Abstract
The reparative properties of amniotic membrane allografts are well-suited for a broad spectrum of specialties. Further enhancement of their utility can be achieved by designing to the needs of each application through the development of novel processing techniques and tissue configurations. As such, this study evaluated the material characteristics and biological properties of two PURION® processed amniotic membrane products, a lyophilized human amnion, intermediate layer, and chorion membrane (LHACM) and a dehydrated human amnion, chorion membrane (DHACM). LHACM is thicker; therefore, its handling properties are ideal for deep, soft tissue deficits; whereas DHACM is more similar to a film-like overlay and may be used for shallow defects or surgical on-lays. Characterization of the similarities and differences between LHACM and DHACM was conducted through a series of in vitro and in vivo studies relevant to the healing cascade
Introduction
The evolution of mammalian reproduction gave rise to the amniotic sac, a biologically complex and dynamic barrier membrane to protect and nourish a developing fetus within the mother. A successful pregnancy is in-part a result of the amniotic sac’s ability to facilitate the transport nutrients, protect against unwanted substances and infections and regulate the immune response at the feto-maternal interface.1–3 The amniotic membrane is a multi-layered matrix comprised of amnion, intermediate layer, and chorion layers. The amnion is the innermost membrane to the fetus, providing tensile strength and acting as a fibrous skeleton and barrier.
4
Toward the maternal side is the chorion, which includes the reticular layer, a pseudobasement membrane, and trophoblast layer that contacts the maternal decidua.
4
The chorion contributes to elasticity and stability and provides a scaffold for native cells.1,4–6 The amnion and chorion attach and interact through a third, middle layer called the intermediate layer (IL), also referred to as the spongy layer or zona spongiosa
Recognition of the unique structure and inherent functions of amniotic tissue prompted a surge in amniotic-derived allografts with the intent of capturing these properties for subsequent clinical application for the treatment of hard-to-heal acute and chronic wounds. The immune-privileged properties of the amniotic sac proved to be the key differentiator from other allogenic or xenogenic tissue sources. The tissue is well-tolerated by an unmatched recipient without the need for decellularization, which can deplete the tissue of key nutrients and signalling proteins.8,9 In 1910, John Davis used amniotic membrane for therapeutic purposes like skin grafts in some skin injuries; later, amniotic membrane was used in various wounds like burns, diabetic foot ulcers, venous ulcers, ocular injuries, and uterine adherences.10–14 Today, amniotic membranes are also thought to improve incisional healing outcomes including in uses where repeated surgical access is required by reduction of fibrotic adhesions.15–17 Additional reports demonstrate that amniotic membranes support wound healing and reduce wound complications, such as surgical site infection, while aiding function post-surgery.15,18,19 The clinical success of amniotic membrane allografts is related to the processing techniques that preserve the membrane properties, including the biophysical structure and bioactivity, while making them safe and readily accessible for the provider.9,20
This study looks to evaluate two unique PURION® processed amniotic tissue allografts to characterize the material properties and the biological activities that are therapeutically relevant to wound healing. Lyophilized human amnion, intermediate layer, and chorion membrane (LHACM) is a freeze-dried tri-layer allograft; whereas, dehydrated human amnion, chorion membrane (DHACM) is an air-dried bi-layer allograft. These unique configurations were developed with the specific intent of creating unique handling properties with similar efficacy. The inclusion of the intermediate layer in conjunction with lyophilization was hypothesized to contribute to the overall thickness of the resultant allograft, but minimally to the biological activity due to the diffuse composition. Previous work with DHACM demonstrated that this patented and proprietary process retains well known regulatory proteins inherent to amniotic tissues, and preserves the bioactivity to stimulate cellular activities, such as proliferation, migration and biosynthesis in multiple cell types.21–26 A compositional analysis of LHACM was performed using immunofluorescence and multiplex enzyme-linked immunosorbent assay (ELISA) for growth factor and regulatory protein content for comparison. Material properties were characterized through histology, thickness measurements and diffusivity through the barrier membrane using size-exclusion equilibrium dialysis. In vitro cellular response was assessed in relevant cell types using cell proliferation, migration, and metabolic assays. Finally, the in vivo cellular response was assessed by evaluation of cell infiltration/ingrowth and tissue remodeling in a mouse subcutaneous implantation model. This study demonstrates the potential for versatility in the use of amniotic membrane allografts in a clinical setting while providing a similar therapeutic impact on tissue repair.
Materials and methods
Human amnion/chorion membrane
Recovered birth tissue, comprised of the placental disc, amniotic sac and umbilical cord, is placed into 18% sodium chloride storage solution under refrigerated conditions. Using aseptic techniques, the amniotic sac is removed from the storage solution and dissected away from the placental disc and further separated into the individual amnion and chorion layers, with attached intermediate layer. Note, the intermediate layer is a jelly-like substance and is evident on the inside facing surface of the amnion and chorion, or the stromal and reticular side respectively. Each layer is subjected to a series of 60-90 min washes with gentle agitation in a hyper-isotonic solution comprised of sodium chloride. Subsequent rinses include an overnight soak in an antibiotic solution and several washes to remove processing reagents, including antibiotics. Remaining blood clots are gently removed from each layer using tweezers.
For DHACM, the intermediate layer is mechanically removed from either side of the amnion and chorion layer using a gentle scraping technique. The layers are reapproximated in their natural orientation and air-dehydrated at room temperature under controlled conditions. For LHACM, the intermediate layer is left intact, and the layers are oriented in a stainless steel tray such that the intermediate layer is sandwiched between the amnion and chorion. A mesh overlay ensures the tissue remains flat during dehydration. The fixture is transferred to a pre-chilled, −40°C, shelf within the GEA lyophilizer unit (FCM-75; GEA Columbia MS). Primary drying is performed at −30°C and 200 mT before moving into secondary drying at 48°C and the vacuum is held at 150 mT. Following dehydration the tissue is cut to size, packaged and terminally sterilized using e-beam irradiation (Sterigenics Oak Brook, IL) Figure 1.27–30 Cross-sectional schematic of amniotic membrane allografts DHACM and LHACM.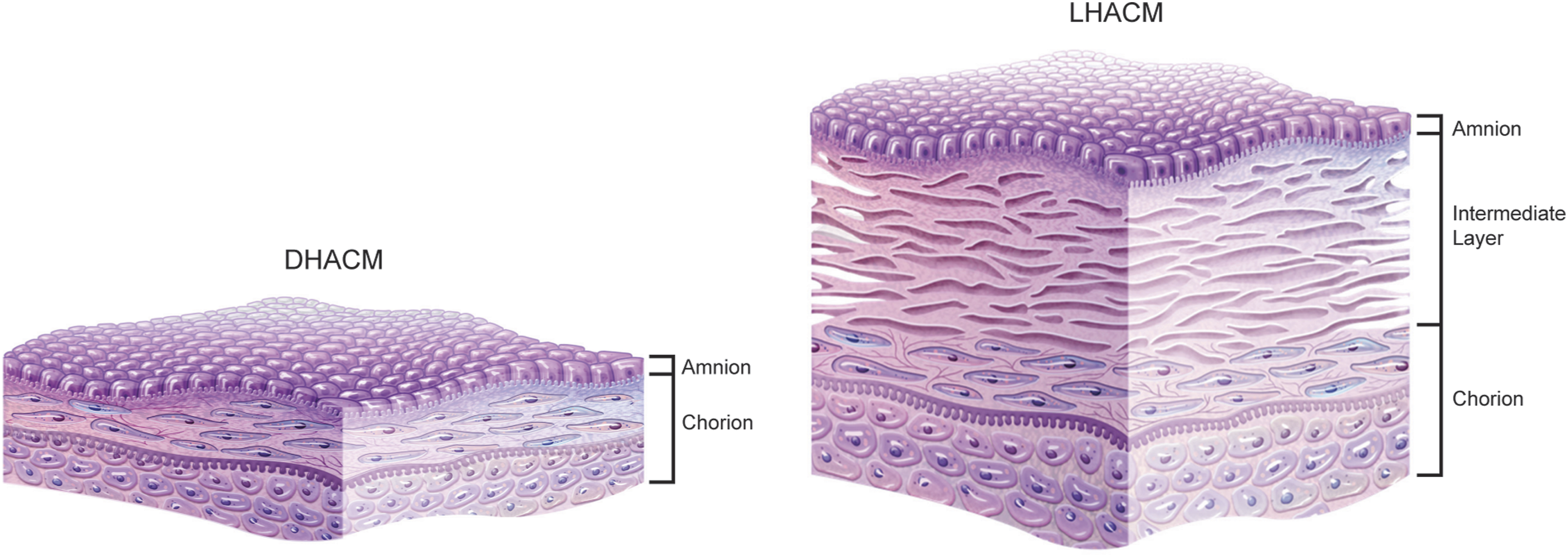
Extract preparation
To prepare soluble extracts of LHACM and DHACM for cell culture experiments, representative samples of amniotic membrane from individual donors of LHACM or DHACM were mechanically minced to an approximate size of 5 mm × 5 mm and extracted overnight at 40 mg of tissue per milliliter of basal DMEM. For experiments with human dermal fibroblasts (HDF, Thermo Fisher Scientific, Waltham, MA), basal media is defined as Dulbecco’s Modified Eagle’s Medium (DMEM) (Thermo Fisher Scientific, Waltham, MA) containing 1% penicillin/streptomycin (Thermo Fisher Scientific, Waltham, MA) and 1% sodium pyruvate (Thermo Fisher Scientific, Waltham, MA). For experiments with human microvascular endothelial cells (HMEC1, ATCC, Manassas, VA), basal media is defined as MCDB 131 medium (Corning, Glendale, AZ) containing 1% Gluta-gro (Corning, Glendale, AZ), and 1% penicillin/streptomycin. The tissue residue was removed by centrifugation and the resultant fluid was passed through 0.22 µm filter (Millipore Sigma, Burlington, MA). The filtrate was collected in a sterile container to serve as the extract for treatment. Prepared extracts, from individual LHACM and DHACM donors, were then diluted in the basal media to the desired testing concentration.
