Abstract
Background:
Mothers with preterm infants may need to express milk for considerable periods. Research to improve breast pump design has focused on compression stimuli, frequencies, and vacuums.
Objective:
This study aimed to compare the effectiveness of 2 electric pumps: Medela Symphony (pump S) and a novel pump (Philips AVENT Twin electronic pump; pump A). Both offer flexibility of rate and suction; pump A also incorporates petal compression cushions. Primary outcomes were (1) milk weight expressed during 10-day study period and (2) weight of milk expressed in a 15-minute test.
Methods:
Seventy-one mothers with preterm infants < 34 weeks were randomized. Mothers completed 10-day diaries including weight of milk expressed. Milk weight expressed during a single 15-minute test period and data on pumping mode, skin-to-skin contact, breastfeeding at infant discharge, and mothers’ opinions of the pump were recorded.
Results:
There was no significant difference in milk expressed during the first 10 days between groups. Pump S mothers expressed significantly more milk during a fixed 15-minute period. Mothers using pump A awarded higher scores for certain characteristics of the pump, notably location of control button and ease of use. Similar proportions of infants received breast milk at discharge, but pump A mothers were more likely to be directly breastfeeding (odds ratio, 4.27 [95% confidence interval, 1.29, 14.1]).
Conclusion:
The breast pumps showed similar effectiveness in terms of milk expression and maternal opinions. The finding that breast pump design may influence breastfeeding at infant discharge merits further investigation.
Keywords
Well Established
Mothers of preterm infants are strongly encouraged to express breast milk for their infant, and most neonatal intensive care units recommend double-pumping using an electric pump. Incorporating petal massage cushions into a manual pump has proven benefits for milk expression in this setting.
Newly Expressed
An electric pump incorporating massage cushions showed similar effectiveness to a standard hospital-grade electric pump for milk expression in the neonatal intensive care unit. Similar proportions of infants received breast milk at discharge, but mothers using the novel pump were more likely to be directly breastfeeding.
Background
The use of human milk is strongly recommended for preterm infants, 1,2 and an increasing number of mothers aim to provide breast milk for their infant. However, these infants are frequently too immature or sick to breastfeed and mothers may need to express milk for a considerable length of time. Breast pumps rely predominantly on suction to extract milk, which may be unphysiological and is sometimes unpleasant for mothers. 3 In recent years, research to improve pump design has focused on the use of compression stimuli 3,4 and different frequency and vacuum patterns. 5 -7 The Medela Symphony pump (pump S; Medela AG, Baar, Switzerland) has an initial “let-down” mode with rapid low suction (120/minute, vacuum -50 to -200 mmHg), followed by “expression mode” (45-78/minute, vacuum -50 to -250 mmHg) with slower rate and deeper suction. The duration of the let-down mode can be altered by the mother and the vacuum strength altered at any time. The Philips AVENT Twin electronic breast pump (pump A; Philips Consumer Lifestyle, Amsterdam, The Netherlands) incorporates a petal massage cushion in the breast shield, designed to massage the areola and surrounding breast during pumping, 3 in an electronic pump that offers flexibility of rate and suction (vacuum range, 0 to -250 mmHg), with the rate/suction strength control button positioned on the breast shield to allow greater ease of control.
The primary objective of this randomized controlled trial was to compare the effectiveness of pump A and pump S in the neonatal intensive care unit (NICU) setting where mothers are expressing milk for their preterm infants.
Methods
The study was conducted in 2 UK NICUs. Mothers were eligible if they delivered their infant(s) < 34 weeks gestational age (including twin and singleton deliveries) and planned to express breast milk. Infant(s) were younger than 72 hours old at randomization and were expected to stay in the NICU for at least 10 days; mothers who delivered at other hospitals but were transferred to a study unit were eligible if recruited by 72 hours postpartum. The study had ethics approval from the UCL Institute of Child Health ethics committee and was registered at www.ClinicalTrials.gov (NCT00887991).
The research nurse approached the mother with information about the study, before delivery where possible but otherwise as soon as was possible postpartum. Following written informed consent, mothers were randomized to use 1 of the study pumps for a 10-day study period; randomization was stratified by the infant’s gestation (≤ 28 weeks, and 29-33 weeks) and by parity. 4,8,9 Randomization schedules (permuted blocks of randomized length) were prepared by a member of the study team who was not involved in practical aspects of the study, and assignments were held in sealed opaque envelopes. Following randomization, mothers were given verbal and written information (Appendix 1 and 2, available online) and help with expressing breast milk by the staff of the NICU or postnatal ward with additional help from the research nurses, who had specific experience in advising on breastfeeding in the NICU setting and who also provided specific instruction on the optimal use of the assigned breast pump. At 1 hospital, manual expression was used during the first 48 hours before introducing a breast pump, while at the other site, mothers started using a breast pump immediately after delivery. Pump S was the standard pump in both NICUs and was therefore used prior to study entry. Breast pumps were located in a designated room in the NICUs but pumps could also be used at the infant’s bedside and were available for home use if a mother was discharged home. After the initial 10-day study period, mothers were encouraged to continue expressing milk using their allocated pump until their infant was discharged.
Primary outcome measures were total weight of milk expressed during the initial study period (to day 10); total weight of milk expressed in a single fixed 15-minute pumping session between 3 and 10 days (physiological test); and the time to first appearance of milk and time taken to express a fixed weight of milk (20 g, 40 g, 60 g) during this test.
