Abstract
Physical frailty is associated with adverse health outcomes in older adults, including cognitive decline; however, the underlying biological and pathophysiological mechanisms linking frailty to neuropathological biomarkers remain underexplored, particularly in older adults without dementia. This study examined associations between physical frailty and cognitive performance and biomarkers based on the amyloid, tau, and neurodegeneration (ATN) framework in a sample of community-dwelling older adults without dementia. Using baseline data from a randomized controlled trial, we conducted a secondary cross-sectional analysis of 137 sedentary adults aged 60-85 years with insomnia symptoms and Montreal Cognitive Assessment scores above 17. Frailty was measured using the Johns Hopkins Frailty Assessment Tool. Cognitive performance was assessed with the CogState computerized battery, and plasma biomarkers—including amyloid beta 42/40 ratio (Aβ42/Aβ40), total tau (t-tau), and neurofilament light chain (NfL)—were quantified using Single Molecule Array (Simoa) technology in a subsample (n = 98). In adjusted models, frail participants demonstrated significantly lower global cognition, processing speed, and attention compared to robust individuals. Prefrail participants did not show significant differences in cognitive performance. For biomarkers, prefrail individuals had significantly lower Aβ42/Aβ40 ratios in unadjusted models, and frail individuals showed significantly higher NfL levels in adjusted models. No significant associations were found for t-tau. These findings suggest that physical frailty is linked to both cognitive impairment and early neurobiological changes in older adults without dementia. Incorporating frailty assessments alongside biomarker evaluation may enhance early detection of Alzheimer’s-related changes and guide timely interventions to mitigate cognitive decline.
Introduction
Physical frailty, defined as a clinical syndrome characterized by a decline in physiological reserves, strength, and endurance, 1 is a prevalent concern among older adults. A substantial body of literature indicates that physical frailty significantly contributes to poor health outcomes, including increased risks of losing independence, hospitalization, disability, and mortality.2–4 Furthermore, physical frailty not only affects physical health but also has been associated with cognitive health.5,6 A systematic review has demonstrated that older adults with physical frailty are more likely to have coexisting cognitive impairment and/or dementia. 7 This connection suggests the possibility of shared underlying biological mechanisms, potentially contributing to both physical and cognitive decline. 8 These findings highlight the need to explore whether physical frailty can act as an early and accessible indicator of cognitive health.
Alzheimer’s disease is the most common cause of dementia and is defined by specific neuropathological changes, including amyloid plaques and tau tangles. Alzheimer’s disease pathology biomarkers classified under the ATN framework, which include Amyloid deposition (A), Tau pathology (T), and Neurodegeneration or neuronal injury (N) are pivotal for diagnosing and monitoring of cognitive decline or neurodegeneration, 9 particularly in Alzheimer’s disease and other neurodegenerative disorders. A growing body of evidence indicates that ATN biomarkers are significantly associated with cognitive performance, thus reinforcing their roles as reliable indicators of cognitive health.10,11
From a pathophysiological perspective, amyloid plaque accumulation in the brain is associated with a decreased cerebrospinal fluid (CSF) amyloid-β42/amyloid-β40 (Aβ42/Aβ40) ratio, serving as a key indicator of amyloid pathology. 12 Concurrently, elevated levels of tau proteins in CSF, such as total tau (t-tau), reflect tau-related neurofibrillary degeneration.12,13 Additionally, CSF neurofilament light chain (NfL), a marker of axonal injury, is increased in neurodegenerative conditions.12,14 Despite their diagnostic significance, the invasive nature of CSF biomarker collection via lumbar puncture limits their accessibility in routine clinical practice due to associated risks such as neurological complications and infection. Blood-based biomarkers, while still involving venipuncture, offer a safer and more accessible alternative. Plasma markers such as Aβ42/Aβ40 ratios, tau, and NfL showed strong correlations with their CSF counterparts,15–17 further supporting their relevance in both clinical and research settings.
Emerging evidence has begun to explore the biological underpinnings of frailty using plasma biomarkers. However, prior studies have often focused on specific subgroups, such as individuals with positive amyloid positron emission tomography (PET)
18
or Hispanic women,
19
limiting the generalizability of the findings. Furthermore, previous studies have frequently restricted their analysis to specific plasma biomarkers like NfL without concurrent amyloid or tau data,
20
or focused on dementia risk rather than the specific characterization of physical frailty.