Histology
LHACM or DHACM samples were sent to Premier Laboratory (Longmont, CO) for processing. Briefly, samples were fixed in 10% formalin and subjected to routine paraffin processing, followed by sectioning into 5 µm thick cross-sections. All sections were mounted in the center of a positively charged, coated slide (Tanner Scientific Sarasota, FL). Slides were air-dried overnight at room temperature and baked at 60°C for 30 min prior to hematoxylin and eosin staining (H&E). Slides were processed on a Sakura Tissue-Tek Prisma Automated Slide Stainer (Sakura Torrance, CA) at room temperature. Staining runs combined both commercial reagents and solutions constituted in-house. All staining runs employed hematoxylin-normal strength (Anatech Battle Creek, MI) and eosin Y, alcoholic (Anatech Battle Creek, MI). Coverslips were applied over stained sections using a Sakura Tissue-Tek Glas cover-slipper (Sakura Torrance, CA). Following routine H&E staining, images were captured with a Leica microscope (DM6 B; Leica Microsystems Deerfield, IL) fitted with a 20x objective.
Thickness measurements
Thickness of 15 grafts, from three different donors of DHACM and LHACM were evaluated using digital calipers (KIT19009; VBX Ball Bearings Anaheim, CA). Three measurements were randomly distributed across each graft were taken.
Membrane permeability
Grafts of LHACM and DHACM (2.5 cm × 2.5 cm) were hydrated in PBS and placed in the dialysis chamber (403170000; SP Bel-Art Wayne, NJ). A volume of 1 mL of a standard molecular weight marker (Thermo Fisher Scientific, Waltham, MA) or phosphate buffered saline (PBS) was added to the cavity on each side of the dialysis chamber with the molecular weight marker being added to the side of the chamber facing the amnion layer of the graft (Figure 2(d)). The dialysis chamber was incubated at 4°C, for 3 days, allowing for diffusion of the various proteins within the molecular weight marker through the grafts. The samples (500 µL) containing the diffused protein were centrifuged in a Pierce™ PES protein concentrator (Thermo Fisher Scientific) with a molecular weight cut-off of 3K at 12,000 g for 30 min. The filtrate, containing the concentrated proteins, was evaluated using 4%–12% Bis Tris Sodium dodecyl-sulfate polyacrylamide (SDS-PAGE). Characteristics of human amniotic membrane allografts. Tissue structure of (a) LHACM and (b) DHACM allografts was visualized using H&E staining and images were captured with a Leica microscope (DM6 B; Leica Microsystems Deerfield, IL) fitted with a 20x objective. (c) Thickness measurements were taken at three different locations along each graft. Five grafts from three different donors were assessed; values are represented as the mean ± the standard deviation (d) Assessment of barrier properties demonstrates that LHACM and DHACM both function as effective barriers to the diffusion of high molecular weight proteins. Left: Experimental apparatus diagram depicting the setup of the dialysis chamber. Figure realized with BioRender.com. Center: Image showing fully assembled dialysis chamber, amnion side. Right: Representative image of SDS-PAGE analysis of permeable proteins that moved into the chamber with PBS after 3 days of dialysis. Scale bar = 100 μm.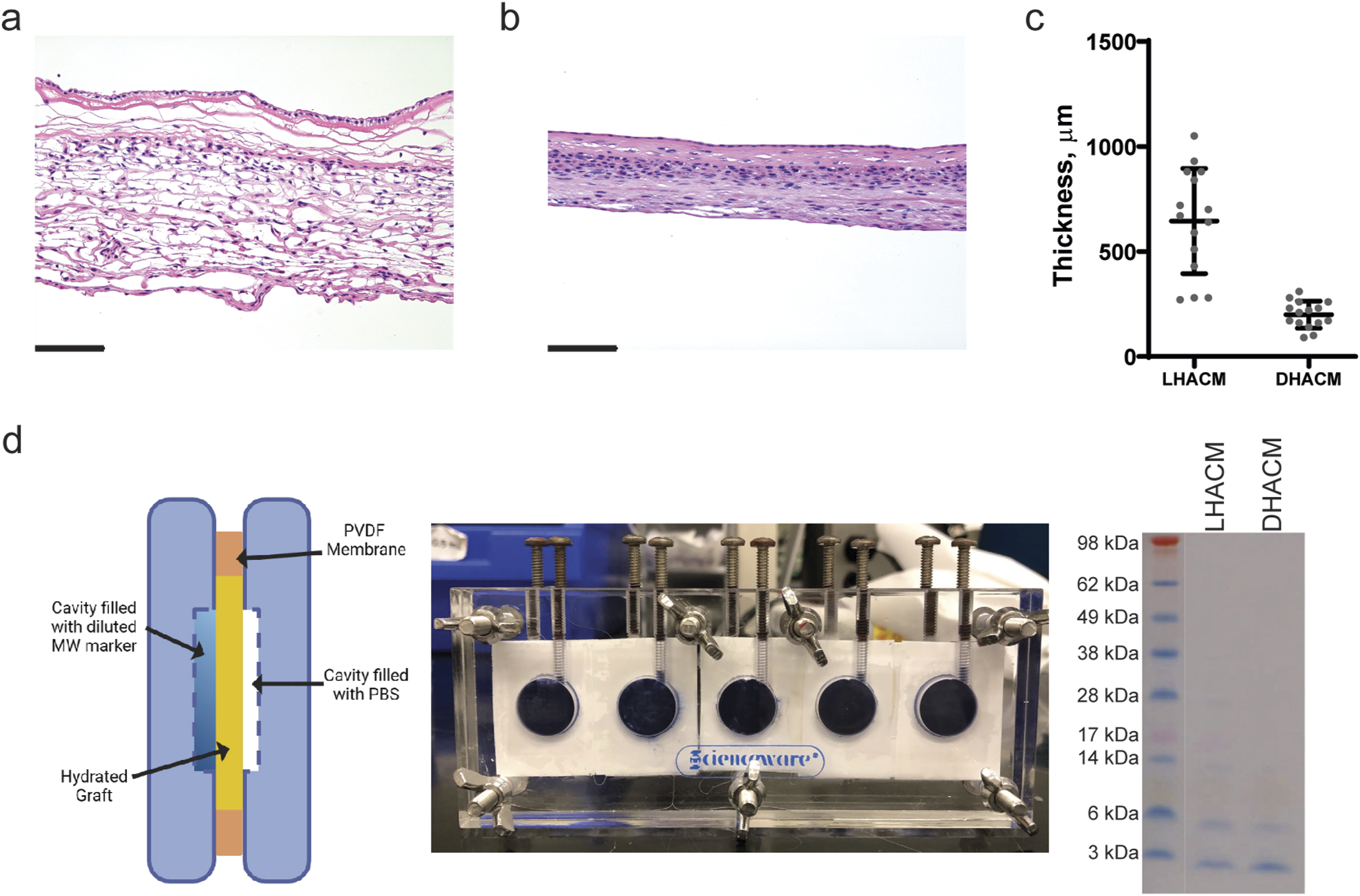
Soluble protein characterization
The LHACM samples (n = 5 donors) were submitted to RayBiotech (Norcross, GA) for analysis. Upon receipt, tissues were minced, weighed, and incubated in lysis buffer containing protease inhibitor at an extraction concentration of 35 mg/mL with gentle agitation overnight at 4°C followed by mechanical homogenization. Lysates were centrifuged at 14,000 r/min for 15 min. Protein concentration was determined and samples were diluted with assay diluent to a testing concentration of 1,000 µg/mL and analyzed on a multiplex ELISA Quantibody® array (Q640 Human Cytokine Antibody Array, RayBiotech). Briefly, slides were blocked with 100 µL of Sample Diluent for 30 min at room temperature. Samples and standards (100 µL) were added to the appropriate slide wells and arrays were incubated at room temperature for 1-2 h. Following wash steps, arrays were incubated with the detection antibody at room temperature for 1-2 h. Fluorescence detection was achieved through labelling of array with Cy3 equivalent dye-streptavidin and visualized through use of a laser scanner equipped with Cy3 wavelength. The Quantibody® array kit assays a total of 640 biomolecular analytes including cytokines, chemokines, receptors, matrix proteins, proteases, and protease inhibitors. Data analysis was performed by RayBiotech. Further analysis, performed by MIMEDX, classified the detected analytes into categories based upon their primary function as defined by RayBiotech and/or literature review.
