Abstract
Background:
Breastfeeding among lactating people with opioid use disorder taking buprenorphine monotherapy is generally accepted, as low concentrations of buprenorphine and metabolites in human milk have been well-established. The use of buprenorphine-naloxone for pregnant and lactating people with opioid use disorder is expanding and there is no information available regarding the concentrations of naloxone and their metabolites in human milk to recommend the use of this combination medication during lactation
Research Aims:
To determine the concentrations of buprenorphine and naloxone and their primary metabolites in human milk, maternal plasma, and infant plasma, among lactating buprenorphine-naloxone maintained people and their infants.
Methods:
Four lactating buprenorphine-naloxone maintained people provided plasma and human milk samples on Days 2, 3, 4, 14, and 30 postpartum. Infant plasma was obtained on Day 14.
Results:
Concentrations of buprenorphine, norbuprenorphine and their glucuronide metabolites were present in maternal plasma and human milk at low concentrations, consistent with previous research in lactating buprenorphine monotherapy participants. Naloxone was not detected, or was detected at concentrations below the limit of quantification, in maternal plasma and in all except one human milk sample at Day 30. Naloxone was not detected or detected at concentrations below the limit of quantification in all infant plasma samples.
Conclusion:
Results support the use of buprenorphine-naloxone by lactating people who meet appropriate criteria for breastfeeding.
Keywords
Key Messages
Despite the combination drug buprenorphine-naloxone’s potential use in the treatment of opioid use disorder in pregnant and postpartum people, safety during lactation has not been established due to the antagonist naloxone component.
Concentrations of buprenorphine and metabolites in human milk, maternal plasma, and infant plasma were low, consistent with previous research.
Concentrations of naloxone in maternal human milk and plasma and infant plasma were generally undetectable.
Results of this small study lend support for the use of buprenorphine-naloxone during breastfeeding.
Background
The persistent concerns related to maternal opioid use disorder (OUD) and effects on the developing fetus and infant have resulted in calls for new and diverse treatment methods. Previously, pregnant and lactating people could receive only methadone for treatment of OUD, but research has revealed generally positive maternal and infant outcomes with buprenorphine monotherapy during pregnancy (Jones et al., 2010). This has resulted in more widespread use of this medication during pregnancy in the United States and internationally. More recently, treatment with buprenorphine-naloxone during pregnancy has become more widely used. This combination product has the potential advantages of acting as a deterrent to injection use and misuse/diversion due to the naloxone component, which, as a potent opioid antagonist when injected but not when taken orally or sublingually, could precipitate withdrawal. Currently it has been indicated that buprenorphine-naloxone is a safe and effective treatment for OUD in pregnant people (Link et al., 2020; Ordean & Tubman-Broeren, 2023). Patient education regarding choice of medication options, to include the effects of available medications on the infant via intrauterine and/or human milk exposure, is important (Substance Abuse and Mental Health Services Administration [SAMHSA], 2018).
Guidelines for breastfeeding in people with substance use disorder, including those on medication therapy, have been established (Reece-Stremtan & Marinelli, 2015). Previous research (Ilett et al., 2012; Jansson et al., 2016) has concluded that concentrations of buprenorphine, norbuprenorphine, and their glucuronide metabolites in maternal plasma and human milk, are very low. Such information is lacking for buprenorphine-naloxone, due to the lack of information regarding naloxone concentrations in human milk. LactMed (2023) reports that “No information is available on the excretion of naloxone into human milk. Because it is not orally bioavailable, it is unlikely to affect the breastfed infant.” Naloxone has no or limited systemic effects when administered sublingually due to its low bioavailability via this route, resulting in an oral bioavailability reported of 10% or less (Harris, 2000). The sublingual bioavailability of naloxone was estimated to be about 3% in the first U.S. Food and Drug Administration (FDA)-approved buprenorphine/naloxone formulation (U.S. FDA/Center for Drug Evaluation and Research, 2002). Given the expanding use of buprenorphine-naloxone and the well-established benefits of human milk and breastfeeding, the purpose of this study was to delineate concentrations of naloxone and its glucuronide component, as well as buprenorphine, norbuprenorphine and their metabolites in human milk, maternal plasma, and infant plasma.
