Abstract
The structural features of polymer-based tissue engineering scaffolds engineered to support cell adhesion, proliferation, and differentiation have been consistently and assiduously studied over the past few decades. It is now well known that scaffolds composed of polymers with ultrafine fibrous morphologies produced via electrospinning and integrated porosity, can positively influence cell response. The primary objective of most studies in tissue engineering scaffold development is to create a scaffold that emulates the native in vivo-like environment of extracellular matrices (ECMs). Achieving an even distribution of cells throughout the scaffold is critical for exactly mimicking the native extracellular matrix environment. However, inadequate cell infiltration towards the center of the scaffolds has been a common issue in many studies. Only a limited subset of researchers has successfully identified the structural features of scaffolds that facilitate cell penetration and has consequently introduced innovative scaffolds. This study aims to identify the critical structural features of polymeric scaffolds that facilitate cell infiltration and presents novel ultrafine fibrous scaffolds engineered to enhance uniform cellular penetration.
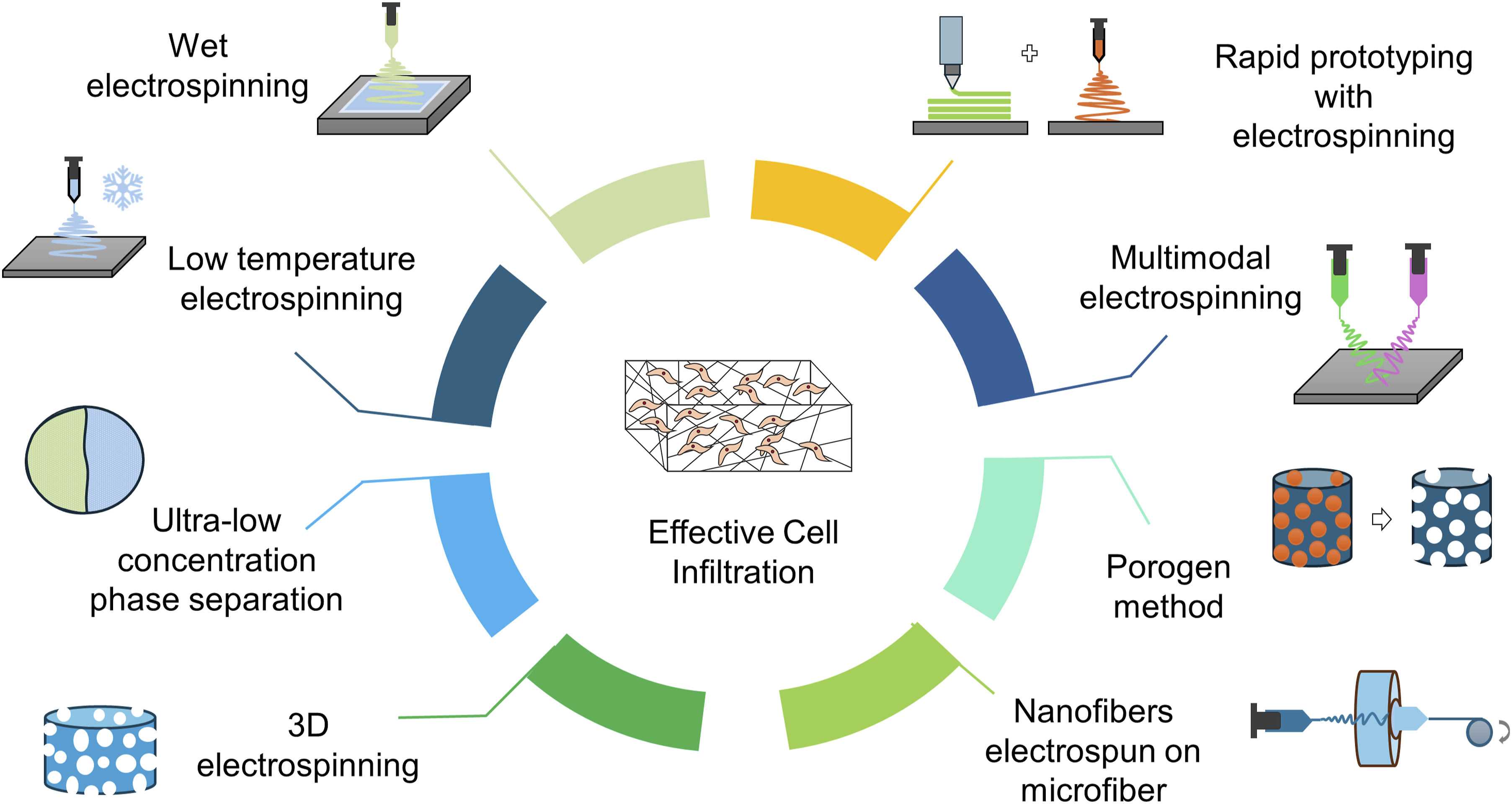
Introduction
Ultrafine fibrous tissue engineering scaffolds can highly support the treatment of the presence of inflammatory cells in human tissue and organ without other evidence of their inflammatory process. Organ and tissue deficiencies in the cardiovascular blood vessels,1–4 neural and neuromuscular, 5 orthopedic6,7 and plastic reconstructive, gastrointestinal, urological, nephrological, and blood transfusion 8 and skin9,10 related human health problems require living tissue regeneration and placement treatment. Tissue engineering works on developing artificial tissues and organs that can replace or augment malfunctioning or damaged ones in the human body.10–12
Tissue engineering faces a range of interdisciplinary challenges spanning cell biology, materials science, fabrication, and translational hurdles. Cell sourcing and culture remain critical due to limitations in availability and aging-related functional decline,13,14 addressed through organ, explant, and organotypic culture techniques.15,16 On the fabrication side, advanced strategies such as electrospinning, 17 3D bioprinting, 18 and decellularization/recellularization 19 aim to mimic the extracellular matrix, though challenges remain in pore interconnectivity and long processing times.20,21 Biomaterial scaffolds must balance biocompatibility, biodegradability, and mechanical integrity, with solutions ranging from polymeric and ceramic scaffolds22,23 to hybrid composites and electrospun designs. 24 Bioreactors, including perfusion and microfluidic systems, help optimize in vitro conditions but remain costly and technically demanding.25,26 At the biochemical level, controlled delivery of growth factors such as TGF-β, VEGF, and BMPs is essential but hampered by instability and inconsistent cellular responses.27–29 Additional hurdles include biomechanical assessment, 30 ensuring immunocompatibility,31,32 and achieving sufficient vascularization, often pursued via angiogenic factor delivery or microfluidic strategies.33,34 Beyond the laboratory, issues of quality control, regulatory compliance, and standardization35,36 complicate scale-up and commercialization, 37 while clinical integration raises patient-specific and long-term viability concerns. 38 Finally, ethical and legal considerations, including sourcing of hESCs, informed consent, and intellectual property rights, continue to shape the societal acceptance of tissue engineering.39,40
An ideal scaffold should hold a number of major characteristics to effectively support tissue regeneration. First, it should have an ultrafine, interconnected three-dimensional porous network that enables cells to attach, migrate, and proliferate throughout the scaffold. Additionally, the scaffold must include proper channels for oxygen and nutrient circulation to ensure cells deep within the scaffold are nourished and to facilitate the removal of cellular waste. Biocompatibility is essential, allowing native tissues or cells to attach and proliferate without adverse reactions. The scaffold should also have the appropriate shape, whether three-dimensional (3D), two-dimensional (2D), or otherwise, depending on the specific application. Finally, it should exhibit suitable biomechanical properties, functionality, and an immunogenicity profile that matches the needs of the tissue it aims to regenerate. 41
A major human health problem is damaged organ or tissue that originated either from a disease or an injury. 8 Numerous works have been published related to the regeneration of tissues offering cell growth on flat glass or polystyrene substrate, with the belief that animal physiology can be truly reproduced using a two-dimensional (2D) cellular monolayer. 42 However, the culture of cells in 2D overlooks many parameters such as cells’ spatial and temporal organization, which are a must to generate a native three-dimensional (3D) extracellular matrix (ECM) like environment. 3D organization of cells can ensure controlled cell functions, i.e. division, through proliferation to differentiation and apoptosis. In this regard, researchers have developed a number of three-dimensional models (i.e. cells seeded within 3D matrices) for a variety of tissues where the culture environment considers the spatial organization of the cells.43–45 In 3D models, in addition to spatial control, cellular aggregates require careful exchange of nutrients and gases. But challenges with cell death become apparent when aggregate thicknesses of 1–2 mm occur through a lack of mass transfer, i.e. reduced exchange of nutrients and waste metabolites.46,47 Thus, in order to create a growth environment that mimics the native 3D ECMs, cells should be introduced into a porous biocompatible substrate. 48 Today, this is done by introducing cells into a biocompatible porous 3D scaffold or matrix, which once seeded can mimic the natural ECM where cells adhere, proliferate and migrate. Apart from using either synthetic or natural polymer scaffolds, special attention has now been paid to the composite materials based on the polymer blends for improved cell functioning with adequate mechanical properties.49–54 Sometimes, additional cross-linking ingredients such as Genipin are used to improve the mechanical properties of the blends. 55 Different types of organs or tissues have different shapes and architectures. As Cellular response on substrates is very reliant on the length scale of substrate’s surface features 56 and scaffolding surfaces, which are directly interacting with cells, should have alike dimensions as the natural ECM, 57 scaffolds of various structure and morphology are necessary for each of the different types of tissues. In the native 3D ECM environment, the 3D configuration and nanometer-scaled fibrous morphology have been observed to influence cell behavior in several tissues.58–61 For this reason, many research works were carried out in designing 3D fibrous scaffolds for TE to mimic the natural 3D ECM. Several different polymeric materials and processing techniques have been proposed to produce 3D scaffolds with Micro- or Nano- or combined Micro-/Nano- hybrid morphology that provide high cell adhesion, proliferation and migration.62–66 But one major problem of most of the approaches, which remains less noticed by the researchers so far, is limited to no spreading of cells inside the scaffold. 67 The key challenge in this respect of tissue engineering is to find an applicable fibrous (as fibers incorporates contact guidance to the cells to orientate and move rapidly along fibers 68 ) scaffold with not only an plenty of active groups and good biocompatibility, but also containing appropriate mechanical properties, biodegradability and porous structures with large interconnected pores as large interconnected pores prevent cells from accumulating at the surface of the scaffold by enabling them to diffuse and distribute throughout the entire scaffold. 69 While existing reviews provide valuable insights into electrospinning techniques and the general application of fibrous scaffolds in tissue engineering, there is a gap in comprehensive analyses focusing specifically on ultrafine fibrous scaffolds and their role in enhancing cell infiltration. This paper aims to fill this gap by representing some original approaches in fibrous scaffold designing, where high cell infiltration so far has been taken under consideration without negotiating critical cell functions. Overall, this review highlights the unique advantages of ultrafine fibers for enhanced cell infiltration, addresses structural limitations in current scaffolds, and integrates insights from material science, bioengineering, and cell biology to develop next-generation ECM-mimicking scaffolds.
