Abstract
Introduction
Inflammatory processes and neurodegeneration are common features of Alzheimer's disease (AD). The systemic immune-inflammation index (SII), neutrophil-to-lymphocyte ratio (NLR), and platelet-to-lymphocyte ratio (PLR) are easily accessible, cost-effective prognostic indicators of inflammation status. We assessed the diagnostic value of SII and compared the efficacies of NLR, PLR, and SII in predicting AD severity.
Materials and Methods
Between January 2021 and December 2022, this prospective, cross-sectional clinical study included 175 patients with AD and 61 controls. SII, NLR, and PLR were calculated.
Results
Age, white blood cell and lymphocyte counts, and NLR and PLR values were significantly different between the patient and control groups (P <.05). Age, hemoglobin, neutrophil, NLR, and SII values were significantly different among dementia severity subgroups (P <.05). Compared with PLR and SII, NLR was more strongly correlated with dementia severity. In the analysis of adjusted data by gender, it was found that hemoglobin level is significantly negatively associated with dementia severity in males, while in females, age and PLR are significantly positively associated with dementia severity, and lymphocyte count and SII are negatively associated.
Conclusion
SII, a novel inflammatory marker, was associated with dementia severity in patients with AD. Compared with PLR and SII, NLR was more strongly correlated with dementia severity. In future studies with larger populations, SII and NLR values can be used to determine dementia severity and establish follow-up plans for patients with high dementia risk.
Keywords
Introduction
Alzheimer’s disease (AD) is the most common cause of dementia, responsible for up to 80% of all dementia diagnoses worldwide, and its prevalence increases with age. 1 β-amyloid plaque deposits and neurofibrillary tangles comprising hyperphosphorylated tau are the classic neuropathological hallmarks of AD. 1 These pathological markers are positively correlated with neuronal degeneration, neuroinflammation, microglia activation, blood–brain barrier dysfunction, and cognitive decline. 2
Neuroinflammation is a prevalent and key component of AD. Microglia, resident macrophage-like immune cells in the brain, play important roles in the inflammatory response and the progression of neurodegenerative disease. Previous studies have shown that the presence of activated microglia and astrocytes surrounding Aβ plaques is correlated with elevated levels of inflammatory cytokines, including interleukins 1 and 6, as well as tumor necrosis factor-α.3,4 Peripheral inflammatory changes during AD include neutrophil hyperactivation and increased lymphocyte migration into the central nervous system across the compromised blood–brain barrier. 5 Because inflammation plays an important role in multiple neurological conditions, including Parkinson’s disease, multiple sclerosis, and stroke, as well as various systemic diseases (eg, atherosclerosis, cancer, cardiovascular disease, and metabolic disorders), recent studies have focused on hematological biomarkers and indexes to determine the risk of inflammation.6-9
Epidemiological data suggest that neuroinflammation is an independent predictor of early death in AD. 10 Thus, it is important to identify patients with high risks of morbidity and mortality. Although the core CSF biomarkers of neurodegeneration (T-tau, P-tau, and Aβ42), CSF NFL, and plasma T-tau are strongly associated with Alzheimer’s disease, 11 they are not very useful markers. However, the neutrophil-to-lymphocyte ratio (NLR), monocyte-to-lymphocyte ratio (MLR), platelet-to-lymphocyte ratio (PLR), and systemic immune-inflammation index (SII) are useful, cost-effective, and commonly used indicators of peripheral systemic inflammation.12-16 Several studies have shown that NLR and PLR are prognostic indicators of cognitive impairment and disease progression in AD.12,17,18
The SII, a novel, cost-effective inflammation marker calculated from hemogram parameters (platelet count × neutrophil count/lymphocyte count) indicates simultaneous immune and inflammatory status; it is a more accurate indicator of overall inflammatory status, compared with NLR and PLR. 15 Furthermore, the SII can predict severity and prognosis in conditions such as stroke, cardiovascular diseases, atherosclerotic diseases, and cancers.6-9,12,14,17
Alzheimer’s disease is highly prevalent in rural areas. However, in these regions, the disease is either not diagnosed or diagnosed only based on cognitive tests. This is due to the lack of resources for amyloid beta or tau pathology testing. Therefore, simple blood tests and more accessible peripheral markers for the diagnosis of Alzheimer’s disease are needed in these areas.
However, the role of SII in AD is not fully understood. Here, we assessed the role of SII in AD pathophysiology and explored the ability of SII to predict disease severity (compared with NLR or PLR) in patients with AD.
Materials and Methods
Study Design
This prospective, cross-sectional, clinical study included all patients consecutively admitted to the neurology outpatient clinic of the Inonu University Faculty of Medicine between January 2021 and December 2022. The study protocol was approved by the Ethics Committee of Inonu University Faculty of Medicine (protocol number: 2022/32), and the study was conducted in accordance with the Declaration of Helsinki. All patients and control participants provided written informed consent after receiving a detailed explanation of the study.
