Abstract
Polypropylene (PP)/microcrystalline cellulose (MCC) composites and PP/MCC composites modified by maleic anhydride grafted PP (PP-g-MA) and methyl acrylic acid glycidyl ester grafted PP (PP-g-GMA) respectively were prepared in a twin-screw extruder. The mechanical properties, morphology, and thermal performance were investigated. The nonisothermal crystallization, melting behavior, and nonisothermal crystallization kinetics were investigated by DSC. The results indicated that the addition of MCC had led to the increase of the tensile strength, impact strength, and flexural strength of PP. PP-g-GMA modification was more conducive to the improvement in tensile strength, impact strength, and flexural strength. The three types of PP/MCC composites have higher thermal decomposition temperatures, Vicat softening temperatures, and dimensional stability. Nonisothermal crystallizations of PP/MCC composites were in accordance with tridimensional growth with heterogeneous nucleation. Meanwhile, MCC was acted as the nucleating agent in PP matrix, which increased the crystallization temperature. PP-g-GMA further increased the crystallization temperature while PP-g-MA weakened the heterogeneous nucleation effect of MCC. Avrami equation and Mo method give a satisfactory description of the crystallization kinetics process. The activation energy of crystallization, nucleation constant, and fold surface free energy of PP were markedly reduced in PP/MCC composites and its compatibilized composites. The value of
Keywords
INTRODUCTION
Polypropylene (PP) has been widely used due to its attractive combination of good processability, mechanical properties, and chemical resistance. However, inadequate stiffness and high brittleness of PP have limited its versatile application to some extent. Thus, a variety of methods such as filling, blending, enhancement, and so on were adopted to improve the performance of PP [1–6]. Microcrystalline cellulose (MCC), whose amorphous regions are removed by acid hydrolysis, can be a very promising cellulosic reinforcement of polymers. MCC is presented as short rod-like or powdered particles with the size of 20–80 µm. Compared with other natural polymers, MCC possesses strong rigidity, high crystallinity, and excellent mechanical properties [7,8]. Therefore, MCC has received a great deal of attention in the preparation and modification of materials. Especially nowadays, when oil fields are becoming depleted, MCC can be used to alleviate the problems of resources and the environment as a renewable resource. Since MCC containing polyhydroxy has inherent hydrophilic property, incompatibility problems would exist when it is introduced into nonpolar PP matrix. Thus, the key issue is to improve the interface compatibility between MCC and thermoplastic resin matrix. In recent years, compatibilizers prepared by grafting polyolefins have been reported [9]. Currently, a good compatibilizer of maleic anhydride grafted polypropylene (PP-g-MA) is being widely used [10]. Due to the fact that glycidyl methacrylate (GMA) with high reactivity of epoxy groups has the advantages of owning a single high boiling point, low toxicity and corrosion for equipment, and so on, GMA grafted polyolefins has also received a lot of attention [11].
The influence of PP and various additives on the crystalline structure and crystallization behavior of PP have been reported by many researchers [12–14], however, the effects of different compatibilizers on the crystallization kinetics of PP/MCC composites have not been reported yet. Taking the actual processing of polymers into account, techniques such as injection molding and other processes are carried out under nonisothermal conditions. Therefore, the nonisothermal crystallization kinetics study is more practically significant. In this article, crystallization behaviors of PP/MCC composites at different cooling rates were investigated. Four dynamic models were employed to deal with the nonisothermal crystallization process of PP/MCC. The kinetic parameters, like the Avrami exponent, was evaluated from the data based on differential scanning calorimeter (DSC) crystallization exotherms for pure PP, PP/MCC composites, PP grafted with maleic anhydride (PP-g-MA), and methyl acrylic acid glycidyl ester grafted PP (PP-g-GMA) modified PP/MCC composites. Furthermore, polarized optical microscopy technique was used to display the effect of MCC and compatibilizer on the nucleation density of PP.
EXPERIMENT
Materials and Equipments
A commercial grade isotactic PP (H030SG, MFR = 3 g/10 min at 200°C) used in this study was supplied by Reliance Industries Limited. MCC was obtained from Guangzhou Gaoli Additives Factory. PP-g-MA and PP-g-GMA were supplied by Guangzhou Lushan Chemical Materials Co. of China with MA and GMA grafting ratio of 1.0% and MFI of 15 g/10 min.
The following equipments were used:
The twin-screw extruder: SHJ-20, Nanjin Jieya Extrusion Equipment Co., Ltd. of China. The injection molding machine: HM7DENKEY, Nissei Plastic Industrial Co., Ltd. The universal testing machine: Zwick/Roell Z005, Zwick Roell Testing Machines Pvt. Ltd. The impact testing machine: ZBC-50, China Shenzhen SANS Testing Machine Co., Ltd. The scanning electron microscope: XL30S-FEG, Netherlands Philips Ltd. The thermogravimetric Analysis: Q5000, TA Instruments USA. The vicat softening machine: GT2HV2000, Taiwan Gotech Testing Machines Co., Ltd.
