Abstract
Gelatin films were prepared from gelatin granules in aqueous medium by casting. Tensile strength, tensile modulus, elongation at break and glass point of the gelatin films were found to be 30 MPa, 450 MPa, 7.1% and 56.5°C, respectively. After irradiation with gamma radiation, the tensile properties increased due to the dense network structure. A series of gelatin solutions was prepared by blending varying percentages (1–10% by wt) of ethylhexyl acrylate and then films were prepared and irradiated with gamma radiation. It was found that the tensile and thermal properties of gelatin films improved significantly. Water uptake of the gelatin films was evaluated.
Introduction
Currently, the idea of replacing commonly used plastics prepared from synthetic polymers with biodegradable plastics prepared from natural polymers is increasingly interesting. Gelatin is one of the biodegradable and environmentally friendly biopolymers, which are now widely studied. It is a high-molecular-weight polypeptide composed of amino acids mainly glycine (27%), hydroxyproline and proline (25%). 1 Because its molecules are tightly bound with hydrogen bonds, pure gelatin films are normally brittle. Gelatin is water soluble and it dissolves in hot water and forms a jelly when cooled. Gelatin is obtained by the thermal denaturation of collagen from animal skin, bones and, rarely, fish scales. Collagen occurs in all types of multicellular animals, invertebrates and vertebrates and forms the major fibrous elements of the extracellular connective tissue. Gelatin is a degraded form of collagen which is a connective tissue protein present in most of the vertebrates, 2 and it has been widely used in the biomedical field due to its advantages, including biological origin, biodegradability, hydrogel properties, commercial availability and relatively low cost. 3 The presence of higher levels of pyrrolidines in gelatin results in the formation of stronger gels. 4 The strength of the gelatin film is due to the presence of triple helixes. 5 Gelation process includes rearrangement of individual molecular chains into ordered, helical arrangement or collagen fold 6 –8 and association of two or three ordered segments to create crystallites. Finally, the structure is stabilized by lateral interchain hydrogen bonding within the helical regions. 9 ,10
Gelatin has also been used in a wide variety of wound dressings. 11 ,12 The selection of gelatin as a component for preparation of the blend film rests up on two reasons. First, gelatin is a connective tissue protein, well known for its nontoxic, nonirritant and biodegradability properties and good living body compatibility; therefore, it has been widely used in food, pharmacology and cosmetic applications. 13 It is most frequently used in hard and soft capsules, microspheres and three-dimensional tissue regenation. 14 Second, the formation of specific intermolecular interaction through hydrogen bond of two or more polymers is responsible for the observed mixing behavior and properties of the blends. 15 The selection of gelatin is also justified with this point of view. One of the drawbacks of gelatin is that the polar groups present in its structure cause a gelatin film to have high moisture absorption. Due to these disadvantages, pure gelatin films are not suitable for many applications. Therefore, gelatin is generally blended or copolymerized with other synthetic polymers. 16 –18 Blending is easy to handle but may encounter the incompatibility between hydrophilic gelatin and hydrophobic synthetic polymers. Graft copolymerization 19,20 is used to improve the properties of gelatin. Researchers have reported the successful graft polymerization on gelatin and subsequent change in the thermal and structural properties of gelatin. 21
Research has been carried out previously to explore the irradiation effects on gelatin. It is reported that gamma radiation increased the cross-linking between protein chains, which in turn increase the mechanical properties of the film. 22 Cheorun et al. 23 observed that gamma radiation enhances the tensile properties (tensile strength [TS] and elongation at break [Eb]) of the pectin- and gelatin-based films. Gamma radiation decontaminates the edible gelatin and is also capable of increasing the digestible energy of agricultural by-products. 24 A great deal of work has also been carried out on gelatin renaturation, 25,26 and it may be mentioned that the gelatin is a denatured product of collagen. Denaturation is a structural change in a protein that results in loss of its biological properties. 27 Due to denaturation, proteins lose its characteristic properties such as solubility in solvent, enzymatic activity, etc. It also affects the functional property of protein. Physical modification of gelatin includes renaturation of the collagen-like helical structure and orientation of gelatin macromolecules. Renaturation is the conversion of denatured protein to its native configuration. The present study deals with the development of a new method to enhance and improve the mechanical properties of gelatin films. The aim of the study was to develop and characterize cross-linked gelatin films with 2-ethylhexyl acrylate (2-EHA) using Co-60 gamma radiation. The thermal properties of the raw and irradiated gelatin films were also investigated.
Experimental
Materials
Gelatin (bloom strength-185; pharmaceutical grade) was obtained from the Global Capsules Ltd (Bangladesh). The monofunctional monomer, 2-EHA, was obtained from Fluka (Switzerland).
Preparation of gelatin films
Granules of gelatin (15 g) were mixed in distilled water (100 ml) and then heated to 90°C for 1 h until it reaches a viscous state. The concentrated gelatin solution was cast onto release film (silicon cloth)-covered frame mounted on a flat glass plate for film formation and then dried in an oven for 8 h at 70°C. Different percentages (1–10%) of monomer 2-EHA were mixed to obtain different formulations and heated at 90°C for 1 h. Five formulations were prepared, which are termed as F-0 to F-4; their compositions are presented in Table 1. The mixture was stirred continuously for homogeneous mixing and gelatin/2-EHA films were prepared following the same technique. The dried films (about 0.25 mm thickness) were peeled off and cut into small pieces of length 50 mm and width 10 mm using conventional scissors. The films were irradiated with gamma radiation (Co-60) with different intensities from 100 to 500 krad at a dose rate of 300 krad/h. Tensile properties of the gelatin films were measured with Universal Testing Machine (Hounsfield Series S, UK). The load range was 500 N; efficiency was within ±1%. The crosshead speed was 10 mm/min. Gauze length was 20 mm. Data presented here are average values of at least five samples.
Composition of 2-EHA blended gelatin formulations (% w/w) for film preparation.