Participants and Eligibility Criteria
Patients who had symptoms consistent with dementia underwent detailed assessments including clinical history, neuropsychological tests including the Mini-Mental Status Examination, and neuroradiological imaging (cranial magnetic resonance imaging or cranial computed tomography). All participants underwent a neurological examination. Alzheimer’s-type dementia was diagnosed in accordance with the criteria of the American Society of Neurology, Language Disorder and Stroke-Alzheimer’s Disease and Related Diseases Working Group. 19 To ensure that the study included patients who only had AD, we excluded patients with secondary dementia and patients in whom SII processes may have played a role, including patients with a history of ischemic or hemorrhagic stroke, possible disease that may affect laboratory results (leukemia, lymphoma, or thrombocytopenia); active infection (pneumonia supported by imaging examination, urinary tract infection by urine examination, chronic active viral hepatitis, etc.) or systemic disease including malignant tumor, uremia, liver cirrhosis, chronic heart, disease, and chronic lung disease. autoimmune or chronic inflammatory disease, organ failure (kidney, liver, lung), endocrine system diseases that may affect routine blood indicators, treatment with immunomodulatory drugs or chronic use of anti-inflammatory drugs, as well as patients with incomplete diagnostic examinations. We also excluded patients with thyroid disease, vitamin B12 deficiency, and acute/chronic psychiatric disease because the corresponding symptoms may be confused with AD.
The control group consisted of healthy older adults with normal cognitive status based on Mini-Mental Status Examination scores. 20 The controls were frequency-matched to patients with AD on the basis of age, sex, education level, and comorbidities. AD severity was classified as mild, moderate, or severe according to neuropsychological test scores and Clinical Dementia Rating scale scores. 21 Patients with a CDR score of 1 or higher were included in the study. A CDR score of 1 is defined as Mild Dementia, 2 as Moderate Dementia, and 3 as Severe Dementia. We are aware that there is a deficiency in our study regarding the definitive diagnosis of Alzheimer’s diseases. But we could not diagnose our patients with cerebrospinal fluid (CSF) biomarkers like amyloid β 42 or hyperphosphorylated tau. Because these tests are performed in only one labarotory in our country and the accessibility of the tests is very difficult.
Biochemical and Hematological Blood Sample Analysis