Human dermal fibroblast migration in vitro
HDFs (Thermo Fisher Scientific, Waltham, MA) were plated at confluence (13,000 cells/well) in a 96-well Image-Lock plate (Sartorius, Ann Arbor, MI) and serum-deprived overnight in DMEM containing 1% fetal bovine serum (FBS, Thermo Fisher Scientific, Waltham, MA) 1% penicillin/streptomycin (Thermo Fisher Scientific, Waltham, MA) and 1% sodium pyruvate (Thermo Fisher Scientific, Waltham, MA) at 37°C and 5% CO2. The following day, a standardized cell-free lane (scratch) was created in each well using a WoundMaker tool (Sartorius, Ann Arbor, MI). The cell-free lanes were washed with basal medium to remove cell fragments and remaining serum and subsequently treated with one of the following: basal medium without serum (negative control), complete medium with 10% FBS (positive control), and extracts at 10 and 1 mg/mL (n = 3 donors; three technical replicates). Extracts were prepared as previously described. The plates were transferred to the IncuCyte S3 imaging system (Software Version 2021A; Sartorius, Ann Arbor, MI) and imaged every 24 h for a total of 120 h. Percent wound confluence was determined using a pre-defined image processing algorithm to consistently identify HDF migration and wound closure.
In vitro proliferation of human dermal fibroblasts
HDFs were plated at 2,500 cells per well of a 96-well plate in DMEM containing 10% FBS, 1% penicillin/streptomycin (Thermo Fisher Scientific, Waltham, MA) and 1% sodium pyruvate (Thermo Fisher Scientific, Waltham, MA) and cultured at 37°C and 5% CO2 for 24 h. After 24 h incubation to allow for cell adhesion, the cells were washed with basal medium and treated with extract at 10 and 1 mg/mL concentrations (n = 4 donors; three technical replicates). After culturing with treatments at 37°C and 5% CO2 \for 72 h, the cells were washed with PBS to remove treatment and cellular debris prior to performing a CyQuant assay (Thermo Fisher Scientific, Waltham, MA) to quantify the number of adherent cells based on cell number standard curve. The fluorescent signal was measured using a fluorescence Biotek Synergy microplate reader (SMATBL; Agilent Technologies, Inc. Santa Clara, CA) using an excitation wavelength of 480 nm and emission wavelength of 520 nm.
Metabolism
HDFs were plated at 15,000 cells per well of a 96-well plate in DMEM containing 10% FBS, 1% penicillin/streptomycin (Thermo Fisher Scientific, Waltham, MA) and 1% sodium pyruvate (Thermo Fisher Scientific, Waltham, MA) and cultured for 24 h at 37°C and 5% CO2. After 24 h incubation to allow for cell adhesion, the cells were serum starved for 24 h in basal DMEM. The cells were cultured with extract at 10 and 1 mg/mL concentrations (n = 3 donors; two technical replicates) at 37°C and 5% CO2. After 48 h treatment,
In vitro cell invasion
Endothelial cell invasion was evaluated using the IncuCyte® Chemotaxis Cell Invasion Assay (Sartorius, Ann Arbor, MI). HMEC1 cells (ATCC, Manassas, VA) were combined with Reduced Growth Factor Matrigel (Corning, Glendale, AZ) and added to the IncuCyte® Clearview insert. The Matrigel was allowed to polymerize at 37°C 5% CO2 for 45 min. Extract was used as the chemoattractant and added to the wells of an IncuCyte® Clearview reservoir plate (n = 3 donors; three technical replicates). Basal media (MCDB 131 medium containing 1% Gluta-gro, and 1% penicillin/streptomycin) and complete media (MCDB 131 medium containing 1% Gluta-gro, 1% penicillin/streptomycin, 10% FBS, 10 ng/mL EGF (Thermo Fisher Scientific, Waltham, MA), and 1 µg/mL hydrocortisone (Millipore Sigma, Burlington, MA)) were used as the negative and positive controls, respectively. The assay was conducted at 37°C 5% CO2 for 24 h. The plates were transferred to the IncuCyte S3 imaging system (Software Version 2021A; Sartorius, Ann Arbor, MI) and imaged every 6 h for a total of 24 h. Invasion was assessed by quantifying total area of ‘objects’ (cells) located on the top surface and the bottom surface of the Clearview membrane. Automated image processing was performed with the Chemotaxis Analysis module (Sartorius, version 2021A) that utilized a pre-defined image processing algorithm. The metric of “Total Phase Object Area Normalized to Initial Top Value” was calculated at each time point by dividing the total area of cells on the bottom surface of the membrane by the initial cell area of the top surface of the membrane.
In vivo mouse model
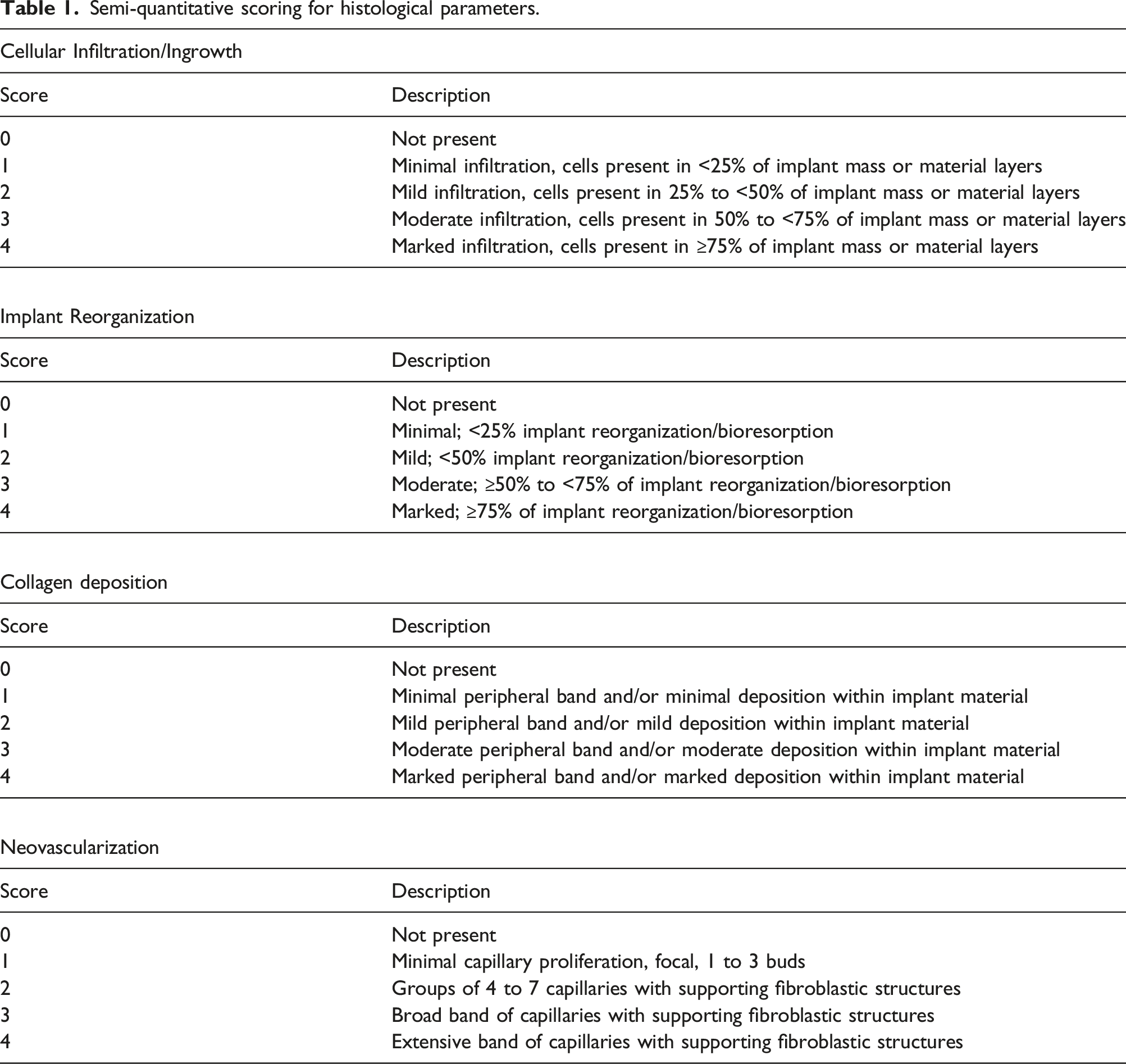
Semi-quantitative scoring for histological parameters.