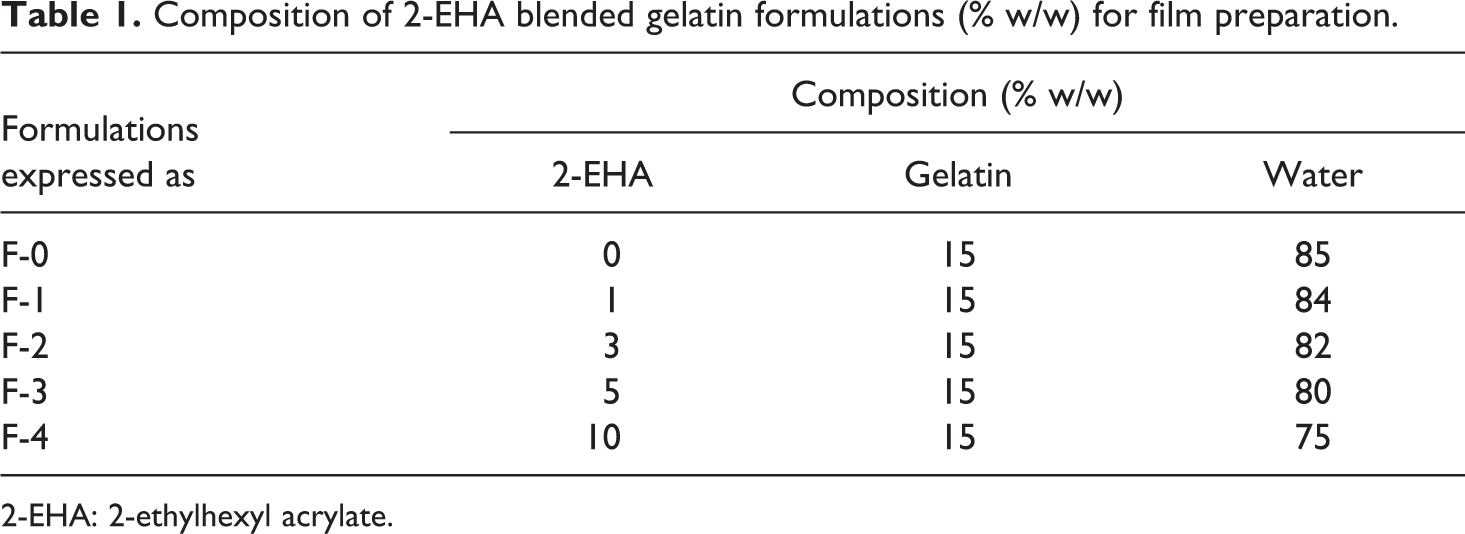
2-EHA: 2-ethylhexyl acrylate.
Thermal analysis of the gelatin films
The thermal properties of the untreated and treated gelatin films were determined using thermomechanical analyzer (LINSEIS TMA, L-77, USA). The experiment was carried out in an inert atmosphere at a heating rate of 5°C/min. The dimension of the sample was 5 × 5 × 1 mm3.
Water uptake
The gelatin films were weighted and then immersed in distilled water for different periods of time at 25°C. Wet samples were wiped with tissue paper to remove the excess liquid and reweighed. The amount of absorbed water was calculated by the following equation: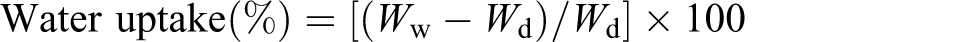
Results and discussion
Tensile properties of pure- and gamma-irradiated gelatin films
Tensile properties (TS, tensile modulus [TM] and Eb) of the gelatin films were investigated before being treated with 2-EHA monomer. TS, TM and Eb of the gelatin films were measured after 3 days of the film formation. The gelatin films were irradiated at different doses (100, 150, 200, 300 and 500 krad) at a dose rate of 300 krad/h. The results are given in Figures 1 and 2. The TS, TM and Eb of the pure gelatin films are found to be 30 MPa, 450 MPa and 7.1%, respectively (expressed as 0 krad in the figures). It was found that the TS and TM of the gelatin films increased with increasing gamma radiation and then decreased. At 100, 150 and 200 krad doses, the TS and TM of the irradiated films were obtained 35, 38, 43 MPa and 720, 756, 828 MPa, respectively, which was about 17, 27, 43% and 60, 68, 84% higher than that of the pure gelatin film. After that, an increase in radiation dose decreases the TS and TM values gradually. At 300 and 500 krad, the TS and TM value goes to 41 and 39 MPa and 747 and 716 MPa, respectively. These values are still higher than the pure gelatin film. So, with the exposure of gamma radiation, the TS and TM of pure gelatin film increases.
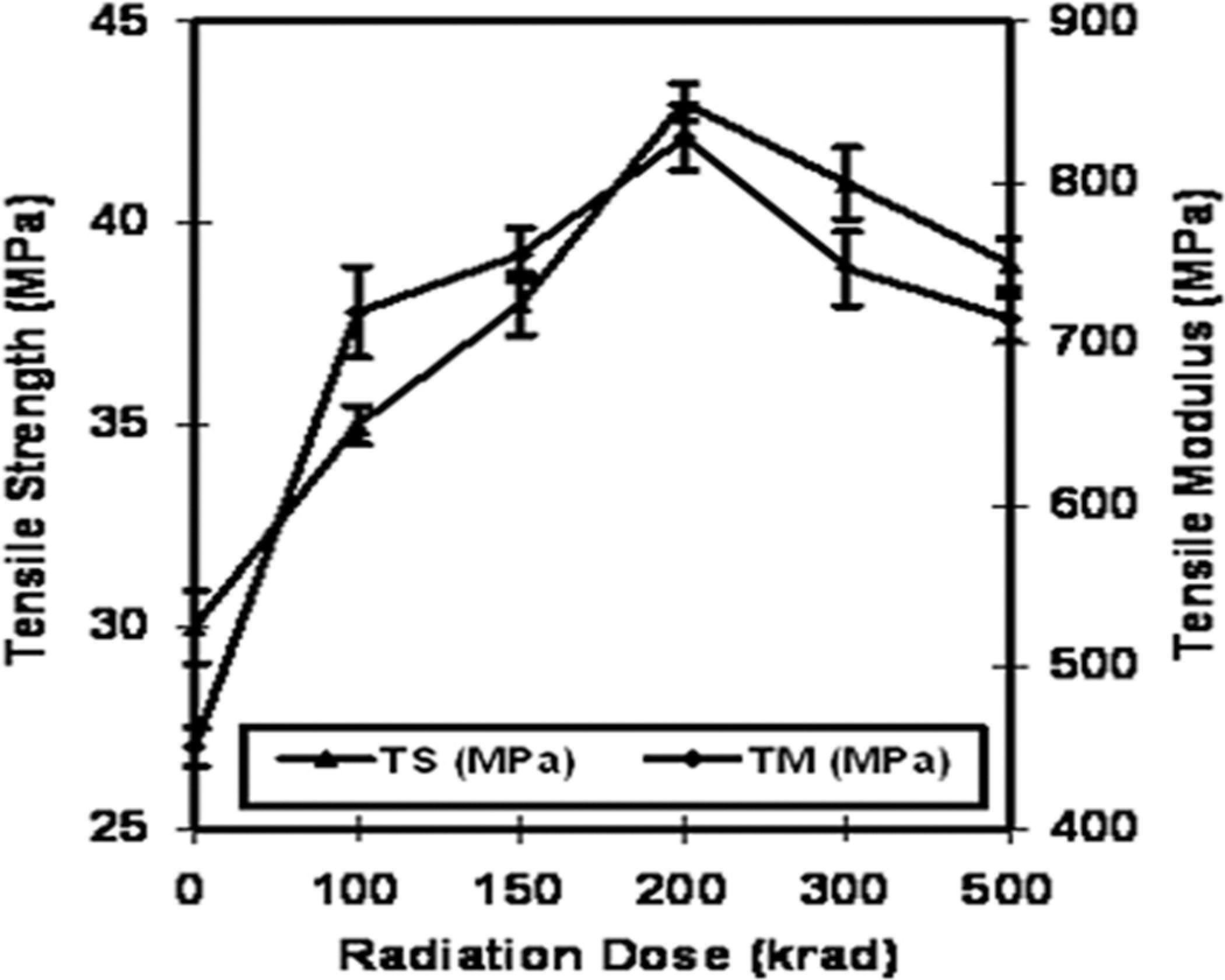
Tensile strength and tensile modulus of pure gelatin films against radiation dose.