Sample Preparation
Composition of PP/MCC composites
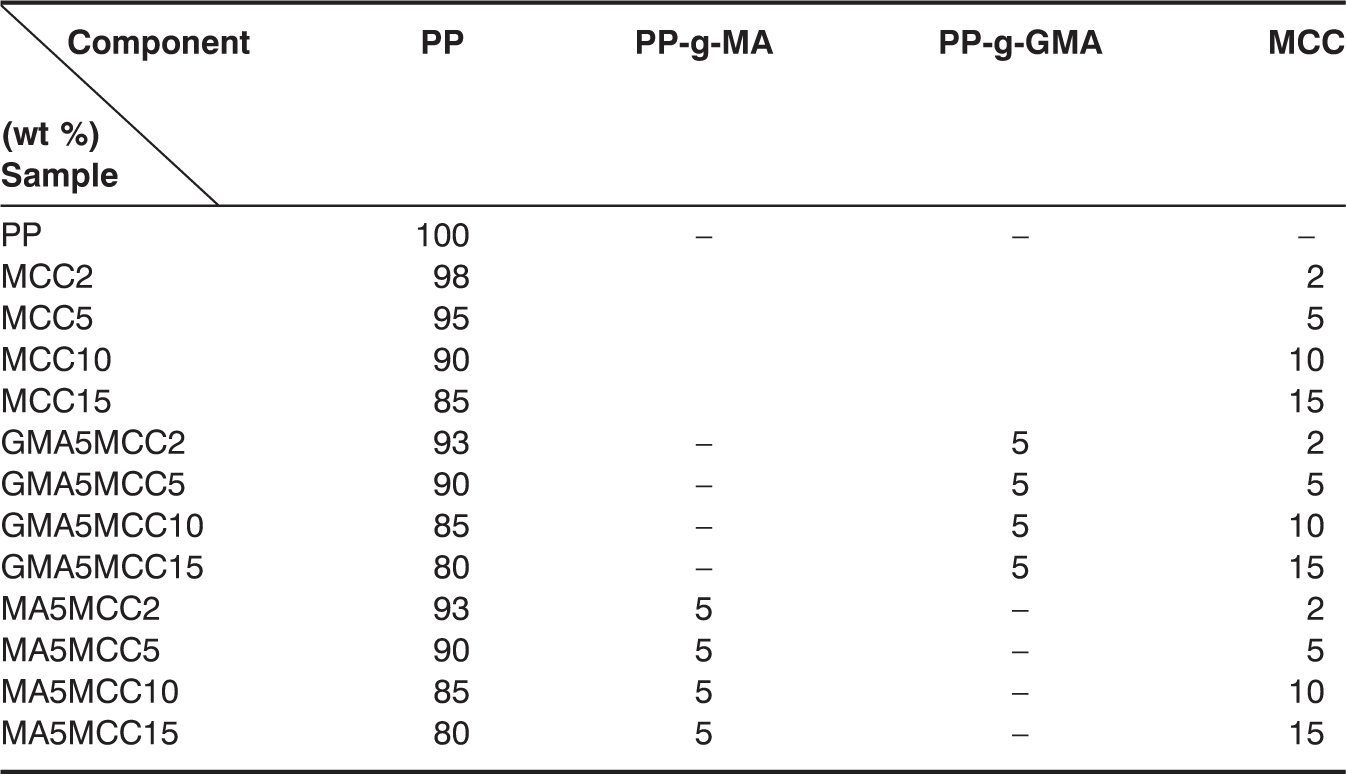
Mechanical Testing
Tensile, flexural, and Izod impact tests were carried out according to ASTM Standard. For each test as well as each type of the composites, five specimens were tested and the average values were reported. Tensile tests were conducted according to ASTM D 638 with a Universal Testing Machine at a crosshead speed of 50 mm/min. Static flexural tests were carried out according to ASTM D 790 with the same Testing Machine mentioned above at a crosshead speed of 2 mm/min. Izod Notch impact tests were conducted according to ISO 179 with a Universal Impact Testing Machine.
Microstructure Analysis
The impact specimens were frozen in liquid nitrogen for 3 h, and then quickly smashed. The fracture surfaces of the specimens were sputter-coated with gold before scanning electron microcopy (SEM) analysis. The fracture surface morphology of the composites was observed with a Philips XL-30 ESEM scanning electron microscope with an acceleration voltage of 15 kV.
DSC Characterization
A TA Instruments Q200 DSC was used to study the thermal behavior of the PP/MCC composites, and was calibrated using the melting temperature and enthalpy of a pure indium standard. About 8–9 mg of the sample was accurately weighted for DSC testing, and all measurements were performed in nitrogen atmosphere.
In nonisothermal crystallization and melting behavior characterization, a sample of the composite was rapidly heated to 220°C and held for 5 min. Subsequently, it was cooled to 60°C at the cooling rate of 10°C/min for crystallization behavior study. And then, it was reheated to 220°C at 10°C/min for melting behavior study.
In nonisothermal crystallization kinetics study, a sample of the composite was rapidly heated to 220°C and held for 5 min. Subsequently, it was cooled to 60°C at selected cooling rates ranging from 5 to 40°C/min. Each sample was used only once.
Polarized Optical Microscopy