Secondary outcome measures were total number of pumping sessions and total time spent expressing milk in the study period; mother’s opinion of the assigned pump; total volume of maternal breast milk expressed and consumed by the infant while in the NICU; number of days taken for the infant to achieve full enteral feeds (150 mL/kg/day); and whether or not the mother was breastfeeding her infant(s) at discharge.
Data Collection
For the first 10 study days, mothers were provided with a diary and asked to record the following every time they used their pump: the weight of milk expressed using scales (CS200E, 200 g × 0.1 g; Ohaus Corporation, Parsippany, New Jersey, USA) provided for this purpose, the time they started and stopped expressing, whether single or double pumping was used, and episodes of kangaroo care and/or the infant being put to the breast. Diaries were checked by the research nurses on a regular basis so that omissions or inconsistencies could be resolved at the time. On day 10, mothers completed a maternal perception questionnaire about their assigned pump, consisting of a series of Likert-type scales, from 1 (very good) to 7 (very bad) with 4 being neutral, on the following parameters: comfort, ease of assembly, ease of use, level of suction, noise level, flexibility regarding the rate and amount of suction, location of control button, speed of milk flow, and overall opinion of pump. Scores were recoded into 3 categories for analysis due to small numbers in some categories; recoding was done in 2 ways, both preserving the symmetrical nature of the data ([a] 1-3 = 1, 4 = 2, 5-7 = 3 and [b] 1-2 = 1, 3-5 = 2, 6-7 = 3). At infant discharge, research nurses collected data on intake of maternal breast milk from the medical charts. Data on volumes of milk expressed (estimated by mothers using bottle markings), duration of expression, and whether the mother breastfed her infant(s) were obtained from diary cards completed by mothers after the initial 10-day study period until the infant was discharged; the research nurses examined these diaries when the infant was discharged but not on a regular basis in the intervening period.
Physiological Study
Between days 3 and 10 (ideally days 5-7) postpartum, each mother was asked to express milk for a single fixed 15-minute period using her assigned breast pump. This measurement was arranged at a time convenient to the mother and research nurse but did not include the first milk expression of the day. Milk was collected and weighed at 1-minute intervals (using portable scale model CS200E, 200 g × 0.1 g) to determine the total weight of milk, the time to the first appearance of milk, and the time taken to produce specific milk weights. All expressed milk was available for consumption by the infant.
Statistics
Sample size
The planned sample size was based on data from our previous randomized controlled trial in preterm mothers comparing a novel hand pump and an electric pump, in which 20% of mothers produced ≤ 150 mL of breast milk per day on average in the first 10 days. We calculated that 76 mothers per group would allow us to detect a reduction in this proportion from 20% to 5%, and we added an additional 15% to this number to allow for dropouts. The total planned sample size was therefore 176 subjects.
Analysis using SPSS version 18 was conducted on an intention-to-treat basis, including data from all mothers who were randomized into the study, regardless of whether they completed the protocol using their assigned breast pump. Group comparisons were made for continuous and categorical variables, respectively, by Student’s t test (or corresponding nonparametric tests for skewed data) and chi-square test, with subsequent regression analysis, allowing control for potential confounders where appropriate.
Results
Subjects
Recruitment was significantly slower than anticipated, due in part to changes in practice in 1 NICU that had a higher-than-expected transfer rate. We were therefore unable within the allocated study period to reach the planned sample size and 71 mothers were randomized (36 pump A and 35 pump S).
Progress of subjects through the study is illustrated in Figure 1. The number of subjects with complete 10-day milk diary data was 33 (92%) versus 29 (83%) for pump A and pump S, respectively, with discharge data available for 30 (83%) versus 25 (74%) subjects. Four infants died (2 pump A, 2 pump S), 7 subjects withdrew before infant discharge (3 pump A, 4 pump S), and 5 subjects (1 pump A, 4 pump S) were lost due to the infant being transferred to another hospital. Eight infants died or the subject withdrew during the first 10 days (3 pump A, 5 pump S); milk diary data for these subjects were included in the primary intention-to-treat analysis where available up to the time of death or withdrawal. There were no significant differences between randomized groups in baseline maternal or infant characteristics (Table 1).