Importance of infiltration and 3D organization of cells in TE scaffolds
3D configuration of the matrix affects both the diffusion of solute and binds several effector proteins, such as growth factors and enzymes. Furthermore, the 3D environment is crucial in morphogenetic and remodeling events in 3D ECMs.70–75 The 3D organization of cells promote proper transmission of mechanical cues and other receptor expressions between cell-cell and cell-substrate, as in native ECM in vivo (Figure 1). Schematic of ECM 3D environment for engineering cell function.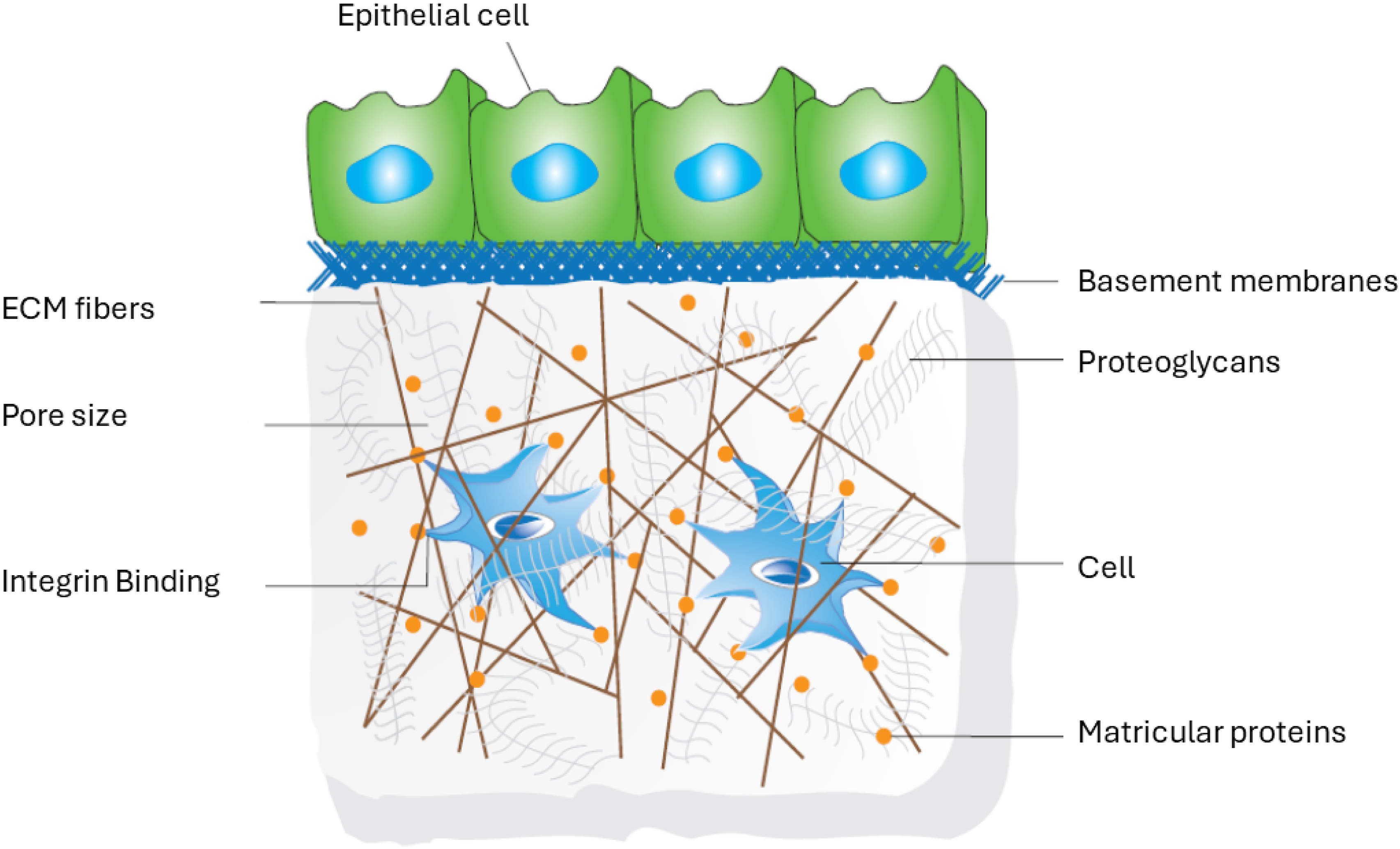
3D organization of cells is also important to achieve the exact shape and structure of their native state when cultured in vitro.48,76 For example, Proper spatial localization of epidermal growth factor receptor signaling activated by cell secreted autocrine ligands denotes a critical factor in embryonic development and tissue organization as cells use autocrine loops (a signaling mode) as sonar system to probe surrounding environment by recapturing the fraction of signals they sent out and thereby helping to define tissue boundary. 77 The idea of incorporating cells on 3D scaffolds comes mainly because, in 3D environments, cells can grow more branches to receive signals from multiple directions than in 2D environments. 78 But poor cell infiltration and nutrient transport inside the 3D scaffolds limit their application in tissue engineering, particularly when used for in vitro culture environment, because, as time passes, the colonized cells seeded and adhered to the scaffold proliferate and migrate only on the surface, resulting in a necrotic core. 79 Therefore, an exact mimic of the native 3D environment can only be done in vitro when cells can penetrate easily inside the 3D scaffolds from the surface and prevent themselves from flattening.
Scaffold parameters required for 3D organization of cells
The characteristic requirement of a scaffold (implantable or injectable form) for effective cell functioning in vivo or in vitro is well known.80,81 Along with the fact that cell adhesion to substrate is ‘cell type’ and ‘scaffolding material’ specific, 82 a nanofibrous scaffold with 3D architecture is well well-defined characteristic for high cell adhesion to the scaffold.83–86 Microfiber scaffolds can produce significantly large pores, but several studies showed nanofibrous scaffolds are better for cell adhesion and proliferation than microfiber scaffolds made from the same component due to their (nanofibers) high surface area to volume ratio.87–93 Moreover, nano-scale architectures make the cells create more filopodia and contribute to the attachment and proliferation of the cells. But cell migration is limited to only on the seeded surface because of small pore size of 2D nanofibrous scaffold94,95 which is not enough for cell infiltration as in many cases, large pore size (several hundreds of micrometer) is necessary for efficient migration of cells throughout the scaffold.90,96–98 Even though high porosity and large pore size have positive effect on cell differentiation and matrix production,99–101 excessive large pores however might not be possible for the cells to bridge over 90 and can cause the loss of extracellular matrix proteins into the medium from the scaffold, 102 hence affecting cell growth negatively.
Another important aspect in this regard is pore interconnectivity and tortuosity (the proportion of the actual path length through connected pores to the shortest linear distance 103 ). Interconnected pores allow efficient transfer of nutrients and waste metabolites and permit matrix production. 104 The interconnected open poor structure can prevent the development of closed-cell morphology and inhomogeneous distribution of cell sizes. 105 Additionally, a porous surface is known to enhance mechanical interlocking between the implanted scaffolds and the surrounding natural tissue in vivo, offering greater mechanical stability at this critical interface 106 and permit facile invasion of blood vessels for the source of nutrients to the transplanted cells in vivo. 107 So, along with 3D ultrafine fibrous (nano- or micro- scaled fibers) morphology, for high cell infiltration, it is very important to develop scaffolds with high porosity and sufficiently large pores where cells, once adhered to the substrate in efficient culture environment, can proliferate and migrate along the fibers through the inter fiber gaps or pores (well defined macro/micro-pores) towards the inside of the scaffold and organize in 3D.
Therefore, for effective 3D cell organization, scaffolds must combine nanofibrous architecture with high porosity, sufficiently large and interconnected pores, and appropriate surface features to enable deep cell infiltration, proliferation, and migration throughout the structure.
Fabrication techniques of fibrous scaffolds for high cell infiltration
Although some cultural environments have shown a significant effect on cell infiltration, such as flow perfusion, 108 but it is only effective for tissue engineering in vitro at the cost of a huge number of cells. An effective method to enhance nutrient and cell transfer to the scaffold center, both in vivo and in vitro, is to model an optimized scaffold with sufficiently large pores. Emphasis has now been given to increasing the pore size of nano-fibrous scaffolds. Several recently published studies show that combining nanofibers and microfibers in a scaffold may increase the pore size of the fibrous construct.65,92 Nanofibers then facilitate surfaces for cell attachment and proliferation, whereas microfibers support the structural environment for cell infiltration. Nano-fibrous scaffolds with macro- (>100 µm) or micro-pores can also be created without incorporating microfibers. Here, some of those noble 3D fibrous scaffolds are represented, where the produced scaffolds nearly bear those properties necessary for cell infiltration. To fabricate noble scaffolds that can provide an isotropic arrangement of nano-/micro-scaled environment throughout the scaffold, researchers are now concentrating on the modification of standard fabrication techniques.