The spherulite morphology and growth of PP/MMC composite were studied using a polarized optical microscope (POM, Zeiss) equipped with a hot stage (Linklam LTM350). The samples were hot pressed into films with the thickness of 0.02 mm. The crystallization process was recorded in the second heating scan at the rate of 0.5°C/min.
TGA Analysis
The thermal decomposition behavior of the composites was studied by a thermogravimetry (TA Instruments) in nitrogen atmosphere with the heating rate of 10°C/min.
3.1 The Effect of Compatibilizers on the Mechanical Properties of PP/MCC Composites
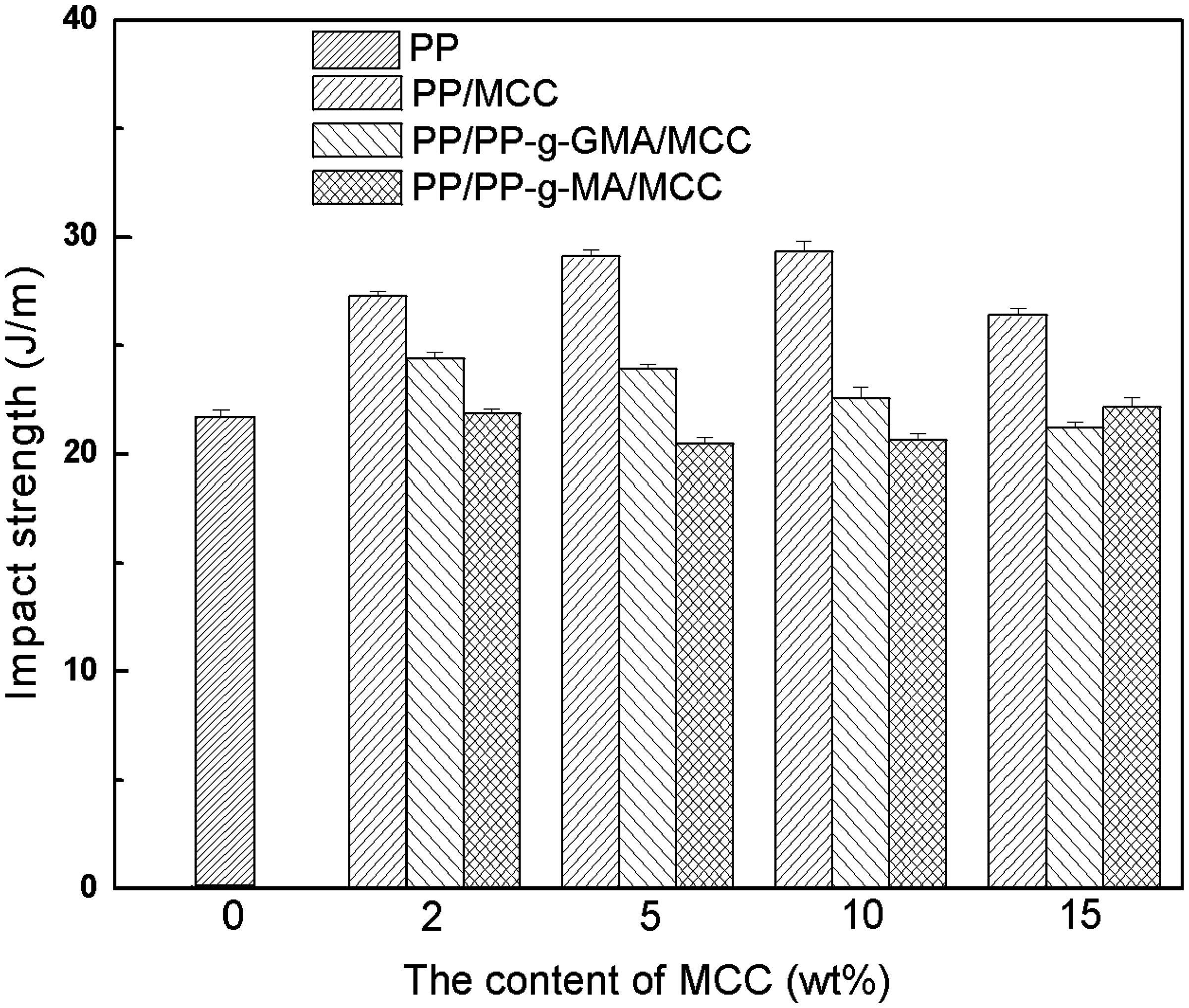
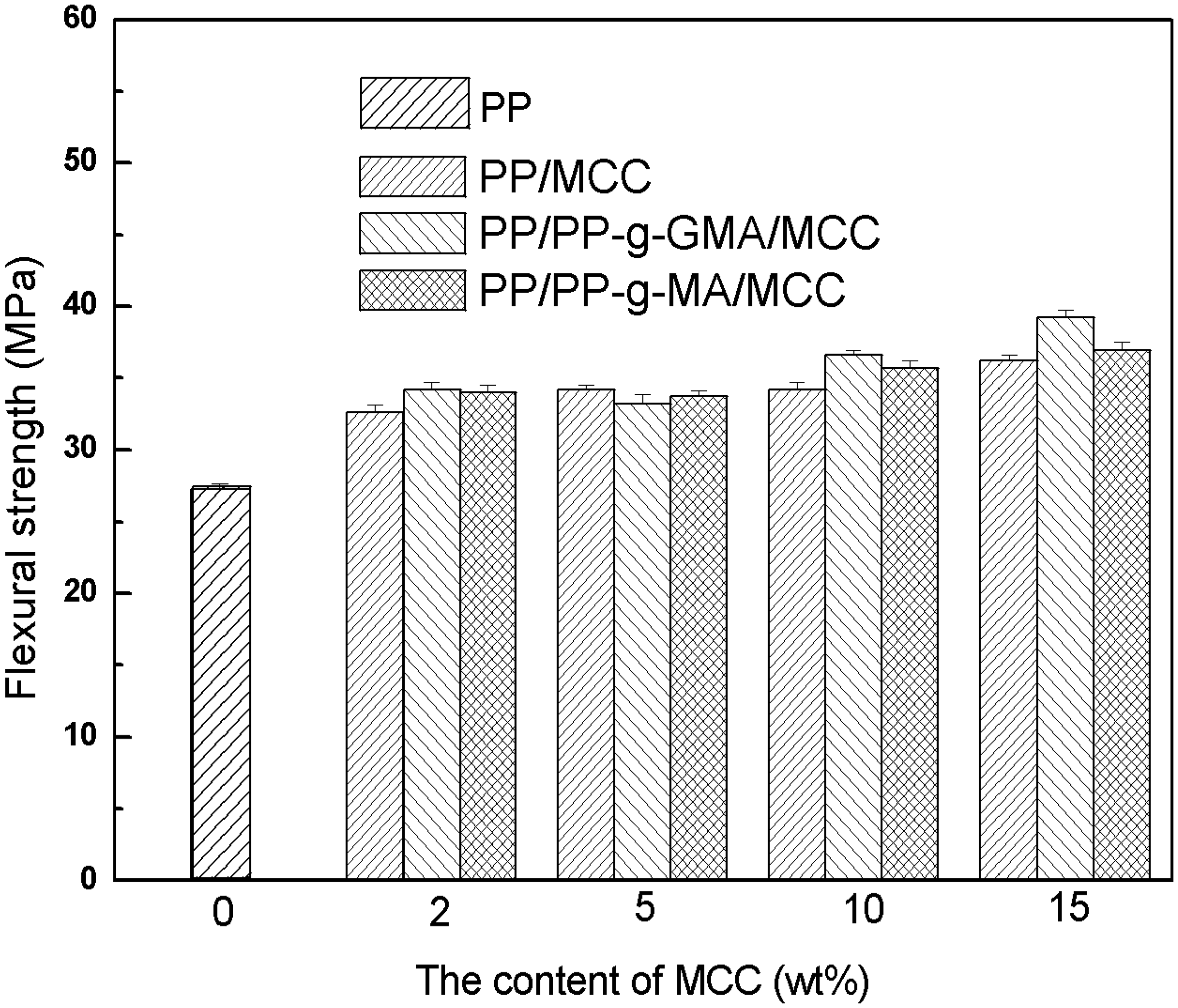
Figures 1–3 show the mechanical properties of unmodified PP/MCC, PP-g-MA modified PP/MCC, and PP-g-GMA modified PP/MCC composites. It can be seen that tensile strength decreases slightly with the increase of MCC content; however, it is still higher than that of pure PP on the whole. Particularly, PP/MCC composites modified by PP-g-GMA have an obvious increase in tensile strength. This indicates that compatibilizer improves the interfacial adhesion of PP and MCC, which is favorable to the mechanical properties of composites. The impact strength of unmodified composites is higher than that of PP/MCC composites modified by compatibilizer while the impact strength of composites modified by PP-g-MA and PP-g-GMA is basically equaled or slightly higher than that of pure PP. The impact strength of PP/MCC composites modified by PP-g-GMA increases obviously. The possible reason may be that the reaction occurred between the epoxy group of PP-g-GMA and hydroxyl groups on MCC particles, which is also effective in improving the compatibility of fibers and plastics. The flexural strength of composites filled with MCC and compatibilizer is better than that of pure PP, and shows a rising trend with the increasing MCC content. The flexural strength of MCC15, GMA5MCC15, and MA5MCC15 has improved by 32.6%, 43.6%, and 35.2%, respectively than those of pure PP at 15% MCC content.