Immunofluorescence
Immunofluorescence was performed on formalin-fixed paraffin-embedded sections. Briefly, sections were deparaffinized, subjected to antigen retrieval followed by blocking in Serum-Free Protein Block (Agilent, Santa Clara, CA) for 1 hour at room temperature. Incubation with primary antibody against human-specific collagen type IV, human-specific collagen type I, mouse-specific collagen type I, and CD31 (Abcam, Waltham, MA) in Antibody Diluent (Agilent, Santa Clara, CA) was carried out overnight at 4°C for in vivo tissue sections. For LHACM and DHACM tissue sections primary antibodies against human-specific collagen type I and type IV (Abcam, Waltham, MA) were utilized. Following incubation, slides were washed in 1X PBS for a total of 75 min. For visualization, cells were incubated with Goat anti-Mouse IgG (H + L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 488 and Goat anti-Rabbit IgG (H + L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor™ 647 (Thermo Fisher Scientific, Waltham, MA). Following washes in 1X PBS, slides were mounted utilizing VectaShield containing DAPI (Vector Laboratories, Newark, CA) to identify the nuclei. Images were acquired on a Leica microscope fitted with 10x and 40x objectives, using Leica Application Suite Advance Fluorescence software and the THUNDER Imager (Leica Microsystems Deerfield, IL).
Statistical analysis
All values are reported as mean ± standard deviation and statistical analyses were performed using GraphPad Prism software (version 9.5.1). Values were compared using a one-way ANOVA. For each ANOVA, pairwise comparisons were made using a Tukey test. Significant difference was assigned when p < 0.05.
Results
Compositional assessment and growth factor content
A histological comparison of PURION® processed LHACM and DHACM revealed the individual layers comprising the grafts. The tri-layer composition of LHACM is approximately three-times thicker than the bi-layer, DHACM. (Figure 2(a)–(c)). Assessment of barrier properties demonstrates that LHACM and DHACM both function as effective barriers to the diffusion of high molecular weight proteins, but permissive to diffusion of proteins 6 kDa or smaller. Minor diffusion of proteins ranging from 6-28 kDa was observed with LHACM (Figure 2(d)).
Immunofluorescent analysis identified matrix components, collagen type I and collagen type IV, in LHACM and DHACM (Figure 3(a)). Type I collagen is primarily localized in the amnion and intermediate layer (LHACM only); while collagen type IV localized to the amnion basement membrane and the chorion layer. Positive DAPI staining throughout both grafts, indicates the presence of intact, but non-viable cells retained in LHACM and DHACM. Characterization of human amniotic tissue allografts. (a) Immunofluorescence staining of LHACM (top) and DHACM (bottom) grafts including type I collagen (red), type IV collagen (green) and cellular nuclei (blue). Type I collagen localized to the amnion and intermediate layer (LHACM only); while collagen type IV localized to the amnion basement membrane and the chorion layer collagen. (b) Protein composition of LHACM. Proteins present in DHACM and LHACM were evaluated using the Human Cytokine Array Q640 (RayBiotech). Total protein from five DHACM and five LHACM tissue grafts were assayed following manufacturer’s protocol. For each category, percentages reported represent the number of proteins in each category normalized to the total number of proteins detected. n = 5; Scale bar = 100 μm.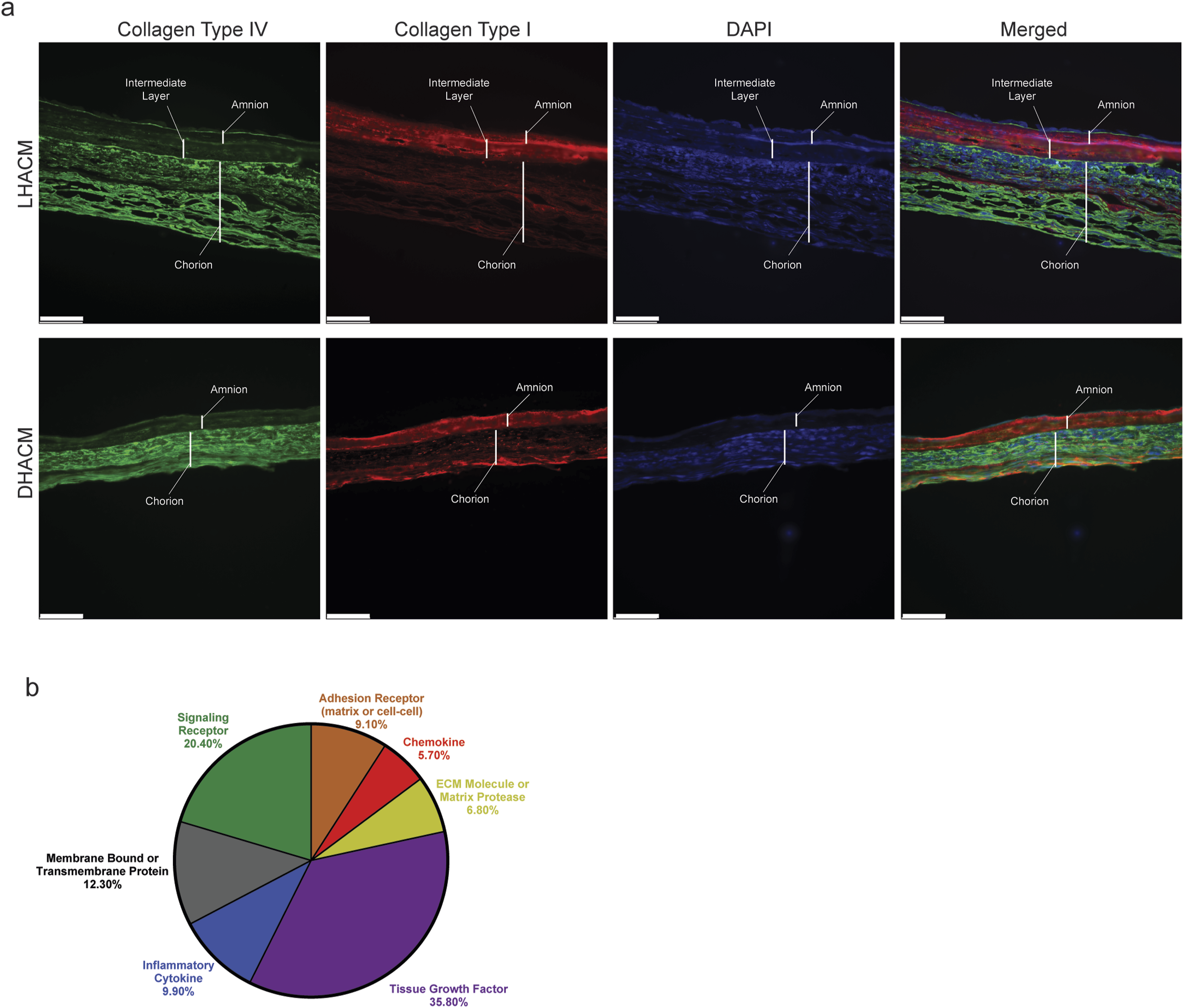
