Abstract
The present study is related to the investigation of flammability properties of poly(methyl methacrylate) (PMMA) after incorporation of clay. PMMA/layered silicate (nanoclay) nanocomposites with 5% loading of nanofiller were prepared using melt intercalation technique. Commercially modified nanoclays such as Cloisite 30B, Cloisite 93A and in-house modified nanoclay (Quaternary ammonium salt modified Na-MMT i.e. OTAB-Na-MMT have been used as organoclays for the preparation of nanocomposites. Thermal characterization of the above prepared nanocomposites was carried out by thermal conductivity, horizontal burning test (UL94HB) and cone calorimeter experiments. The morphological behavior of nanocomposites was characterized by wide angle x-ray diffraction technique. The morphology of charred material obtained after burning was investigated using scanning electron microscopic analysis.
Introduction
There is a high level of interest in using nanoscale reinforcing fillers for making polymeric nanocomposite materials with exceptional properties. 1 –3 Nanocomposites are particle-filled polymers, where at least one dimension of the dispersed particle is on the nanometer scale; these particles are present in the form of sheets/layers, such as layered silicate (nanoclay) or graphite, which are one to a few nanometers thick and hundreds to thousands of nanometers in the other two dimensions. At present, the use of nanoclays is the most common approach for the improvement of performance in flame.
The previous studies 4 confirm that the mechanical properties of PMMA/clay nanocomposites can be considerably enhanced with the addition of 5% clay in PMMA. Therefore, this study comprises the study of PMMA nanocomposites with the loading of 5% clay.
The literature study reveals that thermal degradation initiates due to the heating of sample to a particular temperature. This is the stage at which the burning process of polymeric material begins. Most of the thermal degradation products of polymers possess low boiling temperature when compared with thermal degradation temperature. Therefore, the degradation products get superheated. 5 Subsequently, the bubbles are formed below the heated polymer surface, and the degradation products from the surrounding helps in growth of bubbles. 6 These bubbles evolve into gas phase that act as fuel vapor. The bubbles agitate the outer layer of polymer melt and subsequently interfere with the formation of solid char that acts as a heat-transfer barrier. 5
From several studies, it has been found that the nanoclay particles inhibit vigorous bubbling during combustion, which results in the reduction of flammability. This is because nanocomposites based on nanoclay particles form a continuous protective solid layer on the burning surface or extended island structures made of clay and carbonaceous char during burning. 7 –10 Therefore, it is assumed that in order to attain reduced flammability, formation of the protective layer is important. Clay-particle layers tend to develop large lateral surface cracks in which vigorous bubbling still occurs. Although these nanoparticles are promising flame retardants, further studies are necessary to improve the effectiveness of nanoclay filler and to analyze the responsible factors for this flame-retardant effect.
Several works confirm that in flammability analysis, the cone calorimeter is considered as the most significant instrument. This apparatus has been adopted by the International Organization for Standardization (ISO 5660-1) for measuring heat release rate (HRR) of a sample. HRR is based on the fact that the oxygen consumed during combustion is proportional to the heat released. This device analyses the combustion gases and measures the produced smoke from a test specimen that is being exposed to a certain heat flux. The oxygen concentration alone must be analyzed to calculate the released heat, but to improve the accuracy, carbon monoxide (CO) and carbon dioxide (CO2) concentrations can also be analyzed.
The HRR, especially the peak amount, is the primary characteristic determining the size, growth and suppression requirement of a fire environment. The rate of heat release is determined by the measurement of oxygen consumption as determined by the oxygen concentration and the flow rate in the exhaust product stream. 11 Peak HRR (PHRR) is considered as the parameter that best expresses the maximum intensity of fire, 12 indicating the rate and extent of fire spread. The average HRR is the average value of HRR from the period of ignition to 180 s. 12
Various parameters obtained from the analysis are mentioned in the present study and several investigations report the definition of these parameters. Time to ignition (TTI) (s) is the period required for the entire surface to burn with sustained luminous flame. The HRR is important for characterizing the real-fire response properties of polymeric material. The gas stream containing the combined combustion product is captured through an exhaust duct system, consisting of a high-temperature centrifugal fan, a hood and an orifice plate flow meter. The typical air flow rate is 0.024 m
3
/s. Oxygen concentration in the exhaust stream is measured with an oxygen analyzer with an accuracy of 50 ppm, and the HRR is determined by comparing oxygen concentration with the value obtained when no sample is burning.
11
Parameters related to the smoke evolution, such as total smoke release calculated by integrating the rate of smoke released curve, average of CO emission as well as CO or CO2 weight ratio and smoke density measured by the decrease in transmitted light intensity of a helium–neon laser beam photometer were also obtained and expressed in terms of average smoke extinction area.
13
Smoke parameter ([SP] MW/kg) is defined as the product of average SEA and PHRR. This parameter is indicative of the amount of smoke generated in a fire situation.
14
SP is given as follows
CO and smoke (combination of soot and combustion gases) are primary cause of death in most fires. CO was recognized as one of the most toxic gaseous products in fires and that its concentration was ultimately dependent more on the combustion environment.
Specific extinction area is a property related to the smoke production. The properties of smoke that are of primary interest to the fire community are light extinction, visibility and detection. The most widely measured property of smoke is the light extinction coefficient. The physical basis for light extinction measurements is Bouguer’s law, which relates the intensity,
where K is the light extinction coefficient. When Eq. (2) is expressed in terms of base 10
The quantity D is defined as the optical density per meter, where D = K/2.3. The extinction coefficient, K, is an extensive property and can be expressed as the product of extinction coefficient per unit mass, Km
, and mass concentration of the smoke aerosol, m.
The specific extinction coefficient, Km , depends on the size distribution and optical properties of the smoke. Specific extinction coefficient is also termed as specific extinction area.
Fire performance index ([FPI] m2s/kW) defined as the ratio of TTI to PHRR, which is a parameter related to the time to flashover (or the time available for escape) in a full-scale fire situation.
16
Experimental
Materials
PMMA (LG PMMA IH830), procured from M/s LG chemicals (India), having a melt-flow index value of 3 g/10 min, specific gravity of 1.18 and tensile strength of 750 kg/cm2, has been used as the base matrix. The clay minerals used were commercially available Cloisite 30B ([C30B] Cation exchange capacity (CEC) of 90 meq/100 g clay, d001 = 1.85 nm) and Cloisite 93A ([C93A] 90 meq/100 g clay, d001 = 2.36 nm) modified with organic modifier methyl, tallow, bis-2-hydroxyethyl, quaternary ammonium (MT2EtOH) and methyl, dehydrogenated tallow ammonium (M2HT), respectively. Unmodified sodium montmorillonite (Na-MMT) clay was also used in this work. All the clay materials were procured from Southern clay Products, Inc., USA. Octadecyl trimethyl ammonium bromide (OTAB) used as modifier for in-house modification of clay was procured from Himedia Laboratories Pvt. Ltd. (Gujarat, India).