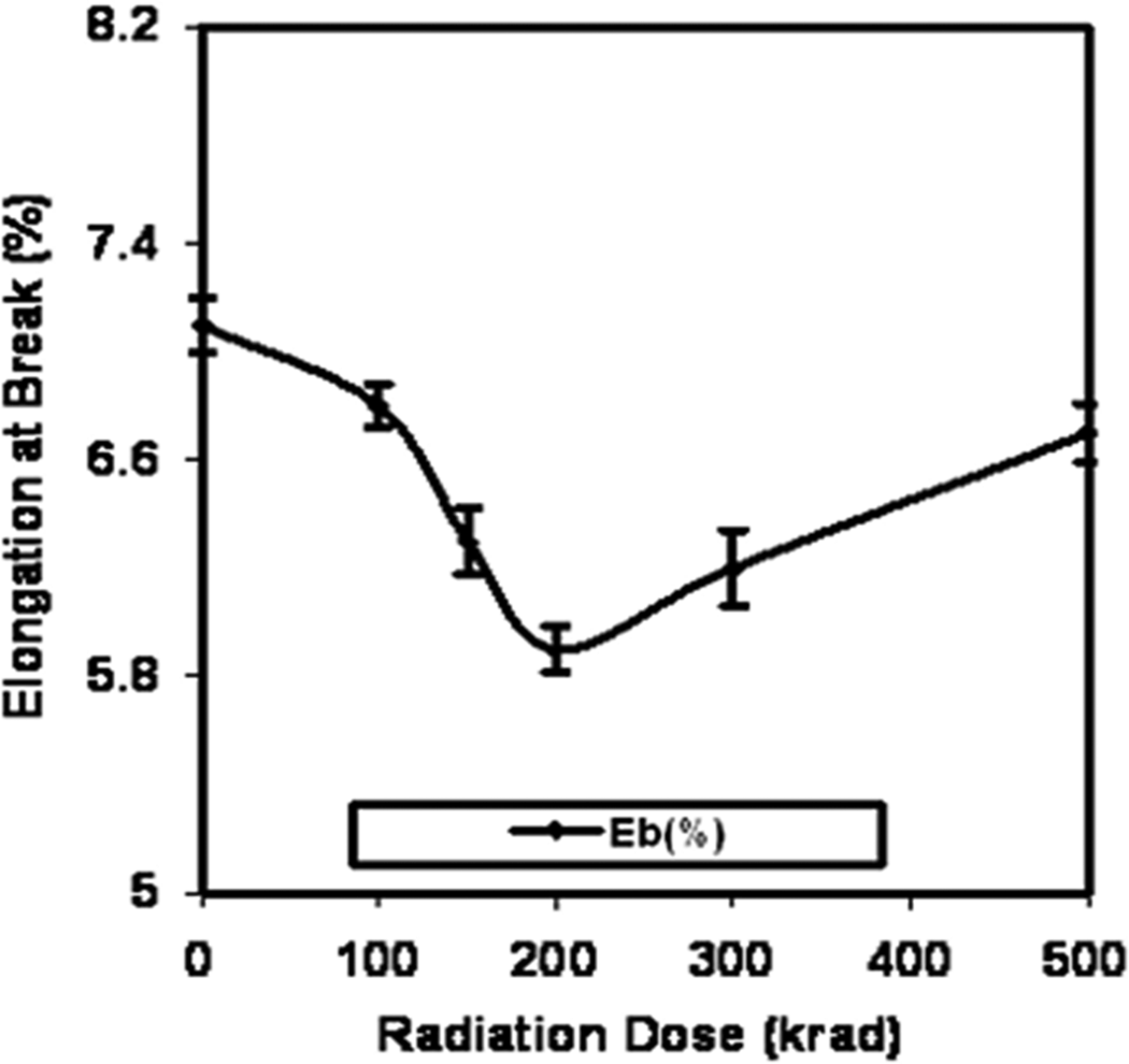
Elongation at break of pure gelatin films against radiation dose.
The Eb values of the irradiated gelatin films are shown in Figure 2. It is found that the Eb value decreases significantly at 200 krad radiation dose. Further increase in radiation dose increases the Eb values but the values are still lower compared with nonirradiated gelatin films. At 100, 150, 200, 300 and 500 krad doses, the Eb values are 6.8, 6.3, 5.9, 6.2 and 6.7%, respectively, which are 4, 11, 17, 13 and 6% lower compared with nonirradiated gelatin films. It is clear that the exposure of gamma radiation on the gelatin films increases the TS and decreases the Eb, which is expected. Gelatin is a natural polymer and gamma radiation has very promising effect on it. Cross-linking and chain scission occurred when polymers are irradiated using gamma radiation. 28 Polysaccharides and other natural polymers generally degrade by breaking the glycosidic linkage with gamma radiation. 29 Gelatin is a natural biopolymer consisting of protein molecules that can easily fragment due to gamma radiation. The nature of probable chain scission of gelatin molecule with the exposure of gamma radiation is given in Figure 3, where some of the probable free radicals are shown. The generated free radicals may cross-link with each other and form more cross-linked sites with the exposure to gamma radiation; as a result, the tensile properties might increase. Khan and coworkers have already discussed some of the probable free radicals. 30
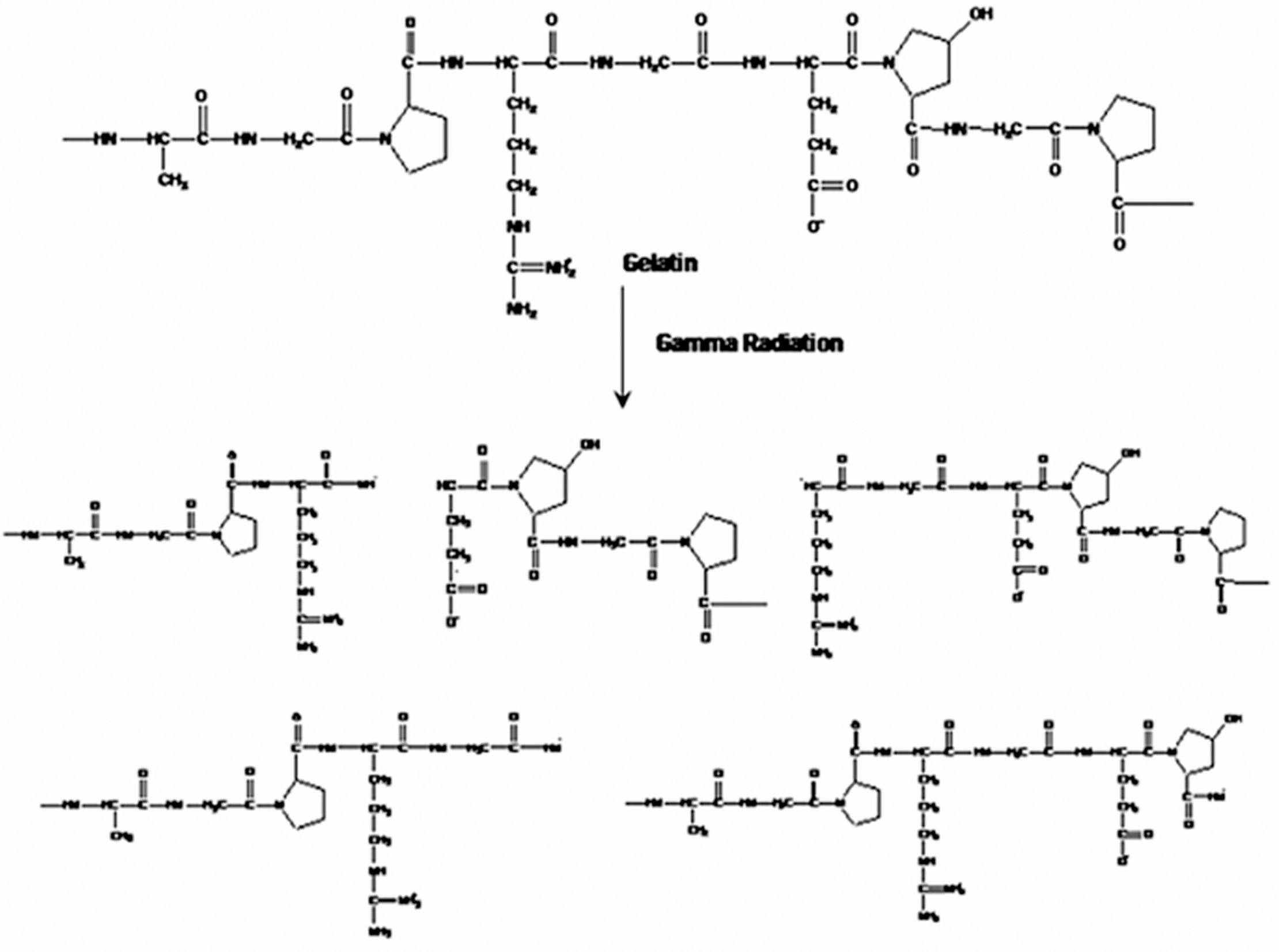