Tensile strength of unmodified PP/MCC composites, PP-g-MA and PP-g-GMA modified PP/MCC composites. Impact strength of unmodified PP/MCC composites, PP-g-MA and PP-g-GMA modified PP/MCC composites. Flexural strength of unmodified PP/MCC composites, PP-g-MA and PP-g-GMA modified PP/MCC composites.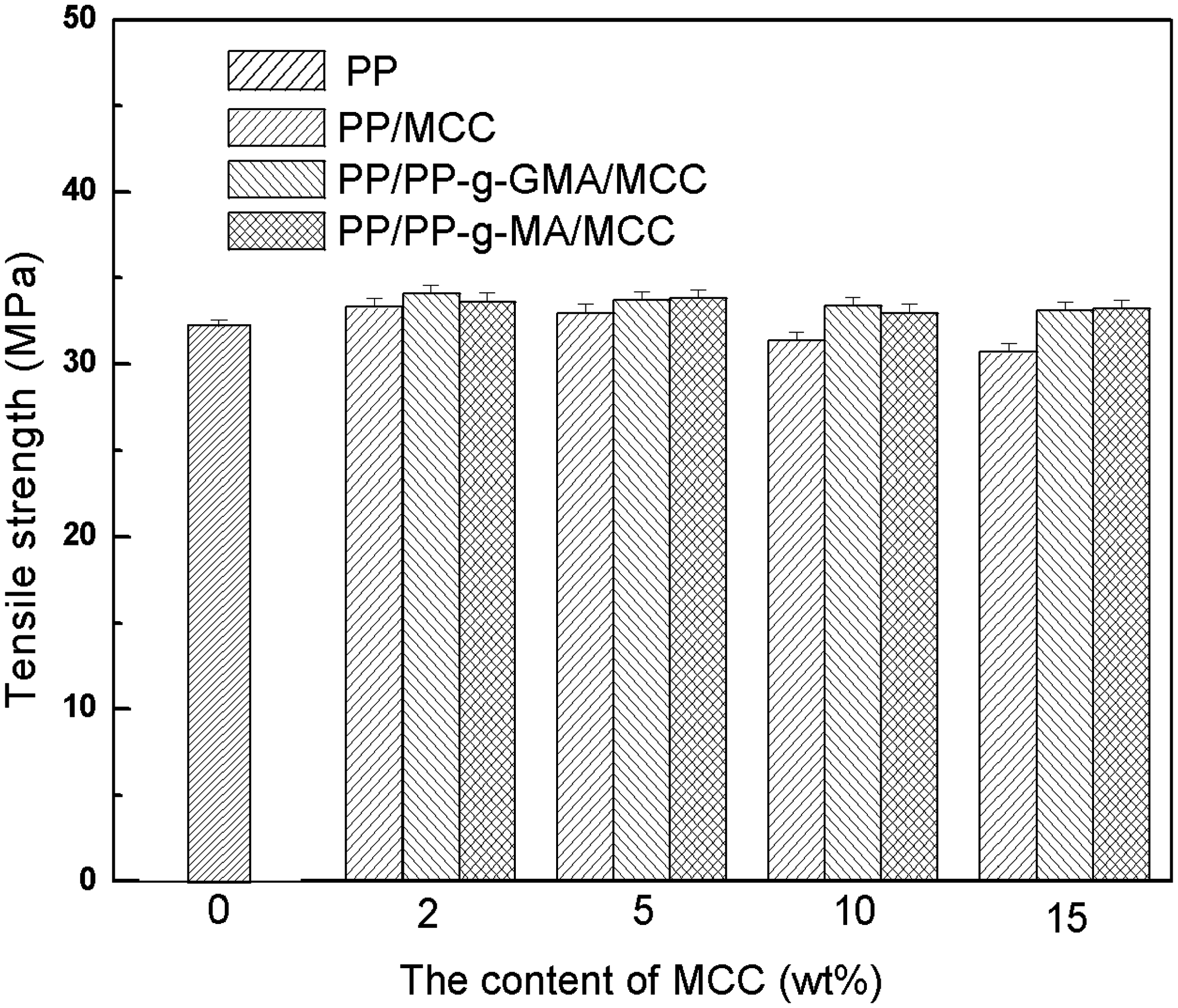
SEM Images of PP/MCC Composites
Figure 4 shows the fracture surface of unmodified PP/MCC, PP-g-MAH modified PP/MCC, and PP-g-GMA modified PP/MCC composites with 2–15 wt% of MCC. It can be seen that unmodified PP/MCC has a rough fracture surface. With increasing MCC content, the MCC agglomeration becomes more severe, and the interface is clearer between MCC particle and PP matrix, which indicates that the decrease of tensile strength is due to the bad interface between MCC and PP. Compared to unmodified PP/MCC composites, the fracture surface of modified composites is relatively smooth. On the whole, massive MCC aggregates cannot be seen, which means only a few MCC particles inlayed in PP matrix can be observed. There is a tight interface between MCC particles and PP matrix, which shows the two compatibilizers can improve the interface bond and increase the tensile strength effectively. The size of MCC particles is observed smaller in the fracture surface of PP-g-GMA modified PP/MCC composites than that of PP-g-MA modified PP/MCC composites, which indicates PP-g-GMA has a more obvious effect on MCC dispersive action and interface modification.