Modification of clay (Na-MMT)
The sodium montmorillonite clay (Na-MMT) was modified by ion exchange process using OTAB modifier. Five parts (by weight, as also for all mixtures herein) of Na-MMT is dispersed in 95 parts of distilled water under vigorous stirring conditions to form a suspension. Three parts of intercalative reagent (OTAB) is then added to the suspension and stirred for 2 h at 80°C. The suspension is filtered and repeatedly washed with distilled water to remove the excess intercalative reagent, until there is no light white precipitate observed in the wash water tested by a 0.1-mol/L AgNO3 solution. The product is then vacuum-dried to a constant weight and ground into powder to get the organically (OTAB) modified Na-MMT (OTAB-Na-MMT).
Nanocomposites and specimen preparation
The preparation of PMMA clay nanocomposites at various composition (with 3 wt% and 5 wt% of clay) was carried out using batch mixer HAAKE PolyLab–Rheomix at (Germany) 180°C at 50 rpm for a mixing time of 20 min. PMMA and nanoclays used for nanocomposites preparation were dried at 80°C in hot air oven for a period of 24 h and 12 h, respectively, prior to processing. The sheet of dimension 160 × 160 × 3 mm was prepared using a compression moulding machine at 200°C for a cycle time of 20 min. Specimens for different characterizations were cut from the sheet of 3 mm thickness.
Characterizations
FTIR spectroscopy
For the confirmation of successful modification of Na-MMT, the Fourier transform infrared (FTIR) spectroscopy was carried out using Thermo scientific FTIR (smart orbit ATR 400–4000 cm−1 with microscope, USA).
Thermal conductivity
Thermal conductivity characterization was carried out according to the American Society for Testing and Materials (ASTM) E1530-06 standard test method using Unitherm™ Model 2022 Anter corporation, based on the guarded heat flow meter technique.
WAXD technique
Wide angle x-ray diffraction (WAXD) analysis was used to analyze the interlayer gallery spacing of nanoclays in the nanocomposites, using Philips X’Pert MPD (Japan), with graphite monochromator and a Cu Kα radiation source operated at 40 kv and 30 mA. The x-ray radiation source having a wavelength of 1.54 Å (copper Kα1 and Ni filter), aperture slit having a width of 0.1 mm and the scanning rate of 0.01° per second over the range of 0.5° < 2θ < 10° was used.
Rate of burning
To evaluate the fire-spread rate, the rate of burning was determined using ATLAS HVUL2-horizontal burning test chamber according to the standard UL94HB with a sample size of 125 × 13 × 3 mm. It measures the fire travel on the horizontal surface including the fire-spread rate, burning behavior, ease of extinction and whether the material burns without dripping.
Cone calorimeter
The cone calorimeter experiments were carried out at 50 kW/m2 heat fluxes with horizontal orientation of the samples (plates with 100 × 100 × 3 mm3), according to ASTM E 1354 using Fire testing technology (FTT) cone calorimeter and dual cone fire testing technology (UK).
Results and discussion
FTIR spectroscopy
The FTIR analysis, represented by Figure 1, reveals the presence of absorbance band in the region 2650–3000 cm−1, namely, at 2847.7 and 2916.6 cm−1. This band occurs due to C–H stretching vibrations of CH3 and methylene (CH2) groups, which confirmed the incorporation of long chain alkyl group into Na-MMT clay. This indicated the successful modification of Na-MMT by the surfactant OTAB.
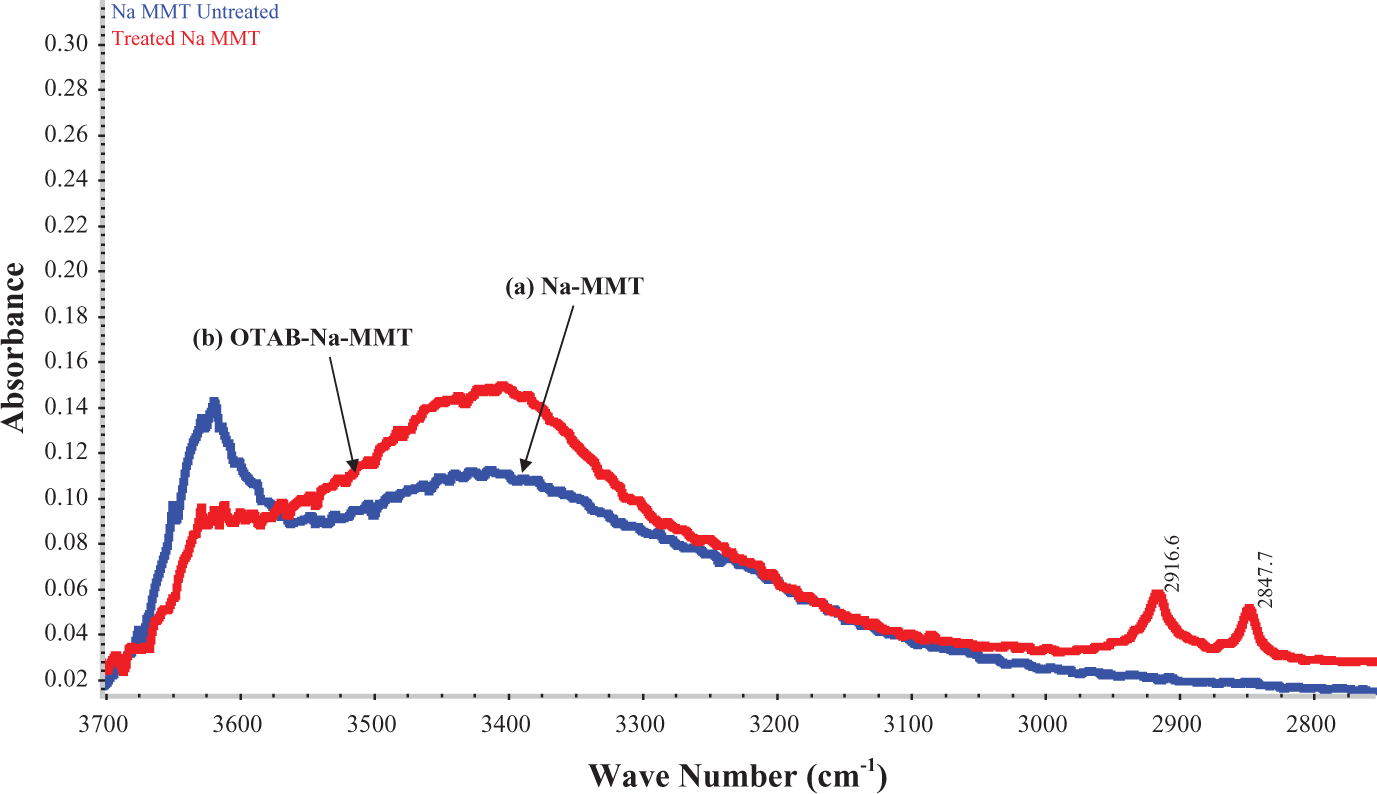
Fourier transform infrared spectra of (a) unmodified Na-MMT clay (Na-MMT) and (b) OTAB-modified Na-MMT clay (OTAB-Na-MMT). Na-MMT: sodium montmorillonite, OTAB: octadecyl trimethyl ammonium bromide.