Probable free radicals generated from gelatin with the exposure of gamma radiation.
Effect of EHA on gelatin film (nonirradiated)
The tensile properties (TS, TM and Eb) of the 2-EHA grafted gelatin films are given in Table 2. From Table 2, it is observed that raw gelatin films grafted with 5% EHA (F-3 formulation) showed the highest TS (51 MPa) and TM (1012 MPa), which is about 70 and 125% enhancement. Monomer concentration plays an important role because it affects the polymerization and the mechanical properties of the cross-linked products. 2-EHA influences the tensile properties of the gelatin films extensively. The TS and TM values increase up to 5% of the monomer concentration and then decrease with increase in monomer concentration which leads to the homopolymer formation. 31 Higher monomer concentration may lead to the homopolymer formation causing the decrease in TS and TM. As the monomer concentration increases, more 2-EHA may undergo polymerization with gelatin to give an extended carbon chain attached to the side group of the gelatin molecules and the TS and TM increased. But at higher concentration, the monomers produced homopolymer (2-EHA + 2-EHA) and the TS and TM decreased. For all formulations, the Eb values decrease and F-3 shows the lowest Eb, which is about 18% lower compared with the gelatin films. As the incorporation of 2-EHA increases the TS and TM of the films, the Eb value decreases.
Effect of 2-EHA on gelatin films.
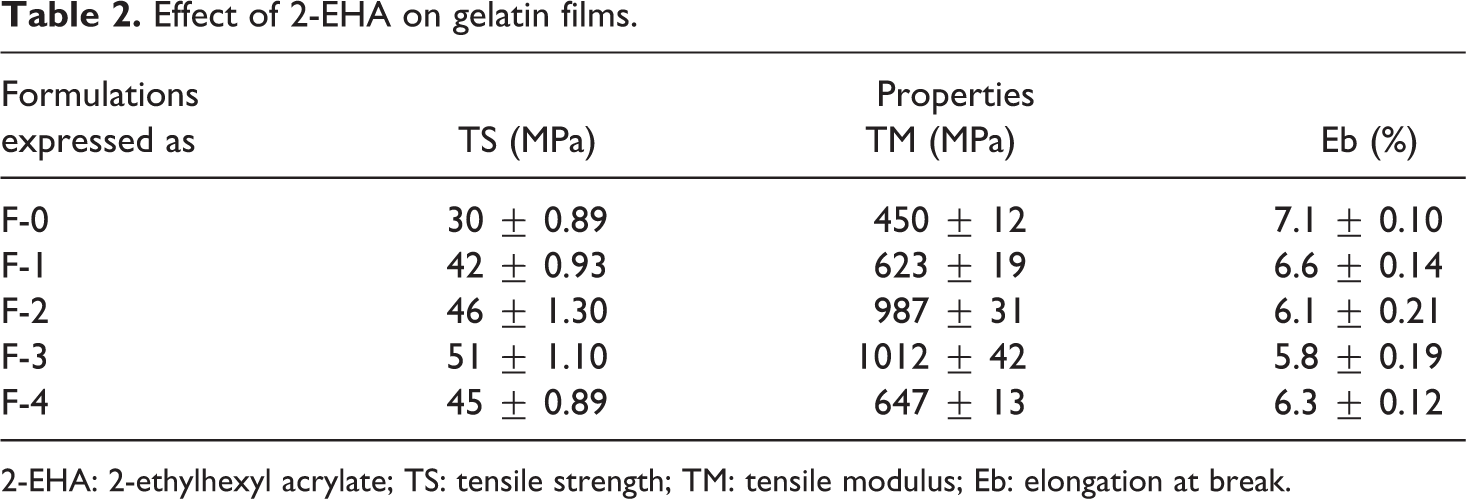
2-EHA: 2-ethylhexyl acrylate; TS: tensile strength; TM: tensile modulus; Eb: elongation at break.