21
Consequently, few studies have simultaneously applied the full plasma ATN framework to determine independent associations with physical frailty in general community-dwelling older adults. (a) To examine the association between physical frailty and cognitive performance, (b) To assess the relationship between physical frailty and plasma biomarkers (ATN: amyloid, tau, and neurofilament light chain) as indicators of cognitive health.
Methods
This study is a secondary analysis of baseline data collected from a randomized controlled trial among community-dwelling older adults without dementia (ClinicalTrials.gov ID# NCT03959202). The trial received IRB approval from Johns Hopkins University (IRB# 00198497).
Participants
From the parent study, 22 participants were recruited based on the following inclusion criteria: a) community-dwelling older adults aged between 60 and 85 years; b) having a sedentary lifestyle over the last 6 months, defined as engaging in fewer than two 20-minute exercise sessions per week and self-reported more than six hours of sedentary behavior per day; c) self-reported insomnia symptoms during the past month (e.g., difficulty in falling asleep, staying asleep, or waking up too early); d) without diagnosed dementia and having a Montreal Cognitive Assessment (MoCA) score greater than 17 and no impairment in daily life function; and e) capable of engaging in mild to moderate intensity exercise. Individuals with untreated sleep apnea were excluded.
Measures
During the baseline data collection visit, trained research team members assessed participants for frailty and cognitive performance. For those who provided consent, blood samples were collected at the end of the visit. Detailed descriptions of the measurements employed in this study are provided below.
Frailty Assessment
Frailty severity was measured using the Johns Hopkins Frailty Assessment Tool, 23 a validated instrument based on the Fried Frailty Phenotype. 24 This tool evaluates five criteria: unintentional weight loss, exhaustion, weakness, slow walking speed, and low physical activity. Unintentional weight loss was assessed by self-reported loss of more than 10 pounds in the past year, while exhaustion was measured through self-reported fatigue or low energy levels. Weakness was evaluated using grip strength measured with a dynamometer, and slow walking speed was assessed by timing participants over a short walking distance (4 meters). Low physical activity was determined based on self-reported physical activity levels. Participants were categorized into three groups based on the number of criteria met: robust (no criteria met), prefrail (1-2 criteria met), or frail (≥3 criteria met).
Cognitive Performance
Cognitive performance was assessed using the CogState Battery, a computer-based cognitive testing tool with established reliability and validity for assessing multiple domains in older adults with normal cognition, mild cognitive impairment (MCI), and dementia.25–27 Studies have demonstrated its concurrent validity, showing significant correlations with standard neuropsychological assessments such as the Trail Making Test, the Hopkins Verbal Learning Test, and the Digit Span task.25,28 The analysis included a global (composite) cognitive score as well as domain-specific scores for the following tasks: • Detection Task (DET): Measures processing speed by assessing reaction time. • Identification Task (IDN): Evaluates attention and processing speed by requiring participants to correctly identify a stimulus. • Groton Maze Learning Task (GML): Assesses executive function and spatial problem-solving through a maze navigation task. • International Shopping List Task (ISL): Measures verbal learning by requiring recall of a list of items immediately after presentation. • International Shopping List Recall Task (ISRL): Evaluates verbal memory by assessing delayed recall of the previously presented list. • One Card Learning Task (OCL): Measures visual learning by requiring participants to recognize previously seen cards. • One Back Task (ONB): Assesses working memory by requiring participants to identify whether a card matches the one presented immediately before it.
Each task score was standardized using z-scores based on the sample mean and standard deviation. A global cognitive score was calculated by averaging the z-scores across all tasks. All cognitive scores, including the global score, were analyzed as continuous variables to provide a comprehensive assessment of cognitive function. All cognitive tests were administered in a single session by trained research staff.
ATN Biomarker
For the current study, neuropathological biomarkers were analyzed using the ATN framework. Amyloid beta (Aβ) biomarkers were measured as the Aβ42/Aβ40 ratio. Tau pathology, representing neuronal damage, was assessed using total tau (t-tau). Neurodegeneration was measured using neurofilament light chain (NfL). All neuropathological biomarkers were quantified from plasma samples processed using Simoa (Single Molecule Array) technology by Quanterix Corporation. 29 All biomarker values (pg/mL) were log-transformed and analyzed as continuous variables, and outliers exceeding 4 standard deviations from the mean were excluded, if any.