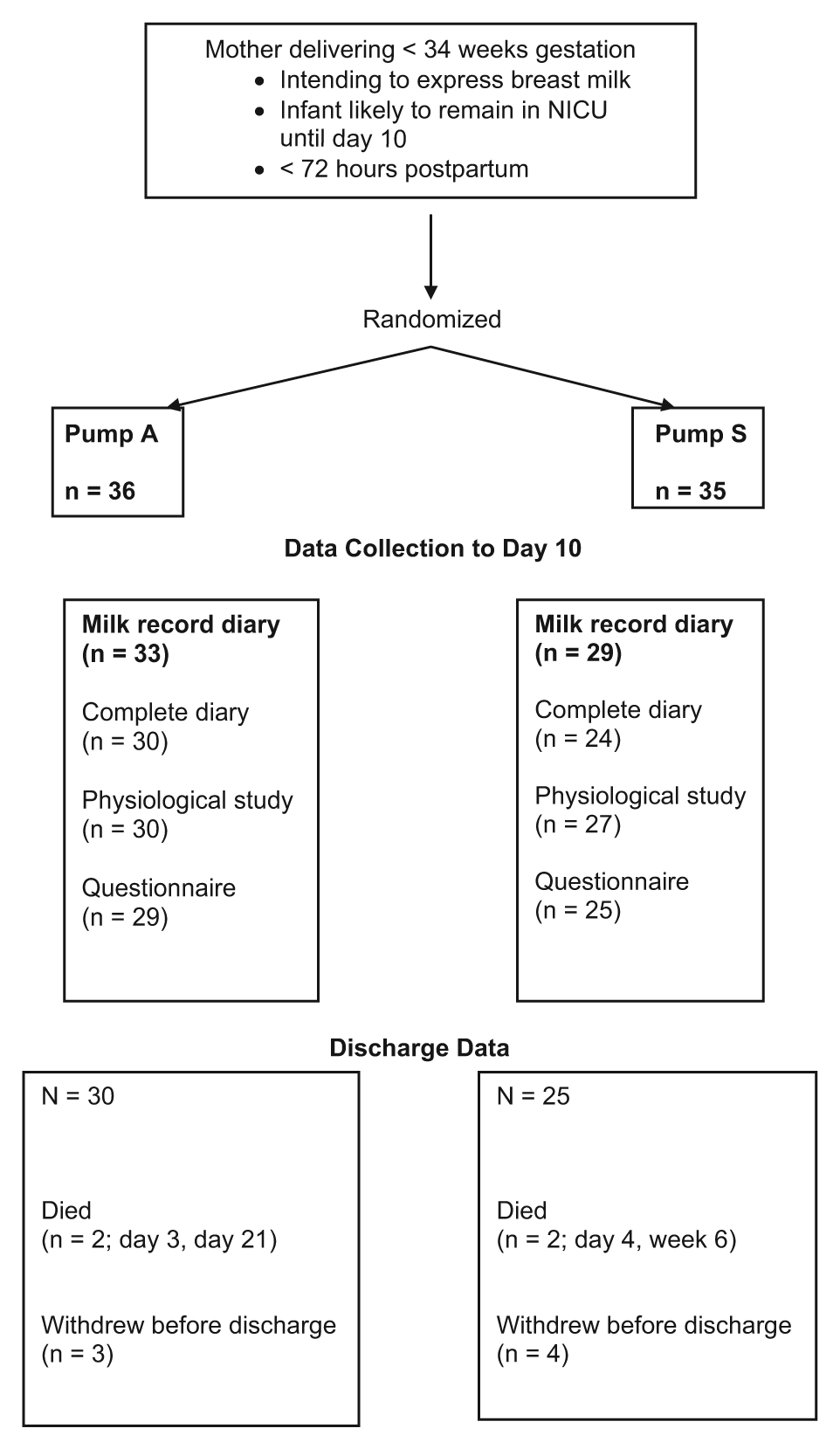
Flow Chart of Subject Progress through Study
Baseline Characteristics According to Randomized Group
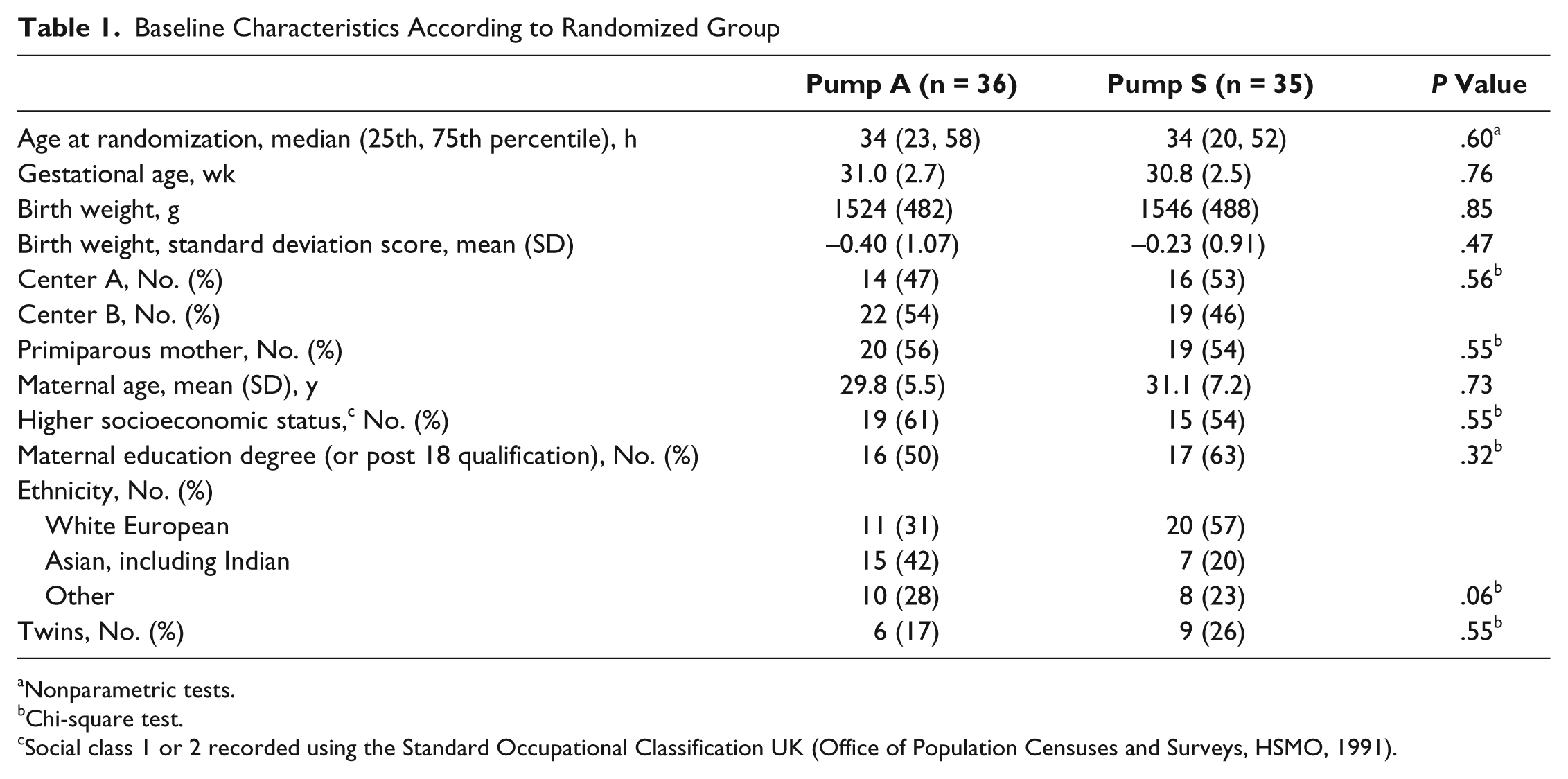
Nonparametric tests.