The Q640 multiplex ELISA Quantibody® array was used to detect growth factors, cytokines, receptors, proteases and inhibitors, transmembrane proteins, and chemokines present in LHACM tissue. The assay detected a total of 383 biomolecules retained in LHACM (Figure 3(b), Supplementary Table 1). Of the targets detected, 137 were identified as tissue growth factors associated with normal physiological mechanisms inherent in remodeling and homeostasis, and contributed 35.8% of the total composition. Inflammatory cytokines (38 of 383 analytes) accounted for 9.9% and chemokines (22 of 383 analytes) accounted for 5.7% of the total composition. Matrix and cell–cell adhesion receptors (35 of analytes) contributed 9.1%, while signaling receptors (78 of 383 analytes) contributed to 20.4% of the analytes detected. Membrane bound and transmembrane proteins (47 of 383 analytes) contributed to 12.3% of the total proteins. ECM molecules and proteases (26 of 383 analytes) accounted for 6.8% of the total soluble protein content. The full list of analytes detected is reported in Supplementrary Table 1. The composition of LHACM is similar to published reports of the composition of DHACM. 31
Effect of amniotic membrane products on HDF activity
Proliferation in response to positive and negative controls and both LHACM and DHACM extracts was tested on HDFs. The 10 mg/mL concentration of LHACM and DHACM extract stimulated significantly greater proliferation of HDFs compared to the basal media group (p < .05). At 1 mg/mL concentration, the effect was no longer observed, indicating dilution of the proliferative factors found in LHACM and DHACM extracts (Figure 4(a)). No significant difference was observed between treatments with LHACM and DHACM. Overall, both LHACM and DHACM extracts demonstrated a dose-dependent mitogenic effect on HDFs. In vitro bioactivity in human dermal fibroblasts. (a) Proliferative response to LHACM and DHACM when HDFs were incubated with extract for 72 h. Cell number was determined using the CyQuant assay (Thermo Fisher Scientific, Waltham, MA). Data normalized to the basal group as average fold change ± the standard deviation. n = 4; three technical replicates. (b) Migration of HDFs in a 2D scratch assay. Live cell label-free imaging of HDF over 120 h under exposure of LHACM or DHACM extracts or media controls. Data presented as % coverage of the initial scratch area at each time point ±standard error. n = 3; three technical replicates. (c) Metabolic response to LHACM and DHACM. Following 48 h treatment with extracts, 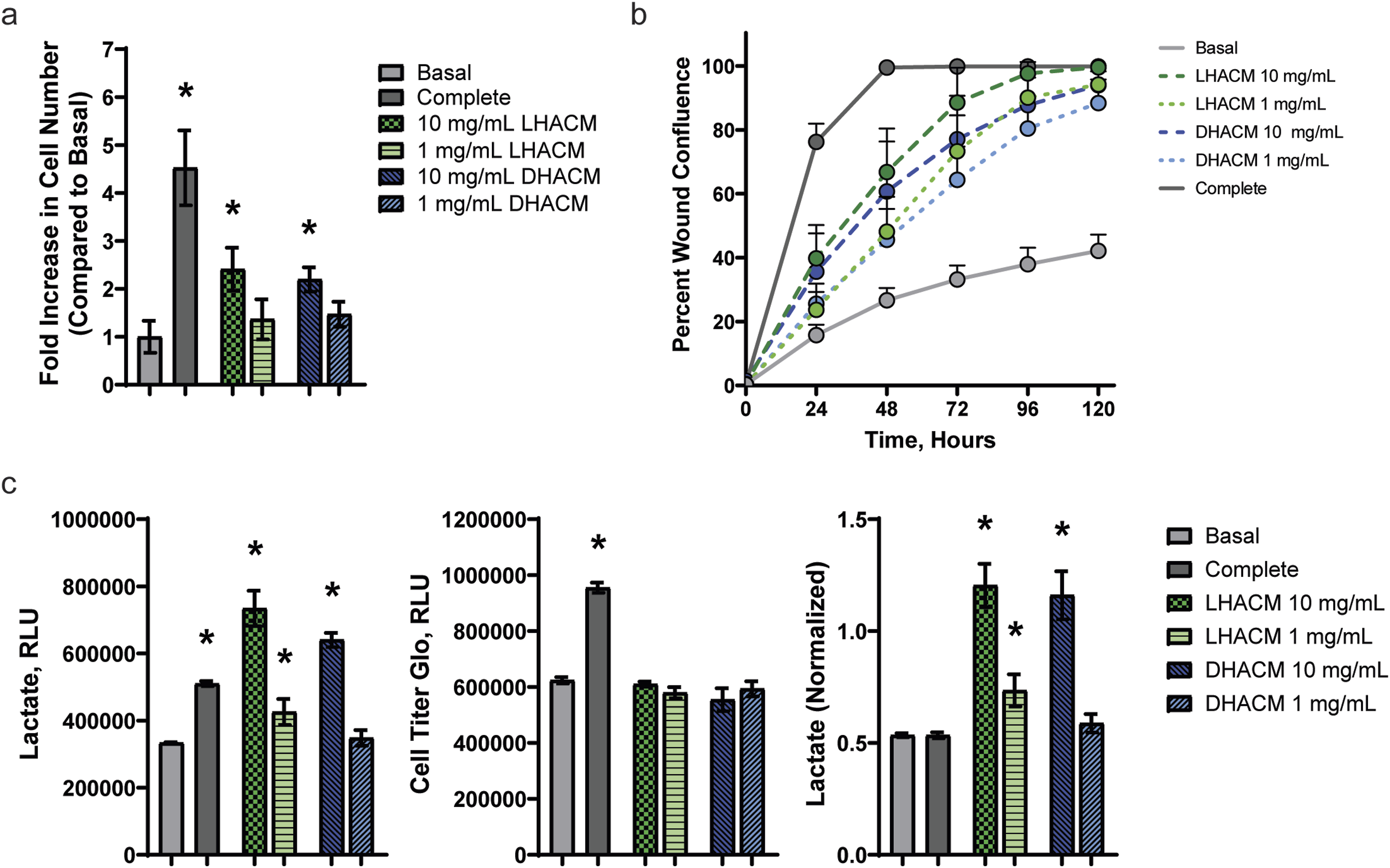
Migration in response to extract treatment and controls was also tested on HDFs. Quantification of cellular ingress into a scratched, cell-free zone was monitored over time and reported as percent scratch (wound) confluence. After 72 h, LHACM and DHACM extracts promoted greater cell migration with an average of 88.5% ± 10.9% and 77.1% ± 13.6% wound closure with 10 mg/mL treatment to 74.1% ± 11.8% and 66.9% ± 7.7% at 1 mg/mL respectively (Figure 4(b)), compared with the basal media group, which sustained cell viability but stimulated a migratory response of only 33.2% ± 4.4% wound closure. In contrast, the positive control (10% FBS in basal media) stimulated greater HDF migration with a 99.8% ± 0.2% wound closure of the scratch compared with both the negative control. However, after 120 h, all treatments with LHACM and DHACM elicited a greater than 85% wound closure compared with the negative control. No differences were observed in the migration of HDFs between LHACM and DHACM extracts.
Metabolic activity in response to extract treatment and controls, as measured by the presence of lactate in the media, was tested in HDFs. The positive control treatment (10% dialyzed FBS in basal media) both LHACM and DHACM extracts at 20 mg/mL significantly increased extracellular lactate compared to the basal media group (p < .05). At 1 mg/mL, LHACM extract continued to induce elevated lactate release; however, DHACM extract was equivalent to the negative control. Despite cells being plated at confluence to minimize the confounding effects of proliferation on the results, the data was normalized to cell number for further analysis. The results were unchanged with the exception of the positive control. The 10% dialyzed FBS treatment increased cell number, but did not increase extracellular lactate. Whereas, LHACM and DHACM both stimulated almost a 2-fold increase in lactate release per cell (Figure 4(c)).