Wet electrospinning
An effective example of wet electrospinning is ‘Layered hydrospinning’ (Figure 2(a)). 3D poly(e-caprolactone) (PCL) nanofibrous scaffold produced by this method has shown a porosity of up to 99% and pores with diameters of over 100 micrometers.
109
This process is fast, and a more than 1 cm thick scaffold can be produced in a reasonably short time. The difference in the pore size between hydrospun and electrospun nano-fiber scaffolds may be due to water exerting tension on fibers, causing them to widen, resulting in larger pores, evidently due to the high ductility of PCL at room temperature. Although not very significant, large pore size can also be clarified by the fact that, in electrospinning, deposited fibers possess charge that causes the newly deposited fiber to move towards the gap between two adjacent fibers deposited earlier and thus making the inter fiber gap almost half resulting in pores with half the size of original one. However, in hydro spinning, fibers are discharged immediately, and next, falling fibers will not repel towards the center of the pore. The thickness difference exists because of the tendency of layers to separate from each other due to tension exerted by water on the nanofibers while evacuating, as they have a large surface area.
109
Furthermore, innovative adaptations combining wet electrospinning and coaxial (core–shell) approaches produced 3D porous PCL scaffolds with protein cargo (e.g., BSA), demonstrating sustained release, biocompatibility, and favorable pore architecture conducive to cartilage tissue regeneration. (a) Step by step method of layered hydrospinning through electrospinning and stacking layers of nanofibers followed by vacuuming, redrawn from,
91
and (b) the concept of core-shell electrospinning.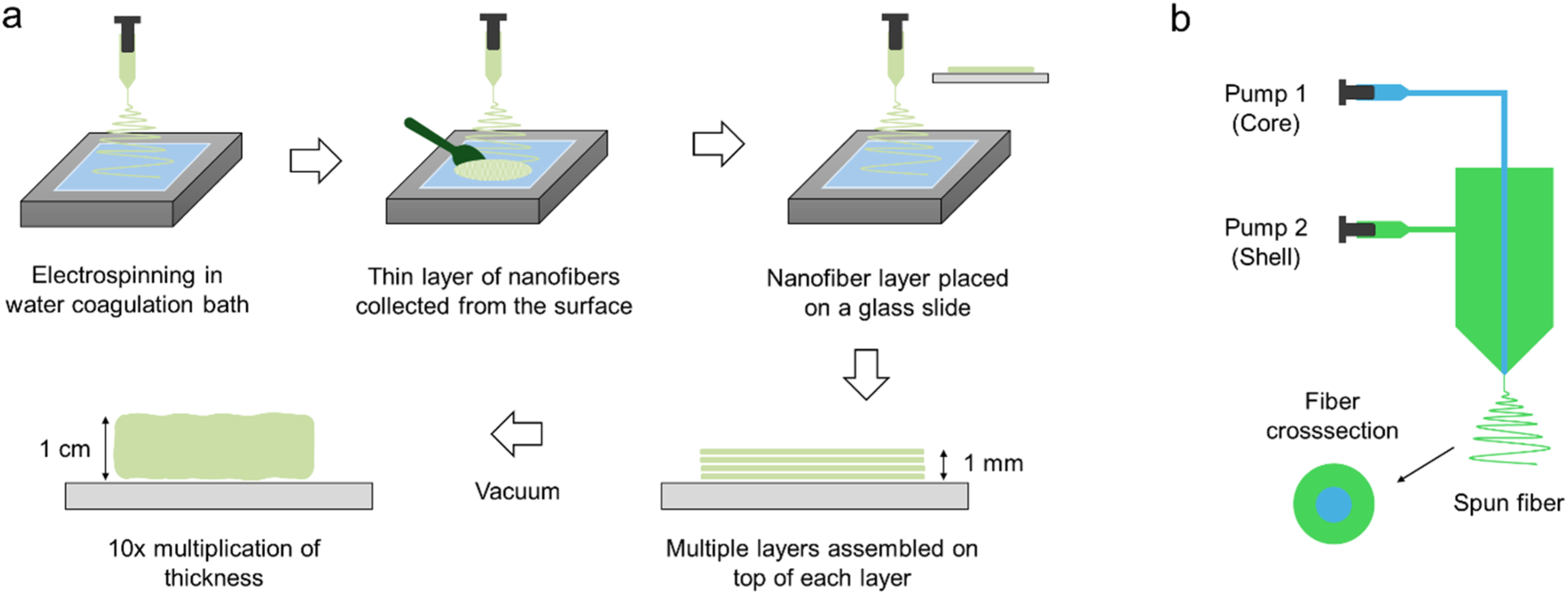
Hydrospinning provides two distinct advantages over typical electrospinning. Those are thicker, porous scaffolds produced in a realistic period and with higher porosity with comparatively large pore size. A similar wet electrospinning method was introduced later by Yang et al., with the exception that no vacuum treatment is necessary.
110
They developed a layer-by-layer cell/fiber deposited 3D scaffold where cells are seeded on consecutive electrospun layers. They used grounded liquid (including culture medium or Phosphate Buffer Saline, or distilled water) as a fiber collection surface instead of using a solid collector. Single cell types, such as incorporation of osteoblastic cells on PCL/chitosan nanofibers for bone-like tissue formation, or multiple cell type for example incorporating dermal fibroblasts and keratinocytes onto PCL/collagen layers in alternating manner for skin tissue engineering can effectively be used in this ‘L-b-L’ bottom up assembly approach.
110
Most importantly, cells can be seeded while spinning fibrous meshes. Scaffolds of controlled thickness can be made by fixing the electrospinning time for each layer, and a uniform distribution of cells into the interior of the scaffold (Figure 3) can be obtained by seeding cells on successive layers. A broader review emphasizes the critical role of scaffold porosity and pore size in tissue engineering, showing how specific pore ranges influence cell behavior and tissue formation. Small pores (∼1–2 µm) support skin epidermal attachment, moderate pores (∼2–60 µm) aid dermal migration, cardiovascular, and lung tissue integration, and larger pores (∼40–400 µm) enhance vascularization, nutrient diffusion, and bone tissue regeneration. Optimizing pore size distributions across different tissues is key to improving scaffold functionality and advancing tissue regeneration strategies.
112
Table 1 summarizes some related methods and key outcomes reported in wet electrospinning. Microscopic images of multilayered cell-fiber constructs: (a) Fluorescent micrograph of DAPI-stained cross-sections of fiber-cell constructs cultured for 2 days. Nuclei = blue. Scale: 200 μm. (b) Confocal microscopic images of cross-sections of formed cell-fiber constructs with a controlled thickness of fiber layer. Scale: 20 μm. Fibers were labelled with FITC (green), and cells were stained blue with DAPI. (c) H&E-stained cross-section of the construct cultured for 7 days. Arrowhead indicates fibroblasts, and asterisks show fibers. Scale: 50 μm. The images shown are representative of three separate experiments. Reprinted from,
111
Copyright (2023), with permission from Elsevier. Wet-electrospinning methods in tissue engineering and key findings.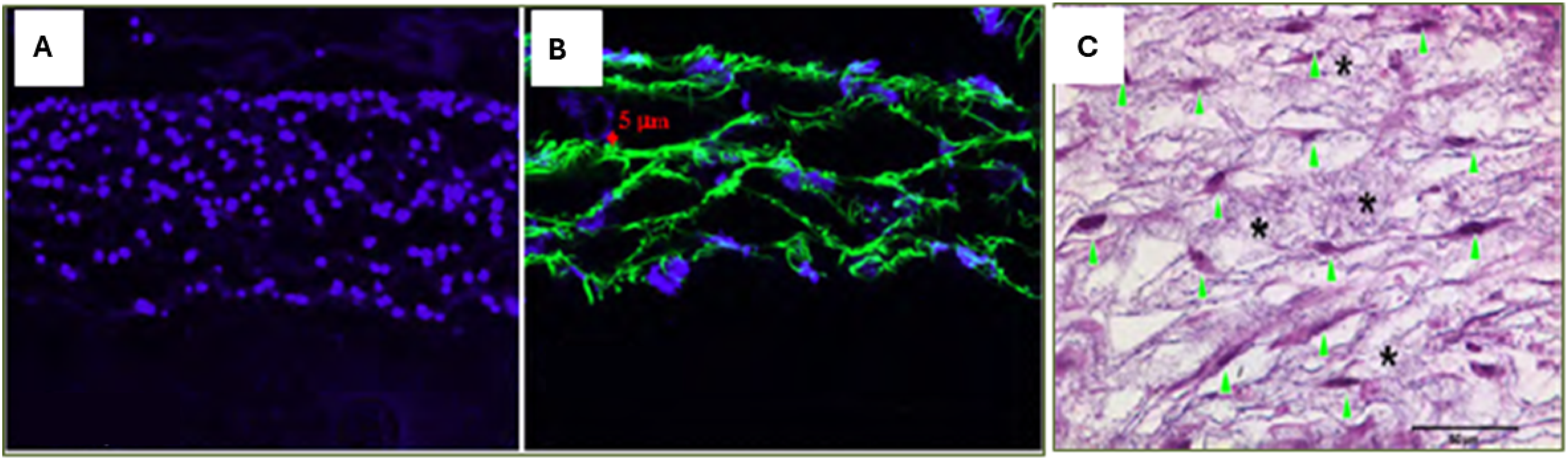

Overall, layered hydrospinning enables the rapid fabrication of thick, highly porous 3D PCL scaffolds with large, well-distributed pores, supports cell seeding during scaffold formation, and allows controlled pore architectures that enhance nutrient diffusion, vascularization, and tissue-specific regeneration.