WAXD technique
The x-ray diffraction pattern of the nanoclays and nanocomposite systems is represented in Figure 2. From the figure, it is revealed that C30B and C93A nanoclays exhibited a single diffraction peak at 2θ = 4.85 and 3.91, corresponding to interlayer d-spacing of 1.85 and 2.56 nm, respectively. This shows typical characteristics of intercalated structure of C30B and C93A in the presence of intercalants MT2EtOH and M2HT, respectively.
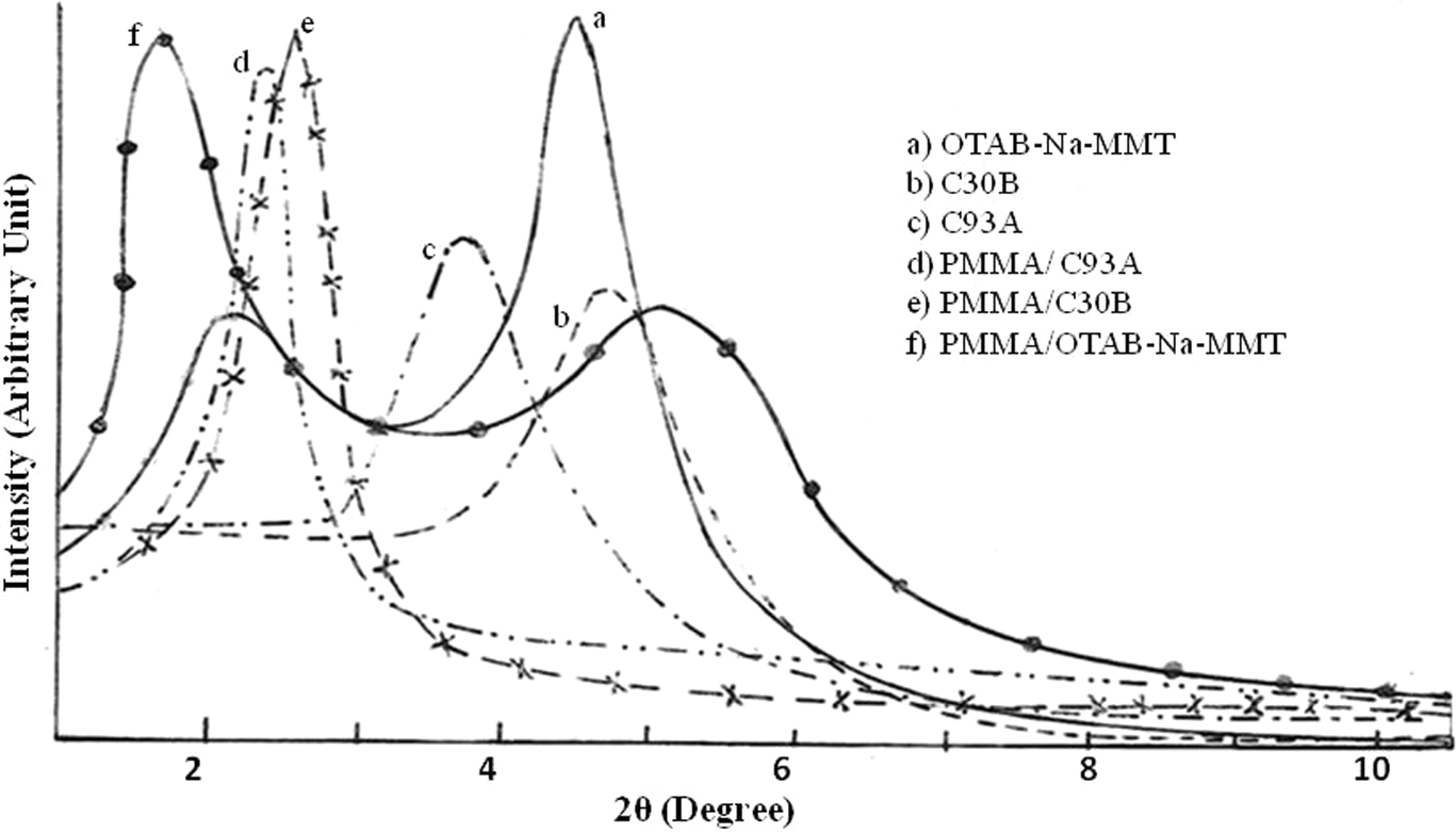
Wide angle x-ray diffraction technique patterns of (a) OTAB-Na-MMT, (b) C30B, (c) C93A, (d) PMMA/C93A, (e) PMMA/C30B, and (f) PMMA/OTAB-Na-MMT. Na-MMT: sodium montmorillonite, OTAB: octadecyl trimethyl ammonium bromide, C30B: Cloisite 30B.
In Na-MMT, the diffraction peak is around 8°, whereas in OTAB-Na-MMT nanoclay, the major diffraction peak shifted to 2θ = 2.3° and a minor peak to 2θ = 4.43, corresponding to the d-spacing of 3.83 and 1.99 nm, respectively. Therefore, it is estimated that octadecyl trimethyl ammonium has entered and expanded the d-spacing of Na-MMT through ion exchange phenomenon. Figure 2d–f represents the x-ray diffractogram of PMMA naocomposites. PMMA/C30B and PMMA/C93A nanocomposites indicated a peak at around 2θ = 2.5 and 2.47, with a d-spacing of 4.16 and 3.58 nm, respectively, which reveals intercalated nanomorphology.
On the contrary, PMMA/OTAB-Na-MMT nanocomposites indicated a peak at 2θ = 1.8°, with a d-spacing of 4.77 nm and a hump around 5.3° at 1.32 nm. This result reveals that although PMMA has been effectively, intercalatedly melt into the lamellar space of OTAB-Na-MMT nanoclay, agglomerates of clay layers are still present within the nanocomposite matrix.
The maximum change of 33.45% in d-spacing was observed between PMMA/Na-MMT nanocomposites and PMMA/OTAB-Na-MMT nanocomposites. This behavior is attributed to the improved interaction between PMMA and clay due to the modification of clay with quaternary ammonium salt. Therefore, all the nanocomposite systems indicated intercalated morphology and quantitative exfoliation was not present in these systems.