Chi-square test.
Social class 1 or 2 recorded using the Standard Occupational Classification UK (Office of Population Censuses and Surveys, HSMO, 1991).
Primary Outcomes
Milk expressed during initial 10-day study period
There was no significant difference in the total weight of milk expressed (Table 2) between randomized groups. The results were unchanged when data from the 8 subjects who had withdrawn from the study or whose infant had died before day 10 were excluded from the analysis (data not shown). The number of mothers achieving a milk weight of at least 500 g/day by day 10 was 11 (31%) versus 9 (26%) for pump A and pump S, respectively, with 20 (61%) versus 17 (59%) expressing a mean of 150 g/day during the 10-day study period. The weight of milk expressed was significantly higher in subjects who predominantly double-pumped a total (median [25th, 75th percentiles]) weight of milk over 10 days of 2238 (1070, 3477) g versus 1210 (560, 2294) g (P = .02), an average daily milk expression of 288 (168, 386) g versus 147 (78, 275) g (P = .04). This effect was seen regardless of the pump used, with no significant difference in the weight of milk expressed between primiparous and multiparous mothers per pumping episode: 175 (78, 332) g versus 198 (99, 294) g, respectively (P = .9).
Results from Mothers’ 10-Day Diaries
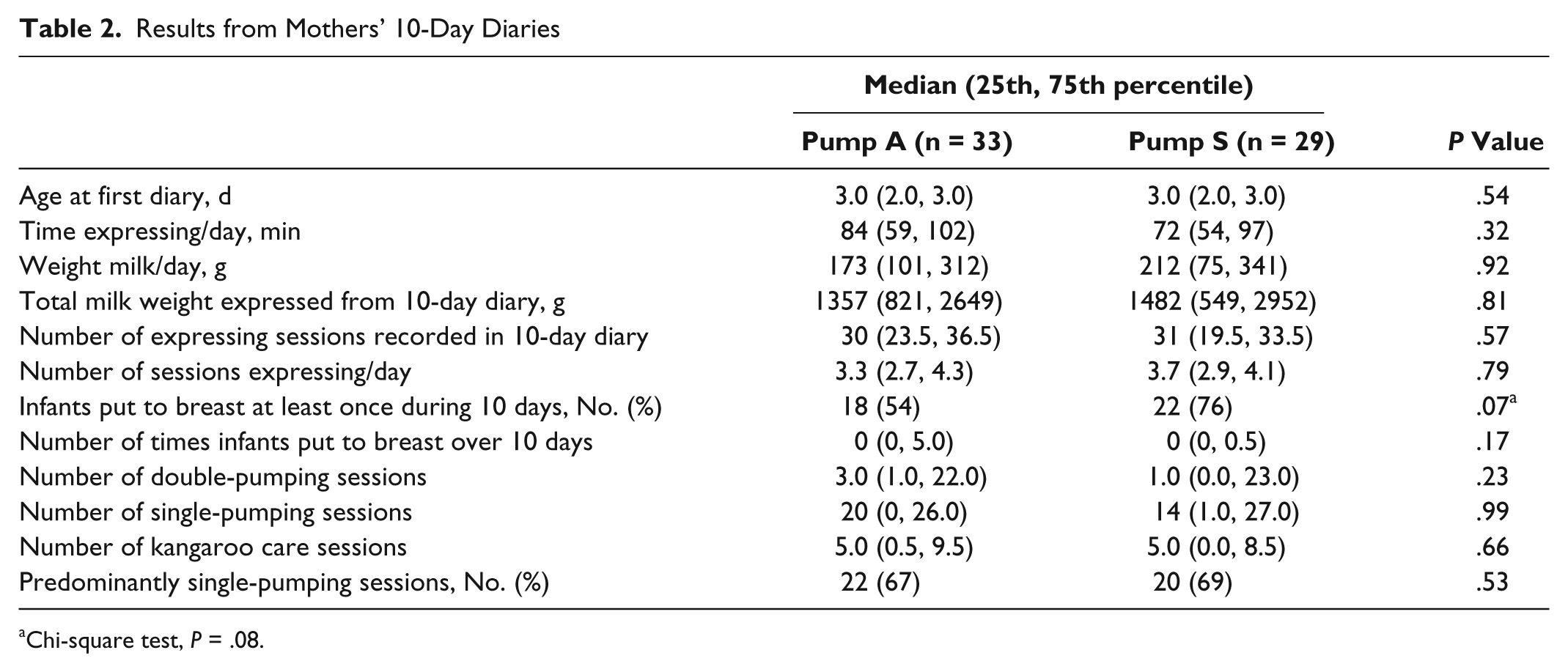
Chi-square test, P = .08.