Statistical Analysis
Descriptive statistics were calculated to summarize participant characteristics, including age, gender, race, education, number of chronic conditions, MoCA score, and MCI status, defined as a MoCA score of 18 to 25, for the overall sample and by physical frailty statuses (Table 1). Group differences by frailty status were examined using ANOVA for continuous variables and Chi-square tests for categorical variables. Frailty was modeled as a categorical independent variable (robust [reference], prefrail, and frail) using dummy variables in the regression analyses. Unadjusted and adjusted linear regression analyses were conducted to examine: 1) The association between frailty severity (independent variable) and cognitive performance (dependent variable); 2) The relationship between frailty severity (independent variable) and ATN biomarkers (dependent variables). We adjusted for age, sex, education, and number of chronic conditions as these are key demographic and health-related factors that can confound the relationship between frailty and cognitive outcomes.
30
All statistical analyses were performed using SPSS version 26, with statistical significance set at
Results
Sample Characteristics
Sample Characteristics of the Total Sample and by Frailty Status.
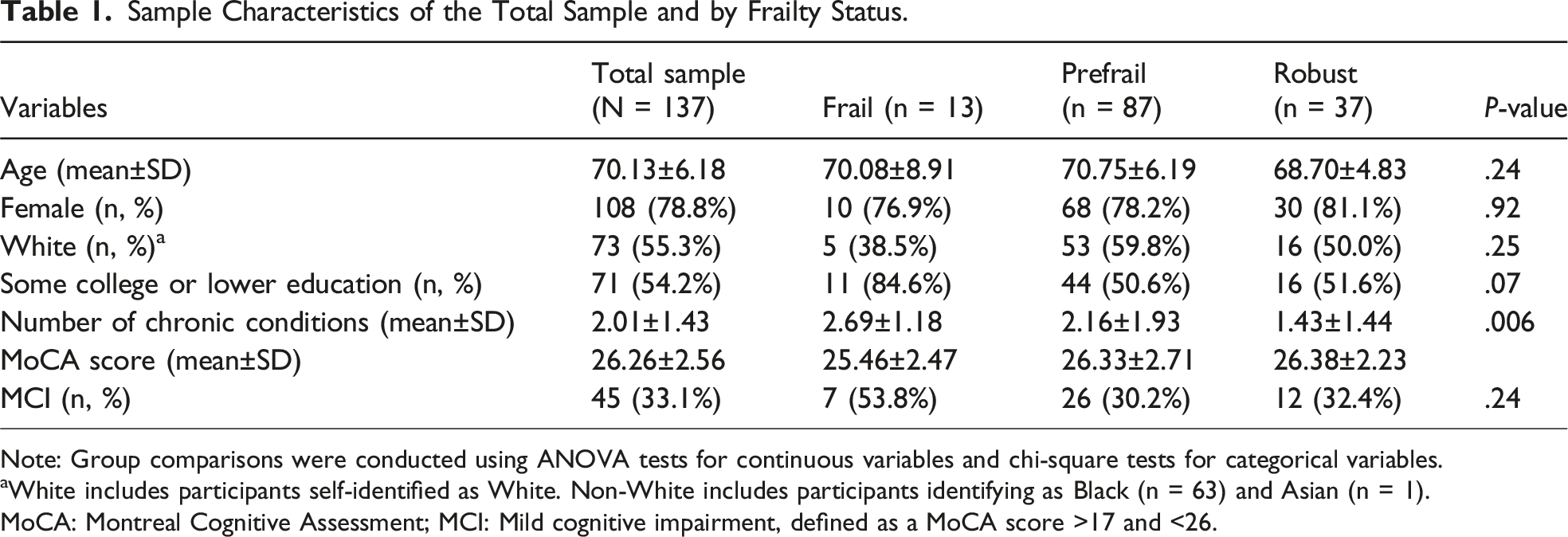
Note: Group comparisons were conducted using ANOVA tests for continuous variables and chi-square tests for categorical variables.
aWhite includes participants self-identified as White. Non-White includes participants identifying as Black (n = 63) and Asian (n = 1).
