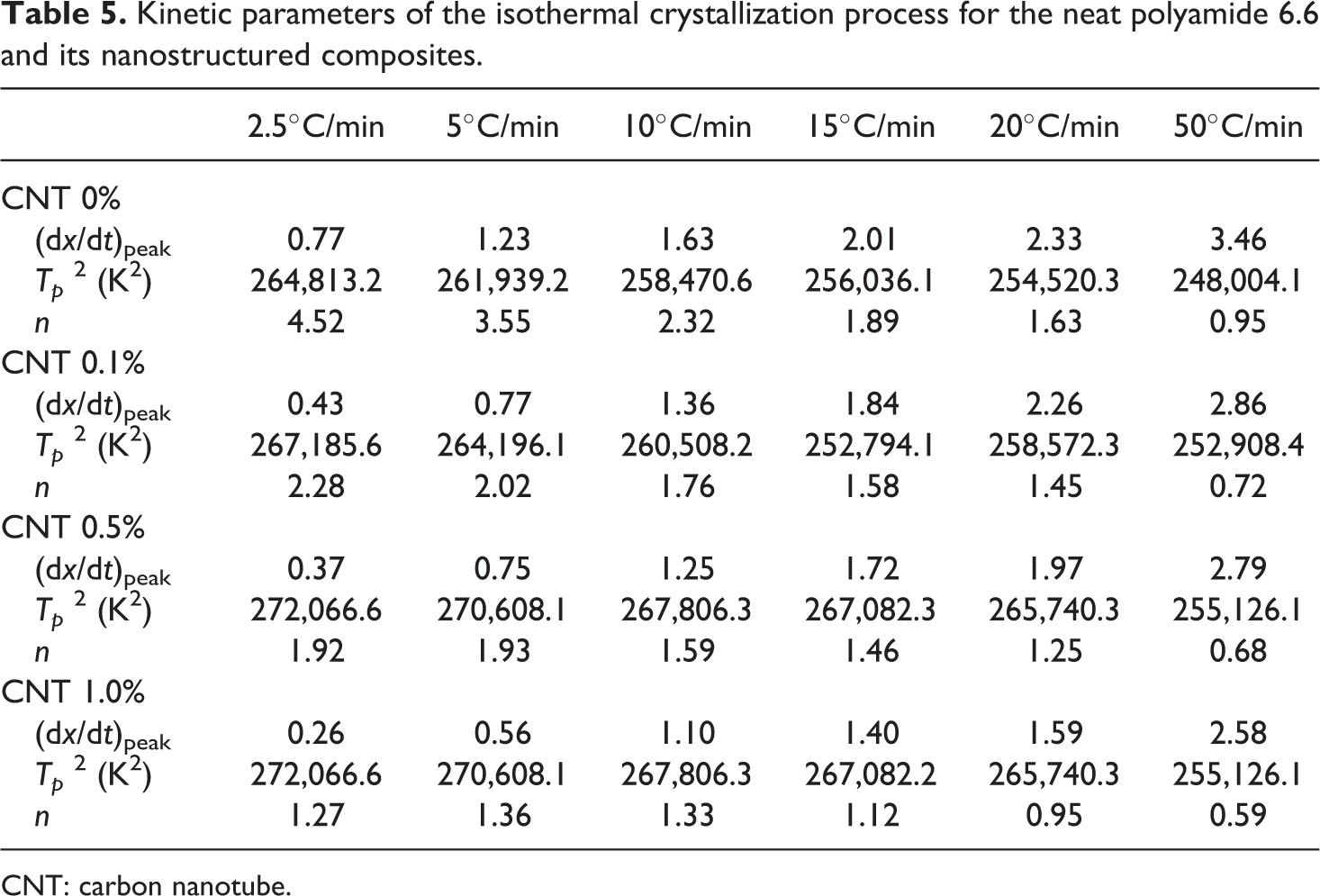
CNT: carbon nanotube.
As can be seen, the
However, the addition of CNTs in the polymeric matrix allows the beginning of nucleation at temperatures close to the melting temperature, or even inhibit the normal growth of the crystal, due to the short distance between the centers of nucleation, affecting the whole crystallization process. The addition of CNT in nanostructured composites provokes a reduction in the Avrami’s exponent values by changing the geometry of the crystals. 34 Thus, it can be concluded that both the cooling rate and the addition of CNTs in nanostructured composites affect the crystallization kinetics of the composites. For the same cooling rate, the CNT concentration in nanostructured composites reduces the Avrami exponent and consequently modifies the crystallization kinetics.
Conclusion
The PA 6.6 crystallization kinetic is influenced by the CNT addition. The presence of CNT in the polymeric matrix reduces the crystallization rate and the Avrami exponent, modifying the formation and growth of crystals in nanostructured composites. This effect is attributed to the formation of a greater number of crystallization nuclei.
From the crystallization kinetic study, under dynamic conditions of nanostructured composites concluded that both the rate of cooling and the addition of CNTs affect the crystallization kinetics of the system. For the same cooling rate, the concentration of CNTs in nanostructured composites reduces the Avrami exponent and consequently its crystallization kinetics. The crystallization time also remains virtually unchanged for the 1.0 wt% concentration, suggesting that the 0.5 wt% concentration is the limit for the variation in the crystallization time of the composite nanostructured.
The crystallization kinetic behavior under isothermal conditions showed the presence of two melting peaks called Tm1 and Tm2 . The addition of CNT in the polymer changed the format and intensity of the first melting peak (Tm1), and the addition of larger quantities of CNTs resulted in the extinction of this peak.
The deformed S-shaped curve format seen in the graph of the crystallization with the isothermal crystalline melt time indicates that the primary stage of crystallization is very fast, becoming slower with increasing isothermal temperature. This same effect is observed on the polymeric matrix with increase in CNT concentration (proved by reducing the slope of the crystallized fraction).
Footnotes
Funding
This research received the financial support from FAPESP (under grants 05/54358-7 and 08/00171-1), CAPES (0023/2008) and CNPq under grants 306053/2006-3 and 151154/2009-0.