Thermal conductivity
Thermal conductivity of virgin PMMA (VPMMA) and PMMA/clay nanocomposites at variable clay loading is represented in Table 1. It is observed that the thermal conductivity value of VPMMA and PMMA/clay nanocomposites system increases with increase in the temperature. Also, a higher thermal conductivity value of PMMA/clay nanocomposites was noticed when compared with that of the VPMMA.
Thermal conductivity of virgin PMMA and PMMA/clay nanocomposites.
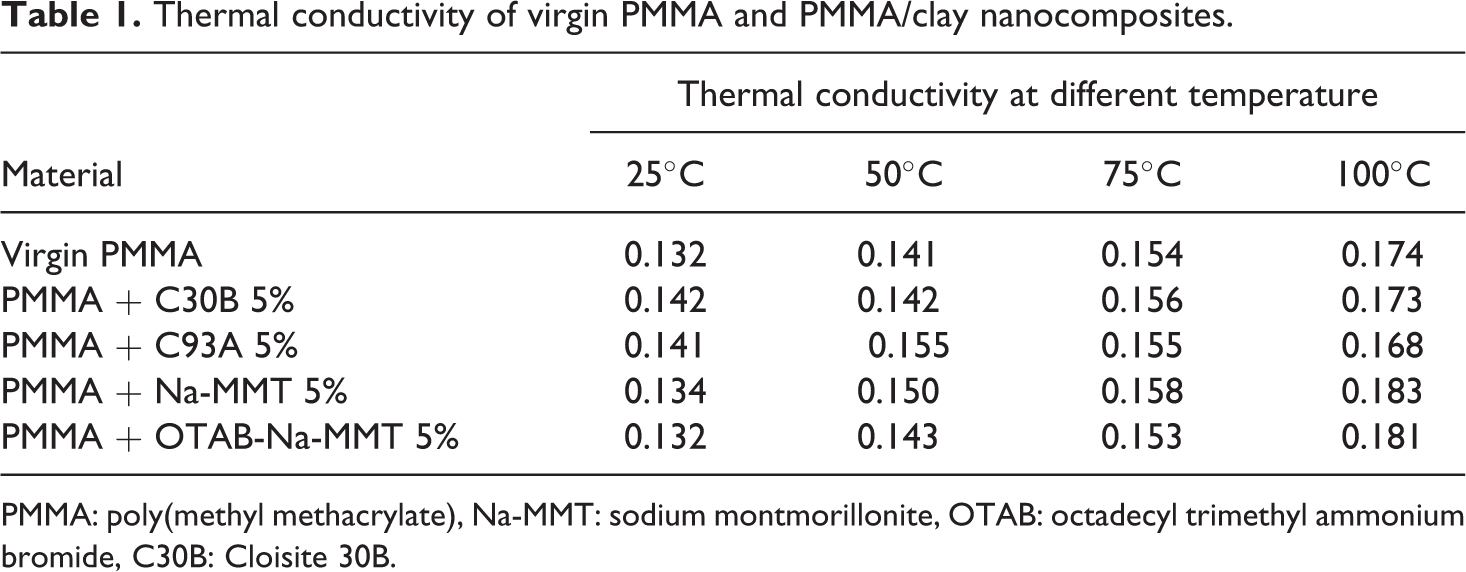
PMMA: poly(methyl methacrylate), Na-MMT: sodium montmorillonite, OTAB: octadecyl trimethyl ammonium bromide, C30B: Cloisite 30B.
This might be due to intercalated morphology of nanocomposites. According to the classic Maxwell thermal conduction model, 17 the presence of clay will result in an increase in the conductivity of the nanocomposites, and this increase in the conductivity value may also be due to the presence of large clay stacks and agglomerates. In this study, similar results were obtained. The PMMA/C30B and PMMA/C93A indicated higher conductivity value when compared with VPMMA.
On the contrary, the analysis indicated that the incorporation of in-house modified clay (OTAB-Na-MMT) does not cause any appreciable increase in the conductivity of PMMA. This may be due to the effective dispersion of the clay layers that restrict the local vibrations of atoms and molecules, which in turn dominate the heat transport in the amorphous polymer. 18
Similar results were shown by Zou et al. Their study reveals that there are two factors that cause decrease in the conductivity value of exfoliated nanocomposites, namely, increase in the surface of clay due to exfoliation and size effect, which results in larger thermal resistance and decrease of thermal conductivity value, respectively. In the present investigation, the increase in thermal conductivity revealed the occurrence of intercalated morphology in the nanocomposites.
Rate of burning
The burning rate data obtained from horizontal burning test (UL94HB) for VPMMA and PMMA/clay nanocomposites are represented in Table 2 and Figure 3. The results revealed that nanocomposite systems exhibit substantially higher burning rates and shorter average times of burning when compared with VPMMA, which indicates their fast burning; moreover, all the nanocomposites burn without dripping; this behavior is very different from VPMMA, which drips significantly during the test. This might be due to “wick effect,” according to which organic/inorganic composite burns out the organic phase and leaves the inorganic phase intact, which leads to a faster burning rate of the composite. 19 Although the burning rate of nanocomposites materials was found higher than VPMMA still the PMMA/clay nanocomposites are more fire safe due to the absence of dripping during burning. Fire safety is the ability of occupant to escape from fire. Dripping contribute to the rapid fire spread; therefore, due to the absence of dripping, the nanocomposites do not contribute to the rapid fire spread.
Burning rate data for virgin PMMA and PMMA/clay nanocomposites.

PMMA: poly(methyl methacrylate), Na-MMT: sodium montmorillonite, OTAB: octadecyl trimethyl ammonium bromide, C30B: Cloisite 30B.

Test specimens after the horizontal burning test (UL94HB). (a) Burned specimen of PMMA and (b) burned specimen of PMMA/nanocomposite. PMMA: poly(methyl methacrylate).
Therefore, it is revealed that the fire spread properties of nanocomposites are less than VPMMA. Thus, it can be concluded that nanocomposites are more fire safe than VPMMA.
Cone calorimeter
The results obtained by the cone calorimeter experiment are represented in Table 3. No significant change in the TTI is noticed for VPMMA and PMMA/clay nanocomposites, which was in accordance with the results reported by Jash and Wilkie, 20 revealing that the time required to burn the entire surface with luminous flame for VPMMA and all the nanocomposite systems does not show considerable difference.
Cone calorimeter data of VPMMA and PMMA/clay nanocomposites.