Effects of LHACM and DHACM on HMEC1 invasion in vitro
HMEC1 invasion in response to treatment was tested in an in vitro transwell system (Figure 5(a)). Invasion was assessed by monitoring chemotactic invasion in real time through a 3D extracellular matrix towards a chemoattractant (LHACM or DHACM extract). Whole-well images of cells on both bottom and top of the membrane allowed for quantitation of cell area on each side of the membrane and reported as an increase in phase object area on the bottom of the membrane (Figure 5(a)). In response to LHACM and DHACM extract, results demonstrate a dose dependent chemotactic migration of HMEC1 cells over that of the negative control (Figure 5(b)). Results demonstrate that LHACM and DHACM extracts stimulate the directional migration of HMECs in vitro. Chemotactic response to human amniotic tissue allografts. HMEC1 invasion was assessed by monitoring chemotactic invasion in real time through a 3D extracellular matrix towards a chemoattractant (LHACM or DHACM extract). Whole-well images of cells on both bottom and top of the membrane allowed for quantitation of cell area on each side of the membrane. (a) left: schematic of the experimental setup; right: Representative 0-h top mask and 24-h bottom mask images. Figure realized with BioRender.com (b) Total phase object area of bottom mask normalized to initial top mask value at various time points ± the standard deviation. n = 3.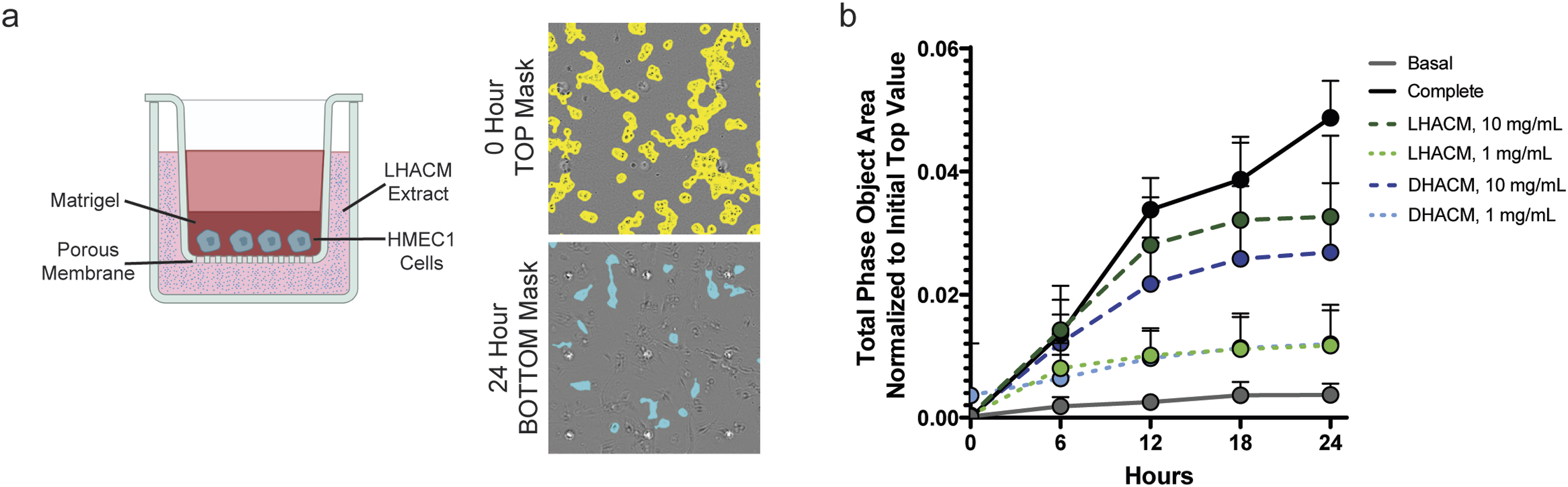
In vivo cellular activity promoted by LHACM and DHACM
The cellular response to LHACM and DHACM implantation was determined in vivo in a subcutaneous nude mouse model over 4 weeks. Histological analysis showed progressive remodeling of LHACM and DHACM throughout the duration of the study, consistent with the expected response to a biocompatible implant. Host cell infiltration was evident with greater than 75% of LHACM and DHACM demonstrating cellular infiltration/ingrowth at two and four weeks post implantation (Figure 6(b) and (d)). Reorganization of the implants, characterized by resorption and deposition of collagenous matrix began as minimal and advanced to mild by weeks two and four (Figure 6). Evidence of neovascularization surrounding the implants and within the implants was observed at week 4 (Figure 6(a) and (c)). No differences were observed in cellular response between LHACM and DHACM. Additionally, histological assessment showed no major inflammatory reaction indicative of a rejection response (Data Not Shown). In vivo cellular response in a subcutaneous implant model. The cellular response to LHACM (a) and DHACM (c) was assessed after (left) 1 week, (middle) 2 weeks, and (right) 4 weeks of subcutaneous implantation in the nude mouse. Explanted tissue was stained by H&E to assess the presence of allografts. LHACM and DHACM allografts are visible at all time points (asterisks). Increased host cell infiltration observed over time (BLACK arrows) and associated with neocollagen. Independent histopathologist score of H&E stained sections for cellular infiltration, implant reorganization, collagen deposition, and neovascularization LHACM (b) and DHACM (d). Data is represented as mean ± the standard deviation. n = 6; Scale bar = 100 µm.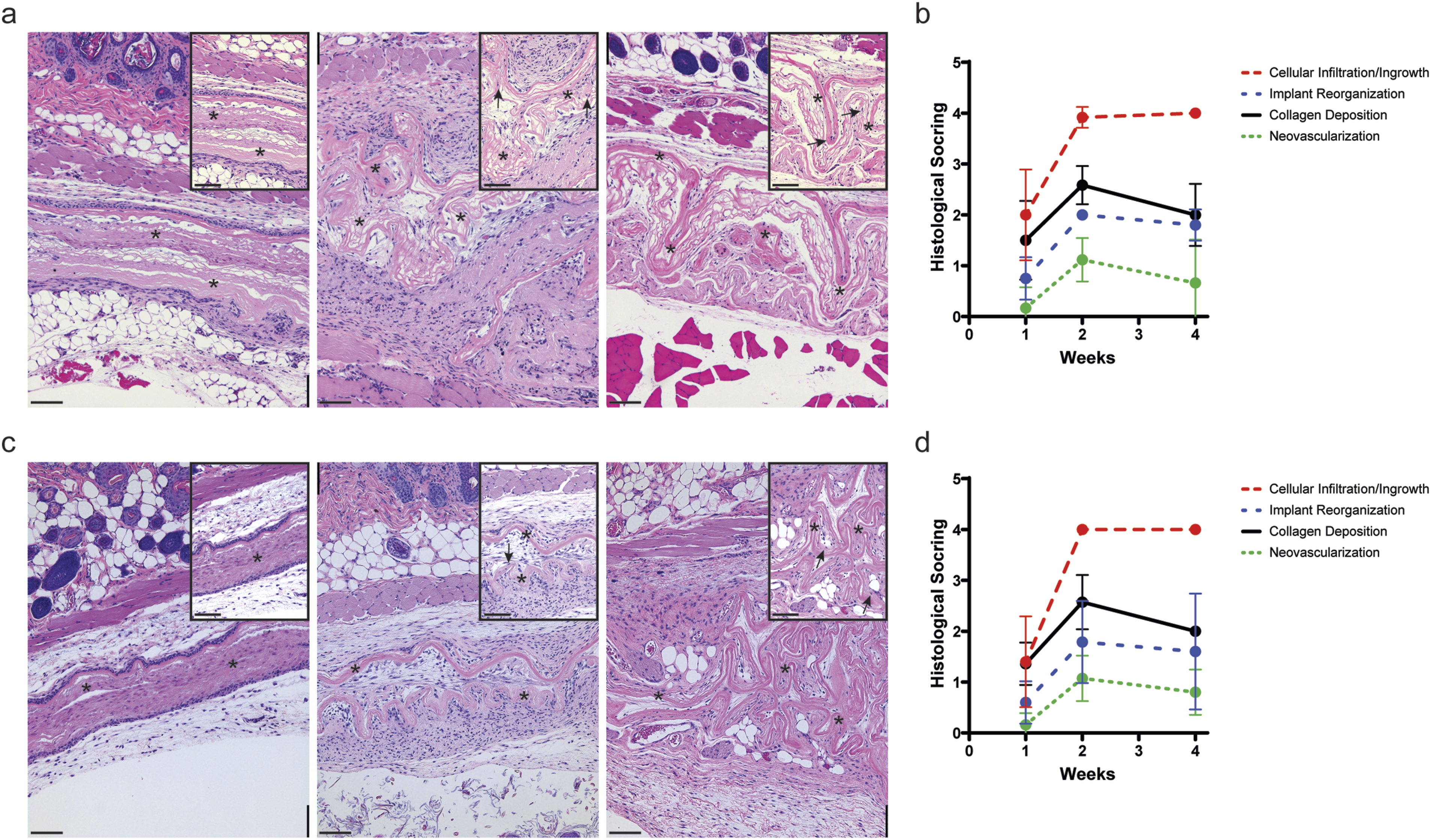
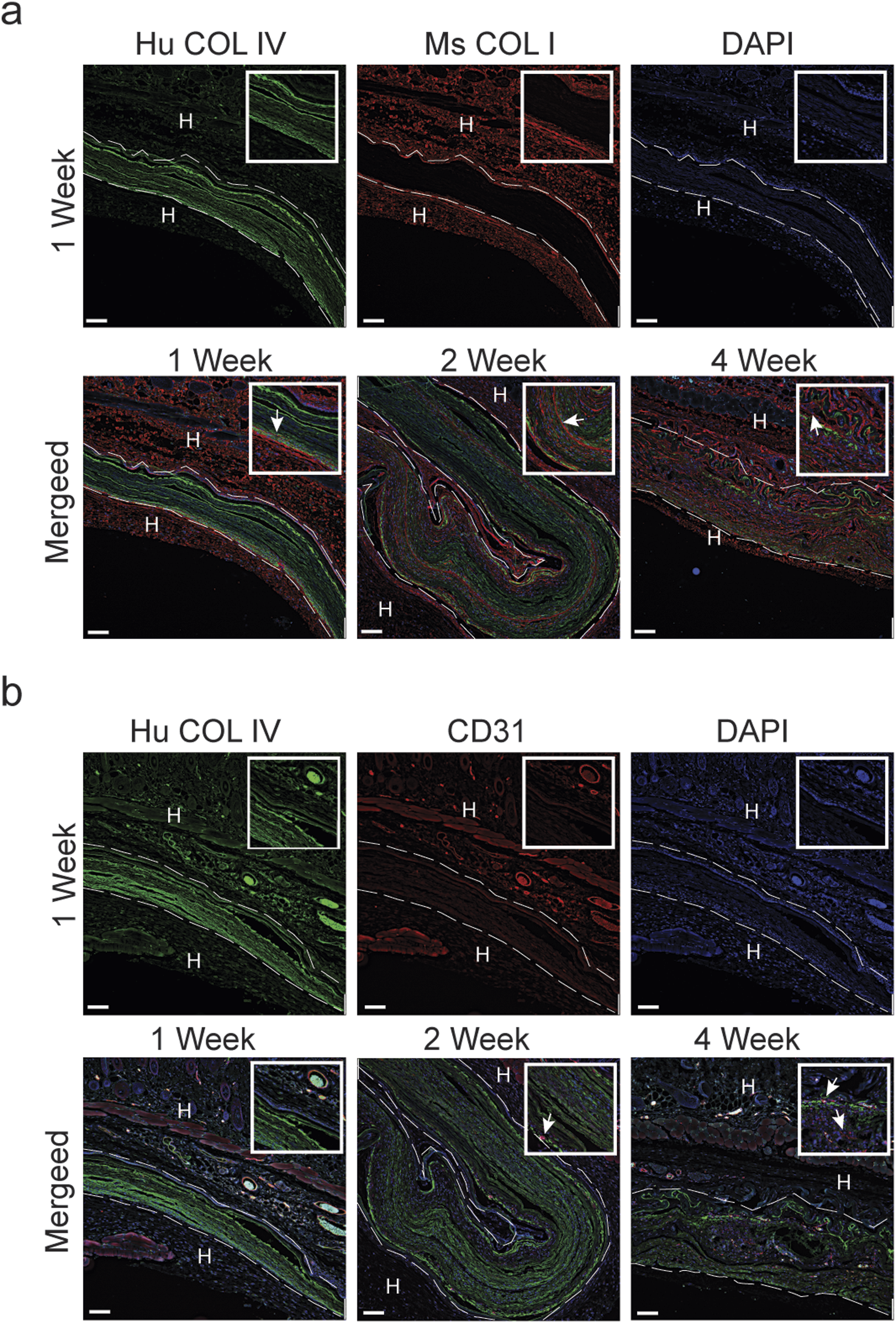