MoCA: Montreal Cognitive Assessment; MCI: Mild cognitive impairment, defined as a MoCA score >17 and <26.
Frailty and Cognitive Performance
Linear Regression Between Frailty and Cognitive Performance.
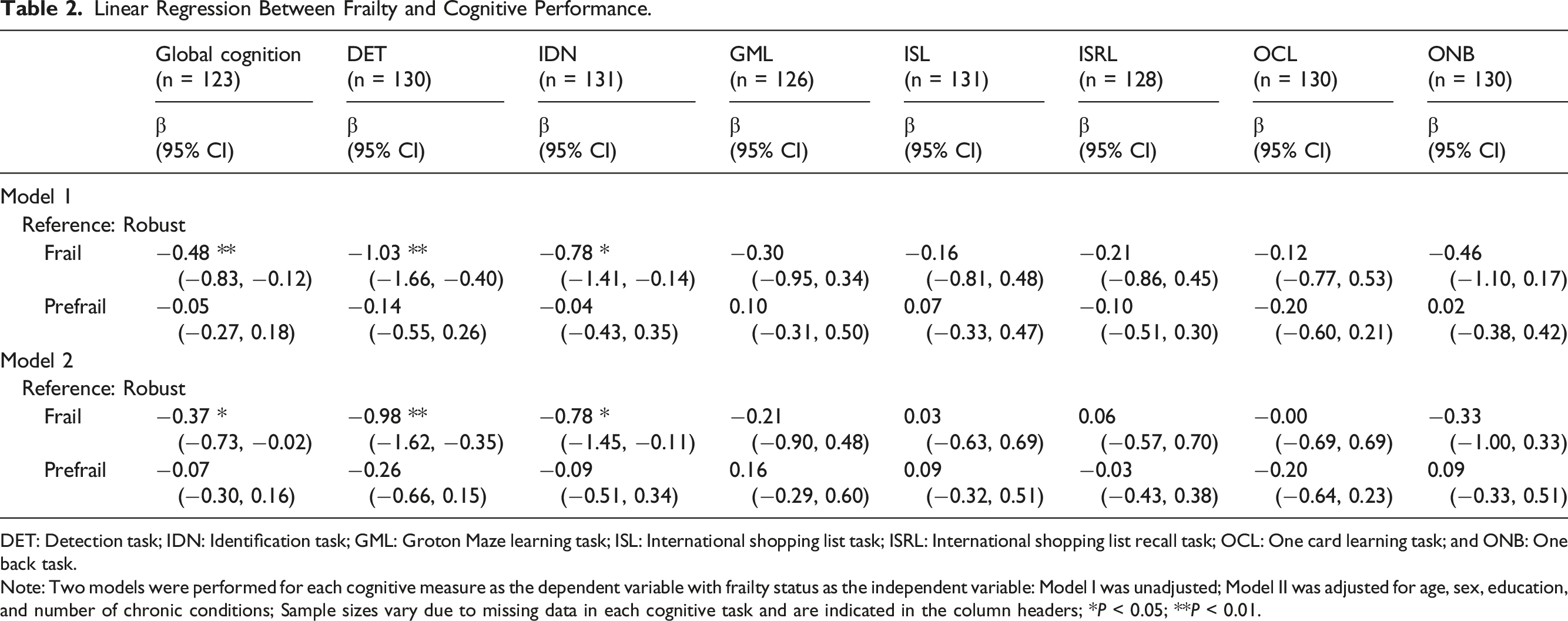
DET: Detection task; IDN: Identification task; GML: Groton Maze learning task; ISL: International shopping list task; ISRL: International shopping list recall task; OCL: One card learning task; and ONB: One back task.
Note: Two models were performed for each cognitive measure as the dependent variable with frailty status as the independent variable: Model I was unadjusted; Model II was adjusted for age, sex, education, and number of chronic conditions; Sample sizes vary due to missing data in each cognitive task and are indicated in the column headers; *
Frailty and ATN Biomarkers
Linear Regression Between Frailty and Cognitive Biomarkers (N = 98).