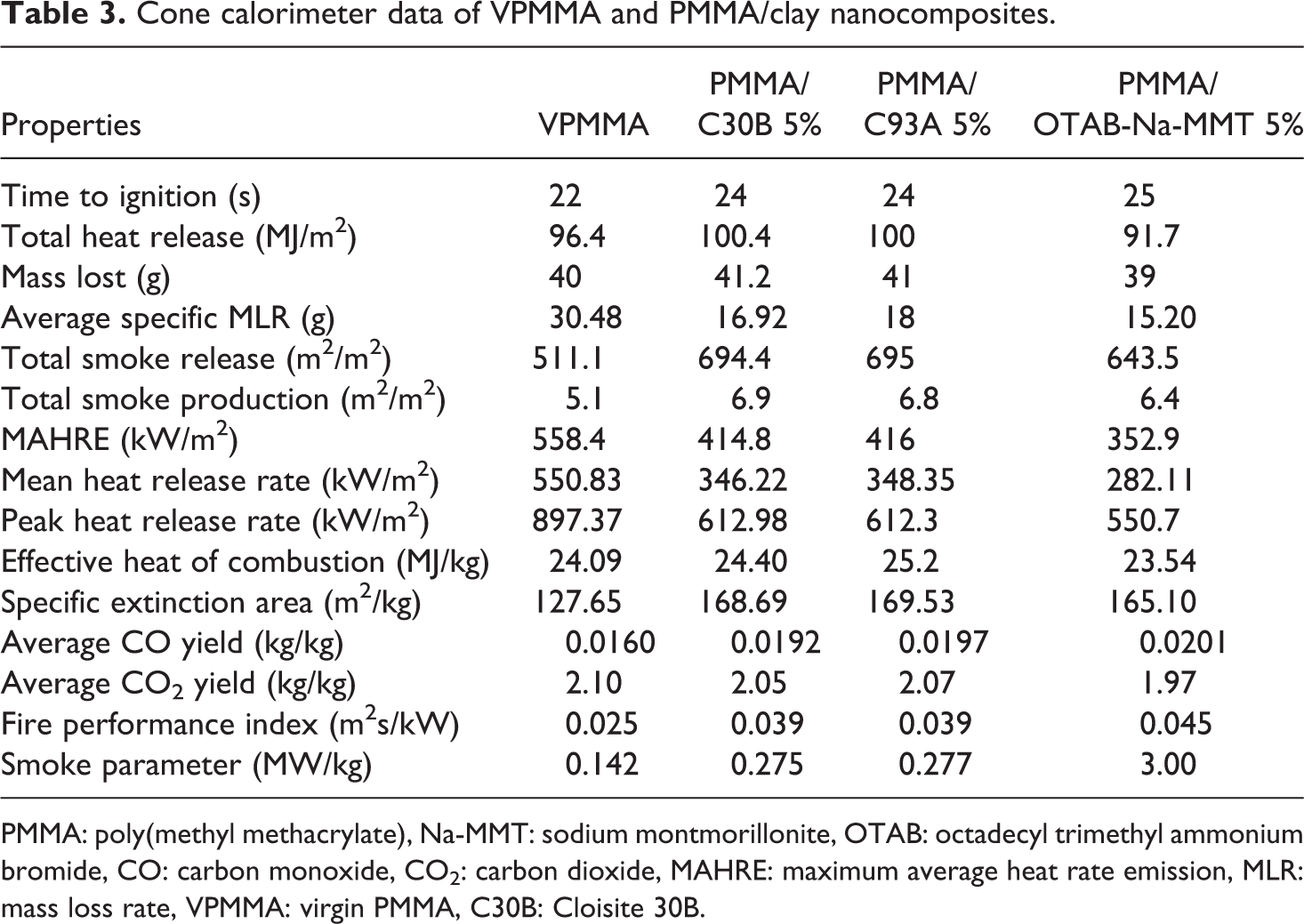
PMMA: poly(methyl methacrylate), Na-MMT: sodium montmorillonite, OTAB: octadecyl trimethyl ammonium bromide, CO: carbon monoxide, CO2: carbon dioxide, MAHRE: maximum average heat rate emission, MLR: mass loss rate, VPMMA: virgin PMMA, C30B: Cloisite 30B.
Mass loss rate (MLR) is another important defining factor in determining the fire-retardant property. The data represented in the Table 3 revealed that the average specific MLR of all the nanocomposite systems was lower than VPMMA and among the nanocomposites, PMMA/OTAB-Na-MMT indicated lowest average specific MLR. This might be due to the formation of a protective layer of char in between flame and sample, thereby reducing the mass transfer in flame and sample. The weights of sample before and after burning were taken wherein the percentage of weight loss on the ignition of PMMA/C30B and PMMA/C93A was found to be 30% and 37%, respectively, which is comparatively less than PMMA/OTAB-Na-MMT. 16 This might be due to the presence of some moities, for example, in the form of free radicals (due to the presence of organic modifier OTAB) which get burned and cause comparatively more weight loss.
From Figure 4, it was noticed that the mass loss of nanocomposite systems shifted toward longer time period than VPMMA. This behavior indicated the slow weight loss of nanocomposites when compared with VPMMA. 21
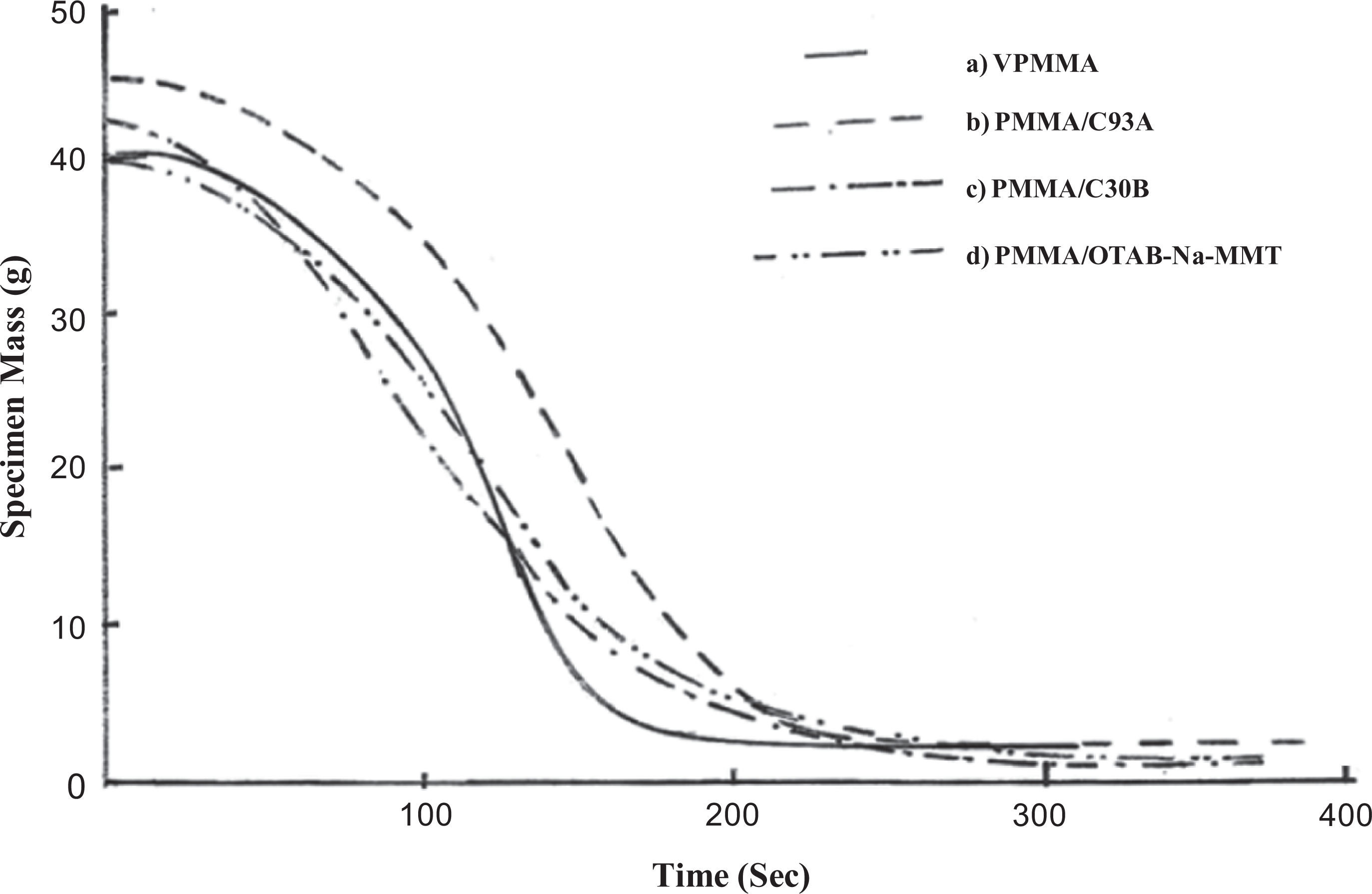
Mass loss rate of PMMA nanocomposite system. (a) PMMA, (b) PMMA/C93A, (c) PMMA/C30B, and (d) PMMA/OTAB-Na-MMT. PMMA: poly(methyl methacrylate), Na-MMT: sodium montmorillonite, OTAB: octadecyl trimethyl ammonium bromide, C30B: Cloisite 30B.