To visualize the histopathology findings, immunofluorescence was employed with human and mouse specific antibodies. Human specific collagen type IV was used to highlight the chorion membrane of the allograft implants. Host cell infiltration over the 4 week period is evident with DAPI staining. The infiltration of host cells was also associated with increased mouse specific collagen type I staining within the allograft implants (Figures 7(a) and 8(a)). Cellular infiltration was primarily observed at the 2 and 4 week time points. Histopathology analysis identified that the host cell infiltration was composed primarily of fibroblasts with minimal neovascularization observed. However, after 4 weeks, CD31 positive cells are present within the implants indicating that endothelial cells were infiltrating the allografts (Figures 7(b) and 8(b)). Immunofluorescence analysis of host cell infiltration in response to LHACM. LHACM after 1 week, 2 weeks, and 4 weeks of subcutaneous implantation in the nude mouse. Immunofluorescence staining, using species specific antibodies, evaluated implant reorganization, collagen formation, and endothelial cell recruitment. Images were acquired on a Leica microscope fitted with 10x and 40x objectives, using Leica Application Suite Advance Fluorescence software and the THUNDER Imager (Leica Microsystems Deerfield, IL). (a) Reorganization and neocollagen formation associated with infiltrating host fibroblast cells (arrows). Immunofluorescence of cellular infiltration and associated neocollagen formation: human collagen type IV (green); mouse collagen type 1 (red); cell nuclei (blue); Dashed line indicated implant boundary. (b) Recruitment of endothelial cells and neovascularization within and surrounding the LHACM implant (arrows). Immunofluorescence of endothelial cells: human collagen type IV (green); Dashed line indicated implant boundary. CD31 (red); cell nuclei (blue). H: host tissue; Hu: human; Ms: mouse; COL IV: collagen type IV; COL I: collagen type I; Scale bar = 100 µm. Immunofluorescence analysis of host cell infiltration in response to DHACM. DHACM after 1 week, 2 weeks, and 4 weeks of subcutaneous implantation in the nude mouse. Immunofluorescence staining, using species specific antibodies, evaluated implant reorganization, collagen formation, and endothelial cell recruitment. Images were acquired on a Leica microscope fitted with 10x and 40x objectives, using Leica Application Suite Advance Fluorescence software and the THUNDER Imager (Leica Microsystems Deerfield, IL). (a) Reorganization and neocollagen formation associated with infiltrating host fibroblast cells (arrows). Immunofluorescence of cellular infiltration and associated neocollagen formation: human collagen type IV (green); mouse collagen type 1 (red); cell nuclei (blue); Dashed line indicated implant boundary. (b) Recruitment of endothelial cells and neovascularization within the LHACM implant (arrows). Immunofluorescence of endothelial cells: human collagen type IV (green); Dashed line indicated implant boundary. CD31 (red); cell nuclei (blue). H: host tissue; Hu: human; Ms: mouse; COL IV: collagen type IV; COL I: collagen type I; Scale bar = 100 µm.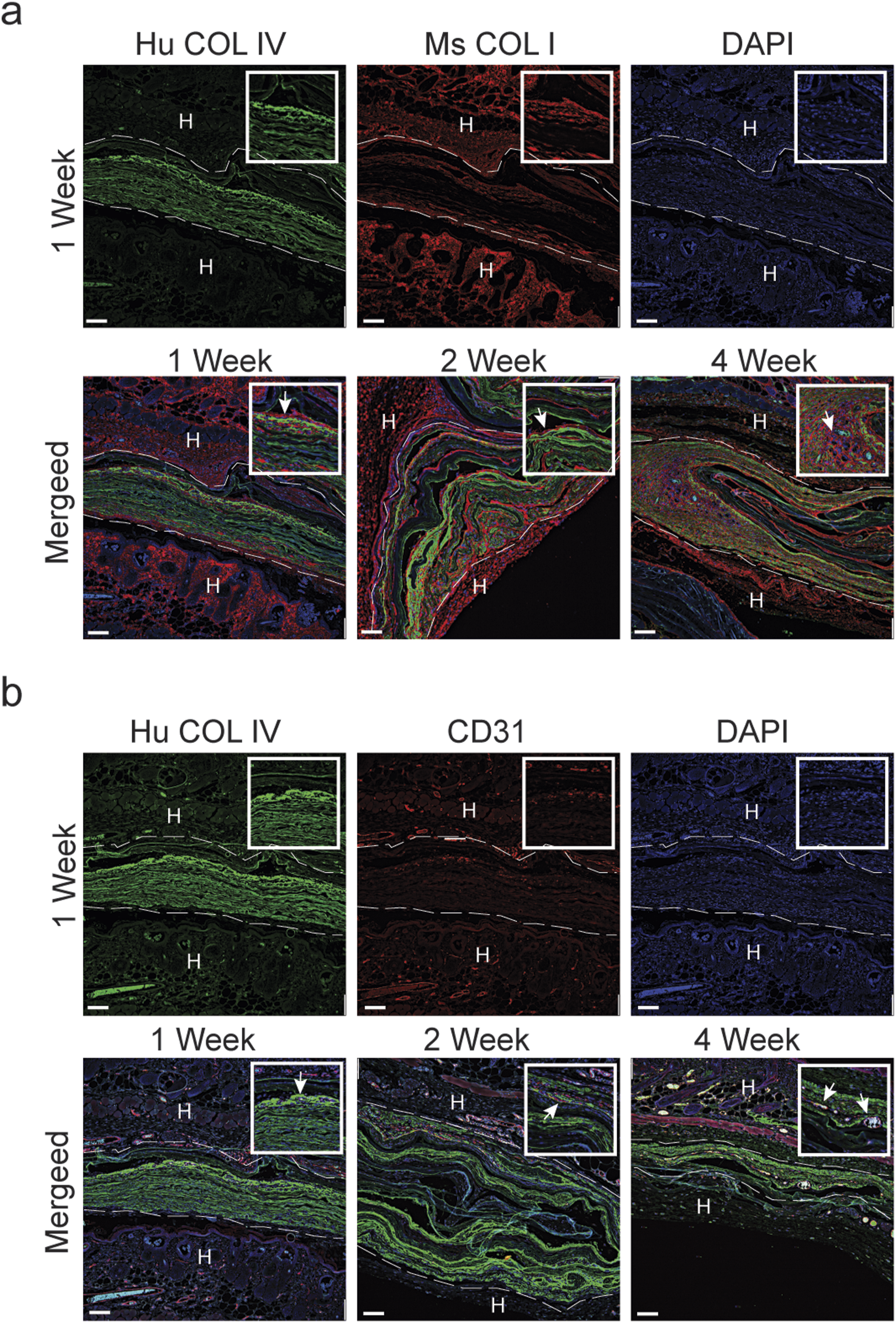
Discussion
The expanded use of amniotic tissue in wound care and other applications over the past decade has spurred the development of novel processing and tissue preservation methods to maintain tissue structure and maximum biological activity while providing versatility in clinical use. Efficacy of these products is a result of the inclusion of all or some of the layers comprising the amniotic sac and the processing techniques employed during manufacturing. When evaluating LHACM, a lyophilized PURION® processed tri-layer allograft and DHACM, a dehydrated bi-layer layer allograft, the differences observed are attributable to the presence or absence of the intermediate layer and the method of dehydration. While the physical characteristics differ, the use of the PURION® process in both configurations preserved the biological activity, both in vitro and in vivo.