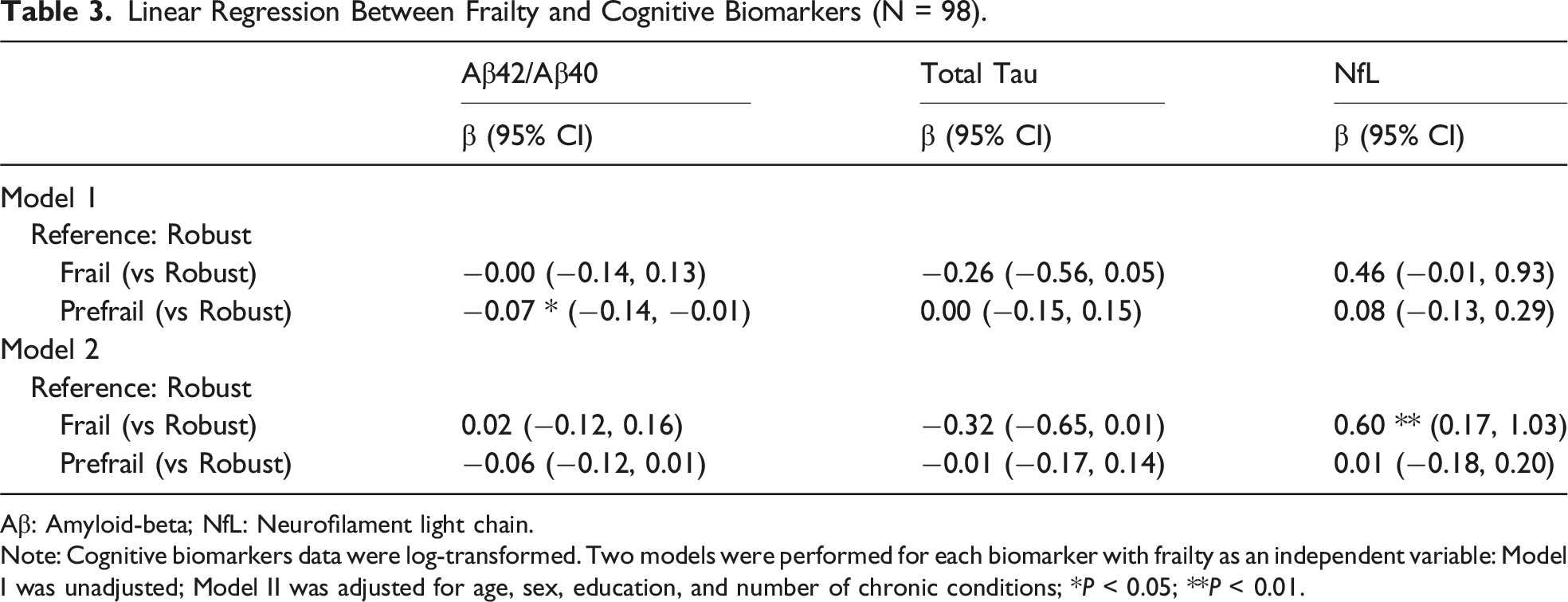
Aβ: Amyloid-beta; NfL: Neurofilament light chain.
Note: Cognitive biomarkers data were log-transformed. Two models were performed for each biomarker with frailty as an independent variable: Model I was unadjusted; Model II was adjusted for age, sex, education, and number of chronic conditions; *
For the prefrail group, Model 1 results indicated a statistically significant negative association with the Aβ42/Aβ40 ratio (β = −0.07, 95% CI: −0.14, −0.01), whereas no significant relationships were observed for t-tau (β = 0.00, 95% CI: −0.15, 0.15) or NfL (β = 0.08, 95% CI: −0.13, 0.29). In the adjusted Model 2, the association between the prefrail group and the Aβ42/Aβ40 ratio was attenuated and no longer statistically significant (β = −0.06, 95% CI: −0.12, 0.01). Similarly, no significant relationships were observed for t-tau (β = −0.01, 95% CI: −0.17, 0.14) or NfL (β = 0.01, 95% CI: −0.18, 0.20).