SEM photos of unmodified PP/MCC composites, PP-g-MA and PP-g-GMA modified PP/MCC composites: (a) MCC2; (b) MCC5; (c) MCC10; (d) MCC15; (e) GMA5MCC2; (f) GMA5MCC5; (g) GMA5MCC10; (h) MA5MCC15; (i) MA5MCC2; (j) MA5MCC5; (k) MA5MCC10; (i) MA5MCC15.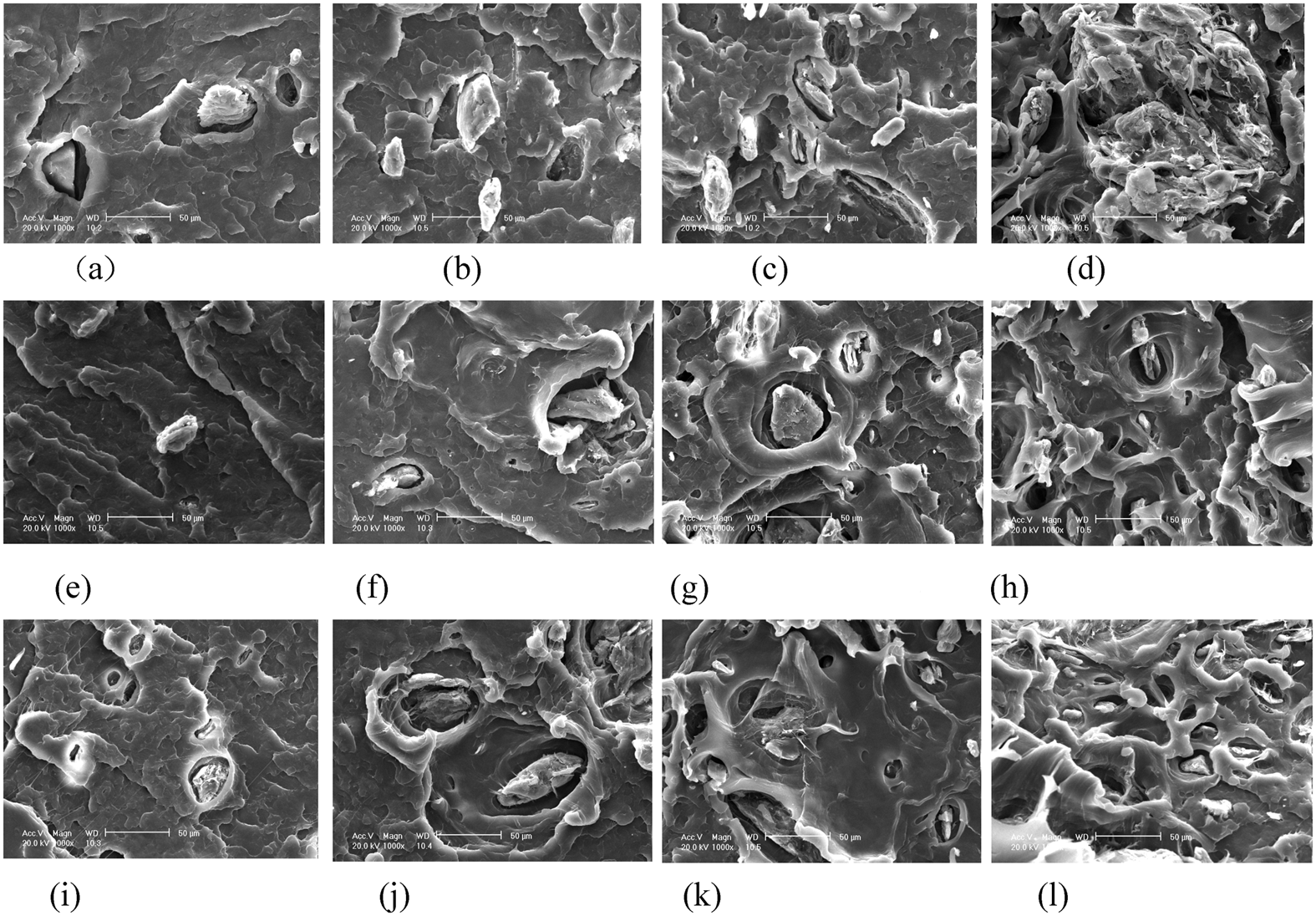
Thermal Stability for PP/MCC Composites
Thermal properties of pure PP, PP/MCC, PP-g-MA, and PP-g-GMA modified PP/MCC composites with the content of 10 wt% of MCC.
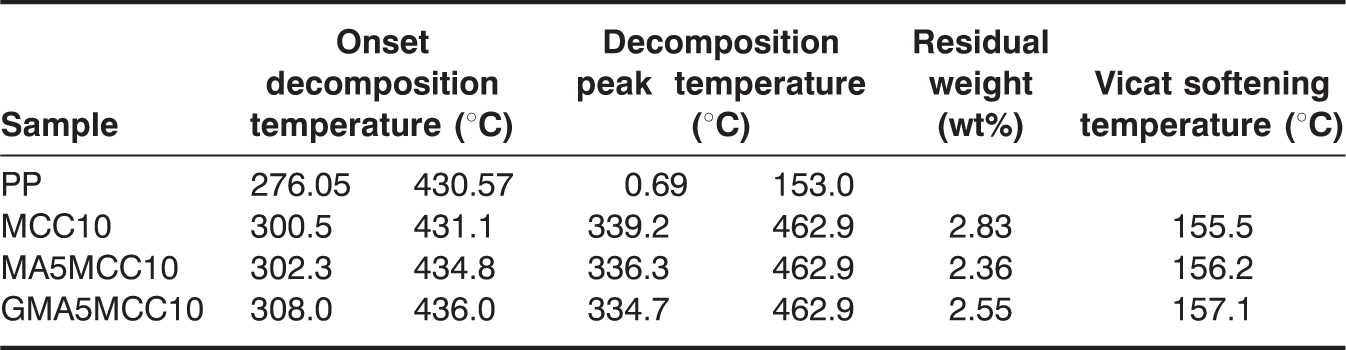
Vicat softening temperatures of unmodified PP/MCC composites, PP-g-MA and PP-g-GMA modified PP/MCC composites.