Physiological measurements
The total weight of milk expressed over the 15-minute test period was significantly greater for mothers using pump S (Table 3). There was a nonsignificant trend for target weights (10 g, 20 g, 40 g) to be reached more quickly by mothers using pump S in comparison with pump A, with a significant difference for the first appearance of milk and for 5 g.
Weight of Milk Expressed by Mothers over a Fixed 15-Minute Period Using Double-Pumping with Their Assigned Breast Pump
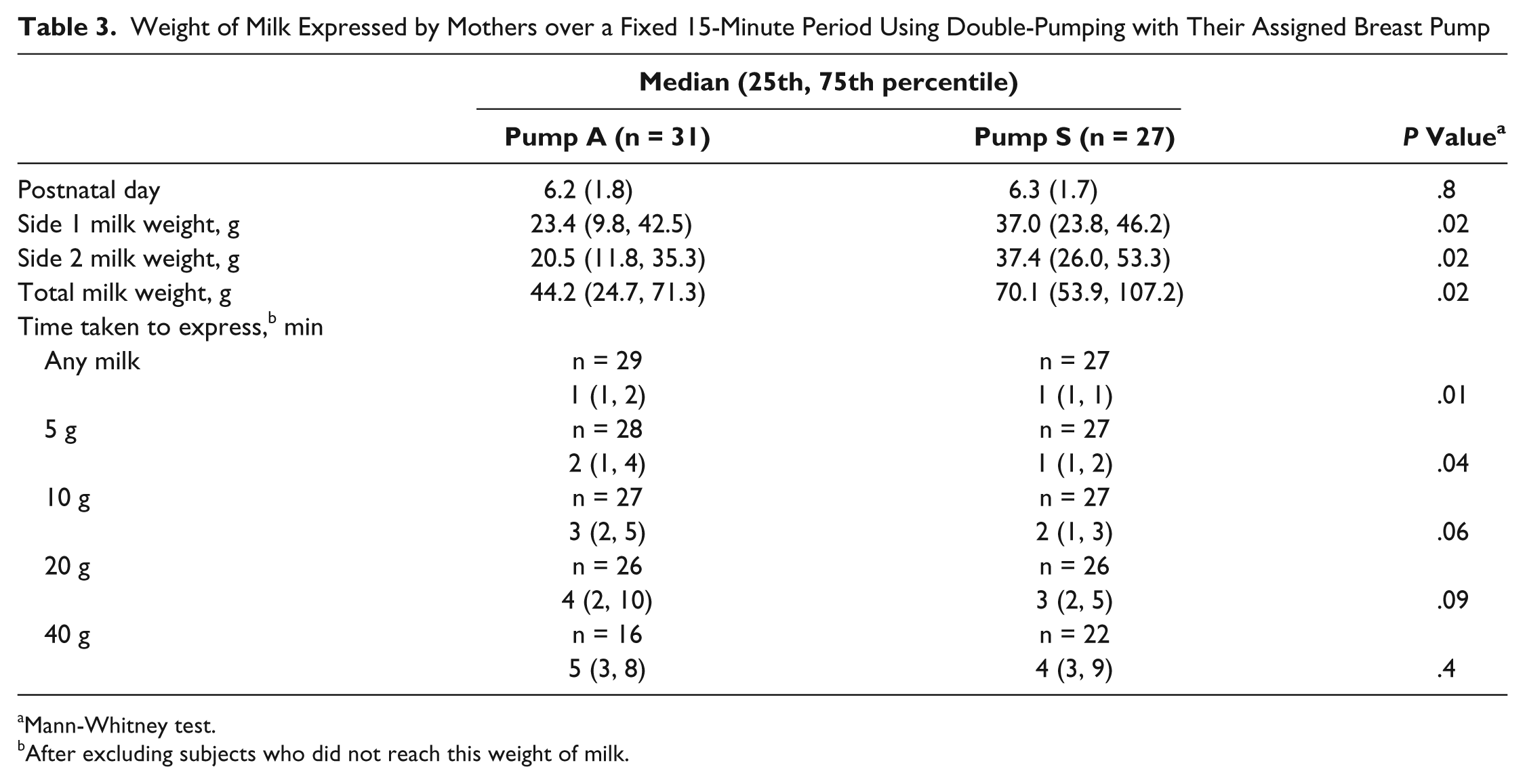
Mann-Whitney test.
After excluding subjects who did not reach this weight of milk.