Sensitivity analyses addressing potential attrition bias revealed no significant differences in frailty status between participants with complete and incomplete data (
Discussion
This study examined associations of physical frailty with cognitive performance and plasma ATN biomarkers, using baseline data collected from the randomized controlled trial of sedentary, community-dwelling older adults with sleep complaints but without dementia—a group considered at high risk for progression to Alzheimer’s disease.31–33 Notably, despite the sedentary nature of the sample, the frailty assessment retained discriminative validity, identifying 27% of participants as robust. This suggests that the measurement did not artificially inflate frailty scores due to physical inactivity, allowing for a reliable examination of its associations. Our findings showed that physical frailty was significantly associated with lower cognitive performance, particularly in global cognition, processing speed (DET), and attention (IDN). For biomarkers, prefrail individuals had lower Aβ42/Aβ40 ratios, while frail individuals had higher plasma NfL levels, suggesting early pathophysiological changes even in the absence of clinical dementia.
These findings are consistent with prior research. A previous meta-analysis reported significantly lower global cognition in both prefrail and frail individuals 34 and another found negative associations with specific cognitive domains such as processing speed and attention. 7 Considering that processing speed reflects the ability to react and execute motor responses, it may be particularly sensitive to physical decline. 35 While other studies have shown associations between frailty and additional cognitive domains (e.g., executive function, immediate memory, and delayed memory), our study did not observe significant differences in these domains beyond DET and IDN, although negative trends were observed. One key strength of this study is the use of a computerized cognitive battery, which provided standardized, objective evaluation across multiple cognitive domains and reduces bias often seen in human-rated tools. 36 Future research should utilize computerized cognitive assessment tools to further validate the accuracy and efficacy of these measures in evaluating cognitive function.
Regarding plasma ATN biomarkers, the prefrail group showed significantly lower Aβ42/Aβ40 ratios in unadjusted models, aligning with studies linking amyloid pathology to frailty.18,37 Plasma Aβ42/Aβ40 ratio tends to decrease in Alzheimer’s pathophysiological states, where amyloid beta accumulates in the brain. 38 Correspondingly, brain imaging studies with positron-emission tomography (PET) have suggested that amyloid beta accumulation in the brain is associated with frailty.39,40 These findings suggest that cognitive decline and physical frailty may share common biological or pathophysiological mechanisms, which need to be explored in future research. We observed significantly lower Aβ42/Aβ40 levels in prefrail compared to robust older adults in the unadjusted model. Notably, this association remained significant in our sensitivity analysis adjusting for global cognition. This suggest that individuals in the prefrail stage may already exhibit early alterations in amyloid processing independent of concurrent cognitive performance, potentially increasing their risk for cognitive decline. This underscores the need for early identification of at-risk individuals and timely intervention during the prefrail stage to help mitigate cognitive decline. However, we did not observe significant differences in Aβ42/Aβ40 between the frail and robust groups, which may be attributed to the small sample size of the participants with frailty, potentially resulting in insufficient statistical power to detect significant association. Thus, further investigations with larger sample size are needed to better understand the underlying pathways and mechanisms linking frailty and amyloid beta.
We did not find a significant association between frailty and t-tau. This aligns with findings from a prior study of community-dwelling older adults, which reported limited specificity of t-tau.
41
Notably, the study found that phosphorylated tau (
In our analysis, a significant association between frailty and elevated NfL levels was observed in the adjusted model. This finding is consistent with previous studies. For example, one study reported a significant positive correlation between NfL levels and frailty among individuals with amyloid deposition.
18
Another study using regression analysis with frailty as the dependent variable found that higher NfL levels were associated with a greater likelihood of pre-frailty and frailty,
20
though these associations did not persist after adjusting for demographics, chronic conditions, and cognitive performance.
20
Similarly, in our sensitive analysis, the association between frailty and NfL was attenuated after adjusting for global cognition. However, given that global cognition itself was not associated with NfL (
Given the limited evidence on the relationship between physical frailty and blood-based ATN biomarkers, our findings contribute to an emerging area of research with significant clinical implications. Since blood biomarkers are more convenient and widely accessible compared to CSF and imaging-based measures, further validation of their utility in detecting neurodegenerative risk in frail older adults could advance early detection and targeted interventions.