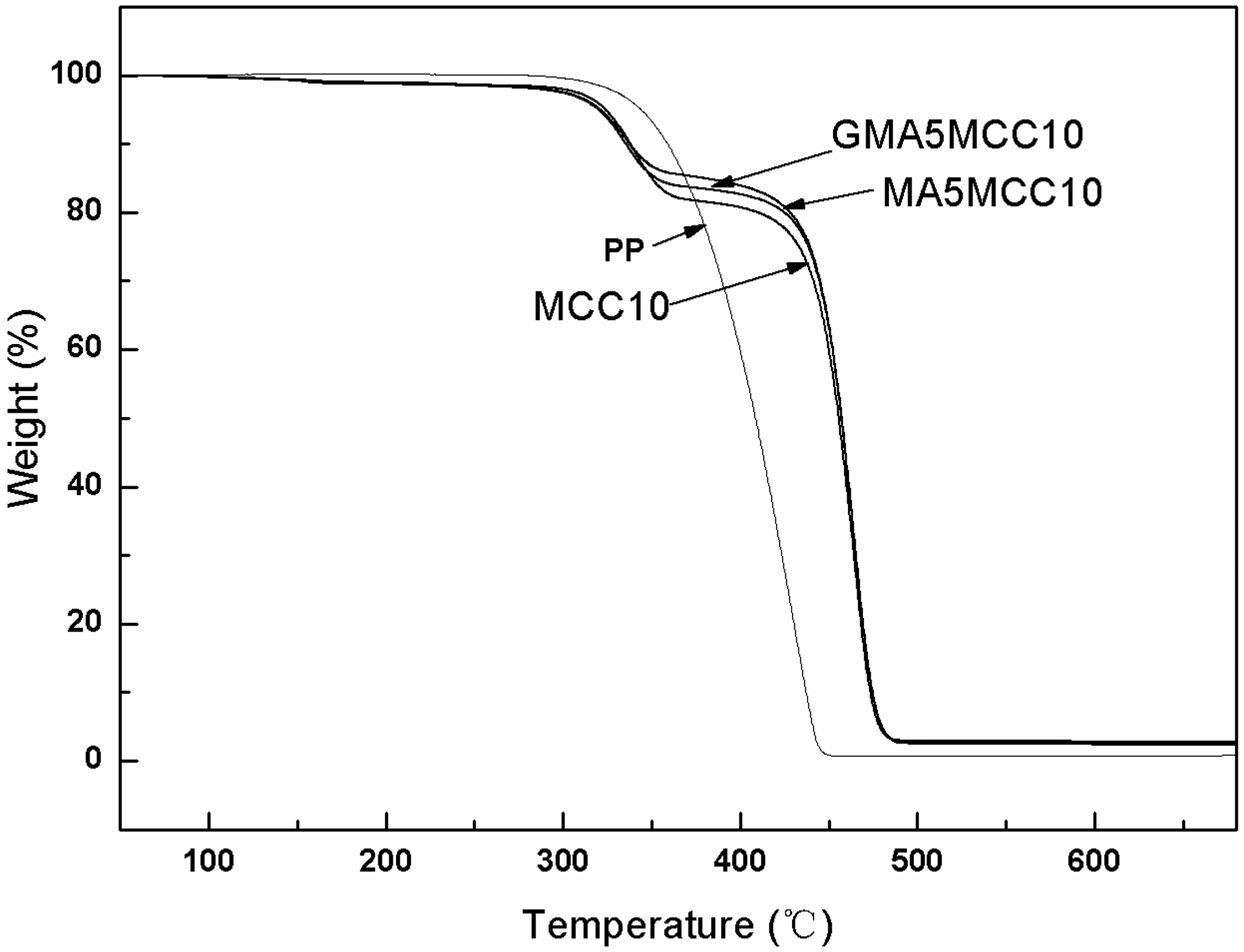
TG curves of pure PP, PP/MCC, PP-g-MA, and PP-g-GMA modified PP/MCC composites with the content of 10 wt% of MCC.
Nonisothermal Crystallization and Melting Behavior
Nonisothermal crystallization and melting parameters of pure PP, PP/MCC, PP-g-GMA, and PP-g-MA modified PP/MCC composites.
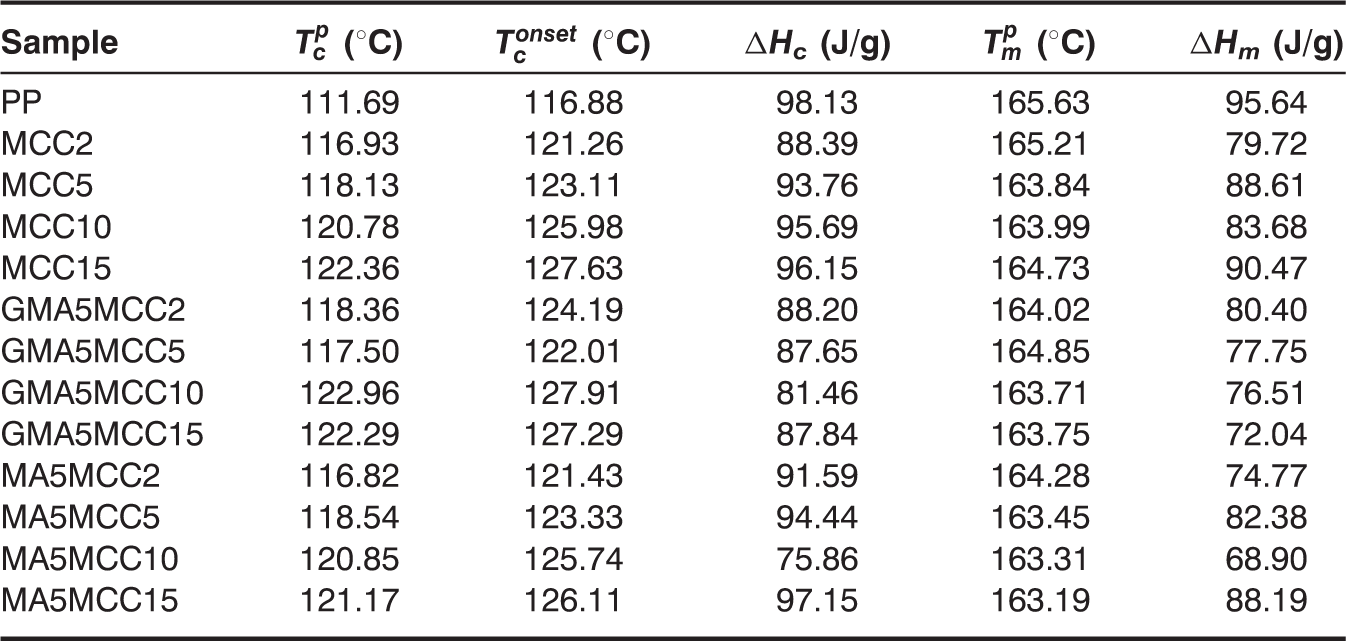
The crystallization and melting behavior of PP-g-GMA modified PP/MCC composites differed from that of PP-g-MA modified PP/MCC composites.