Methods
Research Design
This study was carried out as part of a larger study evaluating the effects of prenatal buprenorphine-naloxone maternal therapy on fetal and maternal physiology and infant outcome. Participants who expressed a desire to breastfeed their infant and met criteria to do so were invited to participate in a secondary lactation study via confidential interview close to the time of delivery. The study was overseen by the university institutional review board (JHUIRB-2; approval number IRB00093171, approved 6/14/2016). Participants received compensation for their involvement in this study.
Setting
Study participants were people attending treatment at an urban comprehensive treatment facility for pregnant and postpartum people with substance use disorder. The treatment facility combined obstetric care with substance use disorder treatment, with a co-located pediatric clinic. Births occurred in an adjacent hospital located on campus. The center treats, in general, people supported by medical assistance funding who face socio-environmental difficulties such as housing instability, lack of education, and unemployment. Breastfeeding is not common in this population for myriad reasons, although breastfeeding is recommended for all women meeting criteria to do so at the center. Lactation support is provided after delivery in the adjacent hospital as well as in the affiliated pediatric clinic for all breastfeeding people.
Sample
Five participants provided consent for procedures and submitted samples. All samples for one participant were lost due to research sample storage unit error. Samples from four participants and their infants made up the final sample.
Study participants received buprenorphine-naloxone sublingual filmstrips in a 4:1 ratio (Suboxone®), 8 mg buprenorphine/2 mg naloxone or 2 mg buprenorphine/0.5 mg naloxone from the time in their pregnancy they entered SUD treatment through the end of study enrollment. To accurately assess exposure, buprenorphine-naloxone dose was abstracted from daily clinical dosing records for each study participant during study enrollment.
Measurement
Maternal demographics were collected via confidential interview as part of the parent study. To assess concentrations of naloxone and its glucuronide component along with concentrations of buprenorphine, norbuprenorphine, and their metabolites, study participants submitted human milk and plasma samples at the time of peak buprenorphine component medication levels (i.e., 2–3 hr after sublingual dose) on Days 2, 3, 4, 14, and 30 postpartum. Plasma was obtained from infants via a heelstick coincident with a routine blood test (PKU screening) on Day 14.
Human milk samples were obtained in one expression via a breast pump (either electric or hand pump) by the research nurse. Milk was obtained at the time of peak maternal medication levels, that is, 2.5 hr after sublingual dosing. Five cc of human milk was collected per expression.
Data Collection
Samples included in this study were collected between January and August, 2019. All participants were provided with information about this sub-study in a confidential interview, and all provided written informed consent for participation. Confidentiality as people in OUD treatment and members of a study was strictly maintained; files were de-identified and kept in locked cabinets in a locked laboratory, and biosamples were similarly de-identified and stored. All study biosamples were obtained by the research registered nurse (RN). All biosamples (maternal plasma, maternal human milk, and infant plasma) were stored in a secure research facility freezer located on campus.
Data Analysis
All human milk and plasma specimens were shipped on dry ice to the analyzing laboratory and stored at −20 °C until analysis. Samples were analyzed using an analytical scheme adapted from Swortwood et al. (2016) with minor modifications. Briefly, proteins were precipitated with acetonitrile prior to solid-phase extraction using Phenomenex Strata-X-C 33 µm Polymeric Strong Cation (60 mg/3 ml) columns (Torrance, CA), that were washed with deionized water, 0.1 M acetic acid, and methanol, before elution with a 70:26:4 (v/v/v) dichloromethane:isopropanol:ammonium hydroxide mixture. The samples were dried down under nitrogen at 35 °C, and reconstituted with 125 μl mobile phase (85:15, A: B, v/v). A volume of 40 μl was injected into the LC-MS/MS. Analysis was performed on an Agilent 1290 Infinity Liquid Chromatograph coupled to an Agilent 6470 Triple Quadrupole Mass spectrometer (Santa Clara, CA, USA). Chromatographic separation was achieved using a Restek Raptor Biphenyl (2.7 μm, 100 × 2.1 mm) column (Bellefonte, PA) with a matching Restek Raptor Biphenyl column guard (5 μm, 5 × 2.1 mm). Mobile Phase A was 0.1% formic acid in water, while mobile Phase B was 0.1% formic acid in methanol. The total run-time per sample was 8 min. The mass spectrometer utilized electrospray ionization operated in positive ionization mode. For identification, multiple reaction monitoring (MRM) was used to monitor two transitions per analyte and internal standard. Data analysis was performed using the Agilent MassHunter Workstation software. This method was fully validated in accordance with national standards and assessed for linearity, accuracy, precision, specificity, sensitivity, and dilution integrity and all acceptability criteria were met. The limit of detection (LOD) was found to be 0.5 ng/ml for the active metabolites of buprenorphine-naloxone: buprenorphine, buprenorphine-glucuronide, norbuprenorphine, norbuprenorphine-glucuronide, naloxone, and 1.25 ng/ml for naloxone-glucuronide. The limit of quantification was determined to be 0.5 ng/ml for buprenorphine, buprenorphine-glucuronide, norbuprenorphine, norbuprenorphine-glucuronide, 1.25 ng/ml for naloxone, and 2.5 ng/ml for naloxone-glucuronide. For reporting, signals lower than the LOD were considered “not detectable” (ND), while signals greater than LOD but lower than LOQ were considered “< LOQ.”