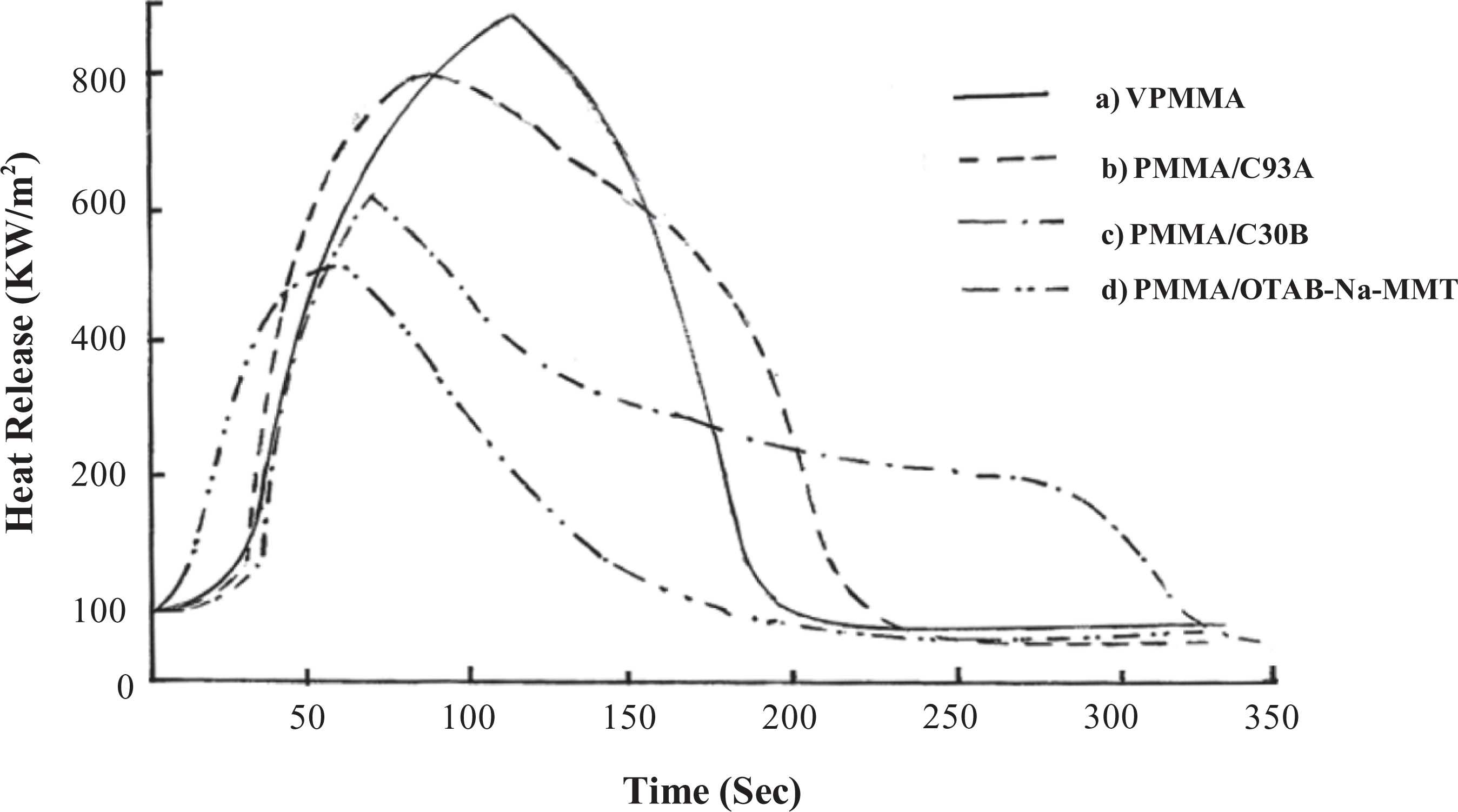
Figure 5 reveals the shift of PHRR in nanocomposites toward longer time when compared with VPMMA. The results indicated that the PHRR and the average HRR value of nanocomposites are lower than VPMMA. The order of average HRR and PHRR value among the nanocomposites is VPMMA > PMMA/C93A > PMMA/C30B > PMMA/OTAB-Na-MMT.
Mass loss rate of PMMA nanocomposite system (a) PMMA, (b) PMMA/C93A, (c) PMMA/C30B, (d) PMMA/OTAB-Na-MMT. PMMA: poly(methyl methacrylate), Na-MMT: sodium montmorillonite, OTAB: octadecyl trimethyl ammonium bromide, C30B: Cloisite 30B.
A decrease in PHRR indicated a reduction in burnable volatiles generated by the degradation of polymer matrix, such drop clearly indicates the flame-retardant effect due to the presence of the organoclays and their ‘molecular’ distribution throughout the matrix. Furthermore, the flame-retardant properties were improved by the fact that the PHRR was spread over a much longer period of time. The reduction in MLR and PHRR revealed the flame-retardant effect of nanoclay in the polymer matrix.