Nonisothermal Crystallization Kinetics
Four methods have been widely adopted to describe the nonisothermal crystallization process, that is, Avrami equation, Ozawa equation, Jeziorny equation, and Mo’s method. The double logarithmic form of Avrami equation is: [16]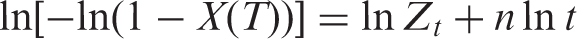
where
Jeziorny [17] calibrated the crystallization kinetic constant of Avrami equation using cooling rate Φ: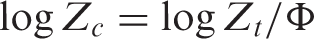
where
Ozawa derived an equation whose double logarithmic form is: [18]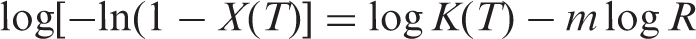
where
Liu and co-workers [19] proposed a different equation by combining the Avrami and Ozawa equations, giving rise to the relationship between cooling rate 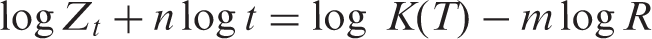
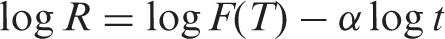
where the kinetic parameter,
Moreover, Kissinger [20] suggested a method to determine the activation energy for the transport of the macromolecular segments to the growing surface, Δ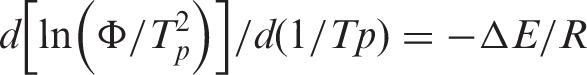
where
Nonisothermal crystallization kinetic parameters of pure PP, PP/MCC, PP-g-GMA, and PP-g-MA modified PP/MCC composites with 10 wt% MCC.
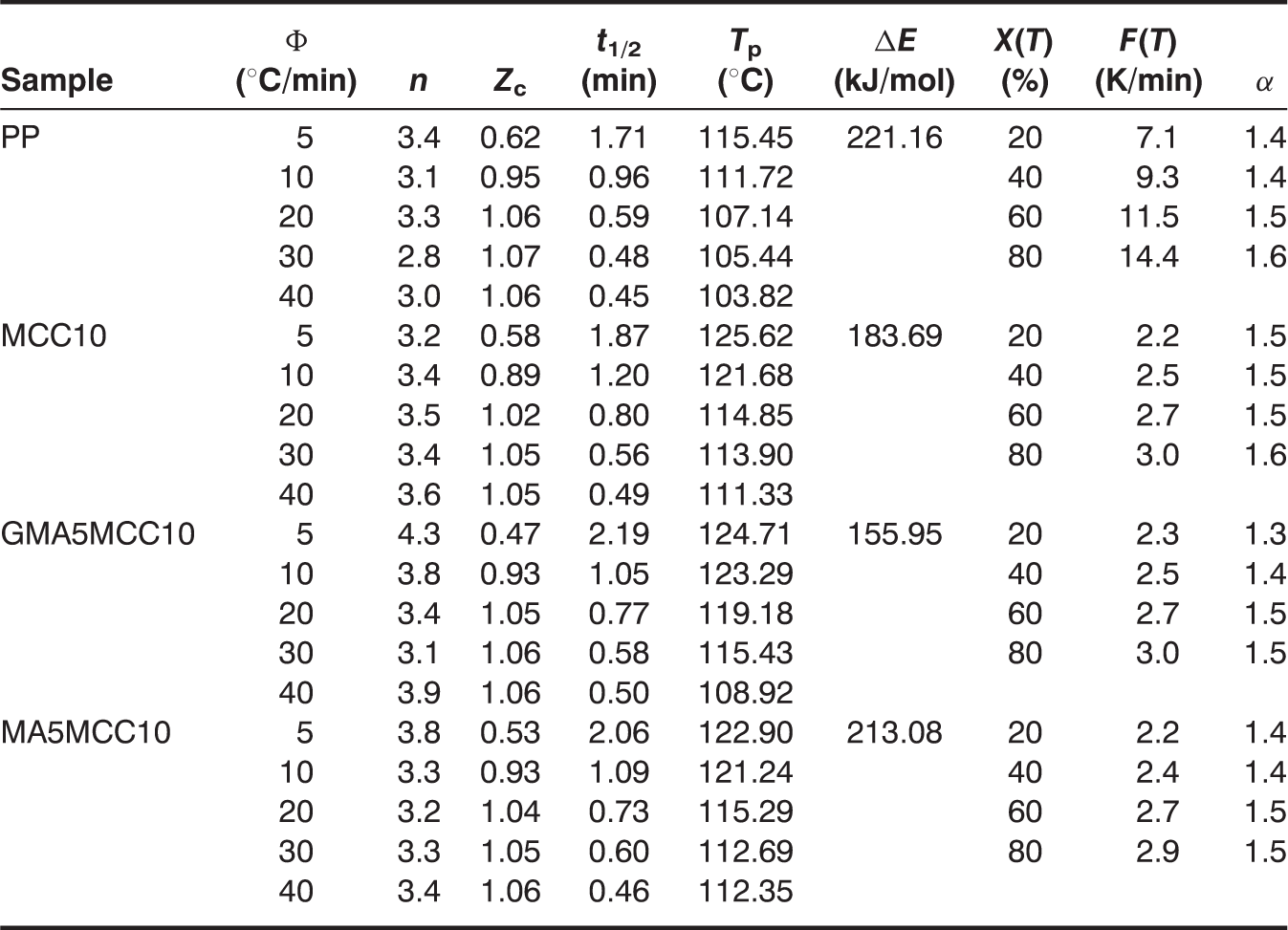
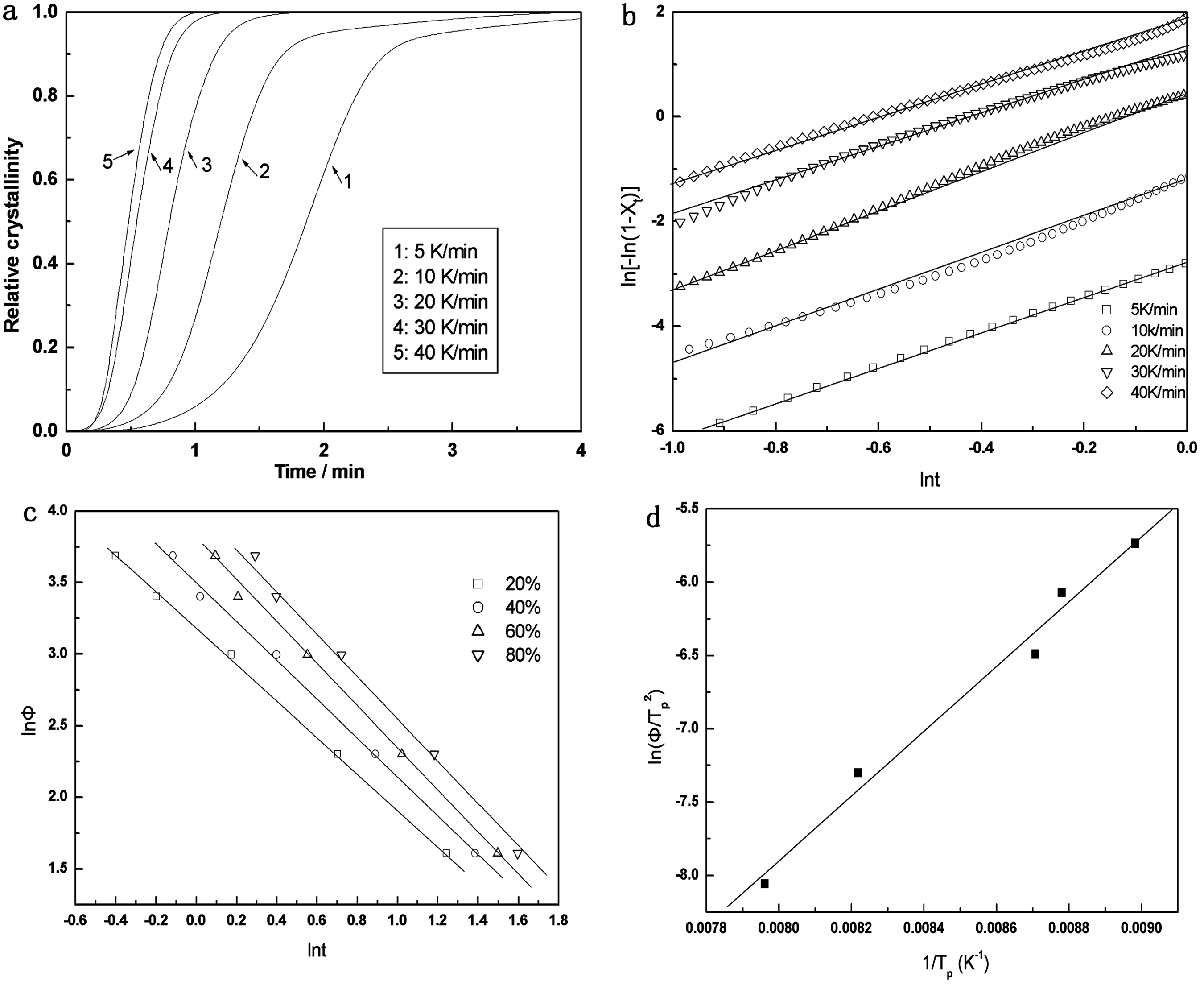
Nonisothermal crystallization kinetic plots of MCC10 composite. (a) the Plot of relative Crystallinity Versus time (b) th Plot of
POM Observation
The final performance of crystalline polymer-matrix composites depends on the crystallization behavior and crystal morphology of polymer-matrix to a large degree. In this article, the crystal morphology of pure PP, unmodified PP/MCC composite, PP-g-GMA, and PP-g-MA modified PP/MCC composite at 10 wt% of MCC were observed by polarized optical microscopy (POM) (Figure 8). The spherulites with black cross extinction phenomena and the obvious crystal interface were observed in pure PP (Figure 8(a)).
POM micrographs of PP/MCC composite, PP-g-GMA and PP-g-MA modified PP/MCC composite with 10 wt% of MCC: (a) PP; (b) MCC10; (c) GMA5MCC10; (d) MA5MCC10.
In Figure 8, it is clear that the nucleation rates of PP/MCC composites are larger than that of PP due to the heterogeneous nucleation of MCC and compatibilizers particles. Crystal growth is ultimately limited by facing spherulite boundary, and it will lead to an irregular final structure and smaller crystalline dimension. It is well known that crystalline morphology affects the mechanical properties of a material significantly since a smaller crystal is prone to develop into a crack to absorb impact energy while a big spherulite tends to form local stress which makes the material brittle. Therefore, the crystalline morphology of PP/MCC is favorable to its mechanical properties.