Effect of EHA on gelatin film (irradiated)
Gelatin films are prepared using various percentages of 2-EHA (1, 3, 5 and 10%) to study the effects. Then the films are irradiated under gamma radiation at different doses (100, 150, 200, 300 and 500 krad) at a dose rate of 300 krad/h and the tensile properties of the films are studied. The TS values of the 2-EHA-treated gelatin films are plotted in Figure 4, where the values are tabulated against total radiation dose as a function of formulation F-3. From the figure, it is clear that the TS value increases up to 150 krad dose and then decreases with increase in radiation doses. The highest TS is found to be 60 MPa for 5% (F-3) 2-EHA-treated gelatin films at 150 krad dose, which is about 100% enhancement compared with pure gelatin films. At 100, 200, 300 and 500 krad radiation doses, the TS values are about 48, 55, 51 and 46 MPa, respectively. When high-energy gamma radiation is applied to the gelatin films using EHA, different types of reactions occur. Protein molecules can easily fragment due to gamma radiation. The reaction may proceed by free radical formation. Free radicals can be produced due to chain scission of gelatin molecule with the exposure of gamma radiation. These free radicals may cross-link with each other and form more cross-linked sites with the exposure of gamma radiation. As a result, mechanical properties might be increased. The proposed reaction mechanism between gelatin and EHA is shown in Figure 5. It is reported in the literature 28 ,32 that cellulose, shellac, chitosan and jute fiber molecules also form free radicals in a similar nature when irradiated by gamma source and may cross-link with monomer. The graft copolymerization of acrylonitrile and methacrylonitrile onto gelatin by gamma radiation has already been discussed. 33 Vinyl monomer treatment increases the TS value due to the cross-linking of amino group in gelatin with vinyl group of 2-EHA. The films became brittle and twisted at higher 2-EHA (10%) due to the homopolymer formation, which decreases the TS. Radiation dose is also very important because at higher radiation dose, TS values decrease because of the radiation degradation of the films. 34
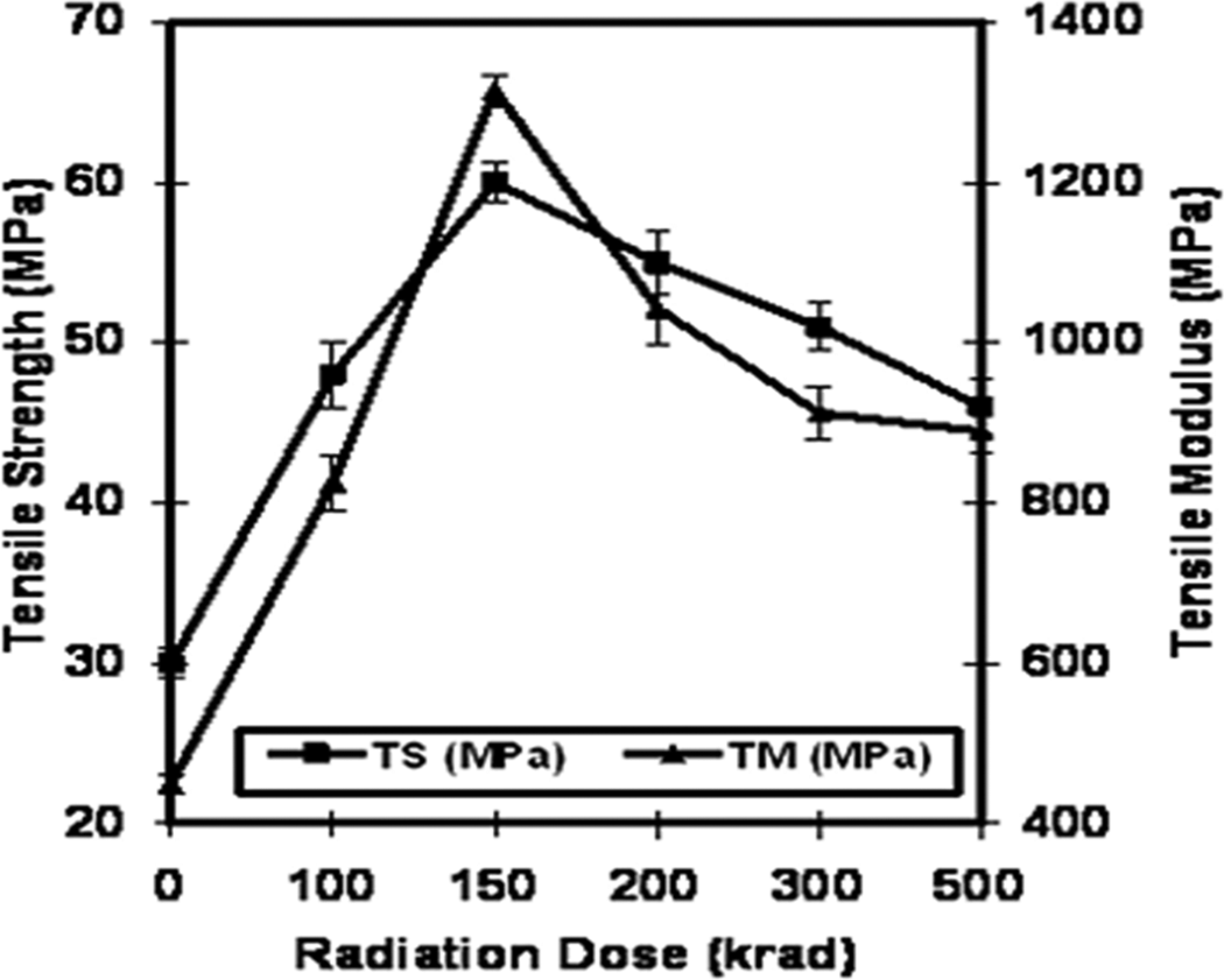
Tensile strength and tensile modulus of gelatin/2-ethylhexyl acrylate (2-EHA) films against radiation dose as a function of F-3 formulation.