Fasting peripheral blood samples were collected from all patients. Hematological parameters, including levels of hemoglobin and platelets, as well as counts of white blood cells (WBCs), neutrophils, lymphocytes, and platelets, were measured using an automated hematology analyzer system (Sysmex Corporation, Kobe, Japan) with kits (Sysmex Corporation) that utilized a fully automatic nephelometric method. Absolute cell counts were used in subsequent analyses. The SII was calculated as platelet count × neutrophil count/lymphocyte count. The NLR and PLR were also calculated.
Statistical Analysis
Statistical analyses were performed using SPSS for Windows version 26.0 (IBM Corp., Armonk, NY, USA). All statistical tests were two-tailed and P-values < .05 were assumed to indicate statistical significance. The normality of continuous variables was verified using the Shapiro–Wilk test (n < 50) and Kolmogorov-Smirnov test (n ≥ 50). Differences between patients with AD and healthy controls were assessed using the Mann–Whitney U test for independent non-normally distributed variables and independent t-tests for normally distributed variables. Dementia severity subgroups were compared using the Kruskal–Wallis test or analysis of variance, depending on data distribution normality. Receiver operating characteristic (ROC) curves were used to determine optimal cutoff values for NLR and SII in terms of predicting disease severity among patients with AD. Spearman’s correlation analysis was used to determine correlations among NLR, PLR, and SII values. The comparison of variables between the dementia severity was conducted using analysis of variance (linear regression), adjusting for age and sex.
Results
Baseline Characteristics
The study included 175 patients (92 men and 83 women) with a mean age of 75.31 ± 9.62 years, (minimum-maximum, 49-102 years) who met all AD diagnostic criteria. AD dementia severity was classified as mild in 61 (35.8%), moderate in 57 (32.6%), and severe in 57 (32.6%) patients. The control group consisted of 61 healthy people (31 men and 30 women) with a mean age of 73.87 ± 7.49 years (minimum-maximum, 60-98 years).
Comparison of Demographic and Hematological Parameters
Demographic and Biochemical Variables According to AD.
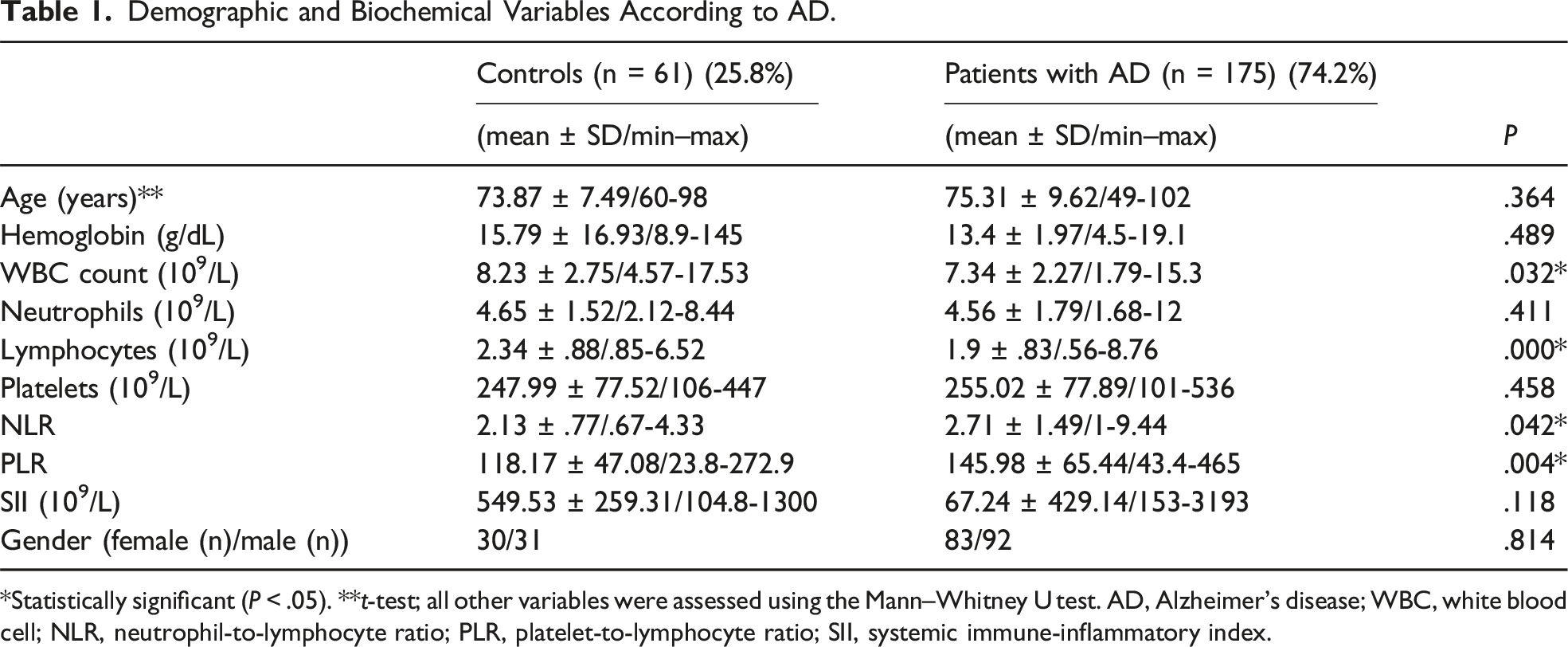
*Statistically significant (P < .05). **t-test; all other variables were assessed using the Mann–Whitney U test. AD, Alzheimer's disease; WBC, white blood cell; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SII, systemic immune-inflammatory index.
Biochemical Variables According to Dementia Severity (Mean ± Standard Deviation/Minimum–Maximum).
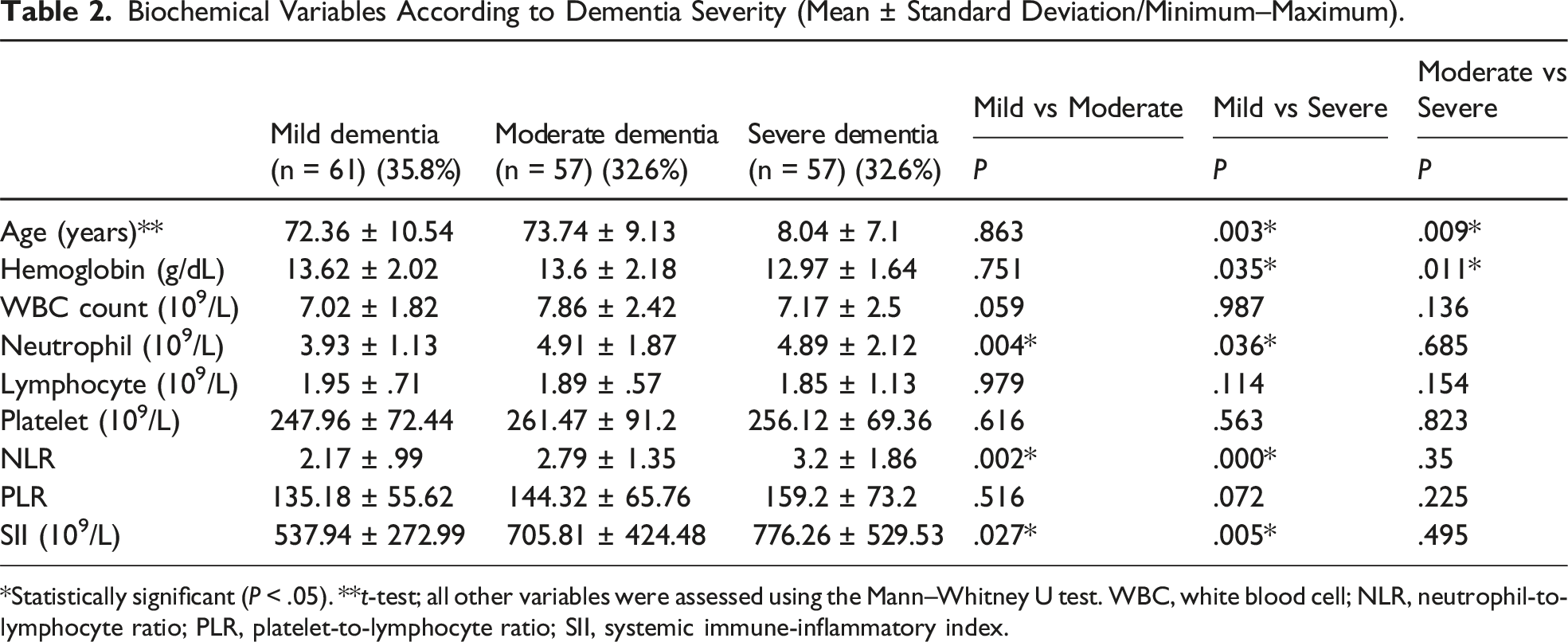
*Statistically significant (P < .05). **t-test; all other variables were assessed using the Mann–Whitney U test. WBC, white blood cell; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SII, systemic immune-inflammatory index.
Biochemical Variables According to Gender (Mean ± Standard Deviation/Minimum–Maximum).