Water Absorption for PP/MCC Composites
The samples (10 mm × 10 mm × 4 mm) were impregnated in the breaker filled with distilled water for 48 h at room temperature after being dried in an oven at 50°C for 24 h, and then, all the water on the sample surface was quickly wiped off with a filter. Subsequently, the sample was quickly weighed to get the weight after water absorption. The percent of weight gain after being soaked is the percentage of water absorption of the sample, as is shown in Table 3. The water absorption of composite remained unchanged with the addition of MCC containing hydroxyl groups, which indicates that MCC has good dimensional stability as the filler in wood-plastic composites. Thus, MCC is of great potential in the application of wood-plastic composites.
CONCLUSIONS
In this study, the PP/MCC composites were prepared by extrusion blending and injection molding. Also, two different types of compatibilizers, that is, PP-g-GMA and PP-g-MA, were used to modify the interface of PP/MCC composites. The results indicated that the addition of MCC led to the increase of tensile strength and impact strength of PP. PP-g-GMA modification is more conducive to the improvement in tensile strength and impact strength while PP-g-GMA modification is more favorable to improve the tensile properties and toughness. PP-g-MAH and PP-g-GMA modification improve the dispersion and interfacial adhesion between MCC and PP, which can explain why mechanical properties of composites are improved. The three types of PP/MCC composites have higher thermal decomposition temperature and Vicat softening temperature and dimensional stability. To sum up, compared to the mineral fiber reinforcements (such as glass fiber), MCC has the advantages of being lightweight, inexpensive, and high-quality as a plastic reinforcement. Four methods were used to deal with the nonisothermal crystallization process of PP/MCC composites and PP-g-GMA and PP-g-MA modified PP/MCC composites. The Avrami equation, Ozawa equation, Jeziorny’s, and Mo’s methods can give a satisfactory description of the nonisothermal crystallization behavior of PP/MCC composites. According to the results obtained by Avrami equation, the primary crystallization stage for nonisothermal melt crystallization might correspond to a three-dimensional spherical growth with heterogeneous nucleation. The values of the kinetics parameter
Footnotes
ACKNOWLEDGMENTS
This work was supported by China Guangdong scientific and technological project (No2010B080701060) and Guangdong natural science fund (No8451063201000041).