Core–shell electrospinning
Core–shell electrospinning is an advanced variation of electrospinning that allows the simultaneous incorporation of two or more materials within a single fiber, thereby integrating complementary properties into a unified structure. In this technique, the core typically provides mechanical stability or controlled drug storage, while the shell offers biological functionality or tailored surface chemistry. Such multi-material fibers enable the fabrication of scaffolds that address both the structural and biological requirements of tissue engineering. Figure 2(b) shows the mechanism of core-shell electrospinning.
Key findings on core-shell electrospinning and their benefits.

3D electrospinning
Though wet electrospinning can physically enhance distances between the electrospun fibers and facilitate deeper penetration of cells into the interior of scaffolds to a certain degree, it cannot alter the planar orientations of the electrospun fibers. 3D zein scaffold developed by Cai et al. using novel 3D electrospinning [electrospinning by repulsive force] showed random orientation of fibers (Figure 4(a) and 4(b)) with fibers in the thickness direction as well, and the pore size is larger than 100 micrometers.
122
They incorporated SDS (sodium dodecyl sulfate) with PEG to reduce the surface resistivity of the fibers, as low surface resistivity can cause faster charge dissipation from the fiber to the collector, resulting in an increase in repulsive force, which thus aligns the fiber in the Z direction (Figure 4(d)). The mechanism of this reduction in resistivity claimed by Cai et al. is that when water evaporates sulfate group of SDS concentrates on the surface of fibers, pointing towards the outside; consequently, a surface water layer is formed on Poly Ethylene Glycol (PEG) where dissociable sodium ions from SDS effectively reduce surface resistivity. They claimed that specific pore volume can be exponentially increased with decreasing surface resistivity of the fiber. (a and b) represent continuous photography of PEG fibers in 2D and 3D electrospinning, respectively, with a 0.125 s time interval between two sequential photographs, where the white arrow represents the PEG fiber depositing on the collector. c & d are schematic diagrams of the deposition of fibers in 2D and 3D electrospinning processes. Dark green color shows the fiber depositing on the collecting board (orange color), light green indicates new fiber depositing on the old one. The black arrow represents quick charge dissipation to the collecting board, resulting in repulsive force generation between the collecting board and the depositing fiber (orange arrow)
122
.
Reported key approaches through 3D electrospinning towards tissue engineering.

In summary, 3D electrospinning techniques, including repulsive-force electrospinning, co-electrospinning with sacrificial fibers, and template- or collector-assisted methods, enable the fabrication of ultrafine fibrous scaffolds with randomly oriented fibers, larger pores, and enhanced interconnectivity, facilitating deeper cell infiltration and improved 3D scaffold architecture.
Multimodal electrospinning
The increased pore size with increasing average fiber diameter65,126,127 gave rise to the idea of mixing microfibers with nano-scale fibers. This is beneficial in such a way that the porosity of the structure can be simply altered by the change of nano-fiber/micro-fiber ratio; hence, it provides expected control over an important parameter. But creating a proper mixing of nano-/micro-fibers is a great challenge. If there is improper mixing, nanofibers can form sheets over microfibers and form a strong barrier for cell infiltration. This is most commonly found in multilayered electrospinning where mixing is carried out by using different scaled fiber layers. Soliman et al. proposed a modified electrospinning technique to produce bio-inspired multi-scale 3D scaffolds endowed with controlled bimodal or multimodal fiber diameter distribution with porosity of about 90%.
128
In this method, a polymer solution of two different polymer concentrations was electrospun from two independently controlled syringe pumps on a single collector situated on a rotating mandrel (Figure 5, left). By using a rotating collector mandrel, it is possible to collect a significant number of nanofibers and microfibers alternately when the collector reaches directly under each syringe by rotation and thus maintain uniform fiber distribution. Large pore size was indicated by human bone marrow-derived mesenchymal stromal cell (mTERT-MSCs) colonization inside the bimodal 3D PCL scaffold after 7 days of culture (Figure 5, right).
128
Schematic representation of multimodal electrospinning technique at left and on the right, immunofluorescence micrographs of DAPI-stained mTERT-MSC as grown after 7 days culture on Mix PCL scaffolds (fiber dia d1 = 3.3 µm and d2 = 0.6 µm). Sectioning depths from the reference surface are (a) 192 µm, (b) 128 µm, (c) 96 µm and (d) 64 µm. The scale bar is 100 µm for (a) but 50 µm for (b–d), Reprinted from,
128
Copyright (2010), with permission from Elsevier.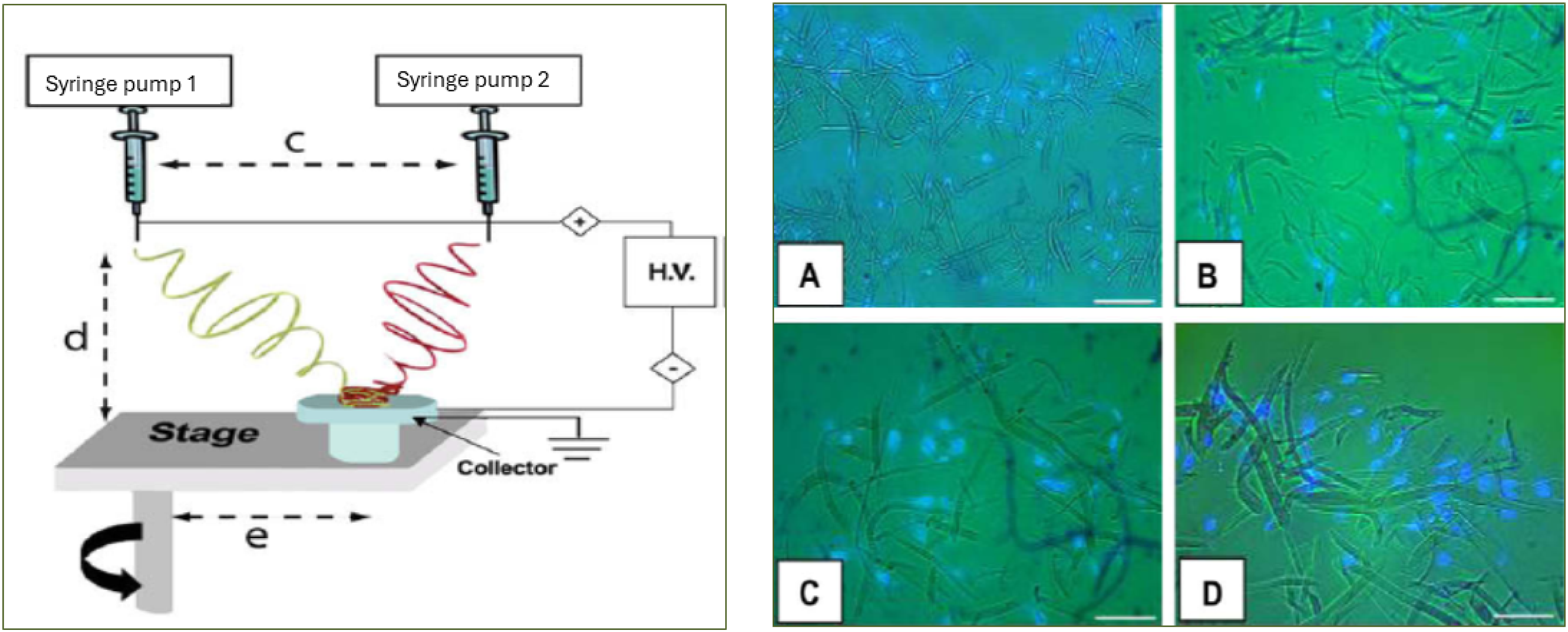
The concept of multimodal electrospinning, mixing fibers of different scales within a single scaffold, enables precise control over porosity and fiber architecture. Soliman et al. implemented this by using two polymer solutions from matched syringe pumps onto a rotating mandrel, achieving a controlled bimodal distribution that supported mesenchymal stromal cell colonization. 128 Another scalable strategy purposely mingles nano- and micrometer fibers to avoid stratified layering, producing a more homogeneous scaffold structure conducive to cell infiltration. 128
Alternatively, melt electrospinning permits the simultaneous deposition of large-diameter and nanoscale fibers without the use of solvents, facilitating full cell penetration and matrix formation while avoiding solvent-related cytotoxicity. Moreover, multi-channel electrospinning setups enable different polymer formulations to be concurrently spun, enhancing scaffold mechanical performance and improving mesenchymal stem cell adhesion through optimized fiber architecture. 129
Overall, multimodal electrospinning combines nano- and microfibers within a single scaffold to precisely control porosity and fiber architecture, enabling enhanced cell infiltration, uniform fiber distribution, and improved scaffold mechanical performance, with techniques including dual-syringe pumps, rotating collectors, melt electrospinning, and multi-channel setups.
Electrospinning of nanofibers onto a single microfiber
Using two spinnerets to simultaneously electrospun both nano- and microfibers is very difficult owing to the repulsive force between two fibers, and also time-consuming. Electrospinning nanofibers directly onto a single microfiber can create highly porous scaffolds with a suitable distribution of combined nano- and micro-fibers throughout the scaffold. Such a flexible technique for creating structures of variable shape, size, porosity and morphology was first introduced by Anna et al., who developed scaffold where a single PLA microfiber was coated with electrospun PCL nanofibers (Figure 6, left).
94
SEM image of electrospun PCL nanofibers coated single PLA microfiber (left) and Scaffolds with chondrocytes after 2 weeks of culturing (right) on PCL coated PLA microfiber scaffold with (a) 97% and (b) 95% porosities and references of (c) PCL only nanofibers and (d) PLA only microfibers, Reprinted (adapted) with permission from,
94
Copyright (2008) American Chemical Society.