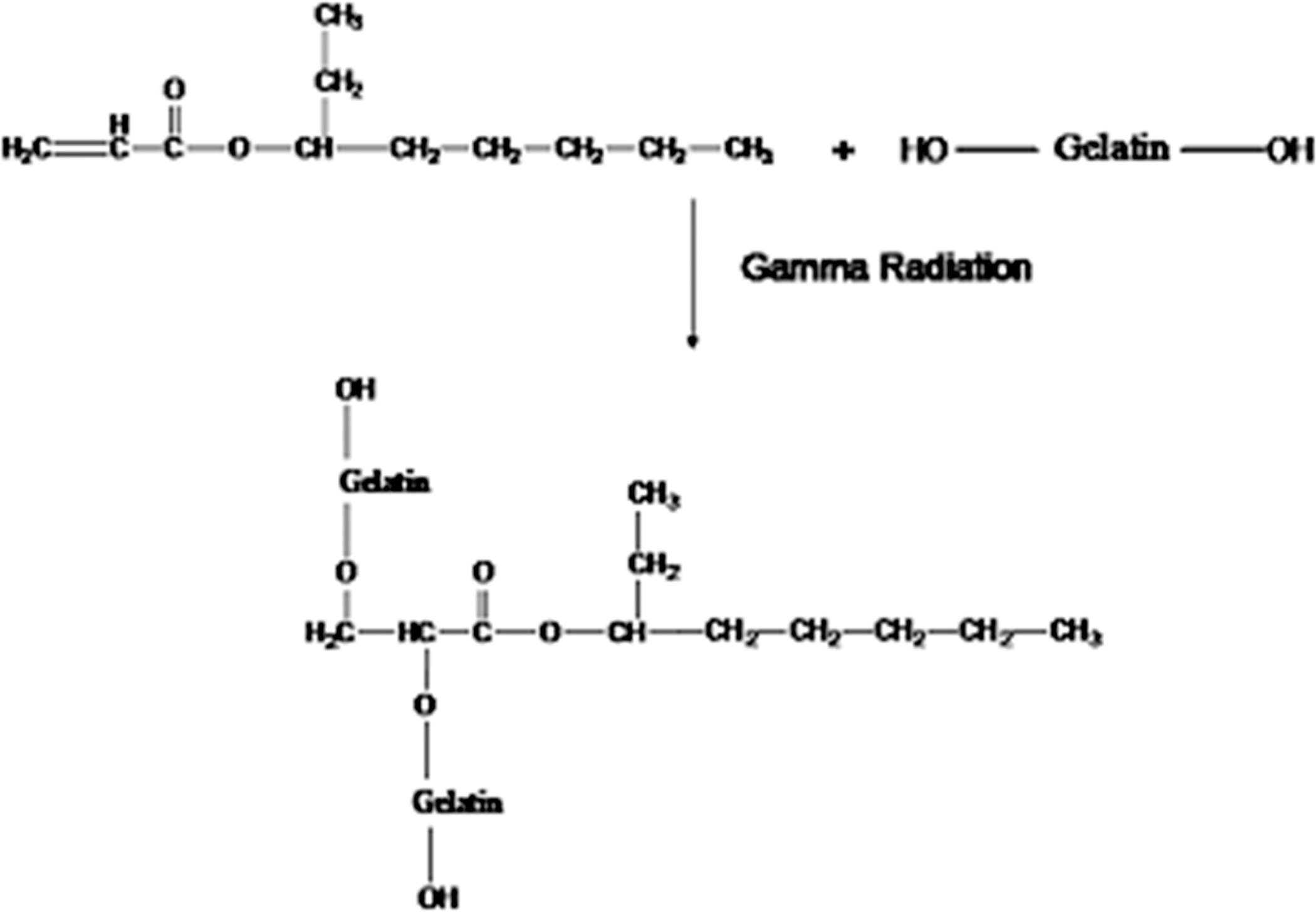
Probable reaction mechanism between 2-ethylhexyl acrylate (2-EHA) and gelatin with the exposure of gamma radiation.
Tensile moduli (TM) of the 2-EHA-treated films are cited in Figure 4 against total radiation doses as a function of formulation F-3. From the figure, it is clear that TM increases with increase in radiation doses up to 150 krad and then decreases with increasing radiation dose. The highest TM is found to be 1314 MPa for 5% (F-3) 2-EHA-treated gelatin films at 150 krad dose, which is about 192% enhancement when compared with pure gelatin films. Further increase in radiation dose decreases the TM due to the radiation degradation of the films. The reason behind increase in TM is the cross-linking of the amino group in gelatin with the vinyl group of 2-EHA.
Figure 6 represents the Eb values of the 2-EHA-treated films against total radiation dose as a function of formulation F-3. It is observed that the Eb values decrease up to 150 krad radiation dose and then increase with increase in radiation dose. This trend is similar for Figure 2, but the values are still lower than that of the nonirradiated films. Formulation F-3 (5% EHA) shows the lowest Eb value of 5.3% at 150 krad radiation dose of gelatin/2-EHA film, which is about 25% lower than the pure gelatin film. A suitable elongation is an important mechanical property in the application of polymer. The TS and TM value increases with increasing gamma radiation but Eb value decreases with increasing 2-EHA. This result indicates that at particular radiation dose, maximum TS, TM and Eb are achieved and further radiation degrades the films. From the above discussion, it is clear that F-3 formulation containing 5% 2-EHA gives the best result.
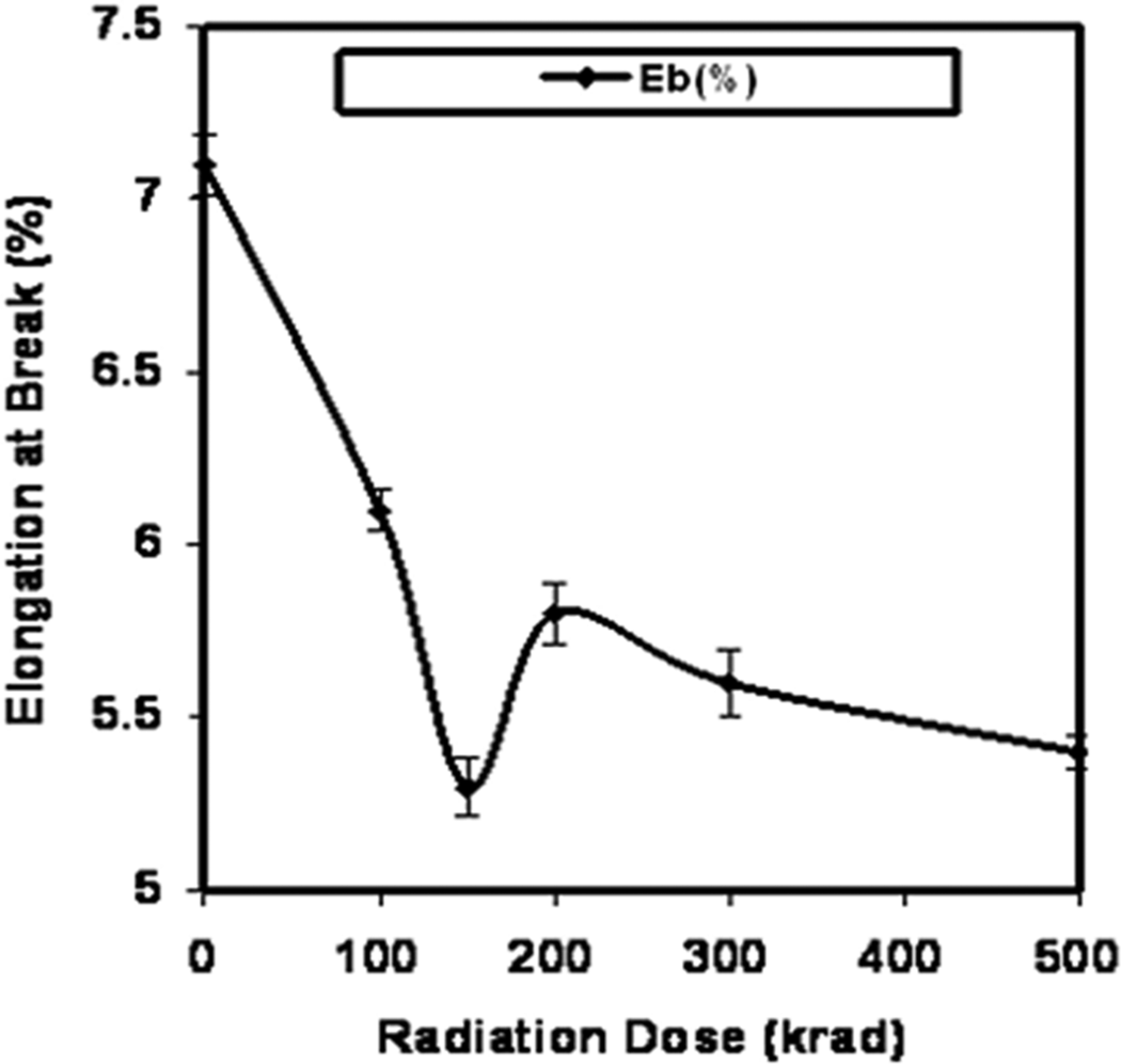
Elongation at break of gelatin/2-ethylhexyl acrylate (2-EHA) films against radiation dose as a function of F-3 formulation.