Secondary Outcomes
Total number of pumping sessions and time spent expressing milk
There were no significant differences in the total number of pumping sessions per day between randomized groups. The median frequency of milk expression was less than 4 times per day; the median values (25th, 75th percentile) were 3.3 (2.7, 4.3) sessions per day for pump A and 3.7 (2.9, 4.1) for pump S (Table 2), considerably lower than recommended (6-8 times per day) in both groups of mothers. There were no significant differences in the time spent expressing milk in total or per day, nor in the efficiency of milk expressed as mL per minute spent expressing between randomized groups (mean time/day expressing [standard deviation (SD)]): 82 (32) minutes for pump A versus 75 (37) for pump S, P = .3; mean mL/min 3.3 (2.9) versus 3.1 (2.3), P = .8.
Mothers’ opinions of the assigned pump
Raw data (using the 7-point scale) are shown in Figure 2. When the data were recoded as 1 = 1-3, 2 = 4, and 3 = 5-7, pump A received significantly better scores for the category “location of control button” (P = .04). When the data were recoded as 1 = 1-2, 2 = 3-5, and 3 = 6-7, pump A scored higher for the category “ease of use.” No other opinions differed significantly between the pump groups (also Figure 2).
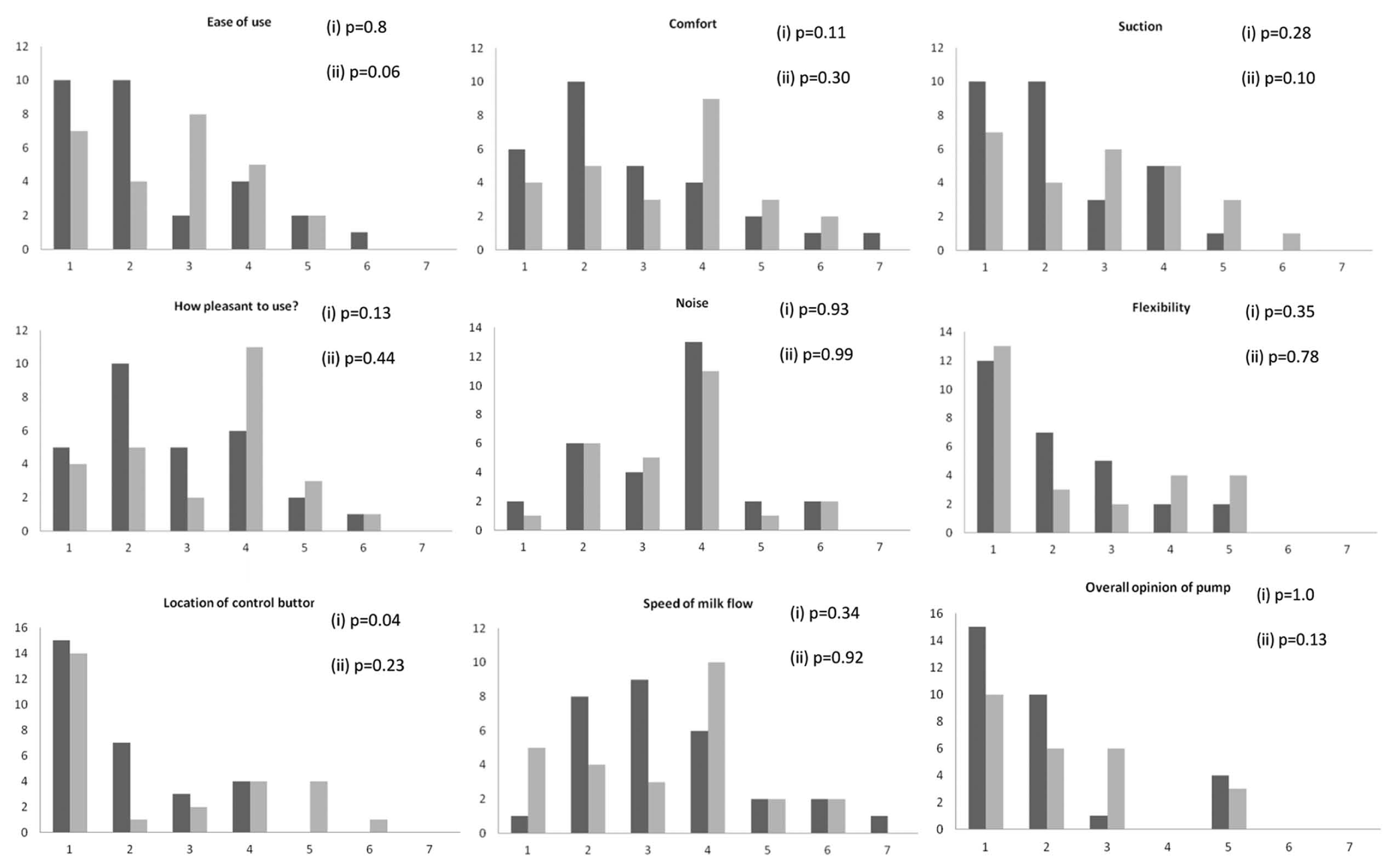
Results of Questionnaires
Analyses beyond initial 10-day study period
There were no significant differences between pump groups in the total volume of milk expressed while infants were in the NICU, volume of milk expressed and fed to infant, time taken for infants to reach full enteral feeds, or the median volume of milk expressed per day (Table 4). While there were no significant differences between groups in the proportion of infants receiving only breast milk (51.7% vs 50.0%), only formula (6.9% vs 19.2%), or a mixture of these (41.4% vs 30.8%) at discharge for pump A and pump S, respectively (P > .05 in all cases), there was a significant association between the randomized pump and whether or not mothers were breastfeeding their infant(s) at the time of discharge from the NICU (P < .01). Twenty-one of 29 group A mothers versus 8 of 21 group S mothers reported breastfeeding at least 1 infant at discharge (odds ratio [OR], 4.27 [95% confidence interval (CI), 1.29, 14.1]). This was also significant when the analysis was restricted to mothers with singletons (16/21 vs 5/13; OR, 5.1 [95% CI, 1.1-23.0]). Using logistic regression to control for potential confounders (birth weight, gestational age, and infant age at discharge), this finding remained significant (adjusted OR, 7.52 [95% CI, 1.79, 32.89]). Gestational age was also a significant predictor of breastfeeding at discharge in this model (OR, 1.37 [95% CI, 1.04, 1.80]).