Results
Characteristics of the Participants
Three buprenorphine-naloxone maintained participants provided complete samples on Days 2, 3, 4, 14, and 30; one participant provided samples only until Day 4 due to treatment program transfer. Maternal participant’s ages ranged from 23 to 35 years, with 12 to 13 years of education. Gestational age upon treatment entry ranged from 22 to 28 weeks. Length of substance use prior to pregnancy ranged from 1 to 5 years. Maternal buprenorphine/naloxone doses ranged from 12 mg/3 mg to 20 mg/5 mg (mean [
One participant exclusively breastfed their infant and the remainder supplemented with formula. All infant participants were born at term in the 39th week of gestation with average Apgar scores of 7.25 and 9.00 at 1 and 5 minutes respectively. Two infant participants required pharmacologic treatment with oral morphine for neonatal abstinence syndrome (NAS) one for 5 days of oral morphine (for a total of 0.94 mg) for mild expression, and one for 25 days for moderate expression receiving a total of 18.62 mg of morphine. The infant participant treated for mild NAS expression was born to the maternal participant who exclusively breastfed and was on the lowest medication dose (12 mg/3 mg); the mother of the infant treated for moderate NAS expression was on the second highest medication dose (16 mg/4 mg). No maternal or infant participants had positive urine toxicology screening for any substances other than buprenorphine at the time of delivery. Plasma was obtained from three infant participants for analysis.
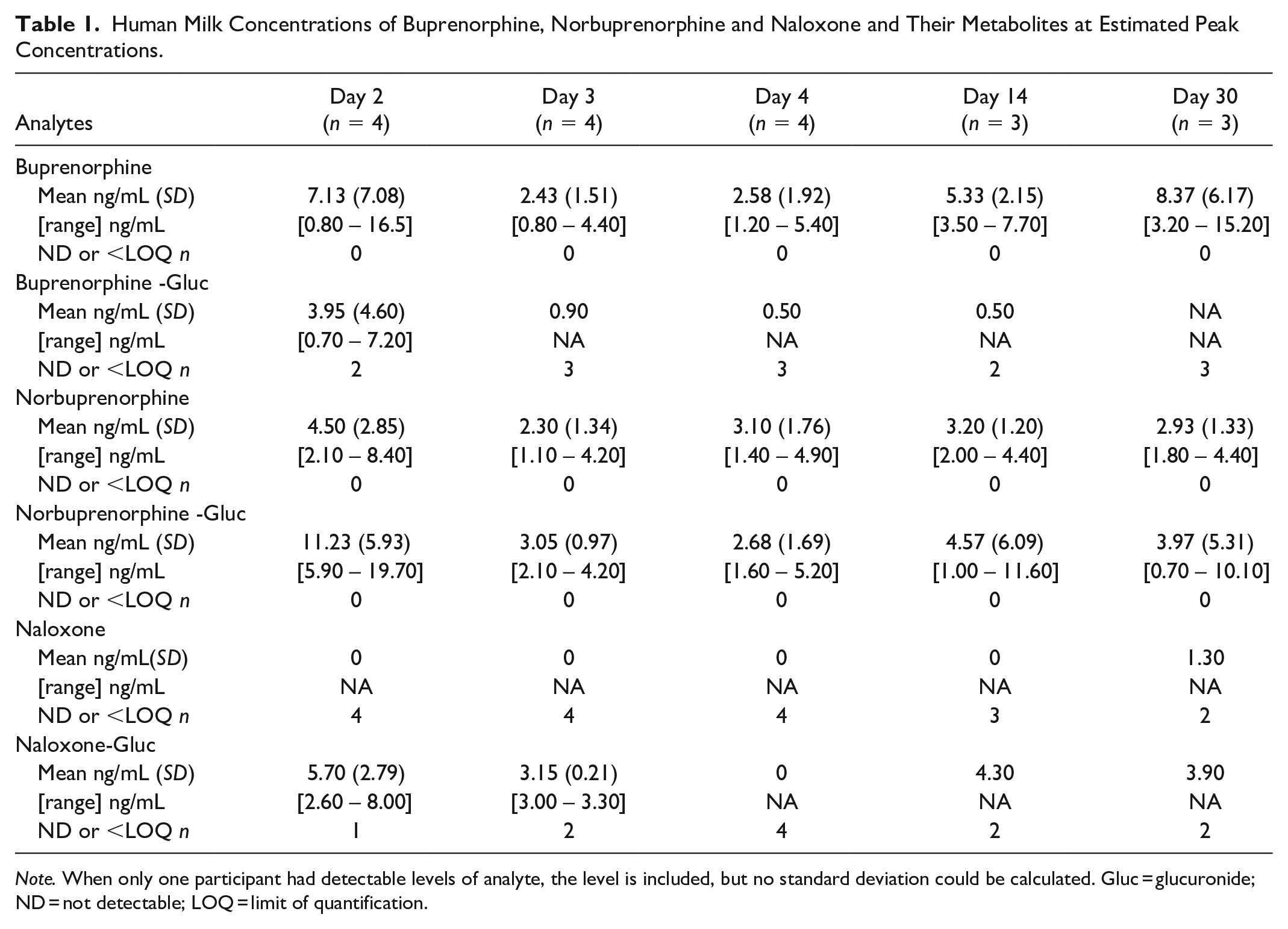
Human Milk Concentrations of Buprenorphine, Norbuprenorphine, and Naloxone
Concentrations of buprenorphine, norbuprenorphine, and naloxone in human milk are described in Table 1. Buprenorphine and norbuprenorphine were detected in all human milk samples as was norbuprenorphine-glucuronide; buprenorphine-glucuronide was variably detected in only three participants on some days. Naloxone was detected in one human milk sample on Day 30; however, its metabolite, naloxone-glucuronide, was variably detectable in some maternal participants (in one participant on Days 2 and 3, in a second maternal participant on Days 2, 14, and 30 and in one participant on Day 3).
Human Milk Concentrations of Buprenorphine, Norbuprenorphine and Naloxone and Their Metabolites at Estimated Peak Concentrations.