They claimed that Scaffolds of randomly distributed mixed fibers and pore sizes of 100 µm or larger can be produced by adjusting the volume% of fibers. In their fabrication method, Poly (lactic acid)
106
microfiber was positioned in the spinning direction from the needle via a centrally located hole in a rotating disk, on which the collector was placed. A Teflon tube was positioned between the syringe needle and the collector to confirm the collection of nanofibers on the collector and microfiber. The nanofiber-coated microfiber was wound onto a rotating wheel stationed behind the collector and then transformed into a scaffold in a separate chamber (Figure 7). Better chondrocyte cell infiltration and uniform cellular distribution were observed throughout the mixed fibers scaffold after 2 days of culture as compared to controls (Figure 6, right). Table 4 provides a summary of some key reports on electrospinning nanofibers onto single microfibers. Electrospinning setup used to direct the nanofibers towards the microfiber (a) and Scaffolding device (b), redrawn from
94
. Some major approaches to electrospinning nanofibers onto microfibers.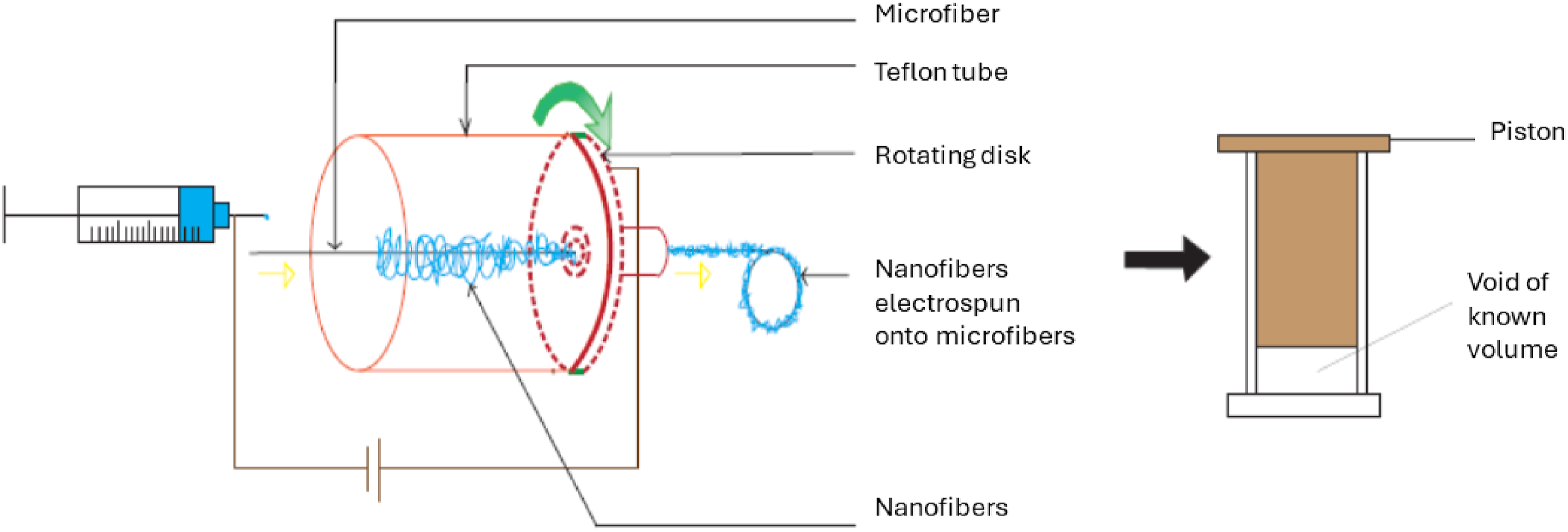

To conclude, electrospinning nanofibers directly onto single microfibers creates highly porous scaffolds with well-distributed nano- and microfibers, allowing adjustable fiber ratios, improved cell infiltration, and uniform cellular distribution, as demonstrated using PLA microfibers coated with PCL nanofibers.
Low temperature electrospinning
Scaffolds with four times higher porosity than standard electrospun scaffolds and with an increased pore size in the thickness direction were achieved by combining ice crystals as a removable void template with low low-temperature electrospinning process. 133 Low temperature electrospinning, along with high relative humidity in the vicinity of the collection drum, ensures ice crystal formation, which consequently results in higher inter-fiber gaps and fibers along the z-direction decrease fiber mesh density. Simonet et al. in their study claimed that LTE may not be scaffolding material specific, as they found similar results for both PLGA and PEU fiber meshes. 134 But Milleret et al. in a separate study found dissimilarities between Polyester Urethane Degrapol (DP) and Poly lactiide-co-glycolic acid (PLGA) scaffolds produced by LTE. They observed a delayed fluorescent bead adsorption in the case of DP scaffolds, which points to more fluffiness in the DP scaffold than in PLGA. 135
More recently, Vieira et al. demonstrated a related approach by embedding glucose crystals within PEO fibers, co-electrospun alongside PLA fibers, to serve as sacrificial porogens. After PEO removal, the resulting scaffold exhibited well-defined, open pore structures (50–150 µm), which notably enabled fibroblast infiltration and colonization throughout the scaffold thickness and maintained structural integrity without triggering inflammatory responses. 136 This pore-engineering strategy complements LTE-based methods and illustrates how combining cryogenic templating with porogen leaching can yield physiologically relevant architectures for enhanced cell infiltration.
Therefore, LTE often combined with ice crystal or porogen templating, produces scaffolds with significantly higher porosity, enlarged pores along the thickness, and well-distributed fibers in the z-direction, enabling enhanced cell infiltration and uniform colonization while maintaining structural integrity.
Rapid prototyping combined with electrospinning
Above-discussed scaffolding techniques lack in mechanical stability and accurate designs when the need for designing complex structures arises, such as scaffolds for heart valves.
137
In this regard, ‘Rapid prototyping’ (RP) can produce computer-designed, highly porous scaffolds of complex architecture with controlled pore size.
59
Among several RP processes, mimicking natural body structure similar to computer tomography can be done by 3D fiber dispensing, where a computer-controlled plotter plots strands of molten polymer layer by layer from an extruder through a movable dispensing needle tip. Although it was thought that RP scaffolds would experience high cell infiltration due to having large, interconnected pores, they failed in terms of initial cell attachment due to a smooth surface and excessively large pore size compared to most cells. Moroni et al. developed Poly(ethylene oxide-terephthalate)/poly(butylene-terephtalate) (PEOT/PBT) scaffold through combined RP and electrospinning technique in a layer by layer assembly manner where macro-scaled (>100 µm) fibers deposited by RP and micro-scaled fibers deposited by electrospinning in alternating manner (Figure 8).
138
A similar study was carried out by Kim et al., who developed a scaffold via this combined approach using Biodegradable PCL.
139
Both studies were able to produce a fibrous 3D scaffold with enhanced cell adhesion and proliferation than single RP scaffolds without compromising mechanical stability. Schematic of rapid prototyping combined with electrospinning technique.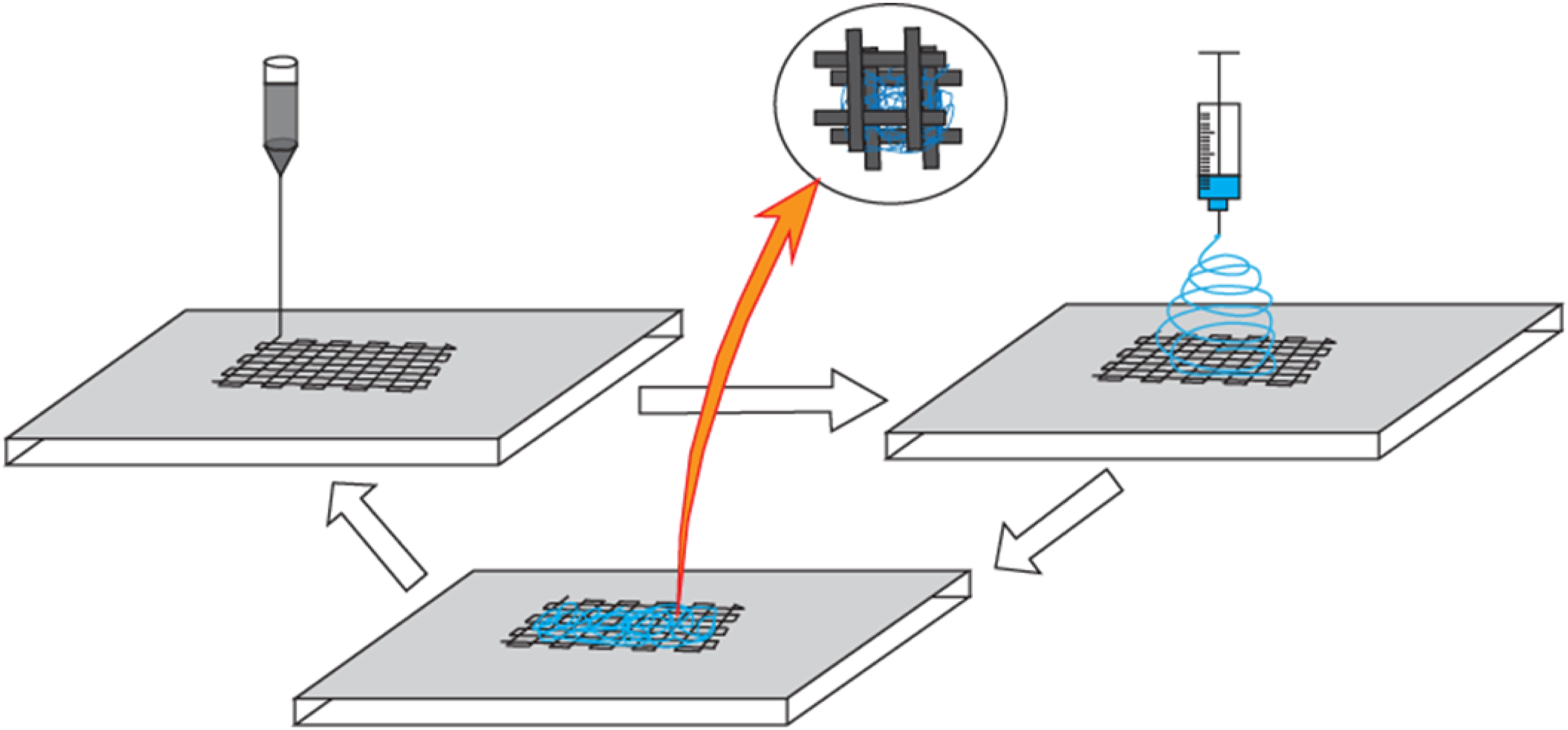
A summary of key material combinations and outcomes based on the combination of RP-electrospinning.