In summary, this study demonstrates the potential of physical frailty as a surrogate marker for cognitive performance and neuropathological biomarkers among older adults without dementia. Specifically, individuals with frailty, compared to robust individuals, demonstrated significant impairments in global cognition, particularly in processing speed and attention cognitive domains. Regarding plasma ATN biomarkers, prefrail status was significantly associated with a lower Aβ42/Aβ40 ratio, while frailty was linked to higher NfL levels. Our findings have important implications for clinical practice, particularly for older adults at high risk of cognitive impairment progression. First, physical frailty—and even prefrail status—may serve as a simple and non-invasive screening tool to identify individuals at high risk for cognitive decline, enabling earlier detection and timely intervention. Regular frailty assessments could be integrated into primary care or geriatric assessment for older adults without dementia. Furthermore, frailty status could guide the use of blood-based biomarkers such as Aβ42/Aβ40 or NfL, improving early detection of individuals more likely to exhibit early Alzheimer’s-related changes. In addition, interventions targeting physical frailty may serve as a critical point for preventing cognitive impairment, given the relationship between physical frailty and cognitive performance.
Despite these contributions, the study has limitations. First, the cross-sectional design restricts the ability to infer causal relationships between frailty, cognitive performance, and neuropathological biomarkers levels. Second, the relatively small sample size, particularly in the frail group may limit statistical power to detect associations, especially for biomarker analyses. Third, as a secondary analysis of a randomized controlled trial, the sample was restricted to older adults with insomnia and a sedentary lifestyle, which may limit generalizability. These specific inclusion criteria may have influenced study outcomes given their association with frailty and cognitive decline. Furthermore, reliance on the parent trial design precluded the exploration of additional relevant variables such as p-tau or longitudinal outcomes. Fourth, while the plasma Aβ42/Aβ40 ratio is widely used as a proxy for amyloid pathology, it may be influenced by various biological and environmental factors, and is subject to measurement variability. The absence of confirmatory biomarkers such as CSF measures or amyloid PET imaging limits our ability to determine true amyloid positivity and may reduce confidence in the interpretation of observed associations.
Future prospective research with larger sample sizes and longitudinal designs is essential to elucidate the mechanisms and causal pathways linking physical frailty, neuropathological biomarkers, and cognitive decline. Specifically, further investigation is needed to verify whether prefrailty serves as an early, independent indicator of amyloid pathology, whereas frailty and neuroaxonal injury represent a more unified phenotype closely coupled with cognitive decline. In addition, considering that the relationship between biomarkers and frailty may vary depending on conditions like amyloid deposition, future research should focus on developing integrated predictive models that combine physical frailty with diverse neuropathological biomarkers. Such models could enhance the accuracy of dementia risk assessment by capturing these underlying complex biophysiological pathways.
Conclusion
This study highlights the potential of physical frailty as a surrogate marker for cognitive decline and biomarkers, suggesting its utility in identifying individuals at increased risk. Associations were observed between frailty, lower cognitive performance and neuropathological plasma biomarkers such as Aβ42/Aβ40 ratio and NfL. These findings suggest clinical implications for using frailty not only as a screening tool but also as an intervention target to prevent or delay cognitive impairment. However, the findings should be interpreted with caution given the limitation of the use of secondary data, the cross-sectional design, and the small sample size nature of the study. Future research should employ longitudinal designs with larger and more diverse samples, and integrated predictive models combining physical frailty with neuropathological biomarkers should be developed to improve early detection and risk assessment for dementia.
Supplemental Material
Supplemental Material - Evaluating Physical Frailty as a Marker of Cognitive Health in Older Adults Without Dementia
Supplemental Material for Evaluating Physical Frailty as a Marker of Cognitive Health in Older Adults Without Dementia by Youngmin Cho, Jing Huang, Sofia Liu, Nada Lukkahatai, Jessica Gill, and Junxin Li in Journal of Geriatric Psychiatry and Neurology
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported from the National Institute of Nursing Research (R00NR016484).
Ethical Considerations
This study was approved by the Johns Hopkins University Institutional Review Board (IRB#00198497).
Consent to participate
All participants provided written informed consent before participating in the study.
Data Availability Statement
The data supporting this study are not publicly available due to privacy and ethical restrictions, as the dataset contains sensitive information from human participants
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