Total Volume of Milk Expressed, Total Volume Fed to Infant, and Time to Full Enteral Feeds While Infants Were in Neonatal Intensive Care Unit
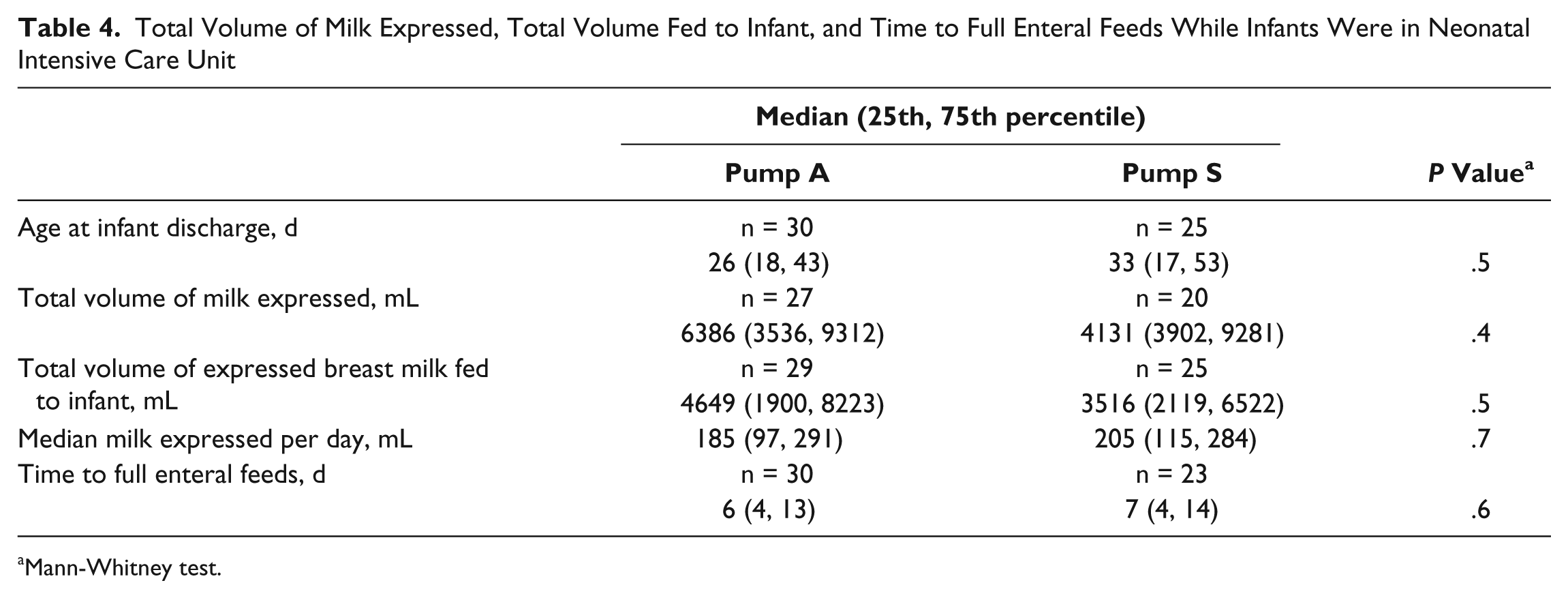
Mann-Whitney test.
Discussion
In the initial 10-day study period, we found no statistically significant difference in the effectiveness of a novel breast pump (pump A) and a pump widely used in many neonatal care units (pump S), in terms of the amount of milk expressed by mothers, time spent expressing, or the amount of milk produced per minute of pumping. This is the most important finding in terms of the provision of breast milk for the infant. Beyond the initial study period, we found similarly no significant differences between breast pumps in the total volume of milk expressed or fed to infants while in the NICU or in time taken to reach full enteral feeds. It has been suggested that breast pump effectiveness may depend more on the stage of milk expression than the type or design of pump. 10 Due to the parallel group design of our study, there was no significant difference in the stage of lactation between randomized groups. However, our ability to draw conclusions is limited by the relatively small sample size; we could exclude a difference of only about 0.8 SD between groups, and it is important to emphasize that this study had insufficient power to detect a smaller, though perhaps biologically or clinically relevant, difference.