Thermal analysis of the gelatin films
The thermal property of onset of melting point of the pure gelatin film and 5% EHA-blended gelatin films irradiated with gamma radiation at different doses were evaluated using thermomechanical analyzer. The results are compared in Figure 7, where the onset of melting point of the gelatin films increased due to the application of gamma radiation. This may be due to the formation of strong intramolecular bond in gelatin because of the exposure to high-radiation gamma dose. The thermal properties such as onset of melting point and glass point and offset of melting point of the treated and untreated gelatin films are measured, and the results are compared in the bar graph (Figure 8). It is seen from the figure that when the pure gelatin films were irradiated at 100 krad after treating with 5% EHA blending, the thermal properties increase. The onset of melting temperature of pure gelatin film was found to be 55.4°C. For modified gelatin films, the onset of melting, the glass point of melting and the offset of melting temperatures shifted to 74.8, 78.6 and 83.5°C, respectively, which is 35, 41 and 51% improvement in the onset of melting, glass point of melting and offset of melting temperatures. From this investigation, it is indicated that when monomer-treated gelatin films irradiated, two types of polymerization reactions occurred, first is a heterogeneous reaction (monomer + gelatin) and the other is heterogeneous reaction (monomer + monomer). Although all monomers have very low melting temperatures, 35 the high-energy gamma radiation enables cross-linking between EHA and gelatin using their double bonds.
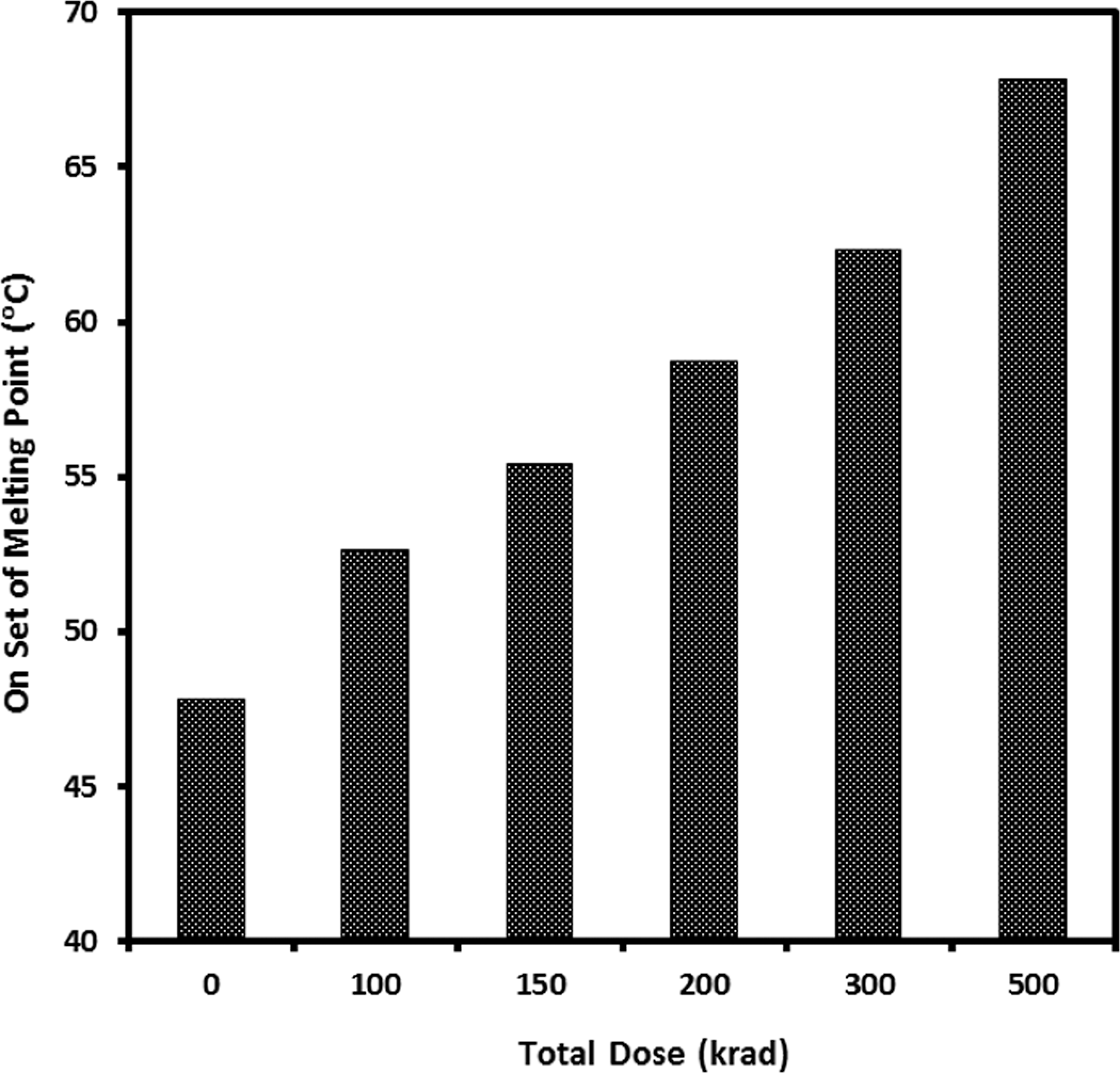
Onset of melting point (°C) of pure gelatin films and gamma irradiated gelatin films at different doses.