Overall, hybrid scaffolds that combine RP with electrospinning successfully bridge the gap between mechanical stability and biological performance. By leveraging macrostructural control from RP and bioactive micro/nanoscale features from electrospinning, these systems provide promising platforms for tissue engineering applications requiring both strength and functionality.
Porogen method
This method involves the incorporation of particulates such as salt or sugar spheres as a void template into either nanofiber meshes (when combined with electrospinning) or into the polymer solution (solvent casting), and later the particulates are leached out, resulting in a highly porous scaffold with interconnected pores. It is possible to create large pores with controlled pore size depending on the size of the particulate used.
67
Lee et al. proposed a technique combining electrospinning and particulate leaching to fabricate a scaffold with both nano-scale and micro-scale structures.
66
But to increase pore interconnectivity, produced scaffolds must be sintered which in turn may adversely affect polymer fibers. Porogen method in association with phase separation can produce interconnected macropores within the scaffolds without this adverse effect, as particulate spheres can be interconnected by applying a certain amount of heat for a certain time before incorporation of polymer/solvent mix. Such approaches developed previously suffered from poor cell adhesion due to the solid pore wall. In order to create fibrous pore morphology, Porogen leaching can be associated with TIPS (Thermally Induced Phase Separation), where particulate spheres are added onto a mold and a polymer/solvent mixture is cast into the mold and quenched.
143
After phase separation, solvent exchange and particulate leaching are carried out by subjecting the composite to water or other chemicals that are immiscible with the polymer but soluble in solvents and lyophilized. Cheng et al. developed a 3D nanofibrous and microporous scaffold by TIPS (thermally induced phase separation) with salt leaching technique (Figure 9).
67
They found that the micro-porous structure aided cell infiltration and multi-cellular organizations in the pores, and the nano-fibrous structures upheld cell differentiation as well as a more in-vivo-like cell-matrix adhesion. Schematic representation of particulate leaching combined with TIPS.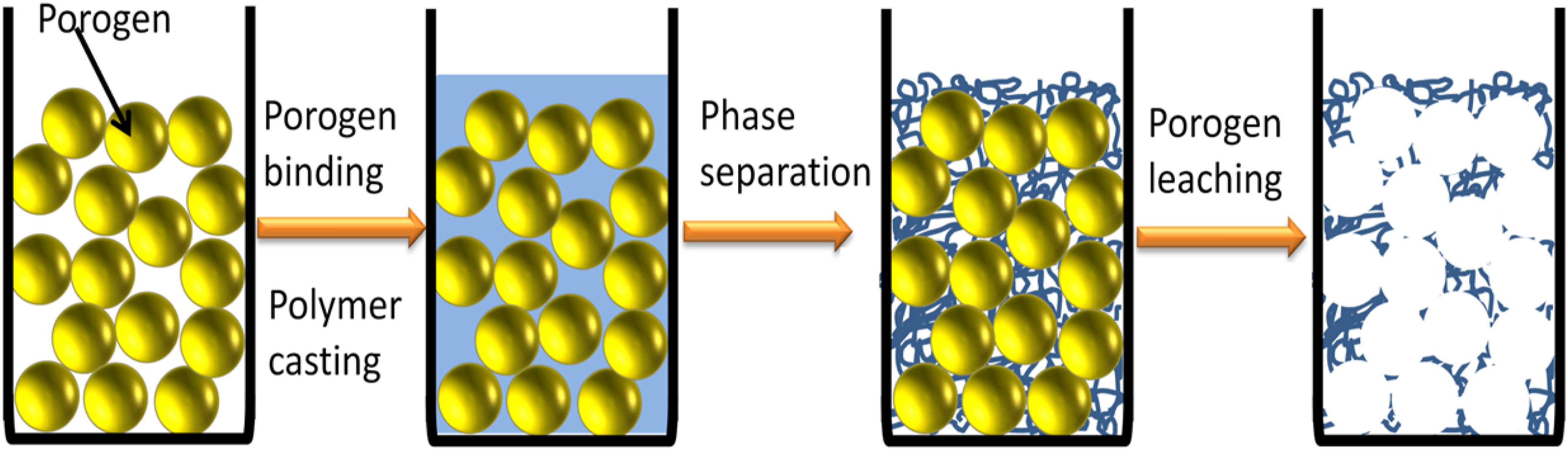
Liumin He et al., who developed PCL-b-PLLA scaffold with a pore size of 144 ± 36 μm through salt leaching and liquid-liquid TIPS, observed abundant filopodia extension, with higher proliferation observed on nano-fibrous walled scaffold. 144 Ma et al. created a 3D macro-porous scaffold with nano-fibrous pore walls by using a technique combining TIPS and paraffin sphere leaching. 145 The pore interconnectivity can also be adjusted by changing heat treatment times of paraffin spheres, as longer heat-treatment times form larger bonding areas between the paraffin spheres, causing larger openings between the macropores of the scaffold. 54 Nevertheless, there are also a few drawbacks of using paraffin spheres, as many organic solvents, e.g., tetrahydrofuran (THF), dioxane, dichloromethane, and pyridine used for polymer dissolution are soluble in paraffin to varying extents. In addition, the presence of a small amount of paraffin in the scaffolds could be unsuitable for the subsequent cell activity because of its hydrophobicity. Hence, paraffin spheres are not appropriate for porogen techniques. Macroporous and nanofibrous PLLA scaffold developed by G. Wei and P. X. Ma using Sugar sphere leaching and TIPS maintained the benefits of interconnected spherical pores, though avoiding the use of paraffin. 146
A summary of key reports on the Porogen technique in tissue engineering.
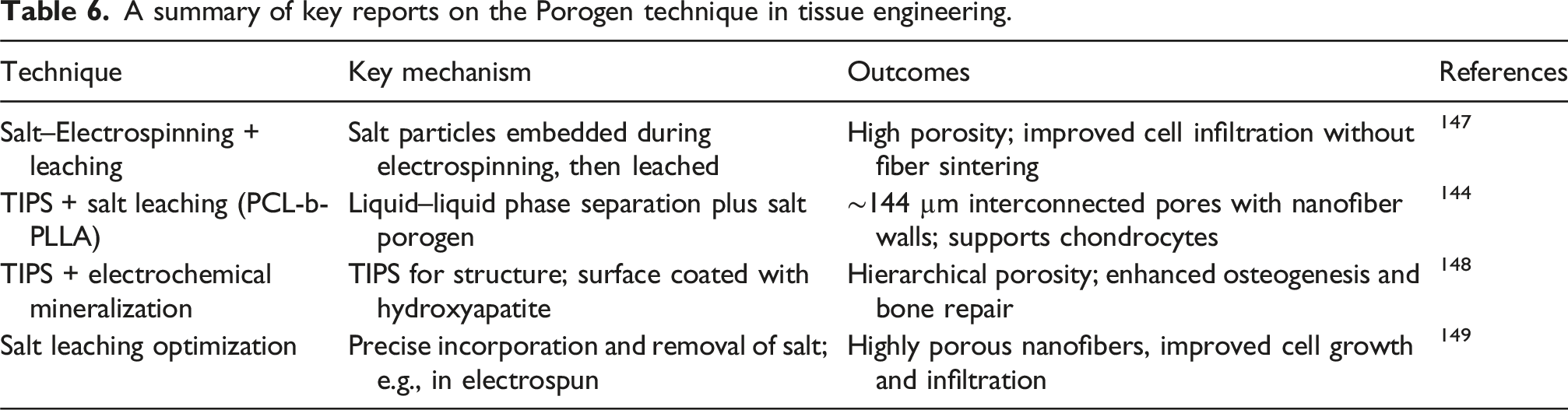
In summary, particulate leaching, often combined with thermally induced phase separation (TIPS) or electrospinning, produces scaffolds with highly interconnected macropores lined with nanofibrous walls, allowing controlled pore size and interconnectivity, which enhances cell infiltration, multi-cellular organization, and in vivo-like cell–matrix interactions while maintaining fiber integrity.
Ultra-low concentration phase separation
Jiang et al. developed an ultra-low concentration phase separation (ULCPS) method to create gelatin ultrafine fibrous scaffolds, where the loose structures of phase-separated (PS) scaffolds with larger pores and weaker fiber entanglement enabled cell infiltration and migration. 150 They found that a lower concentration of gelatin (0.01%) can produce ultrafine fibers, and citric acid crosslinking with a certain amount of catalyst can give stability to the fibers. As compared to typical electrospun gelatin scaffolds, scaffolds produced via ULCPS showed higher cell infiltration. But these PS scaffolds undergo a large amount of shrinkage while soaked in PBS or other medium.