The total weight of milk expressed over a 15-minute fixed test period on a single occasion between day 3 and day 10 was significantly greater and the time taken to produce specific volumes of milk significantly less with pump S than with pump A, indicating that this pump performed better under the test conditions. The default pump for both NICUs was pump S; consequently, this was the pump that the research nurses were familiar with and also the pump that mothers used if they started expressing before randomization into the study. It is also possible that the 2-phase “fixed” expression system in pump S was on average more easily mastered and therefore more effective at obtaining milk under the conditions of the test. Although mothers using pump A should be able to adopt a 2-phase system to suit their own requirements, it may be that they found this difficult under the test conditions or had not yet worked out how to use the pump with greatest effect, which suggests that further education may be required to optimize the use of this pump. Although the mean age at which the physiological test was conducted did not differ between groups, our inability to standardize this age represents a further limitation, reflecting the need to find a convenient time for mothers, many of whom had been discharged from the maternity unit and were visiting from home.
It is interesting, given the results from the physiological test, that on maternal perception questionnaires completed after 10 days of pump use, pump A tended to receive better scores for certain parameters. Two methods were used for recoding the 7-point Likert-type scores, both of which preserved the symmetrical nature of the raw data, in which a score of 4 represented the neutral category, but placed a different emphasis on the more extreme scores. It is important to note that different results were found depending on the method of recoding used, probably reflecting the relatively small sample size. Using the first method, in which scores were grouped as 1-3, 4, and 5-7, a significantly greater proportion of mothers using pump A awarded a higher score for the parameter “location of the control button,” with a similar trend for the parameters “comfort” and “ease of use.” Using the second method, which placed greater emphasis on the extreme scores, grouping 1-2, 3-5, and 6-7 on the basis that a score of 1 or 3 would have a different meaning to mothers, pump A tended to receive higher scores for “ease of use” with a similar trend for “suction” and “overall opinion.” A number of potential issues have been highlighted in drawing conclusions regarding maternal satisfaction in pump studies. 10 For example, it was suggested that the use of pumps for 48 hours only in a previous study from our group 4 gives only an initial impression of the pumps. In the current study, both pumps were used for a period of at least 10 days, allowing mothers to form a longer term opinion of their pump. It would perhaps have been informative to repeat this questionnaire at the point of infant discharge, when mothers had used the pumps for even longer. Other studies have involved descriptive reports of opinions of pumps by mothers 11 and structured interviewing, 12 and this would be worth pursuing in future studies. However, we considered that the use of Likert-type scales was less onerous to mothers who were often experiencing a considerable degree of stress and anxiety over their infants. In the future, it might be preferable to ask mothers to indicate their score by placing a marker on a line of fixed length; the position of their marker could then be quantified, generating data in the form of a continuous variable and avoiding the need for recoding.
As in previous studies, 3,13 we found considerable variability in the weight of milk expressed by individual mothers, regardless of the breast pump used. Mothers who predominantly used double-pumping expressed greater amounts of milk during the initial 10-day study period, perhaps reflecting increased breast stimulation leading to higher prolactin concentrations. Nearly all the mothers in this study were putting their infants to the breast or using kangaroo care during the initial 10-day study period, although most infants were too immature to effectively breastfeed. Although the eligibility criteria for the study included an intention to express milk, we did not ask about the mother’s breastfeeding intentions, and it would be interesting to collect such data in future studies, as highlighted by Becker. 10 It would also be interesting to include a measure of maternal stress, which was not assessed in our study but would be expected to influence milk production and expression.
Data on associations between method of breast milk expression and the likelihood of an infant continuing to receive breast milk and/or transferring to the breast are extremely limited, 13 yet this is an important outcome. The proportion of infants in this study receiving breast milk at discharge and the volumes received during the hospital stay were not significantly different between the pump groups; however, mothers randomized to pump A were significantly more likely to be directly breastfeeding their infant, even after adjusting for potential confounders such as infant gestational age and age at discharge. The odds ratio for this effect following adjustment for confounding factors was 7.5, although the wide confidence intervals (reflecting the inherent variability seen between breastfeeding mothers of preterm infants and the relatively small sample size) limit confidence in the size of the effect. The mechanism for such an effect is unclear. The attainment of breastfeeding in this population is dependent on both infant and maternal characteristics; it is possible that the mother’s experience of using a breast pump influences her motivation to put her infant to the breast and establish breastfeeding or, alternatively, that there is residual confounding by unmeasured infant-related factors. Nevertheless, while receiving breast milk via any route is an important outcome in itself, in the context of current efforts to promote breastfeeding, the finding of a greater likelihood of breastfeeding at infant discharge is worthy of note and merits further investigation.
Conclusion
In conclusion, although pump S performed better during a fixed 15-minute test period, the study showed no significant practically relevant differences between the pumps tested during an initial 10-day study period and during the extended time that infants remained in the NICU. Similar effectiveness of these pumps in everyday clinical practice is potentially important to neonatal units, given differences in the costs of the pumps. Moreover, the finding of an effect of the type of breast pump on the likelihood of breastfeeding at discharge is of importance in the context of the increasing emphasis given to breastfeeding in the NICU.
Footnotes
Acknowledgements
The authors acknowledge the support of the mothers who participated in this study, the research nurses (Sister Anne O’Neill [London] and Sisters Penny Lucas and Janette Clarke [Cambridge]), and nursing and allied professional staff in the participating NICUs.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The study was funded by Philips AVENT. Dr Burton, Dr Fewtrell, and Professor Lucas have also received an unrestricted research grant from Philips AVENT.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from Philips AVENT (Philips Consumer Lifestyle, Amsterdam, The Netherlands), who also provided the breast pumps, and sponsored by the UCL Institute of Child Health. The funders and sponsors were not involved in conducting the study or analyzing or interpreting the data.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