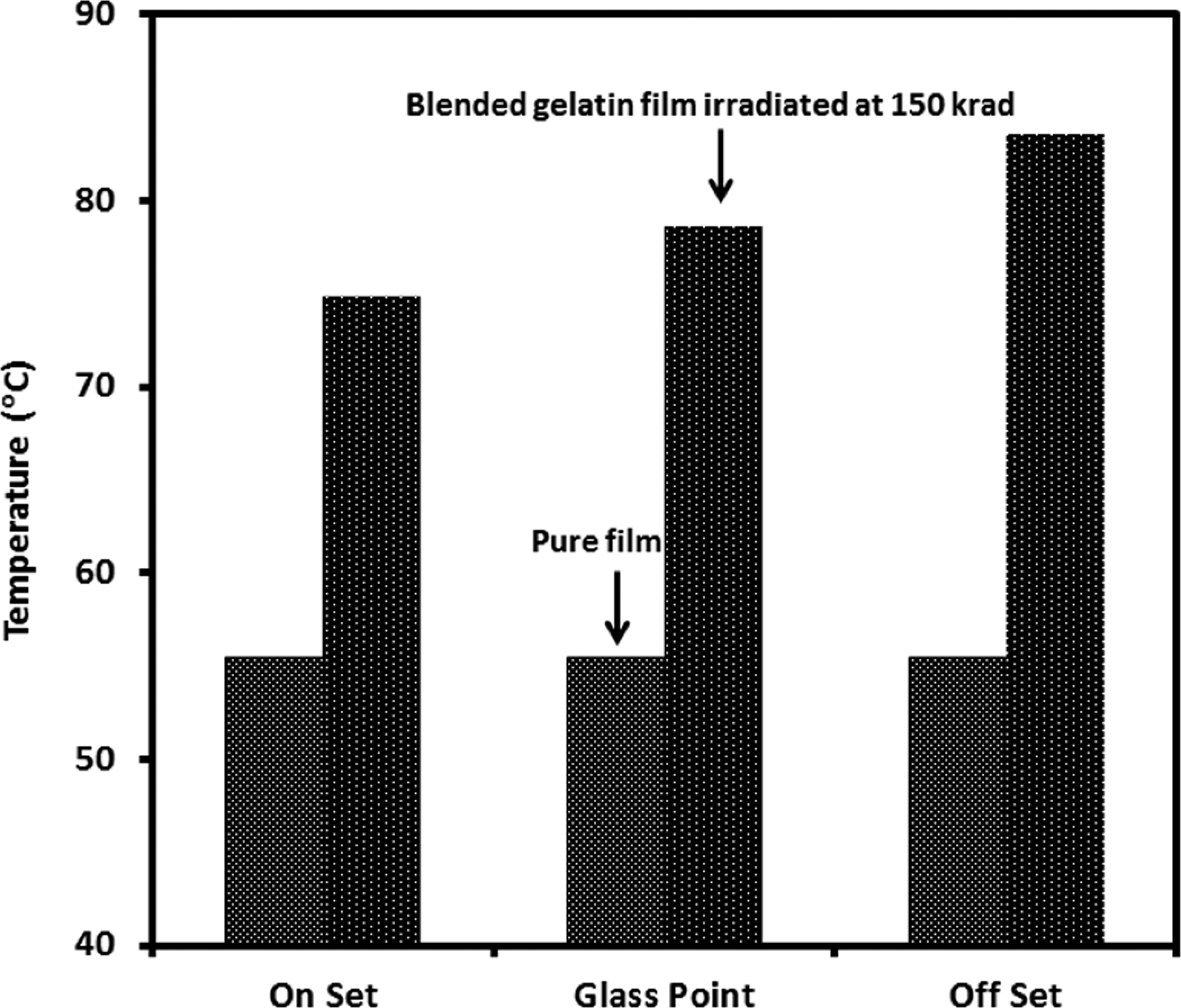
Thermal properties of the pure and blended gelatin films irradiated at 150 krad.
Water uptake of pure and modified gelatin films
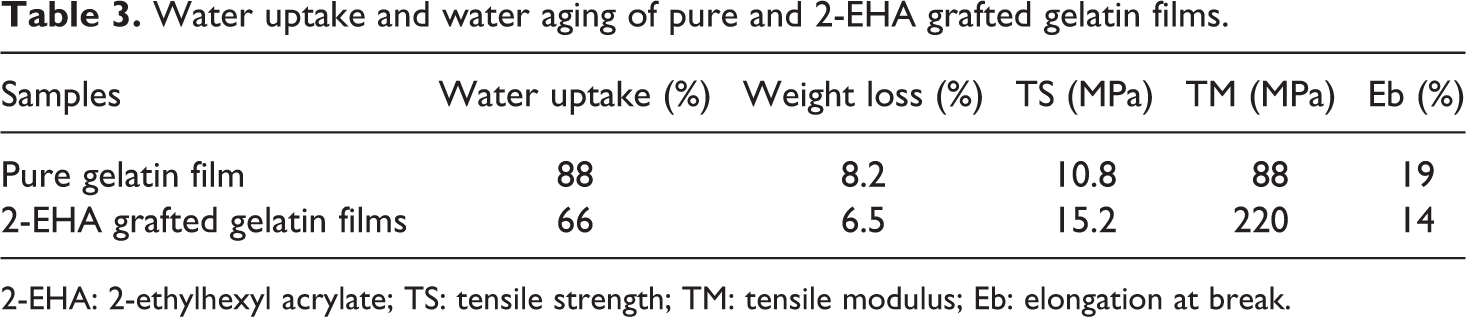
Water uptake of the pure and modified gelatin films were studied for 75 s and the results are shown in Table 3. It was observed that water uptake of pure gelatin film was 88% after 75 s. But the water uptake of modified gelatin film was 66% after 75 s, which is almost 25% lesser. It is revealed that for modified gelatin films, water uptake reduced significantly which supports higher TS value found and explained above.
Water uptake and water aging of pure and 2-EHA grafted gelatin films.
2-EHA: 2-ethylhexyl acrylate; TS: tensile strength; TM: tensile modulus; Eb: elongation at break.
The percentages of weight loss, the changes in TS, TM and Eb of the pure and modified gelatin films due to water aging are shown in Table 3. It is observed that the weight loss and the decrease in TS and TM of pure gelatin films were higher than the modified gelatin films. Pure gelatin film lost 8.2% of its initial weight, whereas modified gelatin film lost 6.5% of its initial weight after 5 min of water aging. The TS and TM of pure gelatin film was found to be 10.8 and 88 MPa, respectively, whereas that of modified gelatin film was found to be 15.2 and 220 MPa, respectively, after 5 min of water aging. It is found that increase in Eb due to water aging was also higher for pure gelatin films than modified gelatin films. Eb of pure gelatin film was found to be 19% and that of modified gelatin film was found to be 14% after 5 min of water aging. The cross-linking between various reactive groups of gelatin and 2-EHA might have made 2-EHA-grafted gelatin films more stable in water than pure gelatin films.
Conclusions
Thin films of gelatin were prepared by casting gelatin solution. It was found that TS, TM and Eb of the gelatin films were 30 MPa, 450 MPa and 7.1%, respectively. Gelatin films were irradiated under gamma radiation at different doses, and it was observed that the tensile properties increased significantly. A series of gelatin solutions was blended with varying percentages (1–10% by weight) of EHA; films were prepared and then irradiated with gamma radiation. TS and TM of the gelatin films were found to be 60 and 1314 MPa, respectively. The present study also figured out that the thermomechanical properties, the onset and offset of melting point and the glass point were increased for the treated films than the untreated films. It can be concluded that gelatin reacted with EHA when exposed to gamma radiation. The reaction between EHA and gelatin is attributed to the improvement in the mechanical properties of the EHA used gelatin films compared with pure gelatin films.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