To mitigate the dimensional instability, subsequent studies adapted phase separation methods. For instance, a study used TIPS on gelatin/PLLA blends, yielding scaffolds with interconnected pores and enhanced resistance to shrinkage. 151 Similarly, Perez-Puyana et al. (2020) incorporated chitosan into phase-separated gelatin scaffolds, which improved structural integrity and reduced collapse during aqueous incubation. 152 More recently, strategies such as collagen–gelatin porous scaffold containing zinc-doped bioactive glass-ceramic nanoparticles were developed as a dermal substitute. The scaffold enhanced cell attachment, proliferation, angiogenesis, and significantly accelerated wound closure, demonstrating its potential for effective cutaneous tissue regeneration.
A summary of the key techniques and findings relevant to ULCPS.
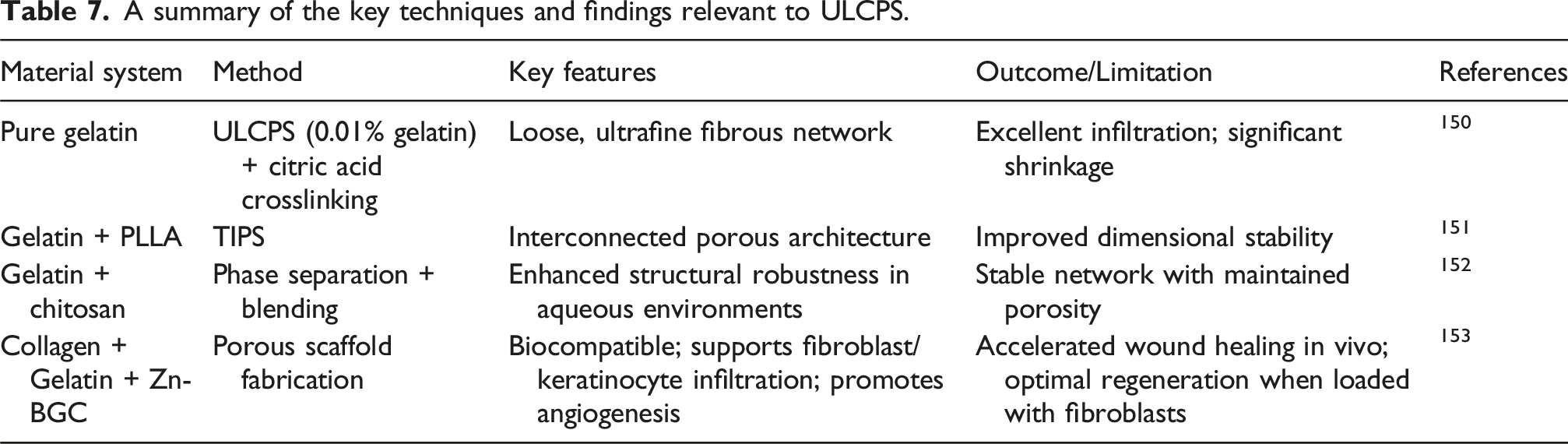
Overall, ULCPS enables the fabrication of ultrafine gelatin scaffolds with enhanced porosity and cell infiltration, closely mimicking native ECM architecture. To ensure their structural integrity and dimensional stability in physiological conditions, however, ULCPS scaffolds must be reinforced through robust crosslinking or blended with complementary polymers.
Polymer–nanoparticle assisted electrospinning for ultra-thin fibers
While ULCPS has enabled the formation of ultrafine fibrous scaffolds, recent research also highlights the potential of polymer–nanoparticle hybrid systems in conventional electrospinning to achieve ultra-thin fibers with tailored properties. Incorporating nanoparticles such as silica, hydroxyapatite, or metallic nanoparticles into polymeric matrices can influence solution conductivity, charge density, and jet stability, all of which are critical parameters for fiber diameter reduction.
For example, Xia et al. (2018) prepared polyvinyl alcohol/silica (PVA/SiO2) hybrid fibers using a sol–gel assisted electrospinning technique for bone tissue engineering applications. At 8% PVA concentration, the fibers exhibited uniform and continuous morphology (∼543 nm diameter) with silica well distributed in the polymer matrix. Upon immersion in simulated body fluid (SBF), the fibers showed strong apatite crystal precipitation, indicating excellent in vitro bioactivity. 154 In another study, Guo et al. designed PLLA scaffolds reinforced with halloysite nanotubes (HNT) modified with in-situ grown SiO2 nano-protrusions (HNT@SiO2). By hydrolyzing tetraethoxysilane (TEOS), irregular SiO2 nano-protrusions were grafted onto HNT surfaces, enhancing dispersion and interfacial adhesion with PLLA. Compared with unmodified HNT, HNT@SiO2 significantly improved scaffold tensile strength, modulus, hydrophilicity, and apatite-forming ability. Furthermore, the scaffolds supported stem cell responses, indicating strong potential in bone tissue engineering. 155 In another study, a ternary nanocomposite of PEEK/PEI reinforced with TiO2 nanoparticles was fabricated via ultrasonication and melt-blending. PEI served as a coupling agent, ensuring homogeneous TiO2 dispersion through π–π stacking and hydrogen bonding. At 4 wt% TiO2, the scaffolds exhibited simultaneous strengthening, stiffening, and toughening, while retaining tensile properties even after steam sterilization or immersion in SBF. The composites demonstrated enhanced thermal stability, dielectric properties, tribological performance, reduced water absorption, and antibacterial activity against both Gram-positive and Gram-negative bacteria. These features highlight their promise for long-term load-bearing implant applications. 156
To conclude, polymer–nanoparticle assisted electrospinning enables the fabrication of ultrathin, bioactive fibrous scaffolds with enhanced mechanical, thermal, and functional properties, where incorporated nanoparticles improve fiber morphology, interfacial adhesion, and cell-material interactions, making these scaffolds particularly suitable for bone tissue engineering and long-term load-bearing implant applications.
Present scenario of cell filtration with scaffold
Cell filtration with scaffolds has come a long way in tissue engineering and regenerative medicine, which has been a significant advancement in the field of biomedical. Recent innovations are mostly focused on designing scaffolds with enhanced porosity and biocompatibility to improve cell filtration efficiency, enabling better nutrient and oxygen diffusion for cells. Lan et al. 157 developed the gelatin-based scaffolds with the main substrate and polyamide layers so that they can make the water channels. That scaffold had the ability to affect the interfacial polymerization so that it could obtain a precise separation layer. Moreover, the membrane demonstrated better high permeability and selectivity.
Techniques like electrospinning, 3D printing, and decellularization are employed to create highly porous and customized scaffolds. Biodegradable and bioactive materials, including collagen, chitosan, and alginate, are widely used due to their ability to promote tissue growth and integration. Erben et al. 158 used 3D printing to develop two-photon stereolithography to print up to mm-sized highly accurate 3D cell scaffolds at a very micrometer resolution. They modified the manufacturing process with two-pass printing and post-print crosslinking, maintaining a variation in Young’s moduli (from 7 to 300 kPa) are printed and quantified through AFM. The effects of different scaffold topographies on the behavior of colonizing cells were studied using mouse myoblast cells and a 3D lung microtissue replica, which was colonized with primary human lung fibroblasts. This method enables a systematic investigation of both single-cell and tissue dynamics in response to specific mechanical and biomolecular signals, with the potential to scale up for application in full organ models. Huang et al. 159 used the biomaterials to improve the functionalities and effect of the therapeutic materials for escalating the process of immunotherapy. They also demonstrated efficient control over surface functionalization, which can impact immune cell modulation. They worked on the development of biocompatible immune cell-engaging particles (ICEp) with synthetic short DNA for fabricating the scaffolds that are capable of controlled and tunable protein loading. They intratumorally injected to ensure the safety of the chimeric antigen receptor (CAR) during T-cell therapies. Moreover, they demonstrated smart signals using an AND-gate on the CAR-T cells. This scaffold unfolded the opportunity for a versatile and modular biomaterial functionalization platform for controlled immunotherapies. Swanson et al. 160 worked on combating craniosynostosis, a severe birth defect related to premature fusion of cranial bones due to early stem cell depletion in the suture tissue between growing bones. They developed biomaterial-based scaffolds that could maintain the stemness of cranial suture cells with an aim to reduce morbidity in craniosynostosis patients. Their findings indicate that the physical properties of synthetic scaffolds influence cell and tissue outcomes. In that study, macro-porous scaffolds with controlled spherical pores were created using a sugar porogen template method. They set up the cell-scaffold constructs in mice for up to 8 weeks and analyzed them for mineralization, vascularization, ECM composition, and gene expression. According to their findings, they showed that pore size affects cell fate: larger pores support bone formation, while smaller pores (<125 μm) maintain stemness and prevent differentiation. Similar outcomes were observed in vitro, linking scaffold pore geometry to differential cell and tissue fate. This suggests that scaffold pore size is a crucial factor in regulating mesenchymal cell fate, offering a new approach to control tissue regeneration and develop stem cell niches both in vivo and in vitro. Research from diverse applications is running current days. The scaffolds possess a variety of functionalities to keep them rolling in different applications.
Overall, cell filtration using scaffolds has advanced significantly in tissue engineering and regenerative medicine, with current research focusing on highly porous, biocompatible scaffolds that improve nutrient and oxygen diffusion, regulate cell fate, and enable precise control over cell behavior; techniques like electrospinning, 3D printing, and decellularization, combined with bioactive materials and innovative designs such as macro-porous structures, functionalized surfaces, and modular biomaterial platforms, are being used to optimize cell infiltration, tissue formation, and therapeutic applications.
Comparative study of performance of different scaffold fabrication method
Comparative study on different fabrication methods of the scaffolds.