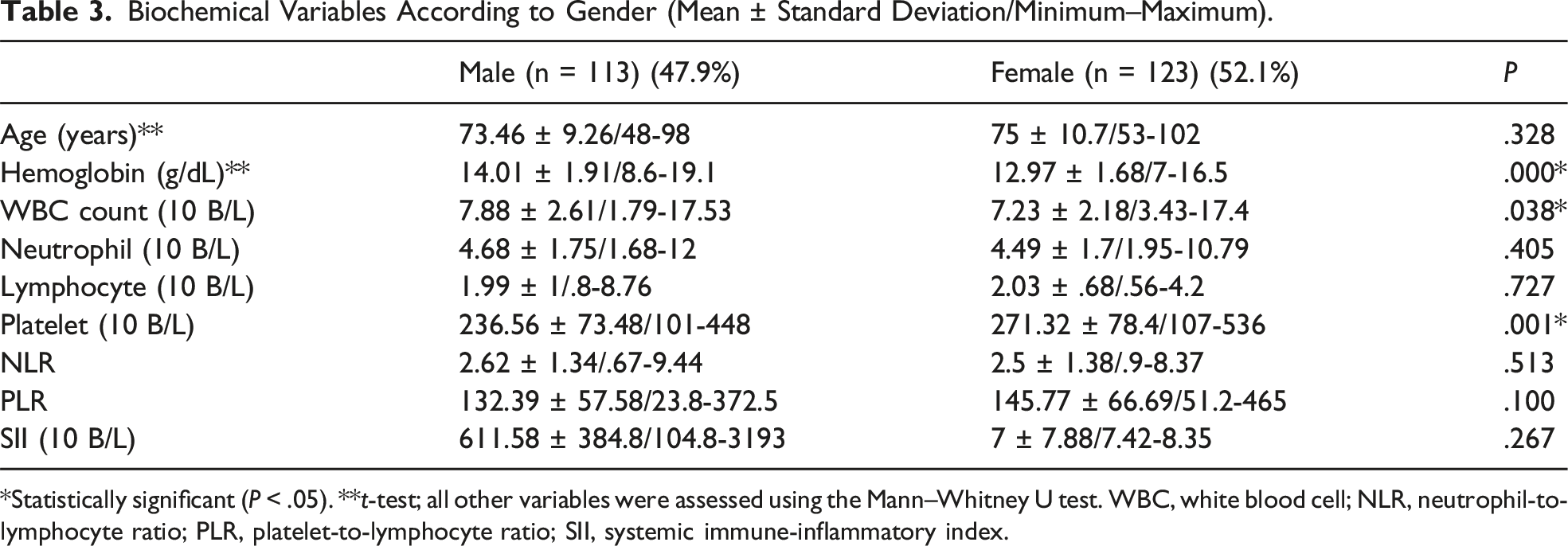
*Statistically significant (P < .05). **t-test; all other variables were assessed using the Mann–Whitney U test. WBC, white blood cell; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SII, systemic immune-inflammatory index.
Association Between Biochemical Variables and Dementia Severity, Adjusted for Gender.
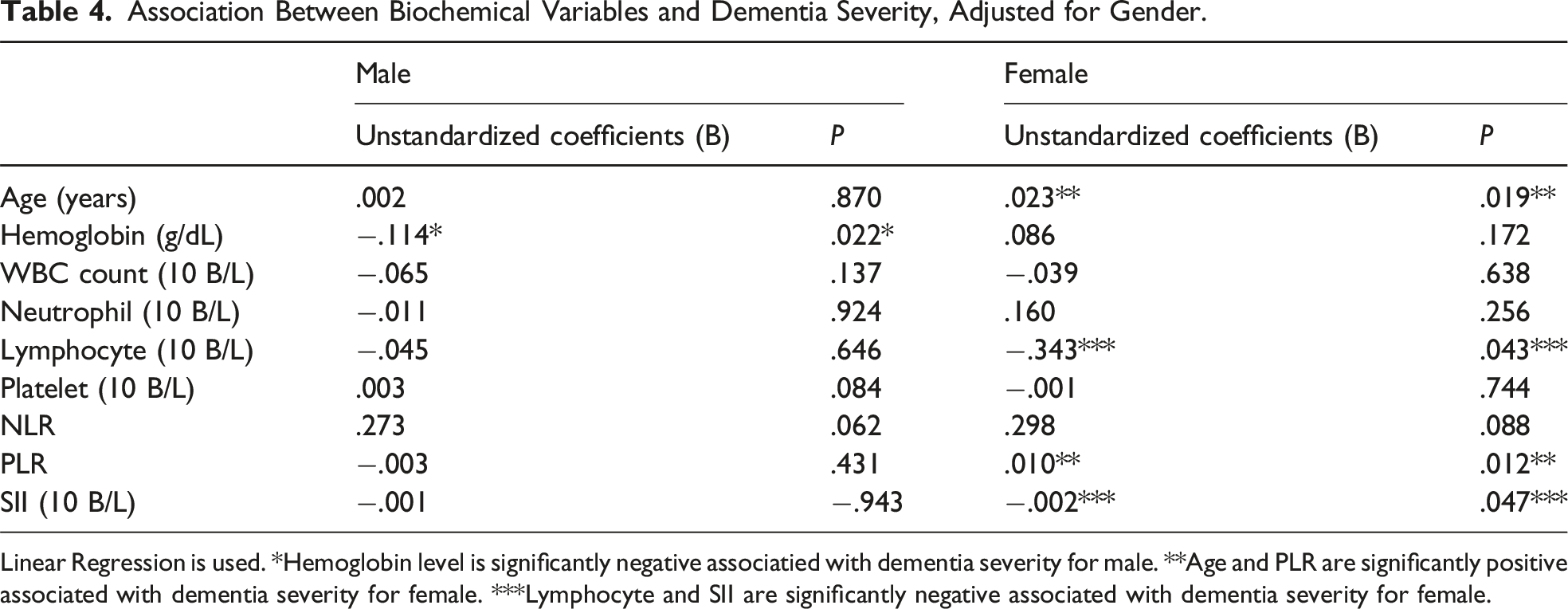
Linear Regression is used. *Hemoglobin level is significantly negative associatied with dementia severity for male. **Age and PLR are significantly positive associated with dementia severity for female. ***Lymphocyte and SII are significantly negative associated with dementia severity for female.