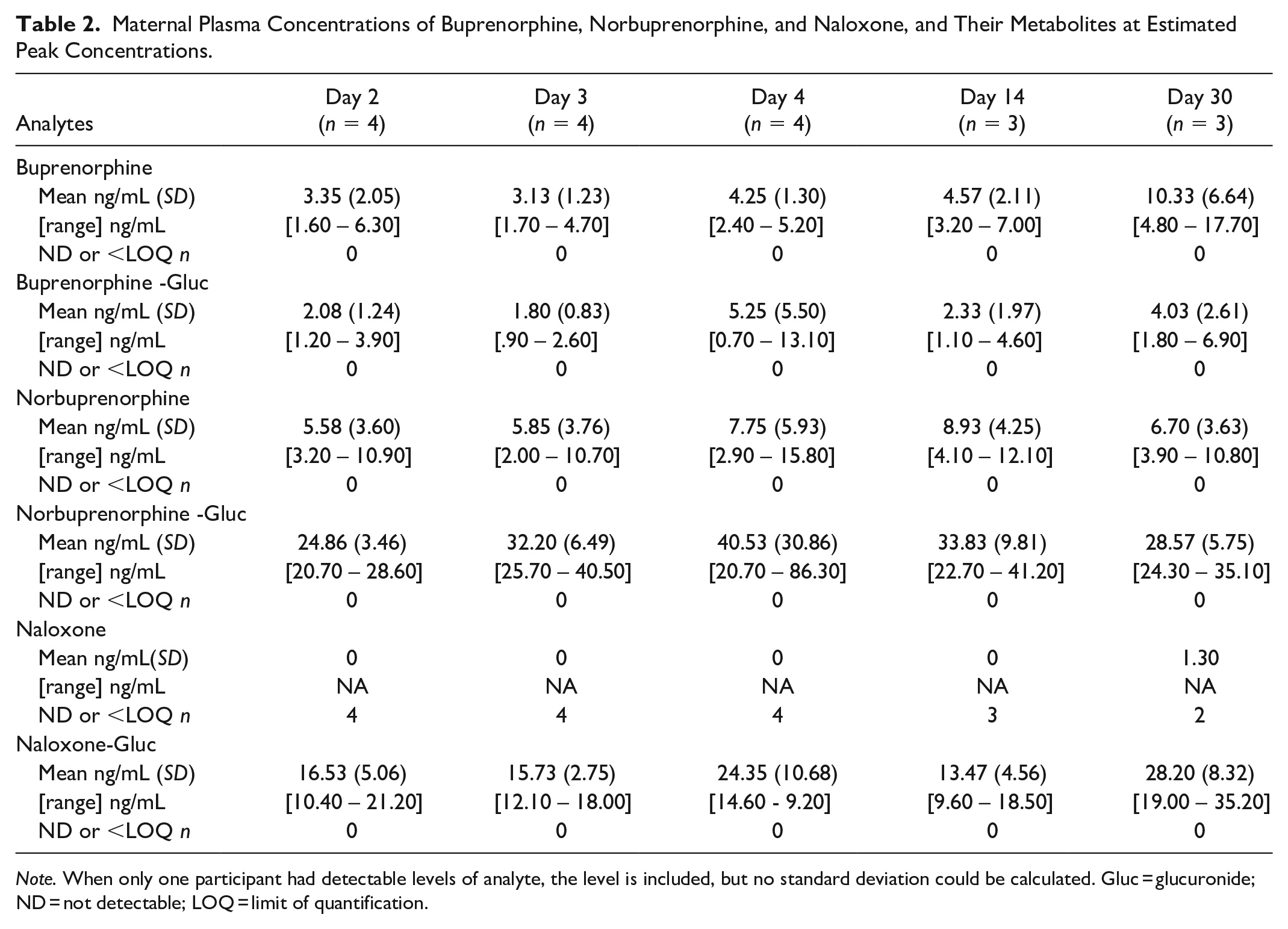
Maternal Plasma Concentrations of Bprenorphine, Norbuprenorphine, and Naloxone
Maternal plasma concentrations of buprenorphine, norbuprenorphine, and naloxone are presented in Table 2. Buprenorphine, buprenorphine-glucuronide, norbuprenorphine, and norbuprenorphine-glucuronide were detected in all samples. Similar to findings for human milk, naloxone was not detected or detected at concentrations below limits of quantification in all plasma samples. Naloxone-glucuronide was detected in all maternal plasma samples.
Maternal Plasma Concentrations of Buprenorphine, Norbuprenorphine, and Naloxone, and Their Metabolites at Estimated Peak Concentrations.
Infant Plasma Concentrations of Buprenorphine, Norbuprenorphine, and Naloxone
Concentrations of buprenorphine, buprenorphine-glucuronide, norbuprenorphine-glucuronide, naloxone, and naloxone-glucuronide were either not detected or detected at concentrations below the limit of quantification in all infant plasma samples at Day 14. Two infant participants had detectable concentrations of norbuprenorphine, 0.9 and 5.4 ng/ml, in plasma at Day 14.