The flame-retardant properties are due to the formation of a char layer during the nanocomposite combustion. This char acts as an insulating and nonburning material and reduces the emission of volatile products (fuel) into the flame area. The silicate layer of the organoclay plays an active role in the formation and strengthening of the char and makes it more resistant to ablation. 22 Study indicated that maximum average heat rate emission (MAHRE) value decreased with clay loading and the value was lowest for PMMA/OTAB-Na-MMT.
Furthermore, investigation revealed the increased total smoke production value for nanocomposites than VPMMA, and the order of amount of smoke produced is VPMMA < PMMA/OTAB-Na-MMT < PMMA/C30B < PMMA/C93A. It was noticed that the specific extinction area calculated according to Eq. (4) related to the amount of smoke produced, followed the same trend as that of the amount of smoke produced.
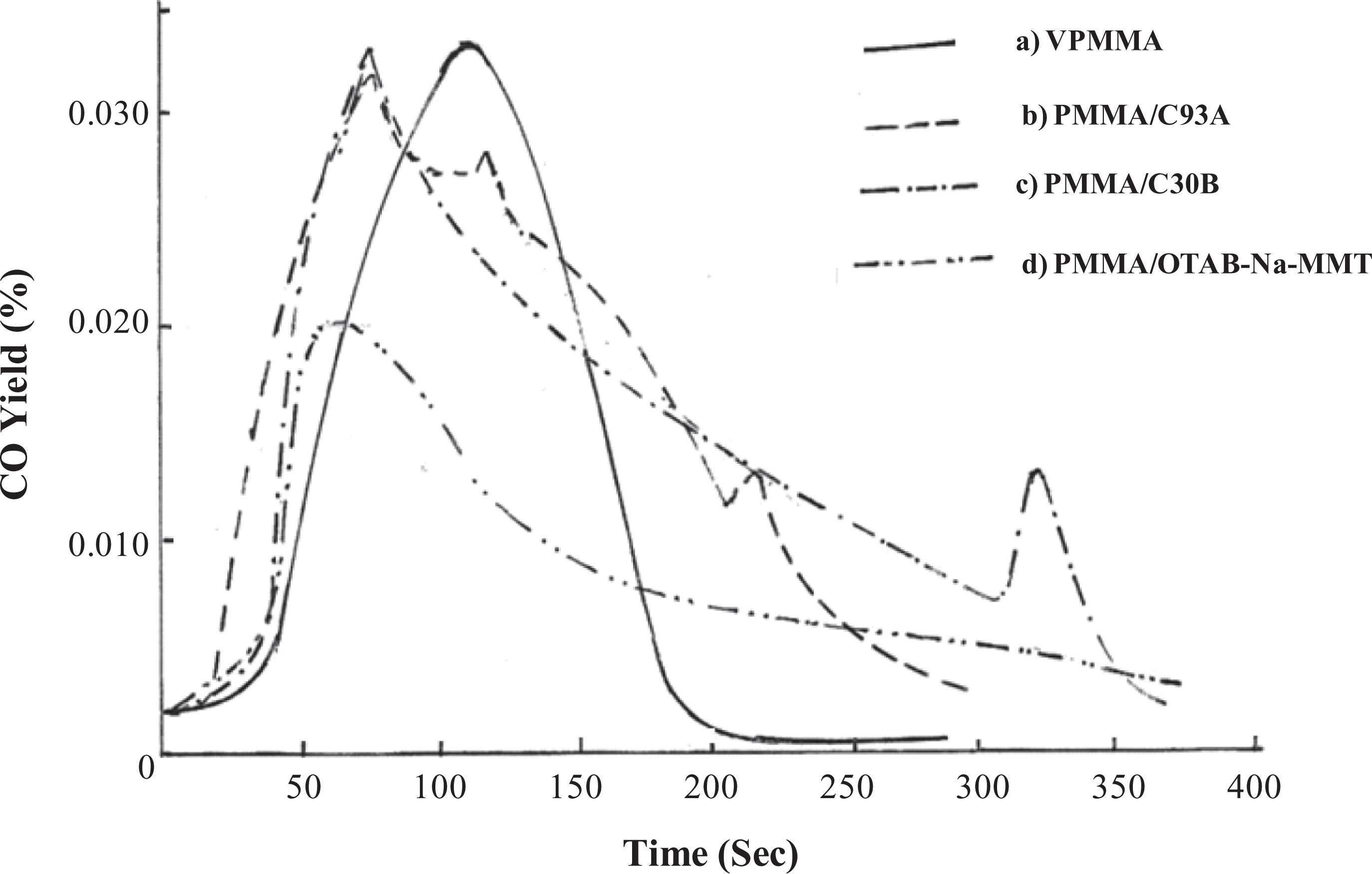
There is a very small difference in the average heat of combustion value of PMMA and its nanocomposites as shown in Table 3. From Figures 6 and 7, a shift in the yield of CO and CO2 nanocomposites toward longer period was revealed, and a very small difference in the average yield of CO and CO2 VPMMA and its nanocomposites was noticed.
Heat release rate of PMMA nanocomposites (a) PMMA, (b) PMMA/C93A, (c) PMMA/C30B, (d) PMMA/OTAB-Na-MMT. PMMA: poly(methyl methacrylate), Na-MMT: sodium montmorillonite, OTAB: Octadecyl trimethyl ammonium bromide, C30B: Cloisite 30B.