Association Between Biochemical Variables and Dementia Severity, Adjusted for Age.
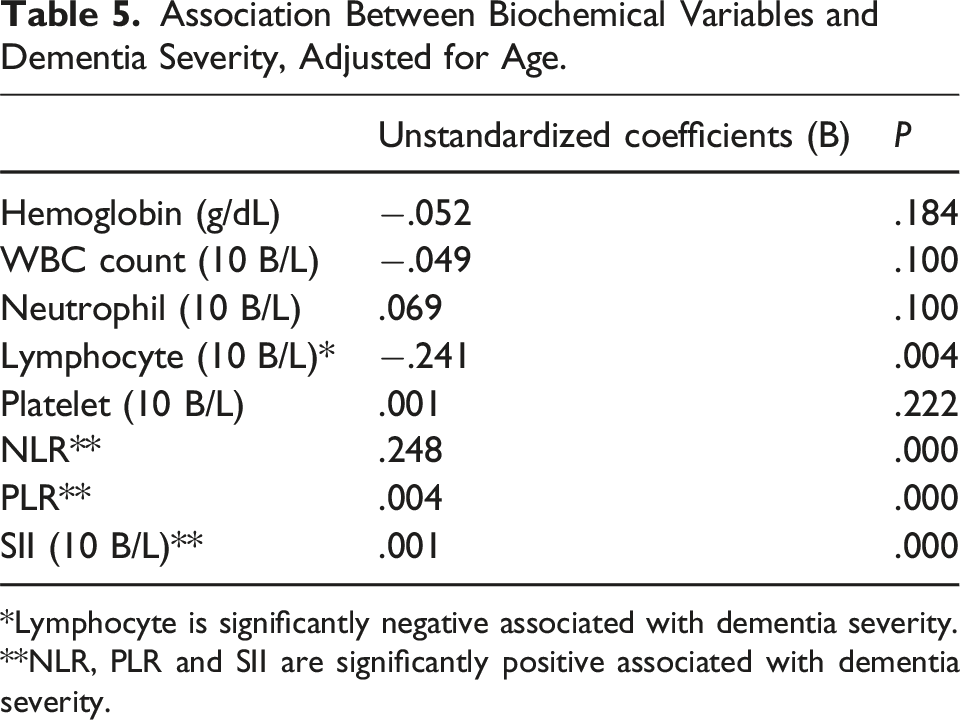
*Lymphocyte is significantly negative associated with dementia severity. **NLR, PLR and SII are significantly positive associated with dementia severity.
Correlations of NLR. PLR. and SII with Dementia Severity.

*Statistically wsignificant (P < .05). NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SII, systemic immune-inflammatory index.
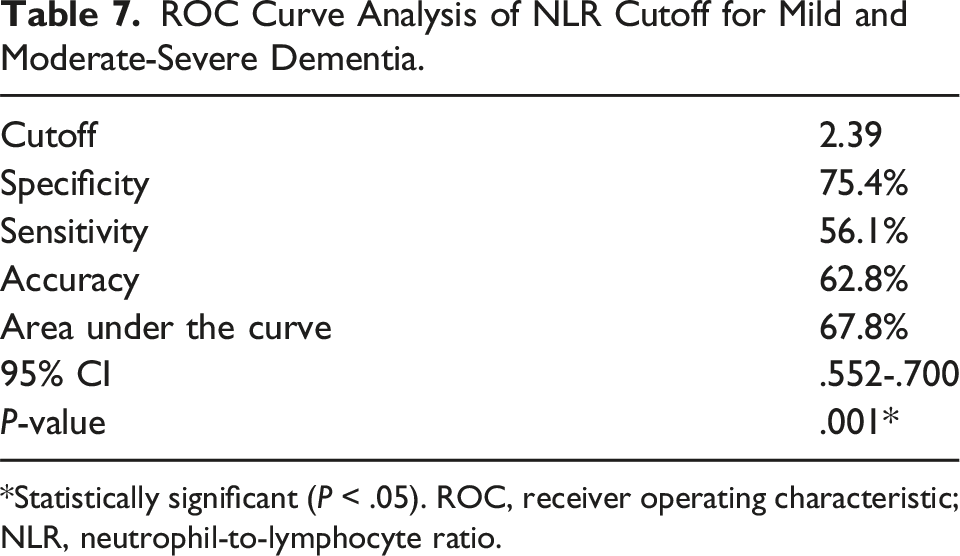
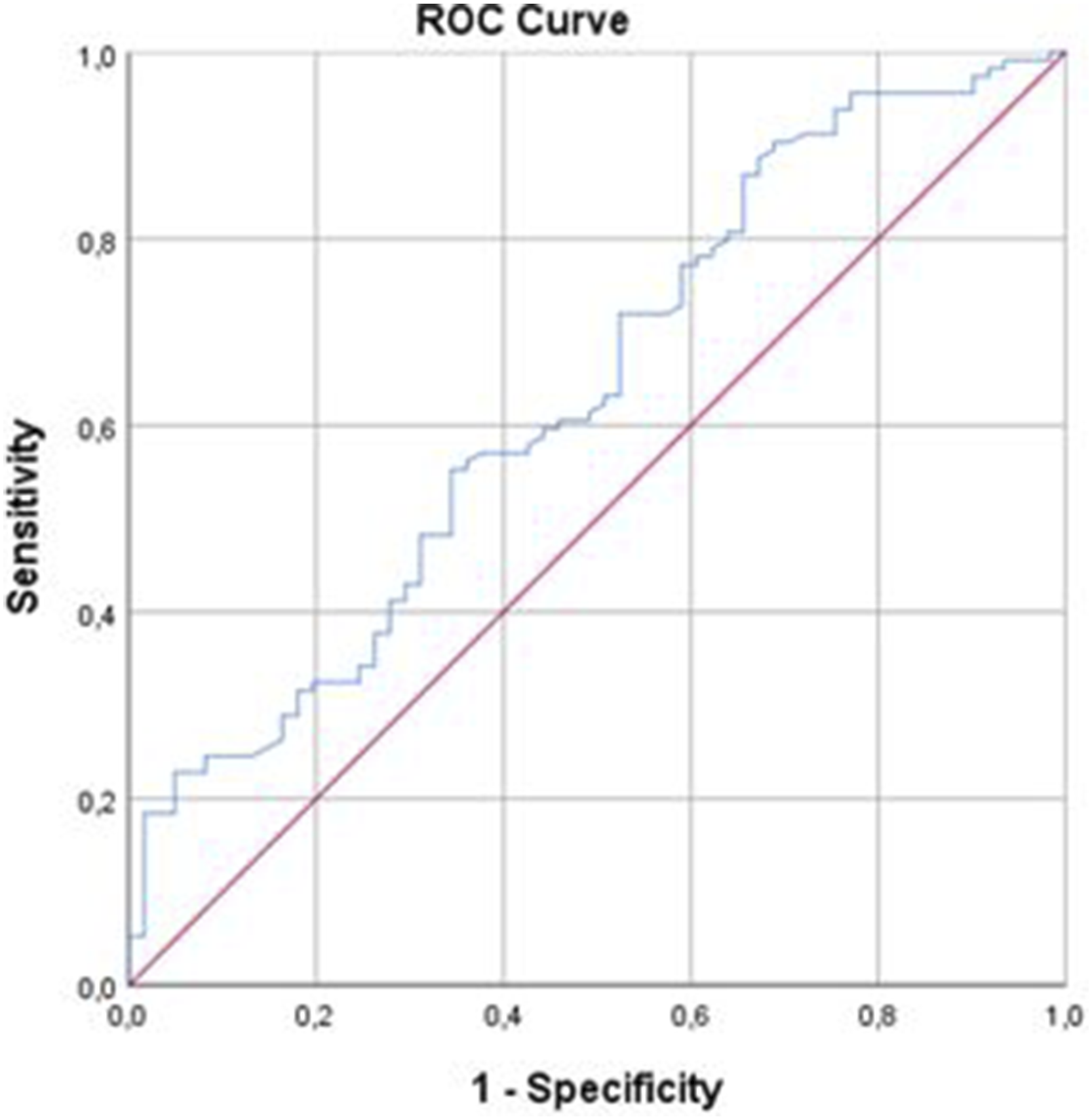
ROC curve analysis and the Youden index were used to determine the optimal serum NLR for distinguishing mild dementia from moderate and severe types of dementia. The area under the ROC curve for NLR was 71.90 (95% confidence interval [CI], .552-.700; Figure 1). ROC curve analysis for moderate-severe dementia revealed that the optimal NLR cutoff value was 2.89 with sensitivity, specificity, and accuracy values of 56.1%, 75.4%, and 62.8%, respectively (Table 7). Furthermore, ROC curve analysis and the Youden index were used to determine the SII value that distinguished mild dementia from moderate and severe types of dementia. The area under the ROC curve for SII was 63.4 (95% CI, .623-.764; Figure 2). ROC curve analysis for moderate-severe dementia showed that the optimal SII cutoff value was 335.15 with sensitivity, specificity, and accuracy values of 90.4%, 31.1%, and 69.7%, respectively (Table 8). ROC curve analysis of NLR to predict mild and moderate-severe