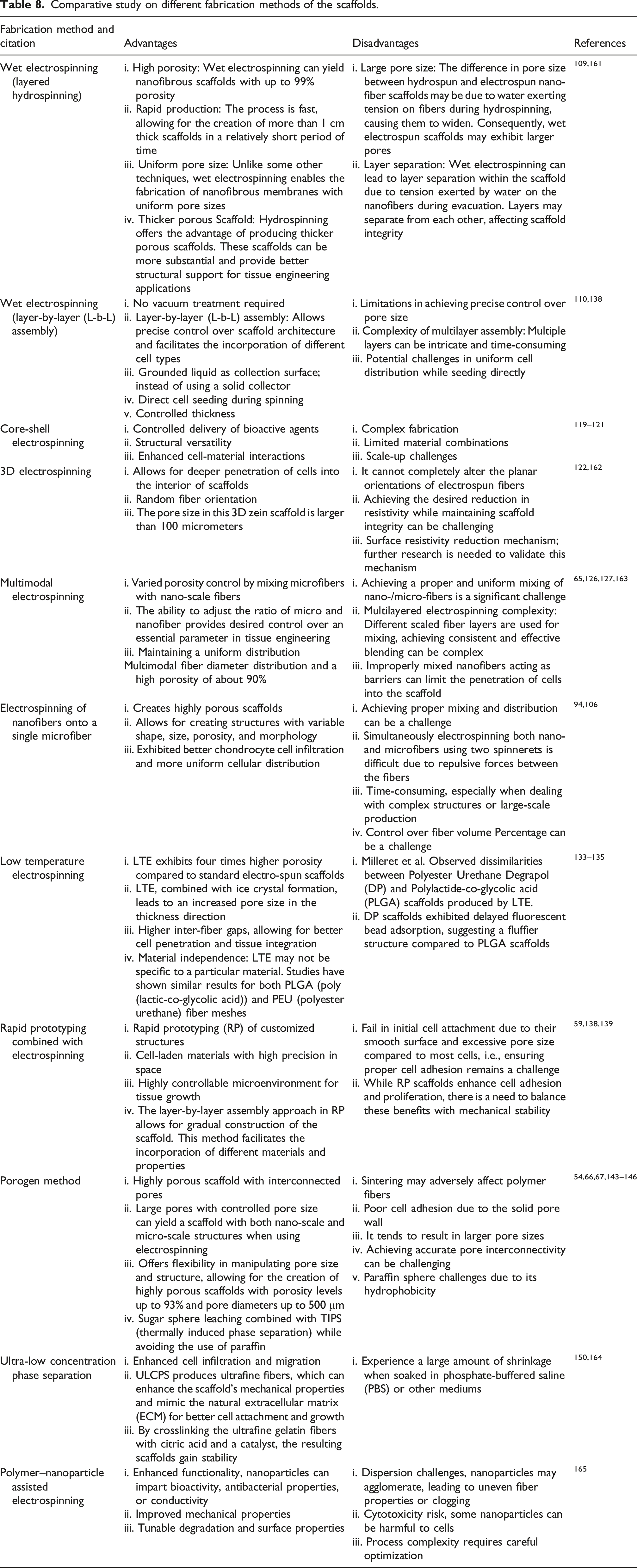
The fabrication of fibrous scaffolds for high cell infiltration is crucial for tissue engineering and regenerative medicine applications. The best method for cell filtration depends on the specific application and requirements, such as the type of cells, the desired purity, the volume of the sample, and whether the cells need to remain viable after filtration. Wet electrospinning (Layered hydrospinning) offers high porosity (up to 99%) for efficient cell filtration, rapid production for high throughput, uniform pore size for consistent filtration, and thicker scaffolds for better structural support, 109 making it the best option when these features are needed. Low-temperature electrospinning (LTE) boasts superior porosity compared to standard methods, increased pore size for better cell penetration, and material independence for broader applicability,133–135 making it the best option when these features are needed. Multimodal electrospinning offers tunable porosity but can be challenging to achieve uniform distribution.65,126,127 Electrospinning nanofibers onto a single microfiber creates highly porous structures, but achieving proper mixing can be difficult.94,106 The porogen method allows for large, controlled pores but may require additional steps for cell adhesion.54,66,67,143–146 Ultra-low concentration phase separation (ULCPS) enhances cell infiltration but experiences shrinkage in certain mediums.150,164
Perspective, summary, and challenges
For high cell infiltration, future research should be conducted on preparing scaffolds using composite materials (as the use of both synthetic and natural biomaterials has shown better cell adhesion and mechanical stability to the scaffold 51 ), either via those scaffolding routes represented in this paper or by introducing new approaches. Nowadays, the use of scaffolds is not limited to skin, bones or cartilage regeneration; its use has expanded to complex tissue engineering fields where aligned fibers or pores for proper cell-matrix interaction are necessary, such as aligned ECM fibrils in tendons, vascular and neural tissues. 166 Many researchers tried to orient scaffold components to the cell supportive direction such as aligning the electrospun nanofibers167,168 or creating unidirectional tubular pores by applying directional temperature gradients while phase separating. 169 But both efforts limit cell infiltration because the earlier effort reduces inter-fiber gaps i.e., decreased pore size, resulting in poor cell ingrowths, and the latter realizes poor cell adhesion due to solid pore walls, resulting in early cell death. Hydro spinning can create uniform cell distribution while seeded during spinning, but might not ensure proper nutrient transport to the cell in the middle of the scaffold due to its layer-by-layer assembly. So, efforts should be given to modify these scaffolds to improve cell viability and distribution by ensuring rapid cell infiltration and efficient nutrient transport. Scaffold surfaces produced via particulate leaching and TIPS can be further modified to be more cell supportive, as different cell types show varying degrees of adhesion and proliferation rates on constructs with different surface topographies.170–172 For example, on a surface with gradient roughness, osteoblasts show a significantly increased proliferation rate with the rise of surface roughness while fibroblasts show the contrasting trend in proliferation rate relative to surface roughness, i.e., slower rate with an enhanced roughness. 172 Designing porous scaffolds with partly nanofibrous and somewhat smooth domains through combined particulate leaching and TIPS may perform as promising 3D matrices for co-culturing diverse cells that prefer varied surface architectures. 173 Efforts should be made to allow active control over substrate properties by generating dynamic gradient substrates for studying directed cell migration. A recent study found that programmed erasure of substrate topography using shape memory polymer causes a rise in angular dispersion with corresponding remodeling of the actin cytoskeleton. 174 The seeding method also has a significant impact on cell distribution throughout the scaffold. For example, Thevenot et al. claimed that cells seeded with orbital shaker result in much more uniform cell distribution throughout the scaffold than any other seeding method. 175 Despite having an ultrafine fibrous structure, all those techniques discussed in this paper either address high cell infiltration or sacrifice mechanical properties. Thus, in the future, efforts should be made to develop methods for producing ultrafine fibrous scaffolds with good mechanical properties that can ensure high cell infiltration.
However, to create a native vivo-like 3D environment, only the preparation of 3D scaffolds is not enough. Ensuring high cell infiltration is a must. For better cell penetration, designing ultrafine fibrous scaffolds with highly porous architecture and proper control over ‘pore size’ and ‘pore interconnectivity’ is obligatory. Fibrous structures, particularly nanofibers, are necessary for proper cell adhesion and migration. Scaffold porosity in particular controls cell dissemination, nutrient supply to cells, metabolite diffusion, local pH stability and cell signaling where the pore size can have an effect on how close the cells are at the early stages of tissue growth (allowing for cell-cell communication in three dimensions) and also manage the amount of space necessary for the cells to have 3D organization in the late stages of tissue regeneration. Pore interconnectivity, on the other hand, ensures proper nutrient delivery inside the scaffold and thus prevents unexpected cell death before expected cell differentiation. So, before designing a scaffold, one should address all these factors discussed here for an effective native ECM environment.
To sum up, future scaffold design for high cell infiltration should focus on composite materials combining synthetic and natural biomaterials, optimized fiber alignment, and controlled pore architecture (size and interconnectivity) to support cell adhesion, migration, and 3D organization, while also considering surface topography, dynamic gradient substrates, and effective seeding methods to balance mechanical properties with nutrient transport and cell viability, ultimately creating scaffolds that closely mimic native ECM environments.
Conclusions
The development of ultrafine fibrous scaffolds with enhanced cell infiltration remains a crucial challenge in tissue engineering. While nanofiber-based scaffolds provide excellent cell adhesion and mimic the extracellular matrix, their dense architecture often limits deep cell penetration. Various fabrication strategies, such as electrospinning, phase separation, and particulate leaching, have been explored to optimize scaffold porosity and interconnectivity. However, many approaches either improve cell infiltration at the expense of mechanical stability or enhance mechanical properties while restricting cell migration. Future research should focus on hybrid scaffold designs incorporating both natural and synthetic biomaterials to balance mechanical strength and bioactivity. Additionally, dynamic scaffold architectures with controlled pore size gradients and shape-memory polymers could provide adaptive microenvironments for improved cell distribution. Advanced seeding techniques, including bioreactors and orbital shaking, may further promote uniform cell infiltration. Ultimately, for scaffolds to successfully replicate native tissue environments, a multifaceted approach integrating material science, bioengineering, and cell biology is essential. By addressing the interplay between fiber morphology, pore structure, and cell-material interactions, researchers can develop next-generation scaffolds that enhance tissue regeneration outcomes. Future efforts should focus on scalable, reproducible fabrication techniques to translate these innovations into clinical applications.
Footnotes
Author contributions
S M Kamrul Hasan: Conceptualization, Writing– original draft, Writing – review & editing, Visualization, Prosenjit Sen: Writing– original draft, Writing – review & editing. Habibur Rahman Anik: Writing– original draft, Writing – review & editing. Md. Redwanul Islam: Visualization. Mowshumi Roy: Writing– original draft, Writing – review & editing. Toufique Ahmed: Writing– original draft, Writing – review & editing. Abu Naser Md Ahsanul Haque: Writing– original draft, Writing – review & editing, Visualization.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