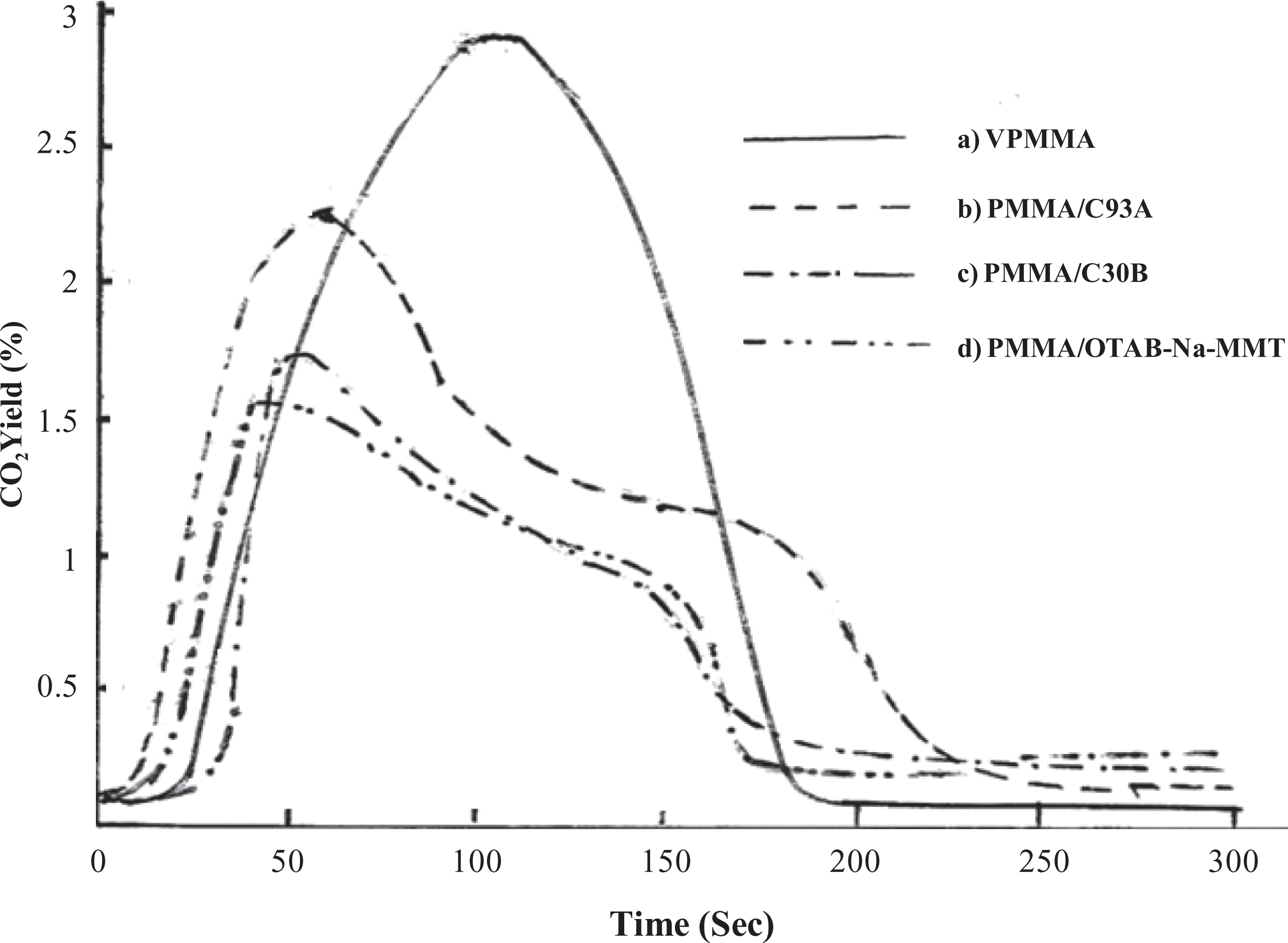
Carbon dioxide emission of PMMA nanocomposites (a) PMMA, (b) PMMA/C93A, (c) PMMA/C30B, (d) PMMA/OTAB-Na-MMT. PMMA: poly(methyl methacrylate), Na-MMT: sodium montmorillonite, OTAB: Octadecyl trimethyl ammonium bromide, C30B: Cloisite 30B.
Because of the char formation and very small change in the heat of combustion, yield of CO and yield of CO2. It is reasonable to conclude that the lower HRR of nanocomposites is due to changes in the condensed phase decomposition processes and not from gas phase effect. 23
The FPI was calculated using Eq. (5). The investigation revealed lower FPI value for VPMMA than the nanocomposites. Among the nanocomposites, the PMMA/C30B and PMMA/C93A showed almost equal FPI value, whereas the PMMA/OTAB-Na-MMT exhibited the highest FPI value when compared with VPMMA and other nanocomposites. That is the time available for escape in high-scale fire situation is highest for PMMA/OTAB-Na-MMT when compared with VPMMA and other nanocomposites.
SP was calculated according to Eq. (1), which indicates the amount of smoke generated in a fire situation. From the investigation, lower SP was noticed for VPMMA when compared with other nanocomposites. Among the nanocomposites, the SP is highest for PMMA/OTAB-Na-MMT nanocomposites. This data support the consideration of high smoke generation by the nanocomposites when compared with VPMMA. This behavior was shown because the char layer formed during the combustion prevents the complete oxidation of degradation products, resulting in the release of only partially oxidized carbonaceous residues. 16
The sample images after burning are represented in Figure 8, which indicated the presence of carbonaceous char on the top surface of the sample. As expected, pristine PMMA did not leave any significant amount of residue. At a low level of the nanoparticle loading in the polymer, the residues did not cover the sample container leaving a pattern consisting of many isolated clumps or islands. An increase in the level of clay led to the conclusion that the residues cover the entire sample container without any cracks or openings.

Charred residues after burning.
Although the smoke production from nanocomposite materials is little higher than VPMMA, the higher availability of time to escape from fire dominates the effect of increased amount of smoke; therefore, the investigations revealed that all the factors determining flammability support nanocomposite material as more fire safe when compared with VPMMA.
Scanning electron microscopy
Morphological properties of PMMA nanocomposite residues from cone calorimetry were studied using scanning electron microscopy (SEM). The SEM micrographs of PMMA/OTAB-Na-MMT, PMMA/C30B and PMMA/C93A are shown in Figures 9a–c, respectively. A continuous network structure was observed in the case of both PMMA/C93A (Figure 9b) and PMMA/OTAB-Na-MMT (Figure 9a) nanocomposites. This may be attributed to the formation of a protective layer on the surface, which shall block the thermal wave from penetrating down the surface and reduces further conduction of heat. Moreover, carbonaceous layer was found to cover the entire surface of the sample, thus reducing their MLR when compared with PMMA/C30B. PMMA/C30B nanocomposite (Figure 9c) exhibited clay platelets with voids among them. The bubbles evolving from the interior of the sample push away the platelets to form openings throughout the layer. These opening may merge at a later stage to form a continuous opening resembling cracks. The presence of these openings/voids may further lead to higher MLR. SEM images indicate that the clay particles migrated toward the surface during the burning of a polymer. They assisted with char formation.

Scanning electron microscopy images of nanocomposites.
Conclusion
The Na-MMT was modified successfully using alkyl ammonium salt. The occurrence of modification was confirmed using FTIR analysis. Thermal conductivity value for intercalated nanocomposite materials is higher than the thermal conductivity value of VPMMA. Burning rate for the PMMA/clay nanocomposites is greater than that of VPMMA but the dripping that occurred in the VPMMA stops by adding the clay into the PMMA. Flammability of nanocomposites was studied using cone calorimeter also. The rate of burning and cone calorimeter analysis reveals that the nanocomposites are more fire safe than VPMMA. The decrease in the PHRR results in the improvement in thermal stability. The variation in the specific extinction area leads to the variation in the amount of smoke evolved by the different material. On the basis of the factors defining flammability, it can be concluded that the loading of clay as nanofiller in the PMMA matrix can help in reducing its flammability. Among all the nanocomposites, the PMMA/OTAB-Na-MMT showed higher performance in flame.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.