dementia. ROC: receiver operating characteristic; NLR: neutrophil-to-lymphocyte ratio. ROC Curve Analysis of NLR Cutoff for Mild and Moderate-Severe Dementia. *Statistically significant (P < .05). ROC, receiver operating characteristic; NLR, neutrophil-to-lymphocyte ratio. ROC curve analysis of SII to predict mild and moderate-severe dementia. ROC: receiver operating characteristic; SII: systemic immune-inflammation index. ROC Curve Analysis of SII Cutoff for Mild and Moderate-Severe Dementia. *Statistically significant (P < .05). ROC, receiver operating characteristic; SII, systemic immune-inflammation index.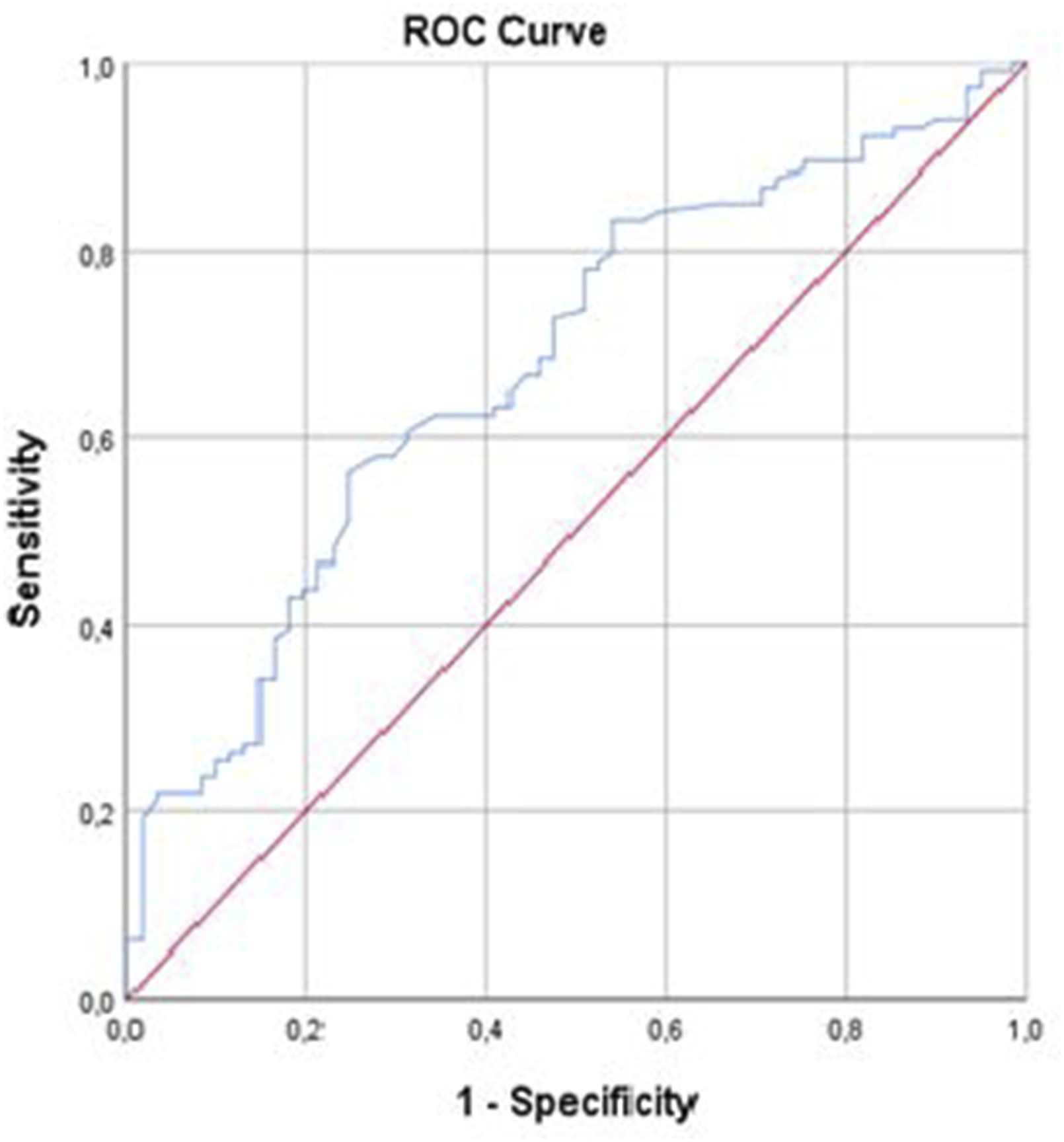

Discussion
We found that SII and NLR values were significantly lower in patients with mild dementia than in patients with moderate or severe dementia. Furthermore, compared with PLR and SII, NLR was a better predictor of dementia. To our knowledge, this study is one of the first to investigate the prognostic values of inflammatory markers in patients with dementia.
Inflammatory processes and neurodegeneration are common features of AD. 22 Serum NLR and PLR,13,23-25 as well as SII, 26 are easily accessible, cost-effective prognostic indicators of inflammation status. The NLR integrates information from two distinct leukocyte subtypes, thereby avoiding the use of misleading information from a single leukocyte subtype, which may be affected by infection or dehydration; thus, it has greater clinical significance than other independent inflammatory biomarkers. 27 The NLR, derived from a widely available complete blood count laboratory test, 18 has been studied as a prognostic factor in diseases such as coronary artery disease,28,29 cancer,30,31 and inflammatory diseases. 32 Kuyumcu et al. 33 reported that the NLR was higher in patients with AD than in controls. By measuring NLR and neocortical amyloid load in patients with AD, Rembach et al. 34 found that the NLR was significantly elevated and was correlated with neocortical amyloid load. In another study, the NLR was significantly elevated among patients with AD and patients with mild cognitive impairment. 17 Another study revealed that the NLR was elevated in patients with AD and mild cognitive impairment, whereas the PLR was not significantly different. 13 Li et al. 26 found that the NLR, but not the PLR, was significantly elevated in patients aged > 60 years who had cognitive impairment. Moreover, individuals with an elevated NLR had greater risk of dementia during a 5.9-year follow-up period. Our findings are consistent with previous reports of an association between the NLR and AD. We also found that the NLR significantly increased with increasing dementia severity. In contrast to previous studies, we found that the PLR significantly differed between AD and control groups. The most notable finding in the present study was that the NLR could more reliably predict dementia severity, compared with PLR and SII, suggesting that the platelet count does not provide additional prognostic information.