During gestation, the amniotic membrane forms a barrier that protects and supports the growing fetus. Tissue processing methods are designed to retain these inherent properties and their ability to do so can be attributed, in part, through the preservation of the ECM. The amnion and chorion layers are composed of collagens, fibronectin and laminin as well as proteoglycans and glycoproteins . 4 This network of macromolecules can facilitate the adhesion of cells and anchor signaling proteins within the matrix, resulting in a framework that confers the barrier function and regulates transport of biomolecules across the membrane . 4 Collagen type I is the primary component of the amnion and intermediate layers and collagen type IV is largely associated with the chorion layer. Using these key components as indicators, the presence of the amnion, intermediate layer and chorion was confirmed in LHACM and the amnion and chorion in DHACM. Visualization of the grafts highlighted another key difference between these two configurations, thickness. The use of lyophilization for LHACM created a thick, open structure; whereas, air-dehydration for DHACM resulted in a thin and dense, laminated matrix. While both grafts are comprised of an intact ECM, the addition of the intermediate layer and lyophilization imparts alternative physical characteristics to LHACM as compared to DHACM.
The clinical impact of amniotic membrane allografts as barriers has been demonstrated in cases to limit postoperative adhesion in abdominal, otolaryngologic, and laminectomy surgery and ocular injuries.32–37 The processing and preservation of LHACM and DHACM produce allografts that function as membrane barriers. However, it is also critical to understand the impact of processing techniques on the biomolecules embedded within the allografts. 20 A multiplex ELISA detected the proteins contained within LHACM and identified a variety of growth factors and cytokines that regulate vital processes in wound healing including cell migration, proliferation, inflammation, angiogenesis and ECM remodeling. 38 Similar factors have previously been identified in DHACM, a dehydrated amniotic membrane allograft.21,24,39–42 The wide range of factors retained in LHACM and DHACM are associated with cellular functions integral to the healing cascade; however, their presence alone cannot predict their activity.
In order to provide the most beneficial and safe amniotic membrane allograft for therapeutic purposes it is necessary to utilize the best methods for collection, processing and storage while preserving the native structure of the amniotic tissue. 20 Extensive analysis of PURION® processed amniotic membrane allografts has demonstrated that this processing method retains well‐known regulatory proteins and preserves the bioactivity to stimulate cellular activities supportive of wound healing. Indeed, increases in proliferation and migration of multiple cell types including stem cells, HDFs, tenocytes, and endothelial cells highlights the ability of amniotic membrane allografts to impact basic cellular functions.21,23,25,26,43,44 Through more advanced analysis, it has been demonstrated that amniotic membrane can not only impact cellular signaling but also that its effects can vary depending upon the environment in which it is placed.43–45 Functional assays derived from the critical steps in the healing cascade were developed to model the role LHACM and DHACM may play in clinical applications. Fibroblast migration and proliferation are essential and rate-limiting steps to repair wounds due to their central role in the formation of granulation tissue which can provide a matrix for additional cells and blood vessels to grow.46,47 It has been demonstrated that fibroblasts respond to cytokines and growth factors such PDGF, transforming growth factor (TGF)-β and bFGF, all of which were identified in both LHACM and DHACM.47,48 Indeed, soluble factors from LHACM and DHACM stimulated human dermal fibroblast proliferation and migration to a similar degree. Upon recruitment of cells to the site of injury, influencing their behavior is the next step in facilitating a healing environment. Cellular metabolic changes are linked to wound healing and alterations in the metabolic requirements can contribute to wound metabolism at different levels and impacts the outcome of the healing response. 49 Chronic wounds are often hypoxic which results in the production and accumulation of lactate that works to regulate the pH of wounds to physiological range while triggering neovascularization and other processes to support wound healing. In normal or acute wound healing, addition of lactate functions to imitate hypoxia which encourages metabolic activity and associated processes to promote wound healing. 50 Increased lactate production by HDFs resulted from treatment of cells with soluble factors from DHACM and LHACM. The two treatments elicited increased production/accumulation of lactate to the same extent. The regulation of the lactate production may influence the healing response through regulation of vascularization and granulation tissue formation through the production of collagen. Indeed, topical application of lactate has been used in many in vitro and in vivo settings and demonstrated that lactate can support wound healing in acute and chronic wounds by instigating signals for angiogenesis and connective tissue deposition.51–54
In a healing wound, the formation of new granulation tissue is accompanied by rapid growth of new capillaries and production of an abundant network of new blood vessels. 55 Previous work demonstrated that DHACM contains both pro- and anti-angiogenic growth factors that differentially regulate angiogenesis depending upon the cellular environment in which it was placed. Additionally, DHACM tissue promoted chemotactic migration of human endothelial cells and endothelial cell proliferation in vitro.22,44 Similarly, LHACM contains a large number of pro-angiogenic growth factors, including angiogenin, angiopoietin-2, EGF, bFGF, HB-EGF, HGF, PDGF-BB, PlGF, TGFβ, and VEGF, and one or more of these factors may have been responsible for promoting chemotactic migration of human endothelial cells in vitro similar to DHACM. The inherent differences in the physical properties of these grafts are apparent, however, use of the PURION® process preserved the regulatory factors such that both LHACM and DHACM elicited similar biological responses in vitro.
Translation of these in vitro observations to clinical outcomes is usually bridged using an animal model for wound healing. However, the use of human-derived allografts in immunocompetent animals introduces the possibility for a xenogeneic response, confounding the interpretation of the results. For the purposes of evaluating LHACM and DHACM, the nude mouse model was used. The lack of a thymus and resulting T-cell deficiency ameliorates the potential for a species-specific rejection response.56–60 Indeed, implantation of DHACM and LHACM resulted in minimal inflammation that is not indicative of a rejection response. However, subcutaneous implantation of LHACM and DHACM tissue allografts in this murine model demonstrated increased cellular ingrowth, collagen deposition, and implant reorganization over 4 weeks. More specifically, immunofluorescence analysis shows a progressive increase in host cell infiltration that is accompanied by increased collagen type I deposition. Using species specific antibodies there is an observed loss of human collagen type IV that coincides with increased mouse collagen type I suggesting a remodeling of the implants along with deposition of new collagen by the infiltrating cells. Furthermore, 4 weeks post implantation, there is a notable presence of CD31 positive cells both within and surrounding the LHACM and DHACM implant. CD31 (also known as platelet endothelial cell adhesion molecule-1) is highly expressed on endothelial cells and to different degrees on several hematopoietic cells, including monocytes, granulocytes, and platelets. 61 The origin of the CD31 positive cells is not known but the delayed appearance of CD31 positive cell suggests that LHACM and DHACM promote the infiltration of fibroblast cells that deposit new collagen into the wound area supporting the formation of granulation tissue which further supports the migration of highly angiogenic CD31 positive cells. This corroborates the data obtained from in vitro studies and further demonstrates the clinical benefit of using LHACM and DHACM in acute and chronic wounds.
Conclusions
Material properties and biomolecule viability can greatly impact the clinical use and efficacy of amniotic membrane allografts. This study characterizes the LHACM and DHACM allografts and demonstrates that although the handling properties are most likely different with LHACM due to its thickness, both allografts still maintain a functional barrier while providing biologically active molecules to support the healing process. LHACM offers an allograft with the biological properties synonymous with the PURION® process, but with alternative structural and material properties relative to DHACM. This data suggests that both LHACM and DHACM provide versatility in the use of amniotic membrane allografts while functioning as a responsive barrier. The equivalent in vitro and in vivo effects between these two allografts suggests that the use of LHACM should provide similar efficacy as seen with DHACM in various clinical and surgical applications.
Supplemental Material
Supplemental Material - PURION® processed human amnion chorion membrane allografts retain material and biological properties supportive of soft tissue repair
Supplemental Material for PURION® processed human amnion chorion membrane allografts retain material and biological properties supportive of soft tissue repair by Sarah Moreno, Michelle Massee, Shauna Campbell, Heather Bara, Thomas J Koob and John R Harper in Journal of Biomaterials Applications
Footnotes
Acknowledgments
The authors thank Lisa Godwin for her technical assistance.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors were full time employees at MIMEDX Group, Inc. at the time experiments were executed. S.M., M.M., H.B., T.K., and J.H. hold equity in MIMEDX Group, Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