Discussion
Buprenorphine-naloxone treatment has become standard care for many people being treated for OUD, including pregnant people (Ordean & Tubman-Broeren, 2023). Both buprenorphine and naloxone are metabolized by the liver primarily by glucuronide conjugation (Dahan et al., 2010). Both are poorly bioavailable when taken orally, and both buprenorphine and buprenorphine-naloxone are given sublingually as OUD treatment medications. Buprenorphine, a partial opioid agonist, allays withdrawal symptoms and cravings in opioid using people, and has a “ceiling effect,” essentially plateauing effects at higher doses (Dahan et al., 2006). Taking more medication once this effect is reached will not produce more euphoria, respiratory depression, or sedation. Naloxone is an opioid antagonist inhibiting all pharmacological effects of opioids. When taken orally it is mostly metabolized by first pass through the liver resulting in poor bioavailability (Harris et al., 2000), therefore not affecting the effectiveness of buprenorphine treatment in the combination product. When injected, it produces withdrawal in opioid-dependent people due to its high affinity for opioid receptors, thereby providing a deterrent to injection use. The combination medication therefore is an effective treatment agent while having features that enhance safety and efficacy—reduced capacity for overdose and diversion/misuse—making it particularly attractive for treatment in pregnant individuals. However, there is little data regarding the effects of buprenorphine-naloxone on the developing fetus and infant, with most reports comparing buprenorphine to buprenorphine-naloxone exposure during pregnancy and generally finding similar or reduced rates of NAS (Mullins et al., 2020) and normal birth parameters (Debelak et al., 2013; Lund et al., 2013). To our knowledge, there is no information regarding buprenorphine-naloxone and human milk/breastfeeding. Concerns exist regarding the potential for the naloxone component expressed in human milk and transmitted to the infant to interfere with opioid agonist (i.e., morphine or methadone) treatment of NAS, despite the fact that naloxone, given orally, is poorly bioavailable and unlikely to affect the infant. However, it is possible that these concerns affect clinical decision-making for prenatally opioid-exposed infants.
Concentrations of buprenorphine and its major metabolites (norbuprenorphine, buprenorphine-glucuronide, and norbuprenorphine-glucuronide) are low in human milk and maternal plasma (Ilett et al., 2012; Jansson et al., 2016) in lactating people maintained on buprenorphine monotherapy. Among infant participants who were breastfed by buprenorphine monotherapy maintained people, plasma concentrations were low or undetectable (Jansson et al., 2016). Among this small sample of buprenorphine-naloxone maintained lactating people and their infants, concentrations of buprenorphine and its major metabolites were similarly low. Naloxone was either not detected or detected at concentrations below the limit of quantification in all maternal plasma samples and all but one human milk sample (one participant at 30 days had a concentration of 1.3 ng/ml, just above the limit of quantification of 1.25 ng/ml). Naloxone-glucuronide, the inactive metabolite, was variably detected in human milk but detected in all maternal plasma samples.
Two of the four infant participants required pharmacotherapy for NAS, one for mild expression, and one for moderate. The mother of the infant participant with moderate NAS was the mother with the barely detectable naloxone concentration in human milk at Day 30. The mother received a buprenorphine-naloxone dose of 16 mg/4 mg. We have no ready explanation for this finding, but it is unlikely to be of clinical significance as this result was obtained at a time after the infant had completed treatment for NAS and was discharged home on Day 29 of life.
Limitations
Major limitations of this study include the small sample size and missing samples. Formula supplementation in unknown quantities occurred in three of the four participants. Further studies evaluating larger populations of lactating people, preferably solely human milk feeding their infants to more fully assess the maximal exposures of buprenorphine-naloxone to the breastfeeding infant, are needed.
Conclusion
Buprenorphine-naloxone has the potential to be a safe and effective treatment medication for this group. Currently, many lactating people are discouraged from the use of this medication due to lack of data. Results of this small sample lend support for the use of buprenorphine-naloxone by lactating people who meet appropriate criteria for breastfeeding.
Footnotes
Author Contributions
Disclosures and Conflicts of Interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors report no disclosures nor conflicts of interest. Munchelou Gomonit is a doctoral student of Madeleine Swortwood.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by NIH/NIDA RO1DA041367, awarded to the first author. Study medication was provided by Indivior, who had no role in study design, collection, analysis, and interpretation of data; in the writing of the manuscript; or in the decision to submit the manuscript for publication.