It is well known that the primary function of platelets is related to the coagulation system and hemostasis. However, they also play a role in inflammatory processes and the proliferation of megakaryocytes, which results in an increase in platelet count. Chemokines, cytokines, and procoagulant molecules released from platelets activated by inflammation are reported to play a role in the immune response as acute phase reactants. Dağıstan et al, Yeşil et al, Chen et al, and Koç et al have shown that an increase in mean platelet volume (MPV) is associated with Alzheimer’s disease (AD),35-38 while Dong et al 17 have found it to be associated with mild cognitive impairment (MCI). Additionally, Liang et al and Dong et al have demonstrated that a decrease in platelet distribution volume (PDV) is significant for AD.17,39 In our study, consistent with the literature, we found that the platelet-lymphocyte ratio (PLR) significantly differed between the AD and control groups. Another result obtained in our study, which differs from other studies in the literature, is that, when the correlation analyzes of the markers are examined, the neutrophil-lymphocyte ratio (NLR) and systemic immune-inflammation index (SII) values were more correlated with dementia levels compared to PLR.
Sex differences in dysregulation of glial cell-mediated neuro-immune responses have been implicated in AD 40 It has been revealed that Alzheimer’s disease is more common in women and the severity of the disease is more severe. 41 Women usually have stronger immune responses to stimulations than men involving different pathways and immune cells, correlating to higher susceptibility to infections in male. In a preclinical study where experimental AD was induced in male and female mice, it was demonstrated that female mice exhibited increased expression of pro-inflammatory cytokines and an enhanced microglial response. 42 In our study, we found that in female patients, age and PLR are significantly positively associated with dementia severity, while lymphocyte count and SII are negatively associated. In contrast, no association was found between peripheral immune system markers and dementia in male patients. These results suggest that, as highlighted in the literature, the differences in immune responses between males and females may contribute to these findings, with immune responses being more pronounced in females compared to males.
Several previous studies have utilized ROC curve analysis to determine diagnostic accuracy for AD. Kuyumcu et al. 33 reported sensitivity of 70% and specificity of 80% for AD diagnosis, whereas another study revealed that the specificity and sensitivity for AD diagnosis were 54% and 83%, respectively. 17 In the present study, ROC curve analysis showed that the sensitivity and specificity of NLR for mild and moderate-severe types of dementia were 56.1% and 75.4%, respectively. Although previous studies utilized ROC curve analyses to distinguish patients with dementia and control participants, we believe that our ROC curve analysis of dementia severity provides novel information.
The SII is an easily measurable, inexpensive novel inflammatory index that can be calculated using the neutrophil, platelet, and lymphocyte counts obtained from routine peripheral blood tests. This biomarker was first proposed by Hu et al. 43 in 2014 to predict the prognosis of hepatocellular carcinoma. A previous study revealed a significant correlation between increased carotid intima-media thickness and SII value in patients with hypertension 15 ; another study showed a positive correlation between SII value and poor prognosis in patients with coronary artery disease. 44 Furthermore, the SII is correlated with disease activity in patients with ankylosing spondylitis and ulcerative colitis.14,45 Li et al. 26 reported that the SII was significantly higher in patients aged > 60 years who had cognitive impairment. In the present study, the SII did not significantly differ between the dementia and control groups; however, SII values differed among dementia severity subgroups. We found that the SII had a diagnostic accuracy of 69.7% for dementia severity. Although this diagnostic accuracy is not high, we found no previous investigations related to SII and diagnostic accuracy for dementia severity; therefore, we present our findings as a contribution to the literature.
Our study had several limitations including the small number of cases, the single-center and cross-sectional study design, and the inability to exclude other factors potentially involved in inflammation. Our study has a cross-sectional design, therefore future studies with longitudinal design are needed to confirm the findings of study.
In conclusion, SII was associated with dementia severity in patients with AD; however, compared with PLR and SII, NLR was more strongly correlated with dementia severity. In future studies with larger populations, SII and NLR values can be used to determine dementia severity and establish follow-up plans for patients with high dementia risk. Moreover, changes in neutrophil and lymphocyte ratios may constitute specific markers of dementia severity.
Supplemental Material
Supplemental Material - Increased Systemic Immune-Inflammation Index as a Novel Indicator of Alzheimer’s Disease Severity
Supplemental Material for Increased Systemic Immune-Inflammation Index as a Novel Indicator of Alzheimer’s Disease Severity by Fatma E. Algul and Yuksel Kaplan in Journal of Geriatric Psychiatry and Neurology
Footnotes
Author Contributions
F.E.A., Y.K.: conceptualized the study. F.E.A.: analyzed the data. Y.K.: inferential statistics; language editing; provided expert advice in clinical pharmacology. F.E.A., Y.K.: provided expert advice inpsychiatry and psychopharmacology. F.E.A.: interpreted the study results, drafted the firstversion of the manuscript, created the tables and figures. F.E.A., Y.K.: assisted with the preparationof the manuscript. Y.K.: supervised the project. All authors: commented on previous versions of the manuscript; read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
